Cellular cardiomyoplasty has emerged as a promising
|
|
|
- Wendy Malone
- 6 years ago
- Views:
Transcription
1 UltraRapid Communication Evidence for Fusion Between Cardiac and Skeletal Muscle Cells Hans Reinecke, Elina Minami, Veronica Poppa, Charles E. Murry Abstract Cardiomyoplasty with skeletal myoblasts may benefit cardiac function after infarction. Recent reports indicate that adult stem cells can fuse with other cell types. Because myoblasts are fusigenic cells by nature, we hypothesized they might be particularly likely to fuse with cardiomyocytes. To test this, neonatal rat cardiomyocytes labeled with LacZ and green fluorescent protein (GFP) were cocultured with unlabeled C2C12 myoblasts. After 3 days, we observed a small population of skeletal myotubes that expressed LacZ and GFP, indicating cell fusion. To test whether such fusion occurred in vivo, LacZ-expressing C2C12 myoblasts were grafted into normal nude mouse hearts. At 2 weeks after grafting, cells at the graft-host interface expressed both LacZ and cardiac-specific myosin light chain 2v (MLC2v). To test more definitively whether fusion between skeletal and cardiac muscle could occur, we used a Cre/lox reporter system that activated LacZ only upon cell fusion. When neonatal cardiomyocytes from -myosin heavy chain promoter ( -MHC)-Cre mice were cocultured with myoblasts from floxed-lacz reporter mice, LacZ was activated in a subset of cells, indicating cell fusion occurred in vitro. Finally, we grafted the floxed-lacz myoblasts into normal hearts of -MHC-Cre and -MHC-Cre mice (n 5 each). Hearts analyzed at 4 days and 1 week after transplantation demonstrated activation of LacZ when the skeletal muscle cells were implanted into hearts of -MHC-Cre mice, but not after implantation into -MHC-Cre mice. These data indicate that skeletal muscle cell grafting gives rise to a subpopulation of skeletal-cardiac hybrid cells with a currently unknown phenotype. The full text of this article is available online at (Circ Res. 2004;94:e56-e60.) Key Words: cardiomyoplasty skeletal myoblast cell transplantation cell fusion Cre/lox Cellular cardiomyoplasty has emerged as a promising therapy for myocardial infarct repair. Skeletal myoblasts and bone marrow derived stem cells have been tried in various animal models and are now undergoing clinical trials. 1 4 A recent study has shown that circulating cells derived from transplanted bone marrow can fuse with cardiomyocytes, Purkinje cells and hepatocytes. 5 Because skeletal myoblasts are fusigenic cells by nature, we tested if myoblasts can fuse with cardiomyocytes. Our study shows that skeletal myoblast grafting into the heart results in fusion with host cardiomyocytes, giving rise to a subpopulation of hybrid cells. Materials and Methods Cell Culture Mouse neonatal cardiomyocytes and primary neonatal skeletal myoblasts were isolated as described previously. 6,7 Spontaneous cell fusion (coculture) experiments were set up in gelatin-coated 6-well plates at a total density of cells/cm 2 (1:1 ratio) in DMEM/ M199 (4:1) supplemented with 10% horse serum, 5% FBS, and 6 g/ml insulin. These conditions were optimized for cardiomyocytes while allowing terminal differentiation of skeletal myoblasts into multinucleated myotubes. Forced fusion was induced by polyethylene glycol (Hybrimax- PEG, 50% solution; Sigma). Neonatal cardiomyocytes were infected with the Cre recombinase adenovirus (AdCre; Microbix Biosystems) and C2C12 skeletal myoblast were infected with Adfloxed-lacZ adenovirus (Microbix Biosystems) at 300 virus particles per cell. After infection, cells were extensively washed, trypsinized, and cocultured overnight at ratios of 10:1 (cardiomyocytes:c2c12 myoblasts) and 1:10, respectively. Cell fusion was induced by addition of PEG (50% solution for 90 seconds). PEG was rinsed off and regular medium was added. After 3 days, cultures were fixed with 2% paraformaldehyde and subjected to X-GAL assays. Animal Procedures The study protocol was approved by the Institutional Animal Care and Use Committee. Alpha-MHC-Cre/FVB mice (kindly provided by Dr M.D. Schneider, Baylor College of Medicine, Houston, Tex) express Cre recombinase only in cardiomyocytes. 8 To achieve graft-host tolerance, -MHC-Cre/FVB mice were crossed with C57Bl/6 wild-type mice, and F1 animals were used as graft recipients. Accordingly, Cre-reporter R26R/C57Bl/6 were crossed with wild-type FVB mice, and F1 animals were used to isolate neonatal skeletal myoblasts as described. 7 The R26R mouse carries a lacz allele that is separated from its constitutively active ROSA26 promoter by a loxp-flanked neomycin cassette (floxed-lacz), thus lacz is expressed only upon Cre-mediated excision of the neomycin cassette. 9 Nude mice were used for grafting of C2C12 myoblasts tagged with a lacz retrovirus (n 4). -MHC-Cre,or -MHC-Cre Original received January 22, 2004; revision received February 18, 2004; accepted February 23, From the Department of Pathology, University of Washington, Seattle, Wash. Correspondence to Hans Reinecke, PhD, University of Washington, Department of Pathology, Box , HSB Room D-514. Seattle, WA hreineck@u.washington.edu 2004 American Heart Association, Inc. Circulation Research is available at DOI: /01.RES
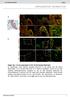
2 2 Circulation Research April 2, 2004 Figure 1. A and B, Coculture of neonatal rat cardiomyocytes and mouse C2C12 skeletal myoblasts. Double-labeled cardiomyocytes (adenoviral nuclear-targeted LacZ, GFP ) were cocultured with nonlabeled C2C12 myoblasts. After 3 days in coculture, multinucleated myotubes with LacZpositive (blue) nuclei and green fluorescent cytoplasm were observed, suggesting cell fusion. Note the presence of a LacZ (blue) gradient in the nuclei, suggesting second-order uptake. C through F, Grafting of nuclear LacZ-tagged C2C12 myoblasts (C; X-GAL assay in vitro) into normal nude mouse hearts (D; X-GAL assay with contrast red). At 2 weeks, we observed cells that were positive for LacZ and the cardiac marker myosin light chain 2v (MLC2v, green fluorescence) (E and F; arrows; note the relatively high background in F results from merging brightfield and fluorescence images). These cells were located at the graft-host interface. G through J, Cre/lox recombination in vitro. Neonatal cardiomyocytes from -MHC-Cre mice were cocultured for 3 days with neonatal skeletal myoblasts from floxed-lacz mice. Activation of LacZ expression (G and H, punctate blue staining, arrows) was observed in cocultures but not in monocultures of the respective cell type (I and J). X-GAL assay with contrast red. Note that in this model Cre-mediated LacZ expression is not nuclear-targeted and punctate LacZ staining is not associated with cell nuclei. (control) mice were used as recipients of floxed-lacz myoblasts (n 8 each). In a separate set of experiments, C2C12 myoblasts were infected with an adenovirus carrying a floxed-lacz gene (AdfloxedlacZ; 300 particles/cell) and grafted into -MHC-Cre and -MHC- Cre (control) mice (n 6 and n 4, respectively). Normal hearts were injected with a standard dose of myoblasts as previously described. 10 Mice were euthanized at 4 days and 1 week ( -MHC- Cre mice) or 2 weeks (nude mice). Hearts from -MHC-Cre mice injected with Adfloxed-lacZ infected C2C12 myoblasts were harvested at 4 days. At this early time point, no immune response to the adenovirus-infected allograft was noted. At the defined time points, hearts were excised and routinely processed for cryosections. Adenovirus Infections Neonatal rat cardiomyocytes were infected at a total of 300 virus particles per cell with adenoviruses encoding nuclear targeted LacZ 11 and cytoplasmic GFP, 12 respectively. Then, cultures were washed extensively and fresh medium was added. Cocultures were set up the following day. The integrity of the reporter cells (floxed-lacz myoblasts) was confirmed by infection with a Cre recombinase adenovirus (AdCre) and subsequent 5-bromo-4-chloro-3-indolyl-b- D-galactoside (X-GAL) staining. When C2C12 myoblasts infected with the Adfloxed-lacZ adenovirus were used for grafting experiments, cells were infected the day before grafting (300 virus particles per cell). On the day of grafting, cultures were washed extensively before cell transplantation. An aliquot of the cells was used to confirm the infection efficiency and integrity of the reporter by infection with the Cre recombinase adenovirus. Results Cell Fusion Detected by Virus-Mediated Cell Tagging Neonatal rat cardiomyocytes tagged with GFP and nuclear LacZ were cocultured with unlabeled C2C12 myoblasts. After 3 days, we observed multinucleated myotubes that carried both the GFP and the nuclear LacZ tag (Figures 1A
