1. Hemoglobin and the Movement of Oxygen. Respirator system/biochemistry
|
|
|
- Cory White
- 6 years ago
- Views:
Transcription
1 1. Hemoglobin and the Movement of Oxygen Respirator system/biochemistry
2 YOU MUST BE ABLE TO: Hemoglobin and the Movement of Oxygen specific aims 1. Compare structure of myoglobin and hemoglobin 2. Understand the mechanism of how oxygen binding to iron affects hemoglobin structure 3. Understand the T and R states of hemoglobin and how they are converted to each other 4. Understand the concept of cooperativity in hemoglobin and the importance of having quaternary structure 5. Differentiate between oxygen saturation curves of myoglobin and hemoglobin and know the implications of these curves on the oxygen delivery (loading and unloading) 6. Understand the effects of H+, CO2, and 2,3BPG on oxygen binding to hemoglobin and what are the important physiological aspects about these effects 7. Know how CO2 is transported by hemoglobin to lungs and exhaled 8. Understand the molecular mechanism of Bohr,s effect 9. Know the difference between the structures of fetal hemoglobin (HbF) and adult hemoglobin (HbA) 10. Understand the importance of 2,3BPG binding to fetal hemoglobin and how this affects fetal life =
3 Introduction Animals Have Widely Varying Needs for Oxygen Demand for Oxygen Can Change in Seconds Basal Needs are Significant - Diffusion not Enough Exercise, Fight/Flight Add to the Need ATP Energy Produced Aerobically 15 Times More Efficiently Than Anaerobically Respiration versus Fermentation Efficient, Adaptable Oxygen Delivery is Necessary
4 Quaternary Structure Interaction of multiple protein subunits Hemoglobin - 4 Subunits (α2β2), 1 Heme Each, 1 O2 Each, 1 Donut Hole Myoglobin - 1 Subunit, 1 Heme, 1 O2 Each
5 Structure and Function Heme Prosthetic Group Ferrous Iron - Methemoglobin Won t Work Only Fe ++ Binds Oxygen
6 Structure and Function Edge-on View Attached to Remainder of Global Subunit Histidine s Movement Changes Global Unit s Shape
7 Cooperativity Entering Lungs No Oxygen (T-State( Affected by O2 Binding (R-State)0 Fully Oxygenated (R-State( Exiting Lungs Oxygen Bound Affected by O2 Binding R-State Binding of the first O2 favors binding of second, etc. - Cooperatively Cooperatively Important as Hemoglobin Rapidly Passes Through Lungs
8 Cooperativity At High O2, Both Hold 100% At Low O2, Myoglobin Holds More than Hemoglobin As Curves Move to Right Less Affinity for Oxygen Sigmoidal Binding Curve Hyperbolic Binding Curve Myoglobin Better for Storing Oxygen Hemoglobin Better at Delivering Oxygen
9 Bohr Effect Less Oxygen Bound at Same Pressure More Affinity Less Affinity Protons Can Bind to Hemoglobin Protons Change Hemoglobin s Shape Reshaped Hemoglobin Loses Oxygen Rapidly Metabolizing Tissues Release Protons Rapidly Metabolizing Tissues Get More Oxygen From Hemoglobin More O2 Required To Have Same Fraction Bound
10 Bohr Effect & CO2 Acid Favors Release of O2 From Hemoglobin CO2 Favors Release of O2 From Hemoglobin Acid and CO2 are Released by Rapidly Metabolizing Tissues
11 2,3 BPG Byproduct of Glycolysis Exercising Muscle Cells Rapid Use Glycolysis Exercising Muscle Cells Produce Acid, CO2, and 2,3 BPG Binds in Hole of Donut Locks Hemoglobin in T-State 2,3 Bisphosphoglycerate
12 2,3 BPG and Oxygen Binding Slow Metabolic Rate Rapidly Metabolizing Cells Produce Acid Rapidly Metabolizing Cells Release CO2 Rapidly Metabolizing Cells Release 2,3 BPG All Favor O2 Release from Hemoglobin So Rapidly Metabolizing Cells Get More O2 Fast Metabolic Rate
13 2,3 BPG and Smoking 2,3 BPG Big Concern for Smokers Blood of Smokers has High Levels of 2,3 BPG Hemoglobin Gets Locked in T-state in Passage Through Lungs Oxygen Carrying Capacity of Blood Reduced Carbon Monoxide Levels Also Higher in Smokers 2,3 Bisphosphoglycerate
14 Movement of CO2 HbO + H + O2 Blood Rapidly Metabolizing Tissue Blood H2O Some Remainder H + :Hb-CO2 HbO HbO Exhaled H +: Hb-CO2 H2O + CO2 O2 + H +: Hb-CO2 Lungs
15 CO2 and O2 Transport are linked to Bohr effect protons
16
17 Carbon Monoxide and Heme An Additional Histidine is Present at the Heme Iron Site Reduces Affinity to CO, but Does Not Eliminate it Carbon Monoxide in Cigarette Smoke Note That CO2 Does Not Bind to Heme, nor do Protons
18 Fetal Hemoglobin The Body Makes Different Globins Over Time Most Variations Centered on Birth Fetal Hemoglobin Mostly α2γ2 High Most of Life Highest in Fetus At Adult Levels by 24 Weeks
19 Fetal Hemoglobin Fetal Hemoglobin Can t Bind to 2,3 BPG Mostly Remains in R-state Increasing Affinity
20 Sickle Cell Anemia Sickle Cell Anemia is a Genetic Disease Affecting Hemoglobin Multiple Forms - Mutation of Glu to Val at Position #6 Most Common Red Blood Cells Lose Rounded Shape and Form Sickles Shape Change Happens in Low O2 Conditions - Exercise Change Caused by Polymerization of Hemoglobin Sickled Cells
21 Sickle Cell Anemia Rounded Cells Move Easily Through Capillaries Sickled Cells Get Stuck Sickled Cells Removed by Spleen
22
23
24 HIGHLIGHTS 1.Myoglobin and hemoglobin are related proteins involved in (respectively) storing and carrying oxygen in the body. 2. Myoglobin is a single subunit protein with high affinity for oxygen. It holds oxygen tighter than hemoglobin and serves in a battery-like capacity in tissues to release oxygen when tissue oxygen concentration is very low. Myoglobin can take oxygen from hemoglobin. 3. Hemoglobin is a four-subunit protein complex (two alpha subunits and two beta subunits) that serves to carry oxygen from the lungs to the tissues where it is needed. Hemoglobin is genetically related to myoglobin and is evolutionarily derived from it.
25 4. Myoglobin and hemoglobin both have porphyrin rings (like in chlorophyll) to hold ferrous (Fe++) iron. The specific porphyrin in these proteins is Protoporphyrin IX. Ferrous iron is the form of iron involved in carrying oxygen. Ferric iron (Fe+++), which is the oxidized form of iron will not carry oxygen. Heme is a term used to describe the protoporphyrin IX complexed with iron. 5. The iron in hemoglobin and myoglobin is held in place by five molecules - four nitrogens of the protoporphyrin IX ring and a histidine (called the proximal histidine). Oxygen is carried between the iron and an additional histidine (called the distal histidine) not involved in holding the iron.
26 6.If one plots the percentage of oxygen sites bound versus partial pressure for myoglobin, a hyperbolic curve is generated, consistent with a molecule with a single binding site and a high affinity for oxygen. The P50, which is the partial pressure of oxygen necessary to fill 50% of the myoglobins with oxygen, is very low for myoglobin, consistent with high affinity. Because myoglobin has high affinity for oxygen, it doesn't release much oxygen until it is in an environment with very low oxygen pressure. For this reason, it would be a poor oxygen transport protein.
