Pain: A Public Health Challenge. NSAIDS for Managing Pain. Iroko: Innovators in Analgesia
|
|
|
- Alexia Franklin
- 5 years ago
- Views:
Transcription
1 Pain: A Public Health Challenge Despite advances in understanding and treatment, pain remains a major public health challenge 1 that exacts a significant personal and economic toll on Americans 2. Pain accounts for an estimated 70 million physician visits every year 3 Each year, an estimated 20 percent of American adults (42 million people) report that pain or physical discomfort disrupts their sleep a few nights a week or more 4 Pain results in more than 50 million lost workdays every year 5 Pain costs the U.S. at least $560-$635 billion annually, or about $2,000 for every American 6 Approximately one half of U.S. adults report pain or discomfort that persists for longer than three months, and an even greater number are likely to have acute pain at any one time 7 More than 76 million people in the U.S. live with chronic pain, and almost half of them receive no treatment 8 Osteoarthritis, the most prevalent pain condition in the U.S., affects approximately 27 million U.S. adults each year 9 Increasing the safe and effective treatment of pain has been identified as a key objective by The Department of Health and Human Services (HSS) Healthy People 2020, which provides science-based, 10-year national objectives for improving the health of Americans. Specific goals outlined include: o Reducing serious injuries from the use of pain medicines o Reducing deaths from the use of pain medicines 10 NSAIDS for Managing Pain Nonsteroidal anti-inflammatory drugs (NSAIDs) are among the most widely used medications for treating pain and inflammation 11. In the U.S. alone, approximately 115 million NSAIDs are prescribed each year 12. NSAIDs work largely by inhibiting cyclooxygenase, an enzyme that controls the production of prostaglandins 13, which are a family of naturally occurring molecules associated with pain and inflammation 14. Though effective, NSAIDs have been associated with significant safety concerns, including cardiovascular thrombotic events, myocardial infarction, stroke 15, gastrointestinal ulcers, gastrointestinal bleeds 16, and renal events such as acute renal failure 17. The risk of these serious adverse events is higher among patients receiving higher doses 18. The U.S. Food and Drug Administration (FDA) and a number of professional medical organizations including the American Heart Association, American Gastroenterological Association, and the American College of Rheumatology recommend that NSAIDs be administered at the lowest effective dose for the shortest duration consistent with individual patient treatment goals 19. Iroko: Innovators in Analgesia Iroko s portfolio includes ZORVOLEX (diclofenac) capsules - the first FDA-approved NSAID developed using SoluMatrix Fine Particle Technology. ZORVOLEX is approved for the management of mild to moderate acute pain and osteoarthritis pain and is now available in pharmacies across the U.S. The second SoluMatrix NSAID, TIVORBEX (indomethacin) capsules, is also FDA-approved for the treatment of mild to moderate acute pain in adults. Our product pipeline consists of several additional low dose NSAIDs created using proprietary SoluMatrix Fine Particle Technology in various stages of nonclinical and clinical development. The safety and efficacy of these investigational products are being evaluated in the therapeutic targets listed.
2 Compound Name Therapeutic Target Nonclinical Phase I Phase II Phase III NDA/sNDA SoluMatrix Meloxicam SoluMatrix Naproxen IP NCB IP NIB *ZORVOLEX is approved for the treatment of mild to moderate acute pain in adults; it is not approved for use in the treatment of osteoarthritis pain. ZORVOLEX is indicated for the management of mild to moderate acute pain and osteoarthritis pain. Important Safety Information About ZORVOLEX Cardiovascular Risk Nonsteroidal anti-inflammatory drugs (NSAIDs) may cause an increased risk of serious cardiovascular thrombotic events, myocardial infarction, and stroke, which can be fatal. This risk may increase with duration of use. Patients with cardiovascular disease or risk factors for cardiovascular disease may be at greater risk. ZORVOLEX is contraindicated for the treatment of perioperative pain in the setting of coronary artery bypass graft (CABG) surgery. Gastrointestinal Risk NSAIDs cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events. ZORVOLEX is contraindicated in patients with: a known hypersensitivity to diclofenac or its inactive ingredients; a history of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. ZORVOLEX should be used at the lowest effective dose for the shortest duration consistent with individual patient treatment goals. Elevation of one or more liver tests may occur during therapy with ZORVOLEX. Physicians should measure transaminases (ALT and AST) periodically in patients receiving long-term therapy with ZORVOLEX. ZORVOLEX should be discontinued immediately if abnormal liver tests persist or worsen. NSAIDS, including ZORVOLEX, can lead to the new onset or worsening of existing hypertension which may contribute to the increased incidence of cardiovascular events. Blood pressure should be monitored closely during treatment with ZORVOLEX. NSAIDs may diminish the antihypertensive activity of thiazides, loop diuretics, ACE inhibitors and angiotensin II antagonists.
3 Fluid retention and edema have been observed in some patients taking NSAIDs. ZORVOLEX should be used with caution in patients with fluid retention or heart failure. Long-term administration of NSAIDs can result in renal papillary necrosis and other renal injury. ZORVOLEX should be used with caution in patients at greatest risk of this reaction, including the elderly, those with impaired renal function, heart failure, liver dysfunction, and those taking diuretics and ACE inhibitors. Treatment with ZORVOLEX in patients with advanced renal disease is not recommended. Anaphylactoid reactions may occur in patients with the aspirin triad or in patients without prior exposure to ZORVOLEX and should be discontinued immediately if an anaphylactoid reaction occurs. NSAIDs can cause serious skin adverse events such as exfoliative dermatitis, Stevens - Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. ZORVOLEX should be discontinued if rash or other signs of local skin reaction occur. Starting at 30 weeks gestation, ZORVOLEX and other NSAIDs should be avoided by pregnant women as premature closure of the ductus arteriosus in the fetus may occur. Concomitant administration of diclofenac and aspirin or anticoagulants is not generally recommended because of the risk of increased GI bleeding that is higher than in users of either drug alone. Most common adverse reactions in clinical trials (incidence 2%) include: edema, nausea, headache, dizziness, vomiting, constipation, pruritus, diarrhea, flatulence, pain in extremity, abdominal pain, sinusitis, alanine aminotransferase increased, blood creatinine increased, hypertension, and dyspepsia. ZORVOLEX capsules do not result in an equivalent systemic exposure to diclofenac as other oral formulations. Therefore, do not substitute similar dosing strengths of other diclofenac products for ZORVOLEX. Please see full Prescribing Information for additional important safety and dosing information. For more information, visit TIVORBEX is indicated for the treatment of mild to moderate acute pain in adults. Important Safety Information about TIVORBEX Cardiovascular Risk Nonsteroidal anti-inflammatory drugs (NSAIDs) may cause an increased risk of serious cardiovascular thrombotic events, myocardial infarction, and stroke, which can be fatal. This risk may increase with duration of use. Patients with cardiovascular disease or risk factors for cardiovascular disease may be at greater risk. TIVORBEX is contraindicated for the treatment of perioperative pain in the setting of coronary artery bypass graft (CABG) surgery. Gastrointestinal Risk NSAIDs cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events.