3 Reinecke et al Fusion Between Cardiac and Skeletal Myocytes 3 Figure 2. A and B, Cre/lox recombination in vivo. Skeletal myoblasts from floxed-lacz mice were grafted into -MHC-Cre hearts. -MHC-Cre hearts served as controls. Activation of LacZ expression (blue punctate staining) was observed in -MHC-Cre but not in -MHC-Cre hearts (not shown). X-GAL assay with contrast red. C through F, Mouse C2C12 myoblasts were infected with Adfloxed-lacZ and grafted into normal hearts of -MHC-Cre or into -MHC-Cre mice (control, not shown). Approximate outline of the graft area showing 2 cells (arrows) with abundant cytoplasmic LacZ expression. The boxed area is shown in D at higher magnification. Some of the hybrid cells exhibited abundant cytoplasmic LacZ expression (D and F). More typically, however, we observed a punctate LacZ staining pattern (E). F, Fusion-derived cell with abundant LacZ expression that was observed outside of the main graft area. Images were derived from 2 hearts of -MHC- Cre mice. No activation of LacZ expression was observed after grafting of -MHC-Cre mice. X-GAL assay with contrast red. and 1B), suggesting fusion of double-labeled cardiomyocytes with unlabeled C2C12 myoblasts. Because replicationdeficient adenoviruses were used for labeling the cardiomyocytes and cells were washed extensively before coculture, it is unlikely that free virus was carried over into cocultures. To test whether fusion between skeletal and cardiac muscle cells could occur in vivo, we generated a C2C12 myoblast clone retrovirally tagged with nuclear-targeted LacZ, and grafted these cells into normal hearts of nude mice (Figures 1C and 1D). At 2 weeks after grafting, we performed X-GAL assays in conjunction with immunostaining for the cardiac-specific myosin light chain 2v (MLC2v). At the border zone between host and graft, we detected a small number of cells that expressed the nuclear LacZ tag and were positive for MLC2v (Figures 1E and 1F), suggesting fusion between myoblasts and cardiomyocytes had occurred in vivo. Cell Fusion Detected by Cre/lox Recombination To test more definitively whether fusion had occurred, we isolated neonatal cardiomyocytes from mice where expression of Cre recombinase was driven by the cardiac-specific -MHC promoter. 8 These cardiomyocytes were cocultured with myoblasts from floxed-lacz reporter mice where the LacZ expression is normally blocked by the presence of a Cre-excisable stuffer sequence. 9 After 3 days in coculture, X-GAL assays revealed the presence of LacZ-positive multinucleated myotubes (Figures 1G and 1H), strongly indicating fusion between the two cell types. Monocultures of the respective cell types did not yield any LacZ-positive cells (Figures 1I and 1J). We then used Cre/lox recombination for cell grafting experiments. Floxed-lacZ skeletal myoblasts were grafted into the uninjured hearts of -MHC-Cre or into -MHC-Cre mice (control). At 4 days and 1 week after cell implantation, LacZ expression was observed in cells at the graft-host interface (Figures 2A and 2B). In contrast, LacZ expression was not detected in grafted hearts of -MHC-Cre mice (data not shown). As a final test for fusion, C2C12 myoblasts were infected with an adenovirus carrying a floxed-lacz gene and grafted into normal hearts of -MHC-Cre or into -MHC-Cre mice (control). Similar to the results with primary floxed-lacz myoblasts, we observed LacZ expression primarily in cells at or close to the graft-host interface (Figures 2C through 2F). In these experiments, some cells displayed punctate LacZ staining (Figure 2E), whereas others showed abundant LacZ staining (Figures 2C, 2D, and 2F). Again, no LacZ expression was detected in grafted hearts of -MHC-Cre mice (data not shown). A final experiment focused on how the stoichiometry of the fusion partners affected morphology of the hybrid cells. We hypothesized that the morphology of fused cells may be dependent on the number of nuclei and/ or cytoplasm contributed by each cell type. Since spontaneous cell fusion was infrequent we explored forced fusion by means of polyethyl-
4 4 Circulation Research April 2, 2004 Figure 3. Forced fusion induced by polyethylene glycol (PEG). Neonatal cardiomyocytes were infected with the Cre recombinase adenovirus (AdCre) and C2C12 skeletal myoblast were infected with Adfloxed-lacZ adenovirus. After infection, cells were extensively washed, trypsinized, and cocultured overnight at ratios of 10:1 and 1:10, respectively. Then, cell fusion was induced by addition of PEG. After 3 days, fused cells were detected by X-GAL assay. Ad-Cre infected neonatal cardiomyocytes were mixed with Adfloxed-lacZ infected C2C12 skeletal at a ratio of 10 cardiomyocytes to 1 myoblast. PEG-forced fusion resulted in Lac-Z positive cells that were mono-, bi-, and trinucleated (left and inset). In contrast, when the ratio was reversed (1 cardiomyocyte:10 C2C12 myoblasts), LacZ-positive fusion cells appeared as typical skeletal myotubes showing multinucleation and branching (right). ene glycol (PEG) as a way to study fusion cell morphology. When Ad-Cre infected neonatal cardiomyocytes were mixed with Adfloxed-lacZ infected C2C12 skeletal myoblasts at a ratio of 10:1, PEG-forced fusion resulted in LacZ-positive cells that were mono-, bi-, and trinucleated (Figure 3). In contrast, when the ratio was reversed (1 cardiomyocyte:10 C2C12 myoblasts) LacZ-positive fusion cells appeared as typical skeletal myotubes with multinucleation and branching (Figure 3). Discussion The major finding of the present study is that skeletal myoblasts can fuse with cardiomyocytes in vitro and in vivo. Cell fusion was detected by using virus-mediated cell labeling (GFP, LacZ) and by Cre/lox recombination. Both coculture models and in vivo cardiac grafting experiments yielded a similarly small (less than 0.01% of seeded or grafted cells) but readily detectable population of hybrid cells. Interestingly, in vitro the fused cells morphologically resembled skeletal myotubes, whereas in vivo they resembled cardiomyocytes. Forced fusion experiments with varying ratios between cardiomyocytes and skeletal myoblasts suggested that the morphology of fused cells was dependent on the stoichiometry of nuclei and/or cytoplasm contributed by each cell type. It should be mentioned that our Cre/lox experiments were not designed to detect a cardiomyogenic transdifferentiation event (eg, from a multipotent progenitor cell) independent of fusion. The studies involving C2C12 myoblasts would not be expected to contain progenitor cells, because they are an immortalized, clonal cell line. Our primary myoblast cultures clearly had a heterogeneous mix (although were 85% desmin-positive) and could have contained progenitor cells. In recent years, it has become apparent that cells of different origin may fuse with one another, thus adopting properties of the fusion partner. Although first described in vitro, 13,14 fusion subsequently was shown to explain the in vivo transdifferentiation of hematopoietic stem cells into hepatocytes. 15,16 The question arises whether cell fusion can explain previous reports of transdifferentiation of various cell types into cardiomyocytes. Our observations are consistent with the very low levels of transdifferentiation reported by Robinson et al 17 (intraarterial delivery of myoblasts), Jackson et al 18 (hematopoietic stem cell transplantation after lethal irradiation), Malouf et al 19 (intramyocardial injection of liver-derived progenitor cells), and Laflamme et al 20 ( transgender cardiac transplantation in humans). They are inconsistent, however, with high level transdifferentiation reported in other studies When the hybrid cells expressed cytoplasmic LacZ from the ROSA26 promoter, we typically observed a punctate LacZ-staining pattern in vitro (Figures 1G and 1H) and in vivo (Figures 2A and 2B). A similar, punctate staining pattern was observed in rare cardiomyocytes ( 0.02%) in the peri-infarct region by Jackson et al, 18 after performing bone marrow transplantation with highly enriched ROSA26-derived (LacZ ) hematopoietic stem cells. Although the authors proposed that the LacZ cardiomyocytes arose via transdifferentiation, one may hypothesize that these cells in fact resulted from cell fusion. Indeed, while our work was in the final phases of preparation, two other studies showed fusion of adult cells with cardiomyocytes. By using Cre/lox recombination, Alvarez-Dolado et al 5 showed that bone marrow derived cells could fuse with cardiomyocytes in the uninjured heart, whereas Oh et al 25 showed that intravenously infused progenitor cells from the adult heart could fuse with cardiomyocytes in the setting of myocardial infarction. Our study extends this work by adding skeletal myoblasts as a clinically relevant cell type that can fuse with cardiac myocytes in vitro and in vivo. Although a rare event, the appearance of cardiac-skeletal hybrid cells with unknown (electrical) properties at the graft-host interface may impact autologous skeletal myoblast grafting for cardiac repair, which is already undergoing clinical trials. 1 3 Acknowledgments This work was supported by NIH grants HL61553, HL64387, and HL References 1. Menasche P, Hagege AA, Vilquin JT, Desnos M, Abergel E, Pouzet B, Bel A, Sarateanu S, Scorsin M, Schwartz K, Bruneval P, Benbunan M, Marolleau JP, Duboc D. Autologous skeletal myoblast transplantation for severe postinfarction left ventricular dysfunction. J Am Coll Cardiol. 2003;41: Hagege AA, Carrion C, Menasche P, Vilquin JT, Duboc D, Marolleau JP, Desnos M, Bruneval P. Viability and differentiation of autologous skeletal myoblast grafts in ischaemic cardiomyopathy. Lancet. 2003;361: Pagani FD, DerSimonian H, Zawadzka A, Wetzel K, Edge AS, Jacoby DB, Dinsmore JH, Wright S, Aretz TH, Eisen HJ, Aaronson KD. Autologous skeletal myoblasts transplanted to ischemia-damaged myocardium in humans: histological analysis of cell survival and differentiation. J Am Coll Cardiol. 2003;41:
5 Reinecke et al Fusion Between Cardiac and Skeletal Myocytes 5 4. Strauer BE, Brehm M, Zeus T, Kostering M, Hernandez A, Sorg RV, Kogler G, Wernet P. Repair of infarcted myocardium by autologous intracoronary mononuclear bone marrow cell transplantation in humans. Circulation. 2002;106: Alvarez-Dolado M, Pardal R, Garcia-Verdugo JM, Fike JR, Lee HO, Pfeffer K, Lois C, Morrison SJ, Alvarez-Buylla A. Fusion of bonemarrow-derived cells with Purkinje neurons, cardiomyocytes and hepatocytes. Nature. 2003;425: Deng XF, Rokosh DG, Simpson PC. Autonomous and growth factorinduced hypertrophy in cultured neonatal mouse cardiac myocytes: comparison with rat. Circ Res. 2000;87: Reinecke H, MacDonald GH, Hauschka SD, Murry CE. Electromechanical coupling between skeletal and cardiac muscle: implications for infarct repair. J Cell Biol. 2000;149: Agah R, Frenkel PA, French BA, Michael LH, Overbeek PA, Schneider MD. Gene recombination in postmitotic cells: targeted expression of Cre recombinase provokes cardiac-restricted, site-specific rearrangement in adult ventricular muscle in vivo. J Clin Invest. 1997;100: Soriano P. Generalized lacz expression with the ROSA26 Cre reporter strain. Nat Genet. 1999;21: Reinecke H, Murry CE. Cell grafting for cardiac repair. Methods Mol Biol. 2003;219: Murry CE, Kay MA, Bartosek T, Hauschka SD, Schwartz SM. Muscle differentiation during repair of myocardial necrosis in rats via gene transfer with MyoD. J Clin Invest. 1996;98: He TC, Zhou S, da Costa LT, Yu J, Kinzler KW, Vogelstein B. A simplified system for generating recombinant adenoviruses. Proc Natl Acad Sci U S A. 1998;95: Terada N, Hamazaki T, Oka M, Hoki M, Mastalerz DM, Nakano Y, Meyer EM, Morel L, Petersen BE, Scott EW. Bone marrow cells adopt the phenotype of other cells by spontaneous cell fusion. Nature. 2002; 416: Ying QL, Nichols J, Evans EP, Smith AG. Changing potency by spontaneous fusion. Nature. 2002;416: Vassilopoulos G, Wang PR, Russell DW. Transplanted bone marrow regenerates liver by cell fusion. Nature. 2003;422: Wang X, Willenbring H, Akkari Y, Torimaru Y, Foster M, Al-Dhalimy M, Lagasse E, Finegold M, Olson S, Grompe M. Cell fusion is the principal source of bone-marrow-derived hepatocytes. Nature. 2003;422: Robinson SW, Cho PW, Levitsky HI, Olson JL, Hruban RH, Acker MA, Kessler PD. Arterial delivery of genetically labelled skeletal myoblasts to the murine heart: long-term survival and phenotypic modification of implanted myoblasts. Cell Transplant. 1996;5: Jackson KA, Majka SM, Wang H, Pocius J, Hartley CJ, Majesky MW, Entman ML, Michael LH, Hirschi KK, Goodell MA. Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells. J Clin Invest. 2001;107: Malouf NN, Coleman WB, Grisham JW, Lininger RA, Madden VJ, Sproul M, Anderson PA. Adult-derived stem cells from the liver become myocytes in the heart in vivo. Am J Pathol. 2001;158: Laflamme MA, Myerson D, Saffitz JE, Murry CE. Evidence for cardiomyocyte repopulation by extracardiac progenitors in transplanted human hearts. Circ Res. 2002;90: Chiu RC, Zibaitis A, Kao RL. Cellular cardiomyoplasty: myocardial regeneration with satellite cell implantation. Ann Thorac Surg. 1995;60: Taylor DA, Atkins BZ, Hungspreugs P, Jones TR, Reedy MC, Hutcheson KA, Glower DD, Kraus WE. Regenerating functional myocardium: improved performance after skeletal myoblast transplantation. Nat Med. 1998;4: Orlic D, Kajstura J, Chimenti S, Jakoniuk I, Anderson SM, Li B, Pickel J, McKay R, Nadal-Ginard B, Bodine DM, Leri A, Anversa P. Bone marrow cells regenerate infarcted myocardium. Nature. 2001;410: Quaini F, Urbanek K, Beltrami AP, Finato N, Beltrami CA, Nadal-Ginard B, Kajstura J, Leri A, Anversa P. Chimerism of the transplanted heart. N Engl J Med. 2002;346: Oh H, Bradfute SB, Gallardo TD, Nakamura T, Gaussin V, Mishina Y, Pocius J, Michael LH, Behringer RR, Garry DJ, Entman ML, Schneider MD. Cardiac progenitor cells from adult myocardium: homing, differentiation, and fusion after infarction. Proc Natl Acad Sci U S A. 2003;100:
Cardiac Myocytes are Recruited by Bone Marrow-Derived Cells in Intact Murine Heart
 Kobe J. Med. Sci., Vol. 48, No. 6, pp. 161-166, 2002 Cardiac Myocytes are Recruited by Bone Marrow-Derived Cells in Intact Murine Heart SEIMI SATOMI-KOBAYASHI 1, SEINOSUKE KAWASHIMA 1*, TSUYOSHI SAKODA
Kobe J. Med. Sci., Vol. 48, No. 6, pp. 161-166, 2002 Cardiac Myocytes are Recruited by Bone Marrow-Derived Cells in Intact Murine Heart SEIMI SATOMI-KOBAYASHI 1, SEINOSUKE KAWASHIMA 1*, TSUYOSHI SAKODA
Stem Cell Therapy for Ischemic Heart Disease : A Status Report
 Stem Cell Therapy for Ischemic Heart Disease : A Status Report Do Sun Lim, M.D. Department of Internal Medicine Korea University College of Medicine Anam Hospital E mail : dslmd@kumc.or.kr Abstract Myocardial
Stem Cell Therapy for Ischemic Heart Disease : A Status Report Do Sun Lim, M.D. Department of Internal Medicine Korea University College of Medicine Anam Hospital E mail : dslmd@kumc.or.kr Abstract Myocardial
Research progress of adult stem cells and clinical applications
 18 4 2006 8 Chinese Bulletin of Life Sciences Vol. 18, No. 4 Aug., 2006 1004-0374(2006)04-0328-05 100850 Q831 A Research progress of adult stem cells and clinical applications XI Jia-Fei, WANG Yun-Fang,
18 4 2006 8 Chinese Bulletin of Life Sciences Vol. 18, No. 4 Aug., 2006 1004-0374(2006)04-0328-05 100850 Q831 A Research progress of adult stem cells and clinical applications XI Jia-Fei, WANG Yun-Fang,
Citation for published version (APA): Velde, S. V. D. (2006). Stem cell-mediated regeneration of the infarcted heart: inflammation rules?. s.n.
 University of Groningen Stem cell-mediated regeneration of the infarcted heart Velde, Susanne van der IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to
University of Groningen Stem cell-mediated regeneration of the infarcted heart Velde, Susanne van der IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to
Resident cardiac stem cells: how to find and use them
 Resident cardiac stem cells: how to find and use them G. Hasenfuß Cardiology and Pneumology Heart Research Center Göttingen Georg-August-University Göttingen Definition: Stem cell Selfrenewal Stem cell
Resident cardiac stem cells: how to find and use them G. Hasenfuß Cardiology and Pneumology Heart Research Center Göttingen Georg-August-University Göttingen Definition: Stem cell Selfrenewal Stem cell
Stem cells of different origins have been observed to
 Both Cell Fusion and Transdifferentiation Account for the Transformation of Human Peripheral Blood CD34-Positive Cells Into Cardiomyocytes In Vivo Sui Zhang, MD, PhD; Dachun Wang, MD; Zeev Estrov, MD;
Both Cell Fusion and Transdifferentiation Account for the Transformation of Human Peripheral Blood CD34-Positive Cells Into Cardiomyocytes In Vivo Sui Zhang, MD, PhD; Dachun Wang, MD; Zeev Estrov, MD;
Paracrine Mechanisms in Adult Stem Cell Signaling and Therapy
 Paracrine Mechanisms in Adult Stem Cell Signaling and Therapy Massimiliano Gnecchi, Zhiping Zhang, Aiguo Ni, Victor J. Dzau Circulation Research 2008 Nov 21;103(11):1204-19 Introduction(1) After AMI all
Paracrine Mechanisms in Adult Stem Cell Signaling and Therapy Massimiliano Gnecchi, Zhiping Zhang, Aiguo Ni, Victor J. Dzau Circulation Research 2008 Nov 21;103(11):1204-19 Introduction(1) After AMI all
Stem Cells. Keith Channon. Department of Cardiovascular Medicine University of Oxford John Radcliffe Hospital, Oxford
 Stem Cells Keith Channon Department of Cardiovascular Medicine University of Oxford John Radcliffe Hospital, Oxford Adult Stem Cells Unique cells that are capable of self-renewal Have the ability to differentiate
Stem Cells Keith Channon Department of Cardiovascular Medicine University of Oxford John Radcliffe Hospital, Oxford Adult Stem Cells Unique cells that are capable of self-renewal Have the ability to differentiate
Supplemental Table 1 Clinical trials of cell-based cardiac repair without controls or with nonrandomized study design
 Cell-Based Therapy for Myocar Ischemia and Infarction: Pathophysiological Mechanisms Supplemental Table 1 Clinical trials of cell-based cardiac repair without s or with nonrandom study design Head-tohead
Cell-Based Therapy for Myocar Ischemia and Infarction: Pathophysiological Mechanisms Supplemental Table 1 Clinical trials of cell-based cardiac repair without s or with nonrandom study design Head-tohead
The concept of regenerative medicine using the body s
 MINI-REVIEW: EXPERT OPINIONS Stem Cell Therapy in Perspective Bodo E. Strauer, MD; Ran Kornowski, MD The concept of regenerative medicine using the body s own stem cells and growth factors to repair tissues
MINI-REVIEW: EXPERT OPINIONS Stem Cell Therapy in Perspective Bodo E. Strauer, MD; Ran Kornowski, MD The concept of regenerative medicine using the body s own stem cells and growth factors to repair tissues
Stem cells: units of development and regeneration. Fernando D. Camargo Ph.D. Whitehead Fellow Whitehead Institute for Biomedical Research.
