Supplementary Appendix
|
|
|
- Debra Lee
- 5 years ago
- Views:
Transcription
1 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Bergenstal RM, Klonoff DC, Garg SK, et al. -based insulin-pump interruption for reduction of hypoglycemia. N Engl J Med 2013;369: DOI: /NEJMoa
2 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Bergenstal RM, et al. -Based Insulin-Pump Interruption for Reduction of Hypoglycemia Contents Members of the ASPIRE In-Home Study Group... 2 Description of Multiple Imputations Method... 3 Figure S1. Study Visits Figure S2. Example of a Sensor-Augmented Pump System... 5 Figure S3. Sensor Glucose Distributions After Nocturnal 2-h Events... 6 Table S1. Primary and Secondary Outcomes... 7 Table S2. Sensor Glucose Distributions... 8 Table S3. Outcomes by Age... 9 Table S4. Outcomes by Glycated Hemoglobin Table S5. Outcomes by Duration of Diabetes Table S6. Device Settings and Behaviors; Glucose Values
3 Members of the ASPIRE In-Home Study Group In addition to the authors, the following investigators participated in the ASPIRE In- Home Study: AMCR Institute, Escondido, CA T. Bailey; Rainier Clinical Research Center, Renton, WA R. Brazg; Texas Diabetes and Endocrinology, Austin, TX L. Casaubon; Palm Beach Diabetes and Endocrine Specialists, West Palm Beach, FL B. Horowitz; Rocky Mountain Diabetes and Osteoporosis Center, Idaho Falls, ID D. Liljenquist; Physicians Research Associates, LLC, Lawrenceville, GA O. Odugbesan; Endocrine Research Solutions, Inc., Roswell, GA J. Reed; Diabetes Ohio University, Athens, OH F. Schwartz; Arkansas Diabetes Clinic and Research Center, Little Rock, AR J. Thrasher; Iowa Diabetes and Endocrinology Center (IDEC), Des Moines, IA A. Bhargava; The Naomi Berrie Diabetes Center, Columbia University, New York, NY: R. Goland; Joslin Diabetes Center (Syracuse University), Syracuse, NY R. Weinstock. 2
4 Description of Multiple Imputations Method The multiple imputations method was adopted to handle missing data in glycated hemoglobin measurements. In each round of imputation, the missing glycated hemoglobin at the end of 3- month study was imputed by a random number which was generated by the MI procedure in SAS 9.2 with imputation regression method. The independent variables in the regression model were age, gender, baseline glycated hemoglobin, body-mass index, and treatment group. The missing glycated hemoglobin data were imputed by yˆ z ˆ, where ŷ is the predicted value from regression, z is a standard normal random variable, and ˆ is the estimated standard deviation of the random residual from the regression model. The imputation was performed five times using the MI procedure and the analysis results were combined to form one inference using the MIANALYZE procedure in SAS 9.2. We also performed analysis on the complete case population where the early withdrawals were excluded. The 95% confidence interval of treatment difference remains the same ([-0.05%, 0.15%]). 3
5 Figure S1. Study Visits. The study included a 2-week Run-In Phase to establish eligibility for the 3-month Study Phase. Blood samples for glycated hemoglobin determination were drawn at Visits 1, 4, and 7. Reported changes in glycated hemoglobin were based on samples drawn at Visits 4 and 7 Pre-Study Phase (6 weeks) Run-in Phase (2 weeks) 2-week run-in phase may be repeated once if Nocturnal Hypoglycemia requirements not met. Visit weeks Pump Training Pump Start Visit weeks Sensor Training Sensor Start Visit weeks Randomization Glycated Hemoglobin Feature Training for Group Visit week Post randomization Phone call Group Group Visit week Post randomization Phone call Study Phase (12 weeks) Visit weeks Post randomization Phone call Visit weeks Post randomization Phone call Visit weeks Post randomization End of Study Glycated Hemoglobin Visit weeks Post randomization End of Study Glycated Hemoglobin Visit 1 Day 0 Baseline Glycated Hemoglobin Screening Informed Consent 4 4
6 Figure S2. Example of a Sensor-Augmented Pump System The hardware used in the Group and in the Group was identical. The threshold suspend feature was an addition to the software used to control the pump. Glucose sensor Infusion site Transmitter Insulin pump with glucose display Group Standard pump software plus threshold suspend algorithm Group Standard pump software 5
7 Figure S3. Sensor Glucose Distributions After Nocturnal 2-hour Events There were at total of hour threshold suspend events that had an event marker in pump uploads, of which 1444 were at night. Not all had SG sensor glucose values at 0, 2, and 4 hours after pump suspension: 1438 had an event marker plus sensor glucose values at 0 hours 1385 had an event marker plus sensor glucose values at 2 hours 1136 had an event marker plus sensor glucose values at 4 hours The percentages of sensor glucose values in various ranges following 2-hour threshold suspend events are shown below for the 1385 events with data at 2 hours (left) and for the 1136 events with data at 4 hours (right). See Figure 3 for mean ± SD sensor glucose values before, during, and after the hour nocturnal threshold suspend events that had an event marker plus sensor glucose values. Sensor Glucose Distribution After 2-Hour Events 100% 2.2% 90% 26.0% 80% Percentage of Values 70% 60% 50% 40% 30% 64.8% >200 mg/dl 70 to 200 mg/dl <70 mg/dl 70.2% 20% 33.1% 10% 0% 2 hours (Time of insulin resumption) 3.9% 4 hours (2 hours after insulin resumption) 6
8 Table S1. Primary and Secondary Outcomes Randomization, Run-In, and Study Phase glycated hemoglobin and hypoglycemia parameters, mean ± SD. Comparisons for glycated hemoglobin and mean AUC of nocturnal hypoglycemic events are shown in Figure 2A and Figure 2B of the article. (n=121) (n=126) Randomization* 7.26 ± ± 0.77 Glycated hemoglobin, % 3-month 7.24 ± ± 0.77 Change 0.00 ± ± 0.42 AUC of nocturnal hypoglycemic events, mg/dl min Nocturnal hypoglycemia events per patient-week AUC of day and night hypoglycemia events, mg/dl min Day and night hypoglycemia events per patient-week Run-In Phase 1547 ± ± 1950 Study Phase 980 ± ± 1995 Run-In Phase 2.4 ± ± 1.5 Study Phase 1.5 ± ± 1.3 Run-In Phase 1153 ± ± 1557 Study Phase 798 ± ± 1590 Run-In Phase 5.0 ± ± 3.0 Study Phase 3.3 ± ± 2.7 * A randomization glycated hemoglobin value was not obtained from one patient in the Group. Based on values from 116 and 124 patients who had available glycated hemoglobin values in the and Groups, respectively. versus, p<
9 Table S2. Sensor Glucose Distributions Sensor glucose distributions, nocturnal and combined day and night. Comparisons for the <50, 50 to <60, and 60 to <70 mg/dl ranges are shown in Figure 2C of the article. Sensor Glucose Range, mg/dl Nocturnal Combined Day and Night <50 1.2%* 2.8% 0.9%* 1.9% 50 to <60 1.8%* 3.1% 1.6%* 2.5% 60 to <70 3.0%* 4.1% 2.8%* 3.7% 70 to < % 62.2% 63.1% 61.4% 180 to < % 19.9% 22.3% 21.0% % 7.9% 9.3% 9.4% *, versus, p<
10 Table S3. Outcomes by Age Glycated hemoglobin and nocturnal hypoglycemia event AUC outcomes for patients of different age categories, mean ± SD. Glycated Hemoglobin, % AUC of nocturnal hypoglycemia events, mg/dl min (n=12) Randomization 7.56 ± month 7.41 ± 0.76 Study Phase 1439 ± 1711 Age Age Age (n=14) 7.64 ± ± ± 2625 (n=79) 7.20 ± ± ± 1117 (n=60) 7.07 ± ± ± 1912 (n=30) 7.28 ± ± ± 1031 (n=52) 7.25 ± ± ±
11 Table S4. Outcomes by Glycated Hemoglobin Glycated hemoglobin and nocturnal hypoglycemia event AUC outcomes for patients of different glycated hemoglobin levels at randomization, mean ± SD Glycated Hemoglobin, % AUC of nocturnal hypoglycemia events, mg/dl min Randomization Glycated Hemoglobin 7% (n=50) (n=58) Randomization Glycated Hemoglobin >7% (n=70) (n=68) Randomization 6.61 ± ± ± ± month 6.75 ± ± ± ± 0.68 Study Phase 1055 ± ± ± ±
