Solution for cardiac perfusion in viaflex plastic container
|
|
|
- Marilyn West
- 6 years ago
- Views:
Transcription
1 CARDIOPLEGIA SOLUTION A Solution for cardiac perfusion in viaflex plastic container DESCRIPTION Cardioplegia Solution A is a sterile, non-pyrogenic solution in a Viaflex bag. It is used to induce cardiac stasis and to protect the myocardium during open-heart surgery. Cardioplegia Solution A is an isotonic crystalloid solution based on extracellular fluid ionic concentrations. Each 1000mL contains: Sodium chloride B.P. 6.43g Potassium chloride B.P. 1.19g Magnesium chloride hexahydrate B.P. 3.25g Calcium chloride dihydrate B.P. 176mg The mixture contains the following ions in 1000mL: Sodium 110mmol Potassium 16mmol Magnesium 16mmol Calcium 1.2mmol Chloride 160mmol Approximate osmolality is 275mOsm/kg and approximate ph is 3.7. Cardioplegia Solution A requires the aseptic addition of 10mL Sodium Bicarbonate 8.4% w/v Injection B.P. prior to use to adjust the ph to Following ph adjustment the total sodium ionic concentration is 120mmol/L. PHARMACOLOGY Perfusion of the coronary circulation by Cardioplegia Solution A allows a quiet, bloodless operative field, a flaccid myocardium and avoidance of air embolism. It protects the myocardium during cardiac surgery by inducing a rapid and complete diastolic arrest, CARDIOPLEGIA SOLUTION A Data Sheet 30 March 2016 Page 1 of 5
2 minimising myocardial energy requirements during arrest, preventing ischaemic damage during the arrest phase and minimising or preventing reperfusion injury once coronary blood flow is restored. Sodium and Chloride These ions have no specific role in producing cardiac arrest but are important in establishing a solution similar to the composition of normal extracellular fluid and sodium is essential in maintaining the ionic integrity of the myocardium and controlling calcium movements. Potassium Potassium is present in Cardioplegia Solution A to induce a rapid diastolic arrest, thereby preserving energy supplies (adenosine triphosphate and creatine phosphate for post-ischaemic activity. Magnesium Addition of magnesium to the solution prevents cellular potassium and magnesium loss, thereby conserving magnesium for its role as an enzymatic cofactor. It appears to counteract the actions of calcium in excitation-contraction coupling, which results in a reduction of energy consumption. It also has a weak arresting action on the heart. Calcium Calcium helps maintain the integrity of the cell membrane and prevents a condition known as calcium paradox occurring during reperfusion. Bicarbonate This is added to adjust the ph, producing a slightly alkaline ( ) solution and to compensate for the metabolic acidosis which accompanies ischaemia. INDICATIONS Following ph adjustment with 10mL of Sodium Bicarbonate 8.4% w/v Injection B.P., Cardioplegia Solution A is used in combination with ischaemia and hypothermia to induce cardiac arrest during open heart surgery and to preserve the myocardium during asystole. CONTRAINDICATIONS Cardioplegia Solution A must not be administered without the prior addition of sodium bicarbonate. PRECAUTIONS Cardioplegia Solution A is intended only for cardiac perfusion when the coronary circulation is isolated from the systemic circulation. It must not be injected intravenously. CARDIOPLEGIA SOLUTION A Data Sheet 30 March 2016 Page 2 of 5
3 It should be used only by those trained in cardiac perfusion techniques and open heart surgery and in the presence of inotropic support drugs and the appropriate defibrillation equipment. The ph of the solution must be adjusted by the aseptic addition of Sodium Bicarbonate Injection. The Cardioplegia Solution A container should then be rapidly inverted five times to ensure complete mixing. It should be cooled to 4 C before use. Once mixed, the solution should be used within 24 hours. It should not be used in serial connections with other containers. The remainder of any partly-used solutions should be discarded. If large volumes of Cardioplegia Solution A are perfused and permitted to return to the extracorporeal circuit without any venting from the right heart, serum magnesium and potassium levels may rise. It is recommended that the right heart be vented. It is important that the appropriate dosage of Cardioplegia Solution A be used (see DOSAGE AND ADMINISTRATION below) to ensure that all areas of the myocardium are cooled evenly, especially those areas distal to arterial obstruction in patients with coronary-artery disease. Inadequate dosage may result in uneven cooling, incomplete arrest and ischaemic injury. Numerous clinical parameters require close monitoring in patients receiving Cardioplegia Solution A. Maintenance of hypothermia is critical and myocardial temperature should be monitored throughout the procedure. Continuous monitoring of myocardial activity is essential. Use in pregnancy (Category B2) Cardioplegia Solution A has not been subjected to animal reproduction studies. Its effect upon the human foetus has not been established nor has its effect upon reproductive capacity. It should be administered to pregnant women only if unavoidable. Use in lactation Use of Cardioplegia Solution A in lactation is not recommended. Carcinogenic potential Cardioplegia Solution A is based on human extracellular fluid. There is no evidence that a carcinogenic potential exists. Genotoxicity Cardioplegia Solution A is based on human extracellular fluid. There is no evidence that a mutagenic potential exists. CARDIOPLEGIA SOLUTION A Data Sheet 30 March 2016 Page 3 of 5
4 ADVERSE EFFECTS The use of Cardioplegia solutions during open-heart surgery has been associated with a number of intraoperative and perioperative risks, including myocardial infarction, ECG abnormalities and arrhythmias (including ventricular fibrillation). Spontaneous recovery after Cardioplegia-induced cardiac arrest may be delayed or absent at reperfusion; shock defibrillation may be required to restore normal cardiac function. In addition, Cardioplegia solutions may cause potential electrolyte and acid-base abnormalities. DOSAGE AND ADMINISTRATION It is important that an appropriate dose of Cardioplegia Solution A is used to ensure that all areas of the myocardium are cooled evenly, especially those areas distal to arterial obstruction in patients with coronary-artery disease. Inadequate dosage may result in uneven cooling, incomplete arrest and ischaemic injury. Dosage may vary depending upon the perfusion technique in use, and the preferences and experience of the surgeon. The volume of solution instilled into the aortic root may vary depending upon the duration or type of open heart surgical procedure. The following information is provided for guidance. The ph must be adjusted with 10mL of Sodium Bicarbonate 8.4% w/v Injection B.P. and the solution cooled to 4 C before use. Following institution of cardiopulmonary bypass at perfusate temperature of 28 to 30 C, and after cross-clamping of the ascending aorta, the ph adjusted solution is administered by rapid infusion into the aortic root. The initial rate of infusion may be 300mL/m 2 /minute (approximately 540mL/min for 173cm, 70kg adult with a body surface area of 1.8m 2 ) given over a period of two to four minutes. Concurrent external cooling (regional hypothermia of the pericardium) may be accomplished by instilling a refrigerated physiological solution such as PLASMALYTE148 (approx. ph 7.4), Sodium Lactate Compound Infusion B.P. (Hartmann s Solution), or a physiological ice slush into the chest cavity. Should myocardial activity persist or recur, Cardioplegia Solution A may be reinfused at a rate of 300mL/m 2 /minute for a period of two minutes. Reinfusion of the solution may be repeated every 20 to 30 minutes or sooner if the myocardial temperature rises above 15 to 20 C or returning cardiac activity is observed. The regional hypothermia solution around the heart may also be replenished continuously or periodically in order to maintain adequate hypothermia. Suction may be used to remove warmed infusates. CARDIOPLEGIA SOLUTION A Data Sheet 30 March 2016 Page 4 of 5
