Appeal decision. Appeal No Canada TRANSLATUM MEDICUS INC. Tokyo, Japan
|
|
|
- Geraldine Rich
- 6 years ago
- Views:
Transcription
1 Appeal decision Appeal No Canada Appellant TRANSLATUM MEDICUS INC Tokyo, Japan Patent Attorney TAKAOKA, Ryoichi The case of appeal against the examiner's decision of refusal of Japanese Patent Application No "Methods for Treating and Diagnosing Blinding Eye Diseases" [international publication date on November 7, 2013, WO2013/ and nationally published on August 13, 2015, National Publication of International Patent Application No ] has resulted in the following appeal decision: Conclusion The appeal of the case was groundless. Reason No. 1 History of the procedures The application was originally filed on April 30, 2013 as an International Patent Application (priority claim under the Paris Convention: May 1, 2012 (US), May 2, 2012 (US), August 24, 2012 (US), March 15, 2013 (US)), and the history of the further procedures is as follows: April 25, 2016 May 24, 2016 August 30, 2016 written amendment notification of reasons for refusal written opinion, written amendment, and supplemental 1 / 25
2 statement October 13, 2016 February 17, 2017 February 17, 2017 March 29, 2017 June 9, 2017 examiner's decision of refusal request for appeal written amendment written amendment (formality) reexamination report by Examiner before appeal No. 2 Decision to Dismiss Amendment submitted on February 17, 2017 [Conclusion of Decision to Dismiss Amendment] The amendment submitted on February 17, 2017 shall be dismissed. [Reason] 1. The details of the amendment submitted on February 17, 2017 The amendment submitted on February 17, 2017 (hereinafter referred to as "the Amendment") is to amend Claim 1 before the Amendment, "[Claim 1] Use of a compound of Formula I or a pharmaceutically acceptable salt thereof for manufacturing a medicament for reducing the rate of progression to dry age-related macular degeneration (AMD) in the hyaloid body, [Chem. 1] C1-C6 alkyl." wherein each of R1 and R2 is independently H or C1-C6 alkyl, and R3 is H or 2 / 25
3 to "[Claim 1] Use of a compound of Formula I or a pharmaceutically acceptable salt thereof for manufacturing a medicament for reducing the rate of progression to dry age-related macular degeneration (AMD) in the hyaloid body, [Chem. 1] wherein each of R1 and R2 is independently H or C1-C6 alkyl, and R3 is H or C1-C6 alkyl, wherein the dry age-related macular degeneration (AMD) is diagnosed by detecting one or more of autofluorescent structures in the retinal pigment epithelium (RPE)." 2. Propriety of amendment The amendment regarding Claim 1 is to limit "dry age-related macular degeneration (AMD)" before the amendment to dry age-related macular degeneration (AMD) "that is diagnosed by detecting one or more of autofluorescent structures in the retinal pigment epithelium (RPE)," and thus the amendment is applicable to restrict the scope of claims. Whether the appellant shall be granted a patent for the invention described in Claim 1 after the Amendment (hereinafter referred to as "Amended Invention") independently at the time of filing the application (whether the Amended Invention falls under the provisions of Article 126(7) of the Patent Act which is applied mutatis 3 / 25
4 mutandis pursuant to the provisions of Article 17-2(6) of the Patent Act) will be examined. (1) Amended Invention As described in the above 1., the Amended Invention is as follows. "[Claim 1] Use of a compound of Formula I or a pharmaceutically acceptable salt thereof for manufacturing a medicament for reducing the rate of progression to dry age-related macular degeneration (AMD) in the hyaloid body, [Chem. 1] wherein each of R1 and R2 is independently H or C1-C6 alkyl, and R3 is H or C1-C6 alkyl, wherein the dry age-related macular degeneration (AMD) is diagnosed by detecting one or more of autofluorescent structures in the retinal pigment epithelium (RPE)." (2) Article 36(4)(i) of the Patent Act A. Introduction Article 36(4) of the Patent Act provides that "The statement of the detailed explanation of the invention as provided in item (iii) of the preceding Paragraph shall comply with each of the following items," and Article 36(4)(i) of the Patent Act requires that "in accordance with Ordinance of the Ministry of Economy, Trade and Industry, the statement shall be clear and sufficient in such a manner as to enable any person skilled 4 / 25
5 in the art to which the invention pertains to work the invention." (what is called enablement requirements) In addition, the Amended Invention relates to "use for manufacturing a medicament for reducing the rate of progression to dry age-related macular degeneration (AMD) in the hyaloid body" and to medicinal use, and as it is generally difficult for medicinal use to predict the utility only from the name or chemical structure, even when the effective amount, method for administration, or matters for formulation are described in the detailed description of the invention, a person skilled in the art cannot understand whether the medicament actually has utility of the medicinal use, and thus there is need to describe pharmacological data or description equivalent to the pharmacological data in the detailed description of the invention for supporting utility of medicinal use, and if such description is not present, it should be said that the application does not meet enablement requirements. This point will be examined as follows. B. The detailed description of the invention The following matters are described in the detailed description of the invention. (a) "[0002] This invention relates to methods and compositions that are useful for the diagnosis, treatment, or prevention of a blinding eye disease, including the discovery of drugs that are efficacious against these diseases." ([0002]) (b) "[0008] In another aspect, the invention provides a method for treating or preventing dry AMD, comprising administering to a subject in need thereof an effective amount of a compound of Formula I: [Chem. 1] 5 / 25
6 or a pharmaceutically acceptable salt thereof, wherein each of R1 and R2 is independently H or a C1-C6 alkyl and R3 is H or a C1-C6 alkyl." ([0008]) (c) "[0031] In another aspect, the invention provides a method for treating or preventing dry AMD, comprising administering to a subject in need thereof an effective amount of a compound of Formula I: [Chem. 2] or a pharmaceutically acceptable salt thereof, wherein each of R1 and R2 is independently H or a C1-C6 alkyl and R3 is H or a C1-C6 alkyl. In one embodiment, the compound of Formula I is bindarit." ([0031]) (d) "[0207] When ophthalmically administered to a human, for example, intravitreally, the 6 / 25
7 dosage of an agent of the invention, including, for example, Formula I, methotrexate or a pharmaceutically acceptable salt thereof and/or additional therapeutic agent is normally mg to 5.0 mg per eye per administration, or 0.03 mg to 3.0 mg per eye per administration, or 0.1 mg to 1.0 mg per eye per administration. In one embodiment, the dosage is 0.03 mg, 0.3 mg, 1.5 mg, or 3.0 mg per eye. In another embodiment, the dosage is 0.5 mg per eye. The dosage can range from 0.01 ml to 0.2 ml administered per eye, or 0.03 ml to 0.15 ml administered per eye, or 0.05 ml to 0.10 ml administered per eye. In one embodiment, the administration is 400 µg of compound, monthly for at least three months. [0208] Generally, when orally administered to a mammal, the dosage of any agent described herein may be mg/kg/day to 100 mg/kg/day, 0.01 mg/kg/day to 50 mg/kg/day, or 0.1 mg/kg/day to 10 mg/kg/day. When orally administered to a human, the dosage of any agent described herein is normally mg to 1000 mg per day, 1 mg to 600 mg per day, or 5 mg to 30 mg per day. In one embodiment, oral dosage is 600 mg per day. In one embodiment, the oral dosage is two 300 mg doses per day. In another embodiment, oral dosage is 7.5 mg per week to 15 mg per week. [0209] For administration of any agent described herein by parenteral injection, the dosage is normally 0.1 mg to 250 mg per day, 1 mg to 20 mg per day, or 3 mg to 5 mg per day. Injections may be given up to four times daily. Generally, when orally or parenterally administered, the dosage of any agent described herein is normally 0.1 mg to 1500 mg per day, or 0.5 mg to 10 mg per day, or 0.5 mg to 5 mg per day. A dosage of up to 3000 mg per day can be administered. [0210] In some embodiments, it may be desirable to administer one or more of any agent described herein to the eye. Administration may be, by way of non-limiting example, intra-ocular, intra-vitreal, topical (including, but not limited to, drops and ointment), sub-conjunctival, sub-tenon's, trans-scleral, suprachoroidal, subretinal, or via iontophoresis. 7 / 25
8 [0211] Other routes of administration may also be used, such as, for example: intradermal, intramuscular, intraperitoneal, intravenous, subcutaneous, intranasal, epidural, oral, sublingual, intranasal, intracerebral, intravaginal, transdermal, rectally, by inhalation, or topically, particularly to the ears, nose, eyes, or skin. [0212] The mode of administration can be left to the discretion of the practitioner, and depends in part upon the site of the medical condition. In most instances, administration results in the release of any agent described herein into the bloodstream. [0213] Any agent described herein can be administered orally. Such agents can also be administered by any other conventional route, for example, by intravenous infusion or bolus injection, by absorption through epithelial or mucocutaneous linings (e.g., oral mucosa, rectal and intestinal mucosa, etc.), and can be administered together with another biologically active agent. Administration can be systemic or local. Various delivery systems are known, e.g., encapsulation in liposomes, microparticles, microcapsules, capsules, etc., and can be used for administration. [0214] Further methods of administration include but are not limited to intra-ocular, intra-vitreal, topical ocular (including but not limited to drops, ointments, and inserts), sub-conjunctival, sub-tenon's, suprachoroidal, trans-scleral, intradermal, intramuscular, intraperitoneal, intravenous, subcutaneous, intranasal, epidural, oral, sublingual, intranasal, intracerebral, intravaginal, transdermal, rectally, by inhalation, or topically, particularly to the ears, nose, eyes, or skin. In some embodiments, more than one of any agent described herein is administered to the eye. Administration may be, by way of non-limiting example, intra-ocular, intra-vitreal, topical (including, but not limited to, drops, and ointment), sub-conjunctival, sub-tenon's, trans-scleral, or iontophoresis. The mode of administration can be left to the discretion of the practitioner, and depends in part upon the site of the medical condition. In most instances, administration results in release into the bloodstream. 8 / 25
