FDA & Transvaginal Mesh: What Happened? What s Next?
|
|
|
- Rosalind Henry
- 5 years ago
- Views:
Transcription
1 FDA & Transvaginal Mesh: What Happened? What s Next? Matthew D. Barber, MD MHS Professor & Vice Chair for Clinical Research Obstetrics Gynecology & Women s Health Institute
2 Disclosures I receive no grants, honoraria or other funding from industry.
3 Objectives To briefly discuss the US FDA regulatory process for medical device approval To review the FDA actions on Graft/Mesh for pelvic organ prolapse and stress urinary incontinence To discuss the impact on current clinical practice with specific emphasis on: Informed Consent Surgeon Credentialing
4 Presentation Title l August 22, 2012 l 4
5 Mesh Timeline First sling kit Protagen (Boston Sci) TVT (Ethicon) First marketed POP kit - IVS Tunneler First POP Mesh - Gynemesh Apogee/Perigee (AMS) introduced Prolift (Ethicon) introduced Non-trocar kits (Elevate, Uphold) Presentation Title l August 22, 2012 l 5
6 POP Repair Surgeries - US Market 2010* 300,000 women *industry source
7 POP Mesh Sales 2010* Total 121,260 *US Markets for Soft Tissue Repair 2010, Millennium Research Group, Inc. All rights reserved. Reproduction, distribution, transmission or publication is prohibited. Reprinted with permission.
8 HOW ARE MEDICAL DEVICES CLEARED IN THE U.S.? Presentation Title l August 22, 2012 l 8
9 Phases of Drug Development
10 FDA Medical Device Regulation Class Regulation Examples Approval Process I General Controls Bandages, Gloves, Surgical instruments II III General and Special Controls Premarket Approval (PMA) Catheters, wheel chairs, infusion pumps, L/S trocars Mesh? Cardiac pacemakers, Interstim, Transurethral bulking agents Labeling, Good manufacturing practices 510(k) PMA study
11 FDA 510 (k) Does not require evidence of clinical safety or efficacy Unlike medications, no phase I, II, or III studies required FDA clearance for many 510 devices (k) approval does requires not evidence necessarily of substantial equivalence to an already approved device indicate safety or efficacy! Same technological characteristics Same intended use Predicate Devices on the market prior to May 28, 1976 or previously cleared Example: Surgical Mesh for Hernia
12 Littman & Culligan, 2009
13 Class III Devices that require Premarket (PMA) studies Urethral Bulking Agents (Coaptite, Durasphere, etc) Tubal ligation devices (Filshie clip, Essure, etc) Without Grandfather Clause there is no doubt Transvaginal Mesh would require Pre-market Studies Endometrial ablation devices Sacral Nerve Stimulation (Interstim)
14 Transvaginal Mesh: Timeline Perigee (AMS) Prolift (Ethicon) Elevate (AMS) UpHold (Bost Sci) Introduced in US First Study Feb 2012 May 2012 First RCT ??
15 Cochrane Review Ogah et al, 2009 Mid-urethral Slings Over 500 articles, 40+ RCTs Similar efficacy to traditional slings but with shorter OR time and less voiding dysfunction and de novo urgency. Similar efficacy to Burch/MMK but with fewer postop complications, less voiding dysfunction, shorter OR time, shorter hospital stay but more bladder perforations Similar efficacy for TVT and TOT but different safety profiles 10+ year data suggest efficacy is durable Definitive Standard of Care
16 FDA timeline 2006 AE signals, safety problems FDA initial review of MAUDE, received over 1,000 reports from nine surgical mesh manufacturers 2008 FDA issues Public Health Notice regarding adverse events associated with surgical mesh when used for repair for SUI and POP
17 FDA 2008 PHN Alert about complications associated with transvaginal placement of surgical mesh to treat Pelvic Organ Prolapse (POP) and Stress Urinary Incontinence (SUI) Although rare, these complications can have serious consequences Recommendations- specialized training, watch for AEs, provide written copy of the patient labeling, inform patients about the potential for serious complications, mesh is permanent, watch for complications Reporting Adverse Events to FDA- requires hospitals and other user facilities to report deaths and serious injuries, report directly to MedWatch
18 Serious complications associated with surgical mesh for transvaginal repair of POP are not rare. Transvaginally placed mesh in POP repair does not conclusively improve clinical outcomes over traditional non-mesh repair.
19 Conclusions from FDA Literature Review Vaginal repair with mesh is main concern serious AEs are not rare effectiveness not superior to traditional repair (possible exception for anterior repair) little known about long term implications safety and effectiveness in question Sacrocolpopexy less concerning lower rates of mesh complications excellent anatomic outcomes low re-surgery rates for recurrent prolapse safety and effectiveness demonstrated in literature
20 Top 10 Adverse Events for POP Reports Rank Adverse Events # of MDRs Percentile Rate 1 Erosion % 2 Pain % 3 Infection % 4 Bleeding % 5 Dyspareunia % 6 Organ Perforation % 7 Urinary Problems % 8 Neuro-muscular problems % 9 Vaginal scarring (41)/ Shrinkage (2) % 10 Recurrence, Prolapse % Total number of adverse events is larger than total number of MDR reports because the majority reported more than one adverse event
21 Transvaginal Mesh for Prolapse 70% decline in sales since July 2011 Over 400 lawsuits nationwide against manufacturers January 2012 FDA issues 522 orders requiring post-marketing surveillance studies for all manufacturers of transvaginal mesh for POP Several companies withdrawing from the market Others investing multi-millions of dollars in research to meet FDA requirements
22
23 The FDA waited a decade to ask mesh makers for safety studies.
24 Response to Updated PHN FDA Convened the Obstetrics-Gynecology Devices Panel of the Medical Device Advisory Committee, on September 8-9, Institutes of Medicine: Made recommendations to the FDA on the 510k process for approval of class II medical devices. Would require an Act of Congress to change device regulation U.S. Congress debating a law that would keep system essentially intact and ratify an agreement between FDA and industry to get devices to market faster.
25 UPDATE 01/04/2012: Based on assessment of Medical Device Reports (adverse event reports) submitted to the FDA, evaluation of the published literature, and the September 2011 Obstetrics-Gynecology Devices Panel meeting, the FDA is considering the recommendation that urogynecologic surgical mesh used for transvaginal repair of pelvic organ prolapse (POP) be reclassified from Class II to Class III.
26 Mandating postmarket surveillance studies ( 522 studies ) by manufacturers of urogynecologic surgical mesh devices. On January 03, 2012, the FDA issued: 88 post-market study orders to 33 manufacturers of urogynecologic surgical mesh for POP; 11 postmarket study orders to seven manufacturers of single-incision mini-slings for SUI.
27 What the FDA has not done: Removed any products from the U.S. market Changed the regulatory pathway for: Midurethral slings Mesh/Devices for Sacrocolpopexy
28 Surgeons performing transvaginal placement of mesh for POP should undergo training specific to each device and have experience with reconstructive surgical procedures and a thorough understanding of pelvic anatomy. Transvaginal placement of mesh for POP should be reserved for high-risk individuals in whom the benefits of mesh placement may justify the risk, such as individuals with recurrent prolapse (particularly of the anterior compartment) or with medical comorbidities that precluded more invasive and lengthier open and endoscopic procedures.
29 Presentation Title l August 22, 2012 l 29
30 Presentation Title l August 22, 2012 l 30
31 FDA Recommended Questions for Patients to Ask Surgeons Are you planning to use mesh in my surgery? Why do you think I am a good candidate for mesh? Why are you choosing surgical mesh for my repair? What are the alternatives to transvaginal surgical mesh repair for POP, including non-surgical options? What are the pros and cons of surgical mesh in my particular case? How likely is it my repair could be successfully performed without mesh? Will my partner be able to feel the surgical mesh during intercourse? What if the surgical mesh erodes?
