DRUG AND THERAPEUTICS NEWSLETTER
|
|
|
- Andrea Francis
- 6 years ago
- Views:
Transcription
1 Oct 2009 Volume 16, Number 3 In This Issue... Changes to Formulary 1 PPI Interchange Policy 2 Filter Required for Diluted Phenytoin 2 Oseltamivir, Lamivudine Expanded Restrictions 2 Narcotic Equivalency: IM/SC versus PO 2 Adding Insulin to TPN 3 PDTM Updates 3 Bowel Protocols: Medicine and Geriatrics 3 Pharmacy Award 3 Errata: Fondaparinux - Oasis-5 trial 3 Clostridium difficile-associated Disease 4 Guidelines All formulary changes and policy/procedure updates have been approved by the Drugs and Therapeutics (D&T) Committee and Medical Advisory Council (MAC). This and other Drug and Therapeutics Newsletters are on the Web at Changes to Formulary Additions 1. Zanamivir 5 mg diskhaler (Relenza ) Antiviral agent added for the treatment and prophylaxis of oseltamivir (Tamiflu ) - resistant influenza Must be approved by the Medical Health Officer or Infection Control 2. Carbamide peroxide 6.5% otic drops (Murine ) Ear wax removal drops Replaces the discontinued product triethanolamine (Cerumenex ) 3. Metoprolol Slow Release 100 mg, 200 mg tablets 4. Esomeprazole (Nexium ) tablets and Rabeprazole tablets (Pariet ) Proton pump inhibitors (PPIs) See Therapeutic Interchange Policy, pg. 2 DRUG AND THERAPEUTICS NEWSLETTER A publication of the CSU Pharmaceutical Sciences Vancouver General Hospital, UBC Hospital, GF Strong 5. Sodium Picosulphate sachets (PicoSalex ) Bowel cleansing agent as an alternative to other bowel preps (See Table 1 below for comparison of agents) Table 1. Adult Bowel Cleansing Agents Drug Sodium Picosulphate (SPS) Polyethylene Glycol (PEG, PegLyte ) Oral Phosphates Solution (Phoslax ) Magnesium Citrate (Citro-Mag ) Dosage Form/ Content Each 16.1 g powder sachet - Magnesium citrate g (in solution) - Sodium Picosulfate 10 mg Each powder sachet - PEG 70 g Each 45 ml liquid - Phosphorus g Each 300 ml liquid - Magnesium citrate 15 g Bowel Cleanser Dose 2 x 150 ml: one in am and pm on day preprocedure (Dissolve each sachet in 150 ml water)* 4 L: 240 ml Q10MIN up to max 4 L or until discharge is clear (Dissolve each sachet in 1 L water) 2 x 45 ml: give 24 hours apart (dilute 45 ml in 120 ml water)* 1 x 300 ml* *patients must be adequately hydrated; follow manufacturer s guidelines EDITORIAL STAFF: Karen Shalansky, Pharm.D., FCSHP Tim Lau, Pharm.D., FCSHP Jane Day, B.Sc.(Pharm.), ACPR Nilu Partovi, Pharm.D., FCSHP Any comments, questions, or concerns with the content of the newsletter should be directed to the editors. Write to CSU Pharmaceutical Sciences Vancouver General Hospital, 855 W12th Ave, Vancouver BC V5Z 1M9, send a FAX to or karen.shalansky@vch.ca Find us on the Web at
2 Drug and Therapeutics Newsletter 2 Volume 16, Number 3 Deletions 1. Protirelin injection (TRH, Relefact ) Available under the Special Access Program 2. Phenazopyridine tablets (Pyridium ) Discontinued by manufacturer 3. Triethanolamine otic (Cerumenex ) Discontinued by manufacturer Updated Policies 1. PROTON PUMP INHIBITOR (PPI) INTERCHANGE POLICY Due to new contract pricing for VCH-PHC Pharmacy, esomeprazole 40 mg is the lowest cost PPI. Since esomeprazole 40 mg cannot be split for lower doses, rabeprazole 10 mg is the PPI chosen for lower dose orders. Esomperazole is also the PPI used for NG/G-tube use. As of August 2009, all PPIs are automatically interchanged to one of these 2 agents according to the table below. Table 2. PPI Equivalent Dose (same interval) Ordered Drug Lansoprazole 15 mg Interchanged to: PO orders Physicians will be called for any PPI doses not contained on this table. 2. FILTER REQUIRED FOR DILUTED PHENYTOIN All diluted phenytoin solutions must be administered with a 0.22 micron in-line filter to remove any phenytoin crystals which may form when diluted. The IV administration sets must be flushed with NS before and after administration. A filter is NOT required for undiluted phenytoin that is administered IV direct. 3. FORMULARY RESTRICTIONS EXPANDED Oseltamivir (Tamiflu ) indications have been expanded to include both the treatment and prophylaxis of influenza virus with the following restrictions: For treatment of individual cases, there are no restrictions. For influenza prophylaxis, approval must be obtained by the Medical Health Officer or Infection Control. Lamivudine can now be ordered by GI, SOT, and Leukemia/BMT services and in patients on high dose chemotherapy for Hepatitis B prophylaxis. Esomeprazole 20 mg Rabeprazole 10 mg PO Omeprazole 10 mg Pantoprazole 20 mg Lansoprazole 30 mg Omeprazole 20 mg Esomeprazole 40 mg PO Pantoprazole 40 mg Rabeprazole 20 mg NG orders Lansoprazole 15 mg Omeprazole 10 mg Esomeprazole 20 mg NG Pantoprazole 20 mg Rabeprazole 10 mg Lansoprazole 30 mg Omeprazole 20 mg Esomeprazole 40 mg NG Pantoprazole 40 mg Rabeprazole 20 mg 4. NARCOTIC EQUIVALENCIES: IM/SC vs. PO Parenteral and oral narcotic doses are not equivalent. For morphine and hydromorphone, the equivalent narcotic dose for IM/SC is generally at least half that of the PO dose: Table 3. Equivalent Narcotic Doses Drug IM/SC PO Morphine 10 mg mg Hydromorphone 2 mg 4 mg Thus, orders that combine IM, SC and PO routes are written incorrectly and should be separated into an IM/SC dose and a PO dose. Incorrect Order Hydromorphone 1 to 4 mg SC/IM/PO Q4H PRN Correct Order Hydromorphone 1 to 2 mg SC/IM Q4H PRN or Hydromorphone 2 to 4 mg PO Q4H PRN
3 Drug and Therapeutics Newsletter 3 Volume 16, Number 3 5. INSULIN GUIDELINES FOR TPN The majority of diabetic patients require supplemental insulin when a high glucose solution is infused. Guidelines for the addition of insulin regular to TPN are as follows: A. Initial insulin dose ordered in TPN: New start TPN in diabetic patient: A common starting point is 0.1 units of insulin per g of dextrose (e.g. 30 units per 300 g dextrose). 1 This ratio of insulin to dextrose is unlikely to cause hypoglycemia. Starting doses may vary based on perceived patient requirements. Patients on TPN with sliding scale: Add up the insulin sliding scale requirements from the previous 24 hours, then add 1/2 to 2/3 of this dose to the TPN. Continue to maintain sliding scale insulin with QID glucometer. B. Increasing the dose of insulin in TPN: If over a 24 hour period glucose values consistently exceed the desired goal, the insulin in the TPN can be increased by 0.05 units per g dextrose each day (e.g. 15 units for 300 g dextrose). In general, doses are increased by increments of 5 to 15 units of insulin per 24 hours. Reference 1. Sacks GS et al. Parenteral nutrition implementation and management. In the A.S.P.E.N. Nutrition Support Manual, PDTM UPDATES Vasopressin infusions for treatment of shock may be administered in CCU, ICU and CSICU If diltiazem or verapamil are administered IV direct on a general nursing unit, a physician must administer the dose and stay on the unit for 15 minutes. BP and HR should be monitored at baseline, then Q5MIN x BOWEL PROTOCOLS: Medicine and Geriatrics Some specialty services do not have a servicespecific bowel protocol. When these services order a bowel protocol but do not specify the type of protocol required, pharmacy will default to the Medicine bowel protocol (for patients < 70 years) or Geriatric bowel protocol (for patients 70 years). Note that suppositories and enemas are contraindicated in Leukemia/BMT patients or those who are pancytopenic or neutropenic. These comments have been added to these protocols. Pharmacy Awards Margie Sidsworth was the 2008/09 recipient of the Resident of the Year award given by the VCH-PHC Pharmacy Practice Residency Program. Errata - FONDAPARINUX: OASIS-5 CLARIFICATION In the last D&T newsletter (2009;16(2):3-4), we published a review of fondaparinux (Arixtra ), a direct inhibitor of Factor Xa, for the treatment of medically managed NSTEMI. The mortality endpoint in the OASIS-5 trial was not sufficiently reported in that article. The OASIS- 5 trial was a non-inferiority study that compared fondaparinux to enoxaparin in unstable angina and NSTEMI patients in a randomized, double-blind, double dummy fashion. 1 The primary endpoint was a composite of death, MI or refractory ischemia at 9 days. Fondaparinux met the criteria for non-inferiority to enoxaparin with the primary endpoint occurring in 5.7% of patients in the enoxaparin arm and 5.8% of patients in the fondaparinux arm, HR 1.01 (95% CI ). The secondary outcomes, composite of death, MI or refractory ischemia at 30 days and 180 days, were also not significantly different between the groups. However, when examining the individual components of the composite endpoint at 30 days, death occurred in 3.5% of enoxaparin patients vs. 2.9% of fondaparinux patients, HR 0.83 (95% CI , p=0.02). Death at 180 days, however, did not reach statistical significance: 6.5% in enoxaparin vs. 5.8% in fondaparinux, HR 0.89 (95% CI , p=0.05). Reference 1. Yusuf S et al. Comparison of fondaparinux and enoxaparin in acute coronary syndromes. NEJM 2006;354:
4 Drug and Therapeutics Newsletter 4 Volume 16, Number 3 Regional Guideline CLOSTRIDIUM DIFFICILE - ASSOCIATED DISEASE Background Clostridium difficile-associated disease (CDAD) is a healthcare-associated infection that is increasing in significance at VCH-PHC. Most notably, the NAP1 strain has emerged in our patient population and is associated with more severe symptoms. CDAD presents as a spectrum of disease, ranging from profuse diarrhea to pseudomembranous colitis and toxic megacolon, which may result in mortality. As such, it is essential that treatment is initiated promptly and stratified appropriately, as patients can deteriorate rapidly. In addition to medical treatment, all patients with CDAD or those with high clinical suspicion of CDAD should be isolated on contact precautions. Patients should be monitored closely and examined on a daily basis by nurses, physicians, and other health care staff. In cases of severe or fulminant disease, infectious diseases, gastroenterology, and/or general surgery consultation is indicated. Guideline Guideline (Figure 1, page 5): Regional Clostridium difficile infection guidelines were created to standardize actions that clinicians and health care staff should follow in the treatment and management of CDAD in adult patients. In particular, this guideline aims to identify those patients who need escalation of therapy to vancomycin and/or urgent surgical intervention. It is expected that the guideline should apply to most patients; individual clinical judgment is always required. This document will be updated periodically as new evidence-based data becomes available. Stakeholders The CDAD management guideline has been reviewed and endorsed by the following stakeholder groups: Infectious Diseases, Medical Microbiology and Infection Control, Pharmacy, Gastroenterology, General Surgery, Critical Care, and Stem Cell Transplantation. Monitoring Daily vital signs (temperature, blood pressure) Daily assessment for number of diarrheal episodes and consistency. Diarrhea should resolve within 4-6 days. Failure to resolve in 6-7 days is considered treatment failure. Daily abdominal examination to determine clinical resolution or progression to more severe disease. Bloodwork for CBC and differential, electrolytes and creatinine as clinically indicated. (A recent albumin should be obtained.) Increasing WBC or left shift, hypotension, or acute renal failure are indications for switching from metronidazole to vancomycin (where applicable), and for ID, GI, and/or general surgery consultation. C. difficile testing should not be used to determine treatment endpoint, as tests may remain positive several months after the episode. Note: Metronidazole IV is considered a secondline agent compared to metronidazole PO and vancomycin PO treatment. Vancomycin IV is not effective for the treatment of CDAD. Alternative therapies (e.g. cholestyramine, probiotics, or other antimicrobial agents) should not be used routinely. References 1. Providence Health Care. Guidelines for management of suspected/confirmed Clostridium difficile-associated diarrhea (CDAD) Providence Health Care pathway. September Zar FA et al. A comparison of vancomycin and metronidazole for the treatment of Clostridium difficile-associated diarrhea, stratified by disease severity. CID 2007;45: Kelly CP et al. Clostridium difficile more difficult than ever. NEJM 2008;359: Gerding DN et al. Treatment of Clostridium difficile infection. CID 2008;46:S Owens RC. Clostridium difficile-associated disease: an emerging threat to patient safety. Pharmacother 2006;26: Henrich TJ et al. Clinical risk factors for severe Clostridium difficile-associated diarrhea. Emerging Infectious Diseases 2009;15:
5 Drug and Therapeutics Newsletter 5 Volume 16, Number 3 Figure 1. Clostridium difficile-associated Disease (CDAD) Guideline
PHARMACY AND THERAPEUTICS NEWSLETTER
 Apr 2015 ` Volume 22 Number 1 In This Issue... Changes to Formulary 1 Comparison of Oral Anticoagulant Agents 2 PDTM 2015 Distribution 3 Pediatric and Neonatal Drug Dosing in PDTM 3 Health Canada MedEffect
Apr 2015 ` Volume 22 Number 1 In This Issue... Changes to Formulary 1 Comparison of Oral Anticoagulant Agents 2 PDTM 2015 Distribution 3 Pediatric and Neonatal Drug Dosing in PDTM 3 Health Canada MedEffect
Clostridium difficile Infection (CDI) Guideline
 Clostridium difficile Infection (CDI) Guideline Site Applicability All VCH PHC acute care and residential sites. Practice Level Physicians basic skill Pharmacists basic skill Nurses (RN, LPN, RPN) basic
Clostridium difficile Infection (CDI) Guideline Site Applicability All VCH PHC acute care and residential sites. Practice Level Physicians basic skill Pharmacists basic skill Nurses (RN, LPN, RPN) basic
PPP 1. Continuation, modification, and discontinuation of a medication
 PRESCRIBING POLICIES: 4.7 PHARMACIST AUTHORITY The College of Pharmacists of BC Professional Practice Policy (PPP) 58 Medication Management (Adapting a Prescription) became effective April 1, 2009. The
PRESCRIBING POLICIES: 4.7 PHARMACIST AUTHORITY The College of Pharmacists of BC Professional Practice Policy (PPP) 58 Medication Management (Adapting a Prescription) became effective April 1, 2009. The
Clostridium difficile Infection (CDI) Management Guideline
 Clostridium difficile Infection (CDI) Management Guideline Do not test all patients with loose or watery stools for CDI o CDI is responsible for
Clostridium difficile Infection (CDI) Management Guideline Do not test all patients with loose or watery stools for CDI o CDI is responsible for
Division of GIM Lecture Series Case Presentation David A. Erickson, M.D October 9th, 2013
 Division of GIM Lecture Series Case Presentation David A. Erickson, M.D October 9th, 2013 Financial Disclosures No financial disclosures Objectives Review a case of recurrent Clostridium difficile infection
Division of GIM Lecture Series Case Presentation David A. Erickson, M.D October 9th, 2013 Financial Disclosures No financial disclosures Objectives Review a case of recurrent Clostridium difficile infection
Updated Clostridium difficile Treatment Guidelines
 Updated Clostridium difficile Treatment Guidelines Arielle Arnold, PharmD, BCPS Clinical Pharmacist Saint Alphonsus Regional Medical Center September 29 th, 2018 Disclosures Nothing to disclose Learning
Updated Clostridium difficile Treatment Guidelines Arielle Arnold, PharmD, BCPS Clinical Pharmacist Saint Alphonsus Regional Medical Center September 29 th, 2018 Disclosures Nothing to disclose Learning
Stony Brook Adult Clostridium difficile Management Guidelines. Discontinue all unnecessary antibiotics
 Stony Brook Adult Clostridium difficile Management Guidelines Summary: Use of the C Diff Infection (CDI) PowerPlan (Adult) Required Patient with clinical findings suggestive of Clostridium difficile infection
Stony Brook Adult Clostridium difficile Management Guidelines Summary: Use of the C Diff Infection (CDI) PowerPlan (Adult) Required Patient with clinical findings suggestive of Clostridium difficile infection
Chapter 5 Gastroenterology. Dose. Route. Units. Given. Dose. Route. Units. Given
 Chapter 5 Gastroenterology Georgia Woodfield TERLIPRESSIN CIPROFLOXACIN 2 initial dose 500 Every 4 hours until bleeding controlled can be reduced to 1 after initial dose if side effects develop or if patient
Chapter 5 Gastroenterology Georgia Woodfield TERLIPRESSIN CIPROFLOXACIN 2 initial dose 500 Every 4 hours until bleeding controlled can be reduced to 1 after initial dose if side effects develop or if patient
! Macrolide antibacterial. Fidaxomicin (Dificid ) package labeling. Optimer Pharmaceuticals, Inc. May 2011.
