Rising to the Challenge of Satisfying Unmet Medical Needs. Back-of-the-eye. Main Back-of-the-Eye Diseases. Uveitis. Behcet s disease.
|
|
|
- Valentine Lucas
- 6 years ago
- Views:
Transcription
1 Feature Rising to the Challenge of Satisfying Unmet Medical Needs Eyeing Further Specialization Back-of-the-eye Diseases Main Back-of-the-Eye Diseases Age-related macular degeneration Uveitis Behcet s disease Detached retina Diabetic retinopathy Macular edema Infectious intraocular inflammation As a specialty company in the eye and other specific fields, we provide products and services to the prescription ophthalmic pharmaceutical market to satisfy a wide range of therapeutic needs, particularly front-of-the-eye diseases such as dry eye and glaucoma. We have established ourselves as the No.1 ophthalmic pharmaceutical company in Japan. We are now providing products and services for back-of-the-eye diseases, with the launch of EYLEA (aflibercept [genetical recombination]) in Japan to treat wet age-related macular degeneration (wet AMD).
2 Retinal Therapeutics Market Potential The number of patients needing retinal disorder treatment is growing rapidly, driven by population aging and other factors. Effective New Drugs Required for Underserved Back-of-the-Eye Field Feature: Rising to the Challenge of Satisfying Unmet Medical Needs There are unmet medical needs for many back-ofthe-eye diseases, including wet AMD, diabetic retinopathy, and macular edema. Patients around the world live in the hope that groundbreaking new treatments will be developed. Overseas, retinal disorder markets are growing due to high unmet medical needs. Santen recognizes the strong potential in these markets to drive its growth going forward. AMD involves abnormalities appearing in the macular area, a region located in the center of the retina that is vital for vision. It occurs in dry and wet forms. With wet AMD, abnormal blood vessels form below the retina in the macular area. Bleeding and leaking from these vessels cause retinal damage and functional impairment. Patients find it hard to see in the center of their vision, resulting in a sharp decline in Quality of Life (QOL). Research has identified vascular endothelial growth factor (VEGF) as the substance that causes blood vessel formation and development and the leaking of blood and exudate. VEGF plays a vital role in the formation and maintenance of normal blood vessels, but it also triggers wet AMD by stimulating the development of unnecessary blood vessels. Around 20 years ago there were no effective treatments for wet AMD, but there have been significant advances in therapeutic methods since then, including laser therapy, antibody drugs, and biopharmaceuticals. Due to the aging population and other factors, the Japanese market for retinal disorder treatments has grown at around 20% per annum and now accounts for approximately 10% of the total prescription ophthalmic pharmaceutical market. Mechanism of Wet AMD These illustrations are images. Image of retina Image of vision New blood vessels Bleeding Macula region In wet AMD, the macular area of the retina is damaged by bleeding and leaking from choroidal blood vessels, resulting in distortion and loss of central vision. Retina Choroid Front view Side view Santen Pharmaceutical Co., Ltd. Annual Report
3 Contributing to Patients with EYLEA Helping patients in the retinal disorder field with the highest unmet needs as a specialized ophthalmic pharmaceutical company. Providing a New Treatment Option for the Rapidly Increasing Number of Wet AMD Patients Santen provides products and services to meet global therapeutic needs in the field of ophthalmology. Our mission is to help improve patients QOL. We have positioned back-of-the-eye diseases, including retinal disorders, as one of the highest priority areas where there are substantial unmet medical needs. The back-of-the-eye disease AMD is the fourth most common cause of vision loss in Japan. Patient numbers are increasing sharply as the population ages, but only a small proportion of patients receive treatment. With few pharmaceutical options for the treatment of wet AMD, healthcare professionals have long hoped for new treatments to become available. To broaden the treatment options available, Santen has launched EYLEA in Japan, a new treatment for wet AMD. As a specialized ophthalmic pharmaceutical company, we consider it extremely important to provide healthcare professionals with products needed by patients. Our goal is to continue supplying unique products, information, and services that satisfy patients unmet needs, in a bid to contribute to better treatment. Sadatoshi Furukado Director Executive Corporate Officer Japan Business and Human Resources Development, Head of Sales and Marketing Division, Prescription Pharmaceuticals Intravitreal VEGF Inhibitor EYLEA
4 Enhancing Patients QOL with New Treatments The Intravitreal VEGF Inhibitor EYLEA, which was co-developed by Regeneron Pharmaceuticals, Inc. and Bayer HealthCare, inhibits the action of VEGF that is one of the causes of wet AMD. Intravitreal injections of EYLEA improves symptoms by suppressing the growth of, and stopping bleeding and leakage from, the new blood vessels. EYLEA is expected to maintain and improve sight through a proactive treatment regimen: once monthly during the first three months (initial phase) and usually once every two months thereafter (maintenance Sales of Intravitreal VEGF Inhibitors in Japan ( Billion) (FY) Source: Santen analysis based on IMS-JPM data phase). EYLEA is rapidly penetrating the market because of its superior product profile and our ophthalmological expertise in the provision of quality medical information. Through the experience with EYLEA, Santen will develop an even more thorough understanding of therapeutic needs in the field of retinal disorders. We are working with our sales partner Bayer Yakuhin, Ltd. to support better patients QOL through the provision of quality medical information. Sales of Intravitreal VEGF Inhibitors in the U.S. (US$ Million) 3,000 1, Source: Based on companies actual results data for ranibizumab and aflibercept intravitreal injection (CY) Feature: Rising to the Challenge of Satisfying Unmet Medical Needs Message I Am Confident We Can Maximize EYLEA s Product Value Through Partnership with Santen Since it was first approved in the U.S. in 2011, EYLEA has been successfully marketed in Australia, Latin America, Europe and some other countries and regions. EYLEA has been well received in these markets, serving as a new treatment option that meets the therapeutic needs of patients around the world. In Japan, EYLEA has seen a smooth uptake in the market in just a few months after its launch, and now contributes to the treatment of many patients. As a specialty company with an unshakeable No.1 share in the Japanese market for prescription ophthalmic pharmaceuticals, Santen is the ideal partner for us as we enter the field of ophthalmology. We feel assured and encouraged by the fact that Santen has established a strong relationship with some 13,000 ophthalmologists across Japan, as well as by the company s in-depth understanding of the needs of the medical community. I am confident that our partnership with Santen will further accelerate the market penetration of EYLEA in Japan. Dr. Carsten Brunn President, Bayer Yakuhin, Ltd.
