Applications of Freezing Point Osmometry
|
|
|
- Rudolf Wilcox
- 6 years ago
- Views:
Transcription
1 Applications of Freezing Point Osmometry
2 Table of Contents Chapter 1 Introduction and Basic Principles 1 Chapter 2 Biological Applications Range of, and reason for, abnormal serum values Osmolality and sodium concentration Urine osmolality Unknown blood solutes Clearance studies Monitoring electrolyte balance Total body water Dehydration ADH Fluid therapy Pediatrics Blood alcohol 10 Chapter 3 Other Applications Various body fluids With an artificial kidney or dialyzer Molecular weight and chemical purity Fetal maturity Chromatographic eluates Quality control Fixing solutions General concentration tests 12
3 1 Introduction and Basic Principles The measurement of the freezing point of a solution is related to the osmotic concentration of that solution. Osmotic concentration can be thought of as the concentration of particles of solute per unit amount of solvent. Sometimes this is called an apparent concentration since the manner in which the solute reacts with the solvent affects the number of particles that are formed. The manner in which the solute and solvent interact is called activity. This refers not only to the degree of dissociation of the solute (if the solute is an ionic salt), but also to a number of other factors including the degree to which the water molecules are free to enter the solution. If you know: and you measure: 1 a. The normal or original freezing The freezing point of the point of a solution solution b. That contamination has taken place 2 a. The weight of solute A The freezing point of A b. The freezing point of plus B solvent B 3 The specific gravity of a The freezing point of the solution solution 4 The freezing point of a The freezing point of solution solution at time A at time B 5 a. The type of dissolved molecule The freezing point of the b. The weight of solute solution c. The weight of solvent 6 a. The molecular weight of the The freezing point of the dissolved solute molecule solution b. The weight of solute c. The weight of solvent 1
4 Applications of Freezing Point Osmometry Some physical chemists prefer to interpret the freezing point of a solution as the concentration of water in that solution. Others relate freezing point to the water potential of the solution. For the purposes of this discussion, it is most convenient to describe freezing point as the concentration of particles in solution. Freezing point won t tell you how big these particles are, or what shape they have, or if they are charged. It only tells you how many. At relatively low concentrations, freezing point is linear with the number of dissolved particles. Twice as many particles will depress the freezing point twice as much, three times as many, three times as much, and so forth. There are six ways in which the knowledge of freezing point can be applied. These are summarized in the table below. you can determine: The percentage dilution or change in concentration. The volume of the container occupied by B The approximate size and number of dissolved molecules. The change in number of molecules due to diastasis or other factors. If freezing point is measured over a number of known time intervals, then the rate of reaction can be measured. The molecular weight of the solute. The osmotic coefficient of the solute in the solvent. the application is: Concentration of body fluids. Quality control of standards and solutions. Measuring volumes of containers, tanks, kettles, pipets, human body, etc. Measuring the dialyzability of solute. Measurement of enzyme activity. Molecular weight determinations. Determination of water binding, activity, etc. 2
5 2 Biological Applications The fluid compartments within the human body as well as most other animals are generally iso-osmotic. The freezing point of the serum is very nearly the same as that of the cerebro-spinal fluid. Intra-cellular and extra-cellular fluid are very close in concentration. Milk serum is in osmotic equilibrium with blood serum. And so forth. Urine, on the other hand, varies widely in concentration. One of the major functions of the kidney is to remove solute or water (depending on whether there is too much of one or the other) from the blood in order to control the concentration of the blood. Sweat glands and the thirst mechanism perform a similar function. Bar graph Gamblegram in meq/l serum (plus mg% of urea (BUN) & glucose) 100 mg% glucose 13 mg% BUN 141 meq/l Na meq/l K + 7 meq/l Ca++ and Mg meq/l Cl - 28 meq/l - HCO3 3 meq/l --- PO4 and -- SO4 3 meq/l acids 15.5 meq/l protein Figure 1: Analysis of solute make-up in normal human serum 3
6 Applications of Freezing Point Osmometry 4 glucose 4 urea 103 Cl Na + 28 HCO3-4.2 K Ca++ and Mg++ Total = mosm/kg H 2 O Figure 2: The same components as in Figure 1 expressed in mosm/kg H 2 O PO4 and -- SO4 1.0 acids 1.9 protein Although the various body compartments are relatively iso-osmotic - contain the same number of particles - the particles which make up the total are different. Serum is one of the most important body compartments. The following is an analysis of the osmotic components of serum and what high and low values mean clinically. Normal value for human serum is 285 to 290 mosm (= to C). 4
7 Biological Applications 2.1 Range of, and reason for, abnormal serum values. 1. What is abnormal - osmolality or osmotic concentration is one of the most highly regulated body variables. Generally a deviation of ±10 from the normal is considered significant, but obviously a knowledge of the patient history and other bio-chemical information must be taken into account before any diagnosis can be made. 2. Low osmolality - an extremely low osmolality would be 230 mosm (this has been reported). The cause of low osmolality is too little sodium (hyponatremia) or too much water. There are no other possibilities. In either case the sodium is low. 3. High osmolality - serum osmolality has been reported as high as 400 mosm in adults, sometimes higher in children. This can be caused by: a. High sodium - hypernatremia b. High glucose - hyperglycemia c. High urea - uremia d. The presence of an unknown molecule e. A combination of the above 2.2 Osmolality and sodium concentration Sodium and osmolality together are helpful in distinguishing among: dilution or water intoxication, true sodium depletion, progressive renal disease, primary water loss, and false hyponatremia due to liquids or artifacts occupying serum space. When used in conjunction with a flame photometer, an osmometer is useful in distinguishing between various types of low sodium---hyponatremia. Since an osmometer measures the concentration of solute per total water rather than per total volume (as a flame photometer), it will not be confused by the presence of something which changes the water content of the solution. Consider the following: 5
