PERIPHERAL US PRODUCT
|
|
|
- Esther Joy Dixon
- 6 years ago
- Views:
Transcription
1 PERIPHERAL US PRODUCT CATALOG July
2 TABLE OF CONTENTS DIRECTIONAL ATHERECTOMY SYSTEMS HawkOne Directional Atherectomy System TurboHawk Peripheral Plaque Excision System SilverHawk Peripheral Plaque Excision System DRUG-COATED BALLOON IN.PACT Admiral Paclitaxel-Coated PTA Balloon Catheter EMBOLIC PROTECTION SpiderFX Embolic Protection Device MO.MA Ultra Proximal Cerebral Protection Device CTO DEVICES Viance Crossing Catheter Enteer Re-entry Catheter Enteer Guidewire STENTS SELF-EXPANDING STENT SYSTEMS EverFlex Self-expanding Peripheral Stent with Entrust Delivery System EverFlex Self-expanding Peripheral Stent System Protégé EverFlex Self-expanding Biliary Stent System Protégé GPS Self-expanding Peripheral and Biliary Stent System Protégé RX Self-expanding Carotid Stent System BALLOON EXPANDABLE STENT SYSTEMS Visi-Pro Balloon-expandable Peripheral Stent System ParaMount Mini GPS Balloon-expandable Biliary Stent System UNMOUNTED STENTS IntraStent DoubleStrut LD Biliary Stent IntraStent Mega LD Biliary Stent IntraStent Max LD Biliary Stent PTA BALLOONS EverCross OTW PTA Balloon Catheter Admiral Xtreme PTA Balloon Catheter Fortrex 0.035" OTW HP PTA Balloon Catheter Pacific Plus PTA Catheter OTW 0.018" Pacific Xtreme PTA Balloon Catheter OTW 0.018" NanoCross Elite 0.014" OTW PTA Balloon Catheter RapidCross PTA Rapid Exchange Balloon Dilatation Catheter Chocolate * PTA Balloon INFUSION THERAPY Cragg-McNamara Valved Infusion Catheters ProStream Infusion Wires MicroMewi Infusion Catheters SNARES Amplatz Goose Neck Snare Kit Amplatz Goose Neck MicroSnare Kit Snare Replacement Catheters GUIDEWIRES Wholey Guidewire System Babywire Double-Ended Nitinol Guidewire Nitrex Guidewire SUPPORT CATHETERS, Y-CONNECTORS & GUIDE CATHETERS TrailBlazer Support Catheter TrailBlazer Angled Support Catheter BigEasy Rotating Y-Connector Sequel Rotating Double Y-Connector Launcher Peripheral Guide Catheter BRIEF STATEMENTS & REFERENCE STATEMENTS
3 DIRECTIONAL ATHERECTOMY HawkOne Directional Atherectomy System 3
4 HawkOne Directional Atherectomy System The HawkOne device is the latest addition to Medtronic s directional atherectomy portfolio, which restores blood flow in PAD patients by removing plaque from blocked arteries. Just as the name implies, the HawkOne device is a comprehensive system that treats all morphologies, including severe calcium, and offers procedural efficiency with enhanced cutting, crossing and cleaning capabilities. Model Name Vessel Diameter Sheath Compatibility (F) Crossing Profile Working 1 Effective 2 Tip Max. Cut Packing Device 7 F HawkOne LS Standard Tip H1-LS 3.5 to F HawkOne LX Extended Tip H1-LX 3.5 to F HawkOne LS Standard Tip H1-M 3.0 to F HawkOne LX Extended Tip H1-S 2.0 to Max guidewire is for HawkOne device. 1 HawkOne Working Distal end of pre-loaded flush tool, in the proximal position, to the distal end of tip. 2 HawkOne Effective Distal end of pre-loaded flush tool, in the proximal position, to the proximal end of cutter window. 4
5 TurboHawk Peripheral Plaque Excision System The TurboHawk plaque excision system treats PAD by removing soft to moderately calcified plaque buildup in leg arteries. This second generation device uses a directional cutting blade to shave plaque from the vessel maximizing luminal gain. The plaque is captured in the nosecone and safely removed from the vessel. Product Name Vessel Diameter Sheath Compatibility 1 (F) Crossing Profile Working 2 Effective 3 Tip Max Cut Packing Device LX-C THS-LX-C 3.5 to LX-M TH-LX-M 3.5 to 7.0 7/ SX-C THS-SX-C 2.0 to SS-CL THS-SS-CL 2.0 to Max guidewire is for all TurboHawk devices. 1 Sheath Compatibility - Per the Instructions For Use, the large vessel devices with smooth cutter are compatible with 8 F sheaths. A physician survey of device usage indicated 7 F sheaths may have an Internal Diameter (ID) that will accommodate the crossing profile of the LX-M device. Data on file with manufacturer. 2 Working - distal end of strain relief to the distal end of tip. 3 Effective - distal end of strain relief to the proximal end of cutter window. 5
6 SilverHawk Peripheral Plaque Excision System The first-generation SilverHawk plaque excision system treats peripheral arterial disease (PAD) by removing soft-to-mild plaque buildup in leg arteries. SilverHawk system technology uses a directional cutting blade to shave plaque from the vessel maximizing luminal gain. The plaque is captured in the nosecone and safely removed from the vessel. Product Name Vessel Diameter Sheath Compatibility 1 (F) Crossing Profile Working 2 Effective 3 Tip Max Cut Packing Device LX-M P to 6.5 7/ MS-M P to 5.0 7/ SS+ P to EXL P to DS P to Max guidewire is for all SilverHawk devices. 1 Sheath Compatibility - Per the Instructions For Use, the medium vessel and large vessel devices are compatible with 8 F sheaths. A physician survey of device usage indicated 7 F sheaths may have an Internal Diameter (ID) that will accommodate the crossing profile of the LX-M and MS-M (non-flush tip) devices. Data on file with manufacturer. 2 Working - distal end of strain relief to the distal end of tip. 3 Effective - distal end of strain relief to the proximal end of cutter window. 6
7 DRUG-COATED BALLOON IN.PACT Admiral Drug-Coated Balloon 7
8 IN.PACT Admiral Drug-Coated Balloon The IN.PACT Admiral Drug-Coated Balloon (DCB) is a clinically proven, primary endovascular therapy that treats femoropopliteal disease, reduces interventions, and preserves future treatment options. TECHNICAL SPECIFICATIONS Paclitaxel drug dose 3.5 μg/mm 2 Excipient Balloon diameters Balloon lengths Balloon fold configuration Catheter design Catheter lengths Urea 4 mm - 7 mm Guidewire compatibility Nominal balloon pressure 40, 60, 80, 120, 150 mm* 4.0 mm: 3 Folds 5.0, 6.0 and 7.0 mm: 6 Folds Over the Wire (OTW) 80 cm and 130 cm 8 atm * NOTE: 120 mm and 150 mm lengths are not offered on the 7.0 mm diameter balloon. Usable 80 cm Usable 130 cm Balloon Diameter Balloon Recommended Introducer Sheath (F) Nominal Pressure (atm) RBP (atm) ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P ADM P
9 EMBOLIC PROTECTION SpiderFX Embolic Protection Device MO.MA Ultra Proximal Cerebral Protection Device 9
10 SpiderFX Embolic Protection Device The SpiderFX device is used to capture and remove debris that becomes dislodged during an interventional procedure. The SpiderFX device is the only embolic protection device that can be delivered over any 0.014" or 0.018" guidewire, or through any 0.035" catheter* for ease of lesion crossing. The SpiderFX device has the broadest indication among distal embolic filters. It is indicated for use in carotid arteries, coronary saphenous vein bypass grafts, and lower extremity procedures. *Lower extremity procedures. Capture Wire Delivery End Recovery End Guide Catheter/ Sheath Filter Size Target Vessel Size Wire OTW/RX Wire Diameter (in/mm) Crossing Profile (F) Diameter (F) Minimum ID (in) SPD2-US SVG & Carotid / SPD2-US SVG & Carotid 320/ / SPD2-US SVG & Carotid / SPD2-US SVG & Carotid 320/ / SPD2-US SPD2-US SPD2-US SPD2-US SPD2-US SPD2-US SVG & Carotid Lower Extremity SVG & Carotid Lower Extremity SVG & Carotid Lower Extremity SVG & Carotid Lower Extremity SVG Carotid Lower Extremity SVG Carotid Lower Extremity / / / / / / / / /
11 MO.MA Ultra Proximal Cerebral Protection Device Use the MO.MA Ultra proximal cerebral protection device to contain and remove all sizes of debris that can dislodge during interventional procedures in the carotid arteries. The Mo.Ma ultra device with double-occlusion balloon system allows for proximal embolic protection to be established prior to crossing a carotid lesion. TECHNICAL SPECIFICATIONS Balloon material Balloon marker distance Recommended guidewire Balloon occlusion range Compliant elastomeric rubber 6 cm (0.89 mm) 5-13 mm diameter (CCA prox. balloon) 3-6 mm diameter (ECA dist. balloon) Minimum Sheath Size (F) Inner Diameter of the Working Channel MUS X "/2.12mm 11
12 CTO DEVICES Enteer Re-entry System Viance Crossing Catheter 12
