Please see Important Safety Information, including Boxed Warnings for Gadavist, Eovist and Magnevist on the following pages.
|
|
|
- Frederica Thomasina Bailey
- 5 years ago
- Views:
Transcription
1 Dear Valued Customer: As a valued partner we want to inform you that a technical issue occurred at our Berlin manufacturing center where final formulation and packaging of Bayer MRI (Magnetic Resonance Imaging) Contrast agents occur. This has caused a near-term impact on the fulfillment of Bayer s MRI contrast agents due to a production stoppage. We sincerely apologize for any inconvenience this may cause. The technical issue has been resolved and production has resumed. There is no risk to patients associated with this issue. Bayer takes patient safety and the quality of its products very seriously. Rest assured that no product you have received was affected by this situation. Released products passed all of our quality tests prior to distribution. We are actively working to find ways to increase production. However, there will be intermittent product shortages over the next several months for the following products: Bayer s Radiology products that are impacted include: Product Description NDC Number McKesson Material Number Comments Gadavist (gadobutrol injection) 1mmol/mL 7.5 ml vial 10 ml vial 30 ml vial (PBP) Shortages expected through May Eovist (gadoxetate disodium) injection 0.25mmol/mL Magnevist (gadopentetate dimeglumine) injection 0.5mmol/mL 15 ml vial ml vial Shortages starting in February. 10mL vial not impacted Possible shortterm shortages starting in mid- April Bayer is committed to working with you to minimize any disruption. All customers are being encouraged to continue to place orders and work with their wholesalers and Bayer Sales Representative to help navigate the situation. You may also contact customer support at Your assistance in this process is also greatly appreciated. As a resource to our customers and to ensure you have on-going and sufficient information, we have set-up a customer support box to help answer customer questions and work with you on potential solutions to meet your ongoing contrast needs. The address is contrastcustomersupport@bayer.com. When ing us, please provide a contact telephone number and the best time to reach you. Someone from our Support Team will be in contact with you. We apologize for any inconvenience this may cause. Although this situation will regretfully cause product shortages, we maintain rigorous product quality standards across all of our products. The decision to stop production was necessary to ensure these standards were being met. We value the trust that has helped to make Bayer s MRI contrast agents so widely used. We are working hard to ensure we are able to continue to supply our customers with the product they need. Thank you for your support and patience as we work to get back to full fulfillment of our contrast agents. Please see Important Safety Information, including Boxed Warnings for Gadavist, Eovist and Magnevist on the following pages. Best regards, Rich Dewit Head, US Sales, Marketing
2 INDICATIONS and IMPORTANT SAFETY INFORMATION INDICATIONS AND USAGE: MAGNEVIST (gadopentetate dimeglumine) Injection Central Nervous System: Magnevist (gadopentetate dimeglumine) injection is indicated for the use with magnetic resonance imaging (MRI) in adults and pediatric patients (2 years of age and older) to visualize lesions with abnormal vascularity in the brain (intracranial lesions), spine and associated tissues. Magnevist has been shown to facilitate visualization of intracranial lesions including but not limited to tumors. Extracranial/Extraspinal Tissues: Magnevist is indicated for use with MRI in adults and pediatric patients (2 years of age and older) to visualize lesions with abnormal vascularity in the head and neck. Body: Magnevist is indicated for use with MRI in adults and pediatric patients (2 years of age and older) to visualize lesions with abnormal vascularity in the body. IMPORTANT SAFETY INFORMATION FOR MAGNEVIST WARNING: NEPHROGENIC SYSTEMIC FIBROSIS (NSF) Gadolinium-based contrast agents (GBCAs) increase the risk for NSF among patients with impaired elimination of the drugs. Avoid use of GBCAs in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. NSF may result in fatal or debilitating fibrosis affecting the skin, muscle and internal organs. Do not administer MAGNEVIST to patients with: o Chronic, severe kidney disease (GFR <30 ml/min/1.73m 2 ), or o Acute kidney injury Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (for example, age >60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing. Do not exceed the recommended MAGNEVIST dose and allow a sufficient period of time for elimination of the drug from the body prior to re-administration. Contraindications: Magnevist is contraindicated in patients with: Chronic, severe kidney disease (glomerular filtration rate, GFR <30 ml/min/1.73m 2 ),or Acute kidney injury, or History of severe hypersensitivity reactions to Magnevist. Hypersensitivity Reactions: Anaphylactoid and anaphylactic reactions with cardiovascular, respiratory and/or cutaneous manifestations rarely resulting in death have occurred. The risk of hypersensitivity reactions is higher in patients with a history of reaction to contrast media, bronchial asthma, or allergic disorders. Hypersensitivity reactions can occur with or without prior exposure to GBCAs. Renal Failure: In patients with renal impairment, acute renal failure (acute kidney injury) requiring dialysis or worsening renal function has occurred, mostly within 48 hours of Magnevist Injection. The risk of acute renal failure is higher with increasing dose of contrast. Use the lowest possible dose, evaluate renal function in patients with renal impairment, and allow sufficient time for contrast elimination before readministration. Elimination half-life in patients with mild or moderate renal impairment is 3 to 4 hours. Elimination half-life in patients with severe renal impairment is about 11 hours. Magnevist is cleared by glomerular filtration and is dialyzable. After 3 dialysis sessions of 3 hours each, about 97% of the administered dose is eliminated from the body; each dialysis session removes about 70% of the circulating drug. Please see additional Important Safety Information for Magnevist (gadopentetate dimeglumine) (continued on the next page)
3 Important Safety Information for Magnevist (gadopentetate dimeglumine) (continued) Injection Site Reaction: Skin and soft tissue necrosis, thrombosis, fasciitis, and compartment syndrome requiring surgical intervention (e.g., compartment release or amputation) have occurred very rarely at the site of the contrast injection or the dosed limb. Total volume and rate of Magnevist Injection, extravasation of contrast agent, and patient susceptibility might contribute to these reactions. Phlebitis and thrombophlebitis may be observed generally within 24 hours after Magnevist Injection and resolve with supportive treatment. Determine the patency and integrity of the intravenous line before administration of Magnevist Injection. Assessment of the dosed limb for the development of injection site reactions is recommended. Interference with Visualization of Lesions with Non-Contrast MRI: As with any paramagnetic contrast agent, Magnevist Injection might impair the visualization of lesions seen on non-contrast MRI. Therefore, caution should be exercised when Magnevist MRI scans are interpreted without a companion noncontrast MRI scan. Adverse Reactions: In clinical trials, the most frequently reported adverse reactions ( 1%) included headache (4.8%), nausea (2.7%), injection site coldness/localized coldness (2.3%) and dizziness (1%). Please see Magnevist Full Prescribing Information INDICATIONS AND USAGE: GADAVIST (gadobutrol) Injection Gadavist (gadobutrol) injection is a gadolinium-based contrast agent indicated for use with magnetic resonance imaging (MRI): To detect and visualize areas with disrupted blood brain barrier (BBB) and/or abnormal vascularity of the central nervous system in adult and pediatric patients (including term neonates) To assess the presence and extent of malignant breast disease Gadavist is indicated for use in magnetic resonance angiography (MRA): To evaluate known or suspected supra-aortic or renal artery disease in adult and pediatric patients (including term neonates) INDICATIONS AND USAGE: EOVIST (gadoxetate disodium) Injection Eovist is indicated for intravenous use in magnetic resonance imaging (MRI) of the liver to detect and characterize lesions in patients with known or suspected focal liver disease. IMPORTANT SAFETY INFORMATION for Gadavist (gadobutrol) and Eovist (gadoxetate disodium) WARNING: NEPHROGENIC SYSTEMIC FIBROSIS (NSF) Gadolinium-based contrast agents (GBCAs) increase the risk for NSF among patients with impaired elimination of the drugs. Avoid use of GBCAs in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. NSF may result in fatal or debilitating fibrosis affecting the skin, muscle and internal organs. The risk for NSF appears highest among patients with: o chronic, severe kidney disease (GFR <30 ml/min/1.73m 2 ), or o acute kidney injury Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (for example, age >60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing. For patients at highest risk for NSF, do not exceed the recommended dose and allow a sufficient period of time for elimination of the drug from the body prior to any readministration. Please see additional Important Safety Information for Gadavist (gadobutrol) and Eovist (gadoxetate disodium) (continued on the next page)
