Date Modified: March 31, Clinical Quality Measures for PQRS
|
|
|
- Melina Carr
- 6 years ago
- Views:
Transcription
1 Date Modified: March 31, Clinical Quality s for PQRS
2 # Domain Title Description Type Denominator Numerator Denominator Exclusions/Exceptions 1 Patient Safety Prostate Biopsy Antibiotic Process Compliance 2 Effective Clinical Care Prostate Biopsy: Unplanned Hospital Admission within 30 Days of TRUS Biopsy 3 Effective Clinical Care Prostate Biopsy: Cancer Detection on Initial Prostate Biopsy 4 Community/Population Health Prostate Biopsy: Proportion of patients undergoing a prostate biopsy with a PSA < 4 5 Effective Clinical Care Prostate Biopsy: Repeat Biopsy for Patients with Atypical Small Acinar Proliferation (ASAP) 102 Efficiency and Cost Reduction 6 Efficiency and Cost Reduction Prostate Cancer: Avoidance of Overuse of Bone Scan for Staging Low Risk Prostate Cancer Patients Prostate Cancer: Avoidance of Overuse of CT Scan for Staging Low Risk Prostate Cancer Patients 104 Effective Clinical Care Prostate Cancer: Adjuvant Hormonal Therapy for High Risk Prostate Cancer Patients Compliance with AUA best practices for antibiotic prophylaxis for transrectal ultrasound-guided (TRUS) biopsy Percentage of biopsies where patients aged 30 years and older had an unplanned hospital admission within 30 days of biopsy Proportion of patients undergoing initial prostate biopsy in the registry found to have Proportion of patients undergoing a prostate biopsy with a PSA < 4 Proportion of patients undergoing a repeat prostate biopsy within 12 months of their initial biopsy in the registry as a result of a finding of atypical small acinar proliferation (ASAP) as per the NCCN guidelines Percentage of patients, regardless of age, with a diagnosis of prostate cancer at low risk of recurrence receiving interstitial prostate brachytherapy, OR external beam radiotherapy to the prostate, OR radical prostatectomy, OR cryotherapy who did not have a bone scan performed at any time since diagnosis of Percentage of patients, regardless of age, with a diagnosis of prostate cancer at low risk of recurrence receiving interstitial prostate brachytherapy, OR external beam radiotherapy to the prostate, OR radical prostatectomy, OR cryotherapy who did not have a CT scan performed at any time since diagnosis of prostate cancer Percentage of patients, regardless of age, with a diagnosis of prostate cancer at high risk of recurrence receiving external beam radiotherapy to the prostate who were prescribed adjuvant hormonal therapy (GnRH agonist or antagonist) Process # of prostate biopsies for patients aged 30 or older # of prostate biopsies for patients aged 30 or older # of patients aged 30 or older undergoing a prostate biopsy # of patients aged 30 or older undergoing a prostate biopsy # of patients aged 30 or older that had a finding of ASAP on their initial prostate biopsy Process Process # of low-risk patients 30 years or older receiving brachytherapy, EBRT, RP, or cryotherapy Process # of biopsies where a compliant antibiotic regimen was given # of biopsies where a hospital admission resulted within 30 days of the procedure # of patients that have an initial biopsy in the registry which shows # of patients undergoing a prostate biopsy with a PSA < 4 # of patients undergoing a repeat prostate biopsy as a result of a finding of ASAP on their initial biopsy See # of low risk patients receiving brachytherapy, EBRT, RP, or cryotherapy who did not have a CT scan since diagnosis of See Non-TRUS biopsies; biopsies performed in patients < 30 years of age; biopsies performed in patients that have Biopsies performed in patients < 30 years of age; biopsies performed in patients that have treatment for treatment for treatment for treatment for Prostate cancer patients < 30 years of age; patients receiving an alternate form of treatment from those specified; patients that have treatment for Rationale evidence-based care safety of medical care clinical effectiveness appropriate utilization of prostate biopsy evidence-based care appropriate resource utilization Michigan Urological Surgery Improvement Collaborative (MUSIC) QCDR Page 1
3 # Domain Title Description Type Denominator Numerator Denominator Exclusions/Exceptions 130 Patient Safety Documentation of Current Process See Medications in the Medical Record 194 Effective Clinical Care Oncology: Cancer Stage Documented 250 Effective Clinical Care Radical Prostatectomy Pathology Reporting 265 Communication and Care Coordination Biopsy Follow-Up 7 Effective Clinical Care Prostate Cancer: Active Surveillance for Low Risk Prostate Cancer Patients 8 Effective Clinical Care Prostate Cancer: Radical Prostatectomy Cases LOS 9 Communication and Care Coordination Prostate Cancer: Unplanned Hospital Readmission After Radical Prostatectomy Percentage of visits for patients aged 18 years and older for which the eligible professional attests to documenting a list of current medications using all immediate resources available on the date of the encounter. This list must include ALL known prescriptions, over-thecounters, herbals, and vitamin/mineral/dietary (nutritional) supplements AND must contain the medications name, dosage, frequency and route of administration. Percentage of patients, regardless of age, with a diagnosis of cancer who are seen in the ambulatory setting who have a baseline American Joint Committee on Cancer (AJCC) cancer stage or documentation that the cancer is metastatic in the medical record at least once during the 12 month reporting period Percentage of radical prostatectomy pathology reports that include the pt category, the pn category, the Gleason score and a statement about margin status Percentage of new patients whose biopsy results have been reviewed and communicated to the primary care/referring physician and patient by the performing physician Proportion of patients with low-risk receiving active surveillance Percentage of radical prostatectomy cases with a length of stay > 2 days Percentage of patients aged 30 years and older who had an unplanned hospital readmission within 30 days of principal procedure Process Process Process # of low-risk patients 30 or older # of patients aged 30 or older who underwent a radical prostatectomy as treatment for # of radical prostatectomies for patients aged 30 or older See See See # of low-risk patients on active surveillance # of patients with a length of stay > 2 days after radical prostatectomy # of radical prostatectomies where a hospital admission resulted within 30 days of the procedure treatment for ; patients that are not being screened or treated for treatment for treatment for treatment for ; patients that are not being screened or treated for prostate cancer Prostate cancer patients < 30 years of age; patients that have Prostate cancer patients < 30 years of age; patients that have Prostatectomies performed in patients < 30 years of age; treatment for Rationale patientcentered decision making appropriate resource utilization safety of medical care Michigan Urological Surgery Improvement Collaborative (MUSIC) QCDR Page 2
4 Non-PQRS #1: Prostate Biopsy Antibiotic Compliance Compliance with AUA best practices for antibiotic prophylaxis for transrectal ultrasound-guided (TRUS) biopsy Patient safety # of biopsies where a compliant antibiotic regimen was given # of prostate biopsies for patients aged 30 or older Excludes non-trus biopsies; biopsies performed in patients < 30 years of age; biopsies performed in treatment for evidence-based care Non-PQRS # 2: Prostate Biopsy: Unplanned Hospital Admissions within 30 days of TRUS Biopsy Percentage of biopsies where patients aged 30 years and older had an unplanned hospital admission within 30 days of biopsy Effective clinical care # of biopsies where a hospital admission resulted within 30 days of the procedure # of prostate biopsies for patients aged 30 or older Excludes non-trus biopsies; biopsies performed in patients < 30 years of age; biopsies performed in treatment for safety of medical care Michigan Urological Surgery Improvement Collaborative (MUSIC) QCDR Page 3
