tlaboratory of Molecular Biology, National Cancer Institute, National Institutes of Health, Bethesda, MD 20892, U.S.A.
|
|
|
- Alison Atkins
- 6 years ago
- Views:
Transcription
1 Biochem. J. (1987) 248, (Printed in Great Britain) The identification of the major excreted protein (MEP) from a transformed mouse fibroblast cell line as a catalytically active precursor form of cathepsin L 449 Robert W. MASON,* Susannah GALt and Michael M. GOTTESMANt *Department of Biochemistry, Strangeways Research Laboratory, Worts Causeway, Cambridge CB1 4RN, U.K., and tlaboratory of Molecular Biology, National Cancer Institute, National Institutes of Health, Bethesda, MD 20892, U.S.A. The major excreted protein (MEP) purified from Kirsten-virus-transformed 3T3 fibroblasts and mature human cathepsin L were compared in respect to a number of catalytic criteria and found to be similar. The Mr of MEP is 39000, whereas that of mature human cathepsin L is Sequence data suggested that MEP could be a pro-form of mouse cathepsin L. Both enzymes acted on the synthetic substrate benzyloxycarbonyl-phe-arg-7-(4-methyl)coumarylamide with similar catalytic constants and acted optimally at ph 5.5. Both were rapidly inactivated by the active-site-directed inhibitors benzyloxycarbonyl- Phe-Phe-diazomethane and L-3-carboxy-trans-2,3-epoxypropionyl-leucylamido-(4-guanidino)butane, and furthermore, 3H-labelled L-3-carboxy-trans-2,3-epoxypropionyl-leucylamido-(4-acetamido)butane, which binds covalently to the heavy chain of mature cathepsin L, also bound to MEP. MEP autolyses rapidly at ph 3.0 to give lower-mr (35000 and 30000) forms, but all forms react with the radiolabelled inhibitor. No autolysis occurred above ph 5.0. MEP hydrolysed azocasein at ph 5.0, demonstrating that it is capable of hydrolysing protein substrates without autolytic activation. Unlike mature forms of cathepsin L, MEP is stable, but not active, at neutral ph. The present work shows that cathepsin L can be secreted as a higher- Mr precursor that is stable in extracellular fluids but only active where local ph values fall below 6.0. These results suggest that the extra N-terminal peptide on MEP is not an activation peptide, but is a regulatory peptide affecting the ph-stability and activity of mouse cathepsin L. INTRODUCTION The cysteine proteinases cathepsins B and L are located in lysosomes, as demonstrated by subcellularfractionation studies and cytochemical and immunochemical staining (Kirschke et al., 1977; Graf et al., 1979; Etherington et al., 1984). This may not be the only location of these enzymes, however. One immunochemical study has also shown an extracellular location of cathepsin B around synovial cells (Poole & Mort, 1981). Furthermore, cathepsin B-like activity has been detected in synovial fluid, bronchoalveolar lavage fluid and sputum (Mort et al., 1983; Chang et al., 1986; Burnett et al., 1983). Precursor forms of cathepsin B have been identified immunochemically in ascites fluid, but have not yet been purified to enable a direct demonstration that these forms are proteolytically active (Mort & Recklies, 1986). The function of these secreted enzymes is not known, but extracellular levels of cathepsin B-like activity have been correlated with metastasis (Sloane & Honn, 1984). A secreted precursor form of a lysosomal cysteine proteinase has now been identified and purified. Kirstenvirus-transformed mouse 3T3 fibroblasts secrete a protein of Mr that was named 'MEP' (Gottesman, 1978). This purified protein exhibited proteolytic activity against bovine serum albumin at ph 3.0. The incubation conditions used resulted in a decrease in the Mr of MEP (Gal & Gottesman, 1986a). At ph 3.0, the enzyme also cleaved the B-chain of insulin, cleaving at the same sites as rat liver cathepsin L at ph 6.0 (Gal & Gottesman, 1986b), suggesting that the two enzymes might be related. The protein sequence derived from the cdna of MEP, and its analogue expressed by mouse peritoneal macrophages, was found to be homologous with the protein sequence of human cathepsin L (Portnoy et al., 1986; Denhardt et al., 1986; Mason et al., 1986; B. Troen, S. Gal & M. M. Gottesman, unpublished work). Over 80% identity of the homologous sequences strongly suggested that these enzymes are species variants of the same protein. MEP has a significantly higher Mr than mature lysosomal forms of cathepsin L and has been shown to be processed to lower-mr forms similar in size to mature cathepsin L (Gal et al., 1985) and hence most probably represents a precursor form of this enzyme. The present paper describes the characterization of the proteolytic activity of MEP and a comparison of its enzymic activity with that of the mature forms of cathepsin L. MATERIALS AND METHODS Materials Human cathepsin L was purified as described by Mason et al. (1985), and MEP was purified as described by Gal & Gottesman (1986a). [3H]Ac-Ep-459 (sp. Abbreviations used: Z-, benzyloxycarbonyl; NHMec, 7-(4-methyl)coumarylamide; -CHN2, diazomethane; MEP, major excreted protein; E-64, L-3-carboxy-trans-2,3-epoxypropionyl-leucylamido-(4-guanidino)butane; Ac-Ep-459, L-3-carboxy-trans-2,3-epoxypropionyl-leucylamido-(4-acetamido)butane. Vol. 248
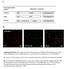
2 450 radioactivity 83 Ci/mol) was prepared as described by Parkes et al. (1985). Dithiothreitol, bovine serum albumin and E-64 [as L-trans-epoxysuccinyl-leucylamido-(4- guanidino)butane] were from Sigma Chemical Co., Poole, Dorset, U.K. Azocasein was prepared as described by Barrett & Kirschke (1981). Z-Phe-Phe-CHN2 and Z- Phe-Ala-CHN2 were gifts from Dr. E. N. Shaw, Friedrich Meischer Institute, Basle, Switzerland. Aminomethylcoumarin substrates were from Cambridge Research Biochemicals, Cambridge, U.K. Methods Activity against synthetic substrates. MEP was routinely assayed for activity against Z-Phe-Arg-NHMec at ph 5.5, as described previously for human cathepsin L (Mason et al., 1985). Identical conditions were employed when Z-Arg-Arg-NHMec and Arg-NHMec were used as substrates. Assays were started by adding enzyme, and initial rates were recorded continuously. Catalytic constants were determined by the method of Wilkinson (1961). For ph profiles, buffers used were 100 mmsodium formate (ph ), 100 mm-sodium acetate (ph ) and 100 mm-sodium phosphate (ph ). For assays of modified (i.e. autolysed) MEP, the enzyme was pretreated at ph 3.0, 37 C for 2 min immediately before assay. Stability of MEP. MEP (1,ug) was incubated in 100 1l of a range of buffers, ph , at 37 C for 1 h. Enzyme was then diluted in 100 mm-sodium acetate buffer, ph 5.5, and assayed as described previously (Mason et al., 1985). Inactivation of MEP by active-site-directed inhibitors. MEP was assayed continuously at ph 5.5 by using standard conditions for the continuous rate assays of human cathepsin L (Mason et al., 1985). After 5 min the inhibitor was added and the rate of inactivation calculated as described previously (Mason et al., 1985). Rates were determined at inhibitor concentrations ranging from 50 to 200 nm in the presence of 1-5 4tM-Z- Phe-Arg-NHMec. Rates of inactivation were dependent upon substrate concentration, and replots of reciprocal rate against substrate concentration gave rates independent of substrate and also gave a separate estimate of Km. Reaction with I3HIAc-Ep-459. MEP (3,tg) was incubated with 200 mm-sodium acetate buffer, ph 5.0, containing 1 mm-edta and 1 mm-dithiothreitol for 30 min at 37 C in a final volume of 15,1. A 5 pul portion of [3H]Ac-Ep-459 (550 #M) was then added and the mixture incubated for a further 30 min. An equal volume of SDS sample buffer (Bury, 1981) was then added and the mixture boiled before it was subjected to 10 % (w/v) polyacrylamide-gel electrophoresis as described previously (Bury, 1981). Other samples of MEP (3,ug) were either preincubated with 200 mm-sodium formate buffer, ph 3.0, containing 1 mm-edta and 1 mm-dithiothreitol for 2 min at 37 C before addition of [3H]Ac-Ep-459, or added to this buffer containing the radiolabelled inhibitor Ġels were sliced for extraction of radioactivity and counted as described by Parkes et al. (1985). Efficiency of extraction of labelled proteins was determined by using active-site-titrated papain as a standard. About 50% of R. W. Mason, S. Gal and M. M. Gottesman the radioactivity could be extracted with an S.D. of no more than 10 %. Autolysis of MEP. MEP was incubated in 100 mmsodium formate buffer, ph 3.0, containing 1 mm-edta at 37 C for 2 min. The reaction mixture was then diluted with 100 mm-sodium acetate buffer containing 1 mm- EDTA, ph 5.5, and stored at 4 C until required. Activity against bovine serum albumin. MEP (0.3,g, 1,u) was added to either 100 mm-sodium formate, ph 3.0, containing 1 mm-edta and 1 mm-dithiothreitol (5,1) or 100 mm-sodium acetate, ph 5.0, containing 1 mm-edta and 1 mm-dithiothreitol (5 IA), and incubated at 37 'C. After 1 min, bovine serum albumin (3,sg, S,ul) was added and incubated at 37 'C for 1 h. A sample of enzyme was preincubated at ph 3.0 with the formate buffer and E-64 (10 /M final concn.) and assayed similarly. The reaction was stopped by boiling in SDS sample