Acid Base Balance. Chapter 26 Balance. ph Imbalances. Acid Base Balance. CO 2 and ph. Carbonic Acid. Part 2. Acid/Base Balance
|
|
|
- Maximillian Hines
- 6 years ago
- Views:
Transcription
1 Acid Base Balance Chapter 26 Balance Part 2. Acid/Base Balance Precisely balances production and loss of hydrogen ions (ph) The body generates acids during normal metabolism, tends to reduce ph Kidneys: Secrete hydrogen ions into urine Generate buffers that enter bloodstream in distal segments of distal convoluted tubule (DCT) and collecting system Lungs: affect ph balance through elimination of carbon dioxide Acid Base Balance phof body fluids is altered by the introduction of acids or bases Acids and bases may be strong or weak Strong acids dissociate completely (only HCl is relevant physiologically) Weak acids do not dissociate completely and thus affect the ph less (e.g. carbonic acid) ph Imbalances Acidosis: physiological state resulting from abnormally low plasma ph Alkalosis: physiological state resulting from abnormally high plasma ph Both are dangerous but acidosis is more common because normal cellular activities generate acids Why is ph so important? Carbonic Acid Carbon Dioxide in solution in peripheral tissues interacts with water to form carbonic acid Carbonic Anhydrase (CA) catalyzes dissociation of carbonic acid into H+ and HCO 3 - Found in: cytoplasm of red blood cells liver and kidney cells parietal cells of stomach many other cells CO 2 and ph Most CO 2 in solution converts to carbonic acid and most carbonic acid dissociates (but not all because it is a weak acid) P CO2 is the most important factor affecting ph in body tissues P CO2 and ph are inversely related Loss of CO 2 at the lungs increases blood ph 1
2 Hydrogen Ions (H + ) Are gained: at digestive tract through cellular metabolic activities Are eliminated: at kidneys and in urine at lungs (as CO 2 + H 2 O) must be neutralized in blood and urine to avoid tissue damage Acids produced in normal metabolic activity are temporarily neutralized by buffers in body fluids Buffers Dissolved compounds that stabilize ph by providing or removing H + Weak acids or weak bases that absorb or release H + are buffers Buffer Systems Buffer Systems in Body Fluids Buffer System: consists of a combination of a weak acid and the anion released by its dissociation (its conjugate base) The anion functions as a weak base: H 2 CO 3 (acid) H + + HCO 3 - (base) In solution, molecules of weak acid exist in equilibrium with its dissociation products (meaning you have all three around in plasma) Figure Major Buffer Systems 1. Protein buffer systems: help regulate ph in ECF and ICF interact extensively with other buffer systems 2. Carbonic acid bicarbonate buffer system: most important in ECF 3. Phosphate buffer system: buffers ph of ICF and urine Protein Buffer Systems Depend on free and terminal amino acids Respond to ph changes by accepting or releasing H + If ph rises: carboxyl group of amino acid dissociates, acting as weak acid, releasing a hydrogen ion If ph drops: carboxylate ion and amino group act as weak bases accept H + form carboxyl group and amino ion Proteins that contribute to buffering capabilities: plasma proteins proteins in interstitial fluid proteins in ICF 2
3 Amino Acids in Protein Buffer Systems The Hemoglobin Buffer System CO 2 diffuses across RBC membrane: no transport mechanism required As carbonic acid dissociates: bicarbonate ions diffuse into plasma in exchange for chloride ions (chloride shift) Hydrogen ions are buffered by hemoglobin molecules the only intracellular buffer system with an immediate effect on ECF ph Helps prevent major changes in ph when plasma P CO2 is rising or falling Figure 27 8 The Carbonic Acid Bicarbonate Buffer System Formed by carbonic acid and its dissociation products Prevents changes in ph caused by organic acids and fixed acids in ECF H+ generated by acid production combines with bicarbonate in the plasma This forms carbonic acid, which dissociates into CO 2 which is breathed out The Carbonic Acid Bicarbonate Buffer System Figure 27 9 Limitations of the Carbonic Acid Buffer System 1. Cannot protect ECF from changes in ph that result from elevated or depressed levels of CO 2 (because CO 2 is part of it) 2. Functions only when respiratory system and respiratory control centers are working normally 3. Ability to buffer acids is limited by availability of bicarbonate ions The Phosphate Buffer System Consists of anion H 2 PO 4 (a weak acid) Works like the carbonic acid bicarbonate buffer system Is important in buffering ph of ICF 3
4 Problems with Buffer Systems Provide only temporary solution to acid base imbalance Do not eliminate H + ions Supply of buffer molecules is limited Maintenance of Acid Base Balance Requires balancing H + gains and losses For homeostasis to be preserved, captured H + must either be: permanently tied up in water molecules through CO 2 removal at lungs OR removed from body fluids through secretion at kidney Thus, problems with either of these organs cause problems with acid/base balance Coordinates actions of buffer systems with: respiratory mechanisms renal mechanisms Respiratory Compensation Is a change in respiratory rate that helps stabilize ph of ECF Occurs whenever body ph moves outside normal limits Directly affects carbonic acid bicarbonate buffer system Respiratory Compensation H 2 CO 3 (acid) H + + HCO 3 - (base) Increasing or decreasing the rate of respiration alters ph by lowering or raising the P CO2 When P CO2 rises, ph falls as addition of CO 2 drives buffer system to the right (adding H+) When P CO2 falls, ph rises as removal of CO 2 drives buffer system to the left (removing H+) Renal Mechanisms Support buffer systems by: 1. secreting or absorbing H + or HCO 3-2. controlling excretion of acids and bases 3. generating additional buffers Renal Compensation Is a change in rates of H + and HCO 3 secretion or reabsorption by kidneys in response to changes in plasma ph Kidneys assist lungs by eliminating any CO 2 that enters renal tubules during filtration or that diffuses into tubular fluid en route to renal pelvis Hydrogen ions are secreted into tubular fluid along: proximal convoluted tubule (PCT) distal convoluted tubule (DCT) collecting system 4
5 Buffers in Urine The ability to eliminate large numbers of H + in a normal volume of urine depends on the presence of buffers in urine (without them, we d need to dilute the H+ with like 1000x more water) 1. Carbonic acid bicarbonate buffer system 2. Phosphate buffer system (these two provided by filtration) 3. Ammonia buffer system: Tubular deamination creates NH 3, which difuses into the tublule and buffers H+ by grabbing it and becoming NH 4 + Bicarbonate is reabsorbed along with Na+ Kidney Tubules and ph Regulation Buffers Figure 27 10a Kidney Tubules and ph Regulation - Renal Responses to Acidosis 1. Secretion of H + 2. Activity of buffers in tubular fluid 3. Removal of CO 2 4. Reabsorption of NaHCO 3 Figure 27 10b Regulation of Plasma ph - Acidosis Renal Responses to Alkalosis 1. Rate of H+ secretion at kidneys declines 2. Tubule cells do not reclaim bicarbonates in tubular fluid 3. Collecting system actually transports HCO 3 - out into tubular fluid while releasing strong acid (HCl) into peritubular fluid Figure 27 11a 5
