Cook Medical. Zenith Flex AAA Endovascular Graft with Z-Trak Introduction System Physician Training
|
|
|
- August Terry
- 6 years ago
- Views:
Transcription
1 Cook Medical Zenith Flex AAA Endovascular Graft with Z-Trak Introduction System Physician Training
2 Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full-thickness, woven polyester graft material Fully supported by self-expanding z-stents Z-Trak Introduction System Flex design, increased gap length provide greater conformability Hydrophilic Flexor Introducer Sheath (main body graft only) Captor Hemostatic Valve
3 Development 1993 Aorto-aortic, uncovered proximal fixation stent 1994 Bifurcated two-piece prosthesis, fully stented; transfer capsule, top cap 1995 External stents, running suture attachment 1996 H&L-B One-Shot Introduction System; longer overlap zone 1997 Bifurcated, 3-piece prosthesis with current proximal fixation stent 1999 Shorter stents in limbs, longer gaps between limb stents 2000 Pivotal U.S. trial began 2003 FDA approval for commercial release (May 23, 2003) 2006 Addition of 36 mm diameter main body 2007 Introduction of Zenith Flex 2008 Introduction of the Z-Trak Introduction System and Flex legs
4 Hydrophilic Flexor Introducer Sheath
5 Captor Hemostatic Valve Indicates valve closed position Indicates valve open position
6 Design Increased gap length incorporated into the proximal segment of the main body provides greater conformance to angulated anatomy. 5 mm gaps (6 mm gaps for 36 mm diameter stent grafts)
7 Main Body Endovascular Graft TFFB-diameter-length-ZT TFLE-diameter-length-ZT Ancillary Components ESC-diameter-diameter-length ZIP-diameter-length ESBE-diameter-length ESLE-diameter-length
8 Main Body Diameter (mm) cl Length (mm) Fr ID (6.0 mm ID/7.1 mm OD) Z-Trak Introduction System 20 Fr ID (6.7 mm ID/7.7 mm OD) Z-Trak Introduction System Fr ID (7.3 mm ID/8.5 mm OD) Z-Trak Introduction System 4 or 5 lengths + 7 diameters = 34 main body sizes Sheath length = 40 cm Sheath + valve length = 51 cm
9 Iliac Legs Length (mm) Diameter (mm) Fr ID (4.7 mm ID/5.3 mm OD) Z-Trak Introduction System Fr ID (5.3 mm ID/6.0 mm OD) Z-Trak Introduction System X X X X X X X X X X 9 diameters + 4 or 6 lengths = 44 iliac leg sizes Sheath length = 71 cm Sheath + valve length = 82 cm
10 Features of the Zenith Flex AAA Endovascular Graft Unique Stent Graft Design Independent, self-expanding stainless steel z-stent configuration provides: graft flexibility secure graft/vessel apposition dimensional stability
11 Features of the Zenith Flex AAA Endovascular Graft Four Gold Proximal Markers 2 mm from most proximal segment of graft material Contralateral Limb Gold Marker Contralateral limb check mark
12 Stent Lengths Main Body Suprarenal stent Proximal sealing stent External stents First stent after bifurcation Distal limb stents 2-5 mm gaps between stents throughout graft for mm diameters 26 17/22 14/17 14/17 14/17 14/17 14/ mm gaps between stents throughout graft for 36 mm diameters NOTE: Measurements specific to the 36 mm diameter stent graft are in red. * 11 mm for stent grafts with a 22 mm proximal diameter * 12 * Lengths in mm
13 Stent Lengths Iliac Leg 12 Sealing stent to main body External stents Distal sealing stent mm gaps between stents throughout graft Lengths in mm
14 Ancillary Components Uses To correct inaccuracies during device size selection To compensate for difficult anatomy encountered during procedure Types Converter (ESC, four sizes) Iliac Plug (ZIP, four sizes) Main Body Extension (ESBE, 14 sizes) Iliac Leg Extension (ESLE, 9 sizes)
15 Deployment Sequence
16 Prior to Use Inspect device and packaging for shipping damage. Do not use if damage has occurred or sterilization barrier has been broken. Verify that correct devices (quantity and size) have been supplied for the patient. Match the device to the prescribed order for each patient. Prepare systems for insertion.
17 Prepare Main Body and Iliac Leg Systems Remove the three shipping protectors. Hubbed shipping stylet Dilator tip protector Peel-Away sheath
18 Prepare Main Body and Iliac Leg Systems (cont.) Elevate distal tip of system and flush: wire lumen through hub sheath through stopcock Wipe Flexor Introducer Sheath to activate hydrophilic coating on the main body delivery system
19 Step 1 Orient main body delivery system using fluoroscopy. Contralateral limb LATERAL = anterior = posterior = lateral LATERAL
20 Step 2 Introduce and position device below the lowest renal artery and verify orientation.* * Perform angiograms as needed throughout procedure.
21 Step 3 Deploy first two covered stents by retracting sheath. Check position relative to renal arteries and aortic bifurcation. Orient using stent markers.
22 Step 4 Continue sheath retraction until contralateral limb is fully deployed. NOTE: Flexor Introducer Sheath must be stabilized at all times.
23 Step 5 Cannulate contralateral limb. Perform angiogram with catheter tip at proximal orifice. Locate renal arteries. Verify catheter position (within graft). Check wire guide position.
24 Step 6 Remove top stent trigger wire. Deploy suprarenal stent (loosen pin vise and advance inner cannula, retighten pin vise).
25 Step 7 Withdraw catheter into body of graft. Advance wire guide through catheter. Perform angiogram to locate contralateral internal iliac artery.
26 Step 8 Introduce/advance leg to achieve stent overlap. Check distal position/deploy. Retract inner cannula; dock and remove grey positioner.
27 Step 9 Fully deploy main body (complete retraction of sheath). Remove ipsilateral limb trigger wire.
28 Step 10 Dock top cap Loosen pin vise and secure both the inner cannula and sheath. Advance and dock grey positioner with top cap. Tighten pin vise. Remove main body delivery system.
29 Step 11 Introduce/advance ipsilateral leg to achieve minimum of one stent overlap. Perform angiogram through large sheath. Check distal position/deploy. Retract inner cannula; dock and remove grey positioner.
30 Step 12 Balloon-mold the graft at vessel seal sites and component overlap sites. Perform final angiogram. Do not inflate the balloon in vessel outside of the graft, as doing so may cause damage to the vessel. Use care in inflating the balloon within the graft in the presence of calcification, as excessive inflation may cause damage to the vessel. NOTE: Remove or replace all stiff wire guides to allow iliac arteries to resume their natural position. NOTE: Confirm there are no endoleaks or kinks and verify position of proximal gold radiopaque markers. BALLOON EXPANSION/GRAFT SEALING SITES
31 Deployment Animation
32 Patient Selection
33 Patient Selection The objective of patient selection is to determine if the patient is: a candidate for AAA repair a candidate for endovascular therapy a candidate for implantation of the Zenith Flex AAA Endovascular Graft Contraindications The Zenith Flex AAA Endovascular Graft with the Z-Trak Introduction System is contraindicated in: patients with known sensitivities or allergies to stainless steel, polyester, solder (tin, silver), polypropylene or gold patients with a systemic infection who may be at increased risk of endovascular graft infection
34 Patient Selection Ideal Morphology Adequate iliac/femoral access compatible with the required introduction systems Nonaneurysmal infrarenal neck that has: length 15 mm diameter, measured outer wall to outer wall, of no greater than 32 mm and no less than 18 mm angle < 60º relative to long axis of aneurysm angle < 45º relative to axis of suprarenal aorta Iliac artery distal fixation site that has: length > 10 mm (preferably > 20 mm) diameter mm (outer wall to outer wall)
35 Patient Selection
36 Device Planning
37 Device Planning Obtain recommended CT and angiography. Follow five recommended steps: 1. Select the side for main body introduction and fixation sites. 2. Obtain and insert anatomical measurements on the worksheet. 3. Select the main body. 4. Select the contralateral iliac leg. 5. Select the ipsilateral iliac leg.
38 Device Planning Measurements needed: 3 diameters (D1, D2, D3) 3 lengths (L1, L2, L3)
39 Device Planning Diameter Measurements outer wall to outer wall D1, D2, D3 Proximal neck and common iliac diameters from axial CT images should be measured from outer wall to outer wall, using shortest axis. EI External iliac diameters from axial CT images should be measured from inner wall to inner wall to assure delivery system access. Vessel size must be compatible with the required introduction systems.
