Transmittal 130 Date: January 14, 2011
|
|
|
- Grant Cross
- 5 years ago
- Views:
Transcription
1 anual ystem Pub edicare National overage Determinations Department of ealth & uman ervices (D) enters for edicare & edicaid ervices () Transmittal 130 Date: January 14, 2011 hange equest 7235 UBJET: ome Oxygen Use to Treat luster eadache (). UY O NGE: fter careful reconsideration, effective for claims with dates of service on and after January 4, 2011, edicare will allow for coverage of home use of oxygen to treat edicare beneficiaries diagnosed with when beneficiaries are enrolled in clinical studies that are approved by for the purpose of gaining further evidence. This revision to the edicare National overage Determinations anual is a national coverage determination (ND). NDs are binding on all carriers, fiscal intermediaries, contractors with the ederal government that review and/or adjudicate claims, determinations, and/or decisions, quality improvement organizations, qualified independent contractors, the edicare appeals council, and administrative law judges (LJs) (see 42 section (a)(4) (2005)). n ND that expands coverage is also binding on a edicare advantage organization. n addition, an LJ may not review an ND. (ee section 1869(f)(1)()(i) of the ocial ecurity ct.) EETVE DTE: January 4, 2011 PLEENTTON DTE: ebruary 15, 2011 Disclaimer for manual changes only: The revision date and transmittal number apply only to red italicized material. ny other material was previously published and remains unchanged. owever, if this revision contains a table of contents, you will receive the new/revised information only, and not the entire table of contents.. NGE N NUL NTUTON: (N/ if manual is not updated) =EVED, N=NEW, D=DELETED-Only One Per ow. /N/D N PTE / ETON / UBETON / TTLE 1/Table of ontents N 1/ /ome Oxygen Use to Treat luster eadache () - Effective January 4, UNDNG: or iscal ntermediaries (s), egional ome ealth ntermediaries (s) and/or arriers: No additional funding will be provided by ; ontractor activities are to be carried out within their operating budgets. or edicare dministrative ontractors (s): The edicare dministrative ontractor is hereby advised that this constitutes technical direction as defined in your contract. does not construe this as a change to the tatement of Work. The contractor is not obligated to incur costs in excess of the amounts allotted in your contract unless and until specifically
2 authorized by the ontracting Officer. f the contractor considers anything provided, as described above, to be outside the current scope of work, the contractor shall withhold performance on the part(s) in question and immediately notify the ontracting Officer, in writing or by , and request formal directions regarding continued performance requirements. V. TTENT: Business equirements anual nstruction *Unless otherwise specified, the effective date is the date of service.
3 ttachment - Business equirements Pub Transmittal: 130 Date: January 14, 2011 hange equest: 7235 UBJET: ome Oxygen Use to Treat luster eadache () Effective Date: January 4, 2011 mplementation Date: ebruary 15, GENEL NOTON. Background: edicare has a National overage Determination (ND) on the home use of oxygen (240.2 dated October 1993) that states it is reasonable and necessary for patients with significant hypoxemia, as evidenced by a blood gas study or a measurement of arterial oxygen saturation. n arch 2006, an internally generated ND at Publication (Pub.) , chapter 1, section , led to coverage with study participation for those beneficiaries who did not qualify for coverage based on the initial criteria for hypoxemia established in ND Pub ,hapter 1, section This expansion in coverage requires that beneficiaries be enrolled subjects in clinical trials sponsored by the National eart, Lung, and Blood nstitute. urrent national policy states that the home use of oxygen is reasonable and necessary for only those patients diagnosed with significant hypoxemia in conjunction with certain health conditions. n pril 2010, the enters for edicare & edicaid ervices () received an external request from the merican eadache ociety and the merican cademy of Neurology requesting an ND to determine if home oxygen therapy is reasonable and necessary for edicare patients diagnosed with cluster headache (). The requestors asked that issue a revision to its current national coverage policy that would add as a covered indication for home oxygen therapy. B. Policy: fter careful reconsideration, effective for claims with dates of service on and after January 4, 2011, edicare will allow for coverage of home use of oxygen to treat edicare beneficiaries diagnosed with when beneficiaries are enrolled in clinical studies that are approved by for the purpose of gaining further evidence. edicare will allow for coverage of the following: Beneficiaries with participating in an approved prospective clinical study comparing normobaric 100% oxygen with at least one clinically appropriate comparator for the treatment of. The clinical study must address whether home use of oxygen improves edicare beneficiaries health outcomes and is subject to the criteria as outlined in Pub , of the ND anual, chapter 1, section NOTE: ontractors shall use existing clinical trial coding conventions to help identify on a claim that the ome use of Oxygen for was provided pursuant to a edicare-approved clinical study under ED, in addition to the D-9- diagnosis codes for (339.00, cluster headache syndrome unspecified, , cluster headache episodic, and , cluster headache, chronic), P code E1399, durable medical equipment, miscellaneous, and place of service (PO) 12, home. The 8-digit clinical trial number is optional. NOTE: urrently, there are no clinical trials approved or pending approval for the home use of oxygen for. Ns are not required in the context of this clinical trial setting. This is a Part B DE benefit only. NOTE: There are no updates to the laims Processing anual at this time. ontractors should refer to the business requirements below, as well as general clinical trial billing requirements at Pub , chapter 1, section 310, and Pub , chapter 32, section 69.
4 . BUNE EQUEENT TBLE Number equirement esponsibility (place an X in each applicable column) / D hared- ystem OT E B E aintainers Effective for claims with dates of service on and after January 4, 2011, edicare will allow for coverage of home use of oxygen for treatment of only for edicare beneficiaries who are diagnosed with and enrolled in an approved clinical trial under ED as specified at Pub , ND anual chapter 1, section ontractors shall pay claims with dates of service on or after January 4, 2011, for home use of oxygen for the treatment of ONLY if they contain all of the following: X X E V W -P code E D-9- diagnosis code(s) , , or linical Trial D-9- diagnosis code V70.7 -linical Trial Procedure ode odifier Q0 -PO 12 (home) 8-digit clinical trial number (optional) ontractors shall deny claims for dates of service on and after January 4, 2011, for home use of oxygen for the treatment of that do not contain all of the coding included in B above using the following messages: X 50 - These are non-covered services because this is not deemed a 'medical necessity' by the payer. Note: efer to the 835 ealthcare Policy dentification egment (loop 2110 ervice Payment nformation E), if present. N386 - This decision was based on a National overage Determination (ND). n ND provides a coverage determination as to whether a particular item or service is covered. copy of this policy is available at f you do not have web access, you may contact the contractor to request a copy of the ND.
5 Number equirement esponsibility (place an X in each applicable column) / D hared- ystem OT E B E aintainers Group ode - Patient esponsibility (P) if BN/NN given, otherwise ontractual Obligation (O) E V W N This service/item was not covered because it was not provided as part of a qualifying trial/study. (Este servicio/artículo no fue cubierto porque no estaba incluido como parte de un ensayo clínico/estudio calificado.) or home oxygen for the treatment of claims with dates of service between January 4, 2011, and the implementation date of this, contractors shall perform necessary adjustments only when affected claims are brought to their attention. X. POVDE EDUTON TBLE Number equirement esponsibility (place an X in each applicable column) / D hared- ystem OT E B E aintainers provider education article related to this instruction will be available at shortly after the is released. You will receive notification of the article release via the established "LN atters" listserv. ontractors shall post this article, or a direct link to this article, on their Web site and include information about it in a listserv message within one week of the availability of the provider education article. n addition, the provider education article shall be included in your next regularly scheduled bulletin. ontractors are free to supplement LN atters articles with localized information that would benefit their provider community in billing and administering the edicare program correctly. X E V W
6 V. UPPOTNG NOTON ection : or any recommendations and supporting information associated with listed requirements, use the box below: N/ X-ef equireme nt Number ecommendations or other supporting information: ection B: or all other recommendations and supporting information, use this space: N V. ONTT Pre-mplementation ontact(s): arah eisenberg, (coverage), , sarah.meisenberg@cms.hhs.gov, Pat Brocato-imons (coverage), , patricia.brocatosimons@cms.hhs.gov, Bobbett Plummer, DE lams Processing, , Bobbett.plummer@cms.hhs.gov. Post-mplementation ontact(s): ppropriate O V. UNDNG ection : or iscal ntermediaries (s), egional ome ealth ntermediaries (s), and/or arriers: No additional funding will be provided by ; contractor activities are to be carried out within their operating budgets. ection B: or edicare dministrative ontractors (s), include the following statement: The edicare dministrative ontractor is hereby advised that this constitutes technical direction as defined in your contract. does not construe this as a change to the tatement of Work. The contractor is not obligated to incur costs in excess of the amounts allotted in your contract unless and until specifically authorized by the ontracting Officer. f the contractor considers anything provided, as described above, to be outside the current scope of work, the contractor shall withhold performance on the part(s) in question and immediately notify the ontracting Officer, in writing or by , and request formal directions regarding continued performance requirements.