6 Kidney Tubules and ph Regulation Regulation of Plasma ph - Alkalosis Figure 27 10c Figure 27 11b Conditions Affecting Acid Base Balance 1. Disorders affecting: circulating buffers respiratory performance renal function 2. Cardiovascular conditions: heart failure hypotension 3. Conditions affecting the CNS: neural damage or disease that affects respiratory and cardiovascular reflexes Disturbances of Acid Base Balance Acute phase: the initial phase in which ph moves rapidly out of normal range Compensated phase: when condition persists, physiological adjustments occur Types of Disorders Respiratory Acid Base Disorders Result from imbalance between CO 2 generation in peripheral tissues and CO 2 excretion at lungs Cause abnormal CO 2 levels in ECF Metabolic Acid Base Disorders Result from one of two things: generation of organic or fixed acids conditions affecting HCO 3 - concentration in ECF Respiratory Acidosis Most common acid/base problem Develops when the respiratory system cannot eliminate all CO 2 generated by peripheral tissues Primary sign: low plasma ph due to hypercapnia Primarily caused by hypoventilation Acute: cardiac arrest, drowning Chronic/compensated: COPD, CHF compensated by: increased respiratory rate, buffering by non-carbonic acid buffers, increased H+ secretion 6
7 Respiratory Acid Base Regulation Respiratory Alkalosis Least clinically relevant Primary sign: high plasma ph due to hypocapnia Primarily caused by hyperventilation Caused by: stress/panic, high altitude hyperventilation Loss of consciousness often resolves or breathing into a bag to increase PCO 2 Only acute, rarely compensated Figure 27 12a Respiratory Acid Base Regulation Figure 27 12b Metabolic Acidosis Caused by: Production of large numbers of fixed or organic acids, H+ overloads buffer system Lactic acidosis: produced by anaerobic cellular respiration Also a complication of hypoxia caused by respiratory acidosis (tissue switched to anaerobic) Ketoacidosis: produced by excess ketone bodies (starvation, untreated diabetes) Impaired H+ excretion at kidneys Caused by kidney damage, overuse of diuretics that stop Na+ at the expense of H+ secretion Severe bicarbonate loss (diarrhea loss of bicarbonate from pancreas, liver that mould have been reabsorbed) Metabolic Acidosis Second most common acid/base problem Respiratory and metabolic acidosis are typically linked: low O 2 generates lactic acid hypoventilation leads to low PO 2 Compensated by Respiratory: increased RR (eliminate CO 2 ) Renal: secrete H+, reabsorb and generate HCO 3 - Responses to Metabolic Acidosis Figure
8 Metabolic Alkalosis Kidney Response to Alkalosis Caused by elevated HCO 3- concentrations Bicarbonate ions interact with H + in solution forming H 2 CO 3 Reduced H + causes alkalosis Causes: Alkaline tide: gastric HCl generation after a meal (temporary) Vomiting: greatly increased HCl generation due to loss in vomit Compensation: Respiratory: reduced RR Increased HCO 3- loss at kidney, Retention of HCl Figure 27 10c Metabolic Alkalosis Detection of Acidosis and Alkalosis Includes blood tests for ph, P CO2 and HCO 3 levels: recognition of acidosis or alkalosis classification as respiratory or metabolic Figure Diseases Kidney Damage: can cause increase in glomerular permeability, plamsa proteins enter capsular space. Causes: Proteinuria Decrease (BCOP), increase CsCOP (result?) Blocked urine flow (kidney stone, etc.) CsHP rises (effect?) Nephritis (inflammation) Causes swelling, also increases CsHP Figure (1 of 2) 8
9 Diuretics Caffeine: reduces sodium reabsorption Alcohol: blocks ADH release at post. pot. Mannitol: adds osmotic particle that must be eliminated with water Loop diuretics: inhibit ion transport in Loop of Henle, short circuit conc grad in medulla Aldosterone blockers e.g. spironolactone (can cause acidosis) Dialysis In chronic renal failure, kidney function can be replaced by filtering the blood through a machine outside of the body. Blood leaves through a catheter, runs through a column with dialysis fluid it, exchange occurs with wastes diffusing out (concentration of urea, creatinine, uric acid, phosphate, sulfate in fluid is zero) Ion Imbalances Hyponatrmia: nausea, lethargy, and apathy, cerebral edema Hypernatremia: neurological damage due to shrinkage of brain cells, confusion, coma Hypokalemia: fibrillation, nervous symptoms such as tingling of the skin, numbness of the hands or feet, weakness Hyperkalemia: cardiac arrhythmia, muscle pain, general discomfort or irritability, weakness, and paralysis Hypocalcemia: brittle bones, parathesias, tetany Hypercalcemia: heart arrhythmias, kidney stones 9
Fluid and Electrolytes P A R T 4
 Fluid and Electrolytes P A R T 4 Mechanisms that control acid-base homeostasis Acids and bases continually enter and leave body Hydrogen ions also result from metabolic activity Acids Hydrogen ion donors
Fluid and Electrolytes P A R T 4 Mechanisms that control acid-base homeostasis Acids and bases continually enter and leave body Hydrogen ions also result from metabolic activity Acids Hydrogen ion donors
Acid-Base Balance 11/18/2011. Regulation of Potassium Balance. Regulation of Potassium Balance. Regulatory Site: Cortical Collecting Ducts.
 Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + )
 Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Fluid, Electrolyte, and Acid Base Balance
 25 Fluid, Electrolyte, and Acid Base Balance Lecture Presentation by Lori Garrett Note to the Instructor: For the third edition of Visual Anatomy & Physiology, we have updated our PowerPoints to fully
25 Fluid, Electrolyte, and Acid Base Balance Lecture Presentation by Lori Garrett Note to the Instructor: For the third edition of Visual Anatomy & Physiology, we have updated our PowerPoints to fully
Acid-Base Balance Dr. Gary Mumaugh
 Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Acid and Base Balance
 Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
Water, Electrolytes, and Acid-Base Balance
 Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Renal Physiology. April, J. Mohan, PhD. Lecturer, Physiology Unit, Faculty of Medical Sciences, U.W.I., St Augustine.
 Renal Physiology April, 2011 J. Mohan, PhD. Lecturer, Physiology Unit, Faculty of Medical Sciences, U.W.I., St Augustine. Office : Room 105, Physiology Unit. References: Koeppen B.E. & Stanton B.A. (2010).
