DVT Primary Care Prescribing Pathway
|
|
|
- Ada Hardy
- 5 years ago
- Views:
Transcription
1 DVT Primary Care Prescribing Pathway Scope Classification Author Health Economy Wide Guideline East Lancashire Medicines Management Board (Reviewed December 207) Authorised by ELMMB Date February 208 Reviewed December 207 Review Date February 2020 Supporting references: Guideline: DVT Primary Care Prescribing Pathway Version Number 7 (final), February 208 Review date February 2020
2 DVT Primary Care Prescribing Pathway Introduction East Lancashire Clinical Commissioning Groups (CCG), working with local hospital specialists, has designed a clinical pathway for the diagnosis and management of DVT in primary care. The pathway supports clinicians to manage patients presenting with a suspected DVT and provides advice which is in line with the relevant NICE guidance and good clinical practice. Background to DVT Venous thromboembolism (VTE) is a condition in which a blood clot (a thrombus) forms in a vein, most commonly in the deep veins of the legs or pelvis. This is known as deep vein thrombosis, or DVT. The thrombus can dislodge and travel in the blood, particularly to the pulmonary arteries. This is known as pulmonary embolism, or PE. Failure to diagnose and treat DVTs correctly can result in fatal PE. However, diagnosis of DVT is not always straightforward. Primary care management of DVT The primary care prescribing pathway guidance document should be read in conjunction with the relevant service specifications covering DVT assessment and management in primary care. To assist clinicians diagnosing and treating DVT the pathway includes reference to the two-level Wells score, and the place of D-dimer testing and ultrasound imaging along with treatment of adults with suspected or confirmed DVT who are suitable for treatment with oral rivaroxaban (Xarelto ). The pathway does not cover children or young people aged under 8, or women who are pregnant or breastfeeding. The pathway will assume that prescribers will use a drug's summary of product characteristics to inform decisions made with individual patients. The Guideline consists of a number of parts: DVT Patient Pathway DVT patient pathway informed by NICE guidance Rivaroxaban for DVT Treatment Prescribing information DVT Investigation & Treatment Proform 0908_v7
3 DVT PATHWAY for GP s in East Lancs. Patient with suspected DVT presents to Healthcare Professional Full History (including provocation factors) clinical assessment and 2 level Wells Score Low and Medium probability WELLS 0- High probability WELLS 2 or more Take blood sample for Quantitative D Dimer and arrange follow-up appointment on the next working day Take blood sample for Quantitative D Dimer and refer for ultrasound scan. Negative Positive- Assess the patient and commence on Rivaroxaban 5mg bd if appropriate* See counselling below. Give alert card. If a proximal leg vein ultrasound scan cannot be carried out within 4 hours of being requested give an interim dose of anticoagulant Assess the patient and commence on Rivaroxaban 5mg bd if appropriate* See counselling below. Give alert card. Refer for Doppler Scan Positive USS Negative USS Unprovoked DVT Refer to NICE Clinical Guideline Negative DVT unlikely Stop Treatment Counselling Checklist for Rivaroxaban. Patient alert card go through each point, -Dose and duration of treatment -Possible side effects and what to do -Impact on dental treatment -Advice on planning pregnancy or becoming pregnant -Effect on sports and travel -When and how to seek medical help 2. Importance of compliance 3. Non-reversibility of rivaroxaban and bleeding risk 4. Missed dose advice 5. Avoid OTC aspirin/ibuprofen where possible 6. Take with food 7. Need to request GP to arrange further supply 8. Expect dose change from 5mg twice daily to Positive Continue Rivaroxaban based on dosing below Continue Rivaroxaban at appropriate dose see dosing footnote Ileofemoral DVT Refer to vascular services. NB. If bloods miss afternoon pick-up refer to Emergency Dept or Urgent Care DVT unlikely Stop Treatment. If D-Dimer positive and ultrasound scan negative Repeat scan in 6-8 days. Rivaroxaban Dosing 5mg bd review 2 days Then 20mg od for 3 months In patients with moderate (creatinine clearance ml/min) or severe (creatinine clearance 5-29 ml/min) renal impairment A reduction of the dose from 20 mg once daily to 5 mg once daily should be considered if the patient's assessed risk for bleeding outweighs the risk for recurrent DVT and PE. The recommendation for the use of 5 mg is based on PK modelling and has not been studied in this clinical setting [NB. Issue 4 tablets as in interim dose] *If Rivaroxaban is contraindicated or clinically unsuitable then refer to Emergency Dep or Urgent Care
4 Notes - All patients presenting with VTE should have a full clinical history and examination undertaken with the aim of detecting underlying conditions contributing to the development of thrombosis and assessing suitability for antithrombotic therapy - Unselective screening for cancer in patients with DVT is not recommended - Patients who have a negative or inadequate initial scan but who have a persisting clinical suspicion of DVT or whose symptoms do not settle should have a repeat ultrasound scan - After deep vein thrombosis affecting a lower limb, the use of well fitted below-knee graduated elastic compression stockings for two years should be encouraged to reduce the risk of post-phlebitic syndrome INDICATIONS for Rivaroxaban covered by this document: Treatment of DVT and prevention of recurrent DVT and pulmonary embolism (PE) following an acute DVT in adults. Rivaroxaban (Xarelto ) patient alert card and patient information The prescriber must provide a patient alert card to each patient prescribed rivaroxaban (Xarelto ) 5mg or 20mg (contained within the patient booklet A Patient s Guide to Deep Vein Thrombosis Treatment available from the manufacturer or CCG). The patient alert card will inform treating physicians and dentists about the patient s anticoagulation treatment and will contain emergency contact information. Please instruct patients to carry the patient alert card with them at all times and present it to every health care provider. Counselling checklist Patient alert card go through each point, keep at hand dose & duration of treatment possible side effects and what to do impact on dental treatment advice on planning pregnancy or become pregnant affect on sports and travel when and how to seek medical help Importance of compliance Non-reversibility of rivaroxaban & bleeding risk Missed dose advice DVT DOSING RECOMMENDATIONS WEEK TO 3 - Initially treat with 5mg twice daily with food for the first three weeks. WEEK 4 ONWARDS - The initial treatment is followed by 20mg once daily with food for another 3 months (3 x 28 tablets of 20mg). (See separate dose adjustment advice for renal impairment on page 5) Duration of treatment 3 months treatment - Confirmed proximal DVT continue the maintenance dose of rivaroxaban for 3 months. Short-term treatment (3 months) is recommended for those with transient risk factors such as recent surgery and trauma. After 3 months treatment at maintenance dose, assess the risks and benefits of continuing treatment. Unprovoked proximal DVT - Consider extending beyond 3 months if their risk of VTE recurrence is high and there is no additional risk of major bleeding. Discuss with the patient the benefits and risks of extending their treatment.