32 FDA Recommended Questions for Patients to Ask Surgeons If you plan to use mesh, how often have you implanted this particular product? What results have your other patients had with this product? What can I expect to feel after surgery and for how long? Which specific side effects should I report to you after surgery? What if the mesh surgery doesn t correct my problem?
33 FDA Recommended Questions for Patients to Ask Surgeons If I develop a complication, will you treat it or will I be referred to a specialist experienced with surgical mesh complications? If I have a complication related to the surgical mesh, how likely is it that the surgical mesh could be removed and what could be the consequences? If you plan to use surgical mesh, is there patient information that comes with the product Informed Consent Checklist
34 Presentation Title l August 22, 2012 l 34 Female Pelvic Med Reconstru Surg 2012; 18:
35 TV Mesh Credentialing Guidelines Surgeons not yet using Mesh for POP Document General Knowledge of Female Pelvic Reconstructive Surgery Fellowship or documentation of appropriate CME Document Specific Knowledge of the Procedure Training in Fellowship or Proctored Cases Experience in treating Pelvic Floor Disorders Fellowship or at least 50% of surgical practice is PFD including at least 30 POP cases/year Current privileges in non-mesh repairs Annual Internal Audit
36 TV Mesh Credentialing Guidelines Surgeons currently using Mesh for POP Continuing Medical Education in female pelvic reconstructive surgery documented annually A minimum of 30 surgical cases annually for pelvic organ prolapse (any route, with or without mesh) Document and maintain privileges in non-mesh vaginal repair of POP Annual Internal Audit Prior to adoption of new technology or device: Demonstrate specific knowledge of device Proctored cases
37 What s Next? All current products stay on market (unless company decides to pull them) Industry to perform 522 postmarket surveillance studies (Start 2013; 3 year follow-up) AUGS in collaboration w/ industry, FDA and others is developing a national PFD registry 5 companies will be performing 522 studies in registry AUGS plans credentialing guidelines for sacrocolpopexy and fundamentals of pelvic reconstructive surgery educational curriculum FDA continues to monitor and will make decision about changing classification of TVM from Class II to Class III
38 Ultimate goals To promote safe and effective are for women with PFD To encourage innovation while maintaining patient safety To maintain a broad range of treatment options to meet the varying needs of our patients
39
US FDA/CDRH: Public Health Notification: Serious Complications Associated with Transvaginal Place... FDA Home Page CDRH Home Page Search A-Z Index
 US FDA/CDRH: Public Health Notification: Serious Complications Associated with Transvaginal Place... http://www.fda.gov/cdrh/safety/102008-surgicalmesh.html Page 1 of 3 FDA Home Page CDRH Home Page Search
US FDA/CDRH: Public Health Notification: Serious Complications Associated with Transvaginal Place... http://www.fda.gov/cdrh/safety/102008-surgicalmesh.html Page 1 of 3 FDA Home Page CDRH Home Page Search
A PATIENT GUIDE TO Understanding Stress Urinary Incontinence
 A PATIENT GUIDE TO Understanding Stress Urinary Incontinence Q: What is SUI? A: Stress urinary incontinence is defined as the involuntary leakage of urine. The problem afflicts approximately 18 million
A PATIENT GUIDE TO Understanding Stress Urinary Incontinence Q: What is SUI? A: Stress urinary incontinence is defined as the involuntary leakage of urine. The problem afflicts approximately 18 million
Avoiding Mesh Disasters: Tips and Tricks for Success and Handling Complications
 Avoiding Mesh Disasters: Tips and Tricks for Success and Handling Complications Karyn S. Eilber, M.D. Cedars-Sinai FPMRS Associate Professor, Cedars-Sinai Dept of Surgery Associate Director, Urology Residency
Avoiding Mesh Disasters: Tips and Tricks for Success and Handling Complications Karyn S. Eilber, M.D. Cedars-Sinai FPMRS Associate Professor, Cedars-Sinai Dept of Surgery Associate Director, Urology Residency
Interventional procedures guidance Published: 12 October 2016 nice.org.uk/guidance/ipg566
 Single-incision short sling mesh insertion for stress urinary incontinence in women Interventional procedures guidance Published: 12 October 2016 nice.org.uk/guidance/ipg566 Your responsibility This guidance
Single-incision short sling mesh insertion for stress urinary incontinence in women Interventional procedures guidance Published: 12 October 2016 nice.org.uk/guidance/ipg566 Your responsibility This guidance
FDA and Mesh Complications in Vaginal Surgery
 FDA and Mesh Complications in Vaginal Surgery Response to FDA Safety Communication dated July 13, 2011 To Our Patients and Women of the Community: As many of you are aware, on July 13, 2011, the FDA released
FDA and Mesh Complications in Vaginal Surgery Response to FDA Safety Communication dated July 13, 2011 To Our Patients and Women of the Community: As many of you are aware, on July 13, 2011, the FDA released
Bard: Continence Therapy. Stress Urinary Incontinence. Regaining Control. Restoring Your Lifestyle.
 Bard: Continence Therapy Stress Urinary Incontinence Regaining Control. Restoring Your Lifestyle. Stress Urinary Incontinence Urinary incontinence is a common problem and one that can be resolved by working
Bard: Continence Therapy Stress Urinary Incontinence Regaining Control. Restoring Your Lifestyle. Stress Urinary Incontinence Urinary incontinence is a common problem and one that can be resolved by working
12/1/13. What are Pelvic Floor Disorders? What is the Pelvic Floor? Facts. Prevalence of Urinary InconOnence. What s New in Pelvic Floor Disorders?
 What are Pelvic Floor Disorders? Urinary Control Problems - InconOnence or leakage of urine Prolapse of pelvic organs - Vagina, bladder, rectum What s New in Pelvic Floor Disorders? Kimberly Kenton MD,
What are Pelvic Floor Disorders? Urinary Control Problems - InconOnence or leakage of urine Prolapse of pelvic organs - Vagina, bladder, rectum What s New in Pelvic Floor Disorders? Kimberly Kenton MD,
Index. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Abdominal mesh background of, 84 85 Age as factor in PFDs, 8 Anal plugs in FI management in women, 107 Anterior compartment native tissue
Index Note: Page numbers of article titles are in boldface type. A Abdominal mesh background of, 84 85 Age as factor in PFDs, 8 Anal plugs in FI management in women, 107 Anterior compartment native tissue
Desara TV and Desara Blue TV
 Desara TV and Desara Blue TV Sling for Female Stress Urinary Incontinence Instructions For Use D I Prescription Use only Do not reuse Sterilized using ethylene oxide Available Electronically M Manufactured
Desara TV and Desara Blue TV Sling for Female Stress Urinary Incontinence Instructions For Use D I Prescription Use only Do not reuse Sterilized using ethylene oxide Available Electronically M Manufactured
Operative Approach to Stress Incontinence. Goals of presentation. Preoperative evaluation: Urodynamic Testing? Michelle Y. Morrill, M.D.