13 Viance Crossing Catheter The Viance catheter is designed to quickly and safely deliver a guidewire via the true lumen. The low-profile Viance catheter with its fast-spin torque handle enables the physician to find small microchannels in a lesion. It is constructed of a multiwire coiled shaft for 1:1 torque and an atraumatic distal tip designed for quick and safe guidewire delivery. Description Working Guidewire Compatibility (in) Crossing Profile (max in) Sheath Compatibility VNC-FX-150 Flexible F VNC-SD-150 Standard F Enteer Re-entry System The Enteer system, consisting of the Enteer re-entry balloon catheter and Enteer guidewire, enables the physician to reliably target the true lumen from the subintimal channel above or below the knee. The distinctive shape of the Enteer balloon causes it to self-orient within a vessel, helping provide reliable true lumen targeting without complete reliance on visualization. Due to its unique design, one flat side of the balloon orients to the true lumen when inflated. Offset exit ports are located on each flat side, allowing the guidewire to be steered through the correct port for successful re-entry. Re-entry Balloon Catheter Balloon Size (W x H X L mm) Working Guidewire Compatibility (in) Crossing Profile (max in) Sheath Compatibility ENB ATK 3.75 x 1.5 x F ENB BTK 2.75 x 1.0 x F Guidewire Description Diameter (in) Tip Reach ENW-FX Flexible ENW-SD Standard ENW-SF Stiff
14 STENTS EverFlex Self-expanding Peripheral Stent System Visi-Pro Balloon-expandable Peripheral Stent System 14
15 EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust delivery system is a one handed stent delivery system with a low 5 F profile. This low profile was achieved without compromising the design of the EverFlex stent or the 0.035" guidewire compatibility. The device was engineered specifically for control and accuracy based on physician feedback during extensive interviews and procedural observations. 80 cm CATHETER LENGTH STENT SIZE COMPATIBILITY 120 cm 150 cm Diameter Recomm. Introducer Sheath (F) Guidewire (in) Recomm. Lumen Size EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD EVD
16 EverFlex Self-expanding Peripheral Stent System The EverFlex stent system is designed for flexibility and durability. It is a second generation stent indicated for use in the superficial femoral and proximal popliteal arteries (SFA/PPA), common iliac, and/or external iliac arteries. The EverFlex stent is available in sizes ranging from 20 mm to 200 mm lengths (SFA/PPA) or mm lengths (iliac). The broad size matrix, all deliverable through a 6 F catheter, provides the most appropriate single-stent fit. Peak-to-peak connection nodes disperse force uniformly enhancing durability while the spiral-cell connection and three-wave peak design optimizes flexibility. 80 cm CATHETER LENGTH STENT SIZE COMPATIBILITY 120 cm Each system includes one stent and delivery catheter system. Diameter Recomm. Introducer Sheath (F) Guidewire (in) Recomm. Lumen Size PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB N/A PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB N/A PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB N/A PRB
17 Protégé EverFlex Self-expanding Biliary Stent System The Protégé EverFlex stent system is designed for the palliative treatment of malignant neoplasms in the biliary tree. The stent is made of nitinol and comes pre-mounted on an over-the-wire delivery system. The proximal and distal ends of the stent have tantalum radiopaque markers for enhanced visibility. Unconstrained Stent Diameter Unconstrained Stent Rec. Lumen Size Usable Sheath Size (F) Guidewire Acceptance (in) Outside Diameter (in) PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB PRB Each system includes one stent and delivery catheter system. 17
18 Protégé GPS Self-expanding Peripheral and Biliary Stent System The Protégé GPS self-expanding peripheral and biliary stent system is a self-expanding nitinol stent system indicated for the common and external iliac arteries, and designed for the palliative treatment of malignant neoplasms in the biliary tree. The stent is made of a nickel titanium alloy (nitinol) and comes pre-mounted on a 6 F over-the-wire delivery system. The stent is cut from a nitinol tube into an open lattice design and has tantalum radiopaque markers at the proximal and distal ends of the stent. Upon deployment, the stent achieves its predetermined diameter and exerts a constant, outward force. Unconstrained Stent Diameter Unconstrained Stent Each system includes one stent and delivery catheter system. Rec. Lumen Size Usable Sheath Size (F) Guidewire Acceptance (in) Outside Diameter (in) SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB Biliary Only SERB SERB SERB SERB SERB SERB SERB SERB SERB SERB
19 Protégé RX Self-expanding Carotid Stent System The Protégé RX stent system is designed for carotid artery stenting. The nitinol stent comes premounted on a 6 F, 0.014" rapid exchange delivery system. The proximal and distal ends of the stent have tantalum radiopaque markers for enhanced visibility. Upon deployment, the stent achieves its predetermined diameter and exerts a constant, gentle outward force to establish patency of the vessel. Unconstrained Stent Diameter Unconstrained Stent Rec. Lumen Size Usable Catheter Sheath Size (F) Guidewire Acceptance (in) Straight SECX SECX SECX SECX SECX SECX SECX SECX SECX SECX SECX SECX SECX SECX SECX SECX SECX SECX SECX SECX Tapered SECX /6 30 ( )-( ) SECX /6 40 ( )-( ) SECX /7 30 ( )-( ) SECX /7 40 ( )-( ) Each system includes one stent and delivery catheter system. 19
20 Visi-Pro Balloon-expandable Peripheral Stent System The Visi-Pro stent system is indicated for use in the common and external iliac arteries. It is made from a stainless steel tube that is cut into an open lattice design and has tantalum radiopaque markers at the proximal and distal ends of the stent. The stent is mounted (crimped) onto a tightly folded balloon catheter and expanded and deployed by inflating the balloon. 80 cm CATHETER LENGTH STENT SIZE BALLOON COMPATIBILITY 135 cm Diameter Balloon Rec. Introducer Sheath (F) Guidewire (in) PXB * 0.035" PXB PXB * 0.035" PXB PXB * 0.035" PXB PXB * 0.035" PXB PXB * 0.035" PXB * 0.035" PXB PXB * 0.035" PXB PXB * 0.035" PXB PXB * 0.035" PXB PXB * 0.035" PXB * 0.035" PXB PXB * 0.035" PXB PXB * 0.035" PXB PXB * 0.035" PXB PXB * 0.035" PXB PXB * 0.035" PXB PXB * 0.035" PXB PXB * 0.035" PXB PXB * 0.035" PXB PXB " PXB PXB " PXB PXB " PXB PXB " PXB PXB " PXB PXB " PXB PXB " PXB PXB " *6 F = I.D. Each system includes one stent and delivery catheter system. 20
21 ParaMount Mini GPS Balloon-expandable Biliary Stent System The ParaMount Mini GPS stent system is designed for high visibility and is compatible with a 6 F and 7 F guide catheter. The proximal and distal ends of the stent have tantalum radiopaque markers for enhanced visiblity. Expanded Stent Diameter Expanded Stent Balloon Recommended Guide Catheter / Sheath Size Guidewire Acceptance (in) PMB / PMB / PMB / PMB */ PMB */ PMB */ PMB / PMB / PMB / Expanded Stent Diameter Expanded Stent Balloon Recommended Guide Catheter / Sheath Size Guidewire Acceptance (in) PMB / PMB / PMB / PMB */ PMB */ PMB */ PMB / PMB / PMB / *6 F = I.D. Each system includes one stent and delivery catheter system. 21
22 IntraStent Biliary Stent System The IntraStent biliary stent system is made up of unmounted large diameter stents designed for flexibility, strength and a low profile. These stents are designed to be manually crimped onto a non-compliant PTA balloon catheter that is indicated for the biliary stent expansion. UNEXPANDED STENT SIZE EXPANDED STENT SIZE Diameter Diameter IntraStent DoubleStrut LD Biliary Stent S15-16 ( ) , 10, 11, S15-26 ( ) , 10, 11, S15-36 ( ) , 10, 11, S15-56 ( ) , 10, 11, S15-76 ( ) , 10, 11, IntraStent Mega LD Biliary Stent S17-16 ( ) , 10, 11, S17-26 ( ) , 10, 11, S17-36 ( ) , 10, 11, IntraStent Max LD Biliary Stent S18-16 ( ) S18-26 ( ) S18-36 ( )
23 PTA BALLOONS Chocolate * PTA Balloon Catheter Fortrex OTW HP PTA Balloon Catheter 23
24 EverCross OTW PTA Balloon Catheter The EverCross balloon catheter is an 0.035" balloon that is specially designed for optimal pushability and trackability. The EverCross balloon catheter delivers both superior performance and a broad range of balloon sizes. Usable 40 cm Usable 80 cm Usable 135 cm Balloon Diameter Balloon Recomm. Introducer Sheath (F) Nominal Pressure (atm) - AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W (continued on next page) - AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W AB35W RBP (atm) 24
25 Admiral Xtreme PTA Balloon Catheter OTW 0.035" Treat longer PAD lesions above the knee with Admiral Xtreme PTA balloon catheter. Offered in 250 and 300 mm lengths, with 0.035" guidewire compatibility, featuring large inflation lumen for rapid inflation and deflation. TECHNICAL SPECIFICATIONS Catheter design Balloon material Balloon coating Balloon marker Usable shaft lengths Shaft diameter Over the wire (OTW) Flexitec Xtreme Hydrophilic 2 swaged (zero profile) Platinum Iridium 80, 130 cm 5 F - 6 F Guidewire compatibility ORDER INFORMATION Usable 80 cm Usable 130 cm Balloon Diameter Balloon Recom. Introducer Sheath (F) Nominal Pressure (atm) ADM ADM ADM ADM ADM ADM ADM ADM ADM ADM ADM ADM ADM ADM RBP (atm) 25
