SOCIETA ITALIANA DI MEDICINA FELINA
|
|
|
- Amberly Reynolds
- 5 years ago
- Views:
Transcription
1 SOCIETA ITALIANA DI MEDICINA FELINA... CREMONA, PALAZZO TRECCHI 9 NOVEMBRE 2014 ATTI
2 SCIVAC Feline Emergency Medicine Course KATE MURPHY BVSC (HONS) DSAM DIP ECVIM-CA MRCVS PGCERT(HE) European Veterinary Specialist in Small Animal Internal Medicine Clinical Lead Highcroft Veterinary Referrals 615 Wells Road Whitchurch Bristol BS149BE Tel :00-10:00 Triage and the unique challenges of the cat 10:00-11:00 Rational approach to fluid therapy and blood transfusions 11:00-11:30 Coffee Break 11: Shock, toxins and CPR :00 Nutritional support of the critically ill cat 13:00-14:00 Lunch Break 14:00-15:00 The dyspnoeic cat (airway disease, parenchymal disease, pleural space disease and cardiac disease) 15:00-16:00 The feline acute abdomen not always so obvious 16:00-16:30 Coffee Break 16: The acute kidney injury cat and other urological emergencies :00 The pale cat 18:00-18:30 Questions? 18:30 end of the day
3 TRIAGE & THE UNIQUE CHALLENGES OF THE CAT Triage is the evaluation and treatment of patients according to a system of priorities. The aim is to identify the critical or unstable patients and to manage their immediate problems before performing a full survey of them or before moving on to the assessment and stabilisation of other patients. Cats are particularly challenging as their signs are often subtle and additional reasons (see table 1) include: Smaller blood volume Response to shock do not tend to get such obviously bounding pulses, more often bradycardic than tachycardic Blood types Mucous membranes normally less pink than dog and when hyperaemic are less obvious An owner may feel that the cat has only become acutely unwell however in reality the disease has been developing gradually but because cats are more subtle in the signs shown than dogs the early phases can be missed. Triage can be performed in different ways; it might be over the telephone, in the waiting area or in a hospital ward. What is always important is to remain calm, logical and to ensure you have asked the necessary questions and made some basic but important evaluation of the cat. Try to remove cats from baskets/blankets to allow better assessment but care with fractious cats. Establish: Nature of the injury Any cardiorespiratory signs Consciousness Presence of bleeding, fractures or severe wounds Any other significant signs If in doubt, always advise the owner to bring the cat for assessment. It can be important to give advice about safe transport of the patient to the clinic e.g. stabilise fractured limbs, transport road accident patients on a board (if they are not already up and walking around), apply direct pressure to bleeding area, keep wounds covered and clean etc. Although owners may want to help their cat it is best not to advise first aid treatment by the owner and to advise clients that their cat may behave differently due to the pain/fear and thus could become aggressive. Initial examination When the patient first arrives it is important to evaluate the major organ systems starting with an ABC (airway, breathing, circulation) approach and obtain a brief history. Age/sex/breed Presenting complaint Last seen normal Last ate/drank/urinated Vomiting/regurgitating/diarrhoea/coughing Toxins/medications/trauma Other conditions/medicationsns
4 It is important to concentrate on abnormalities which might be life threatening to the cat and to stabilise them immediately: Respiratory o Rate, rhythm, effort o If there is any sign of respiratory compromise then consider oxygen supplementation +/- need for intubation / ventilation o Check spo2 o Inspiratory dyspnoea upper airway obstruction o Expiratory dyspnoea lower airway disease eg feline asthma o Rapid shallow breathing pleural space or parenchymal disease Cardiovascular o Mucous membrane colour, capillary refill time, pulse quality & rhythm, heart rate o Consider ECG if rhythm is not regular o Check blood pressure: pulse non-palpable when mean blood pressure <50mmHg. Neurological o Mentation, ability to ambulate, presence of seizure activity o If bradycardic, hypertensive, abnormal breathing pattern and deteriorating mentation consider elevated intracranial pressure Renal o Ability to urinate, ability to palpate urinary bladder Temperature o Active cooling if temperature >41C and active rewarming if temperature < 34.4C If cats have problems with these major organ systems then they should be immediately evaluated and treated. Conditions affecting other body systems are generally not life threatening and can be managed once the main priorities have been addressed. The primary survey builds upon information obtained during triage with repeat assessment of the cardiovascular, respiratory and neurological system and also can involve more detailed assessment e.g. measures of oxygenation, assessment of acid base / lactate to assess perfusion. The secondary survey includes a full physical examination, a detailed history, assessment of response to initial therapy and more in-depth diagnostics including clinical pathology and imaging. It is also important to compile a comprehensive diagnostic and therapeutic plan and to inform owners of the likely costs and the prognosis. Other aspects of triage and emergency care include obtaining vascular access, the emergency database and important therapeutic considerations. Vascular Access Vascular access can be obtained in emergency patients via standard intravenous approach e.g. cephalic, medial saphenous or occasionally jugular veins. Jugular access is contra-indicated if the patient is suspected to have coagulopathy or raised intracranial pressure. In the case of peripheral shut down, a cut-down procedure can be performed to allow visualisation and cannulation of the vessel or intraosseous (IO) access can be obtained. Intraosseous cannulas are useful for providing rapid vascular access in severely collapsed animals and in very small patients where intravenous cannulation can be challenging. Commercially available intraosseous cannulas are available (Global, via Dechra) or a spinal
5 needle with stylet (18-22G) or bone marrow needle can be used. There is also a power driver IO needled placement EZIO system from Mila. Most drugs and fluids which can be administered IV can be safely administered via the intraosseous route. The intraosseous needle can be placed in the trochanteric fossa of the femur, the greater tubercle of the humerus, the wing of the ilium and the tibial plateau. Emergency Database An emergency database will be tailored to the presentation of the case but might include: Packed cell volume and total solids o Providing useful assessment of hydration, presence of anaemia, hypoproteinaemia and in guiding choice of fluid therapy Urea Glucose o Hypoglycaemia Consider sepsis, storage diseases, severe hepatic dysfunction, insulin overdose, severe hypothermia, heat stroke (insulin secreting tumours, addisons, juvenile hypoglycaemia) o Hyperglycaemia Diabetes, stress, seizure activity Blood smear evaluation o Red cell morphology, approximate number and morphology platelets and white cells, any parasites or toxic changes? Electrolytes/ ions e.g. Na/K/Cl /Ca Arterial blood gas Patient-side coagulation tests Urinalysis particularly urine SG Treatment considerations Oxygen consider least stressful way of supplementing Fluid therapy if shocked/hypovolaemic use crystalloid bolus e.g. 5-10ml/kg over 10-20mins and repeat to effect to improve perfusion. Address maintenance needs and ongoing losses. Consider use of colloids if response to crystalloids is either short lived or insufficient. Analgesia opiates and no non-steroidals until patient is fully hydrated and there is no risk of organ damage from their use. Antibiotics use if wounds, compromised GI perfusion with risk of translocation etc Steroids no evidence of benefit unless given before the patient gets shocked Vasopressors/ inotropes used if perfusion cannot be maintained with fluid therapy alone Blood / blood products may be indicated in anaemic, hypovolaemic or coagulopathic patients. The diagnostic /therapeutic plan Generate a problem list and differential diagnosis list. Prioritise the list. Consider further diagnostic tests and treatment plans. Reassess the patient regularly. Ensure monitoring and nursing orders are clear and everyone works as a team, knowing their roles.
6 Table 1. (Modified from JFMS article) Feature Specific feline considerations Significance Shock Uncommon to see hyperdynamic compensatory phase of shock including tachycardia and snappy pulses. Hypothermia, hypotension and bradycardia/normal heart rate are more frequent features. Vascular volume Body surface area Shock organ Occult cardiac disease Susceptibility to volume overload with fluid therapy Stress hyperglycaemia Drug metabolism 66mls/kg versus 90mls/kg in dogs. Higher surface area: body mass ratio is a feature of small patients. Lungs in cats versus (gastrointestinal tract in dogs) Occult cardiomyopathy may be due to subclinical disease or the consequence of systemic illness e.g. hypertension, hyperthyroidism, severe anaemia Development of pulmonary oedema and pleural effusion is a common complication in cats secondary to excessive fluid administration. Cats are less tolerant of large fluid loads. Commonly detected in sick or stressed cats due to increased circulating catecholamine levels (e.g. 16mmol/l, 285 mg/dl in acute stress) Reduced capacity for hepatic glucuronidation and deficiency of thiopurine methyltransferase. Consider whether the heart rate in a collapsed or critically ill patient is appropriate. Assess all perfusion parameters together. Smaller boluses and total doses are used for resuscitative fluid therapy in shock (e.g. 5-10ml/kg bolus, 40-60ml/kg total dose for crystalloid fluids). Risk for rapid heat loss and less efficient compensatory heat production. Frequently re-evaluate the respiratory rate and pattern and reassess lung sounds in critically ill cats; tachypnoea, increased respiratory effort and pulmonary noises ( harsh lung sounds and/or pulmonary crackles) may indicate pulmonary complications of shock, SIRS or sepsis. Increased risk of volume overload and development of pulmonary oedema +/- pleural effusion following intravenous fluid therapy. Frequently reassess the heart and respiratory rate, heart rhythm, pulses and auscultate for heart murmurs, gallop sounds, arrhythmias and pulmonary crackles in critically ill cats. Risk factors include hypothermia (which blunts the vascular response to fluids in cats), occult cardiac disease, pre-existing pulmonary disease (e.g. aspiration pneumonia), SIRS/sepsis, renal failure, acidosis, hypocalcaemia, hypoalbuminaemia, administration of colloids especially haemoglobin-based oxygen-carrying solutions and severe anaemia. Recheck blood glucose levels following a period of stabilisation to determine if there is a persistent elevation; stress hyperglycaemia is typically transient. Reduced hepatic glucuronidation is the cause of paracetamol toxicity at 3-4 fold lower doses in cats versus dogs. Suspected aetiology of species-specific permethrin intolerance.
7 Anaemia Blood types Constant dietary protein requirement Hepatic lipidosis Pain manifestation Fear aggression Anaemia of chronic disease is common (typically a mild anaemia). Patients with chronic non-regenerative anaemia due to primary bone marrow disease frequently present with severely reduced PCVs (<10%). Cats have preformed blood type antibodies snd potent transfusion reactions Inability to down-regulate protein requirements. Risk of development in all cats (regardless of body condition) following even short periods of partial anorexia (<1week). Signs are more subtle including adopting a hunched body position, reduced palpebral fissure, starey coat, lack of interaction, lack of grooming, inappetance, reluctance to move and aggression. Fearful, stressed and painful cats can be challenging and even dangerous for personnel to handle, however fear aggression is an extreme response for a feline patient to mount. Always consider why is the cat demonstrating aggression? Assess PCV in light of hydration-is it overestimated by haemoconcentration? Severe anaemia may be associated with cardiac changes leading to increased risk of volume overload-care with fluid administration. Severity of anaemia on presentation was not found to be associated with survival in a recent study. Blood typing and ideally cross-matching should be performed before ANY transfusion; administration of incompatible blood is potentially fatal. Catabolism of body protein for gluconeogenesis occurs rapidly following a period of anorexia and illness. Assessment of nutritional requirements is a priority following emergency stabilisation and assisted feeding may be necessary in the critically ill patient. Assessment of nutritional requirements is a priority and assisted feeding may be necessary in the critically ill patient. Assessment of response to an analgesic trial using a short acting opioid may be necessary to confirm suspicion of pain. Evaluate for pain using physical examination, heart rate, respiratory rate, rectal temperature and behaviour. Diffuse abdominal pain can be a feature of sepsis, even in the absence of overt abdominal pathology. Efforts should be directed to address pain, and minimise excessive handling and stress in anxious cats. Follow the principles of nursing care and feline handling recommendations made by the AAFP and ISFM. Consider sedatives carefully for extremely fractious patients.