4 Important Safety Information for Gadavist (gadobutrol) and Eovist (gadoxetate disodium) (continued) Additional Important Safety Information for Gadavist (gadobutrol) Contraindication and Important Information about Hypersensitivity Reactions: Gadavist is contraindicated in patients with history of severe hypersensitivity reactions to Gadavist. Anaphylactic and other hypersensitivity reactions with cardiovascular, respiratory, or cutaneous manifestations, ranging from mild to severe, including death, have uncommonly occurred following Gadavist administration. Before Gadavist administration, assess all patients for any history of a reaction to contrast media, bronchial asthma and/or allergic disorders. These patients may have an increased risk for a hypersensitivity reaction to Gadavist. Acute Kidney Injury: In patients with chronic renal impairment, acute kidney injury sometimes requiring dialysis has been observed with the use of some GBCAs. Do not exceed the recommended dose; the risk of acute kidney injury may increase with higher than recommended doses. Extravasation and Injection Site Reactions: Ensure catheter and venous patency before the injection of Gadavist. Extravasation into tissues during Gadavist administration may result in moderate irritation. Overestimation of Extent of Malignant Disease in MRI of the Breast: Gadavist MRI of the breast overestimated the histologically confirmed extent of malignancy in the diseased breast in up to 50% of the patients. Low Sensitivity for Significant Arterial Stenosis The performance of Gadavist MRA for detecting arterial segments with significant stenosis (>50% renal, >70% supra-aortic) has not been shown to exceed 55%. Therefore, a negative MRA study alone should not be used to rule out significant stenosis. Adverse Reactions: The most frequent ( 0.5%) adverse reactions associated with Gadavist in clinical studies were headache (1.5%), nausea (1.1%) and dizziness (0.5%). Please see Full Prescribing Information for Gadavist (Vials and Syringes). Please see Full Prescribing Information for Gadavist (Pharmacy Bulk Package). Additional Important Safety Information for Eovist (gadoxetate disodium) Contraindication and Important Information about Hypersensitivity Reactions Eovist is contraindicated in patients with history of severe hypersensitivity reactions to Eovist. Anaphylactic and other hypersensitivity reactions with cardiovascular, respiratory and cutaneous manifestations, ranging from mild to severe, including shock have uncommonly occurred following Eovist administration. Before Eovist administration, assess all patients for any history of a reaction to contrast media, bronchial asthma and allergic disorders. These patients may have an increased risk for a hypersensitivity reaction to Eovist. Acute Kidney Injury In patients with chronic renal impairment, acute kidney injury sometimes requiring dialysis has been observed with the use of some GBCAs. The risk of acute kidney injury might be lower with Eovist due to its dual excretory pathways. Do not exceed the recommended dose; the risk of acute kidney injury may increase with higher than recommended doses. Extravasation and Injection Site Reactions Ensure catheter and venous patency before the injection of Eovist. Extravasation into tissues during Eovist administration may result in local tissue reactions. Strictly avoid intramuscular administration of Eovist because it may cause myocyte necrosis and inflammation. Interference with Laboratory Tests Serum iron determination using complex metric methods (for example, ferrocene complexation method) may result in falsely high or low values for up to 24 hours after the examination with Eovist because of the caloxetate trisodium excipients. Please see additional Important Safety Information for Eovist (gadoxetate disodium) (continued on the next page)
5 Important Safety Information for Eovist (gadoxetate disodium) (continued) Interference with Visualization of Liver Lesions Severe renal or hepatic failure may impair Eovist imaging performance. In patients with end-stage renal failure, hepatic contrast was markedly reduced and was attributed to elevated serum ferritin levels. In patients with abnormally high (>3 mg/dl) serum bilirubin, reduced hepatic contrast was observed. If Eovist is used in these patients, complete MRI no later than 60 minutes after Eovist administration and use a paired noncontrast and contrast MRI set for diagnosis. Adverse Reactions: The most frequent ( 0.5%) adverse reactions associated with Eovist are nausea (1.1%), headache (1.1%), feeling hot (0.8%), dizziness (0.6%), and back pain (0.6%). Please see Eovist Full Prescribing Information 2018 Bayer. Bayer HealthCare LLC Bayer HealthCare LLC, 100 Bayer Boulevard PO Box 915, Whippany, NJ Bayer, the Bayer Cross, and Eovist Gadavist and Magnevist are trademarks of the Bayer group of companies. PP-CONP-US-0039 March 2018
Important Update - Availability of Bayer s MRI (Magnetic Resonance Imaging) Contrast agents
 Important Update - Availability of Bayer s MRI (Magnetic Resonance Imaging) Contrast agents Certain batches of pharmaceutical products from our company s Supply Center in Berlin where final formulation
Important Update - Availability of Bayer s MRI (Magnetic Resonance Imaging) Contrast agents Certain batches of pharmaceutical products from our company s Supply Center in Berlin where final formulation
Please see Important Safety Information, including Boxed Warnings for Gadavist and Magnevist below.
 August 15 th, 2018 Dear Valued Customer: Thank you for your continued patience over the last several months as we have worked to resolve our contrast supply interruption. The purpose of this letter is
August 15 th, 2018 Dear Valued Customer: Thank you for your continued patience over the last several months as we have worked to resolve our contrast supply interruption. The purpose of this letter is
Billing Guide. For Freestanding Imaging Centers Provided by Bayer HealthCare
 Billing Guide For Freestanding Imaging Centers Provided by Bayer HealthCare Introduction Bayer HealthCare Inc. offers this billing guide to freestanding imaging center customers as part of its comprehensive
Billing Guide For Freestanding Imaging Centers Provided by Bayer HealthCare Introduction Bayer HealthCare Inc. offers this billing guide to freestanding imaging center customers as part of its comprehensive
INTRODUCTION CLINICAL EXPERIENCE CLINICAL EFFICACY PEDIATRIC EXPERIENCE DOSING SUMMARY
 1 2 DOTAREM Administered Doses 3 Molecular Structure of DOTAREM DOTAREM offers high thermodynamic and kinetic stability provided by its macrocyclic and ionic structure* 3 *The clinical significance of
1 2 DOTAREM Administered Doses 3 Molecular Structure of DOTAREM DOTAREM offers high thermodynamic and kinetic stability provided by its macrocyclic and ionic structure* 3 *The clinical significance of
FULL PRESCRIBING INFORMATION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use MAGNEVIST safely and effectively. See full prescribing information for MAGNEVIST. MAGNEVIST (gadopentetate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use MAGNEVIST safely and effectively. See full prescribing information for MAGNEVIST. MAGNEVIST (gadopentetate
Seeing is Believing... Look Beneath the Surface
 References 1. Idee JM, Port C, Raynal I, et al. Clinical and geological consequences of transmetallation induced by contrast agents for magnetic resonance imaging: a review. Fundamental and Clinical Pharmacology.
