Elements for a public summary. VI.2.1 Overview of disease epidemiology
|
|
|
- Harvey Douglas
- 6 years ago
- Views:
Transcription
1 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology The term ocular hypertension usually refers to any situation in which the pressure inside the eye, called intraocular pressure, is higher than normal. Eye pressure is measured in millimeters of mercury (mm Hg). Normal eye pressure ranges from mm Hg. Ocular hypertension is an eye pressure of greater than 21 mm Hg. Ocular hypertension should not be considered a disease by itself. Instead, ocular hypertension is a term that is used to describe individuals who should be observed more closely than the general population for the onset of glaucoma. Recent data on people with ocular hypertension from the Ocular Hypertension Treatment Study have shown that they have an average estimated risk of 10% of developing glaucoma over 5 years. This risk may be decreased to 5% (a 50% decrease in risk) if eye pressure is lowered by medications or laser surgery. However, the risk may become even less than 1% per year because of significantly improved techniques for detecting glaucomatous damage. Glaucoma is the second leading cause of blindness in the world (after cataracts) and the leading cause of blindness among African-Americans. Open-angle glaucoma is the most common type of glaucoma among populations of European or African descent, whereas angle-closure glaucoma is more common among populations of Asian descent. It is estimated that there are 44.7 million people with open-angle glaucoma worldwide in 2010, and that this number will increase to 58.6 million in It is estimated that there are 2.8 million people with open-angle glaucoma in the United States (US) in 2010, and that the number will increase to 3.4 million in The Barbados Eye Study found ocular hypertension present more frequently in women. Mean intraocular pressure (IOP) slowly rises with increasing age. Age older than 40 years is considered a risk factor for the development of ocular hypertension and primary open-angle glaucoma (POAG). Black subjects had almost 3 times the age-adjusted prevalence of glaucoma than white subjects.
2 Aphakia is the absence of the lens of the eye, due to surgical removal, a perforating wound or ulcer, or hereditary anomaly. Short term or permanent increases in intraocular pressure occurs in patients with aphakia, with studies reporting from 6% to 26% of patients developing this problem. The reported incidence varies with the duration of follow-up after cataract surgery. A longer follow-up period has been associated with a higher incidence of glaucoma. Glaucoma can occur at any time after hereditary cataract surgery; therefore, paediatric aphakic patients should be routinely monitored for glaucoma throughout their lives. Secondary glaucoma refers to any form of glaucoma in which there is an identifiable cause of increased eye pressure, resulting in optic nerve damage and vision loss. It may be caused by e.g. an eye injury, inflammation, certain drugs such as steroids, and advanced cases of cataract or diabetes.about 20% of all glaucoma cases may be classed as secondary glaucoma. Glaucomas are a heterogeneous group of eye conditions that may appear from as early as birth to very late age of onset in life. The primary type of glaucomas affecting children and juveniles are less frequent, but their prevalence affecting older people of 70 years progressively increases to ~5%. Primary congenital glaucoma is a rare eye disorder usually manifested at birth or early childhood (before 3 years of age) and accounts for % of total blindness. The only known risk factors of the disease are genetic - consanguinity and affected siblings. The risk of congenital glaucoma in the second child is approximately 5%, and the risk increases to 25% with two affected siblings. Primary juvenile glaucoma is also a rare eye disorder, with an estimated occurrence of 1 per 50,000 persons, when compared in frequency to other types of childhood glaucoma. Juvenileonset glaucoma may appear during the first two decades of life with unknown frequency and is related, like the primary congenital glaucoma, with heredity factors. VI.2.2 Summary of treatment benefits A variety of options are available to treat glaucoma. These include eye drops, laser procedures, and surgery. All are intended to decrease eye pressure and, thereby, protect the optic nerve. Drugs to treat glaucoma are classified by their active ingredient. These include: prostaglandin analogs, beta blockers, alpha agonists, and carbonic anhydrase inhibitors. In addition, combination drugs are available for patients who require more than one type of medication. Beta blockers such as timolol are the second most often used class of medication and work by decreasing production of fluid from the eye. They have systemic side effects that can be minimized by closing the eyes following application or using a technique called punctual occlusion that prevents the drug from entering the tear drainage duct and systemic circulation. In contrast to prostaglandin analogs, they are not associated with changes to the eye itself (e.g. change in iris color and growth of eyelashes). Timolol is also used in combination with a carbonic anhydrase inhibitor (dorzolamide) and also with an alpha agonist (brimonidine) in patients who require more than one type of medication.
3 There are many studies comparing safety and efficacy of timolol maleate with other drugs using to treat glaucoma. Timolol has been shown safe and effective alone or in combination formulations. [Timolol] 2.5 and 5 mg/ml preservative free eye drops, solution does not contain preservative. Thus, there is no risk with contact lenses neither eye irritation that may occur by the preservative himself. VI.2.3 Unknowns relating to treatment benefits Due to limited data in the paediatric population, timolol could only be recommended for use in primary congenital and primary juvenile glaucoma in short term treatment. VI.2.4 Summary of safety concerns Important identified risks Risk What is known Preventability Safety concern in lay language (medical term) Worsening of heart problems (Deterioration of cardiovascular diseases) Patients with poor blood circulation disease (such as Raynaud s disease or Raynaud s syndrome) Brief summary in lay language Inhibition by beta-adrenergic receptor blockade (timolol) may precipitate more severe failure in individuals with a history of severe cardiac diseases such as coronary heart diseases, Prizmental s angina and cardiac failure. In addition, in patients without a history of cardiac failure continued depression of the myocardium with betablocking agents over a period of time can, in some cases, lead to cardiac failure. In cardiac tissues, beta blockade (timolol) causes a reduction in inotropic as well as chronotropic activity, which may further depress cardiac Whether risk can be minimised or mitigated, and how Cardiac failure should be adequately controlled before beginning therapy with the medication. Patients with a history of severe cardiac disease should be watched for signs of cardiac failure and have their pulse rates monitored. Treatment should be discontinued immediately after apparition of symptoms and patient should ask for physician advice.
4 (Peripheral circulatory disorders) Patients with breathing problems, asthma or chronic obstructive pulmonary disease (Bronchospasm) Very low level of blood sugar, diabetes (Acute hypoglycaemia) Overactivity of the thyroid gland (Masking signs) hyperthyroidism output and blood pressure in patients with peripheral circulatory disorders. Timolol is a drug widely used for lowering increased intaocular pressure. It is an effective treatment mode, but as much as 80% of the ophthalmic dose can be absorbed into the systemic circulation. This may cause adverse systemic effects such as bradycardia, lowering of blood pressure, constriction of the airways in the lungs (predominantly in patients with pre-existing disease), difficulty breathing, shortness of breath, wheezing, cough. Beta-adrenergic receptor blocking agents may mask symptoms of hypoglycaemia such as tremors, tachycardia and blood pressure changes. In addition, the nonselective beta-blockers (e.g., propranolol, pindolol, timolol) may potentiate insulin-induced hypoglycaemia and suspending the recovery of normal blood glucose levels. Beta-adrenergic receptor blocking agents (such as timolol) are used to alleviate symptoms of hyperthyroidism such as tachycardia, anxiety, tremor and heat intolerance. Thus, use of timolol eye drops, because of it systemic absorption, may mask hyperthyroidism symptoms. Complications due to hyperthyroidism, such heart problems, brittle bones, thyrotoxic crisis, myasthenia gravis Medication should be used with caution, in patients with mild/moderate chronic obstructive pulmonary disease (COPD) as bronchospasm may be life-threatening in patients with asthma. Treatment should be discontinued immediately after apparition of symptoms and patient should ask for physician advice. Timolol should be administered with caution in patients subject to spontaneous hypoglycaemia or to patients with labile diabetes Physician should be aware if patient had or has overactivity of the thyroid gland
5 Irritation of the eye - dry eyes (Corneal toxicity dry eye) Abnormality in heart s rhythm (Conduction abnormality) Problem in the surface layer of the eye, eye surgery (Choroidal detachment) Signs and symptoms of eye irritation (e.g. burning, stinging, itching, tearing, redness), inflammation of the eyelid, inflammation in the cornea, blurred vision, decreased corneal sensitivity, dry eyes, corneal erosion (damage to the front layer of the eyeball), drooping of the upper eyelid (making the eye stay half closed) double vision, sensitivity to light, discharge from the eye, pain in the eye, have been reported Ophthalmic timolol can induce atrioventricular block, sinus bradycardia demonstrated in ECG, and expressed as syncope or dizziness. The most of them are reversible. Choroidal detachment is the separation of the choroid from the sclera of the eye as a result of leakage of fluid from the vessels of the choroid. It occurs when pressure inside the eyeball is very low, usually after trauma or intraocular surgery. Ophthalmologist should evaluate the risks and benefits of ophthalmic medications before initiating therapy, identify the minimum dosages necessary to achieve a therapeutic benefit, and monitor patients for local and systemic adverse effects Physician should be known if patient low blood pressure. Beta-blocker such as timolol, should not administer with a category of drugs named calcium-channel blockers Choroidal detachment cases have been reported following timolol treatment. Management of eyes with chronic or recurrent choroidal detachment should include stopping all forms of aqueous suppressant therapy. Timolol administration together with adrenaline (Co-administration with adrenaline) In case of surgical anaesthesia, both the anesthesiologist and the ophthalmologist must be aware that eyedrops are readily absorbed through hyperemic incised conjunctivae. Although small in volume, these drops contain highly concentrated medication that can produce systemic results. The anaesthesiologist should be informed when the patient is receiving timolol.