8 LOGICAL INTRAVENOUS FLUID THERAPY In the normal animal fluid homeostasis is a balance between water input (food, drinking water, metabolic water) and fluid output (urine, faeces, evaporation from respiratory tract and skin). In a clinical patient we must decide whether they have inadequate fluid intake or excessive fluid losses, which we must replace. In addition we then must consider the type of loss which is occurring e.g. isotonic, hypotonic, hypertonic, the space from which the fluid loss is occurring (extracellular, intracellular), the degree of fluid imbalance and we use this information to decide the most appropriate fluid replacement therapy. Abnormal fluid losses include vomiting, diarrhoea, polyuria/polydipsia, pyrexia, panting, bleeding, wound/burn exudation, third-spacing (loss into cavities/interstitial space). Good fluid management rests upon understanding why the patient needs fluid therapy and whether volume replacement is urgent/life-saving or whether more gradual correction or maintenance of fluid/electrolyte balance is required. It is important to differentiate between hypovolaemia/hypoperfusion and dehydration. Hypoperfusion refers to local or generalised deficit in tissue blood flow, resulting in inadequate oxygen and nutrient delivery and failure to remove metabolic waste products from the tissues. Generalised hypoperfusion can result from hypovolaemia (reduced effective circulating intravascular volume), reduced cardiac function or maldistribution of blood flow as is seen in systemic inflammatory response syndrome (SIRS). Common causes of hypovolaemia include: Haemorrhage Extracellular fluid loss in excess of intake (vomiting, diarrhoea, polyuria) Internal losses of plasma volume due to exudation or transudation of fluid from the intravascular space ( third spacing ) Dehydration is strictly defined as a net reduction in the free water content of the body; but in veterinary terms is used to refer to combined water and solute loss in excess of intake. Dehydration can ultimately lead to hypovolaemia and hypoperfusion. FLUID SELECTION Fluid homeostasis is important therefore input should balance output. So when considering a patient Do they need fluid therapy o Consider if need to treat hypoperfusion, dehydration or provide maintenance fluids or a combination of the three! Do they have Inadequate intake? Excessive losses? What is the type of fluid loss? Hypotonic, isotonic, hypertonic If the patient requires fluid therapy? What type? What route? How much? How fast? How long?
9 HOW TO ASSESS REQUIREMENTS Perfusion and hydration abnormalities are assessed using the physical examination. Parameters for assessing hydration state include: Moisture of mucous membranes, skin turgor, presence or absence of retraction of the globe Parameters for assessing perfusion status include: Heart rate (& cardiac auscultation), pulse quality (height and width of pulse profile), mucous membrane colour and capillary refill time (& vigour). Uncomplicated hypovolaemia has a relatively straightforward presentation. A normal animal should have pink mucous membranes with rapid CRT (1-1.75s), the pulse height and width should be adequate and heart rate is generally bpm in cats. In early compensated hypovolaemia the general changes are tachycardia, narrower and higher pulse profile than normal ( bounding ), mucous membranes are pink and CRT is rapid. When hypovolaemia becomes complicated or progresses then changes in these parameters are seen with ultimately bradycardia, hypotension, pale membranes with prolonged refill time and short low volume pulses. However remember that cats often do not show the typical signs of early compensated shock. Clinical Assessment of Hydration status % Dehydration Clinical history/findings <5% No physical evidence of dehydration, history suggests increased fluid loss or decreased intake 5-6% Dry mucous membranes, subtle loss of skin elasticity 6-8% Mild to moderate reduction in skin elasticity, mild increase in capillary refill time, dry mucous membranes. Eyes may be sunken in orbits 10-12% Marked reduction in skin elasticity, capillary refill time 2-3 seconds, dry mucous membranes eyes sunken in orbits. Possibly signs of shock : tachycardia, cool extremities, rapid & weak pulses 12-15% Severe signs of shock or dehydration, capillary refill time >3seconds, Severely debilitated >15% Incompatible with life The chart above details the percentage dehydration and the associated clinical signs, which can be used to assess the patient s fluid requirements. This chart underestimates fluid losses due to free water loss and overestimates hypertonic losses. False positive and negative findings can complicate the assessment e.g. obese animals retain normal skin elasticity in the face of significant dehydration, old animals have decreased skin elasticity. Hydration/volume status can also be assessed with urine output and specific gravity, serial measurement of packed cell volume and total protein, bodyweight, serum biochemistry and electrolytes and more advanced monitoring e.g. central venous pressure (CVP).
10 BUT All of these evaluations of fluid requirements are estimates. They are VERY approximate!! HOW MUCH FLUID TO GIVE? Aims: restore circulating volume replace normal maintenance requirements replace continuing losses Methods: Use dehydration chart to estimate % dehydration and then use this equation: A)Fluid deficit in Litres = BWt (kg) x % dehydration as decimal B)Maintenance = sensible losses (27-40ml/kg/day urine) and insensible losses (13-20ml/kg/day faecal/respiratory) often estimated to be 50ml/kg/day C)Ongoing Losses Estimate diarrhoea volume/episode (e.g. Diarrhoea could cause loss of up to 200ml/kg/day) Estimate vomitus volume/episode Wound exudates Total requirement = A+B+C However in clinical practice fluids are often given to effect with a guesstimate rate required to replace deficits. The fluids are administered; the patient is closely monitored and if therapy seems inadequate more is given. RATE & DURATION OF ADMINISTRATION Assess patient s condition using clinical and laboratory parameters. If the patient is in shock then rapid administration is indicated ideally a 5-10ml/kg/bolus of crystalloids is given over minutes (up to 66 ml/kg/hr can be given but it is given as repeat bolus rather than shock dose which has lost favour over bolus therapy with monitoring because of the risk of overinfusion & hypervolaemia). Also small/low volume resuscitation is indicated if the patient has active haemorrhage. Maintenance fluid rates are 2ml/kg /hr or 50ml/kg/day. When replacing deficits and ongoing losses, maintenance (2ml/kg/hr) must be provided plus a multiple of maintenance requirements to replace the deficits. The patient s response to therapy should be monitored and fluids reduced and then stopped when you have controlled excessive losses and normalised intake. Since the incidence of occult heart disease is high in cats, frequent re-evaluation of the thorax is essential particularly in cats with murmurs to monitor for development of murmurs, gallop rhythm and arrhythmias and also to monitor for signs of fluid overload. Hypothermia blunts the cats vascular response to fluid therapy and increases the risk of volume overload, so more conservative doses of fluids should be given whilst warming the cat. MONITORING Monitoring can include: Clinical assessment Clinical assessment:
11 o Heart rate should normalise and pulse rate & quality should improve nb pain, hypoxia, hypothermia, electrolyte disturbances and drugs can also affect heart rate o Respiratory rate should decrease o Mucous membrane colour & capillary refill time should normalise o Urine output 1.5ml/kg/hr = normal >2ml/kg/hr = overhydration/overinfusion o Skin tenting should decrease o Mentation should improve (nb pain, hypoglycaemia, electrolyte changes also affect this) o Temperature should normalise Central venous pressure requires central venous catheter placement and then serial measurements can be used to guide fluid therapy Serial PCV & total protein measurements care as interpretation can be difficult in anaemia or hypoproteinaemic patients. Urea, creatinine, electrolytes and lactate Arterial blood pressure aim to maintain systolic pressure at >90mmHg. If hypotension persists assess for hypoglycaemia, hypoxia, hypothermia, cardiac disease, arrhythmia, bradycardia, electrolyte/acid-base disorders, pleural space disease, cardiac tamponade, sepsis/sirs or brain stem disease. EVIDENCE OF TOO MUCH OR TOO LITTLE FLUID Volume Depletion Volume Overload Weak rapid pulse Coughing Pale, dry/tacky mucous Increased respiratory rate membranes Slow capillary refill time Subcutaneous oedema Poor skin elasticity Ascites / pleural effusion Cool distal extremities Chemosis & Serous nasal discharge Sunken eyes Exophthalmus Reduced urine output Increased urine output Heart small on radiographs Vomiting If there are signs of overinfusion stop fluids! TYPES OF PARENTERAL FLUID There are numerous different fluids each of which has its own indications and contraindications and the underlying disease process and fluid deficit will determine which fluid is the most appropriate for that patient. There are 3 major categories of fluid types: crystalloids, colloids and blood products. Crystalloids are electrolyte solutions that can pass easily out of the vascular space whereas colloids contain macromolecules that are retained within the vascular space for a longer time period. Colloids are synthetic products and are sometimes referred to as plasma substitutes. Blood and blood products (fresh frozen plasma, fresh plasma, packed red cells, cryoprecipitate) were not readily available in the UK and hence colloids were frequently used in situations where blood products would be preferable. The Pet blood bank has been operating in the UK ( for dogs for some time now but does not yet produce feline blood products. Colloids are currently not available in the UK.
12 Plasma is mainly used to manage patients with coagulopathies and packed red cells for anaemic patients. There is also a solution of polymerised bovine haemoglobin, which has colloidal properties but also carries oxygen. Additionally some clinicians consider the use of human serum albumin for compromised hypoalbuminaemic patients. CRYSTALLOIDS These are electrolyte solutions, which freely pass out of the vascular space & equilibrate with the interstitial space % of the fluid leaves the vascular space within 1 hour. They provide short-term intravascular volume expansion and replace interstitial deficits. They can be hypotonic, isotonic or hypertonic and the tonicity of the solution determines its distribution after intravenous infusion. They include: Hartmann s, Ringer s, Normal Saline, Dextrose Saline. REPLACEMENT CRYSTALLOIDS 0.9% SODIUM CHLORIDE An isotonic replacement solution, which contains sodium, chloride and water. Indications: hypochloraemia (short term gastric vomiting) hyponatraemia & hypochloraemia (hypoadrenocorticism) intraoperative fluid therapy. HARTMANN S SOLUTION An isotonic replacement solution that has a similar electrolyte composition to plasma and contains Na, K, Ca, Cl and lactate. Indications: electrolyte loss with acidosis (post pyloric intestinal losses) intraoperative fluid therapy. RINGERS An isotonic replacement solution that contains Na, Cl, K, Ca. Indications: electrolyte & water loss pre-pyloric vomiting patients with liver disease (where Hartmann s is contraindicated as they have reduced ability to metabolise lactate). MAINTENANCE SOLUTIONS 5% DEXTROSE An isotonic solution, which becomes hypotonic once the dextrose has been metabolised. In the animal the dextrose is rapidly metabolised and this leaves free water which rapidly passes out of the intravascular space and distributes across the total body water, therefore it is an ineffective plasma volume expander and should not be used to treat hypovolaemia. There are no electrolytes and rapid infusion of hypotonic solutions can result in electrolyte disorders. Indications: Free water loss e.g. heat stroke, pyrexia, no oral intake It is a glucose source but insufficient to provide the caloric requirements of anorexic patients.