5 Longer duration of treatment - Consider longer treatment for permanent risk factors or idiopathic (unprovoked) deep vein thrombosis if their risk of VTE recurrence is high and there is no additional risk of major bleeding. Discuss with the patient the benefits and risks of extending their treatment. Seek advice from haematology if unsure. General risk factors for VTE recurrence Thrombophilia History/family history of VTE Pregnancy/recent childbirth Bedridden > 3days Age > 60 years Recent surgery in last 4 wks IV drug user Smoker Obesity Travel >4hrs in last 5 wks HRT or oral contraception Cancer Acute medical illness Patients with renal impairment Normal or mild renal impairment egfr >50 ml/min/.73m 2 Moderate to severe renal impairment egfr 5 49 ml/min/.73m 2 Day - 2 Day 22 and onwards Day - 2 Day 22 and onwards 5 mg twice daily for 3 weeks then 20 mg once daily for 3 months (3 x 28 days) 5 mg twice daily for 3 weeks then 20 mg once daily for 3 months (3 x 28 days). A reduction of dose to 5mg once daily should be considered if assessed risk of bleeding outweighs risk of recurrent DVT and PE. Renal impairment egfr <5 ml/min/.73m 2 Not recommended Patients with renal impairment are at a higher risk of haemorrhage. Rivaroxaban (Xarelto ) should also be used with caution in patients with renal impairment taking concomitant potent inhibitors of CYP3A4 (e.g. systemic azole- antimycotics (such as ketoconazole, itraconazole, voriconazole and posaconazole) or HIV protease inhibitors (e.g. ritonavir), or dronaderone. INITIATION OF RIVAROXABAN WHILST AWAITING A SCAN/QUANTITATIVE D-DIMER RESULTS DVT is likely - For those patients in whom the diagnosis of a DVT is likely based on the two-level Wells score, and for whom a proximal leg vein ultrasound scan is not available within 4 hours, week (4 x 5mg tablets) treatment with rivaroxaban should be initiated (as well as performing a D- dimer test). For those patients in whom the diagnosis of a DVT is likely based on the two-level Wells score, and for whom a proximal leg vein ultrasound scan is available within 4 hours, a prescription for week (4 x 5mg tablets) treatment with Rivaroxaban should be issued with instructions only to have dispensed and start treatment if the scan is positive. GP communication with the patient must be within the same day. Check renal function. Remember to stop treatment if the diagnosis of DVT is disproved. DVT is unlikely - For those patients in whom the diagnosis of a DVT is unlikely based on the two-level Wells score, and for whom a proximal leg vein ultrasound scan is not available within 4 hours, but a D-dimer test is positive, week (4 x5mg tablets) treatment with rivaroxaban should be initiated. For those patients in whom the diagnosis of a DVT is unlikely based on the two-level Wells score, and for whom a proximal leg vein ultrasound scan is available within 4 hours, but a D-dimer test is
6 positive, a prescription for week (4 x5mg tablets) treatment with Rivaroxaban should be issued with instructions only to have dispensed and start treatment if the scan is positive. GP communication with the patient must be within the same day. Check renal function. Remember to stop treatment if the diagnosis of DVT is disproved. Missed dose Twice daily treatment period (5mg twice a day for the first three weeks): If a dose is missed, the patient should take rivaroxaban (Xarelto) immediately to ensure intake of 30mg rivaroxaban (Xarelto ) per day. Continue with the regular 5mg twice daily intake on the following day. Once daily treatment period (beyond three weeks): If a dose is missed, the patient should take rivaroxaban (Xarelto ) immediately and continue on the following day with the once daily intake as recommended. The dose should not be doubled within the same day to make up for a missed dose. REVIEW OF PATIENTS Once a diagnosis of DVT is confirmed patients should be reviewed near the end of their 3 month treatment duration. For those patients who continue onto longer durations of treatment, they should be reviewed at least every 3-6 months, depending on the individual clinical circumstances.
7 A. Patient information Name Address GP DOB NHS No. Tel B. Pre-test probability of DVT Two level Wells Score D. Suitable for primary care management with rivaroxaban? Clinical feature Points Active cancer in last 6/2 Paralysis, paresis or recent leg plaster Recently bedridden for 3/7 or more, or major surgery within 4/52 Tenderness along the distribution of the deep venous system Entire leg (calf and thigh) swollen Calf swelling 3 cm larger than asymptomatic side Pitting oedema confined to/worse in the symptomatic leg Collateral superficial veins (non- varicose) Previously documented DVT An alternative diagnosis is at least as -2 likely as DVT Total score =.. Clinical feature YES proceed to consider treatment NO - due to one or more of the following reasons (or another reason) REFER TO SECONDARY CARE IMMEDIATELY Pregnancy or breastfeeding/post-partum Age <8 years Currently on warfarin or low molecular weight heparin (LMWH e.g. tinzaparin) Symptoms of PE Potential bleeding lesions e.g. GI ulcers, GU, pulmonary, intracranial/spinal or GI bleed <4/52 ago, varices, CNS vascular abnormalities, aneurysm, trauma/surgery, vascular retinopathy, bronchiectasis (not exclusive list) Hepatic disease associated with coagulopathy Congenital or acquired bleeding disorders Tick DVT likely 2 points or more Systolic BP >80 or diastolic BP >5 DVT unlikely point or less Active cancer within 6 months (refer for LMWH consideration e.g. tinzaparin) Anticipated compliance problems even with support (e.g. mental illness or alcohol/drug misuse) C. Blood taken sample for quantitative D Dimer Yes/No Severe renal impairment (CKD stage 5) egfr <5 ml/min/.73 m 2 Azole antifungals (e.g. fluconazole, itraconazole etc.) or HIV protease inhibitor or dronaderone
8 E. Treating venous thromboembolism (VTE) Rivaroxaban dosing: Renal function Day Dosing schedule Normal or mild renal impairment egfr >50 ml/min/.73m 2 Day - 2 Day 22 and onwards 5 mg twice daily for 3 weeks then 20 mg once daily for 3 months (3 x 28 days) Moderate to severe renal impairment egfr 5 49 ml/min/.73m 2 Day - 2 Day 22 and onwards 5 mg twice daily for 3 weeks then 20 mg once daily for 3 months (3 x 28 days). A reduction of dose to 5mg once daily should be considered if assessed risk of bleeding outweighs risk of recurrent DVT and PE. Renal impairment egfr <5 ml/min/.73m 2 Not recommended Advice: Missed dose during day 2, two 5 mg tablets may be taken at once (max 30mg/day) F. Duration of treatment 3 months treatment - Confirmed proximal DVT continue the maintenance dose of rivaroxaban for 3 months. Short-term treatment (3 months) is recommended for those with transient risk factors such as recent surgery and trauma. After 3 months treatment at maintenance dose, assess the risks and benefits of continuing treatment. Longer duration of treatment - Consider longer treatment for permanent risk factors or idiopathic (unprovoked) deep vein thrombosis if their risk of VTE recurrence is high and there is no additional risk of major bleeding. Discuss with the patient the benefits and risks of extending their treatment. Seek advice from haematology if unsure. Experience with rivaroxaban in this indication for more than 2 months is limited. Duration.. General risk factors for VTE recurrence for reference Thrombophilia Age > 60 years Obesity History/family history of VTE Recent surgery in last 4 wks Travel >4hrs in last 5 wks Smoker Oral contraception IV drug user Bedridden > 3 days Pregnancy / recent childbirth HRT Acute medical illness Cancer G. Mechanical prophylaxis Proximal deep vein thrombosis Offer below-knee graduated compression stockings with an ankle pressure greater than 23 mmhg (Class 3 stockings, or class 2 stockings if these are poorly tolerated) to patients with proximal DVT a week after diagnosis or when swelling is reduced sufficiently and if there are no contraindications and:
9 Advise patients to continue wearing the stockings for at least 2 years Ensure that the stockings are replaced two or three times per year or according to the manufacturer's instructions Advise patients that the stockings need to be worn only on the affected leg or legs. H. Results & Observations Scan Results Date Result Date of first scan No DVT Confirmed DVT Date of repeat scan (if applicable) No DVT Confirmed DVT
10 DVT Pathway Primary Care - July 207 POPULATIONS POTENTIALLY AT HIGHER RISK OF BLEEDING Any unexplained fall in haemoglobin or blood pressure should lead to a search for a bleeding site. Several sub- groups of patients are at increased risk of bleeding and should be carefully monitored for signs and symptoms of bleeding complications: Patients with hepatic impairment: Rivaroxaban (Xarelto ) is contraindicated in patients with hepatic disease associated with coagulopathy and clinically relevant bleeding risk, including cirrhotic patients with Child Pugh B and C. Patients concomitantly receiving other medicinal products: - Use of rivaroxaban (Xarelto ) is not recommended with systemic azole-antimycotics (such as ketoconazole, itraconazole, voriconazole and posaconazole) or HIV protease inhibitors (e.g. ritonavir), or dronaderone. Take care with drugs affecting haemostasis such as NSAIDs, acetylsalicylic acid, platelet aggregation inhibitors or other antithrombotic agents. Consider GI prophylaxis with a daily proton pump inhibitor (PPI), especially in those on drugs affecting haemostasis, older persons and/or those with additional risk factors. Patients with other haemorrhagic risk factors such as: - uncontrolled severe arterial hypertension (locally defined as systolic BP >80 or diastolic BP >5) - active ulcerative gastrointestinal disease - recent gastrointestinal ulcerations - vascular retinopathy - recent intracranial/intracerebral or genitourinary haemorrhage - intraspinal or intracerebral vascular abnormalities - recent brain, spinal or ophthalmological surgery - bronchiectasis or history of pulmonary bleeding - congenital or acquired bleeding disorders. PREGNANCY/BREASTFEEDING Rivaroxaban (Xarelto ) is contraindicated during pregnancy and breastfeeding. Women of child-bearing potential should avoid becoming pregnant during treatment with Rivaroxaban (Xarelto ). PERIOPERATIVE MANAGEMENT If an invasive procedure or surgical intervention is required, rivaroxaban (Xarelto ) should be stopped at least 24 hours before the intervention, if possible and based on the clinical judgement of the physician. If the procedure cannot be delayed the increased risk of bleeding due to rivaroxaban (Xarelto ) should be assessed against the urgency of the intervention. Rivaroxaban (Xarelto ) should be restarted as soon as possible after the invasive procedure or surgical intervention provided the clinical situation allows and adequate haemostasis has been established. OVERDOSE Due to limited absorption a ceiling effect with no further increase in average plasma exposure is expected at supratherapeutic doses of 50mg rivaroxaban (Xarelto ) and above. The use of activated charcoal to reduce absorption in case of overdose may be considered. HOW TO MANAGE BLEEDING COMPLICATIONS There is currently very limited clinical experience with the use of products in individuals to reverse the effects of rivaroxaban (Xarelto ). Should bleeding complications arise in a patient receiving rivaroxaban (Xarelto ), the next rivaroxaban (Xarelto ) administration should be delayed or treatment discontinued as appropriate. Individualised bleeding management may include: Symptomatic treatment, such as mechanical compression, surgical intervention, fluid replacement Haemodynamic support; blood product or component transfusion For life-threatening bleeding that cannot be controlled with the above measures, administration of a specific procoagulant reversal agent should be considered, such as prothrombin complex concentrate (PCC), activated prothrombin complex concentrate (APCC) or recombinant factor VIIa (r-fviia). Due to the high plasma protein binding rivaroxaban (Xarelto ) is not expected to be dialysable. REPORTING SUSPECTED ADVERSE REACTIONS Rivaroxaban (Xarelto ) is a black triangle drug and all suspected reactions (including those not considered to be serious) should be reported through the Yellow Card Scheme. 9
Xarelto (rivaroxaban) Prescriber Guide
 Xarelto (rivaroxaban) Prescriber Guide October 2018 PP-XAR-IE-0031 Contents Patient Alert Card 4 Dosing Recommendations 4 Stroke prevention in adult patients with non-valvular atrial fibrillation 4 Patients
Xarelto (rivaroxaban) Prescriber Guide October 2018 PP-XAR-IE-0031 Contents Patient Alert Card 4 Dosing Recommendations 4 Stroke prevention in adult patients with non-valvular atrial fibrillation 4 Patients
DVT - initial management NSCCG
 Background information Information resources for patients and carers Updates to this care map Synonyms Below knee DVT and bleeding risks Patient with confirmed DVT Scan confirms superficial thrombophlebitis
Background information Information resources for patients and carers Updates to this care map Synonyms Below knee DVT and bleeding risks Patient with confirmed DVT Scan confirms superficial thrombophlebitis
Xarelto rivaroxaban Prescriber Guide
 Xarelto rivaroxaban Prescriber Guide Patient Alert Card A patient alert card must be provided to each patient who is prescribed Xarelto 2.5 mg, 10 mg, 15 mg or 20 mg and is provided with the product package.