6 Allergic reaction (Hypersensitivity to any allergen) Beta-blockers, such as timolol, in patients with history of atopy or a history of severe anaphylactic reaction to a variety of allergens may be more reactive to repeated challenge with such allergens. Treatment should be discontinued immediately after apparition of an allergic symptom. Patient should ask physician advice. Appearance of calcium on the cornea (Corneal calcification) Allergic reaction such as swelling beneath the skin that can occur in areas such as the face and limbs, and can obstruct the airway which may cause difficulty swallowing or breathing, hives or itchy rash, localized and generalized rash, itchiness may occur Severe sudden life-threatening has been recorded but their frequency (not known) cannot be established from the available data at the moment. Phosphates are widely used as part of the buffering system in ophthalmic drop preparations. However, recent studies have shown that phosphate buffer play a role in the process of corneal calcification. Serious ocular diseases, such as band keratopathy (a corneal disease) derived from the appearance of calcium on the central cornea may occur Cases of corneal calcification have been reported very rarely in association with the use of phosphate eye containing eye drops in some patients with significantly damaged corneas. Thus, of phosphate-buffered medications should be used cautionally. Important potential risks Risk Bacterial keratitis What is known (Including reason why it is considered a potential risk) Bacterial keratitis is an infection of the cornea (the clear, round dome covering the eye's iris and pupil) that causes pain, reduced vision, light sensitivity and tearing or discharge from the eye. It is an important cause of visual loss. The aetiology is diverse: bacterial, fungal, viral, protozoal or it may be polymicrobial in nature. Prescribed topical ocular medications
7 can become contaminated and result in bacterial keratitis especially gram-negative infection with Pseudomonas, Serratia and Proteus species. Missing information Risk No adequate and well controlled studies in pregnant women What is known There are no adequate data for the use of timolol in pregnant women. Timolol should not be used during pregnancy unless clearly necessary. Systemic absorption of topically administered β-blockers can be reduced by nasolacrimal occlusion and by keeping the eyes closed as long as possible (e.g. for 3-5 minutes) after instillation of drops. Epidemiological studies have not revealed malformative effects but show a risk for intra uterine growth retardation when betablockers are administered by the oral route. In addition, signs and symptoms of beta-blockade (e.g. bradycardia, hypotension, respiratory distress and hypoglycaemia) have been observed in the neonate when beta-blockers have been administered until delivery. If timolol is administered until delivery, the neonate should be carefully monitored during the first days of life. No studies in patients wearing contact lenses [Invented name] has not been studied in patients using contact lenses. [Invented name] should not be used while using soft contact lenses. The lenses should only be inserted 15 minutes after the drip. Exposure in paediatric patients There is only very limited data available on the use of Timolol (2.5, 5 mg/ml twice daily one drop) in the paediatric population. In one small, double masked, randomized, published clinical study conducted for a treatment period of up to 12 weeks on 105 children (n=71 on Timolol) aged 12 days 5 years the data have shown to some extent evidence, that Timolol in the indication primary congenital and primary juvenile glaucoma is effective in short term treatment. Thus, timolol could only be recommended for use in primary congenital and primary juvenile glaucoma for a transitional period while a decision is made on a surgical approach and in case of failed surgery while awaiting further options. VI.2.5 Summary of risk minimisation measures by safety concern
8 All medicines have a Summary of Product Characteristics (SmPC) which provides physicians, pharmacists and other health care professionals with details on how to use the medicine, the risks and recommendations for minimising them. An abbreviated version of this in lay language is provided in the form of the package leaflet (PL). The measures in these documents are known as routine risk minimisation measures. This medicine has no additional risk minimisation measures. VI.2.6 Planned post authorisation development plan Not applicable VI.2.7 Summary of changes to the risk management plan over time Version Date Safety concerns Change Important identified risks Deterioration of cardiovascular diseases Peripheral circulatory disorders Bronchospasm Acute hypoglycaemia Masking Hyperthyroidism signs Corneal toxicity dry eye Conduction abnormality adverse events Choroidal detachment Co-administration with epinephrine Hypersensitivity to any allergen Corneal calcification Important potential risks Bacterial keratinitis associated with the use of the multidose containers of topical ophthalmic Initial version
9 products Usage of medicinal product longer than 28 days after first opening the container Missing information No adequate and well controlled studies in pregnant women No studies in patients wearing contact lenses Exposure in paediatric patients Important identified risks Deterioration of cardiovascular diseases Peripheral circulatory disorders Bronchospasm Acute hypoglycaemia Masking Hyperthyroidism signs Corneal toxicity dry eye Conduction abnormality adverse events Choroidal detachment Co-administration with epinephrine Hypersensitivity to any allergen Corneal calcification Bacterial keratitis Important potential risks None Missing information Use in pregnant women No studies in patients wearing contact lenses Implementation of day 106 answers
10 Exposure in paediatric patients Important identified risks Deterioration of cardiovascular diseases Peripheral circulatory disorders Bronchospasm Acute hypoglycaemia Masking Hyperthyroidism signs Corneal toxicity dry eye Conduction abnormality adverse events Choroidal detachment Co-administration with adrenaline Hypersensitivity to any allergen Corneal calcification Important potential risks Bacterial keratitis Implementation of day 160 answers Missing information Use in pregnant women No studies in patients wearing contact lenses Exposure in paediatric patients
Pharmaceutical form(s)/strength: Solution: 5 mg/ml Suspensions: 2.5 and 5 mg/ml P-RMS:
 0BCore Safety Profile Active substance: Betaxolol eyedrops Pharmaceutical form(s)/strength: Solution: 5 mg/ml Suspensions: 2.5 and 5 mg/ml P-RMS: HU/H/PSUR/0010/002 Date of FAR: 20.03.2013 4.2 Posology
0BCore Safety Profile Active substance: Betaxolol eyedrops Pharmaceutical form(s)/strength: Solution: 5 mg/ml Suspensions: 2.5 and 5 mg/ml P-RMS: HU/H/PSUR/0010/002 Date of FAR: 20.03.2013 4.2 Posology
Core Safety Profile. Pharmaceutical form(s)/strength: Sterile eye drops 1%, 2% Date of FAR:
 Core Safety Profile Active substance: Carteolol Pharmaceutical form(s)/strength: Sterile eye drops 1%, 2% P - RMS: SK/H/PSUR/0002/002 Date of FAR: 16.03.2012 4.1 THERAPEUTIC INDICATIONS Ocular hypertension
Core Safety Profile Active substance: Carteolol Pharmaceutical form(s)/strength: Sterile eye drops 1%, 2% P - RMS: SK/H/PSUR/0002/002 Date of FAR: 16.03.2012 4.1 THERAPEUTIC INDICATIONS Ocular hypertension
Brimonidine/timolol Version 1.2, 16Oct, Elements for a public summary. VI.2.1 Overview of disease epidemiology
 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology The estimated number of people with vision loss from glaucoma range from 5.2 to 6.7 million. This is approximately 10% of the
VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology The estimated number of people with vision loss from glaucoma range from 5.2 to 6.7 million. This is approximately 10% of the