4 TIVORBEX is contraindicated in patients with: a known hypersensitivity to indomethacin or its inactive ingredients; a history of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. TIVORBEX should be used at the lowest effective dose for the shortest duration consistent with individual patient treatment goals. Elevation of one or more liver tests may occur during therapy with NSAIDs, including TIVORBEX. Physicians should measure transaminases (ALT and AST) periodically in patients receiving long-term therapy with TIVORBEX. TIVORBEX should be discontinued immediately if abnormal liver tests persist or worsen. NSAIDs, including TIVORBEX, can lead to the new onset or worsening of existing hypertension, which may contribute to the increased incidence of cardiovascular events. Blood pressure should be monitored closely during treatment with TIVORBEX. NSAIDs may diminish the antihypertensive activity of thiazides, loop diuretics, ACE inhibitors and angiotensin II antagonists. Fluid retention and edema have been observed in some patients taking NSAIDs. TIVORBEX should be used with caution in patients with fluid retention or heart failure. Long-term administration of NSAIDs can result in renal papillary necrosis and other renal injury. TIVORBEX should be used with caution in patients at greatest risk of this reaction, including the elderly, those with impaired renal function, heart failure, liver dysfunction, and those taking diuretics and ACE inhibitors. Treatment with TIVORBEX in patients with advanced renal disease is not recommended. Anaphylactic reactions may occur in patients with the aspirin triad or in patients without prior exposure to TIVORBEX and should be discontinued immediately if an anaphylactic reaction occurs. Indomethacin may aggravate depression, and other psychiatric disturbances, epilepsy, or parkinsonism, and should be used with caution in patients with these conditions. Indomethacin may cause drowsiness; therefore patients should be cautioned about engaging in activities requiring mental alertness and motor coordination. Discontinue TIVORBEX if severe central nervous system (CNS) adverse reactions develop. NSAIDs can cause serious skin adverse events such as exfoliative dermatitis, Stevens - Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. TIVORBEX should be discontinued if rash or other signs of local skin reaction occur. Starting at 30 weeks gestation, TIVORBEX and other NSAIDs should be avoided by pregnant women as premature closure of the ductus arteriosus in the fetus may occur. Concomitant administration of indomethacin and aspirin or anticoagulants is not generally recommended because the risk of increased GI bleeding is higher than in users of either drug alone. Most common adverse reactions in clinical trials (incidence >2%) include: nausea, post procedural edema, headache, dizziness, vomiting, post procedural hemorrhage, constipation, pruritus, diarrhea, dyspepsia, post procedural swelling, presyncope, rash, upper abdominal pain, somnolence, generalized pruritus, hyperhidrosis, decreased appetite, hot flush, and syncope.
5 About Iroko Pharmaceuticals, LLC Iroko is a global specialty pharmaceutical company, based in Philadelphia, dedicated to advancing the science of analgesia. The company develops and globally commercializes pharmaceutical products. Iroko is at the forefront of the development of SoluMatrix NSAIDs - new low dose drug products based on existing NSAIDs - using iceutica Inc. s proprietary SoluMatrix Fine Particle Technology exclusively licensed to Iroko for NSAIDs. ZORVOLEX is the first SoluMatrix NSAID and is available in pharmacies; the second SoluMatrix NSAID, TIVORBEX was approved by the FDA in February For more information, visit SoluMatrix Fine Particle Technology is a trademark of iceutica Inc., and the technology is licensed to Iroko for exclusive use in NSAIDs. SoluMatrix is a trademark of iceutica Pty Ltd and is licensed to Iroko. Contact: Jessica Donnelly I For Iroko Pharmaceuticals, LLC I I Jessica.Donnelly@cohnwolfe.com 1. Institute of Medicine of the National Academies Report. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research, The National Academies Press, Washington DC (page 55) Institute of Medicine of the National Academies Report. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research, The National Academies Press, Washington DC (page 20) 3. Schnall (2003). Pain Assessment and the Mental Health Practitioner: A Mind-Body Approach. Einstein J. Biol. Med. 20: American Academy of Pain Medicine. Facts and Figures on Pain The American Pain Society. Media Backgrounder. Available at Accessed 8/14/ Institute of Medicine of the National Academies Report. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research, The National Academies Press, Washington DC. (page 5) 7. Sachs C. (2005 Mar). Oral Analgesics for Acute Nonspecific Pain. Am Fam Physician. 1;71(5): National Institutes of Health: MedLine Plus. Safely Managing Chronic Pain. Available at articles/spring11pg4.html. Accessed 8/14/ Neogi T. et al. (2013 Sep). The epidemiology and impact of pain in osteoarthritis. Osteoarthritis Cartilage. 21(9): Medicinal Product Safety. Healthy People Laine L. (2001 Feb). Review Approaches to Nonsteroidal Anti-Inflammatory Drug Use in the High-Risk Patient. Gastroenterology, 120 (3): IMS National Prescription Audit, Total Prescriptions, Brooks PM. (1991). Non Steroidal Antiinflammatory Drugs Differences and Similarities. New England Journal of Medicine. 324: Rhodus NL (1979 May-Jun). Prostaglandins: Promulgators of Pain. Anesth Prog.,26(3): ZORVOLEX Prescribing Information 2014, Page Rahme E et. al. (2001 Aug). Cost of prescribed NSAID-related gastrointestinal adverse events in elderly patients. Br J Clin Pharmacol. 52(2): Annual Review of Medicine, Nonsteroidal Antiinflammatory Drugs and Renal Function. Vol. 35: DOI: /annurev.med Risser A. (2009 Dec). NSAID Prescribing Precautions. Am Fam Physician. 80(12): U.S. Food and Drug Administration. Public Health Advisory - FDA Announces Important Changes and Additional Warnings for COX-2 Selective and Non- Selective Non-Steroidal Anti-Inflammatory Drugs (NSAIDs). ucm htm Iroko Pharmaceuticals, LLC. All rights reserved. ZOR /2014
TIVORBEX Now Available in U.S. Pharmacies for the Treatment of Acute Pain
 TIVORBEX Now Available in U.S. Pharmacies for the Treatment of Acute Pain Second Low-Dose SoluMatrix NSAID from Iroko Now Available by Prescription PHILADELPHIA, June 29, 2015 Iroko Pharmaceuticals, LLC,
TIVORBEX Now Available in U.S. Pharmacies for the Treatment of Acute Pain Second Low-Dose SoluMatrix NSAID from Iroko Now Available by Prescription PHILADELPHIA, June 29, 2015 Iroko Pharmaceuticals, LLC,
Iroko Pharmaceuticals Gains Additional Patents for ZORVOLEX and TIVORBEX TM
 Iroko Pharmaceuticals Gains Additional Patents for ZORVOLEX and TIVORBEX TM PHILADELPHIA, April 8, 2015 Iroko Pharmaceuticals, LLC, a global specialty pharmaceutical company dedicated to advancing the
Iroko Pharmaceuticals Gains Additional Patents for ZORVOLEX and TIVORBEX TM PHILADELPHIA, April 8, 2015 Iroko Pharmaceuticals, LLC, a global specialty pharmaceutical company dedicated to advancing the
Pain: A Public Health Challenge. NSAIDS for Managing Pain. Iroko: Innovators in Analgesia
 Pain: A Public Health Challenge Despite advances in understanding and treatment, pain remains a major public health challenge 1 that exacts a significant personal and economic toll on Americans. 1 Pain
Pain: A Public Health Challenge Despite advances in understanding and treatment, pain remains a major public health challenge 1 that exacts a significant personal and economic toll on Americans. 1 Pain
Iroko Pharmaceuticals Announces Acceptance for Filing of ZORVOLEX snda for the Treatment of Osteoarthritis Pain in Adults
 Iroko Pharmaceuticals Announces Acceptance for Filing of ZORVOLEX snda for the Treatment of Osteoarthritis Pain in Adults First Lower Dose NSAID Using SoluMatrix Fine Particle Technology to be Reviewed
Iroko Pharmaceuticals Announces Acceptance for Filing of ZORVOLEX snda for the Treatment of Osteoarthritis Pain in Adults First Lower Dose NSAID Using SoluMatrix Fine Particle Technology to be Reviewed
Iroko Pharmaceuticals Receives FDA Approval for VIVLODEX - First Low Dose SoluMatrix Meloxicam for Osteoarthritis Pain
 Iroko Pharmaceuticals Receives FDA Approval for VIVLODEX - First Low Dose SoluMatrix Meloxicam for Osteoarthritis Pain VIVLODEX Developed to Align with FDA NSAID Recommendations Proven Efficacy at Low
Iroko Pharmaceuticals Receives FDA Approval for VIVLODEX - First Low Dose SoluMatrix Meloxicam for Osteoarthritis Pain VIVLODEX Developed to Align with FDA NSAID Recommendations Proven Efficacy at Low
Etoricoxib STADA 30 mg, 60 mg, 90 mg and 120 mg film-coated tablets , Version V1.2 PUBLIC SUMMARY OF THE RISK MANAGEMENT PLAN
 Etoricoxib STADA 30 mg, 60 mg, 90 mg and 120 mg film-coated tablets 23.5.2016, Version V1.2 PUBLIC SUMMARY OF THE RISK MANAGEMENT PLAN VI.2 Elements for a Public Summary Etoricoxib STADA 30 mg film-coated
Etoricoxib STADA 30 mg, 60 mg, 90 mg and 120 mg film-coated tablets 23.5.2016, Version V1.2 PUBLIC SUMMARY OF THE RISK MANAGEMENT PLAN VI.2 Elements for a Public Summary Etoricoxib STADA 30 mg film-coated
Pronaxen 250 mg tablet OTC , Version 1.3 PUBLIC SUMMARY OF THE RISK MANAGEMENT PLAN
 Pronaxen 250 mg tablet OTC 25.9.2015, Version 1.3 PUBLIC SUMMARY OF THE RISK MANAGEMENT PLAN VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Pronaxen 250 mg is indicated for
Pronaxen 250 mg tablet OTC 25.9.2015, Version 1.3 PUBLIC SUMMARY OF THE RISK MANAGEMENT PLAN VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Pronaxen 250 mg is indicated for
Your guide to CELEBREX.