5 Non-PQRS # 3: Prostate Biopsy: Cancer Detection on Initial Prostate Biopsy Proportion of patients undergoing initial prostate biopsy in the registry found to have Effective clinical care # of patients that have an initial biopsy in the registry which shows # of patients aged 30 or older undergoing a prostate biopsy Excludes non-trus biopsies; biopsies performed in patients < 30 years of age; biopsies performed in treatment for clinical effectiveness Non-PQRS # 4: Prostate Biopsy: Proportion of patients undergoing a prostate biopsy with a PSA < 4 Proportion of patients undergoing a prostate biopsy with a PSA < 4 Community/Population Health # of patients undergoing a prostate biopsy with a PSA < 4 # of patients aged 30 or older undergoing a prostate biopsy Excludes non-trus biopsies; biopsies performed in patients < 30 years of age; biopsies performed in treatment for appropriate utilization of prostate biopsy Michigan Urological Surgery Improvement Collaborative (MUSIC) QCDR Page 4
6 Non-PQRS # 5: Prostate Biopsy: Repeat Biopsy for Patients with Atypical Small Acinar Proliferation (ASAP) Proportion of patients undergoing a repeat prostate biopsy within 12 months of their initial biopsy in the registry as a result of a finding of atypical small acinar proliferation (ASAP) as per the NCCN guidelines Effective Clinical Care # of patients undergoing a repeat prostate biopsy as a result of a finding of ASAP on their initial biopsy # of patients undergoing a repeat prostate biopsy as a result of a finding of ASAP on their initial biopsy Excludes non-trus biopsies; biopsies performed in patients < 30 years of age; biopsies performed in treatment for evidence-based care Non-PQRS # 6: Prostate Cancer: Avoidance of Overuse of CT Scan for Staging Low Risk Prostate Cancer Patients Percentage of patients, regardless of age, with a diagnosis of at low risk of recurrence receiving interstitial prostate brachytherapy, OR external beam radiotherapy to the prostate, OR radical prostatectomy, OR cryotherapy who did not have a CT scan performed at any time since diagnosis of Efficiency and cost reduction # of low risk patients receiving brachytherapy, EBRT, RP, or cryotherapy who did not have a CT scan since diagnosis of # of low-risk patients 30 years or older receiving brachytherapy, EBRT, RP, or cryotherapy Excludes patients < 30 years of age; patients receiving an alternate form of treatment from those specified; patients that have appropriate resource utilization Michigan Urological Surgery Improvement Collaborative (MUSIC) QCDR Page 5
7 Non-PQRS # 7: Prostate Cancer: Active Surveillance for Low Risk Prostate Cancer Patients Proportion of patients with low-risk receiving active surveillance Effective clinical care # of low-risk patients on active surveillance # of prostate biopsies for patients aged 30 or older Excludes patients < 30 years of age; treatment for patient-centered decision making Non-PQRS # 8: Prostate Cancer: Radical Prostatectomy Cases LOS Percentage of radical prostatectomy cases with a length of stay > 2 days Effective clinical care # of patients with a length of stay > 2 days after radical prostatectomy # of patients aged 30 or older who underwent a radical prostatectomy as treatment for Excludes patients < 30 years of age; treatment for appropriate resource utilization Michigan Urological Surgery Improvement Collaborative (MUSIC) QCDR Page 6
8 Non-PQRS # 9: Prostate Cancer: Unplanned Hospital Readmission After Radical Prostatectomy Percentage of patients aged 30 years and older who had an unplanned hospital readmission within 30 days of principal procedure Communication and care coordination # of radical prostatectomies where a hospital admission resulted within 30 days of the procedure # of radical prostatectomies for patients aged 30 or older Excludes prostatectomies performed in patients < 30 years of age; treatment for safety of medical care Michigan Urological Surgery Improvement Collaborative (MUSIC) QCDR Page 7
Date Modified: May 29, Clinical Quality Measures for PQRS
 Date Modified: May 29, 2014 Clinical Quality s for PQRS # Domain Type Denominator Numerator Denominator Exclusions/Exceptions Rationale QCDR-1 QCDR-2 Patient Safety 102 Efficiency and Cost Reduction QCDR-3
Date Modified: May 29, 2014 Clinical Quality s for PQRS # Domain Type Denominator Numerator Denominator Exclusions/Exceptions Rationale QCDR-1 QCDR-2 Patient Safety 102 Efficiency and Cost Reduction QCDR-3
Non-QPP Measures. # Measure Title Definition Type Domain. Cryptorchidism: Inappropriate use of scrotal/groin ultrasound on boys
 Non-QPP Measures # Measure Title Definition Type Domain 1 Cryptorchidism: Inappropriate use of scrotal/groin ultrasound on boys Percentage of patients (boys) =< 18 years of age newly diagnosed with undescended
Non-QPP Measures # Measure Title Definition Type Domain 1 Cryptorchidism: Inappropriate use of scrotal/groin ultrasound on boys Percentage of patients (boys) =< 18 years of age newly diagnosed with undescended
creatinine lab order placed abdomen, MRI abdomen, ultrasound abdomen ordered or performed
 Non-QPP Measures # Measure Title Definition Type Domain 1 Cryptorchidism: Inappropriate use of scrotal/groin ultrasound on boys Percentage of patients (boys) =< 18 years of age newly diagnosed with undescended
Non-QPP Measures # Measure Title Definition Type Domain 1 Cryptorchidism: Inappropriate use of scrotal/groin ultrasound on boys Percentage of patients (boys) =< 18 years of age newly diagnosed with undescended
Non-QPP Measures 3 AQUA12. 6 AQUA15 Stones: Urinalysis documented 30 days before
 Non-QPP Measures 1 Measure ID Measure Title Definition Type Domain AQUA3 (inverse) Cryptorchidism: Inappropriate use of scrotal/groin ultrasound on boys Percentage of patients (boys) =< 18 years of age
Non-QPP Measures 1 Measure ID Measure Title Definition Type Domain AQUA3 (inverse) Cryptorchidism: Inappropriate use of scrotal/groin ultrasound on boys Percentage of patients (boys) =< 18 years of age
Percentage of patients who underwent endoscopic procedures following SWL
 Non-QPP Measures Measure ID Measure Title Definition Type Domain 1 AQUA12 Benign Prostate Hyperplasia: IPSS improvement after diagnosis Percentage of patients with NEW diagnosis of clinically significant
Non-QPP Measures Measure ID Measure Title Definition Type Domain 1 AQUA12 Benign Prostate Hyperplasia: IPSS improvement after diagnosis Percentage of patients with NEW diagnosis of clinically significant
2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
 Measure #104 (NQF 0390): Prostate Cancer: Adjuvant Hormonal Therapy for High Risk or Very High Risk Prostate Cancer National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL
Measure #104 (NQF 0390): Prostate Cancer: Adjuvant Hormonal Therapy for High Risk or Very High Risk Prostate Cancer National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL
QUALIFIED CLINICAL DATA REGISTRY (QCDR) 2017 MEASURE SPECIFICATIONS
 QOPI5 Chemotherapy administered to patients with metastatic solid tumor with performance status of 3, 4, or undocumented (Lower Score - Better) Percentage of adult patients with metastatic solid tumors
QOPI5 Chemotherapy administered to patients with metastatic solid tumor with performance status of 3, 4, or undocumented (Lower Score - Better) Percentage of adult patients with metastatic solid tumors
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #102 (NQF 0389): Prostate Cancer: Avoidance of Overuse of Bone Scan for Staging Low Risk Prostate Cancer Patients National Quality Strategy Domain: Efficiency and Cost Reduction 2018 OPTIONS