 Disclosure! I have no conflicts of interest related to this presentation Nina Naeger Murphy, Pharm.D., BCPS Clinical Pharmacy Specialist Infectious Diseases MetroHealth Medical Center Learning Objectives!
Disclosure! I have no conflicts of interest related to this presentation Nina Naeger Murphy, Pharm.D., BCPS Clinical Pharmacy Specialist Infectious Diseases MetroHealth Medical Center Learning Objectives!
Clostridium difficile Infection: Diagnosis and Management
 Clostridium difficile Infection: Diagnosis and Management Brian Viviano D.O. Case study 42 year old female with history of essential hypertension and COPD presents to ED complaining of 24 hours of intractable,
Clostridium difficile Infection: Diagnosis and Management Brian Viviano D.O. Case study 42 year old female with history of essential hypertension and COPD presents to ED complaining of 24 hours of intractable,
Updates to pharmacological management in the prevention of recurrent Clostridium difficile
 Updates to pharmacological management in the prevention of recurrent Clostridium difficile Julia Shlensky, PharmD PGY2 Internal Medicine Resident September 12, 2017 2017 MFMER slide-1 Clinical Impact Increasing
Updates to pharmacological management in the prevention of recurrent Clostridium difficile Julia Shlensky, PharmD PGY2 Internal Medicine Resident September 12, 2017 2017 MFMER slide-1 Clinical Impact Increasing
March 3, To: Hospitals, Long Term Care Facilities, and Local Health Departments
 March 3, 2010 To: Hospitals, Long Term Care Facilities, and Local Health Departments From: NYSDOH Bureau of Healthcare Associated Infections HEALTH ADVISORY: GUIDANCE FOR PREVENTION AND CONTROL OF HEALTHCARE
March 3, 2010 To: Hospitals, Long Term Care Facilities, and Local Health Departments From: NYSDOH Bureau of Healthcare Associated Infections HEALTH ADVISORY: GUIDANCE FOR PREVENTION AND CONTROL OF HEALTHCARE
MAXIMUM ALLOWABLE COST POLICY CHANGES DECEMBER 5, 2016 QUESTIONS AND ANSWERS
 MAXIMUM ALLOWABLE COST POLICY CHANGES DECEMBER 5, 2016 QUESTIONS AND ANSWERS The November 22, 2016 Mid-Year Financial Report referred to changes to drug coverage under the Saskatchewan Drug Plan. What
MAXIMUM ALLOWABLE COST POLICY CHANGES DECEMBER 5, 2016 QUESTIONS AND ANSWERS The November 22, 2016 Mid-Year Financial Report referred to changes to drug coverage under the Saskatchewan Drug Plan. What
Calgary Long Term Care Formulary
 Page 1 of 10 Calgary Long Term Care Formulary Pharmacy & Therapeutics November 2018 Highlights https://www.albertahealthservices.ca/info/page4071.aspx Page 2 of 10 Contents November 2018... 3 Formulary
Page 1 of 10 Calgary Long Term Care Formulary Pharmacy & Therapeutics November 2018 Highlights https://www.albertahealthservices.ca/info/page4071.aspx Page 2 of 10 Contents November 2018... 3 Formulary
EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE
 EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click
EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click
ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE
 ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE The diagnosis of CDI should be based on a combination of clinical and laboratory findings. A case definition for the usual
ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE The diagnosis of CDI should be based on a combination of clinical and laboratory findings. A case definition for the usual
Bezlotoxumab (Zinplava) as Adjunct Treatment for Clostridium difficile. Janel Liane Cala, RPh Medical Center Hospital
 Bezlotoxumab (Zinplava) as Adjunct Treatment for Clostridium difficile Janel Liane Cala, RPh Medical Center Hospital Objectives Review pathophysiology, risk factors, prevention, and treatment options of
Bezlotoxumab (Zinplava) as Adjunct Treatment for Clostridium difficile Janel Liane Cala, RPh Medical Center Hospital Objectives Review pathophysiology, risk factors, prevention, and treatment options of
Acute Gastroenteritis, Adult Emergency Orders
 Form Title Form Number 21003 2018, Alberta Health Services, CKCM This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. The license does not
Form Title Form Number 21003 2018, Alberta Health Services, CKCM This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. The license does not
Calgary Long Term Care Formulary
 Page 1 of 14 Calgary Long Term Care Formulary Pharmacy & Therapeutics Highlights https://www.albertahealthservices.ca/info/page4071.aspx Page 2 of 14 Contents... 3 Formulary Changes (Additions, Changes,
Page 1 of 14 Calgary Long Term Care Formulary Pharmacy & Therapeutics Highlights https://www.albertahealthservices.ca/info/page4071.aspx Page 2 of 14 Contents... 3 Formulary Changes (Additions, Changes,
Clostridium DifficileInfection & Readmissions: An ounce of prevention is worth a pound of cure
 Clostridium DifficileInfection & Readmissions: An ounce of prevention is worth a pound of cure Brian S. Zuckerbraun, MD, FACS Henry T. Bahnson Professor of Surgery University of Pittsburgh Chief, Trauma
Clostridium DifficileInfection & Readmissions: An ounce of prevention is worth a pound of cure Brian S. Zuckerbraun, MD, FACS Henry T. Bahnson Professor of Surgery University of Pittsburgh Chief, Trauma
Long-Term Care Updates
 Long-Term Care Updates April 2018 By Austin Smith, PharmD Candidate and Lindsay Slowiczek, PharmD is the most common healthcare-acquired infection (HAI) in the United States. 1,2 A 2014 prevalence survey
Long-Term Care Updates April 2018 By Austin Smith, PharmD Candidate and Lindsay Slowiczek, PharmD is the most common healthcare-acquired infection (HAI) in the United States. 1,2 A 2014 prevalence survey
Long-Term Care Updates
 Long-Term Care Updates April 2017 Bezlotoxumab to Prevent Recurrent Infection By Amy Wilson, PharmD and Zara Risoldi Cochrane, PharmD, MS, FASCP Introduction The Gram-positive bacteria is a common cause
Long-Term Care Updates April 2017 Bezlotoxumab to Prevent Recurrent Infection By Amy Wilson, PharmD and Zara Risoldi Cochrane, PharmD, MS, FASCP Introduction The Gram-positive bacteria is a common cause
Appropriate Use of Proton Pump Inhibitors (PPIs) Anderson Mabour, Pharm.D., BCPS Clinical Pharmacy Specialist
 Appropriate Use of Proton Pump Inhibitors (PPIs) Anderson Mabour, Pharm.D., BCPS Clinical Pharmacy Specialist Disclosures I have no actual or potential conflicts of interest to report in relation to this
Appropriate Use of Proton Pump Inhibitors (PPIs) Anderson Mabour, Pharm.D., BCPS Clinical Pharmacy Specialist Disclosures I have no actual or potential conflicts of interest to report in relation to this
Pharmacy Coverage Guidelines are subject to change as new information becomes available.