Elements for a Public Summary. Overview of disease epidemiology
 VI.2 VI.2.1 Elements for a Public Summary Overview of disease epidemiology Epidemiology of the disease Incidence and prevalence Increased pressure in the eye occurs in more than 100 million people, and
VI.2 VI.2.1 Elements for a Public Summary Overview of disease epidemiology Epidemiology of the disease Incidence and prevalence Increased pressure in the eye occurs in more than 100 million people, and
TIMALEN 0.5% eye drops
 PACKAGE LEAFLET: INFORMATION FOR THE USER TIMALEN 0.5% eye drops TIMOLOL (AS TIMOLOL MALEATE) This leaflet is a copy of the Summary of Product Characteristics and Patient Information Leaflet for a medicine,
PACKAGE LEAFLET: INFORMATION FOR THE USER TIMALEN 0.5% eye drops TIMOLOL (AS TIMOLOL MALEATE) This leaflet is a copy of the Summary of Product Characteristics and Patient Information Leaflet for a medicine,
Summary of the risk management plan (RMP) for Izba (travoprost)
 EMA/14138/2014 Summary of the risk management plan (RMP) for Izba (travoprost) This is a summary of the risk management plan (RMP) for Izba, which details the measures to be taken in order to ensure that
EMA/14138/2014 Summary of the risk management plan (RMP) for Izba (travoprost) This is a summary of the risk management plan (RMP) for Izba, which details the measures to be taken in order to ensure that
Elements for a public summary. Overview of disease epidemiology
 VI.2 VI.2.1 Elements for a public summary Overview of disease epidemiology Glaucoma is an eye disease that can result in damage to the optic nerve and loss of vision (blindness). It is the major cause
VI.2 VI.2.1 Elements for a public summary Overview of disease epidemiology Glaucoma is an eye disease that can result in damage to the optic nerve and loss of vision (blindness). It is the major cause
[TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN. TRAVOPR-v
![[TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN. TRAVOPR-v [TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN. TRAVOPR-v](/thumbs/94/119684929.jpg) [TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN TRAVOPR-v2-270214 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Studies estimated that 3-6 million people
[TRAVOPROST] 40 MICROGRAMS/ML, EYE DROPS, SOLUTION RISK MANAGEMENT PLAN TRAVOPR-v2-270214 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Studies estimated that 3-6 million people
BETAGAN Allergan Levobunolol HCl Glaucoma Therapy
 BETAGAN Allergan Levobunolol HCl Glaucoma Therapy Action And Clinical Pharmacology: Levobunolol is a noncardioselective beta- adrenoceptor antagonist, equipotent at both beta1 and beta2 receptors. Levobunolol
BETAGAN Allergan Levobunolol HCl Glaucoma Therapy Action And Clinical Pharmacology: Levobunolol is a noncardioselective beta- adrenoceptor antagonist, equipotent at both beta1 and beta2 receptors. Levobunolol
Proposed PIL NL/H/0653/001/IB/024/G. MONOFREE DEXAMETHASON 1 mg/ml, eye drops, solution in single-dose container Dexamethasone phosphate
 Proposed PIL NL/H/0653/001/IB/024/G PACKAGE LEAFLET: INFORMATION FOR THE USER MONOFREE DEXAMETHASON 1 mg/ml, eye drops, solution in single-dose container Dexamethasone phosphate Read all of this leaflet
Proposed PIL NL/H/0653/001/IB/024/G PACKAGE LEAFLET: INFORMATION FOR THE USER MONOFREE DEXAMETHASON 1 mg/ml, eye drops, solution in single-dose container Dexamethasone phosphate Read all of this leaflet
Package leaflet: Information for the user XATABLOC. 50 micrograms/ml + 5 mg/ml eye drops, solution latanoprost / timolol
 Package leaflet: Information for the user XATABLOC 50 micrograms/ml + 5 mg/ml eye drops, solution latanoprost / timolol Read all of this leaflet carefully before you start using this medicine because it
Package leaflet: Information for the user XATABLOC 50 micrograms/ml + 5 mg/ml eye drops, solution latanoprost / timolol Read all of this leaflet carefully before you start using this medicine because it
VI.2.2 Summary of treatment benefits
 EU-Risk Management Plan for Bimatoprost V01 aetiology), both OAG and ACG can be secondary conditions. Secondary glaucoma refers to any case in which another disorder (e.g. injury, inflammation, vascular
EU-Risk Management Plan for Bimatoprost V01 aetiology), both OAG and ACG can be secondary conditions. Secondary glaucoma refers to any case in which another disorder (e.g. injury, inflammation, vascular
Elements for a public summary. VI.2.1 Overview of disease epidemiology
 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Studies estimated that 3-6 million people in the United States alone, including 4-10% of the population older than 40 years, have
VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Studies estimated that 3-6 million people in the United States alone, including 4-10% of the population older than 40 years, have
BRIMOPRESS-T EYE DROPS. COMPOSITION Each ml of BRIMOPRESS-T EYE DROPS contains. Brimonidine Tartarate 0.2% Timolol Maleate 0.5%
 BRIMOPRESS-T EYE DROPS COMPOSITION Each ml of BRIMOPRESS-T EYE DROPS contains Brimonidine Tartarate 0.2% Timolol Maleate 0.5% BRIMOPRESS-T ophthalmic solution is a combination of Brimonidine Tartrate,
BRIMOPRESS-T EYE DROPS COMPOSITION Each ml of BRIMOPRESS-T EYE DROPS contains Brimonidine Tartarate 0.2% Timolol Maleate 0.5% BRIMOPRESS-T ophthalmic solution is a combination of Brimonidine Tartrate,
This leaflet's format was set and its contents inspected and approved by the Ministry of Health
 This leaflet's format was set and its contents inspected and approved by the Ministry of Health Patient leaflet in accordance with the Pharmacists Regulation (Preparations) 1986 The preparation shall be
This leaflet's format was set and its contents inspected and approved by the Ministry of Health Patient leaflet in accordance with the Pharmacists Regulation (Preparations) 1986 The preparation shall be
Summary of the risk management plan (RMP) for Omidria (phenylephrine / ketorolac)
 EMA/368070/2015 Summary of the risk management plan (RMP) for Omidria (phenylephrine / ketorolac) This is a summary of the risk management plan (RMP) for Omidria, which details the measures to be taken
EMA/368070/2015 Summary of the risk management plan (RMP) for Omidria (phenylephrine / ketorolac) This is a summary of the risk management plan (RMP) for Omidria, which details the measures to be taken
Patient Information COSOPT PF (CO-sopt PEA EHF) (dorzolamide hydrochloride-timolol maleate ophthalmic solution) 2%/0.5%
 Patient Information COSOPT PF (CO-sopt PEA EHF) (dorzolamide hydrochloride-timolol maleate ophthalmic solution) 2%/0.5% Read this information before you start using COSOPT PF and each time you get a refill.
Patient Information COSOPT PF (CO-sopt PEA EHF) (dorzolamide hydrochloride-timolol maleate ophthalmic solution) 2%/0.5% Read this information before you start using COSOPT PF and each time you get a refill.
Package leaflet: Information for the patient BETAXOLOL 0.5% EYE DROPS. Betaxolol (as hydrochloride)
 Package leaflet: Information for the patient BETAXOLOL 0.5% EYE DROPS Betaxolol (as hydrochloride) Read all of this leaflet carefully before you starts to using this medicine because it contains important
Package leaflet: Information for the patient BETAXOLOL 0.5% EYE DROPS Betaxolol (as hydrochloride) Read all of this leaflet carefully before you starts to using this medicine because it contains important
D90 (27/10/2005) Final SmPC NL/H/653/01
 1/6 1. NAME OF THE MEDICINAL PRODUCT MONOFREE DEXAMETHASON 1 mg/ml, eye drops, solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 1 mg of dexamethasone phosphate as dexamethasone
1/6 1. NAME OF THE MEDICINAL PRODUCT MONOFREE DEXAMETHASON 1 mg/ml, eye drops, solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 1 mg of dexamethasone phosphate as dexamethasone
INDICATIONS ACULAR 0,5 % is indicated for the relief of inflammation following ocular surgery.
