Full Paper. Koumei SHIRASUNA 1), Takayuki ASAHI 1), Motoki SASAKI 2), Takashi SHIMIZU 1) and Akio MIYAMOTO 1)
|
|
|
- Octavia Warner
- 5 years ago
- Views:
Transcription
1 Journal of Reproduction and Development, Vol. 56, No. 1, 2010, O Full Paper Distribution of Arteriolovenous Vessels, Capillaries and enos Expression in the Bovine Corpus Luteum During the Estrous Cycle: a Possible Implication of Different Sensitivity by Luteal Phase to PGF 2α in the Increase of Luteal Blood Flow Koumei SHIRASUNA 1), Takayuki ASAHI 1), Motoki SASAKI 2), Takashi SHIMIZU 1) and Akio MIYAMOTO 1) 1) Graduate School of Animal and Food Hygiene, Obihiro University of Agriculture and Veterinary Medicine, Obihiro and 2) Department of Basic Veterinary Sciences, Obihiro University of Agriculture and Veterinary Medicine, Obihiro , Japan Abstract. We have shown that luteal blood flow increases in the peripheral vasculature of the mature corpus luteum (CL) prior to the onset of luteolysis in response to prostaglandin F 2α (PGF 2α) in the cow, but this phenomenon does not occur in the early CL. We therefore hypothesize that this acute increase of luteal blood flow occurs by vasodilation of large blood vessels due to local release of nitric oxide (NO). In the present study, we characterized the CL vasculature together with endothelial NO synthase (enos) expression during the estrous cycle in the cow. Immunohistochemistry was used to quantify the number of arteriolovenous vessels (surrounded with smooth muscle actin-positive smooth muscle cells), capillaries (with von Willebrand Factor-positive endothelial cells) and enos protein. The arteriolovenous vessels existed more in the periphery of the matured CL (mid, late and regressing CL) than in the center region. In the early CL, there were as many arteriolovenous vessels in the periphery as in the center, while more capillaries existed in the center than in the periphery of the mid and late CL. Also, enos protein existed in the periphery more than in the center of the matured CL. These results indicate that the early CL has a homogeneous vascular and enos distribution. In contrast, the matured CL is a heterogeneous organ having a higher vascular and enos distribution in the periphery than in the center. In conclusion, the distribution of arteriolovenous vessels and enos in the matured CL was higher in the periphery than in the center of the CL. Thus, this suggests that this structural change from the early (homogeneous) to the mid (heterogeneous) luteal phase is related to the difference in the CL response of blood flow increase due to PGF 2α, which is only observed in the mature CL. Key words: Blood vessels, Corpus luteum, enos, Luteolysis (J. Reprod. Dev. 56: , 2010) rich vascular network is established within the corpus luteum (CL) in order to enable it to function as an endocrine gland. Although the CL is constituted of several types of cells, such as luteal cells, endothelial cells and smooth muscle cells, vascular endothelial cells account for up to 50% of the bovine mature CL [1, 2]. Moreover, endothelin-1 (EDN1) and angiotensin II (Ang II) secreted by endothelial cells in the CL have been shown to inhibit progesterone secretion from luteal cells [3 5]. These findings indicate that luteal blood vessels have a key role in regulation of luteal function. Functionally, luteal blood vessels can be divided roughly into two types of blood vessels. One of these types is an arteriolovenous vessels, i.e., arteriola and venula vessels, which have a smooth muscle layer and receive the vasorelaxant effect. In general, the diameter of an arteriola is about 40 μm (from 20 μm to 100 μm), and the arteriola has an important role in creation of peripheral resistance for local blood flow. In the circulation of blood, the Received: June 15, 2009 Accepted: October 9, 2009 Published online in J-STAGE: November 5, by the Society for Reproduction and Development Correspondence: A Miyamoto ( akiomiya@obihiro.ac.jp) arteriola connects to microcapillary vessels followed by venulae (diameter: about 30 μm) [6]. The venula also has pericyte and/or smooth muscle cells and a vascular tonus function, which includes vasodilation. Indeed, a nitroglycerin, strong vasodilator, can induce vasodilation in not only the arteriola but also in the venula [7]. The other type is capillary vessels, which do not have a smooth muscle layer and receive little vasorelaxant effect. It has been reported that the number of arteriolovenous vessels in the CL increases from the early to mid luteal phase [8, 9]. Additionally, in the rat and rabbit CL, arteriolovenous vessels existed in the periphery of the CL [10, 11], and luteal blood flow is observed mainly in the periphery of the bovine CL in color Doppler images [12, 13]. These findings suggest that the CL is a heterogeneous organ in terms of the distribution of arteriolovenous and capillary vessels. It has been reported in the ewe that blood flow to the CL decreases dramatically in association with luteolysis [14]. More precisely, our recent study indicated that blood flow in the periphery of the CL drastically and transiently increased at h after PGF 2α injection (the dose of PGF 2α analogue recommended by the manufacture) in the mid luteal phase and thereafter gradually decreased over the course of luteolysis [12]. In contrast, PGF 2α induces neither luteal blood flow increase nor luteolysis during the
2 VASCULATURE AND enos IN THE CORPUS LUTEUM 125 early luteal phase [12]. Thus, the acute increase of luteal blood flow induced by PGF 2α may be one of the earliest physiological signs for the luteolytic cascade. We recently demonstrated that the increase in luteal blood flow in response to PGF 2α is induced by vasodilation due to the local action of luteal nitric oxide (NO) [15]. NO is well known as a potent vasodilator, produced by endothelial NO synthase (enos) and inducible NO synthase (inos), and the bovine CL expresses both types of NOS [16]. Furthermore, an immunohistochemical study has shown that the bovine CL expresses NOS at the highest level during the late luteal phase of the estrous cycle [16]. Actually, NO inhibits progesterone secretion from luteal cells [17], and an NOS inhibitor prolongs the estrous cycle and delays luteolysis induced by PGF 2α[16]. We hypothesized that more arteriolovenous vessels, which are capable of vasodilation, should exist in the periphery of the mid CL and that therefore PGF 2α could induce an increase in luteal blood flow in the arteriolovenous vessels, where enos is dominantly expressed compared with the center of the mid CL; they are thereby more sensitive to PGF 2α in vasodilating response. In the present study, we therefore examined the localization of arteriolovenous vessels, capillaries and enos expression in the cyclic bovine CL using immunohistochemistry. Materials and Methods Immunohistochemistry Ovaries with CLs were collected from cows at a local abattoir within min after slaughter. The ovaries were kept in ice-cold phosphate-buffered saline (PBS, ph 7.2) until preparation at the laboratory. The luteal stage was carefully defined by macroscopic observation of the ovaries (follicles and CL) and described in a previous study [18]. The CLs were divided into four groups as follows: early (Days 2 5; n=5), mid (Days 8 12; n=5), late (Days 15 17; n=5) and regressing (after Day 18; n=5). The CLs were enucleated from the ovary and dissected free of connective tissue. Tissue samples were fixed in Bouin s fixative for 24 h at room temperature and then embedded in paraffin wax. Serial sections (5 μm) were mounted onto APS-coated glass microscope slides. The sections were stained with hematoxylin-eosin (H E) for general histological observations. Light microscopic immunohistochemical staining employing the avidin-biotin peroxidase complex (ABC) method [19] was used as in the previous study [20]. We used polyclonal antibodies for von Willebrand Factor (vwf; Dako, diluted 1:200), which is the marker of endothelial cells, smooth muscle actin (SMA; Dako, diluted 1:200), and enos (Affinity BioRegents TM, PA1-037, diluted 1:50) in the present study. As the secondary antibody, we used biotinylated goat anti-rabbit IgG (1:200, BA-1000, Vector Laboratories) for vwf, enos and the negative control and biotinylated goat antimouse IgG (1:200, BA-9200, Vector Laboratories) for SMA. Horseradish peroxidase (HRP) conjugated ABC (1:2 dilution, PK- 6100, Vectastain Elite ABC kit, Vector Laboratories) combined with the secondary antibody were applied to tissue slides at room temperature for 30 min. The binding sites were visualized with 0.02% 3,3 -diaminobenzidine tetrahydrochloride (DAB) in 50 mm Tris-HCL (ph 7.4) containing 0.02% H 2O 2. Decision of the observation area between the periphery and the center of the CL In this study, every observation and quantification was performed in the periphery and center of the CL separately as in our previous study [20]. To examine the local effect of PGF 2α in the CL, tissue samples were collected from regions designated as the periphery of the CL (in the range of 1 mm from the boundary between the luteal tissue and ovarian parenchyma) and center of the CL (in the range of 1.5 mm from the center section of the CL). Measurement of the cross-sectional area of the large luteal cell (LLC) The cross-sectional area of an LLC in each luteal phase was measured separately in the periphery and center of the CL. Five sites were randomly chosen from the pictures of the H-E stained sections among each luteal phase and each luteal site (periphery and center). Five LLCs from each chosen area were measured. The cross-sectional area of an LLC was calculated by assuming that the shape of the cell was an ellipse (S=πab, a=major axis, b= minor axis). Quantification of vwf-positive blood vessels Cells positive for vwf were identified as vascular endothelial cells. A separated block of several endothelial cells was defined as one blood vessel. The number of blood vessels was counted within a unit area (40,000 μm 2 ) in the microscopic field at 200 as described in a previous study [15]. Counting was performed on five randomly chosen areas within the periphery of the CL and in the center of the CL. Quantification was performed independently by three observers. The mean of the three observers data was calculated for each histological section, and the mean of the three observers related means was used as a single observation. Quantification of arteriolovenous vessels In counting of arteriolovenous vessels, arteriolovenous vessels were identified as SMA-positive vascular smooth muscle cells. A vascular lumen that was perfectly covered with a smooth muscle layer was defined as one arteriolovenous vessel. Moreover, the diameter of an arteriolovenous vessel was defined as being from 20 to 50 μm. The number of SMA-positive blood vessels was counted within a unit area (40,000 μm 2 ) in the microscopic field at 200 as described in a previous study [15]. The counted blood vessels included SMA-positive pericytes and vascular smooth muscle cells. As for vwf-positive vessels, quantification was performed independently by three observers. The mean of the three observers data was calculated for each histological section, and the mean of the three observers related means was used as a single observation. Calculation of the number of capillaries The number of capillaries was computed by subtracting the number of SMA-positive blood vessels from the number of vwfpositive blood vessels. The calculation was performed within each of five different fields, and the mean of the five different fields was