Quality ID #102 (NQF 0389): Prostate Cancer: Avoidance of Overuse of Bone Scan for Staging Low Risk Prostate Cancer Patients National Quality Strategy Domain: Efficiency and Cost Reduction 2018 OPTIONS
MEASURE SPECIFICATIONS
 QOPI REPTING REGISTRY (QCDR) 2018 QOPI 5 QOPI 11 Chemotherapy administered to patients with metastatic solid tumor with performance status of 3, 4, or undocumented (Lower Score - Better) Combination chemotherapy
QOPI REPTING REGISTRY (QCDR) 2018 QOPI 5 QOPI 11 Chemotherapy administered to patients with metastatic solid tumor with performance status of 3, 4, or undocumented (Lower Score - Better) Combination chemotherapy
MEASURE SPECIFICATIONS
 QOPI REPTING REGISTRY (QCDR) 2018 QOPI5 Title Chemotherapy administered to patients with metastatic solid tumor with performance status of 3, 4, or undocumented (Lower Score - Better) Description Percentage
QOPI REPTING REGISTRY (QCDR) 2018 QOPI5 Title Chemotherapy administered to patients with metastatic solid tumor with performance status of 3, 4, or undocumented (Lower Score - Better) Description Percentage
Prostate Cancer Dashboard
 Process Risk Assessment Risk assessment: family history assessment of family history of prostate cancer Best Observed: 97 %1 ; Ideal Benchmark:100% measure P8 2 Process Appropriateness of Care Pre-treatment
Process Risk Assessment Risk assessment: family history assessment of family history of prostate cancer Best Observed: 97 %1 ; Ideal Benchmark:100% measure P8 2 Process Appropriateness of Care Pre-treatment
Cancer Endorsement Maintenance 2011-Maintenance Measures
 Measure Number Title Description Measure Steward 0210 Proportion receiving chemotherapy in the last 14 days of life 0211 Proportion with more than one emergency room visit in the last days of life 0212
Measure Number Title Description Measure Steward 0210 Proportion receiving chemotherapy in the last 14 days of life 0211 Proportion with more than one emergency room visit in the last days of life 0212
2011 Physician Quality Reporting System Measures for Consideration by Oncology Providers: Cancer Care Measures
 2011 Physician Quality Reporting System Measures for Consideration by Oncology Providers: Cancer Care Measures The table below includes measures directly relevant to oncology providers as well as general
2011 Physician Quality Reporting System Measures for Consideration by Oncology Providers: Cancer Care Measures The table below includes measures directly relevant to oncology providers as well as general
IQSS 2019 QCDR and MIPS Measure Specifications
 IQSS1 Hypogonadism: Serum T, CBC, PSA, IPSS within 6 months of Rx Percentage of patients with a Effective Clinical Patients with documented new diagnosis of hypogonadism receiving androgen replacement
IQSS1 Hypogonadism: Serum T, CBC, PSA, IPSS within 6 months of Rx Percentage of patients with a Effective Clinical Patients with documented new diagnosis of hypogonadism receiving androgen replacement
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #104 (NQF 0390): Prostate Cancer: Combination Androgen Deprivation Therapy for High Risk or Very High Risk Prostate Cancer National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS
Quality ID #104 (NQF 0390): Prostate Cancer: Combination Androgen Deprivation Therapy for High Risk or Very High Risk Prostate Cancer National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS
2014 Oncology Measures Group Overview
 2014 Oncology Measures Group Overview The Oncology Measures Group is a reporting option that significantly reduces the burden of participation in the Physician Quality Reporting System (PQRS). Source:
2014 Oncology Measures Group Overview The Oncology Measures Group is a reporting option that significantly reduces the burden of participation in the Physician Quality Reporting System (PQRS). Source:
Possible Denominator Codes Applicable to OMS * Le Fort Fractures 21346, 21347, 21348, 21422, 21423, 21432, 21433, 21435, 21436
 2015 Individual PQRS s Eligible OMS #22: Perioperative Care: Discontinuation of Prophylactic Antibiotics (Non- Cardiac Procedures) Percentage of noncardiac surgical patients aged 18 years and older undergoing
2015 Individual PQRS s Eligible OMS #22: Perioperative Care: Discontinuation of Prophylactic Antibiotics (Non- Cardiac Procedures) Percentage of noncardiac surgical patients aged 18 years and older undergoing
NATIONAL QUALITY FORUM
 Cancer Endorsement Maintenance Table of Submitted Measures Phase I 0210 1 Proportion receiving chemotherapy in the last 14 days of life Percentage of patients who died from cancer receiving chemotherapy
Cancer Endorsement Maintenance Table of Submitted Measures Phase I 0210 1 Proportion receiving chemotherapy in the last 14 days of life Percentage of patients who died from cancer receiving chemotherapy
Identifying Initial Populations and Sampling for OCM and EBRT. Henrietta C. Hight, BA, BSN, RN, CCM, CDMS, CPHQ Quality Improvement Specialist
 Identifying Initial Populations and Sampling for OCM and EBRT Henrietta C. Hight, BA, BSN, RN, CCM, CDMS, CPHQ Quality Improvement Specialist January 22, 2015 Learning Objectives Help participants to understand
Identifying Initial Populations and Sampling for OCM and EBRT Henrietta C. Hight, BA, BSN, RN, CCM, CDMS, CPHQ Quality Improvement Specialist January 22, 2015 Learning Objectives Help participants to understand
One Stop Prostate Biopsy Protocol Author Consultation Date Approved
 One Stop Prostate Biopsy Protocol Author Consultation Date Approved Urology Nurse Practioner PROTOCOL FOR MEN ATTENDING A ONE STOP PROSTATE BIOPSY CLINIC RATIONALE Prostate cancer is the most common cancer
One Stop Prostate Biopsy Protocol Author Consultation Date Approved Urology Nurse Practioner PROTOCOL FOR MEN ATTENDING A ONE STOP PROSTATE BIOPSY CLINIC RATIONALE Prostate cancer is the most common cancer
Measure #250 (NQF 1853): Radical Prostatectomy Pathology Reporting National Quality Strategy Domain: Effective Clincial Care
 Measure #250 (NQF 1853): Radical Prostatectomy Pathology Reporting National Quality Strategy Domain: Effective Clincial Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS, REGISTRY DESCRIPTION: Percentage
Measure #250 (NQF 1853): Radical Prostatectomy Pathology Reporting National Quality Strategy Domain: Effective Clincial Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS, REGISTRY DESCRIPTION: Percentage
2014 Physician Quality Reporting System Data Collection Form: Oncology (for patients aged 18 and older)
 2014 Physician Quality Reporting System Data Collection Form: Oncology (for patients aged 18 and older) Physician Name: Patient Name: Last First MI Date of Birth: / / mm dd yyyy Gender: M F Medical Record
2014 Physician Quality Reporting System Data Collection Form: Oncology (for patients aged 18 and older) Physician Name: Patient Name: Last First MI Date of Birth: / / mm dd yyyy Gender: M F Medical Record
Quality ID #250 (NQF 1853): Radical Prostatectomy Pathology Reporting National Quality Strategy Domain: Effective Clinical Care
 Quality ID #250 (NQF 1853): Radical Prostatectomy Pathology Reporting National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process
Quality ID #250 (NQF 1853): Radical Prostatectomy Pathology Reporting National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process
Michigan Urological Surgery Improvement Collaborative
 Michigan Urological Surgery Improvement Collaborative Making Michigan #1 in Prostate Cancer Care Jim Montie, MD Susan Linsell, MHSA February 10, 2015 Vital statistics MUSIC Participants: 42 practices 235
Michigan Urological Surgery Improvement Collaborative Making Michigan #1 in Prostate Cancer Care Jim Montie, MD Susan Linsell, MHSA February 10, 2015 Vital statistics MUSIC Participants: 42 practices 235