 Your guide to CELEBREX. Studies show that staying active can ease arthritis symptoms. But if you have arthritis, staying active can be difficult. CELEBREX can help relieve arthritis pain so your body can
Your guide to CELEBREX. Studies show that staying active can ease arthritis symptoms. But if you have arthritis, staying active can be difficult. CELEBREX can help relieve arthritis pain so your body can
Vimovo (delayed-release enteric-coated naproxen with esomeprazole)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.17.01 Subject: Vimovo Page: 1 of 5 Last Review Date: September 18, 2015 Vimovo Description Vimovo (delayed-release
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.17.01 Subject: Vimovo Page: 1 of 5 Last Review Date: September 18, 2015 Vimovo Description Vimovo (delayed-release
Indications and dosing
 Indications and dosing CELEBREX should be used at the lowest effective dose for the shortest duration consistent with treatment goals for the individual patient CELEBREX can be administered without regard
Indications and dosing CELEBREX should be used at the lowest effective dose for the shortest duration consistent with treatment goals for the individual patient CELEBREX can be administered without regard
Elements for a Public Summary Overview of disease epidemiology
 VI.2 VI.2.1 Elements for a Public Summary Overview of disease epidemiology Acute pain usually responds to medication and should settle in less than three months. Inadequate pain relief may lead to other
VI.2 VI.2.1 Elements for a Public Summary Overview of disease epidemiology Acute pain usually responds to medication and should settle in less than three months. Inadequate pain relief may lead to other
RISK MANAGEMENT PLAN (RMP) PUBLIC SUMMARY ETORICOXIB ORION (ETORICOXIB) 30 MG, 60 MG, 90 MG & 120 MG FILM-COATED TABLET DATE: , VERSION 1.
 RISK MANAGEMENT PLAN (RMP) PUBLIC SUMMARY ETORICOXIB ORION (ETORICOXIB) 30 MG, 60 MG, 90 MG & 120 MG FILM-COATED TABLET DATE: 07-10-2016, VERSION 1.2 VI.2 Elements for a Public Summary Etoricoxib Orion
RISK MANAGEMENT PLAN (RMP) PUBLIC SUMMARY ETORICOXIB ORION (ETORICOXIB) 30 MG, 60 MG, 90 MG & 120 MG FILM-COATED TABLET DATE: 07-10-2016, VERSION 1.2 VI.2 Elements for a Public Summary Etoricoxib Orion
FOR PAIN RELIEF CHOOSE AT THE OPIOID LEVEL DIRECT SAVINGS, DIRECT TO YOUR PATIENTS. Support for Your Practice. Support for Patients
 DIRECT SAVINGS, DIRECT TO YOUR PATIENTS SPRIX Direct provides your patients with their medication EXCLUSIVELY through Cardinal Health Specialty Pharmacy Indications and Usage FOR PAIN RELIEF AT THE OPIOID
DIRECT SAVINGS, DIRECT TO YOUR PATIENTS SPRIX Direct provides your patients with their medication EXCLUSIVELY through Cardinal Health Specialty Pharmacy Indications and Usage FOR PAIN RELIEF AT THE OPIOID
Children Enteric coated tablet : 1-3 mg/kg per day in divided doses.
 Ultrafen Tablet/SR Tablet/Suppository/Gel Description Ultrafen is a preparation of Diclofenac is a non-steroidal antiinflammatory agent with marked analgesic, anti-inflammatory and antipyretic properties.
Ultrafen Tablet/SR Tablet/Suppository/Gel Description Ultrafen is a preparation of Diclofenac is a non-steroidal antiinflammatory agent with marked analgesic, anti-inflammatory and antipyretic properties.
Ask your healthcare provider if SPRIX (ketorolac tromethamine) Nasal Spray is right for you
 For pain that requires opioid-strength relief... You have a non-opioid option RIGHT UNDER YOUR NOSE Ask your healthcare provider if SPRIX (ketorolac tromethamine) Nasal Spray is right for you What is SPRIX?
For pain that requires opioid-strength relief... You have a non-opioid option RIGHT UNDER YOUR NOSE Ask your healthcare provider if SPRIX (ketorolac tromethamine) Nasal Spray is right for you What is SPRIX?
TERICOX. Composition Each film-coated tablet contains 60, 90, or 120 mg of Etoricoxib.
 TERICOX Composition Each film-coated tablet contains 60, 90, or 120 mg of Etoricoxib. Tablets Action Tericox is a nonsteroidal anti-inflammatory drug (NSAID) that exhibits anti-inflammatory, analgesic,
TERICOX Composition Each film-coated tablet contains 60, 90, or 120 mg of Etoricoxib. Tablets Action Tericox is a nonsteroidal anti-inflammatory drug (NSAID) that exhibits anti-inflammatory, analgesic,
REDUCE THE HURT REDUCE THE HARM
 IF YOU HAVE OSTEOARTHRITIS (OA) OR RHEUMATOID ARTHRITIS (RA) REDUCE THE HURT AND REDUCE THE HARM DUEXIS reduces the risk of developing stomach ulcers for patients who are taking ibuprofen for OA/RA INDICATIONS
IF YOU HAVE OSTEOARTHRITIS (OA) OR RHEUMATOID ARTHRITIS (RA) REDUCE THE HURT AND REDUCE THE HARM DUEXIS reduces the risk of developing stomach ulcers for patients who are taking ibuprofen for OA/RA INDICATIONS
CSP Nabumetone ES/H/PSUR/0014/001. January 2010
 CSP Nabumetone ES/H/PSUR/0014/001 January 2010 CLINICAL PARTICULARS 4.1 Therapeutic indications Nabumetone is indicated in the symptomatic treatment of a variety of musculoskeletal disorders requiring
CSP Nabumetone ES/H/PSUR/0014/001 January 2010 CLINICAL PARTICULARS 4.1 Therapeutic indications Nabumetone is indicated in the symptomatic treatment of a variety of musculoskeletal disorders requiring
PUBLIC SUMMARY OF RISK MANAGEMENT PLAN BURANA 40 MG/ML ORAL SUSPENSION DATE: , VERSION 1
 PUBLIC SUMMARY OF RISK MANAGEMENT PLAN BURANA 40 MG/ML ORAL SUSPENSION DATE: 15-5-2015, VERSION 1 VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Musculoskeletal pain: Musculoskeletal
PUBLIC SUMMARY OF RISK MANAGEMENT PLAN BURANA 40 MG/ML ORAL SUSPENSION DATE: 15-5-2015, VERSION 1 VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Musculoskeletal pain: Musculoskeletal
YOUR GUIDE TO. The only prescription nonsteroidal anti-inflammatory patch for acute pain due to minor strains, sprains, and bruises.