9 [0215] In specific embodiments, it may be desirable to administer locally to the area in need of treatment. [0216] In another embodiment, delivery can be in a vesicle, in particular a liposome (see Langer, 1990, Science 249: ; Treat et al., in Liposomes in the Therapy of Infectious Disease and Cancer, Lopez-Berestein and Fidler (eds.), Liss, New York, pp (1989)). In yet another embodiment, delivery can be in a controlled-release system. In one embodiment, a slow release intraocular device may be used. In some embodiments, this device consists of a locally delivered erodible or non-erodable liquid, gel, polymer, etc. [0217] In another embodiment, polymeric materials can be used (see Medical Applications of Controlled Release, Langer and Wise (eds.), CRC Pres., Boca Raton, Florida (1974); Controlled Drug Bioavailability, Drug Product Design and Performance, Smolen and Ball (eds.), Wiley, New York (1984); Ranger and Peppas, 1983, J. Macromol. Sci. Rev. Macromol. Chem. 23:61; see also Levy et al., 1985, Science 228: 190; During et al., 1989, Ann. Neurol. 25:351; Howard et al., 1989, J. Neurosurg. 71: 105). In another embodiment, a controlled-release system can be placed in proximity of the target area to be treated, e.g., the retina, thus requiring only a fraction of the systemic dose (see, e.g., Goodson, in Medical Applications of Controlled Release, supra, vol. 2, pp (1984)). Other controlled-release systems discussed in the review by Langer, 1990, Science 249: ) may also be used. [0218] Administration of any agent described herein can, independently, be one to four times daily or one to four times per month, or one to six times per year, or once every two, three, four, or five years. Administration can be for the duration of one day or one month, two months, three months, six months, one year, two years, three years, and may even be for the life of the subject. Chronic, long-term administration will be indicated in many cases. The dosage may be administered as a single dose or divided into 9 / 25
10 multiple doses. In general, the desired dosage should be administered at set intervals for a prolonged period, usually at least over several weeks or months, although longer periods of administration of several months or years or more may be needed. [0219] The dosage regimen utilizing any agent described herein can be selected in accordance with a variety of factors, including type, species, age, weight, sex, and medical condition of the subject; the severity of the condition to be treated, the route of administration; the renal or hepatic function of the subject; the pharmacogenomic makeup of the individual; and the specific compound of the invention employed. Any agent described herein can be administered in a single daily dose, or the total daily dosage can be administered in divided doses of two, three, or four times daily. Furthermore, any agent described herein can be administered continuously rather than intermittently throughout the dosage regimen. Methods of Treatment [0220] In various aspects, the present invention provides a method for treating or preventing dry AMD and/or RPD. In these aspects, the "agent of the invention" comprises compounds useful for both monotherapy and combination therapy (e.g. as an additional therapeutic agent). In general, monotherapy comprises the use of compounds of Formula I, methotrexate, or their pharmaceutically acceptable salts, while combination therapy comprises compounds of Formula I, methotrexate, or their pharmaceutically acceptable salts in combination with an additional therapeutic agent, including one or more of an anti-vegf agent, an ACE inhibitor, a PPAR-γ agonist, a renin inhibitor, a steroid, an agent that modulates autophagy PPARγ modulator, semapimod, an MIF inhibitor, a CCR2 inhibitor, CKR-2B, a 2-thioimidazole, CAS , CCX140, clodronate, a clodonate-liposome preparation, or gadolinium chloride." ([0207] to [0220]) (e) "[0250] This invention is further illustrated by way of the following non-limiting 10 / 25
11 examples. (Examples) (Example 1): Systemic Injection of the RPE Toxin, NaIO3, induces complex patterns of FAF similar to those of AMD and/or RPD. (...Omitted...) (Example 2): DNIRA of a rat eye after systemic ICG administration identifies the retinal pigment epithelial (RPE) layer in vivo. (...Omitted...) (Example 3): Discovery of compounds for the treatment of blinding eye diseases (...Omitted...) (Example 4): Assessing the activity of compounds for blinding eye diseases (...Omitted...)" ([0250] to [0286]) (f) "(Example 5): Treatment of dry AMD with bindarit [0287] Human subjects, 56 to 100 years of age or more, presented with dry AMD, as diagnosed by one or more of the following clinical tests: clinical examination, FAF (at any wavelength), near infrared and/or red-free photography, fluorescein angiography, which allows for the identification and localization of abnormal vascular processes; OCT, which provides high-resolution, cross-sectional, or face images from within optical scattering media, such as the human retina and choroid; and structured illumination light microscopy, using a specially designed high resolution microscope setup to resolve the fluorescent distribution of small autofluorescent structures (lipofuscin granule) in retinal pigment epithelium tissue sections. [0288] The subjects were administered bindarit in two 300 mg oral doses once a day for 12 weeks. After an initial twelve-week treatment period, the subjects were evaluated for clinical outcomes. Alternatively, patients received intravitreal injection of a vehicle containing bindarit, with or without a drug delivery vehicle. [0289] 11 / 25
12 A first clinical outcome is determined using a standard visual acuity test, as is well known in the art. The subjects are assessed for the ability to clearly see symbols and objects on a Snellen eye chart from a distance. [0290] A second clinical outcome assesses the rate of progression of geographic atrophy. To do so, the subjects' pupils are dilated with 1.0% tropicamide and 2.5% phenylephrine before retinal imaging. Imaging is carried out with an instrument (e.g., Spectralis HRA+OCT; Heidelberg Engineering, Heidelberg, Germany) that allows for simultaneous recording of cslo and spectral-domain optical coherence tomography (SD-OCT) with two independent scanning mirrors, as described in Helb, et al. Acta Ophthalmol Dec; 88(8): Five modes of operation are employed: white light, red-free light, near infrared, FAF, and OCT. [0291] cslo images are obtained according to a standardized operation protocol that includes the acquisition of near-infrared reflectance (λ = 815 nm) and FAF (excitation at λ = 488 nm, emission nm) images. Simultaneous SD-OCT imaging is carried out with an illumination wavelength of 870 nm, an acquisition speed of 40,000 A-scans, and a scan depth of 1.8 mm. Two SD-OCT scans, one vertical and one horizontal, per eye are performed through the approximate foveal center, or in the case of RPD, in proximity to the vascular arcades of the macula. Fluorescein angiography (λ = 488 nm, emission nm, 10% fluorescein dye) is performed as needed. Color fundus photographs are obtained with a fundus camera (e.g. FF 450 Visupac ZK5; Carl Zeiss Meditec AG, Jena, Germany). [0292] Interpretation of clinical outcome data informs a decision for further treatment, if any." ([0286] to [0292]) (g) "(Example 6): Treatment of dry AMD with a combination therapy [0293] Human subjects, 56 to 100 years of age or more, presented with dry AMD, as 12 / 25
13 diagnosed by one or more of the following clinical tests: clinical examination, whitelight fundus imaging, FAF at any wavelength, near infrared and/or red-free photography, blue-light illumination, and/or fluorescein or ICG angiography, which allows for the identification and localization of abnormal vascular processes; OCT, which provides high-resolution, cross-sectional, three-dimensional, and face images from within optical scattering media, such as the human retina and choroid; and structured illumination light microscopy, using a specially designed high resolution microscope or ophthalmoscope set up to resolve the distribution of small autofluorescent structures (lipofuscin, lipofuscin-like, or other granule) in the retinal pigment epithelium or other cells and cell layers. [0294] The subjects were administered bindarit in two 300 mg oral doses once a day for 12 weeks. The subjects were also administered ranibizumab injection once per month (roughly 28 days) in a dose of 0.5 mg per affected eye. After an initial twelve-week treatment period, the subjects were evaluated for clinical outcomes. [0295] A first clinical outcome is determined using a standard visual acuity test, as is well known in the art. The subjects are assessed for the ability to clearly see symbols and objects on a Snellen eye chart from a distance. [0296] A second clinical outcome assesses the rate of progression of geographic atrophy. To do so, the subjects' pupils are dilated with 1.0% tropicamide and 2.5% phenylephrine or a comparable agent before retinal imaging. Imaging is carried out with an instrument (e.g., Spectralis HRA+OCT; Heidelberg Engineering, Heidelberg, Germany) that allows for simultaneous recording of cslo and SD-OCT, as described in Helb, et al. Acta Ophthalmol Dec; 88(8): Multiple modes of operation can be employed: white light, red-free light, blue light, near infrared, and OCT. Similar analysis can be performed with a modified fundus camera. [0297] cslo images are obtained according to protocols known in the art that may 13 / 25
14 include the acquisition of near-infrared reflectance (λ = nm) and FAF (excitation at λ = nm, emission nm) images. Simultaneous SD-OCT imaging is carried out with, for example, an illumination wavelength of 870 nm, an acquisition speed of 40,000 A-scans, and a scan depth of 1.8 mm. Multiple SD-OCT scans per eye are performed through the macula and additionally or in the case of RPD, in proximity to the vascular arcades of the macula. Other OCT imaging, such as, for example, time domain and swept domain, can also be used. Fluorescein angiography (λ = 488 nm, emission nm, 10% fluorescein dye) is performed as needed. Color fundus photographs are obtained with a fundus camera (e.g. FF 450 Visupac ZK5; Carl Zeiss Meditec AG, Jena, Germany). [0298] Interpretation of clinical outcome data informs a decision for further treatment, if any. Illustrative data analysis includes macular cube analysis and 5 line raster." ([0292] to [0298]) (h) "(Example 7): Detection and/or prediction of a blinding eye disease subject response to an agent (...Omitted...) (Example 8): DNIRA of a rat eye after systemic ICG administration labels immune cells in vivo (...Omitted...) Example 9: Clinical in vivo imaging of RPD and Diffuse trickling AMD: (...Omitted...)" ([0298] to [0328]) C. Judgment Regarding methods and compositions that are useful for the diagnosis, treatment, or prevention of a blinding eye disease, including the discovery of drugs that are efficacious against these diseases (summarized matter (a)), a method for treating or preventing dry AMD, comprising administering to a subject in need thereof an effective amount of the compound of Formula I or a pharmaceutically acceptable salt thereof ("a 14 / 25
15 compound of Formula I or a pharmaceutically acceptable salt thereof" may be hereinafter collectively referred to as "a compound of Formula I") is described in the detailed description of the invention (summarized matter (b)), and the effective amount and method for administration are also described to some extent (summarized matter (d)); however as described in the above "A," it cannot be understood whether the compound of Formula I of the Amended Invention is actually useful in "use for manufacturing a medicament for reducing the rate of progression to dry age-related macular degeneration (AMD) in the hyaloid body." In addition, examining whether pharmacological data supporting the utility or description equivalent to the pharmacological data are described in the detailed description of the invention, it is described in Examples 5 and 6 in the detailed description of the invention that human subjects with dry AMD were administered bindarit in two 300 mg oral doses once a day for 12 weeks or received intravitreal injection of a vehicle containing bindarit, and a method for evaluating the administration is also described (summarized matters (f) and (g)), and thus it can be said that a method for administering bindarit, which is a compound of Formula I (summarized matter (c)), to a subject with dry AMD and evaluating the administration is described in the detailed description of the invention. However, it is not described that the evaluation method has actually been performed, and there is no description whether the compound of Formula I has been found capable of "reducing the rates of progression to dry age-related macular degeneration (AMD) in the hyaloid body" from the result of evaluation. That is, it cannot be said that Examples 5 and 6 are pharmacological data actually supporting the utility of "use of the compound of Formula I for manufacturing a medicament for reducing the rates of progression to dry age-related macular degeneration (AMD) in the hyaloid body," and there is no description equivalent to the pharmacological data supporting the utility. Further, Examples other than Examples 5 and 6 are "(Example 1): Systemic Injection of the RPE Toxin, NaIO3, induces complex patterns of FAF similar to those of AMD and/or RPD, (Example 2): DNIRA of a rat eye after systemic ICG administration 15 / 25