26 Fortrex 0.035" OTW HP PTA Balloon Catheter The Fortrex PTA balloon is the next-generation high pressure solution for AV access, and it is also intended for use in the peripheral vascular system. Engineered specifically for peak performance at rated burst pressure, the Fortrex balloon offers the deliverability, predictability and procedural efficiency desired by physicians. Usable 40 cm Usable 80 cm Usable 135 mm Balloon Diameter Balloon Nominal Pressure (atm) Rated Burst Pressure (atm) Recommended Introducer Sheath (F) A35HPV A35HPV A35HPV mm 20 mm A35HPV A35HPV A35HPV mm 40 mm A35HPV A35HPV A35HPV mm 80 mm A35HPV A35HPV A35HPV mm 100 mm A35HPV A35HPV A35HPV mm 20 mm A35HPV A35HPV A35HPV mm 40 mm A35HPV A35HPV A35HPV mm 80 mm A35HPV A35HPV A35HPV mm 100 mm A35HPV A35HPV A35HPV mm 20 mm A35HPV A35HPV A35HPV mm 40 mm A35HPV A35HPV A35HPV mm 80 mm A35HPV A35HPV A35HPV mm 100 mm A35HPV A35HPV A35HPV mm 20 mm A35HPV A35HPV A35HPV mm 40 mm A35HPV A35HPV A35HPV mm 80 mm A35HPV A35HPV A35HPV mm 100 mm A35HPV A35HPV A35HPV mm 40 mm A35HPV A35HPV A35HPV mm 80 mm A35HPV A35HPV A35HPV mm 100 mm A35HPV A35HPV A35HPV mm 40 mm A35HPV A35HPV A35HPV mm 80 mm A35HPV A35HPV A35HPV mm 40 mm A35HPV A35HPV A35HPV mm 80 mm A35HPV A35HPV A35HPV mm 40 mm A35HPV A35HPV A35HPV mm 80 mm
27 Pacific Plus PTA Catheter OTW 0.018" Realize more access options when you choose the Pacific Plus PTA catheter OTW 0.018", with its versatile shaft lengths. The proprietary balloon technology allows for enhanced crossing for treating lesions and the improved shaft design speeds deflation following treatment. TECHNICAL SPECIFICATIONS Catheter design Over the Wire (OTW), Coaxial Shaft Usable shaft lengths 90, 130, 180 cm Balloon material Proprietary Shaft diameter 4 F Balloon coating Hydrophilic Maximum guidewire compatibility Balloon marker 2 Swaged, Platinum Iridium Usable 90 cm Usable 130 cm Usable 180 cm Balloon Diameter Balloon Minimum Sheath Inner Diameter (F) Nominal Pressure (atm) PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP PCP RBP (atm) 27
28 Pacific Xtreme PTA Balloon Catheter OTW 0.018" Treat longer lesions with the Pacific Xtreme PTA balloon catheter OTW 0.018" that is offered in lengths from 150 to 300 mm. These balloons are well suited for use in treating long lesions in the femoral and popliteal vessels. TECHNICAL SPECIFICATIONS Catheter design Over the wire (OTW), coaxial shaft Balloon material Flexitec Xtreme Balloon coating Hydrophilic Balloon marker 2 swaged, Platinum Iridium Usable shaft lengths 90, 130 cm Shaft diameter 3.9 F 4.2 F Guidewire compatibility Usable 90 cm Usable 130 cm Balloon Diameter Balloon Recom. Introducer Sheath (F) Nominal Pressure (atm) PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU PCU AMP AMP / RBP (atm) 28
29 NanoCross Elite 0.014" OTW PTA Balloon Dilatation Catheter Go deeper into the anatomy and across difficult lesions with NanoCross Elite balloon catheter. Its seamless design from balloon to tip offers efficient energy transfer. Usable 90 cm Usable 150 cm Balloon Diameter Balloon Recomm. Introducer Sheath (F) Nominal Pressure (atm) AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W (proximal) / 1.5(distal) AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W (proximal) / 2.0(distal) AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W (proximal) / 2.5(distal) AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W (proximal) / 3.0(distal) AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W (proximal) / 3.5(distal) AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W AB14W RBP (atm) 29
30 RapidCross PTA Rapid Exchange Balloon Dilatation Catheter Reach for the RapidCross PTA rapid exchange balloon dilatation catheter when you need to reach more distal lesions in the small vessels below the knee. This catheter is the Medtronic solution for a rapid exchange balloon with 0.014" guidewire capability. Usable 90 cm Usable 170 cm Balloon Diameter Balloon Recomm. Introducer Sheath (F) Nominal Pressure (atm) A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX (proximal) / 1.5(distal) A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX (proximal) / 2.0(distal) A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX (proximal) / 2.5(distal) A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX (proximal) / 3.0(distal A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX A14BX (proximal) / 3.5(distal) RBP (atm) 30
31 Chocolate * PTA Balloon Catheter Minimize vessel trauma, dissections and the need for bailout stenting above or below the knee with the Chocolate * PTA balloon. The balloon s unique nitinol constraining structure creates pillows and grooves that provide a predictable, uniform and atraumatic dilatation. Usable 80 cm Balloon Diameter Balloon *Third party brands are trademarks of their respective owners. Catheter Guidewire (in) Introducer Sheath (F) Nominal Pressure (atm) CB OTW " CB OTW " CB OTW " CB OTW " CB OTW " CB OTW " CB OTW " CB OTW " CB OTW " CB OTW " CB OTW " CB OTW " CB OTW " CB OTW " CB OTW " CB OTW " CB OTW " CB OTW " RBP (atm) 31
32 INFUSION THERAPY Cragg-McNamara Valved Infusion Catheters 32
33 Cragg-McNamara Valved Infusion Catheter Cragg-McNamara catheters have radiopaque markers proximal and distal to the infusion segments. A Cragg-MicroValve at the distal tip provides end-hole occlusion. Cragg-McNamara catheters are available in a wide variety of side-hole infusion lengths. Diameter Usable Infusion Max. Guidewire (in)
34 ProStream Infusion Wire The ProStream wire is a 0.035" diameter, single lumen, closed-end wire that can be used alone or coaxially to treat two areas simultaneously, anywhere below the knee. Diameter (in) Usable Infusion MicroMewi Infusion Catheter The MicroMewi catheter is a single lumen, 2.9 F flexible catheter that is introduced over an 0.018" guidewire for delivery of pharmaceutical agents directly into a thrombosed segment. Diameter (F) Usable Infusion Max. Guidewire (in)
35 SNARES Amplatz Goose Neck Snare Kit 35
36 Amplatz Goose Neck Snare Kit The Amplatz Goose Neck snare kit is intended for use in the retrieval of atraumatic foreign bodies. Each kit contains one snare and one catheter. The snare is constructed of nitinol cable and a gold-plated tungsten loop. Because of the snare s preformed loop and superelastic construction, it can be introduced through catheters without risk of snare deformation. The snare catheter has a platinum-iridium radiopaque marker band. The Amplatz Goose Neck snare kit is intended for the cardiovascular and peripheral vascular systems. Loop Diameter Snare Catheter Size (F) Catheter GN GN GN GN GN GN GN GN GN Amplatz Goose Neck MicroSnare Kit The Amplatz Goose Neck kit is intended for the coronary and peripheral vascular system and the extra-cranial neurovascular anatomy. Loop Diameter Snare Catheter Size (F) Catheter SK SK SK SK SK SK
37 Snare Replacement Catheters Each Amplatz Goose Neck snare kit contains one snare and one catheter. The snare replacement catheters have a platinum-iridium radiopaque marker band. The Amplatz Goose Neck snare kit is intended for the cardiovascular and peripheral vascular systems. Catheter Outside Diameter (F) Catheter MC MC MC MC Each system includes one snare, one catheter, one introducer, and one torque device. 37
38 GUIDEWIRES Wholey Guidewire System Nitrex Guidewire 38
39 Wholey Guidewire System The Wholey guidewire is intended to facilitate the placement and exchange of interventional devices during diagnostic or therapeutic interventional procedures, Wholey guidewires provide enhanced torqueability and lubricity, alllowing interventionalists to approach challenging cases with confidence. Description Stiffness Profile Tip Style Size Outer Dia. (in) Size Quantity WWFS35145 Floppy Tip, Extension Compatible Floppy Straight/Shapeable /Pkg WWFS35175 Floppy Tip, Extension Compatible Floppy Straight/Shapeable /Pkg WWFS35260 Floppy Tip, Exchange Floppy Straight/Shapeable /Pkg WWFS35300 Floppy Tip, Exchange Floppy Straight/Shapeable /Pkg WWIJ35145 Modified J Tip, Extension Compatible Intermediate Modified J/Shapeable /Pkg WWIJ35175 Modified J Tip, Extension Compatible Intermediate Modified J/Shapeable /Pkg WWIJ35260 Modified J Tip, Exchange Intermediate Modified J/Shapeable /Pkg WWIJ35300 Modified J Tip, Exchange Intermediate Modified J/Shapeable /Pkg WWSS35145 Standard Tip, Extension Compatible Standard Straight/Shapeable /Pkg WWSS35175 Standard Tip, Extension Compatible Standard Straight/Shapeable /Pkg WWSS35260 Standard Tip, Exchange Standard Straight/Shapeable /Pkg WWSS35300 Standard Tip, Exchange Standard Straight/Shapeable /Pkg WWES35001 Extension System Standard Straight/Shapeable /Pkg WWTD35001 Kendall Torque Device NA NA 0.025"-0.038" 10/Pkg Babywire Nitinol Guidewire The Babywire guidewire assists in the placement of initial catheters and exchange of small vessel anatomy. Babywire guidewires are straight nitinol guidewires designed with double-ended round tips and flexible ends. Babywire guidewires are kink resistant and provide 1:1 torque. (10 / Package) Diameter (in) BW BW
