Experimental Physiology
|
|
|
- George Garrett
- 5 years ago
- Views:
Transcription
1 1272 Exp Physiol (2012) pp Research Paper Blood flow in internal carotid and vertebral arteries during orthostatic stress Kohei Sato 1, James P. Fisher 2, Thomas Seifert 3, Morten Overgaard 3,NielsH.Secher 3 and Shigehiko Ogoh 1,4 1 Research Institute of Physical Fitness, Japan Women s College of Physical Education, Tokyo, Japan 2 School of Sport and Exercise Sciences, University of Birmingham, Birmingham, UK 3 Department of Anaesthesia, The Copenhagen Muscle Research Centre, University of Copenhagen, Copenhagen, Denmark 4 Department of Biomedical Engineering, Toyo University, Saitama, Japan Experimental Physiology It remains unclear whether orthostatic stress evokes regional differences in cerebral blood flow. The present study compared blood flow in the internal carotid (ICA) and vertebral arteries (VA) during orthostatic stress (60 deg head-up tilt; HUT) in six healthy young men. The ICA and VA blood flow were measured using Doppler ultrasonography. Dynamic cerebral autoregulation was also determined during supine (Supine) and HUT conditions, from the rate of regulation (RoR) in cerebrovascular conductance of the ICA and VA during acute hypotension induced by the release of bilateral thigh-cuffs. The HUT decreased ICA blood flow by 9.4 ± 1.7% (P < 0.01 versus Supine), leaving ICA conductance unchanged. In contrast, there was no significant difference in VA blood flow between Supine and HUT, and VA conductance increased (+12.9 ± 0.8%, P < 0.01). In addition, dynamic cerebral autoregulation in both the ICA and VA was attenuated during HUT, and the magnitude of the attenuation in RoR was greater in the VA [0.25 ± 0.03 s 1 Supine versus 0.16 ± 0.02 s 1 HUT ( 33.9 ± 5.8%); P < 0.05] compared with the ICA [0.23 ± 0.02 s 1 Supine versus 0.20 ± 0.03 s 1 HUT ( 10.6 ± 13.4%); P > 0.05]. These data indicate that orthostatic stress evokes regional differences in cerebral blood flow and possible differences in dynamic cerebral autoregulation between two main brain vascular areas in response to an acute change in blood pressure during orthostatic stress. (Received 24 January 2012; accepted after revision 6 June 2012; first published online 11 June 2012) Corresponding author K. Sato: Research Institute of Physical Fitness, Japan Women s College of Physical Education, Kita-Karasuyama, Setagaya-ku, Tokyo , Japan. ksato@jwcpe.ac.jp In humans, cerebral blood flow (CBF) is greater when supine compared with when seated or in an upright position (Alperin et al. 2005). Likewise, CBF velocity, measured in the middle cerebral artery (MCA) by transcranial Doppler (TCD), is reduced by 15 20% during simulation of orthostatic stress using head-up tilt (HUT) or lower body negative pressure (Levine et al. 1994; Immink et al. 2006; Zhang & Levine, 2007) and this cannot be explained by the concomitant reduction in the arterial carbon dioxide tension (P aco2 ; Immink et al. 2009). An important question regarding the reduction in CBF during orthostatic stress concerns whether it is a general phenomenon or whether it affects only the anterior part of the circle of Willis (the internal carotid system) as evaluated by mean blood flow velocity in the MCA (MCA V mean ; Madsen & Secher, 1999; Van Lieshout et al. 2003; Panerai, 2009). Sparse information is available on blood flow in the posterior part of the cerebral circulation (the vertebro-basilar system) during orthostatic stress (Haubrich et al. 2004; Sorond et al. 2005; Deegan et al. 2010). The vertebral artery (VA) and offshoots from the VA, including the anterior spinal and the posterior inferior cerebellar arteries, supply blood to the medulla oblongata, which is the location of important cardiac, vasomotor and respiratory control centres (Tatu et al. 1996). Many of the manifestations associated with development of presyncopal symptoms are likely to result from hypoperfusion in the vertebro-basilar system (Shin et al. 1999). Therefore, it is hypothesized that hypoperfusion of the medulla oblongata rather than the cerebral cortex during orthostatic stress could impair cardiac, vasomotor DOI: /expphysiol
2 Exp Physiol (2012) pp Cerebral blood flow during orthostatic stress 1273 and respiratory control, possibly causing many of the haemodynamic changes that take place preceding syncope. Thus, blood flow regulation in the vertebro-basilar system is likely to be particularly important for orthostatic tolerance. Although the relative contribution of the internal carotid artery (ICA) and VA to global CBF at rest is believed to be balanced in resting humans ( 75% in ICA and 25% in VA, respectively), Sato & Sadamoto (2010) and Sato et al. (2011) reported that dynamic exercise evoked different blood flow responses in the internal carotid and vertebro-basilar systems. However, the effect of orthostatic stress on the distribution of blood flow in the ICA and VA remains unclear. There are anatomical and physiological differences between the internal carotid and vertebro-basilar systems, including regional heterogeneity in the sympathetic innervation of intracranial arterioles (Edvinsson et al. 1976; Hamel et al. 1988) and cerebral CO 2 reactivity (Sorond et al. 2005; Reinhard et al. 2008), which may lead to differences in the cerebrovascular responses to an orthostatic challenge. Studies have compared dynamic cerebral autoregulation (CA) during orthostatic stress between the internal carotid and vertebro-basilar system (Haubrich et al. 2004; Deeganet al. 2010). Haubrich et al. (2004) reported that dynamic CA evaluated using transfer function analysis (TFA) was less effective in the posterior cerebral artery (PCA) than in the MCA at rest and during HUT. In contrast, there appears to be no significant difference in dynamic CA between the MCA and VA during combined HUT and lower body negative pressure to presyncope in healthy subjects (Deegan et al. 2010). However, these previous studies used CBF velocity as an index of CBF using TCD measurement. Thus, the actual and global CBF response to orthostatic stress remains unknown but can be evaluated by determination of blood flow in the ICA and VA. Figure 1. Schematic illustration of experimental protocol After baseline measurements of cerebrovascular and cardiorespiratory variables during supine (Supine) and 60 deg head-up tilt (HUT) conditions, subjects underwent a thigh-cuff test in order to assess dynamic cerebral autoregulation (CA). Abbreviations: ICA, internal carotid artery; and VA, vertebral artery. The purpose of the present study was to identify blood flow in the ICA and VA using Doppler ultrasonography and to determine the potential differences in the response of the CBF between the ICA and VA to orthostatic stress in healthy young subjects. As orthostatic syncope is related to a sudden decrease of blood flow and is generated within the vertebro-basilar system (Shin et al. 1999; Haubrich et al. 2004), we hypothesized that the VA would demonstrate a more pronounced reduction in blood flow during orthostatic stress and a lower dynamic CA than the ICA. Methods Six young men (mean ± SD: 26 ± 3 years, 177 ± 7cm and 74 ± 8 kg) participated in this study. The subjects were non-obese, normotensive and free from overt cardiovascular, pulmonary, metabolic or neurological diseases. Subjects were moderately active, non-smokers, who were not taking any medications. Written informed consent was obtained according to the regional Ethics Committee (H-A ), and the study was conducted in accordance with the principles of the Declaration of Helsinki. Subjects were requested to abstain from caffeinated beverages for 12 h and from strenuous physical activity and alcohol for at least 24 h before experimental sessions. On the experimental day, the subjects arrived at the laboratory a minimum of 2 h following a light meal. All subjects were familiarized with the equipment and procedures before any experimental sessions. Experimental protocol Subjects were supine throughout the instrumentation and stabilization period ( 45 min). After supine rest, 2 min of baseline data were recorded (Fig. 1). Subsequently, bilateral thigh-cuffs were inflated to 250 mmhg, maintained at that pressure for 3 min and then rapidly deflated, causing a transient drop in arterial pressure. After thigh-cuff deflation, measurements were continued for 2 min to assess dynamic CA (Aaslid et al. 1989; Ogoh et al. 2008), and the protocol was repeated at least twice in order to measure both ICA and VA blood flow (defined as ICA or VA trials). After recovery to re-establish resting values, the subjects were passively tilted to 60 deg headup tilt (HUT), followed by 2 min of data recording. Then, the thigh-cuff test was repeated. Thigh-cuff tests that were performed during supine rest (Supine) and HUT that resulted in a 15 mmhg or greater decrease in mean arterial pressure (MAP) were used for analysis for dynamic CA. In all experimental protocols, the subjects were asked to maintain their eyes open, and the room was illuminated.