16 identifies the retinal pigment epithelial (RPE) layer in vivo, (Example 3): Discovery of compounds for the treatment of blinding eye diseases, (Example 4): Assessing the activity of compounds for blinding eye diseases, (Example 7): Detection and/or prediction of a blinding eye disease subject response to an agent, (Example 8): DNIRA of a rat eye after systemic ICG administration labels immune cells in vivo, (Example 9): Clinical in vivo imaging of RPD and Diffuse trickling AMD" (summarized matters (e) and (h)), and these Examples are not related to the compound of Formula I, and thus these Examples are not pharmacological data actually supporting the utility of "use of the compound of Formula I for manufacturing a medicament for reducing the rates of progression to dry age-related macular degeneration (AMD) in the hyaloid body," and there is no description equivalent to the pharmacological data supporting the utility. In addition, examining the other detailed description of the invention, there is no description of pharmacological data actually supporting the utility of "use of the compound of Formula I for manufacturing a medicament for reducing the rates of progression to dry age-related macular degeneration (AMD) in the hyaloid body," and there is no description equivalent to the pharmacological data supporting the utility. As described above, since pharmacological data supporting the utility or description equivalent to the pharmacological data is not described in the detailed description of the invention, the detailed description of the invention is not clear and sufficient to enable a person skilled in the art to carry out the Amended Invention of "use of the compound of Formula I for manufacturing a medicament for reducing the rates of progression to dry age-related macular degeneration (AMD) in the hyaloid body." D. Summary Therefore, the detailed description of the invention of the invention does not comply with the provision of Article 36(4)(i) of the Patent Act, and thus the Amended Invention should not be independently patentable at the time of patent application. (3) Article 36(6)(i) of the Patent Act 16 / 25
17 A. Introduction Article 36(6) of the Patent Act provides "The statement of claims as provided in paragraph (2) shall comply with each of the following items," and Article 36(6)(i) of the Patent Act provides "The invention for which a patent is sought is stated in the detailed description of the invention." Whether the statement in the claims satisfies the requirement stipulated in Article 36(6)(i) of the Patent Act (what is called support requirements) is determined by comparing the claimed invention and the detailed description of the invention, and by determining whether the claimed invention is the invention described in the detailed description of the invention and is within the extent in the description to which a person skilled in the art would recognize that a problem to be solved by the invention would actually be solved, or is within the extent in the description to which a person skilled in the art would recognize that a problem to be solved by the invention would actually be solved by taking into account common general technical knowledge at the time of filing the application even if the problem is not described or indicated, and thus this point will be examined. B. Judgment It is described in the detailed description of the invention that the invention provides a method for treating or preventing dry AMD, comprising administering to a subject in need thereof an effective amount of a compound of Formula I (summarized matters (b) and (c)), and taking into account the matters specifying the Amended Invention, it can be said that the problem to be solved by the Amended Invention is to provide "use for manufacturing a medicament for reducing the rate of progression to dry age-related macular degeneration (AMD) in the hyaloid body." However, as described in the above (2)C., there is no description, in the detailed description of the invention, of pharmacological data actually supporting the utility of the compound of Formula I in "use for manufacturing a medicament for reducing the rate of progression to dry age-related macular degeneration (AMD) in the hyaloid body" or description equivalent to the pharmacological data, it cannot be said that a person skilled in the art would recognize that the problem is to be solved with the detailed 17 / 25
18 description of the invention. In addition, even if pharmacological data or description equivalent to the pharmacological data is not described the detailed description of the invention, there was no common general technical knowledge at the time of filing the application that a person skilled in the art was capable of "use of a compound of Formula I for manufacturing a medicament for reducing the rate of progression to dry age-related macular degeneration (AMD) in the hyaloid body," and would recognize that the problem is to be solved. Thus, there is no description in the detailed description of the invention that a person skilled in the art would recognize that the problem is to be solved by the Amended Invention, and there is no description that a person skilled in the art would recognize that the problem is to be solved by taking into account common general technical knowledge at the time of filing the application even if the problem is not described or indicated, and the Amended Invention is not described in the detailed description of the invention. C. Summary Therefore, since the description of claims according to the Amended Invention does not comply with the provision of Article 36(6)(i) of the Patent Act, the Amended Invention should not be independently patentable at the time of patent application. (4) Appellant's allegation The appellant alleges in the written opinion submitted on August 30, 2016 that the detailed description of the invention is clear and sufficient to enable a person skilled in the art to carry out the Amended Invention, and the Amended Invention is described in the detailed description of the invention, pointing out the following points. "As described in Example 5 of the application, on the basis of the evaluation in animal models for dry age-related macular degeneration (AMD) (Examples 1-2), intravitreal injection of bindarit is used to treat a subject with dry AMD. As evidence 18 / 25
19 showing surprising effect of bindarit on treatment of dry AMD, including reducing the rate of progression to dry AMD and the rate of progression of geographic atrophy, the appellant submits experimental data (US written oath (Evidence A No. 1)) by Doctor Boyd, who is the inventor of this application. As is clear from the experimental data, monotherapy by intravitreal injection of bindarit surprisingly and significantly reduced progression of patch of geographic atrophy indicating blinding complication and disease progression after dry AMD. At Time 1 (left panel) of both Figures A and B of the experimental data, patch of geographic atrophy is present in a rat's eye. Figure A shows treatment of patch present in an animal model of the present invention with negative control (saline). As shown at Time 2 of Figure A, the patch treated with saline was expanded (arrow in right panel). In contrast, as shown at Time 2 of Figure B, the patch treated with bindarit was not expanded (no arrow in right panel). These data are summarized in Figure C. There is no progression of geographic atrophy in all animals receiving bindarit in eye. Pharmacological effect of a compound of the Invention can be evaluated by using the animal model described in Examples 1 and 2 and performing the method described in Examples 5 and 6, and thus a person skilled in the art can obtain the above data easily. Therefore, the appellant considers that the detailed description of the invention is clear and sufficient to enable a person skilled in the art to carry out the Invention, and the Invention is described in the detailed description of the invention." However, the above data are not described in the detailed description of the invention and were not common general technical knowledge at the time of filing the application, and thus if the detailed description of the invention meets the requirement stipulated in Article 36(4)(i) of the Patent Act and the claimed invention meets the requirement stipulated in Article 36(6)(i) of the Patent Act by submitting the above data after filing the application, the submission violates the object of Patent Act employing the so-called first-to-file system and the submission is not accepted. 19 / 25
20 Therefore, any of the appellant's allegation cannot be accepted. In addition, the appellant alleges in the grounds for the appeal amended on March 29, 2017 that the detailed description of the invention is clear and sufficient to enable a person skilled in the art to carry out the Amended Invention, and the Amended Invention is described in the detailed description of the invention, pointing out the following points. "According to the above amendment, 'dry age-related macular degeneration (AMD)' described in Claims 1 and 6 is limited to the dry age-related macular degeneration (AMD) 'diagnosed by detecting one or more of autofluorescent structures in the retinal pigment epithelium (RPE).' The amendment is to reflect the matters described in the detailed description of the invention of the application. Namely, the amendment is to reflect the matters for specifically evaluating the effect of a compound of the invention by detecting one or more of autofluorescent structures in the retinal pigment epithelium (RPE) in use of the compound and a method for reducing the rate of progression to dry age-related macular degeneration according to the invention, to the claimed invention 1 and 6. For example, this method is disclosed in Examples 1, 2, 8 and Figures 3A, 3B, 3C, 4A, 4B, 4C, 5, 6, 7A, 7B, 8A, 8B, 8C, 9A, 9B, 10 of the application. Detecting one or more of autofluorescent structures in the retinal pigment epithelium (RPE) is specifically described in Example 1. In addition, on the basis of the evaluation in dry age-related macular degeneration (AMD) disclosed in Example 1, as described in Example 5, it is thought that a person skilled in the art can use intravitreal injection of bindarit for treating patients with dry AMD. Therefore, the appellant believes that the claimed inventions after the Amendment are described in the detailed description of the invention of the application to be clear and sufficient to enable a person skilled in the art to carry out the claimed inventions after the Amendment." Examining the above allegation, even though a method for detecting one or 20 / 25
