Patient details GP details Specialist details Name GP Name Dr Specialist Name Dr R. Horton
|
|
|
- Kristian Hampton
- 6 years ago
- Views:
Transcription
1 Rationale for Initiation, Continuation and Discontinuation (RICaD) Sacubitril/Valsartan (Entresto) For the treatment of symptomatic heart failure with reduced ejection fraction (NICE TA388) This document supports the use and transfer of a treatment initiated by the hospital. It is intended for completion by specialists in order to give Primary Care prescribers a clear indication of the reason for initiating the medication together with suggested criteria for its subsequent continuation or discontinuation. This RICaD should be provided as a supplement to the specialist s clinical letter. Patient details GP details Specialist details Name GP Name Dr Specialist Name Dr R. Horton Hospital Number NHS DOB Patient address GP address I confirm that this patient has been initiated on sacubtril/valsartan for the reasons shown below Signature Date Contact details Heart Failure Nurse Specialists Rationale for Choice and Guidance on initiation Relevant Diagnosis: Symptomatic chronic heart failure with reduced ejection fraction (NICE TA388) Sacubitril valsartan is recommended as an option of treating symptomatic chronic heart failure with reduced ejection fraction, only in people: Reason why sacubitril valsartan has been chosen 1. With NYHA Class II to IV symptoms 2. With left ventricular ejection fraction 35% 3. Who are already taking a stable dose of an ACEI/ARB Yes No Ensure clear instruction has been given regarding the stop date for the ACE/ARB Treatment with sacubitril valsartan will be considered in those patients in whom the above criteria is met. ACEI/ARB to be discontinued: Date ACEI/ARB to be discontinued: Date of Sacubitril valsartan initiation:.... The heart failure multidisciplinary team/specialist to provide the following information: Baseline serum creatinine Estimated GFR (egfr) Date of test Result Pretreatment test results Hepatic function (ALT) Serum potassium Blood pressure Known history of angioedema/hereditary/idiopathic angioedema Yes No Page 1 of 6
2 Guidance on initiation, dose optimisation and stabilisation Recommended starting dose : One tablet of 49 mg/51 mg twice daily, except in situations described below. The dose should be doubled at 2-4 weeks to the target dose of one tablet of 97 mg/103 mg twice daily, as tolerated by the patient Initiation dose Treatment should not be initiated in patients with serum potassium level >5.4 mmol/l or with SBP <100 mmhg. A starting dose of 24 mg/26 mg twice daily should be considered for patients with SBP 100 to 110 mmhg. Sacubitril valsartan should not be co-administered with an ACE inhibitor or an ARB. Due to the potential risk of angioedema when used concomitantly with an ACE inhibitor, it must not be started for at least 36 hours after discontinuing ACE inhibitor therapy. Renal Impairment Hepatic Impairment Blood pressure Monitoring The valsartan contained within sacubitril valsartan is more bioavailable than the valsartan in other marketed tablet formulations. Mild renal impairment (egfr 60-90ml/min/1.73m 2 ) No dose adjustment is required Moderate renal impairment (egfr 30-60ml/min/1.73m 2 An initial starting dose of 24mg/26mg twice daily should ) be considered Severe renal impairment (egfr < 30ml/min/1.73m 2 Use with caution and a starting dose of 24mg/26mg can ) be considered End stage renal disease Not recommended Mild hepatic impairment (Child-Pugh A classification) No dose adjustment is required Moderate hepatic impairment (Child-Pugh B Use with caution, the recommended starting dose is classification) or with AST/ALT values more than twice 24mg/26mg twice daily the upper limit of the normal range Severe hepatic impairment, biliary cirrhosis or Contra-indicated cholestasis (Child-Pugh C classification) Consider a starting dose of 24mg/26mg twice daily with Systolic blood pressure 100 to 110 mmhg slow dose titration After stabilisation monitoring of blood pressure, renal function, serum potassium, liver function, full blood count and adherence to medication should be done at least on an annual basis by the GP. Women of child bearing age should use contraceptive while taking Entresto. This medication should be stopped if the patient becomes pregnant. If concomitant use with lithium preparations (Priadel/Camcolit) is necessary, careful monitoring of lithium levels is advised. If prescribed in combination with sublingual, oral or transdermal nitrate preparations, monitor heart rate. Additional information The initiation of Sacubitril valsartan should be undertaken by heart failure specialists with access to a multidisciplinary heart failure team. The initiating clinician is responsible for ensuring appropriate initiation, optimisation and stabilisation of sacubitril valsartan prior to the transfer of prescribing responsibility to the GP. During this period every effort should be made to reinforce lifestyle advice and the need for adherence to therapy and address any issues relating to non-compliance and/or adverse effects. Page 2 of 6
3 Frequency of review Location Method (what tests are required) Suggested Criteria for Continuation or Discontinuation Assessment of Efficacy 2-4 weeks initially then annually Outpatients department/gp practice Assessment of renal function, hepatic function, blood pressure, serum potassium. Surveillance for signs of angioedema. If combination with lithium necessary, careful monitoring of serum lithium levels. If used in combination with sublingual, oral or transdermal nitrates, monitor heart rate Continuation Criteria Discontinuation Criteria Following stabilisation (over a period of up to 3 months), the responsibility for treatment, including prescribing and monitoring will pass to primary care. Angioedema Deterioration in renal function ( egfr <30 ml/min/1.73 m 2 ) Deterioration in hepatic function Blood pressure - SBP <100 mmhg Increase in serum potassium > 5.4 mmol/l Patients will be reviewed by the heart failure nurse specialists following initiation of sacubitril valsartan by the heart failure MDT. Follow up action Patients will initially be managed by the specialist team for a period of three months after which time they will be prescribed an optimal dose of sacubitril and valsartan and the responsibility for on-going treatment, including prescribing and monitoring will pass to primary care. GPs will be expected to undertake the monitoring outlined above on an annual basis. Ensure any automatic repeat prescriptions for ACEI or ARB are removed from the patient s list. There is a potential risk for patients receiving medicines via monitored dosage systems and dosette boxes. Ensure ACEI or ARB is removed or the contents discarded. References NICE TA Sacubitril valsartan for treating symptomatic chronic heart failure with reduced ejection fraction SmPC Sacubitril valsartan (Entresto ) NICE CG108 - Chronic heart failure in adults: management. The Royal Wolverhampton Hospital NHS Trust Chronic Heart Failure Treatment guideline Page 3 of 6