Best Papers. F. Fusco
 Best Papers UROLOGY F. Fusco Best papers - 2015 RP/RT Oncological outcomes RP/RT IN ct3 Utilization trends RP/RT Complications Evolving role of elnd /Salvage LND This cohort reflects the current clinical
Best Papers UROLOGY F. Fusco Best papers - 2015 RP/RT Oncological outcomes RP/RT IN ct3 Utilization trends RP/RT Complications Evolving role of elnd /Salvage LND This cohort reflects the current clinical
AQUA Registry 2019 Non-QPP Measure Specifications. Denominator Exceptions. IPSS<8 None None Yes Patient Reported Outcome (PRO)
 AQUA12 Benign Prostate Hyperplasia: IPSS improvement after diagnosis with NEW diagnosis of clinically significant BPH who had IPSS (international prostate symptoms score) or AUASS (American urological
AQUA12 Benign Prostate Hyperplasia: IPSS improvement after diagnosis with NEW diagnosis of clinically significant BPH who had IPSS (international prostate symptoms score) or AUASS (American urological
Case Discussions: Prostate Cancer
 Case Discussions: Prostate Cancer Andrew J. Stephenson, MD FRCSC FACS Chief, Urologic Oncology Glickman Urological and Kidney Institute Cleveland Clinic Elevated PSA 1 54 yo, healthy male, family Hx of
Case Discussions: Prostate Cancer Andrew J. Stephenson, MD FRCSC FACS Chief, Urologic Oncology Glickman Urological and Kidney Institute Cleveland Clinic Elevated PSA 1 54 yo, healthy male, family Hx of
Screening and Diagnosis Prostate Cancer
 Screening and Diagnosis Prostate Cancer Daniel Heng MD MPH FRCPC Chair, Genitourinary Tumor Group Tom Baker Cancer Center University of Calgary, Canada @DrDanielHeng Outline Screening Evidence Recommendations
Screening and Diagnosis Prostate Cancer Daniel Heng MD MPH FRCPC Chair, Genitourinary Tumor Group Tom Baker Cancer Center University of Calgary, Canada @DrDanielHeng Outline Screening Evidence Recommendations
Factors Associated with Initial Treatment for Clinically Localized Prostate Cancer
 Factors Associated with Initial Treatment for Clinically Localized Prostate Cancer Preliminary Results from the National Program of Cancer Registries Patterns of Care Study (PoC1) NAACCR Annual Meeting
Factors Associated with Initial Treatment for Clinically Localized Prostate Cancer Preliminary Results from the National Program of Cancer Registries Patterns of Care Study (PoC1) NAACCR Annual Meeting
Clinical Quality Measures for PQRS. Last Updated: June 4, 2014
 Clinical Quality Measures for PQRS Last Updated: June 4, 2014 The Michigan Bariatric Surgery Collaborative (MBSC) Quality Clinical Data Registry will submit the following measures outlined below on behalf
Clinical Quality Measures for PQRS Last Updated: June 4, 2014 The Michigan Bariatric Surgery Collaborative (MBSC) Quality Clinical Data Registry will submit the following measures outlined below on behalf
The Royal Marsden. Prostate case study. Presented by Mr Alan Thompson Consultant Urological Surgeon
 Prostate case study Presented by Mr Alan Thompson Consultant Urological Surgeon 2 Part one Initial presentation A 62 year old male solicitor attends your GP surgery. He has rarely seen you over the last
Prostate case study Presented by Mr Alan Thompson Consultant Urological Surgeon 2 Part one Initial presentation A 62 year old male solicitor attends your GP surgery. He has rarely seen you over the last
Guidelines for the Management of Prostate Cancer West Midlands Expert Advisory Group for Urological Cancer
 Guidelines for the Management of Prostate Cancer West Midlands Expert Advisory Group for Urological Cancer West Midlands Clinical Networks and Clinical Senate Coversheet for Network Expert Advisory Group
Guidelines for the Management of Prostate Cancer West Midlands Expert Advisory Group for Urological Cancer West Midlands Clinical Networks and Clinical Senate Coversheet for Network Expert Advisory Group
Navigating the Stream: Prostate Cancer and Early Detection. Ifeanyi Ani, M.D. TPMG Urology Newport News
 Navigating the Stream: Prostate Cancer and Early Detection Ifeanyi Ani, M.D. TPMG Urology Newport News Understand epidemiology of prostate cancer Discuss PSA screening and PSA controversy Review tools
Navigating the Stream: Prostate Cancer and Early Detection Ifeanyi Ani, M.D. TPMG Urology Newport News Understand epidemiology of prostate cancer Discuss PSA screening and PSA controversy Review tools
The NOF & NBHA Quality Improvement Registry
 In collaboration with CECity The NOF & NBHA Quality Improvement Registry This registry is approved by CMS as a Qualified Clinical Data Registry (QCDR) for Eligible Professionals and GPRO Practices for
In collaboration with CECity The NOF & NBHA Quality Improvement Registry This registry is approved by CMS as a Qualified Clinical Data Registry (QCDR) for Eligible Professionals and GPRO Practices for
The European Board of Urology
 Page 1 of 15 The European Board of Urology Sub-specialty certification application: Prostate cancer, A - General information A1 - APPLICATION IDENTIFICATION 1a. Application code blank 1b. EBU internal
Page 1 of 15 The European Board of Urology Sub-specialty certification application: Prostate cancer, A - General information A1 - APPLICATION IDENTIFICATION 1a. Application code blank 1b. EBU internal
Prostate Overview Quiz
 Prostate Overview Quiz 1. The path report reads: Gleason 3 + 4 = 7. The Gleason s score is a. 3 b. 4 c. 7 d. None of the above 2. The path report reads: Moderately differentiated adenocarcinoma of the
Prostate Overview Quiz 1. The path report reads: Gleason 3 + 4 = 7. The Gleason s score is a. 3 b. 4 c. 7 d. None of the above 2. The path report reads: Moderately differentiated adenocarcinoma of the
Understanding the risk of recurrence after primary treatment for prostate cancer. Aditya Bagrodia, MD
 Understanding the risk of recurrence after primary treatment for prostate cancer Aditya Bagrodia, MD Aditya.bagrodia@utsouthwestern.edu 423-967-5848 Outline and objectives Prostate cancer demographics
Understanding the risk of recurrence after primary treatment for prostate cancer Aditya Bagrodia, MD Aditya.bagrodia@utsouthwestern.edu 423-967-5848 Outline and objectives Prostate cancer demographics
2015 myresearch Science Internship Program: Applied Medicine. Civic Education Office of Government and Community Relations
 2015 myresearch Science Internship Program: Applied Medicine Civic Education Office of Government and Community Relations Harguneet Singh Science Internship Program: Applied Medicine Comparisons of Outcomes
2015 myresearch Science Internship Program: Applied Medicine Civic Education Office of Government and Community Relations Harguneet Singh Science Internship Program: Applied Medicine Comparisons of Outcomes
THE UROLOGY GROUP
 THE UROLOGY GROUP www.urologygroupvirginia.com 1860 Town Center Drive Suite 150/160 Reston, VA 20190 703-480-0220 19415 Deerfield Avenue Suite 112 Leesburg, VA 20176 703-724-1195 224-D Cornwall Street,
THE UROLOGY GROUP www.urologygroupvirginia.com 1860 Town Center Drive Suite 150/160 Reston, VA 20190 703-480-0220 19415 Deerfield Avenue Suite 112 Leesburg, VA 20176 703-724-1195 224-D Cornwall Street,
2015 Physician Quality Reporting System Data Collection Form: Oncology (for patients aged 18 and older)
 2015 Physician Quality Reporting System Data Collection Form: Oncology (for patients aged 18 and older) IMPORTANT: Any measure with a 0% performance rate (100% for inverse measures) is not considered satisfactory
2015 Physician Quality Reporting System Data Collection Form: Oncology (for patients aged 18 and older) IMPORTANT: Any measure with a 0% performance rate (100% for inverse measures) is not considered satisfactory
Patients who receive image or palpation-guided needle biopsy (core or FNA) for the diagnosis of breast cancer.