7 edicare National overage Determinations anual hapter 1, Part 4 (ections ) overage Determinations Table of ontents (ev.130, ssued: ) ome Oxygen Use to Treat luster eadache () (Effective January 4, 2011)
8 ome Oxygen Use to Treat luster eadache () (Effective January 4, 2011) (ev. 130, ssued: , Effective: , mplementation: ). General luster headache (), as described in arrison s Principles of nternal edicine 16 th edition, is an episodic (most common), or chronic unilateral headache syndrome that begins with one to three short-lived headaches per day over many weeks followed by a period of remission. There may be a regular recurrence in the vast majority of attacks. When it becomes chronic, it is characterized by the absence of sustained periods of remission. Generally the cause is unknown but associations can occur with alcohol use which is the only known dietary trigger of. There are other triggers such as strong odors (mainly solvents and cigarette smoke) and napping. is also characterized by unilateral, excruciating pain principally in ocular, frontal, and temporal areas, as well as ipsilateral lacrimation, conjunctival injection, photophobia, and nasal stuffiness. ttacks may happen at precise hours, especially at night. The medical literature includes anecdotal reports of the use of 100% normobaric oxygen for the treatment of. Oxygen is an odorless, colorless gas at room temperature. t can be delivered in a chamber, by compressed air, via oxygen concentrator, or other method. Though often thought of as harmless, oxygen use has been noted to have adverse effects including blindness, pulmonary fibrosis, and suppression of the drive to breathe in patients who have advanced chronic obstructive lung disease. Oxygen is also known to increase fire risk in certain environments. There are a number of drug treatments for, including but not limited to V and sublingual sumatriptan. Effective prophylactic drugs include prednisone, lithium, ethysergide, ergotamine, sodium valproate, and verapamil. t present, there is no curative treatment. B. Nationally overed ndications Effective for claims with dates of services on or after January 4, 2011, the enters for edicare & edicaid ervices () believes that the available evidence suggests that the home use of oxygen to treat is promising and supports further research under 1862(a)(1)(E) of the ocial ecurity ct (the ct) through the overage With tudy Participation (P) form of overage With Evidence Development (ED). The home use of oxygen to treat is covered by edicare only for beneficiaries with participating in an approved prospective clinical study comparing normobaric 100% oxygen (NBOT) with at least one clinically appropriate comparator for the treatment of. The clinical study must address one or more aspects of the following questions:
9 1. Prospectively, compared to individuals with cluster headache who do not receive NBOT, do edicare beneficiaries with who receive NBOT have improved outcomes as indicated by: a. Pain relief b. Time to pain relief c. Durability of pain relief 2. Prospectively, among edicare beneficiaries with cluster headache, which method of oxygen delivery provides the most benefit as indicated by: a. Pain relief b. Time to pain relief c. Durability of pain relief 3. Prospectively, among edicare beneficiaries with cluster headache, what other factors, if any, predict the patient s response to 100% oxygen therapy as indicated by: a. Pain relief b. Time to pain relief c. Durability of pain relief Only those beneficiaries diagnosed with the condition of cluster headache are eligible for participation in a clinical study. adopts the diagnostic criteria used by the nternational eadache ociety to form a definitive diagnosis of. Therefore, the home use of oxygen to treat is covered by edicare only when furnished to edicare beneficiaries who have had at least five severe to very severe unilateral headache attacks lasting minutes when untreated. (ntensity of pain: Degree of pain usually expressed in terms of its functional consequence and scored on a verbal 5-point scale: 0=no pain; 1=mild pain, does not interfere with usual activities; 2=moderate pain, inhibits but does not wholly prevent usual activities; 3=severe pain, prevents all activities; 4=very severe pain. t may also be expressed on a visual analogue scale. The headaches must be accompanied by at least one of the following findings: 1. ipsilateral conjunctival injection and/or lacrimation; or 2. ipsilateral nasal congestion and/or rhinorrhea; or 3. ipsilateral eyelid edema; or 4. ipsilateral forehead and facial sweating; or 5. ipsilateral miosis and/or ptosis; or 6. a sense of restlessness or agitation.
10 The clinical study must adhere to the following standards of scientific integrity and relevance to the edicare population: a. The principal purpose of the research study is to test whether a particular intervention potentially improves the participants health outcomes. b. The research study is well supported by available scientific and medical information or it is intended to clarify or establish the health outcomes of interventions already in common clinical use. c. The research study does not unjustifiably duplicate existing studies. d. The research study design is appropriate to answer the research question being asked in the study. e. The research study is sponsored by an organization or individual capable of executing the proposed study successfully. f. The research study is in compliance with all applicable ederal regulations concerning the protection of human subjects found at 45 Part 46. f a study is regulated by the ood and Drug dministration (D), it must be in compliance with 21 parts 50 and 56. g. ll aspects of the research study are conducted according to appropriate standards of scientific integrity (see h. The research study has a written protocol that clearly addresses, or incorporates by reference, the standards listed here as edicare requirements for ED coverage. i. The clinical research study is not designed to exclusively test toxicity or disease pathophysiology in healthy individuals. Trials of all medical technologies measuring therapeutic outcomes as one of the objectives meet this standard only if the disease or condition being studied is life threatening as defined in (a) and the patient has no other viable treatment options. j. The clinical research study is registered on the linicaltrials.gov website by the principal sponsor/investigator prior to the enrollment of the first study subject. k. The research study protocol specifies the method and timing of public release of all prespecified outcomes to be measured including release of outcomes if outcomes are negative or study is terminated early. The results must be made public within 24 months of the end of data collection. f a report is planned to be published in a peer reviewed journal, then that initial release may be an abstract that meets the requirements of the nternational ommittee of edical Journal Editors ( owever a full report of the outcomes must be made public no later than three (3) years after the end of data collection. l. The research study protocol must explicitly discuss subpopulations affected by the treatment under investigation, particularly traditionally underrepresented groups in clinical studies, how the inclusion and exclusion criteria effect enrollment of these populations, and a plan for the retention and reporting of said populations on the trial. f the inclusion and exclusion criteria are expected to have a negative effect on the recruitment or retention of underrepresented populations, the protocol must discuss why these criteria are necessary. m. The research study protocol explicitly discusses how the results are or are not expected to be generalizable to the edicare population to infer whether edicare patients may
11 benefit from the intervention. eparate discussions in the protocol may be necessary for populations eligible for edicare due to age, disability or edicaid eligibility. onsistent with section 1142 of the ocial ecurity ct, Q supports clinical research studies that determines meet the above-listed standards and address the above-listed research questions.. Nationally Non-overed ndications Effective for claims with dates of service on and after January 4, 2011, believes that the evidence does not demonstrate that the home use of oxygen to treat improves health outcomes in edicare beneficiaries with. Therefore, the home use of oxygen to treat is not reasonable and necessary under 1862(a)(1)() of the ct unless provided in the context of an approved clinical study under ED (see section B. above). D. Other This decision does not modify the existing requirements for coverage of the home use of oxygen currently identified in sections and of this manual. dditionally, the scope of the decision does not include any consideration of hyperbaric oxygen for any indication. (This ND last reviewed January 2011.)