 Stem cells: units of development and regeneration Fernando D. Camargo Ph.D. Whitehead Fellow Whitehead Institute for Biomedical Research Concepts 1. Embryonic vs. adult stem cells 2. Hematopoietic stem
Stem cells: units of development and regeneration Fernando D. Camargo Ph.D. Whitehead Fellow Whitehead Institute for Biomedical Research Concepts 1. Embryonic vs. adult stem cells 2. Hematopoietic stem
The concept of myogenic cell transplantation into the
 Noncultured, Autologous, Skeletal Muscle Cells Can Successfully Engraft Into Ovine Myocardium Nicolas Borenstein, DVM, MSc; Patrick Bruneval, MD; Mehrak Hekmati, PhD; Christophe Bovin; Luc Behr, DVM; Christian
Noncultured, Autologous, Skeletal Muscle Cells Can Successfully Engraft Into Ovine Myocardium Nicolas Borenstein, DVM, MSc; Patrick Bruneval, MD; Mehrak Hekmati, PhD; Christophe Bovin; Luc Behr, DVM; Christian
REVIEW Hematopoietic, vascular and cardiac fates of bone marrow-derived stem cells
 (2002) 9, 648 652 2002 Nature Publishing Group All rights reserved 0969-7128/02 $25.00 www.nature.com/gt REVIEW Hematopoietic, vascular and cardiac fates of bone marrow-derived stem cells KK Hirschi 1
(2002) 9, 648 652 2002 Nature Publishing Group All rights reserved 0969-7128/02 $25.00 www.nature.com/gt REVIEW Hematopoietic, vascular and cardiac fates of bone marrow-derived stem cells KK Hirschi 1
Regeneration Gaps Observations on Stem Cells and Cardiac Repair
 Journal of the American College of Cardiology Vol. 47, No. 9, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2006.02.002
Journal of the American College of Cardiology Vol. 47, No. 9, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2006.02.002
ENDOGENOUS CARDIAC STEM CELLS IN THE REGENERATION OF ACUTE AND CHRONIC ISCHEMIC MYOCARDIUM
 ENDOGENOUS CARDIAC STEM CELLS IN THE REGENERATION OF ACUTE AND CHRONIC ISCHEMIC MYOCARDIUM Bernardo Nadal-Ginard, M.D., Ph.D. New York Medical College Angioplasty Summit 2004, Seoul 04/29/04 MYOCARDIAL
ENDOGENOUS CARDIAC STEM CELLS IN THE REGENERATION OF ACUTE AND CHRONIC ISCHEMIC MYOCARDIUM Bernardo Nadal-Ginard, M.D., Ph.D. New York Medical College Angioplasty Summit 2004, Seoul 04/29/04 MYOCARDIAL
CSP. Cardiopulmonary Support and Physiology
 Direct cell-cell interaction of cardiomyocytes is key for bone marrow stromal cells to go into cardiac lineage in vitro Shinya Fukuhara, MD, a Shinji Tomita, MD, PhD, b Seiji Yamashiro, MD, MSc, c Takayuki
Direct cell-cell interaction of cardiomyocytes is key for bone marrow stromal cells to go into cardiac lineage in vitro Shinya Fukuhara, MD, a Shinji Tomita, MD, PhD, b Seiji Yamashiro, MD, MSc, c Takayuki
Autologous intramyocardial injection of cultured skeletal muscle-derived stem cells in patients with non-acute myocardial infarction
 European Heart Journal (2003) 24, 2012 2020 Clinical research Autologous intramyocardial injection of cultured skeletal muscle-derived stem cells in patients with non-acute myocardial infarction Jesús
European Heart Journal (2003) 24, 2012 2020 Clinical research Autologous intramyocardial injection of cultured skeletal muscle-derived stem cells in patients with non-acute myocardial infarction Jesús
Journal of the American College of Cardiology Vol. 46, No. 9, by the American College of Cardiology Foundation ISSN /05/$30.
 Journal of the American College of Cardiology Vol. 46, No. 9, 2005 2005 by the American College of Cardiology Foundation ISSN 0735-1097/05/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2005.01.069
Journal of the American College of Cardiology Vol. 46, No. 9, 2005 2005 by the American College of Cardiology Foundation ISSN 0735-1097/05/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2005.01.069
Cardiac Regeneration. Piero Anversa, MD, Annarosa Leri, MD, Jan Kajstura, PHD Valhalla, New York THE HEART AS A POST-MITOTIC ORGAN: THE CONTROVERSY
 Journal of the American College of Cardiology Vol. 47, No. 9, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2006.02.003
Journal of the American College of Cardiology Vol. 47, No. 9, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2006.02.003
Transplantation of undifferentiated murine embryonic stem cells in the heart: teratoma formation and immune response
 The FASEB Journal Research Communication Transplantation of undifferentiated murine embryonic stem cells in the heart: teratoma formation and immune response Jeannette Nussbaum,*,,,1 Elina Minami,*,,,,1
The FASEB Journal Research Communication Transplantation of undifferentiated murine embryonic stem cells in the heart: teratoma formation and immune response Jeannette Nussbaum,*,,,1 Elina Minami,*,,,,1
The New England Journal of Medicine EVIDENCE THAT HUMAN CARDIAC MYOCYTES DIVIDE AFTER MYOCARDIAL INFARCTION
 EVIDENCE THAT HUMAN CARDIAC MYOCYTES DIVIDE AFTER MYOCARDIAL INFARCTION ANTONIO P. BELTRAMI, M.D., KONRAD URBANEK, M.D., JAN KAJSTURA, PH.D., SHAO-MIN YAN, M.D., NICOLETTA FINATO, M.D., ROSSANA BUSSANI,
EVIDENCE THAT HUMAN CARDIAC MYOCYTES DIVIDE AFTER MYOCARDIAL INFARCTION ANTONIO P. BELTRAMI, M.D., KONRAD URBANEK, M.D., JAN KAJSTURA, PH.D., SHAO-MIN YAN, M.D., NICOLETTA FINATO, M.D., ROSSANA BUSSANI,
Differential Myocardial Infarct Repair with Muscle Stem Cells Compared to Myoblasts
 doi:10.1016/j.ymthe.2005.07.686 Differential Myocardial Infarct Repair with Muscle Stem Cells Compared to Myoblasts Hideki Oshima, 1,2,3 Thomas R. Payne, 3,4 Kenneth L. Urish, 3,4 Tetsuro Sakai, 2,3,5
doi:10.1016/j.ymthe.2005.07.686 Differential Myocardial Infarct Repair with Muscle Stem Cells Compared to Myoblasts Hideki Oshima, 1,2,3 Thomas R. Payne, 3,4 Kenneth L. Urish, 3,4 Tetsuro Sakai, 2,3,5
Mesenchymal stem cells modified with Akt prevent remodeling and restore performance of infarcted hearts
 Mesenchymal stem cells modified with Akt prevent remodeling and restore performance of infarcted hearts Abeel A Mangi, Nicolas Noiseux, Deling Kong, Huamei He, Mojgan Rezvani, Joanne S Ingwall & Victor
Mesenchymal stem cells modified with Akt prevent remodeling and restore performance of infarcted hearts Abeel A Mangi, Nicolas Noiseux, Deling Kong, Huamei He, Mojgan Rezvani, Joanne S Ingwall & Victor
Meeting Report. From December 8 to 11, 2012 at Atlanta, GA, U.S.A
 Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Stem cell homing and engraftment to the heart and new
 Review: Current Perspective Molecular Genetic Advances in Cardiovascular Medicine Focus on the Myocyte Piero Anversa, MD; Mark A. Sussman, PhD; Roberto Bolli, MD Stem cell homing and engraftment to the
Review: Current Perspective Molecular Genetic Advances in Cardiovascular Medicine Focus on the Myocyte Piero Anversa, MD; Mark A. Sussman, PhD; Roberto Bolli, MD Stem cell homing and engraftment to the
Remodeling of the left ventricle after myocardial infarction
 Clinical Investigation and Reports Repair of Infarcted Myocardium by Autologous Intracoronary Mononuclear Bone Marrow Cell Transplantation in Humans Bodo E. Strauer, MD; Michael Brehm, MD; Tobias Zeus,
Clinical Investigation and Reports Repair of Infarcted Myocardium by Autologous Intracoronary Mononuclear Bone Marrow Cell Transplantation in Humans Bodo E. Strauer, MD; Michael Brehm, MD; Tobias Zeus,
Cell implantation after myocardial infarction: a 10 years experience from the ICREC laboratory
 Cell implantation after myocardial infarction: a 10 years experience from the ICREC laboratory BANFF-SCT Joint Scientific Meeting 2017 Barcelona, 29 th March Santi Roura, PhD Grup ICREC IGTP HuGTiP (Badalona)
Cell implantation after myocardial infarction: a 10 years experience from the ICREC laboratory BANFF-SCT Joint Scientific Meeting 2017 Barcelona, 29 th March Santi Roura, PhD Grup ICREC IGTP HuGTiP (Badalona)
Protocol. Progenitor Cell Therapy for the Treatment of Damaged Myocardium due to Ischemia
 (20218) Medical Benefit Effective Date: 01/01/11 Next Review Date: 07/18 Preauthorization No Review Dates: 09/10, 07/11, 07/12, 07/13, 07/14, 07/15, 07/16, 07/17 This protocol considers this test or procedure
(20218) Medical Benefit Effective Date: 01/01/11 Next Review Date: 07/18 Preauthorization No Review Dates: 09/10, 07/11, 07/12, 07/13, 07/14, 07/15, 07/16, 07/17 This protocol considers this test or procedure
Nature Immunology: doi: /ni Supplementary Figure 1. Huwe1 has high expression in HSCs and is necessary for quiescence.