40 Nitrex Guidewires The 0.014" and 0.018" Nitrex guidewires are intended for use in the peripheral and coronary vasculature. The and Nitrex guidewires are indicated for use in the peripheral vasculature. Nitrex guidewires are kink resistant and provide 1:1 torque. They are constructed of a superelastic nitinol core wire with a gold-plated tungsten coil for enhanced visualization. The wires have proprietary silicone coating for ease in placement. A torque device is packaged with and Nitrex guidewires. (3/Package)* Diameter (in) Tip Style Tip Tip Shape Tip Angle ( ) N INT 5 A 15 N INT 5 A 15 N INT 5 A N INT 5 S 0 N INT 7 S 0 N STD 2 S 0 N INT 5 A 15 N STD 2 S 0 N INT 5 A 15 N Flop 20 A 15 N STD 2 S 0 N INT 5 A N INT 8 A 15 N STD 2 S 0 N INT 8 A Flexible Shaft N INT 15 S 0 N INT 15 A 45 N INT 15 S 0 N INT 15 A 45 N INT 15 S Stiff Shaft N INT 9 S 0 N FLOP 14 A 45 N INT 9 S 0 N FLOP 14 S 0 N INT 9 S 0 N STD 4 A 45 N FLOP 14 S 0 N STD 4 A 45 N INT 9 S 0 N INT 9 S 0 N INT 9 S 0 *Torque devices included on and wire sizes. 40
41 SUPPORT CATHETERS, Y-CONNECTORS, AND GUIDE CATHETERS TrailBlazer Angled Support Catheter 41
TurboHawk. Plaque Excision System
 TurboHawk Plaque Excision System Twelve-month Patency in Diabetics DIABETICS VS. NON-DIABETICS 12-month Primary Patency (%) Diabetic patients show a more positive response to directional atherectomy than
TurboHawk Plaque Excision System Twelve-month Patency in Diabetics DIABETICS VS. NON-DIABETICS 12-month Primary Patency (%) Diabetic patients show a more positive response to directional atherectomy than
Chronic Total Occlusion (CTO) Technologies
 to receive our latest news and key activities. Chronic Total Occlusion (CTO) Technologies Re-open vital channels LinkedIn page Follow us on CORDIS EMEA OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry
to receive our latest news and key activities. Chronic Total Occlusion (CTO) Technologies Re-open vital channels LinkedIn page Follow us on CORDIS EMEA OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES
 MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
Systems for Below the Knee Interventions
 VCULR THERPIE rterial Venous Neurovascular 0.0 + 0.01 ystems for Below the Knee Interventions Nitrex Guidewire TrailBlazer upport Catheter Viance Crossing Catheter Enteer Re-entry ystem TurboHawk Plaque
VCULR THERPIE rterial Venous Neurovascular 0.0 + 0.01 ystems for Below the Knee Interventions Nitrex Guidewire TrailBlazer upport Catheter Viance Crossing Catheter Enteer Re-entry ystem TurboHawk Plaque
Chronic Total Occlusion (CTO) Technologies. Re-open vital channels
 Chronic Total Occlusion (CTO) Technologies Re-open vital channels OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry Technology Get back into the true lumen with ease and precision There are a number of
Chronic Total Occlusion (CTO) Technologies Re-open vital channels OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry Technology Get back into the true lumen with ease and precision There are a number of
NIRxcell CoCr Coronary Stent System
 cordis.com/nirxcell NIRxcell CoCr Coronary Stent System Exceptional Outcomes by Design Exceptionally low TLR rate of 5.1 % at 9 months 1 Superior Deliverability and Crossability 2 Best-in-class Conformability
cordis.com/nirxcell NIRxcell CoCr Coronary Stent System Exceptional Outcomes by Design Exceptionally low TLR rate of 5.1 % at 9 months 1 Superior Deliverability and Crossability 2 Best-in-class Conformability
We Catch. What Others Miss
 We Catch What Others Miss GORE Embolic Filter Diamond Frame Circumferential Filter Attachment eptfe Filter Media with 100-Micron Pores and Hydrophilic Heparin Coating Diamond Frame designed to enhance
We Catch What Others Miss GORE Embolic Filter Diamond Frame Circumferential Filter Attachment eptfe Filter Media with 100-Micron Pores and Hydrophilic Heparin Coating Diamond Frame designed to enhance
The Art of. Flow Restoration. Ikazuchi Zero. Semi-Compliant PTCA Balloon OVERVIEW CROSSABILITY LOW PROFILE CASE STUDY CODES
 The Art of Flow Restoration Japan s Recognized PCI Technology For Both Everyday and Complex CTO Cases* Manufactured by Kaneka Corporation, one of Japan s leading providers of interventional devices. Now
The Art of Flow Restoration Japan s Recognized PCI Technology For Both Everyday and Complex CTO Cases* Manufactured by Kaneka Corporation, one of Japan s leading providers of interventional devices. Now
Phoenix Atherectomy System Product Overview /LB
 Phoenix Atherectomy System Product Overview Phoenix Atherectomy System- The next generation of atherectomy The first hybrid atherectomy system available Combines the benefits of existing atherectomy systems
Phoenix Atherectomy System Product Overview Phoenix Atherectomy System- The next generation of atherectomy The first hybrid atherectomy system available Combines the benefits of existing atherectomy systems
JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW
 JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW DISCOVER THE VALUE OF VERSATILITY Versatility means not having to guess the morphology! Peripheral arterial lesions can present with
JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW DISCOVER THE VALUE OF VERSATILITY Versatility means not having to guess the morphology! Peripheral arterial lesions can present with
NC EMERGE TM PTCA Dilatation Catheter
 LEARN MORE Indications, contraindications, warnings and instructions for use can be found in the product labeling supplied with each device. Information for use only in countries with applicable health
LEARN MORE Indications, contraindications, warnings and instructions for use can be found in the product labeling supplied with each device. Information for use only in countries with applicable health
Guidewire Selection. Making the Most Out of My Guidewire: LINC 2016: Leipzig Interventional Course Leipzig, Germany January 26-29, 2016
 Making the Most Out of My Guidewire: Guidewire Selection LINC 2016: Leipzig Interventional Course Leipzig, Germany January 26-29, 2016 Brian DeRubertis, MD, FACS Associate Professor of Surgery Division
Making the Most Out of My Guidewire: Guidewire Selection LINC 2016: Leipzig Interventional Course Leipzig, Germany January 26-29, 2016 Brian DeRubertis, MD, FACS Associate Professor of Surgery Division
Versatility and Proven Safety
 Versatility and Proven Safety Indicated for: Temporary or Permanent placement Standard or Over-The-Wire Delivery Jugular, Femoral, Antecubital, and Popliteal Access > Proven Safety Initial Clinical Trial
Versatility and Proven Safety Indicated for: Temporary or Permanent placement Standard or Over-The-Wire Delivery Jugular, Femoral, Antecubital, and Popliteal Access > Proven Safety Initial Clinical Trial
RECOMMENDED INSTRUCTIONS FOR USE
 Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
CAROTID ARTERY ANGIOPLASTY
 CAROTID ARTERY ANGIOPLASTY Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
CAROTID ARTERY ANGIOPLASTY Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
Combine the best of both worlds
 S c i e n c e B a s e d E v o l u t i o n s l e a d t o R e v o l u t i o n a r y S o l u t i o n s PIONEER CoCr STENT ON DEB Combine the best of both worlds A next step in cardiovascular treatment should
S c i e n c e B a s e d E v o l u t i o n s l e a d t o R e v o l u t i o n a r y S o l u t i o n s PIONEER CoCr STENT ON DEB Combine the best of both worlds A next step in cardiovascular treatment should
Expanding to every demand: The GORE VIABAHN VBX Stent Graft
 Expanding to every demand: The GORE VIABAHN VBX Stent Graft GORE, VIABAHN, and designs are trademarks of W. L. Gore & Associates. 2017 W. L. Gore & Associates, Inc. Program Faculty Martin Austermann, MD
Expanding to every demand: The GORE VIABAHN VBX Stent Graft GORE, VIABAHN, and designs are trademarks of W. L. Gore & Associates. 2017 W. L. Gore & Associates, Inc. Program Faculty Martin Austermann, MD
Access More Patients. Customize Each Seal.
 Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
2.4 mm deflecting 2.4 mm tracking Above the knee Phoenix Below the knee 2.2 mm tracking 1.8 mm tracking
 Deep femoral artery Superficial femoral artery Popliteal artery Phoenix Above the knee Below the knee Atherectomy system Anterior tibial artery Peroneal artery Posterior tibial artery Dorsalis pedis artery
Deep femoral artery Superficial femoral artery Popliteal artery Phoenix Above the knee Below the knee Atherectomy system Anterior tibial artery Peroneal artery Posterior tibial artery Dorsalis pedis artery
Final Results of the Feasibility Study for the Drug-coated Chocolate Touch PTA balloon. (The ENDURE Trial)
 Final results of the feasibility study for the drug-coated Chocolate Touch PTA balloon of of femoropopliteal Femoropopliteal lesions lesions: (The ENDURE Trial) Final Results of the Feasibility Study for
Final results of the feasibility study for the drug-coated Chocolate Touch PTA balloon of of femoropopliteal Femoropopliteal lesions lesions: (The ENDURE Trial) Final Results of the Feasibility Study for
Final Results of the Feasibility Study for the Drug-coated Chocolate Touch PTA balloon. (The ENDURE Trial)
 Final results of the feasibility study for the drug-coated Chocolate Touch PTA balloon of of femoropopliteal Femoropopliteal lesions lesions: (The ENDURE Trial) Final Results of the Feasibility Study for
Final results of the feasibility study for the drug-coated Chocolate Touch PTA balloon of of femoropopliteal Femoropopliteal lesions lesions: (The ENDURE Trial) Final Results of the Feasibility Study for
Finally, the Control You Need to Deliver Accurate Treatment
 Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
5F Devices with 0.035
 Ambulatory mamagement for PAD endovascular treatment 5F Devices with 0.035 Flavio Airoldi, MD Multmedica IRCCS Milan - ITALY flavio.airoldi@multimedica.it Is there a need for low profile delivery systems?
Ambulatory mamagement for PAD endovascular treatment 5F Devices with 0.035 Flavio Airoldi, MD Multmedica IRCCS Milan - ITALY flavio.airoldi@multimedica.it Is there a need for low profile delivery systems?
Device Evolution. Atherectomy: Where Do We Stand After 12 Years Since FDA Clearance. Where Do We Stand? 4/18/2015
 Disclosure Statement of Financial Interest Atherectomy: Where Do We Stand After 12 Years Since FDA Clearance Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement
Disclosure Statement of Financial Interest Atherectomy: Where Do We Stand After 12 Years Since FDA Clearance Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM
 Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Guide Wires design and selection Abbott Vascular. All rights reserved.
 Guide Wires design and selection the cardiac catheter was...the key in the lock Cournand AF, Nobel lecture December 11, 1956; nowadays, the coronary guidewire is the master key to all locks Colombo et
Guide Wires design and selection the cardiac catheter was...the key in the lock Cournand AF, Nobel lecture December 11, 1956; nowadays, the coronary guidewire is the master key to all locks Colombo et
INTRODUCING. Cordis POWERFLEX Pro PTA Dilatation Catheter
 We Are Helping You Save Patients Limbs by Expanding the Portfolio INTRODUCING Cordis POWERFLEX Pro 0.035 PTA Dilatation Catheter Advanced Crossing Ability. Remarkable Versatility. Cordis POWERFLEX Pro
We Are Helping You Save Patients Limbs by Expanding the Portfolio INTRODUCING Cordis POWERFLEX Pro 0.035 PTA Dilatation Catheter Advanced Crossing Ability. Remarkable Versatility. Cordis POWERFLEX Pro
DURABLE. CONSISTENT. SAFE. IN.PACT Admiral Drug-Coated Balloon
 DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
New Devices Pedro Pinto Cardoso
 New Devices Pedro Pinto Cardoso HSM, CHLN, CALM for CTOs Guidewires Microcatheters Radiofrequency Pharmacotherapy Pedro Pinto Cardoso, Serviço de Cardiologia HSM CHLN New Guidewires SENTAI family Samurai
New Devices Pedro Pinto Cardoso HSM, CHLN, CALM for CTOs Guidewires Microcatheters Radiofrequency Pharmacotherapy Pedro Pinto Cardoso, Serviço de Cardiologia HSM CHLN New Guidewires SENTAI family Samurai
Zenith Alpha T HORACIC ENDOVASCULAR GRAFT
 Device description Zenith Alpha T HORACIC ENDOVASCULAR GRAFT www.cookmedical.com AI-D21181-EN-F Modular design The two-piece modular system allows the physician to customize a graft system to fit each
Device description Zenith Alpha T HORACIC ENDOVASCULAR GRAFT www.cookmedical.com AI-D21181-EN-F Modular design The two-piece modular system allows the physician to customize a graft system to fit each
Turbo-Power. Laser atherectomy catheter. The standard. for ISR
 Turbo-Power Laser atherectomy catheter The standard for ISR Vaporize the ISR challenge In-stent restenosis (ISR) Chance of recurring 7 115,000 + /year (U.S.) 1-6 Repeated narrowing of the arteries after
Turbo-Power Laser atherectomy catheter The standard for ISR Vaporize the ISR challenge In-stent restenosis (ISR) Chance of recurring 7 115,000 + /year (U.S.) 1-6 Repeated narrowing of the arteries after
Vascular Therapies Product Catalogue Arterial/Venous/Neurovascular 2013 Edition. Transforming Total Treatment
 Vascular Therapies Arterial/Venous/Neurovascular Edition Transforming Total Treatment ABOUT COVIDIEN Covidien is a leading global healthcare products company that creates innovative medical solutions for
Vascular Therapies Arterial/Venous/Neurovascular Edition Transforming Total Treatment ABOUT COVIDIEN Covidien is a leading global healthcare products company that creates innovative medical solutions for
CP STENT. Large Diameter, Balloon Expandable Stent
 CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
PrOspective multicenter study of carotid artery stenting Usinng mer Stent OCEANUS study!!!