21 more of autofluorescent structures in the retinal pigment epithelium (RPE) is described in Examples 1, 2, 8 and Figures 3A, 3B, 3C, 4A, 4B, 4C, 5, 6, 7A, 7B, 8A, 8B, 8C, 9A, 9B, 10, there is no relationship between a method for detecting an autofluorescent structure and the actual utility of the compound of Formula I in "use for manufacturing a medicament for reducing the rate of progression to dry age-related macular degeneration (AMD) in the hyaloid body," and on the basis of the description, in the detailed description of the invention, of the method for detecting an autofluorescent structure, it cannot be said that there is description of pharmacological data actually supporting the utility of the compound of Formula I in "use for manufacturing a medicament for reducing the rate of progression to dry age-related macular degeneration (AMD) in the hyaloid body" or description equivalent to the pharmacological data. In addition, even though an animal model is described in Example 1 and a method for evaluation is described in Example 5, as described in the above (2)C, it cannot be said that there is description of pharmacological data actually supporting the utility of the compound of Formula I in "use for manufacturing a medicament for reducing the rate of progression to dry age-related macular degeneration (AMD) in the hyaloid body" or description equivalent to the pharmacological data. Thus, the detailed description of the invention is not clear and sufficient to enable a person skilled in the art to carry out the Amended Invention, and the Amended Invention is not described in the detailed description of the invention. Therefore, any of the appellant's allegations cannot be accepted. 3. Closing regarding the Amendment As described above, since the Amended Invention should not be independently patentable at the time of patent application, the Amendment regarding Claim 1 violates the provisions of Article 126(7) of the Patent Act which is applied mutatis mutandis pursuant to the provisions of Article 17-2(6) of the Patent Act. Therefore, the Amendment shall be dismissed under the provisions of Article 53(1) of the Patent Act applied mutatis mutandis by replacing certain terms pursuant to 21 / 25
22 Article 159(1) of the Patent Act. No. 3 The Invention 1. The Invention As described above, since the Amendment shall be dismissed, the inventions of the application are inventions specified by the matters described in Claims 1 to 10 amended by written amendment submitted on August 30, 2016, and the invention according to Claim 1 (hereinafter referred to as "the Invention") is as follows. "[Claim 1] Use of a compound of Formula I or a pharmaceutically acceptable salt thereof for manufacturing a medicament for reducing the rate of progression to dry age-related macular degeneration (AMD) in the hyaloid body, [Chem. 1] wherein each of R1 and R2 is independently H or C1-C6 alkyl, and R3 is H or C1-C6 alkyl." 2. Reasons for refusal stated in the examiner's decision Refusal stated in the examiner's decision is "The application should be rejected based on Reasons 2 and 3 described in the notification of reasons for refusal issued on May 24, 2016," and the "Reasons 2 and 3" described of reasons for refusal are as follows. 22 / 25
23 "2. (enablement requirements) For the following reasons, the detailed description of the invention of this application does not meet the requirement stipulated in Article 36(4)(i) of the Patent Act. 3. (support requirements) For the following reasons, the claimed invention of this application does not meet the requirement stipulated in Article 36(6)(i) of the Patent Act. (...Omitted...) Note * Regarding Reason 2 (enablement requirements) and Reason 3 (support requirements) - Claims 1 to 10 The inventions according to Claims 1 to 10 are regarded as inventions relating to medicinal use of a compound of Formula (I). However, regarding a pharmacological test supporting the medicinal use, although methods for the pharmacological test are described in Examples 5 and 6 in the detailed description of the invention (paragraphs [0287] to [0298]), the result of pharmacological test is not specifically disclosed. Meanwhile, in the field of pharmaceutical compounds, it is difficult in common general technical knowledge to predict pharmacological effect of a compound from only a structure of the compound, and there is no explanation in the detailed description of the invention based on technical evidence of a relationship between a structure of the compound of Formula (I) and therapeutic activity in dry age-related macular degeneration, and it cannot be said that use of the compound as an agent for treating dry age-related macular degeneration can be predicted with common general technical knowledge at the time of filing the application, and thus it cannot be said that use of the compound as the therapeutic agent is described in the detailed description of the invention. Thus, it cannot be said that the detailed description of the invention of the application is clear and sufficient to enable a person skilled in the art to carry out the inventions according to the claims. 23 / 25
24 In addition, taking into account the detailed description of the invention and common general technical knowledge, it cannot be said that the detailed description of the invention of the application can be understood by a person skilled in the art such that the problem of the invention to provide a compound of Formula (I) as an agent for treating dry age-related macular degeneration is to be solved. Therefore, since the claimed inventions are beyond the range supported by the detailed description of the invention, it cannot be said that the claimed inventions are described in the detailed description of the invention." 3. Judgment by the body As described in the reasons for refusal stated in the examiner's decision, since the detailed description of the invention is not clear and sufficient to enable a person skilled in the art to carry out the Invention, the detailed description of the invention does not comply with provision of Article 36(4)(i) of the Patent Act, and since the Invention is not described in the detailed description of the invention, the claimed invention does not comply with the provision of Article 36(6)(i) of the Patent Act. Thus, the application should be rejected. The reasons are as follows. The Invention does not include the matters that "the dry age-related macular degeneration (AMD) is diagnosed by detecting one or more of autofluorescent structures in the retinal pigment epithelium (RPE)," which is the matters specifying the Amended Invention, and it is obvious that the Invention includes a method "wherein the dry age-related macular degeneration (AMD) is diagnosed by detecting one or more of autofluorescent structures in the retinal pigment epithelium (RPE)." In addition, as described in the above "No (2)," the detailed description of the invention is not clear and sufficient to enable a person skilled in the art to carry out the Amended Invention, and based on a similar reason, the detailed description of the invention is not clear and sufficient to enable a person skilled in the art to carry out the Invention, and thus the detailed description of the invention does not comply with the 24 / 25
25 provision of Article 36(4)(i) of the Patent Act. Further, as described in the above "No (3)," the Amended Invention is not described in the detailed description of the invention, and based on a similar reason, the Invention is not described in the detailed description of the invention, and thus the claimed invention does not comply with the provision of Article 36(6)(i) of the Patent Act. No. 4 Closing As described above, the application should be rejected, since the detailed description of the invention does not comply with provision of Article 36(4)(i) of the Patent Act, and the claimed invention does not comply with the provision of Article 36(6)(i) of the Patent Act. Therefore, the appeal decision shall be made as described in the conclusion. September 11, 2017 Chief administrative judge: NAITO, Shinichi Administrative judge: INOUE, Chiyako Administrative judge: ASANO, Mina 25 / 25
On Different Wavelengths: The Spectrum of Retinal Imaging. On Different Wavelengths: The Spectrum of Retinal Imaging. Wavelength Specific Imaging
 On Different Wavelengths: The Spectrum of Retinal Imaging Timothy J. Bennett, CRA, FOPS, OCT-C Penn State Hershey Eye Center Hershey, PA On Different Wavelengths: The Spectrum of Retinal Imaging Wavelengths
On Different Wavelengths: The Spectrum of Retinal Imaging Timothy J. Bennett, CRA, FOPS, OCT-C Penn State Hershey Eye Center Hershey, PA On Different Wavelengths: The Spectrum of Retinal Imaging Wavelengths
Fundus Autofluorescence
 Brittany Bateman, BS Fundus autofluorescence imaging is used to record fluorescence that may occur naturally in ocular structures or as a byproduct of a disease process. This technique allows the topographic
Brittany Bateman, BS Fundus autofluorescence imaging is used to record fluorescence that may occur naturally in ocular structures or as a byproduct of a disease process. This technique allows the topographic
4/19/2018 FUNDUS AUTOFLUORESCENCE. Fluorescence Imaging. Fundus Autofluorescence (FAF) Fluorescence. Fluorescence
 I have no financial or proprietary interest in the subject matter of this presentation. FUNDUS AUTOFLUORESCENCE Timothy J. Bennett, CRA, OCT-C, FOPS Penn State Eye Center Hershey, PA Fluorescence Imaging
I have no financial or proprietary interest in the subject matter of this presentation. FUNDUS AUTOFLUORESCENCE Timothy J. Bennett, CRA, OCT-C, FOPS Penn State Eye Center Hershey, PA Fluorescence Imaging
Appeal decision. Appeal No USA ATRIUM MEDICAL CORPORATION. Osaka, Japan. Osaka, Japan. Osaka, Japan. Osaka, Japan
 Appeal decision Appeal No. 2014-13097 USA Appellant ATRIUM MEDICAL CORPORATION Osaka, Japan Patent Attorney YAMAMOTO, Shusaku Osaka, Japan Patent Attorney MORISHITA, Natsuki Osaka, Japan Patent Attorney
Appeal decision Appeal No. 2014-13097 USA Appellant ATRIUM MEDICAL CORPORATION Osaka, Japan Patent Attorney YAMAMOTO, Shusaku Osaka, Japan Patent Attorney MORISHITA, Natsuki Osaka, Japan Patent Attorney
Appeal decision. Appeal No USA HILL'S PET NUTRITION, INC. Osaka, Japan
 Appeal decision Appeal No. 2015-17056 USA Appellant HILL'S PET NUTRITION, INC. Osaka, Japan Patent Attorney MURAI, Koji The case of appeal against the examiner's decision of refusal of Japanese Patent
Appeal decision Appeal No. 2015-17056 USA Appellant HILL'S PET NUTRITION, INC. Osaka, Japan Patent Attorney MURAI, Koji The case of appeal against the examiner's decision of refusal of Japanese Patent
Cirrus TM HD-OCT. Details define your decisions
 Cirrus TM HD-OCT Details define your decisions 2 With high-definition OCT Carl Zeiss Meditec takes you beyond standard spectral domain Built on 10 years experience at the vanguard of innovation, Carl Zeiss
Cirrus TM HD-OCT Details define your decisions 2 With high-definition OCT Carl Zeiss Meditec takes you beyond standard spectral domain Built on 10 years experience at the vanguard of innovation, Carl Zeiss
Cirrus TM HD-OCT. Details defi ne your decisions
 Cirrus TM HD-OCT Details defi ne your decisions 2 With high-defi nition OCT Carl Zeiss Meditec takes you beyond standard spectral domain Built on 10 years experience at the vanguard of innovation, Carl
Cirrus TM HD-OCT Details defi ne your decisions 2 With high-defi nition OCT Carl Zeiss Meditec takes you beyond standard spectral domain Built on 10 years experience at the vanguard of innovation, Carl
LEE EYE CENTRE. YOUR VISION, OUR PASSION LEC EyeNews
 LEE EYE CENTRE YOUR VISION, OUR PASSION LEC EyeNews FOR INTERNAL CIRCULATION ONLY www.lec.com.my ISSUE 51/003 SEPT OCT 2017 The American Society of Cataract and Refractive Surgery is one of the leading
LEE EYE CENTRE YOUR VISION, OUR PASSION LEC EyeNews FOR INTERNAL CIRCULATION ONLY www.lec.com.my ISSUE 51/003 SEPT OCT 2017 The American Society of Cataract and Refractive Surgery is one of the leading
HHS Public Access Author manuscript Ophthalmic Surg Lasers Imaging Retina. Author manuscript; available in PMC 2016 January 14.
