Tenapanor, a minimally absorbed NHE3 inhibitor, reduces dietary phosphorus absorption in healthy volunteers
|
|
|
- Adelia Gardner
- 5 years ago
- Views:
Transcription
1 , a minimally absorbed NHE3 inhibitor, reduces dietary phosphorus absorption in healthy volunteers David Rosenbaum, 1 Susanne Johansson, 2 Björn Carlsson, 2 Andrew G Spencer, 1 Bergur Stefansson, 2 Mikael Knutsson, 2 Jeffrey Jacobs, 1 Dominique Charmot 1 1 Ardelyx Inc., Fremont, CA, USA; 2 AstraZeneca R&D, Mölndal, Sweden
2 Disclosures David Rosenbaum, Andrew G Spencer, Jeffrey Jacobs and Dominique Charmot are employees of, and have ownership interest in, Ardelyx Susanne Johansson and Bjӧrn Carlsson are employees of, and have ownership interest in, AstraZeneca. Bergur Stefansson and Mikael Knutsson are employees of AstraZeneca These studies were funded by AstraZeneca and Ardelyx 2
3 reduces sodium and phosphorus absorption (AZD1722) is a first-in-class smallmolecule inhibitor of the Na + /H + exchanger isoform 3 (NHE3) Two healthy volunteer studies 1 show that tenapanor reduces absorption of dietary sodium over 7 days 2 Preclinical data show that tenapanor reduces phosphorus absorption and protects against vascular calcification 3 Sodium excretion (mmol/day) Stool sodium Placebo 3 mg mg 3 mg mg q.d. q.d., once daily 1. Study codes: RDX and RDX5791-2; 2. Spencer AG et al. Sci Transl Med 14;6:227ra36; 3. Charmot D et al. Oral presentation, American Society of Nephrology Kidney Week (abstract 425), 14 3
4 Studies of tenapanor in healthy volunteers Multiple ascending-dose study 1 Once-daily tenapanor HCl 3,, 3 or mg or placebo for 7 days Dose-regimen study 2 HCl 3 mg once daily, 15, 3 or 6 mg twice daily, 3 mg three times daily or placebo for 7 days Formulation study 3 Food-effect study 4 Twice-daily tenapanor HCl 15 mg for 4 days Twice-daily tenapanor HCl 15 mg for 4 days Phosphorus excretion measured daily Participants received a diet standardized for sodium content In each study, all cohorts received the same meals on the same study days Study numbers: 1. RDX5791-1; 2. RDX5791-2; 3. D5611C2; 4. D5611C3 4
5 was generally well tolerated and minimally absorbed No serious adverse events No clinically significant changes in other a serum or urinary electrolytes Serum sodium, potassium, calcium or creatinine Urinary potassium or creatinine No significant changes in other clinical laboratory measurements, vital signs, electrocardiographic parameters or physical examinations Minimal to no systemic availability of tenapanor Plasma concentrations of tenapanor were below the lower limit of quantification (.5 ng/ml) in > 99% of all measured samples (72 samples across four studies) No individual had more than one measurable tenapanor plasma concentration: the highest measured concentration was.792 ng/ml a Other than pharmacodynamic variables: urinary sodium and phosphorus 5
6 Learning from phosphate binders Healthy volunteer studies of sevelamer hydrochloride provide a useful benchmark for developing new treatments for patients with hyperphosphatemia Phosphorus excretion (mmol/day) mmol/day increase (only observed at 5 g t.i.d.) Placebo (n = 6) Stool phosphorus Sevelamer Sevelamer Sevelamer 1 g t.i.d. 2.5 g t.i.d. 5 g t.i.d. (n = 6) (n = 6) (n = 6) Urinary phosphorus mmol/day decrease Placebo Sevelamer Sevelamer Sevelamer (n = 6) 1 g t.i.d. 2.5 g t.i.d. 5 g t.i.d. (n = 6) (n = 6) (n = 6) Data are shown as means. Error bars show one standard deviation. Treatment period was 4 days. t.i.d, three-times daily Adapted from Burke SK et al. Nephrol Dial Transplant 1997;12:
7 increased stool phosphorus over 7 days Phosphorus excretion (mmol/day) Placebo (n = 8) Multiple ascending-dose study (q.d.) 3 mg q.d. (n = 8) Data are daily means over 7 days treatment. Error bars show one standard deviation Study RDX mg q.d. (n = 8) mg q.d. (n = 8) Stool phosphorus increases of mmol/day vs placebo (mean across all tenapanor q.d. doses: 5.9 mmol/day) cf. increase of 7.5 mmol/day with 5 g sevelamer t.i.d. 7
8 increased stool phosphorus over 7 days (continued) Phosphorus excretion (mmol/day) Placebo (n = 21) 3 mg q.d. (n = 11) Dose-regimen study 15 mg b.i.d. (n = 12) 3 mg b.i.d. (n = 12) Data are daily means over 7 days treatment. Error bars show one standard deviation. b.i.d., twice daily Study RDX mg t.i.d. (n = 12) 6 mg b.i.d. (n = 12) Stool phosphorus increases of mmol/day vs placebo (mean across all tenapanor dose regimens: 9.8 mmol/day) cf. increase of 7.5 mmol/day with 5 g sevelamer t.i.d. 8
9 Increases in stool phosphorus were consistent with decreases in urinary phosphorus Formulation study Phosphorus excretion (mmol/day) Stool phosphorus Day 1 (n = 16) 15 mg b.i.d Urinary phosphorus 14.9 mmol/day 12. mmol/day Day 1 15 mg b.i.d. Stool phosphorus Urinary phosphorus Food-effect study Phosphorus excretion (mmol/day) mmol/day Run-in 15 mg b.i.d Run-in 7.7 mmol/day 15 mg b.i.d. Error bars show one standard deviation. Data for tenapanor are means of treatment days 1 4 Formulation study (D5611C2) includes mean of day 1. Data are for tenapanor 15 mg b.i.d. HCl tablet treatment group Food-effect study (D5611C3): run-in comprised mean of days 2 and 1. Data are tenapanor 15 mg b.i.d. preprandial treatment group 9
10 Conclusions, a small-molecule inhibitor of NHE3, was generally well tolerated and minimally absorbed in healthy volunteers reduced absorption of dietary phosphorus Increases in stool phosphorus Concomitant decreases in urinary phosphorus Increases in stool phosphorus were similar to published data for sevelamer hydrochloride in healthy volunteers 1 may provide a new mechanism for the treatment of hyperphosphatemia in patients with chronic kidney disease, with the potential to: Improve phosphate control Reduce pill burden (i.e. mg vs g doses) Reduce sodium overload 1. Burke SK et al. Nephrol Dial Transplant 1997;12:164 4
11 is under investigation for treating hyperphosphatemia in a phase 2 study Patients with chronic kidney disease on maintenance hemodialysis (NCT81534) Data anticipated in first half of 15 Expected number of patients: 15 Randomization 1 mg b.i.d. 3 mg b.i.d. mg b.i.d. Primary endpoint Change in serum phosphorus Screening Washout 3 mg b.i.d. Follow-up Cessation of existing phosphate binders 3 mg q.d. 3 mg q.d. Placebo 1 week 1 3 weeks 4 weeks 2 weeks 11