 Page 1 of 5 SCHEDULING STATUS Schedule 3 PROPRIETARY NAME (AND DOSAGE FORM) ACULAR 0,5 % COMPOSITION ACULAR 0,5 % contains: Preservatives: Benzalkonium chloride 0,01 % m/v Disodium edetate 0,1 % m/v PHARMACOLOGICAL
Page 1 of 5 SCHEDULING STATUS Schedule 3 PROPRIETARY NAME (AND DOSAGE FORM) ACULAR 0,5 % COMPOSITION ACULAR 0,5 % contains: Preservatives: Benzalkonium chloride 0,01 % m/v Disodium edetate 0,1 % m/v PHARMACOLOGICAL
CONSUMER INFORMATION. Sandoz Travoprost / Timolol PQ ophthalmic solution travoprost and timolol ophthalmic solution with POLYQUAD * as preservative
 CONSUMER INFORMATION Pr Sandoz Travoprost / Timolol PQ ophthalmic solution travoprost and timolol ophthalmic solution with POLYQUAD * as preservative This leaflet is designed specifically for Consumers.
CONSUMER INFORMATION Pr Sandoz Travoprost / Timolol PQ ophthalmic solution travoprost and timolol ophthalmic solution with POLYQUAD * as preservative This leaflet is designed specifically for Consumers.
Glaucoma. How is Glaucoma Diagnosed? Glaucoma Testing
 Glaucoma How is Glaucoma Diagnosed? Glaucoma Testing There is no single test for glaucoma. The diagnosis is made by evaluating the patient from a number of perspectives, using specialized instruments.
Glaucoma How is Glaucoma Diagnosed? Glaucoma Testing There is no single test for glaucoma. The diagnosis is made by evaluating the patient from a number of perspectives, using specialized instruments.
Package leaflet: Information for the user. GANFORT 0.3 mg/ml + 5 mg/ml eye drops, solution Bimatoprost/timolol
 Package leaflet: Information for the user GANFORT 0.3 mg/ml + 5 mg/ml eye drops, solution Bimatoprost/timolol Read all of this leaflet carefully before you start using this medicine because it contains
Package leaflet: Information for the user GANFORT 0.3 mg/ml + 5 mg/ml eye drops, solution Bimatoprost/timolol Read all of this leaflet carefully before you start using this medicine because it contains
Glaucoma. What is glaucoma? Eye Words to Know. What causes glaucoma?
 2014 2015 Glaucoma What is glaucoma? Glaucoma is a disease that damages your eye s optic nerve. It usually happens when fluid builds up in the front part of your eye. That extra fluid increases the pressure
2014 2015 Glaucoma What is glaucoma? Glaucoma is a disease that damages your eye s optic nerve. It usually happens when fluid builds up in the front part of your eye. That extra fluid increases the pressure
Package leaflet: Information for the user. GANFORT 0.3 mg/ml + 5 mg/ml eye drops, solution, in single-dose container Bimatoprost/timolol
 Package leaflet: Information for the user GANFORT 0.3 mg/ml + 5 mg/ml eye drops, solution, in single-dose container Bimatoprost/timolol Read all of this leaflet carefully before you start using this medicine
Package leaflet: Information for the user GANFORT 0.3 mg/ml + 5 mg/ml eye drops, solution, in single-dose container Bimatoprost/timolol Read all of this leaflet carefully before you start using this medicine
PATIENT INFORMATION LEAFLET
 Page 1 PATIENT INFORMATION LEAFLET Please read this leaflet carefully before using COMBIGAN. Keep this leaflet. You may need to read it again. If you have any further questions, please ask your doctor
Page 1 PATIENT INFORMATION LEAFLET Please read this leaflet carefully before using COMBIGAN. Keep this leaflet. You may need to read it again. If you have any further questions, please ask your doctor
HIGHLIGHTS OF PRESCRIBING INFORMATION WARNINGS AND PRECAUTIONS
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use BETOPTIC S safely and effectively. See full prescribing information for BETOPTIC S. BETOPTIC S Ophthalmic
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use BETOPTIC S safely and effectively. See full prescribing information for BETOPTIC S. BETOPTIC S Ophthalmic
Summary of the risk management plan (RMP) for Duaklir Genuair (aclidinium / formoterol fumarate dihydrate)
 EMA/605453/2014 Summary of the risk management plan (RMP) for Duaklir Genuair (aclidinium / formoterol fumarate dihydrate) This is a summary of the risk management plan (RMP) for Duaklir Genuair, which
EMA/605453/2014 Summary of the risk management plan (RMP) for Duaklir Genuair (aclidinium / formoterol fumarate dihydrate) This is a summary of the risk management plan (RMP) for Duaklir Genuair, which
East and North Hertfordshire treatment pathway for primary open angle glaucoma and ocular hypertension in adults
 East and North Hertfordshire treatment pathway for primary open angle glaucoma and ocular hypertension in adults Introduction Glaucoma is a group of eye diseases causing optic nerve damage. In most cases
East and North Hertfordshire treatment pathway for primary open angle glaucoma and ocular hypertension in adults Introduction Glaucoma is a group of eye diseases causing optic nerve damage. In most cases
PATIENT INFORMATION LEAFLET. PROPRIETARY NAME, STRENGTH AND PHARMACEUTICAL FORM LUMIGAN 0,01 %, bimatoprost 0,1 mg/ml eye drops
 Page 1 of 6 SCHEDULING STATUS Schedule 4 PATIENT INFORMATION LEAFLET PROPRIETARY NAME, STRENGTH AND PHARMACEUTICAL FORM LUMIGAN 0,01 %, bimatoprost 0,1 mg/ml eye drops Read all of this leaflet carefully
Page 1 of 6 SCHEDULING STATUS Schedule 4 PATIENT INFORMATION LEAFLET PROPRIETARY NAME, STRENGTH AND PHARMACEUTICAL FORM LUMIGAN 0,01 %, bimatoprost 0,1 mg/ml eye drops Read all of this leaflet carefully
Dorzolamide 20 mg/ml Eye Drops, Solution
 PACKAGE LEAFLET: INFORMATION FOR THE USER Dorzolamide 20 mg/ml Eye Drops, Solution (Dorzolamide hydrochloride) Read all of this leaflet carefully before you start using this medicine because it contains
PACKAGE LEAFLET: INFORMATION FOR THE USER Dorzolamide 20 mg/ml Eye Drops, Solution (Dorzolamide hydrochloride) Read all of this leaflet carefully before you start using this medicine because it contains
7.2 Part VI.2 Elements for a Public Summary
 7.2 Part VI.2 Elements for a Public Summary 7.2.1 Part VI.2.1 Overview of disease epidemiology Chronic obstructive pulmonary disease (COPD) is characterized by persistent airflow limitation that is usually
7.2 Part VI.2 Elements for a Public Summary 7.2.1 Part VI.2.1 Overview of disease epidemiology Chronic obstructive pulmonary disease (COPD) is characterized by persistent airflow limitation that is usually
Elements for a public summary
 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology High blood pressure, also known as hypertension, occurs in a large percentage of the adult population. Primary (essential) hypertension
VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology High blood pressure, also known as hypertension, occurs in a large percentage of the adult population. Primary (essential) hypertension
Package Leaflet: Information for the user. LUMIGAN 0.3 mg/ml, eye drops, solution, in single-dose container Bimatoprost
 Package Leaflet: Information for the user LUMIGAN 0.3 mg/ml, eye drops, solution, in single-dose container Bimatoprost Read all of this leaflet carefully before you start using this medicine because it
Package Leaflet: Information for the user LUMIGAN 0.3 mg/ml, eye drops, solution, in single-dose container Bimatoprost Read all of this leaflet carefully before you start using this medicine because it
Package leaflet: Information for the user. COMBIGAN 2 mg/ml + 5 mg/ml eye drops, solution Brimonidine tartrate and timolol
 Package leaflet: Information for the user COMBIGAN 2 mg/ml + 5 mg/ml eye drops, solution Brimonidine tartrate and timolol Read all of this leaflet carefully before you start using this medicine because
Package leaflet: Information for the user COMBIGAN 2 mg/ml + 5 mg/ml eye drops, solution Brimonidine tartrate and timolol Read all of this leaflet carefully before you start using this medicine because
INDICATIONS For steroid responsive inflammation of the palpebral and bulbar conjunctiva, cornea, and anterior segment of the eye globe.