5 OVERDOSAGE Overuse of the solution may result in unnecessary dilation of the myocardial vasculature and leakage into the perivascular myocardium, possibly causing tissue oedema, see PRECAUTIONS and ADVERSE EFFECTS sections. PRESENTATION AND STORAGE CONDITIONS Cardioplegia Solution A is supplied in 1000mL Viaflex plastic containers for single use only. The ph of the solution must be adjusted to between 7.4 and 7.8 with 10mL Sodium Bicarbonate 8.4% w/v Injection BP (not supplied) before use. The product should be stored below 30 C. MEDICINE CLASSIFICATION General Sale Medicine NAME AND ADDRESS Cardioplegia Solution A is distributed in New Zealand by: Healthcare Ltd 33 Vestey Drive Mt Wellington Auckland 1060 Ph: Cardioplegia Solution A is manufactured and distributed in Australia by: Healthcare Pty Ltd 1 Drive Old Toongabbie NSW 2146 DATE OF PREPARATION 30 March 2016 Based on Australian PI most recent amendment 24 February 2015; and CCSI Please refer to the Medsafe website ( for most recent data sheet., Viaflex and Cardioplegia Solution A are trademarks of International Inc. CARDIOPLEGIA SOLUTION A Data Sheet 30 March 2016 Page 5 of 5
PRODUCT INFORMATION. NAME OF THE MEDICINE Compound Sodium Lactate (Hartmann's) Solution for Injection
 PRODUCT INFORMATION NAME OF THE MEDICINE Compound Sodium Lactate (Hartmann's) Solution for Injection DESCRIPTION Molecular formulae. Potassium chloride: KCl; sodium chloride: NaCl; calcium chloride dihydrate:
PRODUCT INFORMATION NAME OF THE MEDICINE Compound Sodium Lactate (Hartmann's) Solution for Injection DESCRIPTION Molecular formulae. Potassium chloride: KCl; sodium chloride: NaCl; calcium chloride dihydrate:
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION
 DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
Each 1000mL of Plasmalyte 56 in 5% Glucose IV infusion contains: q.s. to 1000mL. 401mOsm/kg 835kJ
 PLASMALYTE 56 IN 5% GLUCOSE Plasmalyte 56 in 5% Glucose IV infusion solution (Multiple Electrolyte with Glucose injection) DESCRIPTION Plasmalyte 56 in 5% Glucose IV infusion is a sterile, clear, nonpyrogenic
PLASMALYTE 56 IN 5% GLUCOSE Plasmalyte 56 in 5% Glucose IV infusion solution (Multiple Electrolyte with Glucose injection) DESCRIPTION Plasmalyte 56 in 5% Glucose IV infusion is a sterile, clear, nonpyrogenic
Product Information. Plasma-Lyte 148 Replacement IV Infusion
 Product Information Plasma-Lyte 148 Replacement IV Infusion Name of the medicine Plasma-Lyte 148 Replacement IV Infusion (Multiple Electrolyte Injection). Description Plasma-Lyte 148 Replacement IV Infusion
Product Information Plasma-Lyte 148 Replacement IV Infusion Name of the medicine Plasma-Lyte 148 Replacement IV Infusion (Multiple Electrolyte Injection). Description Plasma-Lyte 148 Replacement IV Infusion
Going on Bypass. What happens before, during and after CPB. Perfusion Dept. Royal Children s Hospital Melbourne, Australia
 Going on Bypass What happens before, during and after CPB. Perfusion Dept. Royal Children s Hospital Melbourne, Australia Circulation Brain Liver Kidneys Viscera Muscle Skin IVC, SVC Pump Lungs R.A. L.V.
Going on Bypass What happens before, during and after CPB. Perfusion Dept. Royal Children s Hospital Melbourne, Australia Circulation Brain Liver Kidneys Viscera Muscle Skin IVC, SVC Pump Lungs R.A. L.V.
Calcium Chloride 10% Injection 1g in 10 ml (as calcium chloride dihydrate containing 6.8 mmol calcium ions in 10 ml)
 10% Injection 1g in 10 ml (as calcium chloride dihydrate containing 6.8 mmol calcium ions in 10 ml) NAME OF THE MEDICINE Name: Calcium chloride dihydrate Molecular formula: CaCl2.2H20 Molecular weight:
10% Injection 1g in 10 ml (as calcium chloride dihydrate containing 6.8 mmol calcium ions in 10 ml) NAME OF THE MEDICINE Name: Calcium chloride dihydrate Molecular formula: CaCl2.2H20 Molecular weight:
SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS PRODUCT SUMMARY 1. NAME OF THE MEDICINAL PRODUCT Sterile Potassium Chloride Concentrate 15%. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 15% of Potassium Chloride in
SUMMARY OF PRODUCT CHARACTERISTICS PRODUCT SUMMARY 1. NAME OF THE MEDICINAL PRODUCT Sterile Potassium Chloride Concentrate 15%. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 15% of Potassium Chloride in
50% Concentrated Injection
 NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
Albumex 4 NAME OF THE MEDICINE DESCRIPTION PHARMACOLOGY CLINICAL TRIALS. New Zealand Data Sheet. Human Albumin 4% (40 g/l)
 New Zealand Data Sheet Albumex 4 Human Albumin 4% (40 g/l) NAME OF THE MEDICINE Human albumin, solution for intravenous infusion. DESCRIPTION Albumex 4 is manufactured from human plasma donated by New
New Zealand Data Sheet Albumex 4 Human Albumin 4% (40 g/l) NAME OF THE MEDICINE Human albumin, solution for intravenous infusion. DESCRIPTION Albumex 4 is manufactured from human plasma donated by New
PRODUCT INFORMATION. DESCRIPTION Addaven is a concentrated trace element solution for infusion which is clear and colourless to slightly yellow.
 PRODUCT INFORMATION Addaven NAME OF MEDICINE Addaven is a concentrated trace element solution containing the following simple salts: chromic chloride hexahydrate (CAS No.:10060-12-5), cupric chloride dihydrate
PRODUCT INFORMATION Addaven NAME OF MEDICINE Addaven is a concentrated trace element solution containing the following simple salts: chromic chloride hexahydrate (CAS No.:10060-12-5), cupric chloride dihydrate
Cardiac anaesthesia. Simon May
 Cardiac anaesthesia Simon May Contents Cardiac: Principles of peri-operative management for cardiac surgery Cardiopulmonary bypass, cardioplegia and off pump cardiac surgery Cardiac disease and its implications
Cardiac anaesthesia Simon May Contents Cardiac: Principles of peri-operative management for cardiac surgery Cardiopulmonary bypass, cardioplegia and off pump cardiac surgery Cardiac disease and its implications
NEW ZEALAND DATA SHEET
 1 0.9% Sodium Chloride (infusion, solution) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredient 9g/L (0.9%) sodium chloride (USP), in water for injection. For the full list of excipients, see
1 0.9% Sodium Chloride (infusion, solution) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredient 9g/L (0.9%) sodium chloride (USP), in water for injection. For the full list of excipients, see
PRODUCT INFORMATION RESONIUM A. Na m
 PRODUCT INFORMATION RESONIUM A NAME OF THE MEDICINE Non-proprietary Name Sodium polystyrene sulfonate Chemical Structure CH - 2 CH SO 3 Na + n CAS Number 28210-41-5 [9003-59-2] CH 2 CH SO - 3 m DESCRIPTION
PRODUCT INFORMATION RESONIUM A NAME OF THE MEDICINE Non-proprietary Name Sodium polystyrene sulfonate Chemical Structure CH - 2 CH SO 3 Na + n CAS Number 28210-41-5 [9003-59-2] CH 2 CH SO - 3 m DESCRIPTION
Thinking outside of the box Perfusion management and myocardial protection strategy for a patient with sickle cell disease
 Thinking outside of the box Perfusion management and myocardial protection strategy for a patient with sickle cell disease Shane Buel MS, RRT 1 Nicole Michaud MS CCP PBMT 1 Rashid Ahmad MD 2 1 Vanderbilt
Thinking outside of the box Perfusion management and myocardial protection strategy for a patient with sickle cell disease Shane Buel MS, RRT 1 Nicole Michaud MS CCP PBMT 1 Rashid Ahmad MD 2 1 Vanderbilt
Demonstration of Uneven. the infusion on myocardial temperature was insufficient
 Demonstration of Uneven in Patients with Coronary Lesions Rolf Ekroth, M.D., HAkan erggren, M.D., Goran Sudow, M.D., Josef Wojciechowski, M.D., o F. Zackrisson, M.D., and Goran William-Olsson, M.D. ASTRACT
Demonstration of Uneven in Patients with Coronary Lesions Rolf Ekroth, M.D., HAkan erggren, M.D., Goran Sudow, M.D., Josef Wojciechowski, M.D., o F. Zackrisson, M.D., and Goran William-Olsson, M.D. ASTRACT
Hartmann s Solution. For Infusion
 0xxxxx1_00_HartmannsSolution_IE_BP 07.02.2017 13:15 Seite 1 PACKAGE LEAFLET: INFORMATION FOR THE USER Hartmann s Solution For Infusion Read all of this leaflet carefully before you start using this medicine
0xxxxx1_00_HartmannsSolution_IE_BP 07.02.2017 13:15 Seite 1 PACKAGE LEAFLET: INFORMATION FOR THE USER Hartmann s Solution For Infusion Read all of this leaflet carefully before you start using this medicine
Plasma-Lyte 56 and 5% Glucose IV Infusion (Multiple Electrolyte and Glucose Injection).