40 Device Planning Diameter Oversizing Diameters for components are oversized. Main body diameters are generally oversized 3-4 mm. Iliac leg diameters are generally oversized 1-2 mm. Use the sizing tables on the Device Planning and Ordering Worksheet to select components with proper oversizing.
41 Device Planning Overlap Contralateral leg stents Ipsilateral 1-2 stents (37, 39, 54 or 56 mm) or 1-3 stents (others)
42 Device Planning Lengths Use CT scan and/or angiography with calibrated catheter. Do not oversize lengths. Use actual lengths. If necessary, select shorter graft length.
43 Device Planning and Ordering Worksheet
44 Device Planning and Ordering Worksheet
45 Device Planning and Ordering Worksheet Step 1 Select the side for main body introduction and fixation sites. Generally, place the main body through the side that has the best access vessel. Factors to consider: Iliac tortuosity Angulation of a distal neck Mural thrombus within the aneurysm Iliac length (short iliac contralateral) Vessel diameter (EI) Mural thrombus within Iliofemoral disease (e.g., stenosis, calcium) Iliac aneurysm (ipsilateral)
46 Device Planning and Ordering Worksheet Step 1
47 Device Planning and Ordering Worksheet Step 2 Obtain anatomical measurements Diameters D1 - Largest aortic neck diameter throughout 15 mm neck length D2 - Largest iliac diameter throughout contralateral distal fixation site D3 - Largest iliac diameter throughout ipsilateral distal fixation site Lengths L1 - Lowest renal artery to aortic bifurcation + lateral deviation/tortuosity L2 - Lowest renal artery to contralateral distal fixation site + lateral deviation/tortuosity L3 - Lowest renal artery to ipsilateral distal fixation site + lateral deviation/tortuosity
48 Device Planning and Ordering Worksheet Step 2
49 Device Planning and Ordering Worksheet Step 3 Select main body From D1, select graft diameter. Table includes oversizing. From L1, select graft lengths. Table provides cl and il lengths, and includes minimum of 5 mm clearance for cl limb. If choice of graft diameter or graft length is affected by other considerations, adjust accordingly (see manual).
50 Device Planning and Ordering Worksheet Step 3
51 Device Planning and Ordering Worksheet Step 4 Select contralateral iliac leg From D2, select graft diameter. Table includes oversizing. L2 cl length = contralateral working length. Using contralateral working length, select graft length. Consider stent overlap and a secure fixation site. If choice is affected by other considerations, adjust accordingly.
52 Device Planning and Ordering Worksheet Step 4
53 Device Planning and Ordering Worksheet Step 5 Select ipsilateral iliac leg From D3, select graft diameter. Table includes oversizing. L3 il length = ipsilateral working length. Using ipsilateral working length, select graft length. Consider stent overlap and a secure fixation site. If choice is affected by other considerations, adjust accordingly.
54 Device Planning and Ordering Worksheet Step 5
55 Device Planning and Ordering Worksheet Devices to Order
56 Ancillary Components
57 Zenith Flex Endovascular Graft Ancillary Components
58 Converter
59 Converter Converters can be used to convert a bifurcated graft into an aorto-uniiliac graft, if necessary (e.g., cases of Type III endoleak, limb occlusion or unattainable contralateral limb cannulation). The converter includes four internal z-stents. The length of the 24, 28 and 32 mm diameter converters is 80 mm. The length of the 36 mm diameter converter is 82 mm. Distal diameter of all converters is 12 mm. Proximal diameters are 24, 28, 32 or 36 mm. A converter, if used, should have a proximal diameter no smaller than that of the main body.
60 Converter (cont.) Place the converter sealing stent exactly within the main body to assure adequate graft-to-graft seal. If implanted properly, the two distal stents on the converter should land within the ipsilateral iliac limb of the main body. Converter introducer sheaths are inserted over the wire and cannot be introduced through the sheath of a main body or an iliac leg. 1. Hub 5. Connecting Tube 9. Hemostatic Valve 2. Pin Vise 6. Sheath 10. Grey Positioner 3. Peel-Away Sheath 7. Dilator Tip 11. Inner Cannula 4. Stopcock 8. Sheath Sideport 12. Converter
61 Converter (cont.) Deployment Step 1 Verify appropriate position to ensure proper sealing (proximal two stents in main body, distal two stents in ipsilateral iliac limb).
62 Converter (cont.) Deployment Step 2 Stabilize grey positioner and retract sheath to deploy converter.
63 Converter (cont.) Deployment Step 3 Deploy until distal stent is uncovered. Retract inner cannula; dock and remove grey positioner.
64 Converter (cont.) Deployment Step 4 Balloon-mold the converter within the proximal segment and then the distal segment. Perform final angiogram. BALLOON EXPANSION/GRAFT SEALING SITES
65 Iliac Plug
66 Iliac Plug The iliac plug is used to occlude an iliac artery when the converter system has been placed and/or is in conjunction with a femoral-to-femoral crossover procedure. The length of all iliac plugs is 30 mm. Available diameters are 14, 16, 20 and 24 mm. Iliac plug diameter should be determined from diameter of the intended fixation site (outer wall to outer wall) and the next larger-sized component selected. An iliac plug should generally have a diameter at least 2 mm larger than that of the iliac artery.
67 Iliac Plug (cont.) The iliac plug is preloaded into 14 or 16 Fr H&L-B One-Shot Introduction Systems. The H&L-B One-Shot Introduction System for the iliac plug has a single trigger-wire release mechanism. Iliac plug introduction systems are inserted over a wire. Do not release and remove the trigger wire until the iliac plug has been deployed from the sheath. 1. Stopcock 5. Connecting Tube 9. Tuohy-Borst Adapter 2. Trigger-Wire Release 6. Sheath 10. Iliac Plug 3. Peel-Away Sheath 7. Hemostatic Valve 4. Stopcock 8. Grey Positioner
68 Iliac Plug (cont.) Deployment Step 1 Verify position with angiography to determine proper placement. Advance the delivery system over the wire to the intended position in the common iliac artery.
69 Iliac Plug (cont.) Deployment Step 2 Stabilize grey positioner and retract sheath to deploy the iliac plug. Withdraw the sheath until the iliac plug is uncovered.
70 Iliac Plug (cont.) Deployment Step 3 Unscrew the trigger-wire release knob. Withdraw the wire completely.
71 Iliac Plug (cont.) Deployment Step 4 Remove wire guide. Ensure graft is not displaced during withdrawal. Remove grey positioner. Ensure graft is not displaced during withdrawal. (Optional) Balloon-mold the expandable length of the iliac plug. Perform final angiogram. BALLOON EXPANSION/GRAFT SEALING SITES
72 Main Body Extension (ESBE)
73 Main Body Extension Used to treat a Type I endoleak, which may be caused by: Improper placement of the main body Tortuosity of the aortic neck Inaccurate selection of main body diameter
74 Main Body Extension (cont.) Main body extensions include two or three z-stents. Main body extensions are 39, 50*, 58 or 73* mm long. Diameters include 22, 24, 26, 28, 30, 32 or 36 mm. A main body extension, if used, should have a diameter no smaller than that of the main body. In selecting the diameter of a main body extension, consider: neck shape neck angulation diameter of existing main body *Lengths for 36 mm diameter only.
75 Main Body Extension (cont.) The H&L-B One-Shot Introduction System for the main body extension has a single trigger-wire release mechanism. Do not release and remove the trigger wire until the main body extension has been deployed from the sheath. Main body extension introduction systems are inserted over a wire and cannot be introduced through the sheath of a main body or an iliac leg. 1. Hub 6. Connecting Tube 11. Grey Positioner 2. Pin Vise 7. Sheath 12. White Trigger-Wire Release Mechanism 3. Safety Lock 8. Dilator Tip 13. Inner Cannula 4. Peel-Away Sheath 9. Sheath Sideport 14. Main Body Extension 5. Stopcock 10. Hemostatic Valve
76 Main Body Extension (cont.) Deployment Step 1 Verify position to ensure proper sealing and resistance to migration. Verify position with angiography to ensure renal arteries remain patent.
77 Main Body Extension (cont.) Deployment Step 2 Stabilize grey positioner and retract sheath to deploy main body extension. Deploy device until the most distal stent is uncovered.
78 Main Body Extension (cont.) Deployment Step 3 Remove white trigger wire. Retract inner cannula; dock and remove grey positioner.
79 Main Body Extension (cont.) Deployment Step 4 Balloon-mold the main body extension within the proximal segment and then the distal segment. Perform final angiogram. BALLOON EXPANSION/GRAFT SEALING SITES
80 Iliac Leg Extension (ESLE)
81 Iliac Leg Extension Used to lengthen and/or bridge the iliac leg(s) of an in situ endovascular graft. Iliac leg extensions include three internal z-stents. Diameters include 8, 10, 12, 14, 16, 18, 20, 22 or 24 mm. An iliac leg extension, if used, should have a diameter no smaller than that of the in situ iliac leg graft. If an iliac leg extension is not available, an iliac leg can be used.