8 Applications of Freezing Point Osmometry 1. Low sodium and normal mosm means high lipids or other fatty artifact. 2. Low sodium and high mosm means that the particle which is increasing the osmolality is occupying volume and thus lowering the apparent concentration of sodium. This would be a high sugar (hyperglycemia), or high urea (uremia). 3. Low sodium and low mosm occur only when the sodium concentration per total water is low. This is either too little sodium or true dilutional hyponatremia---too much water. 2.3 Urine osmolality For accuracy and speed, the Advanced Osmometer is the best way to measure the concentrating power of the kidney. Certain conditions, artifacts, or drugs will falsify the specific gravity. 1. Urine varies widely in concentration in order to control the concentration of the blood. The kidney acts as a selective filter to pass water and hold solute or vice versa. 2. Urine osmolality runs from in a normal adult. In a 24- hour sample, it usually runs between 400 and 600 mosm. Most adult humans have a concentration capability of from mosm. Dogs can concentrate up to about The Australian Kangaroo Rat can concentrate beyond 5000 mosm. 3. Urine/serum ratio: The ratio of urine to serum osmolality can be an important diagnostic tool. a. Greater than 1 On a 24-hour urine sample, the urine/serum ratio should always be greater than 1 (for example, 600/290=2.1). b. Equal to 1 If the urine/serum ratio is only slightly greater than 1 or equal to 1 (for example, 330/320=1.05), then the patient may have severe renal shutdown. The kidney is not acting as a sieve. The closer to 1, the more acute the problem. 6
9 Biological Applications c. Less than 1 In some cases the urine/serum ratio may be less than 1 (for example, 200/300=.67). This occurs when the patient may have diabetes insipidus. 2.4 Unknown blood solutes Some unknown substances in blood raise the osmolality markedly. Other things being known, the Advanced Osmometer is very sensitive to some drug poisonings - salicylate and barbiturates as well as alcohol. Use of the Advanced Osmometer with a refractometer can indicate if the unknown solute can be dialyzed. 2.5 Clearance studies 1. Osmolal clearance: Osmolal clearance is defined as the number of cc s of plasma water which have lost all of their dissolved particles and ions in some measured period of time. If a 24-hour urine sample is available, the procedure for determining osmolal clearance would be as follows: a. Measure the volume of the urine. b. Determine the osmolality of the urine. c. Determine the osmolality of the serum. d. Calculate the urinary output in milliosmoles per day by multiplying the liters of urine by the milliosmolality. e. Divide the total output in milliosmoles by the serum osmolality expressed in milliosmoles per cc. f. Your answer will be osmolal clearance in cc/day. Example Osmolal Clearance: Assume the 24-hour vol. of urine = 2000 cc (2.000 liters) Assume the urine osmolality = 500 mosm/kg Then the osmolal output = x 500 = 1000 mosm/day 7
10 Applications of Freezing Point Osmometry Assume the serum osmolality = 290 mosm/kg (0.29 mosm/cc) Then the osmolal clearance = 1000 = 3448 cc s per day Urea Free Osmolal Clearance: When osmolal clearance values are needed for diagnosis of salt retention or abnormal salt loss, the results are usually of more value if the urine area concentration is determined, converted to millimoles per day (milliosmoles) and this figure then subtracted from the total milliosmoles excreted. The serum osmolality (in mosm/cc) is divided into this figure and the answer is the urea free osmolal clearance. Example Urea Free Osmolal Clearance Assume the 24-hour vol. of urine = 2000 cc (2.000 liters) Assume the urine osmolality = 500 mosm/liter Then the osmolal output = x 500 = 1000 mosm/day Assume the area output = 30 grams/day 30 grams of urea = 30 mole = 1 mole = 500 millimoles = 60 2 about 500 milliosmoles Then the urea free osmolal output = = 500 mosm/day Assume the serum osmolality = 290 mosm/kg (0.29 mosm/cc) Then the urea free osmolal clearance = 500 = 1724 cc s day Normal values: The osmolal clearance = cc s/day (quite variable) The urea free osmolal clearance of an average person on a mixed diet varies from cc s/day. A patient with congestive failure will have an osmolal clearance (urea free) as low as 600 cc s. Diuretics such as Diuril or Diamox cause dramatic increases in osmolal clearance of patients with salt retention. 8
11 Biological Applications 2.6 Monitoring electrolyte balance Since the osmolality of serum and most other body fluids is primarily affected by electrolytes, the Advanced Osmometer provides an excellent means of monitoring electrolyte balance in burn cases, shock or traumatic surgery. An electrolyte series can be run initially (including osmolality), then the osmolality can be measured as often as necessary as a check on the change in patient condition. In some serious acid-base disturbances (resulting from a variety of clinical conditions) the measured osmolality is sometimes remarkedly higher than the calculated osmolality (the sum of the individual electrolytes). Findings indicate that this usually may be fatal. Measured osmolality, using an osmometer, is the prime index. 2.7 Total body water Total body water (TBW) can be determined in a number of ways, but freezing point offers a relatively simple approach. Volume measurement by dilution is the technique. The solute is D 2 O--- heavy water. It quickly diffuses through all the membranes that water does, yet it changes the freezing point markedly. Serum water and the D 2 O are vacuum distilled from serum for the analysis. Pure D 2 O freezes at 3.82 C (above zero). A one percent mixture of D 2 O in water will freeze at C (one percent of 3.82). One mole of D 2 O, or 20 grams, dissolved in 1000 grams of water will elevate the freezing point about C. With a patient whose TBW was 40,000 grams, an injection of 400 grams (1%) of D 2 O would elevate the freezing point C. The Advanced Osmometer measures with a precision of ± C. Therefore, the 400 grams dose would yield an accuracy of ±1%. 80 grams would yield ±5%. 9