 PROTON PUMP INHIBITORS, NON-PREFERRED FORMS: ACIPHEX (rabeprazole sodium EC) oral tablet ACIPHEX SPRINKLE (rabeprazole sodium DR) oral capsule ESOMEPRAZOLE STRONTIUM (esomeprazole strontium DR) oral capsule
PROTON PUMP INHIBITORS, NON-PREFERRED FORMS: ACIPHEX (rabeprazole sodium EC) oral tablet ACIPHEX SPRINKLE (rabeprazole sodium DR) oral capsule ESOMEPRAZOLE STRONTIUM (esomeprazole strontium DR) oral capsule
PEDIATRIC SPINE SURGERY POST-OP PLAN - Phase:.
 - Phase:. PHYSICIAN S Diagnosis Weight Allergies DETAILS Patient Care Patient Activity Bedrest Maintain Surgical Drain Maintain JP Drain, Measure Output q12h, and PRN Convert IV to INT when tolerating
- Phase:. PHYSICIAN S Diagnosis Weight Allergies DETAILS Patient Care Patient Activity Bedrest Maintain Surgical Drain Maintain JP Drain, Measure Output q12h, and PRN Convert IV to INT when tolerating
ADMIT DIABETIC KETOACIDOSIS (DKA) PLAN - Phase: Begin Immediately/Emergency Center
 - Phase: Begin Immediately/Emergency Center Weight PHYSICIAN S Allergies Admit/Discharge/Transfer Patient Status Requested Location: MICU, Pt Status: Inpatient (LOS > 2 midnights) Requested Location: 5E
- Phase: Begin Immediately/Emergency Center Weight PHYSICIAN S Allergies Admit/Discharge/Transfer Patient Status Requested Location: MICU, Pt Status: Inpatient (LOS > 2 midnights) Requested Location: 5E
SUMMARY. Journal of Clinical Pharmacy and Therapeutics, 2015, 40, doi: /jcpt.12329
 Journal of Clinical Pharmacy and Therapeutics, 2015, 40, 655 660 doi: 10.1111/jcpt.12329 Evaluation of a Clostridium difficile infection management policy with clinical pharmacy and medical microbiology
Journal of Clinical Pharmacy and Therapeutics, 2015, 40, 655 660 doi: 10.1111/jcpt.12329 Evaluation of a Clostridium difficile infection management policy with clinical pharmacy and medical microbiology
Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review
 Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review October 18, 2010 James Kahn and Carolyn Kenney, MSIV Overview Burden of disease associated
Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review October 18, 2010 James Kahn and Carolyn Kenney, MSIV Overview Burden of disease associated
Learning Objectives. A Critical Analysis of Recent Fidaxomicin & Nesiritide Trials. Fidaxomicin vs. Vancomycin. Outline 1/3/2012
 Learning Objectives A Critical Analysis of Recent Fidaxomicin & Nesiritide Trials Amy E. Lodolce, PharmD, BCPS Assistant Director, Drug Information Group UIC College of Pharmacy Describe the methods and
Learning Objectives A Critical Analysis of Recent Fidaxomicin & Nesiritide Trials Amy E. Lodolce, PharmD, BCPS Assistant Director, Drug Information Group UIC College of Pharmacy Describe the methods and
Objectives: This presentation will help you to:
 emergency Drugs Objectives: This presentation will help you to: Five rights for medication administration Recognize different cardiac arrhythmias and determine the common drugs used for each one List the
emergency Drugs Objectives: This presentation will help you to: Five rights for medication administration Recognize different cardiac arrhythmias and determine the common drugs used for each one List the
Case 1. Which of the following would be next appropriate investigation/s regarding the pts diarrhoea?
 Case 1 21 yr old HIV +ve, Cd4-100 HAART naïve Profuse diarrhoea for 3/52. Stool MC&S ve Which of the following would be next appropriate investigation/s regarding the pts diarrhoea? Repeat stool MC&S Stool
Case 1 21 yr old HIV +ve, Cd4-100 HAART naïve Profuse diarrhoea for 3/52. Stool MC&S ve Which of the following would be next appropriate investigation/s regarding the pts diarrhoea? Repeat stool MC&S Stool
Modern approach to Clostridium Difficile Infection
 Modern approach to Clostridium Difficile Infection Pseudomembranous Colitis: Principles for diagnosis and treatment Aggelos Stefos Internist, Infectious diseases Specialist Department of Medicine and Research
Modern approach to Clostridium Difficile Infection Pseudomembranous Colitis: Principles for diagnosis and treatment Aggelos Stefos Internist, Infectious diseases Specialist Department of Medicine and Research
BC Cancer Protocol Summary for the Maintenance Therapy of Multiple Myeloma Using Bortezomib for Patients with the High-Risk Chromosome Abnormality
 BC Cancer Protocol Summary for the Maintenance Therapy of Multiple Myeloma Using Bortezomib for Patients with the High-Risk Chromosome Abnormality Protocol Code Tumour Group Contact Physician MYBORMTN
BC Cancer Protocol Summary for the Maintenance Therapy of Multiple Myeloma Using Bortezomib for Patients with the High-Risk Chromosome Abnormality Protocol Code Tumour Group Contact Physician MYBORMTN
Terapia dell infezione da Clostridium difficile. Massimo Coen I Div Mal Inf AO L Sacco
 Terapia dell infezione da Clostridium difficile Massimo Coen I Div Mal Inf AO L Sacco Disease Severity Mild CDI 3 5 BM/day WBC 15,000/mm 3 Defining CDI Disease Severity Mild abdominal pain due to CDI Moderate
Terapia dell infezione da Clostridium difficile Massimo Coen I Div Mal Inf AO L Sacco Disease Severity Mild CDI 3 5 BM/day WBC 15,000/mm 3 Defining CDI Disease Severity Mild abdominal pain due to CDI Moderate
Journey to Decreasing Clostridium Difficile and the Unexpected Twist. Jackie Morton, Infection Prevention Cortney Swiggart, Medication Safety Officer
 Journey to Decreasing Clostridium Difficile and the Unexpected Twist Jackie Morton, Infection Prevention Cortney Swiggart, Medication Safety Officer 4/13/2018 Objectives Discuss the organism and clinical
Journey to Decreasing Clostridium Difficile and the Unexpected Twist Jackie Morton, Infection Prevention Cortney Swiggart, Medication Safety Officer 4/13/2018 Objectives Discuss the organism and clinical
Proton Pump Inhibitors. Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.01 Subject: Proton Pump Inhibitors Page: 1 of 6 Last Review Date: June 24, 2015 Proton Pump Inhibitors
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.01 Subject: Proton Pump Inhibitors Page: 1 of 6 Last Review Date: June 24, 2015 Proton Pump Inhibitors
Drug Class Monograph
 Drug Class Monograph Class: Proton Pump Inhibitors Drugs: Aciphex Sprinkle (rabeprazole), Dexilant (dexlansoprazole), Lansoprazole, Nexium (esomeprazole capsule, esomeprazole granules), Omeprazole, Pantoprazole,
Drug Class Monograph Class: Proton Pump Inhibitors Drugs: Aciphex Sprinkle (rabeprazole), Dexilant (dexlansoprazole), Lansoprazole, Nexium (esomeprazole capsule, esomeprazole granules), Omeprazole, Pantoprazole,
Physician Orders ADULT: Kidney-Panc/PancTransplant Post Op Plan
 Initiate Orders Phase Care Sets/Protocols/PowerPlans Initiate Powerplan Phase T;N, Phase: Kidney-Panc/Panc Transp Post Op Phase, When to Initiate: Kidney-Panc/Panc Transp Post Op Phase Vital Signs Vital
Initiate Orders Phase Care Sets/Protocols/PowerPlans Initiate Powerplan Phase T;N, Phase: Kidney-Panc/Panc Transp Post Op Phase, When to Initiate: Kidney-Panc/Panc Transp Post Op Phase Vital Signs Vital
II. Angiotensin Receptor Blockers (ARBs) Drug Class Review
 DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE DOD BENEFICIARY ADVISORY PANEL I. Uniform Formulary Review Process Under 10 U.S.C. 1074g, as implemented by 32 C.F.R. 199.21,
DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE DOD BENEFICIARY ADVISORY PANEL I. Uniform Formulary Review Process Under 10 U.S.C. 1074g, as implemented by 32 C.F.R. 199.21,
PHYSICIAN SIGNATURE DATE TIME DRUG ALLERGIES WT: KG
 DRUG AND TREATMENT Available ONLY at: BMC-B BMC-D BMC-N BMC-S Nursing Orders Communication Order If CVP unavailable, administer fluid boluses every 30 minutes except monitor O2 requirements Comments: Every