 Operative Approach to Stress Incontinence Goals of presentation Michelle Y. Morrill, M.D. Director of Urogynecology The Permanente Medical Group Kaiser, San Francisco Review preoperative care & evaluation
Operative Approach to Stress Incontinence Goals of presentation Michelle Y. Morrill, M.D. Director of Urogynecology The Permanente Medical Group Kaiser, San Francisco Review preoperative care & evaluation
Adverse Event Reports Relating to Surgical Mesh Implants
 Adverse Event Reports Relating to Surgical Mesh Implants Summary of reports received by August 2017 Page 1 of 26 Document History Version Number Revision Date Summary of Changes 0 Dec-2013 Reports received
Adverse Event Reports Relating to Surgical Mesh Implants Summary of reports received by August 2017 Page 1 of 26 Document History Version Number Revision Date Summary of Changes 0 Dec-2013 Reports received
Karanvir Virk M.D. Minimally Invasive & Pelvic Reconstructive Surgery 01/28/2015
 Karanvir Virk M.D. Minimally Invasive & Pelvic Reconstructive Surgery 01/28/2015 Disclosures I have none Objectives Identify the basic Anatomy and causes of Pelvic Organ Prolapse Examine office diagnosis
Karanvir Virk M.D. Minimally Invasive & Pelvic Reconstructive Surgery 01/28/2015 Disclosures I have none Objectives Identify the basic Anatomy and causes of Pelvic Organ Prolapse Examine office diagnosis
Information from the MAUDE Database
 Information from the MAUDE Database Device mesh, surgical, polymeric Regulation Description Surgical mesh. Product Code FTL Regulation Number 878.3300 Device Class 2 Manufacturer ETHICON Medical Device
Information from the MAUDE Database Device mesh, surgical, polymeric Regulation Description Surgical mesh. Product Code FTL Regulation Number 878.3300 Device Class 2 Manufacturer ETHICON Medical Device
Introduction. Regarding the Section of the UPDATE Entitled Purpose
 Time to Rethink: an Evidence-Based Response from Pelvic Surgeons to the FDA Safety Communication: UPDATE on Serious Complications Associated with Transvaginal Placement of Surgical Mesh for Pelvic Organ
Time to Rethink: an Evidence-Based Response from Pelvic Surgeons to the FDA Safety Communication: UPDATE on Serious Complications Associated with Transvaginal Placement of Surgical Mesh for Pelvic Organ
This information is intended as an overview only
 This information is intended as an overview only Please refer to the INSTRUCTIONS FOR USE included with this device for indications, contraindications, warnings, precautions and other important information
This information is intended as an overview only Please refer to the INSTRUCTIONS FOR USE included with this device for indications, contraindications, warnings, precautions and other important information
AGENDA. 8:00 AM 8:30 AM Pelvic Anatomy of the Lower Urinary Tract and the Anatomy and Physiology of Continence/Incontinence Mickey M.
 Thursday, June 12, 2014 Juniper Ballroom 1: Exhibits AGENDA 6:30 AM 8:00 AM Breakfast and Exhibits EVALUATION AND MANAGEMENT OF LOWER URINARY TRACT SYMPTOMS 8:00 AM 8:30 AM Pelvic Anatomy of the Lower
Thursday, June 12, 2014 Juniper Ballroom 1: Exhibits AGENDA 6:30 AM 8:00 AM Breakfast and Exhibits EVALUATION AND MANAGEMENT OF LOWER URINARY TRACT SYMPTOMS 8:00 AM 8:30 AM Pelvic Anatomy of the Lower
Adverse Event Reports Relating to Surgical Mesh Implants. Summary of reports received by Medsafe
 Adverse Event Reports Relating to Surgical Mesh Implants Summary of reports received by Medsafe December 2016 Page 2 of 25 Document History Version Number Revision Date Summary of Changes 0 Dec-2013 Reports
Adverse Event Reports Relating to Surgical Mesh Implants Summary of reports received by Medsafe December 2016 Page 2 of 25 Document History Version Number Revision Date Summary of Changes 0 Dec-2013 Reports
Labeling for Permanent Hysteroscopically-Placed Tubal Implants Intended for Sterilization
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 Labeling for Permanent Hysteroscopically-Placed Tubal Implants Intended for Sterilization Draft Guidance for Industry and Food and Drug
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 Labeling for Permanent Hysteroscopically-Placed Tubal Implants Intended for Sterilization Draft Guidance for Industry and Food and Drug
Interventional procedures guidance Published: 28 June 2017 nice.org.uk/guidance/ipg583
 Sacrocolpopexy using mesh to repair vaginal vault prolapse Interventional procedures guidance Published: 28 June 2017 nice.org.uk/guidance/ipg583 Your responsibility This guidance represents the view of
Sacrocolpopexy using mesh to repair vaginal vault prolapse Interventional procedures guidance Published: 28 June 2017 nice.org.uk/guidance/ipg583 Your responsibility This guidance represents the view of
q7:480499_P0 6/5/09 10:23 AM Page 1 WHAT YOU SHOULD KNOW ABOUT YOUR DIAGNOSIS OF STRESS URINARY INCONTINENCE
 493495.q7:480499_P0 6/5/09 10:23 AM Page 1 WHAT YOU SHOULD KNOW ABOUT YOUR DIAGNOSIS OF STRESS URINARY INCONTINENCE 493495.q7:480499_P0 6/5/09 10:23 AM Page 2 What is Stress Urinary Incontinence? Urinary
493495.q7:480499_P0 6/5/09 10:23 AM Page 1 WHAT YOU SHOULD KNOW ABOUT YOUR DIAGNOSIS OF STRESS URINARY INCONTINENCE 493495.q7:480499_P0 6/5/09 10:23 AM Page 2 What is Stress Urinary Incontinence? Urinary
Stress Incontinence. Susannah Elvy Urogynaecology CNS
 Stress Incontinence Susannah Elvy Urogynaecology CNS Definitions Prevalence Assessment Investigation Treatment Surgery Men International Continence Society define as the complaint of any involuntary leakage
Stress Incontinence Susannah Elvy Urogynaecology CNS Definitions Prevalence Assessment Investigation Treatment Surgery Men International Continence Society define as the complaint of any involuntary leakage
Facing Gynecologic Surgery?
 Facing Gynecologic Surgery? Domenico Vitobello, MD Domenico Vitobello is the medical director of the Gynecologic Unit at the Humanitas Clinical and Research Center since 2009. He has developed a comprehensive
Facing Gynecologic Surgery? Domenico Vitobello, MD Domenico Vitobello is the medical director of the Gynecologic Unit at the Humanitas Clinical and Research Center since 2009. He has developed a comprehensive
Adverse Event Reports Relating to Surgical Mesh Implants. Summary of reports received by Medsafe
 Adverse Event Reports Relating to Surgical Mesh Implants Summary of reports received by Medsafe September 2016 Page 2 of 25 Document History Version Number Revision Date Summary of Changes 0 Dec-2013 Reports
Adverse Event Reports Relating to Surgical Mesh Implants Summary of reports received by Medsafe September 2016 Page 2 of 25 Document History Version Number Revision Date Summary of Changes 0 Dec-2013 Reports
URINARY INCONTINENCE
 Center for Continence Care and Pelvic Medicine What is urinary incontinence? URINARY INCONTINENCE Urinary incontinence is the uncontrollable loss of urine. The amount of urine leaked can vary from only
Center for Continence Care and Pelvic Medicine What is urinary incontinence? URINARY INCONTINENCE Urinary incontinence is the uncontrollable loss of urine. The amount of urine leaked can vary from only
2012/13 NHS STANDARD CONTRACT FOR ACUTE, AMBULANCE, COMMUNITY AND MENTAL HEALTH AND LEARNING DISABILITY SERVICES (MULTILATERAL)
 E10d 2012/13 NHS STANDARD CONTRACT FOR ACUTE, AMBULANCE, COMMUNITY AND MENTAL HEALTH AND LEARNING DISABILITY SERVICES (MULTILATERAL) SECTION B PART 1 - SERVICE SPECIFICATIONS Service Specification No.
E10d 2012/13 NHS STANDARD CONTRACT FOR ACUTE, AMBULANCE, COMMUNITY AND MENTAL HEALTH AND LEARNING DISABILITY SERVICES (MULTILATERAL) SECTION B PART 1 - SERVICE SPECIFICATIONS Service Specification No.