12 Applications of Freezing Point Osmometry 2.8 Dehydration Infant diarrhea is a major cause of pediatric death. Both in children and in adults, an Advanced Osmometer can be used to accurately indicate the extent of dehydration, to monitor the fluids which are administered, and to monitor the patient s response to the treatment. Particularly in infant diarrhea, but also in many kinds of adult dehydration, continuous investigations of the electrolyte-water ratios in serum and urine are important. The flame photometer tells only part of the story. 2.9 ADH Osmolality can be used for monitoring ADH suppression or secretion through the use of free water clearances Fluid therapy Osmolality is a clinical aid in the regulation of fluid therapy, particularly in post-surgical or trauma patients. Lactic acid may play an important role in this particular application using the results of the formula for calculated osmolality versus measured osmolality Na + Glucose + BUN Pediatrics Pediatric applications include water-loss or water excess syndrome, and diarrhea and vomiting Blood alcohol There are new research correlations between osmolality and blood alcohol levels. 10
13 3 Other Applications Since osmometers measure only dissolved particles in solution (1000 milliosmols = Avogadro s number [6.02 x ] of particles), the number of applications is almost unlimited. 3.1 Various body fluids Osmolality can be quickly determined on saliva, CSF, sweat, gastric juices, or any other body fluid of 0.2ml volume. The sample is not destroyed and can be used for other tests. 3.2 With an artificial kidney or dialyzer Osmolality can be used as a check on the patient s fluids and on the dialyzing media. 3.3 Molecular weight and chemical purity If a certain weight of chemical is made up of small molecules, there will be a much greater change in freezing point than the same weight of large molecules. Thus, the Advanced Cryoscope/Osmometer is a very sensitive tool for low molecular weights and number average molecular weights of mixed substances. The degree of chemical purity can be determined with great precision from the slope of the apparent freezing point versus supercooling curve. 11
14 Applications of Freezing Point Osmometry 3.4 Fetal maturity Osmolality can be used to determine fetal maturity by measuring the concentration of the amniotic fluid. 3.5 Chromatographic eluates Osmometers can monitor eluates from column chromatographic separations. 3.6 Quality control Laboratory reagents can be checked. 3.7 Fixing solutions The proper osmotic concentrations of a fixing solution can increase the resolution and clarity in cell microscopy, preserve enzyme activity, and prevent membrane distortion. For many biologists and anatomical pathologists, the Advanced Osmometer is becoming a necessary aid in the preparation and application of fixatives. 3.8 General concentration tests For dialyzing media, parenteral fluids, fluids administered after surgery, etc., the osmometer provides a fast, simple and accurate check. It can prevent the costly - often fatal - mistake of the administration of a fluid with the wrong concentration. Some hospitals now require the checking of all solutions to be administered after surgery. In addition to checking biological solutions, the osmometer can also be used to check standards for flame photometers and other instruments as well as a quality control check in the preparation of other reagents. 2264_SP Rev1 05/07/01 12
Electrolytes Solution
 Electrolytes Solution Substances that are not dissociated in solution are called nonelectrolytes, and those with varying degrees of dissociation are called electrolytes. Urea and dextrose are examples
Electrolytes Solution Substances that are not dissociated in solution are called nonelectrolytes, and those with varying degrees of dissociation are called electrolytes. Urea and dextrose are examples
Physiology of the body fluids, Homeostasis
 Physiology of the body fluids, Homeostasis Tamas Banyasz The Body as an open system 1. Open system: The body exchanges material and energy with its environment 2. Homeostasis: The process through which
Physiology of the body fluids, Homeostasis Tamas Banyasz The Body as an open system 1. Open system: The body exchanges material and energy with its environment 2. Homeostasis: The process through which
EXCRETION QUESTIONS. Use the following information to answer the next two questions.
 EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
Principles of Fluid Balance
 Principles of Fluid Balance I. The Cellular Environment: Fluids and Electrolytes A. Water 1. Total body water (TBW) = 60% of total body weight 2. Fluid Compartments in the Body a. Intracellular Compartment
Principles of Fluid Balance I. The Cellular Environment: Fluids and Electrolytes A. Water 1. Total body water (TBW) = 60% of total body weight 2. Fluid Compartments in the Body a. Intracellular Compartment
Part 1 The Cell and the Cellular Environment
 1 Chapter 3 Anatomy and Physiology Part 1 The Cell and the Cellular Environment 2 The Human Cell The is the fundamental unit of the human body. Cells contain all the necessary for life functions. 3 Cell
1 Chapter 3 Anatomy and Physiology Part 1 The Cell and the Cellular Environment 2 The Human Cell The is the fundamental unit of the human body. Cells contain all the necessary for life functions. 3 Cell
Fluids and electrolytes
 Body Water Content Fluids and electrolytes Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60% water; healthy females
Body Water Content Fluids and electrolytes Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60% water; healthy females
Urinalysis (Macroscopic Physical Tests) Urinalysis. Basic Urinalysis 12/22/2012. Mohammad Reza Bakhtiari DCLS, PhD
 Urinalysis (Macroscopic Physical Tests) Mohammad Reza Bakhtiari DCLS, PhD Urinalysis Basic Urinalysis CLSI: U/A is the testing of urine with procedures commonly performed in an expeditious, reliable, accurate,
Urinalysis (Macroscopic Physical Tests) Mohammad Reza Bakhtiari DCLS, PhD Urinalysis Basic Urinalysis CLSI: U/A is the testing of urine with procedures commonly performed in an expeditious, reliable, accurate,
Major intra and extracellular ions Lec: 1
 Major intra and extracellular ions Lec: 1 The body fluids are solutions of inorganic and organic solutes. The concentration balance of the various components is maintained in order for the cell and tissue
Major intra and extracellular ions Lec: 1 The body fluids are solutions of inorganic and organic solutes. The concentration balance of the various components is maintained in order for the cell and tissue
Kidney Physiology. Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed
 Kidney Physiology Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed The purpose of tubular secrection To dispose of certain substances that are bound to plasma proteins. To
Kidney Physiology Mechanisms of Urine Formation TUBULAR SECRETION Eunise A. Foster Shalonda Reed The purpose of tubular secrection To dispose of certain substances that are bound to plasma proteins. To