5 Sales Strategy Leveraging Santen s Strengths Taking on the challenge of establishing a presence in a new treatment field based on relationships of trust with ophthalmologists across Japan. Demonstrating Our Strengths as a Specialized Ophthalmic Pharmaceutical Company EYLEA has penetrated the Japanese market much faster than expected. Immediately after launch, the drug was adopted by university hospitals and other Takeshi Ito Corporate Officer Head of Prescription, Pharmaceuticals Sales Department, Sales and Marketing Division, Prescription Pharmaceuticals facilities already using other intravitreal VEGF inhibitors. Santen s sales in Japan in fiscal 2012 reached 3,183 million in only four months from launch. These strong results serve to highlight the significant unmet medical needs for wet AMD in Japan. We expect sales in fiscal 2013 to reach 9,852 million. As well as EYLEA s unique product profile, we attribute this success to our knowledge and expertise built up over many years in the field of ophthalmology and the close collaboration with our sales partner Bayer Yakuhin, Ltd. We have also developed relationships of trust with ophthalmologists over many years and have received feedback from many doctors on the need for a new therapeutic option for wet AMD. EYLEA has penetrated the Japanese market extremely rapidly, even compared with overseas markets, and we feel that we have been able to meet the expectations of patients and healthcare professionals alike. Looking ahead, we will continue to strive to supply products and services to satisfy unmet medical needs so as to allow more patients access to therapies.
6 Creating Products for Unmet Medical Needs Promoting global clinical development for creating products based on global medical treatment needs. Sites for SAKURA 1 global clinical study of DE-109, a treatment for uveitis 150 Around Developing Competitive New Drugs to Address Needs for Treating Retinal Disorders Global R&D bases in Japan, the U.S. and Europe (France and Finland) In the Fiscal Medium-Term Management Plan, Santen proposed a strategic objective of promoting globally oriented research and development. In addition to corneal disorders and glaucoma, areas where Santen has an especially strong presence, we have defined retinal disorders as a key area and are developing products to meet global medical needs in back-of-the-eye diseases. DE-109 has been assigned orphan drug status by the regulatory authorities in the U.S. and Europe for the indication of non-infectious posterior uveitis. A project team has been formed in the U.S. that is managing the SAKURA 1 Phase 3 global study at approximately 150 sites. DE-102 (betamethasone) is undergoing Phase 2/3 studies in Japan for the indication of macular edema associated with diabetes and BRVO 2. Pre-clinical studies demonstrated sustained efficacy when injected around the affected area. Manufacturing methods are being developed for commercial-scale production, a partnership agreement has been signed with Oakwood Laboratories of the U.S., and we are making every effort to successfully 4bases develop this product as quickly as possible. Through our experience of providing medical information and detailing for EYLEA, we are gaining an even deeper understanding of treatment needs, including feedback on treatment methods and challenges with AMD and expectations for product development. We reflect on this feedback from patients and healthcare professionals on their experience with treatment methods and use this information when developing new pharmaceuticals to support better visual and physical health for patients around the world. 1. Study Assessing double-masked Uveitis treatment 2. branch retinal vein occlusion Feature: Rising to the Challenge of Satisfying Unmet Medical Needs Message I Have High Expectations for Santen s Efforts in Retinal Disorders Wet age-related macular degeneration (wet AMD) ranks as the fourth leading cause of acquired blindness in Japan. Around 10 years ago, the number of patients was estimated at approximately 330,000 people. But the number of patients has increased rapidly since then to double that number today due to aging and other factors. Treatment needs are thus rising. Up to now, there were only two intravitreal VEGF (vascular endothelial growth factor) inhibitors that were effective in treating wet AMD. However, the launch of EYLEA has broadened treatment options, raising expectations for maintaining and improving patients sight. As a specialist, I am delighted that a new drug has been developed for retinal disorders where there are high unmet medical needs. I hope that Santen will continue to draw on its knowledge as a specialized ophthalmic pharmaceutical company to develop more outstanding pharmaceuticals sought by patients. Mitsuko Yuzawa, M.D. Professor, Division of Ophthalmology, Department of Visual Sciences, Nihon University School of Medicine
News Release - 1/5 - Bayer Yakuhin, Ltd. Santen Pharmaceutical Co., Ltd.
 News Release Bayer Yakuhin, Ltd. Santen Pharmaceutical Co., Ltd. Intravitreal VEGF Inhibitor EYLEA (aflibercept) Solution for Intravitreal Injection Released on the Market for the Treatment of Wet Age-Related
News Release Bayer Yakuhin, Ltd. Santen Pharmaceutical Co., Ltd. Intravitreal VEGF Inhibitor EYLEA (aflibercept) Solution for Intravitreal Injection Released on the Market for the Treatment of Wet Age-Related
Treatment of visual impairment due to macular edema secondary to branch retinal vein occlusion (BRVO):
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Treatment of visual impairment due to macular edema secondary to branch retinal
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Treatment of visual impairment due to macular edema secondary to branch retinal
Bayer and Regeneron start additional Phase 3 Study for VEGF Trap-Eye in Wet Age-related Macular Degeneration
 Investor News Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer and Regeneron start additional Phase 3 Study for VEGF Trap-Eye in Wet Age-related Macular Degeneration International
Investor News Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer and Regeneron start additional Phase 3 Study for VEGF Trap-Eye in Wet Age-related Macular Degeneration International
VEGF Trap-Eye: New Data Confirm Successes in the Treatment of Age-related Macular Degeneration
 Investor News Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Final Phase 2 Results Presented at Retina Society Meeting VEGF Trap-Eye: New Data Confirm Successes in the Treatment
Investor News Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Final Phase 2 Results Presented at Retina Society Meeting VEGF Trap-Eye: New Data Confirm Successes in the Treatment
Ophthalmic VEGF Inhibitors. Eylea (aflibercept), Macugen (pegaptanib) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
Bayer and Regeneron start clinical Phase III trial to extend ophthalmology research & development program for VEGF Trap- Eye in Asia
 Investor News Not intended for U.S. Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com VEGF Trap-Eye in myopic choroidal neovascularization: Bayer and Regeneron start clinical
Investor News Not intended for U.S. Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com VEGF Trap-Eye in myopic choroidal neovascularization: Bayer and Regeneron start clinical
Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial
 ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
Applications of Sustained Release Delivery Systems in Ocular Disease
 Applications of Sustained Release Delivery Systems in Ocular Disease Formulation and Delivery Systems For Peptide and Protein 2012 December 5, 2012 Christopher A. Rhodes, Ph.D. Principal, Christopher A
Applications of Sustained Release Delivery Systems in Ocular Disease Formulation and Delivery Systems For Peptide and Protein 2012 December 5, 2012 Christopher A. Rhodes, Ph.D. Principal, Christopher A
Bayer s Commitment in Ophthalmology. Zig Lang, MD, PhD Vice President and Head of Medical Affairs & Pharmacovigilance, Bayer HealthCare Company Ltd.