What you should know about your diagnosis of incontinence
 What you should know about your diagnosis of incontinence What is Stress Urinary Incontinence? WHAT IS NORMAL URINARY FUNCTION? Urine is a normal waste product of the body that is manufactured by the kidneys
What you should know about your diagnosis of incontinence What is Stress Urinary Incontinence? WHAT IS NORMAL URINARY FUNCTION? Urine is a normal waste product of the body that is manufactured by the kidneys
What are we talking about? Symptoms. Prolapse Risk Factors. Vaginal bulge 1 Splinting. ?? Pelvic pressure Back pain 1 Urinary complaints 2
 Options for Vaginal Prolapse What are we talking about? Michelle Y. Morrill, M.D. Director of Urogynecology The Permanente Medical Group Kaiser, San Francisco Assistant Professor, Volunteer Faculty Department
Options for Vaginal Prolapse What are we talking about? Michelle Y. Morrill, M.D. Director of Urogynecology The Permanente Medical Group Kaiser, San Francisco Assistant Professor, Volunteer Faculty Department
Pelvic Prolapse. A Patient Guide to Pelvic Floor Reconstruction
 Pelvic Prolapse A Patient Guide to Pelvic Floor Reconstruction Pelvic Prolapse When an organ becomes displaced, or slips down in the body, it is referred to as a prolapse. Your physician has diagnosed
Pelvic Prolapse A Patient Guide to Pelvic Floor Reconstruction Pelvic Prolapse When an organ becomes displaced, or slips down in the body, it is referred to as a prolapse. Your physician has diagnosed
ACC Treatment Injury Claims
 ACC Treatment Injury Claims Surgical Mesh-Related Claim Data From 1 July 25 to 3 June 217 (12 fiscal years) 18/1/217 1 P age Contents Contents... 2 Abbreviations... 2 Colour references... 3 Terminology...
ACC Treatment Injury Claims Surgical Mesh-Related Claim Data From 1 July 25 to 3 June 217 (12 fiscal years) 18/1/217 1 P age Contents Contents... 2 Abbreviations... 2 Colour references... 3 Terminology...
1) What conditions is vaginal mesh used to commonly treat? Vaginal mesh is used to treat two different health issues in women:
 Vaginal Mesh Frequently Asked Questions 1) What conditions is vaginal mesh used to commonly treat? Vaginal mesh is used to treat two different health issues in women: a) stress urinary incontinence (SUI)
Vaginal Mesh Frequently Asked Questions 1) What conditions is vaginal mesh used to commonly treat? Vaginal mesh is used to treat two different health issues in women: a) stress urinary incontinence (SUI)
Sep \8958 Appell Dmochowski.ppt LMF 1
 Surgical Outcomes (How did we get ourselves into this mess?) Roger R. Dmochowski, MD, FACS Department of Urologic Surgery Vanderbilt University School of Medicine Nashville, Tennessee Considerations Evaluation
Surgical Outcomes (How did we get ourselves into this mess?) Roger R. Dmochowski, MD, FACS Department of Urologic Surgery Vanderbilt University School of Medicine Nashville, Tennessee Considerations Evaluation
Laparoscopic Sacrocolpopexy vs Robotic Sacrocolpopexy
 Laparoscopic Sacrocolpopexy vs Robotic Sacrocolpopexy Magnus Murphy FRCSC Clinical Assistant Professor Department of Obstetrics and Gynecology University of Calgary Division of Urogynecology www.pelvicfloor.com
Laparoscopic Sacrocolpopexy vs Robotic Sacrocolpopexy Magnus Murphy FRCSC Clinical Assistant Professor Department of Obstetrics and Gynecology University of Calgary Division of Urogynecology www.pelvicfloor.com
Surgical repair of vaginal wall prolapse using mesh
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Surgical repair of vaginal wall prolapse using mesh Vaginal wall prolapse happens when the normal support
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Surgical repair of vaginal wall prolapse using mesh Vaginal wall prolapse happens when the normal support
Scottish Clinical Coding Standards
 Scottish Clinical Coding Standards Number 16 October 2017 Scottish Clinical Coding Standards ICD-10 Sepsis Sepsis is a serious condition which must always be coded when documented in the medical record.
Scottish Clinical Coding Standards Number 16 October 2017 Scottish Clinical Coding Standards ICD-10 Sepsis Sepsis is a serious condition which must always be coded when documented in the medical record.
Considering Surgery for Vaginal or Uterine Prolapse? Learn why da Vinci Surgery may be your best treatment option.
 Considering Surgery for Vaginal or Uterine Prolapse? Learn why da Vinci Surgery may be your best treatment option. The Condition(s): Vaginal Prolapse, Uterine Prolapse Vaginal prolapse occurs when the
Considering Surgery for Vaginal or Uterine Prolapse? Learn why da Vinci Surgery may be your best treatment option. The Condition(s): Vaginal Prolapse, Uterine Prolapse Vaginal prolapse occurs when the
Polypropylene vaginal mesh implants for vaginal prolapse
 Polypropylene vaginal mesh implants for vaginal prolapse This statement has been developed and reviewed by the Women s Health Committee and approved by the RANZCOG Board and Council. A list of Women s
Polypropylene vaginal mesh implants for vaginal prolapse This statement has been developed and reviewed by the Women s Health Committee and approved by the RANZCOG Board and Council. A list of Women s
Stress Urinary Incontinence in Women. What YOU can do about it...
 Stress Urinary Incontinence in Women What YOU can do about it... www.gynecare.com Stress Urinary Incontinence in Women: It's Common. It's Treatable. Would it surprise you... To learn that more than 13
Stress Urinary Incontinence in Women What YOU can do about it... www.gynecare.com Stress Urinary Incontinence in Women: It's Common. It's Treatable. Would it surprise you... To learn that more than 13
Tension Free vaginal tape. Mrs Ami Shukla, Consultant Urogynaecologist Northampton General Hospital Northampton NN1 5BD
 Tension Free vaginal tape Mrs Ami Shukla, Consultant Urogynaecologist Northampton General Hospital Northampton NN1 5BD What is a TVT procedure? A TVT (Tension Free Vaginal Tape) procedure is an operation
Tension Free vaginal tape Mrs Ami Shukla, Consultant Urogynaecologist Northampton General Hospital Northampton NN1 5BD What is a TVT procedure? A TVT (Tension Free Vaginal Tape) procedure is an operation
Current status in pelvic organ prolapse surgery: an evidence based review
 Current status in pelvic organ prolapse surgery: an evidence based review Christian Falconer, MD, PhD Department of Obstetrics and Gynecology Danderyd University Hospital Stockholm, Sweden Finnish Society
Current status in pelvic organ prolapse surgery: an evidence based review Christian Falconer, MD, PhD Department of Obstetrics and Gynecology Danderyd University Hospital Stockholm, Sweden Finnish Society
Blue Ridge Urogynecology
 Surgery for Stress Urinary Incontinence Surgery has proved to be a very effective treatment for stress incontinence. The best surgical procedures improve or cure the incontinence in 85 to 90 percent of
Surgery for Stress Urinary Incontinence Surgery has proved to be a very effective treatment for stress incontinence. The best surgical procedures improve or cure the incontinence in 85 to 90 percent of
One Slim Needle One Incision. One Simple Solution for Stress Urinary Incontinence. The Difference is in the Data
 CONTINENCE SOLUTIONS One Slim Needle One Incision ordering information Description US International Order Number Order Number One Simple Solution for Stress Urinary Incontinence MiniArc Single-Incision
CONTINENCE SOLUTIONS One Slim Needle One Incision ordering information Description US International Order Number Order Number One Simple Solution for Stress Urinary Incontinence MiniArc Single-Incision
Robotic Ventral Rectopexy
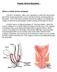 Robotic Ventral Rectopexy What is a robotic ventral rectopexy? The term rectopexy refers to an operation in which the rectum (the part of the bowel nearest the anus) is put back into its normal position
Robotic Ventral Rectopexy What is a robotic ventral rectopexy? The term rectopexy refers to an operation in which the rectum (the part of the bowel nearest the anus) is put back into its normal position
9/24/2015. Pelvic Floor Disorders. Agenda. What is the Pelvic Floor? Pelvic Floor Problems
 Management of Pelvic Floor Disorders Doctor, I don t want THAT mesh! Agenda What are pelvic floor disorders (PFDs)? What are the treatment options? Expectant. Conservative. Surgical. How and when are grafts
Management of Pelvic Floor Disorders Doctor, I don t want THAT mesh! Agenda What are pelvic floor disorders (PFDs)? What are the treatment options? Expectant. Conservative. Surgical. How and when are grafts
Stop Coping. Start Living. Talk to your doctor about pelvic organ prolapse and sacrocolpopexy
 Stop Coping. Start Living Talk to your doctor about pelvic organ prolapse and sacrocolpopexy Did you know? One in three women will suffer from a pelvic health condition in her lifetime. Four of the most
Stop Coping. Start Living Talk to your doctor about pelvic organ prolapse and sacrocolpopexy Did you know? One in three women will suffer from a pelvic health condition in her lifetime. Four of the most
HYSTERECTOMY FOR BENIGN CONDITIONS
 UnitedHealthcare Commercial Medical Policy HYSTERECTOMY FOR BENIGN CONDITIONS Policy Number: 2018T0572G Effective Date: September 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT CONSIDERATIONS...