 Name of the Medicine Plasma-Lyte 56 and 5% Glucose IV Infusion (Multiple Electrolyte and Glucose Injection). Description Plasma-Lyte 56 and 5% Glucose IV Infusion is a sterile, clear, non-pyrogenic hypertonic
Name of the Medicine Plasma-Lyte 56 and 5% Glucose IV Infusion (Multiple Electrolyte and Glucose Injection). Description Plasma-Lyte 56 and 5% Glucose IV Infusion is a sterile, clear, non-pyrogenic hypertonic
Difficult Scenarios for Myocardial Protection SAHA Gil Bolotin M.D., Ph.D. Rambam Medical Center, Haifa, Israel
 Difficult Scenarios for Myocardial Protection SAHA 2017 Gil Bolotin M.D., Ph.D. Rambam Medical Center, Haifa, Israel Difficult Scenarios for Myocardial Protection Stone Heart Nightmare Nightmare of the
Difficult Scenarios for Myocardial Protection SAHA 2017 Gil Bolotin M.D., Ph.D. Rambam Medical Center, Haifa, Israel Difficult Scenarios for Myocardial Protection Stone Heart Nightmare Nightmare of the
NORMOSOL -R MULTIPLE ELECTROLYTES INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid Flexible Plastic Container
 NORMOSOL -R MULTIPLE ELECTROLYTES INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid Flexible Plastic Container R x only DESCRIPTION Normosol-R is a sterile, nonpyrogenic isotonic
NORMOSOL -R MULTIPLE ELECTROLYTES INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid Flexible Plastic Container R x only DESCRIPTION Normosol-R is a sterile, nonpyrogenic isotonic
Product Information. Plasma-Lyte 148 Replacement and 5% Glucose IV Infusion
 Product Information Plasma-Lyte 148 Replacement and 5% Glucose IV Infusion Name of the medicine Plasma-Lyte 148 Replacement and 5% Glucose IV Infusion (Multiple Electrolyte and Glucose Injection) Description
Product Information Plasma-Lyte 148 Replacement and 5% Glucose IV Infusion Name of the medicine Plasma-Lyte 148 Replacement and 5% Glucose IV Infusion (Multiple Electrolyte and Glucose Injection) Description
NEW ZEALAND DATA SHEET
 1. PRODUCT NAME ADDAVEN (infusion, solution concentrate) NEW ZEALAND DATA SHEET 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each 10 ml ampoule of Addaven contains: Chromic chloride hexahydrate 53.33 µg
1. PRODUCT NAME ADDAVEN (infusion, solution concentrate) NEW ZEALAND DATA SHEET 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each 10 ml ampoule of Addaven contains: Chromic chloride hexahydrate 53.33 µg
NEW ZEALAND DATA SHEET
 1 Ringers (infusion, solution) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredients The ingredients in Ringers infusion solution comprise: sodium chloride (8.6g/L), potassium chloride (0.3g/L)
1 Ringers (infusion, solution) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredients The ingredients in Ringers infusion solution comprise: sodium chloride (8.6g/L), potassium chloride (0.3g/L)
Potassium Chloride in 0.9% Sodium Chloride Injection, USP in Plastic Container
 PRESCRIBING INFORMATION Potassium Chloride in 0.9% Sodium Chloride Injection, USP in Plastic Container VIAFLEX Container 20mmol/L Potassium Chloride in Sodium in 0.9% Chloride Injection 40mmol/L Potassium
PRESCRIBING INFORMATION Potassium Chloride in 0.9% Sodium Chloride Injection, USP in Plastic Container VIAFLEX Container 20mmol/L Potassium Chloride in Sodium in 0.9% Chloride Injection 40mmol/L Potassium
NEW ZEALAND DATA SHEET
 1 HEMOSOL B0 (solution, dialysis) NEW ZEALAND DATA SHEET 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Hemosol B0, is a solution for haemodialysis and haemofiltration consisting of a mixture of the following
1 HEMOSOL B0 (solution, dialysis) NEW ZEALAND DATA SHEET 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Hemosol B0, is a solution for haemodialysis and haemofiltration consisting of a mixture of the following
0.45% Sodium Chloride Injection, USP
 PRESCRIBING INFORMATION 0.45% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision: November 22, 2013 Submission Control
PRESCRIBING INFORMATION 0.45% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision: November 22, 2013 Submission Control
COMPOUND SODIUM LACTATE (HARTMANN S SOLUTION) AND 5% GLUCOSE
 COMPOUND SODIUM LACTATE (HARTMANN S SOLUTION) AND 5% GLUCOSE Compound Sodium Lactate (Hartmann s solution) and 5% Glucose intravenous, infusion solution Composition The ingredients in Compound Sodium Lactate
COMPOUND SODIUM LACTATE (HARTMANN S SOLUTION) AND 5% GLUCOSE Compound Sodium Lactate (Hartmann s solution) and 5% Glucose intravenous, infusion solution Composition The ingredients in Compound Sodium Lactate
Cardiothoracic Fellow Expectations Division of Cardiac Anesthesia, Beth Israel Deaconess Medical Center
 The fellowship in Cardiothoracic Anesthesia at the Beth Israel Deaconess Medical Center is intended to provide the foundation for a career as either an academic cardiothoracic anesthesiologist or clinical
The fellowship in Cardiothoracic Anesthesia at the Beth Israel Deaconess Medical Center is intended to provide the foundation for a career as either an academic cardiothoracic anesthesiologist or clinical
SUMMARY OF PRODUCT CHARACTERISTICS. Synthamin 14, 8.5% Amino Acid Intravenous Infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Synthamin 14, 8.5% Amino Acid Intravenous Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION L-Leucine Ph. Eur 0.620% w/v L-Isoleucine
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Synthamin 14, 8.5% Amino Acid Intravenous Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION L-Leucine Ph. Eur 0.620% w/v L-Isoleucine
Myocardial Protection Principles. David J Chambers
 David J Chambers Cardiac Surgical Research/Cardiothoracic Surgery The Rayne Institute (King s College London) Guy s & St Thomas NHS Foundation Trust St Thomas Hospital London UK ScanSect, Aarhus, Denmark.