13 A maintenance solution with few indications. 0.18% SODIUM CHLORIDE & 4% GLUCOSE (GLUCOSE SALINE) This is mainly water with a small amount Na & Cl to replace urinary losses. Indications: patients with primary water loss. It is a maintenance fluid and should be supplemented with potassium in non-hyperkalaemic anorexic patients when being used for maintenance. Few indications to use this fluid. HYPERTONIC SOLUTION % HYPERTONIC SALINE This has a high osmotic potential (compared to normal saline 0.9%, it is 8x more concentrated than plasma). Indications: to improve the circulation by drawing intracellular and interstitial water into intravascular space along a large concentration gradient. patients with gastric dilation and volvulus and severe haemorrhage which benefit from rapid intravascular volume resuscitation lower total volume of resuscitation fluid required It should ALWAYS be used with isotonic crystalloids to avoid depleting the intracellular and interstitial fluids. Duration of action can be prolonged by combining it with colloid. It is contraindicated if patients are dehydrated (no reservoir of interstitial and intracellular fluid), hyperosmolar (e.g. hypernatraemia), volume overload (cardiorespiratory disease) and uncontrolled haemorrhage. COLLOIDS NB NO LONGER ABLE TO ACQUIRE STARCH BASED COLLOIDS IN UK,OTHER COLLOIDS CONTROVERSIAL RE SIDE EFFECTS These are synthetic products that contain macromolecules with a similar molecular size to albumin. These molecules are retained within the vascular space for several hours unless there are leaky capillaries. They provide intravascular volume expansion oncotic support They draw fluid into vessels from interstitial and intracellular spaces. Indicated for: treatment of hypovolaemia (shock, haemorrhage) to provide circulatory support support hypoalbuminaemic patients by providing oncotic support. Small molecular weight substances are excreted/extravasated within hours. The larger molecules remain in the circulation and are enzymatically degraded or removed by the phagocytic system. In humans, they appear to be deleterious in patients with sepsis, capillary leak syndrome and adult respiratory distress syndrome following trauma. The colloid pull relates to the number of molecules rather than the size. There are a number of different types of colloid (Dextrans, Gelatins, Starches) and choice may be influenced by the molecular weight and the duration of action. Gelofusin is a relatively low molecular weight with short duration of maximum benefit and therefore the author prefers to use a starch-based colloid, which has a higher molecular weight and longer duration of action. The dose in an emergency can be 5-10ml/kg given over 30 minutes to 3-6 hours.
14 It can also be used to provide oncotic support at a dose of 10-20ml/kg/day by intravenous infusion (maximum daily dose up to 33ml/kg for pentastarch and 50ml/kg for tetrastarch e.g.voluven). If colloids are given too rapidly/too much it can over-expand the blood volume. They are contraindicated in heart failure patients for this reason. Cats may show restlessness & salivation and are more prone to volume overload. FLUID SUPPLEMENTS POTASSIUM CHLORIDE Hypokalaemia is an important cause of weakness in hospitalised patients. Potassium is predominantly taken in to the body in food thus any anorexic / inappetant patients are at risk of developing hypokalaemia. Certain diseases or drug therapies might accentuate this hypokalaemia due to renal loss e.g. chronic renal failure, hyperaldosteronism or diuretic use (frusemide) or loss secondary to GI disease. Thus it is important to monitor potassium in sick animals and to supplement if necessary as below. Measure serum levels before supplementing intravenous fluids. It should never be supplemented at > 0.5mmol/kg/hour. It is important to ALWAYS mix well (heavy ion which can settle in the bag) and ALWAYS label clearly. DO NOT give until renal function/urine output is established. There is a risk or arrhythmias and possibly cardiac arrest or death with excessive or bolus administration. Maintenance potassium supplementation is often 20mmol of KCl per 1 litre of fluids. Always check the infusion rate to make sure this is safe and if the patient is severely hypokalaemic consult texts for more information on dosing. SODIUM BICARBONATE In most situations the kidney will sort out acid-base disorders if appropriate fluid therapy is given. There are some situations in which the acidosis is severe which require bicarbonate therapy BUT respiratory function must be adequate so that carbon dioxide can be eliminated effectively. It may be indicated for patients with severe metabolic acidosis and also can be used in patients with severe hyperkalaemia. INDICATIONS FOR BLOOD TRANSFUSIONS Restore oxygen carrying capacity e.g. acute haemorrhage, severe anaemia Restore blood volume e.g. acute haemorrhage Provide clotting factors, platelets, plasma proteins Provide specific and non-specific antibodies COLLECTION OF THE BLOOD FROM THE DONOR ASSESSING SUITABILITY OF THE BLOOD Before collecting the blood the suitability of the donor s blood for the recipient should be assessed. This is essential in cats where fatal transfusion reactions can occur with the first transfusion due to the presence of pre-formed anitbodies. BLOOD TYPING/GROUPING Should always be performed before transfusing a cat.
15 Cross matching should also be performed in patients who have previously had a transfusion or who are likely to require more than one. BLOODS AND ITS PRODUCTS WHOLE BLOOD Whole blood can be stored for up to 4 weeks in fridge (CPD) and it is indicated for: Acute haemorrhage o Loss > 30% blood volume Anaemia o? When (controversial, should be based on an individual animal evaluation) If PCV is less than 10-15% Acute clinical deterioration Whole blood is also used is situations where blood components might be more appropriate e.g. coagulopathy because separation of blood into its components is time consuming in practice. PLASMA This is species specific, it is a natural colloid which provides oncotic support (impractical due to volume required, hence use of synthetic colloids) and coagulation factors. It only provides coagulation factors if it is separated from red cells within 8hrs of collection. Fresh frozen plasma (FFP) can be stored for up to 1 year in a freezer. PACKED RED CELLS These are produced when plasma is separated from a whole blood unit. They can be resuspended in 0.9% sodium chloride before administration or follow directions with the product. Because of the citrate anticoagulant, you should not resuspend in Hartmann s or Ringer s as they contain calcium, which reacts with citrate. They can be stored for up to 42 days in fridge. They are indicated for: normovolaemic anaemia o Immune Mediated Haemolytic Anaemia Hypovolaemia anaemia e.g. haemorrhage if plasma volume is also replaced e.g. synthetic colloid, plasma CRYOPRECIPITATE Indicated for von Willebrands disease and also provides fibrinogen, Factor XIII, and Factor VIII. Collected during thawing of frozen plasma. ADMINISTRATION OF A BLOOD TRANSFUSION Aseptically place IV/ intraosseous line into patient Connect RBC bag to specialised giving set (contain filter µm) & run through the giving set o Do not mix with calcium containing solutions e.g. Hartmann s o Use an In line filter for cats if giving blood via syringe Warm blood to 37 o C Connect to patient Rate of administration - varies with reason for transfusion see below Amount of Blood o Acute haemorrhage - rapidly replace volume lost (so long as you have controlled the bleeding)
16 o Anaemia Volume = [(desired PCV-recipient PCV)/PCV of donor] x recipient blood volume Blood volume = 6-8% Body weight 60-70ml/kg cat 2ml/kg whole blood increase recipient PCV by 1% RATE OF ADMINISTRATION In acute haemorrhage blood is given rapidly but in other cases administer at ml/kg/hr for 30 minutes. Monitor every 5 minutes for signs of reaction using TPR, mucous membrane colour etc. If there are no adverse signs then after the first 30 minutes the rate is increased incrementally to 5ml/kg/hr (cats). NB not > 4ml/kg/hr if cardiac disease. The aim is to administer the desired volume of blood within 4 hrs to reduce the risk of bacterial growth if the blood is contaminated (it should not be!). TRANSFUSION REACTIONS Signs of a transfusion reaction include tachycardia, hypotension, urticaria, facial oedema, muscle tremors, panting, pyrexia and vocalization. Immediate reactions can be seen which are haemolytic or non-haemolytic. Haemolytic are unlikely in first transfusion except Group B cats. Delayed reactions can occur up to 2 wks later and often result in decreased survival of the red cells. Transfusion Reactions can also occur due to septicaemia (poor collection technique, storage), via transmission of infectious disease or parasites from donor (e.g. Babesia, Haemobartonella) or through volume overload (normovolaemic patient, patient with cardiac disease). Citrate toxicity is also reported and could be seen in patients with severe hepatic disease who cannot metabolise the citrate and develop hypocalcaemia. OXYGLOBIN Oxyglobin is a solution of polymerised bovine haemoglobin (no cell membrane), which has colloidal properties but also carries oxygen. Oxyglobin has a long shelf life (3 years) if it is in its sealed foil pouch but a very short life once the pouch is opened. There is no requirement for cross matching and can be stored at room temperature. Unfortunately it is relatively expensive. It is cleared from the plasma within 5-9 days and therefore does not have such long lasting benefits for oxygen transport compared with a red cell transfusion. However the clinical benefit is between 74-82hrs after a dose of 30ml/kg. Oxyglobin has been used in dogs and cats. Cats in particular are sensitive to volume overload and can develop pulmonary oedema, particularly when it is used in normovolaemic patients. Samples for laboratory analysis should be collected before administration as the solution will cause discolouration of the sclera and mucous membranes and also will interfere with colourimetric laboratory assays. Indicated to increase oxygen carrying capacity in anaemic animals with no risk of transfusion reaction. It also improves perfusion and is useful in patients with poor microcirculation e.g. sepsis.
17 SHOCK, TOXINS & CPR SHOCK INTRODUCTION Shock is a complex series of physiological events, which can develop from a variety of causes or clinical diseases. It is important to remember that a patient may present in shock, which will require emergency management or may develop shock during hospitalisation. It is vital to try and identify and treat shock as early as possible to reduce the morbidity and mortality of these patients. Shock is essentially an imbalance between oxygen delivery and oxygen consumption such that the tissues are oxygen deficient. Oxygen delivery is dependent upon cardiac output (heart rate and stroke volume) and arterial oxygen content (haemoglobin concentration and saturation with oxygen). Shock can be classified as follows: Hypovolaemic shock which may be secondary to blood loss or salt and water loss (via urine, faeces, vomitus) or third-spacing of fluids in the intestinal tract or body cavities. Traumatic shock often has a component of hypovolaemic shock. Loss of fluid into damaged tissues and pain can alter the vasomotor response. Cardiogenic shock due to failure of the cardiac pump Distributive shock is a loss of adequate peripheral resistance and can be secondary to sepsis, endotoxaemia, anaphylaxis or neurogenic injury. Hypovolaemic and cardiogenic shock are more common in cats. Cats are more typically bradycardic than dogs and bounding pulses and congested mucous membranes are rarely appreciated in cats. Hypothermia is common in shocked cats and blunts the normal vasoconstrictive response to hypovolaemia which results in shocked cats being at high risk of volume overload if hypothermia is not addressed. Occult cardiac disease is also very common either due to primary cardiomyopathy or secondary to systemic disease and thus careful auscultation pre and during fluid therapy is essential. Careful evaluation for heart murmur, gallop rhythm, arrhythmia or crackles should be performed before, during and after fluid therapy. Occult cardiomyopathy can be present without a heart murmur. Pain can be challenging to assess and therefore routine use of analgesics is recommended. Nutrition should be addressed at an early stage (see later notes) The 3 stages of shock (compensatory, early deconpensatory and decompensatroy ro terminal) are less obvious in the cat compared to the dog Shock is associated with distribution of blood to preferred organs e.g. heart and brain. Other tissues are hypoperfused and so become oxygen deficient and switch to anaerobic cell metabolism, which results in lactic acidosis. Inflammatory mediators accumulate and can incite inflammatory responses. Cell damage leads to loss of capillary endothelial integrity and extravasation of fluid into the interstitium, exacerbating the fluid deficit. Reduced perfusion of the gut can lead to bacterial translocation and risk of bacteraemia. Myocardial depressant factors (MDF) may be released by some organs in response to hypoperfusion and can predispose to cardiac arrhythmias. Renal hypoperfusion can lead to reduced urine production and tubular necrosis. Reduced perfusion can also result in microthrombi formation which can result in vascular occlusion and further cell death or they can initiate widespread clotting and consumption of clotting factors (disseminated intravascular coagulation/ DIC). In the late stages of shock, the body has lost autoregulatory control and massive vasodilation occurs in all organs.