Xarelto rivaroxaban Prescriber Guide Patient Alert Card A patient alert card must be provided to each patient who is prescribed Xarelto 2.5 mg, 10 mg, 15 mg or 20 mg and is provided with the product package.
Appendix IV - Prescribing Guidance for Apixaban
 Appendix IV - Prescribing Guidance for Apixaban Patient Factors Dose of Apixaban If your patient has any of the following MAJOR risk factors: Hypersensitivity to the active substance or to any of the excipients
Appendix IV - Prescribing Guidance for Apixaban Patient Factors Dose of Apixaban If your patient has any of the following MAJOR risk factors: Hypersensitivity to the active substance or to any of the excipients
Suspected Deep Vein Thrombosis (DVT) Pathway for Non Pregnant patients Updated November 2016, with new D-dimer reference range
 Suspected Deep Vein Thrombosis (DVT) Pathway for Non Pregnant patients Updated November 2016, with new D-dimer reference range Suspect a DVT? Complete a Two-level DVT Wells score on ICE system (see page
Suspected Deep Vein Thrombosis (DVT) Pathway for Non Pregnant patients Updated November 2016, with new D-dimer reference range Suspect a DVT? Complete a Two-level DVT Wells score on ICE system (see page
Suspected Deep Vein Thrombosis (DVT) Assessment
 CHI no... First name... DOB... /... /... Last name... Sex: c M c F Address...... Telephone... or attach addressograph label here Hospital/Location: c Hairmyres c Monklands c Wishaw Other (specify)... Ward/Base...
CHI no... First name... DOB... /... /... Last name... Sex: c M c F Address...... Telephone... or attach addressograph label here Hospital/Location: c Hairmyres c Monklands c Wishaw Other (specify)... Ward/Base...
Appendix 2H - SECONDARY CARE CONVERSION GUIDELINES ORAL ANTICOAGULANTS
 Appendix 2H - SECONDARY CARE CONVERSION GUIDELINES ORAL ANTICOAGULANTS Please note that newer oral anticoagulants e.g. rivaroxaban, dabigatran and apixiban should be only be considered in patients with
Appendix 2H - SECONDARY CARE CONVERSION GUIDELINES ORAL ANTICOAGULANTS Please note that newer oral anticoagulants e.g. rivaroxaban, dabigatran and apixiban should be only be considered in patients with
Rivaroxaban film coated tablets are available in 2 strengths for this indication: 15mg and 20mg.
 Primary Care Prescriber Information RIVAROXABAN (XARELTO ) Treatment of acute venous thromboembolism and prevention of recurrent venous thromboembolism INDICATION Rivaroxaban is a non-vitamin K antagonist
Primary Care Prescriber Information RIVAROXABAN (XARELTO ) Treatment of acute venous thromboembolism and prevention of recurrent venous thromboembolism INDICATION Rivaroxaban is a non-vitamin K antagonist
Edoxaban Treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism (NICE TA354)
 Rationale for Initiation, Continuation and Discontinuation (RICaD) Edoxaban Treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism (NICE TA354) This document supports the
Rationale for Initiation, Continuation and Discontinuation (RICaD) Edoxaban Treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism (NICE TA354) This document supports the
Southern Trust Anticoagulant Team
 CLINICAL GUIDELINES ID TAG Title: Author: Speciality / Division: Directorate: Anticoagulation- Primary Care Guidance for reviewing patients on DOACs Southern Trust Anticoagulant Team Haematology Acute
CLINICAL GUIDELINES ID TAG Title: Author: Speciality / Division: Directorate: Anticoagulation- Primary Care Guidance for reviewing patients on DOACs Southern Trust Anticoagulant Team Haematology Acute
Deep Vein Thrombosis
 Deep Vein Thrombosis from NHS (UK) guidelines Introduction Deep vein thrombosis (DVT) is a blood clot in one of the deep veins in the body. Blood clots that develop in a vein are also known as venous thrombosis.
Deep Vein Thrombosis from NHS (UK) guidelines Introduction Deep vein thrombosis (DVT) is a blood clot in one of the deep veins in the body. Blood clots that develop in a vein are also known as venous thrombosis.