buffer and samples run in 10 %-(w/v)-polyacrylamide gels as described by Bury (1981). Human cathepsin L (10 ng of active enzyme) was assayed similarly. Activity against azocasein. MEP (0.3,ug) was incubated with azocasein (0.1 %) dissolved in buffer containing 1 mm-edta and 1 mm-dithiothreitol in a total volume of 1.0 ml. Buffers used were 100 mm-sodium acetate (ph ) or 100 mm-sodium phosphate (ph ). Incubation was) for 2 h at 30 'C, and the reaction was stopped by adding 200 1lt of 20 % (w/v) trichloroacetic acid. Soluble fragments of azocasein were measured by their A366 as described previously (Barrett & Kirschke, 1981). For comparison, human cathepsin L (30 ng of active enzyme) was assayed similarly. RESULTS Activity against synthetic substrates MEP was active against the synthetic cathepsin L substrate Z-Phe-Arg-NHMec. The initial activity of MEP against this substrate was found to be maximal at ph 5.5 (Fig. 1). There was a second peak of activity at ph 3.0. This ph-activity profile differs from that of mature human liver cathepsin L, which has a broad initial ph-activity profile, with activity at least 50% of maximal over the range ph (Mason et al., 1985). Treatment of MEP at ph 3.0 has been shown to cause autolysis to yield a smaller protein, similar in size to the mature lysosomal form of the enzyme (Gal & Gottesman, 1986a). To determine if autolysed MEP has the same ph-activity curve as cathepsin L, MEP was incubated at ph 3.0 and 37 'C for 2 min and its initial activity against Z-Phe-Arg-NHMec measured (Fig. 1). The ph-activity profile of this acid-treated protein was similar to that of mature human cathepsin L, and did not exhibit the biphasic profile seen for non-autolysed MEP. The high activity seen for 'intact MEP' at ph 3.0 most probably reflects activity of the protein autolysed during the assay. When activity was measured continuously at ph 4.0, activity slowly increased until the rate was almost equivalent to that of the enzyme assayed at ph 3.0 (Fig. 2). The- most- likely interpretation of this result is that MEP is rapidly converted into a lower-mr catalytically 1987
3 Major excreted protein as active cathepsin L precursor O CLL I 'a. _ w <, O 0) (U c._._ 0 C~- zn>, E 4b, *._ E -5 0 U (U I z i( C( -c N.m C 4m 0) co -o (A V ph Fig. 1. ph-activity NHMec profile of MEP against MEP was diluted in ph 5.5 activating buffer and stored on ice for 10 min. The initial activity of enzyme was measured at a range of ph values as described in the Materials and methods section (0). MEP was autolysed to a lower-mr form, and the initial activity of the modified enzyme was measured at a range of ph values as described in the Materials and methods section (A). -a 900 C, D U. 00 Z-Phe-Arg Time (min) Fig. 2. Activity of MEP against Z-Phe-Arg-NHMec at ph 4.0 MEP was assayed continuously in the presence of 5/ M- Z-Phe-Arg-NHMec in 100 mm-sodium formate buffer, ph 4.0, containing 1 mm-dithiothreitol and 1 mm-edta. active form at ph 3.0, but is activated by autolysis much more slowly at ph 4.0. Although the autolysed form of MEP is catalytically similar to mature forms of cathepsin L, the higher-m, form has a- much more restricted ph-activity range. With respect to proteolytic activity, MEP was found to Vol ph Fig. 3. ph-stability of MEP MEP was preincubated for 1 h at 37 C in a range of buffers (ph ) as described in the Materials and methods section. Activity remaining was assayed at ph 5.5 with Z-Phe-Arg-NHMec as substrate and is expressed as a percentage of activity measured without preincubation (0)^ The stability of human cathepsin L is shown for comparison (A). be stable over the ph range , but unstable below ph 4.5 (Fig. 3). This contrasts with the activity of the mature forms of cathepsin L, which are unstable above ph 6.5 and below ph 4.0 (Mason, 1986). Having established that the activity of MEP against Z- Phe-Arg-NHMec was greatest and stable at ph 5.5, we were then able to determine the Km of MEP for this substrate. A value of ,/M was determined by the method of Wilkinson (1961), a v-alue similar to that found for all mature forms of cathepsin L (Mason, 1986). The kcat of the untreated purified enzyme was determined to be 1.6 s-1, on the basis of total protein. This is approx. 10 times less than that found for mature cathepsin L on the basis of active-site-titrated enzyme. This suggests that either MEP is catalytically less efficient than the mature form of human cathepsin L or that, under these assay conditions (incubation for 30 min at ph 5.5), the purified MEP contains 90% inactive protein. The latter was found to be the case when the enzyme was allowed to react with [3H]Ac-Ep-459 (see below). The cathepsin B and H substrates Z-Arg-Arg-NHMec and Arg-NHMec are poor substrates for cathepsin L (Mason et al., 1985). This was also true for MEP (results not shown). It has therefore been shown that MEP has a similar catalytic activity to cathepsin L against synthetic substrates. The major difference is that mature cathepsin L is active, but unstable, at ph 8.0, whereas MEP is inactive but stable. Inactivation by active-site-directed inhibitors A significant characteristic of mature human cathepsin L is its rate of inactivation by the active-site-directed
4 452 R. W. Mason, S. Gal and M. M. Gottesman y lo-, X Mr Band no i I Lane no Fig. 4. Autolysis of MEP and its reaction with 13HIAc-Ep-459 Lane 1, MEP treated at ph 3.0 for 2 min at 37 C; lane 2, MEP treated at ph 5.0 for 30 min at 37 C; lane 3, MEP + [3H]Ac- Ep-459 (550,uM) treated at ph 3.0 for 2 min at 37 C; lane 4, Mr standards. After pretreatment, enzyme fractions were allowed to react with an excess of [3H]Ac-Ep-459 before running samples in 10 %-(w/v)-polyacrylamide gels in SDS under reducing conditions. Radioactivity (d.p.m.) recovered from each band was as follows: lane 1: band 1, 159; band 2, 267; band 3, 1246; lane 2: band 1, 1519; band 2, 99; band 3, 12; lane 3: band 1, 1025; band 2, 318; band 3, 585. inhibitors Z-Phe-Phe-CHN2 and E-64. These compounds react covalently with the active-site cysteine residue of cysteine proteinases (Leary & Shaw, 1977; Barrett et al., 1982). They are thought to react with active enzyme and are used to titrate cysteine proteinases (Barrett & Kirschke, 1981). The rates of inactivation of MEP by these compounds were determined at ph 5.5 and found to be and Om-1 s-1 respectively. Z-Phe-Ala-CHN2 also rapidly inactivated MEP, with a rate constant of m-'-s-1. Km values for Z-Phe-Arg-NHMec determined from replots of the inverse rate of inactivation against substrate concentration, fell into the range 1-2 #UM, confirming the results obtained by using the Wilkinson (1961) plots (see above). The rates of inactivation were similar to those of species variants of mature cathepsin L (Mason, 1986). Autolysis of MEP and its reaction with a radiolabelied inhibitor It has previously been demonstrated that MEP autolyses at ph 3.0 to yield lower-mr forms (Gal & Gottesman, 1986a). In order to determine whether this could be happening at ph 5.0, MEP was preincubated at this ph for 30 min at 37 C, and then a large excess of [3H]Ac-Ep-459 was added and allowed to react with the enzyme as described in the Materials and methods section. The labelled protein had an Mr identical with that of the purified enzyme (lane 2, Fig. 4). The radioactivity extracted from this band indicated that under these reaction conditions the amount of labelled MEP was only 10 % of the total protein. When MEP was pretreated at ph 3.0 for 2 min at 37 C (lane 1, Fig. 4), most of the MEP was converted into a lower band of M, Total recovery of radioactivity was similar to that seen with enzyme preincubated at ph 5.0. In order to observe any intermediate forms of MEP, inhibitor and ph 3.0 buffer were added to the MEP simultaneously. In this instance, bands of Mr and were seen and, in addition, a band of Mr Again all three bands incorporated radioactivity, indicating that they contained active enzyme. Thus the secreted form of cathepsin L, like the mature form, is labelled by an irreversible active-site-directed inhibitor. The specific activity of intact MEP was determined by using incorporated radioactivity to estimate the concentration of active enzyme and activity against Z-Phe-Arg-NHMec measured as described above. Only 10 % of the MEP reacted with the inhibitor, providing an estimate for kcat of 16 s-5. This is similar to values obtained for mature forms of cathepsin L (Mason, 1986). These results indicate that 90 % of the purified MEP is inactive. This inactive protein does not appear to be latent enzyme, and hence most probably represents either irreversibly oxidized enzyme or denatured protein. This is a common problem when purifying cysteine proteinases by conventional chromatographic techniques. Homogeneous preparations ofhuman cathepsins B, L and H were found to be 50, 60 and 90 % inactive respectively by titration with E-64 (Barrett & Kirschke, 1981; Mason et al., 1985). Activity of MEP against bovine serum albumin MEP was described as an acid-activatable cysteine proteinase on the basis of its ability to cleave bovine serum albumin at ph 3.0 (Gal & Gottesman, 1986a). We 1987