Renal Physiology April, 2011 J. Mohan, PhD. Lecturer, Physiology Unit, Faculty of Medical Sciences, U.W.I., St Augustine. Office : Room 105, Physiology Unit. References: Koeppen B.E. & Stanton B.A. (2010).
Acid/Base Balance. the concentrations of these two ions affect the acidity or alkalinity of body fluids
 Acid/Base Balance some of most critical ions in body fluids are H + (hydrogen) and OH - (hydroxyl) ions the concentrations of these two ions affect the acidity or alkalinity of body fluids acidity/alkalinity
Acid/Base Balance some of most critical ions in body fluids are H + (hydrogen) and OH - (hydroxyl) ions the concentrations of these two ions affect the acidity or alkalinity of body fluids acidity/alkalinity
UNIT VI: ACID BASE IMBALANCE
 UNIT VI: ACID BASE IMBALANCE 1 Objectives: Review the physiological mechanism responsible to regulate acid base balance in the body i.e.: Buffers (phosphate, hemoglobin, carbonate) Renal mechanism Respiratory
UNIT VI: ACID BASE IMBALANCE 1 Objectives: Review the physiological mechanism responsible to regulate acid base balance in the body i.e.: Buffers (phosphate, hemoglobin, carbonate) Renal mechanism Respiratory
Acid-Base Balance * OpenStax
 OpenStax-CNX module: m46409 1 Acid-Base Balance * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will be
OpenStax-CNX module: m46409 1 Acid-Base Balance * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will be
BIO132 Chapter 27 Fluid, Electrolyte and Acid Base Balance Lecture Outline
 BIO132 Chapter 27 Fluid, Electrolyte and Acid Base Balance Lecture Outline Fluid divisions 1. Extracellular fluid (ECF) 2. Intracellular fluid (ICF) Stabilization 1. Fluid balance 2. Electrolyte balance
BIO132 Chapter 27 Fluid, Electrolyte and Acid Base Balance Lecture Outline Fluid divisions 1. Extracellular fluid (ECF) 2. Intracellular fluid (ICF) Stabilization 1. Fluid balance 2. Electrolyte balance
Principles of Anatomy and Physiology
 Principles of Anatomy and Physiology 14 th Edition CHAPTER 27 Fluid, Electrolyte, and Acid Base Fluid Compartments and Fluid In adults, body fluids make up between 55% and 65% of total body mass. Body
Principles of Anatomy and Physiology 14 th Edition CHAPTER 27 Fluid, Electrolyte, and Acid Base Fluid Compartments and Fluid In adults, body fluids make up between 55% and 65% of total body mass. Body
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE
 Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
Acid Base Balance. Professor Dr. Raid M. H. Al-Salih. Clinical Chemistry Professor Dr. Raid M. H. Al-Salih
 Acid Base Balance 1 HYDROGEN ION CONCENTRATION and CONCEPT OF ph Blood hydrogen ion concentration (abbreviated [H + ]) is maintained within tight limits in health, with the normal concentration being between
Acid Base Balance 1 HYDROGEN ION CONCENTRATION and CONCEPT OF ph Blood hydrogen ion concentration (abbreviated [H + ]) is maintained within tight limits in health, with the normal concentration being between
Acids, Bases, and Salts
 Acid / Base Balance Objectives Define an acid, a base, and the measure of ph. Discuss acid/base balance, the effects of acidosis or alkalosis on the body, and the mechanisms in place to maintain balance
Acid / Base Balance Objectives Define an acid, a base, and the measure of ph. Discuss acid/base balance, the effects of acidosis or alkalosis on the body, and the mechanisms in place to maintain balance
Chapter 19 The Urinary System Fluid and Electrolyte Balance
 Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
12/7/10. Excretory System. The basic function of the excretory system is to regulate the volume and composition of body fluids by:
 Excretory System The basic function of the excretory system is to regulate the volume and composition of body fluids by: o o removing wastes returning needed substances to the body for reuse Body systems
Excretory System The basic function of the excretory system is to regulate the volume and composition of body fluids by: o o removing wastes returning needed substances to the body for reuse Body systems
Acids and Bases their definitions and meanings
 Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance
 Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
ACID BASE BALANCE & BODY FLUID. Ani Retno Prijanti Renal and Body Fluids Module Juni 2008
 ACID BASE BALANCE & BODY FLUID Ani Retno Prijanti Renal and Body Fluids Module Juni 2008 1 Continuous Mixing of Body Fluids 2 Water Balance and ECF Osmolality To remain properly hydrated, water intake
ACID BASE BALANCE & BODY FLUID Ani Retno Prijanti Renal and Body Fluids Module Juni 2008 1 Continuous Mixing of Body Fluids 2 Water Balance and ECF Osmolality To remain properly hydrated, water intake
CHAPTER 27 LECTURE OUTLINE
 CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
Carbon Dioxide Transport. Carbon Dioxide. Carbon Dioxide Transport. Carbon Dioxide Transport - Plasma. Hydrolysis of Water
 Module H: Carbon Dioxide Transport Beachey Ch 9 & 10 Egan pp. 244-246, 281-284 Carbon Dioxide Transport At the end of today s session you will be able to : Describe the relationship free hydrogen ions
Module H: Carbon Dioxide Transport Beachey Ch 9 & 10 Egan pp. 244-246, 281-284 Carbon Dioxide Transport At the end of today s session you will be able to : Describe the relationship free hydrogen ions
BCH 450 Biochemistry of Specialized Tissues
 BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
Chapter 15 Fluid and Acid-Base Balance
 Chapter 15 Fluid and Acid-Base Balance by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Fluid Balance Water constitutes ~60% of body weight. All cells and tissues are surrounded by an aqueous environment.
Chapter 15 Fluid and Acid-Base Balance by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Fluid Balance Water constitutes ~60% of body weight. All cells and tissues are surrounded by an aqueous environment.