Transmittal 2127 Date: DECEMBER 29, 2010
 anual ystem Pub 100-04 edicare laims Processing Department of ealth & uman ervices (D) enters for edicare & edicaid ervices () Transmittal 2127 Date: DEEBE 29, 2010 hange equest 7262 UBJET: edical Nutrition
anual ystem Pub 100-04 edicare laims Processing Department of ealth & uman ervices (D) enters for edicare & edicaid ervices () Transmittal 2127 Date: DEEBE 29, 2010 hange equest 7262 UBJET: edical Nutrition
Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 2212 Date: May 5, 2011
 anual ystem Pub 100-04 edicare laims Processing Department of ealth & uman ervices (D) enters for edicare & edicaid ervices () Transmittal 2212 Date: ay 5, 2011 hange equest 7128 Transmittal 2154, dated
anual ystem Pub 100-04 edicare laims Processing Department of ealth & uman ervices (D) enters for edicare & edicaid ervices () Transmittal 2212 Date: ay 5, 2011 hange equest 7128 Transmittal 2154, dated
CMS Manual System. Pub Medicare Claims Processing. Change Request 7580
 anual ystem Pub 100-04 edicare laims Processing Department of ealth & uman ervices (D) enters for edicare & edicaid ervices () Transmittal 2337 Date: October 28, 2011 hange equest 7580 UBJET: New nfluenza
anual ystem Pub 100-04 edicare laims Processing Department of ealth & uman ervices (D) enters for edicare & edicaid ervices () Transmittal 2337 Date: October 28, 2011 hange equest 7580 UBJET: New nfluenza
II. CHANGES IN MANUAL INSTRUCTIONS: (N/A if manual is not updated) R=REVISED, N=NEW, D=DELETED
 anual ystem Pub 100-04 edicare laims Processing Department of Health & Human ervices (DHH) enters for edicare & edicaid ervices () Transmittal 1888 Date: January 6, 2010 hange equest 6753 Transmittal 1879,
anual ystem Pub 100-04 edicare laims Processing Department of Health & Human ervices (DHH) enters for edicare & edicaid ervices () Transmittal 1888 Date: January 6, 2010 hange equest 6753 Transmittal 1879,
Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 1550 Date: July 18, 2008
 anual ystem Pub 100-04 edicare laims Processing Department of ealth & uman ervices (D) enters for edicare & edicaid ervices () Transmittal 1550 Date: July 18, 2008 hange equest 6061 UBJT: larifications
anual ystem Pub 100-04 edicare laims Processing Department of ealth & uman ervices (D) enters for edicare & edicaid ervices () Transmittal 1550 Date: July 18, 2008 hange equest 6061 UBJT: larifications
Transmittal 190 Date: February 5, SUBJECT: Screening for the Human Immunodeficiency Virus (HIV) Infection
 anual ystem Pub 100-03 edicare National overage Determinations Department of ealth & uman ervices (D) enters for edicare & edicaid ervices () Transmittal 190 Date: February 5, 2016 hange Request 9403 UBJET:
anual ystem Pub 100-03 edicare National overage Determinations Department of ealth & uman ervices (D) enters for edicare & edicaid ervices () Transmittal 190 Date: February 5, 2016 hange Request 9403 UBJET:
Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 2824 Date: November 22, 2013
 anual ystem Pub 100-04 edicare laims Processing Department of ealth & uman ervices (D) enters for edicare & edicaid ervices () Transmittal 2824 Date: November 22, 2013 hange equest 8473 UBJET: New nfluenza
anual ystem Pub 100-04 edicare laims Processing Department of ealth & uman ervices (D) enters for edicare & edicaid ervices () Transmittal 2824 Date: November 22, 2013 hange equest 8473 UBJET: New nfluenza
Transmittal 166 Date: April 18, 2014
 S anual System Pub 100-03 edicare National overage Determinations Department of Health & Human Services (DHHS) enters for edicare & edicaid Services (S) Transmittal 166 Date: April 18, 2014 hange Request
S anual System Pub 100-03 edicare National overage Determinations Department of Health & Human Services (DHHS) enters for edicare & edicaid Services (S) Transmittal 166 Date: April 18, 2014 hange Request
Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 2693 Date: May 2, 2013
 anual ystem Pub 100-04 edicare laims Processing Department of Health & Human ervices (DHH) enters for edicare & edicaid ervices () Transmittal 2693 Date: ay 2, 2013 hange equest 8249 NOTE: This Transmittal
anual ystem Pub 100-04 edicare laims Processing Department of Health & Human ervices (DHH) enters for edicare & edicaid ervices () Transmittal 2693 Date: ay 2, 2013 hange equest 8249 NOTE: This Transmittal
Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 2816 Date: November 15, 2013
 S anual System Pub 100-04 edicare laims Processing Department of ealth & uman Services (DS) enters for edicare & edicaid Services (S) Transmittal 2816 Date: November 15, 2013 hange equest 8484 SUBJET:
S anual System Pub 100-04 edicare laims Processing Department of ealth & uman Services (DS) enters for edicare & edicaid Services (S) Transmittal 2816 Date: November 15, 2013 hange equest 8484 SUBJET:
Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 262 Date: January 29, 2016
 CS anual System Pub 100-06 edicare Financial anagement Department of Health & Human Services (DHHS) Centers for edicare & edicaid Services (CS) Transmittal 262 Date: January 29, 2016 Change Request 9355
CS anual System Pub 100-06 edicare Financial anagement Department of Health & Human Services (DHHS) Centers for edicare & edicaid Services (CS) Transmittal 262 Date: January 29, 2016 Change Request 9355
Pharmacogenomic Testing for Warfarin Response (NCD 90.1)
 Policy Number 90.1 Approved By UnitedHealthcare Medicare Reimbursement Policy Committee Current Approval Date 01/08/2014 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to UnitedHealthcare
Policy Number 90.1 Approved By UnitedHealthcare Medicare Reimbursement Policy Committee Current Approval Date 01/08/2014 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable to UnitedHealthcare
CMS Issues New Draft Guidance on Coverage with Evidence Development Policy for National Coverage Determinations
 December 6, 2012 CMS Issues New Draft Guidance on Coverage with Evidence Development Policy for National Coverage Determinations For more information, contact: Seth H. Lundy +1 202 626 2924 slundy@kslaw.com
December 6, 2012 CMS Issues New Draft Guidance on Coverage with Evidence Development Policy for National Coverage Determinations For more information, contact: Seth H. Lundy +1 202 626 2924 slundy@kslaw.com
CMS Manual System Pub Medicare National Coverage Determinations
 CMS Manual System Pub. 100-03 Medicare National Coverage Determinations Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 9 Date: APRIL 1, 2004 CHANGE
CMS Manual System Pub. 100-03 Medicare National Coverage Determinations Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 9 Date: APRIL 1, 2004 CHANGE
Centers for Medicare and Medicaid Services. National Coverage of Transcatheter Valve Technologies December 2015
 Centers for Medicare and Medicaid Services National Coverage of Transcatheter Valve Technologies December 2015 National Coverage Determination (NCD) for Transcatheter Aortic Valve Replacement (TAVR) (20.32)
Centers for Medicare and Medicaid Services National Coverage of Transcatheter Valve Technologies December 2015 National Coverage Determination (NCD) for Transcatheter Aortic Valve Replacement (TAVR) (20.32)
Oxygen and Oxygen Equipment
 Oxygen and Oxygen Equipment Policy Number: Original Effective Date: MM.01.008 12/01/2010 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 08/25/2017 Section: DME Place(s) of Service:
Oxygen and Oxygen Equipment Policy Number: Original Effective Date: MM.01.008 12/01/2010 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 08/25/2017 Section: DME Place(s) of Service:
Headache Master School Japan-Osaka 2016 II. Management of refractory headaches Case Presentation 2. SUNCT/SUNA: concept, management and prognosis
 Headache Master School Japan-Osaka 2016 II. Management of refractory headaches Case Presentation 2. SUNCT/SUNA: concept, management and prognosis Noboru Imai(Department of Neurology, Japanese Red Cross
Headache Master School Japan-Osaka 2016 II. Management of refractory headaches Case Presentation 2. SUNCT/SUNA: concept, management and prognosis Noboru Imai(Department of Neurology, Japanese Red Cross
CHAPTER 7 SECTION 24.1 PHASE I, PHASE II, AND PHASE III CANCER CLINICAL TRIALS TRICARE POLICY MANUAL M, AUGUST 1, 2002 MEDICINE
 MEDICINE CHAPTER 7 SECTION 24.1 ISSUE DATE: AUTHORITY: 32 CFR 199.4(e)(26) I. DESCRIPTION The Department of Defense (DoD) Cancer Prevention and Treatment Clinical Trials Demonstration was conducted from
MEDICINE CHAPTER 7 SECTION 24.1 ISSUE DATE: AUTHORITY: 32 CFR 199.4(e)(26) I. DESCRIPTION The Department of Defense (DoD) Cancer Prevention and Treatment Clinical Trials Demonstration was conducted from
Oxygen and Oxygen Equipment
 Oxygen and Oxygen Equipment Policy Number: Original Effective Date: MM.01.008 12/01/2010 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST 09/01/2013 Section: DME Place(s) of Service: Home I.
Oxygen and Oxygen Equipment Policy Number: Original Effective Date: MM.01.008 12/01/2010 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST 09/01/2013 Section: DME Place(s) of Service: Home I.