27 7. Hemoglobin is much better designed to meet an organism's physiological needs for carrying oxygen than myoglobin. This is due to its four-subunit organization (one heme per subunit and one oxygen carried per subunit) which behaves in a cooperative fashion in binding oxygen. 8. Binding of oxygen by the iron atom causes it to be pulled 'up' slightly. This, in turn, causes the histidine attached to it to change position slightly, which causes all the other amino acids in the subunit to change slightly. The changes in shape (different 'states') result in the protein gaining affinity for oxygen as more oxygen is bound. The phenomenon is referred to as cooperativity. 9. Hemoglobin can exist in a "tight" state, called 'T', which exhibits low oxygen binding affinity. Hemoglobin in the T state will tend to release oxygen.
28 10 A second state of hemoglobin is the "relaxed" or R state, which exhibits increased oxygen binding affinity. Binding of oxygen by hemoglobin helps it to "flip" from the T to the R state and release of oxygen by hemoglobin helps it to flip from R to T. 11. A molecule called 2,3-bisphosphoglycerate (2,3 BPG) is produced by actively respiring tissues. It can bind in the gap in the center of the hemoglobin molecule and in doing so, stabilize the T state and favor the release of oxygen. Thus, tissues that are actively respiring get more oxygen. 2,3 BPG is noteworthy because the blood of smokers has a higher concentration of the molecule than the blood of non-smokers. 12. The Bohr effect describes physiological and molecular responses to changes in ph with respect to oxygen and carbon dioxide in the body. The oxygen effects arise from changes in the teriary structure of hemoglobin arising from binding of protons to histidines in the molecule when under low ph.
29 13. Rapidly metabolizing tissues (such as muscle) generate low phs, due to release of carbon dioxide and the conversion of this to carbonic acid by carbonic anhydrase. Carbonic acid readily loses a proton, becoming bicarbonate. 14. Thus, rapidly metabolizing tissues generate protons, which get absorbed by hemoglobin, which releases oxygen to feed the tissues.
30 15. CO 2 can also be taken up by hemoglobin at amine residues, causing protons to be released. Note that CO 2 binds hemoglobin at a site other than what oxygen binds. Carbon monoxide, however, can compete with oxygen for binding to the heme. 16. In the lungs, a reversal of this process occurs. Remember that the oxygen concentration in the lungs is high, so oxygen forces off the carbon dioxide and the carried protons from the hemoglobin. Addition of a proton to bicarbonate re-creates carbonic acid, which undergoes the reversal of the earlier carbonic anhydrase reaction, causing CO 2 to be released out of the lungs as a gas.
31 Summary Animals Have Widely Varying O2 Needs ATP Generated Much More Efficiently in Presence of O2 Hemoglobin and Myoglobin are Related, but Have Different Functions Hemoglobin has Four Subunits and Hemes. Myoglobin has One of Each Bind of O2 by Heme s Iron Pulls up on a Histidine and Change s Hemoglobin s Shape Changing Hemoglobin s Shape Converts Hemoglobin from T-state to R-state R-state Binds Oxygen Better. T-state Releases O2 Better In the Bohr Effect, Binding of CO2 and H + Favors O2 Release The Bohr Effect Explains How Oxygen and CO2 Exchanged in Lungs 2,3 BPG is Produced by Rapidly Metabolizing Cells. It too Favors O2 Release Fetal Hemoglobin Can t Bind 2,3 BPG and has Greater O2 Affinity Than Adult Hemoglobin Sickle Cell Anemia (SCA) is a Genetic Disease of Hemoglobin In Low O2 Concentration, Red Blood Cells of SCA Sufferers Form Sickle Shapes Sickled Cells Stick in Capillaries and Can be Fatal People Heterozygous for the Mutated Gene Survive Malaria Better Than Others
Chapter 7. Heme proteins Cooperativity Bohr effect
 Chapter 7 Heme proteins Cooperativity Bohr effect Hemoglobin is a red blood cell protein that transports oxygen from the lungs to the tissues. Hemoglobin is an allosteric protein that displays cooperativity
Chapter 7 Heme proteins Cooperativity Bohr effect Hemoglobin is a red blood cell protein that transports oxygen from the lungs to the tissues. Hemoglobin is an allosteric protein that displays cooperativity
Globular proteins Proteins globular fibrous
 Globular proteins Globular proteins Proteins are biochemical compounds consisting of one or more polypeptides typically folded into a globular or fibrous form in a biologically functional way. Globular
Globular proteins Globular proteins Proteins are biochemical compounds consisting of one or more polypeptides typically folded into a globular or fibrous form in a biologically functional way. Globular
2,3BPG & Smoking. When the carve is shifted to the right decreasing in O2 binging affinity b.c the change in the shape &function
 Where do protons bind to Hemoglobin? It could bind in dissociated carboxyl group in Hb or in dissociate amino group ( NH+3, COOH ) Protons change Hb shape, How? Once proton bind to Hb it will produce +
Where do protons bind to Hemoglobin? It could bind in dissociated carboxyl group in Hb or in dissociate amino group ( NH+3, COOH ) Protons change Hb shape, How? Once proton bind to Hb it will produce +
Lecture 5. Dr. Sameh Sarray Hlaoui
 Lecture 5 Myoglobin & Hemoglobin Dr. Sameh Sarray Hlaoui Myoglobin and Hemoglobin Myoglobin - Myoglobin and Hemoglobin are (metalloprotein containing a heme prosthetic group). hemeproteins - Function as
Lecture 5 Myoglobin & Hemoglobin Dr. Sameh Sarray Hlaoui Myoglobin and Hemoglobin Myoglobin - Myoglobin and Hemoglobin are (metalloprotein containing a heme prosthetic group). hemeproteins - Function as
PBL SEMINAR. HEMOGLOBIN, O 2 -TRANSPORT and CYANOSIS An Overview
 1 University of Papua New Guinea School of Medicine and Health Sciences Division of Basic Medical Sciences Discipline of Biochemistry and Molecular Biology PBL SEMINAR HEMOGLOBIN, O 2 -TRANSPORT and CYANOSIS
1 University of Papua New Guinea School of Medicine and Health Sciences Division of Basic Medical Sciences Discipline of Biochemistry and Molecular Biology PBL SEMINAR HEMOGLOBIN, O 2 -TRANSPORT and CYANOSIS
Lecture #1. Done By : Rasha Rakan. Corrected by: Obadah Abubaker
 Lecture #1 Done By : Rasha Rakan Corrected by: Obadah Abubaker مالحظة: المكتوب باللون األسود هو ما كتب ف السال دات وما كتب باللون األزرق فهو كالم الدكتور أثناء المحاضرة وللذ ن قومون بتصو ر الش ت جعلنا
Lecture #1 Done By : Rasha Rakan Corrected by: Obadah Abubaker مالحظة: المكتوب باللون األسود هو ما كتب ف السال دات وما كتب باللون األزرق فهو كالم الدكتور أثناء المحاضرة وللذ ن قومون بتصو ر الش ت جعلنا
The hemoglobin (Hb) can bind a maximum of 220 ml O2 per liter.