3 1274 K. Sato and others Exp Physiol (2012) pp Cerebral blood flow The right-sided ICA and VA blood flow were measured by colour-coded ultrasonography (Vivid-e; GE healthcare, Tokyo, Japan), equipped with a 10 MHz linear transducer. The ICA blood flow measurements were made cm distal to the carotid bifurcation, and VA blood flow was measured between the transverse processes of C3 and the subclavian artery (Fig. 2). Blood flow measurement in the VA is commonly targeted between C3 and C6. The VA runs parallel with the vertebral vein; therefore, it is difficult to measure between C3 and C6 in some subjects. It is also difficult to visualize the entire VA on an ultrasound screen due to acoustic shadow caused by the transverse process of the cervical vertebra. In such cases, it is easier to visualize the blood vessel by measuring between C3 and the subclavian artery. We identified the most clearly visualized blood vessel area between C3 and the subclavian artery for blood flow measurement in each subject. In addition, the right MCA mean blood flow velocity (MCA V mean ) was measured by TCD (Multidop X; DWL, Sipplingen, Germany). A 2 MHz Doppler probe was adjusted over the temporal ultrasound window of the MCA until an acoustical signal was identified. The baseline Supine and HUT ICA and VA blood flow were averaged over 2 min. For blood flow measurements, we used the brightness mode (B mode) to measure the mean diameter of each vessel in a longitudinal section, and the Doppler velocity spectrum was subsequently identified by pulsed wave mode (PW mode), and these data were stored on the hard disk of the machine. To calculate average CBF, we analysed mean vessel diameter and mean blood flow velocity a minimum of three times over 2 min during Supine and HUT. Specifically, the systolic and diastolic diameters were measured and then the mean diameter (D mean ; in centimetres) was taken as D mean = [(systolic diameter 1 / 3 )] + [(diastolic diameter 2 / 3 )]. Moreover, the time-averaged mean flow velocity obtained using the PW mode was defined as the mean blood flow velocity (V mean ; in centimetres per second). The recordings of the V mean were taken from the average of 15 cardiac cycles in order to eliminate effects caused by the breathing cycle. Care was taken to ensure that the probe position was stable, that the insonation angle did not vary, and that the sample volume was positioned in the centre of the vessel and adjusted to cover the width of the vessel diameter. Finally, baseline blood flow in Supine and HUT were calculated by multiplying the cross-sectional area [π (D mean /2) 2 ] by V mean ; blood flow = V mean area 60 (in millilitres per minute). The coefficient of variation in test retest measurements of ICA and VA blood flow was 4.3 ± 2.2 and 3.6 ± 2.2% during Supine, and 4.2 ± 1.8 and 3.9 ± 1.9% during HUT, respectively. For assessment of dynamic CA in ICA and VA, beat-by-beat D mean and V mean in each cardiac cycle were measured using the same Doppler ultrasonography procedures as described above, and beatby-beat ICA and VA blood flows were calculated by multiplying the cross-sectional area by V mean during the thigh-cuff test. Cardiovascular measurements Heart rate (HR) was monitored using a lead II electrocardiogram (ECG; Dialogue-2000; IBC-Danica Electronic, Copenhagen, Denmark). Beat-to-beat arterial blood pressure was measured from a catheter in the left brachial artery and a transducer (Edwards Life Science, Irvine, CA, USA) placed at heart level. Beat-to-beat stroke volume (SV) was estimated from the arterial pressure wave according to the Modelflow method (Beat Scope 1.1; Finapres Medical Systems BV, Amsterdam, The Netherlands), and cardiac output (CO) was SV HR. All cardiovascular data were sampled at 1 khz using an analog-to-digital converter interfaced with a computer (Powerlab; AD Instruments, Colorado Springs, CO, USA). An arterial blood sample was obtained during the 2 min baseline period before thigh-cuff test and immediately analysed for P aco2 using an ABL725 (Radiometer, Copenhagen, Denmark). Data analysis The ratio of ICA and VA blood flow to MAP at each vessel level (flow/map) was used to calculate local cerebrovascular conductance. Estimated MAP at the ICA or VA was calculated by measuring the vertical distance from the heart to the Doppler probe in centimetres and subtracting mmhg cm 1 from MAP at heart level. For evaluation of dynamic CA, beat-by-beat ICA and VA blood flow data were obtained, and their conductance Figure 2. Internal carotid artery (ICA) and vertebral artery (VA) imaging and blood flow velocities measured by Doppler ultrasonograpy
4 Exp Physiol (2012) pp Cerebral blood flow during orthostatic stress 1275 Table 1. Baseline haemodynamic values in each trial in the supine position (Supine) and during 60 deg head-up tilt (HUT) Supine HUT Parameter ICA trial VA trial ICA trial VA trial Cardiorespiratory P aco2 (mmhg) 40.1 ± ± ± ± 0.8 MAP at heart level (mmhg) 88 ± 2 87 ± 3 82 ± 3 81 ± 3 Estimated MAP at ICA level (mmhg) 74 ± 3 Estimated MAP at VA level (mmhg) 75 ± 3 HR (beats min 1 ) 63 ± 2 64 ± 3 78 ± 5 79 ± 5 Cardiac output (l 1 min 1 ) 6.1 ± ± ± ± 0.3 Cerebral blood flow ICA blood flow (ml 1 min 1 ) 346 ± ± 16 Mean blood flow velocity (cm s 1 ) 30.4 ± ± 2.0 Mean diameter (cm) 0.49 ± ± 0.01 VA blood flow (ml 1 min 1 ) 115 ± ± 19 Mean blood flow velocity (cm s 1 ) 18.4 ± ± 2.2 Mean diameter (cm) 0.37 ± ± 0.04 MCA V mean (cm s 1 ) 56.3 ± ± 2.3 Cerebrovascular conductance ICA conductance (ml 1 min 1 mmhg 1 ) 3.95 ± ± 0.24 VA conductance (ml 1 min 1 mmhg 1 ) 1.30 ± ± 0.21 Data are means ± SEM. Abbreviations: HR, heart rate; ICA, internal carotid artery; MAP, mean arterial pressure; MCA V mean, mean blood flow velocity in middle cerebral artery; P aco2, arterial partial pressure of CO 2 ; and VA, vertebral artery. Differences between Supine and HUT are indicated as follows: P < 0.05 and P < was calculated as the ratio between blood flow and MAP. Control values for MAP, ICA and VA blood flow and conductance were defined as their means during the 4 s immediately before the thigh-cuff release. The changes in these variables during recovery from thigh-cuff release were determined relative to their concomitant control values. Although at s following thigh-cuff release the rate of change in vascular conductance expresses its dynamic CA and was used in the original study (Aaslid et al. 1989), we extended this time window to calculate the index of dynamic CA to ensure beat-by-beat blood flow data for at least three cardiac cycles. In detail, the rate of regulation (RoR) was calculated as an index of dynamic CA from the slope of the regression line between ICA or VA conductance and time (t) attime1.0to 4.0 s from thigh-cuff release, normalized by the cuff-release-induced hypotension, as follows: RoR = ( ICA or VA conductance/ t)/ MAP where ICA or VA conductance/ t is the slope of the linear regression between ICA or VA conductance and t, and MAP, the magnitude of the step, was calculated by subtracting control MAP from MAP averaged during the interval from 1.0 to 4.0 s. Consistent with previous studies (Ogoh et al. 2008, 2010; Tzeng et al. 2010), the RoR was calculated using conductance (flow/pressure). Statistics Values are expressed as means ± SEM. Repeated-measures one-way ANOVAs with Bonferoni post hoc tests were used to compare the changes in haemodynamic and dynamic CA variables between Supine and HUT, for the ICA and VA trials. All statistical analyses were conducted using SPSS (19.0, IBM SPSS Statistics, Tokyo, Japan), and P < 0.05 was considered to be a significant difference. Results Cardiorespiratory and cerebrovascular responses to head-up tilt Baseline cardiorespiratory and cerebrovascular data in ICA and VA trials obtained during Supine and HUT are presented in Table 1. When compared with Supine, the P aco2, MAP and CO were reduced (P < 0.05 for P aco2 and CO; and P < 0.01 for MAP at heart, ICA and VA levels) during HUT in both trials, while HR increased (P < 0.05). The ICA blood flow decreased during HUT by 9.4 ± 1.7% (P < 0.01), with no change in conductance (Table 1 and Fig. 3A). The decrease in ICA blood flow (Supine 346 ± 16 versus HUT 314 ± 16 ml min 1 ; P < 0.01) was caused by a decrease in both ICA diameter (Supine 0.49 ± 0.01 versus HUT 0.47 ± 0.01 cm; P < 0.05) and mean blood flow velocity (Supine 30.4 ± 1.3 versus HUT 28.6 ± 2.0 cm s 1 ; P < 0.05; Table 1). Likewise, MCA V mean decreased by 8.8 ± 2.1% (Supine 56.3 ± 3.0 versus HUT 51.2 ± 2.3 cm s 1 ; P < 0.05). In contrast, there was no significant difference in VA blood flow between Supine and HUT, and VA conductance increased (Supine 1.30 ± 0.19 versus HUT 1.46 ± 0.21 ml min 1 mmhg 1 ; P < 0.01; Table 1 and Fig. 3B).
5 1276 K. Sato and others Exp Physiol (2012) pp Thigh-cuff release In both Supine and HUT conditions, thigh-cuff release elicited a decrease in blood pressure (Fig. 4A). Changes in MAP from baseline to nadir in HUT were larger than those in Supine (P < 0.01), yet there were no significant differences between ICA and VA trials (Table 2). The degree of the decrease in ICA blood flow and VA blood flow responses to cuff release was not different during Supine, while during HUT changes in VA blood flow to thigh-cuff release were larger than those in ICA blood flow (VA 32.2 ± 1.7 versus ICA 26.1 ± 1.8%; P < 0.05; Table 2 and Fig. 4B). Immediately after thigh-cuff release, the increase in VA conductance (slope) was attenuated in HUT (Supine ± versus HUT ± 0.004; P < 0.05; Table 2), and RoR in VA was lower (Supine 0.25 ± 0.03 versus HUT 0.16 ± 0.02 s 1 ; P < 0.05; Table 2 and Fig. 5B). In contrast, dynamic CA in the ICA was not significantly attenuated during HUT (Supine 0.23 ± 0.02 versus HUT 0.20 ± 0.03 s 1 ;Table2andFig.5A). Discussion The major novel findings of the present investigation are that there is a differential blood flow response in the ICA and VA in response to orthostatic stress. While blood flow in the ICA and MCA V mean were reduced during HUT, VA blood flow was well maintained. This preservation of VA blood flow may be a beneficial response for regulating the systemic circulation during orthostatic stress. Moreover, the attenuation in RoR during HUT in the VA suggests that there may be differences in dynamic CA between two major brain vascular areas in response to an acute change in perfusion pressure during orthostatic stress. Deegan et al. (2010) attempted to demonstrate the responses of CBF for the different vessels to orthostatic stress in healthy adults. They reported that the decline in blood flow velocity during orthostatic stress was similar in the MCA and VA. Moreover, they did not find any differences in dynamic CA between MCA and VA. This discrepancy may relate to differences in the period of examination and/or the degree of the orthostatic stress, because Deegan et al. (2010) used severe orthostatic stress, which combined HUT at 70 deg with lower body negative pressure to presyncope. Furthermore, in the present study, we evaluated the blood flow extracranially, using the portion of the VA in the cervical region, while Deegan et al. (2010) used TCD measurements to assess intracranial blood flow velocity (from the transforaminal window) in the VA. Given the possibility for differences in regulatory mechanisms controlling intracranial and extracranial blood flow, this may also contribute to inconsistencies between the present study and the work of Deegan et al. (2010). Importantly, Deegan et al. (2010) Figure 3. Change in ICA and VA blood flow from Supine to HUT (A and B) and change (%) in ICA and VA blood flow from Supine to HUT (C) Difference between Supine and HUT (P < 0.01). Difference between VA and ICA (P < 0.05).
6 Exp Physiol (2012) pp Cerebral blood flow during orthostatic stress 1277 identified only CBF velocity during HUT, rather than CBF, because of the limitation of TCD measurement. Indeed, the TCD measurement is useful for identifing changes in CBF only when the arterial diameter is unchanged. Notably, in the present study a change in blood vessel diameter was one of the major causes of the difference between ICA and VA in terms of the blood flow response to HUT (Table 1). These findings raise the possibility that sole examination of CBF velocity is insufficient to characterize CBF regulation fully during orthostatic stress. Thus, the differences in the CBF responses in the ICA and VA that Figure 4. Mean response of mean arterial pressure (A), cerebral blood flow (B) and cerebrovascular conductance (C) to thigh-cuff release during Supine and HUT we report in response to HUT provide new information regarding CBF regulation during orthostatic stress. In this study, the reduction in ICA diameter was probably related to the hydrostatic pressure difference between Supine and HUT. The effect of HUT on carotid artery diameter has been examined (Steinback et al. 2005); however, data concerning the VA are lacking. The absence of a change in VA diameter during HUT may be explained by a difference in mechanical properties of the vessel for a change in hydrostatic pressure between ICA and VA. In addition, the different blood flow responses might be explained by differential autonomic innervations of the ICA and VA, or regional heterogeneity in ion channels and/or reactive oxygen species (Edvinsson et al. 1976; Andresen et al. 2006; Faraci, 2006). The posterior cerebral circulation may have less sympathetic innervation than the anterior cerebral portion (Edvinsson et al. 1976; Hamel et al. 1988). Another possible explanation for the difference between the ICA and VA in terms of blood flow responses to the HUT may be a difference in CO 2 reactivity, although posture might affect the cerebral CO 2 reactivity (Mayberg et al. 1996). Accordingly, these factors may be linked to the lower vascular tone in the VA vascular bed during orthostatic stress, although orthostatic stressinduced reductions in CBF velocity cannot be explained only by sympathetic nerve activity (Zhang & Levine, 2007) or lower P aco2 (Immink et al. 2009). An alternative explanation may be that blood flow and vascular conductance during orthostatic stress are different in the ICA and VA because they are operating in different metabolic states. As neural activity and metabolism are closely related to regional CBF, termed neurovascular coupling, the vascular beds supplying more metabolically active brain regions are likely to be dilated (Nakagawa et al. 2009). Such observations are relevant to the present study, where the territories supplied by the vertebro-basilar system (i.e. medulla oblongata, visual cortex, cerebellum and vestibular regions) are robustly and constantly activated during HUT due to sympathoexcitation, visual stimulation, postural control and gravitational stress. This would place the vertebrobasilar territories in a state of continuous vasodilatation compared with the internal carotid territories (Haubrich et al. 2004; Nakagawa et al. 2009). However, our study did not incorporate direct imaging techniques to assess regional brain metabolic rate for oxygen. Thus, we could not show any direct evidence that the orthostatic condition resulted in regional increases in blood flow within the vertebro-basilar system. The vasodilated vascular beds (increase in VA conductance) in the vertebro-basilar circulation may have induced an impaired dynamic CA response during orthostatic stress. Indeed, the decrease in RoR during orthostatic stress was more pronounced in the VA than that in the ICA (Fig. 5C). Previous study showed that dilated
7 1278 K. Sato and others Exp Physiol (2012) pp Table 2. Haemodynamic response to thigh-cuff test and variables of dynamic cerebral autoregulation in each trial in Supine and HUT conditions Supine HUT Parameter ICA trial VA trial ICA trial VA trial Responses to thigh-cuff release MAP response to cuff release (%) 25.3 ± ± ± ± 3.7 ICA blood flow response to cuff release (%) 16.5 ± ± 1.8 VA blood flow response to cuff release (%) 15.6 ± ± 1.7 Dynamic CA variables Slope of ICA conductance ± ± Slope of VA conductance ± ± MAP (%) 15.1 ± ± ± ± 3.3 ICA RoR (s 1 ) 0.23 ± ± 0.03 VA RoR (s 1 ) 0.25 ± ± 0.02 Data are means ± SEM. Abbreviations: CA, cerebral autoregulation; RoR, rate of regulation; other abbreviations are as for Table 1. Differences between Supine and HUT are indicated as follows: P < 0.05 and P < Differences between ICA trial and VA trial during HUT are indicated as follows: P < 0.05 and P < vascular beds have attenuated dynamic CA compared with constricted beds. Aaslid et al. (1989) reported that the rate of dynamic CA is dependent on basal vascular tone and that the autoregulatory response rate in MCA V mean was slower when cerebral vessels were dilated when subjects breathed a hypercapnic gas mixture. Likewise, Serrador et al. (2005) showed that transfer function gain was higher in subjects with lower cerebrovascular resistance, indicating that dynamic CA is impaired when cerebral vessels are dilated. Thus, we speculate that cerebral vasodilatation in the vascular bed of the VA during orthostatic stress contributed to the slower autoregulatory response. Previous studies have reported that dynamic CA is similar in the MCA and vertebro-basilar circulation during supine rest (Park et al. 2003) and orthostatic stress (Deegan et al. 2010). In contrast, Haubrich et al. (2004) reported that dynamic CA evaluated using TFA was less effective in the PCA than in the MCA during supine rest and orthostatic conditions. Moreover, dynamic CA in the Figure 5. Rate of regulation (RoR) in ICA and VA during Supine and HUT conditions (A and B) and change (%) in RoR from Supine to HUT (C) Difference between Supine and HUT (P < 0.05). Difference between VA and ICA (P < 0.05).