Chapter 27: Fluid, Electrolyte, and Acid Base Balance
 Chapter 27: Fluid, Electrolyte, and Acid Base Balance Fluid, Electrolyte, and Acid Base Balance: An Overview, p. 995 Most of your body weight is water. Water accounts for up to 99 percent of the volume
Chapter 27: Fluid, Electrolyte, and Acid Base Balance Fluid, Electrolyte, and Acid Base Balance: An Overview, p. 995 Most of your body weight is water. Water accounts for up to 99 percent of the volume
Chapter 24 Water, Electrolyte and Acid-Base Balance
 Chapter 24 Water, Electrolyte and Acid-Base Balance Total body water for 150 lb. male = 40L 65% ICF 35% ECF 25% tissue fluid 8% blood plasma, lymph 2% transcellular fluid (CSF, synovial fluid) Water Movement
Chapter 24 Water, Electrolyte and Acid-Base Balance Total body water for 150 lb. male = 40L 65% ICF 35% ECF 25% tissue fluid 8% blood plasma, lymph 2% transcellular fluid (CSF, synovial fluid) Water Movement
DIURETICS-2. Dr. Shariq Syed. Shariq AIKC/TYB/2014
 DIURETICS-2 Dr. Syed Structure of Kidney Blood filtered by functional unit: Nephron Except for cells, proteins, other large molecules, rest gets filtered Structure of Kidney 3 major regions of nephron
DIURETICS-2 Dr. Syed Structure of Kidney Blood filtered by functional unit: Nephron Except for cells, proteins, other large molecules, rest gets filtered Structure of Kidney 3 major regions of nephron
Ch 17 Physiology of the Kidneys
 Ch 17 Physiology of the Kidneys Review Anatomy on your own SLOs List and describe the 4 major functions of the kidneys. List and explain the 4 processes of the urinary system. Diagram the filtration barriers
Ch 17 Physiology of the Kidneys Review Anatomy on your own SLOs List and describe the 4 major functions of the kidneys. List and explain the 4 processes of the urinary system. Diagram the filtration barriers
adam.com (http://www.adam.com/) Benjamin/Cummings Publishing Co (http://www.awl.com/bc) -42-
 Graphics are used with permission of : adam.com (http://www.adam.com/) Benjamin/Cummings Publishing Co (http://www.awl.com/bc) -42-74. (1) Carbon dioxide arrives at the kidney tubule cell in the proximal
Graphics are used with permission of : adam.com (http://www.adam.com/) Benjamin/Cummings Publishing Co (http://www.awl.com/bc) -42-74. (1) Carbon dioxide arrives at the kidney tubule cell in the proximal
PARAMEDIC RESOURCE MANUAL
 ONTARIO BASE HOSPITAL GROUP PARAMEDIC RESOURCE MANUAL ACID-BASE BALANCE SECTION SIX Version 1.1 2010 Update PARAMEDIC RESOURCE MANUAL OBJECTIVES: ACID-BASE BALANCE The objectives indicate what you should
ONTARIO BASE HOSPITAL GROUP PARAMEDIC RESOURCE MANUAL ACID-BASE BALANCE SECTION SIX Version 1.1 2010 Update PARAMEDIC RESOURCE MANUAL OBJECTIVES: ACID-BASE BALANCE The objectives indicate what you should
Acid-Base Tutorial 2/10/2014. Overview. Physiology (2) Physiology (1)
 Overview Acid-Base Tutorial Nicola Barlow Physiology Buffering systems Control mechanisms Laboratory assessment of acid-base Disorders of H + ion homeostasis Respiratory acidosis Metabolic acidosis Respiratory
Overview Acid-Base Tutorial Nicola Barlow Physiology Buffering systems Control mechanisms Laboratory assessment of acid-base Disorders of H + ion homeostasis Respiratory acidosis Metabolic acidosis Respiratory
Chapter 10: Urinary System & Excretion
 Chapter 10: Urinary System & Excretion Organs of Urinary System Kidneys (2) form urine Ureters (2) Carry urine from kidneys to bladder Bladder Stores urine Urethra Carries urine from bladder to outside
Chapter 10: Urinary System & Excretion Organs of Urinary System Kidneys (2) form urine Ureters (2) Carry urine from kidneys to bladder Bladder Stores urine Urethra Carries urine from bladder to outside
CASE 27. What is the response of the kidney to metabolic acidosis? What is the response of the kidney to a respiratory alkalosis?
 CASE 27 A 21-year-old man with insulin-dependent diabetes presents to the emergency center with mental status changes, nausea, vomiting, abdominal pain, and rapid respirations. On examination, the patient
CASE 27 A 21-year-old man with insulin-dependent diabetes presents to the emergency center with mental status changes, nausea, vomiting, abdominal pain, and rapid respirations. On examination, the patient
BUFFERING OF HYDROGEN LOAD
 BUFFERING OF HYDROGEN LOAD 1. Extracellular space minutes 2. Intracellular space minutes to hours 3. Respiratory compensation 6 to 12 hours 4. Renal compensation hours, up to 2-3 days RENAL HYDROGEN SECRETION
BUFFERING OF HYDROGEN LOAD 1. Extracellular space minutes 2. Intracellular space minutes to hours 3. Respiratory compensation 6 to 12 hours 4. Renal compensation hours, up to 2-3 days RENAL HYDROGEN SECRETION
BIOL 2402 Fluid/Electrolyte Regulation
 Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Other Factors Affecting GFR. Chapter 25. After Filtration. Reabsorption and Secretion. 5 Functions of the PCT
 Other Factors Affecting GFR Chapter 25 Part 2. Renal Physiology Nitric oxide vasodilator produced by the vascular endothelium Adenosine vasoconstrictor of renal vasculature Endothelin a powerful vasoconstrictor
Other Factors Affecting GFR Chapter 25 Part 2. Renal Physiology Nitric oxide vasodilator produced by the vascular endothelium Adenosine vasoconstrictor of renal vasculature Endothelin a powerful vasoconstrictor
Excretory System 1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- X- Y- Z- b) Which of the following is not a function of the organ shown? A. to produce
Acid-Base Physiology. Dr. Tamás Bense Dr. Alexandra Turi
 Acid-Base Physiology Dr. Tamás Bense Dr. Alexandra Turi What is a blood gas assessment? We get it from an arterial sample (a.radialis, a. brachialis, a. femoralis) Invasive technique If the patient is
Acid-Base Physiology Dr. Tamás Bense Dr. Alexandra Turi What is a blood gas assessment? We get it from an arterial sample (a.radialis, a. brachialis, a. femoralis) Invasive technique If the patient is
D fini n tion: p = = -log [H+] ph=7 me m an s 10-7 Mol M H+ + (100 nmol m /l); ) p ; H=8 me m an s 10-8 Mol M H+ + (10 (10 n nmol m /l) Nor
![D fini n tion: p = = -log [H+] ph=7 me m an s 10-7 Mol M H+ + (100 nmol m /l); ) p ; H=8 me m an s 10-8 Mol M H+ + (10 (10 n nmol m /l) Nor D fini n tion: p = = -log [H+] ph=7 me m an s 10-7 Mol M H+ + (100 nmol m /l); ) p ; H=8 me m an s 10-8 Mol M H+ + (10 (10 n nmol m /l) Nor](/thumbs/75/72756397.jpg) Definition: ph regulation ph = -log [H + ] ph=7 means 10-7 Mol H + (100 nmol/l); ph=8 means 10 Normal plasma value: 7.35-7.45; 7.45; (H Acidosis: ph7.45 Intracellular ph = 7.1-7.3
Definition: ph regulation ph = -log [H + ] ph=7 means 10-7 Mol H + (100 nmol/l); ph=8 means 10 Normal plasma value: 7.35-7.45; 7.45; (H Acidosis: ph7.45 Intracellular ph = 7.1-7.3
Chapter 21. Diuretic Agents. Mosby items and derived items 2008, 2002 by Mosby, Inc., an affiliate of Elsevier Inc.