 PrOspective multicenter study of carotid artery stenting Usinng mer Stent OCEANUS study!!! Prof Piotr Pieniazek MD PhD Jagiellonian University Institute of Cardiology, John Paul II Hospital Krakow, Poland
PrOspective multicenter study of carotid artery stenting Usinng mer Stent OCEANUS study!!! Prof Piotr Pieniazek MD PhD Jagiellonian University Institute of Cardiology, John Paul II Hospital Krakow, Poland
XIENCE Sierra INTRODUCING
 INTRODUCING Sierra With the safety you ve always relied on and the deliverability you ve always wanted, choosing the right stent is now an easier decision. START 2017 Abbott. All rights reserved. AP2944678-OUS
INTRODUCING Sierra With the safety you ve always relied on and the deliverability you ve always wanted, choosing the right stent is now an easier decision. START 2017 Abbott. All rights reserved. AP2944678-OUS
Vascular V12. Covered Stent. The New Standard of Care
 Vascular V12 Covered Stent The New Standard of Care Innovation Novel Thermo-Conformable Covered Stent 316L stainless steel stent completely encapsulated in a proprietary one piece eptfe film cast covering
Vascular V12 Covered Stent The New Standard of Care Innovation Novel Thermo-Conformable Covered Stent 316L stainless steel stent completely encapsulated in a proprietary one piece eptfe film cast covering
All-In-One. Iliac Branch System. PER F ORM ANC E b y d e s i g n ILIAC BRANCH ENDOPROSTHESIS
 All-In-One Iliac Branch System PER F ORM ANC E b y d e s i g n ILIAC BRANCH ENDOPROSTHESIS Exclusively Designed for Use in The GORE EXCLUDER Iliac Branch designed for the iliac branch, built from the same
All-In-One Iliac Branch System PER F ORM ANC E b y d e s i g n ILIAC BRANCH ENDOPROSTHESIS Exclusively Designed for Use in The GORE EXCLUDER Iliac Branch designed for the iliac branch, built from the same
Balloon Expandable Covered Stents. Suddenly a Crowded Space
 Novel Balloon Expandable Covered Stents for Aorto-iliac Occlusive Disease: New Trial Data makes for an Interesting Balloon Expandable Covered Stents Comparison in Aorto-iliac Occlusive Disease: Andrew
Novel Balloon Expandable Covered Stents for Aorto-iliac Occlusive Disease: New Trial Data makes for an Interesting Balloon Expandable Covered Stents Comparison in Aorto-iliac Occlusive Disease: Andrew
1 Comparison of Warfarin and Aspirin for Symptomatic Intracranial Arterial Stenosis. N Engl J Med 352;13, March 31, 2005
 The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
My idea on intimal tracking guide wire selection for Pop-BTK treatment
 LINC 2017 Leipzig January 24-27, 2017 My idea on intimal tracking guide wire selection for Pop-BTK treatment Jos C. van den Berg, MD PhD Ospedale Regionale di Lugano, sede Civico Lugano University of Bern
LINC 2017 Leipzig January 24-27, 2017 My idea on intimal tracking guide wire selection for Pop-BTK treatment Jos C. van den Berg, MD PhD Ospedale Regionale di Lugano, sede Civico Lugano University of Bern
Advanced Innovation for Exceptional Performance
 Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
THE THE MORE MORE NATURAL APPROACH TO OPTIMAL FIT
 THE THE MORE MORE NATURAL APPROACH Natural Approach TO Optimal Fit TO OPTIMAL FIT Conformability without Compromise THE STANDARD IN Conformability AND Designed for flexibility and conformability in tortuous
THE THE MORE MORE NATURAL APPROACH Natural Approach TO Optimal Fit TO OPTIMAL FIT Conformability without Compromise THE STANDARD IN Conformability AND Designed for flexibility and conformability in tortuous
IVUS Guided Case Review Case Performed by Frank R. Arko III, MD Charlotte, NC
 IVUS Guided Case Review Case Performed by Frank R. Arko III, MD Charlotte, NC The opinions and clinical experiences presented herein are for informational purposes only. Dr. Arko is a paid consultant for
IVUS Guided Case Review Case Performed by Frank R. Arko III, MD Charlotte, NC The opinions and clinical experiences presented herein are for informational purposes only. Dr. Arko is a paid consultant for
SMOOTH VASCULAR ACCESS COMPARATIVE BENCH TOP RESULTS
 SMOOTH VASCULAR ACCESS COMPARATIVE BENCH TOP RESULTS A NEW STANDARD FOR SMOOTH VASCULAR ACCESS NEEDLE PENETRATION FORCE Needle penetration was measured by the amount of force it took for the needle to
SMOOTH VASCULAR ACCESS COMPARATIVE BENCH TOP RESULTS A NEW STANDARD FOR SMOOTH VASCULAR ACCESS NEEDLE PENETRATION FORCE Needle penetration was measured by the amount of force it took for the needle to
Antegrade techniques for CTO recanalization. Dr. George Karavolias, MD, PhD, FESC, FACC Interventional Cardiologist
 Antegrade techniques for CTO recanalization Dr. George Karavolias, MD, PhD, FESC, FACC Interventional Cardiologist can CTOs be reliably opened by PCI? Meta-Analysis of 18,061 Patients Patel V, J Am Coll
Antegrade techniques for CTO recanalization Dr. George Karavolias, MD, PhD, FESC, FACC Interventional Cardiologist can CTOs be reliably opened by PCI? Meta-Analysis of 18,061 Patients Patel V, J Am Coll
INTERVENTIONAL VASCULAR DIAGNOSTICS AND THERAPY. SeQuent Please OTW PERIPHERAL DRUG COATED BALLOON CATHETER
 INTERVENTIONAL VASCULAR DIAGNOSTICS AND THERAPY SeQuent Please OTW PERIPHERAL DRUG COATED BALLOON CATHETER SeQuent Please OTW PERIPHERAL DRUG COATED BALLOON CATHETER The next level in treating peripheral
INTERVENTIONAL VASCULAR DIAGNOSTICS AND THERAPY SeQuent Please OTW PERIPHERAL DRUG COATED BALLOON CATHETER SeQuent Please OTW PERIPHERAL DRUG COATED BALLOON CATHETER The next level in treating peripheral
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU)
 Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Issam D. Moussa, MD. Professor of Medicine Mayo Clinic College of Medicine Chair, Division of Cardiovascular Diseases Mayo Clinic Jacksonville, FL
 Carotid Technologies and Protection Issam D. Moussa, MD Professor of Medicine Mayo Clinic College of Medicine Chair, Division of Cardiovascular Diseases Mayo Clinic Jacksonville, FL Disclosure Statement
Carotid Technologies and Protection Issam D. Moussa, MD Professor of Medicine Mayo Clinic College of Medicine Chair, Division of Cardiovascular Diseases Mayo Clinic Jacksonville, FL Disclosure Statement
Christian Wissgott MD, PhD Assistant Director, Radiology Westküstenkliniken Heide
 2-Year Results Of The Tack Optimized Balloon Angioplasty (TOBA) Trial For Fem- Pop Lesions Demonstrates Safety and Efficacy Of The Tack Endovascular System In Repairing Focal Post-PTA Dissections Christian
2-Year Results Of The Tack Optimized Balloon Angioplasty (TOBA) Trial For Fem- Pop Lesions Demonstrates Safety and Efficacy Of The Tack Endovascular System In Repairing Focal Post-PTA Dissections Christian
Cordis ExoSeal. Maximise Clinical Safety*. The novel approach to secure and easy closure. Vascular Closure Device CORDIS EXOSEAL VCD
 Cordis ExoSeal Vascular Closure Device CORDIS EXOSEAL VCD Maximise Clinical Safety*. The novel approach to secure and easy closure. * Clinical data from the ECLIPSE Trial indicates safety in terms of vascular
Cordis ExoSeal Vascular Closure Device CORDIS EXOSEAL VCD Maximise Clinical Safety*. The novel approach to secure and easy closure. * Clinical data from the ECLIPSE Trial indicates safety in terms of vascular
Ruby Coil. Large Volume Detachable Coils
 Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System
 100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
Zenith Renu AAA Converter Graft. Device Description Planning and Sizing Deployment Sequence Patient Follow-Up
 Zenith Renu AAA Converter Graft Device Description Planning and Sizing Deployment Sequence Patient Follow-Up Device description: Device indications The Zenith Renu AAA Converter Graft with Z-Trak Introduction
Zenith Renu AAA Converter Graft Device Description Planning and Sizing Deployment Sequence Patient Follow-Up Device description: Device indications The Zenith Renu AAA Converter Graft with Z-Trak Introduction
Colonic Metal Stents MAKING A DIFFERENCE TO HEALTH
 Colonic Metal Stents In a fast paced and maturing market, Diagmed Healthcare s Hanarostent has managed to continue to innovate and add unique and clinically superior features to its already premium range.
Colonic Metal Stents In a fast paced and maturing market, Diagmed Healthcare s Hanarostent has managed to continue to innovate and add unique and clinically superior features to its already premium range.