 Hemoglobin Hemoglobin The most important function of the red blood cells is totransport (O2) from the lungs into the tissues, and carbon dioxide (CO2) from the tissues back into the lungs. O2 is poorly
Hemoglobin Hemoglobin The most important function of the red blood cells is totransport (O2) from the lungs into the tissues, and carbon dioxide (CO2) from the tissues back into the lungs. O2 is poorly
Gas Exchange in the Tissues
 Gas Exchange in the Tissues As the systemic arterial blood enters capillaries throughout the body, it is separated from the interstitial fluid by only the thin capillary wall, which is highly permeable
Gas Exchange in the Tissues As the systemic arterial blood enters capillaries throughout the body, it is separated from the interstitial fluid by only the thin capillary wall, which is highly permeable
Key Concepts. Learning Objectives
 Lectures 8 and 9: Protein Function, Ligand Binding -- Oxygen Binding and Allosteric Regulation in Hemoglobin [PDF] Reading: Berg, Tymoczko & Stryer, Chapter 7, pp. 183-199 problems in textbook: chapter
Lectures 8 and 9: Protein Function, Ligand Binding -- Oxygen Binding and Allosteric Regulation in Hemoglobin [PDF] Reading: Berg, Tymoczko & Stryer, Chapter 7, pp. 183-199 problems in textbook: chapter
270,000,000 hemoglobin units are. hemoglobin has 4 heme units; 2 α and 2 β units. Active site of a heme unit has an Iron ion
 RBC strange shape a biconcave disc that is round and flat RBC has no nucleus. The nucleus is extruded from the cell as it matures. An RBC can change shape to an amazing extent, without breaking, as it
RBC strange shape a biconcave disc that is round and flat RBC has no nucleus. The nucleus is extruded from the cell as it matures. An RBC can change shape to an amazing extent, without breaking, as it
Biochemistry. Structure and function of hemoglobin M E D I C I N E. Be like stem cells, differentiate yourself from others! Editing file PO 4.
 HbA NH 2 H 2 O 2 KClO 3 Cl 2 O 7 PO 4 CH2O NAOH KMnO 4 M E D I C I N E KING SAUD UNIVERSITY Co 2 COOH MgCl 2 H 2 O Important Extra Information Doctors slides Doctors notes SO 2 HCN CCl 4 CuCl 2 Biochemistry
HbA NH 2 H 2 O 2 KClO 3 Cl 2 O 7 PO 4 CH2O NAOH KMnO 4 M E D I C I N E KING SAUD UNIVERSITY Co 2 COOH MgCl 2 H 2 O Important Extra Information Doctors slides Doctors notes SO 2 HCN CCl 4 CuCl 2 Biochemistry
OpenStax-CNX module: m Transport of Gases. OpenStax College. Abstract
 OpenStax-CNX module: m46545 1 Transport of Gases OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will
OpenStax-CNX module: m46545 1 Transport of Gases OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will
Biochemistry 15 Doctor /7/2012
 Heme The Heme is a chemical structure that diffracts by light to give a red color. This chemical structure is introduced to more than one protein. So, a protein containing this heme will appear red in
Heme The Heme is a chemical structure that diffracts by light to give a red color. This chemical structure is introduced to more than one protein. So, a protein containing this heme will appear red in
Pharmacist. Drugs. body physiology. ( molecular constituents)
 Why? Pharmacist Drugs body physiology ( molecular constituents) Mechanistic levels of response: Altered patient response physiologic systems Vascular system blood, muscle, liver tissues / organs cellular
Why? Pharmacist Drugs body physiology ( molecular constituents) Mechanistic levels of response: Altered patient response physiologic systems Vascular system blood, muscle, liver tissues / organs cellular
4 Fahed Al Karmi Sufian Alhafez Dr nayef karadsheh
 4 Fahed Al Karmi Sufian Alhafez Dr nayef karadsheh Genetic variants of hemoglobin Hemoglobinopathies (abnormal variants of hemoglobin) are divided into: 1. Structural abnormalities: Any change in the genes
4 Fahed Al Karmi Sufian Alhafez Dr nayef karadsheh Genetic variants of hemoglobin Hemoglobinopathies (abnormal variants of hemoglobin) are divided into: 1. Structural abnormalities: Any change in the genes
May 2003: Hemoglobin Red Blood, Blue Blood Use and Abuse of Hemoglobin
 Red Blood, Blue Blood Ever wondered why blood vessels appear blue? Oxygenated blood is bright red: when you are cut, the blood you see is brilliant red oxygenated blood. Deoxygenated blood is deep purple:
Red Blood, Blue Blood Ever wondered why blood vessels appear blue? Oxygenated blood is bright red: when you are cut, the blood you see is brilliant red oxygenated blood. Deoxygenated blood is deep purple:
2018 Biochemistry 110 California Institute of Technology Lecture 7: Molecular Disease: Sickle-Cell Anemia
 2018 Biochemistry 110 California Institute of Technology Lecture 7: Molecular Disease: Sickle-Cell Anemia James Herrick (1861-1954) Phase-Contrast microscopy image of Sickle Cells intermingled with erythrocytes.
2018 Biochemistry 110 California Institute of Technology Lecture 7: Molecular Disease: Sickle-Cell Anemia James Herrick (1861-1954) Phase-Contrast microscopy image of Sickle Cells intermingled with erythrocytes.
HEMOGLOBINOPATHIES LECTURE OUTLINE. An overview of the structure of hemoglobin. Different types of hemoglobin. Definition of hemoglobinopathies
 Slide 1 HEOGLOBINOPATHIES Slide 2 LETURE OUTLINE An overview of the structure of hemoglobin. Different types of hemoglobin. Definition of hemoglobinopathies Sickle ell Disease and Hemoglobin Slide 3 HEOGLOBIN
Slide 1 HEOGLOBINOPATHIES Slide 2 LETURE OUTLINE An overview of the structure of hemoglobin. Different types of hemoglobin. Definition of hemoglobinopathies Sickle ell Disease and Hemoglobin Slide 3 HEOGLOBIN
Hemoglobin and hemoglobinpathies. Srbová M., Průša R.