 Bayer s Commitment in Ophthalmology Zig Lang, MD, PhD Vice President and Head of Medical Affairs & Pharmacovigilance, Bayer HealthCare Company Ltd. Bayer s commitment to addressing unmet needs in ophthalmology
Bayer s Commitment in Ophthalmology Zig Lang, MD, PhD Vice President and Head of Medical Affairs & Pharmacovigilance, Bayer HealthCare Company Ltd. Bayer s commitment to addressing unmet needs in ophthalmology
Bayer to showcase latest Ophthalmology research at EURETINA 2017
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com 17 th European Society of Retina Specialists (EURETINA) Congress Bayer to showcase
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com 17 th European Society of Retina Specialists (EURETINA) Congress Bayer to showcase
FDA approves Roche s Lucentis (ranibizumab injection) for treatment of diabetic retinopathy in people with diabetic macular edema
 Media Release Basel, 9 February 2015 FDA approves Roche s Lucentis (ranibizumab injection) for treatment of diabetic retinopathy in people with diabetic macular edema First eye medicine approved for treatment
Media Release Basel, 9 February 2015 FDA approves Roche s Lucentis (ranibizumab injection) for treatment of diabetic retinopathy in people with diabetic macular edema First eye medicine approved for treatment
FDA approves Lucentis (ranibizumab injection) for treatment of diabetic macular edema
 Investor Update Basel, 13 August 2012 FDA approves Lucentis (ranibizumab injection) for treatment of diabetic macular edema First Major Treatment Advance in More Than 25 Years for Sight-Threatening Condition
Investor Update Basel, 13 August 2012 FDA approves Lucentis (ranibizumab injection) for treatment of diabetic macular edema First Major Treatment Advance in More Than 25 Years for Sight-Threatening Condition
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Health Technology Appraisal. Aflibercept for treating diabetic macular oedema.
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Health Technology Appraisal Aflibercept for treating diabetic macular oedema Final scope Final remit/appraisal objective To appraise the clinical and cost
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Health Technology Appraisal Aflibercept for treating diabetic macular oedema Final scope Final remit/appraisal objective To appraise the clinical and cost
REFERENCE CODE GDHC482DFR PUBLICAT ION DATE OCTOBER 2014 EYLEA (MACULAR EDEMA AND MACULAR DEGENERATION) - FORECAST AND MARKET ANALYSIS TO 2023
 REFERENCE CODE GDHC482DFR PUBLICAT ION DATE OCTOBER 2014 EYLEA (MACULAR EDEMA AND MACULAR DEGENERATION) - Executive Summary Table below presents the key metrics of Eylea for macular edema (ME) and age-related
REFERENCE CODE GDHC482DFR PUBLICAT ION DATE OCTOBER 2014 EYLEA (MACULAR EDEMA AND MACULAR DEGENERATION) - Executive Summary Table below presents the key metrics of Eylea for macular edema (ME) and age-related
EyeGene Inc. Contact Information Korea Health Industry Development Institute
 EG-Mirotin: First-in-Class recombinant polypeptide novel subcutaneous drug containing RGD-motif targeted to suppress edema and vascular leakage for Nonproliferative Diabetic Retinopathy (NPDR) EyeGene
EG-Mirotin: First-in-Class recombinant polypeptide novel subcutaneous drug containing RGD-motif targeted to suppress edema and vascular leakage for Nonproliferative Diabetic Retinopathy (NPDR) EyeGene
27% Contributions to the World s Glaucoma Patients. Feature million people
 Feature Contributions to the World s Glaucoma Patients Santen will fully harness our strengths as a specialized pharmaceutical company to contribute to patients being identified early and receiving treatment
Feature Contributions to the World s Glaucoma Patients Santen will fully harness our strengths as a specialized pharmaceutical company to contribute to patients being identified early and receiving treatment
Information for Patients undergoing Intravitreal Triamcinolone Acetonide (Kenalog) Injection
 Information for Patients undergoing Intravitreal Triamcinolone Acetonide (Kenalog) Injection Kenalog/SS/ST/04.2012/v1.1 review 05.2013 Page 1 Introduction Your doctor has found that you have leakage of
Information for Patients undergoing Intravitreal Triamcinolone Acetonide (Kenalog) Injection Kenalog/SS/ST/04.2012/v1.1 review 05.2013 Page 1 Introduction Your doctor has found that you have leakage of
VEGF-C shown to have major role in Age-Related Macular Degeneration (AMD)
 ASX and Media release 9 May 2013 VEGF-C shown to have major role in Age-Related Macular Degeneration (AMD) Data presented at the Association for Research in Vision and Ophthalmology (ARVO) 2013 conference
ASX and Media release 9 May 2013 VEGF-C shown to have major role in Age-Related Macular Degeneration (AMD) Data presented at the Association for Research in Vision and Ophthalmology (ARVO) 2013 conference
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at the
Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at the
Clinically Significant Macular Edema (CSME)
 Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Diabetic Retinopathy Treatment - Global Market Outlook ( )
 Published on Market Research Reports Inc. (https://www.marketresearchreports.com) Home > Diabetic Retinopathy Treatment - Global Market Outlook (2015-2022) Diabetic Retinopathy Treatment - Global Market
Published on Market Research Reports Inc. (https://www.marketresearchreports.com) Home > Diabetic Retinopathy Treatment - Global Market Outlook (2015-2022) Diabetic Retinopathy Treatment - Global Market
Treatment of wet macular degeneration by intravitreal injection with Ranibizumab (Lucentis) or Aflibercept (Eylea): Information and consent
 Patient information Treatment of wet macular degeneration by intravitreal injection with Ranibizumab (Lucentis) or Aflibercept (Eylea): Information and consent Introduction You have an eye condition called
Patient information Treatment of wet macular degeneration by intravitreal injection with Ranibizumab (Lucentis) or Aflibercept (Eylea): Information and consent Introduction You have an eye condition called
Ophthalmologic Policy. Vascular Endothelial Growth Factor (VEGF) Inhibitors
 Ophthalmologic Policy UnitedHealthcare Commercial Drug Policy Vascular Endothelial Growth Factor (VEGF) Inhibitors Policy Number: 2016D0042H Effective Date: October 1, 2016 Table of Contents Page INSTRUCTIONS
Ophthalmologic Policy UnitedHealthcare Commercial Drug Policy Vascular Endothelial Growth Factor (VEGF) Inhibitors Policy Number: 2016D0042H Effective Date: October 1, 2016 Table of Contents Page INSTRUCTIONS
There are no published randomized, double-blind trials comparing aflibercept to other therapies in neovascular AMD.
 Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Facts About Diabetic Eye Disease
 Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
RVO RETINAL VEIN OCCLUSION
 RVO RETINAL VEIN OCCLUSION A guide to understanding RVO Take some time to learn about RVO - it may help you hold on to your vision Retinal vein occlusion is a common disorder of the retina and a leading
RVO RETINAL VEIN OCCLUSION A guide to understanding RVO Take some time to learn about RVO - it may help you hold on to your vision Retinal vein occlusion is a common disorder of the retina and a leading
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many as possible the opportunity to get involved
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many as possible the opportunity to get involved
Date approved: 04/18/18. Approved by: Pharmacy and Therapeutics Quality Management Subcommittee Effective Date: Department of Origin: Pharmacy
 Integrated Healthcare Services and Criteria Document: Reference #: PC/V001 Page: 1 of 9 PRODUCT APPLICATION: PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services,
Integrated Healthcare Services and Criteria Document: Reference #: PC/V001 Page: 1 of 9 PRODUCT APPLICATION: PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services,
Introduction How the eye works
 1 Introduction Diabetic retinopathy is a condition that can cause permanent loss of eyesight and even blindness. It is a major cause of loss of vision. But if a person with diabetes receives proper eye
1 Introduction Diabetic retinopathy is a condition that can cause permanent loss of eyesight and even blindness. It is a major cause of loss of vision. But if a person with diabetes receives proper eye
aflibercept 40mg/mL solution for injection (Eylea ) SMC No. (1074/15) Bayer
 aflibercept 40mg/mL solution for injection (Eylea ) SMC No. (1074/15) Bayer 07 August 2015 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS Boards
aflibercept 40mg/mL solution for injection (Eylea ) SMC No. (1074/15) Bayer 07 August 2015 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS Boards
Eylea (aflibercept) Document Number: IC-0026
 Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Growth Conference. October 19, May 2013
 Dawson Corporate James Securities Presentation 3rd Annual Small Cap Growth Conference October 19, 2017 May 2013 Safe Harbor This document contains forward-looking statements within the meaning of the "safe
Dawson Corporate James Securities Presentation 3rd Annual Small Cap Growth Conference October 19, 2017 May 2013 Safe Harbor This document contains forward-looking statements within the meaning of the "safe
Common Drug Review Patient Group Input Submissions
 Common Drug Review Patient Group Input Submissions Aflibercept (Eylea) for Age-related Macular Degeneration Patient group input submissions were received from the following patient groups. Those with permission
Common Drug Review Patient Group Input Submissions Aflibercept (Eylea) for Age-related Macular Degeneration Patient group input submissions were received from the following patient groups. Those with permission
Treatment of Age Related Macular Degeneration (AMD) by Intravitreal Injection
 Information for Patients Manchester Royal Eye Hospital Medical Retina Services Treatment of Age Related Macular Degeneration (AMD) by Intravitreal Injection What is age related macular degeneration (AMD)?
Information for Patients Manchester Royal Eye Hospital Medical Retina Services Treatment of Age Related Macular Degeneration (AMD) by Intravitreal Injection What is age related macular degeneration (AMD)?
Ophthamology Directorate. Eye Injection for Macular Disorders Information for Patients
 Ophthamology Directorate Eye Injection for Macular Disorders Information for Patients As discussed at your appointment today, please call the Medical Retinal Services Coordinator as soon as possible (within
Ophthamology Directorate Eye Injection for Macular Disorders Information for Patients As discussed at your appointment today, please call the Medical Retinal Services Coordinator as soon as possible (within
 measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
Photodynamic Therapy (PDT) for agerelated
 Page 1 of 5 Photodynamic Therapy (PDT) for agerelated eye conditions Introduction This leaflet gives you information about Photodynamic Therapy (PDT) for the age--related eye conditions macular degeneration
Page 1 of 5 Photodynamic Therapy (PDT) for agerelated eye conditions Introduction This leaflet gives you information about Photodynamic Therapy (PDT) for the age--related eye conditions macular degeneration
Andrew Rae, President & CEO / TSX-V: ICO
 ico Therapeutics Andrew Rae, President & CEO 604-602-9414602 www.icotherapeutics.com / TSX-V: ICO Forward Looking Statements Certain of the statements contained in this presentation are forward-looking
ico Therapeutics Andrew Rae, President & CEO 604-602-9414602 www.icotherapeutics.com / TSX-V: ICO Forward Looking Statements Certain of the statements contained in this presentation are forward-looking
GENERAL INFORMATION DIABETIC EYE DISEASE
 GENERAL INFORMATION DIABETIC EYE DISEASE WHAT IS DIABETIC EYE DISEASE? Diabetic eye disease is a term used to describe the common eye complications seen in people with diabetes. It includes: Diabetic retinopathy
GENERAL INFORMATION DIABETIC EYE DISEASE WHAT IS DIABETIC EYE DISEASE? Diabetic eye disease is a term used to describe the common eye complications seen in people with diabetes. It includes: Diabetic retinopathy
Intravitreal Injection
 for patients Eye Clinic Ipswich Hospital Tel: 01473 703230 Intravitreal Injection What is an intravitreal injection? An intravitreal injection is the injection of a drug into the vitreous body (the jelly
for patients Eye Clinic Ipswich Hospital Tel: 01473 703230 Intravitreal Injection What is an intravitreal injection? An intravitreal injection is the injection of a drug into the vitreous body (the jelly
Diabetic Macular Edema Treatment in the 21st Century
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
The Era of anti- - - VEGF Kirk L. Halvorson, OD
 The Era of anti- - - VEGF Kirk L. Halvorson, OD Introduction: Anti- - - Vascular Endothelial Growth Factor (Anti- - - VEGF) medication is a relatively a new line of medications used in treating a variety
The Era of anti- - - VEGF Kirk L. Halvorson, OD Introduction: Anti- - - Vascular Endothelial Growth Factor (Anti- - - VEGF) medication is a relatively a new line of medications used in treating a variety
Dan Roberts. International Low Vision Support Group NEWSLETTER. This Month. In This Issue. 1: This Month 2-5: News
 In This Issue 1: This Month 2-5: News Eye & Body Complete from Biosyntrx Extra Strength AREDS-2 Multisupplement Call for information 800-688-6815 The ILVSG is sponsored by MD Foundation www.eyesight.org
In This Issue 1: This Month 2-5: News Eye & Body Complete from Biosyntrx Extra Strength AREDS-2 Multisupplement Call for information 800-688-6815 The ILVSG is sponsored by MD Foundation www.eyesight.org
VisionNews. Artist asks; What does blindness look like?