12 Table S5. Outcomes by Duration of Diabetes Glycated hemoglobin and nocturnal hypoglycemia event AUC outcomes for patients of different diabetes durations, mean ± SD Glycated Hemoglobin, % AUC of nocturnal hypoglycemia events, mg/dl min Duration of Diabetes 25 years (n=57) (n=56) Duration of Diabetes >25 years (n=63) (n=67) Randomization 7.33 ± ± ± ± month 7.34 ± ± ± ± 0.74 Study Phase 1076 ± ± ± ±
13 Table S6. Device Settings and Behaviors; Glucose Values Insulin Delivery, pump and CGM settings and usage, meter blood glucose and calibration frequencies, and glucose values, Study Phase. Insulin Delivery* Total Insulin Dose, U 47.8 ± ± Basal Insulin, U 22.7 ± ± Bolus Insulin, U 25.1 ± ± Bolus Delivery 6.7 ± ± 2.03 Pump and CGM Settings and Usage* Sensor Wear (%) 88.1 ± ± Sensor Life (days) 4.8 ± ± 1.84 Bolus Calculator Use 5.6 ± ± 2.29 Meter Blood Glucose Frequency* Study Phase (first 2 weeks) 6.2 ± ± 2.39 End of Study Phase (last 2 weeks) 5.6 ± ± 2.03 Calibration Frequency * Study Phase (first 2 weeks) 4.3 ± ± 1.21 End of Study Phase (last 2 weeks) 4.1 ± ± 1.10 Meter Blood Glucose, mg/dl Run-In Phase ± ± 23.6 Study Phase (first 2 weeks) ± ± 26.9 End of Study Phase (last 2 weeks) ± ± 32.1 Sensor Glucose, mg/dl Run-In Phase ± ± 21.0 Study Phase (first 2 weeks) ± ± 20.7 End of Study Phase (last 2 weeks) ± ± 24.9 Weight, kg Beginning of Study Phase 79.6 ± ± End of Study Phase 80.7 ± ± Results are given as mean per patient-day (*) or mean of mean per patient per 2 week interval ( ) 12
Reduction in Hypoglycemia and No Increase in A1C with Threshold-Based Sensor-Augmented Pump (SAP) Insulin Suspension: ASPIRE In-Home
 Reduction in Hypoglycemia and No Increase in A1C with Threshold-Based Sensor-Augmented Pump (SAP) Insulin Suspension: ASPIRE In-Home Richard M. Bergenstal 1, David C. Klonoff 2, Bruce W. Bode 3, Satish
Reduction in Hypoglycemia and No Increase in A1C with Threshold-Based Sensor-Augmented Pump (SAP) Insulin Suspension: ASPIRE In-Home Richard M. Bergenstal 1, David C. Klonoff 2, Bruce W. Bode 3, Satish
PREVENTION OF NOCTURNAL HYPOGLYCEMIA USING PREDICTIVE LOW GLUCOSE SUSPEND (PLGS)
 PREVENTION OF NOCTURNAL HYPOGLYCEMIA USING PREDICTIVE LOW GLUCOSE SUSPEND (PLGS) Pathways for Future Treatment and Management of Diabetes H. Peter Chase, MD Carousel of Hope Symposium Beverly Hilton, Beverly
PREVENTION OF NOCTURNAL HYPOGLYCEMIA USING PREDICTIVE LOW GLUCOSE SUSPEND (PLGS) Pathways for Future Treatment and Management of Diabetes H. Peter Chase, MD Carousel of Hope Symposium Beverly Hilton, Beverly
Artificial Pancreas Device Systems. Populations Interventions Comparators Outcomes. pump. pump
 Protocol Artificial Pancreas Device Systems (10130) Medical Benefit Effective Date: 04/01/18 Next Review Date: 01/19 Preauthorization Yes Review Dates: 03/15, 03/16, 03/17, 01/18 Preauthorization is required.
Protocol Artificial Pancreas Device Systems (10130) Medical Benefit Effective Date: 04/01/18 Next Review Date: 01/19 Preauthorization Yes Review Dates: 03/15, 03/16, 03/17, 01/18 Preauthorization is required.
CAROLINAS CHAPTER/AMERICAN ASSOCIATION OF CLINICAL ENDOCRINOLOGISTS Annual Meeting HILTON HEAD ISLAND FRIDAY PRESENTATION
 CAROLINAS CHAPTER/AMERICAN ASSOCIATION OF CLINICAL ENDOCRINOLOGISTS 2016 Annual Meeting HILTON HEAD ISLAND FRIDAY PRESENTATION September 9-11, 2016 ~ Sonesta Resort ~ Hilton Head Island, SC This continuing
CAROLINAS CHAPTER/AMERICAN ASSOCIATION OF CLINICAL ENDOCRINOLOGISTS 2016 Annual Meeting HILTON HEAD ISLAND FRIDAY PRESENTATION September 9-11, 2016 ~ Sonesta Resort ~ Hilton Head Island, SC This continuing
Artificial Pancreas Device Systems. Populations Interventions Comparators Outcomes Individuals: With type 1 diabetes
 Protocol Artificial Pancreas Device Systems Medical Benefit Effective Date: 07/01/18 Next Review Date: 01/20 Preauthorization Yes Review Dates: 03/15, 03/16, 03/17, 01/18, 05/18, 01/19 Preauthorization
Protocol Artificial Pancreas Device Systems Medical Benefit Effective Date: 07/01/18 Next Review Date: 01/20 Preauthorization Yes Review Dates: 03/15, 03/16, 03/17, 01/18, 05/18, 01/19 Preauthorization
What is a CGM? (Continuous Glucose Monitor) The Bionic Pancreas Is Coming
 The Bionic Pancreas Is Coming Montana Diabetes Professional Conference October 23, 2014 H. Peter Chase, MD Professor of Pediatrics University of Colorado Barbara Davis Center Stanford: Bruce Buckingham,
The Bionic Pancreas Is Coming Montana Diabetes Professional Conference October 23, 2014 H. Peter Chase, MD Professor of Pediatrics University of Colorado Barbara Davis Center Stanford: Bruce Buckingham,
Insulin Pumps and Glucose Sensors in Diabetes Management
 Diabetes Update+ 2014 Congress Whistler, British Columbia Friday March 21, 2014ǀ 8:15 8:45 am Insulin Pumps and Glucose Sensors in Diabetes Management Bruce A Perkins MD MPH Division of Endocrinology Associate
Diabetes Update+ 2014 Congress Whistler, British Columbia Friday March 21, 2014ǀ 8:15 8:45 am Insulin Pumps and Glucose Sensors in Diabetes Management Bruce A Perkins MD MPH Division of Endocrinology Associate
Artificial Pancreas Device System (APDS)
 Medical Policy Manual Durable Medical Equipment, Policy No. 77 Artificial Pancreas Device System (APDS) Next Review: October 2019 Last Review: October 2018 Effective: November 1, 2018 IMPORTANT REMINDER
Medical Policy Manual Durable Medical Equipment, Policy No. 77 Artificial Pancreas Device System (APDS) Next Review: October 2019 Last Review: October 2018 Effective: November 1, 2018 IMPORTANT REMINDER
The In-Clinic Close Loop Experience in the US
 The In-Clinic Close Loop Experience in the US Keystone Symposium, Practical Ways to Achieve Targets in Diabetes Care July 14, 2012 Francine Ratner Kaufman, MD Chief Medical Officer, VP Global Medical,
The In-Clinic Close Loop Experience in the US Keystone Symposium, Practical Ways to Achieve Targets in Diabetes Care July 14, 2012 Francine Ratner Kaufman, MD Chief Medical Officer, VP Global Medical,
CareLink. software REPORT REFERENCE GUIDE. Management Software for Diabetes
 CareLink Management Software for Diabetes software REPORT REFERENCE GUIDE How to use this guide Each type of CareLink report and its components are described in the following sections. Report data used
CareLink Management Software for Diabetes software REPORT REFERENCE GUIDE How to use this guide Each type of CareLink report and its components are described in the following sections. Report data used
1. Continuous Glucose Monitoring
 1. Continuous Glucose Monitoring 1. Physiology of interstitial fluid glucose 2. Comparison of CGM and self-monitored blood glucose (SMBG) data 3. Insulin dosing indication in BGM vs. CGM & the FDA 4. Protection
1. Continuous Glucose Monitoring 1. Physiology of interstitial fluid glucose 2. Comparison of CGM and self-monitored blood glucose (SMBG) data 3. Insulin dosing indication in BGM vs. CGM & the FDA 4. Protection
Presented by Dr. Bruce Perkins, MD MPH Dr. Michael Riddell, PhD
 Type 1 Diabetes and Exercise: Optimizing the Medtronic MiniMed Veo Insulin Pump and Continuous Glucose Monitoring (CGM) for Better Glucose Control 1,2 for Healthcare Professionals Presented by Dr. Bruce
Type 1 Diabetes and Exercise: Optimizing the Medtronic MiniMed Veo Insulin Pump and Continuous Glucose Monitoring (CGM) for Better Glucose Control 1,2 for Healthcare Professionals Presented by Dr. Bruce
Report Reference Guide. THERAPY MANAGEMENT SOFTWARE FOR DIABETES CareLink Report Reference Guide 1
 Report Reference Guide THERAPY MANAGEMENT SOFTWARE FOR DIABETES CareLink Report Reference Guide 1 How to use this guide Each type of CareLink report and its components are described in the following sections.