82 Iliac Leg Extension (cont.) Iliac leg extensions are preloaded into an H&L-B One-Shot Introduction System mm iliac leg extension introducer sheaths can be inserted either through an in situ main body introducer sheath or directly into the vessel. The 22 and 24 mm iliac leg extension introducer sheaths should not be inserted into a main body introducer sheath. 1. Hub 5. Connecting Tube 9. Hemostatic Valve 2. Pin Vise 6. Sheath 10. Grey Positioner 3. Peel-Away Sheath 7. Dilator Tip 11. Inner Cannula 4. Stopcock 8. Sheath Sideport 12. Iliac Leg Extension
83 Iliac Leg Extension (cont.) Deployment Step 1 Verify appropriate stent graft overlap to ensure proper sealing and resistance to migration. Verify placement with angiography to ensure the internal iliacs will remain patent.
84 Iliac Leg Extension (cont.) Deployment Step 2 Stabilize grey positioner and retract sheath to deploy iliac leg extension.
85 Iliac Leg Extension (cont.) Deployment Step 3 Continue to deploy the device until the distal stent is uncovered. Retract inner cannula; dock and remove grey positioner.
86 Iliac Leg Extension (cont.) Deployment Step 4 Balloon-mold the iliac leg extension within the most proximal segment and then the most distal segment. Perform final angiogram.
87 Postoperative Information
88 Follow-up Imaging Guidelines All patients should be advised that: endovascular treatment requires lifelong, regular follow-up to assess performance adhering to the follow-up schedule is critical in ensuring the ongoing safety and effectiveness of endovascular treatment subsequent reinterventions, including catheter-based and open surgical conversion, are possible following endograft placement
89 Minimum Follow-up Imaging Recommendations CT (Contrast and noncontrast)* Abdominal Radiographs Predischarge (within 7 days) X X 1 month X 3 months X if endoleak at previous CT 6 months X X 12 months (annually thereafter) X X * Duplex ultrasound and noncontrast CT may be used for patients who have renal failure or are otherwise unable to have contrast-enhanced CT. * If Type I or III endoleak, prompt intervention and additional postintervention follow-up recommended.
Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully
 Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Ancillary Components with Z-Trak Introduction System
 Ancillary Components with Z-Trak Introduction System Zenith Flex AAA Endovascular Graft Ancillary Components Converter Converters can be used to convert a bifurcated graft into an aortouniiliac graft if
Ancillary Components with Z-Trak Introduction System Zenith Flex AAA Endovascular Graft Ancillary Components Converter Converters can be used to convert a bifurcated graft into an aortouniiliac graft if
Zenith Renu AAA Converter Graft. Device Description Planning and Sizing Deployment Sequence Patient Follow-Up
 Zenith Renu AAA Converter Graft Device Description Planning and Sizing Deployment Sequence Patient Follow-Up Device description: Device indications The Zenith Renu AAA Converter Graft with Z-Trak Introduction
Zenith Renu AAA Converter Graft Device Description Planning and Sizing Deployment Sequence Patient Follow-Up Device description: Device indications The Zenith Renu AAA Converter Graft with Z-Trak Introduction
Talent Abdominal Stent Graft
 Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Zenith Alpha T HORACIC ENDOVASCULAR GRAFT
 Device description Zenith Alpha T HORACIC ENDOVASCULAR GRAFT www.cookmedical.com AI-D21181-EN-F Modular design The two-piece modular system allows the physician to customize a graft system to fit each
Device description Zenith Alpha T HORACIC ENDOVASCULAR GRAFT www.cookmedical.com AI-D21181-EN-F Modular design The two-piece modular system allows the physician to customize a graft system to fit each
Zenith Alpha T HORACIC ENDOVASCULAR GRAFT
 Deployment Sequence Zenith Alpha T HORACIC ENDOVASCULAR GRAFT www.cookmedical.com AI-D21183-EN-F Preparation and flush Proximal and distal components Remove the yellow hubbed inner stylet from the dilator
Deployment Sequence Zenith Alpha T HORACIC ENDOVASCULAR GRAFT www.cookmedical.com AI-D21183-EN-F Preparation and flush Proximal and distal components Remove the yellow hubbed inner stylet from the dilator
Robert F. Cuff, MD FACS SHMG Vascular Surgery
 Robert F. Cuff, MD FACS SHMG Vascular Surgery Objectives To become familiar with the commercially available fenestrated EVAR graft Discuss techniques to increase success Review available data to determine
Robert F. Cuff, MD FACS SHMG Vascular Surgery Objectives To become familiar with the commercially available fenestrated EVAR graft Discuss techniques to increase success Review available data to determine
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Access More Patients. Customize Each Seal.
 Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
Young-Guk Ko, M.D. Severance Cardiovascular Hospital, Yonsei University Health System,
 Young-Guk Ko, M.D., Dangas G, J Am Coll Cardiol Intv 2012;5:1071 All-cause Mortality Dangas Severance G, J Am Coll Cardiovascular Cardiol Intv Hospital, 2012;5:1071 Yonsei University Health System Aneurysm-related
Young-Guk Ko, M.D., Dangas G, J Am Coll Cardiol Intv 2012;5:1071 All-cause Mortality Dangas Severance G, J Am Coll Cardiovascular Cardiol Intv Hospital, 2012;5:1071 Yonsei University Health System Aneurysm-related
Hostile Proximal Neck: A New Conformable EVAR Device
 Hostile Proximal Neck: A New Conformable EVAR Device Young-Guk Ko, M.D., Seoul, Korea Currently Available Devices for EVAR in Korea, 2018 Zenith Flex, Cook Endurant IIs, Medtronic INCRAFT, Cordis AFX2,
Hostile Proximal Neck: A New Conformable EVAR Device Young-Guk Ko, M.D., Seoul, Korea Currently Available Devices for EVAR in Korea, 2018 Zenith Flex, Cook Endurant IIs, Medtronic INCRAFT, Cordis AFX2,
Aortic stents, types, selection, tricks in deployment.
 Aortic stents, types, selection, tricks in deployment. Hamdy Soliman.M.D,FSCAI Consultant of Cardiology&Head of Endovascular Unit National Heart Institute Endovascular Treatment of Thoracic Aortic Aneurysms
Aortic stents, types, selection, tricks in deployment. Hamdy Soliman.M.D,FSCAI Consultant of Cardiology&Head of Endovascular Unit National Heart Institute Endovascular Treatment of Thoracic Aortic Aneurysms
My personal experience with INCRAFT in standard and challenging cases
 My personal experience with INCRAFT in standard and challenging cases G Pratesi, MD Vascular Surgery University of Rome Tor Vergata giovanni.pratesi@uniroma2.it Disclosure Speaker name: Giovanni Pratesi,
My personal experience with INCRAFT in standard and challenging cases G Pratesi, MD Vascular Surgery University of Rome Tor Vergata giovanni.pratesi@uniroma2.it Disclosure Speaker name: Giovanni Pratesi,
Zenith Alpha Thoracic Endovascular Graft
 MEDICAL Zenith Alpha Thoracic Endovascular Graft Instructions for Use Patient I.D. Card Included I-ALPHA-THORACIC-442-03 *442-03* TABLE OF CONTENTS 1 DEVICE DESCRIPTION... 9 1.1 Zenith Alpha Thoracic Endovascular
MEDICAL Zenith Alpha Thoracic Endovascular Graft Instructions for Use Patient I.D. Card Included I-ALPHA-THORACIC-442-03 *442-03* TABLE OF CONTENTS 1 DEVICE DESCRIPTION... 9 1.1 Zenith Alpha Thoracic Endovascular
How to plan a Zenith AAA stent-graft from a CTA: Basic measurements and concepts explained
 How to plan a Zenith AAA stent-graft from a CTA: Basic measurements and concepts explained Poster No.: C-3077 Congress: ECR 2010 Type: Educational Exhibit Topic: Vascular Authors: D. V. Thomas; Northampton/UK
How to plan a Zenith AAA stent-graft from a CTA: Basic measurements and concepts explained Poster No.: C-3077 Congress: ECR 2010 Type: Educational Exhibit Topic: Vascular Authors: D. V. Thomas; Northampton/UK
FROM THE EVERYDAY TO THE EXTRAORDINARY
 FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
Challenging anatomies demand versatility.