Chapter 18 Section 2. EXPIRED - Department Of Defense (DoD) Cancer Prevention And Treatment Clinical Trials Demonstration
 s And Pilot Projects Chapter 18 Section 2 EXPIRED - Department Of Defense (DoD) Cancer Prevention And Treatment Clinical Trials 1.0 PURPOSE The purpose of this demonstration is to improve TRICARE-eligible
s And Pilot Projects Chapter 18 Section 2 EXPIRED - Department Of Defense (DoD) Cancer Prevention And Treatment Clinical Trials 1.0 PURPOSE The purpose of this demonstration is to improve TRICARE-eligible
Inspire Medical Systems. Physician Billing Guide
 Inspire Medical Systems Physician Billing Guide 2019 Inspire Medical Systems Physician Billing Guide This Physician Billing Guide was developed to help providers correctly bill for Inspire Upper Airway
Inspire Medical Systems Physician Billing Guide 2019 Inspire Medical Systems Physician Billing Guide This Physician Billing Guide was developed to help providers correctly bill for Inspire Upper Airway
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: occipital_nerve_stimulation 8/2010 5/2017 5/2018 5/2017 Description of Procedure or Service Occipital nerve
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: occipital_nerve_stimulation 8/2010 5/2017 5/2018 5/2017 Description of Procedure or Service Occipital nerve
Contractor Name: Novitas Solutions, Inc. Contractor Number: Contractor Type: MAC B. LCD ID Number: L34834 Status: A-Approved
 LCD for Blood Glucose Monitoring in a Skilled Nursing Facility (SNF) (L34834) Contractor Name: Novitas Solutions, Inc. Contractor Number: 12502 Contractor Type: MAC B LCD ID Number: L34834 Status: A-Approved
LCD for Blood Glucose Monitoring in a Skilled Nursing Facility (SNF) (L34834) Contractor Name: Novitas Solutions, Inc. Contractor Number: 12502 Contractor Type: MAC B LCD ID Number: L34834 Status: A-Approved
Trigeminal Autonomic Cephalalgias. Disclosures. Objectives 6/20/2018. Rashmi Halker Singh, MD FAHS UCNS Review Course June 2018
 Trigeminal Autonomic Cephalalgias Rashmi Halker Singh, MD FAHS UCNS Review Course June 2018 Disclosures Honoraria from Allergan and Amgen for advisory board, Current Neurology and Neuroscience Reports
Trigeminal Autonomic Cephalalgias Rashmi Halker Singh, MD FAHS UCNS Review Course June 2018 Disclosures Honoraria from Allergan and Amgen for advisory board, Current Neurology and Neuroscience Reports
No An act relating to health insurance coverage for early childhood developmental disorders, including autism spectrum disorders. (S.
 No. 158. An act relating to health insurance coverage for early childhood developmental disorders, including autism spectrum disorders. (S.223) It is hereby enacted by the General Assembly of the State
No. 158. An act relating to health insurance coverage for early childhood developmental disorders, including autism spectrum disorders. (S.223) It is hereby enacted by the General Assembly of the State
HDS PROCEDURE CODE GUIDELINES INTRODUCTION
 The HDS Procedure Code Guidelines (PCG) provides a framework of rules and policies for benefit determination. Please note that specific group contract provisions, limitations, and exclusions take precedence
The HDS Procedure Code Guidelines (PCG) provides a framework of rules and policies for benefit determination. Please note that specific group contract provisions, limitations, and exclusions take precedence
Chronic Daily Headaches
 Chronic Daily Headaches ANWARUL HAQ, MD, MRCP(UK), FAHS DIRECTOR BAYLOR HEADACHE CENTER, DALLAS, TEXAS DISCLOSURES: None OBJECTIVES AT THE CONCLUSION OF THIS ACTIVITY, PARTICIPANTS WILL BE ABLE TO: define
Chronic Daily Headaches ANWARUL HAQ, MD, MRCP(UK), FAHS DIRECTOR BAYLOR HEADACHE CENTER, DALLAS, TEXAS DISCLOSURES: None OBJECTIVES AT THE CONCLUSION OF THIS ACTIVITY, PARTICIPANTS WILL BE ABLE TO: define
Disclosures. Objectives 6/2/2017
 Classification: Migraine and Trigeminal Autonomic Cephalalgias Lauren Doyle Strauss, DO, FAHS Assistant Professor, Child Neurology Assistant Director, Child Neurology Residency @StraussHeadache No disclosures
Classification: Migraine and Trigeminal Autonomic Cephalalgias Lauren Doyle Strauss, DO, FAHS Assistant Professor, Child Neurology Assistant Director, Child Neurology Residency @StraussHeadache No disclosures
DEPARTMENT OF HEALTH AND HUMAN SERVICES Centers for Medicare & Medicaid Services
 DEPARTMENT OF HEALTH AND HUMAN SERVICES Centers for Medicare & Medicaid Services News Flash Looking for the latest Medicare Fee-For-Service (FFS) information? Then subscribe to a Medicare FFS Provider
DEPARTMENT OF HEALTH AND HUMAN SERVICES Centers for Medicare & Medicaid Services News Flash Looking for the latest Medicare Fee-For-Service (FFS) information? Then subscribe to a Medicare FFS Provider
Oxygen and Oxygen Equipment
 Oxygen and Oxygen Equipment I. Policy University Health Alliance (UHA) will reimburse for home oxygen and oxygen equipment when it is determined to be medically necessary and when it meets the medical
Oxygen and Oxygen Equipment I. Policy University Health Alliance (UHA) will reimburse for home oxygen and oxygen equipment when it is determined to be medically necessary and when it meets the medical
Home Sleep Test (HST) Instructions
 Home Sleep Test (HST) Instructions 1. Your physician has ordered an unattended home sleep test (HST) to diagnose or rule out sleep apnea. This test cannot diagnose any other sleep disorders. 2. This device
Home Sleep Test (HST) Instructions 1. Your physician has ordered an unattended home sleep test (HST) to diagnose or rule out sleep apnea. This test cannot diagnose any other sleep disorders. 2. This device
Positive Airway Pressure (PAP) Devices Physician Frequently Asked Questions December 2008
 Positive Airway Pressure (PAP) Devices Physician Frequently Asked Questions December 2008 Based on questions received from the clinical community, the following Frequently Asked Questions will address
Positive Airway Pressure (PAP) Devices Physician Frequently Asked Questions December 2008 Based on questions received from the clinical community, the following Frequently Asked Questions will address
Disease Description and Statistics Current State: Medicare Denial of Coverage Expert Opinion Clinical Studies Gap in Medical Care Cost to US
 Oxygen Therapy Disease Description and Statistics Current State: Medicare Denial of Coverage Expert Opinion Clinical Studies Gap in Medical Care Cost to US Taxpayers Recommendations Cluster headaches are
Oxygen Therapy Disease Description and Statistics Current State: Medicare Denial of Coverage Expert Opinion Clinical Studies Gap in Medical Care Cost to US Taxpayers Recommendations Cluster headaches are
Pulmonary Rehabilitation
 Pulmonary Rehabilitation New Benefit The enactment of HR 6331, the Medicare Improvements for Patients and Providers Act of 2008, established a specific Medicare benefit for pulmonary rehabilitation. The
Pulmonary Rehabilitation New Benefit The enactment of HR 6331, the Medicare Improvements for Patients and Providers Act of 2008, established a specific Medicare benefit for pulmonary rehabilitation. The
RESPIRATORY ASSIST DEVICE E0471
 JURISDICTIONS B &C Bi-Level Pressure Capacity WITH Backup Rate REQUIRED DOCUMENTATION All Claims for E0471 Initial Coverage (1st Three Months) 5 Element Order (5EO) obtained prior to delivery for the E0470
JURISDICTIONS B &C Bi-Level Pressure Capacity WITH Backup Rate REQUIRED DOCUMENTATION All Claims for E0471 Initial Coverage (1st Three Months) 5 Element Order (5EO) obtained prior to delivery for the E0470
Z E N I T H M E D I C A L P R O V I D E R N E T W O R K P O L I C Y Title: Provider Appeal of Network Exclusion Policy