5 Major excreted protein as active cathepsin L precursor 453 Lane no Fig. 5. Activity of MEP and cathepsin L against bovine serum albumin Enzyme and bovine serum albumin were incubated together at 37 C for 1 h as described in the Materials and methods section. Lane 1, MEP and albumin, ph 3.0; lane 2, MEP, E-64 and albumin, ph 3.0; lane 3, albumin alone, ph 3.0; lane 4, MEP and albumin, ph 5.0; lane 5, albumin alone, ph 5.0; lane 6, human cathepsin L and albumin, ph 3.0; lane 7, human cathepsin L, E-64 and albumin, ph 3.0; lane 8, albumin alone, ph 3.0; lane 9, human cathepsin L and albumin, ph 5.0; lane 10, albumin alone, ph 5.0. E ph Fig. 6. Activity of MEP against azocasein Activity of MEP against azocasein was assayed at a range of ph values as described in the Materials and methods section. 0, MEP; A, human cathepsin L (for comparison). have now compared the proteolysis of albumin by MEP directly with that by human cathepsin L (Fig. 5). Activity of MEP at ph 3.0, but not at ph 5.0, against albumin was confirmed. The activity at ph 3.0 was blocked by E- 64, proving that the proteolytic activity was due to a cysteine proteinase. Albumin was similarly resistant to Vol. 248 hydrolysis by mature human cathepsin L at ph 5.0, but was degraded at ph 3.0. Many proteins are resistant to proteolysis in their native state, but are readily hydrolysed when denatured. This most probably explains why this protein is particularly well cleaved by cathepsin L and MEP at low ph and hence it is not a particularly good substrate for measuring the proteolytic activity of MEP at higher ph values. This experiment cannot distinguish between activity of intact or hydrolysed MEP at ph 3.0. Activity against azocasein It has been reported that secreted forms of cathepsin B, a proteinase related to cathepsin L, can hydrolyse synthetic substrates, but not proteins (Mort & Recklies, 1986). We therefore examined the ability of MEP to hydrolyse azocasein, a chromogenic denatured protein substrate. Activity was measured over the ph range 4.5 (the minimum value for solubility of the substrate)-7.5. The optimal ph for hydrolysis of azocasein by MEP was ph 5.0, with no significant activity being measurable above ph 6.0 (Fig. 6). By contrast, human and rabbit cathepsin L are at least 50% as active at ph 6.0 as at ph 5.0 (Mason et al., 1984, 1985). The specific activity of MEP for the hydrolysis of azocasein was compared directly with that of titrated human liver cathepsin L at ph 5.0, and the mouse fibroblast enzyme was found to be twice as active. It has already been demonstrated that MEP does not autolyse significantly at ph 5.0, and we therefore conclude that intact MEP degrades this protein. The data provide further evidence that MEP is an active form of cathepsin L, but with a narrower ph range. DISCUSSION These results show that MEP is catalytically similar to cathepsin L. Taken with the sequence homology and M, differences, it is now clear that MEP (Gottesman, 1978) is a precursor form of mouse cathepsin L (Mason
6 454 R. W. Mason, S. Gal and M. M. Gottesman et al., 1986). The N-terminal extension peptide does not act as a true activation peptide, however, as the precursor enzyme is active. A precursor form of cathepsin D secreted by human breast-cancer cells has also been found to be active (Capony et al., 1987). Thus two of the major lysosomal proteinases have N-terminal extension peptides which do not require cleavage in order for the enzyme to exhibit proteolytic activity. It is likely that these N-terminal extension peptides are regions of the proteins which are sensitive to hydrolysis within the lysosome and hence are not seen on the isolated stable forms of the mature enzymes. The N-terminal extension peptide of mouse cathepsin L (MEP) does seem to have some effects on the enzyme, however. Our results suggest that this peptide stabilizes mouse cathepsin L at neutral ph. Such stability may be required during the normal biosynthesis of cathepsin L as it passes through the Golgi apparatus before transport to lysosomes. The exact ph in the Golgi is uncertain, but is generally considered to be less acidic than in lysosomes, where the mature enzymes are found. Extracellular body fluids are at neutral ph or above, and only the precursor form of cathepsin L, which is the form secreted by transformed mouse cells, would be stable in these fluids. If stabilization were the only function of the N- terminal extension, then the secreted enzyme might be expected to be active at neutral ph, as is the case for the unstable mature form of cathepsin L. The precursor was in fact found to be inactive at ph 7.0, and therefore, although stable at neutral ph, is not functional. This restricts action of the enzyme to sites of low ph within and around cells and tissues (Etherington et al., 1981). Within cells, MEP would be active in acid compartments such as lysosomes, endosomes and some secretory granules, but would be inactive if accidentally released into the cytoplasm. It now appears that cathepsin L may function both intracellularly and extracellularly. The site ofdegradation of proteins hydrolysed by this enzyme will now have to be reconsidered. Chapman & Stone (1984) found that Z- Phe-Ala-CHN2, which has now been shown to be a potent inhibitor of both lysosomal and secreted forms of cathepsin L, could inhibit- elastin degradation by human alveolar macrophages, whereas chloroquine, which only blocks the activity of lysosomal enzymes, was less effective. These results therefore suggest a role for a secreted form of cathepsin L in elastin turnover. Alternatively, the secreted enzyme may have no extracellular proteolytic function and may only be activated when taken up by other cells and packaged into lysosomes. The secreted enzyme can be taken up by other cells with mannose 6-phosphate receptors and be processed intracellularly (S. Gal & M. M. Gottesman, unpublished work). Thus the secreted precursor of cathepsin L could affect proteolysis within cells at some distance from the secreting cell. REFERENCES Barrett, A. J. & Kirschke, H. (1981) Methods Enzymol. 80, Barrett, A. J., Kembhavi, A. A., Brown, M. A., Kirschke, H., Knight, C. G., Tamai, M. & Hanada, K. (1982) Biochem. J. 201, Burnett, D., Crocker, J. & Stockley, R. A. (1983) Am. Rev. Respir. Dis. 128, Bury, A. F. (1981) J. Chromatogr. 213, Capony, F., Morrisset, M., Barrett, A. J., Capony, J. P., Broquet, P., Vignon, F., Chambon, M., Louiaor, P. & Rochefort, H. (1987) J. Cell Biol. 104, Chang, J. C., Lesser, M., Yoo, 0. H. & Orlowski, M. (1986) Am. Rev. Respir. Dis. 134, Chapman, H. A. & Stone, 0. L. (1984) J. Clin. Invest. 74, Denhardt, D. T., Hamilton, R. T., Parfett, C. J. L., Edwards, D. R., St. Pierre, R., Waterhouse, P. & Nilsen-Hamilton, M. (1986) Cancer Res. 46, Etherington, D. J., Pugh, D. & Silver, I. A. (1981) Acta Biol. Med. Germ. 40, Etherington, D. J., Mason, R. W., Taylor, M. A. J. & Wardale, R. J. (1984) Biosci. Rep. 4, Gal, S. & Gottesman, M. M. (1986a) J. Biol. Chem. 261, Gal, S. & Gottesman, M. M. (1986b) Biochem. Biophys. Res. Commun. 139, Gal, S., Willingham, M. C. & Gottesman, M. M. (1985) J. Cell Biol. 100, Gottesman, M. M. (1978) Proc. Natl. Acad. Sci. U.S.A. 75, Graf, M., Leemann, U., Ruch, F. & Strauli, P. (1979) Histochemistry 64, Kirschke, H., Langner, J., Wiederanders, B., Ansorge, S. & Bohley, P. (1977) Eur. J. Biochem. 74, Leary, R. & Shaw, E. N. (1977) Biochem. Biophys. Res. Commun. 79, Mason, R. W. (1986) Biochem. J. 240, Mason, R. W., Taylor, M. A. J. & Etherington, D. J. (1984) Biochem. J. 217, Mason, R. W., Green, G. D. J. & Barrett, A. J. (1985) Biochem. J. 226, Mason, R. W., Walker, J. E. & Northrop, F. D. (1986) Biochem. J. 240, Mort, J. S. & Recklies, A. D. (1986) Biochem. J. 233, Mort, J. S., Leduc, M. S. & Recklies, A. D. (1983) Biochem. J. 230, Parkes, C., Kembhavi, A. A. & Barrett, A. J. (1985) Biochem. J. 230, Poole, A. R. & Mort, J. S. (1981) J. Histochem. Cytochem. 29, Portnoy, D. A., Erickson, A. H., Kockan, J., Ravetch, J. V. & Unkeless, J. C. (1986) J. Biol. Chem. 261, Sloane, B. F. & Honn, K. V. (1984) Cancer Metastasis Rev. 3, Wilkinson, G. N. (1961) Biochem. J. 80, Received 8 May 1987/20 July 1987; accepted 12 August
Collagenase Assay Kit
 Collagenase Assay Kit Catalog # 31 and 32 For Research Use Only - Not Human or Therapeutic Use INTRODUCTION The collagenases are members of the matrix metalloproteinase (MMP) family and degrade collagen
Collagenase Assay Kit Catalog # 31 and 32 For Research Use Only - Not Human or Therapeutic Use INTRODUCTION The collagenases are members of the matrix metalloproteinase (MMP) family and degrade collagen
SUPPLEMENTARY MATERIAL
 SUPPLEMENTARY MATERIAL Purification and biochemical properties of SDS-stable low molecular weight alkaline serine protease from Citrullus Colocynthis Muhammad Bashir Khan, 1,3 Hidayatullah khan, 2 Muhammad
SUPPLEMENTARY MATERIAL Purification and biochemical properties of SDS-stable low molecular weight alkaline serine protease from Citrullus Colocynthis Muhammad Bashir Khan, 1,3 Hidayatullah khan, 2 Muhammad
Collagenase Assay Kit