 Description Measure 0221: Needle biopsy to establish diagnosis of cancer precedes surgical excision/resection (Commission on Cancer, American College of Surgeons) Percentage of patients presenting with
Description Measure 0221: Needle biopsy to establish diagnosis of cancer precedes surgical excision/resection (Commission on Cancer, American College of Surgeons) Percentage of patients presenting with
Prostatectomy as salvage therapy. Cases. Paul Cathcart - Guy s & St Thomas NHS Trust, London
 Prostatectomy as salvage therapy Cases Paul Cathcart - Guy s & St Thomas NHS Trust, London Attributes of brachytherapy appeal to young men who place high utility on genitourinary function At risk of
Prostatectomy as salvage therapy Cases Paul Cathcart - Guy s & St Thomas NHS Trust, London Attributes of brachytherapy appeal to young men who place high utility on genitourinary function At risk of
CLINICAL TRIALS Open clinical uro-oncology trials in Canada George Rodrigues, MD, Eric Winquist, MD
 Open clinical uro-oncology trials in Canada George Rodrigues, MD, Eric Winquist, MD London Health Sciences Centre, London, Ontario, Canada bladder cancer AN OPEN-LABEL, MULTICENTER, RANDOMIZED PHASE II
Open clinical uro-oncology trials in Canada George Rodrigues, MD, Eric Winquist, MD London Health Sciences Centre, London, Ontario, Canada bladder cancer AN OPEN-LABEL, MULTICENTER, RANDOMIZED PHASE II
How do I control (monitor) patients receiving TRT after prostate cancer treatment
 How do I control (monitor) patients receiving TRT after prostate cancer treatment Anthony J. Bella MD, FRCSC Greta and John Hansen Chair in Men s Health Research Division of Urology, Department of Surgery
How do I control (monitor) patients receiving TRT after prostate cancer treatment Anthony J. Bella MD, FRCSC Greta and John Hansen Chair in Men s Health Research Division of Urology, Department of Surgery
March 31, Dear colleagues,
 6300 North River Road Rosemont, IL 60018 p: 847-292-0530 f: 847-292-0531 www.ajrr.net March 31, 2014 Dear colleagues, The American Joint Replacement Registry (AJRR) has submitted materials to be considered
6300 North River Road Rosemont, IL 60018 p: 847-292-0530 f: 847-292-0531 www.ajrr.net March 31, 2014 Dear colleagues, The American Joint Replacement Registry (AJRR) has submitted materials to be considered
Radiation Therapy for Prostate Cancer. Resident Dept of Urology General Surgery Grand Round November 24, 2008
 Radiation Therapy for Prostate Cancer Amy Hou,, MD Resident Dept of Urology General Surgery Grand Round November 24, 2008 External Beam Radiation Advances Improving Therapy Generation of linear accelerators
Radiation Therapy for Prostate Cancer Amy Hou,, MD Resident Dept of Urology General Surgery Grand Round November 24, 2008 External Beam Radiation Advances Improving Therapy Generation of linear accelerators
2015 PQRS Registry. Source Measure Title Measure Description CITIUS1
 1 CQ-IQ covers 65 CMS defined measures that Eligible Providers (EPs) have to report on to assess quality of care provided to the patients. Version Supported: PQRS Registry 2015 65 measures Reporting Period:
1 CQ-IQ covers 65 CMS defined measures that Eligible Providers (EPs) have to report on to assess quality of care provided to the patients. Version Supported: PQRS Registry 2015 65 measures Reporting Period:
Oncology Quality Clinical Data Registry
 Oncology Quality Clinical Data Registry Powered by Premier Inc. This registry has been approved by CMS as a Qualified Clinical Data Registry (QCDR) for eligible clinicians and group practices for the 2019
Oncology Quality Clinical Data Registry Powered by Premier Inc. This registry has been approved by CMS as a Qualified Clinical Data Registry (QCDR) for eligible clinicians and group practices for the 2019
Troubleshooting Audio
 Welcome! Audio for this event is available via ReadyTalk Internet Streaming. No telephone line is required. Computer speakers or headphones are necessary to listen to streaming audio. Limited dial-in lines
Welcome! Audio for this event is available via ReadyTalk Internet Streaming. No telephone line is required. Computer speakers or headphones are necessary to listen to streaming audio. Limited dial-in lines
Clinical Case Conference
 Clinical Case Conference Intermediate-risk prostate cancer 08/06/2014 Long Pham Clinical Case 64 yo man was found to have elevated PSA of 8.65. TRUS-biopies were negative. Surveillance PSA was 7.2 in 3
Clinical Case Conference Intermediate-risk prostate cancer 08/06/2014 Long Pham Clinical Case 64 yo man was found to have elevated PSA of 8.65. TRUS-biopies were negative. Surveillance PSA was 7.2 in 3
Measures Groups Specifications Manual
 2015 Physician Quality Reporting System (PQRS) Measures Groups Specifications Manual Utilized by Individual Eligible Professionals Registry ONLY Reporting 12/22/2014 This manual contains specific guidance
2015 Physician Quality Reporting System (PQRS) Measures Groups Specifications Manual Utilized by Individual Eligible Professionals Registry ONLY Reporting 12/22/2014 This manual contains specific guidance
Diagnosis and management of prostate cancer in the
 Diagnosis and management of prostate cancer in the Jeremy Teoh ( 張源津 ) Assistant Professor, Department of Surgery, The Chinese University of Hong Kong. Email: jeremyteoh@surgery.cuhk.edu.hk Estimated age-standardised
Diagnosis and management of prostate cancer in the Jeremy Teoh ( 張源津 ) Assistant Professor, Department of Surgery, The Chinese University of Hong Kong. Email: jeremyteoh@surgery.cuhk.edu.hk Estimated age-standardised
Compass PTN Core Measures
 Compass PTN Core Measures emeasure ID: CMS122v5 NQF: 0059 QualityID: 001 Diabetes: Hemoglobin A1c (HbA1c) Poor Control (>9%) Patients 18-75 years of age with diabetes with a visit during the measurement
Compass PTN Core Measures emeasure ID: CMS122v5 NQF: 0059 QualityID: 001 Diabetes: Hemoglobin A1c (HbA1c) Poor Control (>9%) Patients 18-75 years of age with diabetes with a visit during the measurement
Protocol. This trial protocol has been provided by the authors to give readers additional information about their work.
 Protocol This trial protocol has been provided by the authors to give readers additional information about their work. Protocol for: Wilt TJ, Brawer MK, Jones KM, et al. Radical prostatectomy versus observation
Protocol This trial protocol has been provided by the authors to give readers additional information about their work. Protocol for: Wilt TJ, Brawer MK, Jones KM, et al. Radical prostatectomy versus observation
Personalized Therapy for Prostate Cancer due to Genetic Testings
 Personalized Therapy for Prostate Cancer due to Genetic Testings Stephen J. Freedland, MD Professor of Urology Director, Center for Integrated Research on Cancer and Lifestyle Cedars-Sinai Medical Center
Personalized Therapy for Prostate Cancer due to Genetic Testings Stephen J. Freedland, MD Professor of Urology Director, Center for Integrated Research on Cancer and Lifestyle Cedars-Sinai Medical Center
Genomics and Genetic Testing. Copyright 2017 Myriad Genetics, Inc., all rights reserved.