12 Acknowledgements The investigators acknowledge and thank the study participants, the study centres and the clinical teams Medical writing support was provided by Richard Claes and Steven Inglis of Oxford PharmaGenesis Ltd, UK, and was funded by AstraZeneca 12
Tenapanor, a gastrointestinal NHE3 inhibitor, reduces serum phosphate in patients with chronic kidney disease stage 5D and hyperphosphatemia
 , a gastrointestinal NHE3 inhibitor, reduces serum phosphate in patients with chronic kidney disease stage 5D and hyperphosphatemia Geoffrey A Block, 1 David P Rosenbaum, 2 Maria Leonsson-Zachrisson, 3
, a gastrointestinal NHE3 inhibitor, reduces serum phosphate in patients with chronic kidney disease stage 5D and hyperphosphatemia Geoffrey A Block, 1 David P Rosenbaum, 2 Maria Leonsson-Zachrisson, 3
Tenapanor, a gastrointestinal NHE3 inhibitor, reduces serum phosphate in patients with chronic kidney disease stage 5D and hyperphosphatemia
 , a gastrointestinal NHE3 inhibitor, reduces serum phosphate in patients with chronic kidney disease stage 5D and hyperphosphatemia Geoffrey A Block, 1 David P Rosenbaum, 2 Maria Leonsson- Zachrisson,
, a gastrointestinal NHE3 inhibitor, reduces serum phosphate in patients with chronic kidney disease stage 5D and hyperphosphatemia Geoffrey A Block, 1 David P Rosenbaum, 2 Maria Leonsson- Zachrisson,
William D Chey, 1 Anthony J Lembo, 2 James A Phillips, 3 David P Rosenbaum 4
 Efficacy and safety of tenapanor in patients with constipationpredominant irritable bowel syndrome: a 12-week, double-blind, placebocontrolled, randomized phase 2b trial William D Chey, 1 Anthony J Lembo,
Efficacy and safety of tenapanor in patients with constipationpredominant irritable bowel syndrome: a 12-week, double-blind, placebocontrolled, randomized phase 2b trial William D Chey, 1 Anthony J Lembo,
A phase 1 study of the safety, tolerability, pharmacodynamics, and pharmacokinetics of tenapanor in healthy Japanese volunteers
 Clin Exp Nephrol (2017) 21:407 416 DOI 10.1007/s10157-016-1302-8 ORIGINAL ARTICLE A phase 1 study of the safety, tolerability, pharmacodynamics, and pharmacokinetics of tenapanor in healthy Japanese volunteers
Clin Exp Nephrol (2017) 21:407 416 DOI 10.1007/s10157-016-1302-8 ORIGINAL ARTICLE A phase 1 study of the safety, tolerability, pharmacodynamics, and pharmacokinetics of tenapanor in healthy Japanese volunteers
Lanthanum Carbonate Reduces Urine Phosphorus Excretion: Evidence of High-Capacity Phosphate Binding
 Renal Failure, 34(3): 263 270, (2012) Copyright Informa Healthcare USA, Inc. ISSN 0886-022X print/1525-6049 online DOI: 10.3109/0886022X.2011.649657 Lanthanum Carbonate Reduces Urine Phosphorus Excretion:
Renal Failure, 34(3): 263 270, (2012) Copyright Informa Healthcare USA, Inc. ISSN 0886-022X print/1525-6049 online DOI: 10.3109/0886022X.2011.649657 Lanthanum Carbonate Reduces Urine Phosphorus Excretion:
The effects of tenapanor on serum fibroblast growth factor 23 in patients receiving hemodialysis with hyperphosphatemia
 Nephrol Dial Transplant (2018) 1 8 doi: 10.1093/ndt/gfy061 The effects of tenapanor on serum fibroblast growth factor 23 in patients receiving hemodialysis with hyperphosphatemia Geoffrey A. Block 1, David
Nephrol Dial Transplant (2018) 1 8 doi: 10.1093/ndt/gfy061 The effects of tenapanor on serum fibroblast growth factor 23 in patients receiving hemodialysis with hyperphosphatemia Geoffrey A. Block 1, David
Velphoro (sucroferric oxyhydroxide)
 STRENGTH DOSAGE FORM ROUTE GPID 500mg chewable tablet oral 36003 MANUFACTURER Fresenius Medical Care North America INDICATION(S) For the control of serum phosphorus levels in patients with chronic kidney
STRENGTH DOSAGE FORM ROUTE GPID 500mg chewable tablet oral 36003 MANUFACTURER Fresenius Medical Care North America INDICATION(S) For the control of serum phosphorus levels in patients with chronic kidney
TRANSPARENCY COMMITTEE OPINION. 22 July 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 22 July 2009 PHOSPHOSORB 660 mg, film-coated tablet Container of 200 (CIP: 381 466-0) Applicant: FRESENIUS MEDICAL
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 22 July 2009 PHOSPHOSORB 660 mg, film-coated tablet Container of 200 (CIP: 381 466-0) Applicant: FRESENIUS MEDICAL
Normal kidneys filter large amounts of organic
 ORIGINAL ARTICLE - NEPHROLOGY Effect Of Lanthanum Carbonate vs Calcium Acetate As A Phosphate Binder In Stage 3-4 CKD- Treat To Goal Study K.S. Sajeev Kumar (1), M K Mohandas (1), Ramdas Pisharody (1),
ORIGINAL ARTICLE - NEPHROLOGY Effect Of Lanthanum Carbonate vs Calcium Acetate As A Phosphate Binder In Stage 3-4 CKD- Treat To Goal Study K.S. Sajeev Kumar (1), M K Mohandas (1), Ramdas Pisharody (1),
FUNCTIONAL GI DISORDERS ORIGINAL CONTRIBUTIONS
 ORIGINAL CONTRIBUTIONS 763 see related editorial on page x Tenapanor Treatment of Patients With Constipation-Predominant Irritable Bowel Syndrome: A Phase 2, Randomized, Placebo-Controlled Efficacy and
ORIGINAL CONTRIBUTIONS 763 see related editorial on page x Tenapanor Treatment of Patients With Constipation-Predominant Irritable Bowel Syndrome: A Phase 2, Randomized, Placebo-Controlled Efficacy and
Effects of Tenapanor on Cytochrome P450-Mediated Drug-Drug Interactions
 Original Manuscript Effects of Tenapanor on Cytochrome P450-Mediated Drug-Drug Interactions Clinical Pharmacology in Drug Development 2017, 6(5) 466 475 C 2017 The Authors. Clinical Pharmacology in Drug
Original Manuscript Effects of Tenapanor on Cytochrome P450-Mediated Drug-Drug Interactions Clinical Pharmacology in Drug Development 2017, 6(5) 466 475 C 2017 The Authors. Clinical Pharmacology in Drug
Protocol GTC : A Randomized, Open Label, Parallel Design Study of Sevelamer Hydrochloride (Renagel ) in Chronic Kidney Disease Patients.
 Protocol GTC-68-208: A Randomized, Open Label, Parallel Design Study of Sevelamer Hydrochloride (Renagel ) in Chronic Kidney Disease Patients. These results are supplied for informational purposes only.