8 Exp Physiol (2012) pp Cerebral blood flow during orthostatic stress 1279 PCA is dependent on the metabolic demand of the visual cortex,andwhensubjectsclosedtheireyestherewasno difference in autoregulation between the PCA and MCA (Nakagawa et al. 2009). This further supports the concept that dynamic CA is altered by metabolic demand in the territories perfused by the vessel under examination. Based on previous studies, a possible reason why dynamic CA is different in the ICA and VA territories may be because they are operating in different metabolic states during HUT. The change in CBF responses observed during acute hypotension is consistent with some previous reports (Aaslid et al. 1989; Ogoh et al. 2008; Tzeng et al. 2010). The decrease in blood flow immediately after cuff release during HUT was more marked in the VA compared with the ICA (Table 1 and Fig. 4B). An attenuated dynamic CA in the VA may mean that flow in this vessel is more sensitive to changes in perfusion pressure. However, VA blood flow regulation in the acute hypotension phase may be different from that over a longer time course. Interestingly, following about the first five heart beats after thigh-cuff release, both ICA and VA blood flow returned to baseline values, probably due to an arterial baroreflexmediated recovery of perfusion pressure (Aaslid et al. 1989; Ogoh et al. 2008, 2010; Tzeng et al. 2010). We observed no significant difference in the increase of blood flow and cerebrovascular conductance between the ICA and VA at this time during HUT. Indeed, a recent study indicated that there is an inverse relationship between dynamic CA and arterial baroreflex sensitivity (Tzeng et al. 2010), but the baroreflex control of cerebral vascular tone in humans and the effect of the baroreflex on dynamic CA remain unknown (Ogoh et al. 2008, 2010; Tzeng et al. 2010). Potential limitations of the present study should be considered. First, the validity and reliability of CBF measurement by ultrasonography are important for our study. Blood flow evaluation by ultrasonography has the advantage of allowing simultaneous evaluation of the vessel diameter and the blood flow velocity. This information cannot be obtained by using TCD. However, measurement using a hand-held transducer without a holder, such as a head band, is expected to be a less reproducible measure because it is difficult to fix the measurement position for several minutes by using this method. However, the coefficient of variation of CBF in test retest measurements was 5% or less, both in the Supine and the HUT positions, and the values recorded were within the range of reported values at rest and during physiological stress (Schöning & Scheel, 1996; Sato & Sadamoto, 2010; Ogoh et al. 2011; Sato et al. 2011). This demonstrates a good reproducibility of the CBF measurement by ultrasonography in our study. Second, calculation of the RoR by using the cuff release method may be considered a less reproducible method for evaluating the dynamic CA (Panerai, 2009). To address this weakness in the evaluation of dynamic CA using the thigh-cuff release test, previous investigations have performed repeated trials to obtain an average RoR value (Aaslid et al. 1989; Panerai, 2009) or used the test in combination with the autoregulatory index (Tiecks et al. 1995) and TFA (Tzeng et al. 2010). The addition of autoregulatory index and/or TFA calculations would have provided support for our results. Unfortunately, this study only assessed dynamic CA using the rate of regulation. However, Haubrich et al. (2004) conducted TFA during orthostatic stress and showed that the vertebro-basilar circulation had a higher gain in blood flow in response to blood pressure variations and a weaker dynamic CA function than the internal carotid circulation. In addition, the autoregulatory index should be reflected by RoR data, and the RoR values are consistent with beat-to-beat changes in CBF within about five beats after cuff release (in the VA, lower dynamic CA probably causes a larger drop in CBF during HUT; Fig. 4B). These findings support the resultsofdynamiccainthepresentstudy. The physiological mechanism for these differential ICA and VA blood flow responses to orthostatic stress is unclear. However, our findings suggest that blood flow regulation in the vertebro-basilar system rather than in the carotid circulation is likely to be more important physiologically for maintenance of systemic circulatory control during orthostatic stress. The vertebrobasilar system provides the blood supply to the medulla oblongata, although there is a possible interaction with the internal carotid circulation. Thus, the blood supply from the basilar artery or the VA could be particularly important for cardiorespiratory regulation. We have also usedvabloodflowasanindexofbasilararterybloodflow, and the basilar artery receive their blood supply from the two VAs. Thus, VA blood flow provides more circulatory information compared with basilar artery blood flow. In summary, VA blood flow regulation during orthostatic stress was not the same as that in ICA. The ICA blood flow was reduced during HUT. In contrast, VA blood flow was preserved during orthostatic stress. This preservation of VA blood flow should be a beneficial response for regulating the systemic circulation in response to orthostatic stress. Thus, even during orthostatic stress, VA blood flow is unchanged by dilatation of territories of the vertebro-basilar system. Furthermore, the attenuation in RoR in the VA suggests possible differences in dynamic CA and CBF regulation between two main brain vascular areas in response to an acute change in perfusion pressure during orthostatic stress. References Aaslid R, Lindegaard KF, Sorteberg W & Nornes H (1989). Cerebral autoregulation dynamics in humans. Stroke 20,
9 1280 K. Sato and others Exp Physiol (2012) pp Alperin N, Lee SH, Sivaramakrishnan A & Husek SG (2005). Quantifying the effect of posture on intracranial physiology in humans by MRI flow indices. J Magn Reson Imagining 22, Andresen J, Shafi NI & Bryan RM Jr (2006). Endothelial influences on cerebrovascular tone. JApplPhysiol100, DeeganBM,CookeJP,LyonsD,ÓlaighinG&SerradorJM (2010). Cerebral autoregulation in the vertebral and middle cerebral arteries during combine head upright tilt and lower body negative pressure in healthy humans. Conf Proc IEEE Eng Med Biol Soc 2010, Edvinsson L, Owman C & Sjöberg NO (1976). Autonomic nerves, mast cells, and amine receptors in human brain vessels: a histochemical and pharmacological study. Brain Res 115, Faraci FM (2006). Reactive oxygen species: influence on cerebral vascular tone. JApplPhysiol100, Hamel E, Edvinsson L & MacKenzie ET (1988). Heterogeneous vasomotor responses of anatomically distinct feline cerebral arteries. Br J Pharmacol 94, Haubrich C, Wendt A, Diehl RR & Klözsch C (2004). Dynamic autoregulation testing in the posterior cerebral artery. Stroke 35, Immink RV, Secher NH, Roos CM, Pott F, Madsen PL & Van Lieshout JJ (2006). The postural reduction in middle cerebral artery blood velocity is not explained by PaCO 2. Eur J Appl Physiol 96, Immink RV, Truijen J, Secher NH & Van Lieshout JJ (2009). Transient influence of end-tidal carbon dioxide tension on the postural restraint in cerebral perfusion. JApplPhysiol 107, Levine BD, Giller CA, Lane LD, Buckey JC & Blomqvist CG (1994). Cerebral versus systemic hemodynamics during graded orthostatic stress in humans. Circulation 90, Madsen PL & Secher NH (1999). Near-infrared oxymetry of the brain. Prog Neurobiol 58, Mayberg TS, Lam AM, Matta BF & Visco E (1996). The variability of cerebrovascular reactivity with posture and time. J Neurosurg Anesthesiol 8, Nakagawa K, Serrador JM, Larose SL, Moslehi F, Lipsitz LA & Sorond FA (2009). Autoregulation in the posterior circulation is altered by the metabolic state of the visual cortex. Stroke 40, Ogoh S, Brothers RM, Eubank WL & Raven PB (2008). Autonomic neural control of the cerebral vasculature: acute hypotension. Stroke 39, Ogoh S, Sato K, Fisher JP, Seifert T, Overgaard M & Secher NH (2011). The effect of phenylephrine on arterial and venous cerebral blood flow in healthy subjects. Clin Physiol Funct Imaging 31, Ogoh S, Tzeng YC, Lucas SJ, Galvin SD & Ainslie PN (2010). Influence of baroreflex-mediated tachycardia on the regulation of dynamic cerebral perfusion during acute hypotension in humans. JPhysiol588, Panerai RB (2009). Transcranial Doppler for evaluation of cerebral autoregulation. Clin Auton Res 19, Park CW, Sturzenegger M, Douville CM, Aaslid R & Newell DW (2003). Autoregulatory response and CO 2 reactivity of the basilar artery. Stroke 34, Reinhard M, Waldkircher Z, Timmer J, Weiller C & Hetzel A (2008). Cerebellar autoregulation dynamics in humans. J Cereb Blood Flow Metab 28, Sato K, Ogoh S, Hirasawa A, Oue A & Sadamoto T (2011). The distribution of blood flow in the carotid and vertebral arteries during dynamic exercise in humans. JPhysiol589, Sato K & Sadamoto T (2010). Different blood flow responses to dynamic exercise between internal carotid and vertebral arteries in women. JApplPhysiol109, Schöning M & Scheel P (1996). Color duplex measurement of cerebral blood flow volume: intra- and interobserver reproducibility and habituation to serial measurements in normal subjects. J Cereb Blood Flow Metab 16, Serrador JM, Sorond FA, Vyas M, Gagnon M, Iloputaife ID & Lipsitz LA (2005). Cerebral pressure-flow relations in hypertensive elderly humans: transfer gain in different frequency domains. JApplPhysiol98, Shin HK, Yoo KM, Chang HM & Caplan LR (1999). Bilateral intracranial vertebral artery disease in the New England Medical Center, Posterior Circulation Registry. Arch Neurol 56, Sorond FA, Khavari R, Serrador JM & Lipsitz LA (2005). Regional cerebral autoregulation during orthostatic stress: age-related differences. JGerontolABiolSciMedSci60, Steinback CD, O Leary DD, Bakker J, Cechetto AD, Ladak HM & Shoemaker JK (2005). Carotid distensibility, baroreflex sensitivity, and orthostatic stress. JApplPhysiol99, Tatu L, Moulin T, Bogousslavsky J & Duvernoy H (1996). Arterial territories of human brain: brainstem and cerebellum. Neurology 47, Tiecks FP, Lam AM, Aaslid R & Newell DW (1995). Comparison of static and dynamic cerebral autoregulation measurements. Stroke 26, Tzeng YC, Lucas SJ, Atkinson G, Willie CK & Ainslie PN (2010). Fundamental relationships between arterial baroreflex sensitivity and dynamic cerebral autoregulation in humans. JApplPhysiol108, Van Lieshout JJ, Weiling W, Kaqremaker JM & Secher NH (2003). Syncope, cerebral perfusion, and oxygenation. JAppl Physiol 94, Zhang R & Levine BD (2007). Autonomic ganglionic blockade does not prevent reduction in cerebral blood flow velocity during orthostasis in humans. Stroke 38, Acknowledgements We appreciate the time and effort spent by our volunteer subjects in the present study. This study was supported by a research grant from Mr Jakob Ehrenreich and Grethe Ehrenreichs Fund (to N.H.S.); the Japanese Ministry of Education, Science, Sports and Culture (grant # to K.S.); and J.P.F. was supported by the Royal Society.