3 126 SHIRASUNA et al. used as a single observation. Percentage area of enos immunostaining The positive staining areas were extracted using PopImaging (Digital Being Kids, Ver. 3.01, Japan) to calculate the percentage area of immunostaining (area of the immunostaining divided by the total area measured 200) as previously described [15, 21]. Areas were analyzed at 200 magnification using one section from each animal and five fields per section. The results were expressed as percentage means ± SEM per unit area. Statistical analysis All data are presented as means ± SEM. Means of the weight of the CL, number of luteal vessels and percentage of immunohistochemical staining areas during the luteal phase were analyzed by ANOVA followed by Bonferroni s multiple comparison test. Probabilities less than 5% (P<0.05) were considered to be significant. Results Weight of the CL during the luteal phase The weight of the CL in the mid (7.9 ± 0.3 g) and late luteal phase (7.7 ± 0.8 g) was about four times heavier than in the early luteal phase (2.1 ± 0.4 g; P<0.05, Fig. 1A). The weight of the CL in the regressing luteal phase (3.9 ± 0.3 g) was half of that of the mid and late luteal phase (P<0.05, Fig. 1A). Change of the cross-sectional area of the LLC In the periphery of the CL, the cross-sectional area of the LLC increased gradually from the early to late luteal phase (Early, ± 2.4 μm 2 ; Mid, ± 15.2 μm 2 ; Late, ± 37.1 μm 2 ; P<0.05, Fig. 1B). In the regressing luteal phase in the periphery of the CL, the cross-sectional area of the LLC was half as large as that in the late luteal phase. In the center of the CL, the cross-sectional area of the LLC in the mid luteal phase was about four times larger than that of the early luteal phase (Early, ± 17.7 μm 2 ; Mid, ± 39.4 μm 2 ; P<0.05, Fig. 1B). Thereafter, the size of cross-sectional area maintained in the late luteal phase decreased by half in the regressing luteal phase compared with the late luteal phase (Late, ± 27.7 μm 2 ; Regressing, ± 43.7 μm 2 ). Comparing the periphery with the center of the CL, the cross-sectional area of the LLC in the center of the mid CL was larger than that in the periphery of the mid CL (P<0.05). Distribution of the arteriolovenous and capillary vessels during the luteal phase Figure 2A shows the changes of the arteriolovenous vessels throughout the luteal phase. In the periphery of the CL, the number of arteriolovenous vessels increased during the early to mid luteal phase (P<0.05) and thereafter decreased in the late luteal phase (P<0.05). During the regressing luteal phase, the number of arteriolovenous vessels increased again (P<0.05). In the center of the CL, the number of arteriolovenous vessels was maintained from the early to the mid luteal phase, and thereafter, decreased in the late luteal phase. However, the number of arteriolovenous vessels in Fig. 1. Changes of the weight of the CL and the cross-sectional area of the large luteal cells throughout the luteal phase. Fig. 1A indicates the changes of the weight of the CL, and Fig. 1B indicates the changes of the cross-sectional area of the large luteal cells throughout the luteal phase. E: Early luteal phase (Days 2 5); M: Mid luteal phase (Days 8 12); L: Late luteal phase (Days 15 17); R: Regressing luteal phase (Days 18 21). In Fig. 1B, black diamonds indicate the periphery of the CL, and white squares indicate the center of the CL. Means ± SEM are presented (n=5). Different superscripts indicate statistically different values (P<0.05). the regressing luteal phase was double that of the late luteal phase (P<0.05). There were many arteriolovenous vessels distributed in the periphery of the CL compared with the center during the mid to regressing luteal phase. Figure 2C-2H shows the immunohistochemical staining for SMA in the periphery and center of the CL. Figure 2B shows the changes of the capillary vessels throughout the luteal phase. In the periphery of the CL, capillaries decreased during the early to mid luteal phase (P<0.05). Thereafter, they increased three-fold during late luteal phase (P<0.05) and decreased again in the regressing luteal phase (P<0.05). In the center of the CL, capillaries increased during the early to mid luteal phase (P<0.05). Thereafter, the number of capillaries were maintained until the late luteal phase and rapidly decreased in the regressing luteal phase (P<0.05). There were many capillaries distributed in the center of the CL compared with the periphery during the mid to late luteal phase. Figure 2I-N shows the immunohistochemical staining for vwf in the periphery and center of the CL, in which vwf was detected during the early to late luteal phase, although it decreased drastically in the regressing luteal phase. In addition, capillary vessels in the center were detected more during the mid and late luteal phase than in the periphery of the CL.
4 VASCULATURE AND enos IN THE CORPUS LUTEUM 127 Fig. 2. Changes of arteriolovenous and capillary vessels in the periphery and the center of the CL throughout the luteal phase. Fig. 2A indicates the changes of the number of arteriolovenous vessels, and Fig. 2B indicates the changes of the capillary vessels in the periphery and center of the CL throughout the luteal phase. In each figure, the black bars indicate the periphery of the CL, and the gray bars indicate the center of the CL. Different superscripts indicate statistically different values (P<0.05). Fig. 2C-H show the immunohistochemical staining of SMA in the periphery and center of the CL during the early luteal phase (C and F), mid luteal phase (D and G) and regressing luteal phase (E and H). Fig. 2I-N show the immunohistochemical staining of vwf in the periphery and center of the CL during the early luteal phase (I and L), mid luteal phase (J and M) and regressing luteal phase (K and N). Scale bars: 50 μm. Distribution of enos during the luteal phase Figure 3A shows the changes in the rate of the enos-positive stained areas throughout the luteal phase. In the periphery of the CL, expression of enos was higher in the early and mid CL and thereafter decreased in the late and regressing luteal phases compared with that in the early and mid luteal phases (P<0.05). In the center of the CL, high expression of enos was detected in the center of the early luteal phase, thereafter decreased during the mid luteal phase (P<0.05) and then was maintained at a stable level until the regressing luteal phase. Although the expression levels of enos were the same in the periphery and center of the early CL, there was more enos distributed in the periphery of the CL compared with the center during the mid to the regressing luteal phase. Figure 3B F shows the immunohistochemical staining for enos in the periphery and center of the CL. Discussion Consistent with the lack of effect of PGF 2α on luteal blood flow in the early CL, PGF 2α did not increase enos expression in the early CL. In contrast, PGF 2α acutely stimulated the expression of enos in the periphery, but not in the center of the mid CL 30 min after PGF 2α administration. Moreover, PGF 2α induces an acute increase in NO in the ovarian circulation [22]. We considered that there were several possibilities concerning why PGF 2α can induce luteal blood flow increase only in the periphery of the mid CL. 1) In the early CL, the number of arteriolovenous vessels was fewer than in the mid CL, and the vessels were distributed equally in the periphery and center of the CL. On the other hand, in the matured CL, more arteriolovenous vessels were presented in the periphery compared with the center of the CL. 2) In the present spontaneous CLs, enos was localized with higher intensity in the periphery than in the center in the matured CLs. However, in the early CLs, the number of arteriolovenous vessels was fewer than in the mid CLs, and the vessels were distributed equally in the periphery and center of the CL. 3) The early CL produces a higher amount of intraluteal PGF 2α than the mid CL, suggesting that desensitization of exogenous PGF 2α occurs in the early CL [23, 24]. These changes in the distribution of the vasculature and enos expression may be one of the reasons why PGF 2α becomes capable of inducing one of the signs of early luteolytic events (an increase in luteal blood flow in the mid luteal phase but not in the early luteal phase), probably by stimulating enos expression in the arteriolovenous vessels. Therefore, a heterogeneous vascular structure may be directly related to luteolytic ability, and PGF 2α may have a siterestricted action depending on not only the luteal phase but also on
5 128 SHIRASUNA et al. Fig. 3. Changes of the enos-positive stained areas and distribution in the periphery and the center of the CL throughout the luteal phase. Fig. 3A indicates the changes of the enos-positive stained areas in the periphery and center of the CL. In the figure, the black bars indicate the periphery of the CL, and the gray bars indicate the center of the CL. Different superscripts indicate statistically different values (P<0.05). Fig. 3B F show the immunohistochemical staining of enos in the periphery and center of the CL during the early luteal phase (B and E), mid luteal phase (C and F) and late luteal phase (D). Fig. 3G shows the negative control. Scale bars: 50 μm. the region in the CL. A rich capillary network is established in the center of the matured CL. In the present study, the number of capillaries increased in the center of the CL but not in the periphery from the early to mid luteal phase. Behind the center of the CL, the number of capillaries in the periphery of the CL increased in the late luteal phase. These differences in timing of the start of increase in cell numbers may be due to heterogeneity in the mitosis of luteal endothelial cells [25]. In fact, from the early to mid luteal phase, the capillary network increases as a result of conspicuous proliferation of endothelial cells [25]. Taken together, the developmental speed of the CL and the timing of angiogenesis within the CL may be regulated differently in the periphery and center regions. The capillary network appears to grow earlier and faster in the center of the CL, followed by the periphery of the CL. In the present study, as synchronized with capillary growth, the cross-sectional area of the large luteal cell also increased faster in the center compared with the periphery of the mid CL. Thereafter, in the late luteal phase, the cross-sectional area of the large luteal cell in the periphery of the CL increased to the same level as that of the center. These findings suggest that the growth of luteal cells may depend on capillary growth because a fully