David J Chambers Cardiac Surgical Research/Cardiothoracic Surgery The Rayne Institute (King s College London) Guy s & St Thomas NHS Foundation Trust St Thomas Hospital London UK ScanSect, Aarhus, Denmark.
DBL NALOXONE HYDROCHLORIDE INJECTION USP
 Name of medicine Naloxone hydrochloride Data Sheet New Zealand DBL NALXNE HYDRCHLRIDE INJECTIN USP Presentation DBL Naloxone Hydrochloride Injection USP is a sterile, clear, colourless solution, free from
Name of medicine Naloxone hydrochloride Data Sheet New Zealand DBL NALXNE HYDRCHLRIDE INJECTIN USP Presentation DBL Naloxone Hydrochloride Injection USP is a sterile, clear, colourless solution, free from
Cardioplegia Circuit Products { ANTEGRADE}
 Cardioplegia Circuit Products { ANTEGRADE} Antegrade cannulae are designed to deliver cardioplegia solution to the heart via the coronary ostia in the normal direction of blood flow (antegrade perfusion).
Cardioplegia Circuit Products { ANTEGRADE} Antegrade cannulae are designed to deliver cardioplegia solution to the heart via the coronary ostia in the normal direction of blood flow (antegrade perfusion).
MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350)
 MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350) NAME OF THE MEDICINE: MOVICOL Liquid Orange Flavour, Concentrate for Oral Solution. DESCRIPTION: A clear colourless solution.
MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350) NAME OF THE MEDICINE: MOVICOL Liquid Orange Flavour, Concentrate for Oral Solution. DESCRIPTION: A clear colourless solution.
Normosol -R and 5% Dextrose Injection MULTIPLE ELECTROLYTES AND 5% DEXTROSE INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid
 Normosol -R and 5% Dextrose Injection MULTIPLE ELECTROLYTES AND 5% DEXTROSE INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid R x only Flexible Plastic Container DESCRIPTION Normosol-R
Normosol -R and 5% Dextrose Injection MULTIPLE ELECTROLYTES AND 5% DEXTROSE INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid R x only Flexible Plastic Container DESCRIPTION Normosol-R
Dianeal Peritoneal Dialysis Solutions
 Dianeal Peritoneal Dialysis Solutions Name of the medicine Dianeal PD-2, PD-4 and 1 mmol/l Calcium Peritoneal Dialysis Solutions. Description Dianeal PD-2, PD-4 and 1 mmol/l Calcium Peritoneal Dialysis
Dianeal Peritoneal Dialysis Solutions Name of the medicine Dianeal PD-2, PD-4 and 1 mmol/l Calcium Peritoneal Dialysis Solutions. Description Dianeal PD-2, PD-4 and 1 mmol/l Calcium Peritoneal Dialysis
Reference ID:
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PRISMASOL and PHOXILLUM safely and effectively. See full prescribing information for PRISMASOL and
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PRISMASOL and PHOXILLUM safely and effectively. See full prescribing information for PRISMASOL and
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Addaven concentrate for solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Addaven contains: 1 ml 1 ampoule (10 ml) Chromic
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Addaven concentrate for solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Addaven contains: 1 ml 1 ampoule (10 ml) Chromic
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Aqupharm 3 Solution for Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredients Sodium Chloride Glucose Anhydrous
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Aqupharm 3 Solution for Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredients Sodium Chloride Glucose Anhydrous
*Sections or subsections omitted from the full prescribing information are not 6 ADVERSE REACTIONS
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PRISMASOL and PHOXILLUM safely and effectively. See full prescribing information for PRISMASOL and
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PRISMASOL and PHOXILLUM safely and effectively. See full prescribing information for PRISMASOL and
Baxter Potassium Chloride and Sodium Chloride Intravenous Infusion 10 mmol/100 ml, 20 mmol/1000 ml, 30 mmol/1000 ml, 40 mmol/1000 ml
 Baxter Potassium Chloride and Sodium Chloride Intravenous Infusion 10 mmol/100 ml, 20 mmol/1000 ml, 30 mmol/1000 ml, 40 mmol/1000 ml Name of the Medicine Potassium Chloride and Sodium Chloride Composition:
Baxter Potassium Chloride and Sodium Chloride Intravenous Infusion 10 mmol/100 ml, 20 mmol/1000 ml, 30 mmol/1000 ml, 40 mmol/1000 ml Name of the Medicine Potassium Chloride and Sodium Chloride Composition:
PACKAGE INSERT CALCIUM GLUCONATE INJECTION, USP 10% Electrolyte Replenisher mosmol/ml 680 mosmol/l Ca meq/ml ph
 CALCIUM GLUCONATE INJECTION, USP 10% Electrolyte Replenisher 0.68 mosmol/ml 680 mosmol/l Ca ++ 0.465 meq/ml ph 6.0 8.2 DESCRIPTION Calcium Gluconate Injection, USP is a sterile, nonpyrogenic supersaturated
CALCIUM GLUCONATE INJECTION, USP 10% Electrolyte Replenisher 0.68 mosmol/ml 680 mosmol/l Ca ++ 0.465 meq/ml ph 6.0 8.2 DESCRIPTION Calcium Gluconate Injection, USP is a sterile, nonpyrogenic supersaturated
Cardiology. Objectives. Chapter
 1:44 M age 1121 Chapter Cardiology Objectives art 1: Cardiovascular natomy and hysiology, ECG Monitoring, and Dysrhythmia nalysis (begins on p. 1127) fter reading art 1 of this chapter, you should be able
1:44 M age 1121 Chapter Cardiology Objectives art 1: Cardiovascular natomy and hysiology, ECG Monitoring, and Dysrhythmia nalysis (begins on p. 1127) fter reading art 1 of this chapter, you should be able
Impact of TOE on delivery of antegrade cardioplegia
 Impact of TOE on delivery of antegrade cardioplegia David Canty Senior Lecturer + Director of Simulation Department of Surgery, University of Melbourne Cardiac Anaesthetist Royal Melbourne Hospital + Monash
Impact of TOE on delivery of antegrade cardioplegia David Canty Senior Lecturer + Director of Simulation Department of Surgery, University of Melbourne Cardiac Anaesthetist Royal Melbourne Hospital + Monash
INTRAVENOUS SOLUTIONS WITH POTASSIUM CHLORIDE POTASSIUM CHLORIDE IN LACTATED RINGER S AND 5% DEXTROSE INJECTION, USP Flexible Plastic Container
 INTRAVENOUS SOLUTIONS WITH POTASSIUM CHLORIDE POTASSIUM CHLORIDE IN LACTATED RINGER S AND 5% DEXTROSE INJECTION, USP Flexible Plastic Container R x only DESCRIPTION Intravenous solutions with potassium
INTRAVENOUS SOLUTIONS WITH POTASSIUM CHLORIDE POTASSIUM CHLORIDE IN LACTATED RINGER S AND 5% DEXTROSE INJECTION, USP Flexible Plastic Container R x only DESCRIPTION Intravenous solutions with potassium
PRODUCT INFORMATION. (sodium bicarbonate) SODIUM BICARBONATE INTRAVENOUS INFUSION BP 8.4% NAME OF THE MEDICINE DESCRIPTION PHARMACOLOGY.
