COIL SYSTEM ORDERING INFORMATION
|
|
|
- Eustace Johnson
- 5 years ago
- Views:
Transcription
1 EMBOLIZATION SYSTEM
2 RUBY COIL SYSTEM FRAME with Ruby Standard FILL with Ruby Soft Now with 3 & 40 mm sizes! SYSTEM ANCHOR with Now designed to embolize 3 4 mm vessels! PACK with Coil Now with WAVE Shape and device
3 VOL UME A DVA N TAGE S OF T NE S S A DVA N TAGE a EMBOLIZ ATION SYS T EM CA SE S Splenic Artery Embolization Left Subclavian Embolization 8-system competitor coil in glass model c Bilateral Iliac Embolization Coil 4 Concerto Coils 4 Penumbra Coils % Dr. Frank Arko Dr. Herbert Cordero Evanston Hospital, IL Atrium Health, NC St. Rose Dominican Siena Campus, NV Pelvic Congestion Syndrome Hypogastric Artery Embolization Hepatic Artery Aneurysm % Dr. Thomas Aquisto 30 large volume soft coil in glass model d 30 Conventional Detachable Coil Soft Coils embolize collateral Soft coils pack, forming dense metal occlusion. & Coils c. Terumo AZUR CX Coil 4 mm x delivered through Terumo Progreat Microcatheter into 4 mm glass tube. d. 30 Coil delivered through Penumbra LANTERN High-Flow Microcatheter into 4 mm glass tube. a. 7. mm glass aneurysm. Tests performed and data on file at Penumbra, Inc. Bench test results may not be indicative of clinical performance. L A N T ERN MICROCAT HE T ER O N LY C O I L TO P ROV I D E 0 C M L EN G T H A N D.0 0" T H I C K N E S S Dr. Parag J. Patel Dr. Corey Teigen Dr. James Benenati Medical College of Wisconsin, WI Sanford Health, ND Miami Cardiac and Vascular Institute, FL Pre Fontan Embolization Pulmonary AVM GDA & Gastric Embolization P R EC I SE C O I L D E TAC H M EN T Coil Thickness LANTERN Microcatheter 0.0" Max. Ruby and are the only coils designed to be delivered through a LANTERN High-Flow Microcatheter. 03 diagnostic catheter Coil is not fully deployed (coil is still within the catheter tip) Coil fully deployed and ready to be detached 0 Coils T I P SH A P E S Conventional 3 Coil deployed from LANTERN High-Flow Microcatheter LANTERN Coil Proximal catheter marker 30º.0" Coil alignment marker 40 Max. Conventional 8 Coil 8 microcatheter b. Based on 8 mm coil. 30 b Max..0" Ruby and are fully retractable and resheathable, enabling user to achieve ideal placement in the target vessel. Coil alignment marker passes proximal catheter marker to form T, indicating that Ruby or can now be detached. Dr. Saar Danon Dr. Mahmood Razavi Dr. Dmitri Samoilov Cardinal Glennon Children s Hospital, MO St. Joseph Hospital, CA Medical Center Radiologists, VA
4 VOL UME A DVA N TAGE S OF T NE S S A DVA N TAGE a EMBOLIZ ATION SYS T EM CA SE S Splenic Artery Embolization Left Subclavian Embolization 8-system competitor coil in glass model c Bilateral Iliac Embolization Coil 4 Concerto Coils 4 Penumbra Coils % Dr. Frank Arko Dr. Herbert Cordero Evanston Hospital, IL Atrium Health, NC St. Rose Dominican Siena Campus, NV Pelvic Congestion Syndrome Hypogastric Artery Embolization Hepatic Artery Aneurysm % Dr. Thomas Aquisto 30 large volume soft coil in glass model d 30 Conventional Detachable Coil Soft Coils embolize collateral Soft coils pack, forming dense metal occlusion. & Coils c. Terumo AZUR CX Coil 4 mm x delivered through Terumo Progreat Microcatheter into 4 mm glass tube. d. 30 Coil delivered through Penumbra LANTERN High-Flow Microcatheter into 4 mm glass tube. a. 7. mm glass aneurysm. Tests performed and data on file at Penumbra, Inc. Bench test results may not be indicative of clinical performance. L A N T ERN MICROCAT HE T ER O N LY C O I L TO P ROV I D E 0 C M L EN G T H A N D.0 0" T H I C K N E S S Dr. Parag J. Patel Dr. Corey Teigen Dr. James Benenati Medical College of Wisconsin, WI Sanford Health, ND Miami Cardiac and Vascular Institute, FL Pre Fontan Embolization Pulmonary AVM GDA & Gastric Embolization P R EC I SE C O I L D E TAC H M EN T Coil Thickness LANTERN Microcatheter 0.0" Max. Ruby and are the only coils designed to be delivered through a LANTERN High-Flow Microcatheter. 03 diagnostic catheter Coil is not fully deployed (coil is still within the catheter tip) Coil fully deployed and ready to be detached 0 Coils T I P SH A P E S Conventional 3 Coil deployed from LANTERN High-Flow Microcatheter LANTERN Coil Proximal catheter marker 30º.0" Coil alignment marker 40 Max. Conventional 8 Coil 8 microcatheter b. Based on 8 mm coil. 30 b Max..0" Ruby and are fully retractable and resheathable, enabling user to achieve ideal placement in the target vessel. Coil alignment marker passes proximal catheter marker to form T, indicating that Ruby or can now be detached. Dr. Saar Danon Dr. Mahmood Razavi Dr. Dmitri Samoilov Cardinal Glennon Children s Hospital, MO St. Joseph Hospital, CA Medical Center Radiologists, VA