4 Appendix: Important information from the Summary of Product Characteristics (SPC) Special precautions Contraindications Hypersensitivity to the active substances or to any of the excipients listed. Concomitant use with ACE inhibitors. Sacubitril valsartan must not be administered until 36 hours after discontinuing ACE inhibitor therapy. Known history of angioedema related to previous ACE inhibitor or ARB therapy. Hereditary or idiopathic angioedema. Concomitant use with aliskiren-containing medicinal products in patients with diabetes mellitus or in patients with renal impairment (egfr <60 ml/min/1.73 m 2 ). Severe hepatic impairment, biliary cirrhosis and cholestasis. Second and third trimester of pregnancy. Treatment should not be initiated if the serum potassium level is >5.4 mmol/l. Cautions Dual blockade of the renin-angiotensin-aldosterone system (RAAS) The combination of sacubitril valsartan with an ACE inhibitor is contraindicated due to the increased risk of angioedema. Sacubitril valsartan must not be initiated until 36 hours after taking the last dose of ACE inhibitor therapy. If treatment with sacubitril valsartan is stopped, ACE inhibitor therapy must not be initiated until 36 hours after the last dose of sacubitril valsartan. The combination of sacubitril valsartan with direct renin inhibitors such as aliskiren is not recommended. The combination of sacubitril valsartan with aliskiren-containing products is contraindicated in patients with diabetes mellitus or in patients with renal impairment (egfr <60 ml/min/1.73 m 2 ). Sacubitril valsartan contains valsartan, and therefore should not be co-administered with another ARB containing product. Hypotension Treatment should not be initiated unless SBP is 100 mmhg. Patients with SBP <100 mmhg were not studied. Cases of symptomatic hypotension have been reported in patients treated with sacubitril valsartan during clinical studies, especially in patients 65 years old, patients with renal disease and patients with low SBP (<112 mmhg). When initiating therapy or during dose titration with sacubitril valsartan, blood pressure should be monitored routinely. If hypotension occurs, temporary down-titration or discontinuation of sacubitril valsartan is recommended. Dose adjustment of diuretics, concomitant antihypertensives and treatment of other causes of hypotension (e.g. hypovolaemia) should be considered. Symptomatic hypotension is more likely to occur if the patient has been volume-depleted, e.g. by diuretic therapy, dietary salt restriction, diarrhoea or vomiting. Sodium and/or volume depletion should be corrected before starting treatment with sacubitril valsartan, however, such corrective action must be carefully weighed against the risk of volume overload. Impaired renal function Evaluation of patients with heart failure should always include assessment of renal function. Patients with mild and moderate renal impairment are more at risk of developing hypotension. There is very limited clinical experience in patients with severe renal impairment (estimated GFR <30 ml/min/1.73m 2 ) and these patients may be at greatest risk of hypotension. There is no experience in patients with end-stage renal disease and use of sacubitril valsartan is not recommended. Worsening renal function Use of sacubitril valsartan may be associated with decreased renal function. The risk may be further increased by dehydration or concomitant use of non-steroidal anti-inflammatory agents (NSAIDs). Down-titration should be considered in patients who develop a clinically significant decrease in renal function. Hyperkalaemia Treatment should not be initiated if the serum potassium level is >5.4 mmol/l. Use of sacubitril valsartan may be associated with an increased risk of hyperkalaemia, although hypokalaemia may also occur. Monitoring of serum potassium is recommended, especially in patients who have risk factors such as renal impairment, diabetes mellitus or hypoaldosteronism or who are on a high potassium diet or on mineralocorticoid antagonists. If patients experience clinically significant hyperkalaemia adjustment of concomitant medicinal products, or temporary down titration or discontinuation is recommended. If serum potassium level is >5.4 mmol/l discontinuation should be considered. Angioedema Angioedema has been reported in patients treated with sacubitril valsartan. If angioedema occurs, sacubitril valsartan should be immediately discontinued and appropriate therapy and monitoring should be provided until complete and sustained resolution of signs and symptoms has occurred. It must not be re-administered. In cases of confirmed angioedema where swelling has been confined to the face and lips, the condition has generally resolved without treatment, although antihistamines have been useful in relieving symptoms. Page 4 of 6
5 Angioedema associated with laryngeal oedema may be fatal. Where there is involvement of the tongue, glottis or larynx likely to cause airway obstruction, appropriate therapy, e.g. adrenaline solution 1 mg/1 ml ( ml), and/or measures necessary to ensure a patent airway, should be promptly administered. Patients with a prior history of angioedema were not studied. As they may be at higher risk for angioedema, caution is recommended if sacubitril valsartan is used in these patients. Sacubitril valsartan is contraindicated in patients with a known history of angioedema related to previous ACE inhibitor or ARB therapy or with hereditary or idiopathic angioedema. Black patients have an increased susceptibility to develop angioedema. Patients with renal artery stenosis Sacubitril valsartan may increase blood urea and serum creatinine levels in patients with bilateral or unilateral renal artery stenosis. Caution is required in patients with renal artery stenosis and monitoring of renal function is recommended. Patients with NYHA functional classification IV Caution should be exercised when initiating sacubitril valsartan in patients with NYHA functional classification IV due to limited clinical experience in this population. B-type natriuretic peptide (BNP) BNP is not a suitable biomarker of heart failure in patients treated with sacubitril valsartan because it is a neprilysin substrate. Drug Interaction (please see SPC for more detail) Patients with hepatic impairment There is limited clinical experience in patients with moderate hepatic impairment (Child-Pugh B classification) or with AST/ALT values more than twice the upper limit of the normal range. In these patients, exposure may be increased and safety is not established. Caution is therefore recommended when using it in these patients. Sacubitril valsartan is contraindicated in patients with severe hepatic impairment, biliary cirrhosis or cholestasis (Child-Pugh C classification). ACE inhibitors The concomitant use of sacubitril valsartan with ACE inhibitors is contraindicated, as the concomitant inhibition of neprilysin (NEP) and ACE may increase the risk of angioedema. Sacubitril valsartan must not be started until 36 hours after taking the last dose of ACE inhibitor therapy. ACE inhibitor therapy must not be started until 36 hours after the last dose of sacubitril valsartan. Aliskiren The concomitant use of sacubitril valsartan with aliskiren-containing products is contraindicated in patients with diabetes mellitus or in patients with renal impairment (egfr <60 ml/min/1.73 m 2 ). The combination of sacubitril valsartan with direct renin inhibitors such as aliskiren is not recommended. Combination of sacubitril valsartan with aliskiren is potentially associated with a higher frequency of adverse events such as hypotension, hyperkalaemia and decreased renal function (including acute renal failure). Sacubitril valsartan contains valsartan, and therefore should not be co-administered with another ARB containing product. Interactions requiring precautions PDE5 inhibitors including sildenafil Caution should be exercised when sildenafil or another PDE5 inhibitor is initiated in patients treated with sacubitril valsartan (a significantly greater blood pressure reduction was observed) Potassium Concomitant use of potassium-sparing diuretics (triamterene, amiloride), mineralocorticoid antagonists (e.g. spironolactone, eplerenone), potassium supplements, salt substitutes containing potassium or other agents (such as heparin) may lead to increases in serum potassium, and to increases in serum creatinine. Monitoring of serum potassium is recommended. Non-steroidal anti-inflammatory agents (NSAIDs), including selective cyclooxygenase-2 (COX-2) inhibitors In elderly patients, volume-depleted patients (including those on diuretic therapy), or patients with compromised renal function, concomitant use of sacubitril valsartan and NSAIDs may lead to an increased risk of worsening of renal function. Therefore, monitoring of renal function is recommended when initiating or modifying treatment in patients on sacubitril valsartan who are taking NSAIDs concomitantly. The use of NSAIDs in all HF patients is discouraged. Alternatives should be used where possible. Lithium Reversible increases in serum lithium concentrations and toxicity have been reported during concomitant administration of lithium with ACE inhibitors or angiotensin II receptor antagonists. Interactions between sacubitril Page 5 of 6