 Challenging anatomies demand versatility. The Distinct Advantages of Separating Seal and Fixation ANATOMICAL FIXATION Unlike proximal fixation designs, the AFX bifurcated unibody endograft allows for natural
Challenging anatomies demand versatility. The Distinct Advantages of Separating Seal and Fixation ANATOMICAL FIXATION Unlike proximal fixation designs, the AFX bifurcated unibody endograft allows for natural
ENG TREO ABDOMINAL STENT-GRAFT SYSTEM TABLE OF CONTENTS
 TREO ABDOMINAL STENT-GRAFT SYSTEM TABLE OF CONTENTS 1. DEVICE DESCRIPTION 28 1.1. Stent-Graft 28 1.1.1. Main Bifurcated Stent-Graft 28 1.1.2. Leg Extensions 28 1.2. Delivery System for the Bifurcated Main
TREO ABDOMINAL STENT-GRAFT SYSTEM TABLE OF CONTENTS 1. DEVICE DESCRIPTION 28 1.1. Stent-Graft 28 1.1.1. Main Bifurcated Stent-Graft 28 1.1.2. Leg Extensions 28 1.2. Delivery System for the Bifurcated Main
A New EVAR Device for Infrarenal AAAs
 A New EVAR Device for Infrarenal AAAs Peter Nelson, MD, MS Assistant Professor of Surgery MM0203 Rev. 01 Current U.S. EVAR Devices Anatomical Fixation Proximal Fixation Powerlink - Endologix Excluder WL
A New EVAR Device for Infrarenal AAAs Peter Nelson, MD, MS Assistant Professor of Surgery MM0203 Rev. 01 Current U.S. EVAR Devices Anatomical Fixation Proximal Fixation Powerlink - Endologix Excluder WL
The clinical update for the Zenith AAA Endovascular Graft has included results from the Zenith AAA Endovascular Graft multi-center clinical study,
 The clinical update for the Zenith AAA Endovascular Graft has included results from the Zenith AAA Endovascular Graft multi-center clinical study, the 36 mm diameter Zenith Flex AAA Endovascular Graft
The clinical update for the Zenith AAA Endovascular Graft has included results from the Zenith AAA Endovascular Graft multi-center clinical study, the 36 mm diameter Zenith Flex AAA Endovascular Graft
Durable outcomes. Proven performance.
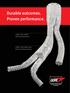 Durable outcomes. Proven performance. GORE EXCLUDER AAA Endoprosthesis GORE EXCLUDER Iliac Branch Endoprosthesis GORE EXCLUDER AAA Endoprosthesis The most-studied* EVAR stent graft designed for durable
Durable outcomes. Proven performance. GORE EXCLUDER AAA Endoprosthesis GORE EXCLUDER Iliac Branch Endoprosthesis GORE EXCLUDER AAA Endoprosthesis The most-studied* EVAR stent graft designed for durable
What's on the Horizon for AAA: Unilateral & Percutaneous, "UP-EVAR" System Zoran Rancic M.D., Ph.D.
 What's on the Horizon for AAA: Unilateral & Percutaneous, "UP-EVAR" System Zoran Rancic M.D., Ph.D. Clinic for Cardiovascular Surgery University Hospital Zurich DISCLOSURES COMMON SITUATIONS FOR UNILATERAL
What's on the Horizon for AAA: Unilateral & Percutaneous, "UP-EVAR" System Zoran Rancic M.D., Ph.D. Clinic for Cardiovascular Surgery University Hospital Zurich DISCLOSURES COMMON SITUATIONS FOR UNILATERAL
Endovascular Repair o Abdominal. Aortic Aneurysms. Cesar E. Mendoza, M.D. Jackson Memorial Hospital Miami, Florida
 Endovascular Repair o Abdominal Aortic Aneurysms Cesar E. Mendoza, M.D. Jackson Memorial Hospital Miami, Florida Disclosure Nothing to disclose. 2 Mr. X AAA Mr. X. Is a 70 year old male who presented to
Endovascular Repair o Abdominal Aortic Aneurysms Cesar E. Mendoza, M.D. Jackson Memorial Hospital Miami, Florida Disclosure Nothing to disclose. 2 Mr. X AAA Mr. X. Is a 70 year old male who presented to
RadRx Your Prescription for Accurate Coding & Reimbursement Copyright All Rights Reserved.
 Interventional Radiology Coding Case Studies Prepared by Stacie L. Buck, RHIA, CCS-P, RCC, CIRCC, AAPC Fellow President & Senior Consultant INDICATION: Abdominal aortic aneurysm. INTERVENTIONAL RADIOLOGIST:
Interventional Radiology Coding Case Studies Prepared by Stacie L. Buck, RHIA, CCS-P, RCC, CIRCC, AAPC Fellow President & Senior Consultant INDICATION: Abdominal aortic aneurysm. INTERVENTIONAL RADIOLOGIST:
LOWERING THE PROFILE RAISING THE BAR
 LOWERING THE PROFILE RAISING THE BAR INNOVATIVE LOW PROFILE. ADVANCED CLINICAL PERFORMANCE. The AFX TM Endovascular AAA System integrates anatomical fixation with an advanced delivery system and graft
LOWERING THE PROFILE RAISING THE BAR INNOVATIVE LOW PROFILE. ADVANCED CLINICAL PERFORMANCE. The AFX TM Endovascular AAA System integrates anatomical fixation with an advanced delivery system and graft
TriVascular Ovation Prime Abdominal Stent Graft System
 TriVascular Ovation Prime Abdominal Stent Graft System Science of the Seal O-Ring Sealing Technology O-Ring Sealing in Proven Engineering Solutions O-rings are designed to seal by blocking the flow of
TriVascular Ovation Prime Abdominal Stent Graft System Science of the Seal O-Ring Sealing Technology O-Ring Sealing in Proven Engineering Solutions O-rings are designed to seal by blocking the flow of
The evolution. AORFIX AAA Stent Graft now with the new AORFLEX Delivery System
 The evolution of AORFIX AORFIX AAA Stent Graft now with the new AORFLEX Delivery System The confident choice for the right angles AORFIX the only AAA stent graft approved for all angulations from 0 90,
The evolution of AORFIX AORFIX AAA Stent Graft now with the new AORFLEX Delivery System The confident choice for the right angles AORFIX the only AAA stent graft approved for all angulations from 0 90,
History of the Powerlink System Design and Clinical Results. Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ
 History of the Powerlink System Design and Clinical Results Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ Powerlink System: Unibody-Bifurcated Design Long Main Body Low-Porosity Proprietary eptfe
History of the Powerlink System Design and Clinical Results Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ Powerlink System: Unibody-Bifurcated Design Long Main Body Low-Porosity Proprietary eptfe
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Durability of The Endurant Stent-Graft through 5 Years
 Durability of The Endurant Stent-Graft through 5 Years Michel S. Makaroun MD Co-Director, UPMC Heart and Vascular Institute Professor and Chair, Division of Vascular Surgery University of Pittsburgh School
Durability of The Endurant Stent-Graft through 5 Years Michel S. Makaroun MD Co-Director, UPMC Heart and Vascular Institute Professor and Chair, Division of Vascular Surgery University of Pittsburgh School
Optimizing Accuracy of Aortic Stent Grafts in Short Necks
 Optimizing Accuracy of Aortic Stent Grafts in Short Necks Venkatesh Ramaiah, MD, FACS Medical Director Arizona Heart Hospital Director Peripheral Vascular and Endovascular Research Arizona Heart Institute
Optimizing Accuracy of Aortic Stent Grafts in Short Necks Venkatesh Ramaiah, MD, FACS Medical Director Arizona Heart Hospital Director Peripheral Vascular and Endovascular Research Arizona Heart Institute
Challenges in the treatment of infrarenal aneurysm & considerations for a durable repair
 Challenges in the treatment of infrarenal aneurysm & considerations for a durable repair Nilo J Mosquera, MD. Head of Department Angiology and Vascular Surgery. CHUO Hospital. Ourense. Spain Disclosure