developed capillary network is necessary for supply of nutrition to support the growth of luteal cells. Thus, it is highly possible that the speed of the constructive development of the bovine CL is different between the periphery and center of the CL and is received in part of the different regulation. The bovine CL has two types of blood vessels, arteriolovenous vessels and capillary vessels. Interestingly, although arteriolovenous vessels increased both in the periphery and center of the CL from the late to regressing luteal phase, the capillary vessels drastically decreased. Indeed, previous reports have indicated that non-capillary vessels having smooth muscle layers increases markedly in the regressing CL compared with the late CL in the cow [8, 26]. During PGF 2α-induced luteolysis in the sheep, the endothelial component of the vascular bed decreases, whereas high numbers of smooth muscle cells are maintained during luteal regression, suggesting that pericytes and vascular smooth muscle cells have a role in luteolysis [27]. Therefore, capillary vessels seem to regress rapidly in the regressing CL due to shut-off of blood supply by vasoconstriction of arterioles due to factors such as EDN1 and Ang II. On the other hand, large arteriolovenous vessels remained intact and exhibited a thickening wall in the regressing CL, suggesting that arteriolovenous vessels may have a role in the expeditious disrupture of the bovine CL. Further studies will be needed to clarify the physiological meaning for the increase in arteriolovenous blood vessels in the regressing CL. It is well considered that spontaneous luteolysis induced by endogenous PGF 2α and pharmacological luteolysis induced by administration of exogenous PGF 2α are different [28 30]. In fact, Skarzynski et al. recently reported that estrus synchronization, depending on the method and PGF 2α analogue, may induce lower P production and decrease the sensitivity of the CL to luteotropic factors compared with those in the spontaneous estrous cycle in the cow [30]. During PGF 2α-induced luteolysis in cattle, systemic PGF 2α treatment induces a transient increase in luteal blood flow [12, 15]. Additionally, during spontaneous CL regression in the cow, an acute increase in luteal blood flow in the periphery of the mature CL is correlated with pulsatile increases in plasma PGFM (a metabolite of PGF 2α produced by the uterus) [15, 31, 32]. A recent report by Ginther et al. provided additional evidence showing that CL blood flow increases with each PGFM pulse during spontaneous luteolysis in cattle [32], but not in mares [33]. Although the luteolytic phenomena observed in spontaneous (endogenous) and PGF 2α-induced (exogenous) luteolysis are not completely the same because excessive PGF 2α doses may stimulate nonphysiologic responses [29], these observations indicate that the phenomenon of an increase in luteal blood flow and its mechanisms may be similar in spontaneous and PGF 2α-induced luteolysis in the cow. In the present study, a high expression of enos was detected in the center of the early CL, although it decreased in the mid, late and regressing luteal phases, confirming previous studies [34, 35]. It has been reported that growth and development of new capillaries occurs in the early CL [36]. In addition, NO has been reported to
6 VASCULATURE AND enos IN THE CORPUS LUTEUM 129 promote angiogenesis in human vascular endothelial cells [37]. Also, a previous study has suggested that enos has a cooperative effect for angiogenesis with VEGF, a potent angiogenic factor [38]. Thus, luteal NO may act as a luteotropic rather than luteolytic factor in the early CL. On the other hand, it has been reported that an NO donor inhibits progesterone secretion [17] and induces apoptosis [39] of mid luteal cells. These results suggest that luteal NO has dual functions according to the luteal phase. Shear stress is generated when the blood flow volume and blood pressure are increased [40]. Shear stress is the frictional tangential force imposed on the vessel wall when blood flows through the vessel. Shear stress modulates many physiological processes and is a potent stimulator for NO production and vasodilation [41]. In fact, NO production is elevated by shear stress in a graded fashion in ovine fetoplacental artery endothelial cells [42, 43]. The acute burst of NO production may relate to an increase in enos activity, probably via an increase in intracellular Ca 2+ and phosphorylation of enos [43]. It is possible that luteolytic pulses of PGF 2α stimulate enos activity by inducing intracellular Ca 2+ and simultaneously increasing luteal blood flow/shear stress in the periphery of the CL. Although we speculated that luteal blood flow and shear stress generated by NO action may have the potential to regulate luteolysis by stimulating other potential luteolytic mediators, such as EDN1 in the cow [15], precise evaluation of the physiological impact of the blood flow increase requires further investigation. In conclusion, the results indicated that the expression pattern of luteal vasculatures and enos was different during the luteal phase in the cow as follows: (1) more arteriolovenous vessels existed in the periphery compared with the center of the mid, late and regressing CL; (2) a larger number of capillaries existed in the center compared with the periphery of the mid and late CL; (3) many enos-positive areas were observed in the periphery of the mid, late and regressing CL, where many arteriolovenous vessels were observed; and (4) in the early CL, both the periphery and center of the CL contained similar distributions of capillaries, arteriolovenous vessels and enos expression. Thus, this structural change from the early (homogeneous) to mid (heterogeneous) luteal phase is related to the difference in the CL response of blood flow increase due to PGF 2α, which is only observed in the mature CL. Acknowledgments This study was supported by a Grant-in-Aid for Scientific Research of the Japan Society for the Promotion of Science (JSPS) and the Global COE Program, Ministry of Education, Culture, Sports, Science and Technology, Japan. KS was supported by JSPS Research Fellowships for Young Scientists. References 1. O Shea JD, Rodgers RJ, D Occhio MJ. Cellular composition of the cyclic corpus luteum of the cow. J Reprod Fertil 1989; 85: Lei ZM, Chegini N, Rao CV. Quantitative cell composition of human and bovine corpora lutea from various reproductive states. Biol Reprod 1991; 44: Girsh E, Milvae RA, Wang W, Meidan R. Effect of endothelin-1 on bovine luteal cell function: role in prostaglandin F2α-induced antisteroidogenic action. Endocrinology 1996; 137: Miyamoto A, Kobayashi S, Arata S, Ohtani M, Fukui Y, Schams D. Prostaglandin F2α promotes the inhibitory action of endothelin-1 on the bovine luteal function in vitro. J Endocrinol 1997; 152: R Hayashi K, Miyamoto A. Angiotensin II interacts with prostaglandin F2α and endothelin-1 as a local luteolytic factor in the bovine corpus luteum in vitro. Biol Reprod 1999; 60: Kashiwagi S, Kajimura M, Yoshimura Y, Suematsu M. Nonendothelial source of nitric oxide in arterioles but not in venules: alternative source revealed in vivo by diaminofluorescein microfluorography. Circ Res 2002; 91: e Chiba Y, Hiramatsu O, Yada T, Nakamoto H, Toyota E, Ebata J, Ogasawara Y, Tsujioka K, Kajiya F. Analysis of the human sublingual microvascular responses after nitroglycerin by a needle-probe CCD microscope. IEICE Technical Report 1995; 95: Bauer M, Schilling N, Spanel-Borowski K. Development and regression of non-capillary vessels in the bovine corpus luteum. Cell Tissue Res 2003; 311: Sugino N, Suzuki T, Sakata A, Miwa I, Asada H, Taketani T, Yamagata Y, Tamura H. Angiogenesis in the human corpus luteum: changes in expression of angiopoietins in the corpus luteum throughout the menstrual cycle and in early pregnancy. J Clin Endocrinol Metab 2005; 90: Amsterdam A, Lindner HR, Groschel-Stewart U. Localization of actin and myosin in the rat oocyte and follicular wall by immunofluorescence. Anat Rec 1977; 187: Wiltbank MC, Dysko RC, Gallagher KP, Keyes PL. Relationship between blood flow and steroidogenesis in the rabbit corpus luteum. J Reprod Fertil 1988; 84: Acosta TJ, Yoshizawa N, Ohtani M, Miyamoto A. Local changes in blood flow within the early and midcycle corpus luteum after prostaglandin F2α injection in the cow. Biol Reprod 2002; 66: Miyamoto A, Shirasuna K, Hayashi KG, Kamada D, Awashima C, Kaneko E, Acosta TJ, Matsui M. A potential use of color ultrasound as a tool for reproductive management: New observations using color ultrasound scanning that were not possible with imaging only in black and white. J Reprod Dev 2006; 52: Niswender GD, Reimers TJ, Diekman MA, Nett TM. Blood flow: a mediator of ovarian function. Biol Reprod 1976; 14: Shirasuna K, Watanabe S, Asahi T, Wijagunawardane MPB, Sasahara K, Jiang C, Matsui M, Sasaki M, Shimizu T, Davis JS, Miyamoto A. Prostaglandin F2α increases endothelial nitric oxide synthase in the periphery of the bovine corpus luteum: the possible regulation of blood flow at an early stage of luteolysis. Reproduction 2008; 135: Skarzynski DJ, Jaroszewski JJ, Bah MM, Deptula KM, Barszczewska B, Gawronska B, Hansel W. Administration of a nitric oxide synthase inhibitor counteracts prostaglandin F2α-induced luteolysis in cattle. Biol Reprod 2003; 68: Skarzynski DJ, Okuda K. Different actions of noradrenaline and nitric oxide on the output of prostaglandins and progesterone in cultured bovine luteal cells. Prostaglandins Other Lipid Mediat 2000; 60: Miyamoto Y, Skarzynski DJ, Okuda K. Is tumor necrosis factor alpha a trigger for the initiation of endometrial prostaglandin F2α release at luteolysis in cattle? Biol Reprod 2000; 62: Hsu SM, Raine L, Fanger H. Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem 1981; 29: Shirasuna K, Shimizu T, Sayama K, Asahi T, Sasaki M, Berisha B, Schams D, Miyamoto A. Expression and localization of apelin and its receptor APJ in the bovine corpus luteum during the estrous cycle and prostaglandin F2α-induced luteolysis. Reproduction 2008; 135: Al-zi abi MO, Watson ED, Fraser HM. Angiogenesis and vascular endothelial growth factor expression in the equine corpus luteum. Reproduction 2003; 125: Acosta TJ, Bah MB, Korzekwa A, Woclawek-Potocka I, Markiewicz W, Jaroszewski JJ, Okuda K, Skarzynski DJ. Acute changes in circulating concentrations of progesterone and nitric oxide and partial pressure of oxygen during prostaglandin F2αinduced luteolysis in cattle. J Reprod Dev 2009; 55: Skarzynski DJ, Okuda K. Sensitivity of bovine corpora lutea to prostaglandin F2α is dependent on progesterone, oxytocin, and prostaglandins. Biol Reprod 1999; 60: Skarzynski DJ, Ferreira-Dias G, Okuda K. Regulation of luteal function and corpus luteum regression in cows: hormonal control, immune mechanisms and intercellular communication. Reprod Domest Anim 2008; 43 (Suppl 2): Zheng J, Redmer DA, Reynolds LP. Vascular development and heparin-binding growth factors in the bovine corpus luteum at several stages of the estrous cycle. Biol Reprod 1993; 49: Hojo T, Al-Zi abi MO, Skarzynski DJ, Acosta TJ, Okuda K. Changes in the vasculature of bovine corpus luteum during the estrous cycle and prostaglandin F2α-induced luteolysis. J Reprod Dev 2009; 55:
7 130 SHIRASUNA et al. 27. Vonnahme KA, Redmer DA, Borowczyk E, Bilski JJ, Luther JS, Johnson ML, Reynolds LP, Grazul-Bilska AT. Vascular composition, apoptosis, and expression of angiogenic factors in the corpus luteum during prostaglandin F2α-induced regression in sheep. Reproduction 2006; 131: Skarzynski DJ, Bogacki M, Kotwica J. Changes in ovarian oxytocin secretion as an indicator of corpus luteum response to prostaglandin F2α treatment in cattle. Theriogenology 1997; 48: Ginther OJ, Araujo RR, Palhao MP, Rodrigues BL, Beg MA. Necessity of sequential pulses of prostaglandin F2α for complete physiologic luteolysis in cattle. Biol Reprod 2009; 80: Skarzynski DJ, Siemieniuch MJ, Pilawski W, Woclawek Potocka I, Bah MM, Majewska M, Jaroszewski JJ. In vitro assessment of progesterone and prostaglandin E2 production by the corpus luteum in cattle following pharmacological synchronization of estrus. J Reprod Dev 2009; 55: Miyamoto A, Shirasuna K, Wijayagunawardane MP, Watanabe S, Hayashi M, Yamamoto D, Matsui M, Acosta TJ. Blood flow: a key regulatory component of corpus luteum function in the cow. Domest Anim Endocrinol 2005; 29: Ginther OJ, Silva LA, Araujo RR, Beg MA. Temporal associations among pulses of 13,14-dihydro-15-keto-PGF2α, luteal blood flow, and luteolysis in cattle. Biol Reprod 2007; 76: Ginther OJ, Gastal EL, Gastal MO, Beg MA. Effect of prostaglandin F2α on ovarian, adrenal, and pituitary hormones and on luteal blood flow in mares. Domest Anim Endocrinol 2007; 32: Rosiansky-Sultan M, Klipper E, Spanel-Borowski K, Meidan R. Inverse relationship between nitric oxide synthases and endothelin-1 synthesis in bovine corpus luteum: interactions at the level of luteal endothelial cell. Endocrinology 2006; 147: Grazul-Bilska AT, Navanukraw C, Johnson ML, Arnold DA, Reynolds LP, Redmer DA. Expression of endothelial nitric oxide synthase in the ovine ovary throughout the estrous cycle. Reproduction 2006; 132: Cavender JL, Murdoch WJ. Morphological studies of the microcirculatory system of periovulatory ovine follicles. Biol Reprod 1988; 39: Pan YM, Yao YZ, Zhu ZH, Sun XT, Qiu YD, Ding YT. Caveolin-1 is important for nitric oxide-mediated angiogenesis in fibrin gels with human umbilical vein endothelial cells. Acta Pharmacol Sin 2006; 27: Pan JW, Zhan RY, Tong Y, Zhou YQ, Zhang M. Expression of endothelial nitric oxide synthase and vascular endothelial growth factor in association with neovascularization in human primary astrocytoma. J Zhejiang Univ Sci B 2005; 6: Korzekwa AJ, Okuda K, Woclawek-Potocka I, Murakami S, Skarzynski DJ. Nitric oxide induces apoptosis in bovine luteal cells. J Reprod Dev 2006; 52: Lie M, Sejersted OM, Kiil F. Local regulation of vascular cross section during changes in femoral arterial blood flow in dogs. Circ Res 1970; 27: Davies PF. Flow-mediated endothelial mechanotransduction. Physiol Rev 1995; 75: Li Y, Zheng J, Bird IM, Magness RR. Effects of pulsatile shear stress on nitric oxide production and endothelial cell nitric oxide synthase expression by ovine fetoplacental artery endothelial cells. Biol Reprod 2003; 69: Li Y, Zheng J, Bird IM, Magness RR. Mechanisms of shear stress-induced endothelial nitric-oxide synthase phosphorylation and expression in ovine fetoplacental artery endothelial cells. Biol Reprod 2004; 70:
Effect of the Dominant Follicle Aspiration before or after Luteinizing Hormone Surge on the Corpus Luteum Formation in the Cow
 Journal of Reproduction and Development, Vol. 52, No. 1, 2006 Research Note Effect of the Dominant Follicle Aspiration before or after Luteinizing Hormone Surge on the Corpus Luteum Formation in the Cow
Journal of Reproduction and Development, Vol. 52, No. 1, 2006 Research Note Effect of the Dominant Follicle Aspiration before or after Luteinizing Hormone Surge on the Corpus Luteum Formation in the Cow
Treatment 3 Days After Ovulation In Mares
 Luteal Regression And Follicle Development Following Prostaglandin-F 2α Treatment 3 Days After Ovulation In Mares D.R. Bergfelt a, R.A. Pierson b, and O.J. Ginther a a University of Wisconsin, Madison,
Luteal Regression And Follicle Development Following Prostaglandin-F 2α Treatment 3 Days After Ovulation In Mares D.R. Bergfelt a, R.A. Pierson b, and O.J. Ginther a a University of Wisconsin, Madison,
JSAR Young Investigator Award. Studies of Follicular Vascularity Associated with Follicle Selection and Ovulation in Cattle
 Journal of Reproduction and Development, Vol. 53, No. 1, 2007 JSAR Young Investigator Award Studies of Follicular Vascularity Associated with Follicle Selection and Ovulation in Cattle Tomas J. ACOSTA
Journal of Reproduction and Development, Vol. 53, No. 1, 2007 JSAR Young Investigator Award Studies of Follicular Vascularity Associated with Follicle Selection and Ovulation in Cattle Tomas J. ACOSTA
Laboratory of Reproductive Endocrinology, Graduate School of Natural Science and Technology, Okayama University, Okayama , Japan
 Journal of Reproduction and Development, Vol. 52, No. 1, 2006 A Potential Use of Color Ultrasound as a Tool for Reproductive Management: New Observations Using Color Ultrasound Scanning that were not Possible
Journal of Reproduction and Development, Vol. 52, No. 1, 2006 A Potential Use of Color Ultrasound as a Tool for Reproductive Management: New Observations Using Color Ultrasound Scanning that were not Possible
GENERAL SUMMARY Corpus luteum is a transient endocrine structure formed from the ruptured ovarian follicle. Its main function is to secrete P 4, a pro
 Corpus luteum is a transient endocrine structure formed from the ruptured ovarian follicle. Its main function is to secrete P 4, a pro-gestational hormone, essential for establishment and maintenance of
Corpus luteum is a transient endocrine structure formed from the ruptured ovarian follicle. Its main function is to secrete P 4, a pro-gestational hormone, essential for establishment and maintenance of
Reduced anti-müllerian hormone (AMH) in mares with hemorrhagic anovulatory follicles
 336 Reduced anti-müllerian hormone (AMH) in mares with hemorrhagic anovulatory follicles Gharagozlou, F. 1 ; Akbarinejad, V. 2* ; Youssefi, R. 1, 3 ; Masoudifard, M. 4 and Hasani, N. 1 1 Department of
336 Reduced anti-müllerian hormone (AMH) in mares with hemorrhagic anovulatory follicles Gharagozlou, F. 1 ; Akbarinejad, V. 2* ; Youssefi, R. 1, 3 ; Masoudifard, M. 4 and Hasani, N. 1 1 Department of
Effect of intraluteal injection o. luteolysis in the cow. Watanabe, Sho, Shirasuna, Koumei, Motozumi, Yamamoto, Dai, Berisha, Author(s)
 ' ' Title type Effect of intraluteal injection o A receptor antagonist on PGF luteolysis in the cow. Watanabe, Sho, Shirasuna, Koumei, Motozumi, Yamamoto, Dai, Berisha, Author(s) Schams, Dieter, Miyamoto,
' ' Title type Effect of intraluteal injection o A receptor antagonist on PGF luteolysis in the cow. Watanabe, Sho, Shirasuna, Koumei, Motozumi, Yamamoto, Dai, Berisha, Author(s) Schams, Dieter, Miyamoto,
Synchronization of Ovulation and Fixed-Time Insemination for Improvement of Conception Rate in Dairy Herds with Poor Estrus Detection Efficiency
 Journal of Reproduction and Development, Vol. 45, No. 1, 1999 Synchronization of Ovulation and Fixed-Time Insemination for Improvement of Conception Rate in Dairy Herds with Poor Estrus Detection Efficiency
Journal of Reproduction and Development, Vol. 45, No. 1, 1999 Synchronization of Ovulation and Fixed-Time Insemination for Improvement of Conception Rate in Dairy Herds with Poor Estrus Detection Efficiency
Animal Reproduction. Reproductive Cyclicity. # lectures for cumulative test # 02 book 12. Reproductive cyclicity: terminology and basic concepts
 Animal Reproduction JP Advis DVM, Ph.D. Bartlett Hall, Animal Sciences, Cook, (732) 932-9240, advis@aesop.rutgers.edu 14 Course website: rci.rutgers.edu/~advis Material to be covered: About lecture Meetings
Animal Reproduction JP Advis DVM, Ph.D. Bartlett Hall, Animal Sciences, Cook, (732) 932-9240, advis@aesop.rutgers.edu 14 Course website: rci.rutgers.edu/~advis Material to be covered: About lecture Meetings
Journal of Dairy Science
 Effects of d-cloprostenol dose and corpus luteum age on ovulation, luteal function and morphology in non-lactating dairy cows with early corpora lutea Journal: Manuscript ID: JDS-11-5284 Article Type:
Effects of d-cloprostenol dose and corpus luteum age on ovulation, luteal function and morphology in non-lactating dairy cows with early corpora lutea Journal: Manuscript ID: JDS-11-5284 Article Type:
Dr. Julio Giordano. Ovulation. Follicle. Corpus Luteum. GnRH
 Dr. Julio Giordano Follicle Corpus Luteum LH FSH E2 Hypothalamic hormones Gonadotropin releasing hormone () Hormone Concentration CL LH (ng/ml) 12 10 8 6 4 2 LH Response Cows Treated with 28 h (22-36)
Dr. Julio Giordano Follicle Corpus Luteum LH FSH E2 Hypothalamic hormones Gonadotropin releasing hormone () Hormone Concentration CL LH (ng/ml) 12 10 8 6 4 2 LH Response Cows Treated with 28 h (22-36)
REPRODUCTION & GENETICS. Hormones
 REPRODUCTION & GENETICS Hormones http://www.youtube.com/watch?v=np0wfu_mgzo Objectives 2 Define what hormones are; Compare and contrast the male and female hormones; Explain what each hormone in the mail
REPRODUCTION & GENETICS Hormones http://www.youtube.com/watch?v=np0wfu_mgzo Objectives 2 Define what hormones are; Compare and contrast the male and female hormones; Explain what each hormone in the mail
Why Cycle Control?" Manipulating Ovulation and Estrous Synchronization" Manipulating Ovulation" Cattle" Principle of PGF 2α Use"
 Why Cycle Control?" Manipulating Ovulation and Estrous Synchronization" John Parrish 1. Group females for parturition: " a) Decrease labor, calving period Reduce calving season" b) More uniform weaning
Why Cycle Control?" Manipulating Ovulation and Estrous Synchronization" John Parrish 1. Group females for parturition: " a) Decrease labor, calving period Reduce calving season" b) More uniform weaning
Influence of large follicles on oestrus induction and ovulation after embryo collection in superovulated Japanese Black cows
 J. Reprod. Engineer. 2015; 17: 1 5. http://sreprod.jp/contents.htm = Original Article = Journal of REPRODUCTION ENGINEERING Influence of large follicles on oestrus induction and ovulation after embryo
J. Reprod. Engineer. 2015; 17: 1 5. http://sreprod.jp/contents.htm = Original Article = Journal of REPRODUCTION ENGINEERING Influence of large follicles on oestrus induction and ovulation after embryo
Review Article Cytokines and Angiogenesis in the Corpus Luteum
 Mediators of Inflammation Volume 2013, Article ID 420186, 11 pages http://dx.doi.org/10.1155/2013/420186 Review Article Cytokines and Angiogenesis in the Corpus Luteum António M. Galvão, 1,2 Graça Ferreira-Dias,
Mediators of Inflammation Volume 2013, Article ID 420186, 11 pages http://dx.doi.org/10.1155/2013/420186 Review Article Cytokines and Angiogenesis in the Corpus Luteum António M. Galvão, 1,2 Graça Ferreira-Dias,
Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle?
 Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle? Introduction: The menstrual cycle (changes within the uterus) is an approximately 28-day cycle that
Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle? Introduction: The menstrual cycle (changes within the uterus) is an approximately 28-day cycle that
LH (Bovine) ELISA Kit
 LH (Bovine) ELISA Kit Catalog Number KA2280 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
LH (Bovine) ELISA Kit Catalog Number KA2280 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
REVIEW Possibility of Diagnosing Uterine Function in Cows
 JARQ 50 (2), 115-119 (2016) http://www.jircas.affrc.go.jp REVIEW Possibility of Diagnosing Uterine Function in Cows Kosuke IGA 1 *, Naoki TAKENOUCHI 2, Manabu SHIMIZU 1 and Yuji HIRAO 3 1 Livestock and
JARQ 50 (2), 115-119 (2016) http://www.jircas.affrc.go.jp REVIEW Possibility of Diagnosing Uterine Function in Cows Kosuke IGA 1 *, Naoki TAKENOUCHI 2, Manabu SHIMIZU 1 and Yuji HIRAO 3 1 Livestock and
Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to Equine Chorionic Gonadotropin
 Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to quine Chorionic Gonadotropin S. L. Pulley, L. D. Wallace, H. I. Mellieon, and J. S. Stevenson
Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to quine Chorionic Gonadotropin S. L. Pulley, L. D. Wallace, H. I. Mellieon, and J. S. Stevenson
Abstracts for the KSAR and JSAR Joint Symposium. Fertility control in female domestic animals: From basic understanding to application
 Abstracts for the KSAR and JSAR Joint Symposium Fertility control in female domestic animals: From basic understanding to application Current Research Orientation in Livestock Reproduction in Korea Choong-Saeng
Abstracts for the KSAR and JSAR Joint Symposium Fertility control in female domestic animals: From basic understanding to application Current Research Orientation in Livestock Reproduction in Korea Choong-Saeng
Cell Localization of ACTH, Dopamine, and GnRH Receptors and PPARγ in Bovine Corpora Lutea during Diestrus
 Acta Scientiae Veterinariae, 203. 4: 29. RESEARCH ARTICLE Pub. 29 ISSN 679-926 Cell Localization of ACTH, Dopamine, and GnRH Receptors and PPARγ in Bovine Corpora Lutea during Diestrus Francesco Parillo,
Acta Scientiae Veterinariae, 203. 4: 29. RESEARCH ARTICLE Pub. 29 ISSN 679-926 Cell Localization of ACTH, Dopamine, and GnRH Receptors and PPARγ in Bovine Corpora Lutea during Diestrus Francesco Parillo,
Histological analysis of arteriovenous anastomosis-like vessels established in the corpus luteum of cows during luteolysis
 Nio-Kobayashi et al. Journal of Ovarian Research (2016) 9:67 DOI 10.1186/s13048-016-0277-0 RESEARCH Open Access Histological analysis of arteriovenous anastomosis-like vessels established in the corpus
Nio-Kobayashi et al. Journal of Ovarian Research (2016) 9:67 DOI 10.1186/s13048-016-0277-0 RESEARCH Open Access Histological analysis of arteriovenous anastomosis-like vessels established in the corpus
Page 1. A wide variety of ovarian abnormalities are encountered in clinical practice
 A wide variety of ovarian abnormalities are encountered in clinical practice Common Problems Anovulatory follicles Persistent anovulatory follicles Hemorrhagic/Luteinized follicles Persistent corpus luteum
A wide variety of ovarian abnormalities are encountered in clinical practice Common Problems Anovulatory follicles Persistent anovulatory follicles Hemorrhagic/Luteinized follicles Persistent corpus luteum
Anti-Apoptotic Roles of Prostaglandin E2 and F2alpha in Bovine Luteal Steroidogenic Cells 1
 BIOLOGY OF REPRODUCTION 79, 310 317 (2008) Published online before print 7 May 2008. DOI 10.1095/biolreprod.107.066084 Anti-Apoptotic Roles of Prostaglandin E2 and F2alpha in Bovine Luteal Steroidogenic
BIOLOGY OF REPRODUCTION 79, 310 317 (2008) Published online before print 7 May 2008. DOI 10.1095/biolreprod.107.066084 Anti-Apoptotic Roles of Prostaglandin E2 and F2alpha in Bovine Luteal Steroidogenic
REGULATION OF MICRO-ENVIRONMENT WITHIN THE OVIDUCT
 REGULATION OF MICRO-ENVIRONMENT WITHIN THE OVIDUCT September, 2016 Yoshihiko KOBAYASHI Graduate School of Environmental and Life Science (Doctor s Course) OKAYAMA UNIVERSITY. CONTENTS PREFACE -------------------------------------------------------------------------------------------------
REGULATION OF MICRO-ENVIRONMENT WITHIN THE OVIDUCT September, 2016 Yoshihiko KOBAYASHI Graduate School of Environmental and Life Science (Doctor s Course) OKAYAMA UNIVERSITY. CONTENTS PREFACE -------------------------------------------------------------------------------------------------
CLARITY reveals dynamics of ovarian follicular architecture and vasculature in three-dimensions
 CLARITY reveals dynamics of ovarian follicular architecture and vasculature in three-dimensions Yi Feng, Peng Cui, Xiaowei Lu, Brian Hsueh, Fredrik Möller Billig, Livia Zarnescu Yanez, Raju Tomer, Derek
CLARITY reveals dynamics of ovarian follicular architecture and vasculature in three-dimensions Yi Feng, Peng Cui, Xiaowei Lu, Brian Hsueh, Fredrik Möller Billig, Livia Zarnescu Yanez, Raju Tomer, Derek
Why Cycle Control? Manipulating Ovulation and Estrous Synchronization. Manipulating Ovulation. Cattle. Principle of PGF 2a Use
 Why Cycle Control? Manipulating and Estrous Synchronization John Parrish 1. Group females for parturition: a) Decrease labor, calving period Reduce calving season b) More uniform weaning weights. 2. Reduce
Why Cycle Control? Manipulating and Estrous Synchronization John Parrish 1. Group females for parturition: a) Decrease labor, calving period Reduce calving season b) More uniform weaning weights. 2. Reduce
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion
 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion A.S. Leaflet R1362 Acacia A. Alcivar, graduate research assistant,
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion A.S. Leaflet R1362 Acacia A. Alcivar, graduate research assistant,
Evaluation of Ovarian Follicular Health with the Markers of Endothelial and Granulosa Cells
 CMU.J.Nat.Sci.Special Issue on Agricultural & Natural Resources (2012) Vol.11 (1) 461 Evaluation of Ovarian Follicular Health with the Markers of Endothelial and Granulosa Cells Tossapol Moonmanee 1, Chainarong
CMU.J.Nat.Sci.Special Issue on Agricultural & Natural Resources (2012) Vol.11 (1) 461 Evaluation of Ovarian Follicular Health with the Markers of Endothelial and Granulosa Cells Tossapol Moonmanee 1, Chainarong
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion
 Beef Research Report, 1996 Animal Science Research Reports 1997 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion Acacia
Beef Research Report, 1996 Animal Science Research Reports 1997 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion Acacia
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH.
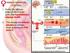 1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
Female Reproductive System. Justin D. Vidal
 Female Reproductive System Justin D. Vidal If you cannot identify the tissue, then it is probably part of the female reproductive system! Introduction The female reproductive system is constantly changing,
Female Reproductive System Justin D. Vidal If you cannot identify the tissue, then it is probably part of the female reproductive system! Introduction The female reproductive system is constantly changing,
CASE 41. What is the pathophysiologic cause of her amenorrhea? Which cells in the ovary secrete estrogen?
 CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
M. Irfan-ur-Rehman Khan, M. A. Rana and N. Ahmad. Department of Theriogenology, University of Veterinary and Animal Sciences, Lahore, Pakistan
 82 ULTRASONIC MONITORING OF FOLLICLES AND CORPORA LUTEA DURING SYNCHRONIZATION IN SUMMER ANOESTROUS NILI RAVI BUFFALOES AND THEIR SUBSEQUENT SUPEROVULATORY RESPONSE M. Irfan-ur-Rehman Khan, M. A. Rana
82 ULTRASONIC MONITORING OF FOLLICLES AND CORPORA LUTEA DURING SYNCHRONIZATION IN SUMMER ANOESTROUS NILI RAVI BUFFALOES AND THEIR SUBSEQUENT SUPEROVULATORY RESPONSE M. Irfan-ur-Rehman Khan, M. A. Rana
Prostaglandin F 2α. J. S. Stevenson, S. L. Pulley, and H. I. Mellieon, Jr.