The Journal of Physiology
 J Physiol 589.11 (2011) pp 2847 2856 2847 The distribution of blood flow in the carotid and vertebral arteries during dynamic exercise in humans Kohei Sato 1, Shigehiko Ogoh 1,2, Ai Hirasawa 1, Anna Oue
J Physiol 589.11 (2011) pp 2847 2856 2847 The distribution of blood flow in the carotid and vertebral arteries during dynamic exercise in humans Kohei Sato 1, Shigehiko Ogoh 1,2, Ai Hirasawa 1, Anna Oue
Posted: 11/27/2011 on Medscape; Published Br J Anaesth. 2011;107(2): Oxford University Press
 Posted: 11/27/2011 on Medscape; Published Br J Anaesth. 2011;107(2):209-217. 2011 Oxford University Press Effect of Phenylephrine and Ephedrine Bolus Treatment on Cerebral Oxygenation in Anaesthetized
Posted: 11/27/2011 on Medscape; Published Br J Anaesth. 2011;107(2):209-217. 2011 Oxford University Press Effect of Phenylephrine and Ephedrine Bolus Treatment on Cerebral Oxygenation in Anaesthetized
Policies and Statements D16. Intracranial Cerebrovascular Ultrasound
 Policies and Statements D16 Intracranial Cerebrovascular Ultrasound SECTION 1: INSTRUMENTATION Policies and Statements D16 Intracranial Cerebrovascular Ultrasound May 2006 (Reaffirmed July 2007) Essential
Policies and Statements D16 Intracranial Cerebrovascular Ultrasound SECTION 1: INSTRUMENTATION Policies and Statements D16 Intracranial Cerebrovascular Ultrasound May 2006 (Reaffirmed July 2007) Essential
Cerebral and cardiovascular dynamics in response to orthostatic stress Harms, M.P.M.
 UvA-DARE (Digital Academic Repository) Cerebral and cardiovascular dynamics in response to orthostatic stress Harms, M.P.M. Link to publication Citation for published version (APA): Harms, M. P. M. (2008).
UvA-DARE (Digital Academic Repository) Cerebral and cardiovascular dynamics in response to orthostatic stress Harms, M.P.M. Link to publication Citation for published version (APA): Harms, M. P. M. (2008).
Cerebral autoregulation (CA) is a homeostatic mechanism
 Heterogeneous Regulation of Brain Blood Flow during Low-Intensity Resistance Exercise AI HIRASAWA 1, KOHEI SATO 2, MARINA YONEYA 2, TOMOKO SADAMOTO 2, DAMIAN M BAILEY 3, and SHIGEHIKO OGOH 4 1 Advanced
Heterogeneous Regulation of Brain Blood Flow during Low-Intensity Resistance Exercise AI HIRASAWA 1, KOHEI SATO 2, MARINA YONEYA 2, TOMOKO SADAMOTO 2, DAMIAN M BAILEY 3, and SHIGEHIKO OGOH 4 1 Advanced
Regulation of Cerebral Blood Flow. Myogenic- pressure autoregulation Chemical: PaCO2, PaO2 Metabolic Neuronal
 Regulation of Cerebral Blood Flow Myogenic- pressure autoregulation Chemical: PaCO2, PaO2 Metabolic Neuronal The Autoregulation, Stupid! Drawing of her daughter (age 7) Flow through rigid tube Mogens Fog
Regulation of Cerebral Blood Flow Myogenic- pressure autoregulation Chemical: PaCO2, PaO2 Metabolic Neuronal The Autoregulation, Stupid! Drawing of her daughter (age 7) Flow through rigid tube Mogens Fog
Diagnosis of Middle Cerebral Artery Occlusion with Transcranial Color-Coded Real-Time Sonography
 Diagnosis of Middle Cerebral Artery Occlusion with Transcranial Color-Coded Real-Time Sonography Kazumi Kimura, Yoichiro Hashimoto, Teruyuki Hirano, Makoto Uchino, and Masayuki Ando PURPOSE: To determine
Diagnosis of Middle Cerebral Artery Occlusion with Transcranial Color-Coded Real-Time Sonography Kazumi Kimura, Yoichiro Hashimoto, Teruyuki Hirano, Makoto Uchino, and Masayuki Ando PURPOSE: To determine
Neuro Quiz 29 Transcranial Doppler Monitoring
 Verghese Cherian, MD, FFARCSI Penn State Hershey Medical Center, Hershey Quiz Team Shobana Rajan, M.D Suneeta Gollapudy, M.D Angele Marie Theard, M.D Neuro Quiz 29 Transcranial Doppler Monitoring This
Verghese Cherian, MD, FFARCSI Penn State Hershey Medical Center, Hershey Quiz Team Shobana Rajan, M.D Suneeta Gollapudy, M.D Angele Marie Theard, M.D Neuro Quiz 29 Transcranial Doppler Monitoring This
Imaging of the Basal Cerebral Arteries and Measurement of Blood Velocity in Adults by Using Transcranial Real-Time Color Flow Doppler Sonography
 497 Imaging of the Basal Cerebral Arteries and Measurement of Blood Velocity in Adults by Using Transcranial Real-Time Color Flow Doppler Sonography Takashi Tsuchiya 1 Masahiro Yasaka Takenori Yamaguchi
497 Imaging of the Basal Cerebral Arteries and Measurement of Blood Velocity in Adults by Using Transcranial Real-Time Color Flow Doppler Sonography Takashi Tsuchiya 1 Masahiro Yasaka Takenori Yamaguchi
USING TRANSCRANIAL Doppler (TCD), Bode conducted
 Acta Anaesthesiol Scand 2002; 46: 393 397 Copyright C Acta Anaesthesiol Scand 2002 Printed in Denmark. All rights reserved ACTA ANAESTHESIOLOGICA SCANDINAVICA 0001-5172 Dynamic cerebral autoregulation
Acta Anaesthesiol Scand 2002; 46: 393 397 Copyright C Acta Anaesthesiol Scand 2002 Printed in Denmark. All rights reserved ACTA ANAESTHESIOLOGICA SCANDINAVICA 0001-5172 Dynamic cerebral autoregulation
The effects of superimposed tilt and lower body negative pressure on anterior and posterior cerebral circulations
 ORIGINAL RESEARCH Physiological Reports ISSN 2051-817X The effects of superimposed tilt and lower body negative pressure on anterior and posterior cerebral circulations Michael M. Tymko 1,2, Caroline A.
ORIGINAL RESEARCH Physiological Reports ISSN 2051-817X The effects of superimposed tilt and lower body negative pressure on anterior and posterior cerebral circulations Michael M. Tymko 1,2, Caroline A.
Functional transcranial Doppler sonography (ftcd) constitutes a
 PERIODICUM BIOLOGORUM UDC 57:61 VOL. 114, No 3, 313 319, 2012 CODEN PDBIAD ISSN 0031-5362 Overview BOJANA @VAN University Medical Centre Ljubljana Clinical Department of Vascular Neurology and Intensive
PERIODICUM BIOLOGORUM UDC 57:61 VOL. 114, No 3, 313 319, 2012 CODEN PDBIAD ISSN 0031-5362 Overview BOJANA @VAN University Medical Centre Ljubljana Clinical Department of Vascular Neurology and Intensive
Is Cerebral Vasomotor Reactivity impaired in Idiopathic Parkinson s Disease?
 Is Cerebral Vasomotor Reactivity impaired in Idiopathic Parkinson s Disease? Martha F Hanby BSc, MB ChB, MRes a*, Ronney B Panerai BSc, MSc, PhD a, b, Thompson G Robinson BSc, MB ChB, MD a, b and Victoria
Is Cerebral Vasomotor Reactivity impaired in Idiopathic Parkinson s Disease? Martha F Hanby BSc, MB ChB, MRes a*, Ronney B Panerai BSc, MSc, PhD a, b, Thompson G Robinson BSc, MB ChB, MD a, b and Victoria
noninvasive, nonionizing, portable, inexpensive, safe for serial or prolonged studies
 TRANS CRANIAL DOPPLER Presented by : Anil Garg Transcranial Doppler 1982, Aaslid and colleagues introduced TCD as a non-invasive technique for monitoring blood flow velocity in basal cerebral arteries
TRANS CRANIAL DOPPLER Presented by : Anil Garg Transcranial Doppler 1982, Aaslid and colleagues introduced TCD as a non-invasive technique for monitoring blood flow velocity in basal cerebral arteries
UvA-DARE (Digital Academic Repository) Cerebral autoregulation: from minutes to seconds Immink, R.V. Link to publication
 UvA-DARE (Digital Academic Repository) Cerebral autoregulation: from minutes to seconds Immink, R.V. Link to publication Citation for published version (APA): Immink, R. V. (2013). Cerebral autoregulation:
UvA-DARE (Digital Academic Repository) Cerebral autoregulation: from minutes to seconds Immink, R.V. Link to publication Citation for published version (APA): Immink, R. V. (2013). Cerebral autoregulation:
Cerebral and cardiovascular dynamics in response to orthostatic stress Harms, M.P.M.
 UvA-DARE (Digital Academic Repository) Cerebral and cardiovascular dynamics in response to orthostatic stress Harms, M.P.M. Link to publication Citation for published version (APA): Harms, M. P. M. (2008).
UvA-DARE (Digital Academic Repository) Cerebral and cardiovascular dynamics in response to orthostatic stress Harms, M.P.M. Link to publication Citation for published version (APA): Harms, M. P. M. (2008).