 Chapter 21 Diuretic Agents Renal Structure and Function Kidneys at level of umbilicus Each weighs 160 to 175 g and is 10 to 12 cm long Most blood flow per gram of weight in body 22% of cardiac output (CO)
Chapter 21 Diuretic Agents Renal Structure and Function Kidneys at level of umbilicus Each weighs 160 to 175 g and is 10 to 12 cm long Most blood flow per gram of weight in body 22% of cardiac output (CO)
RENAL FUNCTION An Overview
 RENAL FUNCTION An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ. Temple 1 Kidneys
RENAL FUNCTION An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ. Temple 1 Kidneys
Metabolic Alkalosis: Vomiting
 RENAL ANL) ACID-BASE PHYSIOLOGY 213 Case 37 Metabolic Alkalosis: Vomiting Maria Cuervo is a 20-year-old philosophy major at a state university. When the "24-hour" stomach flu went around campus during
RENAL ANL) ACID-BASE PHYSIOLOGY 213 Case 37 Metabolic Alkalosis: Vomiting Maria Cuervo is a 20-year-old philosophy major at a state university. When the "24-hour" stomach flu went around campus during
Potassium regulation. -Kidney is a major regulator for potassium Homeostasis.
 Potassium regulation. -Kidney is a major regulator for potassium Homeostasis. Normal potassium intake, distribution, and output from the body. Effects of severe hyperkalemia Partial depolarization of cell
Potassium regulation. -Kidney is a major regulator for potassium Homeostasis. Normal potassium intake, distribution, and output from the body. Effects of severe hyperkalemia Partial depolarization of cell
April 08, biology 2201 ch 11.3 excretion.notebook. Biology The Excretory System. Apr 13 9:14 PM EXCRETORY SYSTEM.
 Biology 2201 11.3 The Excretory System EXCRETORY SYSTEM 1 Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid base concentrations and metabolite concentrations
Biology 2201 11.3 The Excretory System EXCRETORY SYSTEM 1 Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid base concentrations and metabolite concentrations
PRINCIPLES OF DIURETIC ACTIONS:
 DIURETIC: A drug that increases excretion of solutes Increased urine volume is secondary All clinically useful diuretics act by blocking Na + reabsorption Has the highest EC to IC ratio = always more sodium
DIURETIC: A drug that increases excretion of solutes Increased urine volume is secondary All clinically useful diuretics act by blocking Na + reabsorption Has the highest EC to IC ratio = always more sodium
Slide 1. Slide 2. Slide 3. Learning Outcomes. Acid base terminology ARTERIAL BLOOD GAS INTERPRETATION
 Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
The Urinary S. (Chp. 10) & Excretion. What are the functions of the urinary system? Maintenance of water-salt and acidbase
 10.1 Urinary system The Urinary S. (Chp. 10) & Excretion 10.1 Urinary system What are the functions of the urinary system? 1. Excretion of metabolic wastes (urea, uric acid & creatinine) 1. Maintenance
10.1 Urinary system The Urinary S. (Chp. 10) & Excretion 10.1 Urinary system What are the functions of the urinary system? 1. Excretion of metabolic wastes (urea, uric acid & creatinine) 1. Maintenance
Excretory Lecture Test Questions Set 1
 Excretory Lecture Test Questions Set 1 1. The separation and ejection of metabolic wastes, usually in aqueous solution, is: a. reabsorption b. secretion c. filtration d. excretion e. endocrinology 2. Besides
Excretory Lecture Test Questions Set 1 1. The separation and ejection of metabolic wastes, usually in aqueous solution, is: a. reabsorption b. secretion c. filtration d. excretion e. endocrinology 2. Besides
Disorders of Acid-Base
 Disorders of Acid-Base Balance Bởi: OpenStaxCollege Normal arterial blood ph is restricted to a very narrow range of 7.35 to 7.45. A person who has a blood ph below 7.35 is considered to be in acidosis
Disorders of Acid-Base Balance Bởi: OpenStaxCollege Normal arterial blood ph is restricted to a very narrow range of 7.35 to 7.45. A person who has a blood ph below 7.35 is considered to be in acidosis
Renal physiology V. Regulation of acid-base balance. Dr Alida Koorts BMS
 Renal physiology V Regulation of acidbase balance Dr Alida Koorts BMS 712 012 319 2921 akoorts@medic.up.ac.za Hydrogen ions (H + ): Concentration and origin Concentration in arterial blood, resting: [H
Renal physiology V Regulation of acidbase balance Dr Alida Koorts BMS 712 012 319 2921 akoorts@medic.up.ac.za Hydrogen ions (H + ): Concentration and origin Concentration in arterial blood, resting: [H
Chapter 2. Fluid, Electrolyte, and Acid-Base Imbalances
 Chapter 2 Fluid, Electrolyte, and Acid-Base Imbalances Review of Concepts and Processes The major component of the body is water. Water is located in these compartments: Intracellular fluid (ICF) compartment
Chapter 2 Fluid, Electrolyte, and Acid-Base Imbalances Review of Concepts and Processes The major component of the body is water. Water is located in these compartments: Intracellular fluid (ICF) compartment
Renal System and Excretion
 Renal System and Excretion Biology 105 Lecture 19 Chapter 16 Outline Renal System I. Functions II. Organs of the renal system III. Kidneys 1. Structure 2. Function IV. Nephron 1. Structure 2. Function
Renal System and Excretion Biology 105 Lecture 19 Chapter 16 Outline Renal System I. Functions II. Organs of the renal system III. Kidneys 1. Structure 2. Function IV. Nephron 1. Structure 2. Function
ARTERIAL BLOOD GASES PART 1 BACK TO BASICS SSR OLIVIA ELSWORTH SEPT 2017
 ARTERIAL BLOOD GASES PART 1 BACK TO BASICS SSR OLIVIA ELSWORTH SEPT 2017 WHAT INFORMATION DOES AN ABG GIVE US? ph = measure of hydrogen ion concentration (acidity or alkalinity) PaCO2 = partial pressure
ARTERIAL BLOOD GASES PART 1 BACK TO BASICS SSR OLIVIA ELSWORTH SEPT 2017 WHAT INFORMATION DOES AN ABG GIVE US? ph = measure of hydrogen ion concentration (acidity or alkalinity) PaCO2 = partial pressure
WATER, SODIUM AND POTASSIUM
 WATER, SODIUM AND POTASSIUM Attila Miseta Tamás Kőszegi Department of Laboratory Medicine, 2016 1 Average daily water intake and output of a normal adult 2 Approximate contributions to plasma osmolality
WATER, SODIUM AND POTASSIUM Attila Miseta Tamás Kőszegi Department of Laboratory Medicine, 2016 1 Average daily water intake and output of a normal adult 2 Approximate contributions to plasma osmolality
Questions? Homework due in lab 6. PreLab #6 HW 15 & 16 (follow directions, 6 points!)