 Prostaglandin F 2α and GnRH Administration Improved Progesterone tatus, Luteal Number, and Proportion of Ovular and Anovular Dairy Cows with Corpora Lutea efore a Timed Artificial Insemination Program
Prostaglandin F 2α and GnRH Administration Improved Progesterone tatus, Luteal Number, and Proportion of Ovular and Anovular Dairy Cows with Corpora Lutea efore a Timed Artificial Insemination Program
Effects of dbcamp on progesterone synthesis by cultured goat luteal cell subpopulations isolated from early and late luteal stage corpora lutea
 DOI: 10.21451/1984-3143-AR774 Anim. Reprod., Belo Horizonte, v.13, n.2, p.93-99, Apr./Jun. 2016 Effects of dbcamp on progesterone synthesis by cultured goat luteal cell subpopulations isolated from early
DOI: 10.21451/1984-3143-AR774 Anim. Reprod., Belo Horizonte, v.13, n.2, p.93-99, Apr./Jun. 2016 Effects of dbcamp on progesterone synthesis by cultured goat luteal cell subpopulations isolated from early
Proceedings, The Applied Reproductive Strategies in Beef Cattle Workshop, September 5-6, 2002, Manhattan, Kansas
 20 10 0 Proceedings, The Applied Reproductive Strategies in Beef Cattle Workshop, September 5-6, 2002, Manhattan, Kansas REVIEW OF FOLLICULAR GROWTH AND THE BOVINE ESTROUS CYCLE Milo C. Wiltbank Department
20 10 0 Proceedings, The Applied Reproductive Strategies in Beef Cattle Workshop, September 5-6, 2002, Manhattan, Kansas REVIEW OF FOLLICULAR GROWTH AND THE BOVINE ESTROUS CYCLE Milo C. Wiltbank Department
OVARIAN RESPONSES AND CONCEPTION RATES IN RESPONSE TO GnRH, hcg, AND PROGESTERONE 1
 Dairy Research 2006 OVARIAN RESPONSES AND CONCEPTION RATES IN RESPONSE TO GnRH, hcg, AND PROGESTERONE 1 J. S. Stevenson, M. A. Portaluppi, D. E. Tenhouse, A. Lloyd, D. R. Eborn, S. Kacuba 2 and J. M. DeJarnette
Dairy Research 2006 OVARIAN RESPONSES AND CONCEPTION RATES IN RESPONSE TO GnRH, hcg, AND PROGESTERONE 1 J. S. Stevenson, M. A. Portaluppi, D. E. Tenhouse, A. Lloyd, D. R. Eborn, S. Kacuba 2 and J. M. DeJarnette
Reproductive Endocrinology. Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007
 Reproductive Endocrinology Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007 isabelss@hkucc.hku.hk A 3-hormone chain of command controls reproduction with
Reproductive Endocrinology Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007 isabelss@hkucc.hku.hk A 3-hormone chain of command controls reproduction with
are associated with low fertility in dairy cows
 J. Dairy Sci. 95 :2355 2361 http://dx.doi.org/ 10.3168/jds.2011-4325 American Dairy Science Association, 2012. Open access under CC BY-NC-ND license. are associated with low fertility in dairy cows F.
J. Dairy Sci. 95 :2355 2361 http://dx.doi.org/ 10.3168/jds.2011-4325 American Dairy Science Association, 2012. Open access under CC BY-NC-ND license. are associated with low fertility in dairy cows F.
Microvascularization and angiogenic activity of equine corpora lutea throughout the estrous cycle
 Domestic Animal Endocrinology 30 (2006) 247 259 Microvascularization and angiogenic activity of equine corpora lutea throughout the estrous cycle G. Ferreira-Dias a,, P. Pinto Bravo b, L. Mateus a, D.A.
Domestic Animal Endocrinology 30 (2006) 247 259 Microvascularization and angiogenic activity of equine corpora lutea throughout the estrous cycle G. Ferreira-Dias a,, P. Pinto Bravo b, L. Mateus a, D.A.
Available online at Theriogenology xxx (2009) xxx xxx
 Available online at www.sciencedirect.com 1 2 3 4 5 6 7 8 9 10 11 The effect of hormone treatments (hcg and cloprostenol) and season on the incidence of hemorrhagic anovulatory follicles in the mare: A
Available online at www.sciencedirect.com 1 2 3 4 5 6 7 8 9 10 11 The effect of hormone treatments (hcg and cloprostenol) and season on the incidence of hemorrhagic anovulatory follicles in the mare: A
Fixed-Time Artificial Insemination (TAI) in Suckled Beef Cows in Response to Equine Chorionic Gonadotropin (ecg)
 Fixed-Time Artificial Insemination (TAI) in Suckled Beef Cows in Response to Equine Chorionic Gonadotropin (ecg) Guilherme Marquezini 1, Vitor Mercadante 1, Logan Wallace 2, Stacey Pulley 2, KC Olson 2,
Fixed-Time Artificial Insemination (TAI) in Suckled Beef Cows in Response to Equine Chorionic Gonadotropin (ecg) Guilherme Marquezini 1, Vitor Mercadante 1, Logan Wallace 2, Stacey Pulley 2, KC Olson 2,
Embryology Lecture # 4
 1 Quick Review: Oogenesis : - Oogonia start appear in the ovary when the age of the fetus 1 is th (5 week). - Then the Oogonia transformed into 1ry Oocyte. - 1ry Oocyte is surrounded by a follicle (cover).
1 Quick Review: Oogenesis : - Oogonia start appear in the ovary when the age of the fetus 1 is th (5 week). - Then the Oogonia transformed into 1ry Oocyte. - 1ry Oocyte is surrounded by a follicle (cover).
Phytoestrogens and Their Metabolites Inhibit the Sensitivity of the Bovine Corpus Luteum to Luteotropic Factors
 Journal of Reproduction and Development, Vol. 52, No. 1, 2006 Full Paper Phytoestrogens and Their Metabolites Inhibit the Sensitivity of the Bovine Corpus Luteum to Luteotropic Factors Katarzyna K. PIOTROWSKA
Journal of Reproduction and Development, Vol. 52, No. 1, 2006 Full Paper Phytoestrogens and Their Metabolites Inhibit the Sensitivity of the Bovine Corpus Luteum to Luteotropic Factors Katarzyna K. PIOTROWSKA
Ovarian Follicular Development in the Untreated and
 Ovarian Follicular Development in the Untreated and PMSG-treated Cyclic Rat Hajime MIYAMOTO, Goro KATSUURA and Takehiko ISHIBASHI Department of Animal Science, College of Agriculture, Kyoto University,
Ovarian Follicular Development in the Untreated and PMSG-treated Cyclic Rat Hajime MIYAMOTO, Goro KATSUURA and Takehiko ISHIBASHI Department of Animal Science, College of Agriculture, Kyoto University,
Small Ruminant Reproductive Management Workshop
 Small Ruminant Reproductive Management Workshop Animal Nutrition and Physiology Center, North Dakota State University Sponsors: American Sheep and Goat Center, North Dakota State University, University
Small Ruminant Reproductive Management Workshop Animal Nutrition and Physiology Center, North Dakota State University Sponsors: American Sheep and Goat Center, North Dakota State University, University
IGF-1.
 1006 2 *1 1 2 sisaas33@gmail.com.... IGF-1.. - -.. LH LH GnRH.. :.......(1).(2) in vitro 1007..(3) (6) (5) (4).. in vitro. (7)... ) 50. (9) (8) ( 10 (3). (10).(11)...(12).(13) IGF-1. IGF-1..(14).(16).(15)
1006 2 *1 1 2 sisaas33@gmail.com.... IGF-1.. - -.. LH LH GnRH.. :.......(1).(2) in vitro 1007..(3) (6) (5) (4).. in vitro. (7)... ) 50. (9) (8) ( 10 (3). (10).(11)...(12).(13) IGF-1. IGF-1..(14).(16).(15)
Progesterone level does not distinguish the different course of canine ovulation determined by ultrasonography
 Polish Journal of Veterinary Sciences Vol. 17, No. 2 (2014), 293 297 DOI 10.2478/pjvs-2014-0040 Original article Progesterone level does not distinguish the different course of canine ovulation determined
Polish Journal of Veterinary Sciences Vol. 17, No. 2 (2014), 293 297 DOI 10.2478/pjvs-2014-0040 Original article Progesterone level does not distinguish the different course of canine ovulation determined
Manipulation of Ovarian Function for the Reproductive Management of Dairy Cows
 Veterinary Research Communications,28(2004) 111 119 2004 Kluwer Academic Publishers. Printed in the Netherlands Manipulation of Ovarian Function for the Reproductive Management of Dairy Cows W.W. Thatcher1*,
Veterinary Research Communications,28(2004) 111 119 2004 Kluwer Academic Publishers. Printed in the Netherlands Manipulation of Ovarian Function for the Reproductive Management of Dairy Cows W.W. Thatcher1*,
Immunohistochemical Study on the C-cells in the Internal Parathyroid Gland of the Goat
 Immunohistochemical Study on the C-cells in the Internal Parathyroid Gland of the Goat Takeshi TSUCHIYA Department of Animal Morphology, Faculty of Agriculture, Tohoku University, Sendai-Shi 980 (Received
Immunohistochemical Study on the C-cells in the Internal Parathyroid Gland of the Goat Takeshi TSUCHIYA Department of Animal Morphology, Faculty of Agriculture, Tohoku University, Sendai-Shi 980 (Received
Course: Animal Production. Instructor: Ms. Hutchinson. Objectives: After completing this unit of instruction, students will be able to:
 Course: Animal Production Unit Title: Hormones TEKS: 130.3 (C)(6)(A) Instructor: Ms. Hutchinson Objectives: After completing this unit of instruction, students will be able to: A. Define what hormones
Course: Animal Production Unit Title: Hormones TEKS: 130.3 (C)(6)(A) Instructor: Ms. Hutchinson Objectives: After completing this unit of instruction, students will be able to: A. Define what hormones
I. Endocrine System & Hormones Figure 1: Human Endocrine System
 I. Endocrine System & Hormones Figure 1: Human Endocrine System Endocrine System: a) Endocrine glands are ductless since they lack specific vessels for the transport of hormones throughout the body. Instead,
I. Endocrine System & Hormones Figure 1: Human Endocrine System Endocrine System: a) Endocrine glands are ductless since they lack specific vessels for the transport of hormones throughout the body. Instead,
Chapter 14 Reproduction Review Assignment
 Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
Ultrasonographic Observation of Follicular and Luteal Dynamics during the Estrous Cycle in Shiba Goats
 Journal of Reproduction and Development, Vol. 46, No. 1, 2000 Ultrasonographic Observation of Follicular and Luteal Dynamics during the Estrous Cycle in Shiba Goats Jun ORITA, Tomomi TANAKA, Hideo KAMOMAE
Journal of Reproduction and Development, Vol. 46, No. 1, 2000 Ultrasonographic Observation of Follicular and Luteal Dynamics during the Estrous Cycle in Shiba Goats Jun ORITA, Tomomi TANAKA, Hideo KAMOMAE