 Page 1 of 5 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME AND DOSAGE FORM PRED FORTE Sterile Eye Suspension COMPOSITION PRED FORTE Sterile Eye Suspension contains: Prednisolone acetate 10 mg/ml Preservative:
Page 1 of 5 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME AND DOSAGE FORM PRED FORTE Sterile Eye Suspension COMPOSITION PRED FORTE Sterile Eye Suspension contains: Prednisolone acetate 10 mg/ml Preservative:
YOUR VYZULTA TREATMENT GUIDE. Please see Important Safety Information on pages 1, 9, 10, 17 and 18. Please see accompanying Prescribing Information.
 YOUR VYZULTA TREATMENT GUIDE Please see Important Safety Information on pages 1, 9, 10, 17 and 18. Please see accompanying Prescribing Information. INDICATION VYZULTA TM (latanoprostene bunod ophthalmic
YOUR VYZULTA TREATMENT GUIDE Please see Important Safety Information on pages 1, 9, 10, 17 and 18. Please see accompanying Prescribing Information. INDICATION VYZULTA TM (latanoprostene bunod ophthalmic
Core Safety Profile. Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg. Date of FAR:
 Core Safety Profile Active substance: Bisoprolol Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg P - RMS: FI/H/PSUR/0002/002 Date of FAR: 13.12.2011
Core Safety Profile Active substance: Bisoprolol Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg P - RMS: FI/H/PSUR/0002/002 Date of FAR: 13.12.2011
Medical Treatment in Pediatric Glaucoma
 Medical Treatment in Pediatric Glaucoma By Nader Bayoumi, MD Lecturer of Ophthalmology Ophthalmology Department Alexandria University Alexandria, Egypt ESG 2012 Pediatric glaucoma is a surgical disease
Medical Treatment in Pediatric Glaucoma By Nader Bayoumi, MD Lecturer of Ophthalmology Ophthalmology Department Alexandria University Alexandria, Egypt ESG 2012 Pediatric glaucoma is a surgical disease
Treatments for Open-Angle Glaucoma. A Review of the Research for Adults
 Treatments for Open-Angle Glaucoma A Review of the Research for Adults Is This Information Right for Me? Yes, this information is for you if: Your eye doctor has said that you have open-angle glaucoma,
Treatments for Open-Angle Glaucoma A Review of the Research for Adults Is This Information Right for Me? Yes, this information is for you if: Your eye doctor has said that you have open-angle glaucoma,
TIMOPTIC TIMOPTIC-XE Merck Frosst Timolol Maleate Elevated Intraocular Pressure Therapy
 TIMOPTIC TIMOPTIC-XE Merck Frosst Timolol Maleate Elevated Intraocular Pressure Therapy Action and Clinical Timolol is a general beta-adrenergic receptor blocking agent that does not have significant intrinsic
TIMOPTIC TIMOPTIC-XE Merck Frosst Timolol Maleate Elevated Intraocular Pressure Therapy Action and Clinical Timolol is a general beta-adrenergic receptor blocking agent that does not have significant intrinsic
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Dexafree 1 mg/ml, eye drops, solution in single-dose container 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 1 mg
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Dexafree 1 mg/ml, eye drops, solution in single-dose container 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 1 mg
Scrub In. What is the function of vitreous humor? What does the pupil do when exposed to bright light? a. Maintain eye shape and provide color vision
 Scrub In What is the function of vitreous humor? a. Maintain eye shape and provide color vision b. Maintain eye shape and refract light rays c. Provide night vision and color vision d. Provide night vision
Scrub In What is the function of vitreous humor? a. Maintain eye shape and provide color vision b. Maintain eye shape and refract light rays c. Provide night vision and color vision d. Provide night vision
Package leaflet: Information for the patient. Brimonidine Biogaran 2 mg/ml eye drops, solution. brimonidine tartrate
 Package leaflet: Information for the patient Brimonidine Biogaran 2 mg/ml eye drops, solution brimonidine tartrate Read all of this leaflet carefully before you start using this medicine because it contains
Package leaflet: Information for the patient Brimonidine Biogaran 2 mg/ml eye drops, solution brimonidine tartrate Read all of this leaflet carefully before you start using this medicine because it contains
Ophthalmology. Glaucoma
 Ophthalmology Glaucoma The Ophthalmology service offers the latest and most comprehensive eye care for patients. With a dedicated team of eye surgeons and consultants, we treat vision problems ranging
Ophthalmology Glaucoma The Ophthalmology service offers the latest and most comprehensive eye care for patients. With a dedicated team of eye surgeons and consultants, we treat vision problems ranging
Summary of the risk management plan (RMP) for Budesonide/Formoterol Teva (budesonide / formoterol)
 EMA/639304/2014 Summary of the risk management plan (RMP) for Budesonide/Formoterol Teva (budesonide / formoterol) This is a summary of the risk management plan (RMP) for Budesonide/Formoterol Teva, which
EMA/639304/2014 Summary of the risk management plan (RMP) for Budesonide/Formoterol Teva (budesonide / formoterol) This is a summary of the risk management plan (RMP) for Budesonide/Formoterol Teva, which
Timolol 0.25% w/v Eye Drops Solution contains timolol maleate equivalent to 0.25% w/v solution of timolol with preservative.
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Timolol 0.25% w/v Eye Drops Solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Timolol 0.25% w/v Eye Drops Solution contains timolol
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Timolol 0.25% w/v Eye Drops Solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Timolol 0.25% w/v Eye Drops Solution contains timolol
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT GANFORT 0.3 mg/ml + 5 mg/ml eye drops, solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One ml of solution contains 0.3
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT GANFORT 0.3 mg/ml + 5 mg/ml eye drops, solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One ml of solution contains 0.3
MAXITROL Eye Ointment
 MAXITROL Dexamethasone, Neomycin and Polymyxin B CONSUMER MEDICINE INFORMATION What is in this leaflet Read this leaflet carefully before you start to use Maxitrol. This leaflet answers some common questions
MAXITROL Dexamethasone, Neomycin and Polymyxin B CONSUMER MEDICINE INFORMATION What is in this leaflet Read this leaflet carefully before you start to use Maxitrol. This leaflet answers some common questions
SCHEDULING STATUS Schedule 4 PROPRIETARY NAME AND DOSAGE FORM
 Page 1 of 5 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME AND DOSAGE FORM FML Liquifilm Sterile Eye Suspension COMPOSITION FML Liquifilm Sterile Eye Suspension contains: Fluorometholone 1,0 mg/ml Liquifilm
Page 1 of 5 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME AND DOSAGE FORM FML Liquifilm Sterile Eye Suspension COMPOSITION FML Liquifilm Sterile Eye Suspension contains: Fluorometholone 1,0 mg/ml Liquifilm
Package Leaflet: Information for the user. Xalacom 50 micrograms/ml and 5 mg/ml Eye Drops, Solution Latanoprost + Timolol
 Package Leaflet: Information for the user Xalacom 50 micrograms/ml and 5 mg/ml Eye Drops, Solution Latanoprost + Timolol Read all of this leaflet carefully before you start using this medicine because
Package Leaflet: Information for the user Xalacom 50 micrograms/ml and 5 mg/ml Eye Drops, Solution Latanoprost + Timolol Read all of this leaflet carefully before you start using this medicine because
Summary of the risk management plan (RMP) for Vylaer Spiromax (budesonide / formoterol)
 EMA/675937/2014 Summary of the risk management plan (RMP) for Vylaer Spiromax (budesonide / formoterol) This is a summary of the risk management plan (RMP) for Vylaer Spiromax, which details the measures
EMA/675937/2014 Summary of the risk management plan (RMP) for Vylaer Spiromax (budesonide / formoterol) This is a summary of the risk management plan (RMP) for Vylaer Spiromax, which details the measures
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. T-LO Timolol Ophthalmic Solution (0.25%, 0.
 READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr T-LO Timolol Ophthalmic Solution (0.25%, 0.5% w/v) Pr TIMOLOL MALEATE-EX Timolol Ophthalmic Gel Forming Solution
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr T-LO Timolol Ophthalmic Solution (0.25%, 0.5% w/v) Pr TIMOLOL MALEATE-EX Timolol Ophthalmic Gel Forming Solution
LOWPROST Eye Drops (Bimatoprost Ophthalmic Solution 0.01%)
 Published on: 14 Apr 2017 LOWPROST Eye Drops (Bimatoprost Ophthalmic Solution 0.01%) For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only Composition Each ml contains: Bimatoprost...