 Supplementary Figure 1 Huwe1 has high expression in HSCs and is necessary for quiescence. (a) Heat map visualizing expression of genes with a known function in ubiquitin-mediated proteolysis (KEGG: Ubiquitin
Supplementary Figure 1 Huwe1 has high expression in HSCs and is necessary for quiescence. (a) Heat map visualizing expression of genes with a known function in ubiquitin-mediated proteolysis (KEGG: Ubiquitin
Medical Coverage Policy Progenitor Cell Therapy for the Treatment of Damaged Myocardium due to Ischemia
 Medical Coverage Policy Progenitor Cell Therapy for the Treatment of Damaged Myocardium due to Ischemia EFFECTIVE DATE: 02 01 2017 POLICY LAST UPDATED: 02 20 2018 OVERVIEW Progenitor cell therapy describes
Medical Coverage Policy Progenitor Cell Therapy for the Treatment of Damaged Myocardium due to Ischemia EFFECTIVE DATE: 02 01 2017 POLICY LAST UPDATED: 02 20 2018 OVERVIEW Progenitor cell therapy describes
Stem Cells to Regenerate Cardiac Tissue in Heart Failure
 E DITORIALS Stem Cells to Regenerate Cardiac Tissue in Heart Failure Felipe Prósper Cardoso a, Jesús Herreros González, b and Eduardo Alegría Ezquerra b a Servicio de Hematología y Área de Terapia Celular,
E DITORIALS Stem Cells to Regenerate Cardiac Tissue in Heart Failure Felipe Prósper Cardoso a, Jesús Herreros González, b and Eduardo Alegría Ezquerra b a Servicio de Hematología y Área de Terapia Celular,
FEP Medical Policy Manual
 FEP Medical Policy Manual Effective Date: October 15, 2018 Related Policies: 8.01.52 Orthopedic Applications of Stem Cell Therapy (Including Allografts and Bone Substitutes Used With Autologous Bone Marrow)
FEP Medical Policy Manual Effective Date: October 15, 2018 Related Policies: 8.01.52 Orthopedic Applications of Stem Cell Therapy (Including Allografts and Bone Substitutes Used With Autologous Bone Marrow)
Stem Cells for the Heart, Are We There Yet?
 Review Cardiology 2003;100:176 185 DOI: 10.1159/000074811 Received: July 1, 2003 Accepted: September 26, 2003 F. Timmermans J. De Sutter T.C. Gillebert Department of Cardiovascular Diseases, Ghent University,
Review Cardiology 2003;100:176 185 DOI: 10.1159/000074811 Received: July 1, 2003 Accepted: September 26, 2003 F. Timmermans J. De Sutter T.C. Gillebert Department of Cardiovascular Diseases, Ghent University,
Cellular cardiomyoplasty is a cell therapy approach
 In Vitro Preprogramming of Marrow Stromal Cells for Myocardial Regeneration Bindu Bittira, MD, Jin-Qiang Kuang, BS, Abdulaziz Al-Khaldi, MD, Dominique Shum-Tim, MD, and Ray C.-J. Chiu, MD, PhD Division
In Vitro Preprogramming of Marrow Stromal Cells for Myocardial Regeneration Bindu Bittira, MD, Jin-Qiang Kuang, BS, Abdulaziz Al-Khaldi, MD, Dominique Shum-Tim, MD, and Ray C.-J. Chiu, MD, PhD Division
Mesenchymal stem cells from bone marrow and umbilical cord for treating cardiovascular diseases
 Cuban Society of Cardiology Special Article Mesenchymal stem cells from bone marrow and umbilical cord for treating cardiovascular diseases Células madre mesenquimales de médula ósea y de cordón umbilical
Cuban Society of Cardiology Special Article Mesenchymal stem cells from bone marrow and umbilical cord for treating cardiovascular diseases Células madre mesenquimales de médula ósea y de cordón umbilical
Video-Assisted Thoracoscopic Transplantation of Myoblasts Into the Heart
 Video-Assisted Thoracoscopic Transplantation of Myoblasts Into the Heart Richard B. Thompson, MD, Cyrus J. Parsa, MD, Ewout J. van den Bos, MD, Bryce H. Davis, BES, Eric M. Toloza, MD, PhD, Igor Klem,
Video-Assisted Thoracoscopic Transplantation of Myoblasts Into the Heart Richard B. Thompson, MD, Cyrus J. Parsa, MD, Ewout J. van den Bos, MD, Bryce H. Davis, BES, Eric M. Toloza, MD, PhD, Igor Klem,
Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells
 Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells See related Commentary on pages 1355 1356. Kathyjo A. Jackson, 1 Susan M. Majka, 1,2,3 Hongyu Wang, 1 Jennifer Pocius,
Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells See related Commentary on pages 1355 1356. Kathyjo A. Jackson, 1 Susan M. Majka, 1,2,3 Hongyu Wang, 1 Jennifer Pocius,
Myoblast Transplantation. Advanced Angioplasty London, May 18, 2003
 Myoblast Transplantation Advanced Angioplasty London, May 18, 2003 Heart Failure Epidemiology -- High incidence (~ 500,000 per year - in the U.S.) -- High mortality (40% at 1 year in - Class III-IV patients)
Myoblast Transplantation Advanced Angioplasty London, May 18, 2003 Heart Failure Epidemiology -- High incidence (~ 500,000 per year - in the U.S.) -- High mortality (40% at 1 year in - Class III-IV patients)
Cell therapy: enhancing the therapeutic potential of cardiac progenitors for delivery post myocardial infarction. Rita Alonaizan
 Cell therapy: enhancing the therapeutic potential of cardiac progenitors for delivery post myocardial infarction Rita Alonaizan Department of Physiology, Anatomy & Genetics St Catherine s College Supervisor:
Cell therapy: enhancing the therapeutic potential of cardiac progenitors for delivery post myocardial infarction Rita Alonaizan Department of Physiology, Anatomy & Genetics St Catherine s College Supervisor:
Future Perspectives in the Treatment of Heart Failure: From Cell Transplantation to Cardiac Regeneration
 U PDATE Advances in the Management of Heart Failure (IX) Future Perspectives in the Treatment of Heart Failure: From Cell Transplantation to Cardiac Regeneration Felipe Prósper, Jesús Herreros, and Joaquín
U PDATE Advances in the Management of Heart Failure (IX) Future Perspectives in the Treatment of Heart Failure: From Cell Transplantation to Cardiac Regeneration Felipe Prósper, Jesús Herreros, and Joaquín
JACC: CARDIOVASCULAR INTERVENTIONS VOL. 2, NO. 1, PUBLISHED BY ELSEVIER INC. DOI: /j.jcin
 JACC: CARDIOVASCULAR INTERVENTIONS VOL. 2, NO. 1, 2009 2009 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/09/$36.00 PUBLISHED BY ELSEVIER INC. DOI: 10.1016/j.jcin.2008.11.003 CLINICAL
JACC: CARDIOVASCULAR INTERVENTIONS VOL. 2, NO. 1, 2009 2009 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION ISSN 1936-8798/09/$36.00 PUBLISHED BY ELSEVIER INC. DOI: 10.1016/j.jcin.2008.11.003 CLINICAL
Reviews. Neural Stem Cells: An Overview Mesenchymal Stem Cells Myocyte Death, Growth, and Regeneration in Cardiac Hypertrophy and Failure
 Reviews This Review is part of a thematic series on Stem Cells, which includes the following articles: Differentiation of Pluripotent Embryonic Stem Cells Into Cardiomyocytes Derivation and Potential Applications
Reviews This Review is part of a thematic series on Stem Cells, which includes the following articles: Differentiation of Pluripotent Embryonic Stem Cells Into Cardiomyocytes Derivation and Potential Applications
Review. Current perspective of stem cell therapies for cardiac regeneration. Yee Ki Lee 1, Chung-Wah Siu 1,2, Yang Zhang 1 & Hung-Fat Tse 1,2
 Review Current perspective of stem cell therapies for cardiac regeneration Despite recent advances in prevention and treatment of coronary artery disease, it remains the major cause of morbidity and mortality
Review Current perspective of stem cell therapies for cardiac regeneration Despite recent advances in prevention and treatment of coronary artery disease, it remains the major cause of morbidity and mortality
Despite progress during the last several decades in the treatment of
 Evolving Technology Angiogenic pretreatment to enhance myocardial function after cellular cardiomyoplasty with skeletal myoblasts Mauricio A. Retuerto, BS, a James T. Beckmann, BS, a JoAnn Carbray, BS,
Evolving Technology Angiogenic pretreatment to enhance myocardial function after cellular cardiomyoplasty with skeletal myoblasts Mauricio A. Retuerto, BS, a James T. Beckmann, BS, a JoAnn Carbray, BS,
Bone marrow stem cells have myogenic potential and are
 Basic Science Reports Systemic Delivery of Bone Marrow Derived Mesenchymal Stem Cells to the Infarcted Myocardium Feasibility, Cell Migration, and Body Distribution Israel M. Barbash, MD; Pierre Chouraqui,
Basic Science Reports Systemic Delivery of Bone Marrow Derived Mesenchymal Stem Cells to the Infarcted Myocardium Feasibility, Cell Migration, and Body Distribution Israel M. Barbash, MD; Pierre Chouraqui,
The cardiomyocyte cell cycle
 DePauw University Scholarly and Creative Work from DePauw University Biology Faculty publications Biology 2006 The cardiomyocyte cell cycle Pascal J. Lafontant DePauw University, pascallafontant@depauw.edu
DePauw University Scholarly and Creative Work from DePauw University Biology Faculty publications Biology 2006 The cardiomyocyte cell cycle Pascal J. Lafontant DePauw University, pascallafontant@depauw.edu
Local injection of stem cell factor (SCF) improves myocardial homing of systemically delivered c-kit 1 bone marrow-derived stem cells
 Cardiovascular Research (2008) 77, 143 150 doi:10.1093/cvr/cvm027 Local injection of stem cell factor (SCF) improves myocardial homing of systemically delivered c-kit 1 bone marrow-derived stem cells Matthias
Cardiovascular Research (2008) 77, 143 150 doi:10.1093/cvr/cvm027 Local injection of stem cell factor (SCF) improves myocardial homing of systemically delivered c-kit 1 bone marrow-derived stem cells Matthias
Supplemental Information. Myocardial Polyploidization Creates a Barrier. to Heart Regeneration in Zebrafish
 Developmental Cell, Volume 44 Supplemental Information Myocardial Polyploidization Creates a Barrier to Heart Regeneration in Zebrafish Juan Manuel González-Rosa, Michka Sharpe, Dorothy Field, Mark H.