 Collagenase Assay Kit Catalog # 31 and 32 For Research Use Only - Not Human or Therapeutic Use INTRODUCTION Collagenases are members of the matrix metalloproteinase (MMP) family and degrade collagen types
Collagenase Assay Kit Catalog # 31 and 32 For Research Use Only - Not Human or Therapeutic Use INTRODUCTION Collagenases are members of the matrix metalloproteinase (MMP) family and degrade collagen types
Use of double- stranded DNA mini- circles to characterize the covalent topoisomerase- DNA complex
 SUPPLEMENTARY DATA Use of double- stranded DNA mini- circles to characterize the covalent topoisomerase- DNA complex Armêl Millet 1, François Strauss 1 and Emmanuelle Delagoutte 1 1 Structure et Instabilité
SUPPLEMENTARY DATA Use of double- stranded DNA mini- circles to characterize the covalent topoisomerase- DNA complex Armêl Millet 1, François Strauss 1 and Emmanuelle Delagoutte 1 1 Structure et Instabilité
Nature Methods: doi: /nmeth Supplementary Figure 1
 Supplementary Figure 1 Subtiligase-catalyzed ligations with ubiquitin thioesters and 10-mer biotinylated peptides. (a) General scheme for ligations between ubiquitin thioesters and 10-mer, biotinylated
Supplementary Figure 1 Subtiligase-catalyzed ligations with ubiquitin thioesters and 10-mer biotinylated peptides. (a) General scheme for ligations between ubiquitin thioesters and 10-mer, biotinylated
SYNOPSIS STUDIES ON THE PREPARATION AND CHARACTERISATION OF PROTEIN HYDROLYSATES FROM GROUNDNUT AND SOYBEAN ISOLATES
 1 SYNOPSIS STUDIES ON THE PREPARATION AND CHARACTERISATION OF PROTEIN HYDROLYSATES FROM GROUNDNUT AND SOYBEAN ISOLATES Proteins are important in food processing and food product development, as they are
1 SYNOPSIS STUDIES ON THE PREPARATION AND CHARACTERISATION OF PROTEIN HYDROLYSATES FROM GROUNDNUT AND SOYBEAN ISOLATES Proteins are important in food processing and food product development, as they are
INHIBITION OF TRYPSIN AND PAPAIN BY SODIUM AUROTHIOMALATE MEDIATED BY EXCHANGE REACTIONS
 Br. J. Pharmac. (1982), 75, 333-339 INHIBITION OF TRYPSIN AND PAPAIN BY SODIUM AUROTHIOMALATE MEDIATED BY EXCHANGE REACTIONS M.M. GRIFFIN & F.S. STEVEN Department of Medical Biochemistry, University of
Br. J. Pharmac. (1982), 75, 333-339 INHIBITION OF TRYPSIN AND PAPAIN BY SODIUM AUROTHIOMALATE MEDIATED BY EXCHANGE REACTIONS M.M. GRIFFIN & F.S. STEVEN Department of Medical Biochemistry, University of
1. to understand how proteins find their destination in prokaryotic and eukaryotic cells 2. to know how proteins are bio-recycled
 Protein Targeting Objectives 1. to understand how proteins find their destination in prokaryotic and eukaryotic cells 2. to know how proteins are bio-recycled As a protein is being synthesized, decisions
Protein Targeting Objectives 1. to understand how proteins find their destination in prokaryotic and eukaryotic cells 2. to know how proteins are bio-recycled As a protein is being synthesized, decisions
Problem-solving Test: The Mechanism of Protein Synthesis
 Q 2009 by The International Union of Biochemistry and Molecular Biology BIOCHEMISTRY AND MOLECULAR BIOLOGY EDUCATION Vol. 37, No. 1, pp. 58 62, 2009 Problem-based Learning Problem-solving Test: The Mechanism
Q 2009 by The International Union of Biochemistry and Molecular Biology BIOCHEMISTRY AND MOLECULAR BIOLOGY EDUCATION Vol. 37, No. 1, pp. 58 62, 2009 Problem-based Learning Problem-solving Test: The Mechanism
HPLC '88. Poster Presentation. Isolation of Thymosin B4 from Thymosin Fraction 5 by Reverse Phase HPLC
 Essentials in HPLC '88 Poster Presentation Isolation of Thymosin B4 from Thymosin Fraction 5 by Reverse Phase HPLC M. Badamchian, M.P. Strickler, M.J. Stone, A.L. Goldstein for Waters.bioresearchThe absolute,
Essentials in HPLC '88 Poster Presentation Isolation of Thymosin B4 from Thymosin Fraction 5 by Reverse Phase HPLC M. Badamchian, M.P. Strickler, M.J. Stone, A.L. Goldstein for Waters.bioresearchThe absolute,
Broad Spectrum Protease Inhibitor Cocktail
 Broad Spectrum Protease Inhibitor Cocktail Catalog number: AR1182 Boster s Broad Spectrum Protease Inhibitor Cocktail is a complex of various protease inhibitors, which has been tested for inhibiting proteases
Broad Spectrum Protease Inhibitor Cocktail Catalog number: AR1182 Boster s Broad Spectrum Protease Inhibitor Cocktail is a complex of various protease inhibitors, which has been tested for inhibiting proteases
Saccharomyces cerevisiae*
 THE JOURNAL OF BIOLOGICAL CHEMISTRY 1988 by The American Society for Biochemistry and Molecular Biology, Inc. Vol. 263, No. 29, Issue of October 15, pp. 14948-14955, 1988 Printed in U.S.A. Purification
THE JOURNAL OF BIOLOGICAL CHEMISTRY 1988 by The American Society for Biochemistry and Molecular Biology, Inc. Vol. 263, No. 29, Issue of October 15, pp. 14948-14955, 1988 Printed in U.S.A. Purification
Lysosomal Enzymes as Agents of Turnover of Soluble Cytoplasmic Proteins
 Eur. J. Biochem. 58,9-14 (1975) Lysosomal Enzymes as Agents of Turnover of Soluble Cytoplasmic Proteins Roger T. DEAN Department of Experimental Pathology, University College Hospital Medical School, London
Eur. J. Biochem. 58,9-14 (1975) Lysosomal Enzymes as Agents of Turnover of Soluble Cytoplasmic Proteins Roger T. DEAN Department of Experimental Pathology, University College Hospital Medical School, London
Amino acids. Side chain. -Carbon atom. Carboxyl group. Amino group
 PROTEINS Amino acids Side chain -Carbon atom Amino group Carboxyl group Amino acids Primary structure Amino acid monomers Peptide bond Peptide bond Amino group Carboxyl group Peptide bond N-terminal (
PROTEINS Amino acids Side chain -Carbon atom Amino group Carboxyl group Amino acids Primary structure Amino acid monomers Peptide bond Peptide bond Amino group Carboxyl group Peptide bond N-terminal (
and Its Inhibitor Human-Polymorphonuclear-Leucocyte Neutral Protease Studies with Fluorescein-Labelled Polymeric Collagen Fibrils as a Substrate
 Eur. J. Biochem. 67, 165169 (1976) HumanPolymorphonuclearLeucocyte Neutral Protease and Its Inhibitor Studies with FluoresceinLabelled Polymeric Collagen Fibrils as a Substrate Frank S. STEVEN, David W.
Eur. J. Biochem. 67, 165169 (1976) HumanPolymorphonuclearLeucocyte Neutral Protease and Its Inhibitor Studies with FluoresceinLabelled Polymeric Collagen Fibrils as a Substrate Frank S. STEVEN, David W.
FIRST BIOCHEMISTRY EXAM Tuesday 25/10/ MCQs. Location : 102, 105, 106, 301, 302
 FIRST BIOCHEMISTRY EXAM Tuesday 25/10/2016 10-11 40 MCQs. Location : 102, 105, 106, 301, 302 The Behavior of Proteins: Enzymes, Mechanisms, and Control General theory of enzyme action, by Leonor Michaelis
FIRST BIOCHEMISTRY EXAM Tuesday 25/10/2016 10-11 40 MCQs. Location : 102, 105, 106, 301, 302 The Behavior of Proteins: Enzymes, Mechanisms, and Control General theory of enzyme action, by Leonor Michaelis
Purification of carp (Cyprinus carpio) kidney cathepsin C
 Purification of carp (Cyprinus carpio) kidney cathepsin C (Pemurnian enzim cathepsin C dari ginjal ikan mas Cyprinus carpio) Pangkey H. dan Lantu S. ABSTRACT Pemurnian enzim cathepsin C diperoleh melalui
Purification of carp (Cyprinus carpio) kidney cathepsin C (Pemurnian enzim cathepsin C dari ginjal ikan mas Cyprinus carpio) Pangkey H. dan Lantu S. ABSTRACT Pemurnian enzim cathepsin C diperoleh melalui
Trypsin Mass Spectrometry Grade
 058PR-03 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Trypsin Mass Spectrometry Grade A Chemically Modified, TPCK treated, Affinity Purified
058PR-03 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Trypsin Mass Spectrometry Grade A Chemically Modified, TPCK treated, Affinity Purified
PDF hosted at the Radboud Repository of the Radboud University Nijmegen
 PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/142604
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/142604
Dual nucleotide specificity of bovine glutamate dehydrogenase
 Biochem J. (1980) 191, 299-304 Printed in Great Britain 299 Dual nucleotide specificity of bovine glutamate dehydrogenase The role of negative co-operativity Stephen ALX and J. llis BLL Department ofbiochemistry,
Biochem J. (1980) 191, 299-304 Printed in Great Britain 299 Dual nucleotide specificity of bovine glutamate dehydrogenase The role of negative co-operativity Stephen ALX and J. llis BLL Department ofbiochemistry,
SensoLyte 520 Cathepsin K Assay Kit *Fluorimetric*
 SensoLyte 520 Cathepsin K Assay Kit *Fluorimetric* Catalog # 72171 Kit Size 100 Assays (96-well plate) Optimized Performance: This kit is optimized to detect Cathepsin K activity. Enhanced Value: Ample
SensoLyte 520 Cathepsin K Assay Kit *Fluorimetric* Catalog # 72171 Kit Size 100 Assays (96-well plate) Optimized Performance: This kit is optimized to detect Cathepsin K activity. Enhanced Value: Ample
THE EQUILIBRIUM BETWEEN ACTIVE NATIVE TRYPSIN AND INACTIVE DENATURED TRYPSIN
 Published Online: 20 January, 1934 Supp Info: http://doi.org/10.1085/jgp.17.3.393 Downloaded from jgp.rupress.org on November 8, 2018 THE EQUILIBRIUM BETWEEN ACTIVE NATIVE TRYPSIN AND INACTIVE DENATURED
Published Online: 20 January, 1934 Supp Info: http://doi.org/10.1085/jgp.17.3.393 Downloaded from jgp.rupress.org on November 8, 2018 THE EQUILIBRIUM BETWEEN ACTIVE NATIVE TRYPSIN AND INACTIVE DENATURED
