INSTRUCTIONS FOR USE FOR:
|
|
|
- Jared Austin
- 5 years ago
- Views:
Transcription
1 INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
2 INSTRUCTIONS FOR USE FOR GORE TIGRIS Vascular Stent Carefully read all instructions prior to use. Observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. DESCRIPTION The GORE TIGRIS Stent is a flexible, self-expanding endoluminal stent consisting of a nitinol (NiTi = Nickel:Titanium) stent and an external fluoropolymer lattice extending along its entire length (Figure 1). The lumen of the GORE TIGRIS Stent is coated with the CARMEDA BioActive Surface (CBAS Surface) consisting of a stable, covalently bonded reduced molecular weight heparin of porcine origin. The stent is compressed and attached to a tri-axial delivery catheter (Figure 2). The central catheter lumen is used for flushing and guidewire introduction. The delivery catheter handle assembly has one port for flushing and guidewire insertion. To facilitate accurate stent placement, two radiopaque metallic bands are attached to the catheter shaft, marking the ends of the compressed stent. The GORE TIGRIS Stent is supplied STERILE. The GORE TIGRIS Stent should not be resterilized. FIGURE 1: GORE TIGRIS STENT Nitinol Stent Fluoropolymer Lattice FIGURE 2: GORE TIGRIS STENT DELIVERY SYSTEM Radiopaque Marker Locations Constrained Endoprosthesis Deployment Wheel Guidewire / Flushing Port Catheter Shaft Red Deployment Lock Guidewire INTENDED USE / INDICATIONS The GORE TIGRIS Vascular Stent is intended for endovascular stenting of peripheral arteries. CONTRAINDICATIONS The GORE TIGRIS Stent is contraindicated for non-compliant lesions where full expansion of an angioplasty balloon catheter was not achieved during pre-dilatation, or where lesions cannot be dilated sufficiently to allow passage of the delivery system. Do not use the GORE TIGRIS Stent in patients with known hypersensitivity to heparin, including those patients who have had a previous incidence of Heparin-Induced Thrombocytopenia (HIT) type II. 1
3 TABLE 1: SIZING TABLE 1 Labeled Device Diameter (mm) Device Sizing Recommended Vessel Diameter 1 (mm) Introducer Sheath Size (Fr) Guidewire Diameter Recommended Balloon Diameter for Device Touch-up (mm) " (0.889 mm) " (0.889 mm) " (0.889 mm) " (0.889 mm) 8.0 Recommended stent compression within the vessel is approximately 5 20%. PACKAGE HANDLING Store in a cool dry place. This product has an expiration date and should be used before the labeled use by (expiration) date marked on the box. The foil pouch for the GORE TIGRIS Stent is both a moisture barrier and a sterile barrier. DO NOT use or store the device if the foil pouch has been compromised. METHOD Preparation of patients receiving the GORE TIGRIS Stent should include initiation of an appropriate dosage of oral antiplatelet medication prior to and following the procedure. Effective anticoagulation therapy should be maintained throughout the procedure and continued into the postoperative period, as deemed appropriate by the treating physician. The presence of heparin on the GORE TIGRIS Stent is not intended to serve as an alternative to the physician s chosen intraoperative or postoperative anticoagulation regimens. Prior to implantation of the GORE TIGRIS Stent, the physician should refer to the Sizing Table (Table 1) and read the Directions for Use. Proper placement of the stent should be monitored and confirmed using fluoroscopy. Sterile precautions should be the same as for any device implant procedure. Post-deployment stent dilation with an appropriately sized balloon is recommended to ensure complete stent-to-vessel apposition. If performed, select an appropriately sized balloon (Table 1) that matches the size of the reference vessel but one that is not larger than the labeled stent diameter. WARNINGS Do not use the GORE TIGRIS Stent for the treatment of ostial lesions or lesions involving a major side branch that may be crossed by the stent. Do not use in patients with a history of intolerance or adverse reaction to antiplatelet and / or anticoagulation therapies, bleeding diathesis, severe hypertension or renal failure. With any vascular procedure, the possibility of HIT may exist. The incidence of HIT type II is extremely low in vascular patients receiving heparin over a period of several days. If HIT type II is diagnosed, established procedures for the treatment of this condition, including immediate cessation of systemic heparin administration, should be followed. 1,2,3 If symptoms persist, or the health of the patient appears compromised, alternative pharmaceutical or surgical procedures, including removal of the stent, may be considered at the discretion of the attending physician. Do not use a kinked introducer sheath. A kinked introducer sheath may increase the force necessary to deploy the stent and may cause a deployment failure or catheter breakage on removal. Should it become necessary to remove the GORE TIGRIS Stent from the vessel after unlocking the device, DO NOT reuse the GORE TIGRIS Stent. This can result in premature deployment. Inadvertent, partial, or failed deployment or migration of the stent may require surgical intervention. PRECAUTIONS The GORE TIGRIS Stent is designed for single use only; do not reuse device. Gore does not have data regarding reuse of this device. Reuse may cause device failure or procedural complications including device damage, compromised device biocompatibility, and device contamination. Reuse may result in infection, serious injury, or patient death. Do not use the GORE TIGRIS Stent if the sterile package is compromised or the GORE TIGRIS Stent is damaged. Do not use the GORE TIGRIS Stent after the labeled use by (expiration) date. Do not resterilize the GORE TIGRIS Stent. The GORE TIGRIS Stent should only be used by physicians trained in endovascular techniques. Follow the Directions for Use supplied with all accessories used in conjunction with the GORE TIGRIS Stent. The stent is not designed for repositioning once deployment is started. Do not dilate the stent with a balloon longer than the labeled stent length. Refer to Sizing Table (Table 1) for selection of appropriate balloon diameter. Do not attempt to withdraw or reposition a balloon catheter within the lumen of the deployed stent unless the balloon is completely deflated. Antiplatelet medication should be initiated prior to placement of the GORE TIGRIS Stent. Effective anticoagulation therapy should be maintained at a dosage deemed appropriate by the physician. The presence of heparin on the GORE TIGRIS Stent is not intended to serve as an alternative to the physician s chosen intraoperative or postoperative anticoagulation regimens. 2