 PRODUCT INFORMATION SODIUM BICARBONATE INTRAVENOUS INFUSION BP 8.4% (sodium bicarbonate) NAME OF THE MEDICINE Sodium bicarbonate Molecular Formula: NaHCO 3 CAS Number: 144-55-8 DESCRIPTION Description
PRODUCT INFORMATION SODIUM BICARBONATE INTRAVENOUS INFUSION BP 8.4% (sodium bicarbonate) NAME OF THE MEDICINE Sodium bicarbonate Molecular Formula: NaHCO 3 CAS Number: 144-55-8 DESCRIPTION Description
POTASSIUM CHLORIDE, 5% GLUCOSE POTASSIUM CHLORIDE 30MMOL IN 5% GLUCOSE
 POTASSIUM CHLORIDE 20MMOL IN 5% GLUCOSE Potassium Chloride 20mmol (0.15%) in 5% Glucose infusion solution, BP POTASSIUM CHLORIDE 30MMOL IN 5% GLUCOSE Potassium Chloride 30mmol (0.224%) in 5% Glucose infusion
POTASSIUM CHLORIDE 20MMOL IN 5% GLUCOSE Potassium Chloride 20mmol (0.15%) in 5% Glucose infusion solution, BP POTASSIUM CHLORIDE 30MMOL IN 5% GLUCOSE Potassium Chloride 30mmol (0.224%) in 5% Glucose infusion
Sodium Chloride 0.9% w/v Intravenous Infusion BP Solution for Infusion Sodium chloride
 Package leaflet: Information for the user Sodium Chloride 0.9% w/v Intravenous Infusion BP Solution for Infusion Sodium chloride Read all of this leaflet carefully before using this medicine because it
Package leaflet: Information for the user Sodium Chloride 0.9% w/v Intravenous Infusion BP Solution for Infusion Sodium chloride Read all of this leaflet carefully before using this medicine because it
Vamin 18 EF QUALITATIVE AND QUANTITATIVE COMPOSITION
 Title Page Information Dec. 2000 1 (7) Number Vamin 18 EF 121-01 1. NAME OF THE MEDICINAL PRODUCT Vamin 18 g N/l electrolyte-free infusion fluid, solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1000
Title Page Information Dec. 2000 1 (7) Number Vamin 18 EF 121-01 1. NAME OF THE MEDICINAL PRODUCT Vamin 18 g N/l electrolyte-free infusion fluid, solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1000
ALS MODULE 7 Pharmacology
 ALS MODULE 7 Pharmacology Relates to HLT404C Apply Advanced Resuscitation Techniques Introduction There are no studies that addressed the order of drug administration. There is inadequate evidence to define
ALS MODULE 7 Pharmacology Relates to HLT404C Apply Advanced Resuscitation Techniques Introduction There are no studies that addressed the order of drug administration. There is inadequate evidence to define
INTRAVENOUS FLUID THERAPY. Tom Heaps Consultant Acute Physician
 INTRAVENOUS FLUID THERAPY Tom Heaps Consultant Acute Physician LEARNING OBJECTIVES 1. Crystalloids vs colloids 2. Balanced vs non-balanced solutions 3. Composition of various IV fluids 4. What is normal
INTRAVENOUS FLUID THERAPY Tom Heaps Consultant Acute Physician LEARNING OBJECTIVES 1. Crystalloids vs colloids 2. Balanced vs non-balanced solutions 3. Composition of various IV fluids 4. What is normal
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET 1. PRODUCT NAME POTASSIUM CHLORIDE 0.75 g/10 ml Solution for Injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 0.75 g (10 mmol) of Potassium Chloride in 10 ml Sterile Potassium
NEW ZEALAND DATA SHEET 1. PRODUCT NAME POTASSIUM CHLORIDE 0.75 g/10 ml Solution for Injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 0.75 g (10 mmol) of Potassium Chloride in 10 ml Sterile Potassium
The Effect of Acute Coronary Artery Occlusion during Cardioplegic Arrest
 The Effect of Acute Coronary Artery Occlusion during Cardioplegic Arrest and Reperfusion on Myocardial Preservation John H. Rousou, M.D., Richard M. Engelman, M.D., William A. Dobbs, Ph.D., and Mooideen
The Effect of Acute Coronary Artery Occlusion during Cardioplegic Arrest and Reperfusion on Myocardial Preservation John H. Rousou, M.D., Richard M. Engelman, M.D., William A. Dobbs, Ph.D., and Mooideen
2. What you need to know before you use Compound Sodium Lactate
 Package leaflet: Information for the user Compound Sodium Lactate Intravenous Infusion BP, Ecobag (Hartmann s Solution) Read all of this leaflet carefully before you start using this medicine because it
Package leaflet: Information for the user Compound Sodium Lactate Intravenous Infusion BP, Ecobag (Hartmann s Solution) Read all of this leaflet carefully before you start using this medicine because it
PHYSIONEAL 40 GLUCOSE SOLUTION FOR PERITONEAL DIALYSIS
 PHYSIONEAL 40 GLUCOSE SOLUTION FOR PERITONEAL DIALYSIS Name of the Medicine: Physioneal 40 Glucose (%) Solution for peritoneal dialysis Physioneal 40 Glucose 1.36% w/v / 13.6 mg/ml Solution for Peritoneal
PHYSIONEAL 40 GLUCOSE SOLUTION FOR PERITONEAL DIALYSIS Name of the Medicine: Physioneal 40 Glucose (%) Solution for peritoneal dialysis Physioneal 40 Glucose 1.36% w/v / 13.6 mg/ml Solution for Peritoneal
Advanced Resuscitation - Adult
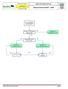 C02A Resuscitation 2017-03-23 17 years & older Office of the Medical Director Advanced Resuscitation - Adult Intermediate Advanced Critical From PRIMARY ASSESSMENT Known or suspected hypothermia Algorithm
C02A Resuscitation 2017-03-23 17 years & older Office of the Medical Director Advanced Resuscitation - Adult Intermediate Advanced Critical From PRIMARY ASSESSMENT Known or suspected hypothermia Algorithm
Diabetic Ketoacidosis
 Diabetic Ketoacidosis Definition: Diabetic Ketoacidosis is one of the most serious acute complications of diabetes. It s more common in young patients with type 1 diabetes mellitus. It s usually characterized
Diabetic Ketoacidosis Definition: Diabetic Ketoacidosis is one of the most serious acute complications of diabetes. It s more common in young patients with type 1 diabetes mellitus. It s usually characterized
DATA SHEET 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 DATA SHEET 1 PRODUCT NAME 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Sachet, each 5.1g sachet contains: Sodium chloride 470mg, potassium chloride 300mg, sodium acid citrate 530mg, glucose 3.56g. Tablet,
DATA SHEET 1 PRODUCT NAME 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Sachet, each 5.1g sachet contains: Sodium chloride 470mg, potassium chloride 300mg, sodium acid citrate 530mg, glucose 3.56g. Tablet,
Advanced Resuscitation - Adolescent
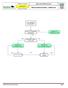 C02B Resuscitation 2017-03-23 10 up to 17 years Office of the Medical Director Advanced Resuscitation - Adolescent Intermediate Advanced Critical From PRIMARY ASSESSMENT Known or suspected hypothermia
C02B Resuscitation 2017-03-23 10 up to 17 years Office of the Medical Director Advanced Resuscitation - Adolescent Intermediate Advanced Critical From PRIMARY ASSESSMENT Known or suspected hypothermia
Advanced Resuscitation - Child
 C02C Resuscitation 2017-03-23 1 up to 10 years Office of the Medical Director Advanced Resuscitation - Child Intermediate Advanced Critical From PRIMARY ASSESSMENT Known or suspected hypothermia Algorithm
C02C Resuscitation 2017-03-23 1 up to 10 years Office of the Medical Director Advanced Resuscitation - Child Intermediate Advanced Critical From PRIMARY ASSESSMENT Known or suspected hypothermia Algorithm
Package leaflet: Information for the user. multibic potassium-free solution for haemodialysis/haemofiltration
 Package leaflet: Information for the user multibic potassium-free solution for haemodialysis/haemofiltration Read all of this leaflet carefully before you start using this medicine because it contains
Package leaflet: Information for the user multibic potassium-free solution for haemodialysis/haemofiltration Read all of this leaflet carefully before you start using this medicine because it contains
New Zealand Data Sheet. LYCINATE Sublingual Tablets contain 0.6mg (600mcg) glyceryl trinitrate.