DRUG AND TREATMENT Available ONLY at: BMC-B BMC-D BMC-N BMC-S Nursing Orders Communication Order If CVP unavailable, administer fluid boluses every 30 minutes except monitor O2 requirements Comments: Every
GUIDELINE FOR THE MANAGEMENT OF ANTIBIOTIC- ASSOCIATED DIARRHOEA IN ADULTS
 GUIDELINE FOR THE MANAGEMENT OF ANTIBIOTIC- ASSOCIATED DIARRHOEA IN ADULTS Version 3.0 Date ratified May 2008 Review date May 2010 Ratified by NUH Antibiotic Guidelines Committee NUH Drugs and Therapeutics
GUIDELINE FOR THE MANAGEMENT OF ANTIBIOTIC- ASSOCIATED DIARRHOEA IN ADULTS Version 3.0 Date ratified May 2008 Review date May 2010 Ratified by NUH Antibiotic Guidelines Committee NUH Drugs and Therapeutics
Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate
 Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate Objectives Summarize the changing epidemiology and demographics of patients at risk for Clostridium
Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate Objectives Summarize the changing epidemiology and demographics of patients at risk for Clostridium
Management of Clostridium Difficile: Total Colectomy versus Colon Sparing Surgery
 Management of Clostridium Difficile: Total Colectomy versus Colon Sparing Surgery Rahul Narang, MD Colon and Rectal Surgery Assistant Professor of Surgery No Disclosure Clostridium Difficile Colitis: Treatments,
Management of Clostridium Difficile: Total Colectomy versus Colon Sparing Surgery Rahul Narang, MD Colon and Rectal Surgery Assistant Professor of Surgery No Disclosure Clostridium Difficile Colitis: Treatments,
ABSTRACT PURPOSE METHODS
 ABSTRACT PURPOSE The purpose of this study was to characterize the CDI population at this institution according to known risk factors and to examine the effect of appropriate evidence-based treatment selection
ABSTRACT PURPOSE The purpose of this study was to characterize the CDI population at this institution according to known risk factors and to examine the effect of appropriate evidence-based treatment selection
BNF CHAPTER 1: GASTRO-INTESTINAL SYSTEM. 1 April 13
 BNF CHAPTER 1: GASTRO-INTESTINAL SYSTEM 1 BNF 1.1 ANTACIDS Mucogel and Peptac are effective antacids and are available over the counter Gaviscon / Gaviscon Advance are available over the counter First
BNF CHAPTER 1: GASTRO-INTESTINAL SYSTEM 1 BNF 1.1 ANTACIDS Mucogel and Peptac are effective antacids and are available over the counter Gaviscon / Gaviscon Advance are available over the counter First
DONATION AFTER CARDIAC DEATH PLAN
 DONATION AFTER CARDIAC DEATH PLAN Diagnosis Weight Allergies Patient Care Core Body Temperature Monitoring Maintain body temp 96-99 degrees Farenheit. Utilize Hyper/Hypothermia blanket prn Insert Gastric
DONATION AFTER CARDIAC DEATH PLAN Diagnosis Weight Allergies Patient Care Core Body Temperature Monitoring Maintain body temp 96-99 degrees Farenheit. Utilize Hyper/Hypothermia blanket prn Insert Gastric
A Pharmacist Perspective
 Leveraging Technology to Reduce CDI A Pharmacist Perspective Ed Eiland, Pharm.D., MBA, BCPS (AQ-ID) Clinical Practice and Business Supervisor Huntsville Hospital System Huntsville Hospital 881 licensed
Leveraging Technology to Reduce CDI A Pharmacist Perspective Ed Eiland, Pharm.D., MBA, BCPS (AQ-ID) Clinical Practice and Business Supervisor Huntsville Hospital System Huntsville Hospital 881 licensed
GOOVIPPC. Protocol Code: Gynecology. Tumour Group: Paul Hoskins. Contact Physician: James Conklin. Contact Pharmacist:
 BCCA Protocol Summary for Primary Treatment of Stage III less than or equal to 1 cm Visible Residual Invasive Epithelial Ovarian Cancer or Stage I Grade 3 or Stage II Grade 3 Papillary Serous Ovarian Cancer
BCCA Protocol Summary for Primary Treatment of Stage III less than or equal to 1 cm Visible Residual Invasive Epithelial Ovarian Cancer or Stage I Grade 3 or Stage II Grade 3 Papillary Serous Ovarian Cancer
DISCLAIMER: ECHO Nevada emphasizes patient privacy and asks participants to not share ANY Protected Health Information during ECHO clinics.
 DISCLAIMER: Video will be taken at this clinic and potentially used in Project ECHO promotional materials. By attending this clinic, you consent to have your photo taken and allow Project ECHO to use this
DISCLAIMER: Video will be taken at this clinic and potentially used in Project ECHO promotional materials. By attending this clinic, you consent to have your photo taken and allow Project ECHO to use this
Stressed Out: Evaluating the Need for Stress Ulcer Prophylaxis in the ICU
 Stressed Out: Evaluating the Need for Stress Ulcer Prophylaxis in the ICU Josh Arnold, PharmD PGY1 Pharmacy Resident Pharmacy Grand Rounds November 8, 2016 2016 MFMER slide-1 Objectives Identify the significance
Stressed Out: Evaluating the Need for Stress Ulcer Prophylaxis in the ICU Josh Arnold, PharmD PGY1 Pharmacy Resident Pharmacy Grand Rounds November 8, 2016 2016 MFMER slide-1 Objectives Identify the significance
Labeled Uses: Treatment of Clostiridum Difficile associated diarrhea (CDAD)
 Brand Name: Dificid Generic Name: fidaxomicin Manufacturer 1,2,3,4,5 : Optimer Pharmaceuticals, Inc. Drug Class 1,2,3,4,5 : Macrolide Antibiotic Uses 1,2,3,4,5 : Labeled Uses: Treatment of Clostiridum
Brand Name: Dificid Generic Name: fidaxomicin Manufacturer 1,2,3,4,5 : Optimer Pharmaceuticals, Inc. Drug Class 1,2,3,4,5 : Macrolide Antibiotic Uses 1,2,3,4,5 : Labeled Uses: Treatment of Clostiridum
Organ Donor Management Recommended Guidelines ADULT CARDIAC DEATH (DCD)
 Date: Time: = Always applicable = Check if applicable ADMISSION INSTRUCTIONS Move to Comfort Care Note in chart. Contact initiated with BC Transplant Consent for Organ Donation obtained Code Status: Full
Date: Time: = Always applicable = Check if applicable ADMISSION INSTRUCTIONS Move to Comfort Care Note in chart. Contact initiated with BC Transplant Consent for Organ Donation obtained Code Status: Full
Proton Pump Inhibitors. Description. Section: Prescription Drugs Effective Date: July 1, 2014
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.09.01 Subject: Proton Pump Inhibitors Page: 1 of 7 Last Review Date: June 12, 2014 Proton Pump Inhibitors
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.09.01 Subject: Proton Pump Inhibitors Page: 1 of 7 Last Review Date: June 12, 2014 Proton Pump Inhibitors
Anti-Influenza Agents Quantity Limit Program Summary
 Anti-Influenza Agents Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1,2 Agent Indication Dosage & Administration Relenza Treatment of influenza in Treatment of influenza: (zanamivir) patients
Anti-Influenza Agents Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1,2 Agent Indication Dosage & Administration Relenza Treatment of influenza in Treatment of influenza: (zanamivir) patients
-2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine. -June 2008: Recurrence of rectal blood loss and urgency
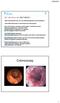 SD, male 40 yrs. old. (680718M467.) -2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine -June 2008: Recurrence of rectal blood loss and urgency Total colonoscopy: ulcerative rectitis,
SD, male 40 yrs. old. (680718M467.) -2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine -June 2008: Recurrence of rectal blood loss and urgency Total colonoscopy: ulcerative rectitis,
Literature Scan: Antibiotics for Clostridium difficile Infection. Month/Year of Review: May 2015 Date of Last Review: April 2012
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
more intense treatments are needed to get rid of the infection.