5 ORDERING INFORMATION COMPLEX STANDARD Frame Diameter Diameter (mm) RBYC030 RBYC03 3 RBYC03 RBYC040 0 RBYC04 4 RBYC043 3 RBYC0 RBYC RBYC0 RBYC RBYC07 7 RBYC08 8 RBYC RBYC RBYC 0 RBYC RBYC0 0 RBYC RBYC0 0 RBYC RBYC RBYC3 3 0 RBYC30 Now! 3 0 RBYC400 Now! 40 0 High-Flow Vessel Sacrifice RUBY COIL SYSTEM COMPLEX SOF T Fill.0 in (. mm) Diameter (mm) RBY4C0 RBY4C0 RBY4C04 4 RBY4C030 3 RBY4C03 RBY4C040 4 RBY4C04 RBY4C0 RBY4C RBY4C RBY4C080 0 RBY4C0 0 RBY4C0 0 DETACHMENT HANDLE Description RH Detachment Handle PACKING COIL Pack Behind Ruby or Backstop The System is indicated for arterial and venous embolizations in the peripheral vasculature. The System should only be used by physicians who have received appropriate training in interventional techniques. The device is intended for single use only. Do not resterilize or reuse. Resterilization and/or reuse may compromise the structural integrity of the device or increase the risk of contamination or infection leading to device failure and/or cross-infection and potential patient injury, illness or death. Do not use kinked or damaged devices. Do not use opened or damaged packages. Return all damaged devices and packaging to the manufacturer/distributor. Use device in conjunction with fluoroscopic guidance. Do not advance or withdraw the device against resistance without careful assessment of the cause using fluoroscopy. For Coils with nominal sizes mm The System is indicated for the embolization of: Intracranial aneurysms Other neurovascular abnormalities such as arteriovenous malformations and arteriovenous fistulae Arterial and venous embolizations in the peripheral vasculature For Coils with nominal sizes > mm The System is indicated for arterial and venous embolizations in the peripheral vasculature The System should only be used by physicians who have received appropriate training in interventional techniques. The device is intended for single use only. Do not resterilize or reuse. Resterilization and/or reuse may compromise the structural integrity of the device or increase the risk of contamination or infection leading to device failure and/or cross-infection and potential patient injury, illness or death. Do not use kinked or damaged devices. Do not use opened or damaged packages. Return all damaged devices and packaging to the manufacturer/distributor. Moving or torquing the device against resistance may result in damage to the vessel or device. Maintain a constant infusion of an appropriate flush solution. Possible complications include but are not limited to: acute occlusion; air embolism; allergic reaction and anaphylaxis from contrast media; aneurysm rupture; arteriovenous fistula; coagulopathy; coil herniation into parent vessel; death; device malfunction; distal embolization; emboli; embolic stroke and other cerebral ischemic events; false aneurysm formation; hematoma or hemorrhage at access site of entry; incomplete aneurysm occlusion; infection; intima dissection; intracranial hemorrhage; ischemia; myocardial infarction; neurological deficits including stroke; parent artery occlusion; peripheral thromboembolic events; post-embolization syndrome; premature device detachment; recanalization; renal failure; respiratory failure; revascularization; thromboembolic episodes; vessel spasm, thrombosis, dissection, or perforation. Use device in conjunction with fluoroscopic guidance. Do not advance or retract the device against resistance without careful assessment of the cause using fluoroscopy. If cannot be advanced or retracted, withdraw the device as a unit with the microcatheter. Moving or torquing the device against resistance may result in damage to the vessel or device. Maintain a constant infusion of an appropriate flush solution. Potential complications include but are not limited to: acute occlusion; air embolism; allergic reaction and anaphylaxis from contrast media; coagulopathy; coil herniation into parent vessel; death; device malfunction; distal embolization; emboli; embolic stroke and other cerebral ischemic events; false aneurysm formation; hematoma or hemorrhage at access site of entry; incomplete aneurysm occlusion; infection; intima dissection; intracranial hemorrhage; ischemia; myocardial infarction; neurological deficits including stroke; parent artery occlusion; peripheral thromboembolic events; post-embolization syndrome; premature device detachment; recanalization; renal failure; respiratory failure; revascularization; thromboembolic episodes; vessel spasm, thrombosis, dissection, or perforation. Target Vessel 7 Product Target Vessel (mm) RBY3 Now! 3 3 RBY RBY 4 30 RBY 0 RBY RBY0 Now! RBY Now! 0 0 RBY4 Now! Product RBYJ Now! Coil J-Soft RBYJ Coil J-Soft RBYJ30 Coil J-Soft RBYJ4 Coil J-Soft 4 4 RBYJ0 Coil J-Soft 0 0 LANTERN Delivery Microcatheter The Penumbra Delivery Microcatheters are intended to assist in the delivery of diagnostic agents, such as contrast media, and therapeutic devices, such as occlusion coils to the peripheral and neuro vasculature. The Penumbra Delivery Microcatheters should only be used by physicians who have received appropriate training in interventional techniques. The devices are intended for single use only. Do not resterilize or reuse. Resterilization and/or reuse may result in ineffective catheter coating lubrication, which may result in high friction and the inability to access the target location. Do not use kinked or damaged devices. Do not use open or damaged packages. Return all damaged devices and packaging to the manufacturer / distributor. Use the Penumbra Delivery Microcatheters in conjunction with fluoroscopic visualization. Do not