Developmental Cell, Volume 44 Supplemental Information Myocardial Polyploidization Creates a Barrier to Heart Regeneration in Zebrafish Juan Manuel González-Rosa, Michka Sharpe, Dorothy Field, Mark H.
Myocardial infarction
 NEW CARDIAC MARKERS AND CARDIAC REGENERATION Päivi Lakkisto, MD, PhD Specialist in Clinical Chemistry Clinical lecturer University of Helsinki and HUSLAB Minerva Institute for Medical Research Myocardial
NEW CARDIAC MARKERS AND CARDIAC REGENERATION Päivi Lakkisto, MD, PhD Specialist in Clinical Chemistry Clinical lecturer University of Helsinki and HUSLAB Minerva Institute for Medical Research Myocardial
Supplemental Experimental Procedures
 Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Xenotransplant Cardiac Chimera: Immune Tolerance of Adult Stem Cells
 Xenotransplant Cardiac Chimera: Immune Tolerance of Adult Stem Cells Takayuki Saito, MD, PhD, Jin-Qiang Kuang, MD, Bindu Bittira, MD, Abdulaziz Al-Khaldi, MD, and Ray C.-J. Chiu, MD, PhD Division of Cardiac
Xenotransplant Cardiac Chimera: Immune Tolerance of Adult Stem Cells Takayuki Saito, MD, PhD, Jin-Qiang Kuang, MD, Bindu Bittira, MD, Abdulaziz Al-Khaldi, MD, and Ray C.-J. Chiu, MD, PhD Division of Cardiac
Skeletal myoblast transplantation is a promising alternative strategy to
 Single fibers of skeletal muscle as a novel graft for cell transplantation to the heart Ken Suzuki, MD, PhD a* Bari Murtuza, MA, FRCS a* Louise Heslop, PhD, b Jennifer E. Morgan, PhD b Ryszard T. Smolenski,
Single fibers of skeletal muscle as a novel graft for cell transplantation to the heart Ken Suzuki, MD, PhD a* Bari Murtuza, MA, FRCS a* Louise Heslop, PhD, b Jennifer E. Morgan, PhD b Ryszard T. Smolenski,
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10188 Supplementary Figure 1. Embryonic epicardial genes are down-regulated from midgestation stages and barely detectable post-natally. Real time qrt-pcr revealed a significant down-regulation
doi:10.1038/nature10188 Supplementary Figure 1. Embryonic epicardial genes are down-regulated from midgestation stages and barely detectable post-natally. Real time qrt-pcr revealed a significant down-regulation
Comparison of Human Skeletal Myoblasts and Bone Marrow-Derived CD133 Progenitors for the Repair of Infarcted Myocardium
 Journal of the American College of Cardiology Vol. 44, No. 2, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2004.03.083
Journal of the American College of Cardiology Vol. 44, No. 2, 2004 2004 by the American College of Cardiology Foundation ISSN 0735-1097/04/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2004.03.083
Coronary heart disease and heart failure continue to be
 CONTROVERSIES IN CARDIOVASCULAR MEDICINE Is stem cell therapy ready for patients? Stem Cell Therapy for Cardiac Repair Ready for the Next Step Andrew J. Boyle, MBBS, PhD; Steven P. Schulman, MD; Joshua
CONTROVERSIES IN CARDIOVASCULAR MEDICINE Is stem cell therapy ready for patients? Stem Cell Therapy for Cardiac Repair Ready for the Next Step Andrew J. Boyle, MBBS, PhD; Steven P. Schulman, MD; Joshua
Mature myocardial tissue does not possess the ability
 Intracardiac Transplantation of Skeletal Myoblasts Yields Two Populations of Striated Cells In Situ B. Zane Atkins, MD, Cleveland W. Lewis, MD, William E. Kraus, MD, Kelley A. Hutcheson, BS, Donald D.
Intracardiac Transplantation of Skeletal Myoblasts Yields Two Populations of Striated Cells In Situ B. Zane Atkins, MD, Cleveland W. Lewis, MD, William E. Kraus, MD, Kelley A. Hutcheson, BS, Donald D.
Mesenchymal Stem Cells and Cancer: Their Interplay
 Mesenchymal Stem Cells and Cancer: Their Interplay Gang Li, MBBS, DPhil (Oxon) Stem Cell and Regeneration Program School of Biomedical Sciences Li Ka Shing Institute of Health Sciences Department of Orthopaedics
Mesenchymal Stem Cells and Cancer: Their Interplay Gang Li, MBBS, DPhil (Oxon) Stem Cell and Regeneration Program School of Biomedical Sciences Li Ka Shing Institute of Health Sciences Department of Orthopaedics
Targeted Cell Delivery Into Infarcted Rat Hearts by Retrograde Intracoronary Infusion: Distribution, Dynamics, and Influence on Cardiac Function
 Targeted Cell Delivery Into Infarcted Rat Hearts by Retrograde Intracoronary Infusion: Distribution, Dynamics, and Influence on Cardiac Function Ken Suzuki, MD, PhD; Bari Murtuza, MD, PhD; Satsuki Fukushima,
Targeted Cell Delivery Into Infarcted Rat Hearts by Retrograde Intracoronary Infusion: Distribution, Dynamics, and Influence on Cardiac Function Ken Suzuki, MD, PhD; Bari Murtuza, MD, PhD; Satsuki Fukushima,
Anti-ceramide Antibody Prevents the Radiation GI Syndrome in Mice
 Anti-ceramide Antibody Prevents the Radiation GI Syndrome in Mice Jimmy A. Rotolo 1, Branka Stancevic 1, Jianjun Zhang 1, Guoqiang Hua 1, John Fuller 1, Xianglei Yin 1, Adriana Haimovitz-Friedman 2, Kisu
Anti-ceramide Antibody Prevents the Radiation GI Syndrome in Mice Jimmy A. Rotolo 1, Branka Stancevic 1, Jianjun Zhang 1, Guoqiang Hua 1, John Fuller 1, Xianglei Yin 1, Adriana Haimovitz-Friedman 2, Kisu
Cell Therapy Attenuates Deleterious Ventricular Remodeling and Improves Cardiac Performance After Myocardial Infarction
 Cell Therapy Attenuates Deleterious Ventricular Remodeling and Improves Cardiac Performance After Myocardial Infarction Mohit Jain*; Harout DerSimonian, PhD*; Daniel A. Brenner, MA; Soeun Ngoy; Paige Teller,
Cell Therapy Attenuates Deleterious Ventricular Remodeling and Improves Cardiac Performance After Myocardial Infarction Mohit Jain*; Harout DerSimonian, PhD*; Daniel A. Brenner, MA; Soeun Ngoy; Paige Teller,
Effect of sphingosine-1-phosphate and myoblast transplantation on rat acute myocardial infarction
 Effect of sphingosine-1-phosphate and myoblast transplantation on rat acute myocardial infarction H. Yu 1, P.L. Chen 2, Y. Zhao 1, X. Gu 3, W. He 1 and Z. Gong 2 1 Key Laboratory of System Biomedicine
Effect of sphingosine-1-phosphate and myoblast transplantation on rat acute myocardial infarction H. Yu 1, P.L. Chen 2, Y. Zhao 1, X. Gu 3, W. He 1 and Z. Gong 2 1 Key Laboratory of System Biomedicine
Supplementary Figure 1 Chemokine and chemokine receptor expression during muscle regeneration (a) Analysis of CR3CR1 mrna expression by real time-pcr
 Supplementary Figure 1 Chemokine and chemokine receptor expression during muscle regeneration (a) Analysis of CR3CR1 mrna expression by real time-pcr at day 0, 1, 4, 10 and 21 post- muscle injury. (b)
Supplementary Figure 1 Chemokine and chemokine receptor expression during muscle regeneration (a) Analysis of CR3CR1 mrna expression by real time-pcr at day 0, 1, 4, 10 and 21 post- muscle injury. (b)
Peritransplant Ischemic Injury Is Associated With Up-Regulation of Stromal Cell-Derived Factor-1
 Journal of the American College of Cardiology Vol. 46, No. 6, 2005 2005 by the American College of Cardiology Foundation ISSN 0735-1097/05/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2005.04.059
Journal of the American College of Cardiology Vol. 46, No. 6, 2005 2005 by the American College of Cardiology Foundation ISSN 0735-1097/05/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2005.04.059
Cellular cardiomyoplasty (CCM) have been the focus of
 Mesenchymal Progenitor Cells Differentiate into an Endothelial Phenotype, Enhance Vascular Density, and Improve Heart Function in a Rat Cellular Cardiomyoplasty Model Siamak Davani, MD; Aliette Marandin,
Mesenchymal Progenitor Cells Differentiate into an Endothelial Phenotype, Enhance Vascular Density, and Improve Heart Function in a Rat Cellular Cardiomyoplasty Model Siamak Davani, MD; Aliette Marandin,
In vitro functional study of Cardiomyocyte
 In vitro functional study of Cardiomyocyte Gwang Hyeon Eom Department of Pharmacology and Medical Research Center for Gene Regulation Chonnam National University Medical School, Gwangju, South Korea Heart
In vitro functional study of Cardiomyocyte Gwang Hyeon Eom Department of Pharmacology and Medical Research Center for Gene Regulation Chonnam National University Medical School, Gwangju, South Korea Heart
Progenitor Cell Therapy for the Treatment of Damaged Myocardium due to Ischemia. Original Policy Date