Water (Dysnatremia) & Sodium (Dysvolemia) Disorders Ahmad Raed Tarakji, MD, MSPH, PGCertMedEd, FRCPC, FACP, FASN, FNKF, FISQua
 Water (Dysnatremia) & Sodium (Dysvolemia) Disorders Ahmad Raed Tarakji, MD, MSPH, PGCertMedEd, FRCPC, FACP, FASN, FNKF, FISQua Assistant Professor Nephrology Unit, Department of Medicine College of Medicine,
Water (Dysnatremia) & Sodium (Dysvolemia) Disorders Ahmad Raed Tarakji, MD, MSPH, PGCertMedEd, FRCPC, FACP, FASN, FNKF, FISQua Assistant Professor Nephrology Unit, Department of Medicine College of Medicine,
Osmoregulation and Osmotic Balance
 OpenStax-CNX module: m44808 1 Osmoregulation and Osmotic Balance OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this
OpenStax-CNX module: m44808 1 Osmoregulation and Osmotic Balance OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this
Disclaimer. Chapter 3 Disorder of Water, Electrolyte and Acid-base Professor A. S. Alhomida. Disorder of Water and Electrolyte
 Disclaimer King Saud University College of Science Department of Biochemistry The texts, tables, figures and images contained in this course presentation (BCH 376) are not my own, they can be found on:
Disclaimer King Saud University College of Science Department of Biochemistry The texts, tables, figures and images contained in this course presentation (BCH 376) are not my own, they can be found on:
Body Water Content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are
 Fluid, Electrolyte, and Acid-Base Balance Body Water Content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60%
Fluid, Electrolyte, and Acid-Base Balance Body Water Content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60%
Cell Processes: Osmosis
 www.mathbench.umd.edu./modules-au Osmosis May 2015 page 1 Cell Processes: Osmosis URL: http://mathbench.org.au/cellular-processes/osmosis/ Learning Outcomes After completing this module you should be able
www.mathbench.umd.edu./modules-au Osmosis May 2015 page 1 Cell Processes: Osmosis URL: http://mathbench.org.au/cellular-processes/osmosis/ Learning Outcomes After completing this module you should be able
IV Fluids. I.V. Fluid Osmolarity Composition 0.9% NaCL (Normal Saline Solution, NSS) Uses/Clinical Considerations
 IV Fluids When administering IV fluids, the type and amount of fluid may influence patient outcomes. Make sure to understand the differences between fluid products and their effects. Crystalloids Crystalloid
IV Fluids When administering IV fluids, the type and amount of fluid may influence patient outcomes. Make sure to understand the differences between fluid products and their effects. Crystalloids Crystalloid
1. 09/07/16 Ch 1: Intro to Human A & P 1
 Table of Contents # Date Title Page # 1. 09/07/16 Ch 1: Intro to Human A & P 1 2. 09/19/16 Ch 18: Water, Electrolyte, and Acid-Base Balance 5 i 1 09/19/16 Chapter 18: Water, Electrolyte, and Acid-Base
Table of Contents # Date Title Page # 1. 09/07/16 Ch 1: Intro to Human A & P 1 2. 09/19/16 Ch 18: Water, Electrolyte, and Acid-Base Balance 5 i 1 09/19/16 Chapter 18: Water, Electrolyte, and Acid-Base
Terminology. Terminology. Terminology. Molarity number of moles of solute / Liter of solution. a) Terminology b) Body Fluid Compartments
 Integrative Sciences: Biological Systems A Fall 2011 Body Fluids Compartments, Renal Clearance and Renal Excretion of Drugs Monday, November 21, 2011 Lisa M. Harrison-Bernard, Ph.D. Department of Physiology;
Integrative Sciences: Biological Systems A Fall 2011 Body Fluids Compartments, Renal Clearance and Renal Excretion of Drugs Monday, November 21, 2011 Lisa M. Harrison-Bernard, Ph.D. Department of Physiology;
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE
 Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
BIOL 221 Chapter 26 Fluids & Electrolytes. 35 slides
 BIOL 221 Chapter 26 Fluids & Electrolytes 35 slides 1 Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender males have higher TBW
BIOL 221 Chapter 26 Fluids & Electrolytes 35 slides 1 Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender males have higher TBW
Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender
 BIOL 221 Chapter 26 Fluids & Electrolytes 35 slides 1 Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender males have higher TBW
BIOL 221 Chapter 26 Fluids & Electrolytes 35 slides 1 Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender males have higher TBW
Water, Electrolytes, and Acid-Base Balance
 Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Ch 17 Physiology of the Kidneys
 Ch 17 Physiology of the Kidneys Review Anatomy on your own SLOs List and describe the 4 major functions of the kidneys. List and explain the 4 processes of the urinary system. Diagram the filtration barriers
Ch 17 Physiology of the Kidneys Review Anatomy on your own SLOs List and describe the 4 major functions of the kidneys. List and explain the 4 processes of the urinary system. Diagram the filtration barriers
UNIVERSITY OF MEDICAL SCIENCES, ONDO DEPARTMENT OF PHYSIOLOGY PHS 212: BLOOD AND BODY FLUID PHYSIOLOGY LECTURER: MR A.O. AKINOLA
 UNIVERSITY OF MEDICAL SCIENCES, ONDO DEPARTMENT OF PHYSIOLOGY PHS 212: BLOOD AND BODY FLUID PHYSIOLOGY LECTURER: MR A.O. AKINOLA OBJECTIVES Introduction Definition of body fluids and body fluid compartments
UNIVERSITY OF MEDICAL SCIENCES, ONDO DEPARTMENT OF PHYSIOLOGY PHS 212: BLOOD AND BODY FLUID PHYSIOLOGY LECTURER: MR A.O. AKINOLA OBJECTIVES Introduction Definition of body fluids and body fluid compartments
Physical Characteristics of
 Physical Characteristics of Urine Bởi: OpenStaxCollege The urinary system s ability to filter the blood resides in about 2 to 3 million tufts of specialized capillaries the glomeruli distributed more or
Physical Characteristics of Urine Bởi: OpenStaxCollege The urinary system s ability to filter the blood resides in about 2 to 3 million tufts of specialized capillaries the glomeruli distributed more or
Amjad Bani Hani Ass.Prof. of Cardiac Surgery & Intensive Care FLUIDS AND ELECTROLYTES
 Amjad Bani Hani Ass.Prof. of Cardiac Surgery & Intensive Care FLUIDS AND ELECTROLYTES Body Water Content Water Balance: Normal 2500 2000 1500 1000 500 Metab Food Fluids Stool Breath Sweat Urine
Amjad Bani Hani Ass.Prof. of Cardiac Surgery & Intensive Care FLUIDS AND ELECTROLYTES Body Water Content Water Balance: Normal 2500 2000 1500 1000 500 Metab Food Fluids Stool Breath Sweat Urine
Acids and Bases their definitions and meanings
 Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
SODIUM BALANCE Overview
 SODIUM BALANCE Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS III Seminar VJ Temple 1 How are solute and solvent related to solution?