4 MR CONDITIONAL MR INFORMATION The GORE TIGRIS Vascular Stent (single and two overlapped versions) was determined to be MR-conditional. Non-clinical testing demonstrated that the GORE TIGRIS Vascular Stent is MR Conditional. A patient with this device can be scanned safely, immediately after placement under the following conditions: Static Magnetic Field - Static magnetic field of 3-Tesla or less - Maximum spatial gradient magnetic field of 720-Gauss/cm or less - Maximum whole-body-averaged specific absorption rate (SAR) of 3.0 W/kg for 15 minutes of scanning MRI-Related Heating In non-clinical testing, the GORE TIGRIS Vascular Stent (single and two overlapped versions) produced the following temperature rises during MRI performed for 15-min of scanning (i.e., per pulse sequence) in 1.5-Tesla/64-MHz (Magnetom, Siemens Medical Solutions, Malvern, PA. Software Numaris/4, Version Syngo MR 2002B DHHS Active-shielded, horizontal field scanner) and 3-Tesla (3-Tesla/128-MHz, Excite, HDx, Software 14X.M5, General Electric Healthcare, Milwaukee, WI) MR systems: 1.5-Tesla 3-Tesla MR system reported, whole body averaged SAR 2.9-W/kg 3.0-W/kg Calorimetry measured values, whole body averaged SAR 2.1-W/kg 2.8-W/kg Highest temperature change, single +1.5 C +2.0 C Highest temperature change, two overlapped +1.6 C +2.2 C These temperature changes should not pose a hazard to a human subject under the conditions indicated above. Artifact Information MR image quality may be affected if the area of interest is in the exact same area or relatively close to the position of the GORE TIGRIS Vascular Stent. Therefore, optimization of MR imaging parameters to compensate for the presence of this device may be necessary. Pulse Sequence T1-SE T1-SE GRE GRE Signal Void Size 1,201-mm mm 2 1,733-mm mm 2 Plane Orientation Parallel Perpendicular Parallel Perpendicular HAZARDS AND ADVERSE EVENTS Procedure Related: As with all procedures that utilize techniques for introducing a catheter into a vessel, complications may be expected. These complications include, but are not limited to: access site infection; entry site bleeding and / or hematoma; vessel thrombosis, occlusion, pseudoaneurysm, and trauma to the vessel wall (including rupture or dissection); distal embolization; arteriovenous fistula formation; transient or permanent contrast induced renal failure; renal toxicity; sepsis; shock; radiation injury; myocardial infarction; fever; pain; malposition; malapposition; inflammation; and / or death. Device Related: Complications and adverse events can occur when using any endovascular device. These complications include, but are not limited to: hematoma; stenosis, thrombosis or occlusion; distal embolism; vessel wall trauma and / or rupture; false aneurysm; infection; inflammation; fever and / or pain in the absence of infection; deployment failure; migration; and device failure. A possible complication which may occur in conjunction with the use of any heparin-containing product: HIT type II. MATERIALS REQUIRED FOR IMPLANTATION GORE TIGRIS Stent Marker guidewire or catheter (for calibrated measurement reference) Syringe filled with heparinized saline Introducer sheath of appropriate size (Table 1) Appropriate guidewire [a 0.035" (0.889 mm) diameter guidewire is recommended] A guidewire with a length at least twice the length of the GORE TIGRIS Stent delivery catheter Appropriate angioplasty balloon catheters and accessories (Table 1) Appropriate diagnostic catheters and accessories DIRECTIONS FOR USE Treatment of Vessel Obstruction A. Access 1. Using standard techniques, gain access to the vessel and insert the vascular introducer sheath. B. Imaging and Measurement 1. To determine the appropriate device size, measure the vessel diameter and lesion length using standard imaging techniques. 3
5 C. Percutaneous Transluminal Angioplasty (PTA) (if required at the discretion of the attending physician) 1. Refer to manufacturer s Directions for Use. 2. Inflate the angioplasty balloon to its nominal pressure according to manufacturer s Directions for Use. Ensure full expansion of the balloon within the lesion. Note: Carefully mark the margins of the angioplasty treatment segment in order to ensure complete coverage with the stent. 