UnitedHealthcare Commercial Medical Policy HYSTERECTOMY FOR BENIGN CONDITIONS Policy Number: 2018T0572G Effective Date: September 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT CONSIDERATIONS...
Considering Surgery for Pelvic Prolapse? Learn about minimally invasive da Vinci Surgery
 Considering Surgery for Pelvic Prolapse? Learn about minimally invasive da Vinci Surgery The Surgery: Pelvic Prolapse Surgery If you have pelvic prolapse symptoms, your doctor may suggest medicine or lifestyle
Considering Surgery for Pelvic Prolapse? Learn about minimally invasive da Vinci Surgery The Surgery: Pelvic Prolapse Surgery If you have pelvic prolapse symptoms, your doctor may suggest medicine or lifestyle
Sling The Mesh The Beginning
 Website: slingthemesh.com @meshcampaign Sling The Mesh The Beginning How it began From 20 members to more than 7000 Media is a powerful campaign tool Taboo busting Understanding science with a drama degree
Website: slingthemesh.com @meshcampaign Sling The Mesh The Beginning How it began From 20 members to more than 7000 Media is a powerful campaign tool Taboo busting Understanding science with a drama degree
Consultation Guide: Specialised gynaecology surgery and complex urogynaecology conditions service specifications
 Consultation Guide: Specialised gynaecology surgery and complex urogynaecology conditions service specifications Consultation guide: Specialised gynaecology surgery and complex urogynaecology conditions
Consultation Guide: Specialised gynaecology surgery and complex urogynaecology conditions service specifications Consultation guide: Specialised gynaecology surgery and complex urogynaecology conditions
HYSTERECTOMY FOR BENIGN CONDITIONS
 HYSTERECTOMY FOR BENIGN CONDITIONS UnitedHealthcare Oxford Clinical Policy Policy Number: SURGERY 104.7 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
HYSTERECTOMY FOR BENIGN CONDITIONS UnitedHealthcare Oxford Clinical Policy Policy Number: SURGERY 104.7 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
INCONTINENCE AND OTHER UROLOGICAL DILEMMAS DR. ANNA LAWRENCE UROLOGIST AUCKLAND HOSPITAL 161 UROLOGY
 INCONTINENCE AND OTHER UROLOGICAL DILEMMAS DR. ANNA LAWRENCE UROLOGIST AUCKLAND HOSPITAL 161 UROLOGY COVERING INCONTINENCE BE ON JUST NAPPIES CATHETERS TYPES AVAILABLE AND WHEN TO USE THEM JJ STENTS???
INCONTINENCE AND OTHER UROLOGICAL DILEMMAS DR. ANNA LAWRENCE UROLOGIST AUCKLAND HOSPITAL 161 UROLOGY COVERING INCONTINENCE BE ON JUST NAPPIES CATHETERS TYPES AVAILABLE AND WHEN TO USE THEM JJ STENTS???
Urethrolysis; When, Why & How. M Karram Professor of Ob/Gyn & Urology University of Cincinnati
 Urethrolysis; When, Why & How M Karram Professor of Ob/Gyn & Urology University of Cincinnati Anatomy Urethra may be fixed to the pubic bone with dense scar tissue Goal of urethrolysis is to completely
Urethrolysis; When, Why & How M Karram Professor of Ob/Gyn & Urology University of Cincinnati Anatomy Urethra may be fixed to the pubic bone with dense scar tissue Goal of urethrolysis is to completely
Surgical treatment of urinary stress incontinence with tension free vaginal tape
 Surgical treatment of urinary stress incontinence with tension free vaginal tape Gynaecology department 01935 384 385 yeovilhospital.nhs.uk Many surgical operations are available for the treatment of
Surgical treatment of urinary stress incontinence with tension free vaginal tape Gynaecology department 01935 384 385 yeovilhospital.nhs.uk Many surgical operations are available for the treatment of
Patient Information Leaflet
 Patient Information Leaflet MID-URETHRAL SLING OPERATION TENSION-FREE VAGINAL TAPE (TVT) TRANSOBTURATOR TAPE (TOT, TVT-O) This information leaflet has been developed to help your understanding of what
Patient Information Leaflet MID-URETHRAL SLING OPERATION TENSION-FREE VAGINAL TAPE (TVT) TRANSOBTURATOR TAPE (TOT, TVT-O) This information leaflet has been developed to help your understanding of what
Complications from permanent synthetic mesh
 Original Research Symptom Resolution After Operative Management of Complications From Transvaginal Mesh Erin C. Crosby, MD, Melinda Abernethy, MD, MPH, Mitchell B. Berger, MD, PhD, John O. DeLancey, MD,
Original Research Symptom Resolution After Operative Management of Complications From Transvaginal Mesh Erin C. Crosby, MD, Melinda Abernethy, MD, MPH, Mitchell B. Berger, MD, PhD, John O. DeLancey, MD,
Use of Power Morcellators: Minimizing Liability, Assuring Safety? By Barbara Youngberg
 EXAM INATIONS Examining the industry market trends that matter most to you February 2015 A Beecher Carlson Publication Use of Power Morcellators: Minimizing Liability, Assuring Safety? By Barbara Youngberg
EXAM INATIONS Examining the industry market trends that matter most to you February 2015 A Beecher Carlson Publication Use of Power Morcellators: Minimizing Liability, Assuring Safety? By Barbara Youngberg
Ralph & Berryl Goldman Fellowship in Neuro urology, voiding dysfunction and bladder reconstruction
 Ralph & Berryl Goldman Fellowship in Neuro urology, voiding dysfunction and bladder reconstruction Length of fellowship: A 1 year Fellowship: This fellowship has been designed for Urology or Gynecology
Ralph & Berryl Goldman Fellowship in Neuro urology, voiding dysfunction and bladder reconstruction Length of fellowship: A 1 year Fellowship: This fellowship has been designed for Urology or Gynecology
PL Narducci Department of Obstetrics and Gynecology General Hospital San Giovanni Battista Foligno, ITALY
 NESA DAYS 2018 New European Surgical Academy Perugia, April 19-21, 2018 EXCELLENCE IN FEMALE SURGERY PROLAPSE RECONSTRUCTIVE SURGERY IN SEXUALLY ACTIVE WOMEN LAPAROSCOPIC ANTERIOR ABDOMINAL WALL COLPOPEXY
NESA DAYS 2018 New European Surgical Academy Perugia, April 19-21, 2018 EXCELLENCE IN FEMALE SURGERY PROLAPSE RECONSTRUCTIVE SURGERY IN SEXUALLY ACTIVE WOMEN LAPAROSCOPIC ANTERIOR ABDOMINAL WALL COLPOPEXY
Registry Protocol Research Registry (PFDR-R) Version 1.3. (August 2016)
 Registry Protocol Research Registry (PFDR-R) Version 1.3 (August 2016) Table of Contents List of Abbreviations... 4 1. Background... 5 2. Rationale... 7 3. Objectives... 8 4. Registry Design... 9 4.1 Registry
Registry Protocol Research Registry (PFDR-R) Version 1.3 (August 2016) Table of Contents List of Abbreviations... 4 1. Background... 5 2. Rationale... 7 3. Objectives... 8 4. Registry Design... 9 4.1 Registry
A Simple Solution. to a Common Problem. Find relief from sudden, unplanned urine leakage.