 Hemoglobin and hemoglobinpathies Srbová M., Průša R. Hemoproteins Consist of hem cyclic tetrapyrrole 1 iron cation Fe 2+ bound in the middle of tetrapyrrole scelet by coordination covalent bonds conjugated
Hemoglobin and hemoglobinpathies Srbová M., Průša R. Hemoproteins Consist of hem cyclic tetrapyrrole 1 iron cation Fe 2+ bound in the middle of tetrapyrrole scelet by coordination covalent bonds conjugated
Introduction To Medical Technology. Hemoglobin Concentration Determination
 Introduction To Medical Technology Hemoglobin Concentration Determination Hemoglobin (Hb) Hemoglobin (Hb) is the standard abbreviation for hemoglobin, the oxygen-carrying pigment and predominant protein
Introduction To Medical Technology Hemoglobin Concentration Determination Hemoglobin (Hb) Hemoglobin (Hb) is the standard abbreviation for hemoglobin, the oxygen-carrying pigment and predominant protein
Haemoglobin BY: MUHAMMAD RADWAN WISSAM MUHAMMAD
 Haemoglobin BY: MUHAMMAD RADWAN WISSAM MUHAMMAD Introduction is the iron-containing oxygen transport metalloprotein in the red blood cells Hemoglobin in the blood carries oxygen from the respiratory organs
Haemoglobin BY: MUHAMMAD RADWAN WISSAM MUHAMMAD Introduction is the iron-containing oxygen transport metalloprotein in the red blood cells Hemoglobin in the blood carries oxygen from the respiratory organs
FUNCTIONS OF HEMOGLOBIN:
 HEMOGLOBIN: Conjugated protein Simple protein combined with a non-protein substance Hemoglobin HEME +GLOBIN nonprotein substance HEME( prosthetic group) Red colour of blood is due to Hb in RBCs Normal
HEMOGLOBIN: Conjugated protein Simple protein combined with a non-protein substance Hemoglobin HEME +GLOBIN nonprotein substance HEME( prosthetic group) Red colour of blood is due to Hb in RBCs Normal
O 2 O 2 O 2. Haemoglobin
 O 2 O 2 O 2 Haemoglobin O 2 O 2 O 2 98% travels in oxyhaemoglobin (in red blood cells) 2% is dissolved in plasma (compared to carbon dioxide, oxygen is relatively insoluble in plasma) O 2 is not very soluble
O 2 O 2 O 2 Haemoglobin O 2 O 2 O 2 98% travels in oxyhaemoglobin (in red blood cells) 2% is dissolved in plasma (compared to carbon dioxide, oxygen is relatively insoluble in plasma) O 2 is not very soluble
Ola Al-juneidi Abdel-Mu'ez Siyam. Dr. Nayef
 3 Ola Al-juneidi Abdel-Mu'ez Siyam Dr. Nayef Transport of CO 2 We have talked previously about the role of hemoglobin in the transport of oxygen and how it is regulated by various allosteric effectors,
3 Ola Al-juneidi Abdel-Mu'ez Siyam Dr. Nayef Transport of CO 2 We have talked previously about the role of hemoglobin in the transport of oxygen and how it is regulated by various allosteric effectors,
Transport of oxygen and carbon dioxide in body fluids. Circulation and Hearts. Circulation in vertebrates and invertebrates
 Circulation Transport of oxygen and carbon dioxide in body fluids Circulation and Hearts Circulation in vertebrates and invertebrates Respiratory pigments Increase the amount of oxygen carried by blood
Circulation Transport of oxygen and carbon dioxide in body fluids Circulation and Hearts Circulation in vertebrates and invertebrates Respiratory pigments Increase the amount of oxygen carried by blood
Mid term Hematology-2011 Lejan 2009\2010
 The correct statment: Maximal Activation of cytotoxic T-Cells involves both interactions through MHC-I & MHC-II 1) about the thalassemia major which of this is not true : a) HbA2 increases in B thalassemia
The correct statment: Maximal Activation of cytotoxic T-Cells involves both interactions through MHC-I & MHC-II 1) about the thalassemia major which of this is not true : a) HbA2 increases in B thalassemia
Amino acids & Protein Structure Chemwiki: Chapter , with most emphasis on 16.3, 16.4 and 16.6
 Amino acids & Protein Structure Chemwiki: Chapter 16. 16.1, 16.3-16.9 with most emphasis on 16.3, 16.4 and 16.6 1 1. Most jobs (except information storage) in cells are performed by proteins. 2. Proteins
Amino acids & Protein Structure Chemwiki: Chapter 16. 16.1, 16.3-16.9 with most emphasis on 16.3, 16.4 and 16.6 1 1. Most jobs (except information storage) in cells are performed by proteins. 2. Proteins
PHAR3316 Pharmacy biochemistry Exam #2 Fall 2010 KEY
 1. How many protons is(are) lost when the amino acid Asparagine is titrated from its fully protonated state to a fully deprotonated state? A. 0 B. 1 * C. 2 D. 3 E. none Correct Answer: C (this question
1. How many protons is(are) lost when the amino acid Asparagine is titrated from its fully protonated state to a fully deprotonated state? A. 0 B. 1 * C. 2 D. 3 E. none Correct Answer: C (this question
Module G: Oxygen Transport. Oxygen Transport. Dissolved Oxygen. Combined Oxygen. Topics to Cover
 Topics to Cover Module G: Oxygen Transport Oxygen Transport Oxygen Dissociation Curve Oxygen Transport Studies Tissue Hypoxia Cyanosis Polycythemia Oxygen Transport Oxygen is carried from the lungs to
Topics to Cover Module G: Oxygen Transport Oxygen Transport Oxygen Dissociation Curve Oxygen Transport Studies Tissue Hypoxia Cyanosis Polycythemia Oxygen Transport Oxygen is carried from the lungs to
Dr. Puntarica Suwanprathes. Version 2007
 Dr. Puntarica Suwanprathes Version 2007 O 2 and CO 2 transport in blood Oxyhemoglobin dissociation curve O 2 consumption (VO 2 ) CO 2 production (VCO 2 ) O 2 capacity O 2 content: CaO 2 or CvO 2 %saturation
Dr. Puntarica Suwanprathes Version 2007 O 2 and CO 2 transport in blood Oxyhemoglobin dissociation curve O 2 consumption (VO 2 ) CO 2 production (VCO 2 ) O 2 capacity O 2 content: CaO 2 or CvO 2 %saturation
Last time we finished the abnormal hemoglobins. We have some hemoglobin. 5 Abdel-muez siyam Abdullah nimer Dr. nayef. Page 1
 Last time we finished the abnormal hemoglobins. We have some hemoglobin 5 Abdel-muez siyam Abdullah nimer Dr. nayef Page 1 derivatives (mostly outside the book) that are of importance: 1. Normal derivatives:
Last time we finished the abnormal hemoglobins. We have some hemoglobin 5 Abdel-muez siyam Abdullah nimer Dr. nayef Page 1 derivatives (mostly outside the book) that are of importance: 1. Normal derivatives:
Hemoglobin and anemia BCH 471
 Hemoglobin and anemia BCH 471 OBJECTIVES Quantitative determination of hemoglobin in a blood sample. Hemoglobin structure Hemoglobin (Hb) is a porphyrin iron (II) protein in RBCs that transport oxygen
Hemoglobin and anemia BCH 471 OBJECTIVES Quantitative determination of hemoglobin in a blood sample. Hemoglobin structure Hemoglobin (Hb) is a porphyrin iron (II) protein in RBCs that transport oxygen
Biology A-level: Transport. Blood. Page 1 of 22 1/18/2009. Red blood cells
 Page 1 of 22 Home A-level Biology Transport Blood Biology A-level: Transport Blood Just over half of the blood volume is made up of a pale yellow fluid called plasma. The rest of the blood is made up of
Page 1 of 22 Home A-level Biology Transport Blood Biology A-level: Transport Blood Just over half of the blood volume is made up of a pale yellow fluid called plasma. The rest of the blood is made up of
Student number. University of Guelph Department of Chemistry and Biochemistry Structure and Function In Biochemistry
 University of Guelph Department of Chemistry and Biochemistry 19356 Structure and Function In Biochemistry Midterm Test, March 3, 1998. Time allowed, 90 min. Answer questions 120 on the computer scoring
University of Guelph Department of Chemistry and Biochemistry 19356 Structure and Function In Biochemistry Midterm Test, March 3, 1998. Time allowed, 90 min. Answer questions 120 on the computer scoring
Question Expected Answers Marks Additional Guidance 1 (a) C ; E ; A ; B ; 4. PhysicsAndMathsTutor.com
 Question Expected Answers Marks Additional Guidance 1 (a) C ; E ; A ; B ; 4 Question Expected Answers Marks Additional Guidance (b) (i) P wave combined with larger peak before QRS complex ; Note: - look
Question Expected Answers Marks Additional Guidance 1 (a) C ; E ; A ; B ; 4 Question Expected Answers Marks Additional Guidance (b) (i) P wave combined with larger peak before QRS complex ; Note: - look
Biological Sciences 4087 Exam I 9/20/11
 Name: Biological Sciences 4087 Exam I 9/20/11 Total: 100 points Be sure to include units where appropriate. Show all calculations. There are 5 pages and 11 questions. 1.(20pts)A. If ph = 4.6, [H + ] =
Name: Biological Sciences 4087 Exam I 9/20/11 Total: 100 points Be sure to include units where appropriate. Show all calculations. There are 5 pages and 11 questions. 1.(20pts)A. If ph = 4.6, [H + ] =
Chapter 34 Active Reading Guide Circulation and Gas Exchange
 Name: AP Biology Mr. Croft Chapter 34 Active Reading Guide Circulation and Gas Exchange Section 1 1. Gaining O 2 and nutrients while shedding CO 2 and other waste products occurs with every cell in the
Name: AP Biology Mr. Croft Chapter 34 Active Reading Guide Circulation and Gas Exchange Section 1 1. Gaining O 2 and nutrients while shedding CO 2 and other waste products occurs with every cell in the
Introduction to Biochemistry Midterm exam )ومن أحياها(
 Introduction to Biochemistry Midterm exam 2016-2017 )ومن أحياها( 1. Which of the following amino (in a peptide chain) would probably be found at a beta bend or turn? a. lysine * b. Gly c. arg d. asn 2.