advance or withdraw the Penumbra Delivery Microcatheters against resistance without careful assessment of the cause using fluoroscopy. If the cause cannot be determined, withdraw the device. Moving or torquing the device against resistance may result in damage to the vessel or device. Maintain a constant infusion of an appropriate flush solution. If flow through the device becomes restricted, do not attempt to clear the lumen by infusion. Remove and replace the device. Possible complications include, but are not limited to, the following: acute occlusion; hematoma or hemorrhage at access site; death; intracranial hemorrhage; hemorrhage; infection (at access site); distal embolization; ischemia (cardiac and/or cerebral); embolus (air, foreign body, thrombus, plaque); aneurysm perforation; false aneurysm formation; neurological deficits including stroke; vessel spasm, thrombosis, dissection, perforation or rupture; air embolism; emboli. Tip Shape ID (in) PXSLIMLANSTR PXSLIMLANT4 PXSLIMLANT90 PXSLIMLAN3STR PXSLIMLAN3T4 PXSLIMLAN3T90 PXSLIMLAN0STR PXSLIMLAN0T4 PXSLIMLAN0T Penumbra, Inc. USA One Penumbra Place Alameda, CA 940 USA T F order@penumbrainc.com info@penumbrainc.com Caution: Federal (USA) law restricts these devices to sale by or on the order of a physician. Prior to use, please refer to the Instructions for Use for System, System, and Penumbra Delivery Microcatheters for complete product indications, contraindications, warnings, precautions, potential adverse events, and detailed instructions for use. Photographs taken by and on file at Penumbra, Inc. Tests performed and data on file at Penumbra, Inc. Bench test results may not be indicative of clinical performance. Renderings for illustrative purposes only. Individual results may vary depending on a variety of patient-specific attributes. Images used with permission. Consents on file at Penumbra, Inc. Please contact your local Penumbra representative for more information. Copyright 7 8 Penumbra, Inc. All rights reserved. The Penumbra P logo, Ruby,, and LANTERN are registered trademarks or trademarks of Penumbra, Inc. in the USA and other countries. All other trademarks are the property of their respective owners. 33, Rev. C 08/8 USA
System.
 System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
Ruby Coil. Large Volume Detachable Coils
 Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
Peripheral Mechanical Thrombectomy
 Peripheral Mechanical Thrombectomy Indigo System Catheters Indigo System Catheters 8 6 5 & D Arterial and venous clot in larger vessels where circumferential aspiration is desired Arterial clot in larger
Peripheral Mechanical Thrombectomy Indigo System Catheters Indigo System Catheters 8 6 5 & D Arterial and venous clot in larger vessels where circumferential aspiration is desired Arterial clot in larger
Single Pass MCA Revascularization with Trevo
 Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
 M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
Peripheral Mechanical Thrombectomy
 Peripheral Mechanical Thrombectomy Indigo System Catheters Indigo System Catheters 6 8 D & Arterial clot in larger arteries including SFA, popliteal, and viscerals Arterial and venous clot in larger vessels
Peripheral Mechanical Thrombectomy Indigo System Catheters Indigo System Catheters 6 8 D & Arterial clot in larger arteries including SFA, popliteal, and viscerals Arterial and venous clot in larger vessels
Peripheral Aneurysm Coiling Using Large Volume Ruby Coils Results from the ACE Study
 Peripheral Aneurysm Coiling Using Large Volume Ruby Coils Results from the ACE Study Corey Teigen, MD 1 1 Sanford Medical Center, Fargo, ND Blaise Baxter, MD 2 ; J. David Moskovitz, MD 3 ; Henry Moyle,
Peripheral Aneurysm Coiling Using Large Volume Ruby Coils Results from the ACE Study Corey Teigen, MD 1 1 Sanford Medical Center, Fargo, ND Blaise Baxter, MD 2 ; J. David Moskovitz, MD 3 ; Henry Moyle,
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
Penumbra, Inc. has been successful in bringing a
 Ruby Coil and POD System: A Coil Platform for Fast and Easy Embolization BY PARAG J. PATEL, MD, AND FRANK R. ARKO III, MD Penumbra, Inc. has been successful in bringing a complete embolization system to
Ruby Coil and POD System: A Coil Platform for Fast and Easy Embolization BY PARAG J. PATEL, MD, AND FRANK R. ARKO III, MD Penumbra, Inc. has been successful in bringing a complete embolization system to
DURABLE. CONSISTENT. SAFE. IN.PACT Admiral Drug-Coated Balloon
 DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
Talent Abdominal Stent Graft
 Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
RECOMMENDED INSTRUCTIONS FOR USE
 Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter
 Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use
 Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use DEVICE DESCRIPTION The Detachable Azur CX 35 Peripheral Coil System (Azur system) consists of a coil implant attached to a delivery system.
Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use DEVICE DESCRIPTION The Detachable Azur CX 35 Peripheral Coil System (Azur system) consists of a coil implant attached to a delivery system.