 Questions? Homework due in lab 6 PreLab #6 HW 15 & 16 (follow directions, 6 points!) Part 3 Variations in Urine Formation Composition varies Fluid volume Solute concentration Variations in Urine Formation
Questions? Homework due in lab 6 PreLab #6 HW 15 & 16 (follow directions, 6 points!) Part 3 Variations in Urine Formation Composition varies Fluid volume Solute concentration Variations in Urine Formation
Dr. Suzana Voiculescu
 Dr. Suzana Voiculescu Definition All the processes inside the body which keep the H+ concentration within normal values. Depends on water and ion balance blood gas homeostasis Blood acidity may be expressed
Dr. Suzana Voiculescu Definition All the processes inside the body which keep the H+ concentration within normal values. Depends on water and ion balance blood gas homeostasis Blood acidity may be expressed
Kidneys and Homeostasis
 16 The Urinary System The Urinary System OUTLINE: Eliminating Waste Components of the Urinary System Kidneys and Homeostasis Urination Urinary Tract Infections Eliminating Waste Excretion Elimination of
16 The Urinary System The Urinary System OUTLINE: Eliminating Waste Components of the Urinary System Kidneys and Homeostasis Urination Urinary Tract Infections Eliminating Waste Excretion Elimination of
Urinary System and Excretion. Bio105 Lecture 20 Chapter 16
 Urinary System and Excretion Bio105 Lecture 20 Chapter 16 1 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of the urinary system
Urinary System and Excretion Bio105 Lecture 20 Chapter 16 1 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of the urinary system
Pharmacology I [PHL 313] Diuretics. Dr. Mohammad Nazam Ansari
![Pharmacology I [PHL 313] Diuretics. Dr. Mohammad Nazam Ansari Pharmacology I [PHL 313] Diuretics. Dr. Mohammad Nazam Ansari](/thumbs/90/104323364.jpg) Pharmacology I [PHL 313] Diuretics Dr. Mohammad Nazam Ansari Renal Pharmacology Kidneys: Each adult kidney weighs 125-170g in males and 115-155g in females, represent 0.5% of total body weight, but receive
Pharmacology I [PHL 313] Diuretics Dr. Mohammad Nazam Ansari Renal Pharmacology Kidneys: Each adult kidney weighs 125-170g in males and 115-155g in females, represent 0.5% of total body weight, but receive
Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender
 BIOL 221 Chapter 26 Fluids & Electrolytes 35 slides!1 Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender males have higher TBW
BIOL 221 Chapter 26 Fluids & Electrolytes 35 slides!1 Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender males have higher TBW
DIURETICS-4 Dr. Shariq Syed
 DIURETICS-4 Dr. Shariq Syed AIKTC - Knowledge Resources & Relay Center 1 Pop Quiz!! Loop diuretics act on which transporter PKCC NKCC2 AIKTCC I Don t know AIKTC - Knowledge Resources & Relay Center 2 Pop
DIURETICS-4 Dr. Shariq Syed AIKTC - Knowledge Resources & Relay Center 1 Pop Quiz!! Loop diuretics act on which transporter PKCC NKCC2 AIKTCC I Don t know AIKTC - Knowledge Resources & Relay Center 2 Pop
The Excretory System. Biology 20
 The Excretory System Biology 20 Introduction Follow along on page 376 What dangers exist if your body is unable to regulate the fluid balance of your tissues? What challenged would the body have to respond
The Excretory System Biology 20 Introduction Follow along on page 376 What dangers exist if your body is unable to regulate the fluid balance of your tissues? What challenged would the body have to respond
EXCRETION QUESTIONS. Use the following information to answer the next two questions.
 EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
Excretion and Waste Management. Biology 30S - Miss Paslawski
 Excretion and Waste Management Biology 30S - Miss Paslawski Lesson 1 Waste Products and Organs 2 3 Excretion Excretion: Process by which dissolved metabolic wastes are separated from body fluids and removed
Excretion and Waste Management Biology 30S - Miss Paslawski Lesson 1 Waste Products and Organs 2 3 Excretion Excretion: Process by which dissolved metabolic wastes are separated from body fluids and removed
There are many buffers in the kidney, but the main one is the phosphate buffer.
 9 Yanal Obada Zalat Renal Control of AcidBase Balance The kidneys play three major roles in the maintenance of normal acidbase balance: 1excretion of H+ (fixed _non volatile H+) 2Reabsorption of filtrated
9 Yanal Obada Zalat Renal Control of AcidBase Balance The kidneys play three major roles in the maintenance of normal acidbase balance: 1excretion of H+ (fixed _non volatile H+) 2Reabsorption of filtrated
Acid-Base Imbalance-2 Lecture 9 (12/4/2015) Yanal A. Shafagoj MD. PhD
 AcidBase Imbalance2 Lecture 9 (12/4/2015) Yanal A. Shafagoj MD. PhD Introduction Disturbance in acidbase balance are common clinical problem that range in severity from mild to life threatening, the acute
AcidBase Imbalance2 Lecture 9 (12/4/2015) Yanal A. Shafagoj MD. PhD Introduction Disturbance in acidbase balance are common clinical problem that range in severity from mild to life threatening, the acute
Acid - base equilibrium
 Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
1. 09/07/16 Ch 1: Intro to Human A & P 1
 Table of Contents # Date Title Page # 1. 09/07/16 Ch 1: Intro to Human A & P 1 2. 09/19/16 Ch 18: Water, Electrolyte, and Acid-Base Balance 5 i 1 09/19/16 Chapter 18: Water, Electrolyte, and Acid-Base
Table of Contents # Date Title Page # 1. 09/07/16 Ch 1: Intro to Human A & P 1 2. 09/19/16 Ch 18: Water, Electrolyte, and Acid-Base Balance 5 i 1 09/19/16 Chapter 18: Water, Electrolyte, and Acid-Base
Part 1 The Cell and the Cellular Environment
 1 Chapter 3 Anatomy and Physiology Part 1 The Cell and the Cellular Environment 2 The Human Cell The is the fundamental unit of the human body. Cells contain all the necessary for life functions. 3 Cell
1 Chapter 3 Anatomy and Physiology Part 1 The Cell and the Cellular Environment 2 The Human Cell The is the fundamental unit of the human body. Cells contain all the necessary for life functions. 3 Cell
ANATOMY & PHYSIOLOGY - CLUTCH CH ACID-BASE BALANCE-- CONTROLLING BLOOD PH
 !! www.clutchprep.com ANATOMY & PHYSIOLOGY - CLUTCH CONCEPT: INTRO. TO DISTURBING AND MAINTAINING BLOOD ph Physiological Sources of Acid (and Base): Acids are molecules/substances that H+ to a solution.