Report Reference Guide THERAPY MANAGEMENT SOFTWARE FOR DIABETES CareLink Report Reference Guide 1 How to use this guide Each type of CareLink report and its components are described in the following sections.
Diabetes Technology Update. Sarah Konigsberg, MD Diabetes & Endocrine Assoc. April 7, 2018
 Diabetes Technology Update Sarah Konigsberg, MD Diabetes & Endocrine Assoc. April 7, 2018 Disclosures None No future technologies are FDA approved Continuous Glucose Monitors Continuous Glucose Monitors
Diabetes Technology Update Sarah Konigsberg, MD Diabetes & Endocrine Assoc. April 7, 2018 Disclosures None No future technologies are FDA approved Continuous Glucose Monitors Continuous Glucose Monitors
Diabetes Management: Current High Tech Innovations
 Diabetes Management: Current High Tech Innovations How Far We ve Come in the Last 40 Years William V. Tamborlane, MD Department of Pediatrics Yale School of Medicine Disclosures I am a consultant for:
Diabetes Management: Current High Tech Innovations How Far We ve Come in the Last 40 Years William V. Tamborlane, MD Department of Pediatrics Yale School of Medicine Disclosures I am a consultant for:
Report Reference Guide
 Report Reference Guide How to use this guide Each type of CareLink report and its components are described in the following sections. Report data used to generate the sample reports was from sample patient
Report Reference Guide How to use this guide Each type of CareLink report and its components are described in the following sections. Report data used to generate the sample reports was from sample patient
Sensor-augmented pump systems provide insulin delivery
 DIABETES TECHNOLOGY & THERAPEUTICS Volume 18, Number 10, 2016 Mary Ann Liebert, Inc. DOI: 10.1089/dia.2016.0216 ORIGINAL ARTICLE Effectiveness of Automated Insulin Management Features of the MiniMed c
DIABETES TECHNOLOGY & THERAPEUTICS Volume 18, Number 10, 2016 Mary Ann Liebert, Inc. DOI: 10.1089/dia.2016.0216 ORIGINAL ARTICLE Effectiveness of Automated Insulin Management Features of the MiniMed c
Advances in Technology in the Treatment of Diabetes Mellitus 2017 How far have we come-how far are we going? Is there a final frontier?
 Advances in Technology in the Treatment of Diabetes Mellitus 2017 How far have we come-how far are we going? Is there a final frontier? Alan B Schorr DO FAAIM FACE www.sugardoc.com abs@sugardoc.com Disclosures
Advances in Technology in the Treatment of Diabetes Mellitus 2017 How far have we come-how far are we going? Is there a final frontier? Alan B Schorr DO FAAIM FACE www.sugardoc.com abs@sugardoc.com Disclosures
Artificial Pancreas Device Systems
 Artificial Pancreas Device Systems Policy Number: 1.01.30 Last Review: 1/2019 Origination: 1/2017 Next Review: 1/2020 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for
Artificial Pancreas Device Systems Policy Number: 1.01.30 Last Review: 1/2019 Origination: 1/2017 Next Review: 1/2020 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for
Medical Policy. MP Artificial Pancreas Device Systems
 Medical Policy MP 1.01.30 BCBSA Ref. Policy: 1.01.30 Last Review: 01/30/2018 Effective Date: 04/01/2018 Section: Durable Medical Equipment Related Policies 1.01.20 Continuous or Intermittent Monitoring
Medical Policy MP 1.01.30 BCBSA Ref. Policy: 1.01.30 Last Review: 01/30/2018 Effective Date: 04/01/2018 Section: Durable Medical Equipment Related Policies 1.01.20 Continuous or Intermittent Monitoring
Threshold-Based Insulin-Pump Interruption for Reduction of Hypoglycemia
 T h e n e w e ngl a nd j o u r na l o f m e dic i n e original article Threshold-Based Insulin-Pump Interruption for Reduction of Hypoglycemia Richard M. Bergenstal, M.D., David C. Klonoff, M.D., Satish
T h e n e w e ngl a nd j o u r na l o f m e dic i n e original article Threshold-Based Insulin-Pump Interruption for Reduction of Hypoglycemia Richard M. Bergenstal, M.D., David C. Klonoff, M.D., Satish
CGM Use in Pregnancy & Unique Populations ELIZABETH O. BUSCHUR, MD THE OHIO STATE UNIVERSITY WEXNER MEDICAL CENTER
 CGM Use in Pregnancy & Unique Populations ELIZABETH O. BUSCHUR, MD THE OHIO STATE UNIVERSITY WEXNER MEDICAL CENTER Case 1: CGM use during pregnancy 29 yo G1P0000 at 10 5/7 weeks gestation presents to set
CGM Use in Pregnancy & Unique Populations ELIZABETH O. BUSCHUR, MD THE OHIO STATE UNIVERSITY WEXNER MEDICAL CENTER Case 1: CGM use during pregnancy 29 yo G1P0000 at 10 5/7 weeks gestation presents to set
THERAPY MANAGEMENT SOFTWARE FOR DIABETES
 THERAPY MANAGEMENT SOFTWARE FOR DIABETES Report Report Interpretation Reference Guide Guide 2007 Medtronic MiniMed. All rights reserved. 6025274-0U2 120707 CareLink Pro Report Reference Guide 0 p.2 Sensor
THERAPY MANAGEMENT SOFTWARE FOR DIABETES Report Report Interpretation Reference Guide Guide 2007 Medtronic MiniMed. All rights reserved. 6025274-0U2 120707 CareLink Pro Report Reference Guide 0 p.2 Sensor
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Artificial Pancreas Device Systems Page 1 of 15 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Artificial Pancreas Device Systems Professional Institutional Original
Artificial Pancreas Device Systems Page 1 of 15 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Artificial Pancreas Device Systems Professional Institutional Original
Welcome to CareLink Pro
 Reference Guide Welcome to CareLink Pro This guide was developed to serve as a reference for obtaining patient data and reviewing CareLink Pro reports. Getting Started with CareLink Pro Adding New Patients
Reference Guide Welcome to CareLink Pro This guide was developed to serve as a reference for obtaining patient data and reviewing CareLink Pro reports. Getting Started with CareLink Pro Adding New Patients
Diabetes Management with Continuous Glucose Monitoring & Multiple Daily Injections. Aaron Michels MD
 Diabetes Management with Continuous Glucose Monitoring & Multiple Daily Injections Aaron Michels MD Outline SMBG & CGM by age group JDRF CGM Trial Sensor Augmented Insulin Pump Therapy for A1c Reduction
Diabetes Management with Continuous Glucose Monitoring & Multiple Daily Injections Aaron Michels MD Outline SMBG & CGM by age group JDRF CGM Trial Sensor Augmented Insulin Pump Therapy for A1c Reduction
Advances Towards the Bionic Pancreas.