Substrate Specificity and Salt Inhibition of Five Proteinases Isolated from the Pyloric Caeca and Stomach of Sardine
 Agric. Biol. Chem., 46 (6), 1565~1569, 1982 1565 Substrate Specificity and Salt Inhibition of Five Proteinases Isolated from the Pyloric Caeca and Stomach of Sardine Minoru Noda, Thanh Vo Van, Isao Kusakabe
Agric. Biol. Chem., 46 (6), 1565~1569, 1982 1565 Substrate Specificity and Salt Inhibition of Five Proteinases Isolated from the Pyloric Caeca and Stomach of Sardine Minoru Noda, Thanh Vo Van, Isao Kusakabe
Kit for assay of thioredoxin
 FkTRX-02-V2 Kit for assay of thioredoxin The thioredoxin system is the major protein disulfide reductase in cells and comprises thioredoxin, thioredoxin reductase and NADPH (1). Thioredoxin systems are
FkTRX-02-V2 Kit for assay of thioredoxin The thioredoxin system is the major protein disulfide reductase in cells and comprises thioredoxin, thioredoxin reductase and NADPH (1). Thioredoxin systems are
COURSE: Medical Microbiology, MBIM 650/720 - Fall TOPIC: Antigen Processing, MHC Restriction, & Role of Thymus Lecture 12
 COURSE: Medical Microbiology, MBIM 650/720 - Fall 2008 TOPIC: Antigen Processing, MHC Restriction, & Role of Thymus Lecture 12 FACULTY: Dr. Mayer Office: Bldg. #1, Rm B32 Phone: 733-3281 Email: MAYER@MED.SC.EDU
COURSE: Medical Microbiology, MBIM 650/720 - Fall 2008 TOPIC: Antigen Processing, MHC Restriction, & Role of Thymus Lecture 12 FACULTY: Dr. Mayer Office: Bldg. #1, Rm B32 Phone: 733-3281 Email: MAYER@MED.SC.EDU
STUDIES OF THE MECHANISM OF ACTION OF COBAMIDE COENZYMES
 STUDIES OF THE MECHANISM OF ACTION OF COBAMIDE COENZYMES R. H. Abeles and H. A. Lee, Jr. University of Michigan Medical School, Ann Arbor, Mich. Aerobacter aerogenes converts propanediol to propionaldehyde,
STUDIES OF THE MECHANISM OF ACTION OF COBAMIDE COENZYMES R. H. Abeles and H. A. Lee, Jr. University of Michigan Medical School, Ann Arbor, Mich. Aerobacter aerogenes converts propanediol to propionaldehyde,
Proteases in germinating finger millet (Eleusine coracana) seeds
 Biosci., Vol. 5, Number 3, September 1983, pp. 219 224. Printed in India. Proteases in germinating finger millet (Eleusine coracana) seeds Introduction U. VIDYAVATHI, B. SHIVARAJ and T. N. PATTABIRAMAN
Biosci., Vol. 5, Number 3, September 1983, pp. 219 224. Printed in India. Proteases in germinating finger millet (Eleusine coracana) seeds Introduction U. VIDYAVATHI, B. SHIVARAJ and T. N. PATTABIRAMAN
SensoLyte Rh110 Cathepsin K Assay Kit *Fluorimetric* Revision#1.2 Last Updated: May 2017 Catalog # Kit Size
 SensoLyte Rh110 Cathepsin K Assay Kit *Fluorimetric* Revision#1.2 Last Updated: May 2017 Catalog # 72152 Kit Size 100 Assays (96-well plate) Optimized Performance: This kit detects Cathepsin K activity.
SensoLyte Rh110 Cathepsin K Assay Kit *Fluorimetric* Revision#1.2 Last Updated: May 2017 Catalog # 72152 Kit Size 100 Assays (96-well plate) Optimized Performance: This kit detects Cathepsin K activity.
Previous Class. Today. Detection of enzymatic intermediates: Protein tyrosine phosphatase mechanism. Protein Kinase Catalytic Properties
 Previous Class Detection of enzymatic intermediates: Protein tyrosine phosphatase mechanism Today Protein Kinase Catalytic Properties Protein Phosphorylation Phosphorylation: key protein modification
Previous Class Detection of enzymatic intermediates: Protein tyrosine phosphatase mechanism Today Protein Kinase Catalytic Properties Protein Phosphorylation Phosphorylation: key protein modification
Supplementary data Inactivation of Human Angiotensin Converting Enzyme by Copper Peptide Complexes Containing ATCUN Motifs.
 This journal is The Royal Society of Chemistry 25 Supplementary data Inactivation of Human Angiotensin Converting Enzyme by Copper Peptide Complexes Containing ATCUN Motifs. Nikhil H. Gokhale and J. A.
This journal is The Royal Society of Chemistry 25 Supplementary data Inactivation of Human Angiotensin Converting Enzyme by Copper Peptide Complexes Containing ATCUN Motifs. Nikhil H. Gokhale and J. A.
Protease Activity Assay Kit (Red)
 Protease Activity Assay Kit (Red) Catalog Number KA2524 500 assays Version: 11 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information... 4
Protease Activity Assay Kit (Red) Catalog Number KA2524 500 assays Version: 11 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information... 4
Homework Hanson section MCB Course, Fall 2014
 Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Homework Hanson section MCB Course, Fall 2014 (1) Antitrypsin, which inhibits certain proteases, is normally secreted into the bloodstream by liver cells. Antitrypsin is absent from the bloodstream of
Protein Cleavage Due to Pro-oxidative Activity in Some Spices
 Protein Cleavage Due to Pro-oxidative Activity in Some Spices Sittiwat Lertsiri Department of Biotechnology Faculty of Science, Mahidol University Phayathai, Bangkok 10400 Thailand Kanchana Dumri Department
Protein Cleavage Due to Pro-oxidative Activity in Some Spices Sittiwat Lertsiri Department of Biotechnology Faculty of Science, Mahidol University Phayathai, Bangkok 10400 Thailand Kanchana Dumri Department
FEBS 1138 January Paul R. Buckland and Bernard Rees Smith
 Volume 166, number 1 FEBS 1138 January 1984 A structural comparison receptors by of guinea pig thyroid and fat TSH photoaffinity labelling Paul R. Buckland and Bernard Rees Smith Endocrine Immunology Unit,
Volume 166, number 1 FEBS 1138 January 1984 A structural comparison receptors by of guinea pig thyroid and fat TSH photoaffinity labelling Paul R. Buckland and Bernard Rees Smith Endocrine Immunology Unit,
PRODUCT INFORMATION & MANUAL
 PRODUCT INFORMATION & MANUAL 0.4 micron for Overall Exosome Isolation (Cell Media) NBP2-49826 For research use only. Not for diagnostic or therapeutic procedures. www.novusbio.com - P: 303.730.1950 - P:
PRODUCT INFORMATION & MANUAL 0.4 micron for Overall Exosome Isolation (Cell Media) NBP2-49826 For research use only. Not for diagnostic or therapeutic procedures. www.novusbio.com - P: 303.730.1950 - P:
Biochem sheet (5) done by: razan krishan corrected by: Shatha Khtoum DATE :4/10/2016
 Biochem sheet (5) done by: razan krishan corrected by: Shatha Khtoum DATE :4/10/2016 Note about the last lecture: you must know the classification of enzyme Sequentially. * We know that a substrate binds
Biochem sheet (5) done by: razan krishan corrected by: Shatha Khtoum DATE :4/10/2016 Note about the last lecture: you must know the classification of enzyme Sequentially. * We know that a substrate binds
SensoLyte Generic MMP Assay Kit *Colorimetric*
 SensoLyte Generic MMP Assay Kit *Colorimetric* Revision#1.2 Catalog # Kit Size Last updated: May2017 AS-72095 100 Assays (96-well plate) Optimized Performance: This kit is optimized to detect MMP activity
SensoLyte Generic MMP Assay Kit *Colorimetric* Revision#1.2 Catalog # Kit Size Last updated: May2017 AS-72095 100 Assays (96-well plate) Optimized Performance: This kit is optimized to detect MMP activity
Purification and some properties of buffalo spleen cathepsin Β
 J. Biosci., Vol. 14, Number 3, September 1989, pp. 261-268. Printed in India. Purification and some properties of buffalo spleen cathepsin Β SARFRAZ AHMAD, SUDHIR K. AGARWAL and M. YAHIYA KHAN* Department
J. Biosci., Vol. 14, Number 3, September 1989, pp. 261-268. Printed in India. Purification and some properties of buffalo spleen cathepsin Β SARFRAZ AHMAD, SUDHIR K. AGARWAL and M. YAHIYA KHAN* Department
Human Cathepsin D ELISA Kit
 GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Human Cathepsin D ELISA Kit Catalog No. GWB-J4JVV9
GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Human Cathepsin D ELISA Kit Catalog No. GWB-J4JVV9
Summary of Endomembrane-system
 Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
Summary of Endomembrane-system 1. Endomembrane System: The structural and functional relationship organelles including ER,Golgi complex, lysosome, endosomes, secretory vesicles. 2. Membrane-bound structures
SensoLyte 490 HIV-1 Protease Assay Kit *Fluorimetric*
 SensoLyte 490 HIV-1 Protease Assay Kit *Fluorimetric* Catalog # 71127 Unit Size Kit Size 1 kit 500 assays (96-well) or 1250 assays (384-well) This kit is optimized to detect the activity of human immunodeficiency
SensoLyte 490 HIV-1 Protease Assay Kit *Fluorimetric* Catalog # 71127 Unit Size Kit Size 1 kit 500 assays (96-well) or 1250 assays (384-well) This kit is optimized to detect the activity of human immunodeficiency
Chapter 10. Regulatory Strategy
 Chapter 10 Regulatory Strategy Regulation of enzymatic activity: 1. Allosteric Control. Allosteric proteins have a regulatory site(s) and multiple functional sites Activity of proteins is regulated by
Chapter 10 Regulatory Strategy Regulation of enzymatic activity: 1. Allosteric Control. Allosteric proteins have a regulatory site(s) and multiple functional sites Activity of proteins is regulated by
protein C (blood clotting/factor V/clotting inhibitors)
 Proc. Nati Acad. Sci. USA Vol. 80, pp. 1584-1588, March 1983 Biochemistry Human coagulation factor Va is a cofactor for the activation of protein C (blood clotting/factor V/clotting inhibitors) HATEM H.