Dr. Riaz JanMohamed Consultant Haematologist The Hillingdon Hospital Foundation Trust
 MANAGEMENT OF PATIENTS WITH DEEP VEIN THROMBOSIS (DVT) IN THE COMMUNITY SETTING & ANTICOAGULATION CLINICS THE PAST, PRESENT AND THE FUTURE Dr. Riaz JanMohamed Consultant Haematologist The Hillingdon Hospital
MANAGEMENT OF PATIENTS WITH DEEP VEIN THROMBOSIS (DVT) IN THE COMMUNITY SETTING & ANTICOAGULATION CLINICS THE PAST, PRESENT AND THE FUTURE Dr. Riaz JanMohamed Consultant Haematologist The Hillingdon Hospital
Edoxaban for the treatment and prevention of venous thromboembolism (DVT or PE) or stroke prevention in non-valvular AF
 Edoxaban for the treatment and prevention of venous thromboembolism (DVT or PE) or stroke prevention in non-valvular AF Traffic light classification- Amber 2 specialist initiation / recommendation Information
Edoxaban for the treatment and prevention of venous thromboembolism (DVT or PE) or stroke prevention in non-valvular AF Traffic light classification- Amber 2 specialist initiation / recommendation Information
Venous thromboembolism - reducing the risk
 Venous thromboembolism - reducing the risk Reducing the risk of venous thromboembolism (deep vein thrombosis and pulmonary embolism) in patients admitted to hospital NICE guideline Draft for consultation,
Venous thromboembolism - reducing the risk Reducing the risk of venous thromboembolism (deep vein thrombosis and pulmonary embolism) in patients admitted to hospital NICE guideline Draft for consultation,
Venous thrombosis is common and often occurs spontaneously, but it also frequently accompanies medical and surgical conditions, both in the community
 Venous Thrombosis Venous Thrombosis It occurs mainly in the deep veins of the leg (deep vein thrombosis, DVT), from which parts of the clot frequently embolize to the lungs (pulmonary embolism, PE). Fewer
Venous Thrombosis Venous Thrombosis It occurs mainly in the deep veins of the leg (deep vein thrombosis, DVT), from which parts of the clot frequently embolize to the lungs (pulmonary embolism, PE). Fewer
Preventing Blood Clots in Adult Patients
 Manchester Royal Eye Hospital Surgical Services Information for Patients Preventing Blood Clots in Adult Patients This leaflet will give you information on how to reduce the risk of developing blood clots
Manchester Royal Eye Hospital Surgical Services Information for Patients Preventing Blood Clots in Adult Patients This leaflet will give you information on how to reduce the risk of developing blood clots
Venous thromboembolism: reducing the risk
 Issue date: January 2010 Venous thromboembolism: reducing the risk Reducing the risk of venous thromboembolism (deep vein thrombosis and pulmonary embolism) in patients admitted to hospital This guideline
Issue date: January 2010 Venous thromboembolism: reducing the risk Reducing the risk of venous thromboembolism (deep vein thrombosis and pulmonary embolism) in patients admitted to hospital This guideline
Primary Care Prescriber Information EDOXABAN (LIXIANA ) Treatment of acute venous thromboembolism and prevention of recurrent venous thromboembolism
 Primary Care Prescriber Information EDOXABAN (LIXIANA ) Treatment of acute venous thromboembolism and prevention of recurrent venous thromboembolism INDICATION Edoxaban is a non-vitamin K antagonist oral
Primary Care Prescriber Information EDOXABAN (LIXIANA ) Treatment of acute venous thromboembolism and prevention of recurrent venous thromboembolism INDICATION Edoxaban is a non-vitamin K antagonist oral
INTRODUCTION Indication and Licensing
 City and Hackney Clinical Commissioning Group Homerton University Hospital Foundation Trust DRUG NAME: Apixaban (Eliquis ) Transfer of Care document Indication: Treatment of acute venous thromboembolism
City and Hackney Clinical Commissioning Group Homerton University Hospital Foundation Trust DRUG NAME: Apixaban (Eliquis ) Transfer of Care document Indication: Treatment of acute venous thromboembolism
Shared Care Protocol for the Prescription and Supply of Low Molecular Weight Heparins
 Tameside Hospital NHS Foundation Trust and NHS Tameside and Glossop Shared Care Protocol for the Prescription and Supply of Low Molecular Weight Heparins Version 5.2 Version: 5.2 Authorised by: Joint Medicines
Tameside Hospital NHS Foundation Trust and NHS Tameside and Glossop Shared Care Protocol for the Prescription and Supply of Low Molecular Weight Heparins Version 5.2 Version: 5.2 Authorised by: Joint Medicines
These are guidelines only and can be deviated from if it is thought to be in the patient s best interest.
 Clinical Guideline Venothromboembolism prophylaxis: Trauma and Orthopaedics Venous thromboembolism (VTE) is a recognised complication associated with inactivity and surgical procedures. Therefore, all
Clinical Guideline Venothromboembolism prophylaxis: Trauma and Orthopaedics Venous thromboembolism (VTE) is a recognised complication associated with inactivity and surgical procedures. Therefore, all
DOAC for VTE. Direct Oral Anticoagulants Clint Shedd DNP, FNP-BC Emory University
 DOAC for VTE Direct Oral Anticoagulants Clint Shedd DNP, FNP-BC Emory University No disclosures Direct Oral Anticoagulants Understand VTE and the ACCP s position on it List the DOACs available in the US
DOAC for VTE Direct Oral Anticoagulants Clint Shedd DNP, FNP-BC Emory University No disclosures Direct Oral Anticoagulants Understand VTE and the ACCP s position on it List the DOACs available in the US
Comparison of novel oral anticoagulants (NOACs)
 Comparison of novel oral anticoagulants (NOACs) For guidance for full information refer to individual SPCs available at www.medicines.org.uk Licensed indications for NOACs Prevention of stroke and systemic
Comparison of novel oral anticoagulants (NOACs) For guidance for full information refer to individual SPCs available at www.medicines.org.uk Licensed indications for NOACs Prevention of stroke and systemic
pat hways Key therapeutic topic Published: 26 February 2016 nice.org.uk/guidance/ktt16
 pat hways Anticoagulants, including non-vitamin K antagonist oral anticoagulants (NOACs) Key therapeutic topic Published: 26 February 2016 nice.org.uk/guidance/ktt16 Options for local implementation NICE
pat hways Anticoagulants, including non-vitamin K antagonist oral anticoagulants (NOACs) Key therapeutic topic Published: 26 February 2016 nice.org.uk/guidance/ktt16 Options for local implementation NICE
1.0 PATIENT CARE Including Physical Healthcare
 SECTION: 1.0 PATIENT CARE Including Physical Healthcare POLICY /PROCEDURE: 1.33 NATURE AND SCOPE: SUBJECT: PROCEDURE - TRUST WIDE VENOUS THROMBOEMBOLISM (VTE) This procedure details Venous Thromboembolism
SECTION: 1.0 PATIENT CARE Including Physical Healthcare POLICY /PROCEDURE: 1.33 NATURE AND SCOPE: SUBJECT: PROCEDURE - TRUST WIDE VENOUS THROMBOEMBOLISM (VTE) This procedure details Venous Thromboembolism
Reducing the risk of venous thrombo-embolism (VTE) in hospital and after discharge
 Reducing the risk of venous thrombo-embolism (VTE) in hospital and after discharge What is a venous thromboembolism (VTE)? This is a medical term that describes a blood clot that develops in a deep vein
Reducing the risk of venous thrombo-embolism (VTE) in hospital and after discharge What is a venous thromboembolism (VTE)? This is a medical term that describes a blood clot that develops in a deep vein
CHAPTER 2 VENOUS THROMBOEMBOLISM
 CHAPTER 2 VENOUS THROMBOEMBOLISM Objectives Venous Thromboembolism (VTE) Prevalence Patho-physiology Risk Factors Diagnosis Pulmonary Embolism (PE) Management of DVT/PE Prevention VTE Patho-physiology
CHAPTER 2 VENOUS THROMBOEMBOLISM Objectives Venous Thromboembolism (VTE) Prevalence Patho-physiology Risk Factors Diagnosis Pulmonary Embolism (PE) Management of DVT/PE Prevention VTE Patho-physiology
Pathology Service User Guide Haematology
 Pathology Service User Guide Haematology St Richard s This section of the Pathology Service User Guide includes: Anticoagulant Therapy Information about the Anticoagulant Clinic Low Molecular Weight Heparin
Pathology Service User Guide Haematology St Richard s This section of the Pathology Service User Guide includes: Anticoagulant Therapy Information about the Anticoagulant Clinic Low Molecular Weight Heparin
Date of preparation: September 2014 L.GB d