 YOUR GUIDE TO The only prescription nonsteroidal anti-inflammatory patch for acute pain due to minor strains, sprains, and bruises. Selected Safety Information Nonsteroidal anti-inflammatory drugs (NSAIDs)
YOUR GUIDE TO The only prescription nonsteroidal anti-inflammatory patch for acute pain due to minor strains, sprains, and bruises. Selected Safety Information Nonsteroidal anti-inflammatory drugs (NSAIDs)
Doctor Discussion Guide
 1 Doctor Discussion Guide Productive talks with your doctor are an important part of helping to manage your arthritis pain. Take the first step and use this guide to help prepare for your next doctor visit.*
1 Doctor Discussion Guide Productive talks with your doctor are an important part of helping to manage your arthritis pain. Take the first step and use this guide to help prepare for your next doctor visit.*
NEOFEN 60 mg suppository
 PACKAGE LEAFLET: INFORMATION FOR THE USER NEOFEN 60 mg suppository IBUPROFEN This leaflet is a copy of the Summary of Product Characteristics and Patient Information Leaflet for a medicine, which outlines
PACKAGE LEAFLET: INFORMATION FOR THE USER NEOFEN 60 mg suppository IBUPROFEN This leaflet is a copy of the Summary of Product Characteristics and Patient Information Leaflet for a medicine, which outlines
Patient Group Direction for the supply and/or administration of Ibuprofen 400mg tablets to patients attending NHS Borders services
 Patient Group Direction for the supply and/or administration of Ibuprofen 400mg tablets to patients attending NHS Borders services This document authorises the supply and/or administration of Ibuprofen
Patient Group Direction for the supply and/or administration of Ibuprofen 400mg tablets to patients attending NHS Borders services This document authorises the supply and/or administration of Ibuprofen
Doctor Discussion Guide
 1 Doctor Discussion Guide Productive talks with your doctor are an important part of helping to manage your arthritis pain. Take the first step and use this guide to help prepare for your next doctor visit.*
1 Doctor Discussion Guide Productive talks with your doctor are an important part of helping to manage your arthritis pain. Take the first step and use this guide to help prepare for your next doctor visit.*
PRODUCT INFORMATION. Sudafed* Sinus + Anti-inflammatory Pain Relief Caplets
 PRODUCT INFORMATION Sudafed* Sinus + Anti-inflammatory Pain Relief Caplets Product description Sudafed* Sinus + Anti-inflammatory Pain Relief caplets contain pseudoephedrine hydrochloride 30 mg and ibuprofen
PRODUCT INFORMATION Sudafed* Sinus + Anti-inflammatory Pain Relief Caplets Product description Sudafed* Sinus + Anti-inflammatory Pain Relief caplets contain pseudoephedrine hydrochloride 30 mg and ibuprofen
Effective pain management begins with OFIRMEV (acetaminophen) injection FIRST Proven efficacy with rapid reduction in pain 1
 Effective pain management begins with OFIRMEV (acetaminophen) injection FIRST Proven efficacy with rapid reduction in pain 1 Fast onset of pain relief with 7% reduction in visual analog scale (VAS) scores
Effective pain management begins with OFIRMEV (acetaminophen) injection FIRST Proven efficacy with rapid reduction in pain 1 Fast onset of pain relief with 7% reduction in visual analog scale (VAS) scores
You May Pay as Little as $4 Per Month with the CELEBREX $4 Co-pay Card* KEEP THESE TIPS IN MIND TO HELP YOU GET BRAND-NAME CELEBREX AT THE PHARMACY.
 You May Pay as Little as $4 Per Month with the CELEBREX $4 Co-pay Card* Bin: 610020 Group: 99990873 ID: Expiration date: 12/31/18 $4 CO-PAY CARD $4 Pay as little as * per month * Eligibility required.
You May Pay as Little as $4 Per Month with the CELEBREX $4 Co-pay Card* Bin: 610020 Group: 99990873 ID: Expiration date: 12/31/18 $4 CO-PAY CARD $4 Pay as little as * per month * Eligibility required.
HIGHLIGHTS OF PRESCRIBING INFORMATION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use CELECOXIB CAPSULES safely and effectively. See full prescribing information for CELECOXIB CAPSULES.
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use CELECOXIB CAPSULES safely and effectively. See full prescribing information for CELECOXIB CAPSULES.
Immodium / loprarmide
 Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS See full prescribing information for complete boxed warning
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use CAMBIA safely and effectively. See full prescribing information for CAMBIA. CAMBIA (diclofenac potassium),
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use CAMBIA safely and effectively. See full prescribing information for CAMBIA. CAMBIA (diclofenac potassium),
CELEBREX (celecoxib) capsules, for oral use Initial U.S. Approval: 1998
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use CELEBREX safely and effectively. See full prescribing information for CELEBREX. CELEBREX (celecoxib)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use CELEBREX safely and effectively. See full prescribing information for CELEBREX. CELEBREX (celecoxib)
Reference ID:
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DUEXIS safely and effectively. See full prescribing information for DUEXIS. DUEXIS (ibuprofen and
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DUEXIS safely and effectively. See full prescribing information for DUEXIS. DUEXIS (ibuprofen and
Page 1 of 43. CELECOXIB capsules, for oral use Initial U.S. Approval: 1998
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use CELECOXIB CAPSULES safely and effectively. See full prescribing information for CELECOXIB CAPSULES.
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use CELECOXIB CAPSULES safely and effectively. See full prescribing information for CELECOXIB CAPSULES.
HIGHLIGHTS OF PRESCRIBING INFORMATION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use VOLTAREN GEL safely and effectively. See full prescribing information for VOLTAREN GEL. VOLTAREN
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use VOLTAREN GEL safely and effectively. See full prescribing information for VOLTAREN GEL. VOLTAREN
HIGHLIGHTS OF PRESCRIBING INFORMATION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use CELECOXIB CAPSULES safely and effectively. See full prescribing information for CELECOXIB CAPSULES.
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use CELECOXIB CAPSULES safely and effectively. See full prescribing information for CELECOXIB CAPSULES.