References 1. Idee JM, Port C, Raynal I, et al. Clinical and geological consequences of transmetallation induced by contrast agents for magnetic resonance imaging: a review. Fundamental and Clinical Pharmacology.
1 INDICATIONS AND USAGE 2 DOSAGE AND ADMINISTRATION. 1.1 Central Nervous System. 1.2 Extracranial/Extraspinal Tissues. 1.3 Body
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use MAGNEVIST safely and effectively. See full prescribing information for MAGNEVIST. MAGNEVIST (gadopentetate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use MAGNEVIST safely and effectively. See full prescribing information for MAGNEVIST. MAGNEVIST (gadopentetate
CONTRAINDICATIONS History of severe hypersensitivity reaction to EOVIST (4)
 HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use EVIST safely and effectively. See full prescribing information for EVIST Injection. EVIST (gadoxetate
HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use EVIST safely and effectively. See full prescribing information for EVIST Injection. EVIST (gadoxetate
Gadolinium-Based Contrast Agents for Magnetic Resonance Imaging (marketed as Magnevist, MultiHance, Omniscan, OptiMARK, ProHance)
 Information for Healthcare Professionals Page 1 of 5 FDA ALERT [6/2006, updated 12/2006 and 5/23/2007: This updated Alert highlights FDA s request for addition of a boxed warning and new warnings about
Information for Healthcare Professionals Page 1 of 5 FDA ALERT [6/2006, updated 12/2006 and 5/23/2007: This updated Alert highlights FDA s request for addition of a boxed warning and new warnings about
Gadolinium Based Contrast Agent Recommendations (Revised 06/07/2018)
 Gadolinium Based Contrast Agent Recommendations (Revised 06/07/2018) Introduction: Nephrogenic systemic fibrosis is the only known adverse health effect related to gadoliniumbased contrast agents (GBCAs).
Gadolinium Based Contrast Agent Recommendations (Revised 06/07/2018) Introduction: Nephrogenic systemic fibrosis is the only known adverse health effect related to gadoliniumbased contrast agents (GBCAs).
CONTRAINDICATIONS History of severe hypersensitivity reaction to EOVIST (4)
 HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use EVIST safely and effectively. See full prescribing information for EVIST Injection. EVIST (gadoxetate
HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use EVIST safely and effectively. See full prescribing information for EVIST Injection. EVIST (gadoxetate
Administration of Gadolinium Contrast in Adults Procedural Guideline
 Administration of Gadolinium Contrast in Adults Procedural Guideline This procedural guideline is designed to assist Radiologists by providing an analytical framework for the evaluation of gadolinium contrast
Administration of Gadolinium Contrast in Adults Procedural Guideline This procedural guideline is designed to assist Radiologists by providing an analytical framework for the evaluation of gadolinium contrast
Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be used
 Contrast vs Non-Contrast When to Order Stephen McManus, M.D. Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be
Contrast vs Non-Contrast When to Order Stephen McManus, M.D. Contrast media Purpose of using contrast Contrast reaction Nephrotoxicity from contrast Nephrogenic systemic fibrosis When should contrast be
Annex III. Amendments to relevant sections of the product information
 Annex III Amendments to relevant sections of the product information Note: These amendments to the relevant sections of the product information are the outcome of the referral procedure. The product information
Annex III Amendments to relevant sections of the product information Note: These amendments to the relevant sections of the product information are the outcome of the referral procedure. The product information
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide. flow rate of approximately 2 ml/second for adults and 1-2 ml/second for pediatric
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
DOTAREM (gadoterate meglumine) Injection for intravenous use Initial U.S. Approval: 2013
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
flow rate of approximately 2 ml/second for adults and 1-2 ml/second for pediatric patients. The dose is delivered by manual or power injection.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
CONTRAINDICATIONS Clinically important hypersensitivity reactions to DOTAREM. (4)
 M120177AB HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
M120177AB HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
Reference ID:
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively. See full prescribing information for DOTAREM. DOTAREM (gadoterate
Radiology Reimbursement Information Setting: Hospital Outpatient
 Radiology Reimbursement Information Setting: Hospital Outpatient About DOTAREM: DOTAREM is a gadolinium-based contrast agent indicated for intravenous use with magnetic resonance imaging (MRI) in brain
Radiology Reimbursement Information Setting: Hospital Outpatient About DOTAREM: DOTAREM is a gadolinium-based contrast agent indicated for intravenous use with magnetic resonance imaging (MRI) in brain
PRODUCT MONOGRAPH GADOVIST 1.0. gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use
 PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) For Professional Use Only Bayer Inc. Date
PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) For Professional Use Only Bayer Inc. Date
DOTAREMÂ - gadoterate meglumineâ injectionâ Guerbet LLC
 DOTAREMÂ - gadoterate meglumineâ injectionâ Guerbet LLC ---------- HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively.
DOTAREMÂ - gadoterate meglumineâ injectionâ Guerbet LLC ---------- HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DOTAREM safely and effectively.
Reference ID: MultiHance Injection, NDA , SAFETY LABELING CHANGES UNDER 505(o)(4) MultiHance PI to FDA_Revised Nov _CLEAN.
 MultiHance Injection, NDA 21-357, SAFETY LABELING CHANGES UNDER 505(o)(4) MultiHance PI to FDA_Revised Nov-18-2010_CLEAN.doc HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the
MultiHance Injection, NDA 21-357, SAFETY LABELING CHANGES UNDER 505(o)(4) MultiHance PI to FDA_Revised Nov-18-2010_CLEAN.doc HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the
PACKAGE LEAFLET: INFORMATION FOR THE USER
 PACKAGE LEAFLET: INFORMATION FOR THE USER Primovist 0.25 mmol/ml, solution for injection Gadoxetate disodium Read all of this leaflet carefully before you are given this medicine. - Keep this leaflet.
PACKAGE LEAFLET: INFORMATION FOR THE USER Primovist 0.25 mmol/ml, solution for injection Gadoxetate disodium Read all of this leaflet carefully before you are given this medicine. - Keep this leaflet.
Gadolinium-Based Contrast Media
 Gadolinium-Based Contrast Media Wm. Faulkner, B.S.,R.T.(R)(MR)CT, FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, R.T.(R)(MR), MRSO (MRSC TM ) Gadolinium ( 64 Gd) 7 un-paired electrons chelate, any of a class
Gadolinium-Based Contrast Media Wm. Faulkner, B.S.,R.T.(R)(MR)CT, FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, R.T.(R)(MR), MRSO (MRSC TM ) Gadolinium ( 64 Gd) 7 un-paired electrons chelate, any of a class
PRODUCT MONOGRAPH DOTAREM. (gadoterate meglumine) Injection. For Intravenous Use
 PRODUCT MONOGRAPH DOTAREM (gadoterate meglumine) Injection For Intravenous Use Gadolinium-Based Contrast Agent For Use with Magnetic Resonance Imaging (MRI) Guerbet LLC Date: August 26, 2013 120 W. 7 th
PRODUCT MONOGRAPH DOTAREM (gadoterate meglumine) Injection For Intravenous Use Gadolinium-Based Contrast Agent For Use with Magnetic Resonance Imaging (MRI) Guerbet LLC Date: August 26, 2013 120 W. 7 th
University of Virginia Institutional Review Board - Health Sciences Research Guidelines for Researchers Using Gadolinium-Enhanced MRI in Research
 University of Virginia Institutional Review Board - Health Sciences Research Guidelines for Researchers Using Gadolinium-Enhanced MRI in Research Page 1 of 16 Table of Contents New Information:... 3 IRB-HSR
University of Virginia Institutional Review Board - Health Sciences Research Guidelines for Researchers Using Gadolinium-Enhanced MRI in Research Page 1 of 16 Table of Contents New Information:... 3 IRB-HSR
PRODUCT MONOGRAPH GADOVIST 1.0. gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use
 PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) Bayer Inc. 77 Belfield Road Toronto, ON M9W
PRODUCT MONOGRAPH GADOVIST 1.0 gadobutrol injection 604 mg/ml (1.0 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance Imaging (MRI) Bayer Inc. 77 Belfield Road Toronto, ON M9W
PATIENT INFORMATION LEAFLET. DOTAREM mg/ml, solution for injection (Gadoteric acid)
 PATIENT INFORMATION LEAFLET DOTAREM 279.32 mg/ml, solution for injection (Gadoteric acid) Read all this leaflet carefully before you start using this medicine because it contains important information
PATIENT INFORMATION LEAFLET DOTAREM 279.32 mg/ml, solution for injection (Gadoteric acid) Read all this leaflet carefully before you start using this medicine because it contains important information
SUBJECT: ASSESSMENT OF LAB RESULTS PRIOR TO INJECTING GADOLINIUM-BASED CONTRAST MEDIA.