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM
 Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
REBEL. Platinum Chromium Coronary Stent System. Patient Information Guide
 REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
CLARIVEIN INFUSION CATHETER
 CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
Instructions for Use 1
 Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER
 Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
1 Comparison of Warfarin and Aspirin for Symptomatic Intracranial Arterial Stenosis. N Engl J Med 352;13, March 31, 2005
 The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
FROM THE EVERYDAY TO THE EXTRAORDINARY
 FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System
 100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
Advanced Innovation for Exceptional Performance
 Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
The New World of (Micro)Plugs. Jafar Golzarian Professor of Radiology and Surgery University of Minnesota Medical Center
 The New World of (Micro)Plugs Jafar Golzarian Professor of Radiology and Surgery University of Minnesota Medical Center Jafar Golzarian, M.D. Stock: Embomedics Speakers Bureau: Boston Scientific, AngioDynamics,
The New World of (Micro)Plugs Jafar Golzarian Professor of Radiology and Surgery University of Minnesota Medical Center Jafar Golzarian, M.D. Stock: Embomedics Speakers Bureau: Boston Scientific, AngioDynamics,
BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ
 BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ 1 Case 1 78 yr. old female Rutherford Class II/III lesion 1 block claudicant 2 Pre Treatment Post Treatment Anterior Tibial Artery Occlusion
BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ 1 Case 1 78 yr. old female Rutherford Class II/III lesion 1 block claudicant 2 Pre Treatment Post Treatment Anterior Tibial Artery Occlusion
As data on coil embolization
 A Complete Embolization System The Ruby coil and POD devices. As data on coil embolization become more A robust, so must the tools and technology we use. Penumbra, Inc. has advanced this technology with
A Complete Embolization System The Ruby coil and POD devices. As data on coil embolization become more A robust, so must the tools and technology we use. Penumbra, Inc. has advanced this technology with
JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW
 JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW DISCOVER THE VALUE OF VERSATILITY Versatility means not having to guess the morphology! Peripheral arterial lesions can present with
JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW DISCOVER THE VALUE OF VERSATILITY Versatility means not having to guess the morphology! Peripheral arterial lesions can present with
Understanding aneurysms and flow diversion treatment
 Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Solitaire FR Revascularization Device
 Solitaire FR Revascularization Device FEATURING PARAMETRIC DESIGN Restore. Retrieve. Revive. 5 Revolutionizing Mechanical Thrombectomy with Parametric Design Solitaire FR Device The Solitaire FR revascularization
Solitaire FR Revascularization Device FEATURING PARAMETRIC DESIGN Restore. Retrieve. Revive. 5 Revolutionizing Mechanical Thrombectomy with Parametric Design Solitaire FR Device The Solitaire FR revascularization
Indications. The AngioVac cannula is intended for use as a venous drainage cannula and for the removal of fresh, soft thrombi or emboli
 Indications Straight Cannula or Cannula with 20 O Angle 17F Working Channel The AngioVac cannula is intended for use as a venous drainage cannula and for the removal of fresh, soft thrombi or emboli Radiopaque
Indications Straight Cannula or Cannula with 20 O Angle 17F Working Channel The AngioVac cannula is intended for use as a venous drainage cannula and for the removal of fresh, soft thrombi or emboli Radiopaque
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU)
 Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
ASAHI Corsair INSTRUCTIONS FOR USE
 ASAHI Corsair INSTRUCTIONS FOR USE SYMBOLS Legal manufacturer Do not use if package is damaged GC Minimum GC I.D. REF Catalogue number Caution, consult accompanying documents Maximum injection pressure
ASAHI Corsair INSTRUCTIONS FOR USE SYMBOLS Legal manufacturer Do not use if package is damaged GC Minimum GC I.D. REF Catalogue number Caution, consult accompanying documents Maximum injection pressure
Neuroform EZ Stent System
 Neuroform EZ Stent System Directions for Use 2 Neuroform EZ Stent System 1 cm 1.9 cm 135 cm 185 cm ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning
Neuroform EZ Stent System Directions for Use 2 Neuroform EZ Stent System 1 cm 1.9 cm 135 cm 185 cm ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp
 Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Chronic Total Occlusion (CTO) Technologies
 to receive our latest news and key activities. Chronic Total Occlusion (CTO) Technologies Re-open vital channels LinkedIn page Follow us on CORDIS EMEA OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry
to receive our latest news and key activities. Chronic Total Occlusion (CTO) Technologies Re-open vital channels LinkedIn page Follow us on CORDIS EMEA OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry
ROTABLATORTM. Peripheral. Rotational Atherectomy System. Quick Reference Cards
 Peripheral ROTABLATORTM Rotational Atherectomy System Quick Reference Cards Technical Assistance: 1.800.949.6708 Customer Service: 1.888.272.1001 Complaints: 1.800.811.3211 Setup Video: www.bostonscientific.com/rotablatorsetup
Peripheral ROTABLATORTM Rotational Atherectomy System Quick Reference Cards Technical Assistance: 1.800.949.6708 Customer Service: 1.888.272.1001 Complaints: 1.800.811.3211 Setup Video: www.bostonscientific.com/rotablatorsetup
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
Peel-Apart Percutaneous Introducer Kits for
 Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
DISRUPT PAD. (( Data Summary )) DISRUPT PAD Data Summary SPL Rev. B 2016 Shockwave Medical Inc. All rights reserved.
 DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
LeMaitre Embolectomy Catheter
 Instructions for Use - English Lemaitre Embolectomy Catheter (Model Numbers 1601-24, e1601-24, 1601-26, e1601-26, 1601-28, e1601-28, 1601-34, e1601-34, 1601-38, e1601-38, 1601-44, e1601-44, 1601-48, e1601-48,
Instructions for Use - English Lemaitre Embolectomy Catheter (Model Numbers 1601-24, e1601-24, 1601-26, e1601-26, 1601-28, e1601-28, 1601-34, e1601-34, 1601-38, e1601-38, 1601-44, e1601-44, 1601-48, e1601-48,
Coils and plugs have emerged as the most commonly
 The MVP Micro Vascular Plug: A New Paradigm in Peripheral Embolization BY RIPAL T. GANDHI, MD, FSVM; BRIAN E. SCHIRF, MD; AND JONATHAN IGLESIAS, MD Coils and plugs have emerged as the most commonly employed
The MVP Micro Vascular Plug: A New Paradigm in Peripheral Embolization BY RIPAL T. GANDHI, MD, FSVM; BRIAN E. SCHIRF, MD; AND JONATHAN IGLESIAS, MD Coils and plugs have emerged as the most commonly employed
Balloon in Balloon (BIB )
 in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
The Continuing Role of Pushable Coils: Advances and Fiscal Responsibilities JAMES F BENENATI MD
 The Continuing Role of Pushable Coils: Advances and Fiscal Responsibilities JAMES F BENENATI MD James Benenati, MD Royalty: Penumbra Stock: Covidien Consultant/Advisory Board: Penumbra, Endoshape, Scientia
The Continuing Role of Pushable Coils: Advances and Fiscal Responsibilities JAMES F BENENATI MD James Benenati, MD Royalty: Penumbra Stock: Covidien Consultant/Advisory Board: Penumbra, Endoshape, Scientia
Figure 1: Revolution TM Peripheral Atherectomy System Diagram. Table 1: Revolution TM Peripheral Atherectomy System Specifications Minimum Burr
 Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
Peripheral Embolization Products
 Peripheral Embolization Products PRODUCT CATALOG AND ORDERING INFORMATION Setting Standards. Exceeding Expectations. BOSTON SCIENTIFIC Peripheral Embolization Solved Complete Offering Throughout our history,
Peripheral Embolization Products PRODUCT CATALOG AND ORDERING INFORMATION Setting Standards. Exceeding Expectations. BOSTON SCIENTIFIC Peripheral Embolization Solved Complete Offering Throughout our history,
New Stroke Interventions. Scott L. Zuckerman M.D. Vanderbilt Neurosurgery
 New Stroke Interventions Scott L. Zuckerman M.D. Vanderbilt Neurosurgery Agenda Clot Retrieval Devices Merci Penumbra Stent Retrievers Solitaire Trevo New Technology Funnel ReCover MERCI Retriever (2004)
New Stroke Interventions Scott L. Zuckerman M.D. Vanderbilt Neurosurgery Agenda Clot Retrieval Devices Merci Penumbra Stent Retrievers Solitaire Trevo New Technology Funnel ReCover MERCI Retriever (2004)
CAROTID ARTERY ANGIOPLASTY
 CAROTID ARTERY ANGIOPLASTY Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
CAROTID ARTERY ANGIOPLASTY Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES. Medtronic Further. Together
 DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
Directions For Use. All directions should be read before use
 Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
22F coil-reinforced cannula. Designed with a balloon actuated, expandable funnel shaped distal tip
 Indication The AngioVac* Cannula is intended for use as a venous drainage cannula for extracorporeal bypass for up to 6 hours. The cannula is also indicated for removal of soft, fresh thrombi or emboli
Indication The AngioVac* Cannula is intended for use as a venous drainage cannula for extracorporeal bypass for up to 6 hours. The cannula is also indicated for removal of soft, fresh thrombi or emboli
ENDOVASCULAR THERAPIES FOR ACUTE STROKE
 ENDOVASCULAR THERAPIES FOR ACUTE STROKE Cerebral Arteriogram Cerebral Anatomy Cerebral Anatomy Brain Imaging Acute Ischemic Stroke (AIS) Therapy Main goal is to restore blood flow and improve perfusion
ENDOVASCULAR THERAPIES FOR ACUTE STROKE Cerebral Arteriogram Cerebral Anatomy Cerebral Anatomy Brain Imaging Acute Ischemic Stroke (AIS) Therapy Main goal is to restore blood flow and improve perfusion
The Challenge of Accurately Stenting Aorto-Ostial Lesions
 The Challenge of Accurately Stenting Aorto-Ostial Lesions In a retrospective study of 100 patients in whom stents were placed using angiographic landmarks, correct stent positioning was achieved in only
The Challenge of Accurately Stenting Aorto-Ostial Lesions In a retrospective study of 100 patients in whom stents were placed using angiographic landmarks, correct stent positioning was achieved in only
Pocket Reference Guide For (CTO) Technologies
 to receive our latest news and key activities. Pocket Reference Guide For (CTO) Technologies LinkedIn page Follow us on CORDIS EMEA Table of Contents Essential prescribing information for both FRONTRUNNER
to receive our latest news and key activities. Pocket Reference Guide For (CTO) Technologies LinkedIn page Follow us on CORDIS EMEA Table of Contents Essential prescribing information for both FRONTRUNNER
Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
 Page 1 of 5 Reprocessed by Instructions for Use Reprocessed Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
Page 1 of 5 Reprocessed by Instructions for Use Reprocessed Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
Innovation by design. Technology that sets a new standard
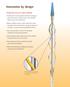 Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
ARROW ENDURANCE. Extended Dwell Peripheral Catheter System. Rx only.