 Advances Towards the Bionic Pancreas. Ron Brazg MD, FACE Rainier Clinical Research Center Renton, WA DISCLOSURES Rainier Clinical Research Center is an independent research facility not directly affiliated
Advances Towards the Bionic Pancreas. Ron Brazg MD, FACE Rainier Clinical Research Center Renton, WA DISCLOSURES Rainier Clinical Research Center is an independent research facility not directly affiliated
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Phillip M, Battelino T, Atlas E, et al. Nocturnal glucose control
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Phillip M, Battelino T, Atlas E, et al. Nocturnal glucose control
THERAPY MANAGEMENT SOFTWARE FOR DIABETES
 THERAPY MANAGEMENT SOFTWARE FOR DIABETES Report Report Interpretation Reference Guide Guide 2009 Medtronic MiniMed. All rights reserved. 6025274-012_a CareLink Pro Report Reference Guide 0 p.2 Adherence
THERAPY MANAGEMENT SOFTWARE FOR DIABETES Report Report Interpretation Reference Guide Guide 2009 Medtronic MiniMed. All rights reserved. 6025274-012_a CareLink Pro Report Reference Guide 0 p.2 Adherence
What is the role of insulin pumps in the modern day care of patients with Type 1 diabetes?
 What is the role of insulin pumps in the modern day care of patients with Type 1 diabetes? Dr. Fiona Wotherspoon Consultant in Diabetes and Endocrinology Dorset County Hospital Fiona.Wotherspoon@dchft.nhs.uk
What is the role of insulin pumps in the modern day care of patients with Type 1 diabetes? Dr. Fiona Wotherspoon Consultant in Diabetes and Endocrinology Dorset County Hospital Fiona.Wotherspoon@dchft.nhs.uk
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Artificial Pancreas Device Systems Page 1 of 16 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Artificial Pancreas Device Systems Professional Institutional Original
Artificial Pancreas Device Systems Page 1 of 16 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Artificial Pancreas Device Systems Professional Institutional Original
Control of Glycemic Variability for Reducing Hypoglycemia Jae Hyeon Kim
 Control of Glycemic Variability for Reducing Hypoglycemia Jae Hyeon Kim Division of Endocrinology and Metabolism, Samsung Medical Center, Sungkyunkwan University School of Medicine Conflict of interest
Control of Glycemic Variability for Reducing Hypoglycemia Jae Hyeon Kim Division of Endocrinology and Metabolism, Samsung Medical Center, Sungkyunkwan University School of Medicine Conflict of interest
INSPIRED BY GIVE HIM MORE. The MiniMed 670G system is the world s first insulin delivery system that automatically adapts to your child s needs.
 INSPIRED BY GIVE HIM MORE. The MiniMed 670G system is the world s first insulin delivery system that automatically adapts to your child s needs. * NOW APPROVED FOR AGES 7 AND UP. Waterproof to a depth
INSPIRED BY GIVE HIM MORE. The MiniMed 670G system is the world s first insulin delivery system that automatically adapts to your child s needs. * NOW APPROVED FOR AGES 7 AND UP. Waterproof to a depth
INSPIRED BY GIVE HER MORE. The MiniMed 670G system is the world s first insulin delivery system that automatically adapts to your child s needs.
 INSPIRED BY GIVE HER MORE. The MiniMed 670G system is the world s first insulin delivery system that automatically adapts to your child s needs. * NOW APPROVED FOR AGES 7 AND UP. MORE TIME HERE TO USING
INSPIRED BY GIVE HER MORE. The MiniMed 670G system is the world s first insulin delivery system that automatically adapts to your child s needs. * NOW APPROVED FOR AGES 7 AND UP. MORE TIME HERE TO USING
CGM and Closing The Loop
 CGM and Closing The Loop Dualities Research: Helmsely Charitable Trust, ADA, JDRF, NIDDK Consulting: Abbott Diabetes Care, Roche, Intarcia, Valeritas, Adocia, Big Foot Like With Pumps, We ve Come A Long
CGM and Closing The Loop Dualities Research: Helmsely Charitable Trust, ADA, JDRF, NIDDK Consulting: Abbott Diabetes Care, Roche, Intarcia, Valeritas, Adocia, Big Foot Like With Pumps, We ve Come A Long
Clinical Policy Title: Artificial pancreas device system
 Clinical Policy Title: Artificial pancreas device system Clinical Policy Number: 08.02.07 Effective Date: April 1, 2016 Initial Review Date: November 18, 2015 Most Recent Review Date: February 6, 2018
Clinical Policy Title: Artificial pancreas device system Clinical Policy Number: 08.02.07 Effective Date: April 1, 2016 Initial Review Date: November 18, 2015 Most Recent Review Date: February 6, 2018
VIRTUAL PATIENTS DERIVED FROM THE
 VIRTUAL PATIENTS DERIVED FROM THE CARELINK DATABASE Benyamin Grosman PhD Senior Principle Scientist Medtronic Diabetes WHY VIRTUAL PATIENT MODELING? Reduce the cost of clinical trials Use models in conjunction
VIRTUAL PATIENTS DERIVED FROM THE CARELINK DATABASE Benyamin Grosman PhD Senior Principle Scientist Medtronic Diabetes WHY VIRTUAL PATIENT MODELING? Reduce the cost of clinical trials Use models in conjunction
QUESTION 4. WHAT CLINICAL DATA ARE CURRENTLY AVAILABLE TO SUPPORT EXPANDED CGM COVERAGE BY PAYERS AS PERTAINS TO QUESTIONS 1 AND 3?