Recommendations for documentation of neurosonographic examinations
 Recommendations for documentation of neurosonographic examinations The documentation of ultrasound examinations is subject to a dynamic development particularly as regards newer applications. The present
Recommendations for documentation of neurosonographic examinations The documentation of ultrasound examinations is subject to a dynamic development particularly as regards newer applications. The present
Carotid Abnormalities Coils, Kinks and Tortuosity David Lorelli M.D., RVT, FACS Michigan Vascular Association Conference Saturday, October 20, 2012
 Carotid Abnormalities Coils, Kinks and Tortuosity David Lorelli M.D., RVT, FACS Michigan Vascular Association Conference Saturday, October 20, 2012 Page 1 Table of Contents Carotid Anatomy Carotid Duplex
Carotid Abnormalities Coils, Kinks and Tortuosity David Lorelli M.D., RVT, FACS Michigan Vascular Association Conference Saturday, October 20, 2012 Page 1 Table of Contents Carotid Anatomy Carotid Duplex
After space flight, there is a wide range of susceptibility to
 Impairment of Cerebral Blood Flow Regulation in Astronauts With Orthostatic Intolerance After Flight Andrew P. Blaber, PhD; Nandu Goswami, MBBS, PhD; Roberta L. Bondar, MD, PhD; Mahmood S. Kassam, DrUniv
Impairment of Cerebral Blood Flow Regulation in Astronauts With Orthostatic Intolerance After Flight Andrew P. Blaber, PhD; Nandu Goswami, MBBS, PhD; Roberta L. Bondar, MD, PhD; Mahmood S. Kassam, DrUniv
Intracranial Cerebrovascular Evaluation Transcranial Doppler (Non-Imaging) and Transcranial Duplex Imaging (TCD-I)
 VASCULAR TECHNOLOGY PROFESSIONAL PERFORMANCE GUIDELINES Intracranial Cerebrovascular Evaluation Transcranial Doppler (Non-Imaging) and Transcranial Duplex Imaging (TCD-I) This Guideline was prepared by
VASCULAR TECHNOLOGY PROFESSIONAL PERFORMANCE GUIDELINES Intracranial Cerebrovascular Evaluation Transcranial Doppler (Non-Imaging) and Transcranial Duplex Imaging (TCD-I) This Guideline was prepared by
Citation for published version (APA): Truijen, J. (2018). Withstanding the flow: Human cardiovascular control during postural challenges
 UvA-DARE (Digital Academic Repository) Withstanding the flow Truijen, J. Link to publication Citation for published version (APA): Truijen, J. (2018). Withstanding the flow: Human cardiovascular control
UvA-DARE (Digital Academic Repository) Withstanding the flow Truijen, J. Link to publication Citation for published version (APA): Truijen, J. (2018). Withstanding the flow: Human cardiovascular control
Dynamic cerebral autoregulation is preserved during isometric handgrip and head-down tilt in healthy volunteers
 ORIGINAL RESEARCH Physiological Reports ISSN 2051-817X Dynamic cerebral autoregulation is preserved during isometric handgrip and head-down tilt in healthy volunteers Maria Skytioti 1, Signe Søvik 2,3
ORIGINAL RESEARCH Physiological Reports ISSN 2051-817X Dynamic cerebral autoregulation is preserved during isometric handgrip and head-down tilt in healthy volunteers Maria Skytioti 1, Signe Søvik 2,3
Protokollanhang zur SPACE-2-Studie Neurology Quality Standards
 Protokollanhang zur SPACE-2-Studie Neurology Quality Standards 1. General remarks In contrast to SPACE-1, the neurological center participating in the SPACE-2 trial will also be involved in the treatment
Protokollanhang zur SPACE-2-Studie Neurology Quality Standards 1. General remarks In contrast to SPACE-1, the neurological center participating in the SPACE-2 trial will also be involved in the treatment
SYNCOPE. Sanjay P. Singh, MD Chairman & Professor, Department of Neurology. Syncope
 SYNCOPE Sanjay P. Singh, MD Chairman & Professor, Department of Neurology. Syncope Syncope is a clinical syndrome characterized by transient loss of consciousness (TLOC) and postural tone that is most
SYNCOPE Sanjay P. Singh, MD Chairman & Professor, Department of Neurology. Syncope Syncope is a clinical syndrome characterized by transient loss of consciousness (TLOC) and postural tone that is most
Lab Period: Name: Physiology Chapter 14 Blood Flow and Blood Pressure, Plus Fun Review Study Guide
 Lab Period: Name: Physiology Chapter 14 Blood Flow and Blood Pressure, Plus Fun Review Study Guide Main Idea: The function of the circulatory system is to maintain adequate blood flow to all tissues. Clinical
Lab Period: Name: Physiology Chapter 14 Blood Flow and Blood Pressure, Plus Fun Review Study Guide Main Idea: The function of the circulatory system is to maintain adequate blood flow to all tissues. Clinical
TCD and cardiac arrest
 TCD and cardiac arrest Background: An effective tissue perfusion has decisive influence on the final prognosis both during cardiopulmonary resuscitation (CPR) and after recovery of spontaneous circulation
TCD and cardiac arrest Background: An effective tissue perfusion has decisive influence on the final prognosis both during cardiopulmonary resuscitation (CPR) and after recovery of spontaneous circulation
ARTICLE IN PRESS. doi: /j.ultrasmedbio DYNAMIC CEREBRAL AUTOREGULATION IN THE OLD USING A REPEATED SIT-STAND MANEUVER
 doi:1.116/j.ultrasmedbio.29.1.11 Ultrasound in Med. & Biol., Vol. -, No. -, pp. 1 1, 21 Copyright Ó 21 World Federation for Ultrasound in Medicine & Biology Printed in the USA. All rights reserved 31-5629/9/$
doi:1.116/j.ultrasmedbio.29.1.11 Ultrasound in Med. & Biol., Vol. -, No. -, pp. 1 1, 21 Copyright Ó 21 World Federation for Ultrasound in Medicine & Biology Printed in the USA. All rights reserved 31-5629/9/$
Radiologic Importance of a High- Resistive Vertebral Artery Doppler Waveform on Carotid Duplex Ultrasonography
 CME Article Radiologic Importance of a High- Resistive Vertebral Artery Doppler Waveform on Carotid Duplex Ultrasonography Esther S. H. Kim, MD, MPH, Megan Thompson, Kristine M. Nacion, BA, Carmel Celestin,
CME Article Radiologic Importance of a High- Resistive Vertebral Artery Doppler Waveform on Carotid Duplex Ultrasonography Esther S. H. Kim, MD, MPH, Megan Thompson, Kristine M. Nacion, BA, Carmel Celestin,
Transcranial Color-Coded Duplex Sonography in Unilateral Flow-Restrictive Extracranial Carotid Artery Disease
 Transcranial Color-Coded Duplex Sonography in Unilateral Flow-Restrictive Extracranial Carotid Artery Disease Ralf W. Baumgartner, Iris Baumgartner, Heinrich P. Mattle, and Gerhard Schroth PURPOSE: To
Transcranial Color-Coded Duplex Sonography in Unilateral Flow-Restrictive Extracranial Carotid Artery Disease Ralf W. Baumgartner, Iris Baumgartner, Heinrich P. Mattle, and Gerhard Schroth PURPOSE: To
Experimental Physiology
 Exp Physiol 99.6 (2014) pp 849 858 849 Research Paper Research Paper Effects of acute hypoxia on cerebrovascular responses to carbon dioxide Shigehiko Ogoh 1, Hidehiro Nakahara 2,ShinyaUeda 2, Kazunobu
Exp Physiol 99.6 (2014) pp 849 858 849 Research Paper Research Paper Effects of acute hypoxia on cerebrovascular responses to carbon dioxide Shigehiko Ogoh 1, Hidehiro Nakahara 2,ShinyaUeda 2, Kazunobu
Faculty Disclosure. Sanjay P. Singh, MD, FAAN. Dr. Singh has listed an affiliation with: Consultant Sun Pharma Speaker s Bureau Lundbeck, Sunovion
 Faculty Disclosure Sanjay P. Singh, MD, FAAN Dr. Singh has listed an affiliation with: Consultant Sun Pharma Speaker s Bureau Lundbeck, Sunovion however, no conflict of interest exists for this conference.
Faculty Disclosure Sanjay P. Singh, MD, FAAN Dr. Singh has listed an affiliation with: Consultant Sun Pharma Speaker s Bureau Lundbeck, Sunovion however, no conflict of interest exists for this conference.
Influence of central command on cerebral blood flow at the onset of exercise in women
 Exp Physiol 94.11 pp 1139 1146 1139 Experimental Physiology Research Paper Influence of central command on cerebral blood flow at the onset of exercise in women Kohei Sato, Mayumi Moriyama and Tomoko Sadamoto
Exp Physiol 94.11 pp 1139 1146 1139 Experimental Physiology Research Paper Influence of central command on cerebral blood flow at the onset of exercise in women Kohei Sato, Mayumi Moriyama and Tomoko Sadamoto
Transcranial Doppler (Basic Step) Dae-il Chang, M.D., Sung Sang Yoon, M.D. Department of Neurology, College of Medicine, Kyunghee university
 Transcranial Doppler (Basic Step) Dae-il Chang, M.D., Sung Sang Yoon, M.D. Department of Neurology, College of Medicine, Kyunghee university Principles of Doppler Ultrasonography Major target Speed & direction
Transcranial Doppler (Basic Step) Dae-il Chang, M.D., Sung Sang Yoon, M.D. Department of Neurology, College of Medicine, Kyunghee university Principles of Doppler Ultrasonography Major target Speed & direction
Noninvasive transcranial Doppler ultrasound recording of flow velocity in basal cerebral arteries
 J Neurosurg 57:769-774, 1982 Noninvasive transcranial Doppler ultrasound recording of flow velocity in basal cerebral arteries RUNE AASLID, PH.D., THOMAS-MARC MARKWALDER, M.D., AND HEt,CE NORNES, M.D.
J Neurosurg 57:769-774, 1982 Noninvasive transcranial Doppler ultrasound recording of flow velocity in basal cerebral arteries RUNE AASLID, PH.D., THOMAS-MARC MARKWALDER, M.D., AND HEt,CE NORNES, M.D.
TRANSCRANIAL DOPPLER ULTRASOUND INTRODUCTION TO TCD INTERPRETATION
 TRANSCRANIAL DOPPLER ULTRASOUND INTRODUCTION TO TCD INTERPRETATION ---Rune Aaslid First TCD Publication 1982 WHAT IS TCD? Uses 2 MHz pulsed Doppler ultrasound Passes through cranial windows Provides information
TRANSCRANIAL DOPPLER ULTRASOUND INTRODUCTION TO TCD INTERPRETATION ---Rune Aaslid First TCD Publication 1982 WHAT IS TCD? Uses 2 MHz pulsed Doppler ultrasound Passes through cranial windows Provides information
Blood flow autoregulation is defined as the ability of a tissue
 Comparison of the Autoregulatory Mechanisms between Middle Cerebral Artery and Ophthalmic Artery after Thigh Cuff Deflation in Healthy Subjects Julia Kolodjaschna, 1 Fatmire Berisha, 1 Solveig Lung, 1
Comparison of the Autoregulatory Mechanisms between Middle Cerebral Artery and Ophthalmic Artery after Thigh Cuff Deflation in Healthy Subjects Julia Kolodjaschna, 1 Fatmire Berisha, 1 Solveig Lung, 1
Detection of impaired cerebral autoregulation improves by increasing arterial blood pressure variability
 Detection of impaired cerebral autoregulation improves by increasing arterial blood pressure variability Emmanuel Katsogridakis 1, Glen Bush 2, Lingke Fan 2, Anthony A. Birch 3, David M. Simpson 4, Robert
Detection of impaired cerebral autoregulation improves by increasing arterial blood pressure variability Emmanuel Katsogridakis 1, Glen Bush 2, Lingke Fan 2, Anthony A. Birch 3, David M. Simpson 4, Robert
Cerebral Blood Flow Velocity Declines Before Arterial Pressure in Patients With Orthostatic Vasovagal Presyncope
 Journal of the American College of Cardiology Vol. 39, No. 6, 2002 2002 by the American College of Cardiology Foundation ISSN 0735-1097/02/$22.00 Published by Elsevier Science Inc. PII S0735-1097(02)01719-9
Journal of the American College of Cardiology Vol. 39, No. 6, 2002 2002 by the American College of Cardiology Foundation ISSN 0735-1097/02/$22.00 Published by Elsevier Science Inc. PII S0735-1097(02)01719-9
NIH Public Access Author Manuscript J Ultrasound Med. Author manuscript; available in PMC 2010 July 20.