6 valsartan and lithium have not been investigated. Therefore, this combination is not recommended. If the combination proves necessary, careful monitoring of serum lithium levels is recommended. If a diuretic is also used, the risk of lithium toxicity may be increased further. Furosemide Co-administration of sacubitril valsartan and furosemide had no effect on the pharmacokinetics of sacubitril valsartan but reduced C max and AUC of furosemide by 50% and 28%, respectively. While there was no relevant change in urine volume, the urinary excretion of sodium was reduced within 4 hours and 24 hours after coadministration. The average daily dose of furosemide was unchanged from baseline until the end of the PARADIGM-HF study in patients treated with sacubitril valsartan. Nitrates, e.g. nitroglycerine There was no drug-drug interaction between sacubitril valsartan and intravenously administered nitroglycerin with regard to blood pressure reduction. Co-administration of nitroglycerin and sacubitril valsartan was associated with a treatment difference of 5 bpm in heart rate compared to the administration of nitroglycerine alone. A similar effect on the heart rate may occur when sacubitril valsartan is co-administered with sublingual, oral or transdermal nitrates. In general no dose adjustment is required. OATP and MRP2 transporters Co-administration of sacubitril valsartan with inhibitors of OATP1B1, OATP1B3, OAT3 (e.g. rifampicin, ciclosporin), OAT1 (e.g. tenofovir, cidofovir) or MRP2 (e.g. ritonavir) may increase the systemic exposure of LBQ657 or valsartan. Appropriate care should be exercised when initiating or ending concomitant treatment with such medicinal products. Metformin Co-administration of sacubitril valsartan with metformin reduced both C max and AUC of metformin by 23%. The clinical relevance of these findings is unknown. Therefore, when initiating therapy with sacubitril valsartan in patients receiving metformin, the clinical status of the patient should be evaluated. Additional info: The valsartan contained within sacubitril valsartan is more bioavailable than the valsartan in other marketed tablet formulations. Sacubitril valsartan may be administered with or without food. The tablets must be swallowed with a glass of water. Please note the information included in this document is correct at the time of writing. The manufacturer s Summary of Product Characteristics (SPC) and the most current edition of the British National Formulary should be consulted for up to date and more detailed prescribing information. Page 6 of 6
Factsheet Sacubitril valsartan (Entresto ) for chronic heart failure with reduced ejection fraction
 North Central London Joint Formulary Committee Factsheet Sacubitril valsartan (Entresto ) for chronic heart failure with reduced ejection fraction Start date: July 2016 Review date: July 2019 Document
North Central London Joint Formulary Committee Factsheet Sacubitril valsartan (Entresto ) for chronic heart failure with reduced ejection fraction Start date: July 2016 Review date: July 2019 Document
Clinical guideline for the introduction of Sacubitril Valsartan in primary and secondary care in Cornwall
 Clinical guideline for the introduction of Sacubitril Valsartan in primary and secondary care in Cornwall Page 1 of 15 Summary Patient clinically assessed and reviewed by a Consultant Cardiologist, Cardiology
Clinical guideline for the introduction of Sacubitril Valsartan in primary and secondary care in Cornwall Page 1 of 15 Summary Patient clinically assessed and reviewed by a Consultant Cardiologist, Cardiology
Guidelines for the Prescribing of Sacubitril / Valsartan
 Hull & East Riding Prescribing Committee Guidelines for the Prescribing of Sacubitril / Valsartan 1. BACKGROUND Sacubitril valsartan is an angiotensin receptor neprilysin inhibitor, including both a neprilysin
Hull & East Riding Prescribing Committee Guidelines for the Prescribing of Sacubitril / Valsartan 1. BACKGROUND Sacubitril valsartan is an angiotensin receptor neprilysin inhibitor, including both a neprilysin
Frimley Health Area Prescribing Committee
 Frimley Health Area Prescribing Committee Frimley Health NHS Foundation Trust North East Hampshire and Farnham CCG East Berkshire CCG Surrey Heath CCG Buckinghamshire CCG SHARED CARE Guideline Amber Traffic
Frimley Health Area Prescribing Committee Frimley Health NHS Foundation Trust North East Hampshire and Farnham CCG East Berkshire CCG Surrey Heath CCG Buckinghamshire CCG SHARED CARE Guideline Amber Traffic
AZMARDA Tablets (Sacubitril/Valsartan)
 Published on: 20 Mar 2017 AZMARDA Tablets (Sacubitril/Valsartan) Composition AZMARDA 50 Each film-coated tablet contains: 24 mg sacubitril and 26 mg valsartan as sodium salt complex AZMARDA 100 Each film-coated
Published on: 20 Mar 2017 AZMARDA Tablets (Sacubitril/Valsartan) Composition AZMARDA 50 Each film-coated tablet contains: 24 mg sacubitril and 26 mg valsartan as sodium salt complex AZMARDA 100 Each film-coated
0BCore Safety Profile. Pharmaceutical form(s)/strength: Film-coated tablet 40, 80, 160, 320 mg SE/H/PSUR/0024/003 Date of FAR:
 0BCore Safety Profile Active substance: Valsartan Pharmaceutical form(s)/strength: Film-coated tablet 40, 80, 160, 320 mg P-RMS: SE/H/PSUR/0024/003 Date of FAR: 28.02.2013 4.2 Posology and method of administration
0BCore Safety Profile Active substance: Valsartan Pharmaceutical form(s)/strength: Film-coated tablet 40, 80, 160, 320 mg P-RMS: SE/H/PSUR/0024/003 Date of FAR: 28.02.2013 4.2 Posology and method of administration
50 mg: Violet white ovaloid biconvex film-coated tablet with beveled edges, unscored, debossed with NVR on one side and LZ on the other side.
 SACUBITRIL-VALSARTAN ENTRESTO 50 mg Film-coated Tablet 100 mg Film-coated Tablet 200 mg Film-coated Tablet Angiotensin Receptor-Neprilysin Inhibitor DESCRIPTION AND COMPOSITION Pharmaceutical form Film-coated
SACUBITRIL-VALSARTAN ENTRESTO 50 mg Film-coated Tablet 100 mg Film-coated Tablet 200 mg Film-coated Tablet Angiotensin Receptor-Neprilysin Inhibitor DESCRIPTION AND COMPOSITION Pharmaceutical form Film-coated
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION
 PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION Pr ENTRESTO TM sacubitril/valsartan film-coated tablets (as sacubitril valsartan sodium hydrate complex) 24 mg sacubitril / 26 mg valsartan 49
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION Pr ENTRESTO TM sacubitril/valsartan film-coated tablets (as sacubitril valsartan sodium hydrate complex) 24 mg sacubitril / 26 mg valsartan 49
Dorset Health Technologies Forum SHARED CARE GUIDELINE FOR PRESCRIBING EPLERENONE (INSPRA )
 INDICATION SHARED CARE GUIDELINE FOR PRESCRIBING EPLERENONE (INSPRA ) Eplerenone is an aldosterone antagonist licensed to be used in addition to standard therapy including beta-blockers, to reduce the
INDICATION SHARED CARE GUIDELINE FOR PRESCRIBING EPLERENONE (INSPRA ) Eplerenone is an aldosterone antagonist licensed to be used in addition to standard therapy including beta-blockers, to reduce the
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Neprilysin Inhibitor (Entresto ) Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Neprilysin Inhibitor (Entresto ) Prime Therapeutics will review Prior
Neprilysin Inhibitor (Entresto ) Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Neprilysin Inhibitor (Entresto ) Prime Therapeutics will review Prior
Neprilysin Inhibitor (Entresto ) Prior Authorization and Quantity Limit Program Summary
 Neprilysin Inhibitor (Entresto ) Prior Authorization and Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1 Indication Entresto Reduce the risk of cardiovascular (sacubitril/valsartan) death
Neprilysin Inhibitor (Entresto ) Prior Authorization and Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1 Indication Entresto Reduce the risk of cardiovascular (sacubitril/valsartan) death
Entresto 24/26 tablets Entresto 49/51 tablets Entresto 97/103 tablets (sacubitril/valsartan)
 1 Entresto 24/26 tablets Entresto 49/51 tablets Entresto 97/103 tablets (sacubitril/valsartan) NAME OF THE MEDICINE Active ingredient: A salt complex of the anionic forms of sacubitril and valsartan, sodium