 500 1 QUESTION 4. WHAT CLINICAL DATA ARE CURRENTLY AVAILABLE TO SUPPORT EXPANDED CGM COVERAGE BY PAYERS AS PERTAINS TO QUESTIONS 1 AND 3? WHAT ADDITIONAL DATA ARE NEEDED? AACE/ACE CGM Consensus Conference:
500 1 QUESTION 4. WHAT CLINICAL DATA ARE CURRENTLY AVAILABLE TO SUPPORT EXPANDED CGM COVERAGE BY PAYERS AS PERTAINS TO QUESTIONS 1 AND 3? WHAT ADDITIONAL DATA ARE NEEDED? AACE/ACE CGM Consensus Conference:
Understanding the CareLinkTM Sensor Meter Overview Report: Page 1
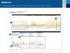 Understanding the CareLinkTM Sensor Meter Overview Report: Page 1 Sensor & Meter Overview (1 of 3) 4-Hour Glucose Sensor Overlay - Readings & Averages (mg/dl) 1 Glucose Sensor Overlay Bedtime to Wake-Up
Understanding the CareLinkTM Sensor Meter Overview Report: Page 1 Sensor & Meter Overview (1 of 3) 4-Hour Glucose Sensor Overlay - Readings & Averages (mg/dl) 1 Glucose Sensor Overlay Bedtime to Wake-Up
Endocrine Update Mary T. Korytkowski MD Division of Endocrinology University of Pittsburgh
 Endocrine Update 2016 Mary T. Korytkowski MD Division of Endocrinology University of Pittsburgh Disclosure of Financial Relationships Mary Korytkowski MD Honoraria British Medical Journal Diabetes Research
Endocrine Update 2016 Mary T. Korytkowski MD Division of Endocrinology University of Pittsburgh Disclosure of Financial Relationships Mary Korytkowski MD Honoraria British Medical Journal Diabetes Research
Norbert Hermanns, PhD 1,2, Beatrix Schumann, MD 2, Bernhard Kulzer, PhD 1,2, and Thomas Haak, MD 1,2. Original Article
 524105DSTXXX10.1177/1932296814524105Journal of Diabetes Science and TechnologyHermanns et al research-article2014 Original Article The Impact of Continuous Glucose Monitoring on Low Interstitial Glucose
524105DSTXXX10.1177/1932296814524105Journal of Diabetes Science and TechnologyHermanns et al research-article2014 Original Article The Impact of Continuous Glucose Monitoring on Low Interstitial Glucose
REPORT INTERPRETATION
 REPORT INTERPRETATION: Interpreting ipro Professional Continuous Glucose Monitoring (CGM) Reports and Making Therapy Adjustments TARGET AUDIENCE The audience for this section is physicians, mid-level practitioners,
REPORT INTERPRETATION: Interpreting ipro Professional Continuous Glucose Monitoring (CGM) Reports and Making Therapy Adjustments TARGET AUDIENCE The audience for this section is physicians, mid-level practitioners,
Continuous Glucose Monitoring System
 Continuous Glucose Monitoring System Policy Number: Original Effective Date: MM.02.003 03/13/2001 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 04/01/2016 Section: DME Place(s)
Continuous Glucose Monitoring System Policy Number: Original Effective Date: MM.02.003 03/13/2001 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 04/01/2016 Section: DME Place(s)
WOULD YOU LIKE MORE CONFIDENCE IN YOUR HYPOGLYCAEMIC CONTROL? SMARTGUARD. PROVIDES PREDICTIVE PROTECTION
 WOULD YOU LIKE MORE CONFIDENCE IN YOUR HYPOGLYCAEMIC CONTROL? SMARTGUARD. PROVIDES PREDICTIVE PROTECTION 1,2 MINIMED 640G WITH SMARTGUARD HAVE YOU CONSIDERED THE REAL IMPACT HYPOGLYCAEMIA HAS ON YOUR LIFE?
WOULD YOU LIKE MORE CONFIDENCE IN YOUR HYPOGLYCAEMIC CONTROL? SMARTGUARD. PROVIDES PREDICTIVE PROTECTION 1,2 MINIMED 640G WITH SMARTGUARD HAVE YOU CONSIDERED THE REAL IMPACT HYPOGLYCAEMIA HAS ON YOUR LIFE?
Preventing Hypoglycemia Using Predictive Alarm Algorithms and Insulin Pump Suspension
 DIABETES TECHNOLOGY & THERAPEUTICS Volume 11, Number 2, 29 Mary Ann Liebert, Inc. DOI: 1.189/dia.28.32 Preventing Hypoglycemia Using Predictive Alarm Algorithms and Insulin Pump Suspension Bruce Buckingham,
DIABETES TECHNOLOGY & THERAPEUTICS Volume 11, Number 2, 29 Mary Ann Liebert, Inc. DOI: 1.189/dia.28.32 Preventing Hypoglycemia Using Predictive Alarm Algorithms and Insulin Pump Suspension Bruce Buckingham,
DISCOVER THE POWER OF CONNECTION MINIMED 640G
 DISCOVER THE POWER OF CONNECTION MINIMED 640G INSULIN PUMP THERAPY CHANGING LIVES TODAY Has your child just been diagnosed with insulin dependent diabetes? Or perhaps they ve been on multiple daily injection
DISCOVER THE POWER OF CONNECTION MINIMED 640G INSULIN PUMP THERAPY CHANGING LIVES TODAY Has your child just been diagnosed with insulin dependent diabetes? Or perhaps they ve been on multiple daily injection
Early Patient and Clinician Experiences with Continuous Glucose Monitoring
 Feature Article / Early Experiences with CGM Early Patient and Clinician Experiences with Continuous Glucose Monitoring David K. Bloomgarden, MD, FACE; Janine Freeman, RD, LD, CDE; and Elizabeth DeRobertis,
Feature Article / Early Experiences with CGM Early Patient and Clinician Experiences with Continuous Glucose Monitoring David K. Bloomgarden, MD, FACE; Janine Freeman, RD, LD, CDE; and Elizabeth DeRobertis,
Diabetes II Insulin pumps; Continuous glucose monitoring system (CGMS) Ernest Asamoah, MD FACE FACP FRCP (Lond)
 Diabetes II Insulin pumps; Continuous glucose monitoring system (CGMS) Ernest Asamoah, MD FACE FACP FRCP (Lond) 9501366-011 20110401 Objectives Understand the need for insulin pumps and CGMS in managing
Diabetes II Insulin pumps; Continuous glucose monitoring system (CGMS) Ernest Asamoah, MD FACE FACP FRCP (Lond) 9501366-011 20110401 Objectives Understand the need for insulin pumps and CGMS in managing
Continuous Glucose Monitoring System
 Continuous Glucose Monitoring System Policy Number: Original Effective Date: MM.02.003 03/13/2001 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 4/1/2018 Section: DME Place(s)
Continuous Glucose Monitoring System Policy Number: Original Effective Date: MM.02.003 03/13/2001 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 4/1/2018 Section: DME Place(s)
Continuous Glucose Monitoring System
 Continuous Glucose Monitoring System Policy Number: Original Effective Date: MM.02.003 03/13/2001 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 04/01/2017 Section: DME Place(s)
Continuous Glucose Monitoring System Policy Number: Original Effective Date: MM.02.003 03/13/2001 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 04/01/2017 Section: DME Place(s)
Medical Education. Personal Continuous Glucose Monitoring Protocol. Innovating for life.
 Personal Continuous Glucose Monitoring Protocol A Guide to Continuous Glucose Monitoring Integrated with Insulin Pump Therapy Includes Alert and Trend Management as well as Threshold Medical Education
Personal Continuous Glucose Monitoring Protocol A Guide to Continuous Glucose Monitoring Integrated with Insulin Pump Therapy Includes Alert and Trend Management as well as Threshold Medical Education
Inpatient Studies of a Kalman-Filter-Based Predictive Pump Shutoff Algorithm
 Journal of Diabetes Science and Technology Volume 6, Issue 5, September 2012 Diabetes Technology Society ORIGINAL ARTICLE Inpatient Studies of a Kalman-Filter-Based Predictive Pump Shutoff Algorithm Fraser,
Journal of Diabetes Science and Technology Volume 6, Issue 5, September 2012 Diabetes Technology Society ORIGINAL ARTICLE Inpatient Studies of a Kalman-Filter-Based Predictive Pump Shutoff Algorithm Fraser,
RELEASED. Clearing your active insulin
 To clear all your settings: 1. Make sure the pump is not connected to your body. 2. Go to the Manage Settings screen. Menu > Utilities > Manage Settings 3. Simultaneously press and hold and until the Manage
To clear all your settings: 1. Make sure the pump is not connected to your body. 2. Go to the Manage Settings screen. Menu > Utilities > Manage Settings 3. Simultaneously press and hold and until the Manage
Female- n (%) 494 (54) White Non-Hispanic 840 (91) Black Non-Hispanic 31 (3) Hispanic 27 (3) Other 21 (2) Age- mean±sd (range) 37.2±18.