Published on: 14 Apr 2017 LOWPROST Eye Drops (Bimatoprost Ophthalmic Solution 0.01%) For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only Composition Each ml contains: Bimatoprost...
PATIENT INFORMATION LEAFLET
 Page 1 of 6 PATIENT INFORMATION LEAFLET SCHEDULING STATUS Schedule 4 Proprietary name, Strength and Pharmaceutical Form LUMIGAN 0,03 % Eye Drops (each ml contains bimatoprost 0,3 mg) Read all of this leaflet
Page 1 of 6 PATIENT INFORMATION LEAFLET SCHEDULING STATUS Schedule 4 Proprietary name, Strength and Pharmaceutical Form LUMIGAN 0,03 % Eye Drops (each ml contains bimatoprost 0,3 mg) Read all of this leaflet
Vision Health: Conditions, Disorders & Treatments GLAUCOMA
 Vision Health: Conditions, Disorders & Treatments GLAUCOMA Glaucoma is a disease of the optic nerve, which transmits the images you see from the eye to the brain. The optic nerve is made up of many nerve
Vision Health: Conditions, Disorders & Treatments GLAUCOMA Glaucoma is a disease of the optic nerve, which transmits the images you see from the eye to the brain. The optic nerve is made up of many nerve
PRODUCT INFORMATION NAME OF THE MEDICINE DESCRIPTION PHARMACOLOGY. BETOPTIC (betaxolol hydrochloride) Eye Drops 0.5%
 PRODUCT INFORMATION BETOPTIC (betaxolol hydrochloride) Eye Drops 0.5% BETOPTIC S (betaxolol hydrochloride) Eye Drops 0.25% NAME OF THE MEDICINE BETOPTIC Eye Drops 0.5% and BETOPTIC S Eye Drops 0.25% contain
PRODUCT INFORMATION BETOPTIC (betaxolol hydrochloride) Eye Drops 0.5% BETOPTIC S (betaxolol hydrochloride) Eye Drops 0.25% NAME OF THE MEDICINE BETOPTIC Eye Drops 0.5% and BETOPTIC S Eye Drops 0.25% contain
Package leaflet: Information for the user. DuoTrav 40 micrograms/ml + 5 mg/ml eye drops, solution travoprost/timolol
 Package leaflet: Information for the user DuoTrav 40 micrograms/ml + 5 mg/ml eye drops, solution travoprost/timolol Read all of this leaflet carefully before you start using this medicine because it contains
Package leaflet: Information for the user DuoTrav 40 micrograms/ml + 5 mg/ml eye drops, solution travoprost/timolol Read all of this leaflet carefully before you start using this medicine because it contains
INDICATIONS ACULAR 0,4% ophthalmic solution is indicated for the reduction of ocular pain and burning/stinging following corneal refractive surgery.
 Page 1 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME (and dosage form) ACULAR 0,4% COMPOSITION ACULAR 0,4% ophthalmic solution contains: Ketorolac tromethamine: 4 mg/ml Preservative: Benzalkonium chloride
Page 1 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME (and dosage form) ACULAR 0,4% COMPOSITION ACULAR 0,4% ophthalmic solution contains: Ketorolac tromethamine: 4 mg/ml Preservative: Benzalkonium chloride
PROPOSED PACKAGE INSERT FOR XALACOM EYE DROPS
 PROPOSED PACKAGE INSERT FOR XALACOM EYE DROPS SCHEDULING STATUS: S4 PROPRIETARY NAME (and dosage form): XALACOM Eye Drops COMPOSITION: Each millilitre contains latanoprost 50 µg and timolol maleate equivalent
PROPOSED PACKAGE INSERT FOR XALACOM EYE DROPS SCHEDULING STATUS: S4 PROPRIETARY NAME (and dosage form): XALACOM Eye Drops COMPOSITION: Each millilitre contains latanoprost 50 µg and timolol maleate equivalent
OCULAR PHARMACOLOGY GLAUCOMA. increased intraocular pressure. normally mm Hg. when to Tx no fixed level.
 OCULAR PHARMACOLOGY GLAUCOMA increased intraocular pressure normally 12 20 mm Hg. when to Tx no fixed level. literature sets ~21 mm Hg as upper limit of normal. some safe at 30 mm Hg some may have damage
OCULAR PHARMACOLOGY GLAUCOMA increased intraocular pressure normally 12 20 mm Hg. when to Tx no fixed level. literature sets ~21 mm Hg as upper limit of normal. some safe at 30 mm Hg some may have damage
The treatment benefit of latanoprost has not been studied in the following populations/patients:
 6.1. ELEMENTS FOR A PUBLIC SUMMARY 6.1.1. Overview of Disease Epidemiology Glaucoma is the second leading cause of blindness worldwide, and affects about 66.8 million people worldwide i,ii. Glaucoma can
6.1. ELEMENTS FOR A PUBLIC SUMMARY 6.1.1. Overview of Disease Epidemiology Glaucoma is the second leading cause of blindness worldwide, and affects about 66.8 million people worldwide i,ii. Glaucoma can
Update on Rhopressa TM QD (netarsudil ophthalmic solution) 0.02% and Roclatan TM (netarsudil/latanoprost ophthalmic solution) 0.02%/0.
 Update on Rhopressa TM QD (netarsudil ophthalmic solution) 0.02% and Roclatan TM (netarsudil/latanoprost ophthalmic solution) 0.02%/0.005% 1 Important Information Any discussion of the potential use or
Update on Rhopressa TM QD (netarsudil ophthalmic solution) 0.02% and Roclatan TM (netarsudil/latanoprost ophthalmic solution) 0.02%/0.005% 1 Important Information Any discussion of the potential use or
PATIENT GUIDE TO LUMIGAN 0.01%
 Treatment Tracker enclosed PATIENT GUIDE TO LUMIGAN 0.01% LUMIGAN (bimatoprost ophthalmic solution) 0.01% is used for the treatment of high eye pressure, also called intraocular pressure (IOP), in people
Treatment Tracker enclosed PATIENT GUIDE TO LUMIGAN 0.01% LUMIGAN (bimatoprost ophthalmic solution) 0.01% is used for the treatment of high eye pressure, also called intraocular pressure (IOP), in people
PACKAGE LEAFLET: INFORMATION FOR THE USER ACULAR 0.5% w/v EYE DROPS, SOLUTION (Ketorolac trometamol)
 PACKAGE LEAFLET: INFORMATION FOR THE USER ACULAR 0.5% w/v EYE DROPS, SOLUTION (Ketorolac trometamol) Read all of this leaflet carefully before you start using this medicine. - Keep this leaflet. You may
PACKAGE LEAFLET: INFORMATION FOR THE USER ACULAR 0.5% w/v EYE DROPS, SOLUTION (Ketorolac trometamol) Read all of this leaflet carefully before you start using this medicine. - Keep this leaflet. You may
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC)
 Guidelines for the medical treatment of chronic open angle glaucoma and ocular hypertension Summary: DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) Diagnosis and management of ocular hypertension (OHT)
Guidelines for the medical treatment of chronic open angle glaucoma and ocular hypertension Summary: DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) Diagnosis and management of ocular hypertension (OHT)
PATIENT INFORMATION LEAFLET. PROPRIETARY NAME, STRENGTH AND PHARMACEUTICAL FORM OZURDEX, dexamethasone 700 μg intravitreal implant
 Page 1 of 6 SCHEDULING STATUS Schedule 4 PATIENT INFORMATION LEAFLET PROPRIETARY NAME, STRENGTH AND PHARMACEUTICAL FORM, dexamethasone 700 μg intravitreal implant Read all of this leaflet carefully before
Page 1 of 6 SCHEDULING STATUS Schedule 4 PATIENT INFORMATION LEAFLET PROPRIETARY NAME, STRENGTH AND PHARMACEUTICAL FORM, dexamethasone 700 μg intravitreal implant Read all of this leaflet carefully before
2 pl-timoptol-la-uk-appr
 Package leaflet: Information for the patient Timoptol-LA 0.25% w/v gel-forming eye drops, solution Timoptol-LA 0.5% w/v gel-forming eye drops, solution timolol (as maleate) Read all of this leaflet carefully
Package leaflet: Information for the patient Timoptol-LA 0.25% w/v gel-forming eye drops, solution Timoptol-LA 0.5% w/v gel-forming eye drops, solution timolol (as maleate) Read all of this leaflet carefully
APPROVED PACKAGE INSERT FOR XALATAN EYE DROPS. Each millilitre contains latanoprost 50 µg and benzalkonium chloride 0,02 % m/v as preservative.