 High-Speed Ultrahigh-Resolution OCT of Bruch s Membrane in Membranoproliferative Glomerulonephritis Type 2 Mehreen Adhi, MD, Sarah P. Read, MD, PhD, Jonathan J. Liu, PhD, James G. Fujimoto, PhD, and Jay
High-Speed Ultrahigh-Resolution OCT of Bruch s Membrane in Membranoproliferative Glomerulonephritis Type 2 Mehreen Adhi, MD, Sarah P. Read, MD, PhD, Jonathan J. Liu, PhD, James G. Fujimoto, PhD, and Jay
THE OPHTHALMOLOGIST S NEEDS FOR THE ANALYSIS OF THE RETINA
 biophotonics end-users needs THE OPHTHALMOLOGIST S NEEDS FOR THE ANALYSIS OF THE RETINA Dr Matonti Frédéric CHU Nord / INT AMU Marseille ANATOMY OF THE RETINA ANATOMY OF THE RETINA ANATOMY OF THE RETINA
biophotonics end-users needs THE OPHTHALMOLOGIST S NEEDS FOR THE ANALYSIS OF THE RETINA Dr Matonti Frédéric CHU Nord / INT AMU Marseille ANATOMY OF THE RETINA ANATOMY OF THE RETINA ANATOMY OF THE RETINA
Flore De Bats, 1 Benjamin Wolff, 2,3 Martine Mauget-Faÿsse, 2 Claire Scemama, 2 and Laurent Kodjikian Introduction
 Case Reports in Medicine Volume 2013, Article ID 260237, 7 pages http://dx.doi.org/10.1155/2013/260237 Case Report B-Scan and En-Face Spectral-Domain Optical Coherence Tomography Imaging for the Diagnosis
Case Reports in Medicine Volume 2013, Article ID 260237, 7 pages http://dx.doi.org/10.1155/2013/260237 Case Report B-Scan and En-Face Spectral-Domain Optical Coherence Tomography Imaging for the Diagnosis
Diagnosis in AMD. Managing your AMD Patients
 Managing your AMD Patients Robert W. Dunphy, O.D., F.A.A.O. Diagnosis in AMD Have suspicion Identify relative risk Conduct surveillance Biometry Utilize technology to facilitate detection of change / stability
Managing your AMD Patients Robert W. Dunphy, O.D., F.A.A.O. Diagnosis in AMD Have suspicion Identify relative risk Conduct surveillance Biometry Utilize technology to facilitate detection of change / stability
Manual. Manual Welsh Eye Care Initiative. A Welsh Eye Care Initiative. Protocol. The Assessment and Management of Age-related Macular Degeneration
 A Protocol 1.0 Definitions The following terms are important in this text: Wet Macular Degeneration Condition caused by the growth of abnormal blood vessels under the retina. Symptoms appear suddenly and
A Protocol 1.0 Definitions The following terms are important in this text: Wet Macular Degeneration Condition caused by the growth of abnormal blood vessels under the retina. Symptoms appear suddenly and
Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D.
 Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D. Disclosure OCT Optical Coherence Tomography No relevant financial relationships I will refer to
Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D. Disclosure OCT Optical Coherence Tomography No relevant financial relationships I will refer to
Fundus Autofluorescence. Jonathan A. Micieli, MD Valérie Biousse, MD
 Fundus Autofluorescence Jonathan A. Micieli, MD Valérie Biousse, MD The retinal pigment epithelium (RPE) has many important functions including phagocytosis of the photoreceptor outer segments Cone Rod
Fundus Autofluorescence Jonathan A. Micieli, MD Valérie Biousse, MD The retinal pigment epithelium (RPE) has many important functions including phagocytosis of the photoreceptor outer segments Cone Rod
Widefield Retinal Imaging with Auto Fluorescence Technology in the Optometric Practice
 Widefield Retinal Imaging with Auto Fluorescence Technology in the Optometric Practice This course will define ultra-widefield retinal imaging and autofluorescence for the attendee. Will show how it is
Widefield Retinal Imaging with Auto Fluorescence Technology in the Optometric Practice This course will define ultra-widefield retinal imaging and autofluorescence for the attendee. Will show how it is
Acquired vitelliform detachment in patients with subretinal drusenoid deposits (reticular pseudodrusen)
 Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2011 Acquired vitelliform detachment in patients with subretinal drusenoid
Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2011 Acquired vitelliform detachment in patients with subretinal drusenoid
R&M Solutions
 Mohamed Hosny El-Bradey, MD., Assistant Professor of Ophthalmology, Tanta University. Wael El Haig, MD., Professor of Ophthalmology. Zagazeeg University. 1 Myopic CNV is considered the most common vision
Mohamed Hosny El-Bradey, MD., Assistant Professor of Ophthalmology, Tanta University. Wael El Haig, MD., Professor of Ophthalmology. Zagazeeg University. 1 Myopic CNV is considered the most common vision
A Comparative Study of Age Related Macular Degeneration In Relation To SD-OCTand Fundus Photography.
 IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 14, Issue 11 Ver. III (Nov. 2015), PP 33-37 www.iosrjournals.org A Comparative Study of Age Related Macular
IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 14, Issue 11 Ver. III (Nov. 2015), PP 33-37 www.iosrjournals.org A Comparative Study of Age Related Macular
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
PRIMUS 200 from ZEISS The essential OCT
 PRIMUS 200 from ZEISS The essential OCT Seeing beyond the surface. ZEISS PRIMUS 200 // INNOVATION MADE BY ZEISS Clear Visualization. Advanced Technology. Reliability. Essential elements of your first OCT.
PRIMUS 200 from ZEISS The essential OCT Seeing beyond the surface. ZEISS PRIMUS 200 // INNOVATION MADE BY ZEISS Clear Visualization. Advanced Technology. Reliability. Essential elements of your first OCT.
The College of Optometrists - Learning outcomes for the Professional Certificate in Medical Retina
 Learning outcomes for the Professional Certificate in Medical Retina, incorporating diabetic retinopathy screening and age related macular degeneration The professional certificate is a prerequisite to
Learning outcomes for the Professional Certificate in Medical Retina, incorporating diabetic retinopathy screening and age related macular degeneration The professional certificate is a prerequisite to
Clinical Trial Endpoints for Macular Diseases
 Clinical Trial Endpoints for Macular Diseases Developed in collaboration Learning Objective Upon completion, participants should be able to: Summarize types of biomarkers of progression and treatment response
Clinical Trial Endpoints for Macular Diseases Developed in collaboration Learning Objective Upon completion, participants should be able to: Summarize types of biomarkers of progression and treatment response
Hydroxychloroquine and Chloroquine Retinopathy: Recommendations on Screening
 linical Guidelines Hydroxychloroquine and hloroquine Retinopathy: Recommendations on Screening February 2018 - Review date: February 2021 Executive Summary Recent data have highlighted that hydroxychloroquine
linical Guidelines Hydroxychloroquine and hloroquine Retinopathy: Recommendations on Screening February 2018 - Review date: February 2021 Executive Summary Recent data have highlighted that hydroxychloroquine
Experience Spectacular Retinal Imaging with the new NIDEK F-10 Digital Ophthalmoscope
 Experience Spectacular Retinal Imaging with the new NIDEK F-10 Digital Ophthalmoscope The F-10 was developed to give Ophthalmologists a high definition (HD) diagnostic imaging system. Designed to provide
Experience Spectacular Retinal Imaging with the new NIDEK F-10 Digital Ophthalmoscope The F-10 was developed to give Ophthalmologists a high definition (HD) diagnostic imaging system. Designed to provide
Appeal Decision. Appeal No USA. Tokyo, Japan. Tokyo, Japan. Tokyo, Japan
 Appeal Decision Appeal No. 2013-4177 USA Appellant Tokyo, Japan Patent Attorney Tokyo, Japan Patent Attorney Tokyo, Japan Patent Attorney THE IAMS COMPANY KATSUNUMA, Hirohito NAKAMURA, Yukitaka ENOKI,
Appeal Decision Appeal No. 2013-4177 USA Appellant Tokyo, Japan Patent Attorney Tokyo, Japan Patent Attorney Tokyo, Japan Patent Attorney THE IAMS COMPANY KATSUNUMA, Hirohito NAKAMURA, Yukitaka ENOKI,
RETINAL CONDITIONS RETINAL CONDITIONS
 GENERAL INFORMATION RETINAL CONDITIONS RETINAL CONDITIONS WHAT ARE RETINAL CONDITIONS? Retinal conditions affect the light-sensitive tissue at the back of eye known as the retina. They include diseases
GENERAL INFORMATION RETINAL CONDITIONS RETINAL CONDITIONS WHAT ARE RETINAL CONDITIONS? Retinal conditions affect the light-sensitive tissue at the back of eye known as the retina. They include diseases
Measurement of Choroidal Thickness in Normal Eyes Using 3D OCT-1000 Spectral Domain Optical Coherence Tomography
 pissn: 111-8942 eissn: 292-9382 Korean J Ophthalmol 212;26(4):255-259 http://dx.doi.org/1.3341/kjo.212.26.4.255 Original Article Measurement of Choroidal Thickness in Normal Eyes Using 3D OCT-1 Spectral
pissn: 111-8942 eissn: 292-9382 Korean J Ophthalmol 212;26(4):255-259 http://dx.doi.org/1.3341/kjo.212.26.4.255 Original Article Measurement of Choroidal Thickness in Normal Eyes Using 3D OCT-1 Spectral
PRIMUS 200 from ZEISS The essential OCT
 EN 00_00I The contents of the brochure may differ from the current status of approval of the product in your country. Please contact your regional representative for more information. Subject to change
EN 00_00I The contents of the brochure may differ from the current status of approval of the product in your country. Please contact your regional representative for more information. Subject to change
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at the
Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at the
(Tentative Translation)