 TheZenith's Z E N I T H M E D I C A L P R O V I D E R N E T W O R K P O L I C Y Title: Provider Appeal of Network Exclusion Policy Application: Zenith Insurance Company and Wholly Owned Subsidiaries Policy
TheZenith's Z E N I T H M E D I C A L P R O V I D E R N E T W O R K P O L I C Y Title: Provider Appeal of Network Exclusion Policy Application: Zenith Insurance Company and Wholly Owned Subsidiaries Policy
ProviderNews FEBRUARY
 ProviderNews FEBRUARY 2017 Reminder: decimal billing required on time-based therapy codes for BadgerCare Plus members In accordance with Forward Health guidelines, Security Health Plan requires decimal
ProviderNews FEBRUARY 2017 Reminder: decimal billing required on time-based therapy codes for BadgerCare Plus members In accordance with Forward Health guidelines, Security Health Plan requires decimal
Clinical Policy: Oxygen Therapy in the Home Reference Number: CP.MP.485
 Clinical Policy: Reference Number: CP.MP.485 Effective Date: 09/04 Last Review Date: 09/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.485 Effective Date: 09/04 Last Review Date: 09/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Outpatient Headache Care Guideline
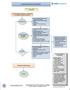 1 Outpatient Care Guideline Inclusion criteria: children > 3 yrs with headaches Is urgent emergency department, neuroimaging, or Neurology consultation indicated? Referral to ED if: New severe headache
1 Outpatient Care Guideline Inclusion criteria: children > 3 yrs with headaches Is urgent emergency department, neuroimaging, or Neurology consultation indicated? Referral to ED if: New severe headache
Tears of Pain SUNCT and SUNA A/PROFESSOR ARUN AGGARWAL RPAH PAIN MANAGEMENT CENTRE
 Tears of Pain SUNCT and SUNA A/PROFESSOR ARUN AGGARWAL RPAH PAIN MANAGEMENT CENTRE IHS Classification 1989 (updated 2004) Primary Headaches 4 categories Migraine Tension-type Cluster and other trigeminal
Tears of Pain SUNCT and SUNA A/PROFESSOR ARUN AGGARWAL RPAH PAIN MANAGEMENT CENTRE IHS Classification 1989 (updated 2004) Primary Headaches 4 categories Migraine Tension-type Cluster and other trigeminal
NEW YORK STATE MEDICAID PROGRAM CHIROPRACTOR MANUAL
 NEW YORK STATE MEDICAID PROGRAM CHIROPRACTOR MANUAL POLICY GUIDELINES Table of Contents SECTION I - REQUIREMENTS FOR PARTICIPATION IN MEDICAID...2 WHO MAY PROVIDE CARE...2 LIMITED CHIROPRACTIC SERVICE
NEW YORK STATE MEDICAID PROGRAM CHIROPRACTOR MANUAL POLICY GUIDELINES Table of Contents SECTION I - REQUIREMENTS FOR PARTICIPATION IN MEDICAID...2 WHO MAY PROVIDE CARE...2 LIMITED CHIROPRACTIC SERVICE
The Third-Party Reimbursement Process for Orthotics
 The Third-Party Reimbursement Process for Orthotics When the foot hits the ground, everything changes. We know that over 90% of the population suffers with overpronation of their feet. Implementing Foot
The Third-Party Reimbursement Process for Orthotics When the foot hits the ground, everything changes. We know that over 90% of the population suffers with overpronation of their feet. Implementing Foot
RESPIRATORY ASSIST DEVICE E0470
 JURISDICTIONS B &C Bi-Level Pressure Capacity WITHOUT Backup Rate REQUIRED DOCUMENTATION All Claims for E0470 Initial Coverage (1st Three Months) 5 Element Order (5EO) obtained prior to delivery for the
JURISDICTIONS B &C Bi-Level Pressure Capacity WITHOUT Backup Rate REQUIRED DOCUMENTATION All Claims for E0470 Initial Coverage (1st Three Months) 5 Element Order (5EO) obtained prior to delivery for the
Respiratory Assist Device E0470:
 Respiratory Assist Device E0470: Bi-Level Pressure Capacity WITHOUT Backup Rate REQUIRED DOCUMENTATION IN SUPPLIER S FILE All Claims for E0470 Initial Coverage (1st Three Months) 5 Element Order obtained
Respiratory Assist Device E0470: Bi-Level Pressure Capacity WITHOUT Backup Rate REQUIRED DOCUMENTATION IN SUPPLIER S FILE All Claims for E0470 Initial Coverage (1st Three Months) 5 Element Order obtained
Professional CGM Reimbursement Guide
 Professional CGM Reimbursement Guide 2015 TABLE OF CONTENTS Coding, Coverage and Payment...2 Coding and Billing...2 CPT Code 95250...3 CPT Code 95251...3 Incident to Billing for Physicians..............................................
Professional CGM Reimbursement Guide 2015 TABLE OF CONTENTS Coding, Coverage and Payment...2 Coding and Billing...2 CPT Code 95250...3 CPT Code 95251...3 Incident to Billing for Physicians..............................................
RESPIRATORY ASSIST DEVICE E0471
 JURISDICTIONS B &C Bi-Level Pressure Capacity WITH Backup Rate REQUIRED DOCUMENTATION All Claims for E0471 Initial Coverage (1st Three Months) 5 Element Order (5EO) obtained prior to delivery for the E0470
JURISDICTIONS B &C Bi-Level Pressure Capacity WITH Backup Rate REQUIRED DOCUMENTATION All Claims for E0471 Initial Coverage (1st Three Months) 5 Element Order (5EO) obtained prior to delivery for the E0470
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Aimovig) Reference Number: CP.PHAR.128 Effective Date: 07.10.18 Last Review Date: 08.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Aimovig) Reference Number: CP.PHAR.128 Effective Date: 07.10.18 Last Review Date: 08.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Managed Health Services (MHS) Candace V. Ervin Market Manager, Indiana Provider Relations October 18, 2017
 Managed Health Services (MHS) Candace V. Ervin Market Manager, Indiana Provider Relations Candace.Ervin@Envolvehealth.com October 18, 2017 1 Today s Agenda MHS ID Card Samples Provider Visits D1110 (Prophylaxis
Managed Health Services (MHS) Candace V. Ervin Market Manager, Indiana Provider Relations Candace.Ervin@Envolvehealth.com October 18, 2017 1 Today s Agenda MHS ID Card Samples Provider Visits D1110 (Prophylaxis
DECISION AND ORDER. After due notice, a telephone hearing was held on. , Medical Director, also testified as a witness for the MHP.
 STATE OF MICHIGAN MICHIGAN ADMINISTRATIVE HEARING SYSTEM FOR THE DEPARTMENT OF HEALTH AND HUMAN SERVICES P.O. Box 30763, Lansing, MI 48909 (517) 373-0722; Fax: (517) 373-4147 IN THE MATTER OF:, MAHS Docket
STATE OF MICHIGAN MICHIGAN ADMINISTRATIVE HEARING SYSTEM FOR THE DEPARTMENT OF HEALTH AND HUMAN SERVICES P.O. Box 30763, Lansing, MI 48909 (517) 373-0722; Fax: (517) 373-4147 IN THE MATTER OF:, MAHS Docket
DELTA DENTAL PREMIER
 DELTA DENTAL PREMIER PARTICIPATING DENTIST AGREEMENT THIS AGREEMENT made and entered into this day of, 20 by and between Colorado Dental Service, Inc. d/b/a Delta Dental of Colorado, as first party, hereinafter
DELTA DENTAL PREMIER PARTICIPATING DENTIST AGREEMENT THIS AGREEMENT made and entered into this day of, 20 by and between Colorado Dental Service, Inc. d/b/a Delta Dental of Colorado, as first party, hereinafter
CMS CLARIFICATION JIMMO VS. SEBELIUS
 CMS CLARIFICATION JIMMO VS. SEBELIUS Liz Almeida-Sanborn, MS, PT, President Maria Maggi, PT, Vice President of Compliance Jodi Wenzel, MPT, Vice President of Operations OBJECTIVES Participants will: Understand
CMS CLARIFICATION JIMMO VS. SEBELIUS Liz Almeida-Sanborn, MS, PT, President Maria Maggi, PT, Vice President of Compliance Jodi Wenzel, MPT, Vice President of Operations OBJECTIVES Participants will: Understand
Corporate Policies. Corporate Billing and Collection Policy Section:
 MedStar Health Title: Purpose: Corporate Policies Corporate Billing and Collection Policy Section: To ensure uniform management of the MedStar Health Corporate Billing and Collection Program for all MedStar
MedStar Health Title: Purpose: Corporate Policies Corporate Billing and Collection Policy Section: To ensure uniform management of the MedStar Health Corporate Billing and Collection Program for all MedStar
Coventry Health Care of Georgia, Inc.