 Genomics and Genetic Testing Copyright 2017 Myriad Genetics, Inc., all rights reserved. www.myriad.com Why is genetic testing important to you? Impact on immediate treatment decisions Identify risks for
Genomics and Genetic Testing Copyright 2017 Myriad Genetics, Inc., all rights reserved. www.myriad.com Why is genetic testing important to you? Impact on immediate treatment decisions Identify risks for
Measure Name: Prostate Cancer: Cancer Surveillance Owner: NQF (#625) Steward: Active Health Management
 Measure Name: Prostate Cancer: Cancer Surveillance Owner: NQF (625) Measure Code: PRS Rule : Applicable Provider Specialty: General Criteria Summary Steward: Active Health Management Lab Data: Y Percentage
Measure Name: Prostate Cancer: Cancer Surveillance Owner: NQF (625) Measure Code: PRS Rule : Applicable Provider Specialty: General Criteria Summary Steward: Active Health Management Lab Data: Y Percentage
Prostate MRI: Who needs it?
 Prostate MRI: Who needs it? Fergus Coakley MD, Professor of Radiology and Urology, Vice Chair for Clinical Services, Chief of Abdominal Imaging, UCSF Abdominal Imaging Magnetic Resonance Science Center
Prostate MRI: Who needs it? Fergus Coakley MD, Professor of Radiology and Urology, Vice Chair for Clinical Services, Chief of Abdominal Imaging, UCSF Abdominal Imaging Magnetic Resonance Science Center
Effective Date: 11/1/2018 Section: SUR Policy No: 420 Medical Policy Committee Approved Date: 8/18; 9/18
 PROVIDENCE HEALTH PLANS MEDICAL (HIFU) (All Lines of Business Except Medicare) Effective Date: 11/1/2018 Section: SUR Policy No: 420 Medical Policy Committee Approved Date: 8/18; 9/18 11/1/18 Medical Officer
PROVIDENCE HEALTH PLANS MEDICAL (HIFU) (All Lines of Business Except Medicare) Effective Date: 11/1/2018 Section: SUR Policy No: 420 Medical Policy Committee Approved Date: 8/18; 9/18 11/1/18 Medical Officer
Measure #265: Biopsy Follow-Up National Quality Strategy Domain: Communication and Care Coordination
 Measure #265: Biopsy Follow-Up National Quality Strategy Domain: Communication and Care Coordination 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY DESCRIPTION: Percentage of new patients whose
Measure #265: Biopsy Follow-Up National Quality Strategy Domain: Communication and Care Coordination 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY DESCRIPTION: Percentage of new patients whose
Commissioning Urology Services in New NHS
 Commissioning Urology Services in New NHS Oct 2013 Commissioning Urology Services NHS England s Role Role of the new Clinical Reference Groups Current Issues / Work Programme for Urology Clinical Reference
Commissioning Urology Services in New NHS Oct 2013 Commissioning Urology Services NHS England s Role Role of the new Clinical Reference Groups Current Issues / Work Programme for Urology Clinical Reference
Technology appraisal guidance Published: 21 November 2018 nice.org.uk/guidance/ta546
 Padeliporfin for untreated localised prostate cancer Technology appraisal guidance Published: 21 November 2018 nice.org.uk/guidance/ta546 NICE 2019. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Padeliporfin for untreated localised prostate cancer Technology appraisal guidance Published: 21 November 2018 nice.org.uk/guidance/ta546 NICE 2019. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
NCDB Special Study: Post-Active Treatment Surveillance in Prostate Cancer. Project Overview Ronald C. Chen, MD MPH
 NCDB Special Study: Post-Active Treatment Surveillance in Prostate Cancer Project Overview Ronald C. Chen, MD MPH Introductions Principal Investigator: Ronald C. Chen, MD MPH ACS Staff: Eileen Tonner,
NCDB Special Study: Post-Active Treatment Surveillance in Prostate Cancer Project Overview Ronald C. Chen, MD MPH Introductions Principal Investigator: Ronald C. Chen, MD MPH ACS Staff: Eileen Tonner,
Localized Prostate Cancer Have we finally got it right? Shingai Mutambirwa Professor & Chair-Division Urology DGMAH & SMU Pretoria SOUTH AFRICA
 Localized Prostate Cancer Have we finally got it right? Shingai Mutambirwa Professor & Chair-Division Urology DGMAH & SMU Pretoria SOUTH AFRICA ESMO Cape Town 14 Feb 2018 Disclosures Advisory boards/lecturer/consultant-
Localized Prostate Cancer Have we finally got it right? Shingai Mutambirwa Professor & Chair-Division Urology DGMAH & SMU Pretoria SOUTH AFRICA ESMO Cape Town 14 Feb 2018 Disclosures Advisory boards/lecturer/consultant-
Consensus and Controversies in Cancer of Prostate BASIS FOR FURHTER STUDIES. Luis A. Linares MD FACRO Medical Director
 BASIS FOR FURHTER STUDIES Main controversies In prostate Cancer: 1-Screening 2-Management Observation Surgery Standard Laparoscopic Robotic Radiation: (no discussion on Cryosurgery-RF etc.) Standard SBRT
BASIS FOR FURHTER STUDIES Main controversies In prostate Cancer: 1-Screening 2-Management Observation Surgery Standard Laparoscopic Robotic Radiation: (no discussion on Cryosurgery-RF etc.) Standard SBRT
Radical Prostatectomy: Management of the Primary From Localized to Oligometasta:c Disease
 Radical Prostatectomy: Management of the Primary From Localized to Oligometasta:c Disease Disclosures I do not have anything to disclose Sexual function causes moderate to severe distress 2 years after
Radical Prostatectomy: Management of the Primary From Localized to Oligometasta:c Disease Disclosures I do not have anything to disclose Sexual function causes moderate to severe distress 2 years after
Attachment #2 Overview of Follow-up
 Attachment #2 Overview of Follow-up Provided below is a general overview of follow-up and this may vary based on specific patient or cancer characteristics. Of note, Labs and imaging can be performed closer
Attachment #2 Overview of Follow-up Provided below is a general overview of follow-up and this may vary based on specific patient or cancer characteristics. Of note, Labs and imaging can be performed closer
2016 Physician Quality Reporting System Data Collection Form: Sinusitis (for patients aged 18 and older)
 2016 Physician Quality Reporting System Data Collection Form: Sinusitis (for patients aged 18 and older) IMPORTANT: Any measure with a 0% performance rate (100% for inverse measures) is not considered
2016 Physician Quality Reporting System Data Collection Form: Sinusitis (for patients aged 18 and older) IMPORTANT: Any measure with a 0% performance rate (100% for inverse measures) is not considered
Prostate Cancer: 2010 Guidelines Update
 Prostate Cancer: 2010 Guidelines Update James L. Mohler, MD Chair, NCCN Prostate Cancer Panel Associate Director for Translational Research, Professor and Chair, Department of Urology, Roswell Park Cancer
Prostate Cancer: 2010 Guidelines Update James L. Mohler, MD Chair, NCCN Prostate Cancer Panel Associate Director for Translational Research, Professor and Chair, Department of Urology, Roswell Park Cancer
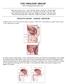
A schematic of the rectal probe in contact with the prostate is show in this diagram.