 NIH Public Access Author Manuscript Published in final edited form as: J Ultrasound Med. 2010 July ; 29(7): 1017 1022. BRAIN BLOOD FLOW AND VELOCITY: CORRELATIONS BETWEEN MAGNETIC RESONANCE IMAGING AND
NIH Public Access Author Manuscript Published in final edited form as: J Ultrasound Med. 2010 July ; 29(7): 1017 1022. BRAIN BLOOD FLOW AND VELOCITY: CORRELATIONS BETWEEN MAGNETIC RESONANCE IMAGING AND
PDF hosted at the Radboud Repository of the Radboud University Nijmegen
 PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/87944
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/87944
Neurocardiogenic syncope
 Neurocardiogenic syncope Syncope Definition Collapse,Blackout A sudden, transient loss of consciousness and postural tone, with spontaneous recovery Very common Syncope Prevalence All age groups (particularly
Neurocardiogenic syncope Syncope Definition Collapse,Blackout A sudden, transient loss of consciousness and postural tone, with spontaneous recovery Very common Syncope Prevalence All age groups (particularly
Chapter 62 Monitoring of Hemodynamic Change in Patients with Carotid Artery Stenosis During the Tilt Test Using Wearable Near-Infrared Spectroscopy
 Chapter 62 Monitoring of Hemodynamic Change in Patients with Carotid Artery Stenosis During the Tilt Test Using Wearable Near-Infrared Spectroscopy Takahiro Igarashi, Kaoru Sakatani, Norio Fujiwara, Yoshihiro
Chapter 62 Monitoring of Hemodynamic Change in Patients with Carotid Artery Stenosis During the Tilt Test Using Wearable Near-Infrared Spectroscopy Takahiro Igarashi, Kaoru Sakatani, Norio Fujiwara, Yoshihiro
ORIGINAL COMMUNICATION. Abstract Reduced cerebrovascular reactivity has been reported in patients with cerebral autosomal
 J Neurol (2005) 252 : 163 167 DOI 10.1007/s00415-005-0624-3 ORIGINAL COMMUNICATION Sumeet Singhal Hugh S. Markus Cerebrovascular reactivity and dynamic autoregulation in nondemented patients with CADASIL
J Neurol (2005) 252 : 163 167 DOI 10.1007/s00415-005-0624-3 ORIGINAL COMMUNICATION Sumeet Singhal Hugh S. Markus Cerebrovascular reactivity and dynamic autoregulation in nondemented patients with CADASIL
The Journal of Physiology
 J Physiol 592.23 (2014) pp 5203 5219 5203 Impact of hypocapnia and cerebral perfusion on orthostatic tolerance NiaC.S.Lewis 1,AnthonyR.Bain 1,DavidB.MacLeod 2,KevinW.Wildfong 1, Kurt J. Smith 1, Christopher
J Physiol 592.23 (2014) pp 5203 5219 5203 Impact of hypocapnia and cerebral perfusion on orthostatic tolerance NiaC.S.Lewis 1,AnthonyR.Bain 1,DavidB.MacLeod 2,KevinW.Wildfong 1, Kurt J. Smith 1, Christopher
The severity of neurologic deficits associated with
 Effect of Internal Carotid Artery Occlusion on Intracranial Hemodynamics Transcranial Doppler Evaluation and Clinical Correlation 589 Peter A. Schneider, MD, Mary E. Rossman, RVT, Eugene F. Bernstein,
Effect of Internal Carotid Artery Occlusion on Intracranial Hemodynamics Transcranial Doppler Evaluation and Clinical Correlation 589 Peter A. Schneider, MD, Mary E. Rossman, RVT, Eugene F. Bernstein,
BIPN100 F15 Human Physiol I (Kristan) Lecture 14 Cardiovascular control mechanisms p. 1
 BIPN100 F15 Human Physiol I (Kristan) Lecture 14 Cardiovascular control mechanisms p. 1 Terms you should understand: hemorrhage, intrinsic and extrinsic mechanisms, anoxia, myocardial contractility, residual
BIPN100 F15 Human Physiol I (Kristan) Lecture 14 Cardiovascular control mechanisms p. 1 Terms you should understand: hemorrhage, intrinsic and extrinsic mechanisms, anoxia, myocardial contractility, residual
Akira YOSHIOKA, Kazuki NISHIMURA, Kazutoshi SEKI, Keita ARAKANE, Tatsuya SAITO, Terumasa TAKAHARA and Sho ONODERA
 Kawasaki Journal of Medical Welfare Vol. 17, No. 1, 2011 9-13 Akira YOSHIOKA, Kazuki NISHIMURA, Kazutoshi SEKI, Keita ARAKANE, Tatsuya SAITO, Terumasa TAKAHARA and Sho ONODERA (Accepted May 20, 2011) inferior
Kawasaki Journal of Medical Welfare Vol. 17, No. 1, 2011 9-13 Akira YOSHIOKA, Kazuki NISHIMURA, Kazutoshi SEKI, Keita ARAKANE, Tatsuya SAITO, Terumasa TAKAHARA and Sho ONODERA (Accepted May 20, 2011) inferior
Title. Authors. Jatinder S Minhas 134, Nazia F Syed 1, Victoria J Haunton 13, Ronney B Panerai 23, Thompson G Robinson 134, and Amit K Mistri 14
 Title Is Dynamic Cerebral Autoregulation Measurement Using Transcranial Doppler Ultrasound Reproducible in the Presence of High Concentration Oxygen and Carbon Dioxide? Authors Jatinder S Minhas 134, Nazia
Title Is Dynamic Cerebral Autoregulation Measurement Using Transcranial Doppler Ultrasound Reproducible in the Presence of High Concentration Oxygen and Carbon Dioxide? Authors Jatinder S Minhas 134, Nazia
Note: At the end of the instructions, you will find a table which must be filled in to complete the exercise.
 Autonomic Nervous System Theoretical foundations and instructions for conducting practical exercises carried out during the course List of practical exercises 1. Deep (controlled) breath test 2. Cold pressor
Autonomic Nervous System Theoretical foundations and instructions for conducting practical exercises carried out during the course List of practical exercises 1. Deep (controlled) breath test 2. Cold pressor
19. Monitoring of CA using TCD- Mx and Sx
 19. Monitoring of CA using TCD- Mx and Sx Drawing the autoregulatory curve: in clinical practice such dramatic changes in CPP are not permissible Monitoring cerebral autoregulation - certainly not a new
19. Monitoring of CA using TCD- Mx and Sx Drawing the autoregulatory curve: in clinical practice such dramatic changes in CPP are not permissible Monitoring cerebral autoregulation - certainly not a new
Cardiac Pathophysiology
 Cardiac Pathophysiology Evaluation Components Medical history Physical examination Routine laboratory tests Optional tests Medical History Duration and classification of hypertension. Patient history of
Cardiac Pathophysiology Evaluation Components Medical history Physical examination Routine laboratory tests Optional tests Medical History Duration and classification of hypertension. Patient history of
Dynamic autoregulatory response after severe head injury
 J Neurosurg 97:1054 1061, 2002 Dynamic autoregulatory response after severe head injury ROMAN HLATKY, M.D., YU FURUYA, M.D., PH.D., ALEX B. VALADKA, M.D., JORGE GONZALEZ, M.D., ARI CHACKO, M.D., YASU MIZUTANI,
J Neurosurg 97:1054 1061, 2002 Dynamic autoregulatory response after severe head injury ROMAN HLATKY, M.D., YU FURUYA, M.D., PH.D., ALEX B. VALADKA, M.D., JORGE GONZALEZ, M.D., ARI CHACKO, M.D., YASU MIZUTANI,
Dynamic Cerebral Autoregulation in Acute Ischemic Stroke Assessed From Spontaneous Blood Pressure Fluctuations
 Dynamic Cerebral Autoregulation in Acute Ischemic Stroke Assessed From Spontaneous Blood Pressure Fluctuations M. Reinhard, MD; M. Roth, PhD; B. Guschlbauer; A. Harloff, MD; J. Timmer, PhD; M. Czosnyka,
Dynamic Cerebral Autoregulation in Acute Ischemic Stroke Assessed From Spontaneous Blood Pressure Fluctuations M. Reinhard, MD; M. Roth, PhD; B. Guschlbauer; A. Harloff, MD; J. Timmer, PhD; M. Czosnyka,
Blood Pressure Regulation. Faisal I. Mohammed, MD,PhD
 Blood Pressure Regulation Faisal I. Mohammed, MD,PhD 1 Objectives Outline the short term and long term regulators of BP Know how baroreceptors and chemoreceptors work Know function of the atrial reflex.
Blood Pressure Regulation Faisal I. Mohammed, MD,PhD 1 Objectives Outline the short term and long term regulators of BP Know how baroreceptors and chemoreceptors work Know function of the atrial reflex.
Physiology Unit 3 CARDIOVASCULAR PHYSIOLOGY: THE VASCULAR SYSTEM
 Physiology Unit 3 CARDIOVASCULAR PHYSIOLOGY: THE VASCULAR SYSTEM In Physiology Today Hemodynamics F = ΔP/R Blood flow (F) High to low pressure Rate = L/min Pressure (P) Hydrostatic pressure Pressure exerted
Physiology Unit 3 CARDIOVASCULAR PHYSIOLOGY: THE VASCULAR SYSTEM In Physiology Today Hemodynamics F = ΔP/R Blood flow (F) High to low pressure Rate = L/min Pressure (P) Hydrostatic pressure Pressure exerted
Physician s Vascular Interpretation Examination Content Outline
 Physician s Vascular Interpretation Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 2 3 4 5 6 Cerebrovascular Abdominal Peripheral Arterial - Duplex Imaging Peripheral Arterial
Physician s Vascular Interpretation Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 2 3 4 5 6 Cerebrovascular Abdominal Peripheral Arterial - Duplex Imaging Peripheral Arterial
Blood Supply. Allen Chung, class of 2013
 Blood Supply Allen Chung, class of 2013 Objectives Understand the importance of the cerebral circulation. Understand stroke and the types of vascular problems that cause it. Understand ischemic penumbra
Blood Supply Allen Chung, class of 2013 Objectives Understand the importance of the cerebral circulation. Understand stroke and the types of vascular problems that cause it. Understand ischemic penumbra
The Exercise Pressor Reflex
 The Exercise Pressor Reflex Dr. James P. Fisher School of Sport, Exercise & Rehabilitation Sciences College of Life & Environmental Sciences University of Birmingham, UK Copenhagen, 2018 Based on work
The Exercise Pressor Reflex Dr. James P. Fisher School of Sport, Exercise & Rehabilitation Sciences College of Life & Environmental Sciences University of Birmingham, UK Copenhagen, 2018 Based on work
The Journal of Physiology
 J Physiol 592.14 (2014) pp 3143 3160 3143 Dehydration affects cerebral blood flow but not its metabolic rate for oxygen during maximal exercise in trained humans Steven J. Trangmar 1, Scott T. Chiesa 1,
J Physiol 592.14 (2014) pp 3143 3160 3143 Dehydration affects cerebral blood flow but not its metabolic rate for oxygen during maximal exercise in trained humans Steven J. Trangmar 1, Scott T. Chiesa 1,
Changes in Linear Dynamics of Cerebrovascular System After Severe Traumatic Brain Injury
 Changes in Linear Dynamics of Cerebrovascular System After Severe Traumatic Brain Injury M. Müller, MD; O. Bianchi; S. Erülkü; C. Stock; K. Schwerdtfeger, MD; for the Homburg Traumatic Brain Injury Group
Changes in Linear Dynamics of Cerebrovascular System After Severe Traumatic Brain Injury M. Müller, MD; O. Bianchi; S. Erülkü; C. Stock; K. Schwerdtfeger, MD; for the Homburg Traumatic Brain Injury Group
PHYSIOLOGY MeQ'S (Morgan) All the following statements related to blood volume are correct except for: 5 A. Blood volume is about 5 litres. B.