Introduction to Biochemistry Midterm exam 2016-2017 )ومن أحياها( 1. Which of the following amino (in a peptide chain) would probably be found at a beta bend or turn? a. lysine * b. Gly c. arg d. asn 2.
GAS EXCHANGE IB TOPIC 6.4 CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM. Terminal bronchiole Nasal cavity. Pharynx Left lung Alveoli.
 IB TOPIC 6.4 GAS EXCHANGE CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM Branch from the pulmonary artery (oxygen-poor blood) Branch from the pulmonary vein (oxygen-rich blood) Terminal bronchiole Nasal
IB TOPIC 6.4 GAS EXCHANGE CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM Branch from the pulmonary artery (oxygen-poor blood) Branch from the pulmonary vein (oxygen-rich blood) Terminal bronchiole Nasal
IB TOPIC 6.4 GAS EXCHANGE
 IB TOPIC 6.4 GAS EXCHANGE CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM Branch from the pulmonary artery (oxygen-poor blood) Branch from the pulmonary vein (oxygen-rich blood) Terminal bronchiole Nasal
IB TOPIC 6.4 GAS EXCHANGE CARDIOPULMONARY SYSTEM CARDIOPULMONARY SYSTEM Branch from the pulmonary artery (oxygen-poor blood) Branch from the pulmonary vein (oxygen-rich blood) Terminal bronchiole Nasal
Rama Nada. -Ensherah Mokheemer. 1 P a g e
 - 3 - Rama Nada -Ensherah Mokheemer - 1 P a g e Don t forget to refer to page index wherever you see * Quick revision: In the previous lecture we said that: - your body contains 4-5g of iron (4g in females
- 3 - Rama Nada -Ensherah Mokheemer - 1 P a g e Don t forget to refer to page index wherever you see * Quick revision: In the previous lecture we said that: - your body contains 4-5g of iron (4g in females
ATP. Chapter 7, parts of 48 Cellular Respiration: Gas Exchange, Other Metabolites & Control of Respiration. Cellular Respiration
 Chapter 7, parts of 48 Cellular Respiration: Gas Exchange, Other Metabolites & Control of Respiration Cellular Respiration ATP Gas Exchange O 2 & CO 2 exchange provides O 2 for aerobic cellular respiration
Chapter 7, parts of 48 Cellular Respiration: Gas Exchange, Other Metabolites & Control of Respiration Cellular Respiration ATP Gas Exchange O 2 & CO 2 exchange provides O 2 for aerobic cellular respiration
Cellular Respiration Notes. Biology - Mrs. Kaye
 Cellular Respiration Notes Biology - Mrs. Kaye Energy Transfer In cellular respiration, chemical energy is converted into usable energy which is converted into heat energy. ATP and ADP ATP acts as an energy
Cellular Respiration Notes Biology - Mrs. Kaye Energy Transfer In cellular respiration, chemical energy is converted into usable energy which is converted into heat energy. ATP and ADP ATP acts as an energy
االمتحان النهائي لعام 1122
 االمتحان النهائي لعام 1122 Amino Acids : 1- which of the following amino acid is unlikely to be found in an alpha-helix due to its cyclic structure : -phenylalanine -tryptophan -proline -lysine 2- : assuming
االمتحان النهائي لعام 1122 Amino Acids : 1- which of the following amino acid is unlikely to be found in an alpha-helix due to its cyclic structure : -phenylalanine -tryptophan -proline -lysine 2- : assuming
Biochemistry by Mary K. Campbell & Shawn O. Farrell
 4 Biochemistry by Mary K. Campbell & Shawn O. Farrell 4-1 4 The ThreeDimensional Structure of Proteins 4-2 4 Learning Objectives 1. How does the Structure of Proteins Determine Their Function? 2. What
4 Biochemistry by Mary K. Campbell & Shawn O. Farrell 4-1 4 The ThreeDimensional Structure of Proteins 4-2 4 Learning Objectives 1. How does the Structure of Proteins Determine Their Function? 2. What
Porphyrins: Chemistry and Biology
 Porphyrins: Chemistry and Biology 20.109 Lecture 6 24 February, 2011 Goals Explore some essential roles of heme in biology Appreciate how ature has used the same cofactor to achieve diverse functions Gain
Porphyrins: Chemistry and Biology 20.109 Lecture 6 24 February, 2011 Goals Explore some essential roles of heme in biology Appreciate how ature has used the same cofactor to achieve diverse functions Gain
بسم هللا الرحمن الرحيم
 بسم هللا الرحمن الرحيم Q1: the overall folding of a single protein subunit is called : -tertiary structure -primary structure -secondary structure -quaternary structure -all of the above Q2 : disulfide
بسم هللا الرحمن الرحيم Q1: the overall folding of a single protein subunit is called : -tertiary structure -primary structure -secondary structure -quaternary structure -all of the above Q2 : disulfide
Hemoglobin. Each alpha subunit has 141 amino acids, and each beta subunit has 146 amino acids.
 In the previous lecture we talked about erythropoiesis and its regulation by many vitamins like vitamin B12 and folic acid, proteins, iron and trace elements copper and cobalt. Also we talked about pernicious
In the previous lecture we talked about erythropoiesis and its regulation by many vitamins like vitamin B12 and folic acid, proteins, iron and trace elements copper and cobalt. Also we talked about pernicious
Nafith Abu Tarboush DDS, MSc, PhD
 Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Types of proteins Proteins can be divided into two groups according to structure: Fibrous (fiber-like with a uniform secondary-structure
Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Types of proteins Proteins can be divided into two groups according to structure: Fibrous (fiber-like with a uniform secondary-structure
Cellular Respiration Assignment
 I. MULTIPLE CHOICES. Select the best answer. Write the letter of your choice on the space provided. 1. Electron transport chain allows the electron to. a) transfer from grana to stroma. b) transfer from
I. MULTIPLE CHOICES. Select the best answer. Write the letter of your choice on the space provided. 1. Electron transport chain allows the electron to. a) transfer from grana to stroma. b) transfer from
OVERVIEW OF RESPIRATION AND LOOSE ENDS. What agents? What war?