 A Simple Solution to a Common Problem Find relief from sudden, unplanned urine leakage. A Common Problem Millions of women suffer from stress urinary incontinence (SUI). This condition results in accidental
A Simple Solution to a Common Problem Find relief from sudden, unplanned urine leakage. A Common Problem Millions of women suffer from stress urinary incontinence (SUI). This condition results in accidental
Prolapse & Stress Incontinence
 Advanced Pelvic Floor Course Prolapse & Stress Incontinence OVERVIEW Day One and morning of Day Two- Pelvic Organ Prolapse The Prolapse component covers the detailed anatomy of POP including the DeLancey
Advanced Pelvic Floor Course Prolapse & Stress Incontinence OVERVIEW Day One and morning of Day Two- Pelvic Organ Prolapse The Prolapse component covers the detailed anatomy of POP including the DeLancey
CHAU KHAC TU M.D., Ph.D.
 CHAU KHAC TU M.D., Ph.D. Hue Central Hospital Vietnam LAPAROSCOPIC PROMONTOFIXATION FOR THE GENITAL PROLAPSE TREATMENT Chau Khac Tu MD.PhD. Hue central hospital CONTENT 3 1 INTRODUCTION 2 OBJECTIVE AND
CHAU KHAC TU M.D., Ph.D. Hue Central Hospital Vietnam LAPAROSCOPIC PROMONTOFIXATION FOR THE GENITAL PROLAPSE TREATMENT Chau Khac Tu MD.PhD. Hue central hospital CONTENT 3 1 INTRODUCTION 2 OBJECTIVE AND
Advanced Care for Female Overactive Bladder & Urinary Incontinence. Department of Urology Kaiser Permanente Santa Rosa
 Advanced Care for Female Overactive Bladder & Urinary Incontinence Department of Urology Kaiser Permanente Santa Rosa Goals Participants will: Review normal urinary tract anatomy and function Understand
Advanced Care for Female Overactive Bladder & Urinary Incontinence Department of Urology Kaiser Permanente Santa Rosa Goals Participants will: Review normal urinary tract anatomy and function Understand
DENOMINATOR: All patients undergoing anterior or apical pelvic organ prolapse (POP) surgery
 Quality ID #428: Pelvic Organ Prolapse: Preoperative Assessment of Occult Stress Urinary Incontinence National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY
Quality ID #428: Pelvic Organ Prolapse: Preoperative Assessment of Occult Stress Urinary Incontinence National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY
Anatomical and Functional Results of Pelvic Organ Prolapse Mesh Repair: A Prospective Study of 105 Cases
 International Journal of Clinical Urology 2018; 2(1): 20-24 http://www.sciencepublishinggroup.com/j/ijcu doi: 10.11648/j.ijcu.20180201.14 Anatomical and Functional Results of Pelvic Organ Prolapse Mesh
International Journal of Clinical Urology 2018; 2(1): 20-24 http://www.sciencepublishinggroup.com/j/ijcu doi: 10.11648/j.ijcu.20180201.14 Anatomical and Functional Results of Pelvic Organ Prolapse Mesh
International Federation of Gynecology and Obstetrics
 International Federation of Gynecology and Obstetrics COMMITTEE FOR UROGYNAECOLOGY AND PELVIC FLOOR MEMBER: TSUNG-HSIEN (CHARLES) SU, CHAIR (TAIWAN) DAVID RICHMOND, CO-CHAIR (UK) CHITTARANJAN PURANDARE,
International Federation of Gynecology and Obstetrics COMMITTEE FOR UROGYNAECOLOGY AND PELVIC FLOOR MEMBER: TSUNG-HSIEN (CHARLES) SU, CHAIR (TAIWAN) DAVID RICHMOND, CO-CHAIR (UK) CHITTARANJAN PURANDARE,
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Outcome
 Quality ID #432: Proportion of Patients Sustaining a Bladder Injury at the Time of any Pelvic Organ Prolapse Repair National Quality Strategy Domain: Patient Safety 2018 OPTIONS FOR INDIVIDUAL MEASURES:
Quality ID #432: Proportion of Patients Sustaining a Bladder Injury at the Time of any Pelvic Organ Prolapse Repair National Quality Strategy Domain: Patient Safety 2018 OPTIONS FOR INDIVIDUAL MEASURES:
Las Vegas Urogynecology
 Las Vegas Urogynecology 7500 Smoke Ranch Road - #200 Las Vegas, NV 89128 Telephone: (702) 233-0727 Fax: (702) 233-4799 Physician's Surgical Procedure Disclosure and Patient s Consent TO THE PATIENT: You
Las Vegas Urogynecology 7500 Smoke Ranch Road - #200 Las Vegas, NV 89128 Telephone: (702) 233-0727 Fax: (702) 233-4799 Physician's Surgical Procedure Disclosure and Patient s Consent TO THE PATIENT: You
Understanding Pelvic Organ Prolapse. Stephanie Pickett, MD, MS Female Pelvic Medicine and Reconstructive Surgery
 Understanding Pelvic Organ Prolapse Stephanie Pickett, MD, MS Female Pelvic Medicine and Reconstructive Surgery Disclosures None I am the daughter of a physician assistant. Objectives List types of pelvic
Understanding Pelvic Organ Prolapse Stephanie Pickett, MD, MS Female Pelvic Medicine and Reconstructive Surgery Disclosures None I am the daughter of a physician assistant. Objectives List types of pelvic
Clinical Policy: Urinary Incontinence Devices and Treatments
 Clinical Policy: Reference Number: CP.MP.142 Last Review Date: 03/18 See Important Reminder at the end of this policy for important regulatory and legal information. Coding Implications Revision Log Description
Clinical Policy: Reference Number: CP.MP.142 Last Review Date: 03/18 See Important Reminder at the end of this policy for important regulatory and legal information. Coding Implications Revision Log Description
Current trend in anti-incontinence surgery
 Current trend in anti-incontinence surgery 吳銘斌醫師 (Ming-Ping Wu, M.D., Ph.D.) 奇美醫院婦產部婦女泌尿暨骨盆醫學科主任台北醫學大學醫學院婦產學科副教授成功大學醫學院臨床醫學所博士 Anti-incontinence surgery Bladder buttress operation: Kelly plication Needle
Current trend in anti-incontinence surgery 吳銘斌醫師 (Ming-Ping Wu, M.D., Ph.D.) 奇美醫院婦產部婦女泌尿暨骨盆醫學科主任台北醫學大學醫學院婦產學科副教授成功大學醫學院臨床醫學所博士 Anti-incontinence surgery Bladder buttress operation: Kelly plication Needle
INTERNATIONAL UROGYNAECOLOGICAL ASSOCIATION (IUGA) JOINT REPORT ON THE TERMINOLOGY FOR SURGICAL PROCEDURES TO
 AN AMERICAN UROGYNECOLOGIC SOCIETY (AUGS) / INTERNATIONAL UROGYNAECOLOGICAL ASSOCIATION (IUGA) JOINT REPORT ON THE TERMINOLOGY FOR SURGICAL PROCEDURES TO TREAT PELVIC ORGAN PROLAPSE NEED FOR A WORKING
AN AMERICAN UROGYNECOLOGIC SOCIETY (AUGS) / INTERNATIONAL UROGYNAECOLOGICAL ASSOCIATION (IUGA) JOINT REPORT ON THE TERMINOLOGY FOR SURGICAL PROCEDURES TO TREAT PELVIC ORGAN PROLAPSE NEED FOR A WORKING
Desara and Desara Blue
 Desara and Desara Blue Sling for Female Stress Urinary Incontinence Instructions For Use D I Prescription Use only Do not reuse Sterilized using ethylene oxide M Manufactured by: Caldera Medical, Inc.