 Coventry Health Care of Georgia, Inc. PRESCRIPTION DRUG RIDER (for High Deductible Health Plans) This Prescription Drug Rider is an attachment to the Coventry Health Care of Georgia, Inc. ( Health Plan
Coventry Health Care of Georgia, Inc. PRESCRIPTION DRUG RIDER (for High Deductible Health Plans) This Prescription Drug Rider is an attachment to the Coventry Health Care of Georgia, Inc. ( Health Plan
A A ~l~js AM f'ricj\n ACADBl\IY OF 0RTllOPAEDIC SURGEONS ~ J AMERICAN A SOCIATION OF ORTHOPAEDIC SURGEONS. Therapy billing for beginners
 Therapy billing for beginners http://www.aaos.org/news/aaosnow/nov10/managing1.asp 1 of 4 3/25/2014 2:56 PM A A ~l~js AM f'ricj\n ACADBl\IY OF 0RTllOPAEDIC SURGEONS ~ J AMERICAN A SOCIATION OF ORTHOPAEDIC
Therapy billing for beginners http://www.aaos.org/news/aaosnow/nov10/managing1.asp 1 of 4 3/25/2014 2:56 PM A A ~l~js AM f'ricj\n ACADBl\IY OF 0RTllOPAEDIC SURGEONS ~ J AMERICAN A SOCIATION OF ORTHOPAEDIC
EXCERPTED FROM REHABILITATION ASSOCIATION OF VIRGINIA, INC. V. KOZLOWSKI, ET AL., 42 F. 3D 1444,1450 (4 TH CIR. 1994)
 There can be no doubt but that the statutes and provisions in question, involving the financing of Medicare, are among the most completely impenetrable texts within human experience... one approaches [them]
There can be no doubt but that the statutes and provisions in question, involving the financing of Medicare, are among the most completely impenetrable texts within human experience... one approaches [them]
Authorized By: Elizabeth Connolly, Acting Commissioner, Department of Human
 HUMAN SERVICES 49 NJR 6(1) June 5, 2017 Filed May 10, 2017 DIVISION OF MENTAL HEALTH AND ADDICTION SERVICES Medical Necessity Review Tool for Substance Use Disorders Proposed New Rules: N.J.A.C. 10:163
HUMAN SERVICES 49 NJR 6(1) June 5, 2017 Filed May 10, 2017 DIVISION OF MENTAL HEALTH AND ADDICTION SERVICES Medical Necessity Review Tool for Substance Use Disorders Proposed New Rules: N.J.A.C. 10:163
WATCHMAN. For questions regarding WATCHMAN reimbursement, please contact:
 WATCHMAN IMPORTANCE OF DOCUMENTATION & THE IMPACT ON MS- DRG ASSIGNMENT This guide stresses the importance of documentation in capturing the appropriate acuity level for patients considered WATCHMAN candidates.
WATCHMAN IMPORTANCE OF DOCUMENTATION & THE IMPACT ON MS- DRG ASSIGNMENT This guide stresses the importance of documentation in capturing the appropriate acuity level for patients considered WATCHMAN candidates.
Outpatient Therapy Functional Reporting Requirements. Provider Types Affected
 DEPARTMENT OF HEALTH AND HUMAN SERVICES Centers for Medicare & Medicaid Services The Centers for Medicare & Medicaid Services (CMS) is launching a new instrument for 2013 called the MAC Satisfaction Indicator
DEPARTMENT OF HEALTH AND HUMAN SERVICES Centers for Medicare & Medicaid Services The Centers for Medicare & Medicaid Services (CMS) is launching a new instrument for 2013 called the MAC Satisfaction Indicator
Glucose Monitors Policy Pearls
 Glucose Monitors Policy Pearls Length: 20:31 Date Recorded: 1.1.17 Hello and welcome to Medicare Minute MD, a video and podcast series produced by the DME MACs for the benefit of physicians and healthcare
Glucose Monitors Policy Pearls Length: 20:31 Date Recorded: 1.1.17 Hello and welcome to Medicare Minute MD, a video and podcast series produced by the DME MACs for the benefit of physicians and healthcare
Electrical Stimulation Device Used for Cancer Treatment
 Electrical Stimulation Device Used for Cancer Treatment OPTUNE (NOVOTTF 100A SYSTEM) For any item to be covered by The Health Plan, it must: 1. Be eligible for a defined Medicare or The Health Plan benefit
Electrical Stimulation Device Used for Cancer Treatment OPTUNE (NOVOTTF 100A SYSTEM) For any item to be covered by The Health Plan, it must: 1. Be eligible for a defined Medicare or The Health Plan benefit
Medicare Myths-Busters: Dispelling Common Compliance Misconceptions. Learner Objectives. Learner Objectives
 Medicare Myths-Busters: Dispelling Common Compliance Misconceptions Rick Gawenda, PT President Gawenda Seminars & Consulting, Inc. September 30, 2017 Learner Objectives Identify the supervision requirements
Medicare Myths-Busters: Dispelling Common Compliance Misconceptions Rick Gawenda, PT President Gawenda Seminars & Consulting, Inc. September 30, 2017 Learner Objectives Identify the supervision requirements
MICHIGAN OFFICE OF SERVICES TO THE AGING. Operating Standards For Service Programs
 Community SERVICE NAME Congregate Meals SERVICE NUMBER C-3 SERVICE CATEGORY SERVICE DEFINITION UNIT OF SERVICE Community The provision of nutritious meals to older individuals in congregate settings. Each
Community SERVICE NAME Congregate Meals SERVICE NUMBER C-3 SERVICE CATEGORY SERVICE DEFINITION UNIT OF SERVICE Community The provision of nutritious meals to older individuals in congregate settings. Each
Physician s Compliance Guide
 Physician s Compliance Guide Updates to this guide will be posted on the Optum website and can be found at: http://www.optumcoding.com/product/updates/2013pcg/pcg13 Please use the following password to
Physician s Compliance Guide Updates to this guide will be posted on the Optum website and can be found at: http://www.optumcoding.com/product/updates/2013pcg/pcg13 Please use the following password to
Exhibit 2 RFQ Engagement Letter
 Exhibit 2 RFQ 17-25 Engagement Letter The attached includes the 6 page proposed engagement letter to be used by HCC. ENGAGEMENT LETTER Dear: [Lead Counsel/Partner] We are pleased to inform you that your
Exhibit 2 RFQ 17-25 Engagement Letter The attached includes the 6 page proposed engagement letter to be used by HCC. ENGAGEMENT LETTER Dear: [Lead Counsel/Partner] We are pleased to inform you that your
Pulmonary Rehabilitation. Palmetto GBA, Jurisdiction 11 MAC Provider Outreach and Education
 Pulmonary Rehabilitation Palmetto GBA, Jurisdiction 11 MAC Provider Outreach and Education Pulmonary Rehabilitation Pulmonary Rehabilitation is a multi-disciplinary program of care for patients with chronic
Pulmonary Rehabilitation Palmetto GBA, Jurisdiction 11 MAC Provider Outreach and Education Pulmonary Rehabilitation Pulmonary Rehabilitation is a multi-disciplinary program of care for patients with chronic
SENATE BILL No. 501 AMENDED IN SENATE MAY 1, 2017 AMENDED IN SENATE APRIL 20, 2017 AMENDED IN SENATE APRIL 17, Introduced by Senator Glazer
 AMENDED IN SENATE MAY 1, 2017 AMENDED IN SENATE APRIL 20, 2017 AMENDED IN SENATE APRIL 17, 2017 SENATE BILL No. 501 Introduced by Senator Glazer February 16, 2017 An act to amend Sections 1601.4, 1646,
AMENDED IN SENATE MAY 1, 2017 AMENDED IN SENATE APRIL 20, 2017 AMENDED IN SENATE APRIL 17, 2017 SENATE BILL No. 501 Introduced by Senator Glazer February 16, 2017 An act to amend Sections 1601.4, 1646,
Florida Medicaid. Dental Services Coverage Policy. Agency for Health Care Administration
 Florida Medicaid Agency for Health Care Administration Table of Contents 1.0 Introduction... 1 1.1 Description... 1 1.2 Legal Authority... 1 1.3 Definitions... 1 2.0 Eligible Recipient... 2 2.1 General
Florida Medicaid Agency for Health Care Administration Table of Contents 1.0 Introduction... 1 1.1 Description... 1 1.2 Legal Authority... 1 1.3 Definitions... 1 2.0 Eligible Recipient... 2 2.1 General
tation DEVELOPMENTAL PROGRAMS BULLETIN COMMONWEALTH OF PENNSYLVANIA DEPARTMENT OF PUBLIC WELFARE EFFECTIVE DATE: April 8, 2014 BY:
 tation DEVELOPMENTAL PROGRAMS BULLETIN COMMONWEALTH OF PENNSYLVANIA DEPARTMENT OF PUBLIC WELFARE DATE OF ISSUE: April 8, 2014 EFFECTIVE DATE: April 8, 2014 NUMBER 00-14-04 SUBJECT: Accessibility of Intellectual
tation DEVELOPMENTAL PROGRAMS BULLETIN COMMONWEALTH OF PENNSYLVANIA DEPARTMENT OF PUBLIC WELFARE DATE OF ISSUE: April 8, 2014 EFFECTIVE DATE: April 8, 2014 NUMBER 00-14-04 SUBJECT: Accessibility of Intellectual
KANSAS MEDICAL ASSISTANCE PROGRAM. Fee-for-Service Provider Manual. Rehabilitative Therapy Services
 Fee-for-Service Provider Manual Rehabilitative Therapy Services Updated 12.2015 PART II (PHYSICAL THERAPY, OCCUPATIONAL THERAPY, SPEECH/LANGUAGE PATHOLOGY) Introduction Section BILLING INSTRUCTIONS Page