 ASSESSMENT OF PATIENTS: IMAGING SERVICES Policy #: 300.02 Effective Date: 6/29/09 Last Revision Date: 10/07 Page 1 of 5 SUBJECT: ASSESSMENT OF LAB RESULTS PRIOR TO INJECTING GADOLINIUM-BASED CONTRAST MEDIA
ASSESSMENT OF PATIENTS: IMAGING SERVICES Policy #: 300.02 Effective Date: 6/29/09 Last Revision Date: 10/07 Page 1 of 5 SUBJECT: ASSESSMENT OF LAB RESULTS PRIOR TO INJECTING GADOLINIUM-BASED CONTRAST MEDIA
PRODUCT MONOGRAPH PRIMOVIST. gadoxetate disodium injection mg/ml (0.25 mmol/ml)
 PRODUCT MONOGRAPH PRIMOVIST gadoxetate disodium injection 181.43 mg/ml (0.25 mmol/ml) Intravenous contrast enhancement agent for magnetic resonance imaging (MRI) For Professional Use Only Distributed and
PRODUCT MONOGRAPH PRIMOVIST gadoxetate disodium injection 181.43 mg/ml (0.25 mmol/ml) Intravenous contrast enhancement agent for magnetic resonance imaging (MRI) For Professional Use Only Distributed and
Protocol for iv. iodine and gadolinium contrast studies
 Protocol for iv. iodine and gadolinium contrast studies Royal College of Radiologists Standard The individual administering the contrast agent must ensure that the patient understands that it is to be
Protocol for iv. iodine and gadolinium contrast studies Royal College of Radiologists Standard The individual administering the contrast agent must ensure that the patient understands that it is to be
Instructions for the safe administration of gadolinium based contrast agents (GBCA). 1. PREADMINISTRATION PRECAUTIONS:
 POLICY NUMBER: RM 6-18 CATEGORY: Patient Care DATE: June 2016 NEXT REVIEW DATE: April 2017 SUBJECT: PURPOSE: POLICY: Intravenous Gadolinium Based Contrast Administration To prevent complications associated
POLICY NUMBER: RM 6-18 CATEGORY: Patient Care DATE: June 2016 NEXT REVIEW DATE: April 2017 SUBJECT: PURPOSE: POLICY: Intravenous Gadolinium Based Contrast Administration To prevent complications associated
PRODUCT INFORMATION DOTAREM
 PRODUCT INFORMATION DOTAREM NAME OF THE MEDICINE DOTAREM (gadoteric acid) paramagnetic contrast medium for Magnetic Resonance Imaging (MRI). The structural formula of gadoteric acid is shown below: The
PRODUCT INFORMATION DOTAREM NAME OF THE MEDICINE DOTAREM (gadoteric acid) paramagnetic contrast medium for Magnetic Resonance Imaging (MRI). The structural formula of gadoteric acid is shown below: The
WARNING, CONTRAINDICATIONS, WARNINGS AND PRECAUTIONS,
 Celgene Corporation 86 Morris Avenue Summit, New Jersey 07901 Tel 908-673-9000 Fax 908-673-9001 October 2012 NEW Indication Announcement for ABRAXANE for Injectable Suspension (paclitaxel protein-bound
Celgene Corporation 86 Morris Avenue Summit, New Jersey 07901 Tel 908-673-9000 Fax 908-673-9001 October 2012 NEW Indication Announcement for ABRAXANE for Injectable Suspension (paclitaxel protein-bound
Package Leaflet: Information for the user. MAGNEVIST 2 mmol/l solution for injection dimeglumine gadopentetate
 Due to regulatory changes, the content of the following Patient Information Leaflet may vary from the one found in your medicine pack. Please compare the 'Leaflet prepared/revised date' towards the end
Due to regulatory changes, the content of the following Patient Information Leaflet may vary from the one found in your medicine pack. Please compare the 'Leaflet prepared/revised date' towards the end
PRIMOVIST 5 ml, 7,5 ml, 10 ml
 REGISTERED PACKAGE INSERT SCHEDULING STATUS S4 PROPRIETARY NAMES AND DOSAGE FORMS PRIMOVIST 5 ml, 7,5 ml, 10 ml Solution for injection COMPOSITION 1 ml of the MRI contract agent Primovist contains 181,43
REGISTERED PACKAGE INSERT SCHEDULING STATUS S4 PROPRIETARY NAMES AND DOSAGE FORMS PRIMOVIST 5 ml, 7,5 ml, 10 ml Solution for injection COMPOSITION 1 ml of the MRI contract agent Primovist contains 181,43
DIVISION OF MEDICAL IMAGING AND HEMATOLOGY PRODUCTS
 DIVISION OF MEDICAL IMAGING AND HEMATOLOGY PRODUCTS MEMORANDUM TO THE FILE i. NDA: 20-123, 22-066 Product: Omniscan ii. NDA: 19-596, 21-037 Product: Magnevist iii. NDA: 20-976, 20-937,20-975 Product: OptiMARK
DIVISION OF MEDICAL IMAGING AND HEMATOLOGY PRODUCTS MEMORANDUM TO THE FILE i. NDA: 20-123, 22-066 Product: Omniscan ii. NDA: 19-596, 21-037 Product: Magnevist iii. NDA: 20-976, 20-937,20-975 Product: OptiMARK
Package leaflet: Information for the patient. Gadovist 1.0 mmol/ml solution for injection. Gadobutrol
 Due to regulatory changes, the content of the following Patient Information Leaflet may vary from the one found in your medicine pack. Please compare the 'Leaflet prepared/revised date' towards the end
Due to regulatory changes, the content of the following Patient Information Leaflet may vary from the one found in your medicine pack. Please compare the 'Leaflet prepared/revised date' towards the end
MR Contrast Agents. Why Use Contrast Agents in MRI? Why Use Contrast Agents in MRI? US Agents. Understanding and Embracing Change
 Why Use Contrast Agents in MRI? Improve disease detection and characterization Increase sensitivity to extent of disease MR Contrast Agents Understanding and Embracing Change Kristan Harrington, MBA, RT
Why Use Contrast Agents in MRI? Improve disease detection and characterization Increase sensitivity to extent of disease MR Contrast Agents Understanding and Embracing Change Kristan Harrington, MBA, RT
FDA Advisory Committee Briefing Document. Medical Imaging Drugs Advisory Committee. Meeting to be held on February 14, 2013
 FDA Advisory Committee Briefing Document Medical Imaging Drugs Advisory Committee Meeting to be held on February 14, 2013 New Drug Application 204-781 Gadoterate meglumine Injection (Dotarem ), sponsored
FDA Advisory Committee Briefing Document Medical Imaging Drugs Advisory Committee Meeting to be held on February 14, 2013 New Drug Application 204-781 Gadoterate meglumine Injection (Dotarem ), sponsored
Data Sheet PRIMOVIST. Name of the Medicine. Solution for Intravenous Injection disodium gadoxetate mg/ml (0.25M)
 Data Sheet PRIMVIST Solution for Intravenous Injection disodium gadoxetate 181.43 mg/ml (0.25M) Name of the Medicine Primovist 0.25 mmol/ml, solution for injection is an injectable contrast media agent
Data Sheet PRIMVIST Solution for Intravenous Injection disodium gadoxetate 181.43 mg/ml (0.25M) Name of the Medicine Primovist 0.25 mmol/ml, solution for injection is an injectable contrast media agent
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION DOTAREM. Gadoterate meglumine injection. (376.9 mg/ml, equivalent to 0.
 PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION DOTAREM Gadoterate meglumine injection (376.9 mg/ml, equivalent to 0.5 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION DOTAREM Gadoterate meglumine injection (376.9 mg/ml, equivalent to 0.5 mmol/ml) For Intravenous Use Contrast Enhancement Agent for Magnetic Resonance
DATA SHEET MAGNEVIST. Solution for Intravenous Injection Dimeglumine gadopentetate 0.5 mmol/ml PRESENTATION
 DATA SHEET MAGNEVIST Solution for Intravenous Injection Dimeglumine gadopentetate 0.5 mmol/ml PRESENTATION Magnevist injection is a sterile, clear, colourless to slightly yellow solution for intravenous
DATA SHEET MAGNEVIST Solution for Intravenous Injection Dimeglumine gadopentetate 0.5 mmol/ml PRESENTATION Magnevist injection is a sterile, clear, colourless to slightly yellow solution for intravenous
PATIENT CARE AND MRI SAFETY Module 7
 PATIENT CARE AND MRI SAFETY Module 7 1 Biological Considerations There are no reported adverse biological effects of extended exposure to MRI. However, several inconsequential and reversible effects of
PATIENT CARE AND MRI SAFETY Module 7 1 Biological Considerations There are no reported adverse biological effects of extended exposure to MRI. However, several inconsequential and reversible effects of
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT DOTAREM 0.5 mmol/ml solution for injection in vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution for injection contains
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT DOTAREM 0.5 mmol/ml solution for injection in vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution for injection contains
Beyond NSF: Acute GBCA adverse reactions
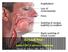 Source images Beyond NSF: Acute GBCA adverse reactions Martin R. Prince, MD, PhD, FACR Disclosures Patent Agreements: GE, Siemens, Philips, Hitachi, Toshiba, Bayer, Bracco, Mallinckrodt, Medrad, Nemoto,
Source images Beyond NSF: Acute GBCA adverse reactions Martin R. Prince, MD, PhD, FACR Disclosures Patent Agreements: GE, Siemens, Philips, Hitachi, Toshiba, Bayer, Bracco, Mallinckrodt, Medrad, Nemoto,
MAGNEVIST 0.5 mmol/ml BAYER MIDDLE EAST
 08-15 MAGNEVIST 0.5 mmol/ml BAYER MIDDLE EAST Solution for injection Dimeglumine gadopentetate, for use in infants, children and adults 1. WHAT MAGNEVIST IS AND WHAT IT IS USED FOR Magnevist is a contrast
08-15 MAGNEVIST 0.5 mmol/ml BAYER MIDDLE EAST Solution for injection Dimeglumine gadopentetate, for use in infants, children and adults 1. WHAT MAGNEVIST IS AND WHAT IT IS USED FOR Magnevist is a contrast
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT DOTAREM 0.5 mmol/ml (for multiple use) solution for injection in vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution for
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT DOTAREM 0.5 mmol/ml (for multiple use) solution for injection in vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution for
Policy #: 291 Latest Review Date: February 2013
 Effective for dates of service on or after April 1, 2013, refer to: https://www.bcbsal.org/providers/policies/carecore.cfm Name of Policy: Magnetic Resonance Angiography (MRA) of the Chest (excluding the
Effective for dates of service on or after April 1, 2013, refer to: https://www.bcbsal.org/providers/policies/carecore.cfm Name of Policy: Magnetic Resonance Angiography (MRA) of the Chest (excluding the
Gadovist. Prescribing information on all Bayer Contrast Media products March 2015, L.GB a
 Prescribing information on all Bayer Contrast Media products March 2015, L.GB.01.2015.9569a Gadovist Gadovist 1.0 mmol/ml solution for injection (gadobutrol) and Gadovist 1.0 mmol/ml solution for injection
Prescribing information on all Bayer Contrast Media products March 2015, L.GB.01.2015.9569a Gadovist Gadovist 1.0 mmol/ml solution for injection (gadobutrol) and Gadovist 1.0 mmol/ml solution for injection
Each ml contains 0.25 mmol gadoxetate disodium (Gd-EOB-DTPA disodium), equivalent to mg gadoxetate disodium.