 ARROW ENDURANCE Extended Dwell Peripheral Catheter System Rx only. Product Description: The ARROW Endurance catheter system is a sterile, single use peripheral intravascular device designed to permit access
ARROW ENDURANCE Extended Dwell Peripheral Catheter System Rx only. Product Description: The ARROW Endurance catheter system is a sterile, single use peripheral intravascular device designed to permit access
Per-Q-Cath* PICC Catheters with Excalibur Introducer* System
 Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
CY2015 Hospital Outpatient: Endovascular Procedure APCs and Complexity Adjustments
 CY2015 Hospital Outpatient: Endovascular Procedure APCs Complexity Adjustments Comprehensive Ambulatory Payment Classifications (c-apcs) CMS finalized the implementation of 25 Comprehensive APC to further
CY2015 Hospital Outpatient: Endovascular Procedure APCs Complexity Adjustments Comprehensive Ambulatory Payment Classifications (c-apcs) CMS finalized the implementation of 25 Comprehensive APC to further
Advancing Lives and the Delivery of Health Care. The High-Flow Port Designed for Apheresis
 Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
Pipeline Embolization Device
 Pipeline Embolization Device The power to redefine aneurysm treatment. REDEFINE The Pipeline device redefines treatment for large or giant wide-necked aneurysms by reconstructing the parent artery and
Pipeline Embolization Device The power to redefine aneurysm treatment. REDEFINE The Pipeline device redefines treatment for large or giant wide-necked aneurysms by reconstructing the parent artery and
Sustained Release. Superior Results.
 ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
Repair of Intracranial Vessel Perforation with Onyx-18 Using an Exovascular Retreating Catheter Technique
 Repair of Intracranial Vessel Perforation with Onyx-18 Using an Exovascular Retreating Catheter Technique Michael Horowitz M.D. Pittsburgh, Pennsylvania Background Iatrogenic intraprocedural rupture rates
Repair of Intracranial Vessel Perforation with Onyx-18 Using an Exovascular Retreating Catheter Technique Michael Horowitz M.D. Pittsburgh, Pennsylvania Background Iatrogenic intraprocedural rupture rates
Navigate Complex Interventions at Every Turn
 Navigate Complex Interventions at Every Turn Keep Driving Forward The portfolio of Catheters places the power in your hands. Navigate tortuous anatomy with agility and precision. No matter the challenge,
Navigate Complex Interventions at Every Turn Keep Driving Forward The portfolio of Catheters places the power in your hands. Navigate tortuous anatomy with agility and precision. No matter the challenge,
NEUROVASCULAR 2018 PRICE LIST
 NEUROVASCULAR 2018 PRICE LIST TABLE OF CONTENTS GUIDEWIRES Avígo.014" Hydrophilic Guidewire...1 Mirage.008" Hydrophilic Guidewire...1 SilverSpeed Hydrophilic Guidewire...1 X-Pedion Hydrophilic Guidewire...1
NEUROVASCULAR 2018 PRICE LIST TABLE OF CONTENTS GUIDEWIRES Avígo.014" Hydrophilic Guidewire...1 Mirage.008" Hydrophilic Guidewire...1 SilverSpeed Hydrophilic Guidewire...1 X-Pedion Hydrophilic Guidewire...1
Interventional Radiology in Trauma. Vikash Prasad, MD, FRCPC Vascular and Interventional Radiology The Moncton Hospital
 Interventional Radiology in Trauma Vikash Prasad, MD, FRCPC Vascular and Interventional Radiology The Moncton Hospital Disclosures None relevant to this presentation Shareholder Johnson and Johnson Goal
Interventional Radiology in Trauma Vikash Prasad, MD, FRCPC Vascular and Interventional Radiology The Moncton Hospital Disclosures None relevant to this presentation Shareholder Johnson and Johnson Goal
RUBY. Large Volume Detachable Coil. Copyright 2014 Penumbra, Inc. All rights reserved. 7516, Rev.A
 RUBY TM Large Volume Detachable Coil TM Copyright 2014 Penumbra, Inc. All rights reserved 7516, Rev.A RUBY NEW SOLUTIONS IN PERIPHERAL THERAPY THE PERFECT DEVICE FOR VISCERAL ANEURYSMS Lorenzo Paolo Moramarco,
RUBY TM Large Volume Detachable Coil TM Copyright 2014 Penumbra, Inc. All rights reserved 7516, Rev.A RUBY NEW SOLUTIONS IN PERIPHERAL THERAPY THE PERFECT DEVICE FOR VISCERAL ANEURYSMS Lorenzo Paolo Moramarco,
The Importance of Coil Packing
 The Importance of Coil Packing Keigo Osuga, MD, PhD Department of Diagnostic and Interventional Radiology, Osaka University Graduate School of Medicine Keigo Osuga, M.D., Ph.D. No relevant financial relationship
The Importance of Coil Packing Keigo Osuga, MD, PhD Department of Diagnostic and Interventional Radiology, Osaka University Graduate School of Medicine Keigo Osuga, M.D., Ph.D. No relevant financial relationship
Chronic Total Occlusion (CTO) Technologies. Re-open vital channels
 Chronic Total Occlusion (CTO) Technologies Re-open vital channels OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry Technology Get back into the true lumen with ease and precision There are a number of
Chronic Total Occlusion (CTO) Technologies Re-open vital channels OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry Technology Get back into the true lumen with ease and precision There are a number of
NAVIGATE DISTAL AND TORTUOUS ANATOMY
 PROGREAT ALPHA PUSHINGBOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging cases. We are relentlessly
PROGREAT ALPHA PUSHINGBOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging cases. We are relentlessly
Optimized for Performance and Ease of Use. Designed with Titanium Oxide Surface. References
 References MicroVention data on file low Re-direction Endoluminal Device in treatment of cerebral aneurysms: initial experience with short-term follow-up results, Author: Naci Kocer et al. The RED and
References MicroVention data on file low Re-direction Endoluminal Device in treatment of cerebral aneurysms: initial experience with short-term follow-up results, Author: Naci Kocer et al. The RED and
GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER Section I Clinical experience. Section II Worldwide commercial experience
 GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER 2018 Abstract This annual clinical update provides a review of the ongoing experience with the GORE EXCLUDER AAA Endoprosthesis used in the
GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER 2018 Abstract This annual clinical update provides a review of the ongoing experience with the GORE EXCLUDER AAA Endoprosthesis used in the
Watchman. Left Atrial Appendage Closure Device. Uniquely engineered for the LAA 1-3 with proven safety and longterm efficacy. 4-8
 TM Watchman Left Atrial Appendage Closure Device PROOF OFLEADERSHIP Uniquely engineered for the LAA 1-3 with proven safety and longterm efficacy. 4-8 Patients with AF have a 5x increased risk of stroke.