 VisionNews WINTER 2012 Diabetic retinopathy affects 93 million worldwide Accomplished neuroscientist joins CERA Early treatment leads to better outcomes for common eye disease Global indicators for blindness
VisionNews WINTER 2012 Diabetic retinopathy affects 93 million worldwide Accomplished neuroscientist joins CERA Early treatment leads to better outcomes for common eye disease Global indicators for blindness
New STAIRWAY study data shows potential for extended durability with Roche s faricimab in neovascular age-related macular degeneration (namd)
 Media Release New STAIRWAY study data shows potential for extended durability with Roche s faricimab in neovascular age-related macular degeneration (namd) Faricimab the first bispecific antibody designed
Media Release New STAIRWAY study data shows potential for extended durability with Roche s faricimab in neovascular age-related macular degeneration (namd) Faricimab the first bispecific antibody designed
OPHTHALMOLOGIC POLICY: VASCULAR ENDOTHELIAL GROWTH FACTOR (VEGF) INHIBITORS
 UnitedHealthcare Commercial Medical Benefit Drug Policy OPHTHALMOLOGIC POLICY: VASCULAR ENDOTHELIAL GROWTH FACTOR (VEGF) INHIBITORS Policy Number: PHA020 Effective Date: September 1, 2018 Table of Contents
UnitedHealthcare Commercial Medical Benefit Drug Policy OPHTHALMOLOGIC POLICY: VASCULAR ENDOTHELIAL GROWTH FACTOR (VEGF) INHIBITORS Policy Number: PHA020 Effective Date: September 1, 2018 Table of Contents
A Patient s Guide to Diabetic Retinopathy
 Diabetic Retinopathy A Patient s Guide to Diabetic Retinopathy 840 Walnut Street, Philadelphia PA 19107 www.willseye.org Diabetic Retinopathy 1. Definition Diabetic retinopathy is a complication of diabetes
Diabetic Retinopathy A Patient s Guide to Diabetic Retinopathy 840 Walnut Street, Philadelphia PA 19107 www.willseye.org Diabetic Retinopathy 1. Definition Diabetic retinopathy is a complication of diabetes
Bank of America Merrill Lynch HealthCare Conference Bayer HealthCare Dr. Jörg Reinhardt. September 14, 2011
 Bank of America Merrill Lynch HealthCare Conference Bayer HealthCare Dr. Jörg Reinhardt Chairman of the Board of Management of Bayer HealthCare AG and Chairman of the Bayer HealthCare Executive Committee
Bank of America Merrill Lynch HealthCare Conference Bayer HealthCare Dr. Jörg Reinhardt Chairman of the Board of Management of Bayer HealthCare AG and Chairman of the Bayer HealthCare Executive Committee
Brampton Hurontario Street Brampton, ON L6Y 0P6
 Diabetic Retinopathy What is Diabetic Retinopathy Diabetic retinopathy is one of the leading causes of blindness world-wide. Diabetes damages blood vessels in many organs of the body including the eyes.
Diabetic Retinopathy What is Diabetic Retinopathy Diabetic retinopathy is one of the leading causes of blindness world-wide. Diabetes damages blood vessels in many organs of the body including the eyes.
Diabetic Retinopathy A Presentation for the Public
 Diabetic Retinopathy A Presentation for the Public Ray M. Balyeat, MD The Eye Institute Tulsa, Oklahoma The Healthy Eye Light rays enter the eye through the cornea, pupil and lens. These light rays are
Diabetic Retinopathy A Presentation for the Public Ray M. Balyeat, MD The Eye Institute Tulsa, Oklahoma The Healthy Eye Light rays enter the eye through the cornea, pupil and lens. These light rays are
Another Record Year for Bayer HealthCare Pharmaceuticals in Asia Pacific
 News Release Bayer (South East Asia) Pte Ltd Communications 63 Chulia Street OCBC Centre East, 14 th Floor Singapore 049514 Tel. +65 6496 1888 Co. No.: 197901344Z Another Record Year for Bayer HealthCare
News Release Bayer (South East Asia) Pte Ltd Communications 63 Chulia Street OCBC Centre East, 14 th Floor Singapore 049514 Tel. +65 6496 1888 Co. No.: 197901344Z Another Record Year for Bayer HealthCare
Dr Dianne Sharp Ophthalmologist Retina Specialists, Parnell Greenlane Clinical Centre
 Dr Dianne Sharp Ophthalmologist Retina Specialists, Parnell Greenlane Clinical Centre 11:00-11:55 WS #115: The Revolution in Macular Degeneration Management 12:05-13:00 WS #127: The Revolution in Macular
Dr Dianne Sharp Ophthalmologist Retina Specialists, Parnell Greenlane Clinical Centre 11:00-11:55 WS #115: The Revolution in Macular Degeneration Management 12:05-13:00 WS #127: The Revolution in Macular
FROM OUTDATED TO UPDATED Eminence-Based Medicine
 FROM OUTDATED TO UPDATED Eminence-Based Medicine Evidence-Based Medicine A REVIEW OF KEY CLINICAL TRIALS Anthony DeWilde, OD FAAO 1 EMINENCE BASED MEDICINE 2 EVIDENCE BASED MEDICINE 3 4 CLINICAL TRIALS
FROM OUTDATED TO UPDATED Eminence-Based Medicine Evidence-Based Medicine A REVIEW OF KEY CLINICAL TRIALS Anthony DeWilde, OD FAAO 1 EMINENCE BASED MEDICINE 2 EVIDENCE BASED MEDICINE 3 4 CLINICAL TRIALS
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Arkadin Managed Calls
 Arkadin Managed Calls ThromboGenics Conference Call FY 2017 Business Update & Results Thursday 15.03.2018 18u30 CET Speakers: Patrik De Haes, MD, CEO and Dominique Vanfleteren, CFO Call Duration: 19:05
Arkadin Managed Calls ThromboGenics Conference Call FY 2017 Business Update & Results Thursday 15.03.2018 18u30 CET Speakers: Patrik De Haes, MD, CEO and Dominique Vanfleteren, CFO Call Duration: 19:05
Treatment of Retinal Vein Occlusion (RVO)
 Manchester Royal Eye Hospital Medical Retina Services Information for Patients Treatment of Retinal Vein Occlusion (RVO) What is a Retinal Vein Occlusion (RVO)? The retina is the light sensitive layer
Manchester Royal Eye Hospital Medical Retina Services Information for Patients Treatment of Retinal Vein Occlusion (RVO) What is a Retinal Vein Occlusion (RVO)? The retina is the light sensitive layer
Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England. Retinal Vein Occlusion (RVO) with Macular oedema (MO)
 Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England (Royal Victoria Infirmary, Sunderland Eye Infirmary, James Cook University Hospital, Darlington Memorial Hospital, University Hospital
Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England (Royal Victoria Infirmary, Sunderland Eye Infirmary, James Cook University Hospital, Darlington Memorial Hospital, University Hospital
Diabetic Retinopathy
 Diabetic Retinopathy Diabetes mellitus is one of the leading causes of irreversible blindness worldwide. In the United States, it is the most common cause of blindness in people younger than 65 years.