 SCHEDULING STATUS: S4 APPROVED PACKAGE INSERT FOR XALATAN EYE DROPS PROPRIETARY NAME (and dosage form): XALATAN Eye Drops COMPOSITION: Each millilitre contains latanoprost 50 µg and benzalkonium chloride
SCHEDULING STATUS: S4 APPROVED PACKAGE INSERT FOR XALATAN EYE DROPS PROPRIETARY NAME (and dosage form): XALATAN Eye Drops COMPOSITION: Each millilitre contains latanoprost 50 µg and benzalkonium chloride
Summary of the risk management plan (RMP) for Ristempa (pegfilgrastim)
 EMA/183255/2015 Summary of the risk management plan (RMP) for Ristempa (pegfilgrastim) This is a summary of the risk management plan (RMP) for Ristempa, which details the measures to be taken in order
EMA/183255/2015 Summary of the risk management plan (RMP) for Ristempa (pegfilgrastim) This is a summary of the risk management plan (RMP) for Ristempa, which details the measures to be taken in order
PATIENT INFORMATION LEAFLET
 Page 1 of 6 PATIENT INFORMATION LEAFLET SCHEDULING STATUS Schedule 4 PROPRIETARY NAME, STRENGTH AND PHARMACEUTICAL FORM PRED FORTE Sterile Eye Suspension, prednisolone acetate 10 mg/ml sterile eye suspension
Page 1 of 6 PATIENT INFORMATION LEAFLET SCHEDULING STATUS Schedule 4 PROPRIETARY NAME, STRENGTH AND PHARMACEUTICAL FORM PRED FORTE Sterile Eye Suspension, prednisolone acetate 10 mg/ml sterile eye suspension
Elements for a Public Summary
 VI.2 Elements for a Public Summary Fluticasone propionate / formoterol fumarate are available in pressarised metered dose inhalers (pmdi) under the brand names Flutiform, and in breath actuated inhalers
VI.2 Elements for a Public Summary Fluticasone propionate / formoterol fumarate are available in pressarised metered dose inhalers (pmdi) under the brand names Flutiform, and in breath actuated inhalers
New Zealand Datasheet. Dorzolamide 2% w/v and Timolol 0.5% w/v ophthalmic solution
 New Zealand Datasheet Arrow-Dortim Dorzolamide 2% w/v and Timolol 0.5% w/v ophthalmic solution Presentation A clear, colourless slightly viscous solution. Each ml contains 20 mg of dorzolamide (as hydrochloride)
New Zealand Datasheet Arrow-Dortim Dorzolamide 2% w/v and Timolol 0.5% w/v ophthalmic solution Presentation A clear, colourless slightly viscous solution. Each ml contains 20 mg of dorzolamide (as hydrochloride)
THE CHRONIC GLAUCOMAS
 THE CHRONIC GLAUCOMAS WHAT IS GLAUCOMA? People with glaucoma have lost some of their field of all round vision. It is often the edge or periphery that is lost. That is why the condition can be missed until
THE CHRONIC GLAUCOMAS WHAT IS GLAUCOMA? People with glaucoma have lost some of their field of all round vision. It is often the edge or periphery that is lost. That is why the condition can be missed until
Angina pectoris due to coronary atherosclerosis : Atenolol is indicated for the long term management of patients with angina pectoris.
 Lonet Tablet Description Lonet contains Atenolol, a synthetic β1 selective (cardioselective) adrenoreceptor blocking agent without membrane stabilising or intrinsic sympathomimetic (partial agonist) activity.
Lonet Tablet Description Lonet contains Atenolol, a synthetic β1 selective (cardioselective) adrenoreceptor blocking agent without membrane stabilising or intrinsic sympathomimetic (partial agonist) activity.
O NH CH 3. soluble in water with a molecular weight of Structural formula: Empirical formula: C18H29N03.HCl
 PRODUCT INFORMATION NAME OF MEDICINE BETOQUIN Eye Drops Betaxolol 0.5% (5 mg/ml) as 0.56% betaxolol hydrochloride DESCRIPTION BETOQUIN Eye Drops 0.5% contain betaxolol hydrochloride (HCl), a cardioselective
PRODUCT INFORMATION NAME OF MEDICINE BETOQUIN Eye Drops Betaxolol 0.5% (5 mg/ml) as 0.56% betaxolol hydrochloride DESCRIPTION BETOQUIN Eye Drops 0.5% contain betaxolol hydrochloride (HCl), a cardioselective
WORLD GLAUCOMA WEEK What is Glaucoma? Can I develop glaucoma if I have increased eye pressure?
 WORLD GLAUCOMA WEEK What is Glaucoma? Glaucoma is a group of diseases that damage the optic nerve and can result in gradual vision loss and blindness. However, with early detection and treatment, you can
WORLD GLAUCOMA WEEK What is Glaucoma? Glaucoma is a group of diseases that damage the optic nerve and can result in gradual vision loss and blindness. However, with early detection and treatment, you can
Chemical Name: (-)-(S)-5-[3-(tert-Butylamino)-2-hydroxypropoxy]-3,4-dihydro-1(2H)- naphthalenone hydrochloride.
![Chemical Name: (-)-(S)-5-[3-(tert-Butylamino)-2-hydroxypropoxy]-3,4-dihydro-1(2H)- naphthalenone hydrochloride. Chemical Name: (-)-(S)-5-[3-(tert-Butylamino)-2-hydroxypropoxy]-3,4-dihydro-1(2H)- naphthalenone hydrochloride.](/thumbs/76/74231442.jpg) BETAGAN (levobunolol hydrochloride ophthalmic solution, USP) sterile DESCRIPTION BETAGAN (levobunolol hydrochloride ophthalmic solution, USP) sterile is a noncardioselective beta-adrenoceptor blocking
BETAGAN (levobunolol hydrochloride ophthalmic solution, USP) sterile DESCRIPTION BETAGAN (levobunolol hydrochloride ophthalmic solution, USP) sterile is a noncardioselective beta-adrenoceptor blocking
ALPHAGAN EYE DROPS BRIMONIDINE TARTRATE
 PACKAGE LEAFLET: INFORMATION FOR THE USER ALPHAGAN EYE DROPS BRIMONIDINE TARTRATE This leaflet is a copy of the Summary of Product Characteristics and Patient Information Leaflet for a medicine, which
PACKAGE LEAFLET: INFORMATION FOR THE USER ALPHAGAN EYE DROPS BRIMONIDINE TARTRATE This leaflet is a copy of the Summary of Product Characteristics and Patient Information Leaflet for a medicine, which
Summary of the risk management plan (RMP) for DuoResp Spiromax (budesonide / formoterol)
 EMA/126654/2014 Summary of the risk management plan (RMP) for DuoResp Spiromax (budesonide / formoterol) This is a summary of the risk management plan (RMP) for DuoResp Spiromax, which details the measures
EMA/126654/2014 Summary of the risk management plan (RMP) for DuoResp Spiromax (budesonide / formoterol) This is a summary of the risk management plan (RMP) for DuoResp Spiromax, which details the measures
GLAUCOMA. An Overview
 GLAUCOMA An Overview Compiled by Campbell M Gold (2004) CMG Archives http://campbellmgold.com --()-- IMPORTANT The health information contained herein is not meant as a substitute for advice from your
GLAUCOMA An Overview Compiled by Campbell M Gold (2004) CMG Archives http://campbellmgold.com --()-- IMPORTANT The health information contained herein is not meant as a substitute for advice from your
HIDROKORTIZON SA HLORAMFENIKOLOM (10 mg + 2 mg) / ml, eye drops, suspension
 PACKAGE LEAFLET: INFORMATION FOR THE USER HIDROKORTIZON SA HLORAMFENIKOLOM (10 mg + 2 mg) / ml, eye drops, suspension HYDROCORTISONE, CHLORAMPHENICOL This leaflet is a copy of the Summary of Product Characteristics
PACKAGE LEAFLET: INFORMATION FOR THE USER HIDROKORTIZON SA HLORAMFENIKOLOM (10 mg + 2 mg) / ml, eye drops, suspension HYDROCORTISONE, CHLORAMPHENICOL This leaflet is a copy of the Summary of Product Characteristics
Elements for a Public Summary
 VI.2 VI.2.1 Elements for a Public Summary Overview of disease epidemiology Alzheimer's disease is the most common form of dementia, a general term for memory loss and other intellectual abilities serious
VI.2 VI.2.1 Elements for a Public Summary Overview of disease epidemiology Alzheimer's disease is the most common form of dementia, a general term for memory loss and other intellectual abilities serious