TM Watchman Left Atrial Appendage Closure Device PROOF OFLEADERSHIP Uniquely engineered for the LAA 1-3 with proven safety and longterm efficacy. 4-8 Patients with AF have a 5x increased risk of stroke.
CY2017 Hospital Outpatient: Vascular Procedure APCs and Complexity Adjustments
 CY2017 Hospital Outpatient: Vascular Procedure APCs and Complexity Adjustments Comprehensive Ambulatory Payment Classifications (c-apcs) In CY2015 and in an effort to help pay providers for quality, not
CY2017 Hospital Outpatient: Vascular Procedure APCs and Complexity Adjustments Comprehensive Ambulatory Payment Classifications (c-apcs) In CY2015 and in an effort to help pay providers for quality, not
Every heartbeat is precious. Thanks to you, he ll have 100 million more.
 Every heartbeat is precious. Thanks to you, he ll have 100 million more. Master the Complex Thrombectomy Portfolio FETCH 2 Aspiration Catheter ANGIOJET Ultra Thrombectomy System EVERY HEARTBEAT IS PRECIOUS
Every heartbeat is precious. Thanks to you, he ll have 100 million more. Master the Complex Thrombectomy Portfolio FETCH 2 Aspiration Catheter ANGIOJET Ultra Thrombectomy System EVERY HEARTBEAT IS PRECIOUS
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Cardiva Catalyst III INSTRUCTIONS FOR USE
 Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Usefulness of Coil-assisted Technique in Treating Wide-neck Intracranial Aneurysms: Neck-bridge Procedure Using the Coil Mass as a Support
 Journal of Neuroendovascular Therapy 2017; 11: 220 225 Online December 14, 2016 DOI: 10.5797/jnet.tn.2016-0081 Usefulness of Coil-assisted Technique in Treating Wide-neck Intracranial Aneurysms: Neck-bridge
Journal of Neuroendovascular Therapy 2017; 11: 220 225 Online December 14, 2016 DOI: 10.5797/jnet.tn.2016-0081 Usefulness of Coil-assisted Technique in Treating Wide-neck Intracranial Aneurysms: Neck-bridge
Treating Pulmonary Arteriovenous Malformations: What to Do and What Not to Do
 Baptist Health South Florida Scholarly Commons @ Baptist Health South Florida All Publications 2016 Treating Pulmonary Arteriovenous Malformations: What to Do and What Not to Do Ripal Gandhi Baptist Hospital
Baptist Health South Florida Scholarly Commons @ Baptist Health South Florida All Publications 2016 Treating Pulmonary Arteriovenous Malformations: What to Do and What Not to Do Ripal Gandhi Baptist Hospital
The Endurant Stent Graft System
 The Stent Graft System Instructions for Use (IFU) IMPORTANT! Do not attempt to use the Stent Graft System before completely reading and understanding the information contained in the Instructions for Use.
The Stent Graft System Instructions for Use (IFU) IMPORTANT! Do not attempt to use the Stent Graft System before completely reading and understanding the information contained in the Instructions for Use.
Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters
 Reprocessed by Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a
Reprocessed by Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a
2019 ABBOTT REIMBURSEMENT GUIDE CMS Physician Fee Schedule
 ABBOTT REIMBURSEMENT GUIDE CMS Physician Fee Schedule This document and the information contained herein is for general information purposes only and is not intended and does not constitute legal, reimbursement,
ABBOTT REIMBURSEMENT GUIDE CMS Physician Fee Schedule This document and the information contained herein is for general information purposes only and is not intended and does not constitute legal, reimbursement,
The Art of. Flow Restoration. Ikazuchi Zero. Semi-Compliant PTCA Balloon OVERVIEW CROSSABILITY LOW PROFILE CASE STUDY CODES
 The Art of Flow Restoration Japan s Recognized PCI Technology For Both Everyday and Complex CTO Cases* Manufactured by Kaneka Corporation, one of Japan s leading providers of interventional devices. Now
The Art of Flow Restoration Japan s Recognized PCI Technology For Both Everyday and Complex CTO Cases* Manufactured by Kaneka Corporation, one of Japan s leading providers of interventional devices. Now
ULTRA ICE PLUS ULTRASOUND IMAGING CATHETER. Know where you are. See what you want to avoid.