Name Brufen Flu Tablets & Suspension Description For the relief of the symptoms of colds and flu. Active Ingredients:
 Name Brufen Flu Tablets & Suspension Description For the relief of the symptoms of colds and flu Active Ingredients: Brufen Flu each tablet (Film-coated) contains: Ibuprofen 200 mg Pseudoephedrine hydrochloride
Name Brufen Flu Tablets & Suspension Description For the relief of the symptoms of colds and flu Active Ingredients: Brufen Flu each tablet (Film-coated) contains: Ibuprofen 200 mg Pseudoephedrine hydrochloride
PROFESSIONAL INFORMATION
 SCHEDULING STATUS: S1 PROPRIETARY NAME AND DOSAGE FORM: Aleve Tablets COMPOSITION: Each tablet contains naproxen sodium 220 mg (equivalent to 200 mg naproxen) CATEGORY AND CLASS: A / 2.7 Antipyretic or
SCHEDULING STATUS: S1 PROPRIETARY NAME AND DOSAGE FORM: Aleve Tablets COMPOSITION: Each tablet contains naproxen sodium 220 mg (equivalent to 200 mg naproxen) CATEGORY AND CLASS: A / 2.7 Antipyretic or
According to the Indications and Usage section of the approved product labeling (PI): 1
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Silver Spring, MD 20993 TRANSMITTED BY FACSIMILE George Marchesini Associate Director, Global Regulatory Affairs
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Silver Spring, MD 20993 TRANSMITTED BY FACSIMILE George Marchesini Associate Director, Global Regulatory Affairs
MESULID 100 REVISED PRODUCT INFORMATION
 MESULID 100 REVISED PRODUCT INFORMATION COMPOSITION Each caplet of MESULID 100 contains: Nimesulide 100 mg. Inactive Ingredients Lactose, microcrystalline cellulose, sodium starch glycolate, hydrogenated
MESULID 100 REVISED PRODUCT INFORMATION COMPOSITION Each caplet of MESULID 100 contains: Nimesulide 100 mg. Inactive Ingredients Lactose, microcrystalline cellulose, sodium starch glycolate, hydrogenated
STARTING THE CONVERSATION
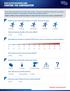 YOUR DOCTOR DISCUSSION GUIDE STARTING THE CONVERSATION If you have acute pain due to a minor strain, sprain, or bruise, it s important to talk with your doctor. Use this tool to start the conversation
YOUR DOCTOR DISCUSSION GUIDE STARTING THE CONVERSATION If you have acute pain due to a minor strain, sprain, or bruise, it s important to talk with your doctor. Use this tool to start the conversation
Elements for a Public Summary
 VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Osteoarthritis Osteoarthritis is the most common type of joint disease. It represents a group of conditions that result in changes
VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Osteoarthritis Osteoarthritis is the most common type of joint disease. It represents a group of conditions that result in changes
PACKAGE LEAFLET: INFORMATION FOR THE USER. EASOFEN MAX STRENGTH 400mg FILM-COATED TABLETS. Ibuprofen
 PACKAGE LEAFLET: INFORMATION FOR THE USER EASOFEN MAX STRENGTH 400mg FILM-COATED TABLETS Ibuprofen Read all of this leaflet carefully before you start taking Easofen Max Strength Tablets because it contains
PACKAGE LEAFLET: INFORMATION FOR THE USER EASOFEN MAX STRENGTH 400mg FILM-COATED TABLETS Ibuprofen Read all of this leaflet carefully before you start taking Easofen Max Strength Tablets because it contains
Voltaren 75 mg and 100 mg Prolongedrelease
 Voltaren 75 mg and 100 mg Prolongedrelease tablets NSAIDs: non-steroid anti-inflammatory drug DESCRIPTION AND COMPOSITION Pharmaceutical form Prolonged-release tablets Active substance The active substance
Voltaren 75 mg and 100 mg Prolongedrelease tablets NSAIDs: non-steroid anti-inflammatory drug DESCRIPTION AND COMPOSITION Pharmaceutical form Prolonged-release tablets Active substance The active substance
NEW ZEALAND DATA SHEET
 IBUPROFEN (Arrowcare) Ibuprofen Tablets 200mg Presentation NEW ZEALAND DATA SHEET White, capsule shaped, coated tablets with no markings. Uses Actions Ibuprofen is a non-steroidal anti-inflammatory agent.
IBUPROFEN (Arrowcare) Ibuprofen Tablets 200mg Presentation NEW ZEALAND DATA SHEET White, capsule shaped, coated tablets with no markings. Uses Actions Ibuprofen is a non-steroidal anti-inflammatory agent.
DOSAGE FORMS AND STRENGTHS ARTHROTEC (diclofenac/misoprostol) tablets: 50 mg/200 mcg and 75 mg/200 mcg (3)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ARTHROTEC safely and effectively. See full prescribing information for ARTHROTEC. ARTHROTEC (diclofenac
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ARTHROTEC safely and effectively. See full prescribing information for ARTHROTEC. ARTHROTEC (diclofenac
Durlaza. Durlaza (aspirin) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.85.13 Subject: Durlaza Page: 1 of 4 Last Review Date: September 15, 2016 Durlaza Description Durlaza
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.85.13 Subject: Durlaza Page: 1 of 4 Last Review Date: September 15, 2016 Durlaza Description Durlaza
Action Naproxen sodium, the active principle of Naproxan, has been developed as an analgesic because it is more rapidly absorbed than Naproxen.
 NAPROXAN Composition Each tablet contains Naproxen sodium 275 mg. Tablets Action Naproxen sodium, the active principle of Naproxan, has been developed as an analgesic because it is more rapidly absorbed
NAPROXAN Composition Each tablet contains Naproxen sodium 275 mg. Tablets Action Naproxen sodium, the active principle of Naproxan, has been developed as an analgesic because it is more rapidly absorbed
Please note that the information highlighted in grey colour is the additional information specific to the concerned dosage form.
 Proposed Core Safety Profile for diclofenac systemic formulations, including solution for injection, sugar-coated tablets, prolonged release tablets, dispersible tablets, gastro-resistant tablets and suppositories
Proposed Core Safety Profile for diclofenac systemic formulations, including solution for injection, sugar-coated tablets, prolonged release tablets, dispersible tablets, gastro-resistant tablets and suppositories
FELDENE (piroxicam) capsules, for oral use Initial U.S. Approval: 1982
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use FELDENE safely and effectively. See full prescribing information for FELDENE. FELDENE (piroxicam)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use FELDENE safely and effectively. See full prescribing information for FELDENE. FELDENE (piroxicam)
HIGHLIGHTS OF PRESCRIBING INFORMATION WARNINGS AND PRECAUTIONS
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use FLECTOR PATCH safely and effectively. See full prescribing information for FLECTOR PATCH. FLECTOR
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use FLECTOR PATCH safely and effectively. See full prescribing information for FLECTOR PATCH. FLECTOR
Part VI: Summary of the risk management plan by product
 Version 1.3, 28-Dec-2015 Risk Management Plan of a fixed combination of Dexketoprofen trometamol and Tramadol hydrochloride (oral formulation) Part VI: Summary of the risk management plan by product VI.1
Version 1.3, 28-Dec-2015 Risk Management Plan of a fixed combination of Dexketoprofen trometamol and Tramadol hydrochloride (oral formulation) Part VI: Summary of the risk management plan by product VI.1
DAYPRO (oxaprozin) caplets, for oral use Initial U.S. Approval: 1992
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DAYPRO safely and effectively. See full prescribing information for DAYPRO. DAYPRO (oxaprozin) caplets,
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DAYPRO safely and effectively. See full prescribing information for DAYPRO. DAYPRO (oxaprozin) caplets,