Protocol GTC-68-208: A Randomized, Open Label, Parallel Design Study of Sevelamer Hydrochloride (Renagel ) in Chronic Kidney Disease Patients. These results are supplied for informational purposes only.
The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. doi: /ajg.2017.
 Tenapanor Treatment of Patients With Constipation-Predominant Irritable Bowel Syndrome: A Phase 2, Randomized, Placebo- Controlled Efficacy and Safety Trial The Harvard community has made this article
Tenapanor Treatment of Patients With Constipation-Predominant Irritable Bowel Syndrome: A Phase 2, Randomized, Placebo- Controlled Efficacy and Safety Trial The Harvard community has made this article
Cowen Healthcare Conference March 12, 2018
 Cowen Healthcare Conference March 12, 2018 Forward-Looking Statements Some of the statements included in this presentation, particularly those regarding the commercialization and ongoing clinical development
Cowen Healthcare Conference March 12, 2018 Forward-Looking Statements Some of the statements included in this presentation, particularly those regarding the commercialization and ongoing clinical development
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Hyperphosphatemia is associated with a
 TREATMENT OPTIONS IN THE MANAGEMENT OF PHOSPHATE RETENTION * George A. Porter, MD, FACP, and Hartmut H. Malluche, MD, FACP ABSTRACT Hyperphosphatemia is an independent risk factor for mortality and cardiovascular
TREATMENT OPTIONS IN THE MANAGEMENT OF PHOSPHATE RETENTION * George A. Porter, MD, FACP, and Hartmut H. Malluche, MD, FACP ABSTRACT Hyperphosphatemia is an independent risk factor for mortality and cardiovascular
ADVERSE REACTIONS The most common (>10%) adverse reactions are hypercalcemia, nausea, and diarrhea. (6.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOSLYRA safely and effectively. See full prescribing information for PHOSLYRA. PHOSLYRA (calcium
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOSLYRA safely and effectively. See full prescribing information for PHOSLYRA. PHOSLYRA (calcium
Randomized Clinical Trial of the Iron-Based Phosphate Binder PA21 in Hemodialysis Patients
 Article Randomized Clinical Trial of the Iron-Based Phosphate Binder PA21 in Hemodialysis Patients Rudolf P. Wüthrich,* Michel Chonchol, Adrian Covic, Sylvain Gaillard, Edward Chong, and James A. Tumlin
Article Randomized Clinical Trial of the Iron-Based Phosphate Binder PA21 in Hemodialysis Patients Rudolf P. Wüthrich,* Michel Chonchol, Adrian Covic, Sylvain Gaillard, Edward Chong, and James A. Tumlin
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 28 March 2012
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 28 March 2012 OSVAREN 435 mg/235 mg, film-coated tablet Bottle of 180 (CIP: 382 886 3) Applicant: FRESENIUS MEDICAL
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 28 March 2012 OSVAREN 435 mg/235 mg, film-coated tablet Bottle of 180 (CIP: 382 886 3) Applicant: FRESENIUS MEDICAL
2.0 Synopsis. Paricalcitol Capsules M Clinical Study Report R&D/15/0380. (For National Authority Use Only)
 2.0 Synopsis AbbVie Inc. Name of Study Drug: ABT-358/Zemplar (paricalcitol) Capsules Name of Active Ingredient: paricalcitol Individual Study Table Referring to Part of Dossier: Volume: Page: (For National
2.0 Synopsis AbbVie Inc. Name of Study Drug: ABT-358/Zemplar (paricalcitol) Capsules Name of Active Ingredient: paricalcitol Individual Study Table Referring to Part of Dossier: Volume: Page: (For National
Improved Assessment of Aortic Calcification in Japanese Patients Undergoing Maintenance Hemodialysis
 ORIGINAL ARTICLE Improved Assessment of Aortic Calcification in Japanese Patients Undergoing Maintenance Hemodialysis Masaki Ohya 1, Haruhisa Otani 2,KeigoKimura 3, Yasushi Saika 4, Ryoichi Fujii 4, Susumu
ORIGINAL ARTICLE Improved Assessment of Aortic Calcification in Japanese Patients Undergoing Maintenance Hemodialysis Masaki Ohya 1, Haruhisa Otani 2,KeigoKimura 3, Yasushi Saika 4, Ryoichi Fujii 4, Susumu
Tenapanor for irritable bowel syndrome with constipation
 NIHR Innovation Observatory Evidence Briefing: February 2018 Tenapanor for irritable bowel syndrome with constipation NIHRIO (HSRIC) ID: 6704 NICE ID: 9736 LAY SUMMARY Irritable bowel syndrome with constipation
NIHR Innovation Observatory Evidence Briefing: February 2018 Tenapanor for irritable bowel syndrome with constipation NIHRIO (HSRIC) ID: 6704 NICE ID: 9736 LAY SUMMARY Irritable bowel syndrome with constipation
Month/Year of Review: May 2014 Date of Last Review: September New Drug Evaluation: Sucrofferic Oxyhydroxide (Velphoro )
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Secondary Hyperparathyroidism: Where are we now?
 Secondary Hyperparathyroidism: Where are we now? Dylan M. Barth, Pharm.D. PGY-1 Pharmacy Resident Mayo Clinic 2017 MFMER slide-1 Objectives Identify risk factors for the development of complications caused
Secondary Hyperparathyroidism: Where are we now? Dylan M. Barth, Pharm.D. PGY-1 Pharmacy Resident Mayo Clinic 2017 MFMER slide-1 Objectives Identify risk factors for the development of complications caused
The effect of sevelamer carbonate and lanthanum carbonate on the pharmacokinetics of oral calcitriol
 Nephrol Dial Transplant (2011) 26: 1615 1621 doi: 10.1093/ndt/gfq598 Advance Access publication 4 October 2010 The effect of sevelamer carbonate and lanthanum carbonate on the pharmacokinetics of oral
Nephrol Dial Transplant (2011) 26: 1615 1621 doi: 10.1093/ndt/gfq598 Advance Access publication 4 October 2010 The effect of sevelamer carbonate and lanthanum carbonate on the pharmacokinetics of oral
Month/Year of Review: September 2012 Date of Last Review: September 2010
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Jose A. Diaz-Buxo Jonathan Greenberg Chiang-Hong Ho Laura Howson Olga Sergeyeva Punit Hinsu Claudy Mullon
 A Randomized, Controlled, Three-arm, Open-label, Cross-over Bioequivalence Study Comparing Calcium Acetate Oral Solution and Calcium Acetate Gelcaps in Healthy Volunteers Jose A. Diaz-Buxo Jonathan Greenberg
A Randomized, Controlled, Three-arm, Open-label, Cross-over Bioequivalence Study Comparing Calcium Acetate Oral Solution and Calcium Acetate Gelcaps in Healthy Volunteers Jose A. Diaz-Buxo Jonathan Greenberg
HYDROCHLORIDE FOR THE TREATMENT OF SECONDARY HYPERPARATHYROIDISM IN PATIENTS WITH END-STAGE RENAL DISEASE ON MAINTENANCE DIALYSIS THERAPY
 UK RENAL PHARMACY GROUP SUBMISSION TO THE NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE on CINACALCET HYDROCHLORIDE FOR THE TREATMENT OF SECONDARY HYPERPARATHYROIDISM IN PATIENTS WITH END-STAGE RENAL DISEASE
UK RENAL PHARMACY GROUP SUBMISSION TO THE NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE on CINACALCET HYDROCHLORIDE FOR THE TREATMENT OF SECONDARY HYPERPARATHYROIDISM IN PATIENTS WITH END-STAGE RENAL DISEASE
The impact of improved phosphorus control: use of sevelamer hydrochloride in patients with chronic renal failure
 Nephrol Dial Transplant (2002) 17: 340 345 The impact of improved phosphorus control: use of sevelamer hydrochloride in patients with chronic renal failure Naseem Amin Genzyme Corporation, Cambridge, MA,
Nephrol Dial Transplant (2002) 17: 340 345 The impact of improved phosphorus control: use of sevelamer hydrochloride in patients with chronic renal failure Naseem Amin Genzyme Corporation, Cambridge, MA,
T. Andersson 1, P. Nagy 1, M. Niazi 1, S. Nylander 1, L. Wallentin 2. ESC 2012, Munich, Germany. (1) AstraZeneca R&D, Mölndal, Sweden
 Impact of omeprazole, esomeprazole +/- acetylsalicylic acid and lansoprazole on the pharmacodynamics and pharmacokinetics of clopidogrel in healthy volunteers T. Andersson 1, P. Nagy 1, M. Niazi 1, S.