1 Entresto 24/26 tablets Entresto 49/51 tablets Entresto 97/103 tablets (sacubitril/valsartan) NAME OF THE MEDICINE Active ingredient: A salt complex of the anionic forms of sacubitril and valsartan, sodium
PRODUCT CIRCULAR. Tablets COZAAR (losartan potassium) I. THERAPEUTIC CLASS II. INDICATIONS III. DOSAGE AND ADMINISTRATION PAK-CZR-T
 PRODUCT CIRCULAR Tablets I. THERAPEUTIC CLASS, the first of a new class of agents for the treatment of hypertension, is an angiotensin II receptor (type AT 1 ) antagonist. also provides a reduction in
PRODUCT CIRCULAR Tablets I. THERAPEUTIC CLASS, the first of a new class of agents for the treatment of hypertension, is an angiotensin II receptor (type AT 1 ) antagonist. also provides a reduction in
New Zealand Data sheet
 1. PRODUCT NAME Entresto 24/26 film-coated tablets Entresto 49/51 film-coated tablets Entresto 97/103 film-coated tablets (sacubitril/valsartan) 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredient:
1. PRODUCT NAME Entresto 24/26 film-coated tablets Entresto 49/51 film-coated tablets Entresto 97/103 film-coated tablets (sacubitril/valsartan) 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredient:
The P&T Committee Lisinopril (Qbrelis )
 Situation Background Assessment The P&T Committee Lisinopril (Qbrelis ) Qbrelis, 1 mg/ml lisinopril oral solution, has recently become an FDA- approved formulation. Current practice at UK Chandler Medical
Situation Background Assessment The P&T Committee Lisinopril (Qbrelis ) Qbrelis, 1 mg/ml lisinopril oral solution, has recently become an FDA- approved formulation. Current practice at UK Chandler Medical
Byvalson. (nebivolol, valsartan) New Product Slideshow
 Byvalson (nebivolol, valsartan) New Product Slideshow Introduction Brand name: Byvalson Generic name: Nebivolol, valsartan Pharmacological class: Beta-blocker + angiotensin II receptor blocker (ARB) Strength
Byvalson (nebivolol, valsartan) New Product Slideshow Introduction Brand name: Byvalson Generic name: Nebivolol, valsartan Pharmacological class: Beta-blocker + angiotensin II receptor blocker (ARB) Strength
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS. Medicinal product no longer authorised
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Sprimeo 150 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 150 mg aliskiren
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Sprimeo 150 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 150 mg aliskiren
Initiating New Medications in the Management of Heart Failure
 Initiating New Medications in the Management of Heart Failure Sandra Oliver-McNeil DNP, MSN, ACNP-BC, CHFN Associate Professor (Clinical) Wayne State University College of Nursing Objectives The participant
Initiating New Medications in the Management of Heart Failure Sandra Oliver-McNeil DNP, MSN, ACNP-BC, CHFN Associate Professor (Clinical) Wayne State University College of Nursing Objectives The participant
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Rasilez 150 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 150 mg aliskiren
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Rasilez 150 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 150 mg aliskiren
Heart Failure (HF) - Primary Care Flow Charts. Pre diagnosis Symptoms or signs suggestive of HF
 Heart Failure (HF) - Primary Care Flow Charts Pre diagnosis Symptoms or signs suggestive of HF 12 lead ECG Normal examination and 12 lead ECG HF highly unlikely Abnormal 12 lead ECG HF Possible Arrange
Heart Failure (HF) - Primary Care Flow Charts Pre diagnosis Symptoms or signs suggestive of HF 12 lead ECG Normal examination and 12 lead ECG HF highly unlikely Abnormal 12 lead ECG HF Possible Arrange
Heart Failure (HF) - Primary Care Flow Charts. Symptoms or signs suggestive of HF. Pre diagnosis. Refer to the Heart Failure Clinic at VHK for
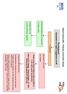 Heart Failure (HF) - Primary Care Flow Charts Pre diagnosis Symptoms or signs suggestive of HF 12 lead ECG Normal examination and 12 lead ECG HF highly unlikely Abnormal 12 lead ECG HF Possible Arrange
Heart Failure (HF) - Primary Care Flow Charts Pre diagnosis Symptoms or signs suggestive of HF 12 lead ECG Normal examination and 12 lead ECG HF highly unlikely Abnormal 12 lead ECG HF Possible Arrange
PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) CANDESARTAN/HYDROCHLOROTHIAZIDE ORION 8 MG/12.5 MG, 16 MG/12.5 MG, 32 MG/12.5 MG, 32 MG/25 MG ORION
 PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) CANDESARTAN/HYDROCHLOROTHIAZIDE ORION 8 MG/12.5 MG, 16 MG/12.5 MG, 32 MG/12.5 MG, 32 MG/25 MG ORION CORPORATION DATE: 17-04-2015, VERSION 2 VI.2 Elements for
PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) CANDESARTAN/HYDROCHLOROTHIAZIDE ORION 8 MG/12.5 MG, 16 MG/12.5 MG, 32 MG/12.5 MG, 32 MG/25 MG ORION CORPORATION DATE: 17-04-2015, VERSION 2 VI.2 Elements for
7.3 Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) Including WARNING: FETAL TOXICITY
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ENTRESTO safely and effectively. See full prescribing information for ENTRESTO. ENTRESTO (sacubitril
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ENTRESTO safely and effectively. See full prescribing information for ENTRESTO. ENTRESTO (sacubitril
Lessons and initiatives for driving success in heart failure
 Lessons and initiatives for driving success in heart failure Saturday 11 th November 2017 09:50 10:35 East Midlands Conference Centre (EMCC) Nottingham This meeting is organised and funded by Novartis
Lessons and initiatives for driving success in heart failure Saturday 11 th November 2017 09:50 10:35 East Midlands Conference Centre (EMCC) Nottingham This meeting is organised and funded by Novartis
Treating HF Patients with ARNI s Why, When and How?
 Treating HF Patients with ARNI s Why, When and How? 19 th Annual San Diego Heart Failure Symposium for Primary Care Physicians January 11-12, 2019 La Jolla, CA Barry Greenberg M.D. Distinguished Professor
Treating HF Patients with ARNI s Why, When and How? 19 th Annual San Diego Heart Failure Symposium for Primary Care Physicians January 11-12, 2019 La Jolla, CA Barry Greenberg M.D. Distinguished Professor
keyword: diuretics Drug monitoring Monitoring diuretics in primary care 2 March 2009 best tests
 www.bpac.org.nz keyword: diuretics Drug monitoring Monitoring diuretics in primary care 2 March 2009 best tests Why do we monitor patients taking diuretics and what do we monitor? Monitoring a person on
www.bpac.org.nz keyword: diuretics Drug monitoring Monitoring diuretics in primary care 2 March 2009 best tests Why do we monitor patients taking diuretics and what do we monitor? Monitoring a person on
Ivabradine for treating chronic heart failure: NICE technology appraisal guidance 267 Information for East Kent GPs revised July 2015
 Ivabradine for treating chronic heart failure: NICE technology appraisal guidance 267 Information for East Kent GPs revised July 2015 In November 2012 NICE recommended that: 1.1 Ivabradine is recommended
Ivabradine for treating chronic heart failure: NICE technology appraisal guidance 267 Information for East Kent GPs revised July 2015 In November 2012 NICE recommended that: 1.1 Ivabradine is recommended
Sacubitril valsartan (Entresto ) for patients with symptomatic chronic heart failure
 North Central London Joint Formulary Committee Sacubitril valsartan (Entresto ) for patients with symptomatic chronic heart failure North East and North Central London Position statement for the period
North Central London Joint Formulary Committee Sacubitril valsartan (Entresto ) for patients with symptomatic chronic heart failure North East and North Central London Position statement for the period
Heart Failure: Current Management Strategies
 Heart Failure: Current Management Strategies CSHP Fall Education Session- September 30th, 2017 Carolyn MacKinnon & Tamara Matchett BscPharm, ACPR Candidates Objectives 1. Describe the pathophysiology &
Heart Failure: Current Management Strategies CSHP Fall Education Session- September 30th, 2017 Carolyn MacKinnon & Tamara Matchett BscPharm, ACPR Candidates Objectives 1. Describe the pathophysiology &
Akash Ghai MD, FACC February 27, No Disclosures