 (Tentative Translation) Public Notice of the Ministry of Education, Culture, Sports, Science and Technology (MEXT) and the Ministry of Health, Labour and Welfare (MHLW) No. 2 of December 17, 2010 Table
(Tentative Translation) Public Notice of the Ministry of Education, Culture, Sports, Science and Technology (MEXT) and the Ministry of Health, Labour and Welfare (MHLW) No. 2 of December 17, 2010 Table
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
DOME SHAPED MACULOPATHY. Ιωάννης Ν. Βαγγελόπουλος Χειρ. Οφθαλμίατρος - Βόλος
 DOME SHAPED MACULOPATHY Ιωάννης Ν. Βαγγελόπουλος Χειρ. Οφθαλμίατρος - Βόλος DOME SHAPED MACULOPATHY-DEFINITIONS The entity Dome Shaped Macula ( DSM ) was first described by Gaucher and associates in 2008
DOME SHAPED MACULOPATHY Ιωάννης Ν. Βαγγελόπουλος Χειρ. Οφθαλμίατρος - Βόλος DOME SHAPED MACULOPATHY-DEFINITIONS The entity Dome Shaped Macula ( DSM ) was first described by Gaucher and associates in 2008
Macular Morphology and Visual Acuity in the Comparison of Age-related Macular Degeneration Treatments Trials
 Macular Morphology and Visual Acuity in the Comparison of Age-related Macular Degeneration Treatments Trials Glenn J. Jaffe, MD, 1 Daniel F. Martin, MD, 2 Cynthia A. Toth, MD, 1 Ebenezer Daniel, MPH, PhD,
Macular Morphology and Visual Acuity in the Comparison of Age-related Macular Degeneration Treatments Trials Glenn J. Jaffe, MD, 1 Daniel F. Martin, MD, 2 Cynthia A. Toth, MD, 1 Ebenezer Daniel, MPH, PhD,
Fundus autofluorescence in exudative age-related macular degeneration
 Fundus autofluorescence in exudative age-related macular degeneration Q. Peng*, Y. Dong* and P.Q. Zhao Department of Ophthalmology, Xinhua Hospital Affiliated to Shanghai JiaoTong University School of
Fundus autofluorescence in exudative age-related macular degeneration Q. Peng*, Y. Dong* and P.Q. Zhao Department of Ophthalmology, Xinhua Hospital Affiliated to Shanghai JiaoTong University School of
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care
 The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Medical Retina November 2016 Association of Health Professions in Ophthalmology General basic
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Medical Retina November 2016 Association of Health Professions in Ophthalmology General basic
Spectral-domain Optical Coherence Tomography Imaging of Age-related Macular Degeneration
 Imaging Spectral-domain Optical Coherence Tomography Imaging of Age-related Macular egeneration Carlos Alexandre de Amorim Garcia Filho, 1 Philip J Rosenfeld, 2 Zohar Yehoshua 3 and Giovanni Gregori 3
Imaging Spectral-domain Optical Coherence Tomography Imaging of Age-related Macular egeneration Carlos Alexandre de Amorim Garcia Filho, 1 Philip J Rosenfeld, 2 Zohar Yehoshua 3 and Giovanni Gregori 3
Case Report Peripapillary Intrachoroidal Cavitation in Myopia Evaluated with Multimodal Imaging Comprising (En-Face) Technique
 Case Reports in Ophthalmological Medicine Volume 2015, Article ID 890876, 5 pages http://dx.doi.org/10.1155/2015/890876 Case Report Peripapillary Intrachoroidal Cavitation in Myopia Evaluated with Multimodal
Case Reports in Ophthalmological Medicine Volume 2015, Article ID 890876, 5 pages http://dx.doi.org/10.1155/2015/890876 Case Report Peripapillary Intrachoroidal Cavitation in Myopia Evaluated with Multimodal
Swept-Source OCT Angiography: SS OCT Angio TM
 Swept-Source OCT Angiography: SS OCT Angio TM Not available in all countries, please check with your distributor. 2015.09 Swept-Source OCT Angiography: SS OCT Angio TM Introduction Optical coherence tomography
Swept-Source OCT Angiography: SS OCT Angio TM Not available in all countries, please check with your distributor. 2015.09 Swept-Source OCT Angiography: SS OCT Angio TM Introduction Optical coherence tomography
Optical Coherence Tomograpic Features in Idiopathic Retinitis, Vasculitis, Aneurysms and Neuroretinitis (IRVAN)
 Columbia International Publishing Journal of Ophthalmic Research (2014) Research Article Optical Coherence Tomograpic Features in Idiopathic Retinitis, Vasculitis, Aneurysms and Neuroretinitis (IRVAN)
Columbia International Publishing Journal of Ophthalmic Research (2014) Research Article Optical Coherence Tomograpic Features in Idiopathic Retinitis, Vasculitis, Aneurysms and Neuroretinitis (IRVAN)
Optical Coherence Tomography (OCT) in Uveitis Piergiorgio Neri, BMedSc, MD, PhD Head Ocular Immunology Unit
 The Eye Clinic Polytechnic University of Marche Head: Prof Alfonso Giovannini November, 1991 Optical Coherence Tomography (OCT) in Uveitis Piergiorgio Neri, BMedSc, MD, PhD Head Ocular Immunology Unit
The Eye Clinic Polytechnic University of Marche Head: Prof Alfonso Giovannini November, 1991 Optical Coherence Tomography (OCT) in Uveitis Piergiorgio Neri, BMedSc, MD, PhD Head Ocular Immunology Unit
SUMMARY. Heather Casparis, MD,* and Neil M. Bressler, MD MARINA AND ANCHOR
 The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
Pharmaceutics I صيدالنيات 1. Unit 2 Route of Drug Administration
 Pharmaceutics I صيدالنيات 1 Unit 2 Route of Drug Administration 1 Routs of Drug administration The possible routes of drug entry into the body may be divided into two classes: Parenteral Rout Enteral Rout
Pharmaceutics I صيدالنيات 1 Unit 2 Route of Drug Administration 1 Routs of Drug administration The possible routes of drug entry into the body may be divided into two classes: Parenteral Rout Enteral Rout
Trilateral Project WM4
 ANNEX 2: Comments of the JPO Trilateral Project WM4 Comparative studies in new technologies Theme: Comparative study on protein 3-dimensional (3-D) structure related claims 1. Introduction As more 3-D
ANNEX 2: Comments of the JPO Trilateral Project WM4 Comparative studies in new technologies Theme: Comparative study on protein 3-dimensional (3-D) structure related claims 1. Introduction As more 3-D
Incorporating OCT Angiography Into Patient Care
 Incorporating OCT Angiography Into Patient Care Beth A. Steele, OD, FAAO OCT A: Introduction Isolates microvascular circulation from OCT image data Axial resolution = 5 microns (i.e. fine capillaries visible)
Incorporating OCT Angiography Into Patient Care Beth A. Steele, OD, FAAO OCT A: Introduction Isolates microvascular circulation from OCT image data Axial resolution = 5 microns (i.e. fine capillaries visible)
The legally binding text is the original French version
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 12 September 2007 INFRACYANINE 25 mg/10 ml, powder and solvent for solution for injection 25 mg vial with one 10 ml
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 12 September 2007 INFRACYANINE 25 mg/10 ml, powder and solvent for solution for injection 25 mg vial with one 10 ml
Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration
 Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration Marilita M. Moschos Abstract- Purpose: To evaluate by OCT and mf-erg the macular function in eyes with CNV due to ARMD
Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration Marilita M. Moschos Abstract- Purpose: To evaluate by OCT and mf-erg the macular function in eyes with CNV due to ARMD
Age-Related Macular Degeneration (AMD)
 Age-Related Macular Degeneration (AMD) What is the Macula? What is Dry AMD (Age-related Macular Degeneration)? Dry AMD is an aging process that causes accumulation of waste product under the macula leading
Age-Related Macular Degeneration (AMD) What is the Macula? What is Dry AMD (Age-related Macular Degeneration)? Dry AMD is an aging process that causes accumulation of waste product under the macula leading
Ultrahigh Speed Imaging of the Rat Retina Using Ultrahigh Resolution Spectral/Fourier Domain OCT
 Ultrahigh Speed Imaging of the Rat Retina Using Ultrahigh Resolution Spectral/Fourier Domain OCT The MIT Faculty has made this article openly available. Please share how this access benefits you. Your
Ultrahigh Speed Imaging of the Rat Retina Using Ultrahigh Resolution Spectral/Fourier Domain OCT The MIT Faculty has made this article openly available. Please share how this access benefits you. Your
Posterior Segment Age-related Macular Degeneration
 Posterior Segment Age-related Macular egeneration Spectral-domain Optical oherence Tomography Imaging of Age-related Macular egeneration arlos Alexandre de Amorim Garcia Filho, M, 1 Philip J Rosenfeld,
Posterior Segment Age-related Macular egeneration Spectral-domain Optical oherence Tomography Imaging of Age-related Macular egeneration arlos Alexandre de Amorim Garcia Filho, M, 1 Philip J Rosenfeld,
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
ZEISS AngioPlex OCT Angiography Making the revolutionary, routine.
 ZEISS AngioPlex OCT Angiography Making the revolutionary, routine. The moment that revolutionary insight becomes routine. // OCT ANGIOGRAPHY MADE BY ZEISS CIRRUS with AngioPlex creates a new era in both
ZEISS AngioPlex OCT Angiography Making the revolutionary, routine. The moment that revolutionary insight becomes routine. // OCT ANGIOGRAPHY MADE BY ZEISS CIRRUS with AngioPlex creates a new era in both
Retinal. Next Generation Retinal Imaging: cslo Combined With Spectral Domain OCT. PHYSICIAN January/February 2008
 Retinal PHYSICIAN January/February 2008 Next Generation Retinal Imaging: cslo Combined With Spectral Domain OCT Highlights of a Breakfast Symposium held during The Retina Society meeting, Boston, Mass.
Retinal PHYSICIAN January/February 2008 Next Generation Retinal Imaging: cslo Combined With Spectral Domain OCT Highlights of a Breakfast Symposium held during The Retina Society meeting, Boston, Mass.