Chapter 27 The Reproductive System. MDufilho
 Chapter 27 The Reproductive System 1 Figure 27.19 Events of oogenesis. Before birth Meiotic events 2n Oogonium (stem cell) Mitosis Follicle development in ovary Follicle cells Oocyte 2n Primary oocyte
Chapter 27 The Reproductive System 1 Figure 27.19 Events of oogenesis. Before birth Meiotic events 2n Oogonium (stem cell) Mitosis Follicle development in ovary Follicle cells Oocyte 2n Primary oocyte
INDUCTIONS OF SUPEROVULATION USING SEVERAL FSH REGIMENS IN HOLSTEIN-FRIESIAN HEIFERS
 lpn. l. Vet. Res., 33, 45-50 (1985) INDUCTIONS OF SUPEROVULATION USING SEVERAL FSH REGIMENS IN HOLSTEIN-FRIESIAN HEIFERS Yoshiyuki TAKAHASHI and Hiroshi KANAGAWA (Received for publication, February 2,
lpn. l. Vet. Res., 33, 45-50 (1985) INDUCTIONS OF SUPEROVULATION USING SEVERAL FSH REGIMENS IN HOLSTEIN-FRIESIAN HEIFERS Yoshiyuki TAKAHASHI and Hiroshi KANAGAWA (Received for publication, February 2,
Physiology of Male Reproductive System
 Physiology of Male Reproductive System the anterior pituitary gland serves as the primary control of reproductive function at puberty Ant Pituitary secretes FSH & large amounts of LH (ICSH) FSH & LH cause
Physiology of Male Reproductive System the anterior pituitary gland serves as the primary control of reproductive function at puberty Ant Pituitary secretes FSH & large amounts of LH (ICSH) FSH & LH cause
Effects of modified FSH surges on follicle selection and codominance in heifers
 Anim. Reprod., v.2. n.1, p.28-40, Jan./March 2005 Effects of modified FSH surges on follicle selection and codominance in heifers T.J. Acosta 1,2, M.A. Beg 1, O.J. Ginther 1, 3 1 Department of Animal Health
Anim. Reprod., v.2. n.1, p.28-40, Jan./March 2005 Effects of modified FSH surges on follicle selection and codominance in heifers T.J. Acosta 1,2, M.A. Beg 1, O.J. Ginther 1, 3 1 Department of Animal Health
Hai V Vu, Tomas J Acosta *, Shin Yoshioka, Hironori Abe and Kiyoshi Okuda
 Vu et al. Reproductive Biology and Endocrinology 2012, 10:87 RESEARCH Open Access Roles of prostaglandin F2alpha and hydrogen peroxide in the regulation of Copper/Zinc superoxide dismutase in bovine corpus
Vu et al. Reproductive Biology and Endocrinology 2012, 10:87 RESEARCH Open Access Roles of prostaglandin F2alpha and hydrogen peroxide in the regulation of Copper/Zinc superoxide dismutase in bovine corpus
Sow Reproduction and Seasonal Infertility. Darlington Pig Discussion Group 13 th March 2014 Richard Bull
 Sow Reproduction and Seasonal Infertility Darlington Pig Discussion Group 13 th March 2014 Richard Bull Richard Bull Taurus Concepts Ltd Sow Reproduction Endogenous Hormones Gland Hormone Function Hypothalamus
Sow Reproduction and Seasonal Infertility Darlington Pig Discussion Group 13 th March 2014 Richard Bull Richard Bull Taurus Concepts Ltd Sow Reproduction Endogenous Hormones Gland Hormone Function Hypothalamus
Phyto- and Endogenous Estrogens Differently Activate Intracellular Calcium Ion Mobilization in Bovine Endometrial Cells
 Journal of Reproduction and Development, Vol. 52, No. 6, 2006 Full Paper Phyto- and Endogenous Estrogens Differently Activate Intracellular Calcium Ion Mobilization in Bovine Endometrial Cells Izabela
Journal of Reproduction and Development, Vol. 52, No. 6, 2006 Full Paper Phyto- and Endogenous Estrogens Differently Activate Intracellular Calcium Ion Mobilization in Bovine Endometrial Cells Izabela
THIS ARTICLE IS SPONSORED BY THE MINNESOTA DAIRY HEALTH CONFERENCE.
 THIS ARTICLE IS SPONSORED BY THE MINNESOTA DAIRY HEALTH CONFERENCE. ST. PAUL, MINNESOTA UNITED STATES OF MINNESOTA Reproductive Research in Jersey Cows Ricardo C. Chebel, DVM, MPVM Introduction A very
THIS ARTICLE IS SPONSORED BY THE MINNESOTA DAIRY HEALTH CONFERENCE. ST. PAUL, MINNESOTA UNITED STATES OF MINNESOTA Reproductive Research in Jersey Cows Ricardo C. Chebel, DVM, MPVM Introduction A very
Theriogenology Department, Faculty of Veterinary Medicine, Beni Suef University, Egypt 2
 Theriogenology Insight: 3(1):11-16. April, 2013 Ultrasonic monitoring and biometry of ovaries and ovarian structures during superovulation following transvagianl follicle ablation in Murrah buffaloes S.M.
Theriogenology Insight: 3(1):11-16. April, 2013 Ultrasonic monitoring and biometry of ovaries and ovarian structures during superovulation following transvagianl follicle ablation in Murrah buffaloes S.M.
REPRODUCCIÓN. La idea fija. Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
A comparison of the effects of estrus cow. nuclear maturation of bovine oocytes
 A comparison of the effects of estrus cow serum and fetal calf serum on in vitro nuclear maturation of bovine oocytes J Spiropoulos, SE Long University of Bristol, School of Veterinary Science, Department
A comparison of the effects of estrus cow serum and fetal calf serum on in vitro nuclear maturation of bovine oocytes J Spiropoulos, SE Long University of Bristol, School of Veterinary Science, Department
REGULATION OF PROSTAGLANDIN F 2α BIOSYNTHESIS BY LONG CHAIN FATTY ACIDS IN CATTLE
 REGULATION OF PROSTAGLANDIN F 2α BIOSYNTHESIS BY LONG CHAIN FATTY ACIDS IN CATTLE By CARLOS J. RODRÍGUEZ SALLABERRY A DISSERTATION PRESENTED TO THE GRADUATE SCHOOL OF THE UNIVERSITY OF FLORIDA IN PARTIAL
REGULATION OF PROSTAGLANDIN F 2α BIOSYNTHESIS BY LONG CHAIN FATTY ACIDS IN CATTLE By CARLOS J. RODRÍGUEZ SALLABERRY A DISSERTATION PRESENTED TO THE GRADUATE SCHOOL OF THE UNIVERSITY OF FLORIDA IN PARTIAL
Abstract ZORRILLA, LEAH M. Control of Luteolytic Sensitivity in the Porcine Corpus Luteum. (Under the direction of Dr. John E. Gadsby).
 Abstract ZORRILLA, LEAH M. Control of Luteolytic Sensitivity in the Porcine Corpus Luteum. (Under the direction of Dr. John E. Gadsby). The porcine corpus luteum (CL) is unusual in that it does not show
Abstract ZORRILLA, LEAH M. Control of Luteolytic Sensitivity in the Porcine Corpus Luteum. (Under the direction of Dr. John E. Gadsby). The porcine corpus luteum (CL) is unusual in that it does not show
Concentrations of luteinizing hormone and ovulatory responses in dairy cows before timed artificial insemination
 Kansas Agricultural Experiment Station Research Reports Volume 0 Issue Dairy Research (98-0) Article 8 0 Concentrations of luteinizing hormone and ovulatory responses in dairy cows before timed artificial
Kansas Agricultural Experiment Station Research Reports Volume 0 Issue Dairy Research (98-0) Article 8 0 Concentrations of luteinizing hormone and ovulatory responses in dairy cows before timed artificial
Deficient Proliferation and Apoptosis in the Granulosa and Theca Interna Cells of the Bovine Cystic Follicle
 Journal of Reproduction and Development, Vol. 53, No. 5, 2007 Research Note Deficient Proliferation and Apoptosis in the Granulosa and Theca Interna Cells of the Bovine Cystic Follicle Naoki ISOBE 1) and
Journal of Reproduction and Development, Vol. 53, No. 5, 2007 Research Note Deficient Proliferation and Apoptosis in the Granulosa and Theca Interna Cells of the Bovine Cystic Follicle Naoki ISOBE 1) and
INDUCING DOUBLE OVULATIONS IN BEEF CATTLE VIA SIMULTANEOUS LUTEAL REGRESSION AND FOLLICLE WAVE EMERGENCE IN A LOW PROGESTERONE ENVIRONMENT
 INDUCING DOUBLE OVULATIONS IN BEEF CATTLE VIA SIMULTANEOUS LUTEAL REGRESSION AND FOLLICLE WAVE EMERGENCE IN A LOW PROGESTERONE ENVIRONMENT E M M A M. J I N K S Honors Research Thesis Research Advisor:
INDUCING DOUBLE OVULATIONS IN BEEF CATTLE VIA SIMULTANEOUS LUTEAL REGRESSION AND FOLLICLE WAVE EMERGENCE IN A LOW PROGESTERONE ENVIRONMENT E M M A M. J I N K S Honors Research Thesis Research Advisor:
LH (Rodent) ELISA Kit
 LH (Rodent) ELISA Kit Catalog Number KA2332 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
LH (Rodent) ELISA Kit Catalog Number KA2332 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
Concentrations of Circulating Gonadotropins During. Various Reproductive States in Mares
 BIOLOGY OF REPRODUCTION, 744-75 (19) Concentrations of Circulating Gonadotropins During Various Reproductive States in Mares KURT F. MILLER, S. L. BERG, D. C. SHARP and. J. GINTHER Department of Veterinary
BIOLOGY OF REPRODUCTION, 744-75 (19) Concentrations of Circulating Gonadotropins During Various Reproductive States in Mares KURT F. MILLER, S. L. BERG, D. C. SHARP and. J. GINTHER Department of Veterinary
Supplementary Figure 1
 Supplementary Figure 1 The average sigmoid parametric curves of capillary dilation time courses and average time to 50% peak capillary diameter dilation computed from individual capillary responses averaged
Supplementary Figure 1 The average sigmoid parametric curves of capillary dilation time courses and average time to 50% peak capillary diameter dilation computed from individual capillary responses averaged
Nutrient partitioning in dairy cattle. Matthew C. Lucy. Department of Animal Sciences, University of Missouri
 NUTRIENT PARTITIONING AND REPRODUCTIVE PERFORMANCE IN DAIRY COWS Matthew C. Lucy Department of Animal Sciences, University of Missouri Take Home Messages Blood growth hormone (GH) concentrations increase
NUTRIENT PARTITIONING AND REPRODUCTIVE PERFORMANCE IN DAIRY COWS Matthew C. Lucy Department of Animal Sciences, University of Missouri Take Home Messages Blood growth hormone (GH) concentrations increase
Effects of Label-Dose Permethrin Administration on Reproductive Function and Embryo Quality on Superovulated Beef Heifers
 Animal Industry Report AS 662 ASL R3050 2016 Effects of Label-Dose Permethrin Administration on Reproductive Function and Embryo Quality on Superovulated Beef Heifers Tyler M. Dohlman Iowa State University,