FLECTOR PATCH (diclofenac epolamine patch) 1.3%, for topical use Initial U.S. Approval: 1988
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use FLECTOR PATCH safely and effectively. See full prescribing information for FLECTOR PATCH. FLECTOR
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use FLECTOR PATCH safely and effectively. See full prescribing information for FLECTOR PATCH. FLECTOR
CLINICAL PHARMACOLOGY
 INDOCIN - indomethacin suppository Iroko Pharmaceuticals, LLC ---------- Cardiovas cular Thrombotic Events Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular
INDOCIN - indomethacin suppository Iroko Pharmaceuticals, LLC ---------- Cardiovas cular Thrombotic Events Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular
Celecoxib Powder, Diclofenac Powder, Flurbiprofen Powder, Ibuprofen Powder, Ketoprofen Powder, Meloxicam Powder, Tramadol Powder
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.02.26 Subject: Anti-Inflammatory Pain Powders Page: 1 of 5 Last Review Date: December 3, 2015 Anti-Inflammatory
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.02.26 Subject: Anti-Inflammatory Pain Powders Page: 1 of 5 Last Review Date: December 3, 2015 Anti-Inflammatory
Indomethacin Capsules, USP 25 mg
 Indomethacin Capsules, USP 25 mg Rx only WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS Cardiovascular Thrombotic Events Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased
Indomethacin Capsules, USP 25 mg Rx only WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS Cardiovascular Thrombotic Events Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased
EMEA PUBLIC STATEMENT ON PARECOXIB SODIUM (Dynastat/Rayzon/Xapit) RISK OF SERIOUS HYPERSENSITIVITY AND SKIN REACTIONS
 The European Agency for the Evaluation of Medicinal Products Post-authorisation evaluation of medicines for human use London, 22 October 2002 EMEA/25175/02 EMEA PUBLIC STATEMENT ON PARECOXIB SODIUM (Dynastat/Rayzon/Xapit)
The European Agency for the Evaluation of Medicinal Products Post-authorisation evaluation of medicines for human use London, 22 October 2002 EMEA/25175/02 EMEA PUBLIC STATEMENT ON PARECOXIB SODIUM (Dynastat/Rayzon/Xapit)
APO-CELECOXIB CAPSULES. 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzenesulfonamide
![APO-CELECOXIB CAPSULES. 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzenesulfonamide APO-CELECOXIB CAPSULES. 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzenesulfonamide](/thumbs/90/102200709.jpg) APO-CELECOXIB CAPSULES NAME OF THE MEDICINE Celecoxib Chemical Name: 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzenesulfonamide Structural Formula: Molecular Formula: C17H14F3N3O2S Molecular
APO-CELECOXIB CAPSULES NAME OF THE MEDICINE Celecoxib Chemical Name: 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzenesulfonamide Structural Formula: Molecular Formula: C17H14F3N3O2S Molecular
WHERE IT HURTS GET RELIEF THAT WORKS GET TARGETED, TOPICAL PENNSAID 2% FOR OSTEOARTHRITIS KNEE PAIN, SELECT IMPORTANT SAFETY INFORMATION
 FOR OSTEOARTHRITIS KNEE PAIN, GET RELIEF THAT WORKS WHERE IT HURTS GET TARGETED, TOPICAL PENNSAID 2% PENNSAID (diclofenac sodium topical solution) 2% w/w is a nonsteroidal anti-inflammatory drug (NSAID)
FOR OSTEOARTHRITIS KNEE PAIN, GET RELIEF THAT WORKS WHERE IT HURTS GET TARGETED, TOPICAL PENNSAID 2% PENNSAID (diclofenac sodium topical solution) 2% w/w is a nonsteroidal anti-inflammatory drug (NSAID)
500 mg Tablets, 50 mg/5 ml Suspension 500 mg Capsules 250 mg Capsules
 MEFENAMIC ACID PONSTAN PONSTAN SF 500 mg Tablets, 50 mg/5 ml Suspension 500 mg Capsules 250 mg Capsules Absolute contraindications: Not to be given to those patients who have history of: Stroke: cerebrovascular
MEFENAMIC ACID PONSTAN PONSTAN SF 500 mg Tablets, 50 mg/5 ml Suspension 500 mg Capsules 250 mg Capsules Absolute contraindications: Not to be given to those patients who have history of: Stroke: cerebrovascular
Migraleve, Migraleve Pink and Migraleve Yellow Product Information
 Migraleve, Migraleve Pink and Migraleve Yellow Product Information Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard Adverse events should also
Migraleve, Migraleve Pink and Migraleve Yellow Product Information Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard Adverse events should also
PRODUCT CIRCULAR. Tablets COZAAR (losartan potassium) I. THERAPEUTIC CLASS II. INDICATIONS III. DOSAGE AND ADMINISTRATION PAK-CZR-T
 PRODUCT CIRCULAR Tablets I. THERAPEUTIC CLASS, the first of a new class of agents for the treatment of hypertension, is an angiotensin II receptor (type AT 1 ) antagonist. also provides a reduction in
PRODUCT CIRCULAR Tablets I. THERAPEUTIC CLASS, the first of a new class of agents for the treatment of hypertension, is an angiotensin II receptor (type AT 1 ) antagonist. also provides a reduction in
Ketorolac Tromethamine Injection, USP FOR INTRAVENOUS/INTRAMUSCULAR USE (15 mg and 30 mg) FOR INTRAMUSCULAR USE ONLY (60 mg)
 Ketorolac Tromethamine Injection, USP FOR INTRAVENOUS/INTRAMUSCULAR USE (15 mg and 30 mg) FOR INTRAMUSCULAR USE ONLY (60 mg) Rx only WARNING Ketorolac tromethamine, a nonsteroidal anti-inflammatory drug
Ketorolac Tromethamine Injection, USP FOR INTRAVENOUS/INTRAMUSCULAR USE (15 mg and 30 mg) FOR INTRAMUSCULAR USE ONLY (60 mg) Rx only WARNING Ketorolac tromethamine, a nonsteroidal anti-inflammatory drug
Strength: 7.5 mg and 15 mg Pack Size: 90, 100 and 500 Tablets per bottle Revision No.: 02
 EMERGENCY OVERVIEW Each Meloxicam Tablets intended for oral administration contains Meloxicam and excipients generally considered to be non- toxic and non-hazardous in small quantities and under conditions
EMERGENCY OVERVIEW Each Meloxicam Tablets intended for oral administration contains Meloxicam and excipients generally considered to be non- toxic and non-hazardous in small quantities and under conditions
Diclofenac Potassium Tablets, USP. 50 mg. Rx Only
 Diclofenac Potassium Tablets, USP 50 mg Rx Only Cardiovascular Risk NSAIDs may cause an increased risk of serious cardiovascular thrombotic events, myocardial infarction, and stroke, which can be fatal.
Diclofenac Potassium Tablets, USP 50 mg Rx Only Cardiovascular Risk NSAIDs may cause an increased risk of serious cardiovascular thrombotic events, myocardial infarction, and stroke, which can be fatal.
Package Insert. Spasfree
 Package Insert Spasfree Product Summary 1. Name of the medicinal product Spasfree 2. Qualitative and quantitative composition Each film-coated tablet contains Aceclofenac 100 mg and drotaverine hydrochloride
Package Insert Spasfree Product Summary 1. Name of the medicinal product Spasfree 2. Qualitative and quantitative composition Each film-coated tablet contains Aceclofenac 100 mg and drotaverine hydrochloride
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Melfen 200 mg Film-coated Tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 200 mg ibuprofen. Excipients:
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Melfen 200 mg Film-coated Tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 200 mg ibuprofen. Excipients:
INDICATIONS Treatment of Chronic Iron Overload Due to Blood Transfusions (Transfusional Iron Overload) EXJADE (deferasirox) is indicated for the
 INDICATIONS Treatment of Chronic Iron Overload Due to Blood Transfusions (Transfusional Iron Overload) EXJADE (deferasirox) is indicated for the treatment of chronically elevated levels of iron in the