!! www.clutchprep.com ANATOMY & PHYSIOLOGY - CLUTCH CONCEPT: INTRO. TO DISTURBING AND MAINTAINING BLOOD ph Physiological Sources of Acid (and Base): Acids are molecules/substances that H+ to a solution.
Chapter 12. Excretion and the Interaction of Systems
 Chapter 12 Excretion and the Interaction of Systems 1 2 Goals for This Chapter 1. Identify the main structures and functions of the human excretory system 2. Explain the function of the nephron 3. Describe
Chapter 12 Excretion and the Interaction of Systems 1 2 Goals for This Chapter 1. Identify the main structures and functions of the human excretory system 2. Explain the function of the nephron 3. Describe
Chapter 20 8/23/2016. Fluids and Electrolytes. Fluid (Water) Fluid (Water) (Cont.) Functions
 Chapter 20 Fluids and Electrolytes All items and derived items 2015, 2011, 2006 by Mosby, Inc., an imprint of Elsevier Inc. All rights reserved. Fluid (Water) Functions Provides an extracellular transportation
Chapter 20 Fluids and Electrolytes All items and derived items 2015, 2011, 2006 by Mosby, Inc., an imprint of Elsevier Inc. All rights reserved. Fluid (Water) Functions Provides an extracellular transportation
5/18/2017. Specific Electrolytes. Sodium. Sodium. Sodium. Sodium. Sodium
 Specific Electrolytes Hyponatremia Hypervolemic Replacing water (not electrolytes) after perspiration Freshwater near-drowning Syndrome of Inappropriate ADH Secretion (SIADH) Hypovolemic GI disease (decreased
Specific Electrolytes Hyponatremia Hypervolemic Replacing water (not electrolytes) after perspiration Freshwater near-drowning Syndrome of Inappropriate ADH Secretion (SIADH) Hypovolemic GI disease (decreased
Renal Physiology Part II. Bio 219 Napa Valley College Dr. Adam Ross
 Renal Physiology Part II Bio 219 Napa Valley College Dr. Adam Ross Fluid and Electrolyte balance As we know from our previous studies: Water and ions need to be balanced in order to maintain proper homeostatic
Renal Physiology Part II Bio 219 Napa Valley College Dr. Adam Ross Fluid and Electrolyte balance As we know from our previous studies: Water and ions need to be balanced in order to maintain proper homeostatic
Principles of Fluid Balance
 Principles of Fluid Balance I. The Cellular Environment: Fluids and Electrolytes A. Water 1. Total body water (TBW) = 60% of total body weight 2. Fluid Compartments in the Body a. Intracellular Compartment
Principles of Fluid Balance I. The Cellular Environment: Fluids and Electrolytes A. Water 1. Total body water (TBW) = 60% of total body weight 2. Fluid Compartments in the Body a. Intracellular Compartment
Instrumental determination of electrolytes in urine. Amal Alamri
 Instrumental determination of electrolytes in urine Amal Alamri What is the Electrolytes? Electrolytes are positively and negatively chargedions, Found in Within body's cells extracellular fluids, including
Instrumental determination of electrolytes in urine Amal Alamri What is the Electrolytes? Electrolytes are positively and negatively chargedions, Found in Within body's cells extracellular fluids, including
Urinary System. Dr. ZHANG Xiong. Dept. of Physiology. ZJU School of Medicine. QUESTION 6
 Urinary System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine http://10.71.121.158 Copyright@ Xiong Zhang QUESTION 6 How is the filtrate reabsorbed in tubular system? Copyright@ Xiong Zhang
Urinary System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine http://10.71.121.158 Copyright@ Xiong Zhang QUESTION 6 How is the filtrate reabsorbed in tubular system? Copyright@ Xiong Zhang
PARTS OF THE URINARY SYSTEM
 EXCRETORY SYSTEM Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid-base concentrations and metabolite concentrations 1 ORGANS OF EXCRETION Skin and
EXCRETORY SYSTEM Excretory System How does the excretory system maintain homeostasis? It regulates heat, water, salt, acid-base concentrations and metabolite concentrations 1 ORGANS OF EXCRETION Skin and
The Urinary System 15PART B. PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART B Ureters Slender tubes attaching the kidney to the bladder Continuous with
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART B Ureters Slender tubes attaching the kidney to the bladder Continuous with
1. Urinary System, General
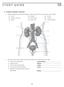 S T U D Y G U I D E 16 1. Urinary System, General a. Label the figure by placing the numbers of the structures in the spaces by the correct labels. 7 Aorta 6 Kidney 8 Ureter 2 Inferior vena cava 4 Renal
S T U D Y G U I D E 16 1. Urinary System, General a. Label the figure by placing the numbers of the structures in the spaces by the correct labels. 7 Aorta 6 Kidney 8 Ureter 2 Inferior vena cava 4 Renal
Dr. Suzana Voiculescu Discipline of Physiology and Fundamental Neurosciences Carol Davila Univ. of Medicine and Pharmacy
 Dr. Suzana Voiculescu Discipline of Physiology and Fundamental Neurosciences Carol Davila Univ. of Medicine and Pharmacy Definition All the processes inside the body which keep the H+ concentration within
Dr. Suzana Voiculescu Discipline of Physiology and Fundamental Neurosciences Carol Davila Univ. of Medicine and Pharmacy Definition All the processes inside the body which keep the H+ concentration within
Outline Urinary System
 Urinary System and Excretion Bio105 Lecture Packet 20 Chapter 16 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure B. Urine formation 1. Hormonal regulation
Urinary System and Excretion Bio105 Lecture Packet 20 Chapter 16 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure B. Urine formation 1. Hormonal regulation
ACID-BASE BALANCE URINE BLOOD AIR
 ACIDBASE BALANCE URINE BLOOD AIR H 2 PO 4 NH 4 HCO 3 KIDNEY H H HCO 3 CELLS Hb H LUNG H 2 CO 3 HHb CO 2 H 2 O ph = 7.4 [HCO 3 ] = 24 meq/l PCO 2 = 40 mm Hg CO 2 PRIMARY RENAL MECHANISMS INVOLVED IN ACIDBASE
ACIDBASE BALANCE URINE BLOOD AIR H 2 PO 4 NH 4 HCO 3 KIDNEY H H HCO 3 CELLS Hb H LUNG H 2 CO 3 HHb CO 2 H 2 O ph = 7.4 [HCO 3 ] = 24 meq/l PCO 2 = 40 mm Hg CO 2 PRIMARY RENAL MECHANISMS INVOLVED IN ACIDBASE
Chapter 13 The Urinary System
 Biology 12 Name: Urinary System Per: Date: Chapter 13 The Urinary System Complete using BC Biology 12, page 408-435 13.1 The Urinary System pages 412-413 1. As the kidneys produce urine, they carry out