18 Many organs can become affected if shock continues Deteriorating kidney function Respiratory compromised Ischaemia of gut translocation of bacteria and endotoxin sepsis Liver hypoperfusion reduced drug metabolism/excretion, increased risk infection Myocardial depression Assessment of Shock: Clinical Sign Mental State Mucous membrane colour Capillary Refill Time Heart Rate Pulse Quality Temperature of extremities Skin Turgor Mucous membrane moistness Alteration Reduced perfusion can result in depression of the mental status due to the high energy demands of the brain Amount and composition (haemoglobin, oxygen) of blood in the underlying capillaries determines this. Pallor/white = severe vasoconstriction or anaemia Red = vasodilation & venous pooling, common in sepsis Prolonged with vasoconstriction Shortened with vasodilation Tachycardia due to volume loss, pain, fever, hypoxaemia, hypercapnia but more often bradycardia in cat Reflects the difference between systolic and diastolic pressure and the duration of the waveform. Poor pulse quality with hypovolaemia and loss of circulating volume. Hyperdynamic pulse with loss of vasomotor tone/vasodilation is uncommonly felt in the cat. Decreased with vasoconstriction, increased with vasodilation. Hypothermia common in cat. Prolonged skin tent with interstitial dehydration Decreased with interstitial fluid deficits. Altered by increased oral secretions e.g. nausea, oral disease, hypersalivation Clinical Evaluation Haematocrit / PCV/Hb Total Protein Urea/ Creatinine / Electrolytes Acid Base/ arterial blood gases Alteration PCV should be kept at least at23-25% to maintain oxygenation. Acute changes in blood volume are not reflected Consider colloids if TP >35g/l as risk of interstitial fluid accumulation Can be used to monitor hydration and renal function. Some abnormalities require intervention e.g. severe hyperkalaemia Acidosis as consequence of hypoperfusion can increase risk of arrhythmias and reduced cardiac performance
19 Arterial blood Pressure Central Venous Pressure Urine Output Pulse Oximetry Loss of autoregulation when systolic <80mmHg use fluids and possibly vasopressors to support Gives information about right ventricular function and ability of body to cope with fluid load Should be >1ml/kg/hr. If <1ml/kg/hr and no history renal disease, likely secondary to decreased cardiac output Monitor of oxygen saturation, can be misleading if poor perfusion or pigmentation Clinical evaluation can be extended to involve more advanced monitoring and laboratory assessments. Treatment of Shock As with all critical patients it is important to remember the ABC s of resuscitation (airway, breathing, bleeding and circulation) in addition to assessing major organ function (heart, lungs, kidneys, brain). If a primary cause can be identified for the development of shock then this should be treated specifically e.g. control bleeding. However often the underlying cause is unknown or cannot be treated quickly enough to reverse the situation. In this situation symptomatic and supportive therapy is required. The main goals of therapy are: to restore arterial blood pressure and circulating volume to sustain cardiac output and this will ensure adequate tissue perfusion & oxygenation. In most cases fluid therapy will achieve restoration of circulating volume and is all that is required. Crystalloids, colloids and oxyglobin may be considered refer to Fluid therapy
20 notes for further information. However treatment of shock is more challenging in cats because of: smaller blood volume (-use smaller fluid boluses eg 5ml/kg) frequent existence of occult cardiac disease greater susceptibility to volume overload (develop pulmonary oedema and pleural effusion which may relate to occult cardiac disease or to hypothermia affecting vascular tone, or multi-organ failure) NB in the cat the shock organ is considered to be the lung and particularly at risk of injury with sepsis. There are some other aims of treatment which include: Reduced viscosity of the blood fluids help to achieve this. Improved oxygenation supplement oxygen in a non stressful way (oxygen tent, oxygen collar, mask, nasal oxygen catheter, nasal prongs, flow by). Flow rates of 1-2L/min by flow by or T-piece. Cardiac support using vasopressor agents e.g. norepinephrine at 0.1-3mcg/kg/min. Treat metabolic acidosis once shock is established metabolism switches to anaerobic due to the hypoperfusion. This results in metabolic acidosis, which increases blood viscosity and impairs myocardial function. Sodium bicarbonate can be used as described in fluid therapy lectures. Thermoregulation body heat should be conserved as body temperature falls in shock. If the patient is hypothermic avoid further heat loss by covering the animal but do not rewarm directly as this will cause peripheral vasodilation and has haemodynamic consequences. Warm the ambient environment and warm IV fluids to 37C before administration. A Bair-hugger or similar device (or incubator) is more beneficial than heating mat. Peripheries can be wrapped in bubble wrap or soffban to reduce heat loss. Minimise use of spirit and clipping. Analgesia opioids preferred. Pain and response to it are detrimental so needs treatment. Consider methadone mg/kg slow IV or IM, morphine mg/kg IM, buprenorphine mg/kg IV or IM (onset action is 30 minutes and duration 6-8hr) or pethidine 3-5mg/kg IM. Antibiotics If sepsis is suspected or evident antibiosis is indicated particularly. IV therapy should be started and organ function (renal, hepatic) considered with respect to dose and interval. Corticosteroids This is a very controversial area and has been extensively studied. They have been shown to be beneficial if given BEFORE the patient has shock induced. In high doses they improve tissue perfusion and enhance myocardial contractility and stabilise cell membranes. The beneficial effects seem much less significant if they are given after induction of shock. Current preference is not to use corticosteroids. There is a rationale for using physiological doses in patients suspected to have relative adrenal insufficiency e.g. refractory hypotension despite appropriate fluid therapy, normothermia and vasopressors Non-steroidal anti-inflammatory drugs Prostaglandins mediate some of the deleterious effects of shock and therefore NSAID may be partially protective. Their use in hypovolaemic patients requires care since they may enhance renal damage. Not recommended. Anticoagulant therapy if disseminated intravascular coagulation or microvascular thrombosis is documented then heparin may be beneficial. Vasopressor therapy dopamine or noradrenaline Anti-endotoxic shock there are several potential treatments including monoclonal antibodies and mediator antagonists (antitnf, antipaf, NOS inhibitors)
21 Antioxidants/oxygen radical scavengers - in protection from reperfusion injury e.g. vitamin E, DMSO, allopurinol Careful attention to glucose levels in humans normoglycaemia improves outcomes in critically ill patients. Persistent hyperglycaemia may be managed with judicious insulin therapy. Hypoglycaemia could indicate sepsis and needs addressing with glucose boluses followed by infusions. Monitor and manage blood pressure FLUID THERAPY Intravenous access must be obtained, initially a peripheral venous catheter is placed in either the cephalic or saphenous (medial in cat) veins. A large gauge, short catheter is desirable for rapid fluid administration. In some cases it may be advisable to place an additional peripheral catheter or to place a central venous or jugular catheter. In severely hypovolaemic patients if may be necessary to perform a venous cut down or to place an intraosseous needle to achieve access for fluid resuscitation. Crystalloids are used to replace interstitial deficits since 75-85% of isotonic crystalloids will move to the interstitial space within 1 hour of intravenous (IV) administration. This is important since crystalloids as single therapy are not suitable to provide long-term intravascular support for shocked patients. Excessive administration of crystalloids decreases the intravascular oncotic pressure, which promotes further loss of fluids into the interstitial space from the intravascular space. Therefore colloids can have an important role to play in shock management. Synthetic colloids contain molecules with a similar molecular weight to albumin. This is beneficial in certain shock states whereby body composition is altered (decreased colloid oncotic pressure) particularly with systemic inflammatory response syndrome. Whole blood and plasma are natural colloids whereas the synthetic colloids include hetastarch, dextrans, haemaccel, oxyglobin. Whole blood and plasma have specific indications but are rarely used in shock treatment. Hetastarch was the recommended colloid as it had less effect on haemostasis than dextrans but starches are not currently available in UK. The aim of colloid therapy is to provide intravascular volume expansion and maintain oncotic pressure by remaining within the vascular space and attracting sodium and water into the vasculature. Remember that colloids only replace the intravascular volume deficit and so crystalloids must also be given to replace interstitial deficits. Oxyglobin is a haemoglobin-based oxygen carrying solution, which is suitable for use in the shock patient as a colloid with the additional benefit of its oxygen carrying capacity. If fluid support is ineffective in restoring circulating volume, blood pressure etc. then additional therapy must be considered e.g. positive inotropic drugs or vasopresors MONITORING This should include heart & respiratory rate, pulse rate and quality/pressure, CRT, body temperature (& extremities temperature) and demeanour/mental state. In addition further monitoring might include: central venous pressure, arterial blood pressure, electrocardiography, urine output, haematocrit, total proteins and glucose (+/- urea, creatinine and electrolytes), arterial blood gas analysis and pulse oximetry. A monitoring chart can be useful to detect trends in the patient s parameters rather than simply relying on the numbers. It is important to consider end-points which will allow you to assess if the therapy has been successful, and to base treatment decisions upon and frequency of monitoring.