Desara and Desara Blue Sling for Female Stress Urinary Incontinence Instructions For Use D I Prescription Use only Do not reuse Sterilized using ethylene oxide M Manufactured by: Caldera Medical, Inc.
2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Measure #428: Pelvic Organ Prolapse: Preoperative Assessment of Occult Stress Urinary Incontinence National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY
Measure #428: Pelvic Organ Prolapse: Preoperative Assessment of Occult Stress Urinary Incontinence National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY
Urogynaecology Update. Andrew Tapp Consultant Obstetrician and Gynaecologist Shrewsbury & Telford Hospital NHS Trust
 Urogynaecology Update Andrew Tapp Consultant Obstetrician and Gynaecologist Shrewsbury & Telford Hospital NHS Trust Urogynaecology Training Core module 18. ATSM. Sub-specialty training. Urogynaecology
Urogynaecology Update Andrew Tapp Consultant Obstetrician and Gynaecologist Shrewsbury & Telford Hospital NHS Trust Urogynaecology Training Core module 18. ATSM. Sub-specialty training. Urogynaecology
Introduction to GYN Specialties
 Outline Introduction to GYN Specialties Gynecologic Oncology* Female Pelvic Medicine and Reconstructive Surgery* Reproductive Endocrinology and Infertility* Pediatric and Adolescent Gynecology** Family
Outline Introduction to GYN Specialties Gynecologic Oncology* Female Pelvic Medicine and Reconstructive Surgery* Reproductive Endocrinology and Infertility* Pediatric and Adolescent Gynecology** Family
PRACTICE BULLETIN Female Pelvic Medicine & Reconstructive Surgery Volume 23, Number 4, July/August 2017
 PRACTICE BULLETIN Number 176, April 2017 (Replaces Committee Opinion Number 513, December 2011) Pelvic Organ Prolapse Pelvic organ prolapse (POP) is a common, benign condition in women. For many women
PRACTICE BULLETIN Number 176, April 2017 (Replaces Committee Opinion Number 513, December 2011) Pelvic Organ Prolapse Pelvic organ prolapse (POP) is a common, benign condition in women. For many women
Evidence Summary: The inflow Urinary Prosthesis FDA De Novo Approval DEN130044
 Evidence Summary: The inflow Urinary Prosthesis FDA De Novo Approval DEN130044 Unique alternative to urinary catheters for women with permanent urinary retention Therapeutic benefits o Highly effective
Evidence Summary: The inflow Urinary Prosthesis FDA De Novo Approval DEN130044 Unique alternative to urinary catheters for women with permanent urinary retention Therapeutic benefits o Highly effective
Medical Devices and the Public s Health: The FDA 510(k) Clearance Process at 35 Years. Written Statement of
 Medical Devices and the Public s Health: The FDA 510(k) Clearance Process at 35 Years Written Statement of Dr. David R. Challoner Vice President for Health Affairs, Emeritus University of Florida and Chair,
Medical Devices and the Public s Health: The FDA 510(k) Clearance Process at 35 Years Written Statement of Dr. David R. Challoner Vice President for Health Affairs, Emeritus University of Florida and Chair,
Infracoccygeal sacropexy using mesh for uterine prolapse repair
 Infracoccygeal sacropexy using mesh for uterine Issued: January 2009 www.nice.org.uk/ipg280 NHS Evidence has accredited the process used by the NICE Interventional Procedures Programme to produce interventional
Infracoccygeal sacropexy using mesh for uterine Issued: January 2009 www.nice.org.uk/ipg280 NHS Evidence has accredited the process used by the NICE Interventional Procedures Programme to produce interventional
INSTRUCTIONS FOR USE FOR:
 INSTRUCTIONS FOR USE FOR: en English INSTRUCTIONS FOR USE GORE ENFORM PREPERITONEAL BIOMATERIAL Carefully read all instructions prior to use. Observe all instructions, warnings, and precautions noted throughout.
INSTRUCTIONS FOR USE FOR: en English INSTRUCTIONS FOR USE GORE ENFORM PREPERITONEAL BIOMATERIAL Carefully read all instructions prior to use. Observe all instructions, warnings, and precautions noted throughout.
Urogynecology: Evidence-Based Clinical Practice
 Urogynecology: Evidence-Based Clinical Practice Kate H. Moore Urogynecology: Evidence-Based Clinical Practice Second Edition Kate H. Moore, MBBS, FRCOG, FRANZCOG, MD, CU Department Obstetrics & Gynaecology
Urogynecology: Evidence-Based Clinical Practice Kate H. Moore Urogynecology: Evidence-Based Clinical Practice Second Edition Kate H. Moore, MBBS, FRCOG, FRANZCOG, MD, CU Department Obstetrics & Gynaecology
NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE SCOPE. Urinary incontinence: the management of urinary incontinence in women
 NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE 1 Guideline title SCOPE Urinary incontinence: the management of urinary incontinence in women 1.1 Short title Urinary incontinence 2 Background a) The National
NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE 1 Guideline title SCOPE Urinary incontinence: the management of urinary incontinence in women 1.1 Short title Urinary incontinence 2 Background a) The National
DENOMINATOR: All patients undergoing anterior or apical pelvic organ prolapse (POP) surgery
 Measure #428: Pelvic Organ Prolapse: Preoperative Assessment of Occult Stress Urinary Incontinence National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTY
Measure #428: Pelvic Organ Prolapse: Preoperative Assessment of Occult Stress Urinary Incontinence National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTY
Tension-free Vaginal Tape (TVT)
 Page 1 of 7 Tension-free Vaginal Tape (TVT) Introduction This leaflet will provide you with basic information about the Tension--free Vaginal Tape (TVT) procedure. What is a TVT? TVT is an operation to
Page 1 of 7 Tension-free Vaginal Tape (TVT) Introduction This leaflet will provide you with basic information about the Tension--free Vaginal Tape (TVT) procedure. What is a TVT? TVT is an operation to
In the August 2016 issue of OBG Management,
 GYN coding changes to note for your maximized reimbursement Revised ICD-10 gynecologic diagnostic codes go into effect October 1. Here is a look at the added, expanded, and revised codes you will need
GYN coding changes to note for your maximized reimbursement Revised ICD-10 gynecologic diagnostic codes go into effect October 1. Here is a look at the added, expanded, and revised codes you will need
Female Pelvic Medicine & Reconstructive Surgery
 Female Pelvic Medicine & Reconstructive Surgery APPLICATION FOR NEW FELLOWSHIP Name of Institution: McGill University Location: Royal Victoria Hospital (Glen Site), St Mary s Hospital Centre Type of Fellowship:
Female Pelvic Medicine & Reconstructive Surgery APPLICATION FOR NEW FELLOWSHIP Name of Institution: McGill University Location: Royal Victoria Hospital (Glen Site), St Mary s Hospital Centre Type of Fellowship:
2/17/2018. Prof. Steven S. Saliterman Department of Biomedical Engineering, University of Minnesota
 Department of Biomedical Engineering, University of Minnesota http://saliterman.umn.edu/ FDA Authority Medical Device Act of 1976 FDA Modernization Act of 1997 Federal Food, Drug and Cosmetic Act (FFDCA)
Department of Biomedical Engineering, University of Minnesota http://saliterman.umn.edu/ FDA Authority Medical Device Act of 1976 FDA Modernization Act of 1997 Federal Food, Drug and Cosmetic Act (FFDCA)
Prof. Steven S. Saliterman. Department of Biomedical Engineering, University of Minnesota
 Department of Biomedical Engineering, University of Minnesota http://saliterman.umn.edu/ FDA Authority Medical Device Act of 1976 FDA Modernization Act of 1997 Federal Food, Drug and Cosmetic Act (FFDCA)
Department of Biomedical Engineering, University of Minnesota http://saliterman.umn.edu/ FDA Authority Medical Device Act of 1976 FDA Modernization Act of 1997 Federal Food, Drug and Cosmetic Act (FFDCA)
Modern methods of imaging in urogynecology when do we really need them?