THE CHRONIC GLAUCOMAS
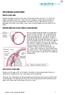 THE CHRONIC GLAUCOMAS WHAT IS GLAUCOMA People with glaucoma have lost some of their field of all round vision. It is often the edge or periphery that is lost. That is why the condition can be missed until
THE CHRONIC GLAUCOMAS WHAT IS GLAUCOMA People with glaucoma have lost some of their field of all round vision. It is often the edge or periphery that is lost. That is why the condition can be missed until
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Xatabloc 50 micrograms/ml + 5 mg/ml eye drops, solution. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains latanoprost
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Xatabloc 50 micrograms/ml + 5 mg/ml eye drops, solution. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains latanoprost
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT GANFORT 0.3 mg/ml + 5 mg/ml eye drops, solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One ml of solution contains 0.3
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT GANFORT 0.3 mg/ml + 5 mg/ml eye drops, solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One ml of solution contains 0.3
Package leaflet: Information for the user. Latanoprost Pfizer 50 micrograms/ml Eye drops, solution Latanoprost
 Package leaflet: Information for the user Latanoprost Pfizer 50 micrograms/ml Eye drops, solution Latanoprost Read all of this leaflet carefully before you start using this medicine because it contains
Package leaflet: Information for the user Latanoprost Pfizer 50 micrograms/ml Eye drops, solution Latanoprost Read all of this leaflet carefully before you start using this medicine because it contains
SCHEDULING STATUS Schedule 4. PROPRIETARY NAME (AND DOSAGE FORM) GANFORT Eye Drops
 Page 1 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME (AND DOSAGE FORM) GANFORT Eye Drops COMPOSITION Each ml contains: Bimatoprost 0,3 mg and timolol maleate equivalent to 5 mg timolol Preservative: Benzalkonium
Page 1 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME (AND DOSAGE FORM) GANFORT Eye Drops COMPOSITION Each ml contains: Bimatoprost 0,3 mg and timolol maleate equivalent to 5 mg timolol Preservative: Benzalkonium
OOGZIEKTEN VOOR DE HUISARTS F. GOES, JR.
 OOGZIEKTEN VOOR DE HUISARTS F. GOES, JR. HET RODE OOG F. GOES, JR. Condition Signs Symptoms Causes Conjunctivitis Viral Normal vision, normal pupil size Mild to no pain, diffuse Adenovirus (most common),
OOGZIEKTEN VOOR DE HUISARTS F. GOES, JR. HET RODE OOG F. GOES, JR. Condition Signs Symptoms Causes Conjunctivitis Viral Normal vision, normal pupil size Mild to no pain, diffuse Adenovirus (most common),
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Xalcom 50 micrograms/ml + 5 mg/ml eye drops, solution. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains latanoprost
SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Xalcom 50 micrograms/ml + 5 mg/ml eye drops, solution. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains latanoprost
TENOPT timolol maleate
 TENOPT timolol maleate Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about It does not contain all the available information. It does not take the place
TENOPT timolol maleate Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about It does not contain all the available information. It does not take the place
IOP measurements: 8.00 am (trough drug levels) and am (peak drug levels)(2 hours post dose)
 This overview of 2% eye drops was presented by Dr. Ravin N. Das, at Hotel Satya Ashoka, on 19-6-2004 in place of Dr. H. S. Ray due to unforseen circumstances. This dinner meeting was sponsored by Cipla
This overview of 2% eye drops was presented by Dr. Ravin N. Das, at Hotel Satya Ashoka, on 19-6-2004 in place of Dr. H. S. Ray due to unforseen circumstances. This dinner meeting was sponsored by Cipla
Summary of the risk management plan (RMP) for Laventair (umeclidinium bromide and vilanterol)
 EMA/258177/2014 Summary of the risk management plan (RMP) for Laventair (umeclidinium bromide and vilanterol) This is a summary of the risk management plan (RMP) for Laventair, which details the measures
EMA/258177/2014 Summary of the risk management plan (RMP) for Laventair (umeclidinium bromide and vilanterol) This is a summary of the risk management plan (RMP) for Laventair, which details the measures
PRESCRIBING INFORMATION AND CONSUMER INFORMATION ISOPTO * CARPINE. pilocarpine hydrochloride ophthalmic solution, USP.
 PRESCRIBING INFORMATION AND CONSUMER INFORMATION Pr ISOPTO * CARPINE pilocarpine hydrochloride ophthalmic solution, USP 1%, 2% and 4% w/v Parasympathomimetic Alcon Canada Inc. 2665 Meadowpine Blvd. Mississauga,
PRESCRIBING INFORMATION AND CONSUMER INFORMATION Pr ISOPTO * CARPINE pilocarpine hydrochloride ophthalmic solution, USP 1%, 2% and 4% w/v Parasympathomimetic Alcon Canada Inc. 2665 Meadowpine Blvd. Mississauga,
PRODUCT INFORMATION ALCAINE. Proparacaine Hydrochloride Sterile Ophthalmic Solution, USP. 5 mg/ml. Topical Anesthetic
 PRODUCT INFORMATION ALCAINE Proparacaine Hydrochloride Sterile Ophthalmic Solution, USP 5 mg/ml Topical Anesthetic Alcon Canada Inc. 2665 Meadowpine Blvd. Mississauga, ON L5N 8C7 www.alcon.ca Date of Preparation:
PRODUCT INFORMATION ALCAINE Proparacaine Hydrochloride Sterile Ophthalmic Solution, USP 5 mg/ml Topical Anesthetic Alcon Canada Inc. 2665 Meadowpine Blvd. Mississauga, ON L5N 8C7 www.alcon.ca Date of Preparation:
Glaucoma. Cornea. Iris
 Glaucoma Introduction Glaucoma is a group of eye diseases that can lead to blindness if not treated. Openangle glaucoma, the most common form of glaucoma, affects about 3 million Americans. Half of those
Glaucoma Introduction Glaucoma is a group of eye diseases that can lead to blindness if not treated. Openangle glaucoma, the most common form of glaucoma, affects about 3 million Americans. Half of those
Pilocarpine 2% w/v Eye Drops, solution. Pilocarpine 4% w/v Eye Drops, solution. Pilocarpine Hydrochloride
 PACKAGE LEAFLET: INFORMATION FOR THE USER Pilocarpine 2% w/v Eye Drops, solution Pilocarpine 4% w/v Eye Drops, solution Pilocarpine Hydrochloride (Referred to as Pilocarpine Eye Drops in this leaflet)
PACKAGE LEAFLET: INFORMATION FOR THE USER Pilocarpine 2% w/v Eye Drops, solution Pilocarpine 4% w/v Eye Drops, solution Pilocarpine Hydrochloride (Referred to as Pilocarpine Eye Drops in this leaflet)
Divakar Gupta Glaucoma Fellow, Duke Eye Center 5/14/16
 Divakar Gupta Glaucoma Fellow, Duke Eye Center 5/14/16 Pathophysiology of glaucoma Consider risk factors of glaucoma Understand the side effects of glaucoma medications Diagnostic testing Leading cause
Divakar Gupta Glaucoma Fellow, Duke Eye Center 5/14/16 Pathophysiology of glaucoma Consider risk factors of glaucoma Understand the side effects of glaucoma medications Diagnostic testing Leading cause
Page 1 of 9. Timolol Maleate Ophthalmic Solution, USP 0.5% Initial U.S. Approval: 1978 STERILE
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Timolol Maleate Ophthalmic Solution, USP 0.5% safely and effectively. See full prescribing information
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Timolol Maleate Ophthalmic Solution, USP 0.5% safely and effectively. See full prescribing information