Taking the shock factor out of shock
 Taking the shock factor out of shock Julie Antonellis, BS, LVT, VTS (ECC) Northern Virginia Regional Director for the VALVT Technician Supervisor VCA Animal Emergency Critical Care Business owner Antonellis
Taking the shock factor out of shock Julie Antonellis, BS, LVT, VTS (ECC) Northern Virginia Regional Director for the VALVT Technician Supervisor VCA Animal Emergency Critical Care Business owner Antonellis
Printed copies of this document may not be up to date, obtain the most recent version from
 Children s Acute Transport Service Clinical Guidelines Septic Shock Document Control Information Author Claire Fraser P.Ramnarayan Author Position tanp CATS Consultant Document Owner E. Polke Document
Children s Acute Transport Service Clinical Guidelines Septic Shock Document Control Information Author Claire Fraser P.Ramnarayan Author Position tanp CATS Consultant Document Owner E. Polke Document
KASHVET VETERINARIAN RESOURCES FLUID THERAPY AND SELECTION OF FLUIDS
 KASHVET VETERINARIAN RESOURCES FLUID THERAPY AND SELECTION OF FLUIDS INTRODUCTION Formulating a fluid therapy plan for the critical small animal patient requires careful determination of the current volume
KASHVET VETERINARIAN RESOURCES FLUID THERAPY AND SELECTION OF FLUIDS INTRODUCTION Formulating a fluid therapy plan for the critical small animal patient requires careful determination of the current volume
IV Fluids. I.V. Fluid Osmolarity Composition 0.9% NaCL (Normal Saline Solution, NSS) Uses/Clinical Considerations
 IV Fluids When administering IV fluids, the type and amount of fluid may influence patient outcomes. Make sure to understand the differences between fluid products and their effects. Crystalloids Crystalloid
IV Fluids When administering IV fluids, the type and amount of fluid may influence patient outcomes. Make sure to understand the differences between fluid products and their effects. Crystalloids Crystalloid
-Cardiogenic: shock state resulting from impairment or failure of myocardium
 Shock chapter Shock -Condition in which tissue perfusion is inadequate to deliver oxygen, nutrients to support vital organs, cellular function -Affects all body systems -Classic signs of early shock: Tachycardia,tachypnea,restlessness,anxiety,
Shock chapter Shock -Condition in which tissue perfusion is inadequate to deliver oxygen, nutrients to support vital organs, cellular function -Affects all body systems -Classic signs of early shock: Tachycardia,tachypnea,restlessness,anxiety,
12/1/2009. Chapter 19: Hemorrhage. Hemorrhage and Shock Occurs when there is a disruption or leak in the vascular system Internal hemorrhage
 Chapter 19: Hemorrhage Hemorrhage and Shock Occurs when there is a disruption or leak in the vascular system External hemorrhage Internal hemorrhage Associated with higher morbidity and mortality than
Chapter 19: Hemorrhage Hemorrhage and Shock Occurs when there is a disruption or leak in the vascular system External hemorrhage Internal hemorrhage Associated with higher morbidity and mortality than
SHOCK AETIOLOGY OF SHOCK (1) Inadequate circulating blood volume ) Loss of Autonomic control of the vasculature (3) Impaired cardiac function
 SHOCK Shock is a condition in which the metabolic needs of the body are not met because of an inadequate cardiac output. If tissue perfusion can be restored in an expeditious fashion, cellular injury may
SHOCK Shock is a condition in which the metabolic needs of the body are not met because of an inadequate cardiac output. If tissue perfusion can be restored in an expeditious fashion, cellular injury may
Paediatric Shock. Dr Andrew Pittaway Department of Anaesthesia Bristol Royal Hospital for Children Bristol, UK
 Paediatric Shock Dr Andrew Pittaway Department of Anaesthesia Bristol Royal Hospital for Children Bristol, UK Self-assessment: 1. What is the definition of shock? 2. List the different pathophysiological
Paediatric Shock Dr Andrew Pittaway Department of Anaesthesia Bristol Royal Hospital for Children Bristol, UK Self-assessment: 1. What is the definition of shock? 2. List the different pathophysiological
SHOCK. Pathophysiology
 SHOCK Dr. Ahmed Saleem FICMS TUCOM / 3rd Year / 2015 Shock is the most common and therefore the most important cause of death of surgical patients. Death may occur rapidly due to a profound state of shock,
SHOCK Dr. Ahmed Saleem FICMS TUCOM / 3rd Year / 2015 Shock is the most common and therefore the most important cause of death of surgical patients. Death may occur rapidly due to a profound state of shock,
Critical Care of the Post-Surgical Patient
 Critical Care of the Post-Surgical Patient, Dr med vet, DEA, DECVIM-CA Many critically ill patients require surgical treatments. These patients often have multisystem abnormalities during the immediate
Critical Care of the Post-Surgical Patient, Dr med vet, DEA, DECVIM-CA Many critically ill patients require surgical treatments. These patients often have multisystem abnormalities during the immediate
Printed copies of this document may not be up to date, obtain the most recent version from
 Children s Acute Transport Service Clinical Guidelines Septic Shock Document Control Information Author Shruti Dholakia L Chigaru Author Position Fellow CATS Consultant Document Owner E. Polke Document
Children s Acute Transport Service Clinical Guidelines Septic Shock Document Control Information Author Shruti Dholakia L Chigaru Author Position Fellow CATS Consultant Document Owner E. Polke Document
Fluids and electrolytes: the basics
 Fluids and electrolytes: the basics This document is based on the handout from the Surgery for Finals course. The notes provided here summarise key aspects, focusing on areas that are popular in clinical
Fluids and electrolytes: the basics This document is based on the handout from the Surgery for Finals course. The notes provided here summarise key aspects, focusing on areas that are popular in clinical
INTRAVENOUS FLUID THERAPY. Tom Heaps Consultant Acute Physician
 INTRAVENOUS FLUID THERAPY Tom Heaps Consultant Acute Physician LEARNING OBJECTIVES 1. Crystalloids vs colloids 2. Balanced vs non-balanced solutions 3. Composition of various IV fluids 4. What is normal
INTRAVENOUS FLUID THERAPY Tom Heaps Consultant Acute Physician LEARNING OBJECTIVES 1. Crystalloids vs colloids 2. Balanced vs non-balanced solutions 3. Composition of various IV fluids 4. What is normal
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Aqupharm 3 Solution for Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredients Sodium Chloride Glucose Anhydrous
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Aqupharm 3 Solution for Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredients Sodium Chloride Glucose Anhydrous
Physiological Response to Hypovolemic Shock Dr Khwaja Mohammed Amir MD Assistant Professor(Physiology) Objectives At the end of the session the
 Physiological Response to Hypovolemic Shock Dr Khwaja Mohammed Amir MD Assistant Professor(Physiology) Objectives At the end of the session the students should be able to: List causes of shock including
Physiological Response to Hypovolemic Shock Dr Khwaja Mohammed Amir MD Assistant Professor(Physiology) Objectives At the end of the session the students should be able to: List causes of shock including
Chapter 3 MAKING THE DECISION TO TRANSFUSE
 Chapter 3 MAKING THE DECISION TO TRANSFUSE PRACTICE POINTS Determine the best treatment for the patient which may include transfusion. Treat the cause of cytopenia (anaemia or thrombocytopenia) or plasma
Chapter 3 MAKING THE DECISION TO TRANSFUSE PRACTICE POINTS Determine the best treatment for the patient which may include transfusion. Treat the cause of cytopenia (anaemia or thrombocytopenia) or plasma
HOW LOW CAN YOU GO? HYPOTENSION AND THE ANESTHETIZED PATIENT.
 HOW LOW CAN YOU GO? HYPOTENSION AND THE ANESTHETIZED PATIENT. Donna M. Sisak, CVT, LVT, VTS (Anesthesia/Analgesia) Seattle Veterinary Specialists Kirkland, WA dsisak@svsvet.com THE ANESTHETIZED PATIENT
HOW LOW CAN YOU GO? HYPOTENSION AND THE ANESTHETIZED PATIENT. Donna M. Sisak, CVT, LVT, VTS (Anesthesia/Analgesia) Seattle Veterinary Specialists Kirkland, WA dsisak@svsvet.com THE ANESTHETIZED PATIENT
FLUID THERAPY: IT S MORE THAN JUST LACTATED RINGERS
 FLUID THERAPY: IT S MORE THAN JUST LACTATED RINGERS Elisa M. Mazzaferro, MS, DVM, PhD, DACVECC Cornell University Veterinary Specialists, Stamford, CT, USA Total body water constitutes approximately 60%
FLUID THERAPY: IT S MORE THAN JUST LACTATED RINGERS Elisa M. Mazzaferro, MS, DVM, PhD, DACVECC Cornell University Veterinary Specialists, Stamford, CT, USA Total body water constitutes approximately 60%
What would be the response of the sympathetic system to this patient s decrease in arterial pressure?
 CASE 51 A 62-year-old man undergoes surgery to correct a herniated disc in his spine. The patient is thought to have an uncomplicated surgery until he complains of extreme abdominal distention and pain
CASE 51 A 62-year-old man undergoes surgery to correct a herniated disc in his spine. The patient is thought to have an uncomplicated surgery until he complains of extreme abdominal distention and pain
COMPANY CORE PACKAGE INSERT CCPI (PI/CORE/ENGLISH)
 COMPANY CORE PACKAGE INSERT CCPI (PI/CORE/ENGLISH) HUMAN ALBUMIN 20 % BEHRING Rev.: 05-MAR-2008 / PEI approval 26.02.08 Supersedes previous versions Rev.: 28-NOV-2007 / Adaptation to Core SPC Rev.: 02-JAN-2007
COMPANY CORE PACKAGE INSERT CCPI (PI/CORE/ENGLISH) HUMAN ALBUMIN 20 % BEHRING Rev.: 05-MAR-2008 / PEI approval 26.02.08 Supersedes previous versions Rev.: 28-NOV-2007 / Adaptation to Core SPC Rev.: 02-JAN-2007
SEPSIS SYNDROME
 INTRODUCTION Sepsis has been defined as a life threatening condition that arises when the body s response to an infection injures its own tissues and organs. Sepsis may lead to shock, multiple organ failure
INTRODUCTION Sepsis has been defined as a life threatening condition that arises when the body s response to an infection injures its own tissues and organs. Sepsis may lead to shock, multiple organ failure
HEAT STROKE. Lindsay VaughLindsay Vaughn, DVM, DACVECCDVM, DACVECC
 HEAT STROKE Lindsay VaughLindsay Vaughn, DVM, DACVECCDVM, DACVECC Heat Stroke More Preventable Than Treatable Heat Stroke A form of hyperthermia associated with a systemic inflammatory response leading
HEAT STROKE Lindsay VaughLindsay Vaughn, DVM, DACVECCDVM, DACVECC Heat Stroke More Preventable Than Treatable Heat Stroke A form of hyperthermia associated with a systemic inflammatory response leading
SLCOA National Guidelines
 SLCOA National Guidelines Peri - operative Fluid & Electrolyte Management SLCOA National Guidelines Contents List of Contributors 92 Paediatric fasting guidelines for elective procedures 93 Guidelines
SLCOA National Guidelines Peri - operative Fluid & Electrolyte Management SLCOA National Guidelines Contents List of Contributors 92 Paediatric fasting guidelines for elective procedures 93 Guidelines
Nursing the critical care patient part 1: triage
 Nursing the critical care patient part 1: triage EJCAP 26(3) Autumn 2016 P 51 Commissioned paper* Nursing the critical care patient part 1: triage Katherine Howie 1 SUMMARY Good nursing of the emergency
Nursing the critical care patient part 1: triage EJCAP 26(3) Autumn 2016 P 51 Commissioned paper* Nursing the critical care patient part 1: triage Katherine Howie 1 SUMMARY Good nursing of the emergency
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Albunorm 20%, 200 g/l, solution for infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Albunorm 20% is a solution containing 200 g/l
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Albunorm 20%, 200 g/l, solution for infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Albunorm 20% is a solution containing 200 g/l
Colic: medical treatment and management in horses
 Vet Times The website for the veterinary profession https://www.vettimes.co.uk Colic: medical treatment and management in horses Author : Nicola Menzies-Gow Categories : Equine, Vets Date : July 4, 2016
Vet Times The website for the veterinary profession https://www.vettimes.co.uk Colic: medical treatment and management in horses Author : Nicola Menzies-Gow Categories : Equine, Vets Date : July 4, 2016
SHOCK. Emergency pediatric PICU division Pediatric Department Medical Faculty, University of Sumatera Utara H. Adam Malik Hospital
 SHOCK Emergency pediatric PICU division Pediatric Department Medical Faculty, University of Sumatera Utara H. Adam Malik Hospital 1 Definition Shock is an acute, complex state of circulatory dysfunction
SHOCK Emergency pediatric PICU division Pediatric Department Medical Faculty, University of Sumatera Utara H. Adam Malik Hospital 1 Definition Shock is an acute, complex state of circulatory dysfunction
SHOCK Susanna Hilda Hutajulu, MD, PhD
 SHOCK Susanna Hilda Hutajulu, MD, PhD Div Hematology and Medical Oncology Department of Internal Medicine Universitas Gadjah Mada Yogyakarta Outline Definition Epidemiology Physiology Classes of Shock
SHOCK Susanna Hilda Hutajulu, MD, PhD Div Hematology and Medical Oncology Department of Internal Medicine Universitas Gadjah Mada Yogyakarta Outline Definition Epidemiology Physiology Classes of Shock
SUMMARY OF PRODUCT CHARACTERISTICS. Flexbumin 200 g/l is a solution containing 200 g/l (20%) of total protein of which at least 95% is human albumin.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Flexbumin 200g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Flexbumin 200 g/l is a solution containing 200 g/l
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Flexbumin 200g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Flexbumin 200 g/l is a solution containing 200 g/l
Surgical Care at the District Hospital. EMERGENCY & ESSENTIAL SURGICAL CARE
 Surgical Care at the District Hospital 1 13 Resuscitation and Preparation for Anesthesia & Surgery Key Points 2 13.1 Management of Emergencies and Cardiopulmonary Resuscitation The emergency measures that
Surgical Care at the District Hospital 1 13 Resuscitation and Preparation for Anesthesia & Surgery Key Points 2 13.1 Management of Emergencies and Cardiopulmonary Resuscitation The emergency measures that
3. PHARMACEUTICAL FORM Solution for infusion. A clear, slightly viscous liquid; it is almost colourless, yellow, amber or green.