Animal Industry Report AS 662 ASL R3050 2016 Effects of Label-Dose Permethrin Administration on Reproductive Function and Embryo Quality on Superovulated Beef Heifers Tyler M. Dohlman Iowa State University,
A differential equation model to investigate the dynamics of the bovine estrous cycle
 A differential equation model to investigate the dynamics of the bovine estrous cycle H. M. T. Boer 1,2, C. Stötzel 3, S. Röblitz 3, H. Woelders 1 1 Animal Breeding and Genomics Centre, Wageningen UR Livestock
A differential equation model to investigate the dynamics of the bovine estrous cycle H. M. T. Boer 1,2, C. Stötzel 3, S. Röblitz 3, H. Woelders 1 1 Animal Breeding and Genomics Centre, Wageningen UR Livestock
Female Reproductive System. Lesson 10
 Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated,
 1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
Follicle profile and plasma gonadotropin concentration in pubertal female ponies
 Brazilian Journal of Medical and Biological Research (0) 37: 913-9 Puberty in female pony ISSN 00-79X 913 Follicle profile and plasma gonadotropin concentration in pubertal female ponies Departamento de
Brazilian Journal of Medical and Biological Research (0) 37: 913-9 Puberty in female pony ISSN 00-79X 913 Follicle profile and plasma gonadotropin concentration in pubertal female ponies Departamento de
EVALUATION OF FOLLICULAR ATRESIA AND ELECTROPHORETIC PATTERN OF FOLLICULAR FLUID PROTEINS IN ACYCLIC BUFFALO (Bubalus bubalis)
 Original Article Buffalo Bulletin (December 2013) Vol.32 No.4 EVALUATION OF FOLLICULAR ATRESIA AND ELECTROPHORETIC PATTERN OF FOLLICULAR FLUID PROTEINS IN ACYCLIC BUFFALO (Bubalus bubalis) F.A. Khan 1,*,
Original Article Buffalo Bulletin (December 2013) Vol.32 No.4 EVALUATION OF FOLLICULAR ATRESIA AND ELECTROPHORETIC PATTERN OF FOLLICULAR FLUID PROTEINS IN ACYCLIC BUFFALO (Bubalus bubalis) F.A. Khan 1,*,
SISTEMA REPRODUCTOR (LA IDEA FIJA) Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
FSH (Rodent) ELISA Kit
 FSH (Rodent) ELISA Kit Catalog Number KA2330 96 assays Version: 06 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
FSH (Rodent) ELISA Kit Catalog Number KA2330 96 assays Version: 06 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
Hormonal Control of Human Reproduction
 Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting. protein3) regulate autophagy and mitophagy in renal tubular cells in. acute kidney injury
 Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and
 This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
Concentrations of Luteinizing Hormone and Ovulatory Responses in Dairy Cows Before Timed Artificial Insemination
 Concentrations of Luteinizing Hormone and Ovulatory Responses in Dairy Cows Before Timed Artificial Insemination S. L. Pulley, D. H. Keisler, S. L. Hill, and J. S. Stevenson Summary The objective of this
Concentrations of Luteinizing Hormone and Ovulatory Responses in Dairy Cows Before Timed Artificial Insemination S. L. Pulley, D. H. Keisler, S. L. Hill, and J. S. Stevenson Summary The objective of this
The reproductive lifespan
 The reproductive lifespan Reproductive potential Ovarian cycles Pregnancy Lactation Male Female Puberty Menopause Age Menstruation is an external indicator of ovarian events controlled by the hypothalamicpituitary
The reproductive lifespan Reproductive potential Ovarian cycles Pregnancy Lactation Male Female Puberty Menopause Age Menstruation is an external indicator of ovarian events controlled by the hypothalamicpituitary
Three-dimensional modeling of color Doppler images: a new approach to study follicular vascularization in cattle
 Anim. Reprod., v.10, n.4, p.662-669, Oct./Dec. 2013. Three-dimensional modeling of color Doppler images: a new approach to study follicular vascularization in cattle E.K.N. Arashiro 1, M.P. Palhao 2, J.R.L.
Anim. Reprod., v.10, n.4, p.662-669, Oct./Dec. 2013. Three-dimensional modeling of color Doppler images: a new approach to study follicular vascularization in cattle E.K.N. Arashiro 1, M.P. Palhao 2, J.R.L.
S. M. Quirk, G. J. Hickey and J. E. Fortune. Summary. Ultrasonography was used to monitor the growth, ovulation and regression
 Growth and regression of ovarian follicles during the follicular phase of the oestrous cycle in heifers undergoing spontaneous and PGF-2\g=a\-induced luteolysis S. M. Quirk, G. J. Hickey and J. E. Fortune
Growth and regression of ovarian follicles during the follicular phase of the oestrous cycle in heifers undergoing spontaneous and PGF-2\g=a\-induced luteolysis S. M. Quirk, G. J. Hickey and J. E. Fortune
FIXED-TIME INSEMINATION OF PORCINE LUTEINIZING HORMONE- TREATED SUPEROVULATED BEEF COWS AND THE RESYNCHRONIZATION OF BEEF COWS FOR FIXED-TIME EMBRYO
 FIXED-TIME INSEMINATION OF PORCINE LUTEINIZING HORMONE- TREATED SUPEROVULATED BEEF COWS AND THE RESYNCHRONIZATION OF BEEF COWS FOR FIXED-TIME EMBRYO TRANSFER A Dissertation by JOHN STEPHEN NELSON Submitted
FIXED-TIME INSEMINATION OF PORCINE LUTEINIZING HORMONE- TREATED SUPEROVULATED BEEF COWS AND THE RESYNCHRONIZATION OF BEEF COWS FOR FIXED-TIME EMBRYO TRANSFER A Dissertation by JOHN STEPHEN NELSON Submitted
Novel prospects for evaluation of follicle wall blood flow using color-doppler ultrasonography
 DOI: 10.21451/1984-3143-AR806 Anim. Reprod., v.13, n.4, p.762-771, Oct./Dec. 2016 Novel prospects for evaluation of follicle wall blood flow using color-doppler ultrasonography S.G.S. de Tarso, G.M. Ishak,
DOI: 10.21451/1984-3143-AR806 Anim. Reprod., v.13, n.4, p.762-771, Oct./Dec. 2016 Novel prospects for evaluation of follicle wall blood flow using color-doppler ultrasonography S.G.S. de Tarso, G.M. Ishak,
Vascular endothelial growth factor receptor 2 mediated angiogenesis is essential for gonadotropin-dependent follicle development
 Vascular endothelial growth factor receptor 2 mediated angiogenesis is essential for gonadotropin-dependent follicle development Ralf C. Zimmermann, 1 Tipton Hartman, 1 Suzanne Kavic, 1 Samuel A. Pauli,
Vascular endothelial growth factor receptor 2 mediated angiogenesis is essential for gonadotropin-dependent follicle development Ralf C. Zimmermann, 1 Tipton Hartman, 1 Suzanne Kavic, 1 Samuel A. Pauli,
Ovarian and PGF2 alpha responses to stimulation of endogenous PRL pulses during the estrous cycle in mares
 Universidade de São Paulo Biblioteca Digital da Produção Intelectual - BDPI Departamento de Zootecnia - FZEA/ZAZ Artigos e Materiais de Revistas Científicas - FZEA/ZAZ 212 Ovarian and PGF2 alpha responses
Universidade de São Paulo Biblioteca Digital da Produção Intelectual - BDPI Departamento de Zootecnia - FZEA/ZAZ Artigos e Materiais de Revistas Científicas - FZEA/ZAZ 212 Ovarian and PGF2 alpha responses
Management of recipients for an embryo transfer program in dromedary camels (Camelus dromedarius)
 Management of recipients for an embryo transfer program in dromedary camels (Camelus dromedarius) J.A. Skidmore* and M. Billah Camel Reproduction Centre, P.O. Box 79914, Dubai, United Arab Emirates Summary
Management of recipients for an embryo transfer program in dromedary camels (Camelus dromedarius) J.A. Skidmore* and M. Billah Camel Reproduction Centre, P.O. Box 79914, Dubai, United Arab Emirates Summary
Effect of repeated laparoscopic ovum pick-up on yield and quality of oocytes in goats
 Effect of repeated laparoscopic ovum pick-up on yield and quality of oocytes in goats Nor Farizah, A.H., Rahman, M.M., Wan Khadijah, W.E. and Abdullah, R.B.* Animal Biotechnology-Embryo Laboratory (ABEL),
Effect of repeated laparoscopic ovum pick-up on yield and quality of oocytes in goats Nor Farizah, A.H., Rahman, M.M., Wan Khadijah, W.E. and Abdullah, R.B.* Animal Biotechnology-Embryo Laboratory (ABEL),
Ovarian follicular development in cattle
 Ovarian follicular development in cattle John P Kastelic Professor of Theriogenology Head, Department of Production Animal Health University of Calgary Calgary, Alberta, Canada Overview Prenatal development
Ovarian follicular development in cattle John P Kastelic Professor of Theriogenology Head, Department of Production Animal Health University of Calgary Calgary, Alberta, Canada Overview Prenatal development
Comparison of endocrine and cellular mechanisms regulating the corpus luteum of primates and ruminants
 Anim Reprod, v.9, n.3, p.242-259, Jul./Sept. 2012 Comparison of endocrine and cellular mechanisms regulating the corpus luteum of primates and ruminants M.C. Wiltbank 1,8,9, S.M. Salih 2, M.O. Atli 1,3,
Anim Reprod, v.9, n.3, p.242-259, Jul./Sept. 2012 Comparison of endocrine and cellular mechanisms regulating the corpus luteum of primates and ruminants M.C. Wiltbank 1,8,9, S.M. Salih 2, M.O. Atli 1,3,
Effects of superovulation with FSH. Abstract
 Chapter 2 Effects of superovulation with ofsh and norgestomet/gnrh-controlled release of the LH surge on hormone concentrations, and yield of oocytes and embryos at specific developmental stages Effects
Chapter 2 Effects of superovulation with ofsh and norgestomet/gnrh-controlled release of the LH surge on hormone concentrations, and yield of oocytes and embryos at specific developmental stages Effects
SUMMARY. Keywords: quail, Coturnix japonica, morphology, ovary, oviduct, neurotrophins, immunohistochemistry
 SUMMARY Keywords: quail, Coturnix japonica, morphology, ovary, oviduct, neurotrophins, immunohistochemistry Studies on the development of biological systems have expanded using animal models, always to
SUMMARY Keywords: quail, Coturnix japonica, morphology, ovary, oviduct, neurotrophins, immunohistochemistry Studies on the development of biological systems have expanded using animal models, always to