 LYCINATE New Zealand Data Sheet Glyceryl trinitrate 600mcg Tablets Presentation LYCINATE Sublingual Tablets contain 0.6mg (600mcg) glyceryl trinitrate. Uses Actions Glyceryl trinitrate causes smooth muscle
LYCINATE New Zealand Data Sheet Glyceryl trinitrate 600mcg Tablets Presentation LYCINATE Sublingual Tablets contain 0.6mg (600mcg) glyceryl trinitrate. Uses Actions Glyceryl trinitrate causes smooth muscle
PRODUCT INFORMATION UROCIT -K. Wax matrix tablets
 PRODUCT INFORMATION UROCIT -K Wax matrix tablets NAME OF DRUG Potassium Citrate CAS number- 6100-05-6 The empirical formula of potassium citrate is K 3 C 6 H 5 0 7.H 2 O, and its structural formula is:
PRODUCT INFORMATION UROCIT -K Wax matrix tablets NAME OF DRUG Potassium Citrate CAS number- 6100-05-6 The empirical formula of potassium citrate is K 3 C 6 H 5 0 7.H 2 O, and its structural formula is:
MOVICOL Lemon-Lime Flavour Powder for Solution (macrogol 3350)
 MOVICOL Lemon-Lime Flavour Powder for Solution (macrogol 3350) Product Name: MOVICOL Lemon-Lime Flavour Product Description: Each sachet of MOVICOL Lemon-Lime contains: Macrogol 3350 Sodium chloride Sodium
MOVICOL Lemon-Lime Flavour Powder for Solution (macrogol 3350) Product Name: MOVICOL Lemon-Lime Flavour Product Description: Each sachet of MOVICOL Lemon-Lime contains: Macrogol 3350 Sodium chloride Sodium
NEW ZEALAND DATA SHEET
 1 PHOXILIUM 1.2MMOL/L, PHOSPHATE (solution, dialysis) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Phoxilium 1.2mmol/L phosphate solution for haemodialysis and haemofiltration consists of a mixture of the
1 PHOXILIUM 1.2MMOL/L, PHOSPHATE (solution, dialysis) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Phoxilium 1.2mmol/L phosphate solution for haemodialysis and haemofiltration consists of a mixture of the
Effect of Four Crystalloid Cardioplegias on Immature Rabbit Hearts During Global Ischaemia
 Original Article Effect of Four Crystalloid Cardioplegias on Immature Rabbit Hearts During Global Ischaemia Yunqing Mei, Hua Liu, Cun Long, Bangchang Cheng, Shangzhi Gao and Dayi Hu, Department of Thoracic
Original Article Effect of Four Crystalloid Cardioplegias on Immature Rabbit Hearts During Global Ischaemia Yunqing Mei, Hua Liu, Cun Long, Bangchang Cheng, Shangzhi Gao and Dayi Hu, Department of Thoracic
PRODUCT INFORMATION INFLUVAC. NAME OF THE MEDICINE Influvac inactivated influenza vaccine (surface antigen)
 PRODUCT INFORMATION INFLUVAC NAME OF THE MEDICINE Influvac inactivated influenza vaccine (surface antigen) DESCRIPTION Influvac is a clear colourless suspension for injection. It is an egg-grown, inactivated
PRODUCT INFORMATION INFLUVAC NAME OF THE MEDICINE Influvac inactivated influenza vaccine (surface antigen) DESCRIPTION Influvac is a clear colourless suspension for injection. It is an egg-grown, inactivated
Chapter 16 Nutrition, Fluids and Electrolytes, and Acid-Base Balance Nutrition Nutrients Water o Functions Promotes metabolic processes Transporter
 Chapter 16 Nutrition, Fluids and Electrolytes, and Acid-Base Balance Nutrition Nutrients Water o Functions Promotes metabolic processes Transporter for nutrients and wastes Lubricant Insulator and shock
Chapter 16 Nutrition, Fluids and Electrolytes, and Acid-Base Balance Nutrition Nutrients Water o Functions Promotes metabolic processes Transporter for nutrients and wastes Lubricant Insulator and shock
PRODUCT INFORMATION. NAME OF MEDICINE Glamin is an amino acid / dipeptide 13.4% solution for intravenous infusion.
 PRODUCT INFORMATION GLAMIN NAME OF MEDICINE Glamin is an amino acid / dipeptide 13.4% solution for intravenous infusion. The active ingredients are: alanine (CAS No.:56-41-7), arginine (CAS No.:74-79-3),
PRODUCT INFORMATION GLAMIN NAME OF MEDICINE Glamin is an amino acid / dipeptide 13.4% solution for intravenous infusion. The active ingredients are: alanine (CAS No.:56-41-7), arginine (CAS No.:74-79-3),
Package leaflet: information for the user. Prismasol 2 mmol/l Potassium Solution for haemodialysis/haemofiltration
 Package leaflet: information for the user Prismasol 2 mmol/l Potassium Solution for haemodialysis/haemofiltration Calcium chloride dihydrate/ magnesium chloride hexahydrate/ glucose monohydrate/ lactic
Package leaflet: information for the user Prismasol 2 mmol/l Potassium Solution for haemodialysis/haemofiltration Calcium chloride dihydrate/ magnesium chloride hexahydrate/ glucose monohydrate/ lactic
Acid-base management during hypothermic CPB alpha-stat and ph-stat models of blood gas interpretation
 Acid-base management during hypothermic CPB alpha-stat and ph-stat models of blood gas interpretation Michael Kremke Department of Anaesthesiology and Intensive Care Aarhus University Hospital, Denmark
Acid-base management during hypothermic CPB alpha-stat and ph-stat models of blood gas interpretation Michael Kremke Department of Anaesthesiology and Intensive Care Aarhus University Hospital, Denmark
SUMMARY OF PRODUCT CHARACTERISTICS. Potassium Chloride 0.15% w/v & Sodium Chloride 0.9% w/v Solution for Infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Potassium Chloride 0.15% w/v & Sodium Chloride 0.9% w/v Solution for Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Potassium Chloride
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Potassium Chloride 0.15% w/v & Sodium Chloride 0.9% w/v Solution for Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Potassium Chloride
Sodium Bicarbonate Injection, USP
 Sodium Bicarbonate Injection, USP FOR THE CORRECTION OF METABOLIC ACIDOSIS AND OTHER CONDITIONS REQUIRING SYSTEMIC ALKALINIZATION. Plastic Ansyr II Syringe R x only DESCRIPTION Sodium Bicarbonate Injection,
Sodium Bicarbonate Injection, USP FOR THE CORRECTION OF METABOLIC ACIDOSIS AND OTHER CONDITIONS REQUIRING SYSTEMIC ALKALINIZATION. Plastic Ansyr II Syringe R x only DESCRIPTION Sodium Bicarbonate Injection,
PRODUCT INFORMATION INFLUVAC. NAME OF THE MEDICINE Influvac inactivated influenza vaccine (surface antigen)
 PRODUCT INFORMATION INFLUVAC NAME OF THE MEDICINE Influvac inactivated influenza vaccine (surface antigen) DESCRIPTION Influvac is a clear colourless suspension for injection. It is an egg-grown, inactivated
PRODUCT INFORMATION INFLUVAC NAME OF THE MEDICINE Influvac inactivated influenza vaccine (surface antigen) DESCRIPTION Influvac is a clear colourless suspension for injection. It is an egg-grown, inactivated
DBL CALCIUM GLUCONATE INJECTION BP
 Description DBL CALCIUM GLUCONATE INJECTION BP DBL Calcium Gluconate Injection BP is a clear, colourless solution containing in each 10 ml, Calcium Gluconate BP 953 mg and Calcium Saccharate U.S.P. 30