 Akash Ghai MD, FACC February 27, 2015 No Disclosures Epidemiology Lifetime risk is > 20% for American s older than 40 years old. > 650,000 new cases diagnosed each year. Incidence increases with age: 2%
Akash Ghai MD, FACC February 27, 2015 No Disclosures Epidemiology Lifetime risk is > 20% for American s older than 40 years old. > 650,000 new cases diagnosed each year. Incidence increases with age: 2%
Dosing Information. The First & Only FDA-approved Spironolactone Oral Suspension P HARM A. Exclusively from
 Dosing Information The First & Only FDA-approved Spironolactone Oral Suspension Exclusively from P HARM A The Formulation That s Easy To Prescribe CAROSPIR is an antagonist of aldosterone indicated for:
Dosing Information The First & Only FDA-approved Spironolactone Oral Suspension Exclusively from P HARM A The Formulation That s Easy To Prescribe CAROSPIR is an antagonist of aldosterone indicated for:
Drug Name: Lithium Clinical Indications: Treatment and prophylaxis of mania; bipolar disorder; augmentation therapy in treatment resistant depression
 SHARED CARE PROTOCOL AND INFORMATION FOR GPS Drug Name: Lithium Clinical Indications: Treatment and prophylaxis of mania; bipolar disorder; augmentation therapy in treatment resistant depression Version:
SHARED CARE PROTOCOL AND INFORMATION FOR GPS Drug Name: Lithium Clinical Indications: Treatment and prophylaxis of mania; bipolar disorder; augmentation therapy in treatment resistant depression Version:
Amlodipine plus Lisinopril Tablets AMLOPRES-L
 Amlodipine plus Lisinopril Tablets AMLOPRES-L COMPOSITION AMLOPRES-L Each uncoated tablet contains: Amlodipine besylate equivalent to Amlodipine 5 mg and Lisinopril USP equivalent to Lisinopril (anhydrous)
Amlodipine plus Lisinopril Tablets AMLOPRES-L COMPOSITION AMLOPRES-L Each uncoated tablet contains: Amlodipine besylate equivalent to Amlodipine 5 mg and Lisinopril USP equivalent to Lisinopril (anhydrous)
Sacubitril/valsartan: A New Management Strategy for the Treatment of Heart Failure. Elizabeth Pogge, PharmD, MPH, BCPS, FASCP
 Sacubitril/valsartan: A New Management Strategy for the Treatment of Heart Failure Elizabeth Pogge, PharmD, MPH, BCPS, FASCP Disclosure Elizabeth Pogge reports no actual or potential conflicts of interest
Sacubitril/valsartan: A New Management Strategy for the Treatment of Heart Failure Elizabeth Pogge, PharmD, MPH, BCPS, FASCP Disclosure Elizabeth Pogge reports no actual or potential conflicts of interest
Long-Term Care Updates
 Long-Term Care Updates July 2015 By Amy Friedman Wilson, PharmD Heart failure (HF) is a clinical condition in which ventricular filling or ejection of blood is structurally or functionally impaired. 1
Long-Term Care Updates July 2015 By Amy Friedman Wilson, PharmD Heart failure (HF) is a clinical condition in which ventricular filling or ejection of blood is structurally or functionally impaired. 1
Entresto. Summary of the Risk Management Plan (RMP) for Entresto (sacubitril/valsartan)
 Drug Regulatory Affairs Entresto 50 mg, 100 mg and 200 mg film-coated tablets Summary of the Risk Management Plan (RMP) for Entresto (sacubitril/valsartan) Document version: 02 Document status: Document
Drug Regulatory Affairs Entresto 50 mg, 100 mg and 200 mg film-coated tablets Summary of the Risk Management Plan (RMP) for Entresto (sacubitril/valsartan) Document version: 02 Document status: Document
Cardiovascular Pharmacotherapy
 Cardiovascular Pharmacotherapy Overview Mechanism of cardiovascular drugs Indications and clinical use in cardiology Renin-Angiotensin Inhibitors: Angiotensin-Converting Enzyme Inhibitors, Angiotensin
Cardiovascular Pharmacotherapy Overview Mechanism of cardiovascular drugs Indications and clinical use in cardiology Renin-Angiotensin Inhibitors: Angiotensin-Converting Enzyme Inhibitors, Angiotensin
Shared Care Guideline Metolazone for fluid management in CKD (Adults)
 Shared Care Guideline Metolazone for fluid management in CKD (Adults) It is vital for safe and appropriate patient care that there is a clear understanding of where clinical and prescribing responsibility
Shared Care Guideline Metolazone for fluid management in CKD (Adults) It is vital for safe and appropriate patient care that there is a clear understanding of where clinical and prescribing responsibility
Beyond ACE-inhibitors for Heart Failure. Jacob Townsend, MD NCVH Birmingham 2015
 Beyond ACE-inhibitors for Heart Failure Jacob Townsend, MD NCVH Birmingham 2015 % Decrease in Mortality Current Therapy HFrEF 0% Angiotensin receptor blocker ACE inhibitor Beta blocker Mineralocorticoid
Beyond ACE-inhibitors for Heart Failure Jacob Townsend, MD NCVH Birmingham 2015 % Decrease in Mortality Current Therapy HFrEF 0% Angiotensin receptor blocker ACE inhibitor Beta blocker Mineralocorticoid
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 9 March 2011
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 9 March 2011 TAREG 3 mg/ml oral solution B/1 160 ml (CIP code: 491 474-8) Applicant: NOVARTIS PHARMA SAS valsartan
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 9 March 2011 TAREG 3 mg/ml oral solution B/1 160 ml (CIP code: 491 474-8) Applicant: NOVARTIS PHARMA SAS valsartan
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Valsartan Sandoz 40 mg film-coated tablets Valsartan Sandoz 80 mg film-coated tablets Valsartan Sandoz 160 mg film-coated tablets Valsartan
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Valsartan Sandoz 40 mg film-coated tablets Valsartan Sandoz 80 mg film-coated tablets Valsartan Sandoz 160 mg film-coated tablets Valsartan
ARNI (Angiotensin Receptor blocker / Neprilysin Inhibitors [Sacubutril/Valsartan]) Heart Failure Medication Initiation and Titration
![ARNI (Angiotensin Receptor blocker / Neprilysin Inhibitors [Sacubutril/Valsartan]) Heart Failure Medication Initiation and Titration ARNI (Angiotensin Receptor blocker / Neprilysin Inhibitors [Sacubutril/Valsartan]) Heart Failure Medication Initiation and Titration](/thumbs/77/76069192.jpg) ARNI (Angiotensin Receptor blocker / Neprilysin Inhibitors [Sacubutril/Valsartan]) Heart Failure Medication and Symptomatic HF despite ACEI/ARB and B-blocker therapy Bilateral renal artery stenosis Moderate/Severe
ARNI (Angiotensin Receptor blocker / Neprilysin Inhibitors [Sacubutril/Valsartan]) Heart Failure Medication and Symptomatic HF despite ACEI/ARB and B-blocker therapy Bilateral renal artery stenosis Moderate/Severe
Heart Failure Clinician Guide JANUARY 2016
 Kaiser Permanente National CLINICAL PRACTICE GUIDELINES Heart Failure Clinician Guide JANUARY 2016 Introduction This evidence-based guideline summary is based on the 2016 National Heart Failure Guideline.
Kaiser Permanente National CLINICAL PRACTICE GUIDELINES Heart Failure Clinician Guide JANUARY 2016 Introduction This evidence-based guideline summary is based on the 2016 National Heart Failure Guideline.
INSPRA 25 & 50 mg TABLETS
 INSPRA 25 & 50 mg TABLETS SCHEDULING STATUS: Schedule 4 PROPRIETARY NAMES (and dosage forms): INSPRA 25 (Tablets) INSPRA 50 (Tablets) COMPOSITION: INSPRA 25: INSPRA 50: Each tablet contains 25 mg eplerenone
INSPRA 25 & 50 mg TABLETS SCHEDULING STATUS: Schedule 4 PROPRIETARY NAMES (and dosage forms): INSPRA 25 (Tablets) INSPRA 50 (Tablets) COMPOSITION: INSPRA 25: INSPRA 50: Each tablet contains 25 mg eplerenone
Eplerenon Medical Valley + Eplerenon Stada
 VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Heart failure is a complex syndrome, clinically characterized by signs and symptoms secondary to abnormal cardiac function. It
VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Heart failure is a complex syndrome, clinically characterized by signs and symptoms secondary to abnormal cardiac function. It
Younger adults with a family history of premature artherosclerotic disease should have their cardiovascular risk factors measured.
 Appendix 2A - Guidance on Management of Hypertension Measurement of blood pressure All adults from 40 years should have blood pressure measured as part of opportunistic cardiovascular risk assessment.
Appendix 2A - Guidance on Management of Hypertension Measurement of blood pressure All adults from 40 years should have blood pressure measured as part of opportunistic cardiovascular risk assessment.