 1. NAME OF THE MEDICINAL PRODUCT Primovist 0.25 mmol/ml, solution for injection, pre-filled syringe 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains 0.25 mmol gadoxetate disodium (Gd-EOB-DTPA
1. NAME OF THE MEDICINAL PRODUCT Primovist 0.25 mmol/ml, solution for injection, pre-filled syringe 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains 0.25 mmol gadoxetate disodium (Gd-EOB-DTPA
IMPORTANT DRUG WARNING
 IMPORTANT DRUG INFORMATION UPDATE IMPORTANT DRUG WARNING Rifampin for Injection USP, 600 mg/vial See Page 4 for Lots Pfizer NDC: 0069-0112-01; NOVAPLUS NDC: 0069-0141-01 December 4, 2013 Attention: Product
IMPORTANT DRUG INFORMATION UPDATE IMPORTANT DRUG WARNING Rifampin for Injection USP, 600 mg/vial See Page 4 for Lots Pfizer NDC: 0069-0112-01; NOVAPLUS NDC: 0069-0141-01 December 4, 2013 Attention: Product
Billing and Coding Information
 Billing and Coding Information The information provided is for educational purposes only. The healthcare provider is fully responsible for billing and coding determinations. INDICATIONS Injectafer (ferric
Billing and Coding Information The information provided is for educational purposes only. The healthcare provider is fully responsible for billing and coding determinations. INDICATIONS Injectafer (ferric
Conversion Dosing Guide:
 Conversion Dosing Guide: From epoetin alfa to Aranesp in patients with anemia due to CKD on dialysis Indication Aranesp (darbepoetin alfa) is indicated for the treatment of anemia due to chronic kidney
Conversion Dosing Guide: From epoetin alfa to Aranesp in patients with anemia due to CKD on dialysis Indication Aranesp (darbepoetin alfa) is indicated for the treatment of anemia due to chronic kidney
The P&T Committee Lisinopril (Qbrelis )
 Situation Background Assessment The P&T Committee Lisinopril (Qbrelis ) Qbrelis, 1 mg/ml lisinopril oral solution, has recently become an FDA- approved formulation. Current practice at UK Chandler Medical
Situation Background Assessment The P&T Committee Lisinopril (Qbrelis ) Qbrelis, 1 mg/ml lisinopril oral solution, has recently become an FDA- approved formulation. Current practice at UK Chandler Medical
Local Natalizumab Treatment Protocol
 Local Natalizumab Treatment Protocol 1. New medicine name: Natalizumab 300mg concentrate for solution for infusion (Natalizumab ) 2. Licensed indication(s): Natalizumab is indicated for single disease
Local Natalizumab Treatment Protocol 1. New medicine name: Natalizumab 300mg concentrate for solution for infusion (Natalizumab ) 2. Licensed indication(s): Natalizumab is indicated for single disease
NEW ZEALAND DATA SHEET
 1. PRODUCT NAME Dotarem (bottles and pre-filled syringes) 0.5 mmol/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Per 100 ml of solution: Gadoteric acid*... 27.932 g corresponding
1. PRODUCT NAME Dotarem (bottles and pre-filled syringes) 0.5 mmol/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Per 100 ml of solution: Gadoteric acid*... 27.932 g corresponding
A Safety Update on the Gadolinium Chelates
 Control # 1029 A Safety Update on the Gadolinium Chelates Val M. Runge, MD Universitätsinstitut für Diagnostische, Interventionelle und Pädiatrische Radiologie University Hospital Bern Allergic Reactions
Control # 1029 A Safety Update on the Gadolinium Chelates Val M. Runge, MD Universitätsinstitut für Diagnostische, Interventionelle und Pädiatrische Radiologie University Hospital Bern Allergic Reactions
TIVORBEX Now Available in U.S. Pharmacies for the Treatment of Acute Pain
 TIVORBEX Now Available in U.S. Pharmacies for the Treatment of Acute Pain Second Low-Dose SoluMatrix NSAID from Iroko Now Available by Prescription PHILADELPHIA, June 29, 2015 Iroko Pharmaceuticals, LLC,
TIVORBEX Now Available in U.S. Pharmacies for the Treatment of Acute Pain Second Low-Dose SoluMatrix NSAID from Iroko Now Available by Prescription PHILADELPHIA, June 29, 2015 Iroko Pharmaceuticals, LLC,
Iroko Pharmaceuticals Gains Additional Patents for ZORVOLEX and TIVORBEX TM
 Iroko Pharmaceuticals Gains Additional Patents for ZORVOLEX and TIVORBEX TM PHILADELPHIA, April 8, 2015 Iroko Pharmaceuticals, LLC, a global specialty pharmaceutical company dedicated to advancing the
Iroko Pharmaceuticals Gains Additional Patents for ZORVOLEX and TIVORBEX TM PHILADELPHIA, April 8, 2015 Iroko Pharmaceuticals, LLC, a global specialty pharmaceutical company dedicated to advancing the
Global reports and findings of NSF and its effects
 Safety Forum: MR Safety Update - SMRT 2008 Sunday May 8, 2:25 p.m. Global reports and findings of NSF and its effects Tim Leiner, MD PhD Assistant Professor of Radiology, Department of Radiology, Maastricht
Safety Forum: MR Safety Update - SMRT 2008 Sunday May 8, 2:25 p.m. Global reports and findings of NSF and its effects Tim Leiner, MD PhD Assistant Professor of Radiology, Department of Radiology, Maastricht
PRAC recommendations on signals for update of the product information
 Thursday 22 January 2015 EMA/PRAC/62352/2015 Pharmacovigilance Risk Assessment Committee PRAC recommendations on signals for update of the product information Adopted at the 6-9 January 2015 PRAC 1. Atorvastatin,
Thursday 22 January 2015 EMA/PRAC/62352/2015 Pharmacovigilance Risk Assessment Committee PRAC recommendations on signals for update of the product information Adopted at the 6-9 January 2015 PRAC 1. Atorvastatin,
Information for Healthcare Professionals and other Stakeholders
 Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, February 2014
Information for Healthcare Professionals and other Stakeholders NEPHROGENIC SYSTEMIC FIBROSIS: AN UNCOMMON AND DEBILITATING DISEASE POSSIBLY ASSOCIATED WITH GADOLINIUM CHELATES Villepinte, February 2014
PRODUCT INFORMATION NAME OF THE MEDICINE DESCRIPTION. GADOVIST 1.0 (gadobutrol) Physico-chemical properties
 PRODUCT INFORMATION GADOVIST 1.0 (gadobutrol) NAME OF THE MEDICINE Gadovist 1.0 injection is a 1.0 mmol/ml solution of 10-(2,3-Dihydroxy-1- hydroxymethylpropyl)-1,4,7,10-tetraazacyclododecane-1,4,7-triacetic
PRODUCT INFORMATION GADOVIST 1.0 (gadobutrol) NAME OF THE MEDICINE Gadovist 1.0 injection is a 1.0 mmol/ml solution of 10-(2,3-Dihydroxy-1- hydroxymethylpropyl)-1,4,7,10-tetraazacyclododecane-1,4,7-triacetic
PACKAGE LEAFLET: INFORMATION FOR THE USER. Fibrovein 3%, 1%, 0.5% and 0.2% Solution for Injection Sodium tetradecyl sulphate
 PACKAGE LEAFLET: INFORMATION FOR THE USER Fibrovein 3%, 1%, 0.5% and 0.2% Solution for Injection Sodium tetradecyl sulphate Read all of this leaflet carefully before you start using this medicine. Keep
PACKAGE LEAFLET: INFORMATION FOR THE USER Fibrovein 3%, 1%, 0.5% and 0.2% Solution for Injection Sodium tetradecyl sulphate Read all of this leaflet carefully before you start using this medicine. Keep
NSF Coming and Going
 NSF Coming and Going Martin R. Prince, MD, PhD Cornell and Columbia Universities Patent agreements: GE, Philips, Siemens, Hitachi, Medrad, Epix, Lantheus, Bayer, Bracco, Nemoto, Mallinckrodt and Topspins
NSF Coming and Going Martin R. Prince, MD, PhD Cornell and Columbia Universities Patent agreements: GE, Philips, Siemens, Hitachi, Medrad, Epix, Lantheus, Bayer, Bracco, Nemoto, Mallinckrodt and Topspins
Finish the course for as little as $ Start the course for as little as
 HELP YOUR PATIENTS ACCESS IRON* INJECTAFER Restrictions apply. For Adult IDA Patients With Iron Deficits, Consider the Injectafer Savings Program* Start the course for as little as $ 50 FIRST DOSE up to
HELP YOUR PATIENTS ACCESS IRON* INJECTAFER Restrictions apply. For Adult IDA Patients With Iron Deficits, Consider the Injectafer Savings Program* Start the course for as little as $ 50 FIRST DOSE up to
Calcium Folinate Solution for Injection