Challenges in the treatment of infrarenal aneurysm & considerations for a durable repair Nilo J Mosquera, MD. Head of Department Angiology and Vascular Surgery. CHUO Hospital. Ourense. Spain Disclosure
GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER Section I Clinical experience. Section II Worldwide commercial experience
 GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER 2018 Abstract This annual clinical update provides a review of the ongoing experience with the GORE EXCLUDER AAA Endoprosthesis used in the
GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER 2018 Abstract This annual clinical update provides a review of the ongoing experience with the GORE EXCLUDER AAA Endoprosthesis used in the
Right Choice for Right Angles
 Right Choice for Right Angles The Anatomy of Technology Aorfix gives you technology that conforms to patient anatomy, optimising both procedure and post-operative performance. Fishmouth for optimum neck
Right Choice for Right Angles The Anatomy of Technology Aorfix gives you technology that conforms to patient anatomy, optimising both procedure and post-operative performance. Fishmouth for optimum neck
Anatomical challenges in EVAR
 Anatomical challenges in EVAR M.H. EL DESSOKI, MD,FRCS PROFESSOR OF VASCULAR SURGERY CAIRO UNIVERSITY Disclosure Speaker name:... I have the following potential conflicts of interest to report: Consulting
Anatomical challenges in EVAR M.H. EL DESSOKI, MD,FRCS PROFESSOR OF VASCULAR SURGERY CAIRO UNIVERSITY Disclosure Speaker name:... I have the following potential conflicts of interest to report: Consulting
Abdominal Aortic Aneurysm 가천대길병원 이상준
 Abdominal Aortic Aneurysm 가천대길병원 이상준 1 Definition Diameter of the aorta 1.5 times greater than normal. Most are infrarenal, and a significant number extend down into one or both iliac arteries Abdominal
Abdominal Aortic Aneurysm 가천대길병원 이상준 1 Definition Diameter of the aorta 1.5 times greater than normal. Most are infrarenal, and a significant number extend down into one or both iliac arteries Abdominal
The Ventana Off-the-Shelf Graft for Pararenal AAA. Andrew Holden Associate Professor of Radiology Auckland Hospital
 The Ventana Off-the-Shelf Graft for Pararenal AAA Andrew Holden Associate Professor of Radiology Auckland Hospital Disclosures Andrew Holden, MBChB, FRANZCR Investigator in Nellix and Ventana Trials Clinical
The Ventana Off-the-Shelf Graft for Pararenal AAA Andrew Holden Associate Professor of Radiology Auckland Hospital Disclosures Andrew Holden, MBChB, FRANZCR Investigator in Nellix and Ventana Trials Clinical
TRAINING DOCUMENT, FOR INTERNAL USE ONLY. Pre-case Planning
 TRINING DOCUMENT, FOR INTERNL USE ONLY Pre-case Planning Imaging Specifications CONTRST ENHNCED CT-SCN CT angiography date should be within 3 months of implant date CT slices thickness not exceeding 3
TRINING DOCUMENT, FOR INTERNL USE ONLY Pre-case Planning Imaging Specifications CONTRST ENHNCED CT-SCN CT angiography date should be within 3 months of implant date CT slices thickness not exceeding 3
Indications for use. Contraindications within the United States
 Indications for use Indications within the United States The GORE TAG Thoracic Endoprosthesis is intended for endovascular repair of all lesions of the descending thoracic aorta, including: Isolated lesions
Indications for use Indications within the United States The GORE TAG Thoracic Endoprosthesis is intended for endovascular repair of all lesions of the descending thoracic aorta, including: Isolated lesions
Abdominal and thoracic aneurysm repair
 Abdominal and thoracic aneurysm repair William A. Gray MD Director, Endovascular Intervention Cardiovascular Research Foundation Columbia University Medical Center Abdominal Aortic Aneurysm Endografts
Abdominal and thoracic aneurysm repair William A. Gray MD Director, Endovascular Intervention Cardiovascular Research Foundation Columbia University Medical Center Abdominal Aortic Aneurysm Endografts
Current Status of Abdominal Aortic Stent Grafts. John R. Laird Professor of Medicine Director of the Vascular Center UC Davis Medical Center
 Current Status of Abdominal Aortic Stent Grafts John R. Laird Professor of Medicine Director of the Vascular Center UC Davis Medical Center Autumn Greetings Disclosure Statement of Financial Interest Within
Current Status of Abdominal Aortic Stent Grafts John R. Laird Professor of Medicine Director of the Vascular Center UC Davis Medical Center Autumn Greetings Disclosure Statement of Financial Interest Within
ABDOMINAL XT ABDOMINAL STENT GRAFT SYSTEM
 ABDOMINAL XT ABDOMINAL STENT GRAFT SYSTEM Pure performance and quality: Stent graft systems by JOTEC JOTEC is an innovation-oriented company with consistent focus on its core competencies: the development
ABDOMINAL XT ABDOMINAL STENT GRAFT SYSTEM Pure performance and quality: Stent graft systems by JOTEC JOTEC is an innovation-oriented company with consistent focus on its core competencies: the development
2014 ANNUAL CLINICAL UPDATE. TriVascular Inc. Ovation and Ovation Prime Abdominal Stent Graft Systems
 2014 ANNUAL CLINICAL UPDATE TriVascular Inc. Ovation and Ovation Prime Abdominal Stent Graft Systems 2014 ANNUAL CLINICAL UPDATE TriVascular Inc. Ovation and Ovation Prime Abdominal Stent Graft Systems
2014 ANNUAL CLINICAL UPDATE TriVascular Inc. Ovation and Ovation Prime Abdominal Stent Graft Systems 2014 ANNUAL CLINICAL UPDATE TriVascular Inc. Ovation and Ovation Prime Abdominal Stent Graft Systems
Considerations for a Durable Repair
 Considerations for a Durable Repair Eric Verhoeven, MD, PhD, A. Katsargyris, MD Department of Vascular and Endovascular Surgery, Paracelsus Medical University, Nuremberg, Germany Disclosures William Cook
Considerations for a Durable Repair Eric Verhoeven, MD, PhD, A. Katsargyris, MD Department of Vascular and Endovascular Surgery, Paracelsus Medical University, Nuremberg, Germany Disclosures William Cook
2015 Clinical Update Endovascular Systems for AAA Repair
 Endovascular Systems for AAA Repair 2 Musick Irvine, CA 92618 Tel 949.595.7200 Fax 949.612.1893 www.endologix.com TABLE OF CONTENTS Section Page ABSTRACT... 3 READER S GUIDE... 4 SECTION 1: US PIVOTAL
Endovascular Systems for AAA Repair 2 Musick Irvine, CA 92618 Tel 949.595.7200 Fax 949.612.1893 www.endologix.com TABLE OF CONTENTS Section Page ABSTRACT... 3 READER S GUIDE... 4 SECTION 1: US PIVOTAL
Nellix Endovascular System: Clinical Outcomes and Device Overview
 Nellix Endovascular System: Clinical Outcomes and Device Overview Jeffrey P. Carpenter, MD Professor and Chief, Department of Surgery CAUTION: Investigational device. This product is under clinical investigation
Nellix Endovascular System: Clinical Outcomes and Device Overview Jeffrey P. Carpenter, MD Professor and Chief, Department of Surgery CAUTION: Investigational device. This product is under clinical investigation
Technique and Tips for Complicated AAA Cases with Stent Graft