LXIV: DRUGS: 4. RAS BLOCKADE
 LXIV: DRUGS: 4. RAS BLOCKADE ACE Inhibitors Components of RAS Actions of Angiotensin i II Indications for ACEIs Contraindications RAS blockade in hypertension RAS blockade in CAD RAS blockade in HF Limitations
LXIV: DRUGS: 4. RAS BLOCKADE ACE Inhibitors Components of RAS Actions of Angiotensin i II Indications for ACEIs Contraindications RAS blockade in hypertension RAS blockade in CAD RAS blockade in HF Limitations
Heart Failure Clinician Guide JANUARY 2018
 Kaiser Permanente National CLINICAL PRACTICE GUIDELINES Heart Failure Clinician Guide JANUARY 2018 Introduction This evidence-based guideline summary is based on the 2018 National Heart Failure Guideline.
Kaiser Permanente National CLINICAL PRACTICE GUIDELINES Heart Failure Clinician Guide JANUARY 2018 Introduction This evidence-based guideline summary is based on the 2018 National Heart Failure Guideline.
Diagnosis and management of Chronic Heart Failure in 2018: What does NICE say? PCCS Meeting Issues and Answers Conference Nottingham
 Diagnosis and management of Chronic Heart Failure in 2018: What does NICE say? PCCS Meeting Issues and Answers Conference Nottingham NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Chronic heart failure
Diagnosis and management of Chronic Heart Failure in 2018: What does NICE say? PCCS Meeting Issues and Answers Conference Nottingham NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Chronic heart failure
BNSSG Shared Care Guidance Please complete all sections
 NHS Bristol CCG NHS North Somerset CCG NHS South Gloucestershire CCG North Bristol NHS Trust University Hospitals Bristol NHS Foundation Trust Weston Area Health NHS Trust BNSSG Shared Care Guidance Please
NHS Bristol CCG NHS North Somerset CCG NHS South Gloucestershire CCG North Bristol NHS Trust University Hospitals Bristol NHS Foundation Trust Weston Area Health NHS Trust BNSSG Shared Care Guidance Please
Package leaflet: Information for the patient
 Package leaflet: Information for the patient Entresto 24 mg/26 mg film-coated tablets Entresto 49 mg/51 mg film-coated tablets Entresto 97 mg/103 mg film-coated tablets sacubitril/valsartan This medicine
Package leaflet: Information for the patient Entresto 24 mg/26 mg film-coated tablets Entresto 49 mg/51 mg film-coated tablets Entresto 97 mg/103 mg film-coated tablets sacubitril/valsartan This medicine
REVIEW ARTICLE. Sacubitril/valsartan Use for the Hospitalist Mitchell Padkins 1, James Hart 1, Rachel Littrell 2
 Sacubitril/valsartan Use for the Hospitalist Mitchell Padkins 1, James Hart 1, Rachel Littrell 2 1 University of Missouri School of Medicine, Columbia, MO 2 Division of Cardiovascular Medicine, Department
Sacubitril/valsartan Use for the Hospitalist Mitchell Padkins 1, James Hart 1, Rachel Littrell 2 1 University of Missouri School of Medicine, Columbia, MO 2 Division of Cardiovascular Medicine, Department
ESCA: Cinacalcet (Mimpara )
 ESCA: Cinacalcet (Mimpara ) Effective Shared Care Agreement for the Treatment of Primary hyperparathyroidism when parathyroidectomy is contraindicated or not clinically appropriate. Specialist details
ESCA: Cinacalcet (Mimpara ) Effective Shared Care Agreement for the Treatment of Primary hyperparathyroidism when parathyroidectomy is contraindicated or not clinically appropriate. Specialist details
eplerenone 25, 50mg film-coated tablets (Inspra ) SMC No. (793/12) Pfizer Ltd
 eplerenone 25, 50mg film-coated tablets (Inspra ) SMC No. (793/12) Pfizer Ltd 08 June 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS Boards
eplerenone 25, 50mg film-coated tablets (Inspra ) SMC No. (793/12) Pfizer Ltd 08 June 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS Boards
Chronic heart failure: management of chronic heart failure in adults in primary and secondary care (partial update)
 Chronic heart failure: management of chronic heart failure in adults in primary and secondary care (partial update) NICE guideline Apendix C The algorithms Draft for consultation, January 2010 Chronic
Chronic heart failure: management of chronic heart failure in adults in primary and secondary care (partial update) NICE guideline Apendix C The algorithms Draft for consultation, January 2010 Chronic
azilsartan medoxomil
 azilsartan medoxomil edarbi 40mg Tablet 80mg Tablet ANTIHYPERTENSIVE Angiotensin II Receptor Antagonist FORMULATION: Each tablet contains 40mg Azilsartan medoxomil (as potassium) Each tablet contains 80mg
azilsartan medoxomil edarbi 40mg Tablet 80mg Tablet ANTIHYPERTENSIVE Angiotensin II Receptor Antagonist FORMULATION: Each tablet contains 40mg Azilsartan medoxomil (as potassium) Each tablet contains 80mg
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Rasilez 150 mg film-coated tablets Rasilez 300 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Rasilez 150
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Rasilez 150 mg film-coated tablets Rasilez 300 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Rasilez 150
Scientific conclusions and detailed explanation of the scientific grounds for the differences from the PRAC recommendation
 Annex I Scientific conclusions, grounds for variation to the terms of the marketing authorisations and detailed explanation of the scientific grounds for the differences from the PRAC recommendation 1
Annex I Scientific conclusions, grounds for variation to the terms of the marketing authorisations and detailed explanation of the scientific grounds for the differences from the PRAC recommendation 1
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Diovan 40 mg film-coated tablets Diovan 80 mg film-coated tablets Diovan 160 mg film-coated tablets Diovan 320 mg film-coated tablets
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Diovan 40 mg film-coated tablets Diovan 80 mg film-coated tablets Diovan 160 mg film-coated tablets Diovan 320 mg film-coated tablets
INSTRUCTION for medical use KLOSART
 APPROVED The Order of Ministry of Health of Ukraine 13.07.2018 1309 Registration certificate UA/8765/01/01 UA/8765/01/02 UA/8765/01/03 INSTRUCTION for medical use KLOSART Composition: active substance:
APPROVED The Order of Ministry of Health of Ukraine 13.07.2018 1309 Registration certificate UA/8765/01/01 UA/8765/01/02 UA/8765/01/03 INSTRUCTION for medical use KLOSART Composition: active substance:
HYPERTENSION IN CKD. LEENA ONGAJYOOTH, M.D., Dr.med RENAL UNIT SIRIRAJ HOSPITAL
 HYPERTENSION IN CKD LEENA ONGAJYOOTH, M.D., Dr.med RENAL UNIT SIRIRAJ HOSPITAL Stages in Progression of Chronic Kidney Disease and Therapeutic Strategies Complications Normal Increased risk Damage GFR
HYPERTENSION IN CKD LEENA ONGAJYOOTH, M.D., Dr.med RENAL UNIT SIRIRAJ HOSPITAL Stages in Progression of Chronic Kidney Disease and Therapeutic Strategies Complications Normal Increased risk Damage GFR
SHARED CARE GUIDELINE
 SHARED CARE GUIDELINE Title: Lithium Treatment in Adults aged 18-65 years Scope: Pennine Care NHS Foundation Trust NHS Bury NHS Oldham NHS Heywood, Middleton and Rochdale NHS Stockport NHS Tameside & Glossop
SHARED CARE GUIDELINE Title: Lithium Treatment in Adults aged 18-65 years Scope: Pennine Care NHS Foundation Trust NHS Bury NHS Oldham NHS Heywood, Middleton and Rochdale NHS Stockport NHS Tameside & Glossop
PHARMACEUTICAL INFORMATION AZILSARTAN
 AZEARLY Tablets Each Tablet Contains Azilsartan 20/40/80 mg PHARMACEUTICAL INFORMATION AZILSARTAN Generic name: Azilsartan Chemical name: 2-Ethoxy-1-{[2'-(5-oxo-2,5-dihydro-1,2,4-oxadiazol-3-yl)-4-biphenylyl]methyl}-
AZEARLY Tablets Each Tablet Contains Azilsartan 20/40/80 mg PHARMACEUTICAL INFORMATION AZILSARTAN Generic name: Azilsartan Chemical name: 2-Ethoxy-1-{[2'-(5-oxo-2,5-dihydro-1,2,4-oxadiazol-3-yl)-4-biphenylyl]methyl}-
COMPOSITION. A film coated tablet contains. Active ingredient: irbesartan 75 mg, 150 mg or 300 mg. Rotazar (Film coated tablets) Irbesartan
 Rotazar (Film coated tablets) Irbesartan Rotazar 75 mg, 150 mg, 300 mg COMPOSITION A film coated tablet contains Active ingredient: irbesartan 75 mg, 150 mg or 300 mg. Rotazar 75 mg, 150 mg, 300 mg PHARMACOLOGICAL
Rotazar (Film coated tablets) Irbesartan Rotazar 75 mg, 150 mg, 300 mg COMPOSITION A film coated tablet contains Active ingredient: irbesartan 75 mg, 150 mg or 300 mg. Rotazar 75 mg, 150 mg, 300 mg PHARMACOLOGICAL
Denosumab (Prolia 60 mg) Effective Shared Care Agreement For the treatment of Osteoporosis. Date: Date:
 Denosumab (Prolia 60 mg) Effective Shared Care Agreement For the treatment of Osteoporosis Section 1: Shared care arrangements and responsibilities Section 1.1 Agreement for transfer of prescribing to