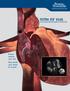 ULTRA ICE PLUS ULTRASOUND IMAGING CATHETER Know where you are. See what you want to avoid. ULTRA ICE PLUS for Transseptal Puncture Know where you are. See what you want to avoid. Fluoroscopy alone provides
ULTRA ICE PLUS ULTRASOUND IMAGING CATHETER Know where you are. See what you want to avoid. ULTRA ICE PLUS for Transseptal Puncture Know where you are. See what you want to avoid. Fluoroscopy alone provides
NIRxcell CoCr Coronary Stent System
 cordis.com/nirxcell NIRxcell CoCr Coronary Stent System Exceptional Outcomes by Design Exceptionally low TLR rate of 5.1 % at 9 months 1 Superior Deliverability and Crossability 2 Best-in-class Conformability
cordis.com/nirxcell NIRxcell CoCr Coronary Stent System Exceptional Outcomes by Design Exceptionally low TLR rate of 5.1 % at 9 months 1 Superior Deliverability and Crossability 2 Best-in-class Conformability
Case Report 1. CTA head. (c) Tele3D Advantage, LLC
 Case Report 1 CTA head 1 History 82 YEAR OLD woman with signs and symptoms of increased intra cranial pressure in setting of SAH. CT Brain was performed followed by CT Angiography of head. 2 CT brain Extensive
Case Report 1 CTA head 1 History 82 YEAR OLD woman with signs and symptoms of increased intra cranial pressure in setting of SAH. CT Brain was performed followed by CT Angiography of head. 2 CT brain Extensive
INSTRUCTIONS FOR USE
 PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent
 Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion
Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion
INSTRUCTIONS FOR USE FOR:
 INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
SEE IT. BELIEVE IT. THE CONFIDENCE OF CLARITY. Solitaire Platinum. Revascularization Device
 SEE IT. BELIEVE IT. THE CONFIDENCE OF CLARITY. Revascularization THE CONFIDENCE OF CLARITY. The key features that make the device effective have been retained including our unique Parametric overlapping
SEE IT. BELIEVE IT. THE CONFIDENCE OF CLARITY. Revascularization THE CONFIDENCE OF CLARITY. The key features that make the device effective have been retained including our unique Parametric overlapping
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE
 CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE INDICATIONS FOR USE The LATERA Absorbable Nasal Implant is indicated for supporting upper and lower lateral nasal cartilage. CAUTION: Federal law restricts
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE INDICATIONS FOR USE The LATERA Absorbable Nasal Implant is indicated for supporting upper and lower lateral nasal cartilage. CAUTION: Federal law restricts
AERO DV Tracheobronchial Direct Visualization Stent System
 AERO DV Tracheobronchial Direct Visualization Stent System Review Instructions For Use Before Using This System. Single Use Only Non-sterile MR Conditional CONTENTS Instructions for Use...............................
AERO DV Tracheobronchial Direct Visualization Stent System Review Instructions For Use Before Using This System. Single Use Only Non-sterile MR Conditional CONTENTS Instructions for Use...............................
Management of Acute DVT Extending From the Tibial Veins to the Common Iliac Vein Using the AngioJet Thrombectomy System
 FEATURED TECHNOLOGY: ANGIOJET THROMBECTOMY SYSTEM Sponsored by Boston Scientific Corporation Management of Acute DVT Extending From the Tibial Veins to the Common Iliac Vein Using the AngioJet Thrombectomy
FEATURED TECHNOLOGY: ANGIOJET THROMBECTOMY SYSTEM Sponsored by Boston Scientific Corporation Management of Acute DVT Extending From the Tibial Veins to the Common Iliac Vein Using the AngioJet Thrombectomy
Exceptional Performance and Ease of Placement
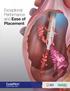 Exceptional Performance and Ease of Placement S IMU LATED FLO W PERFO RMANC E Higher Flow Rates Higher is Better GLIDEPATH catheters demonstrated on average in forward Precision catheters Lower Pressures
Exceptional Performance and Ease of Placement S IMU LATED FLO W PERFO RMANC E Higher Flow Rates Higher is Better GLIDEPATH catheters demonstrated on average in forward Precision catheters Lower Pressures
Quality Metrics. Stroke Related Procedure Outcomes
 Quality Metrics Stroke Related Procedure Outcomes Below is a description of some of the stroke-related procedures performed at St. Dominic Hospital in Jackson, with quality information on the complication
Quality Metrics Stroke Related Procedure Outcomes Below is a description of some of the stroke-related procedures performed at St. Dominic Hospital in Jackson, with quality information on the complication
2018 Diagnosis Coding Fact Sheet
 The information contained in this document is provided for informational purposes only and represents no statement, promise, or guarantee by Cordis Corporation concerning levels of reimbursement, payment,
The information contained in this document is provided for informational purposes only and represents no statement, promise, or guarantee by Cordis Corporation concerning levels of reimbursement, payment,