 Technique and Tips for Complicated AAA Cases with Stent Graft Seung-Woon Rha, MD, PhD FACC, FAHA, FESC, FSCAI, FAPSIC Cardiovascular Center, Korea University Guro Hospital Mar 15, 2018 LINC AP 2018 Endoleak;
Technique and Tips for Complicated AAA Cases with Stent Graft Seung-Woon Rha, MD, PhD FACC, FAHA, FESC, FSCAI, FAPSIC Cardiovascular Center, Korea University Guro Hospital Mar 15, 2018 LINC AP 2018 Endoleak;
Chungbuk Regional Cardiovascular Center, Division of Cardiology, Departments of Internal Medicine, Chungbuk National University Hospital Sangmin Kim
 Endovascular Procedures for Isolated Common Iliac and Internal Iliac Aneurysm Chungbuk Regional Cardiovascular Center, Division of Cardiology, Departments of Internal Medicine, Chungbuk National University
Endovascular Procedures for Isolated Common Iliac and Internal Iliac Aneurysm Chungbuk Regional Cardiovascular Center, Division of Cardiology, Departments of Internal Medicine, Chungbuk National University
Reimbursement Guide Zenith Fenestrated AAA Endovascular Graft
 MEDICAL Reimbursement Guide Zenith Fenestrated AAA Endovascular Graft Disclaimer: The information provided herein reflects Cook s understanding of the procedure(s) and/or device(s) from sources that may
MEDICAL Reimbursement Guide Zenith Fenestrated AAA Endovascular Graft Disclaimer: The information provided herein reflects Cook s understanding of the procedure(s) and/or device(s) from sources that may
GORE EXCLUDER AAA Endoprosthesis demonstrates long-term durability. Michel Reijnen Rijnstate Hospital Arnhem, The Netherlands
 GORE EXCLUDER AAA Endoprosthesis demonstrates long-term durability Michel Reijnen Rijnstate Hospital Arnhem, The Netherlands Disclosure Speaker name: Michel Reijnen I have the following potential conflicts
GORE EXCLUDER AAA Endoprosthesis demonstrates long-term durability Michel Reijnen Rijnstate Hospital Arnhem, The Netherlands Disclosure Speaker name: Michel Reijnen I have the following potential conflicts
Acute dissections of the descending thoracic aorta (Debakey
 Endovascular Treatment of Acute Descending Thoracic Aortic Dissections Nimesh D. Desai, MD, PhD, and Joseph E. Bavaria, MD Acute dissections of the descending thoracic aorta (Debakey type III or Stanford
Endovascular Treatment of Acute Descending Thoracic Aortic Dissections Nimesh D. Desai, MD, PhD, and Joseph E. Bavaria, MD Acute dissections of the descending thoracic aorta (Debakey type III or Stanford
From 1996 to 1999, a total of 1,193 patients with
 THE ANEURX CLINICAL TRIAL AT 8 YEARS Lessons learned following the US AneuRx clinical trial from 1996 to 2004. BY CHRISTOPHER K. ZARINS, MD From 1996 to 1999, a total of 1,193 patients with infrarenal
THE ANEURX CLINICAL TRIAL AT 8 YEARS Lessons learned following the US AneuRx clinical trial from 1996 to 2004. BY CHRISTOPHER K. ZARINS, MD From 1996 to 1999, a total of 1,193 patients with infrarenal
How to select FEVAR versus EVAR + endoanchors in short-necked AAAs
 How to select FEVAR versus EVAR + endoanchors in short-necked AAAs Jean-Paul P.M. de Vries, Richte C.L. Schuurmann St. Antonius Hospital Nieuwegein, The Netherlands 21st Critical Issues Congress Nuernberg,
How to select FEVAR versus EVAR + endoanchors in short-necked AAAs Jean-Paul P.M. de Vries, Richte C.L. Schuurmann St. Antonius Hospital Nieuwegein, The Netherlands 21st Critical Issues Congress Nuernberg,
Obesity, Scaring, Access in EVAR. Kiskinis D, Melas N, Ktenidis K. 1 st Department of Surgery Aristotle University of Thessaloniki, Greece
 Obesity, Scaring, Access in EVAR Kiskinis D, Melas N, Ktenidis K. 1 st Department of Surgery Aristotle University of Thessaloniki, Greece Obesity Decreased radiolucency (visibility) Max weight load < 160
Obesity, Scaring, Access in EVAR Kiskinis D, Melas N, Ktenidis K. 1 st Department of Surgery Aristotle University of Thessaloniki, Greece Obesity Decreased radiolucency (visibility) Max weight load < 160
Annual Clinical Update
 Zenith TX2 TAA Endovascular Graft (P716) Annual Clinical Update (216) Page i Annual Clinical Update Abstract Cook is pleased to provide you with this clinical update on the Zenith TX2 TAA Endovascular
Zenith TX2 TAA Endovascular Graft (P716) Annual Clinical Update (216) Page i Annual Clinical Update Abstract Cook is pleased to provide you with this clinical update on the Zenith TX2 TAA Endovascular
Endovascular Management of Thoracic Aortic Pathology Stéphan Haulon, J Sobocinski, B Maurel, T Martin-Gonzalez, R Spear, A Hertault, R Azzaoui
 Endovascular Management of Thoracic Aortic Pathology Stéphan Haulon, J Sobocinski, B Maurel, T Martin-Gonzalez, R Spear, A Hertault, R Azzaoui Aortic Center, Lille University Hospital, France Disclosures
Endovascular Management of Thoracic Aortic Pathology Stéphan Haulon, J Sobocinski, B Maurel, T Martin-Gonzalez, R Spear, A Hertault, R Azzaoui Aortic Center, Lille University Hospital, France Disclosures
Increased Flexibility of AneuRx Stent-Graft Reduces Need for Secondary Intervention Following Endovascular Aneurysm Repair
 583 Increased Flexibility of AneuRx Stent-Graft Reduces Need for Secondary Intervention Following Endovascular Aneurysm Repair Frank R. Arko, MD; W. Anthony Lee, MD; Bradley B. Hill, MD; Paul Cipriano,
583 Increased Flexibility of AneuRx Stent-Graft Reduces Need for Secondary Intervention Following Endovascular Aneurysm Repair Frank R. Arko, MD; W. Anthony Lee, MD; Bradley B. Hill, MD; Paul Cipriano,
Abdominal Aortic Aneurysms. A Surgeons Perspective Dr. Derek D. Muehrcke
 Abdominal Aortic Aneurysms A Surgeons Perspective Dr. Derek D. Muehrcke Aneurysm Definition The abnormal enlargement or bulging of an artery caused by an injury or weakness in the blood vessel wall A localized
Abdominal Aortic Aneurysms A Surgeons Perspective Dr. Derek D. Muehrcke Aneurysm Definition The abnormal enlargement or bulging of an artery caused by an injury or weakness in the blood vessel wall A localized
Anatomy-Driven Endograft Selection for Abdominal Aortic Aneurysm Repair S. Jay Mathews, MD, MS, FACC
 Anatomy-Driven Endograft Selection for Abdominal Aortic Aneurysm Repair S. Jay Mathews, MD, MS, FACC Interventional Cardiologist/Endovascular Specialist Bradenton Cardiology Center Bradenton, FL, USA Disclosures
Anatomy-Driven Endograft Selection for Abdominal Aortic Aneurysm Repair S. Jay Mathews, MD, MS, FACC Interventional Cardiologist/Endovascular Specialist Bradenton Cardiology Center Bradenton, FL, USA Disclosures
How to do AAA EVAR: Tips and Tricks
 How to do AAA EVAR: Tips and Tricks June 14, 2007 Jon S. Matsumura, MD Associate Professor of Surgery Division of Vascular Surgery Northwestern University Medical School Disclosure Information Jon Matsumura,
How to do AAA EVAR: Tips and Tricks June 14, 2007 Jon S. Matsumura, MD Associate Professor of Surgery Division of Vascular Surgery Northwestern University Medical School Disclosure Information Jon Matsumura,
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
Citation for published version (APA): Tielliu, I. F. J. (2010). Endovascular repair of peripheral artery aneurysms Groningen: s.n.