WITH APPLICATIONS TO : ; : GENERAL AND SPECIALTY AREAS
 f Fourth Edition WITH APPLICATIONS TO : ; : GENERAL AND SPECIALTY AREAS Joyce LeFever Kee, RN, MS Associate Professor Emerita College of Health and Nursing Sciences University of Delaware Newark, Delaware
f Fourth Edition WITH APPLICATIONS TO : ; : GENERAL AND SPECIALTY AREAS Joyce LeFever Kee, RN, MS Associate Professor Emerita College of Health and Nursing Sciences University of Delaware Newark, Delaware
ZEISS AngioPlex OCT Angiography. Clinical Case Reports
 Clinical Case Reports Proliferative Diabetic Retinopathy (PDR) Case Report 969 PROLIFERATIVE DIABETIC RETINOPATHY 1 1-year-old diabetic female presents for follow-up of proliferative diabetic retinopathy
Clinical Case Reports Proliferative Diabetic Retinopathy (PDR) Case Report 969 PROLIFERATIVE DIABETIC RETINOPATHY 1 1-year-old diabetic female presents for follow-up of proliferative diabetic retinopathy
OCT Interpretation in Retinal Disease
 OCT Interpretation in Retinal Disease Jay M. Haynie, OD, FAAO Financial Disclosure I have received honoraria or am on the advisory board for the following companies: Carl Zeiss Meditec Advanced Ocular
OCT Interpretation in Retinal Disease Jay M. Haynie, OD, FAAO Financial Disclosure I have received honoraria or am on the advisory board for the following companies: Carl Zeiss Meditec Advanced Ocular
Course # Getting to Know Your OCT
 Course # 140 Getting to Know Your OCT Course Title: Lecturer: Getting to Know Your OCT Brad Sutton, OD, FAAO IU School of Optometry Financial Disclosures No financial disclosures Optical Coherence Tomography-OCT
Course # 140 Getting to Know Your OCT Course Title: Lecturer: Getting to Know Your OCT Brad Sutton, OD, FAAO IU School of Optometry Financial Disclosures No financial disclosures Optical Coherence Tomography-OCT
Comparative Study of Experimental Choroidal Neovascularization by Optical Coherence Tomography and Histopathology
 Comparative Study of Experimental Choroidal Neovascularization by Optical Coherence Tomography and Histopathology Toshio Fukuchi, Kanji Takahashi, Masanobu Uyama and Miyo Matsumura Department of Ophthalmology,
Comparative Study of Experimental Choroidal Neovascularization by Optical Coherence Tomography and Histopathology Toshio Fukuchi, Kanji Takahashi, Masanobu Uyama and Miyo Matsumura Department of Ophthalmology,
Mariam Raouf Fadel M.B., B.Ch. M.Sc., Cairo University. A thesis. Submitted by. For partial fulfillment of. MD Degree in Ophthalmology
 Correlation of fundus autofluorescence and spectral domain OCT findings of the macula with visual outcome after successful repair of rhegmatogenous retinal detachment A thesis Submitted by Mariam Raouf
Correlation of fundus autofluorescence and spectral domain OCT findings of the macula with visual outcome after successful repair of rhegmatogenous retinal detachment A thesis Submitted by Mariam Raouf
The Foundation WHAT IS THE RETINA?
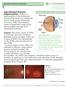 Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
SECOND MEDICAL USE CLAIMS
 SECOND MEDICAL USE CLAIMS AIPPI Mari Korsten 18-1-2017 Second medical use claims (EPO perspective) Background of medical use claims; Legal basis of medical use claims Novelty and Inventive step of medical
SECOND MEDICAL USE CLAIMS AIPPI Mari Korsten 18-1-2017 Second medical use claims (EPO perspective) Background of medical use claims; Legal basis of medical use claims Novelty and Inventive step of medical
ZEISS presents innovations that support ophthalmologists in their work
 ZEISS presents innovations that support ophthalmologists in their work At the 2015 Annual Meeting of the American Academy of Ophthalmology (AAO) in Las Vegas, ZEISS announces innovations for clinical excellence
ZEISS presents innovations that support ophthalmologists in their work At the 2015 Annual Meeting of the American Academy of Ophthalmology (AAO) in Las Vegas, ZEISS announces innovations for clinical excellence
NIH Public Access Author Manuscript JAMA Ophthalmol. Author manuscript; available in PMC 2013 September 10.
 NIH Public Access Author Manuscript Published in final edited form as: JAMA Ophthalmol. 2013 May ; 131(5): 693 694. doi:10.1001/jamaophthalmol.2013.692. Effect of Intravitreous Anti Vascular Endothelial
NIH Public Access Author Manuscript Published in final edited form as: JAMA Ophthalmol. 2013 May ; 131(5): 693 694. doi:10.1001/jamaophthalmol.2013.692. Effect of Intravitreous Anti Vascular Endothelial
Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial
 ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
Indocyanine Green Angiographic Findings of Chorioretinal Folds
 Indocyanine Green Angiographic Findings of Chorioretinal Folds Miho Haruyama, Mitsuko Yuzawa, Akiyuki Kawamura, Chikayo Yamazaki and Youko Matsumoto Department of Ophthalmology, Nihon University School
Indocyanine Green Angiographic Findings of Chorioretinal Folds Miho Haruyama, Mitsuko Yuzawa, Akiyuki Kawamura, Chikayo Yamazaki and Youko Matsumoto Department of Ophthalmology, Nihon University School
OCT Image Analysis System for Grading and Diagnosis of Retinal Diseases and its Integration in i-hospital
 Progress Report for1 st Quarter, May-July 2017 OCT Image Analysis System for Grading and Diagnosis of Retinal Diseases and its Integration in i-hospital Milestone 1: Designing Annotation tool extraction
Progress Report for1 st Quarter, May-July 2017 OCT Image Analysis System for Grading and Diagnosis of Retinal Diseases and its Integration in i-hospital Milestone 1: Designing Annotation tool extraction
RXi Pharmaceuticals. sd-rxrna Demonstrate Robust Efficacy in the Eye. Dr. Geert Cauwenbergh President & CEO OTCQX: RXII. Next Generation in RNAi
 RXi Pharmaceuticals sd-rxrna Demonstrate Robust Efficacy in the Eye Next Generation in RNAi Dr. Geert Cauwenbergh President & CEO OTCQX: RXII 2 Forward Looking Statements This presentation contains forward-looking
RXi Pharmaceuticals sd-rxrna Demonstrate Robust Efficacy in the Eye Next Generation in RNAi Dr. Geert Cauwenbergh President & CEO OTCQX: RXII 2 Forward Looking Statements This presentation contains forward-looking
International Journal of Health Sciences and Research ISSN:
 International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
Diabetic Retinopathy Clinical Research Network
 Diabetic Retinopathy Clinical Research Network Comparison of Time Domain OCT and Spectral Domain OCT Retinal Thickness Measurement in Diabetic Macular Edema Version 1.0 June 16, 2009 comparison of td vs
Diabetic Retinopathy Clinical Research Network Comparison of Time Domain OCT and Spectral Domain OCT Retinal Thickness Measurement in Diabetic Macular Edema Version 1.0 June 16, 2009 comparison of td vs
Although photocoagulation and photodynamic PROCEEDINGS PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION *
 PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
Clinically Significant Macular Edema (CSME)
 Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Michael P. Blair, MD Retina Consultants, Ltd Libertyville/Des Plaines, Illinois Clinical Associate University of Chicago 17 October 2015
 Michael P. Blair, MD Retina Consultants, Ltd Libertyville/Des Plaines, Illinois Clinical Associate University of Chicago 17 October 2015 So What Parts of the Eye Retina are Affected by VHL Neural tissue
Michael P. Blair, MD Retina Consultants, Ltd Libertyville/Des Plaines, Illinois Clinical Associate University of Chicago 17 October 2015 So What Parts of the Eye Retina are Affected by VHL Neural tissue
Routes of drug administration
 Routes of drug administration Definition:- A route of administration in pharmacy is the path by which a drug is taken into the body. Classification:- The various routes of administrations are classified
Routes of drug administration Definition:- A route of administration in pharmacy is the path by which a drug is taken into the body. Classification:- The various routes of administrations are classified
Katsuhiko, Fukui ; Akitoshi, Yoshida ; Hiromasa, Igarashi ; Hong-Ming, Cheng ; Hironori, Isobe
 The Journal of ophthalmic photography (1995) 17(2):54-60. Anterior Segment Fluorescein Angiography Using a Modified Photo Slit- Lamp Katsuhiko, Fukui ; Akitoshi, Yoshida ; Hiromasa, Igarashi ; Hong-Ming,
The Journal of ophthalmic photography (1995) 17(2):54-60. Anterior Segment Fluorescein Angiography Using a Modified Photo Slit- Lamp Katsuhiko, Fukui ; Akitoshi, Yoshida ; Hiromasa, Igarashi ; Hong-Ming,
Appeal decision. Appeal No Germany. Osaka, Japan. Osaka, Japan
 Appeal decision Appeal No. 2012-9689 Germany Appellant PAT GMBH Osaka, Japan Patent Attorney ISEKI, Katsumori Osaka, Japan Patent Attorney TANAKA, Yonezo The case of appeal against the examiner's decision
Appeal decision Appeal No. 2012-9689 Germany Appellant PAT GMBH Osaka, Japan Patent Attorney ISEKI, Katsumori Osaka, Japan Patent Attorney TANAKA, Yonezo The case of appeal against the examiner's decision
The MP-1 Microperimeter Clinical Applications in Retinal Pathologies
 The MP-1 Microperimeter Clinical Applications in Retinal Pathologies Nelson R. Sabates, MD Director, Retina/Vitreous Service Vice-Chairman Department of Ophthalmology University of Missouri Kansas City
The MP-1 Microperimeter Clinical Applications in Retinal Pathologies Nelson R. Sabates, MD Director, Retina/Vitreous Service Vice-Chairman Department of Ophthalmology University of Missouri Kansas City
Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187
 Clinical Policy: (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Optical Coherence Tomography in Diabetic Retinopathy. Mrs Samantha Mann Consultant Ophthalmologist Clinical Lead of SEL-DESP
 Optical Coherence Tomography in Diabetic Retinopathy Mrs Samantha Mann Consultant Ophthalmologist Clinical Lead of SEL-DESP Content OCT imaging Retinal layers OCT features in Diabetes Some NON DR features
Optical Coherence Tomography in Diabetic Retinopathy Mrs Samantha Mann Consultant Ophthalmologist Clinical Lead of SEL-DESP Content OCT imaging Retinal layers OCT features in Diabetes Some NON DR features
Dehiscence of detached internal limiting membrane in eyes with myopic traction maculopathy with spontaneous resolution
 Hirota et al. BMC Ophthalmology 2014, 14:39 RESEARCH ARTICLE Open Access Dehiscence of detached internal limiting membrane in eyes with myopic traction maculopathy with spontaneous resolution Kazunari
Hirota et al. BMC Ophthalmology 2014, 14:39 RESEARCH ARTICLE Open Access Dehiscence of detached internal limiting membrane in eyes with myopic traction maculopathy with spontaneous resolution Kazunari
Contents. Chapter 1 Common Pitfalls in the Use of Optical Coherence Tomography for Macular Diseases Lihteh Wu and Teodoro Evans
 Contents Chapter 1 Common Pitfalls in the Use of Optical Coherence Tomography for Macular Diseases Lihteh Wu and Teodoro Evans 1.1 Introduction... 1 1.2 Limitations of Time-Domain OCT... 2 1.2.1 Acquisition
Contents Chapter 1 Common Pitfalls in the Use of Optical Coherence Tomography for Macular Diseases Lihteh Wu and Teodoro Evans 1.1 Introduction... 1 1.2 Limitations of Time-Domain OCT... 2 1.2.1 Acquisition
Clinical Policy: Aflibercept (Eylea) Reference Number: CP.PHAR.184
 Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
OCT Angiography in Primary Eye Care
 OCT Angiography in Primary Eye Care An Image Interpretation Primer Julie Rodman, OD, MS, FAAO and Nadia Waheed, MD, MPH Table of Contents Diabetic Retinopathy 3-6 Choroidal Neovascularization 7-9 Central
OCT Angiography in Primary Eye Care An Image Interpretation Primer Julie Rodman, OD, MS, FAAO and Nadia Waheed, MD, MPH Table of Contents Diabetic Retinopathy 3-6 Choroidal Neovascularization 7-9 Central
Multimodal Imaging of Geographic Atrophy
 A PROMOTIONAL SUPPLEMENT TO June 2017 C u t t i n g - e d g e A d v a n c e m e n t s Multimodal Imaging of Geographic Atrophy PROCEEDINGS FROM A SYMPOSIUM HELD DURING THE EURETINA MEETING OF THE EUROPEAN
A PROMOTIONAL SUPPLEMENT TO June 2017 C u t t i n g - e d g e A d v a n c e m e n t s Multimodal Imaging of Geographic Atrophy PROCEEDINGS FROM A SYMPOSIUM HELD DURING THE EURETINA MEETING OF THE EUROPEAN
RETINA 2018 OBJECTIVES OCT VERY USEFUL INFORMATION SAFE AND FRIENDLY 1/11/2018 KELLY MITCHELL
 RETINA 2018 KELLY MITCHELL OBJECTIVES HIGHLIGHT NEW DIAGNOSTIC & TREATMENT OPTIONS REVIEW DIAGNOSTIC KEYS OF SELECT RETINAL DISEASES DISCUSS USE OF IMAGING AND REFERRAL RECOURSES FOR PATIENT BENEFIT OCT
RETINA 2018 KELLY MITCHELL OBJECTIVES HIGHLIGHT NEW DIAGNOSTIC & TREATMENT OPTIONS REVIEW DIAGNOSTIC KEYS OF SELECT RETINAL DISEASES DISCUSS USE OF IMAGING AND REFERRAL RECOURSES FOR PATIENT BENEFIT OCT
Trial decision SAWAI PHARMACEUTICAL CO., LTD. TEVA PHARMACEUTICAL INDUSTRIES LTD.