3. Following deflation of the angioplasty balloon, evaluate the results angiographically. D. Sizing and Selection of the GORE TIGRIS Stent 1. Prior to opening the Sterile Package, check that the diameter and length of the stent as well as the delivery catheter length are correct. a. In selecting the appropriate size stent, a careful assessment of the vessel is necessary. In general, to assure adequate anchoring, the diameter of the stent should be approximately 5 20% larger than the healthy vessel diameter immediately proximal and distal to the lesion (Table 1). b. Where possible it is recommended to extend the stent into healthy vessel at least 1 cm beyond the proximal and distal margins of the lesion. c. Verify that there is sufficient catheter length to access the treatment site. 2. When overlapping (telescoping) multiple devices, the following are suggested: Balloon touch-up (post-dilatation) should be performed on the first device prior to placing the second device. To ensure proper seating, at least 1 cm of overlap between devices is suggested. Overlapping devices should not differ by more than 1 mm in diameter. If unequal device diameters are used, the smaller device should be placed first and then the larger device should be placed inside of the smaller device. E. Preparation of the GORE TIGRIS Stent 1. Open the sterile package. Carefully inspect the packaging for damage to the sterile barrier. Do not use the GORE TIGRIS Stent after the use by (expiration) date. Peel back the outer pouch and remove the sterile inner pouch, coil and backer card containing the GORE TIGRIS Stent. Beginning at one corner, peel back the edge of the inner pouch and gently remove the GORE TIGRIS Stent. 2. Inspection Prior to Use. Prior to using the GORE TIGRIS Stent, all materials and equipment to be used for the procedure should be carefully examined for bends, kinks, or other damage. Do not manipulate the constrained stent. Do not use any defective equipment. Do not use the GORE TIGRIS Stent if the sterile package is compromised or the GORE TIGRIS Stent is damaged. 3. Preparation of the GORE TIGRIS Stent delivery catheter. a. Flush the delivery catheter by attaching a syringe of heparinized saline to the flushing port on the handle (Figure 2). Continue flushing until a steady stream of fluid exits the tip of the catheter. b. After flushing the catheter, remove the syringe. 4. Do not let the surface of the GORE TIGRIS Stent dry once it has been wetted. F. Introduction and Positioning of the GORE TIGRIS Stent 1. Select the compatible introducer sheath size from Table Ensure the guidewire is 0.035" (0.889 mm). The guidewire must have a length at least twice that of the delivery catheter. 3. Be sure to remove the balloon catheter while maintaining the position of the guidewire beyond the target lesion. 4. Do not manipulate the constrained stent. 5. With the delivery catheter as straight as possible, insert the guidewire into the tip of the delivery catheter while supporting the delivery catheter and the compressed stent. Carefully advance the stent in small increments (approximately 0.5 cm) over the guidewire, through the hemostasis valve and introducer sheath, and into the access vessel. Note: If excessive resistance is felt as the GORE TIGRIS Stent is introduced through the hemostasis valve, remove and inspect the delivery system for damage. Do not reuse the GORE TIGRIS Stent if damaged. Ensure a compatible introducer sheath size (Table 1), and that the introducer sheath is free of kinks. To decrease introduction force, consider GENTLY moistening constrained stent using standard techniques. DO NOT TWIST THE DEVICE WHILE INSERTING IT THROUGH THE INTRODUCER SHEATH. 6. Using fluoroscopic guidance, advance the delivery catheter over the guidewire via the sheath. Advance cautiously, especially if resistance is felt. If excessive resistance is felt, remove the delivery catheter. 7. Position the GORE TIGRIS Stent across the target lesion using the radiopaque hub and tip markers on the catheter. These markers identify the proximal and distal ends of the stent, respectively. Note: If PTA is performed, the stent length should cover the entire vessel segment treated with balloon angioplasty. For treatment of stenotic or occlusive lesions, the stent should extend at least 1 cm proximal and distal to the margins of the lesion. 4
6 G. Final Positioning and Deployment of the GORE TIGRIS Stent 1. While keeping the extracorporeal segment of the catheter as straight as possible (Figure 3A), unlock the device by pushing the deployment lock on the handle inward and simultaneously sliding toward the rear of the handle until it reaches its full extent of travel (Figure 3B). Note that the nose of the handle will retract during the unlocking operation. Note: If resistance is encountered during the unlock procedure, gently push the nose of the handle toward the handle and continue to deploy. 2. After the unlock procedure is completed, finalize the position of the stent across the target lesion and verify fluorscopically. 3. Stabilize the handle against the table and the delivery catheter at the hemostasis valve of the introducer sheath. It is also important to stabilize the delivery catheter and introducer sheath relative to the patient. This will minimize catheter movement during deployment and ensure accurate stent positioning. 4. Rotate the deployment wheel as shown (Figure 3C) to deploy the stent. Note: Deployment of the stent will occur from the tip of the delivery catheter toward the hub. Note: Once deployment has started, repositioning of the stent or advancing the delivery system should not be attempted. Note: Should it become necessary to remove the GORE TIGRIS Stent from the vessel after unlocking the device, DO NOT reuse the GORE TIGRIS Stent. This can result in premature deployment. FIGURE 3A: FIGURE 3B: LOCKED UNLOCKED FIGURE 3C: 5. While maintaining the position of the guidewire across the treated lesion, carefully withdraw the delivery catheter through the lumen of the stent and remove it via the introducer sheath. Moderate resistance may be felt when the distal tip is withdrawn through the introducer sheath. Excessive or abrupt force during catheter removal may damage the stent, delivery catheter, or introducer sheath. 6. After deployment, it is recommended to inflate an angioplasty balloon within the stent to ensure complete apposition to the vessel wall. Balloon diameter should be selected according to Table 1 and should be inflated to the desired diameter along the entire length of the stent. If the stent length exceeds that of the balloon, multiple inflations may be needed. After the balloon is inflated throughout the stent, attention is required to ensure complete deflation of the balloon prior to cautious removal of the balloon catheter to prevent stent displacement. Do not extend balloon dilatation beyond the ends of the device and into healthy vessel as this may induce restenosis. 7. Using contrast angiography, evaluate the treated segment prior to completing the procedure. A final angiographic run to evaluate vessel patency is recommended. 8. When clinically appropriate, remove the introducer sheath and achieve hemostasis of the puncture site. 5