Proc. Nati Acad. Sci. USA Vol. 80, pp. 1584-1588, March 1983 Biochemistry Human coagulation factor Va is a cofactor for the activation of protein C (blood clotting/factor V/clotting inhibitors) HATEM H.
Amylase: a sample enzyme
 Amylase: a sample enzyme Objectives: After completion of this laboratory exercise you will be able to: 1. Explain the importance of enzymes in biology. 2. Explain the basic properties of an enzyme as a
Amylase: a sample enzyme Objectives: After completion of this laboratory exercise you will be able to: 1. Explain the importance of enzymes in biology. 2. Explain the basic properties of an enzyme as a
number Done by Corrected by Doctor Dr. Diala
 number 30 Done by Dergam Al-Tarawneh Corrected by Zaid Emad Doctor Dr. Diala 1 After we ve finished talking about lipids metabolism pathways, today we will start talking about another pathway that takes
number 30 Done by Dergam Al-Tarawneh Corrected by Zaid Emad Doctor Dr. Diala 1 After we ve finished talking about lipids metabolism pathways, today we will start talking about another pathway that takes
Edman Sample Preparation Part 2. Bill Henzel Genentech Inc.
 Edman Sample Preparation Part 2 Bill Henzel Genentech Inc. Determining a Protein is Blocked Estimate the amount of protein applied to the protein sequencer. Compare the staining intensity of the band of
Edman Sample Preparation Part 2 Bill Henzel Genentech Inc. Determining a Protein is Blocked Estimate the amount of protein applied to the protein sequencer. Compare the staining intensity of the band of
v o = V max [S] rate = kt[s] e V max = k cat E t ΔG = -RT lnk eq K m + [S]
![v o = V max [S] rate = kt[s] e V max = k cat E t ΔG = -RT lnk eq K m + [S] v o = V max [S] rate = kt[s] e V max = k cat E t ΔG = -RT lnk eq K m + [S]](/thumbs/81/84404059.jpg) Exam 3 Spring 2017 Dr. Stone 8:00 Name There are 100 possible points on this exam. -ΔG / RT v o = V max [S] rate = kt[s] e V max = k cat E t ΔG = -RT lnk eq K m + [S] h rate forward = k forward [reactants]
Exam 3 Spring 2017 Dr. Stone 8:00 Name There are 100 possible points on this exam. -ΔG / RT v o = V max [S] rate = kt[s] e V max = k cat E t ΔG = -RT lnk eq K m + [S] h rate forward = k forward [reactants]
Four melanocyte-stimulating hormones have the following amino acid sequences:
 Assignment 14: Melanocyte-stimulating hormone belongs to a group called the melanocortins. This group includes ACTH, alpha-msh, beta-msh and gamma-msh; these peptides are all cleavage products of a large
Assignment 14: Melanocyte-stimulating hormone belongs to a group called the melanocortins. This group includes ACTH, alpha-msh, beta-msh and gamma-msh; these peptides are all cleavage products of a large
REGULATION OF ENZYME ACTIVITY. Medical Biochemistry, Lecture 25
 REGULATION OF ENZYME ACTIVITY Medical Biochemistry, Lecture 25 Lecture 25, Outline General properties of enzyme regulation Regulation of enzyme concentrations Allosteric enzymes and feedback inhibition
REGULATION OF ENZYME ACTIVITY Medical Biochemistry, Lecture 25 Lecture 25, Outline General properties of enzyme regulation Regulation of enzyme concentrations Allosteric enzymes and feedback inhibition
TECHNICAL BULLETIN. R 2 GlcNAcβ1 4GlcNAcβ1 Asn
 GlycoProfile II Enzymatic In-Solution N-Deglycosylation Kit Product Code PP0201 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description Glycosylation is one of the most common posttranslational
GlycoProfile II Enzymatic In-Solution N-Deglycosylation Kit Product Code PP0201 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description Glycosylation is one of the most common posttranslational
Page 1 of 5 Biochemistry I Fall 2017 Practice for Exam2 Dr. Stone Name
 Page 1 of 5 Biochemistry I Fall 2017 Practice for Exam2 Dr. Stone ame o answers will be provided. ere are some constants and equations that may be useful: K a = [+][A-]/[A] p = pka + log [A-]/[A] K a for
Page 1 of 5 Biochemistry I Fall 2017 Practice for Exam2 Dr. Stone ame o answers will be provided. ere are some constants and equations that may be useful: K a = [+][A-]/[A] p = pka + log [A-]/[A] K a for
DEGRADATION OF CONNECTIVE TISSUE MATRICES BY MACROPHAGES II. Influence of Matrix Composition on Proteolysis of Glycoproteins,
 DEGRADATION OF CONNECTIVE TISSUE MATRICES BY MACROPHAGES II. Influence of Matrix Composition on Proteolysis of Glycoproteins, Elastin, and Collagen by Macrophages in Culture* BY PETER A. JONES AND ZENA
DEGRADATION OF CONNECTIVE TISSUE MATRICES BY MACROPHAGES II. Influence of Matrix Composition on Proteolysis of Glycoproteins, Elastin, and Collagen by Macrophages in Culture* BY PETER A. JONES AND ZENA
TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
 Journal of Supramolecular Structure 4:441 (401)-447 (407) (1976) TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
Journal of Supramolecular Structure 4:441 (401)-447 (407) (1976) TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
PROTAZYME AK TABLETS
 www.megazyme.com ASSAY OF endo-protease using PROTAZYME AK TABLETS T-PRAK 05/16 Megazyme International Ireland 2016 SUBSTRATE: The substrate employed is Azurine-crosslinked casein (AZCL-casein). This substrate
www.megazyme.com ASSAY OF endo-protease using PROTAZYME AK TABLETS T-PRAK 05/16 Megazyme International Ireland 2016 SUBSTRATE: The substrate employed is Azurine-crosslinked casein (AZCL-casein). This substrate
Fatty-Acid Synthesis in Lactating-Goat Mammary Gland
 Eur. J. Biochem. 95, 503507 (1979) FattyAcid Synthesis in LactatingGoat Mammary Gland 2. MediumChain Fatty Acid Synthesis Inger GRUNNET and Jens KNUDSEN Institute of Biochemistry, Odense University (Received
Eur. J. Biochem. 95, 503507 (1979) FattyAcid Synthesis in LactatingGoat Mammary Gland 2. MediumChain Fatty Acid Synthesis Inger GRUNNET and Jens KNUDSEN Institute of Biochemistry, Odense University (Received
Cell Quality Control. Peter Takizawa Department of Cell Biology
 Cell Quality Control Peter Takizawa Department of Cell Biology Cellular quality control reduces production of defective proteins. Cells have many quality control systems to ensure that cell does not build
Cell Quality Control Peter Takizawa Department of Cell Biology Cellular quality control reduces production of defective proteins. Cells have many quality control systems to ensure that cell does not build
ab SREBP-2 Translocation Assay Kit (Cell-Based)
 ab133114 SREBP-2 Translocation Assay Kit (Cell-Based) Instructions for Use For analysis of translocation of SREBP-2 into nuclei. This product is for research use only and is not intended for diagnostic
ab133114 SREBP-2 Translocation Assay Kit (Cell-Based) Instructions for Use For analysis of translocation of SREBP-2 into nuclei. This product is for research use only and is not intended for diagnostic
Expression constructs
 Gene expressed in bebe3 ZmBEa Expression constructs 35S ZmBEa Pnos:Hygromycin r 35S Pnos:Hygromycin r 35S ctp YFP Pnos:Hygromycin r B -1 Chl YFP- Merge Supplemental Figure S1: Constructs Used for the Expression
Gene expressed in bebe3 ZmBEa Expression constructs 35S ZmBEa Pnos:Hygromycin r 35S Pnos:Hygromycin r 35S ctp YFP Pnos:Hygromycin r B -1 Chl YFP- Merge Supplemental Figure S1: Constructs Used for the Expression
Previous Class. Today. Term test I discussions. Detection of enzymatic intermediates: chymotrypsin mechanism
 Term test I discussions Previous Class Today Detection of enzymatic intermediates: chymotrypsin mechanism Mechanistic Understanding of Enzymemediated Reactions Ultimate goals: Identification of the intermediates,
Term test I discussions Previous Class Today Detection of enzymatic intermediates: chymotrypsin mechanism Mechanistic Understanding of Enzymemediated Reactions Ultimate goals: Identification of the intermediates,
Protein Trafficking in the Secretory and Endocytic Pathways
 Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Inhibition of reverse transcriptases by seminalplasmin
 Biochem. J. (1983) 29, 183-188 183 Printed in Great Britain Inhibition of reverse transcriptases by seminalplasmin E. Shyam Prasad REDDY,* M. Ramachandra DAS,* E. Premkumar REDDYt and Pushpa M. BHARGAVA*
Biochem. J. (1983) 29, 183-188 183 Printed in Great Britain Inhibition of reverse transcriptases by seminalplasmin E. Shyam Prasad REDDY,* M. Ramachandra DAS,* E. Premkumar REDDYt and Pushpa M. BHARGAVA*
N-Glycosidase F Deglycosylation Kit
 For life science research only. Not for use in diagnostic procedures. FOR IN VITRO USE ONLY. N-Glycosidase F Deglycosylation Kit Kit for the deglycosylation of asparagine-linked glycan chains on glycoproteins.
For life science research only. Not for use in diagnostic procedures. FOR IN VITRO USE ONLY. N-Glycosidase F Deglycosylation Kit Kit for the deglycosylation of asparagine-linked glycan chains on glycoproteins.