 5.19.06 OVERVIEW OF RESPIRATION AND LOOSE ENDS What agents? What war? 1 Ubiquinone or Coenzyme Q: small hydrophobic molecule that can pick up or donate electrons The respiratory chain contains 3 large
5.19.06 OVERVIEW OF RESPIRATION AND LOOSE ENDS What agents? What war? 1 Ubiquinone or Coenzyme Q: small hydrophobic molecule that can pick up or donate electrons The respiratory chain contains 3 large
Protein Structure and Function
 Protein Structure and Function Protein Structure Classification of Proteins Based on Components Simple proteins - Proteins containing only polypeptides Conjugated proteins - Proteins containing nonpolypeptide
Protein Structure and Function Protein Structure Classification of Proteins Based on Components Simple proteins - Proteins containing only polypeptides Conjugated proteins - Proteins containing nonpolypeptide
Red Blood Cells (Erythrocytes) Lecture-2
 Red Blood Cells (Erythrocytes) Lecture-2 Functions Transport hemoglobin, which in turn carries oxygen from the lungs to the tissues. RBCs contain a large quantity of carbonic anhydrase, an enzyme that
Red Blood Cells (Erythrocytes) Lecture-2 Functions Transport hemoglobin, which in turn carries oxygen from the lungs to the tissues. RBCs contain a large quantity of carbonic anhydrase, an enzyme that
G = Energy of activation
 Biochemistry I, CHEM 4400 Exam 2 10:00 Fall 2015 Dr. Stone Name rate forward = k forward [reactants] rate reverse = k reverse [products] K eq = [products]/[reactants] ΔG = ΔH - TΔS ΔG = ΔG + RT ln Q Q
Biochemistry I, CHEM 4400 Exam 2 10:00 Fall 2015 Dr. Stone Name rate forward = k forward [reactants] rate reverse = k reverse [products] K eq = [products]/[reactants] ΔG = ΔH - TΔS ΔG = ΔG + RT ln Q Q
4. THE THREE-DIMENSIONAL STRUCTURE OF PROTEINS
 4. THE THREE-DIMENSIONAL STRUCTURE OF PROTEINS 4.1 Proteins Structures and Function Levels of Structure in Proteins Native conformation - Biological activity - Random structure: no obvious regular repeating
4. THE THREE-DIMENSIONAL STRUCTURE OF PROTEINS 4.1 Proteins Structures and Function Levels of Structure in Proteins Native conformation - Biological activity - Random structure: no obvious regular repeating
Around million aged erythrocytes/hour are broken down.
 Anemia Degradation ofheme Around 100 200 million aged erythrocytes/hour are broken down. The degradation process starts in reticuloendothelial cells in the spleen, liver, and bone marrow. [1] The tetrapyrrole
Anemia Degradation ofheme Around 100 200 million aged erythrocytes/hour are broken down. The degradation process starts in reticuloendothelial cells in the spleen, liver, and bone marrow. [1] The tetrapyrrole
Batool Emad. Marah Karablieh. - Nayef Karadsheh
 4 4 1 P a g e Batool Emad Marah Karablieh - Nayef Karadsheh ***Topics that will be discussed in this Lecture: 1) Globin gene organization 2) Hemoglobinopathies 3) HbS (sickle cell disease) 4) HbC and HbSC
4 4 1 P a g e Batool Emad Marah Karablieh - Nayef Karadsheh ***Topics that will be discussed in this Lecture: 1) Globin gene organization 2) Hemoglobinopathies 3) HbS (sickle cell disease) 4) HbC and HbSC
Sheet #8 Dr. Nafeth Abu-Tarboush 13/07/2014
 Done by 1 Ali Khresat Structure-function relationship of proteins we have talked about proteins, the structure of proteins and features of proteins now we will talk about how this structure is related
Done by 1 Ali Khresat Structure-function relationship of proteins we have talked about proteins, the structure of proteins and features of proteins now we will talk about how this structure is related
The Respiratory System
 I. Organization of the Respiratory System The Respiratory System The respiratory system allows the circulation of air and the gas exchange between the body and the outside environment. It consists of the
I. Organization of the Respiratory System The Respiratory System The respiratory system allows the circulation of air and the gas exchange between the body and the outside environment. It consists of the
Disaccharides. Compound dehydration synthesis puts sugars together Hydrolysis (hydro-water, lysisbreakdown)
 Carbohydrate Carbo-hydrate -carbon, water Cn(H2O) n Monosaccharides Hexose hex = 6 [carbons], "-ose" means sugar Glucose monosaccaccharide usually assume a ring structure Disaccharides Compound dehydration
Carbohydrate Carbo-hydrate -carbon, water Cn(H2O) n Monosaccharides Hexose hex = 6 [carbons], "-ose" means sugar Glucose monosaccaccharide usually assume a ring structure Disaccharides Compound dehydration
Introductory slide with title and major topics. Footer with last name, course and year on each slide
 Introductory slide with title and major topics Footer with last name, course and year on each slide From your undergraduate education, summer biochemistry pre- matriculation studies or prep work for this
Introductory slide with title and major topics Footer with last name, course and year on each slide From your undergraduate education, summer biochemistry pre- matriculation studies or prep work for this
Mammalian body fluids - Blood A suspension of cells in a pale yellow fluid called plasma
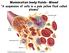 Mammalian body fluids - Blood A suspension of cells in a pale yellow fluid called plasma A = Red blood cell B, C and D = White blood cells Composition of blood: The blood - carries out several vital jobs:
Mammalian body fluids - Blood A suspension of cells in a pale yellow fluid called plasma A = Red blood cell B, C and D = White blood cells Composition of blood: The blood - carries out several vital jobs:
2008MSC'STRUCTURAL'BIOCHEMISTRY'
 LearningObjectives: 2008MSC'STRUCTURAL'BIOCHEMISTRY' Module'1' 'Amino'Acids'and'Proteins:'Structure'and'Function' 1 Understandthestructureandstereochemistryofalpha1aminoacids 1 Beabletodescribetheclassesofaminoacidsandtheirproperties
LearningObjectives: 2008MSC'STRUCTURAL'BIOCHEMISTRY' Module'1' 'Amino'Acids'and'Proteins:'Structure'and'Function' 1 Understandthestructureandstereochemistryofalpha1aminoacids 1 Beabletodescribetheclassesofaminoacidsandtheirproperties
v o = V max [S] rate = kt[s] e V max = k cat E t ΔG = -RT lnk eq K m + [S]
![v o = V max [S] rate = kt[s] e V max = k cat E t ΔG = -RT lnk eq K m + [S] v o = V max [S] rate = kt[s] e V max = k cat E t ΔG = -RT lnk eq K m + [S]](/thumbs/81/84404059.jpg) Exam 3 Spring 2017 Dr. Stone 8:00 Name There are 100 possible points on this exam. -ΔG / RT v o = V max [S] rate = kt[s] e V max = k cat E t ΔG = -RT lnk eq K m + [S] h rate forward = k forward [reactants]
Exam 3 Spring 2017 Dr. Stone 8:00 Name There are 100 possible points on this exam. -ΔG / RT v o = V max [S] rate = kt[s] e V max = k cat E t ΔG = -RT lnk eq K m + [S] h rate forward = k forward [reactants]
Table of Contents. Section 1 Glycolysis and Fermentation. Section 2 Aerobic Respiration
 Table of Contents Section 1 Glycolysis and Fermentation Section 2 Aerobic Respiration Objectives Identify the two major steps of cellular respiration. Describe the major events in glycolysis. Compare lactic
Table of Contents Section 1 Glycolysis and Fermentation Section 2 Aerobic Respiration Objectives Identify the two major steps of cellular respiration. Describe the major events in glycolysis. Compare lactic
Anatomy and Physiology
 Anatomy and Physiology For The First Class 2 nd Semester Erythrocytes = Red Blood Cells (RBC) Erythrocytes = Red Blood Cells Red blood cells are biconcave discs, they have no nucleus and cytoplasmic organelles.
Anatomy and Physiology For The First Class 2 nd Semester Erythrocytes = Red Blood Cells (RBC) Erythrocytes = Red Blood Cells Red blood cells are biconcave discs, they have no nucleus and cytoplasmic organelles.