Impact of omeprazole, esomeprazole +/- acetylsalicylic acid and lansoprazole on the pharmacodynamics and pharmacokinetics of clopidogrel in healthy volunteers T. Andersson 1, P. Nagy 1, M. Niazi 1, S.
Effectiveness and tolerability of sevelamer in the treatment of hyperphosphatemia in hemodialysis patients
 Open Access Journal of Journal of Parathyroid Disease 2017,5(1),17 24 http://www.jparathyroid.com Original Effectiveness and tolerability of sevelamer in the treatment of hyperphosphatemia in hemodialysis
Open Access Journal of Journal of Parathyroid Disease 2017,5(1),17 24 http://www.jparathyroid.com Original Effectiveness and tolerability of sevelamer in the treatment of hyperphosphatemia in hemodialysis
Jefferies Healthcare Conference June 6, 2018
 Jefferies Healthcare Conference June 6, 2018 Forward-Looking Statements Some of the statements included in this presentation, particularly those regarding the commercialization and ongoing clinical development
Jefferies Healthcare Conference June 6, 2018 Forward-Looking Statements Some of the statements included in this presentation, particularly those regarding the commercialization and ongoing clinical development
CKD FOR INTERNISTS. Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College
 CKD FOR INTERNISTS Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College INTRODUCTION In 2002, the National Kidney Foundation s Kidney Disease Outcomes Quality Initiative(KDOQI)
CKD FOR INTERNISTS Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College INTRODUCTION In 2002, the National Kidney Foundation s Kidney Disease Outcomes Quality Initiative(KDOQI)
Revised: 11/2017 FULL PRESCRIBING INFORMATION: CONTENTS* 10 OVERDOSAGE 1 INDICATIONS AND USAGE 11 DESCRIPTION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AURYXIA safely and effectively. See full prescribing information for AURYXIA. AURYXIA (ferric citrate)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AURYXIA safely and effectively. See full prescribing information for AURYXIA. AURYXIA (ferric citrate)
02/27/2018. Objectives. To Replace or Not to Replace: Nutritional Vitamin D in Dialysis.
 To Replace or Not to Replace: Nutritional Vitamin D in Dialysis. Michael Shoemaker-Moyle, M.D. Assistant Professor of Clinical Medicine Objectives Review Vitamin D Physiology Review Current Replacement
To Replace or Not to Replace: Nutritional Vitamin D in Dialysis. Michael Shoemaker-Moyle, M.D. Assistant Professor of Clinical Medicine Objectives Review Vitamin D Physiology Review Current Replacement
Clinical benefits of an adherence monitoring program in the management of secondary hyperparathyroidism with cinacalcet:
 Clinical benefits of an adherence monitoring program in the management of secondary hyperparathyroidism with cinacalcet: Results of a prospective randomized controlled study Forni Valentina¹, Pruijm Menno¹,
Clinical benefits of an adherence monitoring program in the management of secondary hyperparathyroidism with cinacalcet: Results of a prospective randomized controlled study Forni Valentina¹, Pruijm Menno¹,
Drug drug interactions between sucroferric oxyhydroxide and losartan, furosemide, omeprazole, digoxin and warfarin in healthy subjects
 J Nephrol (2014) 27:659 666 DOI 10.1007/s40620-014-0080-1 ORIGINAL ARTICLE Drug drug interactions between sucroferric oxyhydroxide and losartan, furosemide, omeprazole, digoxin and warfarin in healthy
J Nephrol (2014) 27:659 666 DOI 10.1007/s40620-014-0080-1 ORIGINAL ARTICLE Drug drug interactions between sucroferric oxyhydroxide and losartan, furosemide, omeprazole, digoxin and warfarin in healthy
Phosphate binders and metabolic acidosis in patients undergoing maintenance hemodialysis sevelamer hydrochloride, calcium carbonate, and bixalomer
 Hemodialysis International 2015; 19:5459 Phosphate binders and metabolic acidosis in patients undergoing maintenance hemodialysis sevelamer hydrochloride, calcium carbonate, and bixalomer Toru SANAI, 1
Hemodialysis International 2015; 19:5459 Phosphate binders and metabolic acidosis in patients undergoing maintenance hemodialysis sevelamer hydrochloride, calcium carbonate, and bixalomer Toru SANAI, 1
2017 KDIGO Guidelines Update
 2017 KDIGO Guidelines Update Clinic for Hemodialysis Clinical Center University of Sarajevo 13 th Congress of the Balkan cities Association of Nephrology, Dialysis, and Artificial Organs Transplantation
2017 KDIGO Guidelines Update Clinic for Hemodialysis Clinical Center University of Sarajevo 13 th Congress of the Balkan cities Association of Nephrology, Dialysis, and Artificial Organs Transplantation
Draft Labeling Package Insert Not Actual Size. BRAINTREE LABORATORIES, INC. PhosLo Capsules (Calcium Acetate)
 Draft Labeling Package Insert Not Actual Size BRAINTREE LABORATORIES, INC. PhosLo Capsules (Calcium Acetate) DESCRIPTION: Full Size: Each opaque capsule with a white cap and white body is spin printed
Draft Labeling Package Insert Not Actual Size BRAINTREE LABORATORIES, INC. PhosLo Capsules (Calcium Acetate) DESCRIPTION: Full Size: Each opaque capsule with a white cap and white body is spin printed
The Parsabiv Beginner s Book
 The Parsabiv Beginner s Book A quick guide to help you learn about your treatment with Parsabiv and what to expect Indication Parsabiv (etelcalcetide) is indicated for the treatment of secondary hyperparathyroidism
The Parsabiv Beginner s Book A quick guide to help you learn about your treatment with Parsabiv and what to expect Indication Parsabiv (etelcalcetide) is indicated for the treatment of secondary hyperparathyroidism
Clinical Study Synopsis for Public Disclosure
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of the clinical
PRODUCT MONOGRAPH. sevelamer carbonate tablets. 800 mg. Phosphate Binder. ATC code: VO3A EO2
 PRODUCT MONOGRAPH Pr RENVELA sevelamer carbonate tablets 800 mg Phosphate Binder ATC code: VO3A EO2 sanofi-aventis Canada Inc. 2905 Place Louis-R.-Renaud Laval, Quebec H7V 0A3 Date of Revision: March 13,
PRODUCT MONOGRAPH Pr RENVELA sevelamer carbonate tablets 800 mg Phosphate Binder ATC code: VO3A EO2 sanofi-aventis Canada Inc. 2905 Place Louis-R.-Renaud Laval, Quebec H7V 0A3 Date of Revision: March 13,
The hart and bone in concert
 The hart and bone in concert Piotr Rozentryt III Department of Cardiology, Silesian Centre for Heart Disease, Silesian Medical University, Zabrze, Poland Disclosure Research grant, speaker`s fee, travel
The hart and bone in concert Piotr Rozentryt III Department of Cardiology, Silesian Centre for Heart Disease, Silesian Medical University, Zabrze, Poland Disclosure Research grant, speaker`s fee, travel
Effect of lanthanum carbonate and calcium acetate in the treatment of hyperphosphatemia in patients of chronic kidney disease