The Utility of Atherectomy and the Jetstream Atherectomy System
 The Utility of Atherectomy and the Jetstream Atherectomy System William A. Gray, MD Columbia University Medical Center 2014 Boston Scientific Corporation or its affiliates. All rights reserved. IMPORTANT
The Utility of Atherectomy and the Jetstream Atherectomy System William A. Gray, MD Columbia University Medical Center 2014 Boston Scientific Corporation or its affiliates. All rights reserved. IMPORTANT
Appropriate Device Selection for Endovascular Procedures
 Appropriate Device Selection for Endovascular Procedures Thomas M. Shimshak, MD Florida Hospital Heartland Medical Center Sebring, Florida Disclosures Speaker s Bureau: Abbott Vascular Boston Scientific
Appropriate Device Selection for Endovascular Procedures Thomas M. Shimshak, MD Florida Hospital Heartland Medical Center Sebring, Florida Disclosures Speaker s Bureau: Abbott Vascular Boston Scientific
NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER
 Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Pocket Reference Guide For (CTO) Technologies
 to receive our latest news and key activities. Pocket Reference Guide For (CTO) Technologies LinkedIn page Follow us on CORDIS EMEA Table of Contents Essential prescribing information for both FRONTRUNNER
to receive our latest news and key activities. Pocket Reference Guide For (CTO) Technologies LinkedIn page Follow us on CORDIS EMEA Table of Contents Essential prescribing information for both FRONTRUNNER
Maximise Clinical Safety*. Cordis ExoSeal. The novel approach to secure and easy closure. Vascular Closure Device CORDIS EXOSEAL VCD
 Cordis ExoSeal Vascular Closure Device Cardiology and Endovascular CORDIS EXOSEAL VCD Maximise Clinical Safety*. The novel approach to secure and easy closure. * Clinical data from the ECLIPSE Trial indicates
Cordis ExoSeal Vascular Closure Device Cardiology and Endovascular CORDIS EXOSEAL VCD Maximise Clinical Safety*. The novel approach to secure and easy closure. * Clinical data from the ECLIPSE Trial indicates
Selection and Use of Basic Equipment : Guiding Catheters, Wires and Balloons
 SCAI Interventional Cardiology Fall Fellows Course 2014 Selection and Use of Basic Equipment : Guiding Catheters, Wires and Balloons December 7, 2014 8:30 am John S. Douglas Jr, MD Professor of Medicine
SCAI Interventional Cardiology Fall Fellows Course 2014 Selection and Use of Basic Equipment : Guiding Catheters, Wires and Balloons December 7, 2014 8:30 am John S. Douglas Jr, MD Professor of Medicine
Solving the Dilemma of Ostial Stenting: A Case Series Illustrating the Flash Ostial System
 Volume 1, Issue 1 Case Report Solving the Dilemma of Ostial Stenting: A Case Series Illustrating the Flash Ostial System Robert F. Riley * and Bill Lombardi University of Washington Medical Center, Division
Volume 1, Issue 1 Case Report Solving the Dilemma of Ostial Stenting: A Case Series Illustrating the Flash Ostial System Robert F. Riley * and Bill Lombardi University of Washington Medical Center, Division
CTO: Technique and Tools
 CTO: Technique and Tools S. Hinan Ahmed, MD Associate Professor: Cardiology and Cardiothoracic Surgery Program Director: Interventional Fellowship Program Associate Editor: Cath Cardiov Interventions UT
CTO: Technique and Tools S. Hinan Ahmed, MD Associate Professor: Cardiology and Cardiothoracic Surgery Program Director: Interventional Fellowship Program Associate Editor: Cath Cardiov Interventions UT
William A. Gray MD System Chief of Cardiovascular Services, Main Line Health President, Lankenau Heart Institute Wynnewood, PA USA
 William A. Gray MD System Chief of Cardiovascular Services, President, Wynnewood, PA USA Why atherectomy? Calcification is the norm not the exception Most trials do not include heavy calcification There
William A. Gray MD System Chief of Cardiovascular Services, President, Wynnewood, PA USA Why atherectomy? Calcification is the norm not the exception Most trials do not include heavy calcification There
Indications. The AngioVac cannula is intended for use as a venous drainage cannula and for the removal of fresh, soft thrombi or emboli
 Indications Straight Cannula or Cannula with 20 O Angle 17F Working Channel The AngioVac cannula is intended for use as a venous drainage cannula and for the removal of fresh, soft thrombi or emboli Radiopaque
Indications Straight Cannula or Cannula with 20 O Angle 17F Working Channel The AngioVac cannula is intended for use as a venous drainage cannula and for the removal of fresh, soft thrombi or emboli Radiopaque
Straub Endovascular System &
 Straub Endovascular System & S t r a u b E n d o v a s c u l a r To o l s Straub Endovascular System Effective debulking in occluded arteries and veins Effective debulking in many indications Rotarex
Straub Endovascular System & S t r a u b E n d o v a s c u l a r To o l s Straub Endovascular System Effective debulking in occluded arteries and veins Effective debulking in many indications Rotarex
Is a Stent or Scaffold Necessary in The SFA?
 1 2 3 Is a Stent or Scaffold Necessary in The SFA? Stents were developed to optimize acute results after angioplasty Specifically, stents are universally accepted to manage flow limiting dissections and
1 2 3 Is a Stent or Scaffold Necessary in The SFA? Stents were developed to optimize acute results after angioplasty Specifically, stents are universally accepted to manage flow limiting dissections and
PERFORMANCE YOU CAN TRUST. EverFlex Self-expanding Peripheral Stent with Entrust Delivery System
 PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
Clinically proven. ordering info. Vascular Intervention // Peripheral Self-Expanding Stent System/0.018 /OTW. Pulsar-18
 140 μm thin struts Clinically proven 4F low profile Vascular Intervention // Peripheral Self-Expanding Stent System/0.018 /OTW Pulsar-18 Technical data / ordering info 140 μm thin struts - thinner than
140 μm thin struts Clinically proven 4F low profile Vascular Intervention // Peripheral Self-Expanding Stent System/0.018 /OTW Pulsar-18 Technical data / ordering info 140 μm thin struts - thinner than
Micromesh technology in carotid artery treatment what is next? Max Amor MD Clinique Louis Pasteur, Essey-Les-Nancy France
 Micromesh technology in carotid artery treatment what is next? Max Amor MD Clinique Louis Pasteur, Essey-Les-Nancy France www.incathlab.com Disclosure Speaker name: Max Amor, MD.... I have the following
Micromesh technology in carotid artery treatment what is next? Max Amor MD Clinique Louis Pasteur, Essey-Les-Nancy France www.incathlab.com Disclosure Speaker name: Max Amor, MD.... I have the following
New Drug Coated Balloons in Femoro-Popliteal Disease in Korea
 New Drug Coated Balloons in Femoro-Popliteal Disease in Korea Seung-Woon Rha, MD, PhD, FACC, FAHA, FSCAI, FESC, FAPSIC Div of Cardiovascular Intervention and Research Cardiovascular Center, Korea University
New Drug Coated Balloons in Femoro-Popliteal Disease in Korea Seung-Woon Rha, MD, PhD, FACC, FAHA, FSCAI, FESC, FAPSIC Div of Cardiovascular Intervention and Research Cardiovascular Center, Korea University
Intervention for Lower Extremity PAD: When, why and what?! Robert F Cuff, MD FACS RVT RPVI
 Intervention for Lower Extremity PAD: When, why and what?! Robert F Cuff, MD FACS RVT RPVI 1 Disclosures I have no financial disclosures related to this talk Objectives 1. Discuss indications for intervention
Intervention for Lower Extremity PAD: When, why and what?! Robert F Cuff, MD FACS RVT RPVI 1 Disclosures I have no financial disclosures related to this talk Objectives 1. Discuss indications for intervention
ABDOMINAL XT ABDOMINAL STENT GRAFT SYSTEM
 ABDOMINAL XT ABDOMINAL STENT GRAFT SYSTEM Pure performance and quality: Stent graft systems by JOTEC JOTEC is an innovation-oriented company with consistent focus on its core competencies: the development
ABDOMINAL XT ABDOMINAL STENT GRAFT SYSTEM Pure performance and quality: Stent graft systems by JOTEC JOTEC is an innovation-oriented company with consistent focus on its core competencies: the development
Gastric / EUS Metal Stents
 Gastric / EUS Metal Stents In a fast paced and maturing market, Diagmed Healthcare s Hanarostent has managed to continue to innovate and add unique and clinically superior features to its already premium
Gastric / EUS Metal Stents In a fast paced and maturing market, Diagmed Healthcare s Hanarostent has managed to continue to innovate and add unique and clinically superior features to its already premium
Indications for use. Contraindications within the United States
 Indications for use Indications within the United States The GORE TAG Thoracic Endoprosthesis is intended for endovascular repair of all lesions of the descending thoracic aorta, including: Isolated lesions
Indications for use Indications within the United States The GORE TAG Thoracic Endoprosthesis is intended for endovascular repair of all lesions of the descending thoracic aorta, including: Isolated lesions
CAROTID ARTERY ANGIOPLASTY
 CAROTID ARTERY ANGIOPLASTY Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and
CAROTID ARTERY ANGIOPLASTY Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and
Klinikum Rosenheim Department of Diagnostic and Interventional Radiology
 Klinikum Rosenheim Department of Diagnostic and Interventional Radiology Is Directional Atherectomy (Silverhawk Or Turbohawk) With DEB, Better Than DEB Alone: Based On The DEFINITIVE AR RCT: 1-Year Results
Klinikum Rosenheim Department of Diagnostic and Interventional Radiology Is Directional Atherectomy (Silverhawk Or Turbohawk) With DEB, Better Than DEB Alone: Based On The DEFINITIVE AR RCT: 1-Year Results
The evolution. AORFIX AAA Stent Graft now with the new AORFLEX Delivery System
 The evolution of AORFIX AORFIX AAA Stent Graft now with the new AORFLEX Delivery System The confident choice for the right angles AORFIX the only AAA stent graft approved for all angulations from 0 90,
The evolution of AORFIX AORFIX AAA Stent Graft now with the new AORFLEX Delivery System The confident choice for the right angles AORFIX the only AAA stent graft approved for all angulations from 0 90,
Superficial Femoral Artery Intervention: The gift that keeps on giving! Wm. Britton Eaves,MD WKHSC Bossier City, LA
 : The gift that keeps on giving! Wm. Britton Eaves,MD WKHSC Bossier City, LA Peripheral arterial disease (PAD) of the superficial femoral artery (SFA) is the most common cause of intermittent claudication
: The gift that keeps on giving! Wm. Britton Eaves,MD WKHSC Bossier City, LA Peripheral arterial disease (PAD) of the superficial femoral artery (SFA) is the most common cause of intermittent claudication
Protecting Patients. Preserving Access.