SODIUM BALANCE Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS III Seminar VJ Temple 1 How are solute and solvent related to solution?
Body fluids. Lecture 13:
 Lecture 13: Body fluids Body fluids are distributed in compartments: A. Intracellular compartment: inside the cells of the body (two thirds) B. Extracellular compartment: (one third) it is divided into
Lecture 13: Body fluids Body fluids are distributed in compartments: A. Intracellular compartment: inside the cells of the body (two thirds) B. Extracellular compartment: (one third) it is divided into
WATER, SODIUM AND POTASSIUM
 WATER, SODIUM AND POTASSIUM Attila Miseta Tamás Kőszegi Department of Laboratory Medicine, 2016 1 Average daily water intake and output of a normal adult 2 Approximate contributions to plasma osmolality
WATER, SODIUM AND POTASSIUM Attila Miseta Tamás Kőszegi Department of Laboratory Medicine, 2016 1 Average daily water intake and output of a normal adult 2 Approximate contributions to plasma osmolality
Chapter 19 The Urinary System Fluid and Electrolyte Balance
 Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Lab 4: Osmosis and Diffusion
 Page 4.1 Lab 4: Osmosis and Diffusion Cells need to obtain water and other particles from the fluids that surround them. Water and other particles also move out of cells. Osmosis (for water) and diffusion
Page 4.1 Lab 4: Osmosis and Diffusion Cells need to obtain water and other particles from the fluids that surround them. Water and other particles also move out of cells. Osmosis (for water) and diffusion
Expressing the Amount of Solute by Weight:
 Expressing the Concentration of Substances in Solution A solution consists of one or more substances (Solutes) that are dissolved in a liquid (Solvent). If the liquid is water, it is called an aqueous
Expressing the Concentration of Substances in Solution A solution consists of one or more substances (Solutes) that are dissolved in a liquid (Solvent). If the liquid is water, it is called an aqueous
Fahed alkarmi. Bahaa najjar. Muhammad khatatbeh
 13 Fahed alkarmi Bahaa najjar Muhammad khatatbeh We have said before that we have a certain amount of water in our bodies, this amount is distributed as follows: 1- Two thirds (2/3) of that water is inside
13 Fahed alkarmi Bahaa najjar Muhammad khatatbeh We have said before that we have a certain amount of water in our bodies, this amount is distributed as follows: 1- Two thirds (2/3) of that water is inside
For more information about how to cite these materials visit
 Author(s): Michael Heung, M.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Noncommercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Author(s): Michael Heung, M.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Noncommercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Fal Fal P h y s i o l o g y 6 1 1, S a n F r a n c i s c o S t a t e U n i v e r s i t y
 Fall 12 OSMOTIC REGULATION OF THE RENAL SYSTEM: Effects of fasting and ingestion of water, coke, or Gatorade on urine flow rate and specific gravity Dorette Franks The purpose of the physiology experiment
Fall 12 OSMOTIC REGULATION OF THE RENAL SYSTEM: Effects of fasting and ingestion of water, coke, or Gatorade on urine flow rate and specific gravity Dorette Franks The purpose of the physiology experiment
INTRAVENOUS FLUID THERAPY
 INTRAVENOUS FLUID THERAPY PRINCIPLES Postnatal physiological weight loss is approximately 5 10% in first week of life Preterm neonates have more total body water and may lose 10 15% of their weight in
INTRAVENOUS FLUID THERAPY PRINCIPLES Postnatal physiological weight loss is approximately 5 10% in first week of life Preterm neonates have more total body water and may lose 10 15% of their weight in
Instrumental determination of electrolytes in urine. Amal Alamri
 Instrumental determination of electrolytes in urine Amal Alamri What is the Electrolytes? Electrolytes are positively and negatively chargedions, Found in Within body's cells extracellular fluids, including
Instrumental determination of electrolytes in urine Amal Alamri What is the Electrolytes? Electrolytes are positively and negatively chargedions, Found in Within body's cells extracellular fluids, including
The Urinary System 15PART B. PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART B Ureters Slender tubes attaching the kidney to the bladder Continuous with
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART B Ureters Slender tubes attaching the kidney to the bladder Continuous with
Dr. Dafalla Ahmed Babiker Jazan University
 Dr. Dafalla Ahmed Babiker Jazan University objectives Overview Definition of dehydration Causes of dehydration Types of dehydration Diagnosis, signs and symptoms Management of dehydration Complications
Dr. Dafalla Ahmed Babiker Jazan University objectives Overview Definition of dehydration Causes of dehydration Types of dehydration Diagnosis, signs and symptoms Management of dehydration Complications