 1. NAME OF THE MEDICINAL PRODUCT Albutein 250 g/l, solution for infusion. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Human albumin Albutein 250 g/l is a solution containing 250 g/l of total protein of
1. NAME OF THE MEDICINAL PRODUCT Albutein 250 g/l, solution for infusion. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Human albumin Albutein 250 g/l is a solution containing 250 g/l of total protein of
Introduction to Emergency Medical Care 1
 Introduction to Emergency Medical Care 1 OBJECTIVES 6.1 Define key terms introduced in this chapter. Slides 11, 15, 17, 26, 27, 31, 33, 37, 40 42, 44, 45, 51, 58 6.2 Describe the basic roles and structures
Introduction to Emergency Medical Care 1 OBJECTIVES 6.1 Define key terms introduced in this chapter. Slides 11, 15, 17, 26, 27, 31, 33, 37, 40 42, 44, 45, 51, 58 6.2 Describe the basic roles and structures
Fluid Management in Dengue Fever and Dengue Haemorrhagic Fever
 Fluid Management in Dengue Fever and Dengue Haemorrhagic Fever Dengue infection Dr. A LakKumar Fernando, Consultant Paediatrician Dengue is a disease which is silently transmitted in the community. For
Fluid Management in Dengue Fever and Dengue Haemorrhagic Fever Dengue infection Dr. A LakKumar Fernando, Consultant Paediatrician Dengue is a disease which is silently transmitted in the community. For
Transfusion Requirements and Management in Trauma RACHEL JACK
 Transfusion Requirements and Management in Trauma RACHEL JACK Overview Haemostatic resuscitation Massive Transfusion Protocol Overview of NBA research guidelines Haemostatic resuscitation Permissive hypotension
Transfusion Requirements and Management in Trauma RACHEL JACK Overview Haemostatic resuscitation Massive Transfusion Protocol Overview of NBA research guidelines Haemostatic resuscitation Permissive hypotension
Australian College of Veterinary Scientists. Membership Examinationn. Veterinary Emergency and Critical Care Paper 1
 Australian College of Veterinary Scientists Membership Examinationn June 2011 Veterinary Emergency and Critical Care Paper 1 Perusal time: Fifteen (15) minutes Time allowed: Two (2) hours after perusal
Australian College of Veterinary Scientists Membership Examinationn June 2011 Veterinary Emergency and Critical Care Paper 1 Perusal time: Fifteen (15) minutes Time allowed: Two (2) hours after perusal
INTRAVENOUS FLUIDS PRINCIPLES
 INTRAVENOUS FLUIDS PRINCIPLES Postnatal physiological weight loss is approximately 5-10% Postnatal diuresis is delayed in Respiratory Distress Syndrome (RDS) Preterm babies have limited capacity to excrete
INTRAVENOUS FLUIDS PRINCIPLES Postnatal physiological weight loss is approximately 5-10% Postnatal diuresis is delayed in Respiratory Distress Syndrome (RDS) Preterm babies have limited capacity to excrete
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Albunorm 5%, 50 g/l, solution for infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Albunorm 5% is a solution containing 50 g/l of total
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Albunorm 5%, 50 g/l, solution for infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Albunorm 5% is a solution containing 50 g/l of total
Brief summary of the NICE guidelines December 2013
 Brief summary of the NICE guidelines December 2013 Intravenous fluid therapy in adults in hospital the relevance to Emergency Department Care Applicable to patients 16 years and older receiving i.v. fluids
Brief summary of the NICE guidelines December 2013 Intravenous fluid therapy in adults in hospital the relevance to Emergency Department Care Applicable to patients 16 years and older receiving i.v. fluids
Acute Kidney Injury. Eleanor Haskey BSc(hons) RVN VTS(ECC) VPAC A1
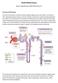 Acute Kidney Injury Eleanor Haskey BSc(hons) RVN VTS(ECC) VPAC A1 Anatomy and Physiology The role of the kidneys is to filter the blood through the glomerulus to form filtrate. The filtrate is then reabsorbed
Acute Kidney Injury Eleanor Haskey BSc(hons) RVN VTS(ECC) VPAC A1 Anatomy and Physiology The role of the kidneys is to filter the blood through the glomerulus to form filtrate. The filtrate is then reabsorbed
Fluid assessment, monitoring and therapy for the acute nurse
 Fluid assessment, monitoring and therapy for the acute nurse Kelly Wright Lead Nurse for AKI King s College Hospital Aims and objectives Aims and objectives Why do we worry about volume assessment? Completing
Fluid assessment, monitoring and therapy for the acute nurse Kelly Wright Lead Nurse for AKI King s College Hospital Aims and objectives Aims and objectives Why do we worry about volume assessment? Completing
HYPOVOLEMIA AND HEMORRHAGE UPDATE ON VOLUME RESUSCITATION HEMORRHAGE AND HYPOVOLEMIA DISTRIBUTION OF BODY FLUIDS 11/7/2015
 UPDATE ON VOLUME RESUSCITATION HYPOVOLEMIA AND HEMORRHAGE HUMAN CIRCULATORY SYSTEM OPERATES WITH A SMALL VOLUME AND A VERY EFFICIENT VOLUME RESPONSIVE PUMP. HOWEVER THIS PUMP FAILS QUICKLY WITH VOLUME
UPDATE ON VOLUME RESUSCITATION HYPOVOLEMIA AND HEMORRHAGE HUMAN CIRCULATORY SYSTEM OPERATES WITH A SMALL VOLUME AND A VERY EFFICIENT VOLUME RESPONSIVE PUMP. HOWEVER THIS PUMP FAILS QUICKLY WITH VOLUME
Staging Sepsis for the Emergency Department: Physician
 Staging Sepsis for the Emergency Department: Physician Sepsis Continuum 1 Sepsis Continuum SIRS = 2 or more clinical criteria, resulting in Systemic Inflammatory Response Syndrome Sepsis = SIRS + proven/suspected
Staging Sepsis for the Emergency Department: Physician Sepsis Continuum 1 Sepsis Continuum SIRS = 2 or more clinical criteria, resulting in Systemic Inflammatory Response Syndrome Sepsis = SIRS + proven/suspected
INTRAVENOUS FLUID THERAPY
 INTRAVENOUS FLUID THERAPY PRINCIPLES Postnatal physiological weight loss is approximately 5 10% in first week of life Preterm neonates have more total body water and may lose 10 15% of their weight in
INTRAVENOUS FLUID THERAPY PRINCIPLES Postnatal physiological weight loss is approximately 5 10% in first week of life Preterm neonates have more total body water and may lose 10 15% of their weight in
(human albumin solution) POM SUMMARY OF PRODUCT CHARACTERISTICS
 albunorm TM 20% (human albumin solution) POM SUMMARY OF PRODUCT CHARACTERISTICS UK IRELAND and Ireland Octapharma Limited The Zenith Building, 26 Spring Gardens Manchester M2 1AB United Kingdom 1. Name
albunorm TM 20% (human albumin solution) POM SUMMARY OF PRODUCT CHARACTERISTICS UK IRELAND and Ireland Octapharma Limited The Zenith Building, 26 Spring Gardens Manchester M2 1AB United Kingdom 1. Name
How and why I give IV fluid Disclosures SCA Fluids and public health 4/1/15. Andrew Shaw MB FRCA FCCM FFICM
 How and why I give IV fluid Andrew Shaw MB FRCA FCCM FFICM Professor and Chief Cardiothoracic Anesthesiology Vanderbilt University Medical Center 2015 Disclosures Consultant for Grifols manufacturer of
How and why I give IV fluid Andrew Shaw MB FRCA FCCM FFICM Professor and Chief Cardiothoracic Anesthesiology Vanderbilt University Medical Center 2015 Disclosures Consultant for Grifols manufacturer of
Exotic Animal Physical Exams and Nursing
 Exotic Animal Physical Exams and Nursing By: Stephen Cital RVT, RLAT, SRA www.stephencital.com Signalment Complete description of the animal Species, Breed, Age, Sex, Reproductive status, other distinguishing
Exotic Animal Physical Exams and Nursing By: Stephen Cital RVT, RLAT, SRA www.stephencital.com Signalment Complete description of the animal Species, Breed, Age, Sex, Reproductive status, other distinguishing
Unit 5: Blood Transfusion
 Unit 5: Blood Transfusion Blood transfusion (BT) therapy: Involves transfusing whole blood or blood components (specific portion or fraction of blood lacking in patient). Learn the concepts behind blood
Unit 5: Blood Transfusion Blood transfusion (BT) therapy: Involves transfusing whole blood or blood components (specific portion or fraction of blood lacking in patient). Learn the concepts behind blood
(human albumin solution) POM SUMMARY OF PRODUCT CHARACTERISTICS
 albunorm TM 5% (human albumin solution) POM SUMMARY OF PRODUCT CHARACTERISTICS UK IRELAND and Ireland Octapharma Limited The Zenith Building, 26 Spring Gardens Manchester M2 1AB United Kingdom 1. Name
albunorm TM 5% (human albumin solution) POM SUMMARY OF PRODUCT CHARACTERISTICS UK IRELAND and Ireland Octapharma Limited The Zenith Building, 26 Spring Gardens Manchester M2 1AB United Kingdom 1. Name
Daniel A. Beals MD, FACS, FAAP Pediatric Surgery and Urology Community Medical Center Associate Professor of Surgery and Pediatrics University of
 Daniel A. Beals MD, FACS, FAAP Pediatric Surgery and Urology Community Medical Center Associate Professor of Surgery and Pediatrics University of Washington Seattle Children s Hospital Objectives Define
Daniel A. Beals MD, FACS, FAAP Pediatric Surgery and Urology Community Medical Center Associate Professor of Surgery and Pediatrics University of Washington Seattle Children s Hospital Objectives Define
Proceeding of the LAVC Latin American Veterinary Conference Oct , 2011 Lima, Peru
 Close this window to return to IVIS www.ivis.org Proceeding of the LAVC Latin American Veterinary Conference Oct. 24-26, 2011 Lima, Peru Next LAVC Conference: Apr. 24-26, 2012 Lima, Peru Reprinted in the
Close this window to return to IVIS www.ivis.org Proceeding of the LAVC Latin American Veterinary Conference Oct. 24-26, 2011 Lima, Peru Next LAVC Conference: Apr. 24-26, 2012 Lima, Peru Reprinted in the
Standard Operating Procedure (SOP) Management of intervention group patients SOP 001
 ` Standard Operating Procedure (SOP) Management of intervention group patients SOP 001 Authors: Mark Edwards & Rupert Pearse Authorisation: Rupert Pearse (Chief Investigator) Scope To provide guidance
` Standard Operating Procedure (SOP) Management of intervention group patients SOP 001 Authors: Mark Edwards & Rupert Pearse Authorisation: Rupert Pearse (Chief Investigator) Scope To provide guidance
Fine-tuning Management in Dengue Fever
 Fine-tuning Management in Dengue Fever Annual Scientific Meeting On Intensive Care 16 August 2014 Eg Kah Peng University of Malaya Medical Centre Rise In Dengue Infection New Straits Times 8 July 2014
Fine-tuning Management in Dengue Fever Annual Scientific Meeting On Intensive Care 16 August 2014 Eg Kah Peng University of Malaya Medical Centre Rise In Dengue Infection New Straits Times 8 July 2014
Care of the Deteriorating Patient in Recovery NADIA TICEHURST : CLINICAL NURSE EDUCATOR PERI ANAESTHETICS BENDIGO HEALTH
 Care of the Deteriorating Patient in Recovery NADIA TICEHURST : CLINICAL NURSE EDUCATOR PERI ANAESTHETICS BENDIGO HEALTH Intended learning outcomes Describe the components of a comprehensive clinician
Care of the Deteriorating Patient in Recovery NADIA TICEHURST : CLINICAL NURSE EDUCATOR PERI ANAESTHETICS BENDIGO HEALTH Intended learning outcomes Describe the components of a comprehensive clinician
Reverse (fluid) resuscitation Should we be doing it? NAHLA IRTIZA ISMAIL
 Reverse (fluid) resuscitation Should we be doing it? NAHLA IRTIZA ISMAIL 65 Male, 60 kg D1 in ICU Admitted from OT intubated Diagnosis : septic shock secondary to necrotising fasciitis of the R lower limb