INDICATIONS Treatment of Chronic Iron Overload Due to Blood Transfusions (Transfusional Iron Overload) EXJADE (deferasirox) is indicated for the treatment of chronically elevated levels of iron in the
PRODUCT MONOGRAPH. FLOCTAFENINE Floctafenine Tablets 200 mg and 400 mg THERAPEUTIC CLASSIFICATION. Anti-inflammatory, Analgesic
 0 PRODUCT MONOGRAPH FLOCTAFENINE Floctafenine Tablets 200 mg and 400 mg THERAPEUTIC CLASSIFICATION Anti-inflammatory, Analgesic INFORMATION FOR THE PATIENT FLOCTAFENINE, which has been prescribed to you
0 PRODUCT MONOGRAPH FLOCTAFENINE Floctafenine Tablets 200 mg and 400 mg THERAPEUTIC CLASSIFICATION Anti-inflammatory, Analgesic INFORMATION FOR THE PATIENT FLOCTAFENINE, which has been prescribed to you
Pharmaceutical form(s)/strength: Capsules, 200mg, 400mg, Oral suspensions, 90mg/5ml, 180mg/5ml, 36 mg/ml SI/H/PSUR/0002/002 Date of FAR:
 0BCore Safety Profile Active substance: Ceftibuten Pharmaceutical form(s)/strength: Capsules, 200mg, 400mg, Oral suspensions, 90mg/5ml, 180mg/5ml, 36 mg/ml P-RMS: SI/H/PSUR/0002/002 Date of FAR: 14.02.2013
0BCore Safety Profile Active substance: Ceftibuten Pharmaceutical form(s)/strength: Capsules, 200mg, 400mg, Oral suspensions, 90mg/5ml, 180mg/5ml, 36 mg/ml P-RMS: SI/H/PSUR/0002/002 Date of FAR: 14.02.2013
Fact Sheet. Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII
 Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII Fact Sheet Zohydro ER (hydrocodone bitartrate) Extended-Release Capsule, CII, is a long-acting (extendedrelease) type of pain medication
Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII Fact Sheet Zohydro ER (hydrocodone bitartrate) Extended-Release Capsule, CII, is a long-acting (extendedrelease) type of pain medication
PRODUCT MONOGRAPH. VOLTAREN SR (diclofenac sodium)
 PRODUCT MONOGRAPH Pr VOLTAREN Pr VOLTAREN SR (diclofenac sodium) 50 mg Enteric-Coated Tablets 75 and 100 mg Slow-Release Tablets 50 and 100 mg Suppositories Acetic Acid Derivatives and Related Substances
PRODUCT MONOGRAPH Pr VOLTAREN Pr VOLTAREN SR (diclofenac sodium) 50 mg Enteric-Coated Tablets 75 and 100 mg Slow-Release Tablets 50 and 100 mg Suppositories Acetic Acid Derivatives and Related Substances
PRODUCT MONOGRAPH SANDOZ DICLOFENAC. (diclofenac sodium) 50 mg Enteric-Coated Tablets 75 and 100 mg Slow-Release Tablets 50 and 100 mg Suppositories
 PRODUCT MONOGRAPH Pr SANDOZ DICLOFENAC Pr SANDOZ DICLOFENAC SR (diclofenac sodium) 50 mg Enteric-Coated Tablets 75 and 100 mg Slow-Release Tablets 50 and 100 mg Suppositories Acetic Acid Derivatives and
PRODUCT MONOGRAPH Pr SANDOZ DICLOFENAC Pr SANDOZ DICLOFENAC SR (diclofenac sodium) 50 mg Enteric-Coated Tablets 75 and 100 mg Slow-Release Tablets 50 and 100 mg Suppositories Acetic Acid Derivatives and
GENRX PIROXICAM CAPSULES
 GENRX PIROXICAM CAPSULES NAME OF THE MEDICINE Piroxicam. Chemical name: 4-hydroxy-2-methyl-N-2-pyridinyl-2H-1, 2-benzothiazine-3-carboxamide 1, 1- dioxide. It is a member of a new chemical class of non-steroidal
GENRX PIROXICAM CAPSULES NAME OF THE MEDICINE Piroxicam. Chemical name: 4-hydroxy-2-methyl-N-2-pyridinyl-2H-1, 2-benzothiazine-3-carboxamide 1, 1- dioxide. It is a member of a new chemical class of non-steroidal
Risk Management Plan Etoricoxib film-coated tablets
 VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Osteoarthritis (OA): OA is a condition in which the cartilage of the joints is broken down. This causes stiffness, pain and leads
VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Osteoarthritis (OA): OA is a condition in which the cartilage of the joints is broken down. This causes stiffness, pain and leads
Celecoxib is a diaryl substituted pyrazole and has the following chemical structure and formula:
 PRODUCT INFORMATION CELEBREX 100 mg, CELEBREX 200 mg and CELEBREX 400 mg Capsules NAME OF THE MEDICINE Celecoxib 100 mg Celecoxib 200 mg Celecoxib 400 mg Celecoxib is a diaryl substituted pyrazole and
PRODUCT INFORMATION CELEBREX 100 mg, CELEBREX 200 mg and CELEBREX 400 mg Capsules NAME OF THE MEDICINE Celecoxib 100 mg Celecoxib 200 mg Celecoxib 400 mg Celecoxib is a diaryl substituted pyrazole and
JUN Re : Docket No. 2005P-0072/CP1
 , J~F~ySERVIC g,g~' 0 b < x 0 Y,` I h~byd3~ DEPARTMENT OF HEALTH & HUMAN SERVICES JUN 2 2 2006 Public Health Service Food and Drug Administration Rockvilie MD 20857 Roger E, Salisbury, MD Professor of
, J~F~ySERVIC g,g~' 0 b < x 0 Y,` I h~byd3~ DEPARTMENT OF HEALTH & HUMAN SERVICES JUN 2 2 2006 Public Health Service Food and Drug Administration Rockvilie MD 20857 Roger E, Salisbury, MD Professor of
PRODUCT MONOGRAPH. (diclofenac potassium) 50 mg Sugar-Coated Tablets. Nonsteroidal Anti-Inflammatory Drug (NSAID)
 PRODUCT MONOGRAPH Pr SANDOZ DICLOFENAC RAPIDE (diclofenac potassium) 50 mg Sugar-Coated Tablets Nonsteroidal Anti-Inflammatory Drug (NSAID) Sandoz Canada Inc. Date of Revision: September 30, 2014 145 Jules
PRODUCT MONOGRAPH Pr SANDOZ DICLOFENAC RAPIDE (diclofenac potassium) 50 mg Sugar-Coated Tablets Nonsteroidal Anti-Inflammatory Drug (NSAID) Sandoz Canada Inc. Date of Revision: September 30, 2014 145 Jules
Read all of this leaflet carefully because it contains important information for you.
 Nurofen Plus Tablets Ibuprofen 200mg, Codeine Phosphate Hemihydrate 12.8mg ADVANCED DUAL ACTION FOR POWERFUL RELIEF Read all of this leaflet carefully because it contains important information for you.
Nurofen Plus Tablets Ibuprofen 200mg, Codeine Phosphate Hemihydrate 12.8mg ADVANCED DUAL ACTION FOR POWERFUL RELIEF Read all of this leaflet carefully because it contains important information for you.
PRODUCT MONOGRAPH SANDOZ DICLOFENAC. (diclofenac sodium)
 PRODUCT MONOGRAPH Pr SANDOZ DICLOFENAC Pr SANDOZ DICLOFENAC SR (diclofenac sodium) 25 mg and 50 mg Enteric-Coated Tablets 75 and 100 mg Slow-Release Tablets 50 and 100 mg Suppositories Nonsteroidal Anti-Inflammatory
PRODUCT MONOGRAPH Pr SANDOZ DICLOFENAC Pr SANDOZ DICLOFENAC SR (diclofenac sodium) 25 mg and 50 mg Enteric-Coated Tablets 75 and 100 mg Slow-Release Tablets 50 and 100 mg Suppositories Nonsteroidal Anti-Inflammatory
FDA strengthens warning that non-aspirin nonsteroidal antiinflammatory drugs (NSAIDs) can cause heart attacks or strokes
 FDA strengthens warning that non-aspirin nonsteroidal antiinflammatory drugs (NSAIDs) can cause heart attacks or strokes Safety Announcement [7-9-2015] The U.S. Food and Drug Administration (FDA) is strengthening
FDA strengthens warning that non-aspirin nonsteroidal antiinflammatory drugs (NSAIDs) can cause heart attacks or strokes Safety Announcement [7-9-2015] The U.S. Food and Drug Administration (FDA) is strengthening
PRODUCT INFORMATION Celaxib Celecoxib
 PRODUCT INFORMATION Celaxib Celecoxib NAME OF THE MEDICINE Active ingredient: Chemical name: Celecoxib 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl] benzenesulfonamide Structural formula:
PRODUCT INFORMATION Celaxib Celecoxib NAME OF THE MEDICINE Active ingredient: Chemical name: Celecoxib 4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl] benzenesulfonamide Structural formula:
PRODUCT MONOGRAPH. (diclofenac potassium) 50 mg Tablets. Teva Standard
 PRODUCT MONOGRAPH Pr TEVA-DICLOFENAC-K (diclofenac potassium) 50 mg Tablets Teva Standard Acetic Acid Derivatives and Related Substances Teva Canada Limited 30 Novopharm Court Toronto, Ontario Canada M1B
PRODUCT MONOGRAPH Pr TEVA-DICLOFENAC-K (diclofenac potassium) 50 mg Tablets Teva Standard Acetic Acid Derivatives and Related Substances Teva Canada Limited 30 Novopharm Court Toronto, Ontario Canada M1B
NEW ZEALAND DATA SHEET. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each coated tablet contains 200 mg of ibuprofen.