 What Is Clostridium Difficile (C. Diff)? Clostridium difficile, or C. diff for short, is an infection from a bacterium that can grow in your intestines and cause bad GI symptoms. The main risk of getting
What Is Clostridium Difficile (C. Diff)? Clostridium difficile, or C. diff for short, is an infection from a bacterium that can grow in your intestines and cause bad GI symptoms. The main risk of getting
Organ Donor Management Recommended Guidelines ADULT Brain Death (NDD)
 Date: Time: = Always applicable = Check if applicable ADMISSION INSTRUCTIONS Neurological Determination of Death (NDD) has been performed by at least 2 licensed physicians Contact initiated with BC Transplant
Date: Time: = Always applicable = Check if applicable ADMISSION INSTRUCTIONS Neurological Determination of Death (NDD) has been performed by at least 2 licensed physicians Contact initiated with BC Transplant
PEDIATRIC SPINE SURGERY POST-OP PLAN - Phase: Pediatric Spine Surgery General Orders
 - Phase: Pediatric Spine Surgery General Orders PHYSICIAN S Diagnosis Weight Allergies Patient Care Patient Activity Bedrest Maintain Surgical Drain Maintain JP Drain, Measure Output q12h, and PRN Convert
- Phase: Pediatric Spine Surgery General Orders PHYSICIAN S Diagnosis Weight Allergies Patient Care Patient Activity Bedrest Maintain Surgical Drain Maintain JP Drain, Measure Output q12h, and PRN Convert
Proton Pump Inhibitors:
 Proton Pump Inhibitors: How bad could they be? Andrea Flanagan, Pharm.D. Iowa City VA Medical Center PGY-1 Pharmacy Resident Objectives for Pharmacists At the end of this presentation PHARMACISTS should
Proton Pump Inhibitors: How bad could they be? Andrea Flanagan, Pharm.D. Iowa City VA Medical Center PGY-1 Pharmacy Resident Objectives for Pharmacists At the end of this presentation PHARMACISTS should
Inflammatory Bowel Disease
 + Inflammatory Bowel Disease Christina Kalafsky, Dietetic Intern University of Maryland College Park Children s National Medical Center Case Study January 31, 2014 + Outline n Inflammatory Bowel Disease
+ Inflammatory Bowel Disease Christina Kalafsky, Dietetic Intern University of Maryland College Park Children s National Medical Center Case Study January 31, 2014 + Outline n Inflammatory Bowel Disease
DRUG ORDERING & DISPENSING:
 STUDY DRUG: List the study drugs that will be used at your site based on your hospital formulary 1. Insulin Humulin R 2. Insulin Humalog 3. Insulin Lantus DRUG SUPPLY & STORAGE: Since study drugs are not
STUDY DRUG: List the study drugs that will be used at your site based on your hospital formulary 1. Insulin Humulin R 2. Insulin Humalog 3. Insulin Lantus DRUG SUPPLY & STORAGE: Since study drugs are not
Sept 2004 Volume 11, Number 3. Non-micronized
 Sept 2004 Volume 11, Number 3 In This Issue... Changes to Formulary... 1 Pharmacists Ordering Drug Levels.. 2 Revised Drug Administration Policies. 2 Tiotropium.. 2 Bisoprolol. 4 Pharmacy Residency Accreditation
Sept 2004 Volume 11, Number 3 In This Issue... Changes to Formulary... 1 Pharmacists Ordering Drug Levels.. 2 Revised Drug Administration Policies. 2 Tiotropium.. 2 Bisoprolol. 4 Pharmacy Residency Accreditation
POST-OP CARDIAC SURGERY PHYSICIAN S ORDER SHEET USE BALLPOINT PEN ONLY. CARDIAC INTENSIVE CARE UNIT
 PHYSICIAN S SHEET Automatically Activate, if not in agreement, cross out and initial Activated by Checking Box ALLERGIES: None known YES Patient s Height: Patient s Weight: ALL MEDICATION and INTRAVENOUS
PHYSICIAN S SHEET Automatically Activate, if not in agreement, cross out and initial Activated by Checking Box ALLERGIES: None known YES Patient s Height: Patient s Weight: ALL MEDICATION and INTRAVENOUS
Omeprazole 10mg. Name, Restriction, Manner of administration and form OMEPRAZOLE omeprazole 10 mg enteric tablet, 30 (8332M) Max. Qty.
 Omeprazole 10mg Name, Restriction, Manner of administration and form omeprazole 10 mg enteric tablet, 30 (8332M) Gastro-oesophageal reflux disease Name, Restriction, Manner of administration and form omeprazole
Omeprazole 10mg Name, Restriction, Manner of administration and form omeprazole 10 mg enteric tablet, 30 (8332M) Gastro-oesophageal reflux disease Name, Restriction, Manner of administration and form omeprazole
PHARMACY AND THERAPEUTICS NEWSLETTER
 Feb 2017 ` Volume 24 Number 1 In This Issue... Changes to Formulary 1 Lidocaine Infusions for Pain Management 1 Hyperkalemia Management PPO 2 Buprenorphine-Naloxone (Suboxone ) Tablets 2 Fentanyl Subcutaneous
Feb 2017 ` Volume 24 Number 1 In This Issue... Changes to Formulary 1 Lidocaine Infusions for Pain Management 1 Hyperkalemia Management PPO 2 Buprenorphine-Naloxone (Suboxone ) Tablets 2 Fentanyl Subcutaneous
Proton Pump Inhibitor De-prescribing Guidance
 Amendment History Proton Pump Inhibitor De-prescribing Guidance VERSION DATE AMENDMENT HISTORY 1.0 2013 Previous version 2.0 September 2015 Comments Amendment to Flow chart and addition of Rationale page
Amendment History Proton Pump Inhibitor De-prescribing Guidance VERSION DATE AMENDMENT HISTORY 1.0 2013 Previous version 2.0 September 2015 Comments Amendment to Flow chart and addition of Rationale page
Family Medicine Clinical Pharmacy Forum Vol. 4, Issue 5 (September/October 2008)
 1 Family Medicine Clinical Pharmacy Forum Vol. 4, Issue 5 (September/October 2008) Family Medicine Clinical Pharmacy Forum is a brief bi-monthly publication from the Family Medicine clinical pharmacists
1 Family Medicine Clinical Pharmacy Forum Vol. 4, Issue 5 (September/October 2008) Family Medicine Clinical Pharmacy Forum is a brief bi-monthly publication from the Family Medicine clinical pharmacists
ORTHO TOTAL KNEE REPLACEMENT POST-OP PLAN - Phase: PACU Ortho Phase
 - Phase: PACU Ortho Phase PHYSICIAN S Diagnosis Weight Allergies DETAILS Admit/Discharge/Transfer Patient Status Patient Condition Acuity Level Floor Status Acuity Level Critical Acuity Level Intermediate
- Phase: PACU Ortho Phase PHYSICIAN S Diagnosis Weight Allergies DETAILS Admit/Discharge/Transfer Patient Status Patient Condition Acuity Level Floor Status Acuity Level Critical Acuity Level Intermediate
Physician Orders ADULT: Acute MI/Acute Coronary Syndrome Adult Plan
 Initiate Orders Phase Care Sets/Protocols/PowerPlans Initiate Powerplan Phase Phase: Acute MI/Acute Coronary Syndrome Adult Phase, When to Initiate: Acute MI/Acute Coronary Syndrome Adlt Phase Non Categorized
Initiate Orders Phase Care Sets/Protocols/PowerPlans Initiate Powerplan Phase Phase: Acute MI/Acute Coronary Syndrome Adult Phase, When to Initiate: Acute MI/Acute Coronary Syndrome Adlt Phase Non Categorized
Bariatric Surgery Post Op Day Version 2 Approved 11/13/2017
 Patient Name: Diagnosis: Allergies with reaction type: Bariatric Surgery Post Op Day Version 2 Approved 11/13/2017 Diagnosis Preferred Location/Unit Surgical ICU Code Status: Full Code Activity Ambulate
Patient Name: Diagnosis: Allergies with reaction type: Bariatric Surgery Post Op Day Version 2 Approved 11/13/2017 Diagnosis Preferred Location/Unit Surgical ICU Code Status: Full Code Activity Ambulate