 Research Article Effect of lanthanum and calcium acetate in the treatment of hyperphosphatemia in patients of chronic kidney disease P. Thomas Scaria, Reneega Gangadhar 1, Ramdas Pisharody 2 ABSTRACT Departments
Research Article Effect of lanthanum and calcium acetate in the treatment of hyperphosphatemia in patients of chronic kidney disease P. Thomas Scaria, Reneega Gangadhar 1, Ramdas Pisharody 2 ABSTRACT Departments
Advances in Peritoneal Dialysis, Vol. 29, 2013
 Advances in Peritoneal Dialysis, Vol. 29, 2013 Takeyuki Hiramatsu, 1 Takahiro Hayasaki, 1 Akinori Hobo, 1 Shinji Furuta, 1 Koki Kabu, 2 Yukio Tonozuka, 2 Yoshiyasu Iida 1 Icodextrin Eliminates Phosphate
Advances in Peritoneal Dialysis, Vol. 29, 2013 Takeyuki Hiramatsu, 1 Takahiro Hayasaki, 1 Akinori Hobo, 1 Shinji Furuta, 1 Koki Kabu, 2 Yukio Tonozuka, 2 Yoshiyasu Iida 1 Icodextrin Eliminates Phosphate
New Medicines Profile. December 2013 Issue No. 13/04. Colestilan
 New Medicines Profile December 2013 Issue. 13/04 Concise evaluated information to support the managed entry of new medicines in the NHS Summary (BindRen ) is an oral, non-absorbed, non-calcium, nonmetallic
New Medicines Profile December 2013 Issue. 13/04 Concise evaluated information to support the managed entry of new medicines in the NHS Summary (BindRen ) is an oral, non-absorbed, non-calcium, nonmetallic
THE IMPACT OF SERUM PHOSPHATE LEVELS IN CKD-MBD PROGRESSION
 THE IMPACT OF SERUM PHOSPHATE LEVELS IN CKD-MBD PROGRESSION Mario Cozzolino, MD, PhD, Fellow of the European Renal Association Department of Health Sciences University of Milan Renal Division & Laboratory
THE IMPACT OF SERUM PHOSPHATE LEVELS IN CKD-MBD PROGRESSION Mario Cozzolino, MD, PhD, Fellow of the European Renal Association Department of Health Sciences University of Milan Renal Division & Laboratory
( ) , (Donabedian, 1980) We would not choose any treatment with poor outcomes
 ..., 2013 Amgen. 1 ? ( ), (Donabedian, 1980) We would not choose any treatment with poor outcomes 1. :, 2. ( ): 3. :.,,, 4. :, [Biomarkers Definitions Working Group, 2001]., (William M. Bennet, Nefrol
..., 2013 Amgen. 1 ? ( ), (Donabedian, 1980) We would not choose any treatment with poor outcomes 1. :, 2. ( ): 3. :.,,, 4. :, [Biomarkers Definitions Working Group, 2001]., (William M. Bennet, Nefrol
Pediatric Nutrition and Kidney Disease
 Pediatric Nutrition and Kidney Disease Loai Eid, MD, MSHS, FAAP Consultant Pediatric Nephrologist Pediatric Nephrology & Hypertension Division Chief Dubai Hospital - DHA 26 th October, 2017 Objectives
Pediatric Nutrition and Kidney Disease Loai Eid, MD, MSHS, FAAP Consultant Pediatric Nephrologist Pediatric Nephrology & Hypertension Division Chief Dubai Hospital - DHA 26 th October, 2017 Objectives
What is renal failure?
 What is renal failure? The kidney is a very important organ, and cannot be restored to health once it fails. However, it is possible to avoid or retard the deterioration of its functionality if therapy
What is renal failure? The kidney is a very important organ, and cannot be restored to health once it fails. However, it is possible to avoid or retard the deterioration of its functionality if therapy
SCIENTIFIC DISCUSSION
 European Medicines Agency London, 01 June 2007 Product Name : Renagel Procedure No: EMEA/H/C/000254/II/56 SCIENTIFIC DISCUSSION 1/11 1. Introduction Renagel (sevelamer), a non-absorbed, calcium and metal-free
European Medicines Agency London, 01 June 2007 Product Name : Renagel Procedure No: EMEA/H/C/000254/II/56 SCIENTIFIC DISCUSSION 1/11 1. Introduction Renagel (sevelamer), a non-absorbed, calcium and metal-free
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Reference Number: CP.PMN.04 Effective Date: 11.15.17 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: Reference Number: CP.PMN.04 Effective Date: 11.15.17 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy for important
Treatment of hyperphosphatemia in hemodialysis patients: The Calcium Acetate Renagel Evaluation (CARE Study)
 Kidney International, Vol. 65 (2004), pp. 1914 1926 Treatment of hyperphosphatemia in hemodialysis patients: The Calcium Acetate Renagel Evaluation (CARE Study) WAJEH Y. QUNIBI,ROBERT E. HOOTKINS,LAVETA
Kidney International, Vol. 65 (2004), pp. 1914 1926 Treatment of hyperphosphatemia in hemodialysis patients: The Calcium Acetate Renagel Evaluation (CARE Study) WAJEH Y. QUNIBI,ROBERT E. HOOTKINS,LAVETA
Report on the Deliberation Results
 Report on the Deliberation Results March 3, 2014 Evaluation and Licensing Division, Pharmaceutical and Food Safety Bureau Ministry of Health, Labour and Welfare [Brand name] (a) Samsca Tablets 7.5 mg (b)
Report on the Deliberation Results March 3, 2014 Evaluation and Licensing Division, Pharmaceutical and Food Safety Bureau Ministry of Health, Labour and Welfare [Brand name] (a) Samsca Tablets 7.5 mg (b)
Do We Do Too Many Parathyroidectomies in Dialysis? Sagar Nigwekar MD, MMSc Massachusetts General Hospital
 Do We Do Too Many Parathyroidectomies in Dialysis? Sagar Nigwekar MD, MMSc Massachusetts General Hospital E-mail: snigwekar@mgh.harvard.edu March 13, 2017 Disclosures statement: Consultant: Allena, Becker
Do We Do Too Many Parathyroidectomies in Dialysis? Sagar Nigwekar MD, MMSc Massachusetts General Hospital E-mail: snigwekar@mgh.harvard.edu March 13, 2017 Disclosures statement: Consultant: Allena, Becker
Incorporating K/DOQI Using a Novel Algorithm Approach: Regina Qu Appelle s Experience
 Incorporating K/DOQI Using a Novel Algorithm Approach: Regina Qu Appelle s Experience Michael Chan, Renal Dietitian Regina Qu Appelle Health Region BC Nephrology Days There is a strong association among
Incorporating K/DOQI Using a Novel Algorithm Approach: Regina Qu Appelle s Experience Michael Chan, Renal Dietitian Regina Qu Appelle Health Region BC Nephrology Days There is a strong association among
PROs for Drug Development. Melanie Blank, MD
 PROs for Drug Development in Chronic Kidney Disease Melanie Blank, MD Disclaimer The views expressed here represent my opinions and do not necessarily represent the views of the FDA. Overview Stagnation
PROs for Drug Development in Chronic Kidney Disease Melanie Blank, MD Disclaimer The views expressed here represent my opinions and do not necessarily represent the views of the FDA. Overview Stagnation
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Sevelamer carbonate Zentiva 800 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 800
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Sevelamer carbonate Zentiva 800 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 800
Should cinacalcet be used in patients who are not on dialysis?