 1. PRODUCT NAME IBUPROFEN, coated tablets 200 mg, Teva NEW ZEALAND DATA SHEET 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each coated tablet contains 200 mg of ibuprofen. Excipient with known effect: Lactose
1. PRODUCT NAME IBUPROFEN, coated tablets 200 mg, Teva NEW ZEALAND DATA SHEET 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each coated tablet contains 200 mg of ibuprofen. Excipient with known effect: Lactose
Sun Pharma had acquired YONSA commercialize YONSA in the U.S. from Churchill Pharmaceuticals and will
 Sun Pharma Announces USFDA Approval of YONSA (abiraterone acetate) To Treat Metastatic Castration- Resistant Prostate Cancer In Combination With Methylprednisolone YONSA was shown in clinical studies to
Sun Pharma Announces USFDA Approval of YONSA (abiraterone acetate) To Treat Metastatic Castration- Resistant Prostate Cancer In Combination With Methylprednisolone YONSA was shown in clinical studies to
פורמט עלון זה נקבע ע"י משרד הבריאות ותוכנו נבדק ואושר SUMMARY OF PRODUCT CHARACTERISTICS
 פורמט עלון זה נקבע ע"י משרד הבריאות ותוכנו נבדק ואושר SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT FELDENE GEL 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each gram contains 5mg
פורמט עלון זה נקבע ע"י משרד הבריאות ותוכנו נבדק ואושר SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT FELDENE GEL 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each gram contains 5mg
The intranasal Nonsteroidal Anti-Inflammatory Drug (NSAID) that provides pain relief at the opioid level
 The intranasal Nonsteroidal Anti-Inflammatory Drug (NSAID) that provides pain relief at the opioid level Instructions for the use of SPRIX Nasal Spray About SPRIX A Before you use SPRIX for the first time,
The intranasal Nonsteroidal Anti-Inflammatory Drug (NSAID) that provides pain relief at the opioid level Instructions for the use of SPRIX Nasal Spray About SPRIX A Before you use SPRIX for the first time,
disease or in clients who consume alcohol on a regular basis. bilirubin
 NON-OPIOID Acetaminophen(Tylenol) Therapeutic class: Analgesic, antipyretic Aspirin (ASA, Acetylsalicylic Acid) Analgesic, NSAID, antipyretic Non-Opioid Analgesics COMMON USES WHAT I NEED TO KNOW AS A
NON-OPIOID Acetaminophen(Tylenol) Therapeutic class: Analgesic, antipyretic Aspirin (ASA, Acetylsalicylic Acid) Analgesic, NSAID, antipyretic Non-Opioid Analgesics COMMON USES WHAT I NEED TO KNOW AS A
Salicylates: Interactions 10/14/2009. Salicylates DRUGS USED IN THE MANAGEMENT OF MUSCULOSKELETAL DISORDERS. Chapters 17, 18, 34 & Pages 577 &
 DRUGS USED IN THE MANAGEMENT OF MUSCULOSKELETAL DISORDERS Chapters 17, 18, 34 & Pages 577 & 579-586 Salicylates aspirin Have analgesic, antipyretic, and anti-inflammatory effects. Inhibits the production
DRUGS USED IN THE MANAGEMENT OF MUSCULOSKELETAL DISORDERS Chapters 17, 18, 34 & Pages 577 & 579-586 Salicylates aspirin Have analgesic, antipyretic, and anti-inflammatory effects. Inhibits the production
Etodolac Capsules, USP 200 and 300 mg. Rx only
 Etodolac Capsules, USP 200 and 300 mg Rx only Cardiovascular Thrombotic Events Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including
Etodolac Capsules, USP 200 and 300 mg Rx only Cardiovascular Thrombotic Events Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including
Translation from Latvian Approved by SAM on PACKAGE LEAFLET: INFORMATION FOR THE USER. IBUPROFEN 200 mg film-coated tablets Ibuprofenum
 Translation from Latvian Approved by SAM on 31-01-2013 PACKAGE LEAFLET: INFORMATION FOR THE USER IBUPROFEN 200 mg film-coated tablets Ibuprofenum Read all of this leaflet carefully because it contains
Translation from Latvian Approved by SAM on 31-01-2013 PACKAGE LEAFLET: INFORMATION FOR THE USER IBUPROFEN 200 mg film-coated tablets Ibuprofenum Read all of this leaflet carefully because it contains
AUSTRALIAN PRODUCT INFORMATION CALDOLOR
 AUSTRALIAN CALDOLOR (IBUPROFEN) 100 mg/ml CONCENTRATED INJECTION 1 NAME OF THE MEDICINE Ibuprofen 2 QUALITATIVE AND QUANTITATIVE COMPOSITION concentrated injection contains the active ingredient ibuprofen,
AUSTRALIAN CALDOLOR (IBUPROFEN) 100 mg/ml CONCENTRATED INJECTION 1 NAME OF THE MEDICINE Ibuprofen 2 QUALITATIVE AND QUANTITATIVE COMPOSITION concentrated injection contains the active ingredient ibuprofen,
NOTOPAIN CAPLETS. Diclofenac Sodium + Paracetamol. Composition. Each tablet contains: Diclofenac Sodium BP 50mg Paracetamol BP 500mg.
 NOTOPAIN CAPLETS Diclofenac Sodium + Paracetamol Composition Each tablet contains: Diclofenac Sodium BP 50mg Paracetamol BP 500mg Pharmacology Phamacodynamics Diclofenac relieves pain and inflammation
NOTOPAIN CAPLETS Diclofenac Sodium + Paracetamol Composition Each tablet contains: Diclofenac Sodium BP 50mg Paracetamol BP 500mg Pharmacology Phamacodynamics Diclofenac relieves pain and inflammation
Excipients: Each diclofenac potassium 50 mg tablet contains 5.8 mg of potassium.
 AUSTRALIAN PI VOLTAREN RAPID (DICLOFENAC POTASSIUM) TABLETS 1 NAME OF THE MEDICINE Active ingredient: Diclofenac Potassium 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Voltaren Rapid tablets contain 50 mg
AUSTRALIAN PI VOLTAREN RAPID (DICLOFENAC POTASSIUM) TABLETS 1 NAME OF THE MEDICINE Active ingredient: Diclofenac Potassium 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Voltaren Rapid tablets contain 50 mg
Core Safety Profile. Pharmaceutical form(s)/strength: Film-coated tablet UK/H/PSUR/0032/001 Date of FAR:
 Core Safety Profile Active substance: Etoricoxib Pharmaceutical form(s)/strength: Film-coated tablet P-RMS: UK/H/PSUR/0032/001 Date of FAR: 25.03.2009 4.2 Posology and method of administration /.../ is
Core Safety Profile Active substance: Etoricoxib Pharmaceutical form(s)/strength: Film-coated tablet P-RMS: UK/H/PSUR/0032/001 Date of FAR: 25.03.2009 4.2 Posology and method of administration /.../ is
FDA Approves Carnexiv (carbamazepine) injection as Intravenous Short-Term Replacement Therapy for Certain Seizure Types
 FOR IMMEDIATE RELEASE FDA Approves Carnexiv (carbamazepine) injection as Intravenous Short-Term Replacement Therapy for Certain Seizure Types Carnexiv is the first FDA-approved intravenous carbamazepine
FOR IMMEDIATE RELEASE FDA Approves Carnexiv (carbamazepine) injection as Intravenous Short-Term Replacement Therapy for Certain Seizure Types Carnexiv is the first FDA-approved intravenous carbamazepine