 Archives of Perinatal Medicine 23(2), 77 81, 2017 ORIGINAL PAPER Modern methods of imaging in urogynecology when do we really need them? GRZEGORZ SURKONT, EDYTA WLAŹLAK Abstract Imaging is more often used
Archives of Perinatal Medicine 23(2), 77 81, 2017 ORIGINAL PAPER Modern methods of imaging in urogynecology when do we really need them? GRZEGORZ SURKONT, EDYTA WLAŹLAK Abstract Imaging is more often used
Surgical mesh implants for treatment of urinary incontinence and pelvic organ prolapse. ANSM review of French market between 2014 and 2017
 Surgical mesh implants for treatment of urinary incontinence and pelvic organ prolapse ANSM review of French market between 2014 and 2017 November 2018 Summary Introduction... 2 Methodology... 4 Results
Surgical mesh implants for treatment of urinary incontinence and pelvic organ prolapse ANSM review of French market between 2014 and 2017 November 2018 Summary Introduction... 2 Methodology... 4 Results
Medical Review Criteria Invasive Treatment for Urinary Incontinence
 Medical Review Criteria Invasive Treatment for Urinary Incontinence Effective Date: December 21, 2016 Subject: Invasive Treatment for Urinary Incontinence Background: Urinary incontinence (the involuntary
Medical Review Criteria Invasive Treatment for Urinary Incontinence Effective Date: December 21, 2016 Subject: Invasive Treatment for Urinary Incontinence Background: Urinary incontinence (the involuntary
Stress Urinary Incontinence in Women
 Stress Urinary Incontinence in Women Stress Urinary Incontinence in Women: It's Common. It's Treatable. Would it surprise you... To learn that more than 3 million women in the United Kingdom have urinary
Stress Urinary Incontinence in Women Stress Urinary Incontinence in Women: It's Common. It's Treatable. Would it surprise you... To learn that more than 3 million women in the United Kingdom have urinary
Clinical Curriculum: Urogynecology
 Updated July 201 Clinical Curriculum: Urogynecology GOAL: The primary goal of the Urogynecology rotation at the University of Alabama at Birmingham (UAB) is to train physicians to have a broad knowledge
Updated July 201 Clinical Curriculum: Urogynecology GOAL: The primary goal of the Urogynecology rotation at the University of Alabama at Birmingham (UAB) is to train physicians to have a broad knowledge
Clinical Policy: Essure Removal Reference Number: CP.MP.131
 Clinical Policy: Reference Number: CP.MP.131 Effective Date: 11/16 Last Review Date: 11/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.131 Effective Date: 11/16 Last Review Date: 11/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
ARTIFICIAL MESH REPAIR FOR TREATMENT OF PELVIC ORGAN PROLAPSE
 Pelvic Floor Unit / Department of Gynaecology Ward 17, Singleton Hospital, Sketty, Swansea, SA2 8QA 01792 205666 Secretary Direct Line: 01792 285688. Fax: 01792 285874 ARTIFICIAL MESH REPAIR FOR TREATMENT
Pelvic Floor Unit / Department of Gynaecology Ward 17, Singleton Hospital, Sketty, Swansea, SA2 8QA 01792 205666 Secretary Direct Line: 01792 285688. Fax: 01792 285874 ARTIFICIAL MESH REPAIR FOR TREATMENT
T h e C o m p l e t e Tr e a t m e n t o f P e l v i c F l o o r P r o l a p s e by Laparoscopy Technique, Tips and Tricks
 T h e C o m p l e t e Tr e a t m e n t o f P e l v i c F l o o r P r o l a p s e by Laparoscopy Technique, Tips and Tricks R Botchorishvili, A Wattiez, G Mage, M Canis, B Rabischong, K Jardon, C Rivoire,
T h e C o m p l e t e Tr e a t m e n t o f P e l v i c F l o o r P r o l a p s e by Laparoscopy Technique, Tips and Tricks R Botchorishvili, A Wattiez, G Mage, M Canis, B Rabischong, K Jardon, C Rivoire,
Women s Health. Product innovation. Commitment. We are committed to you... and advancing the quality of your patient care.
 II Commitment We are committed to you... and advancing the quality of your patient care. Boston Scientific is always there for you, your physicians and your patients. We are anchored in our guiding principle
II Commitment We are committed to you... and advancing the quality of your patient care. Boston Scientific is always there for you, your physicians and your patients. We are anchored in our guiding principle
Urinary Incontinence. Biopolymer GmbH & Co.KG - Germany
 Urinary Incontinence Treatment Options: 1. Physical Therapy: a conservative early stage treatment for UI, including pelvic floor muscles training known as Kegel exercise. 2. Medications: by means of pharmaceutical
Urinary Incontinence Treatment Options: 1. Physical Therapy: a conservative early stage treatment for UI, including pelvic floor muscles training known as Kegel exercise. 2. Medications: by means of pharmaceutical
ROBOTIC PRECISION. HUMAN COMPASSION.
 ROBOTIC PRECISION. HUMAN COMPASSION. Find out how robotic surgery can help you. Find a surgeon, attend a class, or learn more at adena.org/robot or by calling 877-779-7585. ADENA S NEW DA VINCI SI ROBOTIC
ROBOTIC PRECISION. HUMAN COMPASSION. Find out how robotic surgery can help you. Find a surgeon, attend a class, or learn more at adena.org/robot or by calling 877-779-7585. ADENA S NEW DA VINCI SI ROBOTIC
NOTICE OF FILING. Details of Filing. Kathryn Gill & Ors v Ethicon Sarl & Ors NEW SOUTH WALES REGISTRY - FEDERAL COURT OF AUSTRALIA
 NOTICE OF FILING This document was lodged electronically in the FEDERAL COURT OF AUSTRALIA (FCA) on 13/04/2018 3:35:30 PM AEST and has been accepted for filing under the Court s Rules. Details of filing
NOTICE OF FILING This document was lodged electronically in the FEDERAL COURT OF AUSTRALIA (FCA) on 13/04/2018 3:35:30 PM AEST and has been accepted for filing under the Court s Rules. Details of filing