 MAHURKAR * Family of Acute Care Catheters Protecting Patients. Preserving Access. A full spectrum of acute care catheters for use in hemodialysis, apheresis, infusion and renal replacement therapy The
MAHURKAR * Family of Acute Care Catheters Protecting Patients. Preserving Access. A full spectrum of acute care catheters for use in hemodialysis, apheresis, infusion and renal replacement therapy The
RX Biliary System. Start
 Start We re in! When you control the wire, efficiency comes along for the ride. The of Physician-Controlled Wireguided Cannulation Physician-controlled wireguided cannulation (WGC) facilitates deep biliary
Start We re in! When you control the wire, efficiency comes along for the ride. The of Physician-Controlled Wireguided Cannulation Physician-controlled wireguided cannulation (WGC) facilitates deep biliary
There are multiple endovascular options for treatment
 Peripheral Rotablator Atherectomy: The Below-the-Knee Approach to Address Calcium Head On Peripheral Rotablator s front-cutting, diamond-tipped burr provides stable rotation in calcified lesions. BY SONYA
Peripheral Rotablator Atherectomy: The Below-the-Knee Approach to Address Calcium Head On Peripheral Rotablator s front-cutting, diamond-tipped burr provides stable rotation in calcified lesions. BY SONYA
Penumbra INTRACRANIAL ACCESS REDEFINED
 Penumbra INTRACRANIAL ACCESS REDEFINED BENCHMARK Case Examples Stability & Compatibility Atraumatic Navigation Flow Diverter Delivery Pipeline BENCHMARK Stability Without Surpass Full distal shaft radiopacity
Penumbra INTRACRANIAL ACCESS REDEFINED BENCHMARK Case Examples Stability & Compatibility Atraumatic Navigation Flow Diverter Delivery Pipeline BENCHMARK Stability Without Surpass Full distal shaft radiopacity
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
Proven Performance Through Innovative Design *
 Proven Performance Through Innovative Design * Deliver Our Next Generation AV Covered Stent Results The COVERA Vascular Covered Stent builds upon proven technologies from the category leader in AV Access.
Proven Performance Through Innovative Design * Deliver Our Next Generation AV Covered Stent Results The COVERA Vascular Covered Stent builds upon proven technologies from the category leader in AV Access.
Endovascular Treatment of Aortoiliac Occlusive Disease: What s in My Toolbox in Jade S. Hiramoto, MD, MAS UCSF Vascular Symposium April 20, 2018
 Endovascular Treatment of Aortoiliac Occlusive Disease: What s in My Toolbox in 2018 Jade S. Hiramoto, MD, MAS UCSF Vascular Symposium April 20, 2018 Disclosures Research support and royalties, Cook Inc.
Endovascular Treatment of Aortoiliac Occlusive Disease: What s in My Toolbox in 2018 Jade S. Hiramoto, MD, MAS UCSF Vascular Symposium April 20, 2018 Disclosures Research support and royalties, Cook Inc.
Introduction 3. What is Peripheral Vascular Disease? 5. What Are Some of the Symptoms of Peripheral Vascular Disease? 6
 Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
Drug-Coated Balloon Treatment for Patients with Intermittent Claudication: Insights from the IN.PACT Global Full Clinical Cohort
 Drug-Coated Balloon Treatment for Patients with Intermittent Claudication: Insights from the IN.PACT Global Full Clinical Cohort a.o. Univ. Prof. Dr. Marianne Brodmann Medical University of Graz Graz,
Drug-Coated Balloon Treatment for Patients with Intermittent Claudication: Insights from the IN.PACT Global Full Clinical Cohort a.o. Univ. Prof. Dr. Marianne Brodmann Medical University of Graz Graz,
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
Calcified lesions optimal treatment
 Calcified lesions optimal treatment I give you the latest innovation in balloon angioplasty. Dr. Mathias TISCHLER Hanusch Krankenhaus, Vienna, Austria 1 Nothing to disclose 2 Dienstag, 2. Februar 16 Calcification
Calcified lesions optimal treatment I give you the latest innovation in balloon angioplasty. Dr. Mathias TISCHLER Hanusch Krankenhaus, Vienna, Austria 1 Nothing to disclose 2 Dienstag, 2. Februar 16 Calcification
Disclosures. Tips and Tricks for Tibial Intervention. Tibial intervention overview
 Tips and Tricks for Tibial Intervention Donald L. Jacobs, MD C Rollins Hanlon Endowed Professor and Chair Chair of Surgery Saint Louis University SSM-STL Saint Louis University Hospital Disclosures Abbott
Tips and Tricks for Tibial Intervention Donald L. Jacobs, MD C Rollins Hanlon Endowed Professor and Chair Chair of Surgery Saint Louis University SSM-STL Saint Louis University Hospital Disclosures Abbott
Iliac Solutions. Arterial Venous Neurovascular. Wholey Guidewire System. Nitrex Guidewires. TrailBlazer Support Catheter
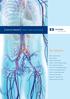 VCULR THERPIE rterial Venous Neurovascular Iliac olutions Wholey Guidewire ystem Nitrex Guidewires TrailBlazer upport Catheter EverCross.03 OTW PT Balloon Catheter VisiPro Balloon Expandable tent Protege
VCULR THERPIE rterial Venous Neurovascular Iliac olutions Wholey Guidewire ystem Nitrex Guidewires TrailBlazer upport Catheter EverCross.03 OTW PT Balloon Catheter VisiPro Balloon Expandable tent Protege
Radiation Safety Abbott Vascular. All rights reserved.
 Radiation Safety More and more complex cases are performed Complexity Index and Fluoroscopy Time 2 3 Collimators / Distances The intensity of scattered radiation is a function of exposed field size Use
Radiation Safety More and more complex cases are performed Complexity Index and Fluoroscopy Time 2 3 Collimators / Distances The intensity of scattered radiation is a function of exposed field size Use
Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully
 Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Endovascular Catalog. Endovascular Catalog Page 1 cordis.com
 Endovascular Catalog Endovascular Catalog Page 1 cordis.com Table of Contents About Us 4 CORDIS Access Portfolio 5 AVANTI + Sheath Introducer 6 BRITE TIP Catheter Sheath Introducer 7 Obturators 8 Vessel
Endovascular Catalog Endovascular Catalog Page 1 cordis.com Table of Contents About Us 4 CORDIS Access Portfolio 5 AVANTI + Sheath Introducer 6 BRITE TIP Catheter Sheath Introducer 7 Obturators 8 Vessel
CHALLENGES IN FEMORO-POPLITEAL STENTING
 CHALLENGES IN FEMORO-POPLITEAL STENTING Karathanos C MD, MSc, PhD, Athanasoulas A, Rousas N, Spanos K, Giannoukas A Department of Vascular Surgery, University Hospital of Larissa, Faculty of Medicine,
CHALLENGES IN FEMORO-POPLITEAL STENTING Karathanos C MD, MSc, PhD, Athanasoulas A, Rousas N, Spanos K, Giannoukas A Department of Vascular Surgery, University Hospital of Larissa, Faculty of Medicine,
Leg arteries : MANAGEMENT and STRATEGY
 Leg arteries : MANAGEMENT and STRATEGY Prof E. Ducasse Unit of vascular surgery BORDEAUX ESVB May 14th 2011 BARD Symposium CLI : definition Fontaine Rutherford ABI Symptoms class category Asymptomatic
Leg arteries : MANAGEMENT and STRATEGY Prof E. Ducasse Unit of vascular surgery BORDEAUX ESVB May 14th 2011 BARD Symposium CLI : definition Fontaine Rutherford ABI Symptoms class category Asymptomatic
Biliary Metal Stents MAKING A DIFFERENCE TO HEALTH
 Biliary Metal Stents In a fast paced and maturing market, Diagmed Healthcare s Hanarostent has managed to continue to innovate and add unique and clinically superior features to its already premium range.
Biliary Metal Stents In a fast paced and maturing market, Diagmed Healthcare s Hanarostent has managed to continue to innovate and add unique and clinically superior features to its already premium range.
Introduction What Causes Peripheral Vascular Disease? How Do Doctors Treat Peripheral Vascular Disease?... 9
 Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
Ureteroscopy solutions for your most challenging cases.
 Ureteroscopy solutions for your most challenging cases. IT TAKES INSIGHT TO SEE THAT NO TWO STONE PATIENTS ARE ALIKE. THAT S THE DIFFERENCE BETWEEN MAKING DEVICES AND MAKING PROGRESS. There is nothing
Ureteroscopy solutions for your most challenging cases. IT TAKES INSIGHT TO SEE THAT NO TWO STONE PATIENTS ARE ALIKE. THAT S THE DIFFERENCE BETWEEN MAKING DEVICES AND MAKING PROGRESS. There is nothing