 Date of preparation: September 2014 L.GB.12.2013.4666d This medicine is subject to additional monitoring. This will allow quick identification of new safety information. If you get any side effects, talk
Date of preparation: September 2014 L.GB.12.2013.4666d This medicine is subject to additional monitoring. This will allow quick identification of new safety information. If you get any side effects, talk
UHL Guideline for Treatment of Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE) in adults, with Direct Oral Anti-Coagulants
 UHL Guideline for Treatment of Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE) in adults, with Direct Oral Anti-Coagulants Trust Ref B11/2018 1. Introduction and Who Guideline applies to The introduction
UHL Guideline for Treatment of Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE) in adults, with Direct Oral Anti-Coagulants Trust Ref B11/2018 1. Introduction and Who Guideline applies to The introduction
North East Essex Medicines Management Committee
 Colchester Hospital University NHS Foundation Trust North East Essex Clinical Commissioning Group North East Essex Medicines Management Committee ORAL ANTICOAGULANT (Vit K antagonist only) MANAGEMENT GUIDELINES
Colchester Hospital University NHS Foundation Trust North East Essex Clinical Commissioning Group North East Essex Medicines Management Committee ORAL ANTICOAGULANT (Vit K antagonist only) MANAGEMENT GUIDELINES
Prescriber Guide Date of preparation: January 2018
 Prescriber Guide Date of preparation: January 2018 Table of Contents Patient Information Booklet 2 Patient Alert Card 2 Assessing stroke risk CHA 2DS 2-VASc 3 Assessing bleeding risk HAS-BLED 3 Therapeutic
Prescriber Guide Date of preparation: January 2018 Table of Contents Patient Information Booklet 2 Patient Alert Card 2 Assessing stroke risk CHA 2DS 2-VASc 3 Assessing bleeding risk HAS-BLED 3 Therapeutic
DEEP VEIN THROMBOSIS (DVT): TREATMENT
 DEEP VEIN THROMBOSIS (DVT): TREATMENT OBJECTIVE: To provide an evidence-based approach to treatment of patients presenting with deep vein thrombosis (DVT). BACKGROUND: An estimated 45,000 patients in Canada
DEEP VEIN THROMBOSIS (DVT): TREATMENT OBJECTIVE: To provide an evidence-based approach to treatment of patients presenting with deep vein thrombosis (DVT). BACKGROUND: An estimated 45,000 patients in Canada
Edoxaban For preventing stroke and systemic embolism in people with non-valvular atrial fibrillation (NICE TA 355)
 Rationale for Initiation, Continuation and Discontinuation (RICaD) Edoxaban For preventing stroke and systemic embolism in people with non-valvular atrial fibrillation (NICE TA 355) This document supports
Rationale for Initiation, Continuation and Discontinuation (RICaD) Edoxaban For preventing stroke and systemic embolism in people with non-valvular atrial fibrillation (NICE TA 355) This document supports
Venothromboembolism prophylaxis: Trauma and Orthopaedics Clinical guideline, V2
 Clinical Guideline Venothromboembolism prophylaxis: Trauma and Orthopaedics 11/11/11 TEMPORARY GUIDANCE There is no prophylactic tinzaparin available in the Trust currently. Please substitute enoxaparin
Clinical Guideline Venothromboembolism prophylaxis: Trauma and Orthopaedics 11/11/11 TEMPORARY GUIDANCE There is no prophylactic tinzaparin available in the Trust currently. Please substitute enoxaparin
XARELTO PRESCRIBER GUIDE
 XARELTO PRESCRIBER GUIDE This booklet provides important information to assist prescribers in the use of Xarelto (rivaroxaban) with their patients. This guide is intended for Health Care Professionals
XARELTO PRESCRIBER GUIDE This booklet provides important information to assist prescribers in the use of Xarelto (rivaroxaban) with their patients. This guide is intended for Health Care Professionals
Deep Vein Thrombosis and Pulmonary Embolism: Risks, Prevention & Treatment
 Deep Vein Thrombosis and Pulmonary Embolism: Risks, Prevention & Treatment Other formats If you need this information in another format such as audio tape or computer disk, Braille, large print, high contrast,
Deep Vein Thrombosis and Pulmonary Embolism: Risks, Prevention & Treatment Other formats If you need this information in another format such as audio tape or computer disk, Braille, large print, high contrast,
DRUG NAME: EDOXABAN (LIXIANA ) Transfer of Care document Treatment of acute venous thromboembolism and prevention of recurrent venous thromboembolism
 City and Hackney Clinical Commissioning Group Homerton University Hospital Foundation Trust DRUG NAME: EDOXABAN (LIXIANA ) Treatment of acute venous thromboembolism and prevention of recurrent venous thromboembolism
City and Hackney Clinical Commissioning Group Homerton University Hospital Foundation Trust DRUG NAME: EDOXABAN (LIXIANA ) Treatment of acute venous thromboembolism and prevention of recurrent venous thromboembolism
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Xarelto 10 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 10 mg rivaroxaban.
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Xarelto 10 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 10 mg rivaroxaban.
DIRECT ORAL ANTICOAGULANTS: WHEN TO USE, WHICH TO CHOOSE AND MANAGEMENT OF BLEEDING
 DIRECT ORAL ANTICOAGULANTS: WHEN TO USE, WHICH TO CHOOSE AND MANAGEMENT OF BLEEDING KATHERINE STIRLING CONSULTANT PHARMACIST ANTICOAGULATION AND THROMBOSIS DR LISHEL HORN CONSULTANT HAEMATOLOGIST HAEMOSTASIS
DIRECT ORAL ANTICOAGULANTS: WHEN TO USE, WHICH TO CHOOSE AND MANAGEMENT OF BLEEDING KATHERINE STIRLING CONSULTANT PHARMACIST ANTICOAGULATION AND THROMBOSIS DR LISHEL HORN CONSULTANT HAEMATOLOGIST HAEMOSTASIS
Deep vein thrombosis (DVT) and pulmonary embolism (PE) advice for ophthalmic surgery patients
 Deep vein thrombosis (DVT) and pulmonary embolism (PE) advice for ophthalmic surgery patients What is a deep vein thrombosis (DVT)? A DVT is a blood clot that forms within a vein deep in the leg but can
Deep vein thrombosis (DVT) and pulmonary embolism (PE) advice for ophthalmic surgery patients What is a deep vein thrombosis (DVT)? A DVT is a blood clot that forms within a vein deep in the leg but can
Implementation of NICE TA 249 and NICE TA 256
 Implementation of NICE TA 249 and NICE TA 256 Dabigatran and rivaroxaban for the prevention of stroke and systemic embolism in atrial fibrillation Version 1.0 Background NICE has recently issued guidance
Implementation of NICE TA 249 and NICE TA 256 Dabigatran and rivaroxaban for the prevention of stroke and systemic embolism in atrial fibrillation Version 1.0 Background NICE has recently issued guidance
MMP016 POLICY FOR PRIMARY THROMBOPROPHYLAXIS FOR PATIENTS ADMITTED TO NHFT
 MMP016 POLICY FOR PRIMARY THROMBOPROPHYLAXIS FOR PATIENTS ADMITTED TO NHFT MMP016 Policy for primary thromboprophylaxis for patients admitted to NHFT (rev Sept 2020) Page 1 of 21 Table of Contents Why
MMP016 POLICY FOR PRIMARY THROMBOPROPHYLAXIS FOR PATIENTS ADMITTED TO NHFT MMP016 Policy for primary thromboprophylaxis for patients admitted to NHFT (rev Sept 2020) Page 1 of 21 Table of Contents Why
VENOUS THROMBOEMBOLISM: DURATION OF TREATMENT
 VENOUS THROMBOEMBOLISM: DURATION OF TREATMENT OBJECTIVE: To provide guidance on the recommended duration of anticoagulant therapy for venous thromboembolism (VTE). BACKGROUND: Recurrent episodes of VTE
VENOUS THROMBOEMBOLISM: DURATION OF TREATMENT OBJECTIVE: To provide guidance on the recommended duration of anticoagulant therapy for venous thromboembolism (VTE). BACKGROUND: Recurrent episodes of VTE
To prevent blood clots after hip or knee replacement surgery This booklet contains information for those who have been prescribed ELIQUIS (apixaban)
 To prevent blood clots after hip or knee replacement surgery This booklet contains information for those who have been prescribed ELIQUIS (apixaban) after hip or knee replacement surgery Always read the
To prevent blood clots after hip or knee replacement surgery This booklet contains information for those who have been prescribed ELIQUIS (apixaban) after hip or knee replacement surgery Always read the
How to prevent blood clots whilst in hospital and after your return home
 How to prevent blood clots whilst in hospital and after your return home Patient information WHAT What IS is DEEP deep VEIN vein THROMBOSIS? thrombosis? Deep Vein Thrombosis DVT is a blood clot within