 Should cinacalcet be used in patients who are not on dialysis? Jorge B Cannata-Andía and José Luis Fernández-Martín Affiliations: Bone and Mineral Research Unit. Hospital Universitario Central de Asturias.
Should cinacalcet be used in patients who are not on dialysis? Jorge B Cannata-Andía and José Luis Fernández-Martín Affiliations: Bone and Mineral Research Unit. Hospital Universitario Central de Asturias.
Sensipar (cinacalcet) Tablets
 DESCRIPTION Sensipar (cinacalcet) is a calcimimetic agent that increases the sensitivity of the calcium-sensing receptor to activation by extracellular calcium. Its empirical formula is C 22 H 22 F 3 N
DESCRIPTION Sensipar (cinacalcet) is a calcimimetic agent that increases the sensitivity of the calcium-sensing receptor to activation by extracellular calcium. Its empirical formula is C 22 H 22 F 3 N
Managing Acid Base and Electrolyte Disturbances with RRT
 Managing Acid Base and Electrolyte Disturbances with RRT John R Prowle MA MSc MD MRCP FFICM Consultant in Intensive Care & Renal Medicine RRT for Regulation of Acid-base and Electrolyte Acid base load
Managing Acid Base and Electrolyte Disturbances with RRT John R Prowle MA MSc MD MRCP FFICM Consultant in Intensive Care & Renal Medicine RRT for Regulation of Acid-base and Electrolyte Acid base load
PART FOUR. Metabolism and Nutrition
 PART FOUR Metabolism and Nutrition Advances in Peritoneal Dialysis, Vol. 22, 2006 Costas Fourtounas, Eirini Savidaki, Marilena Roumelioti, Periklis Dousdampanis, Andreas Hardalias, Pantelitsa Kalliakmani,
PART FOUR Metabolism and Nutrition Advances in Peritoneal Dialysis, Vol. 22, 2006 Costas Fourtounas, Eirini Savidaki, Marilena Roumelioti, Periklis Dousdampanis, Andreas Hardalias, Pantelitsa Kalliakmani,
4Q and Full Year 2017 Financial Results Call February 7, 2018
 4Q and Full Year 2017 Financial Results Call February 7, 2018 Agenda Topic Welcome Opening Remarks 4Q 2017 Results Commercial Progress Question & Answer Speakers Amy Sullivan, SVP, Corporate Affairs Greg
4Q and Full Year 2017 Financial Results Call February 7, 2018 Agenda Topic Welcome Opening Remarks 4Q 2017 Results Commercial Progress Question & Answer Speakers Amy Sullivan, SVP, Corporate Affairs Greg
CKD: Bone Mineral Metabolism. Peter Birks, Nephrology Fellow
 CKD: Bone Mineral Metabolism Peter Birks, Nephrology Fellow CKD - KDIGO Definition and Classification of CKD CKD: abnormalities of kidney structure/function for > 3 months with health implications 1 marker
CKD: Bone Mineral Metabolism Peter Birks, Nephrology Fellow CKD - KDIGO Definition and Classification of CKD CKD: abnormalities of kidney structure/function for > 3 months with health implications 1 marker
Dr. M.Mothilal Assistant professor
 Dr. M.Mothilal Assistant professor Bioavailability is a measurement of the rate and extent of drug that reaches the systemic circulation from a drug product or a dosage form. There are two different types
Dr. M.Mothilal Assistant professor Bioavailability is a measurement of the rate and extent of drug that reaches the systemic circulation from a drug product or a dosage form. There are two different types
Clinical Study Synopsis for Public Disclosure
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data. The synopsis
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data. The synopsis
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Renvela 800 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 800 mg sevelamer carbonate.
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Renvela 800 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 800 mg sevelamer carbonate.