 University of Groningen Endovascular repair of peripheral artery aneurysms Tielliu, Ignace François Jacques IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish
University of Groningen Endovascular repair of peripheral artery aneurysms Tielliu, Ignace François Jacques IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish
Case Report Early and Late Endograft Limb Proximal Migration with Resulting Type 1b Endoleak following an EVAR for Ruptured AAA
 Hindawi Case Reports in Vascular Medicine Volume 2017, Article ID 4931282, 5 pages https://doi.org/10.1155/2017/4931282 Case Report Early and Late Endograft Limb Proximal Migration with Resulting Type
Hindawi Case Reports in Vascular Medicine Volume 2017, Article ID 4931282, 5 pages https://doi.org/10.1155/2017/4931282 Case Report Early and Late Endograft Limb Proximal Migration with Resulting Type
14F OD Ovation Abdominal Stent Graft System
 14F OD Ovation Abdominal Stent Graft System 2 Year Results from the European Study Dierk Scheinert, MD Center for Vascular Medicine Angiology and Vascular Surgery Park Hospital Leipzig, Germany Disclosure
14F OD Ovation Abdominal Stent Graft System 2 Year Results from the European Study Dierk Scheinert, MD Center for Vascular Medicine Angiology and Vascular Surgery Park Hospital Leipzig, Germany Disclosure
ENCORE, a Study to Investigate the Durability of Polymer EVAR with Ovation A Contemporary Review of 1296 Patients
 ENCORE, a Study to Investigate the Durability of Polymer EVAR with Ovation A Contemporary Review of 1296 Patients The Ovation System is approved to treat infrarenal abdominal aortic aneurysms and is not
ENCORE, a Study to Investigate the Durability of Polymer EVAR with Ovation A Contemporary Review of 1296 Patients The Ovation System is approved to treat infrarenal abdominal aortic aneurysms and is not
Challenges. 1. Sizing. 2. Proximal landing zone 3. Distal landing zone 4. Access vessels 5. Spinal cord ischemia 6. Endoleak
 Disclosure I have the following potential conflicts of interest to report: Consulting: Medtronic, Gore Employment in industry Stockholder of a healthcare company Owner of a healthcare company Other(s)
Disclosure I have the following potential conflicts of interest to report: Consulting: Medtronic, Gore Employment in industry Stockholder of a healthcare company Owner of a healthcare company Other(s)
Bilateral use of the Gore IBE device for bilateral CIA aneurysms and a first interim analysis of the prospective Iceberg registry
 Bilateral use of the Gore IBE device for bilateral CIA aneurysms and a first interim analysis of the prospective Iceberg registry Michel MPJ Reijnen, MD, PhD Department of Vascular Surgery, Rijnstate Hospital
Bilateral use of the Gore IBE device for bilateral CIA aneurysms and a first interim analysis of the prospective Iceberg registry Michel MPJ Reijnen, MD, PhD Department of Vascular Surgery, Rijnstate Hospital
Management of Endoleaks. Michael Meuse, M.D Vascular and Interventional Radiology 12/14/09
 Management of Endoleaks Michael Meuse, M.D Vascular and Interventional Radiology 12/14/09 Endoleak Failure to totally exclude the abdominal aortic aneurysm (AAA) from continued perfusion and pressurization
Management of Endoleaks Michael Meuse, M.D Vascular and Interventional Radiology 12/14/09 Endoleak Failure to totally exclude the abdominal aortic aneurysm (AAA) from continued perfusion and pressurization
MODERN METHODS FOR TREATING ABDOMINAL ANEURYSMS AND THORACIC AORTIC DISEASE
 MODERN METHODS FOR TREATING ABDOMINAL ANEURYSMS AND THORACIC AORTIC DISEASE AAA FACTS 200,000 New Cases Each Year Ruptured AAA = 15,000 Deaths per Year in U.S. 13th Leading Cause of Death 80% Chance of
MODERN METHODS FOR TREATING ABDOMINAL ANEURYSMS AND THORACIC AORTIC DISEASE AAA FACTS 200,000 New Cases Each Year Ruptured AAA = 15,000 Deaths per Year in U.S. 13th Leading Cause of Death 80% Chance of
THE THE MORE MORE NATURAL APPROACH TO OPTIMAL FIT
 THE THE MORE MORE NATURAL APPROACH Natural Approach TO Optimal Fit TO OPTIMAL FIT Conformability without Compromise THE STANDARD IN Conformability AND Designed for flexibility and conformability in tortuous
THE THE MORE MORE NATURAL APPROACH Natural Approach TO Optimal Fit TO OPTIMAL FIT Conformability without Compromise THE STANDARD IN Conformability AND Designed for flexibility and conformability in tortuous
The Endurant Stent Graft System
 The Stent Graft System Instructions for Use (IFU) IMPORTANT! Do not attempt to use the Stent Graft System before completely reading and understanding the information contained in the Instructions for Use.
The Stent Graft System Instructions for Use (IFU) IMPORTANT! Do not attempt to use the Stent Graft System before completely reading and understanding the information contained in the Instructions for Use.
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
CHALLENGING ILIAC ACCESSES AND THROMBOSIS PREVENTION
 CHALLENGING ILIAC ACCESSES AND THROMBOSIS PREVENTION ARMANDO MANSILHA MD, PhD, FEBVS UNIVERSITY HOSPITAL - PORTO Disclosure of Interest Speaker name: ARMANDO MANSILHA I have the following potential conflicts
CHALLENGING ILIAC ACCESSES AND THROMBOSIS PREVENTION ARMANDO MANSILHA MD, PhD, FEBVS UNIVERSITY HOSPITAL - PORTO Disclosure of Interest Speaker name: ARMANDO MANSILHA I have the following potential conflicts
3. Endoluminal Treatment of Infrarenal Abdominal Aortic Aneurysm
 3. Endoluminal Treatment of Infrarenal Abdominal Aortic Aneurysm Hence J. M. Verhagen, Geoffrey H. White, Tom Daly and Theodossios Perdikides A 78-year-old male was referred for investigation and management
3. Endoluminal Treatment of Infrarenal Abdominal Aortic Aneurysm Hence J. M. Verhagen, Geoffrey H. White, Tom Daly and Theodossios Perdikides A 78-year-old male was referred for investigation and management
(EU), FACC (USA), FSCAI (USA)
 How to reduce vascular complications of TAVI Paul TL Chiam MBBS (S pore), MMed, MRCP (UK), FAMS FRCP (Edin), FESC (EU), FACC (USA), FSCAI (USA) Cardiologist Mount Elizabeth Hospital Singapore Definition
How to reduce vascular complications of TAVI Paul TL Chiam MBBS (S pore), MMed, MRCP (UK), FAMS FRCP (Edin), FESC (EU), FACC (USA), FSCAI (USA) Cardiologist Mount Elizabeth Hospital Singapore Definition
INCRAFT system: Update from the Pivotal INSPIRATION Study
 INCRAFT system: Update from the Pivotal INSPIRATION Study Michel S. Makaroun MD Co-Director, UPMC Heart and Vascular Institute Professor and Chair, Division of Vascular Surgery University of Pittsburgh
INCRAFT system: Update from the Pivotal INSPIRATION Study Michel S. Makaroun MD Co-Director, UPMC Heart and Vascular Institute Professor and Chair, Division of Vascular Surgery University of Pittsburgh
Case Study of Implantation of a VICI VENOUS STENT - Combined NIVL and PTS Stenting
 Case Study of Implantation of a VICI VENOUS STENT - Combined NIVL and PTS Stenting Courtesy of Mr. Stephen Black United kingdom Patient History 25 y/o female 2011: Conservatively treated ilio-femoral DVT
Case Study of Implantation of a VICI VENOUS STENT - Combined NIVL and PTS Stenting Courtesy of Mr. Stephen Black United kingdom Patient History 25 y/o female 2011: Conservatively treated ilio-femoral DVT
Ovation. Sean Lyden, MD Department Chair, Vascular Surgery Cleveland Clinic
 Ovation Sean Lyden, MD Department Chair, Vascular Surgery Cleveland Clinic Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement
Ovation Sean Lyden, MD Department Chair, Vascular Surgery Cleveland Clinic Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement
Challenges with Complex Anatomies Advancing Care in Endovascular Aortic Treatment
 Challenges with Complex Anatomies Advancing Care in Endovascular Aortic Treatment Robert Y. Rhee, MD Chief, Vascular and Endovascular Surgery Director, Aortic Center Maimonides Medical Center Brooklyn,
Challenges with Complex Anatomies Advancing Care in Endovascular Aortic Treatment Robert Y. Rhee, MD Chief, Vascular and Endovascular Surgery Director, Aortic Center Maimonides Medical Center Brooklyn,
Feasibility of aortic neck anatomy for endovascular aneurysm repair in Korean patients with abdominal aortic aneurysm
 LINC 2019 Leipzig, Germany Feasibility of aortic neck anatomy for endovascular aneurysm repair in Korean patients with abdominal aortic aneurysm Deokbi Hwang, Sujin Park, Hyung-Kee Kim, Seung Huh Division
LINC 2019 Leipzig, Germany Feasibility of aortic neck anatomy for endovascular aneurysm repair in Korean patients with abdominal aortic aneurysm Deokbi Hwang, Sujin Park, Hyung-Kee Kim, Seung Huh Division
Malperfusion Syndromes Type B Aortic Dissection with Malperfusion
 Malperfusion Syndromes Type B Aortic Dissection with Malperfusion Jade S. Hiramoto, MD, MAS April 27, 2012 Associated with early mortality Occurs when there is end organ ischemia secondary to aortic branch
Malperfusion Syndromes Type B Aortic Dissection with Malperfusion Jade S. Hiramoto, MD, MAS April 27, 2012 Associated with early mortality Occurs when there is end organ ischemia secondary to aortic branch
BC Vascular Day. Contents. November 3, Abdominal Aortic Aneurysm 2 3. Peripheral Arterial Disease 4 6. Deep Venous Thrombosis 7 8
 BC Vascular Day Contents Abdominal Aortic Aneurysm 2 3 November 3, 2018 Peripheral Arterial Disease 4 6 Deep Venous Thrombosis 7 8 Abdominal Aortic Aneurysm Conservative Management Risk factor modification
BC Vascular Day Contents Abdominal Aortic Aneurysm 2 3 November 3, 2018 Peripheral Arterial Disease 4 6 Deep Venous Thrombosis 7 8 Abdominal Aortic Aneurysm Conservative Management Risk factor modification
symptomatic aneurysms or aneurysms that grow >1cm/yr
 1. Elective repair for aneurysm >5.5 cm, symptomatic aneurysms or aneurysms that grow >1cm/yr 2. Ruptured AAA Aneurysm Detection and Management Study (ADAM) and UK Small Aneurysm Trial early open surgery
1. Elective repair for aneurysm >5.5 cm, symptomatic aneurysms or aneurysms that grow >1cm/yr 2. Ruptured AAA Aneurysm Detection and Management Study (ADAM) and UK Small Aneurysm Trial early open surgery
Length Measurements of the Aorta After Endovascular Abdominal Aortic Aneurysm Repair
 Eur J Vasc Endovasc Surg 18, 481 486 (1999) Article No. ejvs.1999.0882 Length Measurements of the Aorta After Endovascular Abdominal Aortic Aneurysm Repair J. J. Wever, J. D. Blankensteijn, I. A. M. J.