 Supplementary Table 1. Participant Characteristics Total- n 919 Female- n (%) 494 (54) Race- n (%) White Non-Hispanic 840 (91) Black Non-Hispanic 31 (3) Hispanic 27 (3) Other 21 (2) Age- mean±sd (range)
Supplementary Table 1. Participant Characteristics Total- n 919 Female- n (%) 494 (54) Race- n (%) White Non-Hispanic 840 (91) Black Non-Hispanic 31 (3) Hispanic 27 (3) Other 21 (2) Age- mean±sd (range)
RELEASED. first steps. Icon Icon name What it means
 Icon Icon name What it means Connection The connection icon appears green when the Sensor feature is on and your transmitter is successfully communicating with your pump. The connection icon appears gray
Icon Icon name What it means Connection The connection icon appears green when the Sensor feature is on and your transmitter is successfully communicating with your pump. The connection icon appears gray
The Impact of Accelerometer Use in Exercise-Associated Hypoglycemia Prevention in Type 1 Diabetes
 551045DSTXXX10.1177/1932296814551045Journal of Diabetes Science and TechnologyStenerson et al research-article2014 Original Article The Impact of Accelerometer Use in Exercise-Associated Hypoglycemia Prevention
551045DSTXXX10.1177/1932296814551045Journal of Diabetes Science and TechnologyStenerson et al research-article2014 Original Article The Impact of Accelerometer Use in Exercise-Associated Hypoglycemia Prevention
NEW TECHNOLOGIES FOR MANAGING DIABETES ANGELA THOMPSON DNP, FNP-C, BC-ADM, CDE, FAANP
 NEW TECHNOLOGIES FOR MANAGING DIABETES ANGELA THOMPSON DNP, FNP-C, BC-ADM, CDE, FAANP No commercial support or sponsorship was received for this project I have nothing to disclose OBJECTIVES Identify at
NEW TECHNOLOGIES FOR MANAGING DIABETES ANGELA THOMPSON DNP, FNP-C, BC-ADM, CDE, FAANP No commercial support or sponsorship was received for this project I have nothing to disclose OBJECTIVES Identify at
Today s Goals 10/6/2017. New Frontiers in Diabetes Technology. Disclosures
 New Frontiers in Diabetes Technology Marie E. McDonnell, MD Director, Brigham and Women's Diabetes Program Division of Endocrinology, Diabetes and Hypertension Brigham and Women s Hospital Today s Goals
New Frontiers in Diabetes Technology Marie E. McDonnell, MD Director, Brigham and Women's Diabetes Program Division of Endocrinology, Diabetes and Hypertension Brigham and Women s Hospital Today s Goals
Subject Index. Breastfeeding, self-monitoring of blood glucose 56
 Subject Index Animas Vibe 86, 130 Artificial pancreas clinical studies inpatient studies 175 180 outpatient studies outcome assessment 182, 183 technology 180, 181 telemedicine 182 components glucose sensor
Subject Index Animas Vibe 86, 130 Artificial pancreas clinical studies inpatient studies 175 180 outpatient studies outcome assessment 182, 183 technology 180, 181 telemedicine 182 components glucose sensor
Insulin Delivery and Glucose Monitoring Methods for Diabetes Mellitus: Comparative Effectiveness
 Insulin Delivery and Glucose Monitoring Methods for Diabetes Mellitus: Comparative Effectiveness Prepared for: Agency for Healthcare Research and Quality (AHRQ) www.ahrq.gov Outline of Material Introduction
Insulin Delivery and Glucose Monitoring Methods for Diabetes Mellitus: Comparative Effectiveness Prepared for: Agency for Healthcare Research and Quality (AHRQ) www.ahrq.gov Outline of Material Introduction
PATIENT TRAINING CHECKLIST MINIMED 670G SYSTEM
 Technical Training Insulin Pump Infusion Set: Insulin Pump Model: BASIC FEATURES Patient has demonstrated understanding of: Button Functions Home Screen Menu review Status Screens Battery type/insertion
Technical Training Insulin Pump Infusion Set: Insulin Pump Model: BASIC FEATURES Patient has demonstrated understanding of: Button Functions Home Screen Menu review Status Screens Battery type/insertion
Continuous Glucose Monitoring (CGM)
 Continuous Glucose Monitoring (CGM) Date of Origin: 02/2001 Last Review Date: 07/26/2017 Effective Date: 07/26/2017 Dates Reviewed: 04/2004, 04/2005, 03/2006, 11/2006, 12/2007, 03/2008, 09/2008, 04/2009,
Continuous Glucose Monitoring (CGM) Date of Origin: 02/2001 Last Review Date: 07/26/2017 Effective Date: 07/26/2017 Dates Reviewed: 04/2004, 04/2005, 03/2006, 11/2006, 12/2007, 03/2008, 09/2008, 04/2009,
Insulin Pump Therapy in children. Prof. Abdulmoein Al-Agha, FRCPCH(UK)
 Insulin Pump Therapy in children Prof. Abdulmoein Al-Agha, FRCPCH(UK) aagha@kau.edu.sa Highlights Evolution of insulin pump Pumps mimics Pancreas Goals of diabetes care What lowers HbA1c Criteria for selection
Insulin Pump Therapy in children Prof. Abdulmoein Al-Agha, FRCPCH(UK) aagha@kau.edu.sa Highlights Evolution of insulin pump Pumps mimics Pancreas Goals of diabetes care What lowers HbA1c Criteria for selection
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Stewart ZA, Wilinska ME, Hartnell S, et al. Closed-loop insulin
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Stewart ZA, Wilinska ME, Hartnell S, et al. Closed-loop insulin
GIVE YOUR PATIENTS CERTAINTY
 GIVE CERTAINTY Pictured: The Renouf brothers all live with T1D TECH UPGRADE CGM SUBSCRIPTION INTRODUCING THE TECH UPGRADE CGM SUBSCRIPTION FROM MEDTRONIC HOW SMARTGUARD TM WORKS In Australia people generally
GIVE CERTAINTY Pictured: The Renouf brothers all live with T1D TECH UPGRADE CGM SUBSCRIPTION INTRODUCING THE TECH UPGRADE CGM SUBSCRIPTION FROM MEDTRONIC HOW SMARTGUARD TM WORKS In Australia people generally
Understanding the Assessment and Progress Report
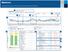 Understanding the ssessment and Progress Report ssessment and Progress 8//2-8/7/2 (7 Days) 3 7/2/2-7/27/2 (7 Days) 4 25-75% -% verage 2 4: M- 5:4 M Hyperglycemic patterns() :45 PM- :3 PM 2 3 :2 M- :5 M
Understanding the ssessment and Progress Report ssessment and Progress 8//2-8/7/2 (7 Days) 3 7/2/2-7/27/2 (7 Days) 4 25-75% -% verage 2 4: M- 5:4 M Hyperglycemic patterns() :45 PM- :3 PM 2 3 :2 M- :5 M
Continuous or Intermittent Glucose Monitoring in Interstitial Fluid
 Continuous or Intermittent Glucose Monitoring in Interstitial Fluid Policy Number: 1.01.20 Last Review: 9/2014 Origination: 11/2001 Next Review: 9/2015 Policy Blue Cross and Blue Shield of Kansas City
Continuous or Intermittent Glucose Monitoring in Interstitial Fluid Policy Number: 1.01.20 Last Review: 9/2014 Origination: 11/2001 Next Review: 9/2015 Policy Blue Cross and Blue Shield of Kansas City
THE MINIMED 670G SYSTEM SCHOOL NURSE GUIDE
 THE MINIMED 670G SYSTEM SCHOOL NURSE GUIDE Indicated for type 1 patients 14 and over. Prescription required. WARNING: Medtronic performed an evaluation of the MiniMed 670G system and determined that it