Description DBL CALCIUM GLUCONATE INJECTION BP DBL Calcium Gluconate Injection BP is a clear, colourless solution containing in each 10 ml, Calcium Gluconate BP 953 mg and Calcium Saccharate U.S.P. 30
Coronary Circulation Under normal conditions cardiac muscle metabolism is almost exclusively aerobic depending on oxidative phosorylation to
 Coronary Circulation Under normal conditions cardiac muscle metabolism is almost exclusively aerobic depending on oxidative phosorylation to resynthesis the ATP continuously utilized by repetitive, excitation,
Coronary Circulation Under normal conditions cardiac muscle metabolism is almost exclusively aerobic depending on oxidative phosorylation to resynthesis the ATP continuously utilized by repetitive, excitation,
MWLCEMS SYSTEM Continuing Education Packet Management of the Acute MI Patient
 MWLCEMS SYSTEM Continuing Education Packet Management of the Acute MI Patient In this CE we will discuss the patient presenting with an acute ST-Elevation Myocardial Infarction (STEMI) Definition: Myocardial
MWLCEMS SYSTEM Continuing Education Packet Management of the Acute MI Patient In this CE we will discuss the patient presenting with an acute ST-Elevation Myocardial Infarction (STEMI) Definition: Myocardial
BRICANYL INJECTION. terbutaline sulfate PRODUCT INFORMATION
 BRICANYL INJECTION terbutaline sulfate PRODUCT INFORMATION NAME OF THE MEDICINE Terbutaline sulfate, 2-(tert-butylamino)-1-(3,5-dihydroxyphenyl) ethanol sulfate, a sympathomimetic bronchodilator with a
BRICANYL INJECTION terbutaline sulfate PRODUCT INFORMATION NAME OF THE MEDICINE Terbutaline sulfate, 2-(tert-butylamino)-1-(3,5-dihydroxyphenyl) ethanol sulfate, a sympathomimetic bronchodilator with a
Product Information BROWN SNAKE ANTIVENOM AUST R 74897
 Product Information APPROVED NAME BROWN SNAKE ANTIVENOM AUST R 74897 DESCRIPTION BROWN SNAKE ANTIVENOM is prepared from the plasma of horses immunised with the venom of the brown snake (Pseudonaja textilis).
Product Information APPROVED NAME BROWN SNAKE ANTIVENOM AUST R 74897 DESCRIPTION BROWN SNAKE ANTIVENOM is prepared from the plasma of horses immunised with the venom of the brown snake (Pseudonaja textilis).
DATA SHEET ADENOCOR NAME OF THE MEDICINE DESCRIPTION PHARMACOLOGY
 DATA SHEET ADENOCOR NAME OF THE MEDICINE Adenocor (adenosine) 6 mg/ 2 ml solution for intravenous infusion. Adenosine is designated chemically as 6-amino-9-ß-D-ribofuranosyl-9-H-purine and has the following
DATA SHEET ADENOCOR NAME OF THE MEDICINE Adenocor (adenosine) 6 mg/ 2 ml solution for intravenous infusion. Adenosine is designated chemically as 6-amino-9-ß-D-ribofuranosyl-9-H-purine and has the following
Steph ani eph ani Mi M ck i MD Cleveland Clinic
 Stephanie Mick MD Stephanie Mick MD Cleveland Clinic Upper hemisternotomy AVR Ascending Aorta MVr Thoracotomy Based Anterior AVR Lateral Thoracotomy Mitral/Tricuspid surgery Robotically assisted surgery
Stephanie Mick MD Stephanie Mick MD Cleveland Clinic Upper hemisternotomy AVR Ascending Aorta MVr Thoracotomy Based Anterior AVR Lateral Thoracotomy Mitral/Tricuspid surgery Robotically assisted surgery
NEW ZEALAND DATA SHEET
 1 MILRINONE-CLARIS (as lactate) 10 mg/ml concentrate for injection Milrinone-Claris (as lactate) 10 mg/10ml concentrate for injection. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Milrinone-Claris is a sterile,
1 MILRINONE-CLARIS (as lactate) 10 mg/ml concentrate for injection Milrinone-Claris (as lactate) 10 mg/10ml concentrate for injection. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Milrinone-Claris is a sterile,
PACKAGE LEAFLET: INFORMATION FOR THE USER. Glucose Intravenous Infusion BP 10% w/v solution for infusion Glucose (as glucose monohydrate)
 PACKAGE LEAFLET: INFORMATION FOR THE USER Glucose Intravenous Infusion BP 10% w/v solution for infusion Glucose (as glucose monohydrate) Read all of this leaflet carefully before you start using this medicine
PACKAGE LEAFLET: INFORMATION FOR THE USER Glucose Intravenous Infusion BP 10% w/v solution for infusion Glucose (as glucose monohydrate) Read all of this leaflet carefully before you start using this medicine
MOVICOL Junior Chocolate Flavour Powder for Solution (macrogol 3350)
 MOVICOL Junior Chocolate Flavour Powder for Solution (macrogol 3350) Product Name: MOVICOL Junior Chocolate Flavour Product Description: Each sachet of MOVICOL Junior Chocolate contains: Macrogol 3350
MOVICOL Junior Chocolate Flavour Powder for Solution (macrogol 3350) Product Name: MOVICOL Junior Chocolate Flavour Product Description: Each sachet of MOVICOL Junior Chocolate contains: Macrogol 3350
PACKAGE LEAFLET: INFORMATION FOR THE USER. SODIPHOS 22mEq / 10ml Concentrate for solution for infusion. Disodium phosphate dihydrate
 PACKAGE LEAFLET: INFORMATION FOR THE USER SODIPHOS 22mEq / 10ml Concentrate for solution for infusion Disodium phosphate dihydrate Read all of this leaflet carefully before you start using this medicine.
PACKAGE LEAFLET: INFORMATION FOR THE USER SODIPHOS 22mEq / 10ml Concentrate for solution for infusion Disodium phosphate dihydrate Read all of this leaflet carefully before you start using this medicine.
Neosynephrine. Name of the Medicine
 Name of the Medicine Neosynephrine Phenylephrine hydrochloride 1% injection Neosynephrine Presentation Neosynephrine is a clear, colourless, aqueous solution, free from visible particulates, in sterile
Name of the Medicine Neosynephrine Phenylephrine hydrochloride 1% injection Neosynephrine Presentation Neosynephrine is a clear, colourless, aqueous solution, free from visible particulates, in sterile
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Centre for Clinical Practice SCOPE Clinical guideline title: Intravenous fluid therapy in adults in hospital Quality standard title: Intravenous fluid
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Centre for Clinical Practice SCOPE Clinical guideline title: Intravenous fluid therapy in adults in hospital Quality standard title: Intravenous fluid
MOVICOL Junior Powder for Solution (macrogol 3350)
 MOVICOL Junior Powder for Solution (macrogol 3350) Product Name: MOVICOL Junior Product Description: Each sachet of MOVICOL Junior contains: Macrogol 3350 Sodium chloride Sodium bicarbonate Potassium chloride
MOVICOL Junior Powder for Solution (macrogol 3350) Product Name: MOVICOL Junior Product Description: Each sachet of MOVICOL Junior contains: Macrogol 3350 Sodium chloride Sodium bicarbonate Potassium chloride
PACKAGE LEAFLET: INFORMATION FOR THE USER. Nutriflex plus Solution for Infusion Amino acids / Glucose / Electrolytes
 PACKAGE LEAFLET: INFORMATION FOR THE USER Nutriflex plus Solution for Infusion Amino acids / Glucose / Electrolytes Read all of this leaflet carefully. It provides a summary of information about your medicine.