 MP 2.02.14 Progenitor Cell Therapy for the Treatment of Damaged Myocardium due to Ischemia Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed with
MP 2.02.14 Progenitor Cell Therapy for the Treatment of Damaged Myocardium due to Ischemia Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed with
Stem cells in the dog heart are self-renewing, clonogenic, and multipotent and regenerate infarcted myocardium, improving cardiac function
 Stem cells in the dog heart are self-renewing, clonogenic, and multipotent and regenerate infarcted myocardium, improving cardiac function Axel Linke*, Patrick Müller*, Daria Nurzynska*, Claudia Casarsa*,
Stem cells in the dog heart are self-renewing, clonogenic, and multipotent and regenerate infarcted myocardium, improving cardiac function Axel Linke*, Patrick Müller*, Daria Nurzynska*, Claudia Casarsa*,
Transplant of autologous bone marrow stem cells in ischemic heart disease
 22 I. Jaafar et al. / Brunei Darussalam Journal of Health Volume 1, 2006 Transplant of autologous bone marrow stem cells in ischemic heart disease Isham Jaafar 1,6, Reiji Hattori¹, Azhari Yakub¹, Jeswant
22 I. Jaafar et al. / Brunei Darussalam Journal of Health Volume 1, 2006 Transplant of autologous bone marrow stem cells in ischemic heart disease Isham Jaafar 1,6, Reiji Hattori¹, Azhari Yakub¹, Jeswant
Quantitative analysis of survival of transplanted smooth muscle cells with real-time polymerase chain reaction
 Cardiothoracic Transplantation Quantitative analysis of survival of transplanted smooth muscle cells with real-time polymerase chain reaction Tamotsu Yasuda, MD, PhD Richard D. Weisel, MD Chris Kiani,
Cardiothoracic Transplantation Quantitative analysis of survival of transplanted smooth muscle cells with real-time polymerase chain reaction Tamotsu Yasuda, MD, PhD Richard D. Weisel, MD Chris Kiani,
Promoting Fracture Healing Through Systemic or Local Administration of Allogeneic Mesenchymal Stem Cells
 Promoting Fracture Healing Through Systemic or Local Administration of Allogeneic Mesenchymal Stem Cells Gang Li Dept. of Orthopaedics and Traumatology School of Biomedical Sciences, The Chinese University
Promoting Fracture Healing Through Systemic or Local Administration of Allogeneic Mesenchymal Stem Cells Gang Li Dept. of Orthopaedics and Traumatology School of Biomedical Sciences, The Chinese University
Protocol. Progenitor Cell Therapy for the Treatment of Damaged Myocardium Due to Ischemia
 (20218) Medical Benefit Effective Date: 01/01/11 Next Review Date: 07/15 Preauthorization No Review Dates: 09/10, 07/11, 07/12, 07/13, 07/14 The following Protocol contains medical necessity criteria that
(20218) Medical Benefit Effective Date: 01/01/11 Next Review Date: 07/15 Preauthorization No Review Dates: 09/10, 07/11, 07/12, 07/13, 07/14 The following Protocol contains medical necessity criteria that
Devices are So Old School: The New World of Myocardial Regeneration
 Devices are So Old School: The New World of Myocardial Regeneration Todd K. Rosengart, M.D. Professor and Chairman DeBakey-Bard Chair of Surgery Michael E. DeBakey Department of Surgery Professor, Texas
Devices are So Old School: The New World of Myocardial Regeneration Todd K. Rosengart, M.D. Professor and Chairman DeBakey-Bard Chair of Surgery Michael E. DeBakey Department of Surgery Professor, Texas
Nature Neuroscience: doi: /nn Supplementary Figure 1. MADM labeling of thalamic clones.
 Supplementary Figure 1 MADM labeling of thalamic clones. (a) Confocal images of an E12 Nestin-CreERT2;Ai9-tdTomato brain treated with TM at E10 and stained for BLBP (green), a radial glial progenitor-specific
Supplementary Figure 1 MADM labeling of thalamic clones. (a) Confocal images of an E12 Nestin-CreERT2;Ai9-tdTomato brain treated with TM at E10 and stained for BLBP (green), a radial glial progenitor-specific
Electromechanical Coupling between Skeletal and Cardiac Muscle: Implications for Infarct Repair
 Electromechanical Coupling between Skeletal and Cardiac Muscle: Implications for Infarct Repair Hans Reinecke,* Glen H. MacDonald, Stephen D. Hauschka, and Charles E. Murry* *Department of Pathology, Department
Electromechanical Coupling between Skeletal and Cardiac Muscle: Implications for Infarct Repair Hans Reinecke,* Glen H. MacDonald, Stephen D. Hauschka, and Charles E. Murry* *Department of Pathology, Department
Expression of cardiomyocytic markers on adipose tissue-derived cells in a murine model of acute myocardial injury
 Cytotherapy (2005) Vol. 7, No. 3, 282/291 Expression of cardiomyocytic markers on adipose tissue-derived cells in a murine model of acute myocardial injury BM Strem 1,2, M Zhu 1, Z Alfonso 1, EJ Daniels
Cytotherapy (2005) Vol. 7, No. 3, 282/291 Expression of cardiomyocytic markers on adipose tissue-derived cells in a murine model of acute myocardial injury BM Strem 1,2, M Zhu 1, Z Alfonso 1, EJ Daniels
DOWNLOAD PDF CARDIAC REMODELING AND CELL DEATH IN HEART FAILURE
 Chapter 1 : The fibrosis-cell death axis in heart failure Remodeling may be defined as changes in the morphology, structure, and function of the heart related to alterations in loading conditions and/or
Chapter 1 : The fibrosis-cell death axis in heart failure Remodeling may be defined as changes in the morphology, structure, and function of the heart related to alterations in loading conditions and/or
1. Cardiomyocytes and nonmyocyte. 2. Extracellular Matrix 3. Vessels שאלה 1. Pathobiology of Heart Failure Molecular and Cellular Mechanism
 Pathobiology of Heart Failure Molecular and Cellular Mechanism Jonathan Leor Neufeld Cardiac Research Institute Tel-Aviv University Sheba Medical Center, Tel-Hashomer שאלה 1 התא הנפוץ ביותר (75%~) בלב
Pathobiology of Heart Failure Molecular and Cellular Mechanism Jonathan Leor Neufeld Cardiac Research Institute Tel-Aviv University Sheba Medical Center, Tel-Hashomer שאלה 1 התא הנפוץ ביותר (75%~) בלב
Dr. Alexander Lyon Senior Lecturer and Consultant Cardiologist Clinical Lead in Cardio-Oncology Royal Brompton Hospital, London UK
 Advanced heart failure - devices, mechanical circulatory support and cardiac transplantation Monday 30 January 2017 Stem cell and gene therapies for heart failure Dr. Alexander Lyon Senior Lecturer and
Advanced heart failure - devices, mechanical circulatory support and cardiac transplantation Monday 30 January 2017 Stem cell and gene therapies for heart failure Dr. Alexander Lyon Senior Lecturer and
Ischemic heart disease accounts for more than 50% of the reported cases of heart
 Cardiopulmonary Support and Physiology Bone marrow cells have a potent anti-ischemic effect against myocardial cell death in humans Chandrashekhar Kubal, MS, MRCS, a Kamlesh Sheth, MCh, FRCS, a Bernardo
Cardiopulmonary Support and Physiology Bone marrow cells have a potent anti-ischemic effect against myocardial cell death in humans Chandrashekhar Kubal, MS, MRCS, a Kamlesh Sheth, MCh, FRCS, a Bernardo
Neural stem cells and the neurobiology of ageing. Chen Siyun 1, Dawe G.S. 2
 ABSTRACT Neural stem cells and the neurobiology of ageing Chen Siyun 1, Dawe G.S. 2 Department of Physics, Faculty of Science, National University of Singapore 10 Kent Ridge Road, Singapore 117546 The
ABSTRACT Neural stem cells and the neurobiology of ageing Chen Siyun 1, Dawe G.S. 2 Department of Physics, Faculty of Science, National University of Singapore 10 Kent Ridge Road, Singapore 117546 The
Cardiac repair with thymosin 4 and cardiac reprogramming factors
 Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Thymosins in Health and Disease Cardiac repair with thymosin 4 and cardiac reprogramming factors Deepak Srivastava,
Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Thymosins in Health and Disease Cardiac repair with thymosin 4 and cardiac reprogramming factors Deepak Srivastava,
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
Probe. Hind III Q,!?R'!! /0!!!!D1"?R'! vector. Homologous recombination
 Supple-Zhang Page 1 Wild-type locus Targeting construct Targeted allele Exon Exon3 Exon Probe P1 P P3 FRT FRT loxp loxp neo vector amh I Homologous recombination neo P1 P P3 FLPe recombination Q,!?R'!!