Fluid and Electrolytes P A R T 4
 Fluid and Electrolytes P A R T 4 Mechanisms that control acid-base homeostasis Acids and bases continually enter and leave body Hydrogen ions also result from metabolic activity Acids Hydrogen ion donors
Fluid and Electrolytes P A R T 4 Mechanisms that control acid-base homeostasis Acids and bases continually enter and leave body Hydrogen ions also result from metabolic activity Acids Hydrogen ion donors
Acids, Bases, and Salts
 Acid / Base Balance Objectives Define an acid, a base, and the measure of ph. Discuss acid/base balance, the effects of acidosis or alkalosis on the body, and the mechanisms in place to maintain balance
Acid / Base Balance Objectives Define an acid, a base, and the measure of ph. Discuss acid/base balance, the effects of acidosis or alkalosis on the body, and the mechanisms in place to maintain balance
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance
 Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
Principles of Anatomy and Physiology
 Principles of Anatomy and Physiology 14 th Edition CHAPTER 27 Fluid, Electrolyte, and Acid Base Fluid Compartments and Fluid In adults, body fluids make up between 55% and 65% of total body mass. Body
Principles of Anatomy and Physiology 14 th Edition CHAPTER 27 Fluid, Electrolyte, and Acid Base Fluid Compartments and Fluid In adults, body fluids make up between 55% and 65% of total body mass. Body
Body Water ANS 215 Physiology and Anatomy of Domesticated Animals
 Body Water ANS 215 Physiology and Anatomy of Domesticated Animals I. Body Water A. Water is the most abundant constituent comprising 60% of total body weight. 1. Solvent for many chemicals of the body
Body Water ANS 215 Physiology and Anatomy of Domesticated Animals I. Body Water A. Water is the most abundant constituent comprising 60% of total body weight. 1. Solvent for many chemicals of the body
Basic Fluid and Electrolytes
 Basic Fluid and Electrolytes Chapter 22 Basic Fluid and Electrolytes Introduction Infants and young children have a greater need for water and are more vulnerable to alterations in fluid and electrolyte
Basic Fluid and Electrolytes Chapter 22 Basic Fluid and Electrolytes Introduction Infants and young children have a greater need for water and are more vulnerable to alterations in fluid and electrolyte
Module 8: Electrolyte Solutions
 PHARMACEUTICAL CALCULATIONS FALL 207 Contents General Vocab & Definitions... 2 Milliequivalent Calculations... 2 Millimole and Micromole Calculations... 5 Osmolarity... 6 Daily Water Requirement Calculations
PHARMACEUTICAL CALCULATIONS FALL 207 Contents General Vocab & Definitions... 2 Milliequivalent Calculations... 2 Millimole and Micromole Calculations... 5 Osmolarity... 6 Daily Water Requirement Calculations
INTRAVENOUS FLUIDS PRINCIPLES
 INTRAVENOUS FLUIDS PRINCIPLES Postnatal physiological weight loss is approximately 5-10% Postnatal diuresis is delayed in Respiratory Distress Syndrome (RDS) Preterm babies have limited capacity to excrete
INTRAVENOUS FLUIDS PRINCIPLES Postnatal physiological weight loss is approximately 5-10% Postnatal diuresis is delayed in Respiratory Distress Syndrome (RDS) Preterm babies have limited capacity to excrete
Water compartments inside and outside cells maintain a balanced distribution of total body water.
 Chapter 9 Water Balance Chapter 9 Lesson 9.1 Key Concepts Water compartments inside and outside cells maintain a balanced distribution of total body water. The concentration of various solute particles
Chapter 9 Water Balance Chapter 9 Lesson 9.1 Key Concepts Water compartments inside and outside cells maintain a balanced distribution of total body water. The concentration of various solute particles
The table indicates how changing the variable listed alone will alter diffusion rate.
 Rate of Diffusion (flux) Concentration gradient substance x surface area of membrane x lipid solubility = Distance (thickness of membrane) x molecular weight Table 3-1: Factors Influencing the Rate of
Rate of Diffusion (flux) Concentration gradient substance x surface area of membrane x lipid solubility = Distance (thickness of membrane) x molecular weight Table 3-1: Factors Influencing the Rate of
Glucosuria: Diabetes Mellitus
 172 PHYSIOLOGY CASES AND PROBLEMS Case 30 Glucosuria: Diabetes Mellitus David Mandel was diagnosed with type I (insulin-dependent) diabetes mellitus when he was 12 years old, right after he started middle
172 PHYSIOLOGY CASES AND PROBLEMS Case 30 Glucosuria: Diabetes Mellitus David Mandel was diagnosed with type I (insulin-dependent) diabetes mellitus when he was 12 years old, right after he started middle
Faculty version with model answers
 Faculty version with model answers Urinary Dilution & Concentration Bruce M. Koeppen, M.D., Ph.D. University of Connecticut Health Center 1. Increased urine output (polyuria) can result in a number of