Diabetic Retinopathy Diabetes mellitus is one of the leading causes of irreversible blindness worldwide. In the United States, it is the most common cause of blindness in people younger than 65 years.
Management of Neovascular AMD
 Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
Diagnosis and treatment of diabetic retinopathy. Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City
 Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
What is Age-Related Macular Degeneration?
 Intravitreal Injections Eylea / Lucentis Patient Information What is Age-Related Macular Degeneration? Age-related macular degeneration (AMD) is an eye condition found in older people, which may lead to
Intravitreal Injections Eylea / Lucentis Patient Information What is Age-Related Macular Degeneration? Age-related macular degeneration (AMD) is an eye condition found in older people, which may lead to
REFERENCE CODE GDHC317DFR PUBLICAT ION DATE DECEMBER 2013 LUCENTIS (MICROVASCULAR COMPLICATIONS OF DIABETES) - FORECAST AND MARKET ANALYSIS TO 2022
 REFERENCE CODE GDHC317DFR PUBLICAT ION DATE DECEMBER 2013 LUCENTIS Executive Summary The table below presents a summary of the key metrics for Lucentis (Microvascular Complications for Diabetes) for recombinant
REFERENCE CODE GDHC317DFR PUBLICAT ION DATE DECEMBER 2013 LUCENTIS Executive Summary The table below presents a summary of the key metrics for Lucentis (Microvascular Complications for Diabetes) for recombinant
psivida Transforms into Commercial Stage Specialty BioPharmaceutical Company AAO October 25, 2018 NASDAQ: EYPT
 psivida Transforms into Commercial Stage Specialty BioPharmaceutical Company OIS @ AAO October 25, 2018 NASDAQ: EYPT Forward Looking SAFE HARBOR STATEMENTS UNDER THE PRIVATE SECURITIES LITIGATION REFORM
psivida Transforms into Commercial Stage Specialty BioPharmaceutical Company OIS @ AAO October 25, 2018 NASDAQ: EYPT Forward Looking SAFE HARBOR STATEMENTS UNDER THE PRIVATE SECURITIES LITIGATION REFORM
Information for patients
 Information for patients Intravitreal Anti-VEGF Treatment (vascular endothelial growth factor) This leaflet gives you information that will help you decide whether to have intravitreal treatment. It also
Information for patients Intravitreal Anti-VEGF Treatment (vascular endothelial growth factor) This leaflet gives you information that will help you decide whether to have intravitreal treatment. It also
VISIONCARE S IMPLANTABLE MINIATURE TELESCOPE (by Dr. Isaac Lipshitz)
 PATIENT INFORMATION BOOKLET PAGE 1 OF 32 VISIONCARE S IMPLANTABLE MINIATURE TELESCOPE (by Dr. Isaac Lipshitz) AN INTRAOCULAR TELESCOPE FOR TREATING SEVERE TO PROFOUND VISION IMPAIRMENT DUE TO BILATERAL
PATIENT INFORMATION BOOKLET PAGE 1 OF 32 VISIONCARE S IMPLANTABLE MINIATURE TELESCOPE (by Dr. Isaac Lipshitz) AN INTRAOCULAR TELESCOPE FOR TREATING SEVERE TO PROFOUND VISION IMPAIRMENT DUE TO BILATERAL
The Human Eye. Cornea Iris. Pupil. Lens. Retina
 The Retina Thin layer of light-sensitive tissue at the back of the eye (the film of the camera). Light rays are focused on the retina then transmitted to the brain. The macula is the very small area in
The Retina Thin layer of light-sensitive tissue at the back of the eye (the film of the camera). Light rays are focused on the retina then transmitted to the brain. The macula is the very small area in
The Foundation WHAT IS THE RETINA? continued next page. RETINA HEALTH SERIES Facts from the ASRS
 The Foundation American Society of Retina Specialists Committed to improving the quality of life of all people with retinal disease. Intravitreal Injections An intravitreal (pronounced in tra VIT re al)
The Foundation American Society of Retina Specialists Committed to improving the quality of life of all people with retinal disease. Intravitreal Injections An intravitreal (pronounced in tra VIT re al)
Ophthalmology. Juliette Stenz, MD
 Ophthalmology Juliette Stenz, MD Required Slide Disclosures NO SIGNIFICANT FINANCIAL, GENERAL, OR OBLIGATION INTERESTS TO REPORT Required Slide At the end of this session, students will be able to: 1.
Ophthalmology Juliette Stenz, MD Required Slide Disclosures NO SIGNIFICANT FINANCIAL, GENERAL, OR OBLIGATION INTERESTS TO REPORT Required Slide At the end of this session, students will be able to: 1.
INFORMED CONSENT FOR AVASTIN TM (BEVACIZUMAB) INTRAVITREAL INJECTION
 INFORMED CONSENT FOR AVASTIN TM (BEVACIZUMAB) INTRAVITREAL INJECTION INDICATIONS: Age-related macular degeneration (AMD) is the leading cause of blindness in people over 50 years of age. It is caused by
INFORMED CONSENT FOR AVASTIN TM (BEVACIZUMAB) INTRAVITREAL INJECTION INDICATIONS: Age-related macular degeneration (AMD) is the leading cause of blindness in people over 50 years of age. It is caused by
The Trust does not record electronically the number of patients who are treated by laser for a condition.