Biomolecules: amino acids
 Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
BIL 256 Cell and Molecular Biology Lab Spring, Tissue-Specific Isoenzymes
 BIL 256 Cell and Molecular Biology Lab Spring, 2007 Background Information Tissue-Specific Isoenzymes A. BIOCHEMISTRY The basic pattern of glucose oxidation is outlined in Figure 3-1. Glucose is split
BIL 256 Cell and Molecular Biology Lab Spring, 2007 Background Information Tissue-Specific Isoenzymes A. BIOCHEMISTRY The basic pattern of glucose oxidation is outlined in Figure 3-1. Glucose is split
General Principles of Endocrine Physiology
 General Principles of Endocrine Physiology By Dr. Isabel S.S. Hwang Department of Physiology Faculty of Medicine University of Hong Kong The major human endocrine glands Endocrine glands and hormones
General Principles of Endocrine Physiology By Dr. Isabel S.S. Hwang Department of Physiology Faculty of Medicine University of Hong Kong The major human endocrine glands Endocrine glands and hormones
ASSAY OF USING BETA-GLUCAZYME TABLETS
 ASSAY OF endo-β-glucanases USING BETA-GLUCAZYME TABLETS T-BGZ 12/12 Note: Changed assay format for malt β-glucanase Megazyme International Ireland 2012 SUBSTRATE: The substrate employed is Azurine-crosslinked
ASSAY OF endo-β-glucanases USING BETA-GLUCAZYME TABLETS T-BGZ 12/12 Note: Changed assay format for malt β-glucanase Megazyme International Ireland 2012 SUBSTRATE: The substrate employed is Azurine-crosslinked
Enzymatic Assay of POLYGALACTURONASE (EC )
 PRINCIPLE: Polygalacturonic Acid + H 2 O PG > Reducing Sugars Abbreviations: PG = Polygalacturonase CONDITIONS: T = 30 C, ph 5.0, A 540nm, Light path = 1 cm METHOD: Colorimetric REAGENTS: A. 50 mm Sodium
PRINCIPLE: Polygalacturonic Acid + H 2 O PG > Reducing Sugars Abbreviations: PG = Polygalacturonase CONDITIONS: T = 30 C, ph 5.0, A 540nm, Light path = 1 cm METHOD: Colorimetric REAGENTS: A. 50 mm Sodium
1. For the following reaction, at equilibrium [S] = 5 mm, [P] = 0.5 mm, and k f = 10 s -1. k f
![1. For the following reaction, at equilibrium [S] = 5 mm, [P] = 0.5 mm, and k f = 10 s -1. k f 1. For the following reaction, at equilibrium [S] = 5 mm, [P] = 0.5 mm, and k f = 10 s -1. k f](/thumbs/77/75734355.jpg) 1. For the following reaction, at equilibrium [S] = 5 mm, [P] = 0.5 mm, and k f = 10 s -1. S k f k r P a) Calculate K eq (the equilibrium constant) and k r. b) A catalyst increases k f by a factor of 10
1. For the following reaction, at equilibrium [S] = 5 mm, [P] = 0.5 mm, and k f = 10 s -1. S k f k r P a) Calculate K eq (the equilibrium constant) and k r. b) A catalyst increases k f by a factor of 10
PHRM 836 September 1, 2015
 PRM 836 September 1, 2015 Protein structure- function relationship: Catalysis example of serine proteases Devlin, section 9.3 Physiological processes requiring serine proteases Control of enzymatic activity
PRM 836 September 1, 2015 Protein structure- function relationship: Catalysis example of serine proteases Devlin, section 9.3 Physiological processes requiring serine proteases Control of enzymatic activity
General information. Cell mediated immunity. 455 LSA, Tuesday 11 to noon. Anytime after class.
 General information Cell mediated immunity 455 LSA, Tuesday 11 to noon Anytime after class T-cell precursors Thymus Naive T-cells (CD8 or CD4) email: lcoscoy@berkeley.edu edu Use MCB150 as subject line
General information Cell mediated immunity 455 LSA, Tuesday 11 to noon Anytime after class T-cell precursors Thymus Naive T-cells (CD8 or CD4) email: lcoscoy@berkeley.edu edu Use MCB150 as subject line
OxisResearch A Division of OXIS Health Products, Inc.
 OxisResearch A Division of OXIS Health Products, Inc. BIOXYTECH pl GPx Enzyme Immunoassay Assay for Human Plasma Glutathione Peroxidase For Research Use Only. Not For Use In Diagnostic Procedures. Catalog
OxisResearch A Division of OXIS Health Products, Inc. BIOXYTECH pl GPx Enzyme Immunoassay Assay for Human Plasma Glutathione Peroxidase For Research Use Only. Not For Use In Diagnostic Procedures. Catalog
7.06 Cell Biology EXAM #3 April 24, 2003
 7.06 Spring 2003 Exam 3 Name 1 of 8 7.06 Cell Biology EXAM #3 April 24, 2003 This is an open book exam, and you are allowed access to books and notes. Please write your answers to the questions in the
7.06 Spring 2003 Exam 3 Name 1 of 8 7.06 Cell Biology EXAM #3 April 24, 2003 This is an open book exam, and you are allowed access to books and notes. Please write your answers to the questions in the
CRYSTALLINE PEPSIN BY JOHN H. NORTHROP. (From the Laboratories of The Rockefeller Institute for Medical Research, Princeton, iv. J.
 CRYSTALLINE PEPSIN III. PREPARATION OF ACTIVE CRYSTALLINE PEPSIN FROM INACTIVE DENATURED PEPSIN BY JOHN H. NORTHROP (From the Laboratories of The Rockefeller Institute for Medical Research, Princeton,
CRYSTALLINE PEPSIN III. PREPARATION OF ACTIVE CRYSTALLINE PEPSIN FROM INACTIVE DENATURED PEPSIN BY JOHN H. NORTHROP (From the Laboratories of The Rockefeller Institute for Medical Research, Princeton,
Regulation of Lamp2a Levels in the Lysosomal Membrane
 Traffic 2000 1: 570 583 Munksgaard International Publishers Regulation of Lamp2a Levels in the Lysosomal Membrane A.M. Cuervo and J.F. Dice* Department of Physiology, Tufts University School of Medicine,
Traffic 2000 1: 570 583 Munksgaard International Publishers Regulation of Lamp2a Levels in the Lysosomal Membrane A.M. Cuervo and J.F. Dice* Department of Physiology, Tufts University School of Medicine,
UNIVERSITY OF GUELPH CHEM 4540 ENZYMOLOGY Winter 2005 Quiz #2: March 24, 2005, 11:30 12:50 Instructor: Prof R. Merrill ANSWERS
 UNIVERSITY F GUELPH CHEM 4540 ENZYMLGY Winter 2005 Quiz #2: March 24, 2005, 11:30 12:50 Instructor: Prof R. Merrill ANSWERS Instructions: Time allowed = 80 minutes. Total marks = 30. This quiz represents
UNIVERSITY F GUELPH CHEM 4540 ENZYMLGY Winter 2005 Quiz #2: March 24, 2005, 11:30 12:50 Instructor: Prof R. Merrill ANSWERS Instructions: Time allowed = 80 minutes. Total marks = 30. This quiz represents
Chapter 2 Transport Systems
 Chapter 2 Transport Systems The plasma membrane is a selectively permeable barrier between the cell and the extracellular environment. It permeability properties ensure that essential molecules such as
Chapter 2 Transport Systems The plasma membrane is a selectively permeable barrier between the cell and the extracellular environment. It permeability properties ensure that essential molecules such as
Conversion of green note aldehydes into alcohols by yeast alcohol dehydrogenase
 Conversion of green note aldehydes into alcohols by yeast alcohol dehydrogenase M.-L. Fauconnier 1, A. Mpambara 1, J. Delcarte 1, P. Jacques 2, P. Thonart 2 & M. Marlier 1 1 Unité de Chimie Générale et
Conversion of green note aldehydes into alcohols by yeast alcohol dehydrogenase M.-L. Fauconnier 1, A. Mpambara 1, J. Delcarte 1, P. Jacques 2, P. Thonart 2 & M. Marlier 1 1 Unité de Chimie Générale et
Synthesis and Degradation of Liver Acetyl Coenzyme A Carboxylase
 Proc. Nat. Acad. Sci. USA Vol. 68, No. 9, pp. 2288-2292, September 1971 Synthesis and Degradation of Liver Acetyl Coenzyme A Carboxylase in Genetically Obese Mice (increased hepatic lipogenesis/immunochemical
Proc. Nat. Acad. Sci. USA Vol. 68, No. 9, pp. 2288-2292, September 1971 Synthesis and Degradation of Liver Acetyl Coenzyme A Carboxylase in Genetically Obese Mice (increased hepatic lipogenesis/immunochemical
Chemical Mechanism of Enzymes
 Chemical Mechanism of Enzymes Enzyme Engineering 5.2 Definition of the mechanism 1. The sequence from substrate(s) to product(s) : Reaction steps 2. The rates at which the complex are interconverted 3.
Chemical Mechanism of Enzymes Enzyme Engineering 5.2 Definition of the mechanism 1. The sequence from substrate(s) to product(s) : Reaction steps 2. The rates at which the complex are interconverted 3.