Denosumab (Prolia 60 mg) Effective Shared Care Agreement For the treatment of Osteoporosis Section 1: Shared care arrangements and responsibilities Section 1.1 Agreement for transfer of prescribing to
The first and only FDA-approved lisinopril oral solution for pediatric patients 6 years of age and older. WARNING: FETAL TOXICITY
 MEDICAS DIFFER FROM 5 drug products that are essentially copies of commercially available LIPRIL ORAL AF IMPORAN SAFEY INA WARNING: FEAL OXIC 5 drug products that are essentially copies of commercially
MEDICAS DIFFER FROM 5 drug products that are essentially copies of commercially available LIPRIL ORAL AF IMPORAN SAFEY INA WARNING: FEAL OXIC 5 drug products that are essentially copies of commercially
What s New in Heart Failure? Marie-France Gauthier, BSc, PharmD, ACPR Clinical Pharmacist at Montfort Hospital
 What s New in Heart Failure? Marie-France Gauthier, BSc, PharmD, ACPR Clinical Pharmacist at Montfort Hospital Disclosures I have no current or past relationships with commercial entities Learning objectives
What s New in Heart Failure? Marie-France Gauthier, BSc, PharmD, ACPR Clinical Pharmacist at Montfort Hospital Disclosures I have no current or past relationships with commercial entities Learning objectives
Disclosures. This speaker has indicated there are no relevant financial relationships to be disclosed.
 Disclosures This speaker has indicated there are no relevant financial relationships to be disclosed. And the Beat Goes On: New Medications for Heart Failure Alison M. Walton, PharmD, BCPS The Case of
Disclosures This speaker has indicated there are no relevant financial relationships to be disclosed. And the Beat Goes On: New Medications for Heart Failure Alison M. Walton, PharmD, BCPS The Case of
Appendix IV - Prescribing Guidance for Apixaban
 Appendix IV - Prescribing Guidance for Apixaban Patient Factors Dose of Apixaban If your patient has any of the following MAJOR risk factors: Hypersensitivity to the active substance or to any of the excipients
Appendix IV - Prescribing Guidance for Apixaban Patient Factors Dose of Apixaban If your patient has any of the following MAJOR risk factors: Hypersensitivity to the active substance or to any of the excipients
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS. Medicinal product no longer authorised
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Rasilamlo 150 mg/5 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 150
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Rasilamlo 150 mg/5 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 150
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS. Medicinal product no longer authorised
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
Summary/Key Points Introduction
 Summary/Key Points Introduction Scope of Heart Failure (HF) o 6.5 million Americans 20 years of age have HF o 960,000 new cases of HF diagnosed annually o 5-year survival rate for HF is ~50% Classification
Summary/Key Points Introduction Scope of Heart Failure (HF) o 6.5 million Americans 20 years of age have HF o 960,000 new cases of HF diagnosed annually o 5-year survival rate for HF is ~50% Classification
The Failing Heart in Primary Care
 The Failing Heart in Primary Care Hamid Ikram How fares the Heart Failure Epidemic? 4357 patients, 57% women, mean age 74 years HFSA 2010 Practice Guideline (3.1) Heart Failure Prevention A careful and
The Failing Heart in Primary Care Hamid Ikram How fares the Heart Failure Epidemic? 4357 patients, 57% women, mean age 74 years HFSA 2010 Practice Guideline (3.1) Heart Failure Prevention A careful and
Factsheet LINACLOTIDE (Constella ) Irritable Bowel Syndrome constipation predominant (IBS-C)
 North Central London Joint Formulary Committee Factsheet LINACLOTIDE (Constella ) Irritable Bowel Syndrome constipation predominant (IBS-C) Start date: September 2018 Review date: September 2021 Document
North Central London Joint Formulary Committee Factsheet LINACLOTIDE (Constella ) Irritable Bowel Syndrome constipation predominant (IBS-C) Start date: September 2018 Review date: September 2021 Document
PRESCRIPTION DRUG. classification). Double the dose of ENTRESTO every 2 to 4 weeks to the target maintenance dose of 200 mg twice
 ENTRESTO 50mg film-coated tablets Local license number: 026670 ENTRESTO 100mg film-coated tablets Local license number: 026672 ENTRESTO 200mg film-coated tablets Local license number: 026671 PRESCRPTON
ENTRESTO 50mg film-coated tablets Local license number: 026670 ENTRESTO 100mg film-coated tablets Local license number: 026672 ENTRESTO 200mg film-coated tablets Local license number: 026671 PRESCRPTON
From PARADIGM-HF to Clinical Practice. Waleed AlHabeeb, MD, MHA Associate Professor of Medicine President of the Saudi Heart Failure Group
 From PARADIGM-HF to Clinical Practice Waleed AlHabeeb, MD, MHA Associate Professor of Medicine President of the Saudi Heart Failure Group PARADIGM-HF: Inclusion Criteria Chronic HF NYHA FC II IV with LVEF
From PARADIGM-HF to Clinical Practice Waleed AlHabeeb, MD, MHA Associate Professor of Medicine President of the Saudi Heart Failure Group PARADIGM-HF: Inclusion Criteria Chronic HF NYHA FC II IV with LVEF
Edoxaban For preventing stroke and systemic embolism in people with non-valvular atrial fibrillation (NICE TA 355)
 Rationale for Initiation, Continuation and Discontinuation (RICaD) Edoxaban For preventing stroke and systemic embolism in people with non-valvular atrial fibrillation (NICE TA 355) This document supports
Rationale for Initiation, Continuation and Discontinuation (RICaD) Edoxaban For preventing stroke and systemic embolism in people with non-valvular atrial fibrillation (NICE TA 355) This document supports
Pharmacology. Update. Learning Objectives. Learning Objectives. for Advanced Practice Nurses
 Pharmacology Update for Advanced Practice Nurses Trish Freeman, RPh, PhD Associate Professor, Pharmacy Practice and Science University of Kentucky College of Pharmacy University of Southern Indiana September
Pharmacology Update for Advanced Practice Nurses Trish Freeman, RPh, PhD Associate Professor, Pharmacy Practice and Science University of Kentucky College of Pharmacy University of Southern Indiana September
Summary 1. Comparative effectiveness
 Cost-effectiveness of Sacubitril/Valsartan (Entresto) for the treatment of symptomatic chronic heart failure in adult patients with reduced ejection fraction. The NCPE has issued a recommendation regarding
Cost-effectiveness of Sacubitril/Valsartan (Entresto) for the treatment of symptomatic chronic heart failure in adult patients with reduced ejection fraction. The NCPE has issued a recommendation regarding
PRODUCT MONOGRAPH. (Lisinopril Tablets, USP) 5 mg, 10 mg and 20 mg. Angiotensin-Converting Enzyme Inhibitor
 PRODUCT MONOGRAPH Pr SANDOZ LISINOPRIL (Lisinopril Tablets, USP) 5 mg, 10 mg and 20 mg Angiotensin-Converting Enzyme Inhibitor Sandoz Canada Inc. Date of Revision: August 16, 2018 110 de Lauzon Boucherville,
PRODUCT MONOGRAPH Pr SANDOZ LISINOPRIL (Lisinopril Tablets, USP) 5 mg, 10 mg and 20 mg Angiotensin-Converting Enzyme Inhibitor Sandoz Canada Inc. Date of Revision: August 16, 2018 110 de Lauzon Boucherville,
LOSARTAN + HYDROCHLOROTHIAZIDE HYZAAR /HYZAAR DS
 PRODUCT CIRCULAR WPC-HYZ-T-092007 THERAPEUTIC CLASS LOSARTAN + HYDROCHLOROTHIAZIDE HYZAAR /HYZAAR DS Film-Coated Tablet Antihypertensive LOSARTAN + HYDROCHLOROTHIAZIDE (HYZAAR) is the first combination
PRODUCT CIRCULAR WPC-HYZ-T-092007 THERAPEUTIC CLASS LOSARTAN + HYDROCHLOROTHIAZIDE HYZAAR /HYZAAR DS Film-Coated Tablet Antihypertensive LOSARTAN + HYDROCHLOROTHIAZIDE (HYZAAR) is the first combination
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Micardis 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg telmisartan. Excipients with known
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Micardis 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg telmisartan. Excipients with known
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Telmisartan Teva 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg telmisartan Excipients
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Telmisartan Teva 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg telmisartan Excipients
1/4/18. Heart Failure Guideline Review and Update. Disclosure. Pharmacist Objectives. Pharmacy Technician Objectives. What is Heart Failure?