Fee-for-Service Provider Manual Rehabilitative Therapy Services Updated 12.2015 PART II (PHYSICAL THERAPY, OCCUPATIONAL THERAPY, SPEECH/LANGUAGE PATHOLOGY) Introduction Section BILLING INSTRUCTIONS Page
GENERAL TERMS AND CONDITIONS WCO PUBLICATIONS
 Version 1.0 GENERAL TERMS AND CONDITIONS WCO PUBLICATIONS These General Terms and Conditions govern all sales of Publications, on paper and/or in digital format whether directly at the Sales Counter or
Version 1.0 GENERAL TERMS AND CONDITIONS WCO PUBLICATIONS These General Terms and Conditions govern all sales of Publications, on paper and/or in digital format whether directly at the Sales Counter or
Medical Necessity Guidelines: Applied Behavioral Analysis (ABA) including Early Intervention for RITogether
 Medical Necessity Guidelines: Applied Behavioral Analysis (ABA) including Effective: August 1, 2017 Clinical Documentation and Prior Authorization Required Applies to: Coverage Guideline, No prior Authorization
Medical Necessity Guidelines: Applied Behavioral Analysis (ABA) including Effective: August 1, 2017 Clinical Documentation and Prior Authorization Required Applies to: Coverage Guideline, No prior Authorization
MedStar Health, Inc. POLICY AND PROCEDURE MANUAL Policy Number: PA.010.MH Last Review Date: 05/11/2017 Effective Date: 07/01/2017
 MedStar Health, Inc. POLICY AND PROCEDURE MANUAL Last Review Date: 05/11/2017 Effective Date: 07/01/2017 PA.010.MH Durable Medical Equipment, Corrective Appliances and This policy applies to the following
MedStar Health, Inc. POLICY AND PROCEDURE MANUAL Last Review Date: 05/11/2017 Effective Date: 07/01/2017 PA.010.MH Durable Medical Equipment, Corrective Appliances and This policy applies to the following
Coverage of Orthodontic Specific Examination
 Medical Strategy & Development Disclaimer: By accessing Daman Adjudication Rules, you acknowledge that you have read and understood the terms of use set out in the disclaimer below: Daman Adjudication
Medical Strategy & Development Disclaimer: By accessing Daman Adjudication Rules, you acknowledge that you have read and understood the terms of use set out in the disclaimer below: Daman Adjudication
32 CFR (a)(4), (a)(6)(iii), and (a)(6)(iv)
 CHAPTER 15 SECTION 1 ISSUE DATE: November 6, 2007 AUTHORITY: 32 CFR 199.14(a)(4), (a)(6)(iii), and (a)(6)(iv) I. APPLICABILITY This policy is mandatory for the reimbursement of services provided either
CHAPTER 15 SECTION 1 ISSUE DATE: November 6, 2007 AUTHORITY: 32 CFR 199.14(a)(4), (a)(6)(iii), and (a)(6)(iv) I. APPLICABILITY This policy is mandatory for the reimbursement of services provided either
Medical Policy Original Effective Date: 01/23/2019
 Page 1 of 12 Disclaimer Refer to the member s specific benefit plan and Schedule of Benefits to determine coverage. This may not be a benefit on all plans or the plan may have broader or more limited benefits
Page 1 of 12 Disclaimer Refer to the member s specific benefit plan and Schedule of Benefits to determine coverage. This may not be a benefit on all plans or the plan may have broader or more limited benefits
Diabetes Management, Equipment and Supplies
 Coverage Summary Diabetes Management, Equipment and Supplies Policy Number: D-001 Products: UnitedHealthcare Medicare Advantage Plans Original Approval Date: 11/01/2006 Approved by: UnitedHeatlhcare Medicare
Coverage Summary Diabetes Management, Equipment and Supplies Policy Number: D-001 Products: UnitedHealthcare Medicare Advantage Plans Original Approval Date: 11/01/2006 Approved by: UnitedHeatlhcare Medicare
GILMER COUNTY SCHOOLS Policy No POLICY MANUAL
 GILMER COUNTY SCHOOLS Policy No. 4080 POLICY MANUAL TITLE: DRUG FREE WORKPLACE POLICY 1. General Policy. The Board believes it is imperative to establish, promote and maintain a safe, healthy working and
GILMER COUNTY SCHOOLS Policy No. 4080 POLICY MANUAL TITLE: DRUG FREE WORKPLACE POLICY 1. General Policy. The Board believes it is imperative to establish, promote and maintain a safe, healthy working and
MEDICAID PRIOR AUTHORIZATION TRANSITION
 MEDICAID PRIOR AUTHORIZATION TRANSITION Prepared for: Mississippi Medicaid Hearing Providers November 2013 December 1, 2013 The Road Ahead 12/8/2013 HEARING PROVIDER PRESENTATION 2 Today s Goals and Objectives
MEDICAID PRIOR AUTHORIZATION TRANSITION Prepared for: Mississippi Medicaid Hearing Providers November 2013 December 1, 2013 The Road Ahead 12/8/2013 HEARING PROVIDER PRESENTATION 2 Today s Goals and Objectives
Common Diagnosis Codes and Tips for Coding Nicotine Use/
 ICD-10 Tobacco Billing Guide ICD-10 and Tobacco Common Diagnosis Codes and Tips for Coding Nicotine Use/ Dependence Tobacco use is the leading cause of preventable disease and death in the United States,
ICD-10 Tobacco Billing Guide ICD-10 and Tobacco Common Diagnosis Codes and Tips for Coding Nicotine Use/ Dependence Tobacco use is the leading cause of preventable disease and death in the United States,
Oxygen Therapy Coverage Guidelines for Non-Medicare Advantage Members
 Origination: 05/06/04 Revised: 08/02/17 Annual Review: 11/02/17 Purpose: To provide oxygen therapy guidelines for non-medicare Advantage Members for the Medical Department staff to reference when making
Origination: 05/06/04 Revised: 08/02/17 Annual Review: 11/02/17 Purpose: To provide oxygen therapy guidelines for non-medicare Advantage Members for the Medical Department staff to reference when making
OREGON MEDICAL MARIJUANA ACT
 OREGON MEDICAL MARIJUANA ACT 475.300 Findings. The people of the state of Oregon hereby find that: (1) Patients and doctors have found marijuana to be an effective treatment for suffering caused by debilitating
OREGON MEDICAL MARIJUANA ACT 475.300 Findings. The people of the state of Oregon hereby find that: (1) Patients and doctors have found marijuana to be an effective treatment for suffering caused by debilitating
Medicare Podiatry Services: Information for Medicare Fee-For-Service Health Care Professionals
 R DEPARTMENT OF HEALTH AND HUMAN SERVICES Centers for Medicare & Medicaid Services FACT SHEET Medicare Podiatry Services: Information for Medicare Fee-For-Service Health Care Professionals Overview This
R DEPARTMENT OF HEALTH AND HUMAN SERVICES Centers for Medicare & Medicaid Services FACT SHEET Medicare Podiatry Services: Information for Medicare Fee-For-Service Health Care Professionals Overview This
Certification in Lower Extremity Geriatric Medicine Handbook
 Certification in Lower Extremity Geriatric Medicine Handbook 555 8 th Ave, Ste 1902, New York, NY 10018 888 852 1442 1 Mission Statement We exist to protect and improve the podiatric health and welfare
Certification in Lower Extremity Geriatric Medicine Handbook 555 8 th Ave, Ste 1902, New York, NY 10018 888 852 1442 1 Mission Statement We exist to protect and improve the podiatric health and welfare
and at the same patient encounter. Code has been deleted. For scanning computerized ophthalmic diagnostic imaging of optic nerve and retin
 92227: Remote imaging for detection of retinal disease (eg, retinopathy in a patient with diabetes) with analysis and report under physician supervision, unilateral or bilateral. For Medicare, bill only
92227: Remote imaging for detection of retinal disease (eg, retinopathy in a patient with diabetes) with analysis and report under physician supervision, unilateral or bilateral. For Medicare, bill only
C. No employee shall report to work or remain on duty while having a detectable blood alcohol concentration.
 1 Series 3000 Personnel Section 3100 General Provisions Policy 3101 Drug Free Work Place File: 3101 3101.1 STATEMENT OF PURPOSE: The WV Board of Education and the Harrison County Board of Education recognize
1 Series 3000 Personnel Section 3100 General Provisions Policy 3101 Drug Free Work Place File: 3101 3101.1 STATEMENT OF PURPOSE: The WV Board of Education and the Harrison County Board of Education recognize
NOTE: The first appearance of terms in bold in the body of this document (except titles) are defined terms please refer to the Definitions section.