Energy Production In A Cell (Chapter 25 Metabolism)
 Energy Production In A Cell (Chapter 25 Metabolism) Large food molecules contain a lot of potential energy in the form of chemical bonds but it requires a lot of work to liberate the energy. Cells need
Energy Production In A Cell (Chapter 25 Metabolism) Large food molecules contain a lot of potential energy in the form of chemical bonds but it requires a lot of work to liberate the energy. Cells need
Chapter 10 Respiration
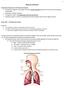 1 Chapter 10 Respiration Introduction/Importance of the Respiratory System All eukaryotic organisms need oxygen to perform cellular respiration (production of ATP), either aerobically or anaerobically.
1 Chapter 10 Respiration Introduction/Importance of the Respiratory System All eukaryotic organisms need oxygen to perform cellular respiration (production of ATP), either aerobically or anaerobically.
Human Circulation and Respiration Chapter 38
 Human Circulation and Respiration Chapter 38 Goals: 1. Compare closed and open circulatory systems. 2. Label the parts of the human heart. 3. Explain how blood travels through the heart; identify chambers
Human Circulation and Respiration Chapter 38 Goals: 1. Compare closed and open circulatory systems. 2. Label the parts of the human heart. 3. Explain how blood travels through the heart; identify chambers
How Cells Release Chemical Energy. Chapter 8
 How Cells Release Chemical Energy Chapter 8 Impacts, Issues: When Mitochondria Spin Their Wheels More than forty disorders related to defective mitochondria are known (such as Friedreich s ataxia); many
How Cells Release Chemical Energy Chapter 8 Impacts, Issues: When Mitochondria Spin Their Wheels More than forty disorders related to defective mitochondria are known (such as Friedreich s ataxia); many
Find this material useful? You can help our team to keep this site up and bring you even more content consider donating via the link on our site.
 Find this material useful? You can help our team to keep this site up and bring you even more content consider donating via the link on our site. Still having trouble understanding the material? Check
Find this material useful? You can help our team to keep this site up and bring you even more content consider donating via the link on our site. Still having trouble understanding the material? Check
Topics of this lecture : RBC. Structural characteristics Hemoglobin Erythropoiesis Erythrocytes destruction
 Topics of this lecture : RBC Structural characteristics Hemoglobin Erythropoiesis Erythrocytes destruction Structural characteristics Its small size and biconcave shape provides more surface area than
Topics of this lecture : RBC Structural characteristics Hemoglobin Erythropoiesis Erythrocytes destruction Structural characteristics Its small size and biconcave shape provides more surface area than
thebiotutor.com AS Biology Unit 2 Exchange & Transport
 thebiotutor.com AS Biology Unit 2 Exchange & Transport 1 Exchange of materials Oxygen and Carbon dioxide are obtained passively by simple diffusion Fick s law The rate of diffusion = concentration difference
thebiotutor.com AS Biology Unit 2 Exchange & Transport 1 Exchange of materials Oxygen and Carbon dioxide are obtained passively by simple diffusion Fick s law The rate of diffusion = concentration difference
Student number. University of Guelph Department of Chemistry and Biochemistry Structure and Function In Biochemistry
 University of Guelph Department of Chemistry and Biochemistry 19356 Structure and Function In Biochemistry Midterm Test, March 3, 1998. Time allowed, 90 min. Answer questions 120 on the computer scoring
University of Guelph Department of Chemistry and Biochemistry 19356 Structure and Function In Biochemistry Midterm Test, March 3, 1998. Time allowed, 90 min. Answer questions 120 on the computer scoring
Chapter 15. Enzyme Regulation. Activity? Part 1 Factors that influence enzymatic activity
 Chapter 15 Enzyme Regulation http://lms.ls.ntou.edu.tw/course/106ls tw/course/106 hanjia@mail.ntou.edu.tw Reginald H. Garrett Charles M. Grisham Essential Questions Before this class, ask your self the
Chapter 15 Enzyme Regulation http://lms.ls.ntou.edu.tw/course/106ls tw/course/106 hanjia@mail.ntou.edu.tw Reginald H. Garrett Charles M. Grisham Essential Questions Before this class, ask your self the
Mammalian Transport and The Heart
 Cardiovascular System AS-G, Chapters 8-9 Blood flows through the body in a closed system (circuit) driven by the pumping power of the heart Closed vs open: does the system have vessels contained the entire
Cardiovascular System AS-G, Chapters 8-9 Blood flows through the body in a closed system (circuit) driven by the pumping power of the heart Closed vs open: does the system have vessels contained the entire
Haemoglobinopathies case studies 11 th Annual Sickle Cell and Thalassaemia Conference October 2017
 Haemoglobinopathies case studies 11 th Annual Sickle Cell and Thalassaemia Conference 11 13 October 2017 Chris Lambert Haematology Service Delivery Manager Viapath Laboratories Kings College Hospital HUMAN
Haemoglobinopathies case studies 11 th Annual Sickle Cell and Thalassaemia Conference 11 13 October 2017 Chris Lambert Haematology Service Delivery Manager Viapath Laboratories Kings College Hospital HUMAN
Chapter Seven (Cellular Respiration)
 Chapter Seven (Cellular Respiration) 1 SECTION ONE: GLYCOLYSIS AND FERMENTATION HARVESTING CHEMICAL ENERGY Cellular respiration is the process in which cells make adenosine triphosphate (ATP) by breaking
Chapter Seven (Cellular Respiration) 1 SECTION ONE: GLYCOLYSIS AND FERMENTATION HARVESTING CHEMICAL ENERGY Cellular respiration is the process in which cells make adenosine triphosphate (ATP) by breaking
Bio Factsheet April 2000 Number 66
 April Number 66 The Physiology of Exercise This factsheet summarises the aspects of exercise physiology that relate to skeletal muscles on current syllabuses. The student should have a basic knowledge
April Number 66 The Physiology of Exercise This factsheet summarises the aspects of exercise physiology that relate to skeletal muscles on current syllabuses. The student should have a basic knowledge
Factors affecting oxygen dissociation curve
 P a g e 1 Factors affecting oxygen dissociation curve As you know, hemoglobin contains 4 heme molecules that bind 4 oxygen molecules (8 atoms). These 4 heme molecules, however, do not bind oxygen all at
P a g e 1 Factors affecting oxygen dissociation curve As you know, hemoglobin contains 4 heme molecules that bind 4 oxygen molecules (8 atoms). These 4 heme molecules, however, do not bind oxygen all at
BIOLOGY - CLUTCH CH.42 - CIRCULATORY SYSTEM.