 Ref: FOI/CAD/ID 3224 06 April 20 Please reply to: FOI Administrator Trust Management Maidstone Hospital Hermitage Lane Maidstone Kent ME 9QQ Email: mtw-tr.foiadmin@nhs.net Freedom of Information Act 2000
Ref: FOI/CAD/ID 3224 06 April 20 Please reply to: FOI Administrator Trust Management Maidstone Hospital Hermitage Lane Maidstone Kent ME 9QQ Email: mtw-tr.foiadmin@nhs.net Freedom of Information Act 2000
Anti-VEGF drugs & CATT Results. in AMD. Apollo Hospitals, Hyderabad. Mallika Goyal, MD
 Anti-VEGF drugs & CATT Results in AMD Mallika Goyal, MD Apollo Hospitals, Hyderabad Vascular Endothelial Growth Factor Napoleone Ferrara at Genentech 1989 identified & cloned the gene Protein that causes
Anti-VEGF drugs & CATT Results in AMD Mallika Goyal, MD Apollo Hospitals, Hyderabad Vascular Endothelial Growth Factor Napoleone Ferrara at Genentech 1989 identified & cloned the gene Protein that causes
REFERENCE CODE GDHC319DFR PUBLICAT ION DATE DECEMBER 2013 EYLEA (MICROVASCULAR COMPLICATIONS OF DIABETES) - FORECAST AND MARKET ANALYSIS TO 2022
 REFERENCE CODE GDHC319DFR PUBLICAT ION DATE DECEMBER 2013 EYLEA Executive Summary The table below presents a summary of the key metrics for Eylea (Microvascular Complications for Diabetes) for recombinant
REFERENCE CODE GDHC319DFR PUBLICAT ION DATE DECEMBER 2013 EYLEA Executive Summary The table below presents a summary of the key metrics for Eylea (Microvascular Complications for Diabetes) for recombinant
Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis)
 Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
Updates to the Alberta Drug Benefit List. Effective August 1, 2017
 Updates to the Alberta Drug Benefit List Effective August 1, 2017 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780) 498-8370
Updates to the Alberta Drug Benefit List Effective August 1, 2017 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780) 498-8370
RETINAL CONDITIONS RETINAL CONDITIONS
 GENERAL INFORMATION RETINAL CONDITIONS RETINAL CONDITIONS WHAT ARE RETINAL CONDITIONS? Retinal conditions affect the light-sensitive tissue at the back of eye known as the retina. They include diseases
GENERAL INFORMATION RETINAL CONDITIONS RETINAL CONDITIONS WHAT ARE RETINAL CONDITIONS? Retinal conditions affect the light-sensitive tissue at the back of eye known as the retina. They include diseases
Clinical Policy: Aflibercept (Eylea) Reference Number: CP.PHAR.184
 Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
f.a.q s Q. Why should I have an iwellness exam?
 f.a.q s The iwellness exam is a quick and non-invasive scan of your eye that lets your doctor see the layers of your retina to aid in the diagnosis of sight-threatening eye disease. Q. Why should I have
f.a.q s The iwellness exam is a quick and non-invasive scan of your eye that lets your doctor see the layers of your retina to aid in the diagnosis of sight-threatening eye disease. Q. Why should I have
Retinab for Diabetic Retinopathy
 Retinab for Diabetic Retinopathy Tarnhelm Therapeutics January 2014 2 2013 Tarnhelm Therapeutics Company Background Virtual company based in Bryn Mawr, PA Developing biologics for diabetic retinopathy
Retinab for Diabetic Retinopathy Tarnhelm Therapeutics January 2014 2 2013 Tarnhelm Therapeutics Company Background Virtual company based in Bryn Mawr, PA Developing biologics for diabetic retinopathy
Vision and eye healthcare study in residential aged care facilities
 Vision and eye healthcare study in residential aged care facilities Study report Report prepared by: Rob Cummins, Director, Research & Policy Julie Heraghty, Former Chief Executive Officer Macular Disease
Vision and eye healthcare study in residential aged care facilities Study report Report prepared by: Rob Cummins, Director, Research & Policy Julie Heraghty, Former Chief Executive Officer Macular Disease
Outline. Preventing & Treating Diabetes Related Blindness. Eye Care Center Doctors. Justin Kanoff, MD. Eye Care Center of Northern Colorado
 Outline Preventing & Treating Diabetes Related Blindness Justin Kanoff, MD Eye Care Center of Northern Colorado 303 974 4302 Introduction to Eye Care Center of Northern Colorado How the eye works Eye problems
Outline Preventing & Treating Diabetes Related Blindness Justin Kanoff, MD Eye Care Center of Northern Colorado 303 974 4302 Introduction to Eye Care Center of Northern Colorado How the eye works Eye problems
What you can expect with OZURDEX
 Important Information About Macular Edema Following Branch or Central Retinal Vein Occlusion (RVO) and Treatment For patients with RVO What you can expect with OZURDEX Approved Use OZURDEX (dexamethasone
Important Information About Macular Edema Following Branch or Central Retinal Vein Occlusion (RVO) and Treatment For patients with RVO What you can expect with OZURDEX Approved Use OZURDEX (dexamethasone
CENTENE PHARMACY AND THERAPEUTICS NEW DRUG REVIEW 2Q17 April May
 BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY
 EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY Diwakar chaudhary *1, 2, Hu shuqiong, Long Yuan and Xiong kun 1 Yangtze University, 1 Nanhuan Road
EFFICACY OF ANTI-VASCULAR ENDOTHELIAL GROWTH FACTOR AGENTS IN RETINAL DISORDER FOR BETTER VISUAL ACUITY Diwakar chaudhary *1, 2, Hu shuqiong, Long Yuan and Xiong kun 1 Yangtze University, 1 Nanhuan Road
EYLEA. (aflibercept solution for injection) Patient Guide
 EYLEA (aflibercept solution for injection) Patient Guide Eylea is used to treat Wet Age-Related Macular Degeneration (Wet AMD), macular oedema secondary to Branch and Central Retinal Vein Occlusion (BRVO
EYLEA (aflibercept solution for injection) Patient Guide Eylea is used to treat Wet Age-Related Macular Degeneration (Wet AMD), macular oedema secondary to Branch and Central Retinal Vein Occlusion (BRVO
Ophthalmology Macular Pathways
 Ophthalmology Macular Pathways Age related Macular Degeneration Diabetic Macular Oedema Macular Oedema secondary to Central Retinal Macular Oedema secondary to Branch Retinal CNV associated with pathological
Ophthalmology Macular Pathways Age related Macular Degeneration Diabetic Macular Oedema Macular Oedema secondary to Central Retinal Macular Oedema secondary to Branch Retinal CNV associated with pathological
From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014
 From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014 1. This course is designed to review the important ophthalmic literature that was released between October 2013
From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014 1. This course is designed to review the important ophthalmic literature that was released between October 2013
Eyes on Diabetics: How to Avoid Blindness in Diabetic Patient
 Eyes on Diabetics: How to Avoid Blindness in Diabetic Patient Rova Virgana FK Unpad Pusat Mata Nasional RS Mata Cicendo Bandung Eye Center (Hospital and Clinic) PIT IDI Jabar 2018 Keys Facts from WHO
Eyes on Diabetics: How to Avoid Blindness in Diabetic Patient Rova Virgana FK Unpad Pusat Mata Nasional RS Mata Cicendo Bandung Eye Center (Hospital and Clinic) PIT IDI Jabar 2018 Keys Facts from WHO
Wednesday 26 th November 2008 The ASX Company Announcements office: Chairman s AGM Address
 Advanced Molecular Diagnostic Systems The Walter and Eliza Hall Biotechnology Centre, 4 Research Avenue, La Trobe R&D Park, Bundoora, Victoria 3083 T: +61 (0)3 9345 2127 F: +61 (0)3 9345 2242 Wednesday
Advanced Molecular Diagnostic Systems The Walter and Eliza Hall Biotechnology Centre, 4 Research Avenue, La Trobe R&D Park, Bundoora, Victoria 3083 T: +61 (0)3 9345 2127 F: +61 (0)3 9345 2242 Wednesday
The Foundation WHAT IS THE RETINA?