 TITLE SCOPE Provincial APPROVAL AUTHORITY Clinical Operations Executive Committee SPONSOR Pharmacy Services PARENT DOCUMENT TITLE, TYPE AND NUMBER Not applicable DOCUMENT # INITIAL EFFECTIVE DATE REVISION
TITLE SCOPE Provincial APPROVAL AUTHORITY Clinical Operations Executive Committee SPONSOR Pharmacy Services PARENT DOCUMENT TITLE, TYPE AND NUMBER Not applicable DOCUMENT # INITIAL EFFECTIVE DATE REVISION
Purpose of Session. Discuss. Review. Medicare audiology coverage policy. Issues raised by transmittals Possible outcomes 11/24/2008
 Purpose of Session Review Medicare audiology coverage policy 2008 Medicare audiologytransmittals Discuss Issues raised by transmittals Possible outcomes 1 Three Audiology Update Transmittals Transmittal
Purpose of Session Review Medicare audiology coverage policy 2008 Medicare audiologytransmittals Discuss Issues raised by transmittals Possible outcomes 1 Three Audiology Update Transmittals Transmittal
Claim Submission. Agenda 1/31/2013. Payment Basics
 February 2013 Jean C. Russell, MS, RHIT jrussell@epochhealth.com Richard Cooley, BA, CCS rcooley@epochhealth.com 518-430-1144 2 Payment Basics Agenda 2013 PT / OT / SP Codes Deleted Codes New Codes Significant
February 2013 Jean C. Russell, MS, RHIT jrussell@epochhealth.com Richard Cooley, BA, CCS rcooley@epochhealth.com 518-430-1144 2 Payment Basics Agenda 2013 PT / OT / SP Codes Deleted Codes New Codes Significant
Workplace Health, Safety & Compensation Review Division
 Workplace Health, Safety & Compensation Review Division WHSCRD Case No: WHSCC Claim No: Decision Number: 15240 Bruce Peckford Review Commissioner The Review Proceedings 1. The worker applied for a review
Workplace Health, Safety & Compensation Review Division WHSCRD Case No: WHSCC Claim No: Decision Number: 15240 Bruce Peckford Review Commissioner The Review Proceedings 1. The worker applied for a review
Vermont Recovery Network
 Vermont Recovery Network 200 Olcott Drive White River Junction, VT 05001 vtrecoverynetwork@gmail.com www.vtrecoverynetwork.org 802-738-8998 The Vermont Recovery Network currently consists of 12 recovery
Vermont Recovery Network 200 Olcott Drive White River Junction, VT 05001 vtrecoverynetwork@gmail.com www.vtrecoverynetwork.org 802-738-8998 The Vermont Recovery Network currently consists of 12 recovery
Chapter 15 Section 1
 Chapter 15 Section 1 Issue Date: November 6, 2007 Authority: 32 CFR 199.14(a)(3) and (a)(6)(ii) 1.0 APPLICABILITY This policy is mandatory for the reimbursement of services provided either by network or
Chapter 15 Section 1 Issue Date: November 6, 2007 Authority: 32 CFR 199.14(a)(3) and (a)(6)(ii) 1.0 APPLICABILITY This policy is mandatory for the reimbursement of services provided either by network or
Counseling to Prevent Tobacco Use
 News Flash Vaccination is the Best Protection Against the Flu. This year, the Centers for Disease Control and Prevention (CDC) is encouraging everyone 6 months of age and older to get vaccinated against
News Flash Vaccination is the Best Protection Against the Flu. This year, the Centers for Disease Control and Prevention (CDC) is encouraging everyone 6 months of age and older to get vaccinated against
ANNEX A. STATEMENT OF WORK Task Authorization (TA) 22 FOR SUB CONTRACT WITH CIMVHR
 1. NUMBER TITLE OF TASK AUTHORIZATION 1.1 TA 22 A pilot study to understand how equine-assisted therapy may assist Veterans in living with mental health conditions. 2. VALIDATION OF SCOPE OF CONTRACT 2.1
1. NUMBER TITLE OF TASK AUTHORIZATION 1.1 TA 22 A pilot study to understand how equine-assisted therapy may assist Veterans in living with mental health conditions. 2. VALIDATION OF SCOPE OF CONTRACT 2.1
ENROLMENT FORM. Title: First Name: Surname: Postal Address: Postcode: Emergency Contact: Relationship: Phone: What is your main fitness goal?
 ENROLMENT FORM Personal Information Title: First Name: Surname: Date of Birth: Sex: Female Male Postal Address: Postcode: Phone: Home: Work: Mobile: Email: Preferred method of contact: Letter Phone Email
ENROLMENT FORM Personal Information Title: First Name: Surname: Date of Birth: Sex: Female Male Postal Address: Postcode: Phone: Home: Work: Mobile: Email: Preferred method of contact: Letter Phone Email
Cluster headache (CH): epidemiology, classification and clinical picture
 Cluster headache (CH): epidemiology, classification and clinical picture Toomas Toomsoo, M.D. Head of the Center of Neurology East Tallinn Central Hospital 1 INTRODUCTION Cluster headache - known as trigeminal
Cluster headache (CH): epidemiology, classification and clinical picture Toomas Toomsoo, M.D. Head of the Center of Neurology East Tallinn Central Hospital 1 INTRODUCTION Cluster headache - known as trigeminal
Employment Contract. This sample employment contract is from Self-Employment vs. Employment Status, CDHA (no date available)
 Employment Contract This sample employment contract is from Self-Employment vs. Employment Status, CDHA (no date available (NOTE: This is only one example of an employment contract. This example is meant
Employment Contract This sample employment contract is from Self-Employment vs. Employment Status, CDHA (no date available (NOTE: This is only one example of an employment contract. This example is meant
CERT Oxygen Errors: The DME CERT Outreach and Education Task Force Responds
 CERT Oxygen Errors: The DME CERT Outreach and Education Task Force Responds DME CERT Outreach and Education Task Force National Oxygen Webinar, July 22, 2014 1 Today s Presenters Michael Hanna, CERT Task
CERT Oxygen Errors: The DME CERT Outreach and Education Task Force Responds DME CERT Outreach and Education Task Force National Oxygen Webinar, July 22, 2014 1 Today s Presenters Michael Hanna, CERT Task
WHEREAS, the Tennessee General Assembly finds that thousands of Tennesseans are
 AN ACT to license sign language interpreters WHEREAS, the Tennessee General Assembly finds that thousands of Tennesseans are individuals who are Deaf, Deaf-Blind, or Hard of Hearing; and WHEREAS, the General
AN ACT to license sign language interpreters WHEREAS, the Tennessee General Assembly finds that thousands of Tennesseans are individuals who are Deaf, Deaf-Blind, or Hard of Hearing; and WHEREAS, the General
Professional CGM Reimbursement Guide
 Professional CGM Reimbursement Guide 2017 TABLE OF CONTENTS Coding, Coverage and Payment...2 Coding and Billing...2 CPT Code 95250...3 CPT Code 95251...3 Incident to Billing for Physicians..............................................
Professional CGM Reimbursement Guide 2017 TABLE OF CONTENTS Coding, Coverage and Payment...2 Coding and Billing...2 CPT Code 95250...3 CPT Code 95251...3 Incident to Billing for Physicians..............................................