20S Proteasome Activity Assay Kit
 20S Proteasome Activity Assay Kit For 100 Assays Cat. No. APT280 FOR RESEARCH USE ONLY NOT FOR USE IN DIAGNOSTIC PROCEDURES USA & Canada Phone: +1(800) 437-7500 Fax: +1 (951) 676-9209 Europe +44 (0) 23
20S Proteasome Activity Assay Kit For 100 Assays Cat. No. APT280 FOR RESEARCH USE ONLY NOT FOR USE IN DIAGNOSTIC PROCEDURES USA & Canada Phone: +1(800) 437-7500 Fax: +1 (951) 676-9209 Europe +44 (0) 23
Assay Kit for Measurement of Proteoglycan. (Sulfated Glycosaminoglycan Quantification Kit)
 Assay Kit for Measurement of Proteoglycan. (Sulfated Glycosaminoglycan Quantification Kit) Cat. No. 280560-N INTRODUCTION Glycosaminoglycans (GAGs) are a major component of the extracellular matrix (ECM)
Assay Kit for Measurement of Proteoglycan. (Sulfated Glycosaminoglycan Quantification Kit) Cat. No. 280560-N INTRODUCTION Glycosaminoglycans (GAGs) are a major component of the extracellular matrix (ECM)
SUPPLEMENTARY INFORMATION
 Figure S1. Cleavage of uniquitin AAA -CPP TAT in vitro and in cells. a, b. In vitro two-dimensional 1 H- 15 N correlation spectrum of ubiquitin AAA -CPP TAT before (a) and after (b) Yeast Ubiquitin Hydrolase
Figure S1. Cleavage of uniquitin AAA -CPP TAT in vitro and in cells. a, b. In vitro two-dimensional 1 H- 15 N correlation spectrum of ubiquitin AAA -CPP TAT before (a) and after (b) Yeast Ubiquitin Hydrolase
Role of the Carbohydrate Moiety in Phospholipase B from Torulaspora delbrueckii
 Agric. Bioi Chern., 54 (3), 599-603, 1990 599 Role of the Carbohydrate Moiety in Phospholipase B from Torulaspora delbrueckii Masafumi Maruyama, Hideki Kadowaki, Yasuo Watanabe and Youichi Tamai Department
Agric. Bioi Chern., 54 (3), 599-603, 1990 599 Role of the Carbohydrate Moiety in Phospholipase B from Torulaspora delbrueckii Masafumi Maruyama, Hideki Kadowaki, Yasuo Watanabe and Youichi Tamai Department
Improve Protein Analysis with the New, Mass Spectrometry- Compatible ProteasMAX Surfactant
 Improve Protein Analysis with the New, Mass Spectrometry- Compatible Surfactant ABSTRACT Incomplete solubilization and digestion and poor peptide recovery are frequent limitations in protein sample preparation
Improve Protein Analysis with the New, Mass Spectrometry- Compatible Surfactant ABSTRACT Incomplete solubilization and digestion and poor peptide recovery are frequent limitations in protein sample preparation
Role of metabolism in Drug-Induced Liver Injury (DILI) Drug Metab Rev. 2007;39(1):
 Role of metabolism in Drug-Induced Liver Injury (DILI) Drug Metab Rev. 2007;39(1):159-234 Drug Metab Rev. 2007;39(1):159-234 Drug Metab Rev. 2007;39(1):159-234 A schematic representation of the most relevant
Role of metabolism in Drug-Induced Liver Injury (DILI) Drug Metab Rev. 2007;39(1):159-234 Drug Metab Rev. 2007;39(1):159-234 Drug Metab Rev. 2007;39(1):159-234 A schematic representation of the most relevant
Ferritin-Conjugated Antibodies Used for Labeling of Organelles Involved
 Proc. Nat. Acad. Sci. USA Vol. 71, No. 5, pp. 2033-2037, May 1974 Ferritin-Conjugated Antibodies Used for Labeling of Organelles Involved in the Cellular Synthesis and Transport of Procollagen (ferritin-antibody
Proc. Nat. Acad. Sci. USA Vol. 71, No. 5, pp. 2033-2037, May 1974 Ferritin-Conjugated Antibodies Used for Labeling of Organelles Involved in the Cellular Synthesis and Transport of Procollagen (ferritin-antibody
Ubiquitin-aldehyde: A general inhibitor of ubiquitinrecycling
 Proc. Nati. Acad. Sci. USA Vol. 84, pp. 1829-1833, April 1987 Biochemistry Ubiquitin-aldehyde: A general inhibitor of ubiquitinrecycling processes (protein breakdown/isopeptidase/c-terminal hydrolase)
Proc. Nati. Acad. Sci. USA Vol. 84, pp. 1829-1833, April 1987 Biochemistry Ubiquitin-aldehyde: A general inhibitor of ubiquitinrecycling processes (protein breakdown/isopeptidase/c-terminal hydrolase)
PYRROLE AS A CATALYST FOR CERTAIN BIOLOGICAL OXIDATIONS
 PYRROLE AS A CATALYST FOR CERTAIN BIOLOGICAL OXIDATIONS BY FREDERICK BERNHEIM AND MARY L. C. BERNHEIM* (From the Departments of Physiology and Biochemistry, Duke University School of Medicine, Durham)
PYRROLE AS A CATALYST FOR CERTAIN BIOLOGICAL OXIDATIONS BY FREDERICK BERNHEIM AND MARY L. C. BERNHEIM* (From the Departments of Physiology and Biochemistry, Duke University School of Medicine, Durham)
Self-association of α-chymotrypsin: Effect of amino acids
 J. Biosci., Vol. 13, Number 3, September 1988, pp. 215 222. Printed in India. Self-association of α-chymotrypsin: Effect of amino acids T. RAMAKRISHNA and M. W. PANDIT* Centre for Cellular and Molecular
J. Biosci., Vol. 13, Number 3, September 1988, pp. 215 222. Printed in India. Self-association of α-chymotrypsin: Effect of amino acids T. RAMAKRISHNA and M. W. PANDIT* Centre for Cellular and Molecular
Mouse Cathepsin B ELISA Kit
 GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Mouse Cathepsin B ELISA Kit Catalog No. GWB-ZZD154
GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Mouse Cathepsin B ELISA Kit Catalog No. GWB-ZZD154
SensoLyte 520 HIV-1 Protease Assay Kit *Fluorimetric*
 SensoLyte 520 HIV-1 Protease Assay Kit *Fluorimetric* Catalog # 71147 Kit Size 100 assays (96-well) or 500 assays (384-well) Convenient Format: Complete kit including all the assay components. Optimized
SensoLyte 520 HIV-1 Protease Assay Kit *Fluorimetric* Catalog # 71147 Kit Size 100 assays (96-well) or 500 assays (384-well) Convenient Format: Complete kit including all the assay components. Optimized
Structural Characterization of Prion-like Conformational Changes of the Neuronal Isoform of Aplysia CPEB
 Structural Characterization of Prion-like Conformational Changes of the Neuronal Isoform of Aplysia CPEB Bindu L. Raveendra, 1,5 Ansgar B. Siemer, 2,6 Sathyanarayanan V. Puthanveettil, 1,3,7 Wayne A. Hendrickson,
Structural Characterization of Prion-like Conformational Changes of the Neuronal Isoform of Aplysia CPEB Bindu L. Raveendra, 1,5 Ansgar B. Siemer, 2,6 Sathyanarayanan V. Puthanveettil, 1,3,7 Wayne A. Hendrickson,
MCB130 Midterm. GSI s Name:
 1. Peroxisomes are small, membrane-enclosed organelles that function in the degradation of fatty acids and in the degradation of H 2 O 2. Peroxisomes are not part of the secretory pathway and peroxisomal
1. Peroxisomes are small, membrane-enclosed organelles that function in the degradation of fatty acids and in the degradation of H 2 O 2. Peroxisomes are not part of the secretory pathway and peroxisomal
Part IV Antithrombotics, Anticoagulants and Fibrinolytics
 Part IV Antithrombotics, Anticoagulants and Fibrinolytics "The meaning of good and bad, of better and worse, is simply helping or hurting" Emerson Chapter 16: Blood Coagulation and Fibrinolytic System
Part IV Antithrombotics, Anticoagulants and Fibrinolytics "The meaning of good and bad, of better and worse, is simply helping or hurting" Emerson Chapter 16: Blood Coagulation and Fibrinolytic System
10-6 M) or freeze-dried normal rat plasma (35 jug mg-' wet wt. tissue) did not show
 J. Physiol. (1981), 315, pp. 413-419 413 With 1 text-figure Printed in Great Britain ROLE OF A RENAL ARGINYLESTEROPEPTIDASE IN THE PRODUCTION OF A RENOTROPHIC FACTOR IN UNILATERALLY NEPHRECTOMIZED RATS
J. Physiol. (1981), 315, pp. 413-419 413 With 1 text-figure Printed in Great Britain ROLE OF A RENAL ARGINYLESTEROPEPTIDASE IN THE PRODUCTION OF A RENOTROPHIC FACTOR IN UNILATERALLY NEPHRECTOMIZED RATS
[GANN, 59, ; October, 1968] CHANGES IN ALDOLASE ISOZYME PATTERNS OF HUMAN CANCEROUS TISSUES
![[GANN, 59, ; October, 1968] CHANGES IN ALDOLASE ISOZYME PATTERNS OF HUMAN CANCEROUS TISSUES [GANN, 59, ; October, 1968] CHANGES IN ALDOLASE ISOZYME PATTERNS OF HUMAN CANCEROUS TISSUES](/thumbs/87/96205960.jpg) [GANN, 59, 415-419; October, 1968] UDC 616-006-092.18 CHANGES IN ALDOLASE ISOZYME PATTERNS OF HUMAN CANCEROUS TISSUES Kiyoshi TSUNEMATSU, Shin-ichi YOKOTA, and Tadao SHIRAISHI (Third Department of Internal
[GANN, 59, 415-419; October, 1968] UDC 616-006-092.18 CHANGES IN ALDOLASE ISOZYME PATTERNS OF HUMAN CANCEROUS TISSUES Kiyoshi TSUNEMATSU, Shin-ichi YOKOTA, and Tadao SHIRAISHI (Third Department of Internal
MAXIMIZATION OF PRODUCTION OF PROTEIN HYDROLYSATES BY USING IMMOBILIZED PAPAIN
 Int. J. Chem. Sci.: 7(4), 2009, 2624-2632 MAXIMIZATION OF PRODUCTION OF PROTEIN HYDROLYSATES BY USING IMMOBILIZED PAPAIN T. SATHISH a and N. Y. S. MURTHY * Department of Biotechnology, Malla Reddy Engineering
Int. J. Chem. Sci.: 7(4), 2009, 2624-2632 MAXIMIZATION OF PRODUCTION OF PROTEIN HYDROLYSATES BY USING IMMOBILIZED PAPAIN T. SATHISH a and N. Y. S. MURTHY * Department of Biotechnology, Malla Reddy Engineering