Supple-Zhang Page 1 Wild-type locus Targeting construct Targeted allele Exon Exon3 Exon Probe P1 P P3 FRT FRT loxp loxp neo vector amh I Homologous recombination neo P1 P P3 FLPe recombination Q,!?R'!!
With the recent advent in stem cell biology, it has been shown
 Cardiopulmonary Support and Physiology Transcoronary implantation of bone marrow stromal cells ameliorates cardiac function after myocardial infarction Takayuki Saito, MD, PhD a,b Jin-Qiang Kuang, MD a
Cardiopulmonary Support and Physiology Transcoronary implantation of bone marrow stromal cells ameliorates cardiac function after myocardial infarction Takayuki Saito, MD, PhD a,b Jin-Qiang Kuang, MD a
W J C. World Journal of Cardiology. Heart regeneration: Past, present and future INTRODUCTION. Abstract PAST EDITORIAL
 W J C World Journal of Cardiology Online Submissions: http://www.wjgnet.com/1949-8462office wjc@wjgnet.com doi:10.4330/wjc.v2.i5.107 World J Cardiol 2010 May 26; 2(5): 107-111 ISSN 1949-8462 (online) 2010
W J C World Journal of Cardiology Online Submissions: http://www.wjgnet.com/1949-8462office wjc@wjgnet.com doi:10.4330/wjc.v2.i5.107 World J Cardiol 2010 May 26; 2(5): 107-111 ISSN 1949-8462 (online) 2010
In vivo and in vitro studies have strengthened the notion that
 bcl-2 overexpression promotes myocyte proliferation Federica Limana*, Konrad Urbanek*, Stefano Chimenti*, Federico Quaini*, Annarosa Leri*, Jan Kajstura*, Bernardo Nadal-Ginard*, Seigo Izumo, and Piero
bcl-2 overexpression promotes myocyte proliferation Federica Limana*, Konrad Urbanek*, Stefano Chimenti*, Federico Quaini*, Annarosa Leri*, Jan Kajstura*, Bernardo Nadal-Ginard*, Seigo Izumo, and Piero
Therapeutic Potential of Human Umbilical Cord Derived Stem Cells in a Rat Myocardial Infarction Model
 Therapeutic Potential of Human Umbilical Cord Derived Stem Cells in a Rat Myocardial Infarction Model Kai Hong Wu, MD, PhD,* Bin Zhou, PhD,* Cun Tao Yu, MD, Bin Cui, MD, Shi Hong Lu, BS, Zhong Chao Han,
Therapeutic Potential of Human Umbilical Cord Derived Stem Cells in a Rat Myocardial Infarction Model Kai Hong Wu, MD, PhD,* Bin Zhou, PhD,* Cun Tao Yu, MD, Bin Cui, MD, Shi Hong Lu, BS, Zhong Chao Han,
Review Article Therapeutic Application of Cardiac Stem Cells and Other Cell Types
 BioMed Research International Volume 2013, Article ID 736815, 6 pages http://dx.doi.org/10.1155/2013/736815 Review Article Therapeutic Application of Cardiac Stem Cells and Other Cell Types Emiko Hayashi
BioMed Research International Volume 2013, Article ID 736815, 6 pages http://dx.doi.org/10.1155/2013/736815 Review Article Therapeutic Application of Cardiac Stem Cells and Other Cell Types Emiko Hayashi
Engraftment Is Optimal When Myoblasts Are Transplanted Early: The Role of Hepatocyte Growth Factor
 Engraftment Is Optimal When Myoblasts Are Transplanted Early: The Role of Hepatocyte Growth Factor Stacy B. O Blenes, MD, Audrey W. Li, PhD, Robert Chen, MD, Rakesh C. Arora, MD, PhD, and Magda Horackova,
Engraftment Is Optimal When Myoblasts Are Transplanted Early: The Role of Hepatocyte Growth Factor Stacy B. O Blenes, MD, Audrey W. Li, PhD, Robert Chen, MD, Rakesh C. Arora, MD, PhD, and Magda Horackova,
Cardiomyocytes can be formed, at least ex vivo, from diverse
 Cardiac progenitor cells from adult myocardium: Homing, differentiation, and fusion after infarction Hidemasa Oh ab, Steven B. Bradfute cd, Teresa D. Gallardo e, Teruya Nakamura ab, Vinciane Gaussin f,
Cardiac progenitor cells from adult myocardium: Homing, differentiation, and fusion after infarction Hidemasa Oh ab, Steven B. Bradfute cd, Teresa D. Gallardo e, Teruya Nakamura ab, Vinciane Gaussin f,
Supporting Information. Calculation of the relative contributions of myocyte proliferation, stem cell. Supporting Information Fig 1 (page 9)
 Supporting Information Table of contents Calculation of the relative contributions of myocyte proliferation, stem cell differentiation and cardioprotection (page 2) Supporting Information Fig 1 (page 9)
Supporting Information Table of contents Calculation of the relative contributions of myocyte proliferation, stem cell differentiation and cardioprotection (page 2) Supporting Information Fig 1 (page 9)
Myocardial Regeneration and Stem Cell Repair
 Myocardial Regeneration and Stem Cell Repair Annarosa Leri, MD, Jan Kajstura, MD, Piero Anversa, MD, and William H. Frishman, MD Abstract: Recent evidence would suggest that the heart is not a terminally
Myocardial Regeneration and Stem Cell Repair Annarosa Leri, MD, Jan Kajstura, MD, Piero Anversa, MD, and William H. Frishman, MD Abstract: Recent evidence would suggest that the heart is not a terminally
Journal Club WS 2012/13 Stefanie Nickl
 Journal Club WS 2012/13 Stefanie Nickl Background Mesenchymal Stem Cells First isolation from bone marrow 30 ys ago Isolation from: spleen, heart, skeletal muscle, synovium, amniotic fluid, dental pulp,
Journal Club WS 2012/13 Stefanie Nickl Background Mesenchymal Stem Cells First isolation from bone marrow 30 ys ago Isolation from: spleen, heart, skeletal muscle, synovium, amniotic fluid, dental pulp,
SUPPLEMENTARY INFORMATION
 Suppl. Fig. 1 in vivo expression of ISL1 in the human fetal heart. a, Hematoxylin eosin staining showing structures of left atrium and left atrium appendage (*) of a human fetal heart at 11 weeks of gestation.
Suppl. Fig. 1 in vivo expression of ISL1 in the human fetal heart. a, Hematoxylin eosin staining showing structures of left atrium and left atrium appendage (*) of a human fetal heart at 11 weeks of gestation.
Review Article Present and Future Perspectives on Cell Sheet-Based Myocardial Regeneration Therapy
 BioMed Volume 2013, Article ID 583912, 6 pages http://dx.doi.org/10.1155/2013/583912 Review Article Present and Future Perspectives on Cell Sheet-Based Myocardial Regeneration Therapy Yoshiki Sawa and
BioMed Volume 2013, Article ID 583912, 6 pages http://dx.doi.org/10.1155/2013/583912 Review Article Present and Future Perspectives on Cell Sheet-Based Myocardial Regeneration Therapy Yoshiki Sawa and
How many skeletal muscles are present in our body? Muscles are excitable & contractile, extensible and elastic to some extent.
 Muscles How many skeletal muscles are present in our body? -646 muscles The functions of the muscles are: Movement Maintenance of posture Generation of heat Stabilization of joints : amount of muscle surrounding
Muscles How many skeletal muscles are present in our body? -646 muscles The functions of the muscles are: Movement Maintenance of posture Generation of heat Stabilization of joints : amount of muscle surrounding
Mass Histology Service
 Mass Histology Service A complete anatomical pathology laboratory www.masshistology.com Telephone: (877) 286-6004 Report on Pathology A Time Course Study of the Local Effects of Intramuscular XXXXXXX Injection
Mass Histology Service A complete anatomical pathology laboratory www.masshistology.com Telephone: (877) 286-6004 Report on Pathology A Time Course Study of the Local Effects of Intramuscular XXXXXXX Injection
Cardiomyocyte proliferation and progenitor cell recruitment underlie therapeutic regeneration after myocardial infarction in the adult mouse heart
 OPEN ACCESS Cardiomyocyte proliferation and progenitor cell recruitment underlie therapeutic regeneration after myocardial infarction in the adult mouse heart Konstantinos Malliaras, Yiqiang Zhang, Jeffrey
OPEN ACCESS Cardiomyocyte proliferation and progenitor cell recruitment underlie therapeutic regeneration after myocardial infarction in the adult mouse heart Konstantinos Malliaras, Yiqiang Zhang, Jeffrey
Muscles, muscle fibres and myofibrils
 Muscles, muscle fibres and myofibrils Properties of Muscle Fiber Types Fast fibers Slow fibers Characteristic IIb IIx IIa Type I V max (speed of shortening) Highest Intermediate Low Resistance to fatigue
Muscles, muscle fibres and myofibrils Properties of Muscle Fiber Types Fast fibers Slow fibers Characteristic IIb IIx IIa Type I V max (speed of shortening) Highest Intermediate Low Resistance to fatigue