 PHYSIOLOGY MeQ'S (Morgan) Chapter 5 All the following statements related to capillary Starling's forces are correct except for: 1 A. Hydrostatic pressure at arterial end is greater than at venous end.
PHYSIOLOGY MeQ'S (Morgan) Chapter 5 All the following statements related to capillary Starling's forces are correct except for: 1 A. Hydrostatic pressure at arterial end is greater than at venous end.
Posterior Cerebral Artery Aneurysms with Common Carotid Artery Occlusion: A Report of Two Cases
 Journal of Neuroendovascular Therapy 2017; 11: 371 375 Online March 3, 2017 DOI: 10.5797/jnet.cr.2016-0114 Posterior Cerebral Artery Aneurysms with Common Carotid Artery Occlusion: A Report of Two Cases
Journal of Neuroendovascular Therapy 2017; 11: 371 375 Online March 3, 2017 DOI: 10.5797/jnet.cr.2016-0114 Posterior Cerebral Artery Aneurysms with Common Carotid Artery Occlusion: A Report of Two Cases
Therefore MAP=CO x TPR = HR x SV x TPR
 Regulation of MAP Flow = pressure gradient resistance CO = MAP TPR Therefore MAP=CO x TPR = HR x SV x TPR TPR is the total peripheral resistance: this is the combined resistance of all blood vessels (remember
Regulation of MAP Flow = pressure gradient resistance CO = MAP TPR Therefore MAP=CO x TPR = HR x SV x TPR TPR is the total peripheral resistance: this is the combined resistance of all blood vessels (remember
Blood Pressure. a change in any of these could cause a corresponding change in blood pressure
 Blood Pressure measured as mmhg Main factors affecting blood pressure: 1. cardiac output 2. peripheral resistance 3. blood volume a change in any of these could cause a corresponding change in blood pressure
Blood Pressure measured as mmhg Main factors affecting blood pressure: 1. cardiac output 2. peripheral resistance 3. blood volume a change in any of these could cause a corresponding change in blood pressure
Arterial baroreflex regulation of cerebral blood flow in humans
 J Phys Fitness Sports Med, 1(4): 631-636 (2012) JPFSM: Review Article Arterial baroreflex regulation of cerebral blood flow in humans Shigehiko Ogoh 1*, Ai Hirasawa 1 and James P. Fisher 2 1 Department
J Phys Fitness Sports Med, 1(4): 631-636 (2012) JPFSM: Review Article Arterial baroreflex regulation of cerebral blood flow in humans Shigehiko Ogoh 1*, Ai Hirasawa 1 and James P. Fisher 2 1 Department
Regional Cerebral Blood Flow Circulation Differences Between the Middle Cerebral Artery and the Basilar Artery. Emma Quigley
 Regional Cerebral Blood Flow Circulation Differences Between the Middle Cerebral Artery and the Basilar Artery. by Emma Quigley A thesis presented to the University of Waterloo in fulfilment of the thesis
Regional Cerebral Blood Flow Circulation Differences Between the Middle Cerebral Artery and the Basilar Artery. by Emma Quigley A thesis presented to the University of Waterloo in fulfilment of the thesis
Continuous Monitoring of Blood Pressure Based on Heart Pulse Analysis
 Journal of Physics: Conference Series PAPER OPEN ACCESS Continuous Monitoring of Blood Pressure Based on Heart Pulse Analysis To cite this article: Valerie Tan et al 2018 J. Phys.: Conf. Ser. 1049 012062
Journal of Physics: Conference Series PAPER OPEN ACCESS Continuous Monitoring of Blood Pressure Based on Heart Pulse Analysis To cite this article: Valerie Tan et al 2018 J. Phys.: Conf. Ser. 1049 012062
SYMPATHETIC STRESSORS AND SYMPATHETIC FAILURES
 SYMPATHETIC STRESSORS AND SYMPATHETIC FAILURES Any discussion of sympathetic involvement in circulation, and vasodilation, and vasoconstriction requires an understanding that there is no such thing as
SYMPATHETIC STRESSORS AND SYMPATHETIC FAILURES Any discussion of sympathetic involvement in circulation, and vasodilation, and vasoconstriction requires an understanding that there is no such thing as
GUNDERSEN/LUTHERAN ULTRASOUND DEPARTMENT POLICY AND PROCEDURE MANUAL
 GUNDERSEN/LUTHERAN ULTRASOUND DEPARTMENT POLICY AND PROCEDURE MANUAL SUBJECT: Carotid Duplex Ultrasound SECTION: Vascular Ultrasound ORIGINATOR: Deborah L. Richert, BSVT, RDMS, RVT DATE: October 15, 2015
GUNDERSEN/LUTHERAN ULTRASOUND DEPARTMENT POLICY AND PROCEDURE MANUAL SUBJECT: Carotid Duplex Ultrasound SECTION: Vascular Ultrasound ORIGINATOR: Deborah L. Richert, BSVT, RDMS, RVT DATE: October 15, 2015
Orthostatic Fluid Shifts
 Orthostatic Fluid Shifts 65 Assessment of orthostatic fluid shifts with strain gauge plethysmography Roland D. Thijs, 1 Maaike Bruijnzeels, 1 Adriaan M. Kamper, 2 Arjan D. van Dijk, 3 J. Gert van Dijk
Orthostatic Fluid Shifts 65 Assessment of orthostatic fluid shifts with strain gauge plethysmography Roland D. Thijs, 1 Maaike Bruijnzeels, 1 Adriaan M. Kamper, 2 Arjan D. van Dijk, 3 J. Gert van Dijk
Veins. VENOUS RETURN = PRELOAD = End Diastolic Volume= Blood returning to heart per cardiac cycle (EDV) or per minute (Venous Return)
 Veins Venous system transports blood back to heart (VENOUS RETURN) Capillaries drain into venules Venules converge to form small veins that exit organs Smaller veins merge to form larger vessels Veins
Veins Venous system transports blood back to heart (VENOUS RETURN) Capillaries drain into venules Venules converge to form small veins that exit organs Smaller veins merge to form larger vessels Veins
Cerebral hemodynamic effects of Cheyne-Stokes respiration in a patient with stroke.
 *Marked Revision Click here to download Marked Revision: manuscript_marked changes_final.docx Cerebral hemodynamic effects of Cheyne-Stokes respiration in a patient with stroke. Nogueira RC 1, Panerai
*Marked Revision Click here to download Marked Revision: manuscript_marked changes_final.docx Cerebral hemodynamic effects of Cheyne-Stokes respiration in a patient with stroke. Nogueira RC 1, Panerai
Analysis of CO 2 Vasomotor Reactivity and Vessel Diameter Changes by Simultaneous Venous and Arterial Doppler Recordings
 Analysis of CO 2 Vasomotor Reactivity and Vessel Diameter Changes by Simultaneous Venous and Arterial Doppler Recordings José Manuel Valdueza, MD; Bogdan Draganski; Olaf Hoffmann; Ulrich Dirnagl, MD; Karl
Analysis of CO 2 Vasomotor Reactivity and Vessel Diameter Changes by Simultaneous Venous and Arterial Doppler Recordings José Manuel Valdueza, MD; Bogdan Draganski; Olaf Hoffmann; Ulrich Dirnagl, MD; Karl
Heat stress redistributes blood flow in arteries of the brain during dynamic exercise
 J Appl Physiol : 766 773, 2016. First published February 4, 2016; doi:10.1152/japplphysiol.00353.2015. Heat stress redistributes blood flow in arteries of the brain during dynamic exercise Kohei Sato,
J Appl Physiol : 766 773, 2016. First published February 4, 2016; doi:10.1152/japplphysiol.00353.2015. Heat stress redistributes blood flow in arteries of the brain during dynamic exercise Kohei Sato,
Statistical analysis
 Abstract Background: We tested the hypothesis that paradigms from the Addenbrooke s Cognitive Examination (ACE-III), including those that had not been studied using TCD previously (novel) versus those
Abstract Background: We tested the hypothesis that paradigms from the Addenbrooke s Cognitive Examination (ACE-III), including those that had not been studied using TCD previously (novel) versus those
Enhancement of Cranial US: Utility of Supplementary Acoustic Windows and Doppler Harriet J. Paltiel, MD
 Enhancement of Cranial US: Utility of Supplementary Acoustic Windows and Doppler Harriet J. Paltiel, MD Boston Children s Hospital Harvard Medical School None Disclosures Conventional US Anterior fontanelle
Enhancement of Cranial US: Utility of Supplementary Acoustic Windows and Doppler Harriet J. Paltiel, MD Boston Children s Hospital Harvard Medical School None Disclosures Conventional US Anterior fontanelle
MRI MEASUREMENTS OF CRANIOSPINAL AND INTRACRANIAL VOLUME CHANGE IN HEALTHY AND HEAD TRAUMA CASES
 1of 4 MRI MEASUREMENTS OF CRANIOSPINAL AND INTRACRANIAL VOLUME CHANGE IN HEALTHY AND HEAD TRAUMA CASES N. Alperin, Y. Kadkhodayan, B. Varadarajalu, C. Fisher, B. Roitberg Department of Radiology and Neurosurgery,
1of 4 MRI MEASUREMENTS OF CRANIOSPINAL AND INTRACRANIAL VOLUME CHANGE IN HEALTHY AND HEAD TRAUMA CASES N. Alperin, Y. Kadkhodayan, B. Varadarajalu, C. Fisher, B. Roitberg Department of Radiology and Neurosurgery,
EFFECTS OF SUDDEN AND PROLONGED STANDING FROM SUPINE POSTURE ON HEART RATE, ECG-PATTERN AND BLOOD PRESSURE
 J. Human Ergol.,17: 3-12,1988 Center for Academic Publications Japan. Printed in Japan. EFFECTS OF SUDDEN AND PROLONGED STANDING FROM SUPINE POSTURE ON HEART RATE, ECG-PATTERN AND BLOOD PRESSURE Satipati
J. Human Ergol.,17: 3-12,1988 Center for Academic Publications Japan. Printed in Japan. EFFECTS OF SUDDEN AND PROLONGED STANDING FROM SUPINE POSTURE ON HEART RATE, ECG-PATTERN AND BLOOD PRESSURE Satipati
to Regulation of the Brain Vessels
 Short Communication Japanese Journal of Physiology, 34,193-197,1984 The Relevance of Cardio-pulmonary-vascular Reflex to Regulation of the Brain Vessels Masatsugu NAKAI and Koichi OGINO Department of Cardiovascular
Short Communication Japanese Journal of Physiology, 34,193-197,1984 The Relevance of Cardio-pulmonary-vascular Reflex to Regulation of the Brain Vessels Masatsugu NAKAI and Koichi OGINO Department of Cardiovascular
Hypertension The normal radial artery blood pressures in adults are: Systolic arterial pressure: 100 to 140 mmhg. Diastolic arterial pressure: 60 to
 Hypertension The normal radial artery blood pressures in adults are: Systolic arterial pressure: 100 to 140 mmhg. Diastolic arterial pressure: 60 to 90 mmhg. These pressures are called Normal blood pressure
Hypertension The normal radial artery blood pressures in adults are: Systolic arterial pressure: 100 to 140 mmhg. Diastolic arterial pressure: 60 to 90 mmhg. These pressures are called Normal blood pressure