7 REFERENCES 1. Ortel TL, Chong BH. New treatment options for heparin-induced thrombocytopenia. Seminars in Hematology 1998; 35 (4): Almeida JI, Coats R, Liem TK, Silver D. Reduced morbidity and mortality rates of heparin-induced thrombocytopenia. Journal of Vascular Surgery 1998; 27 (2): Warkentin TE, Grienacher A, Koster A, Lincoff AM. Treatment and prevention of heparin-induced thrombocytopenia: American College of Chest Physicians evidence-based clinical practice guidelines (8th edition). Chest 2008;133:340S-380S. DEFINITIONS Use By Caution Consult Instructions for Use 2 STERILIZE Do Not Resterilize V Do Not Reuse Catalogue Number Batch Code Authorised Representative in the European Community MR Conditional CAUTION: USA Federal Law restricts the sale, distribution, or use of this device to, by, or on the order of a physician. Sterile Sterilized using Ethylene Oxide Do Not Use if Package is Damaged Keep Dry Store in a Cool Place Diameter Manufacturer Catheter Working Length Delivery Profile Guidewire Compatibility Vessel Diameter 6
8 AN0265-ML1 Manufacturer W. L. Gore & Associates, Inc. Flagstaff, Arizona USA Order Information: Tel.: Tel.: Technical Information: Tel.: Tel.: For international contact and additional product information, visit GORE, PROPATEN, TIGRIS, and designs are trademarks of W. L. Gore & Associates. CARMEDA and CBAS are trademarks of Carmeda AB Corp., a wholly owned subsidiary of W. L. Gore & Associates, Inc W. L. Gore & Associates, Inc. Printed on recyclable paper AUGUST 2011
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
RECOMMENDED INSTRUCTIONS FOR USE
 Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System
 100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Balloon in Balloon (BIB )
 Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER
 Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
Allium Trans-hepatic Biliary Stent (BIS)
 Allium Trans-hepatic Biliary (BIS) Instructions for Use Manufactured by Allium Ltd. DEVICE NAME Allium Trans-hepatic Biliary (BIS) DEVICE DESCRIPTION The Trans-hepatic BIS is a flexible, self-expanding
Allium Trans-hepatic Biliary (BIS) Instructions for Use Manufactured by Allium Ltd. DEVICE NAME Allium Trans-hepatic Biliary (BIS) DEVICE DESCRIPTION The Trans-hepatic BIS is a flexible, self-expanding
DISTAL RADIUS. Instructions for Use
 DISTAL RADIUS Instructions for Use CAUTION: FEDERAL LAW RESTRICTS THESE DEVICES TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Table of Contents 1. INTENDED USE... 3 2. DEVICE DESCRIPTION... 3 3. METHOD OF
DISTAL RADIUS Instructions for Use CAUTION: FEDERAL LAW RESTRICTS THESE DEVICES TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Table of Contents 1. INTENDED USE... 3 2. DEVICE DESCRIPTION... 3 3. METHOD OF
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
North Medical Endoscopic Biliary Stent (BilS)
 North Medical Endoscopic Biliary Stent (BilS) Instructions for Use Manufactured by North Medical DEVICE NAME North Medical Endoscopic Biliary Stent (BilS) DEVICE DESCRIPTION North Medical's BilS stent
North Medical Endoscopic Biliary Stent (BilS) Instructions for Use Manufactured by North Medical DEVICE NAME North Medical Endoscopic Biliary Stent (BilS) DEVICE DESCRIPTION North Medical's BilS stent
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
Balloon in Balloon (BIB )
 in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM
 Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
 M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
WallFlex Biliary RX Partially Covered Stent System Prescriptive Information
 Caution/Rx Only: Federal Law (USA) restricts this device to sale by or on the order of a physician. Intended Use/Indications for Use The WallFlex Biliary RX is indicated for use in the palliative treatment
Caution/Rx Only: Federal Law (USA) restricts this device to sale by or on the order of a physician. Intended Use/Indications for Use The WallFlex Biliary RX is indicated for use in the palliative treatment
Introduction What Causes Peripheral Vascular Disease? How Do Doctors Treat Peripheral Vascular Disease?... 9
 Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
DURABLE. CONSISTENT. SAFE. IN.PACT Admiral Drug-Coated Balloon
 DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
Talent Abdominal Stent Graft
 Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
MULTILAYER FLOW MODULATOR (MFM ) FOR PERIPHERAL ANEURYSMS TREATMENT (LOWER LIMBS, VISCERALS AND SUBCLAVIAN)
 0459 MULTILAYER FLOW MODULATOR (MFM ) FOR PERIPHERAL ANEURYSMS TREATMENT (LOWER LIMBS, VISCERALS AND SUBCLAVIAN) WITH ITS ANGIOGRAPHIC CONTROL DELIVERY SYSTEM INSTRUCTIONS FOR USE (I.F.U.) English Français
0459 MULTILAYER FLOW MODULATOR (MFM ) FOR PERIPHERAL ANEURYSMS TREATMENT (LOWER LIMBS, VISCERALS AND SUBCLAVIAN) WITH ITS ANGIOGRAPHIC CONTROL DELIVERY SYSTEM INSTRUCTIONS FOR USE (I.F.U.) English Français
CLARIVEIN INFUSION CATHETER
 CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
Peel-Apart Percutaneous Introducer Kits for
 Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Directions For Use. All directions should be read before use
 Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Introduction 3. What is Peripheral Vascular Disease? 5. What Are Some of the Symptoms of Peripheral Vascular Disease? 6
 Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
REBEL. Platinum Chromium Coronary Stent System. Patient Information Guide
 REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
Advanced Innovation for Exceptional Performance
 Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter
 Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
1 Comparison of Warfarin and Aspirin for Symptomatic Intracranial Arterial Stenosis. N Engl J Med 352;13, March 31, 2005
 The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
Instructions for Use 1
 Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU)
 Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER. Instructions for Use
 Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional
Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE
 CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE INDICATIONS FOR USE The LATERA Absorbable Nasal Implant is indicated for supporting upper and lower lateral nasal cartilage. CAUTION: Federal law restricts
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE INDICATIONS FOR USE The LATERA Absorbable Nasal Implant is indicated for supporting upper and lower lateral nasal cartilage. CAUTION: Federal law restricts
INSTRUCTIONS FOR USE FOR: English
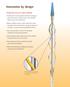
 INSTRUCTIONS FOR USE FOR: H E P A R I N B I O A C T I V E S U R F A C E en English INSTRUCTIONS FOR USE FOR Endoprosthesis Endoprosthesis with Heparin Bioactive Surface Carefully read all instructions
INSTRUCTIONS FOR USE FOR: H E P A R I N B I O A C T I V E S U R F A C E en English INSTRUCTIONS FOR USE FOR Endoprosthesis Endoprosthesis with Heparin Bioactive Surface Carefully read all instructions
PTCA Dilatation Catheter. Instructions for Use
 PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
Single Pass MCA Revascularization with Trevo
 Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp
 Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use
 Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use DEVICE DESCRIPTION The Detachable Azur CX 35 Peripheral Coil System (Azur system) consists of a coil implant attached to a delivery system.
Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use DEVICE DESCRIPTION The Detachable Azur CX 35 Peripheral Coil System (Azur system) consists of a coil implant attached to a delivery system.
Figure 1: Revolution TM Peripheral Atherectomy System Diagram. Table 1: Revolution TM Peripheral Atherectomy System Specifications Minimum Burr
 Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
PTCA Dilatation Catheter. Instructions for Use
 PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
INSTRUCTIONS FOR USE (IFU)
 INSTRUCTIONS FOR USE (IFU) B05696 Vers.3/11-09 BARD E LUMINEXX Vascular Stent Delivery System Diagrams BARD S.A.F.E. Delivery System with the PERFORMAXX Grip BARD S.A.F.E. Delivery System after removal
INSTRUCTIONS FOR USE (IFU) B05696 Vers.3/11-09 BARD E LUMINEXX Vascular Stent Delivery System Diagrams BARD S.A.F.E. Delivery System with the PERFORMAXX Grip BARD S.A.F.E. Delivery System after removal
Document No. BMB/IFU/40 Rev No. & Date 00 & 15/11/2017 Issue No & Date 01 & 15/11/2017
 Central Venous Catheter Device Description Multi-lumen catheters incorporate separate, non-communicating vascular access lumens within a single catheter body. Minipunctur Access Sets And Trays: Used for
Central Venous Catheter Device Description Multi-lumen catheters incorporate separate, non-communicating vascular access lumens within a single catheter body. Minipunctur Access Sets And Trays: Used for
Bard LifeStar Vascular Stent System
 Bard LifeStar Vascular Stent System Instructions For Use (IFU) Bard LifeStar Vascular Stent System Delivery System Diagram 8x60 B05685 Vers.2/12-11 1 Instructions for use Read the Bard LifeStar Vascular
Bard LifeStar Vascular Stent System Instructions For Use (IFU) Bard LifeStar Vascular Stent System Delivery System Diagram 8x60 B05685 Vers.2/12-11 1 Instructions for use Read the Bard LifeStar Vascular
The following languages are included in this packet:
 OSTEOSET XR BONE VOID FILLER 150841-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese
OSTEOSET XR BONE VOID FILLER 150841-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese
CARILLON Mitral Contour System (XE2)
 CARILLON Mitral Contour System (XE2) Instructions for Use 0 3 4 4 Manufacturer: Cardiac Dimensions, Inc. 5540 Lake Washington Blvd. NE Kirkland, WA 98033 United States of America EC Representative: MedPass
CARILLON Mitral Contour System (XE2) Instructions for Use 0 3 4 4 Manufacturer: Cardiac Dimensions, Inc. 5540 Lake Washington Blvd. NE Kirkland, WA 98033 United States of America EC Representative: MedPass
Directions For Use. All directions should be read before use. Page 1 of 8
 Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
System.
 System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
Patient Brochure. Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland. PK Rev. 0 05/17
 Patient Brochure Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland PK1411100 Rev. 0 05/17 LIFESTREAM Patient Brochure If you or a member of your family has been diagnosed with
Patient Brochure Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland PK1411100 Rev. 0 05/17 LIFESTREAM Patient Brochure If you or a member of your family has been diagnosed with
Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully
 Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Endorectal Balloon (ERB) Endorectal Balloon (ERB) Instructions for Use
 Endorectal Balloon (ERB) Instructions for Use GRF-0004-00 Rev. 08 Page 1 of 9 Contents Instructions for Use... 3 Indications for Use... 3 Contraindications... 3 Storage... 4 Warnings... 4 Accessories Provided...