AURYXIA IS ON FORMULARY AT ALL MAJOR MEDICARE PART D PLANS
 Hyperphosphatemia CKD On Dialysis AURYXIA IS ON FORMULARY AT ALL MAJOR MEDICARE PART D PLANS Account Common Plan Names Reimbursement Status Caremark OptumRx/ UnitedHealthcare Humana Express Scripts Aetna
Hyperphosphatemia CKD On Dialysis AURYXIA IS ON FORMULARY AT ALL MAJOR MEDICARE PART D PLANS Account Common Plan Names Reimbursement Status Caremark OptumRx/ UnitedHealthcare Humana Express Scripts Aetna
Modeling & Simulation to support evaluation of Safety and Efficacy of Drugs in Older Patients
 Modeling & Simulation to support evaluation of Safety and Efficacy of Drugs in Older Patients Eva Bredberg, Director Global Clinical Pharmacology, AstraZeneca On behalf of EFPIA EMA Geriatrics Workshop
Modeling & Simulation to support evaluation of Safety and Efficacy of Drugs in Older Patients Eva Bredberg, Director Global Clinical Pharmacology, AstraZeneca On behalf of EFPIA EMA Geriatrics Workshop
Stages of Chronic Kidney Disease (CKD)
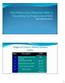 Early Treatment is the Key Stages of Chronic Kidney Disease (CKD) Stage Description GFR (ml/min/1.73 m 2 ) >90 1 Kidney damage with normal or GFR 2 Mild decrease in GFR 60-89 3 Moderate decrease in GFR
Early Treatment is the Key Stages of Chronic Kidney Disease (CKD) Stage Description GFR (ml/min/1.73 m 2 ) >90 1 Kidney damage with normal or GFR 2 Mild decrease in GFR 60-89 3 Moderate decrease in GFR
on metabolic acidosis in dialysis p Harada, Takashi; Kohno, Shigeru
 NAOSITE: Nagasaki University's Ac Title Author(s) Citation Effect of switching from sevelamer on metabolic acidosis in dialysis p Minami, Kana; Nishino, Tomoya; Arai Yamashita, Hiroshi; Uramatsu, Tadas
NAOSITE: Nagasaki University's Ac Title Author(s) Citation Effect of switching from sevelamer on metabolic acidosis in dialysis p Minami, Kana; Nishino, Tomoya; Arai Yamashita, Hiroshi; Uramatsu, Tadas
Sevelamer Decreases Serum Uric Acid Concentration through Adsorption of Uric Acid in Maintenance Hemodialysis Patients
 ORIGINAL ARTICLE Sevelamer Decreases Serum Uric Acid Concentration through Adsorption of Uric Acid in Maintenance Hemodialysis Patients Iwao Ohno 1, Yuichiro Yamaguchi 1, Hajime Saikawa 1, Daijiro Uetake
ORIGINAL ARTICLE Sevelamer Decreases Serum Uric Acid Concentration through Adsorption of Uric Acid in Maintenance Hemodialysis Patients Iwao Ohno 1, Yuichiro Yamaguchi 1, Hajime Saikawa 1, Daijiro Uetake
Clinical Guideline Bone chemistry management in adult renal patients on dialysis
 Clinical Guideline Bone chemistry management in adult renal patients on dialysis This guidance covers how to: Maintain serum phosphate 0.8 to 1.7mmol/L 1 Maintain serum corrected calcium 2.1 to 2.5mmol/L
Clinical Guideline Bone chemistry management in adult renal patients on dialysis This guidance covers how to: Maintain serum phosphate 0.8 to 1.7mmol/L 1 Maintain serum corrected calcium 2.1 to 2.5mmol/L
BONE AND MINERAL METABOLISM in the PD PATIENT
 BONE AND MINERAL METABOLISM in the PD PATIENT John Burkart, MD Professor of Medicine/Nephrology Wake Forest University Baptist Medical Center Chief Medical Officer Health Systems Management Maria V. DeVita,
BONE AND MINERAL METABOLISM in the PD PATIENT John Burkart, MD Professor of Medicine/Nephrology Wake Forest University Baptist Medical Center Chief Medical Officer Health Systems Management Maria V. DeVita,
Ferric citrate in end-stage kidney disease as a phosphate binder and source of iron: a review of clinical trials
 Ferric citrate in end-stage kidney disease as a phosphate binder and source of iron: a review of clinical trials Hyperphosphatemia is highly prevalent among patients with end-stage kidney disease and is
Ferric citrate in end-stage kidney disease as a phosphate binder and source of iron: a review of clinical trials Hyperphosphatemia is highly prevalent among patients with end-stage kidney disease and is
SUPPLEMENTAL MATERIAL
 SUPPLEMENTAL MATERIAL Randomized Controlled Trial of to Increase Serum Bicarbonate in Chronic Kidney Disease Patients David A. Bushinsky a, Thomas Hostetter b, Gerrit Klaerner c, Yuri Stasiv c, Claire
SUPPLEMENTAL MATERIAL Randomized Controlled Trial of to Increase Serum Bicarbonate in Chronic Kidney Disease Patients David A. Bushinsky a, Thomas Hostetter b, Gerrit Klaerner c, Yuri Stasiv c, Claire
Case Study: Chronic Kidney Disease
 Taylor Zwimpfer Joan Rupp Nutrition 409 23 September 2014 Case Study: Chronic Kidney Disease 1. Kidneys act to maintain the balance of fluids, electrolytes and organic solutes in the body through filtration
Taylor Zwimpfer Joan Rupp Nutrition 409 23 September 2014 Case Study: Chronic Kidney Disease 1. Kidneys act to maintain the balance of fluids, electrolytes and organic solutes in the body through filtration
Therapeutic golas in the treatment of CKD-MBD
 Therapeutic golas in the treatment of CKD-MBD Hemodialysis clinic Clinical University Center Sarajevo Bantao, 04-08.10.2017, Sarajevo Abbvie Satellite symposium 06.10.2017 Chronic Kidney Disease Mineral
Therapeutic golas in the treatment of CKD-MBD Hemodialysis clinic Clinical University Center Sarajevo Bantao, 04-08.10.2017, Sarajevo Abbvie Satellite symposium 06.10.2017 Chronic Kidney Disease Mineral
Sodium and Potassium Intake and Cardiovascular and Bone Health:
 Sodium and Potassium Intake and Cardiovascular and Bone Health: How Important is the Ratio? Connie M. Weaver Nutrition Science Purdue University Disclosures Boards/Scientific Advisory Committees ILSI Showalter
Sodium and Potassium Intake and Cardiovascular and Bone Health: How Important is the Ratio? Connie M. Weaver Nutrition Science Purdue University Disclosures Boards/Scientific Advisory Committees ILSI Showalter
Section Questions Answers
 Section Questions Answers Guide to CKD Screening and Evaluation -Alec Otteman, MD Delaying Progression - Paul Drawz, MD, MHS, MS 1. Modifiable risk factors for CKD include: a. Diabetes b. Hypertension
Section Questions Answers Guide to CKD Screening and Evaluation -Alec Otteman, MD Delaying Progression - Paul Drawz, MD, MHS, MS 1. Modifiable risk factors for CKD include: a. Diabetes b. Hypertension
Prurisol: A New Small Molecule under investigation for the treatment of Psoriasis
 Prurisol: A New Small Molecule under investigation for the treatment of Psoriasis Krishna Menon, RCM, PhD, VMD, President of Research and Chief Scientific Officer MEDICAL DERMATOLOGY THERAPEUTIC R&D AND
Prurisol: A New Small Molecule under investigation for the treatment of Psoriasis Krishna Menon, RCM, PhD, VMD, President of Research and Chief Scientific Officer MEDICAL DERMATOLOGY THERAPEUTIC R&D AND
COMPOSITION. A film coated tablet contains. Active ingredient: irbesartan 75 mg, 150 mg or 300 mg. Rotazar (Film coated tablets) Irbesartan
 Rotazar (Film coated tablets) Irbesartan Rotazar 75 mg, 150 mg, 300 mg COMPOSITION A film coated tablet contains Active ingredient: irbesartan 75 mg, 150 mg or 300 mg. Rotazar 75 mg, 150 mg, 300 mg PHARMACOLOGICAL
Rotazar (Film coated tablets) Irbesartan Rotazar 75 mg, 150 mg, 300 mg COMPOSITION A film coated tablet contains Active ingredient: irbesartan 75 mg, 150 mg or 300 mg. Rotazar 75 mg, 150 mg, 300 mg PHARMACOLOGICAL
BASELINE CHARACTERISTICS OF THE STUDY POPULATION