 Disclosure Heart Failure Guideline Review and Update I have had no financial relationship over the past 12 months with any commercial sponsor with a vested interest in this presentation. Natalie Beiter,
Disclosure Heart Failure Guideline Review and Update I have had no financial relationship over the past 12 months with any commercial sponsor with a vested interest in this presentation. Natalie Beiter,
Annex I. Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s)
 Annex I Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s) 1 Scientific conclusions Taking into account the PRAC Assessment Report on the PSUR(s) for cilazapril,
Annex I Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s) 1 Scientific conclusions Taking into account the PRAC Assessment Report on the PSUR(s) for cilazapril,
Olmesartan is used for treating high blood pressure (hypertension) in adult patients.
 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Olmesartan is used for treating high blood pressure (hypertension) in adult patients. Essential hypertension 1 Worldwide, it is
VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Olmesartan is used for treating high blood pressure (hypertension) in adult patients. Essential hypertension 1 Worldwide, it is
Drugs acting on the reninangiotensin-aldosterone
 Drugs acting on the reninangiotensin-aldosterone system John McMurray Eugene Braunwald Scholar in Cardiovascular Diseases, Brigham and Women s Hospital, Boston & Visiting Professor, Harvard Medical School
Drugs acting on the reninangiotensin-aldosterone system John McMurray Eugene Braunwald Scholar in Cardiovascular Diseases, Brigham and Women s Hospital, Boston & Visiting Professor, Harvard Medical School
NICE Chronic Heart Failure Guidelines in Adults 2018
 NICE Chronic Heart Failure Guidelines in Adults 2018 The Whys, Whats and Hows of the importance of effectively managing heart failure in Primary Care and the community. Foreword Dr Clare J Taylor, General
NICE Chronic Heart Failure Guidelines in Adults 2018 The Whys, Whats and Hows of the importance of effectively managing heart failure in Primary Care and the community. Foreword Dr Clare J Taylor, General
ACCUPRIL (Quinapril)
 APPROVED PACKAGE INSERT ACCUPRIL (Quinapril) SCHEDULING STATUS: S3 PROPRIETARY NAMES (AND DOSAGE FORMS): ACCUPRIL 5 mg (tablet) ACCUPRIL 10 mg (tablet) ACCUPRIL 20 mg (tablet) ACCUPRIL 40 mg (tablet) COMPOSITION:
APPROVED PACKAGE INSERT ACCUPRIL (Quinapril) SCHEDULING STATUS: S3 PROPRIETARY NAMES (AND DOSAGE FORMS): ACCUPRIL 5 mg (tablet) ACCUPRIL 10 mg (tablet) ACCUPRIL 20 mg (tablet) ACCUPRIL 40 mg (tablet) COMPOSITION:
New NICE Heart Failure Guidelines What do they mean for primary and secondary care, and patients?
 New NICE Heart Failure Guidelines 2018 - What do they mean for primary and secondary care, and patients? Prof Ahmet Fuat PhD FRCGP FRCP PG Dip (Cardiology) GP & GPSI Cardiology Darlington Professor of
New NICE Heart Failure Guidelines 2018 - What do they mean for primary and secondary care, and patients? Prof Ahmet Fuat PhD FRCGP FRCP PG Dip (Cardiology) GP & GPSI Cardiology Darlington Professor of
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Telmisartan Actavis 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg telmisartan For the
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Telmisartan Actavis 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg telmisartan For the
ENTRESTO (sacubitril and valsartan) oral tablet
 ENTRESTO (sacubitril and valsartan) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This
ENTRESTO (sacubitril and valsartan) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Edarbi 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg of azilsartan medoxomil (as potassium).
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Edarbi 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg of azilsartan medoxomil (as potassium).
Updates in Heart Failure (HF) 2016: ACC / AHA and ESC
 Updates in Heart Failure (HF) 2016: ACC / AHA and ESC Patrick McBride, MD, MPH Professor of Medicine & Family Medicine, UW School of Medicine and Public Health Special thanks to: Clyde W. Yancy, MD, MSc
Updates in Heart Failure (HF) 2016: ACC / AHA and ESC Patrick McBride, MD, MPH Professor of Medicine & Family Medicine, UW School of Medicine and Public Health Special thanks to: Clyde W. Yancy, MD, MSc
MICARDIS Composition Pharmacological properties Pharmacokinetics
 MICARDIS Boehringer (Tablet) Composition 1 tablet contains 40 or 80 mg [1,1 -biphenyl]-2-carboxylic acid, 4 -[(1,4 dime-thyl- 2 -propyl[2,6-bi-1h-benzimidazole]-1 -yl)methyl] (= telmisartan) Excipients:
MICARDIS Boehringer (Tablet) Composition 1 tablet contains 40 or 80 mg [1,1 -biphenyl]-2-carboxylic acid, 4 -[(1,4 dime-thyl- 2 -propyl[2,6-bi-1h-benzimidazole]-1 -yl)methyl] (= telmisartan) Excipients:
Shared Care Agreement for Donepezil
 ESCA: for the treatment of Alzheimer s disease SECONDARY CARE SECTION TO BE COMPLETED BY INITIATING DOCTOR Patient s Name: Date of Birth: NHS Number: ESCA Date: One copy of information leaflet given to
ESCA: for the treatment of Alzheimer s disease SECONDARY CARE SECTION TO BE COMPLETED BY INITIATING DOCTOR Patient s Name: Date of Birth: NHS Number: ESCA Date: One copy of information leaflet given to
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Pritor 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg telmisartan. Excipients: Each tablet
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Pritor 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg telmisartan. Excipients: Each tablet
PART VI: SUMMARY OF THE RISK MANAGEMENT PLAN
 PART VI: SUMMARY OF THE RISK MANAGEMENT PLAN VI.1 Summary of activities in the risk management plan The summary below was prepared based on the information included in Part II, IV and V of the present
PART VI: SUMMARY OF THE RISK MANAGEMENT PLAN VI.1 Summary of activities in the risk management plan The summary below was prepared based on the information included in Part II, IV and V of the present