 Hello. My name is William Osai. I am a nurse practitioner in the GU Medical Oncology Department at The University of Texas MD Anderson Cancer Center in Houston. Today s presentation is Part 2 of the Overview
Hello. My name is William Osai. I am a nurse practitioner in the GU Medical Oncology Department at The University of Texas MD Anderson Cancer Center in Houston. Today s presentation is Part 2 of the Overview
Measure #72 (NQF 0385): Colon Cancer: Chemotherapy for AJCC Stage III Colon Cancer Patients National Quality Strategy Domain: Effective Clinical Care
 Measure #72 (NQF 0385): Colon Cancer: Chemotherapy for AJCC Stage III Colon Cancer Patients National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS F INDIVIDUAL MEASURES: CLAIMS, REGISTRY
Measure #72 (NQF 0385): Colon Cancer: Chemotherapy for AJCC Stage III Colon Cancer Patients National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS F INDIVIDUAL MEASURES: CLAIMS, REGISTRY
Meaningful Use Clinical Quality Measures for Eligible Professionals
 Meaningful Use Clinical Quality Measures for Eligible Professionals Measure Type NQF ID CMS ID Description Title: Adult Weight Screening and Follow-Up 1 NQF 0421 PQRI 128 calculated BMI in the past six
Meaningful Use Clinical Quality Measures for Eligible Professionals Measure Type NQF ID CMS ID Description Title: Adult Weight Screening and Follow-Up 1 NQF 0421 PQRI 128 calculated BMI in the past six
Prostate Cancer Case Study 1. Medical Student Case-Based Learning
 Prostate Cancer Case Study 1 Medical Student Case-Based Learning The Case of Mr. Powers Prostatic Nodule The effervescent Mr. Powers is found by his primary care provider to have a prostatic nodule. You
Prostate Cancer Case Study 1 Medical Student Case-Based Learning The Case of Mr. Powers Prostatic Nodule The effervescent Mr. Powers is found by his primary care provider to have a prostatic nodule. You
Clinically Localized Prostate Cancer: AUA/ASTRO/SUO Guideline High-Risk Disease
 Clinically Localized Prostate Cancer: AUA/ASTRO/SUO Guideline High-Risk Disease Mary-Ellen Taplin, MD Associate Professor of Medicine Harvard Medical School Director of Clinical Research, Lank Center for
Clinically Localized Prostate Cancer: AUA/ASTRO/SUO Guideline High-Risk Disease Mary-Ellen Taplin, MD Associate Professor of Medicine Harvard Medical School Director of Clinical Research, Lank Center for
THE HISTORY AND EVOLUTION OF PROSTATE CANCER DIAGNOSIS AND TREATMENT BY: DR. ANDREW GROLLMAN ALBUQUERQUE UROLOGY ASSOCIATES
 THE HISTORY AND EVOLUTION OF PROSTATE CANCER DIAGNOSIS AND TREATMENT BY: DR. ANDREW GROLLMAN ALBUQUERQUE UROLOGY ASSOCIATES OVERVIEW Diagnosis Laboratory Tests PSA Free and Total PSA PCA-3 4K Score The
THE HISTORY AND EVOLUTION OF PROSTATE CANCER DIAGNOSIS AND TREATMENT BY: DR. ANDREW GROLLMAN ALBUQUERQUE UROLOGY ASSOCIATES OVERVIEW Diagnosis Laboratory Tests PSA Free and Total PSA PCA-3 4K Score The
EORTC radiation Oncology Group Intergroup collaboration with RTOG EORTC 1331-ROG; RTOG 0924
 EORTC radiation Oncology Group Intergroup collaboration with RTOG EORTC 1331-ROG; RTOG 0924 Title of the Study Medical Condition Androgen deprivation therapy and high dose radiotherapy with or without
EORTC radiation Oncology Group Intergroup collaboration with RTOG EORTC 1331-ROG; RTOG 0924 Title of the Study Medical Condition Androgen deprivation therapy and high dose radiotherapy with or without
To treat or not to treat: When to treat! A case presentation
 To treat or not to treat: When to treat! A case presentation Filip Ameye, MD,Phd Universitary Hospitals Leuven, Belgium Departement of Urology Prostate Center A case presentation Pt. 76 y. Mild LUTS (07/1999)
To treat or not to treat: When to treat! A case presentation Filip Ameye, MD,Phd Universitary Hospitals Leuven, Belgium Departement of Urology Prostate Center A case presentation Pt. 76 y. Mild LUTS (07/1999)
Causes of Raised PSA A very large prostate Gland Infection of urine or Prostate Gland Possibility of prostate Cancer
 Causes of Raised PSA A very large prostate Gland Infection of urine or Prostate Gland Possibility of prostate Cancer Gleason score Gleason score 2-4: well differentiated (seldom reported now): Low risk
Causes of Raised PSA A very large prostate Gland Infection of urine or Prostate Gland Possibility of prostate Cancer Gleason score Gleason score 2-4: well differentiated (seldom reported now): Low risk
Outcomes of Radical Prostatectomy in Thai Men with Prostate Cancer
 Original Article Outcomes of Radical Prostatectomy in Thai Men with Prostate Cancer Sunai Leewansangtong, Suchai Soontrapa, Chaiyong Nualyong, Sittiporn Srinualnad, Tawatchai Taweemonkongsap and Teerapon
Original Article Outcomes of Radical Prostatectomy in Thai Men with Prostate Cancer Sunai Leewansangtong, Suchai Soontrapa, Chaiyong Nualyong, Sittiporn Srinualnad, Tawatchai Taweemonkongsap and Teerapon
MR-US Fusion Guided Biopsy: Is it fulfilling expectations?
 MR-US Fusion Guided Biopsy: Is it fulfilling expectations? Kenneth L. Gage MD, PhD Assistant Member Department of Diagnostic Imaging and Interventional Radiology 4 th Annual New Frontiers in Urologic Oncology
MR-US Fusion Guided Biopsy: Is it fulfilling expectations? Kenneth L. Gage MD, PhD Assistant Member Department of Diagnostic Imaging and Interventional Radiology 4 th Annual New Frontiers in Urologic Oncology
MEDICAL POLICY. SUBJECT: BRACHYTHERAPY OR RADIOACTIVE SEED IMPLANTATION FOR PROSTATE CANCER POLICY NUMBER: CATEGORY: Technology Assessment
 MEDICAL POLICY SUBJECT: BRACHYTHERAPY OR PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy
MEDICAL POLICY SUBJECT: BRACHYTHERAPY OR PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy
2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
 Measure #258: Rate of Open Repair of Small or Moderate Non-Ruptured Abdominal Aortic Aneurysms (AAA) without Major Complications (Discharged to Home by Post-Operative Day #7) National Quality Strategy
Measure #258: Rate of Open Repair of Small or Moderate Non-Ruptured Abdominal Aortic Aneurysms (AAA) without Major Complications (Discharged to Home by Post-Operative Day #7) National Quality Strategy
Active surveillance for low-risk Prostate Cancer Compared with Immediate Treatment: A Canadian cost evaluation
 Active surveillance for low-risk Prostate Cancer Compared with Immediate Treatment: A Canadian cost evaluation Alice Dragomir, PhD Fabio Cury, MD Armen Aprikian, MD Introduction Clinical and economic burden
Active surveillance for low-risk Prostate Cancer Compared with Immediate Treatment: A Canadian cost evaluation Alice Dragomir, PhD Fabio Cury, MD Armen Aprikian, MD Introduction Clinical and economic burden
Subject Index. Androgen antiandrogen therapy, see Hormone ablation therapy, prostate cancer synthesis and metabolism 49
 OOOOOOOOOOOOOOOOOOOOOOOOOOOOOO Subject Index Androgen antiandrogen therapy, see Hormone ablation therapy, synthesis and metabolism 49 Bacillus Calmette-Guérin adjunct therapy with transurethral resection
OOOOOOOOOOOOOOOOOOOOOOOOOOOOOO Subject Index Androgen antiandrogen therapy, see Hormone ablation therapy, synthesis and metabolism 49 Bacillus Calmette-Guérin adjunct therapy with transurethral resection
Radical Prostatectomy:
 Overtreatment and undertreatment Radical Prostatectomy: An Emerging Standard of Care for High Risk Prostate Cancer Matthew R. Cooperberg, MD,MPH UCSF Radiation Oncology Update San Francisco, CA April 2,