 COMPARISON OF TREATING METABOLIC ACIDOSIS IN CKD STAGE 4 HYPERTENSIVE KIDNEY DISEASE WITH FRUITS & VEGETABLES OR SODIUM BICARBONATE This was a 1-year, single-center, prospective, randomized, interventional
COMPARISON OF TREATING METABOLIC ACIDOSIS IN CKD STAGE 4 HYPERTENSIVE KIDNEY DISEASE WITH FRUITS & VEGETABLES OR SODIUM BICARBONATE This was a 1-year, single-center, prospective, randomized, interventional
Original epidemiologic studies 1 have suggested that approximately
 Factors for Increased Morbidity and Mortality in Uremia: Hyperphosphatemia Nathan W. Levin, Frank A. Gotch, and Martin K. Kuhlmann Hyperphosphatemia is a metabolic abnormality present in the majority of
Factors for Increased Morbidity and Mortality in Uremia: Hyperphosphatemia Nathan W. Levin, Frank A. Gotch, and Martin K. Kuhlmann Hyperphosphatemia is a metabolic abnormality present in the majority of
VA/DoD Clinical Practice Guideline for the Management of Chronic Kidney Disease in Primary Care (2008) PROVIDER REFERENCE CARDS Chronic Kidney Disease
 VA/DoD Clinical Practice Guideline for the Management of Chronic Kidney Disease in Primary Care (2008) PROVIDER REFERECE CARDS Chronic Kidney Disease CKD VA/DoD Clinical Practice Guideline for the Management
VA/DoD Clinical Practice Guideline for the Management of Chronic Kidney Disease in Primary Care (2008) PROVIDER REFERECE CARDS Chronic Kidney Disease CKD VA/DoD Clinical Practice Guideline for the Management
Hyperparathyroidism: Operative Considerations. Financial Disclosures: None. Hyperparathyroidism. Hyperparathyroidism 11/10/2012
 Hyperparathyroidism: Operative Considerations Financial Disclosures: None Steven J Wang, MD FACS Associate Professor Dept of Otolaryngology-Head and Neck Surgery University of California, San Francisco
Hyperparathyroidism: Operative Considerations Financial Disclosures: None Steven J Wang, MD FACS Associate Professor Dept of Otolaryngology-Head and Neck Surgery University of California, San Francisco
New aspects of acid-base disorders
 New aspects of acid-base disorders I. David Weiner, M.D. C. Craig and Audrae Tisher Chair in Nephrology Division of Nephrology, Hypertension and Transplantation University of Florida College of Medicine
New aspects of acid-base disorders I. David Weiner, M.D. C. Craig and Audrae Tisher Chair in Nephrology Division of Nephrology, Hypertension and Transplantation University of Florida College of Medicine
Literature Scan: Phosphate Binders
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Prevalence of malnutrition in dialysis
 ESPEN Congress Cannes 2003 Organised by the Israel Society for Clinical Nutrition Education and Clinical Practice Programme Session: Nutrition and the Kidney Malnutrition and Haemodialysis Doctor Noël
ESPEN Congress Cannes 2003 Organised by the Israel Society for Clinical Nutrition Education and Clinical Practice Programme Session: Nutrition and the Kidney Malnutrition and Haemodialysis Doctor Noël
Thus, we can group the entire loading dose together as though it was given as a single dose, all administered when the first dose was given.
 PHA 5128 Dose Optimization II, Spring 2012, Case Study V Solution If you have any questions regarding this case study, do not hesitate to contact Benjamin Weber (benjaminweber@ufl.edu). Please remember
PHA 5128 Dose Optimization II, Spring 2012, Case Study V Solution If you have any questions regarding this case study, do not hesitate to contact Benjamin Weber (benjaminweber@ufl.edu). Please remember
Corporate Presentation January 2013
 Corporate Presentation January 2013 0 Forward-Looking Statements Certain statements and information included in this presentation are forwardlooking statements under the Private Securities Litigation Reform
Corporate Presentation January 2013 0 Forward-Looking Statements Certain statements and information included in this presentation are forwardlooking statements under the Private Securities Litigation Reform
RENAL FAILURE IN CHILDREN Dr. Mai Mohamed Elhassan Assistant Professor Jazan University
 RENAL FAILURE IN CHILDREN Dr. Mai Mohamed Elhassan Assistant Professor Jazan University OBJECTIVES By the end of this lecture each student should be able to: Define acute & chronic kidney disease(ckd)
RENAL FAILURE IN CHILDREN Dr. Mai Mohamed Elhassan Assistant Professor Jazan University OBJECTIVES By the end of this lecture each student should be able to: Define acute & chronic kidney disease(ckd)
2.0 Synopsis. ABT-358/Paricalcitol M Clinical Study Report R&D/09/1255. (For National Authority Use Only) to Part of Dossier: Volume:
 2.0 Synopsis Title of Study: Late Phase II Study of Paricalcitol Injection Dose-response study of paricalcitol injection in chronic kidney disease subjects receiving hemodialysis with secondary hyperparathyroidism
2.0 Synopsis Title of Study: Late Phase II Study of Paricalcitol Injection Dose-response study of paricalcitol injection in chronic kidney disease subjects receiving hemodialysis with secondary hyperparathyroidism
Important aspects of acid-base disorders
 Important aspects of acid-base disorders I. David Weiner, M.D. Co-holder, C. Craig and Audrae Tisher Chair in Nephrology Division of Nephrology, Hypertension and Transplantation University of Florida College
Important aspects of acid-base disorders I. David Weiner, M.D. Co-holder, C. Craig and Audrae Tisher Chair in Nephrology Division of Nephrology, Hypertension and Transplantation University of Florida College
Clinical rationale of sucroferric oxyhydroxide for controlling hyperphosphatemia in patients with chronic kidney disease
 oxide ients Clinical rationale of sucroferric oxyhydroxide for controlling hyperphosphatemia in patients with chronic kidney disease Sucroferric oxyhydroxide (Velphoro ; Vifor Fresenius Medical Care Renal
oxide ients Clinical rationale of sucroferric oxyhydroxide for controlling hyperphosphatemia in patients with chronic kidney disease Sucroferric oxyhydroxide (Velphoro ; Vifor Fresenius Medical Care Renal
Effect of Patiromer on Urinary Ion Excretion in Healthy Adults
 Article Effect of Patiromer on Urinary Ion Excretion in Healthy Adults David A. Bushinsky,* David M. Spiegel, Coleman Gross, Wade W. Benton, Jeanene Fogli, Kathleen M. Hill Gallant, Charles Du Mond, Geoffrey
Article Effect of Patiromer on Urinary Ion Excretion in Healthy Adults David A. Bushinsky,* David M. Spiegel, Coleman Gross, Wade W. Benton, Jeanene Fogli, Kathleen M. Hill Gallant, Charles Du Mond, Geoffrey
The Management of Phosphataemia in Renal Dogs or Cats
 The Management of Phosphataemia in Renal Dogs or Cats improves both the quality of life and the life expectancy of animals. THE CARDIOLOGY - NEPHROLOGY DIMENSION PAIN INFLAMMATION ANTI-INFECTIVE PHOSPHATAEMIA
The Management of Phosphataemia in Renal Dogs or Cats improves both the quality of life and the life expectancy of animals. THE CARDIOLOGY - NEPHROLOGY DIMENSION PAIN INFLAMMATION ANTI-INFECTIVE PHOSPHATAEMIA
The CARI Guidelines Caring for Australasians with Renal Impairment. ACE Inhibitor and Angiotensin II Antagonist Combination Treatment GUIDELINES
 ACE Inhibitor and Angiotensin II Antagonist Combination Treatment Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES No recommendations possible based on Level
ACE Inhibitor and Angiotensin II Antagonist Combination Treatment Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES No recommendations possible based on Level