Reverse (fluid) resuscitation Should we be doing it? NAHLA IRTIZA ISMAIL 65 Male, 60 kg D1 in ICU Admitted from OT intubated Diagnosis : septic shock secondary to necrotising fasciitis of the R lower limb
Proceeding of the LAVECCS
 Close this window to return to IVIS Proceeding of the LAVECCS Congreso Latinoamericano de Emergencia y Cuidados Intensivos Ju1. 28-30, 2011 Santiago de Chile, Chile www.laveccs.org Reprinted in IVIS with
Close this window to return to IVIS Proceeding of the LAVECCS Congreso Latinoamericano de Emergencia y Cuidados Intensivos Ju1. 28-30, 2011 Santiago de Chile, Chile www.laveccs.org Reprinted in IVIS with
SUMMARY OF PRODUCT CHARACTERISTICS. Albuman 40 g/l is a solution containing 40 g/l (4%) of total protein of which at least 95% is human albumin.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albuman 40 g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albuman 40 g/l is a solution containing 40 g/l (4%)
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albuman 40 g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albuman 40 g/l is a solution containing 40 g/l (4%)
ISOVALERIC ACIDAEMIA -ACUTE DECOMPENSATION (standard version)
 Contact Details Name: Hospital Telephone: This protocol has 5 pages ISOVALERIC ACIDAEMIA -ACUTE DECOMPENSATION (standard version) Please read carefully. Meticulous treatment is very important as there
Contact Details Name: Hospital Telephone: This protocol has 5 pages ISOVALERIC ACIDAEMIA -ACUTE DECOMPENSATION (standard version) Please read carefully. Meticulous treatment is very important as there
Pediatric Code Blue. Goals of Resuscitation. Focus Conference November Ensure organ perfusion
 Pediatric Code Blue Focus Conference November 2015 Duane C. Williams, MD Pediatric Critical Care Department of Pediatrics Children s Hospital of Richmond at VCU Goals of Resuscitation Ensure organ perfusion
Pediatric Code Blue Focus Conference November 2015 Duane C. Williams, MD Pediatric Critical Care Department of Pediatrics Children s Hospital of Richmond at VCU Goals of Resuscitation Ensure organ perfusion
Vasoactive Medications. Matthew J. Korobey Pharm.D., BCCCP Critical Care Clinical Specialist Mercy St. Louis
 Vasoactive Medications Matthew J. Korobey Pharm.D., BCCCP Critical Care Clinical Specialist Mercy St. Louis Objectives List components of physiology involved in blood pressure Review terminology related
Vasoactive Medications Matthew J. Korobey Pharm.D., BCCCP Critical Care Clinical Specialist Mercy St. Louis Objectives List components of physiology involved in blood pressure Review terminology related
How Normal Body Processes Are Altered By Disease and Injury
 1 Chapter 4, GENERAL PRINCIPLES OF PATHOPHYSIOLOGY. Part 1 How Normal Body Processes Are Altered By Disease and Injury 2 How Cells Respond to Change and Injury 3 Pathology & Pathophysiology : the study
1 Chapter 4, GENERAL PRINCIPLES OF PATHOPHYSIOLOGY. Part 1 How Normal Body Processes Are Altered By Disease and Injury 2 How Cells Respond to Change and Injury 3 Pathology & Pathophysiology : the study
Pediatric emergencies (SHOCK & COMA) Dr Mubarak Abdelrahman Assistant Professor Jazan University
 Pediatric emergencies (SHOCK & COMA) Dr Mubarak Abdelrahman Assistant Professor Jazan University SHOCK Definition: Shock is a syndrome = inability to provide sufficient oxygenated blood to tissues. Oxygen
Pediatric emergencies (SHOCK & COMA) Dr Mubarak Abdelrahman Assistant Professor Jazan University SHOCK Definition: Shock is a syndrome = inability to provide sufficient oxygenated blood to tissues. Oxygen
European Resuscitation Council
 European Resuscitation Council Incidence of Trauma in Childhood Leading cause of death and disability in children older than one year all over the world Structured approach Primary survey and resuscitation
European Resuscitation Council Incidence of Trauma in Childhood Leading cause of death and disability in children older than one year all over the world Structured approach Primary survey and resuscitation
Fundamentals of Pharmacology for Veterinary Technicians Chapter 19
 Figure 19-1 Figure 19-2A Figure 19-2B Figure 19-3 Figure 19-4A1 Figure 19-4A2 Figure 19-4B Figure 19-4C Figure 19-4D Figure 19-5 Figure 19-6A Figure 19-6B A Figure 19-7A B Figure 19-7B C Figure 19-7C D
Figure 19-1 Figure 19-2A Figure 19-2B Figure 19-3 Figure 19-4A1 Figure 19-4A2 Figure 19-4B Figure 19-4C Figure 19-4D Figure 19-5 Figure 19-6A Figure 19-6B A Figure 19-7A B Figure 19-7B C Figure 19-7C D
Post Resuscitation Care
 Princess Margaret Hospital f Children PAEDIATRIC ACUTE CARE GUIDELINE Post Resuscitation Care Scope (Staff): Scope (Area): All Emergency Department Clinicians Emergency Department This document should
Princess Margaret Hospital f Children PAEDIATRIC ACUTE CARE GUIDELINE Post Resuscitation Care Scope (Staff): Scope (Area): All Emergency Department Clinicians Emergency Department This document should
MANAGEMENT OF DENGUE INFECTION IN ADULTS (Revised 2 nd Edition) QUICK REFERENCE FOR HEALTHCARE PROVIDERS
 1 KEY MESSAGES Dengue is a dynamic disease and presented in three phases - febrile phase, critical phase and recovery phase. Clinical deterioration often occurs in the critical phase and is marked by plasma
1 KEY MESSAGES Dengue is a dynamic disease and presented in three phases - febrile phase, critical phase and recovery phase. Clinical deterioration often occurs in the critical phase and is marked by plasma
How Normal Body Processes Are Altered By Disease and Injury
 1 Chapter 4, General Principles of Pathophysiology Part 1 How Normal Body Processes Are Altered By Disease and Injury 2 How Cells Respond to Change and Injury 3 Pathology & Pathophysiology : the study
1 Chapter 4, General Principles of Pathophysiology Part 1 How Normal Body Processes Are Altered By Disease and Injury 2 How Cells Respond to Change and Injury 3 Pathology & Pathophysiology : the study
Chapter 8 ADMINISTRATION OF BLOOD COMPONENTS
 Chapter 8 ADMINISTRATION OF BLOOD COMPONENTS PRACTICE POINTS Give the right blood product to the right patient at the right time. Failure to correctly check the patient or the pack can be fatal. At the
Chapter 8 ADMINISTRATION OF BLOOD COMPONENTS PRACTICE POINTS Give the right blood product to the right patient at the right time. Failure to correctly check the patient or the pack can be fatal. At the
Major Haemorrhage Protocol. Commentary
 Hairmyres Hospital Monklands Hospital Wishaw General Hospital Major Haemorrhage Protocol Commentary N.B. There is a separate NHSL protocol for the Management of Obstetric Haemorrhage Authors Dr Tracey
Hairmyres Hospital Monklands Hospital Wishaw General Hospital Major Haemorrhage Protocol Commentary N.B. There is a separate NHSL protocol for the Management of Obstetric Haemorrhage Authors Dr Tracey
Bachelor of Chinese Medicine Shock
 BCM Year 2 Dr. Irene Ng Jan 28, 2003 9:30 am 1:00 pm Rm 004 UPB Bachelor of Chinese Medicine 2002 2003 Shock Learning objectives Be able to: know the definition of shock know the classification and causes
BCM Year 2 Dr. Irene Ng Jan 28, 2003 9:30 am 1:00 pm Rm 004 UPB Bachelor of Chinese Medicine 2002 2003 Shock Learning objectives Be able to: know the definition of shock know the classification and causes
CLINICAL GUIDELINES ID TAG
 CLINICAL GUIDELINES ID TAG Title: Author: Speciality / Division: Directorate: Guideline for the perioperative fluid management in children Kieran O Connor Anaesthetics ATICS Date Uploaded: 26/04/2016 Review
CLINICAL GUIDELINES ID TAG Title: Author: Speciality / Division: Directorate: Guideline for the perioperative fluid management in children Kieran O Connor Anaesthetics ATICS Date Uploaded: 26/04/2016 Review
Going on Bypass. What happens before, during and after CPB. Perfusion Dept. Royal Children s Hospital Melbourne, Australia
 Going on Bypass What happens before, during and after CPB. Perfusion Dept. Royal Children s Hospital Melbourne, Australia Circulation Brain Liver Kidneys Viscera Muscle Skin IVC, SVC Pump Lungs R.A. L.V.
Going on Bypass What happens before, during and after CPB. Perfusion Dept. Royal Children s Hospital Melbourne, Australia Circulation Brain Liver Kidneys Viscera Muscle Skin IVC, SVC Pump Lungs R.A. L.V.
Department of Pediatrics, Faculty of Medicine Siriraj Hospital, Mahidol University, Bangkok 10700, Thailand.
 SIRIRAJ MEDICAL LIBRARY SpecialIssue Clinical Practice Guide for the Management of Dengue Hemorrhagic Fever (DHF), Siriraj Hospital Kulkanya Chokephaibulkit, M.D., Wanee Wisuthsarewong, M.D., Gavivann
SIRIRAJ MEDICAL LIBRARY SpecialIssue Clinical Practice Guide for the Management of Dengue Hemorrhagic Fever (DHF), Siriraj Hospital Kulkanya Chokephaibulkit, M.D., Wanee Wisuthsarewong, M.D., Gavivann
Proceeding of the NAVC North American Veterinary Conference Jan. 8-12, 2005, Orlando, Florida
 Proceeding of the NAVC North American Veterinary Conference Jan. 8-12, 2005, Orlando, Florida Reprinted in the IVIS website with the permission of the NAVC http:/// The North American Veterinary Conference
Proceeding of the NAVC North American Veterinary Conference Jan. 8-12, 2005, Orlando, Florida Reprinted in the IVIS website with the permission of the NAVC http:/// The North American Veterinary Conference
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Alburex 5, 50 g/l, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Alburex 5 is a solution containing 50 g/l of total
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Alburex 5, 50 g/l, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Alburex 5 is a solution containing 50 g/l of total
Pediatric Shock. Hypovolemia. Sepsis. Most common cause of pediatric shock Small blood volumes (80cc/kg)
 Critical Concepts: Shock Inadequate peripheral perfusion where oxygen delivery does not meet metabolic demand Adult vs Pediatric Shock - Same causes/different frequencies Pediatric Shock Hypovolemia Most
Critical Concepts: Shock Inadequate peripheral perfusion where oxygen delivery does not meet metabolic demand Adult vs Pediatric Shock - Same causes/different frequencies Pediatric Shock Hypovolemia Most
Proceeding of the NAVC North American Veterinary Conference Jan. 8-12, 2005, Orlando, Florida
 Proceeding of the NAVC North American Veterinary Conference Jan. 8-12, 2005, Orlando, Florida Reprinted in the IVIS website with the permission of the NAVC http:/// The North American Veterinary Conference
Proceeding of the NAVC North American Veterinary Conference Jan. 8-12, 2005, Orlando, Florida Reprinted in the IVIS website with the permission of the NAVC http:/// The North American Veterinary Conference
Human Albumin 200 g/l Baxter is a solution containing 200 g/l of total protein of which at least 95% is human albumin.
 1. NAME OF THE MEDICINAL PRODUCT Human Albumin 200 g/l Baxter Solution for Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Human Albumin 200 g/l Baxter is a solution containing 200 g/l of total protein
1. NAME OF THE MEDICINAL PRODUCT Human Albumin 200 g/l Baxter Solution for Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Human Albumin 200 g/l Baxter is a solution containing 200 g/l of total protein
Shock Quiz! By Clare Di Bona
 Shock Quiz! By Clare Di Bona Test Question What is Mr Burns full legal name? Answer Charles Montgomery Plantagenet Schicklgruber Burns. (Season 22, episode 11) Question 1. What is the definition of shock?