Eur J Vasc Endovasc Surg 18, 481 486 (1999) Article No. ejvs.1999.0882 Length Measurements of the Aorta After Endovascular Abdominal Aortic Aneurysm Repair J. J. Wever, J. D. Blankensteijn, I. A. M. J.
Instructions for Use
 Ovation ix Abdominal Stent Graft System Instructions for Use EN (810-0016-01-01) 0 0 5 0 810-0016-01-01 Rev A Table of Contents 1. Device Description... 3 1.1. Delivery System... 4 1.2. Fill Kit and Autoinjector...
Ovation ix Abdominal Stent Graft System Instructions for Use EN (810-0016-01-01) 0 0 5 0 810-0016-01-01 Rev A Table of Contents 1. Device Description... 3 1.1. Delivery System... 4 1.2. Fill Kit and Autoinjector...
All-In-One. Iliac Branch System. PER F ORM ANC E b y d e s i g n ILIAC BRANCH ENDOPROSTHESIS
 All-In-One Iliac Branch System PER F ORM ANC E b y d e s i g n ILIAC BRANCH ENDOPROSTHESIS Exclusively Designed for Use in The GORE EXCLUDER Iliac Branch designed for the iliac branch, built from the same
All-In-One Iliac Branch System PER F ORM ANC E b y d e s i g n ILIAC BRANCH ENDOPROSTHESIS Exclusively Designed for Use in The GORE EXCLUDER Iliac Branch designed for the iliac branch, built from the same
Peripheral Arterial Disease: A Practical Approach
 Peripheral Arterial Disease: A Practical Approach Sanjoy Kundu BSc, MD, FRCPC, DABR, FASA, FCIRSE, FSIR The Scarborough Hospital Toronto Endovascular Centre The Vein Institute of Toronto Scarborough Vascular
Peripheral Arterial Disease: A Practical Approach Sanjoy Kundu BSc, MD, FRCPC, DABR, FASA, FCIRSE, FSIR The Scarborough Hospital Toronto Endovascular Centre The Vein Institute of Toronto Scarborough Vascular
Tips and techniques for optimal stent graft placement in angulated aneurysm necks
 VASCULAR AND ENDOVASCULAR TECHNIQUES Thomas L. Forbes, MD, Section Editor Tips and techniques for optimal stent graft placement in angulated aneurysm necks Jasper W. van Keulen, MD, Frans L. Moll, MD,
VASCULAR AND ENDOVASCULAR TECHNIQUES Thomas L. Forbes, MD, Section Editor Tips and techniques for optimal stent graft placement in angulated aneurysm necks Jasper W. van Keulen, MD, Frans L. Moll, MD,
Endoanchor-assisted TEVAR
 Endoanchor-assisted TEVAR May 29, 2015 NCVH2015 Grayson H. Wheatley III, MD Director of Aortic and Endovascular Surgery Associate Professor of Surgery Temple University School of Medicine Disclosures Consultant
Endoanchor-assisted TEVAR May 29, 2015 NCVH2015 Grayson H. Wheatley III, MD Director of Aortic and Endovascular Surgery Associate Professor of Surgery Temple University School of Medicine Disclosures Consultant
Current Status of EVAR for Infrarenal AAA. 31 st Annual Florida Vascular Society. PENN Surgery
 Current Status of EVAR for Infrarenal AAA 31 st Annual Florida Vascular Society PENN Surgery No Disclosures Stent Grafts Design Related Differences What really matters? Modular Unibody Supported Unsupported
Current Status of EVAR for Infrarenal AAA 31 st Annual Florida Vascular Society PENN Surgery No Disclosures Stent Grafts Design Related Differences What really matters? Modular Unibody Supported Unsupported
Case Report 1. CTA head. (c) Tele3D Advantage, LLC
 Case Report 1 CTA head 1 History 82 YEAR OLD woman with signs and symptoms of increased intra cranial pressure in setting of SAH. CT Brain was performed followed by CT Angiography of head. 2 CT brain Extensive
Case Report 1 CTA head 1 History 82 YEAR OLD woman with signs and symptoms of increased intra cranial pressure in setting of SAH. CT Brain was performed followed by CT Angiography of head. 2 CT brain Extensive
Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System
 100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
Instructions for Use
 with the Ovation ix Iliac Stent Graft Instructions for Use EN (810-0009-01-04) 0 0 5 0 810-0009-01-04 Rev A Table of Contents 1. Device Description... 3 1.1. Delivery System... 4 1.2. Fill Kit and Autoinjector...
with the Ovation ix Iliac Stent Graft Instructions for Use EN (810-0009-01-04) 0 0 5 0 810-0009-01-04 Rev A Table of Contents 1. Device Description... 3 1.1. Delivery System... 4 1.2. Fill Kit and Autoinjector...
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
 M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
Important Update to Field Safety Notice Nellix EndoVascular Aneurysm Sealing System Updated Instructions for Use (IFU)
 October 6, 2017 Important Update to Field Safety Notice Nellix EndoVascular Aneurysm Sealing System Updated Instructions for Use (IFU) Dear Physician, This notification is to provide you with further information
October 6, 2017 Important Update to Field Safety Notice Nellix EndoVascular Aneurysm Sealing System Updated Instructions for Use (IFU) Dear Physician, This notification is to provide you with further information
How to manage TAVI related vascular complications. Paul TL Chiam MBBS, FRCP, FESC, FACC, FSCAI
 How to manage TAVI related vascular complications Paul TL Chiam MBBS, FRCP, FESC, FACC, FSCAI Definition VARC-2 consensus statement Complications caused by: Wire Catheter Anything related to vascular access
How to manage TAVI related vascular complications Paul TL Chiam MBBS, FRCP, FESC, FACC, FSCAI Definition VARC-2 consensus statement Complications caused by: Wire Catheter Anything related to vascular access
FLEXIBLE, BALOON EXPANDABLE
 EARLY RESULTS OF A CLINICAL TRIAL OF FLEXIBLE, BALOON EXPANDABLE COVERED STENT GRAFT IN ILIAC OCCLUSIVE DISEASE Chris LeCroy Coastal Vascular and Interventional Pensacola, Florida Clinical Trial WL GORE
EARLY RESULTS OF A CLINICAL TRIAL OF FLEXIBLE, BALOON EXPANDABLE COVERED STENT GRAFT IN ILIAC OCCLUSIVE DISEASE Chris LeCroy Coastal Vascular and Interventional Pensacola, Florida Clinical Trial WL GORE
An endoleak is radiographic or ultrasonic evidence
 Complex Coil Embolization of Multiple Type II Endoleaks Liquid embolics, detachable coils, and plugs to repair an enlarging abdominal aortic aneurysm sac 5 years after EVAR. BY FRANK R. ARKO, MD; ABRAHAM
Complex Coil Embolization of Multiple Type II Endoleaks Liquid embolics, detachable coils, and plugs to repair an enlarging abdominal aortic aneurysm sac 5 years after EVAR. BY FRANK R. ARKO, MD; ABRAHAM
Vascular V12. Covered Stent. The New Standard of Care
 Vascular V12 Covered Stent The New Standard of Care Innovation Novel Thermo-Conformable Covered Stent 316L stainless steel stent completely encapsulated in a proprietary one piece eptfe film cast covering
Vascular V12 Covered Stent The New Standard of Care Innovation Novel Thermo-Conformable Covered Stent 316L stainless steel stent completely encapsulated in a proprietary one piece eptfe film cast covering
RadRx Your Prescription for Accurate Coding & Reimbursement Copyright All Rights Reserved.
 Interventional Radiology Coding Case Studies Prepared by Stacie L. Buck, RHIA, CCS-P, RCC, CIRCC, AAPC Fellow President & Senior Consultant Week of November 19, 2018 Abdominal Aortogram, Bilateral Runoff
Interventional Radiology Coding Case Studies Prepared by Stacie L. Buck, RHIA, CCS-P, RCC, CIRCC, AAPC Fellow President & Senior Consultant Week of November 19, 2018 Abdominal Aortogram, Bilateral Runoff