Biology 12 Name: Urinary System Per: Date: Chapter 13 The Urinary System Complete using BC Biology 12, page 408-435 13.1 The Urinary System pages 412-413 1. As the kidneys produce urine, they carry out
Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance
 Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance Question No. 1 of 10 Which of the following statements about the functions of the urinary system
Human Anatomy and Physiology - Problem Drill 23: The Urinary System, Fluid, Electrolyte and Acid-Base Balance Question No. 1 of 10 Which of the following statements about the functions of the urinary system
Outline Urinary System. Urinary System and Excretion. Urine. Urinary System. I. Function II. Organs of the urinary system
 Outline Urinary System Urinary System and Excretion Bio105 Chapter 16 Renal will be on the Final only. I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of
Outline Urinary System Urinary System and Excretion Bio105 Chapter 16 Renal will be on the Final only. I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of
Nephron Function and Urine Formation. Ms. Kula December 1, 2014 Biology 30S
 Nephron Function and Urine Formation Ms. Kula December 1, 2014 Biology 30S The Role of the Nephron In order for the body to properly function and maintain homeostasis, the amount of dissolved substances
Nephron Function and Urine Formation Ms. Kula December 1, 2014 Biology 30S The Role of the Nephron In order for the body to properly function and maintain homeostasis, the amount of dissolved substances
Answers and Explanations
 Answers and Explanations 1. The answer is D [V B 4 b]. Distal K + secretion is decreased by factors that decrease the driving force for passive diffusion of K + across the luminal membrane. Because spironolactone
Answers and Explanations 1. The answer is D [V B 4 b]. Distal K + secretion is decreased by factors that decrease the driving force for passive diffusion of K + across the luminal membrane. Because spironolactone
The Urinary System. BIOLOGY OF HUMANS Concepts, Applications, and Issues. Judith Goodenough Betty McGuire
 BIOLOGY OF HUMANS Concepts, Applications, and Issues Fifth Edition Judith Goodenough Betty McGuire 16 The Urinary System Lecture Presentation Anne Gasc Hawaii Pacific University and University of Hawaii
BIOLOGY OF HUMANS Concepts, Applications, and Issues Fifth Edition Judith Goodenough Betty McGuire 16 The Urinary System Lecture Presentation Anne Gasc Hawaii Pacific University and University of Hawaii
Major intra and extracellular ions Lec: 1
 Major intra and extracellular ions Lec: 1 The body fluids are solutions of inorganic and organic solutes. The concentration balance of the various components is maintained in order for the cell and tissue
Major intra and extracellular ions Lec: 1 The body fluids are solutions of inorganic and organic solutes. The concentration balance of the various components is maintained in order for the cell and tissue
1/3/2008. Karen Burke Priscilla LeMone Elaine Mohn-Brown. Medical-Surgical Nursing Care, 2e Karen Burke, Priscilla LeMone, and Elaine Mohn-Brown
 Medical-Surgical Nursing Care Second Edition Karen Burke Priscilla LeMone Elaine Mohn-Brown Chapter 7 Caring for Clients with Altered Fluid, Electrolyte, or Acid-Base Balance Water Primary component of
Medical-Surgical Nursing Care Second Edition Karen Burke Priscilla LeMone Elaine Mohn-Brown Chapter 7 Caring for Clients with Altered Fluid, Electrolyte, or Acid-Base Balance Water Primary component of
Urinary bladder provides a temporary storage reservoir for urine
 Urinary System Organs Kidney Filters blood, allowing toxins, metabolic wastes, and excess ions to leave the body in urine Urinary bladder provides a temporary storage reservoir for urine Paired ureters
Urinary System Organs Kidney Filters blood, allowing toxins, metabolic wastes, and excess ions to leave the body in urine Urinary bladder provides a temporary storage reservoir for urine Paired ureters
Disorders of Acid-Base Balance
 OpenStax-CNX module: m46413 1 Disorders of Acid-Base Balance OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section,
OpenStax-CNX module: m46413 1 Disorders of Acid-Base Balance OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section,
Acid Base Balance by: Susan Mberenga RN, BSN, MSN
 Acid Base Balance by: Susan Mberenga RN, BSN, MSN Acid Base Balance Refers to hydrogen ions as measured by ph Normal range: 7.35-7.45 Acidosis/acidemia: ph is less than 7.35 Alkalosis/alkalemia: ph is
Acid Base Balance by: Susan Mberenga RN, BSN, MSN Acid Base Balance Refers to hydrogen ions as measured by ph Normal range: 7.35-7.45 Acidosis/acidemia: ph is less than 7.35 Alkalosis/alkalemia: ph is
Urine Formation. Urinary Physiology Urinary Section pages Urine Formation. Glomerular Filtration 4/24/2016
 Urine Formation Urinary Physiology Urinary Section pages 9-17 Filtrate Blood plasma minus most proteins Urine
Urine Formation Urinary Physiology Urinary Section pages 9-17 Filtrate Blood plasma minus most proteins Urine
Physio 12 -Summer 02 - Renal Physiology - Page 1
 Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Urinary System Organization. Urinary System Organization. The Kidneys. The Components of the Urinary System
 Urinary System Organization The Golden Rule: The Job of The Urinary System is to Maintain the Composition and Volume of ECF remember this & all else will fall in place! Functions of the Urinary System
Urinary System Organization The Golden Rule: The Job of The Urinary System is to Maintain the Composition and Volume of ECF remember this & all else will fall in place! Functions of the Urinary System
There are number of parameters which are measured: ph Oxygen (O 2 ) Carbon Dioxide (CO 2 ) Bicarbonate (HCO 3 -) AaDO 2 O 2 Content O 2 Saturation
 Arterial Blood Gases (ABG) A blood gas is exactly that...it measures the dissolved gases in your bloodstream. This provides one of the best measurements of what is known as the acid-base balance. The body
Arterial Blood Gases (ABG) A blood gas is exactly that...it measures the dissolved gases in your bloodstream. This provides one of the best measurements of what is known as the acid-base balance. The body