How to prevent blood clots whilst in hospital and after your return home Patient information WHAT What IS is DEEP deep VEIN vein THROMBOSIS? thrombosis? Deep Vein Thrombosis DVT is a blood clot within
Venous Thromboembolism Prophylaxis
 Approved by: Venous Thromboembolism Prophylaxis Vice President and Chief Medical Officer; and Vice President and Chief Operating Officer Corporate Policy & Procedures Manual Number: Date Approved January
Approved by: Venous Thromboembolism Prophylaxis Vice President and Chief Medical Officer; and Vice President and Chief Operating Officer Corporate Policy & Procedures Manual Number: Date Approved January
Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital
 Please Note: This policy is currently under review and is still fit for purpose. Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital This procedural document supersedes
Please Note: This policy is currently under review and is still fit for purpose. Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital This procedural document supersedes
HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) DABIGATRAN RECOMMENDED What it is Indications Date decision last revised
 Name: generic (trade) Dabigatran etexilate (Pradaxa ) HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) DABIGATRAN RECOMMENDED What it is Indications Date decision last revised Direct thrombin inhibitor
Name: generic (trade) Dabigatran etexilate (Pradaxa ) HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) DABIGATRAN RECOMMENDED What it is Indications Date decision last revised Direct thrombin inhibitor
Approach to Thrombosis
 Approach to Thrombosis Theera Ruchutrakool, M.D. Division of Hematology Department of Medicine Siriraj Hospital Faculty of Medicine Mahidol University Approach to Thrombosis Thrombosis: thrombus formation
Approach to Thrombosis Theera Ruchutrakool, M.D. Division of Hematology Department of Medicine Siriraj Hospital Faculty of Medicine Mahidol University Approach to Thrombosis Thrombosis: thrombus formation
New Oral Anticoagulants in treatment of VTE, PE DR.AMR HANAFY (LECTURER OF CARDIOLOGY ) ASWAN UNIVERSITY
 New Oral Anticoagulants in treatment of VTE, PE DR.AMR HANAFY (LECTURER OF CARDIOLOGY ) ASWAN UNIVERSITY Fact VTE is deadly! It nibbles after it bites! The 30-day mortality rates for first-time DVT or
New Oral Anticoagulants in treatment of VTE, PE DR.AMR HANAFY (LECTURER OF CARDIOLOGY ) ASWAN UNIVERSITY Fact VTE is deadly! It nibbles after it bites! The 30-day mortality rates for first-time DVT or
Disclosures. DVT: Diagnosis and Treatment. Questions To Ask. Dr. Susanna Shin - DVT: Diagnosis and Treatment. Acute Venous Thromboembolism (VTE) None
 Disclosures DVT: Diagnosis and Treatment None Susanna Shin, MD, FACS Assistant Professor University of Washington Acute Venous Thromboembolism (VTE) Deep Venous Thrombosis (DVT) Pulmonary Embolism (PE)
Disclosures DVT: Diagnosis and Treatment None Susanna Shin, MD, FACS Assistant Professor University of Washington Acute Venous Thromboembolism (VTE) Deep Venous Thrombosis (DVT) Pulmonary Embolism (PE)
Anticoagulant treatment for DVT
 Anticoagulant treatment for DVT A guide to your diagnosis and the choice of treatment For more information, please contact: Please see contact details on page 23 of this booklet Caring with pride 2 Caring
Anticoagulant treatment for DVT A guide to your diagnosis and the choice of treatment For more information, please contact: Please see contact details on page 23 of this booklet Caring with pride 2 Caring
INDICATIONS FOR THROMBO-PROPHYLAXIS AND WHEN TO STOP ANTICOAGULATION BEFORE ELECTIVE SURGERY
 INDICATIONS FOR THROMBO-PROPHYLAXIS AND WHEN TO STOP ANTICOAGULATION BEFORE ELECTIVE SURGERY N.E. Pearce INTRODUCTION Preventable death Cause of morbidity and mortality Risk factors Pulmonary embolism
INDICATIONS FOR THROMBO-PROPHYLAXIS AND WHEN TO STOP ANTICOAGULATION BEFORE ELECTIVE SURGERY N.E. Pearce INTRODUCTION Preventable death Cause of morbidity and mortality Risk factors Pulmonary embolism
2 Summary of NICE TA 249: Atrial fibrillation - Dabigatran Etexilate
 Service Notification in response to DHSSPS endorsed NICE Technology Appraisals NICE TA 249: Atrial fibrillation - Dabigatran Etexilate 1 Name of Commissioning Team Long Term Conditions Commissioning Team
Service Notification in response to DHSSPS endorsed NICE Technology Appraisals NICE TA 249: Atrial fibrillation - Dabigatran Etexilate 1 Name of Commissioning Team Long Term Conditions Commissioning Team
New v1.0 Date: December 2015 Patricia Wain - Associate Director Physical Care. Kenny Laing - Deputy Director of Nursing
 Clinical Venous Thromboembolism: Standing Operating Procedure Document Control Summary Status: Version: Author/Title: Owner/Title: New v1.0 Date: December 2015 Patricia Wain - Associate Director Physical
Clinical Venous Thromboembolism: Standing Operating Procedure Document Control Summary Status: Version: Author/Title: Owner/Title: New v1.0 Date: December 2015 Patricia Wain - Associate Director Physical
Preventing blood clots while you are in hospital and after you leave. Information for patients Pharmacy
 Preventing blood clots while you are in hospital and after you leave Information for patients Pharmacy Why does blood clot? When we cut ourselves, we bleed. To stop us from bleeding too much, chemicals
Preventing blood clots while you are in hospital and after you leave Information for patients Pharmacy Why does blood clot? When we cut ourselves, we bleed. To stop us from bleeding too much, chemicals
Inhixa (Enoxaparin Sodium)
 Inhixa (Enoxaparin Sodium) P R E V ENTIS SAFETY D E V I C E P R E V E N T I S I S A N AU TO M AT I C N E E D L E S H I E L D I N G S Y S T E M, W H I C H H A S A C O V E R T H AT E X T E N D S O V E R
Inhixa (Enoxaparin Sodium) P R E V ENTIS SAFETY D E V I C E P R E V E N T I S I S A N AU TO M AT I C N E E D L E S H I E L D I N G S Y S T E M, W H I C H H A S A C O V E R T H AT E X T E N D S O V E R
THROMBOSIS RISK FACTOR ASSESSMENT
 Name: Procedure: Doctor: Date: THROMBOSIS RISK FACTOR ASSESSMENT CHOOSE ALL THAT APPLY EACH RISK FACTOR REPRESENTS 1 POINT Age 41 60 years Minor Surgery Planned History of Prior Major Surgery (< 1 month)
Name: Procedure: Doctor: Date: THROMBOSIS RISK FACTOR ASSESSMENT CHOOSE ALL THAT APPLY EACH RISK FACTOR REPRESENTS 1 POINT Age 41 60 years Minor Surgery Planned History of Prior Major Surgery (< 1 month)
Primary Care Prescriber Information RIVAROXABAN (XARELTO ) Prevention of stroke and embolism for nonvalvular atrial fibrillation
 Primary Care Prescriber Information RIVAROXABAN (XARELTO ) Prevention of stroke and embolism for nonvalvular atrial fibrillation INDICATION Rivaroxaban is a non-vitamin K antagonist oral anticoagulant
Primary Care Prescriber Information RIVAROXABAN (XARELTO ) Prevention of stroke and embolism for nonvalvular atrial fibrillation INDICATION Rivaroxaban is a non-vitamin K antagonist oral anticoagulant
PULMONARY EMBOLISM (PE): DIAGNOSIS AND TREATMENT
 PULMONARY EMBOLISM (PE): DIAGNOSIS AND TREATMENT OBJECTIVE: To provide a diagnostic algorithm and treatment options for patients with acute pulmonary embolism (PE). BACKGROUND: Venous thromboembolism (VTE)
PULMONARY EMBOLISM (PE): DIAGNOSIS AND TREATMENT OBJECTIVE: To provide a diagnostic algorithm and treatment options for patients with acute pulmonary embolism (PE). BACKGROUND: Venous thromboembolism (VTE)
Guidance for management of bleeding in patients taking the new oral anticoagulant drugs: rivaroxaban, dabigatran or apixaban
 Guidance for management of bleeding in patients taking the new oral anticoagulant drugs: rivaroxaban, dabigatran or apixaban Purpose The aim of this guidance is to outline the management of patients presenting
Guidance for management of bleeding in patients taking the new oral anticoagulant drugs: rivaroxaban, dabigatran or apixaban Purpose The aim of this guidance is to outline the management of patients presenting
NEW/NOVEL ORAL ANTICOAGULANTS (NOACS): COMPARISON AND FREQUENTLY ASKED QUESTIONS
 NEW/NOVEL ORAL ANTICOAGULANTS (NOACS): COMPARISON AND FREQUENTLY ASKED QUESTIONS OBJECTIVES: To provide a comparison of the new/novel oral anticoagulants (NOACs) currently available in Canada. To address
NEW/NOVEL ORAL ANTICOAGULANTS (NOACS): COMPARISON AND FREQUENTLY ASKED QUESTIONS OBJECTIVES: To provide a comparison of the new/novel oral anticoagulants (NOACs) currently available in Canada. To address