Endorectal Balloon (ERB) Instructions for Use GRF-0004-00 Rev. 08 Page 1 of 9 Contents Instructions for Use... 3 Indications for Use... 3 Contraindications... 3 Storage... 4 Warnings... 4 Accessories Provided...
ASAHI Corsair INSTRUCTIONS FOR USE
 ASAHI Corsair INSTRUCTIONS FOR USE SYMBOLS Legal manufacturer Do not use if package is damaged GC Minimum GC I.D. REF Catalogue number Caution, consult accompanying documents Maximum injection pressure
ASAHI Corsair INSTRUCTIONS FOR USE SYMBOLS Legal manufacturer Do not use if package is damaged GC Minimum GC I.D. REF Catalogue number Caution, consult accompanying documents Maximum injection pressure
INSTRUCTIONS FOR USE
 PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
CELLPLEX TCP SYNTHETIC CANCELLOUS BONE
 CELLPLEX TCP SYNTHETIC CANCELLOUS BONE 129257-9 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) -
CELLPLEX TCP SYNTHETIC CANCELLOUS BONE 129257-9 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) -
Neuroform EZ Stent System
 Neuroform EZ Stent System Directions for Use 2 Neuroform EZ Stent System 1 cm 1.9 cm 135 cm 185 cm ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning
Neuroform EZ Stent System Directions for Use 2 Neuroform EZ Stent System 1 cm 1.9 cm 135 cm 185 cm ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning
CP STENT. Large Diameter, Balloon Expandable Stent
 CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
INSTRUCTIONS FOR USE FOR: English
 INSTRUCTIONS FOR USE FOR: en English dk TABLE OF CONTENTS DESCRIPTION...1 INTENDED USE/INDICATIONS FOR USE...2 CONTRAINDICATIONS...2 WARNINGS...2 PRECAUTIONS...3 MAGNETIC RESONANCE IMAGING (MRI)...3 ADVERSE
INSTRUCTIONS FOR USE FOR: en English dk TABLE OF CONTENTS DESCRIPTION...1 INTENDED USE/INDICATIONS FOR USE...2 CONTRAINDICATIONS...2 WARNINGS...2 PRECAUTIONS...3 MAGNETIC RESONANCE IMAGING (MRI)...3 ADVERSE
Cardiva Catalyst III INSTRUCTIONS FOR USE
 Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
AERO DV Tracheobronchial Direct Visualization Stent System
 AERO DV Tracheobronchial Direct Visualization Stent System Review Instructions For Use Before Using This System. Single Use Only Non-sterile MR Conditional CONTENTS Instructions for Use...............................
AERO DV Tracheobronchial Direct Visualization Stent System Review Instructions For Use Before Using This System. Single Use Only Non-sterile MR Conditional CONTENTS Instructions for Use...............................
Per-Q-Cath* PICC Catheters with Excalibur Introducer* System
 Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
Innovation by design. Technology that sets a new standard
 Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Zenith Alpha T HORACIC ENDOVASCULAR GRAFT
 Deployment Sequence Zenith Alpha T HORACIC ENDOVASCULAR GRAFT www.cookmedical.com AI-D21183-EN-F Preparation and flush Proximal and distal components Remove the yellow hubbed inner stylet from the dilator
Deployment Sequence Zenith Alpha T HORACIC ENDOVASCULAR GRAFT www.cookmedical.com AI-D21183-EN-F Preparation and flush Proximal and distal components Remove the yellow hubbed inner stylet from the dilator
MRI Information for Medtronic Spinal and Biologics Devices
 M708348B263E Rev C Release Date: 2013-02 MRI Information for Medtronic Spinal and Biologics Devices Not for Distribution in the U.S or its territories. 1 P age Table of Contents MRI Information for Medtronic
M708348B263E Rev C Release Date: 2013-02 MRI Information for Medtronic Spinal and Biologics Devices Not for Distribution in the U.S or its territories. 1 P age Table of Contents MRI Information for Medtronic
Instructions for Use
 Ovation ix Abdominal Stent Graft System Instructions for Use EN (810-0016-01-01) 0 0 5 0 810-0016-01-01 Rev A Table of Contents 1. Device Description... 3 1.1. Delivery System... 4 1.2. Fill Kit and Autoinjector...
Ovation ix Abdominal Stent Graft System Instructions for Use EN (810-0016-01-01) 0 0 5 0 810-0016-01-01 Rev A Table of Contents 1. Device Description... 3 1.1. Delivery System... 4 1.2. Fill Kit and Autoinjector...
Finally, the Control You Need to Deliver Accurate Treatment
 Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
Instructions for Use
 with the Ovation ix Iliac Stent Graft Instructions for Use EN (810-0009-01-04) 0 0 5 0 810-0009-01-04 Rev A Table of Contents 1. Device Description... 3 1.1. Delivery System... 4 1.2. Fill Kit and Autoinjector...
with the Ovation ix Iliac Stent Graft Instructions for Use EN (810-0009-01-04) 0 0 5 0 810-0009-01-04 Rev A Table of Contents 1. Device Description... 3 1.1. Delivery System... 4 1.2. Fill Kit and Autoinjector...
Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent. Michael K. W. Lichtenberg MD, FESC
 Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution
Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution
FROM THE EVERYDAY TO THE EXTRAORDINARY
 FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES
 MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
Pocket Reference Guide For (CTO) Technologies
 to receive our latest news and key activities. Pocket Reference Guide For (CTO) Technologies LinkedIn page Follow us on CORDIS EMEA Table of Contents Essential prescribing information for both FRONTRUNNER
to receive our latest news and key activities. Pocket Reference Guide For (CTO) Technologies LinkedIn page Follow us on CORDIS EMEA Table of Contents Essential prescribing information for both FRONTRUNNER
INSTRUCTIONS FOR USE FOR:
 INSTRUCTIONS FOR USE FOR: en English INSTRUCTIONS FOR USE GORE ENFORM PREPERITONEAL BIOMATERIAL Carefully read all instructions prior to use. Observe all instructions, warnings, and precautions noted throughout.
INSTRUCTIONS FOR USE FOR: en English INSTRUCTIONS FOR USE GORE ENFORM PREPERITONEAL BIOMATERIAL Carefully read all instructions prior to use. Observe all instructions, warnings, and precautions noted throughout.
St. Jude Medical 8-Channel Adapter. Clinician's Manual
 St. Jude Medical 8-Channel Adapter Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. For U.S. California Only: Proposition 65, a State of California
St. Jude Medical 8-Channel Adapter Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. For U.S. California Only: Proposition 65, a State of California
Ruby Coil. Large Volume Detachable Coils
 Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ
 BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ 1 Case 1 78 yr. old female Rutherford Class II/III lesion 1 block claudicant 2 Pre Treatment Post Treatment Anterior Tibial Artery Occlusion
BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ 1 Case 1 78 yr. old female Rutherford Class II/III lesion 1 block claudicant 2 Pre Treatment Post Treatment Anterior Tibial Artery Occlusion
CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN.
 Mounted CP Stent COARCTATION OF THE AORTA INSTRUCTIONS FOR USE General Instructions.. Page 2 Stent Sizing Charts and Information..Page 21 CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR
Mounted CP Stent COARCTATION OF THE AORTA INSTRUCTIONS FOR USE General Instructions.. Page 2 Stent Sizing Charts and Information..Page 21 CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING
 Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
 Page 1 of 5 Reprocessed by Instructions for Use Reprocessed Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
Page 1 of 5 Reprocessed by Instructions for Use Reprocessed Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
Single Use Curlew TM Multiple Biopsy Forceps
 Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
AXIOS Stent and Delivery System
 PRESCRIPTIVE INFORMATION AXIOS Stent and Delivery System Warning REFER TO THE DEVICE DIRECTIONS FOR USE FOR COMPLETE INSTRUCTIONS ON DEVICE USE. RX ONLY. CAUTION: FEDERAL LAW (USA) RESTRICTS THIS DEVICE
PRESCRIPTIVE INFORMATION AXIOS Stent and Delivery System Warning REFER TO THE DEVICE DIRECTIONS FOR USE FOR COMPLETE INSTRUCTIONS ON DEVICE USE. RX ONLY. CAUTION: FEDERAL LAW (USA) RESTRICTS THIS DEVICE
Understanding aneurysms and flow diversion treatment
 Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Neuroform Microdelivery Stent System
 Neuroform Microdelivery Stent System Patient Information Booklet TABLE OF CONTENTS What is the Purpose of This Booklet?...2 What is a Wide Neck, Intracranial Aneurysm?...2 What is the Treatment for Wide
Neuroform Microdelivery Stent System Patient Information Booklet TABLE OF CONTENTS What is the Purpose of This Booklet?...2 What is a Wide Neck, Intracranial Aneurysm?...2 What is the Treatment for Wide
Ancillary Components with Z-Trak Introduction System
 Ancillary Components with Z-Trak Introduction System Zenith Flex AAA Endovascular Graft Ancillary Components Converter Converters can be used to convert a bifurcated graft into an aortouniiliac graft if
Ancillary Components with Z-Trak Introduction System Zenith Flex AAA Endovascular Graft Ancillary Components Converter Converters can be used to convert a bifurcated graft into an aortouniiliac graft if
ROTABLATORTM. Peripheral. Rotational Atherectomy System. Quick Reference Cards
 Peripheral ROTABLATORTM Rotational Atherectomy System Quick Reference Cards Technical Assistance: 1.800.949.6708 Customer Service: 1.888.272.1001 Complaints: 1.800.811.3211 Setup Video: www.bostonscientific.com/rotablatorsetup
Peripheral ROTABLATORTM Rotational Atherectomy System Quick Reference Cards Technical Assistance: 1.800.949.6708 Customer Service: 1.888.272.1001 Complaints: 1.800.811.3211 Setup Video: www.bostonscientific.com/rotablatorsetup
Chronic Total Occlusion (CTO) Technologies
 to receive our latest news and key activities. Chronic Total Occlusion (CTO) Technologies Re-open vital channels LinkedIn page Follow us on CORDIS EMEA OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry
to receive our latest news and key activities. Chronic Total Occlusion (CTO) Technologies Re-open vital channels LinkedIn page Follow us on CORDIS EMEA OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry
Lutonix AV Clinical Trial
 Lutonix AV Clinical Trial Long Term Effects of LUTONIX 035 DCB Catheter 18 month Interim Results Scott O. Trerotola, MD Stanley Baum Professor of Radiology Professor of Surgery Associate Chair and Chief,