Overtreatment and undertreatment Radical Prostatectomy: An Emerging Standard of Care for High Risk Prostate Cancer Matthew R. Cooperberg, MD,MPH UCSF Radiation Oncology Update San Francisco, CA April 2,
Clinically Localized Prostate Cancer: AUA/ASTRO/SUO Guideline Very Low-/Low-Risk Disease
 Clinically Localized Prostate Cancer: AUA/ASTRO/SUO Guideline Very Low-/Low-Risk Disease Jeffrey A. Cadeddu, MD Professor, Department of Urology UT Southwestern Medical Center Vice-Chair, AUA/ASTRO/SUO
Clinically Localized Prostate Cancer: AUA/ASTRO/SUO Guideline Very Low-/Low-Risk Disease Jeffrey A. Cadeddu, MD Professor, Department of Urology UT Southwestern Medical Center Vice-Chair, AUA/ASTRO/SUO
Department of Urology, Cochin hospital Paris Descartes University
 Technical advances in the treatment of localized prostate cancer Pr Michaël Peyromaure Department of Urology, Cochin hospital Paris Descartes University Introduction Curative treatments of localized prostate
Technical advances in the treatment of localized prostate cancer Pr Michaël Peyromaure Department of Urology, Cochin hospital Paris Descartes University Introduction Curative treatments of localized prostate
Physician Self-Referral: Recent Research from the Government Accountability Office (GAO)
 Physician Self-Referral: Recent Research from the Government Accountability Office (GAO) James C. Cosgrove, Ph.D. National Health Policy Forum July 18, 2014 Self-Referral: Background Physician self-referral
Physician Self-Referral: Recent Research from the Government Accountability Office (GAO) James C. Cosgrove, Ph.D. National Health Policy Forum July 18, 2014 Self-Referral: Background Physician self-referral
National Prostate Cancer Audit. Bill Cross June 2015
 National Prostate Cancer Audit Bill Cross June 2015 National Prostate Cancer Audit aim of assessing the process of care and its outcomes in men diagnosed with prostate cancer in England and Wales National
National Prostate Cancer Audit Bill Cross June 2015 National Prostate Cancer Audit aim of assessing the process of care and its outcomes in men diagnosed with prostate cancer in England and Wales National
Guideline Prostate cancer: diagnosis and management (update)
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Guideline Prostate cancer: diagnosis and management (update) Draft for consultation, December 0 This guideline covers diagnosing and managing prostate
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Guideline Prostate cancer: diagnosis and management (update) Draft for consultation, December 0 This guideline covers diagnosing and managing prostate
Prostate Cancer in men with germline DNA repair deficiency
 Prostate Cancer in men with germline DNA repair deficiency Bruce Montgomery, MD Professor, Medicine and Urology Univ Washington, Fred Hutchinson CRC VA Puget Sound HCS Disclosures Company Tokai, ESSA,
Prostate Cancer in men with germline DNA repair deficiency Bruce Montgomery, MD Professor, Medicine and Urology Univ Washington, Fred Hutchinson CRC VA Puget Sound HCS Disclosures Company Tokai, ESSA,
MEDICAL POLICY Genetic and Protein Biomarkers for Diagnosis and Risk Assessment of
 POLICY: PG0367 ORIGINAL EFFECTIVE: 08/26/16 LAST REVIEW: 09/27/18 MEDICAL POLICY Genetic and Protein Biomarkers for Diagnosis and Risk Assessment of Prostate Cancer GUIDELINES This policy does not certify
POLICY: PG0367 ORIGINAL EFFECTIVE: 08/26/16 LAST REVIEW: 09/27/18 MEDICAL POLICY Genetic and Protein Biomarkers for Diagnosis and Risk Assessment of Prostate Cancer GUIDELINES This policy does not certify
Focal Therapy is a Fool s Paradise : The whole prostate must be treated!
 Focal Therapy is a Fool s Paradise : The whole prostate must be treated! Ofer Yossepowitch, MD Head, Department of Urology Tel Aviv Sourasky Medical Center Preaching against focal therapy in a focal therapy
Focal Therapy is a Fool s Paradise : The whole prostate must be treated! Ofer Yossepowitch, MD Head, Department of Urology Tel Aviv Sourasky Medical Center Preaching against focal therapy in a focal therapy
NPQR Quality Payment Program (QPP) Measures 21_18247_LS.
 NPQR Quality Payment Program (QPP) Measures 21_18247_LS MEASURE ID: QPP 99 MEASURE TITLE: Breast Cancer Resection Pathology Reporting pt Category (Primary Tumor) and pn Category (Regional Lymph Nodes)
NPQR Quality Payment Program (QPP) Measures 21_18247_LS MEASURE ID: QPP 99 MEASURE TITLE: Breast Cancer Resection Pathology Reporting pt Category (Primary Tumor) and pn Category (Regional Lymph Nodes)
2017 Merit-based Incentive Payment System. Avoiding the Penalty
 2017 Merit-based Incentive Payment System Avoiding the Penalty 1 What is the Quality Reporting Program? Quality Payment Program (also known as MACRA) Advanced Alternative Payment Models (APMs) Merit-based
2017 Merit-based Incentive Payment System Avoiding the Penalty 1 What is the Quality Reporting Program? Quality Payment Program (also known as MACRA) Advanced Alternative Payment Models (APMs) Merit-based
PET imaging of cancer metabolism is commonly performed with F18
 PCRI Insights, August 2012, Vol. 15: No. 3 Carbon-11-Acetate PET/CT Imaging in Prostate Cancer Fabio Almeida, M.D. Medical Director, Arizona Molecular Imaging Center - Phoenix PET imaging of cancer metabolism
PCRI Insights, August 2012, Vol. 15: No. 3 Carbon-11-Acetate PET/CT Imaging in Prostate Cancer Fabio Almeida, M.D. Medical Director, Arizona Molecular Imaging Center - Phoenix PET imaging of cancer metabolism
When PSA fails. Urology Grand Rounds Alexandra Perks. Rising PSA after Radical Prostatectomy
 When PSA fails Urology Grand Rounds Alexandra Perks Rising PSA after Radical Prostatectomy Issues Natural History Local vs Metastatic Treatment options 1 10 000 men / year in Canada 4000 RRP 15-year PSA
When PSA fails Urology Grand Rounds Alexandra Perks Rising PSA after Radical Prostatectomy Issues Natural History Local vs Metastatic Treatment options 1 10 000 men / year in Canada 4000 RRP 15-year PSA
American College of Physicians Genesis Registry
 Powered by Premier American College of Physicians Genesis Registry This registry has been approved by CMS as a Qualified Clinical Data Registry (QCDR) for Eligible Clinicians and group practices for the
Powered by Premier American College of Physicians Genesis Registry This registry has been approved by CMS as a Qualified Clinical Data Registry (QCDR) for Eligible Clinicians and group practices for the
Measure ID Domain Measure Title Measure Description/Definition Numerator Denominator Denominator Exclusions Denominator Exceptions
 n-qpp s ID Domain Title Description/Definition Numerator Denominator Denominator Exclusions Denominator Exceptions Riskadjustment AQUA3 (inverse) Cryptorchidism: Inappropriate use of scrotal/groin ultrasound
n-qpp s ID Domain Title Description/Definition Numerator Denominator Denominator Exclusions Denominator Exceptions Riskadjustment AQUA3 (inverse) Cryptorchidism: Inappropriate use of scrotal/groin ultrasound
Radical prostate surgery?
 Decipher enables personalized and actionable treatment after surgery Radical prostate surgery? The Decipher Prostate Cancer Classifier can help you and your doctor decide on important next steps in your
Decipher enables personalized and actionable treatment after surgery Radical prostate surgery? The Decipher Prostate Cancer Classifier can help you and your doctor decide on important next steps in your