THE MINIMED 670G SYSTEM SCHOOL NURSE GUIDE Indicated for type 1 patients 14 and over. Prescription required. WARNING: Medtronic performed an evaluation of the MiniMed 670G system and determined that it
Insulin Pumps and Continuous Glucose Sensors- Embracing Technology. Susan Cavalier, BS, RN, CDE Manager, Diabetes Educator Sanford Diabetes Education
 Insulin Pumps and Continuous Glucose Sensors- Embracing Technology Susan Cavalier, BS, RN, CDE Manager, Diabetes Educator Sanford Diabetes Education Diabetes Management Tools in 1974 Insulin pump history
Insulin Pumps and Continuous Glucose Sensors- Embracing Technology Susan Cavalier, BS, RN, CDE Manager, Diabetes Educator Sanford Diabetes Education Diabetes Management Tools in 1974 Insulin pump history
DIABETES & ENDOCRINE DIABETES TECHNOLOGY: HOW TO STAY CURRENT WITH ONGOING TECHNOLOGY ADVANCEMENT
 DIABETES TECHNOLOGY: HOW TO STAY CURRENT WITH ONGOING TECHNOLOGY ADVANCEMENT Min-Jye Chen, MD Endocrinology Diabetes Management of the School-Aged Child Provided by Texas Children s Hospital Provider #18-267764-A
DIABETES TECHNOLOGY: HOW TO STAY CURRENT WITH ONGOING TECHNOLOGY ADVANCEMENT Min-Jye Chen, MD Endocrinology Diabetes Management of the School-Aged Child Provided by Texas Children s Hospital Provider #18-267764-A
CARELINK PERSONAL ACTIONABLE INSIGHTS FOR BETTER DIABETES MANAGEMENT CARELINK REPORTS GUIDE
 CARELINK PERSONAL ACTIONABLE INSIGHTS FOR BETTER DIABETES MANAGEMENT CARELINK REPORTS GUIDE CARELINK PERSONAL ACTIONABLE INSIGHTS FOR BETTER DIABETES MANAGEMENT As part of your diabetes therapy you have
CARELINK PERSONAL ACTIONABLE INSIGHTS FOR BETTER DIABETES MANAGEMENT CARELINK REPORTS GUIDE CARELINK PERSONAL ACTIONABLE INSIGHTS FOR BETTER DIABETES MANAGEMENT As part of your diabetes therapy you have
King s Research Portal
 King s Research Portal DOI: 10.1089/dia.2015.0324 Document Version Publisher's PDF, also known as Version of record Link to publication record in King's Research Portal Citation for published version (APA):
King s Research Portal DOI: 10.1089/dia.2015.0324 Document Version Publisher's PDF, also known as Version of record Link to publication record in King's Research Portal Citation for published version (APA):
Paolo Di Bartolo U.O di Diabetologia Dip. Malattie Digestive & Metaboliche AULS Prov. di Ravenna. Ipoglicemie e Monitoraggio Glicemico
 Paolo Di Bartolo U.O di Diabetologia Dip. Malattie Digestive & Metaboliche AULS Prov. di Ravenna Ipoglicemie e Monitoraggio Glicemico Management of Hypoglycaemia.if hypoglycemia is a problem, the principles
Paolo Di Bartolo U.O di Diabetologia Dip. Malattie Digestive & Metaboliche AULS Prov. di Ravenna Ipoglicemie e Monitoraggio Glicemico Management of Hypoglycaemia.if hypoglycemia is a problem, the principles
Diagnostics guidance Published: 12 February 2016 nice.org.uk/guidance/dg21
 Integrated sensor-augmented pump therapy systems for managing blood glucose levels els in type 1 diabetes (the MiniMed Paradigm adigm Veo system and the Vibe and G4 PLATINUM CGM system) Diagnostics guidance
Integrated sensor-augmented pump therapy systems for managing blood glucose levels els in type 1 diabetes (the MiniMed Paradigm adigm Veo system and the Vibe and G4 PLATINUM CGM system) Diagnostics guidance
Original Article. Nicholas B. Argento, MD 1 ; Katherine Nakamura, PhD 2 ABSTRACT
 Original Article Nicholas B. Argento, MD 1 ; Katherine Nakamura, PhD 2 ABSTRACT Objective: Little information is available on personal real-time continuous glucose monitoring (PCGM) in patients 65 years
Original Article Nicholas B. Argento, MD 1 ; Katherine Nakamura, PhD 2 ABSTRACT Objective: Little information is available on personal real-time continuous glucose monitoring (PCGM) in patients 65 years
Insulin Pumps - External
 Insulin Pumps - External Policy Number: Original Effective Date: MM.01.004 04/01/2011 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 04/01/20174/1/2018 Section: DME Place(s) of
Insulin Pumps - External Policy Number: Original Effective Date: MM.01.004 04/01/2011 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 04/01/20174/1/2018 Section: DME Place(s) of
How to Transfer Your Settings
 How to Transfer Your Settings for your Replacement MiniMed 530G System Let s get started! Step 1: Before you send your original pump back, copy your current settings onto the Insulin Pump Settings Form.
How to Transfer Your Settings for your Replacement MiniMed 530G System Let s get started! Step 1: Before you send your original pump back, copy your current settings onto the Insulin Pump Settings Form.
Diabetes Devices Workshop Angela Aldrich, PharmD, PhC April Mott, PharmD, PhC, BCPS Presbyterian Medical Group 28 January 2018
 Diabetes Devices Workshop Angela Aldrich, PharmD, PhC April Mott, PharmD, PhC, BCPS Presbyterian Medical Group 28 January 2018 Pumps & Sensors & Meters, Oh My! A Tale of Two Meters Technology for glucometers
Diabetes Devices Workshop Angela Aldrich, PharmD, PhC April Mott, PharmD, PhC, BCPS Presbyterian Medical Group 28 January 2018 Pumps & Sensors & Meters, Oh My! A Tale of Two Meters Technology for glucometers
06/13/17. A. Completed a comprehensive diabetes education program within the past two years; and
 Reference #: MC/L011 Page 1 of 4 PRODUCT APPLICATION: PreferredOne Community Health Plan (PCHP) PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services, Inc. (PAS) Non-ERISA
Reference #: MC/L011 Page 1 of 4 PRODUCT APPLICATION: PreferredOne Community Health Plan (PCHP) PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services, Inc. (PAS) Non-ERISA
State of California Health and Human Services Agency Department of Health Care Services
 State of California Health and Human Services Agency Department of Health Care Services JENNIFER KENT Director EDMOND G. BROWN JR Governor DATE: N.L.: 03-0317 Index: Benefits TO: ALL COUNTY CALIFORNIA
State of California Health and Human Services Agency Department of Health Care Services JENNIFER KENT Director EDMOND G. BROWN JR Governor DATE: N.L.: 03-0317 Index: Benefits TO: ALL COUNTY CALIFORNIA
Understanding the CareLink TM Therapy Management Dashboard Report
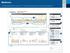 3 2 1 5 6 7 4 1 This is a great place to start recognizing trends. In this example you may see plenty of events that result in a low glucose trend. A low glucose is identified as a value less than 70mg/dL.
3 2 1 5 6 7 4 1 This is a great place to start recognizing trends. In this example you may see plenty of events that result in a low glucose trend. A low glucose is identified as a value less than 70mg/dL.