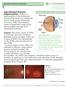 Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
New Insights on Treating Diabetic Retinopathy & Diabetic Macular Edema from the 2018 AAO Meeting
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care
 The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Medical Retina November 2016 Association of Health Professions in Ophthalmology General basic
The Common Clinical Competency Framework for Non-medical Ophthalmic Healthcare Professionals in Secondary Care Medical Retina November 2016 Association of Health Professions in Ophthalmology General basic
Major new Macular Degeneration Foundation research initiative
 Research Update 2010 Global research on the causes, genetics and treatment of Macular Degeneration continues at a hectic pace. In fact, no other area of ophthalmology is receiving as much attention at
Research Update 2010 Global research on the causes, genetics and treatment of Macular Degeneration continues at a hectic pace. In fact, no other area of ophthalmology is receiving as much attention at
A Letter from the Medical Director. Retina Consultants of Hawaii Newsletter VOLUME THREE ISSUE 1 ~ Fall 2014
 Retina Consultants of Hawaii Newsletter VOLUME THREE ISSUE 1 ~ Fall 2014 A Letter from the Medical Director to this potential new treatment for dry age related macular degeneration to our patients here
Retina Consultants of Hawaii Newsletter VOLUME THREE ISSUE 1 ~ Fall 2014 A Letter from the Medical Director to this potential new treatment for dry age related macular degeneration to our patients here
Patient AB. Born in 1961 PED
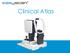 Clinical Atlas Patient AB Born in 1961 PED Autofluorescence Dilated 45 EasyScan Zero-dilation IR 45 Fundus Dilated 45 In the fundus photos (Canon CX1) the PED is not able to be seen. However, the extent
Clinical Atlas Patient AB Born in 1961 PED Autofluorescence Dilated 45 EasyScan Zero-dilation IR 45 Fundus Dilated 45 In the fundus photos (Canon CX1) the PED is not able to be seen. However, the extent
Abicipar pegol for wet age related macular degeneration
 EVIDENCE BRIEFING AUGUST 2018 Abicipar pegol for wet age related macular degeneration NIHRIO ID 7084 NICE ID 9724 Developer/Company Allergan Ltd and Molecular Partners UKPS ID 646978 Licensing and market
EVIDENCE BRIEFING AUGUST 2018 Abicipar pegol for wet age related macular degeneration NIHRIO ID 7084 NICE ID 9724 Developer/Company Allergan Ltd and Molecular Partners UKPS ID 646978 Licensing and market
Anti-epilepsy Drug VIMPAT for I.V. infusion 200mg Launched in Japan
 Press Release March 25, 2019 UCB Japan Co., Ltd. Daiichi Sankyo Company, Limited Anti-epilepsy Drug VIMPAT for I.V. infusion 200mg Launched in Japan UCB Japan Co., Ltd. (headquarters: Shinjuku-ku, Tokyo;
Press Release March 25, 2019 UCB Japan Co., Ltd. Daiichi Sankyo Company, Limited Anti-epilepsy Drug VIMPAT for I.V. infusion 200mg Launched in Japan UCB Japan Co., Ltd. (headquarters: Shinjuku-ku, Tokyo;
GENERAL INFORMATION FOOTSCRAY CLINIC
 GENERAL INFORMATION FOOTSCRAY CLINIC visioneyeinstitute.com.au Quality Management. ISO 9001 SPECIALIST EYE CARE FOR PATIENTS Vision Eye Institute Footscray is a well-established private ophthalmology clinic
GENERAL INFORMATION FOOTSCRAY CLINIC visioneyeinstitute.com.au Quality Management. ISO 9001 SPECIALIST EYE CARE FOR PATIENTS Vision Eye Institute Footscray is a well-established private ophthalmology clinic
Telemedicine Diagnostic Challenges for Diabetic Retinopathy
 Telemedicine Diagnostic Challenges for Diabetic Retinopathy Ingrid Zimmer-Galler, M.D. Johns Hopkins Office of Telemedicine Financial Disclosures None 1 Telemedicine Diabetic Retinopathy Screening Performed
Telemedicine Diagnostic Challenges for Diabetic Retinopathy Ingrid Zimmer-Galler, M.D. Johns Hopkins Office of Telemedicine Financial Disclosures None 1 Telemedicine Diabetic Retinopathy Screening Performed
OPHTHALMOLOGY DEPARTMENT Primary care referral guidelines
 OPHTHALMOLOGY DEPARTMENT Primary care referral guidelines Contents REFERRAL CATEGIES... 2 Emergency... 2 Urgent... 2 Semi urgent/routine... 2 Not accepted... 2 OPHTHALMOLOGY CONDITIONS NOT ACCEPTED...
OPHTHALMOLOGY DEPARTMENT Primary care referral guidelines Contents REFERRAL CATEGIES... 2 Emergency... 2 Urgent... 2 Semi urgent/routine... 2 Not accepted... 2 OPHTHALMOLOGY CONDITIONS NOT ACCEPTED...