Lutonix AV Clinical Trial Long Term Effects of LUTONIX 035 DCB Catheter 18 month Interim Results Scott O. Trerotola, MD Stanley Baum Professor of Radiology Professor of Surgery Associate Chair and Chief,
Advancing Lives and the Delivery of Health Care. The High-Flow Port Designed for Apheresis
 Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
AXIOS Stent and Electrocautery Enhanced Delivery System. Quick Reference Guide
 AXIOS Stent and Electrocautery Enhanced Delivery System Quick Reference Guide AXIOS Stent (front and side views) UPN Codes Flange Diameter (mm) Lumen Diamter (mm) Saddle Length (mm) Catheter OD (Fr) Catheter
AXIOS Stent and Electrocautery Enhanced Delivery System Quick Reference Guide AXIOS Stent (front and side views) UPN Codes Flange Diameter (mm) Lumen Diamter (mm) Saddle Length (mm) Catheter OD (Fr) Catheter
Cook Medical. Zenith Flex AAA Endovascular Graft with Z-Trak Introduction System Physician Training
 Cook Medical Zenith Flex AAA Endovascular Graft with Z-Trak Introduction System Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full-thickness,
Cook Medical Zenith Flex AAA Endovascular Graft with Z-Trak Introduction System Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full-thickness,
Express LD Iliac. Premounted Stent System ONLY. over-the-wire
 50455844-01 STERILE - DO NOT RESTERILIZE - SINGLE USE ONLY ONLY 2017-03 < EN > Express LD Iliac over-the-wire Premounted Stent System Caution: Federal Law (USA) restricts this device to sale by or on the
50455844-01 STERILE - DO NOT RESTERILIZE - SINGLE USE ONLY ONLY 2017-03 < EN > Express LD Iliac over-the-wire Premounted Stent System Caution: Federal Law (USA) restricts this device to sale by or on the
FLEXIC ATH LTD. Peripherally Inserted. Instructions n For Use.
 FLEXIC ATH LTD * M/29M Peripherally Inserted Catheter Instructions n For Use This leaflet contains instructions for both stan- dard needle-introducer er and protection con- tained M/29 models, i.e., with
FLEXIC ATH LTD * M/29M Peripherally Inserted Catheter Instructions n For Use This leaflet contains instructions for both stan- dard needle-introducer er and protection con- tained M/29 models, i.e., with
Nit-Occlud. Coil System for PDA Closure IMPLANTATION POCKET GUIDE. Rx only CV / B. Braun Interventional Systems Inc.
 Refer to the Nit-Occlud PDA Instructions for Use for relevant warnings, precautions, complications and contraindications. This device has been designed for single use only. Nit-Occlud Coil System for PDA
Refer to the Nit-Occlud PDA Instructions for Use for relevant warnings, precautions, complications and contraindications. This device has been designed for single use only. Nit-Occlud Coil System for PDA
Emerge. PTCA Dilatation Catheter ONLY. monorail. over-the-wire
 90990229-01 Emerge monorail over-the-wire PTCA Dilatation Catheter Table of Contents 2014-09 < EN > warning...1 device description...1 Table 1. Emerge Balloon Coatings...1 Contents...1 Intended use/indications
90990229-01 Emerge monorail over-the-wire PTCA Dilatation Catheter Table of Contents 2014-09 < EN > warning...1 device description...1 Table 1. Emerge Balloon Coatings...1 Contents...1 Intended use/indications
PERFORMANCE YOU CAN TRUST. EverFlex Self-expanding Peripheral Stent with Entrust Delivery System
 PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
Access More Patients. Customize Each Seal.
 Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
Device Preparation (all steps to be performed per standard interventional technique)
 sertera Biopsy Device Instructions for Use Please read all information carefully. Failure to properly follow the instructions may lead to unintended consequences. Important: This package insert is designed
sertera Biopsy Device Instructions for Use Please read all information carefully. Failure to properly follow the instructions may lead to unintended consequences. Important: This package insert is designed
Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use. Polyurethane Dual Lumen Occlusion Catheter
 Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use Polyurethane Dual Lumen Occlusion Catheter Polyurethane Dual Lumen Occlusion Catheter (Model Numbers 4F-2L-40, 4F-2L-80, 5F-2L-40,
Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use Polyurethane Dual Lumen Occlusion Catheter Polyurethane Dual Lumen Occlusion Catheter (Model Numbers 4F-2L-40, 4F-2L-80, 5F-2L-40,
Understanding Peripheral
 Patient Information Guide Understanding Peripheral Artery Disease Innova Vascular Self-Expanding Stent System Table of Contents Glossary... 2 What is Peripheral Artery Disease?... 4 Treating Peripheral
Patient Information Guide Understanding Peripheral Artery Disease Innova Vascular Self-Expanding Stent System Table of Contents Glossary... 2 What is Peripheral Artery Disease?... 4 Treating Peripheral
ENG TREO ABDOMINAL STENT-GRAFT SYSTEM TABLE OF CONTENTS
 TREO ABDOMINAL STENT-GRAFT SYSTEM TABLE OF CONTENTS 1. DEVICE DESCRIPTION 28 1.1. Stent-Graft 28 1.1.1. Main Bifurcated Stent-Graft 28 1.1.2. Leg Extensions 28 1.2. Delivery System for the Bifurcated Main
TREO ABDOMINAL STENT-GRAFT SYSTEM TABLE OF CONTENTS 1. DEVICE DESCRIPTION 28 1.1. Stent-Graft 28 1.1.1. Main Bifurcated Stent-Graft 28 1.1.2. Leg Extensions 28 1.2. Delivery System for the Bifurcated Main
CATHETER ACCESS KIT. For use with Prometra Programmable Infusion Systems
 CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