 1 of 7 6/10/2016 4:14 PM Dosage Form: injection, solution Calcium Folinate Solution for Injection URGENT: LEUCOVORIN UPDATE June 2011 Dear Healthcare Professional: Due to the current critical shortage
1 of 7 6/10/2016 4:14 PM Dosage Form: injection, solution Calcium Folinate Solution for Injection URGENT: LEUCOVORIN UPDATE June 2011 Dear Healthcare Professional: Due to the current critical shortage
Hepatobiliary Contrast Agents for Liver MRI
 Hepatobiliary Contrast Agents for Liver MRI Scott B. Reeder, MD, PhD International Society for Magnetic Resonance in Medicine Sociedad Mexicana de Radiologia e Imagen (SMRI) Mexico City June 4, 2014 Department
Hepatobiliary Contrast Agents for Liver MRI Scott B. Reeder, MD, PhD International Society for Magnetic Resonance in Medicine Sociedad Mexicana de Radiologia e Imagen (SMRI) Mexico City June 4, 2014 Department
Gadolinium-Based MR Contrast Agents
 Gadolinium-Based MR Contrast Agents Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) 0.35 T 0.35
Gadolinium-Based MR Contrast Agents Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) 0.35 T 0.35
PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker
 PACKAGE INSERT Pr PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker ACTIONS AND CLINICAL PHARMACOLOGY Phentolamine produces an alpha-adrenergic
PACKAGE INSERT Pr PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker ACTIONS AND CLINICAL PHARMACOLOGY Phentolamine produces an alpha-adrenergic
Package leaflet: Information for the patient. Gadovist 1.0 mmol/ml solution for injection in prefilled syringe. Gadobutrol
 Package leaflet: Information for the patient Gadovist 1.0 mmol/ml solution for injection in prefilled syringe Gadobutrol Read all of this leaflet carefully before you are given this medicine because it
Package leaflet: Information for the patient Gadovist 1.0 mmol/ml solution for injection in prefilled syringe Gadobutrol Read all of this leaflet carefully before you are given this medicine because it
Patients with Parkinson s disease treated with Neupro (rotigotine) showed low rates of dyskinesias with long term treatment
 For the attention of accredited medical writers only Patients with Parkinson s disease treated with Neupro (rotigotine) showed low rates of dyskinesias with long term treatment Data presented at the 7
For the attention of accredited medical writers only Patients with Parkinson s disease treated with Neupro (rotigotine) showed low rates of dyskinesias with long term treatment Data presented at the 7
MabThera. SC. The wait is over. MabThera delivered in just 5 minutes. SC= subcutaneous injection
 MabThera SC. The wait is over. MabThera delivered in just 5 minutes Abbreviated Prescribing Information MabThera 1400 mg solution for subcutaneous (SC) injection (Rituximab) Indications: Indicated in adults
MabThera SC. The wait is over. MabThera delivered in just 5 minutes Abbreviated Prescribing Information MabThera 1400 mg solution for subcutaneous (SC) injection (Rituximab) Indications: Indicated in adults
Atrovent Administration
 Atrovent Administration ICEMA Training 2007 Sherri Shimshy RN OBJECTIVES Describe the pharmacology of Atrovent Identify the indications for use of Atrovent in the Adult Population Identify the indications
Atrovent Administration ICEMA Training 2007 Sherri Shimshy RN OBJECTIVES Describe the pharmacology of Atrovent Identify the indications for use of Atrovent in the Adult Population Identify the indications
Temporary interruption in supply of APIDRA (insulin glulisine) - OPTISET and SOLOSTAR prefilled pens and CLIKSTAR reusable cartridges.
 September 2011 Dear Health Care Professional, Temporary interruption in supply of APIDRA (insulin glulisine) - OPTISET and SOLOSTAR prefilled pens and CLIKSTAR reusable cartridges. We would like to inform
September 2011 Dear Health Care Professional, Temporary interruption in supply of APIDRA (insulin glulisine) - OPTISET and SOLOSTAR prefilled pens and CLIKSTAR reusable cartridges. We would like to inform
The first and only FDA-approved lisinopril oral solution for pediatric patients 6 years of age and older. WARNING: FETAL TOXICITY
 MEDICAS DIFFER FROM 5 drug products that are essentially copies of commercially available LIPRIL ORAL AF IMPORAN SAFEY INA WARNING: FEAL OXIC 5 drug products that are essentially copies of commercially
MEDICAS DIFFER FROM 5 drug products that are essentially copies of commercially available LIPRIL ORAL AF IMPORAN SAFEY INA WARNING: FEAL OXIC 5 drug products that are essentially copies of commercially
WARNING LETTER. MD-Gastroview (Diatrizoate Meglumine and Diatrizoate Sodium Solution USP)
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration TRANSMITTED BY FACSIMILE Steven Hanley President Imaging Division Mallinckrodt Inc. 675 McDonnell Boulevard P.O.
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration TRANSMITTED BY FACSIMILE Steven Hanley President Imaging Division Mallinckrodt Inc. 675 McDonnell Boulevard P.O.
Gadolinium-Based MR Contrast Agents
 Gadolinium-Based MR Contrast Agents Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 0.35 T 35/1600
Gadolinium-Based MR Contrast Agents Wm. Faulkner, BS,RT(R)(MR)(CT), FSMRT, MRSO (MRSC ) Kristan Harrington, MBA, RT(R)(MR) ARRT, MRSO (MRSC ) Magnetic Resonance Imaging, Vol 3, pp27-35, 1985 0.35 T 35/1600
SUMMARY OF PRODUCT CHARACTERISTICS. Each ml solution for injection contains phenylephrine hydrochloride corresponding to 0.1 mg phenylephrine.
 1. NAME OF THE MEDICINAL PRODUCT Fenylefrin Abcur 0.05 mg/ml, solution for injection Fenylefrin Abcur 0.1 mg/ml, solution for injection SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE
1. NAME OF THE MEDICINAL PRODUCT Fenylefrin Abcur 0.05 mg/ml, solution for injection Fenylefrin Abcur 0.1 mg/ml, solution for injection SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE
Medical Review Guidelines Magnetic Resonance Angiography
 Medical Review Guidelines Magnetic Resonance Angiography Medical Guideline Number: MRG2001-05 Effective Date: 2/13/01 Revised Date: 2/14/2006 OHCA Reference OAC 317:30-5-24. Radiology. (f) Magnetic Resonance
Medical Review Guidelines Magnetic Resonance Angiography Medical Guideline Number: MRG2001-05 Effective Date: 2/13/01 Revised Date: 2/14/2006 OHCA Reference OAC 317:30-5-24. Radiology. (f) Magnetic Resonance
M120211AA V6.pdf 1 18/10/ :22 M120211AA
 M120211AA V6.pdf 1 18/10/2018 09:22 M120211AA HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LIPIODOL safely and effectively. See full prescribing
M120211AA V6.pdf 1 18/10/2018 09:22 M120211AA HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LIPIODOL safely and effectively. See full prescribing
Abdominal Imaging Update. Tom Sutherland MBBS MMed FRANZCR
 Abdominal Imaging Update Tom Sutherland MBBS MMed FRANZCR Objectives Review selected radiological abdominal studies. CT Colonography Rectal MRI Small bowel Imaging Liver Imaging. Discuss limitations, advantages
Abdominal Imaging Update Tom Sutherland MBBS MMed FRANZCR Objectives Review selected radiological abdominal studies. CT Colonography Rectal MRI Small bowel Imaging Liver Imaging. Discuss limitations, advantages
New data show sustained 5-year benefit of Neupro (Rotigotine Transdermal System) for symptoms of Restless Legs Syndrome
 New data show sustained 5-year benefit of Neupro (Rotigotine Transdermal System) for symptoms of Restless Legs Syndrome Latest safety and efficacy results for rotigotine in the treatment of moderate to
New data show sustained 5-year benefit of Neupro (Rotigotine Transdermal System) for symptoms of Restless Legs Syndrome Latest safety and efficacy results for rotigotine in the treatment of moderate to
DATA SHEET. GADOVIST 1.0 is available as a 1.0 mmol/ml solution and each ml of GADOVIST 1.0 contains mg gadobutrol.