 !! www.clutchprep.com CONCEPT: GAS EXCHANGE AND CIRCULATION Respiratory system draws in gases from the environment, intakes O2 and outputs CO2 Circulatory system transports O2, CO2, nutrients, hormones,
!! www.clutchprep.com CONCEPT: GAS EXCHANGE AND CIRCULATION Respiratory system draws in gases from the environment, intakes O2 and outputs CO2 Circulatory system transports O2, CO2, nutrients, hormones,
Lec.2 Medical Physiology Blood Physiology Z.H.Kamil
 Destruction of Red Blood Cells When red blood cells are delivered from the bone marrow into the circulatory system, they normally circulate an average of 120 days before being destroyed. Even though mature
Destruction of Red Blood Cells When red blood cells are delivered from the bone marrow into the circulatory system, they normally circulate an average of 120 days before being destroyed. Even though mature
3.7 CELLULAR RESPIRATION. How are these two images related?
 3.7 CELLULAR RESPIRATION How are these two images related? CELLULAR RESPIRATION Cellular respiration is the process whereby the body converts the energy that we get from food (glucose) into an energy form
3.7 CELLULAR RESPIRATION How are these two images related? CELLULAR RESPIRATION Cellular respiration is the process whereby the body converts the energy that we get from food (glucose) into an energy form
Chem*3560 Lecture 4: Inherited modifications in hemoglobin
 Chem*3560 Lecture 4: Inherited modifications in hemoglobin Genetic modifications fall into two classes: Thalassemias, which are the result of failure to express globin genes. Thalassa is Greek for the
Chem*3560 Lecture 4: Inherited modifications in hemoglobin Genetic modifications fall into two classes: Thalassemias, which are the result of failure to express globin genes. Thalassa is Greek for the
3.7.1 Define cell respiration [Cell respiration is the controlled release of energy from organic compounds in cells to form ATP]
![3.7.1 Define cell respiration [Cell respiration is the controlled release of energy from organic compounds in cells to form ATP] 3.7.1 Define cell respiration [Cell respiration is the controlled release of energy from organic compounds in cells to form ATP]](/thumbs/72/66350985.jpg) 3.7 Cell respiration ( Chapter 9 in Campbell's book) 3.7.1 Define cell respiration [Cell respiration is the controlled release of energy from organic compounds in cells to form ATP] Organic compounds store
3.7 Cell respiration ( Chapter 9 in Campbell's book) 3.7.1 Define cell respiration [Cell respiration is the controlled release of energy from organic compounds in cells to form ATP] Organic compounds store
Respiratory System. Introduction. Atmosphere. Some Properties of Gases. Human Respiratory System. Introduction
 Introduction Respiratory System Energy that we consume in our food is temporarily stored in the bonds of ATP (adenosine triphosphate) before being used by the cell. Cells use ATP for movement and to drive
Introduction Respiratory System Energy that we consume in our food is temporarily stored in the bonds of ATP (adenosine triphosphate) before being used by the cell. Cells use ATP for movement and to drive
AEROBIC RESPIRATION. Chapter 8
 AEROBIC RESPIRATION Chapter 8 AEROBIC RESPIRATION Aerobic respiration is the next step after Glycolysis if the cell can obtain oxygen. We won t need it until the last step but we still need it. Remember
AEROBIC RESPIRATION Chapter 8 AEROBIC RESPIRATION Aerobic respiration is the next step after Glycolysis if the cell can obtain oxygen. We won t need it until the last step but we still need it. Remember
CELLULAR RESPIRATION. Chapter 7
 CELLULAR RESPIRATION Chapter 7 7.1 GLYCOLYSIS AND FERMENTATION If I have a $10.00 bill and a $10.00 check, which is better? ATP is like cash in the cell Glucose, NADH, FADH2 are like checks in a cell.
CELLULAR RESPIRATION Chapter 7 7.1 GLYCOLYSIS AND FERMENTATION If I have a $10.00 bill and a $10.00 check, which is better? ATP is like cash in the cell Glucose, NADH, FADH2 are like checks in a cell.
10. The diagram below shows two different kinds of substances, A and B, entering a cell.
 1. In the binomial system of nomenclature, which two classification groups provide the scientific name of an organism? A) kingdom and phylum B) phylum and species C) kingdom and genus D) genus and species
1. In the binomial system of nomenclature, which two classification groups provide the scientific name of an organism? A) kingdom and phylum B) phylum and species C) kingdom and genus D) genus and species
CIE Biology A-level Topic 8: Transport in mammals
 CIE Biology A-level Topic 8: Transport in mammals Notes Circulatory systems can either be open, for instance in insects, or closed, like in fish and mammals where the blood is confined to blood vessels
CIE Biology A-level Topic 8: Transport in mammals Notes Circulatory systems can either be open, for instance in insects, or closed, like in fish and mammals where the blood is confined to blood vessels
Cellular Respiration
 Cellular Respiration How do living things use chemical reactions to get energy from food? Version November 2017 1 How do we get our energy from food? 1 The connection between food and energy We ve figured
Cellular Respiration How do living things use chemical reactions to get energy from food? Version November 2017 1 How do we get our energy from food? 1 The connection between food and energy We ve figured
4/5/17. Blood. Blood. Outline. Blood: An Overview. Functions of Blood
 Outline Blood Biol 105 Chapter 11 I. Overview of blood II. Functions of blood III. Composition of blood IV. Composition of plasma V. Composition of formed elements VI. Platelets VII. White blood cells
Outline Blood Biol 105 Chapter 11 I. Overview of blood II. Functions of blood III. Composition of blood IV. Composition of plasma V. Composition of formed elements VI. Platelets VII. White blood cells
Ch. 9 Cell Respiration. Title: Oct 15 3:24 PM (1 of 53)
 Ch. 9 Cell Respiration Title: Oct 15 3:24 PM (1 of 53) Essential question: How do cells use stored chemical energy in organic molecules and to generate ATP? Title: Oct 15 3:28 PM (2 of 53) Title: Oct 19
Ch. 9 Cell Respiration Title: Oct 15 3:24 PM (1 of 53) Essential question: How do cells use stored chemical energy in organic molecules and to generate ATP? Title: Oct 15 3:28 PM (2 of 53) Title: Oct 19
MULTIPLE CHOICE QUESTIONS
 MULTIPLE CHOICE QUESTIONS 1. Which of the following statements concerning anabolic reactions is FALSE? A. They are generally endergonic. B. They usually require ATP. C. They are part of metabolism. D.
MULTIPLE CHOICE QUESTIONS 1. Which of the following statements concerning anabolic reactions is FALSE? A. They are generally endergonic. B. They usually require ATP. C. They are part of metabolism. D.
Cellular Respiration: Obtaining Energy from Food
 Chapter 6 Cellular Respiration: Obtaining Energy from Food Lectures by Chris C. Romero, updated by Edward J. Zalisko PowerPoint Lectures for Campbell Essential Biology, Fourth Edition Eric Simon, Jane
Chapter 6 Cellular Respiration: Obtaining Energy from Food Lectures by Chris C. Romero, updated by Edward J. Zalisko PowerPoint Lectures for Campbell Essential Biology, Fourth Edition Eric Simon, Jane
Name Class Date. 1. Cellular respiration is the process by which the of "food"
 Name Class Date Cell Respiration Introduction Cellular respiration is the process by which the chemical energy of "food" molecules is released and partially captured in the form of ATP. Carbohydrates,
Name Class Date Cell Respiration Introduction Cellular respiration is the process by which the chemical energy of "food" molecules is released and partially captured in the form of ATP. Carbohydrates,
Hemoglobin Properties of Egyptian Obese Smokers: Effect of Combination of Obesity and Smoking
 Australian Journal of Basic and Applied Sciences, (4): 135-1330, 008 ISSN 1991-8178 Hemoglobin Properties of Egyptian Obese Smokers: Effect of Combination of Obesity and Smoking Nahed S. Hassan Department
Australian Journal of Basic and Applied Sciences, (4): 135-1330, 008 ISSN 1991-8178 Hemoglobin Properties of Egyptian Obese Smokers: Effect of Combination of Obesity and Smoking Nahed S. Hassan Department
DONE BY : RaSHA RAKAN & Bushra Saleem
 DONE BY : RaSHA RAKAN & Bushra Saleem Hemolytic anemias (2 of 2) Sickle Cell Anemia The most common familial hemolytic anemia in the world Sickle cell anemia is the prototypical (and most prevalent) hemoglobinopathy
DONE BY : RaSHA RAKAN & Bushra Saleem Hemolytic anemias (2 of 2) Sickle Cell Anemia The most common familial hemolytic anemia in the world Sickle cell anemia is the prototypical (and most prevalent) hemoglobinopathy