PACKAGE LEAFLET: INFORMATION FOR THE USER Nutriflex plus Solution for Infusion Amino acids / Glucose / Electrolytes Read all of this leaflet carefully. It provides a summary of information about your medicine.
Sodium Chloride 0.9% Injection, FK Std.
 Text for Prescribing Information: Sodium Chloride 0.9% Injection, FK Std. DESCRIPTION Sodium Chloride 0.9% Injection is a sterile nonpyrogenic intravenous solution that contains no bacteriostatic or antimicrobial
Text for Prescribing Information: Sodium Chloride 0.9% Injection, FK Std. DESCRIPTION Sodium Chloride 0.9% Injection is a sterile nonpyrogenic intravenous solution that contains no bacteriostatic or antimicrobial
PEDIATRIC CARDIAC RHYTHM DISTURBANCES. -Jason Haag, CCEMT-P
 PEDIATRIC CARDIAC RHYTHM DISTURBANCES -Jason Haag, CCEMT-P General: CARDIAC RHYTHM DISTURBANCES - More often the result and not the cause of acute cardiovascular emergencies - Typically the end result
PEDIATRIC CARDIAC RHYTHM DISTURBANCES -Jason Haag, CCEMT-P General: CARDIAC RHYTHM DISTURBANCES - More often the result and not the cause of acute cardiovascular emergencies - Typically the end result
Objectives: This presentation will help you to:
 emergency Drugs Objectives: This presentation will help you to: Five rights for medication administration Recognize different cardiac arrhythmias and determine the common drugs used for each one List the
emergency Drugs Objectives: This presentation will help you to: Five rights for medication administration Recognize different cardiac arrhythmias and determine the common drugs used for each one List the
Package leaflet: Information for the patient. Trasylol 10,000 KIU/ml solution for injection or infusion. Aprotinin
 Package leaflet: Information for the patient Trasylol 10,000 KIU/ml solution for injection or infusion Aprotinin Read all of this leaflet carefully before you are given this medicine because it contains
Package leaflet: Information for the patient Trasylol 10,000 KIU/ml solution for injection or infusion Aprotinin Read all of this leaflet carefully before you are given this medicine because it contains
Heart transplantation is the gold standard treatment for
 Organ Care System for Heart Procurement and Strategies to Reduce Primary Graft Failure After Heart Transplant Masaki Tsukashita, MD, PhD, and Yoshifumi Naka, MD, PhD Primary graft failure is a rare, but
Organ Care System for Heart Procurement and Strategies to Reduce Primary Graft Failure After Heart Transplant Masaki Tsukashita, MD, PhD, and Yoshifumi Naka, MD, PhD Primary graft failure is a rare, but
Vasoactive Medications. Matthew J. Korobey Pharm.D., BCCCP Critical Care Clinical Specialist Mercy St. Louis
 Vasoactive Medications Matthew J. Korobey Pharm.D., BCCCP Critical Care Clinical Specialist Mercy St. Louis Objectives List components of physiology involved in blood pressure Review terminology related
Vasoactive Medications Matthew J. Korobey Pharm.D., BCCCP Critical Care Clinical Specialist Mercy St. Louis Objectives List components of physiology involved in blood pressure Review terminology related
Chapter 9. Learning Objectives. Learning Objectives 9/11/2012. Cardiac Arrhythmias. Define electrical therapy
 Chapter 9 Cardiac Arrhythmias Learning Objectives Define electrical therapy Explain why electrical therapy is preferred initial therapy over drug administration for cardiac arrest and some arrhythmias
Chapter 9 Cardiac Arrhythmias Learning Objectives Define electrical therapy Explain why electrical therapy is preferred initial therapy over drug administration for cardiac arrest and some arrhythmias
Intra-operative Echocardiography: When to Go Back on Pump
 Intra-operative Echocardiography: When to Go Back on Pump GREGORIO G. ROGELIO, MD., F.P.C.C. OUTLINE A. Indications for Intraoperative Echocardiography B. Role of Intraoperative Echocardiography C. Criteria
Intra-operative Echocardiography: When to Go Back on Pump GREGORIO G. ROGELIO, MD., F.P.C.C. OUTLINE A. Indications for Intraoperative Echocardiography B. Role of Intraoperative Echocardiography C. Criteria
Electrolytes Solution
 Electrolytes Solution Substances that are not dissociated in solution are called nonelectrolytes, and those with varying degrees of dissociation are called electrolytes. Urea and dextrose are examples
Electrolytes Solution Substances that are not dissociated in solution are called nonelectrolytes, and those with varying degrees of dissociation are called electrolytes. Urea and dextrose are examples
ELAPRASE Idursulfase 6 mg/3 ml, Concentrate for Solution for Infusion
 ELAPRASE Idursulfase 6 mg/3 ml, Concentrate for Solution for Infusion Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about Elaprase. It does not contain
ELAPRASE Idursulfase 6 mg/3 ml, Concentrate for Solution for Infusion Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about Elaprase. It does not contain
NHS Grampian Staff Guidance For The Management Of Hypomagnesaemia In Adults. Consultation Group: See Page 4. Review Date: June 2021
 NHS...... Grampian NHS Grampian Staff Guidance For The Management Of Hypomagnesaemia In Adults Co-ordinators: Consultation Group: Approver:. Senior Medicines Information Pharmacist See Page 4 Medicine
NHS...... Grampian NHS Grampian Staff Guidance For The Management Of Hypomagnesaemia In Adults Co-ordinators: Consultation Group: Approver:. Senior Medicines Information Pharmacist See Page 4 Medicine
POTASSIUM DIHYDROGEN PHOSPHATE 13.6% CONCENTRATED INJECTION
 POTASSIUM DIHYDROGEN PHOSPHATE 13.6% CONCENTRATED INJECTION NAME OF THE MEDICINE Potassium Dihydrogen Phosphate Synonyms: potassium biphosphate, potassium acid phosphate, monopotassium phosphate, or monoibasic
POTASSIUM DIHYDROGEN PHOSPHATE 13.6% CONCENTRATED INJECTION NAME OF THE MEDICINE Potassium Dihydrogen Phosphate Synonyms: potassium biphosphate, potassium acid phosphate, monopotassium phosphate, or monoibasic
for Improved Topical Car & ac Hypothermia
 A Recirtrulating Cooling S stem for Improved Topical Car & ac Hypothermia F. L. Rosenfeldt, F.R.C.S.E., A. Fambiatos, B.Sc., J. PastorizaPinol, C.C.P., and G. R. Stirling, F.R.A.C.S. ABSTRACT A simple
A Recirtrulating Cooling S stem for Improved Topical Car & ac Hypothermia F. L. Rosenfeldt, F.R.C.S.E., A. Fambiatos, B.Sc., J. PastorizaPinol, C.C.P., and G. R. Stirling, F.R.A.C.S. ABSTRACT A simple