 Trial decision Invalidation No. 2013-800139 Osaka, Japan Demandant Tokyo, Japan Attorney Tokyo, Japan Attorney Tokyo, Japan Attorney Israel Intervenor Tokyo, Japan Patent Attorney Tokyo, Japan Attorney
Trial decision Invalidation No. 2013-800139 Osaka, Japan Demandant Tokyo, Japan Attorney Tokyo, Japan Attorney Tokyo, Japan Attorney Israel Intervenor Tokyo, Japan Patent Attorney Tokyo, Japan Attorney
Eye injection treatment costs and rebates
 Eye injection treatment costs and rebates This fact sheet provides general information on the bill you receive from your ophthalmologist for eye injections for the management of wet macular degeneration,
Eye injection treatment costs and rebates This fact sheet provides general information on the bill you receive from your ophthalmologist for eye injections for the management of wet macular degeneration,
Spontaneous Large Serous Retinal Pigment Epithelial Tear
 This is an Open Access article licensed under the terms of the Creative Commons Attribution-NonCommercial-NoDerivs 3.0 License (www.karger.com/oa-license), applicable to the online version of the article
This is an Open Access article licensed under the terms of the Creative Commons Attribution-NonCommercial-NoDerivs 3.0 License (www.karger.com/oa-license), applicable to the online version of the article
Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis)
 Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
RetCAD 1.3.0: White paper
 RetCAD 1.3.0: White paper 18 July 2018 Computer aided detection for Age-related Macular Degeneration and Diabetic Retinopathy About this white paper This white paper applies to RetCAD 1.3.0 and describes
RetCAD 1.3.0: White paper 18 July 2018 Computer aided detection for Age-related Macular Degeneration and Diabetic Retinopathy About this white paper This white paper applies to RetCAD 1.3.0 and describes
Reports of Adverse Drug Reactions, etc. of Pharmaceuticals
 (Attachment) Reports of Adverse Drug Reactions, etc. of Pharmaceuticals 1 GLOSSARY (1) Drugs, quasi-drugs, and cosmetics -1- Related to Article 228-20, paragraph 1, item 1 of the Enforcement Ordinance
(Attachment) Reports of Adverse Drug Reactions, etc. of Pharmaceuticals 1 GLOSSARY (1) Drugs, quasi-drugs, and cosmetics -1- Related to Article 228-20, paragraph 1, item 1 of the Enforcement Ordinance
Trial decision. Invalidation No Gunma, Japan. Tokyo, Japan YOSHINAGA, Takahiro. Tokyo, Japan. Tokyo, Japan.
 Trial decision Invalidation No. 2011-800233 Gunma, Japan Demandant HAMA, Michihisa Tokyo, Japan Attorney YOSHINAGA, Takahiro Tokyo, Japan Demandee KAO CORPORATION Tokyo, Japan Attorney HANADA, Yoshiaki
Trial decision Invalidation No. 2011-800233 Gunma, Japan Demandant HAMA, Michihisa Tokyo, Japan Attorney YOSHINAGA, Takahiro Tokyo, Japan Demandee KAO CORPORATION Tokyo, Japan Attorney HANADA, Yoshiaki
These issues are covered in more detail below.
 26.3.07 Comments from Novartis on the Assessment Report for the Health Technology Appraisal of Pegaptinib and Ranibizumab for the treatment of age-related macular degeneration In general we feel that the
26.3.07 Comments from Novartis on the Assessment Report for the Health Technology Appraisal of Pegaptinib and Ranibizumab for the treatment of age-related macular degeneration In general we feel that the
Tips and Tactics for Retinal Imaging
 Tips and Tactics for Retinal Imaging Retinal Imaging Fundus camera Scanning Laser Ophthalmoscope (cslo) SD-Optical Coherence Tomography Timothy J. Bennett, CRA, OCT-C, FOPS Penn State Eye Center Hershey,
Tips and Tactics for Retinal Imaging Retinal Imaging Fundus camera Scanning Laser Ophthalmoscope (cslo) SD-Optical Coherence Tomography Timothy J. Bennett, CRA, OCT-C, FOPS Penn State Eye Center Hershey,
Technologies and Methods for Visualizing the Retina
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/revealing-retina/technologies-and-methods-for-visualizing-theretina/3663/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/revealing-retina/technologies-and-methods-for-visualizing-theretina/3663/
Mark Dunbar: Disclosure
 Important Things to Understand About OCT Mark T. Dunbar, O.D., F.A.A.O. Bascom Palmer Eye Institute University of Miami, School of Medicine Mark Dunbar: Disclosure Optometry Advisory Board for: Allergan
Important Things to Understand About OCT Mark T. Dunbar, O.D., F.A.A.O. Bascom Palmer Eye Institute University of Miami, School of Medicine Mark Dunbar: Disclosure Optometry Advisory Board for: Allergan
Citation. As Published Publisher. Version
 Effect of Intravitreous Anti Vascular Endothelial Growth Factor Therapy on Choroidal Thickness in Neovascular Age-Related Macular Degeneration Using Spectral-Domain The MIT Faculty has made this article
Effect of Intravitreous Anti Vascular Endothelial Growth Factor Therapy on Choroidal Thickness in Neovascular Age-Related Macular Degeneration Using Spectral-Domain The MIT Faculty has made this article
Clinical Study Choroidal Thickness in Eyes with Unilateral Ocular Ischemic Syndrome
 Hindawi Publishing Corporation Journal of Ophthalmology Volume 215, Article ID 62372, 5 pages http://dx.doi.org/1.1155/215/62372 Clinical Study Choroidal Thickness in Eyes with Unilateral Ocular Ischemic
Hindawi Publishing Corporation Journal of Ophthalmology Volume 215, Article ID 62372, 5 pages http://dx.doi.org/1.1155/215/62372 Clinical Study Choroidal Thickness in Eyes with Unilateral Ocular Ischemic
Retinal pigment epithelial atrophy over polypoidal choroidal vasculopathy lesions during ranibizumab monotherapy
 Hikichi et al. BMC Ophthalmology (2016) 16:55 DOI 10.1186/s12886-016-0237-x RESEARCH ARTICLE Retinal pigment epithelial atrophy over polypoidal choroidal vasculopathy lesions during ranibizumab monotherapy
Hikichi et al. BMC Ophthalmology (2016) 16:55 DOI 10.1186/s12886-016-0237-x RESEARCH ARTICLE Retinal pigment epithelial atrophy over polypoidal choroidal vasculopathy lesions during ranibizumab monotherapy
Report on FY2015 Annual User Satisfaction Survey on Patent Examination Quality
 Report on FY2015 Annual User Satisfaction Survey on Patent Examination Quality June 2016 Japan Patent Office ABSTRACT I. Introduction Globally reliable high-quality patent examination and proper patent-granting
Report on FY2015 Annual User Satisfaction Survey on Patent Examination Quality June 2016 Japan Patent Office ABSTRACT I. Introduction Globally reliable high-quality patent examination and proper patent-granting
Drug CHAPTER 2. Pharmacologic Principles. NDEG 26A Eliza Rivera-Mitu, RN, MSN. Pharmacology. Drug Names. Pharmacologic Principles. Drug Names (cont'd)
 CHAPTER 2 Pharmacologic Principles NDEG 26A Eliza Rivera-Mitu, RN, MSN Drug Any chemical that affects the physiologic processes of a living organism Pharmacology The study or science of drugs Drug Names
CHAPTER 2 Pharmacologic Principles NDEG 26A Eliza Rivera-Mitu, RN, MSN Drug Any chemical that affects the physiologic processes of a living organism Pharmacology The study or science of drugs Drug Names