500,000 29,000. New 2015 Data. Lessa et al, N Eng J Med 2015: 34.2% of CDI cases were considered community-acquired
 Cost-effective Treatment of Clostridium difficile Infection in the ICU Kevin W. Garey, PharmD, MS. Professor and Chair University of Houston College of Pharmacy New 2015 Data 500,000 29,000 Lessa et al,
Cost-effective Treatment of Clostridium difficile Infection in the ICU Kevin W. Garey, PharmD, MS. Professor and Chair University of Houston College of Pharmacy New 2015 Data 500,000 29,000 Lessa et al,
INTERNATIONAL SOCIETY FOR HEART AND LUNG TRANSPLANTATION a Society that includes Basic Science, the Failing Heart, and Advanced Lung Disease
 International Society of Heart and Lung Transplantation Advisory Statement on the Implications of Pandemic Influenza for Thoracic Organ Transplantation This advisory statement has been produced by the
International Society of Heart and Lung Transplantation Advisory Statement on the Implications of Pandemic Influenza for Thoracic Organ Transplantation This advisory statement has been produced by the
ELIGIBILITY: Newly diagnosed acute promyelocytic leukemia (APL) with low to intermediate risk (WBC less than 10 x 10 9 /L)
 BCCA Protocol Summary for First-Line Induction and Consolidation Therapy of Acute Promyelocytic Leukemia Using Arsenic Trioxide and Tretinoin (All-Trans Retinoic Acid) Protocol Code Tumour Group Contact
BCCA Protocol Summary for First-Line Induction and Consolidation Therapy of Acute Promyelocytic Leukemia Using Arsenic Trioxide and Tretinoin (All-Trans Retinoic Acid) Protocol Code Tumour Group Contact
APIDRA (insulin glulisine) injection vial APIDRA SOLOSTAR (insulin glulisine) subcutaneous solution pen-injector
 APIDRA SOLOSTAR (insulin glulisine) subcutaneous solution pen-injector Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
APIDRA SOLOSTAR (insulin glulisine) subcutaneous solution pen-injector Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
Sample. Fractured Hip Post-Operative Orders. Legend < Mandatory fields o Optional fields. Height Allergies: List or o Up to date in electronic system
 Legend Mandatory fields o Optional fields Height Allergies: List or o Up to date in electronic system cm Weight Diagnosis kg Date (yyyy-mon-dd) Time (hh:mm) Anticipated Date Of Discharge (ADOD) o Greater
Legend Mandatory fields o Optional fields Height Allergies: List or o Up to date in electronic system cm Weight Diagnosis kg Date (yyyy-mon-dd) Time (hh:mm) Anticipated Date Of Discharge (ADOD) o Greater
Attach patient label here. Physician Orders ADULT: Palliative Care Plan
 Initiate Orders Phase Care Sets/Protocols/PowerPlans Initiate Powerplan Phase T;N, Phase: Palliative Care Phase, When to Initiate: Palliative Care Phase Admission/Transfer/Discharge Patient Status Initial
Initiate Orders Phase Care Sets/Protocols/PowerPlans Initiate Powerplan Phase T;N, Phase: Palliative Care Phase, When to Initiate: Palliative Care Phase Admission/Transfer/Discharge Patient Status Initial
MEDICATIONS IN PREGNANCY
 MEDICATIONS IN PREGNANCY If patient presents with significant past history of drug addiction pain control should be closely monitored due to increased tolerance to narcotics. Consult Pain Specialist and
MEDICATIONS IN PREGNANCY If patient presents with significant past history of drug addiction pain control should be closely monitored due to increased tolerance to narcotics. Consult Pain Specialist and
SUBMISSION FOR RECLASSIFICATION
 SUBMISSION FOR RECLASSIFICATION OF SODIUM PICOSULPHATE (PICOPREP Powder) July 2002 Submission Prepared by: Contact: Baxter Healthcare Ltd. Auckland Mr. Alan Stanley Technical Manager - 1 - PART A 1. International
SUBMISSION FOR RECLASSIFICATION OF SODIUM PICOSULPHATE (PICOPREP Powder) July 2002 Submission Prepared by: Contact: Baxter Healthcare Ltd. Auckland Mr. Alan Stanley Technical Manager - 1 - PART A 1. International
Colonoscopy Preparation. Dean N. Silas, M.D. Advocate Lutheran General Hospital
 Colonoscopy Preparation Dean N. Silas, M.D. Advocate Lutheran General Hospital Disclosure NO relevant financial conflicts of interest I will be mentioning off-label uses of medications For clarity I will
Colonoscopy Preparation Dean N. Silas, M.D. Advocate Lutheran General Hospital Disclosure NO relevant financial conflicts of interest I will be mentioning off-label uses of medications For clarity I will
IV Acetaminophen Pain Study for Neurosurgery Post Op Non ICU Admit Plan Begin Immediately/PACU
 Post Op Non ICU Admit Plan Begin Immediately/PACU PHYSICIAN S Weight Allergies Admit/Discharge/Transfer Request Patient Bed Requested Location: 3W, Pt Status: Inpatient (LOS > 2 midnights) Patient Condition
Post Op Non ICU Admit Plan Begin Immediately/PACU PHYSICIAN S Weight Allergies Admit/Discharge/Transfer Request Patient Bed Requested Location: 3W, Pt Status: Inpatient (LOS > 2 midnights) Patient Condition
SAALT3W Sarcoma Dr. Meg Knowling. Protocol Code Tumour Group Contact Physician
 BCCA Protocol Summary for Etoposide, Ifosfamide-Mesna (SAIME) Alternating with vincristine, DOXOrubicin and Cyclophosphamide (with or without Mesna)(SAVAC or SAVACM) with Filgrastim Support at a THREE
BCCA Protocol Summary for Etoposide, Ifosfamide-Mesna (SAIME) Alternating with vincristine, DOXOrubicin and Cyclophosphamide (with or without Mesna)(SAVAC or SAVACM) with Filgrastim Support at a THREE
Drug Max dose approved for IVP Dilution Rate Monitoring Parameters. Dilution not necessary (Available in prefilled syringe)
 Drug Max dose approved for IVP Dilution Rate Monitoring Parameters Acetazolamide 500 mg Reconstitute with at least 5ml sterile water (max concentration should not exceed 100mg/ml) 100-500 mg/min Hypotension
Drug Max dose approved for IVP Dilution Rate Monitoring Parameters Acetazolamide 500 mg Reconstitute with at least 5ml sterile water (max concentration should not exceed 100mg/ml) 100-500 mg/min Hypotension
The Infection Control Doctor and Clostridium difficile infection. Dr David R Jenkins University Hospitals of Leicester NHS Trust, England
 The Infection Control Doctor and Clostridium difficile infection Dr David R Jenkins University Hospitals of Leicester NHS Trust, England 250 200 150 100 50 0 Monthly cases of Clostridium difficile (UHL
The Infection Control Doctor and Clostridium difficile infection Dr David R Jenkins University Hospitals of Leicester NHS Trust, England 250 200 150 100 50 0 Monthly cases of Clostridium difficile (UHL
5 MUSCULOSKELETAL SYSTEM
 5 MUSCULOSKELETAL SYSTEM 5.01 NON-STEROIDAL ANTIILAMMATORY DRUGS (NSAIDS) *Acetylsalicylic Acid (Aspirin) Tab Soluble 300mg Diclofenac Sodium Tab 25mg, Supp 25mg, 50mg & 100mg (Voltaren) 300-900mg every
5 MUSCULOSKELETAL SYSTEM 5.01 NON-STEROIDAL ANTIILAMMATORY DRUGS (NSAIDS) *Acetylsalicylic Acid (Aspirin) Tab Soluble 300mg Diclofenac Sodium Tab 25mg, Supp 25mg, 50mg & 100mg (Voltaren) 300-900mg every
PHYSICIAN S ORDERS Page 1 of 1 Providence Hospital ICU Insulin Drip Protocol