 DATA SHEET 1. PRODUCT NAME (strength pharmaceutical form) GADOVIST 1.0 (1.0 mmol/ml) Solution for Intravenous Injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Gadobutrol 604.72 mg/ml (1.0 mmol/ml)
DATA SHEET 1. PRODUCT NAME (strength pharmaceutical form) GADOVIST 1.0 (1.0 mmol/ml) Solution for Intravenous Injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Gadobutrol 604.72 mg/ml (1.0 mmol/ml)
ROYAL WOLVERHAMPTON HOSPITALS NHS TRUST
 ROYAL WOLVERHAMPTON HOSPITALS NHS TRUST SHARED CARE PROTOCOL FOR ERYTHROPOIETIN USE 2016 New Cross Hospital Dr J Odum Dr P B Rylance Dr P Carmichael Dr S Acton Dr B Ramakrishna Walsall Manor Hospital Dr
ROYAL WOLVERHAMPTON HOSPITALS NHS TRUST SHARED CARE PROTOCOL FOR ERYTHROPOIETIN USE 2016 New Cross Hospital Dr J Odum Dr P B Rylance Dr P Carmichael Dr S Acton Dr B Ramakrishna Walsall Manor Hospital Dr
Cetirizine Proposed Core Safety Profile
 Cetirizine Proposed Core Safety Profile Posology and method of administration Elderly subjects: data do not suggest that the dose needs to be reduced in elderly subjects provided that the renal function
Cetirizine Proposed Core Safety Profile Posology and method of administration Elderly subjects: data do not suggest that the dose needs to be reduced in elderly subjects provided that the renal function
DESCRIPTION Sodium tetradecyl sulfate is an anionic surfactant which occurs as a white, waxy solid. The structural formula is as follows:
 SOTRADECOL- tetradecyl hydrogen sulfate (ester) injection, solution Mylan Ins titutional LLC ---------- SOTRADECOL (Sodium Tetradecyl Sulfate Injection) 1% 20 mg/2 ml (10 mg/ml) and 3% 60 mg/2 ml (30 mg/ml)
SOTRADECOL- tetradecyl hydrogen sulfate (ester) injection, solution Mylan Ins titutional LLC ---------- SOTRADECOL (Sodium Tetradecyl Sulfate Injection) 1% 20 mg/2 ml (10 mg/ml) and 3% 60 mg/2 ml (30 mg/ml)
Contrast Materials Patient Safety: What are contrast materials and how do they work?
 Contrast Materials Patient Safety: What are contrast materials and how do they work? Which imaging exams use contrast materials? How safe are contrast materials? How should I prepare for my imaging procedure
Contrast Materials Patient Safety: What are contrast materials and how do they work? Which imaging exams use contrast materials? How safe are contrast materials? How should I prepare for my imaging procedure
Have you seen a patient like Carol *?
 (linagliptin) 5mg tablets Have you seen a patient like Carol *? *Hypothetical patient profile Carol * : 70 years old Retired schoolteacher *Hypothetical patient profile CAROL*: T2D patient with moderate
(linagliptin) 5mg tablets Have you seen a patient like Carol *? *Hypothetical patient profile Carol * : 70 years old Retired schoolteacher *Hypothetical patient profile CAROL*: T2D patient with moderate
Elements for a public summary. VI.2.1 Overview of disease epidemiology
 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Coronary artery disease and as anticoagulant (inhibiting the clotting of the blood) in patients undergoing surgery to treat blockages
VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Coronary artery disease and as anticoagulant (inhibiting the clotting of the blood) in patients undergoing surgery to treat blockages
PHARMACY DOSING AND ORDERING GUIDE
 PHARMACY DOSING AND ORDERING GUIDE FIRST AND ONLY APPROVED TREATMENT FOR PATIENTS WITH VOD WITH RENAL OR PULMONARY DYSFUNCTION POST HSCT VOD=veno-occlusive disease Indication Defitelio (defibrotide sodium)
PHARMACY DOSING AND ORDERING GUIDE FIRST AND ONLY APPROVED TREATMENT FOR PATIENTS WITH VOD WITH RENAL OR PULMONARY DYSFUNCTION POST HSCT VOD=veno-occlusive disease Indication Defitelio (defibrotide sodium)
Vascular Imaging in the Pediatric Abdomen. Jonathan Swanson, MD
 Vascular Imaging in the Pediatric Abdomen Jonathan Swanson, MD Goals and Objectives To understand the imaging approach, appearance, and clinical manifestations of the common pediatric abdominal vascular
Vascular Imaging in the Pediatric Abdomen Jonathan Swanson, MD Goals and Objectives To understand the imaging approach, appearance, and clinical manifestations of the common pediatric abdominal vascular
Cardiac MRI in ACHD What We. ACHD Patients
 Cardiac MRI in ACHD What We Have Learned to Apply to ACHD Patients Faris Al Mousily, MBChB, FAAC, FACC Consultant, Pediatric Cardiology, KFSH&RC/Jeddah Adjunct Faculty, Division of Pediatric Cardiology
Cardiac MRI in ACHD What We Have Learned to Apply to ACHD Patients Faris Al Mousily, MBChB, FAAC, FACC Consultant, Pediatric Cardiology, KFSH&RC/Jeddah Adjunct Faculty, Division of Pediatric Cardiology
Iroko Pharmaceuticals Announces Acceptance for Filing of ZORVOLEX snda for the Treatment of Osteoarthritis Pain in Adults
 Iroko Pharmaceuticals Announces Acceptance for Filing of ZORVOLEX snda for the Treatment of Osteoarthritis Pain in Adults First Lower Dose NSAID Using SoluMatrix Fine Particle Technology to be Reviewed
Iroko Pharmaceuticals Announces Acceptance for Filing of ZORVOLEX snda for the Treatment of Osteoarthritis Pain in Adults First Lower Dose NSAID Using SoluMatrix Fine Particle Technology to be Reviewed
Elements for a Public Summary
 VI.2 VI.2.1 Elements for a Public Summary Overview of disease epidemiology Alzheimer's disease is the most common form of dementia, a general term for memory loss and other intellectual abilities serious
VI.2 VI.2.1 Elements for a Public Summary Overview of disease epidemiology Alzheimer's disease is the most common form of dementia, a general term for memory loss and other intellectual abilities serious
Fact Sheet. Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII
 Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII Fact Sheet Zohydro ER (hydrocodone bitartrate) Extended-Release Capsule, CII, is a long-acting (extendedrelease) type of pain medication
Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII Fact Sheet Zohydro ER (hydrocodone bitartrate) Extended-Release Capsule, CII, is a long-acting (extendedrelease) type of pain medication
INTRODUCING. Potential Monotherapy Option 5 Provided in Convenient Vials 1 Reimbursable J-Code Available 3
 INTRODUCING The only FDA-approved morphine sulfate for intrathecal and epidural pain management for use in continuous microinfusion devices, packaged in convenient, easy-to-use vials. Available, Agreed-Upon
INTRODUCING The only FDA-approved morphine sulfate for intrathecal and epidural pain management for use in continuous microinfusion devices, packaged in convenient, easy-to-use vials. Available, Agreed-Upon
P-RMS: IE/H/PSUR/0014/002
 Core Safety Profile Active substance: Nitroglycerin Pharmaceutical form(s)/strength: Transdermal patch 25mg, 50mg, 75mg (corresponding to 5, 10 and 15mg per 24 hours respectively P-RMS: IE/H/PSUR/0014/002
Core Safety Profile Active substance: Nitroglycerin Pharmaceutical form(s)/strength: Transdermal patch 25mg, 50mg, 75mg (corresponding to 5, 10 and 15mg per 24 hours respectively P-RMS: IE/H/PSUR/0014/002