Prevention and treatment of venous thromboembolic disease
 REVIEW Prevention and treatment of venous thromboembolic disease SUSAN McNEILL AND CATHERINE BAGOT Awareness of the risk factors for venous thromboembolic (VTE) disease and timely administration of thromboprophylaxis
REVIEW Prevention and treatment of venous thromboembolic disease SUSAN McNEILL AND CATHERINE BAGOT Awareness of the risk factors for venous thromboembolic (VTE) disease and timely administration of thromboprophylaxis
Treatment of deep vein thrombosis and pulmonary embolism with low molecular weight heparin
 Treatment of deep vein thrombosis and pulmonary embolism with low molecular weight heparin You have been given this leaflet because you have a blood clot. Normally blood flows freely around the body without
Treatment of deep vein thrombosis and pulmonary embolism with low molecular weight heparin You have been given this leaflet because you have a blood clot. Normally blood flows freely around the body without
PULMONARY EMBOLISM MANAGEMENT GUIDELINES
 PULMONARY EMBOLISM MANAGEMENT GUIDELINES This document is adapted from the NICE guidelines titled Venous thromboembolic diseases: the management of venous thromboembolic diseases and the role of thrombophilia
PULMONARY EMBOLISM MANAGEMENT GUIDELINES This document is adapted from the NICE guidelines titled Venous thromboembolic diseases: the management of venous thromboembolic diseases and the role of thrombophilia
The legally binding text is the original French version. Opinion 15 May 2013
 The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 15 May 2013 ARIXTRA 2.5 mg/0.5 ml, solution for injection in pre-filled syringe B/2 (CIP: 34009 359 225 4 0) B/7 (CIP:
The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 15 May 2013 ARIXTRA 2.5 mg/0.5 ml, solution for injection in pre-filled syringe B/2 (CIP: 34009 359 225 4 0) B/7 (CIP:
CURRENT & FUTURE THERAPEUTIC MANAGEMENT OF VENOUS THROMBOEMBOLISM. Gordon Lowe Professor of Vascular Medicine University of Glasgow
 CURRENT & FUTURE THERAPEUTIC MANAGEMENT OF VENOUS THROMBOEMBOLISM Gordon Lowe Professor of Vascular Medicine University of Glasgow VENOUS THROMBOEMBOLISM Common cause of death and disability 50% hospital-acquired
CURRENT & FUTURE THERAPEUTIC MANAGEMENT OF VENOUS THROMBOEMBOLISM Gordon Lowe Professor of Vascular Medicine University of Glasgow VENOUS THROMBOEMBOLISM Common cause of death and disability 50% hospital-acquired
VTE Prophylaxis. NEW NICE guidance. Providing venous thromboembolism (VTE) prophylaxis to all at risk hospital patients
 NEW NICE guidance NICE guidance (MTG19) supports the use of the geko device for people who have a high risk of VTE and for whom pharmacological or other mechanical methods of VTE prevention are impractical
NEW NICE guidance NICE guidance (MTG19) supports the use of the geko device for people who have a high risk of VTE and for whom pharmacological or other mechanical methods of VTE prevention are impractical
Confirmed blood clot
 n The Leeds Teaching Hospitals NHS Trust Confirmed blood clot (Deep vein thrombosis and pulmonary embolism) Information for patients Please read this leaflet carefully. It will give you information about
n The Leeds Teaching Hospitals NHS Trust Confirmed blood clot (Deep vein thrombosis and pulmonary embolism) Information for patients Please read this leaflet carefully. It will give you information about
Treatment Options and How They Work
 Treatment Options and How They Work Robin Offord Director of Clinical Pharmacy UCL Hospitals NHS Foundation Trust robin.offord@uclh.nhs.uk Introducing the term anticoagulant... What they do Inhibit the
Treatment Options and How They Work Robin Offord Director of Clinical Pharmacy UCL Hospitals NHS Foundation Trust robin.offord@uclh.nhs.uk Introducing the term anticoagulant... What they do Inhibit the
PRACTICAL GUIDE LIXIANA (edoxaban)
 NICE RECOMMENDED AND SMC ACCEPTED IN NVAF AND VTE 1-4 PRACTICAL GUIDE LIXIANA (edoxaban) For clinical data and prescriber resources please visit www.lixiana.co.uk Please refer to the Prescribing Information
NICE RECOMMENDED AND SMC ACCEPTED IN NVAF AND VTE 1-4 PRACTICAL GUIDE LIXIANA (edoxaban) For clinical data and prescriber resources please visit www.lixiana.co.uk Please refer to the Prescribing Information
Deep vein thrombosis: diagnosis, prevention and treatment
 Deep vein thrombosis: diagnosis, prevention and treatment Catherine Bagot BSc, MD, MRCP, FRCPath and Campbell Tait BSc, FRCP, FRCPath Deep vein thrombosis can lead to significant morbidity and has well-recognised
Deep vein thrombosis: diagnosis, prevention and treatment Catherine Bagot BSc, MD, MRCP, FRCPath and Campbell Tait BSc, FRCP, FRCPath Deep vein thrombosis can lead to significant morbidity and has well-recognised
Direct Oral Anticoagulants (DOACs). Dr GM Benson Director NI Haemophilia Comprehensive Care Centre and Thrombosis Unit BHSCT
 Direct Oral Anticoagulants (DOACs). Dr GM Benson Director NI Haemophilia Comprehensive Care Centre and Thrombosis Unit BHSCT OAC WARFARIN Gold standard DABIGATRAN RIVAROXABAN APIXABAN EDOXABAN BETRIXABAN
Direct Oral Anticoagulants (DOACs). Dr GM Benson Director NI Haemophilia Comprehensive Care Centre and Thrombosis Unit BHSCT OAC WARFARIN Gold standard DABIGATRAN RIVAROXABAN APIXABAN EDOXABAN BETRIXABAN
Pulmonary Embolism Pathway
 Pulmonary Embolism Pathway Ambulatory Care Pathway Dr. A. Zafar, Dr. A. Rehman, Dr. T. Malik September, 2011. Patient Identification Label Pulmonary Embolism Pathway Clinical History Comments Hospital
Pulmonary Embolism Pathway Ambulatory Care Pathway Dr. A. Zafar, Dr. A. Rehman, Dr. T. Malik September, 2011. Patient Identification Label Pulmonary Embolism Pathway Clinical History Comments Hospital
MEDICAL ASSISTANCE HANDBOOK PRIOR AUTHORIZATION OF PHARMACEUTICAL SERVICES. A. Prescriptions That Require Prior Authorization
 MEDICAL ASSISTANCE HBOOK PRI AUTHIZATION OF PHARMACEUTICAL SERVICES I. Requirements for Prior Authorization of Anticoagulants A. Prescriptions That Require Prior Authorization Prescriptions for Anticoagulants
MEDICAL ASSISTANCE HBOOK PRI AUTHIZATION OF PHARMACEUTICAL SERVICES I. Requirements for Prior Authorization of Anticoagulants A. Prescriptions That Require Prior Authorization Prescriptions for Anticoagulants
PART VI Summary of Activities in the Risk Management Plan by Product
 PART VI Summary of Activities in the Risk Management Plan by Product Active substance(s) (INN or common name): Rivaroxaban Medicinal products to which this RMP refers: 1 Name of Marketing Authorisation
PART VI Summary of Activities in the Risk Management Plan by Product Active substance(s) (INN or common name): Rivaroxaban Medicinal products to which this RMP refers: 1 Name of Marketing Authorisation
Technology appraisal guidance Published: 26 June 2013 nice.org.uk/guidance/ta287
 Rivaroxaban for treating pulmonary embolism and preventing enting recurrent venous thromboembolism Technology appraisal guidance Published: 26 June 2013 nice.org.uk/guidance/ta287 NICE 2018. All rights
Rivaroxaban for treating pulmonary embolism and preventing enting recurrent venous thromboembolism Technology appraisal guidance Published: 26 June 2013 nice.org.uk/guidance/ta287 NICE 2018. All rights
NICE Guidance: Venous thromboembolism (deep vein thrombosis and pulmonary embolism) in patients admitted to hospital 1
 The College of Emergency Medicine Patron: HRH The Princess Royal Churchill House Tel +44 (0)207 404 1999 35 Red Lion Square Fax +44 (0)207 067 1267 London WC1R 4SG www.collemergencymed.ac.uk CLINICAL EFFECTIVENESS
The College of Emergency Medicine Patron: HRH The Princess Royal Churchill House Tel +44 (0)207 404 1999 35 Red Lion Square Fax +44 (0)207 067 1267 London WC1R 4SG www.collemergencymed.ac.uk CLINICAL EFFECTIVENESS
Deep Vein Thrombosis
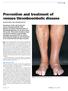
 Deep Vein Thrombosis Introduction Deep vein thrombosis (DVT) is a blood clot in a vein. This condition can affect men and women of any age and race. DVT is a potentially serious condition. If not treated,
Deep Vein Thrombosis Introduction Deep vein thrombosis (DVT) is a blood clot in a vein. This condition can affect men and women of any age and race. DVT is a potentially serious condition. If not treated,
Venous Thromboembolism National Hospital Inpatient Quality Measures