Chapter 24 Vital Signs. Copyright 2011 Wolters Kluwer Health Lippincott Williams & Wilkins
 Chapter 24 Vital Signs Vital Signs Temperature Pulse Respiration Blood pressure When to Assess Vital Signs Upon admission to any healthcare agency Based on agency institutional policy and procedures Anytime
Chapter 24 Vital Signs Vital Signs Temperature Pulse Respiration Blood pressure When to Assess Vital Signs Upon admission to any healthcare agency Based on agency institutional policy and procedures Anytime
The effects of reduced end-tidal carbon dioxide tension on cerebral blood flow during heat stress
 J Physiol 587.15 (2009) pp 3921 3927 3921 The effects of reduced end-tidal carbon dioxide tension on cerebral blood flow during heat stress R. Matthew Brothers 1,2,JonathanE.Wingo 1,2, Kimberly A. Hubing
J Physiol 587.15 (2009) pp 3921 3927 3921 The effects of reduced end-tidal carbon dioxide tension on cerebral blood flow during heat stress R. Matthew Brothers 1,2,JonathanE.Wingo 1,2, Kimberly A. Hubing
Dynamic Cerebral Autoregulation Changes during Sub- Maximal Handgrip Maneuver
 Dynamic Cerebral Autoregulation Changes during Sub- Maximal Handgrip Maneuver Ricardo C. Nogueira 1, Edson Bor-Seng-Shu 2, Marcelo R. Santos 3, Carlos E. Negrão 3, Manoel J. Teixeira 2, Ronney B. Panerai
Dynamic Cerebral Autoregulation Changes during Sub- Maximal Handgrip Maneuver Ricardo C. Nogueira 1, Edson Bor-Seng-Shu 2, Marcelo R. Santos 3, Carlos E. Negrão 3, Manoel J. Teixeira 2, Ronney B. Panerai
Physiology of Circulation
 Physiology of Circulation Dr. Ali Ebneshahidi Blood vessels Arteries: Blood vessels that carry blood away from the heart to the lungs and tissues. Arterioles are small arteries that deliver blood to the
Physiology of Circulation Dr. Ali Ebneshahidi Blood vessels Arteries: Blood vessels that carry blood away from the heart to the lungs and tissues. Arterioles are small arteries that deliver blood to the
Chapter 14 Blood Vessels, Blood Flow and Pressure Exam Study Questions
 Chapter 14 Blood Vessels, Blood Flow and Pressure Exam Study Questions 14.1 Physical Law Governing Blood Flow and Blood Pressure 1. How do you calculate flow rate? 2. What is the driving force of blood
Chapter 14 Blood Vessels, Blood Flow and Pressure Exam Study Questions 14.1 Physical Law Governing Blood Flow and Blood Pressure 1. How do you calculate flow rate? 2. What is the driving force of blood
Three questions to the human brain
 PERIODICUM BIOLOGORUM UDC 57:61 VOL. 116, No 1, 9 14, 2014 CODEN PDBIAD ISSN 0031-5362 Three questions to the human brain NIELS H. SECHER Department of Anesthesia Rigshospitalet 2041 Blegdamsvej 9 DK-2100
PERIODICUM BIOLOGORUM UDC 57:61 VOL. 116, No 1, 9 14, 2014 CODEN PDBIAD ISSN 0031-5362 Three questions to the human brain NIELS H. SECHER Department of Anesthesia Rigshospitalet 2041 Blegdamsvej 9 DK-2100
The relationship of blood flow velocity fluctuations to intracranial pressure B waves
 J Neurosurg 76:415-421, 1992 The relationship of blood flow velocity fluctuations to intracranial pressure B waves DAVID W. NEWELL, M.D., RUNE AASLID, Ph.D., RENATE STOOSS, R.N., AND HANS J. REULEN, M.D.
J Neurosurg 76:415-421, 1992 The relationship of blood flow velocity fluctuations to intracranial pressure B waves DAVID W. NEWELL, M.D., RUNE AASLID, Ph.D., RENATE STOOSS, R.N., AND HANS J. REULEN, M.D.
Blood Pressure and its Regulation
 Blood Pressure and its Regulation Blood pressure in your blood vessels is closely monitored by baroreceptors; they send messages to the cardio regulatory center of your medulla oblongata to regulate your
Blood Pressure and its Regulation Blood pressure in your blood vessels is closely monitored by baroreceptors; they send messages to the cardio regulatory center of your medulla oblongata to regulate your
Precision Medicine in Neurocritical Care: Should we individualize care?
 Precision Medicine in Neurocritical Care: Should we individualize care? Victoria McCredie Toronto Western Hospital Critical Care Canada Forum 2 nd November 2016 Conflicts of interest None Outline 1. Precision
Precision Medicine in Neurocritical Care: Should we individualize care? Victoria McCredie Toronto Western Hospital Critical Care Canada Forum 2 nd November 2016 Conflicts of interest None Outline 1. Precision
What is the mechanism of the audible carotid bruit? How does one calculate the velocity of blood flow?
 CASE 8 A 65-year-old man with a history of hypertension and coronary artery disease presents to the emergency center with complaints of left-sided facial numbness and weakness. His blood pressure is normal,
CASE 8 A 65-year-old man with a history of hypertension and coronary artery disease presents to the emergency center with complaints of left-sided facial numbness and weakness. His blood pressure is normal,
Any vascular studies performed should be as a result of, or to complement, a thorough patient evaluation and neurological examination.
 National Imaging Associates, Inc. Clinical guidelines NON-INVASIVE CEREBROVASCULAR ARTERIALS TUDIES Original Date: October 2015 Page 1 of 8 FOR CMS (MEDICARE) MEMBERS ONLY CPT4 Codes: Please refer to page
National Imaging Associates, Inc. Clinical guidelines NON-INVASIVE CEREBROVASCULAR ARTERIALS TUDIES Original Date: October 2015 Page 1 of 8 FOR CMS (MEDICARE) MEMBERS ONLY CPT4 Codes: Please refer to page
Special circulations, Coronary, Pulmonary. Faisal I. Mohammed, MD,PhD
 Special circulations, Coronary, Pulmonary Faisal I. Mohammed, MD,PhD 1 Objectives Describe the control of blood flow to different circulations (Skeletal muscles, pulmonary and coronary) Point out special
Special circulations, Coronary, Pulmonary Faisal I. Mohammed, MD,PhD 1 Objectives Describe the control of blood flow to different circulations (Skeletal muscles, pulmonary and coronary) Point out special
Effects of dominant and non-dominant passive arm manoeuvres on the neurovascular coupling response
 Effects of dominant and non-dominant passive arm manoeuvres on the neurovascular coupling response Osian Llwyd 1,2, Ronney B. Panerai 1,2 and Thompson G. Robinson 1,2 1 Department of Cardiovascular Sciences,
Effects of dominant and non-dominant passive arm manoeuvres on the neurovascular coupling response Osian Llwyd 1,2, Ronney B. Panerai 1,2 and Thompson G. Robinson 1,2 1 Department of Cardiovascular Sciences,
Exercise plus volume loading prevents orthostatic intolerance but not reduction in cerebral blood flow velocity after bed rest
 Am J Physiol Heart Circ Physiol 302: H489 H497, 2012. First published November 11, 2011; doi:10.1152/ajpheart.00427.2011. Exercise plus volume loading prevents orthostatic intolerance but not reduction
Am J Physiol Heart Circ Physiol 302: H489 H497, 2012. First published November 11, 2011; doi:10.1152/ajpheart.00427.2011. Exercise plus volume loading prevents orthostatic intolerance but not reduction
Department of Intensive Care Medicine UNDERSTANDING CIRCULATORY FAILURE IN SEPSIS
 Department of Intensive Care Medicine UNDERSTANDING CIRCULATORY FAILURE IN SEPSIS UNDERSTANDING CIRCULATORY FAILURE IN SEPSIS a mismatch between tissue perfusion and metabolic demands the heart, the vasculature
Department of Intensive Care Medicine UNDERSTANDING CIRCULATORY FAILURE IN SEPSIS UNDERSTANDING CIRCULATORY FAILURE IN SEPSIS a mismatch between tissue perfusion and metabolic demands the heart, the vasculature
Assumption of the upright position is associated with a
 Muscle Tensing During Standing Effects on Cerebral Tissue Oxygenation and Cerebral Artery Blood Velocity Johannes J. van Lieshout, MD, PhD; Frank Pott, MD; Per Lav Madsen, MD; Jeroen van Goudoever, MSc,
Muscle Tensing During Standing Effects on Cerebral Tissue Oxygenation and Cerebral Artery Blood Velocity Johannes J. van Lieshout, MD, PhD; Frank Pott, MD; Per Lav Madsen, MD; Jeroen van Goudoever, MSc,
Transcranial Doppler In Cerebral Vasospasm
 Cerebral Vasospasm 1042-3680/90 $0.00 +.20 Transcranial Doppler In Cerebral Vasospasm David W. Newell, MD,* and H. Richard Winn, MDt The confirmation of cerebral vasospasm following subarachnoid hemorrhage,
Cerebral Vasospasm 1042-3680/90 $0.00 +.20 Transcranial Doppler In Cerebral Vasospasm David W. Newell, MD,* and H. Richard Winn, MDt The confirmation of cerebral vasospasm following subarachnoid hemorrhage,
Principles Arteries & Veins of the CNS LO14
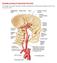 Principles Arteries & Veins of the CNS LO14 14. Identify (on cadaver specimens, models and diagrams) and name the principal arteries and veins of the CNS: Why is it important to understand blood supply
Principles Arteries & Veins of the CNS LO14 14. Identify (on cadaver specimens, models and diagrams) and name the principal arteries and veins of the CNS: Why is it important to understand blood supply
Chapter 9. Body Fluid Compartments. Body Fluid Compartments. Blood Volume. Blood Volume. Viscosity. Circulatory Adaptations to Exercise Part 4
 Body Fluid Compartments Chapter 9 Circulatory Adaptations to Exercise Part 4 Total body fluids (40 L) Intracellular fluid (ICF) 25 L Fluid of each cell (75 trillion) Constituents inside cell vary Extracellular
Body Fluid Compartments Chapter 9 Circulatory Adaptations to Exercise Part 4 Total body fluids (40 L) Intracellular fluid (ICF) 25 L Fluid of each cell (75 trillion) Constituents inside cell vary Extracellular
Cardiac Output MCQ. Professor of Cardiovascular Physiology. Cairo University 2007
 Cardiac Output MCQ Abdel Moniem Ibrahim Ahmed, MD Professor of Cardiovascular Physiology Cairo University 2007 90- Guided by Ohm's law when : a- Cardiac output = 5.6 L/min. b- Systolic and diastolic BP
Cardiac Output MCQ Abdel Moniem Ibrahim Ahmed, MD Professor of Cardiovascular Physiology Cairo University 2007 90- Guided by Ohm's law when : a- Cardiac output = 5.6 L/min. b- Systolic and diastolic BP