DISCOVER THE POWER OF CONNECTION MINIMED 640G
 DISCOVER THE POWER OF CONNECTION MINIMED 640G INSULIN PUMP THERAPY CHANGING LIVES TODAY Have you just been diagnosed with insulin dependent diabetes? Perhaps you ve been on multiple daily injection therapy
DISCOVER THE POWER OF CONNECTION MINIMED 640G INSULIN PUMP THERAPY CHANGING LIVES TODAY Have you just been diagnosed with insulin dependent diabetes? Perhaps you ve been on multiple daily injection therapy
USING THE MINIMED 670G SYSTEM
 USING THE MINIMED 670G SYSTEM Instructions for the Experienced MiniMed 630G System User USING THE MINIMED 670G SYSTEM: INSTRUCTIONS FOR THE EXPERIENCED USER Checklist for starting on your MiniMed 670G
USING THE MINIMED 670G SYSTEM Instructions for the Experienced MiniMed 630G System User USING THE MINIMED 670G SYSTEM: INSTRUCTIONS FOR THE EXPERIENCED USER Checklist for starting on your MiniMed 670G
USING THE MINIMED 670G SYSTEM. Instructions for the Experienced MiniMed 630G System User
 USING THE MINIMED 670G SYSTEM Instructions for the Experienced MiniMed 630G System User This is a supplemental guide. For complete instructions and safety information, please see your MiniMed 670G System
USING THE MINIMED 670G SYSTEM Instructions for the Experienced MiniMed 630G System User This is a supplemental guide. For complete instructions and safety information, please see your MiniMed 670G System
Supplementary Data. Formula S1. Calculation of IG fluctuation from continuous glucose monitoring profiles Z T 1 T
 Supplementary Data Formula S1. Calculation of IG fluctuation from continuous glucose monitoring profiles 1 T Z T O jig(t) IGjdt IG, interstitial glucose; IG(t), IG value at time t; IG, mean IG from the
Supplementary Data Formula S1. Calculation of IG fluctuation from continuous glucose monitoring profiles 1 T Z T O jig(t) IGjdt IG, interstitial glucose; IG(t), IG value at time t; IG, mean IG from the
Diabetes Technology Continuous Subcutaneous Insulin Infusion Therapy And Continuous Glucose Monitoring In Adults: An Endocrine Society Clinical
 Diabetes Technology Continuous Subcutaneous Insulin Infusion Therapy And Continuous Glucose Monitoring In Adults: An Endocrine Society Clinical Practice Guideline Task Force Members Anne Peters, MD (Chair)
Diabetes Technology Continuous Subcutaneous Insulin Infusion Therapy And Continuous Glucose Monitoring In Adults: An Endocrine Society Clinical Practice Guideline Task Force Members Anne Peters, MD (Chair)
Policy for Continuous Glucose Monitoring for Type 1 Diabetic Paediatric Patients (<18 years of age)
 Policy for Continuous Glucose Monitoring for Type 1 Diabetic Paediatric Patients (
Policy for Continuous Glucose Monitoring for Type 1 Diabetic Paediatric Patients (
Continuous Glucose Monitoring
 Continuous Glucose Monitoring What is Continuous Glucose Monitoring? Blood glucose meters measure glucose in your blood and glucose sensors measure glucose levels in the fluid around the cells They are
Continuous Glucose Monitoring What is Continuous Glucose Monitoring? Blood glucose meters measure glucose in your blood and glucose sensors measure glucose levels in the fluid around the cells They are
MEDICAL POLICY Continuous Glucose Monitoring Systems and Insulin Pumps
 POLICY: PG0177 ORIGINAL EFFECTIVE: 12/01/08 LAST REVIEW: 02/13/18 MEDICAL POLICY Continuous Glucose Monitoring Systems and Insulin Pumps GUIDELINES This policy does not certify benefits or authorization
POLICY: PG0177 ORIGINAL EFFECTIVE: 12/01/08 LAST REVIEW: 02/13/18 MEDICAL POLICY Continuous Glucose Monitoring Systems and Insulin Pumps GUIDELINES This policy does not certify benefits or authorization
Effective Health Care Program
 Comparative Effectiveness Review Number 57 Effective Health Care Program Methods for Insulin Delivery and Glucose Monitoring: Comparative Effectiveness Executive Summary Background Diabetes mellitus is
Comparative Effectiveness Review Number 57 Effective Health Care Program Methods for Insulin Delivery and Glucose Monitoring: Comparative Effectiveness Executive Summary Background Diabetes mellitus is
Diabetes Medical Management Plan (DMMP)
 Diabetes Medical Management Plan (DMMP) This plan should be completed by the student s personal diabetes health care team, including the parents/guardians. It should be reviewed with relevant school staff
Diabetes Medical Management Plan (DMMP) This plan should be completed by the student s personal diabetes health care team, including the parents/guardians. It should be reviewed with relevant school staff
CURRICULUM VITAE. School of Medicine Cleveland, Ohio; MD, 1970
 CURRICULUM VITAE Robert M. Bernstein, MD, FACE Regional Endocrinology Associates, PC Southwest Clinical Research Center 1533 St. Francis Drive, Santa Fe, New Mexico 87505 Phone: (505) 982-2860 Fax: (505)
CURRICULUM VITAE Robert M. Bernstein, MD, FACE Regional Endocrinology Associates, PC Southwest Clinical Research Center 1533 St. Francis Drive, Santa Fe, New Mexico 87505 Phone: (505) 982-2860 Fax: (505)
10:20 AM March 5, B 100% 235 u. 124 mg/dl 3 HRS INSULIN ON BOARD:
 DEXCOM G5 MOBILE CGM COMPATIBLE B 100% 235 u INSULIN ON BOARD: OPTIONS 10:20 AM March 5, 2017 400 350 300 250 200 150 100 50 1.1 u 1:09 hrs BOLUS 124 mg/dl 3 HRS The pump that gets updated, not outdated.
DEXCOM G5 MOBILE CGM COMPATIBLE B 100% 235 u INSULIN ON BOARD: OPTIONS 10:20 AM March 5, 2017 400 350 300 250 200 150 100 50 1.1 u 1:09 hrs BOLUS 124 mg/dl 3 HRS The pump that gets updated, not outdated.
31 days Wed Nov 15, Fri Dec 15, 2017 N/A. Hypoglycemia. Sensor usage (CGM) Mahlon's glucose data was in the target range about 77% of the day.
 Mahlon Lovett Overview Report Generated at: Thu, Feb 8, 2018 12:42 PM EST 137 26 N/A 18% 75% 7% Days with CGM data Avg. calibrations per day 3% 1 / 31 0.0 0% Average glucose Standard Hypoglycemia Time
Mahlon Lovett Overview Report Generated at: Thu, Feb 8, 2018 12:42 PM EST 137 26 N/A 18% 75% 7% Days with CGM data Avg. calibrations per day 3% 1 / 31 0.0 0% Average glucose Standard Hypoglycemia Time
Pumps & Sensors made easy. OPADA ALZOHAILI MD FACE Endocrinology Assistant Professor Wayne State University
 Pumps & Sensors made easy OPADA ALZOHAILI MD FACE Endocrinology Assistant Professor Wayne State University DeFronzo RA. Diabetes. 2009;58:773-795. Ominous Octet Relationship of b-cell Dysfunction and Development
Pumps & Sensors made easy OPADA ALZOHAILI MD FACE Endocrinology Assistant Professor Wayne State University DeFronzo RA. Diabetes. 2009;58:773-795. Ominous Octet Relationship of b-cell Dysfunction and Development
Artificial Pancreas, shortly close to home? BC Pediatric Diabetes Day, Feb 26, 2016
 Artificial Pancreas, shortly close to home? BC Pediatric Diabetes Day, Feb 26, 2016 Rémi Rabasa- Lhoret, M.D. (Endocrinology), Ph.D. CSPQ Director Pla:orm for Research in Obesity, Metabolism and Diabetes
Artificial Pancreas, shortly close to home? BC Pediatric Diabetes Day, Feb 26, 2016 Rémi Rabasa- Lhoret, M.D. (Endocrinology), Ph.D. CSPQ Director Pla:orm for Research in Obesity, Metabolism and Diabetes
DIABETES TECHNOLOGY: WHERE ARE WE NOW AND WHERE ARE WE GOING? Presented by: Tom Brobson
 DIABETES TECHNOLOGY: WHERE ARE WE NOW AND WHERE ARE WE GOING? Presented by: Tom Brobson March 3 rd, 2018 Who is this guy? Accelerating Progress 1 Oh that guy! Accelerating Progress 2 STATE OF T1D CARE
DIABETES TECHNOLOGY: WHERE ARE WE NOW AND WHERE ARE WE GOING? Presented by: Tom Brobson March 3 rd, 2018 Who is this guy? Accelerating Progress 1 Oh that guy! Accelerating Progress 2 STATE OF T1D CARE
Dexcom Current and Future Technology Innovations. Jorge Valdes Chief Technical Officer
 Dexcom Current and Future Technology Innovations 2015 Jorge Valdes Chief Technical Officer G5 App Sensor (Same as G4) Indicated for adults and children down to Age 2 years Tiny insertion needle (26Ga)
Dexcom Current and Future Technology Innovations 2015 Jorge Valdes Chief Technical Officer G5 App Sensor (Same as G4) Indicated for adults and children down to Age 2 years Tiny insertion needle (26Ga)