 Venous Thromboembolism National Hospital Inpatient Quality Measures Presentation Overview Review venous thromboembolism as a new mandatory measure set Outline measures with exclusions and documentation
Venous Thromboembolism National Hospital Inpatient Quality Measures Presentation Overview Review venous thromboembolism as a new mandatory measure set Outline measures with exclusions and documentation
PRESENTATION TITLE. Case Studies
 PRESENTATION TITLE Case Studies 1) SH is a 67 year old male. He has a history of type 2 diabetes, controlled hypertension and peripheral artery disease. He takes naproxen 500mg bd for arthritis and admits
PRESENTATION TITLE Case Studies 1) SH is a 67 year old male. He has a history of type 2 diabetes, controlled hypertension and peripheral artery disease. He takes naproxen 500mg bd for arthritis and admits
Medicines Management Group
 Title SHARED CARE PROTOCOL for Extended Treatment of symptomatic VTE and prevention of its recurrence in Cancer Patients with Solid and Haematological Tumours Scope: Dalteparin (Fragmin ) may be considered
Title SHARED CARE PROTOCOL for Extended Treatment of symptomatic VTE and prevention of its recurrence in Cancer Patients with Solid and Haematological Tumours Scope: Dalteparin (Fragmin ) may be considered
NHS Kent and Medway Medicines Management. Dronedarone (Multaq ) Shared Care Guideline For Prescribing
 NHS Kent and Medway Medicines Management Dronedarone (Multaq ) Shared Care Guideline For Prescribing Issue No: 2 Review Date (If Applicable): Accountable Officer: Heather Lucas Contact Details: 01233 618158
NHS Kent and Medway Medicines Management Dronedarone (Multaq ) Shared Care Guideline For Prescribing Issue No: 2 Review Date (If Applicable): Accountable Officer: Heather Lucas Contact Details: 01233 618158
Primary Care Prescriber Information EDOXABAN (LIXIANA ) Prevention of stroke and embolism for nonvalvular atrial fibrillation
 Primary Care Prescriber Information EDOXABAN (LIXIANA ) Prevention of stroke and embolism for nonvalvular atrial fibrillation INDICATION Edoxaban is a non-vitamin K antagonist oral anticoagulant (NOAC)
Primary Care Prescriber Information EDOXABAN (LIXIANA ) Prevention of stroke and embolism for nonvalvular atrial fibrillation INDICATION Edoxaban is a non-vitamin K antagonist oral anticoagulant (NOAC)
Rivaroxaban (Xarelto) in the management of stroke and DVT
 Rivaroxaban (Xarelto) in the management of stroke and DVT Steve Chaplin MSc, MRPharmS, Victoria Haunton BM, DGM, MRCP, Thompson Robinson BMedSci, MD, FRCP and Catherine Bagot MD, FRCPath KEY POINTS is
Rivaroxaban (Xarelto) in the management of stroke and DVT Steve Chaplin MSc, MRPharmS, Victoria Haunton BM, DGM, MRCP, Thompson Robinson BMedSci, MD, FRCP and Catherine Bagot MD, FRCPath KEY POINTS is
XARELTO 15 Film-coated tablets XARELTO 20 Film-coated tablets
 SCHEDULING STATUS: S4 PROPRIETARY NAME AND DOSAGE FORM: XARELTO 15 Film-coated tablets XARELTO 20 Film-coated tablets COMPOSITION: XARELTO 15: Each film-coated tablet contains rivaroxaban 15 mg. XARELTO
SCHEDULING STATUS: S4 PROPRIETARY NAME AND DOSAGE FORM: XARELTO 15 Film-coated tablets XARELTO 20 Film-coated tablets COMPOSITION: XARELTO 15: Each film-coated tablet contains rivaroxaban 15 mg. XARELTO
Understanding thrombosis in venous thromboembolism. João Morais Head of Cardiology Division and Research Centre Leiria Hospital Centre Portugal
 Understanding thrombosis in venous thromboembolism João Morais Head of Cardiology Division and Research Centre Leiria Hospital Centre Portugal Disclosures João Morais On the last year JM received honoraria
Understanding thrombosis in venous thromboembolism João Morais Head of Cardiology Division and Research Centre Leiria Hospital Centre Portugal Disclosures João Morais On the last year JM received honoraria
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE. Single Technology Appraisal (STA)
 Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Misunderstandings of Venous thromboembolism prophylaxis
 Misunderstandings of Venous thromboembolism prophylaxis Veerendra Chadachan Senior Consultant Dept of General Medicine (Vascular Medicine and Hypertension) Tan Tock Seng Hospital, Singapore Case scenario
Misunderstandings of Venous thromboembolism prophylaxis Veerendra Chadachan Senior Consultant Dept of General Medicine (Vascular Medicine and Hypertension) Tan Tock Seng Hospital, Singapore Case scenario
Tablet core: Microcrystalline cellulose, croscarmellose sodium, lactose, hypromellose, sodium lauryl sulfate, magnesium stearate.
 PRODUCT INFORMATION XARELTO (rivaroxaban) NAME OF THE MEDICINE Xarelto (rivaroxaban) is a selective, direct acting Factor Xa inhibitor. Rivaroxaban is 5-Chloro-N-({(5S)-2-oxo-3-[4-(3-oxo-4-morpholinyl)phenyl]-1,3-oxazolidin-5-
PRODUCT INFORMATION XARELTO (rivaroxaban) NAME OF THE MEDICINE Xarelto (rivaroxaban) is a selective, direct acting Factor Xa inhibitor. Rivaroxaban is 5-Chloro-N-({(5S)-2-oxo-3-[4-(3-oxo-4-morpholinyl)phenyl]-1,3-oxazolidin-5-
Bortezomib, Thalidomide & Dexamethasone
 DRUG ADMINISTRATION SCHEDULE Cumbria, Northumberland, Tyne & Wear Area Team Day Drug Dose Route Diluent Rate 1, 4, 8, & 11 2 Bortezomib 1.3mg/m IV bolus/ SC injection* None Fast bolus: 3 to 5 seconds 1
DRUG ADMINISTRATION SCHEDULE Cumbria, Northumberland, Tyne & Wear Area Team Day Drug Dose Route Diluent Rate 1, 4, 8, & 11 2 Bortezomib 1.3mg/m IV bolus/ SC injection* None Fast bolus: 3 to 5 seconds 1
Venous Thrombo-Embolism (VTE)
 Venous Thrombo-Embolism (VTE) Information for service users and carers RDaSH leading the way with care Older People s Mental Health Services Reducing risk of unwanted blood clots whilst in hospital About
Venous Thrombo-Embolism (VTE) Information for service users and carers RDaSH leading the way with care Older People s Mental Health Services Reducing risk of unwanted blood clots whilst in hospital About
Rivaroxaban to prevent blood clots for patients who have a lower limb plaster cast. Information for patients Pharmacy
 Rivaroxaban to prevent blood clots for patients who have a lower limb plaster cast Information for patients Pharmacy Your doctor has prescribed a tablet called rivaroxaban. This leaflet tells you about
Rivaroxaban to prevent blood clots for patients who have a lower limb plaster cast Information for patients Pharmacy Your doctor has prescribed a tablet called rivaroxaban. This leaflet tells you about
Risk factors for DVT. Venous thrombosis & pulmonary embolism. Anticoagulation (cont d) Diagnosis 1/5/2018. Ahmed Mahmoud, MD
 Risk factors for DVT Venous thrombosis & pulmonary embolism Ahmed Mahmoud, MD Surgery ; post op especially for long cases, pelvic operations (THR), Trauma ; long bone fractures, pelvic fractures (posterior
Risk factors for DVT Venous thrombosis & pulmonary embolism Ahmed Mahmoud, MD Surgery ; post op especially for long cases, pelvic operations (THR), Trauma ; long bone fractures, pelvic fractures (posterior
Venous thrombosis & pulmonary embolism. Ahmed Mahmoud, MD
 Venous thrombosis & pulmonary embolism Ahmed Mahmoud, MD Risk factors for DVT Surgery ; post op especially for long cases, pelvic operations (THR), Trauma ; long bone fractures, pelvic fractures (posterior
Venous thrombosis & pulmonary embolism Ahmed Mahmoud, MD Risk factors for DVT Surgery ; post op especially for long cases, pelvic operations (THR), Trauma ; long bone fractures, pelvic fractures (posterior
Venous Thromboembolism Policy (VTE)
 Venous Thromboembolism Policy (VTE) DOCUMENT CONTROL: Version: v3 Ratified by: Clinical Quality Group Date ratified: 2 February 2016 Name of originator/author: Modern Matron St John s Hospice Name of responsible
Venous Thromboembolism Policy (VTE) DOCUMENT CONTROL: Version: v3 Ratified by: Clinical Quality Group Date ratified: 2 February 2016 Name of originator/author: Modern Matron St John s Hospice Name of responsible
Deep Vein Thrombosis and Pulmonary Embolism: Patient Information
 Deep Vein Thrombosis and Pulmonary Embolism: Patient Information A Deep Vein Thrombosis (DVT) and a Pulmonary Embolism (PE) are both disorders of unwanted blood clotting. Unwanted blood clots can occur
Deep Vein Thrombosis and Pulmonary Embolism: Patient Information A Deep Vein Thrombosis (DVT) and a Pulmonary Embolism (PE) are both disorders of unwanted blood clotting. Unwanted blood clots can occur