Shock Quiz! By Clare Di Bona Test Question What is Mr Burns full legal name? Answer Charles Montgomery Plantagenet Schicklgruber Burns. (Season 22, episode 11) Question 1. What is the definition of shock?
BREAK 11:10-11:
 1. Sepsis Tom Heaps 09:30-10:20 2. Oncological Emergencies Clare Pollard 10:20-11:10 ------------------------ BREAK 11:10-11:30 ------------------------ 3. Diabetic Ketoacidosis Tom Heaps 11:30-12:20 4.
1. Sepsis Tom Heaps 09:30-10:20 2. Oncological Emergencies Clare Pollard 10:20-11:10 ------------------------ BREAK 11:10-11:30 ------------------------ 3. Diabetic Ketoacidosis Tom Heaps 11:30-12:20 4.
Presented by: Indah Dwi Pratiwi
 Presented by: Indah Dwi Pratiwi Normal Fluid Requirements Resuscitation Fluids Goals of Resuscitation Maintain normal body temperature In most cases, elevate the feet and legs above the level of the heart
Presented by: Indah Dwi Pratiwi Normal Fluid Requirements Resuscitation Fluids Goals of Resuscitation Maintain normal body temperature In most cases, elevate the feet and legs above the level of the heart
Case TWO. Vital Signs: Temperature 36.6degC BP 137/89 HR 110 SpO2 97% on Room Air
 Mr N is a 64year old Chinese gentleman who is a heavy drinker, still actively drinking, and chronic smoker of >40pack year history. He has a past medical history significant for Hypertension, Hyperlipidemia,
Mr N is a 64year old Chinese gentleman who is a heavy drinker, still actively drinking, and chronic smoker of >40pack year history. He has a past medical history significant for Hypertension, Hyperlipidemia,
Circulatory shock. Types, Etiology, Pathophysiology. Physiology of Circulation: The Vessels. 600,000 miles of vessels containing 5-6 liters of blood
 Circulatory shock Types, Etiology, Pathophysiology Blagoi Marinov, MD, PhD Pathophysiology Dept. Physiology of Circulation: The Vessels 600,000 miles of vessels containing 5-6 liters of blood Vessel tone
Circulatory shock Types, Etiology, Pathophysiology Blagoi Marinov, MD, PhD Pathophysiology Dept. Physiology of Circulation: The Vessels 600,000 miles of vessels containing 5-6 liters of blood Vessel tone
Feline Anesthesia Fluid Therapy Treatment of Anesthetic Complications
 Feline Anesthesia Fluid Therapy Treatment of Anesthetic Complications Rebecca A. Krimins, DVM, MS April 29, 2018 DCVR Annual Spring Symposium Email: drkrimins@gmail.com Cat Anesthesia There is no single
Feline Anesthesia Fluid Therapy Treatment of Anesthetic Complications Rebecca A. Krimins, DVM, MS April 29, 2018 DCVR Annual Spring Symposium Email: drkrimins@gmail.com Cat Anesthesia There is no single
DOCUMENT CONTROL PAGE
 DOCUMENT CONTROL PAGE Title Title: UNDERGOING SPINAL DEFORMITY SURGERY Version: 2 Reference Number: Supersedes Supersedes: all other versions Description of Amendment(s): Revision of analgesia requirements
DOCUMENT CONTROL PAGE Title Title: UNDERGOING SPINAL DEFORMITY SURGERY Version: 2 Reference Number: Supersedes Supersedes: all other versions Description of Amendment(s): Revision of analgesia requirements
Fluids in Sepsis: How much and what type? John Fowler, MD, FACEP Kent Hospital, İzmir Eisenhower Medical Center, USA American Hospital Dubai, UAE
 Fluids in Sepsis: How much and what type? John Fowler, MD, FACEP Kent Hospital, İzmir Eisenhower Medical Center, USA American Hospital Dubai, UAE In critically ill patients: too little fluid Low preload,
Fluids in Sepsis: How much and what type? John Fowler, MD, FACEP Kent Hospital, İzmir Eisenhower Medical Center, USA American Hospital Dubai, UAE In critically ill patients: too little fluid Low preload,
Acute Kidney Injury (AKI) Undergraduate nurse education
 Acute Kidney Injury (AKI) Undergraduate nurse education Year Three Developed Summer 2017 Objectives Understand Acute Kidney Injury and its relevance to patient care. Brief revision of the Anatomy and physiology
Acute Kidney Injury (AKI) Undergraduate nurse education Year Three Developed Summer 2017 Objectives Understand Acute Kidney Injury and its relevance to patient care. Brief revision of the Anatomy and physiology
Amjad Bani Hani Ass.Prof. of Cardiac Surgery & Intensive Care FLUIDS AND ELECTROLYTES
 Amjad Bani Hani Ass.Prof. of Cardiac Surgery & Intensive Care FLUIDS AND ELECTROLYTES Body Water Content Water Balance: Normal 2500 2000 1500 1000 500 Metab Food Fluids Stool Breath Sweat Urine
Amjad Bani Hani Ass.Prof. of Cardiac Surgery & Intensive Care FLUIDS AND ELECTROLYTES Body Water Content Water Balance: Normal 2500 2000 1500 1000 500 Metab Food Fluids Stool Breath Sweat Urine
TRIAGE AND INITIAL ASSESSMENT. Elisa A. Rogers CVT, VTS(ECC) MJR Veterinary Hospital University of Pennsylvania Philadelphia Pa
 TRIAGE AND INITIAL ASSESSMENT Elisa A. Rogers CVT, VTS(ECC) MJR Veterinary Hospital University of Pennsylvania Philadelphia Pa Triage and Initial Assessment An emergency can be described as any situation
TRIAGE AND INITIAL ASSESSMENT Elisa A. Rogers CVT, VTS(ECC) MJR Veterinary Hospital University of Pennsylvania Philadelphia Pa Triage and Initial Assessment An emergency can be described as any situation
Division 1 Introduction to Advanced Prehospital Care
 Division 1 Introduction to Advanced Prehospital Care Topics Fluids & fluid imbalances IV Therapy Hypoperfusion Shock Fluids and Fluid Imbalances 1 Water is the most abundant substance in the human body.
Division 1 Introduction to Advanced Prehospital Care Topics Fluids & fluid imbalances IV Therapy Hypoperfusion Shock Fluids and Fluid Imbalances 1 Water is the most abundant substance in the human body.
12/29/2014. IV/IO Therapy & Fluid Administration. Objectives. Cleansing of the soul
 IV/IO Therapy & Fluid Administration Gary Hoertz, EMT-P Spokane County EMS Indications for IV Access Types of Intravenous Access IV fluids Flow Rates Fluid resuscitation Objectives Cleansing of the soul
IV/IO Therapy & Fluid Administration Gary Hoertz, EMT-P Spokane County EMS Indications for IV Access Types of Intravenous Access IV fluids Flow Rates Fluid resuscitation Objectives Cleansing of the soul
ELECTROLYTES RENAL SHO TEACHING
 ELECTROLYTES RENAL SHO TEACHING Metabolic Alkalosis 2 factors are responsible for generation and maintenance of metabolic alkalosis this includes a process that raises serum bicarbonate and a process that
ELECTROLYTES RENAL SHO TEACHING Metabolic Alkalosis 2 factors are responsible for generation and maintenance of metabolic alkalosis this includes a process that raises serum bicarbonate and a process that
Maternal Collapse Guideline
 Maternal Collapse Guideline Guideline Number: 664 Supersedes: Classification Clinical Version No: Date of EqIA: Approved by: Date Approved: Date made active: Review Date: 1 Obstetric Written Documentation
Maternal Collapse Guideline Guideline Number: 664 Supersedes: Classification Clinical Version No: Date of EqIA: Approved by: Date Approved: Date made active: Review Date: 1 Obstetric Written Documentation
INTENSIVE CARE MEDICINE CPD EVENING. Dr Alastair Morgan Wednesday 13 th September 2017
 INTENSIVE CARE MEDICINE CPD EVENING Dr Alastair Morgan Wednesday 13 th September 2017 WHAT IS NEW IN ICU? (RELEVANT TO ANAESTHETISTS) Not much! SURVIVING SEPSIS How many deaths in England were thought
INTENSIVE CARE MEDICINE CPD EVENING Dr Alastair Morgan Wednesday 13 th September 2017 WHAT IS NEW IN ICU? (RELEVANT TO ANAESTHETISTS) Not much! SURVIVING SEPSIS How many deaths in England were thought
Date written: April 2014 Review date: April 2016 Related documents: Paediatric Sepsis 6
 Scottish Paediatric Retrieval Service (Edinburgh) www.paedsretrieval.com Clinical Guideline SEPSIS Date written: April 2014 Review date: April 2016 Related documents: Paediatric Sepsis 6 Author: Steve
Scottish Paediatric Retrieval Service (Edinburgh) www.paedsretrieval.com Clinical Guideline SEPSIS Date written: April 2014 Review date: April 2016 Related documents: Paediatric Sepsis 6 Author: Steve
SHOCK. May 12, 2011 Body and Disease
 SHOCK May 12, 2011 Body and Disease Shock Definition of shock Pathophysiology Types of shock Management of shock Shock Definition? Shock What the Duke Community would have experienced if Gordon Hayward
SHOCK May 12, 2011 Body and Disease Shock Definition of shock Pathophysiology Types of shock Management of shock Shock Definition? Shock What the Duke Community would have experienced if Gordon Hayward
There are number of parameters which are measured: ph Oxygen (O 2 ) Carbon Dioxide (CO 2 ) Bicarbonate (HCO 3 -) AaDO 2 O 2 Content O 2 Saturation
 Arterial Blood Gases (ABG) A blood gas is exactly that...it measures the dissolved gases in your bloodstream. This provides one of the best measurements of what is known as the acid-base balance. The body
Arterial Blood Gases (ABG) A blood gas is exactly that...it measures the dissolved gases in your bloodstream. This provides one of the best measurements of what is known as the acid-base balance. The body
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albunorm 20%, 200g/l, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albunorm 20% is a solution containing 200 g/l
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albunorm 20%, 200g/l, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albunorm 20% is a solution containing 200 g/l
The Septic Patient. Dr Arunraj Navaratnarajah. Renal SpR Imperial College NHS Healthcare Trust
 The Septic Patient Dr Arunraj Navaratnarajah Renal SpR Imperial College NHS Healthcare Trust Objectives of this session Define SIRS / sepsis / severe sepsis / septic shock Early recognition of Sepsis The
The Septic Patient Dr Arunraj Navaratnarajah Renal SpR Imperial College NHS Healthcare Trust Objectives of this session Define SIRS / sepsis / severe sepsis / septic shock Early recognition of Sepsis The
Australian and New Zealand College of Veterinary Scientists. Membership Examination. Veterinary Emergency and Critical Care Paper 1
 Australian and New Zealand College of Veterinary Scientists Membership Examination June 2017 Veterinary Emergency and Critical Care Paper 1 Perusal time: Fifteen (15) minutes Time allowed: Two (2) hours
Australian and New Zealand College of Veterinary Scientists Membership Examination June 2017 Veterinary Emergency and Critical Care Paper 1 Perusal time: Fifteen (15) minutes Time allowed: Two (2) hours
Module D Sepsis, Septic Shock, and DIC NUR 203. Page 1 of 24
 Module D Sepsis, Septic Shock, and DIC NUR 203 Page 1 of 24 Shock Patho: a response to poor tissue oxygenation Etiology: Any problem that impairs oxygen delivery to tissues and organs can start the syndrome
Module D Sepsis, Septic Shock, and DIC NUR 203 Page 1 of 24 Shock Patho: a response to poor tissue oxygenation Etiology: Any problem that impairs oxygen delivery to tissues and organs can start the syndrome