 DTE PROVIDENCE HOSPITL 6801 irport Boulevard, Mobile L 36608, PHYSICIN S ORDERS Page 1 of 1 Providence Hospital ICU Insulin Drip Protocol 1. Discontinue all previous insulin and oral diabetic medications
DTE PROVIDENCE HOSPITL 6801 irport Boulevard, Mobile L 36608, PHYSICIN S ORDERS Page 1 of 1 Providence Hospital ICU Insulin Drip Protocol 1. Discontinue all previous insulin and oral diabetic medications
DRUG AND THERAPEUTICS NEWSLETTER
 DRUG AND THERAPEUTICS NEWSLETTER A publication of the CSU Pharmaceutical Sciences Vancouver General Hospital, UBC Hospital, GF Strong July 2005 Volume 12, Number 3 In This Issue... Changes to Formulary
DRUG AND THERAPEUTICS NEWSLETTER A publication of the CSU Pharmaceutical Sciences Vancouver General Hospital, UBC Hospital, GF Strong July 2005 Volume 12, Number 3 In This Issue... Changes to Formulary
Proton Pump Inhibitors Drug Class Prior Authorization Protocol
 Proton Pump Inhibitors Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: November 15, 2017 Effective Date: January 1, 2018 This policy has been developed through review
Proton Pump Inhibitors Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: November 15, 2017 Effective Date: January 1, 2018 This policy has been developed through review
Peramivir IV Questions and Answers for Health Care Providers
 Drugs Peramivir IV Questions and Answers for Health Care Providers Q1. What action is FDA taking regarding Peramivir IV? A. As part of the federal government s response to the 2009 H1N1 public health emergency,
Drugs Peramivir IV Questions and Answers for Health Care Providers Q1. What action is FDA taking regarding Peramivir IV? A. As part of the federal government s response to the 2009 H1N1 public health emergency,
Vanderbilt University Medical Center Trauma ICU Nutrition Management Guidelines
 Vanderbilt University Medical Center Trauma ICU Nutrition Management Guidelines Trauma Critical Care Nutrition Guidelines Clinical judgment may supersede guidelines as patient circumstances warrant ASSESSMENT
Vanderbilt University Medical Center Trauma ICU Nutrition Management Guidelines Trauma Critical Care Nutrition Guidelines Clinical judgment may supersede guidelines as patient circumstances warrant ASSESSMENT
BCCA Protocol Summary for Treatment of Advanced Squamous Cell Carcinoma of the Head and Neck Cancer Using Fluorouracil and Platinum
 BCCA Protocol Summary for Treatment of Advanced Squamous Cell Carcinoma of the Head and Neck Cancer Using Fluorouracil and Platinum Protocol Code: Tumour Group: Contact Physician: HNAVFUP Head and Neck
BCCA Protocol Summary for Treatment of Advanced Squamous Cell Carcinoma of the Head and Neck Cancer Using Fluorouracil and Platinum Protocol Code: Tumour Group: Contact Physician: HNAVFUP Head and Neck
Juluca (dolutegravir, rilpivirine) NEW PRODUCT SLIDESHOW
 Juluca (dolutegravir, rilpivirine) NEW PRODUCT SLIDESHOW Introduction Brand name: Juluca Generic name: Dolutegravir, rilpivirine Pharmacological class: HIV-1 integrase strand transfer inhibitor (INSTI)
Juluca (dolutegravir, rilpivirine) NEW PRODUCT SLIDESHOW Introduction Brand name: Juluca Generic name: Dolutegravir, rilpivirine Pharmacological class: HIV-1 integrase strand transfer inhibitor (INSTI)
Xifaxan. Xifaxan (rifaximin) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.34 Subject: Xifaxan Page: 1 of 6 Last Review Date: December 8, 2017 Xifaxan Description Xifaxan (rifaximin)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.34 Subject: Xifaxan Page: 1 of 6 Last Review Date: December 8, 2017 Xifaxan Description Xifaxan (rifaximin)
Treatment of Clostridium Difficile Infection in Community Teaching Hospital: A Retrospective Study
 International Journal of Infectious Diseases and Therapy 2018; 3(3): 52-61 http://www.sciencepublishinggroup.com/j/ijidt doi: 10.11648/j.ijidt.20180303.12 ISSN: 2578-9651 (Print); ISSN: 2578-966X (Online)
International Journal of Infectious Diseases and Therapy 2018; 3(3): 52-61 http://www.sciencepublishinggroup.com/j/ijidt doi: 10.11648/j.ijidt.20180303.12 ISSN: 2578-9651 (Print); ISSN: 2578-966X (Online)
Dosing and Administration Guide for ARZERRA
 Dosing and Administration Guide for ARZERRA Indications ARZERRA (ofatumumab) is indicated: In combination with chlorambucil, for the treatment of previously untreated patients with chronic lymphocytic
Dosing and Administration Guide for ARZERRA Indications ARZERRA (ofatumumab) is indicated: In combination with chlorambucil, for the treatment of previously untreated patients with chronic lymphocytic
Admit date (YYYY/MM/DD): Cardiologist On-Call: Diagnosis: Lab Tests. CBC, Electrolytes, Urea, Creatinine, Glucose, INR, PTT, Urinalysis
 of nurse 1. Admit under ward Attending Physician: Dr. Admit date (YYYY/MM/DD): Cardiologist On-Call: Diagnosis: Lab Tests 2. On admission (if not already performed in Emergency Department or in Coronary
of nurse 1. Admit under ward Attending Physician: Dr. Admit date (YYYY/MM/DD): Cardiologist On-Call: Diagnosis: Lab Tests 2. On admission (if not already performed in Emergency Department or in Coronary
ADJUVANT TIGECYCLINE FOR SEVERE CLOSTRIDIUM DIFFICILE-ASSOCIATED DIARRHEA
 ADJUVANT TIGECYCLINE FOR SEVERE CLOSTRIDIUM DIFFICILE-ASSOCIATED DIARRHEA Candace Marr, DO Catholic Health System University at Buffalo, NY Kevin Shiley, MD Catholic Health System Buffalo, NY FINANCIAL
ADJUVANT TIGECYCLINE FOR SEVERE CLOSTRIDIUM DIFFICILE-ASSOCIATED DIARRHEA Candace Marr, DO Catholic Health System University at Buffalo, NY Kevin Shiley, MD Catholic Health System Buffalo, NY FINANCIAL
IV Vancomycin dosing and monitoring Antibiotic Guidelines. Contents. Intro
 IV Vancomycin dosing and Antibiotic Guidelines Classification: Clinical Guideline Lead Author: Antibiotic Steering Committee Additional author(s): as above Authors Division: DCSS & Tertiary Medicine Unique
IV Vancomycin dosing and Antibiotic Guidelines Classification: Clinical Guideline Lead Author: Antibiotic Steering Committee Additional author(s): as above Authors Division: DCSS & Tertiary Medicine Unique
Acute Stroke with Alteplase Administration Order Set
 Review Due Date: 2017 October PATIENT CARE DERS Weight: Adverse Reactions or Intolerances Drug No Yes (list) Food No Yes (list) _ Latex No Yes Admission Admit to Neurology service: Dr. Critical Care Diagnosis:
Review Due Date: 2017 October PATIENT CARE DERS Weight: Adverse Reactions or Intolerances Drug No Yes (list) Food No Yes (list) _ Latex No Yes Admission Admit to Neurology service: Dr. Critical Care Diagnosis:
daily; available as 10- mg g PO
 Overview of the PRN: The Pain and Palliative Care PRN of ACCP is an organization of pharmacy practitioners, clinical scientists, pharmacy educators, and others. Its mission is to advance pain and palliative
Overview of the PRN: The Pain and Palliative Care PRN of ACCP is an organization of pharmacy practitioners, clinical scientists, pharmacy educators, and others. Its mission is to advance pain and palliative