Faculty version with model answers Urinary Dilution & Concentration Bruce M. Koeppen, M.D., Ph.D. University of Connecticut Health Center 1. Increased urine output (polyuria) can result in a number of
CHAPTER. Movement Across Plasma Membrane. Chapter 6 Outline. Diffusion Osmosis. Membrane Potential Cell Signaling
 CHAPTER 6 Interaction Between Cells and the Extracellular Environment Chapter 6 Outline Extracellular Environment Diffusion Osmosis Carrier-Mediated Carrier Mediated Transport Membrane Potential Cell Signaling
CHAPTER 6 Interaction Between Cells and the Extracellular Environment Chapter 6 Outline Extracellular Environment Diffusion Osmosis Carrier-Mediated Carrier Mediated Transport Membrane Potential Cell Signaling
URINE CONCENTRATION AND REGULATION OF ECF OSMOLARITY
 URINE CONCENTRATION AND REGULATION OF ECF OSMOLARITY Dilute and concentrated urine 1-Dilute urine : Nephron function continuous reabsorption. Solutes while failing to reabsorbe water in distal tubule and
URINE CONCENTRATION AND REGULATION OF ECF OSMOLARITY Dilute and concentrated urine 1-Dilute urine : Nephron function continuous reabsorption. Solutes while failing to reabsorbe water in distal tubule and
Acid Base Balance. Chapter 26 Balance. ph Imbalances. Acid Base Balance. CO 2 and ph. Carbonic Acid. Part 2. Acid/Base Balance
 Acid Base Balance Chapter 26 Balance Part 2. Acid/Base Balance Precisely balances production and loss of hydrogen ions (ph) The body generates acids during normal metabolism, tends to reduce ph Kidneys:
Acid Base Balance Chapter 26 Balance Part 2. Acid/Base Balance Precisely balances production and loss of hydrogen ions (ph) The body generates acids during normal metabolism, tends to reduce ph Kidneys:
Don t Forget the Strong Ions
 Don t Forget the s Rich Erdman University of Maryland Department of Animal & Avian Sciences erdman@umd.edu Don t Forget the s Rich Erdman Department of Animal & Avian Sciences erdman@umd.edu K + 39.1 Na
Don t Forget the s Rich Erdman University of Maryland Department of Animal & Avian Sciences erdman@umd.edu Don t Forget the s Rich Erdman Department of Animal & Avian Sciences erdman@umd.edu K + 39.1 Na
Hyponatremia. Mis-named talk? Basic Pathophysiology
 Hyponatremia Great Lakes Hospital Medicine Symposium by Brian Wolfe, MD Assistant Professor of Internal Medicine University of Colorado Denver Mis-named talk? Why do we care about Hyponatremia? concentration
Hyponatremia Great Lakes Hospital Medicine Symposium by Brian Wolfe, MD Assistant Professor of Internal Medicine University of Colorado Denver Mis-named talk? Why do we care about Hyponatremia? concentration
** Accordingly GFR can be estimated by using one urine sample and do creatinine testing.
 This sheet includes the lecture and last year s exam. When a patient goes to a clinic, we order 2 tests: 1) kidney function test: in which we measure UREA and CREATININE levels, and electrolytes (Na+,
This sheet includes the lecture and last year s exam. When a patient goes to a clinic, we order 2 tests: 1) kidney function test: in which we measure UREA and CREATININE levels, and electrolytes (Na+,
Fluid, Electrolyte, and Acid Base Balance
 Chapter 26 Fluid, Electrolyte, and Acid Base Balance Bi 233 Body Water Content Largest component of the body Infants have low body fat, low bone mass, and are 73% or more water Healthy males are about
Chapter 26 Fluid, Electrolyte, and Acid Base Balance Bi 233 Body Water Content Largest component of the body Infants have low body fat, low bone mass, and are 73% or more water Healthy males are about
Fluid, Electrolyte, and Acid Base Balance
 25 Fluid, Electrolyte, and Acid Base Balance Lecture Presentation by Lori Garrett Note to the Instructor: For the third edition of Visual Anatomy & Physiology, we have updated our PowerPoints to fully
25 Fluid, Electrolyte, and Acid Base Balance Lecture Presentation by Lori Garrett Note to the Instructor: For the third edition of Visual Anatomy & Physiology, we have updated our PowerPoints to fully
Extracellular fluid (ECF) compartment volume control
 Water Balance Made Easier Joon K. Choi, DO. Extracellular fluid (ECF) compartment volume control Humans regulate ECF volume mainly by regulating body sodium content. Several major systems work together
Water Balance Made Easier Joon K. Choi, DO. Extracellular fluid (ECF) compartment volume control Humans regulate ECF volume mainly by regulating body sodium content. Several major systems work together
Regulation of fluid and electrolytes balance
 Regulation of fluid and electrolytes balance Three Compartment Fluid Compartments Intracellular = Cytoplasmic (inside cells) Extracellular compartment is subdivided into Interstitial = Intercellular +
Regulation of fluid and electrolytes balance Three Compartment Fluid Compartments Intracellular = Cytoplasmic (inside cells) Extracellular compartment is subdivided into Interstitial = Intercellular +
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D.
 RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
Excretory System-Training Handout
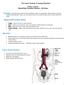 Excretory System-Training Handout Karen L. Lancour National Rules Committee Chairman Life Science Excretion - Excretion is the removal of the metabolic wastes of an organism. Wastes that are removed include
Excretory System-Training Handout Karen L. Lancour National Rules Committee Chairman Life Science Excretion - Excretion is the removal of the metabolic wastes of an organism. Wastes that are removed include
Fluid and electrolyte balance, imbalance
 Fluid and electrolyte balance, imbalance Body fluid The fluids are distributed throughout the body in various compartments. Body fluid is composed primarily of water Water is the solvent in which all solutes
Fluid and electrolyte balance, imbalance Body fluid The fluids are distributed throughout the body in various compartments. Body fluid is composed primarily of water Water is the solvent in which all solutes
November 30, 2016 & URINE FORMATION
 & URINE FORMATION REVIEW! Urinary/Renal System 200 litres of blood are filtered daily by the kidneys Usable material: reabsorbed back into blood Waste: drained into the bladder away from the heart to the
& URINE FORMATION REVIEW! Urinary/Renal System 200 litres of blood are filtered daily by the kidneys Usable material: reabsorbed back into blood Waste: drained into the bladder away from the heart to the
Osmotic Regulation and the Urinary System. Chapter 50
 Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
BIOL 2402 Fluid/Electrolyte Regulation
 Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Chapter 12. Excretion and the Interaction of Systems
 Chapter 12 Excretion and the Interaction of Systems 1 2 Goals for This Chapter 1. Identify the main structures and functions of the human excretory system 2. Explain the function of the nephron 3. Describe
Chapter 12 Excretion and the Interaction of Systems 1 2 Goals for This Chapter 1. Identify the main structures and functions of the human excretory system 2. Explain the function of the nephron 3. Describe
Module 8: Practice Problems
 Module 8: Practice Problems 1. Convert a blood plasma level range of 5 to 20 µg/ml of tobramycin (Z = 467.52) to µmol/l. 5 µg/ml = 10.7 µmol/l 20 µg/ml = 42.8 µmol/l 2. A preparation contains in each milliliter,
Module 8: Practice Problems 1. Convert a blood plasma level range of 5 to 20 µg/ml of tobramycin (Z = 467.52) to µmol/l. 5 µg/ml = 10.7 µmol/l 20 µg/ml = 42.8 µmol/l 2. A preparation contains in each milliliter,
Chapter 23. Composition and Properties of Urine
 Chapter 23 Composition and Properties of Urine Composition and Properties of Urine (1 of 2) urinalysis the examination of the physical and chemical properties of urine appearance - clear, almost colorless
Chapter 23 Composition and Properties of Urine Composition and Properties of Urine (1 of 2) urinalysis the examination of the physical and chemical properties of urine appearance - clear, almost colorless
CHAPTER 27 LECTURE OUTLINE
 CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
LAB 4: OSMOSIS AND DIFFUSION
 Page 4.1 LAB 4: OSMOSIS AND DIFFUSION Cells need to obtain water and other particles from the fluids that surround them. Water and other particles also move out of cells. Osmosis (for water) and diffusion
Page 4.1 LAB 4: OSMOSIS AND DIFFUSION Cells need to obtain water and other particles from the fluids that surround them. Water and other particles also move out of cells. Osmosis (for water) and diffusion
BIO132 Chapter 27 Fluid, Electrolyte and Acid Base Balance Lecture Outline
 BIO132 Chapter 27 Fluid, Electrolyte and Acid Base Balance Lecture Outline Fluid divisions 1. Extracellular fluid (ECF) 2. Intracellular fluid (ICF) Stabilization 1. Fluid balance 2. Electrolyte balance
BIO132 Chapter 27 Fluid, Electrolyte and Acid Base Balance Lecture Outline Fluid divisions 1. Extracellular fluid (ECF) 2. Intracellular fluid (ICF) Stabilization 1. Fluid balance 2. Electrolyte balance
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + )
 Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Urea Nitrogen (BUN) detection Kit
 K-ASSAY KAMIYA BIOMEDICAL COMPANY KAMIYA BIOMEDICAL COMPANY Urea Nitrogen (BUN) detection Kit For the quantitative determination of urea nitrogen in saliva and TCM Cat. No. KT-747 For Research Use Only.
K-ASSAY KAMIYA BIOMEDICAL COMPANY KAMIYA BIOMEDICAL COMPANY Urea Nitrogen (BUN) detection Kit For the quantitative determination of urea nitrogen in saliva and TCM Cat. No. KT-747 For Research Use Only.
Chapter 4 Cell Membrane Transport
 Chapter 4 Cell Membrane Transport Plasma Membrane Review o Functions Separate ICF / ECF Allow exchange of materials between ICF / ECF such as obtaining O2 and nutrients and getting rid of waste products
Chapter 4 Cell Membrane Transport Plasma Membrane Review o Functions Separate ICF / ECF Allow exchange of materials between ICF / ECF such as obtaining O2 and nutrients and getting rid of waste products
Dr. Carlos Fernando Estrada Garzona. Departamento de Farmacología Universidad de Costa Rica
 Dr. Carlos Fernando Estrada Garzona Departamento de Farmacología Universidad de Costa Rica OBJETIVOS FISIOLOGIA LIQUIDOS CORPORALES SOLUCIONES PARENTERALES PRINCIPIOS DE FLUIDOTERAPIA CRISTALOIDE VS COLOIDE
Dr. Carlos Fernando Estrada Garzona Departamento de Farmacología Universidad de Costa Rica OBJETIVOS FISIOLOGIA LIQUIDOS CORPORALES SOLUCIONES PARENTERALES PRINCIPIOS DE FLUIDOTERAPIA CRISTALOIDE VS COLOIDE
Managing Acid Base and Electrolyte Disturbances with RRT
 Managing Acid Base and Electrolyte Disturbances with RRT John R Prowle MA MSc MD MRCP FFICM Consultant in Intensive Care & Renal Medicine RRT for Regulation of Acid-base and Electrolyte Acid base load
Managing Acid Base and Electrolyte Disturbances with RRT John R Prowle MA MSc MD MRCP FFICM Consultant in Intensive Care & Renal Medicine RRT for Regulation of Acid-base and Electrolyte Acid base load
Body water content. Fluid compartments. Regulation of water output. Water balance and ECF osmolallty. Regulation of water intake
 Body water content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60% water; females 50% This difference reflects
Body water content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60% water; females 50% This difference reflects
BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1
 BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1 1. a. Proximal tubule. b. Proximal tubule. c. Glomerular endothelial fenestrae, filtration slits between podocytes of Bowman's capsule.
BIPN100 F15 Human Physiology (Kristan) Problem Set #8 Solutions p. 1 1. a. Proximal tubule. b. Proximal tubule. c. Glomerular endothelial fenestrae, filtration slits between podocytes of Bowman's capsule.
Outline Urinary System. Urinary System and Excretion. Urine. Urinary System. I. Function II. Organs of the urinary system
 Outline Urinary System Urinary System and Excretion Bio105 Chapter 16 Renal will be on the Final only. I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of
Outline Urinary System Urinary System and Excretion Bio105 Chapter 16 Renal will be on the Final only. I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of
Chapter 44. Regulating the Internal Environment. AP Biology
 Chapter 44. Regulating the Internal Environment Homeostasis Living in the world organisms had a choice: regulate their internal environment maintain relatively constant internal conditions conform to the
Chapter 44. Regulating the Internal Environment Homeostasis Living in the world organisms had a choice: regulate their internal environment maintain relatively constant internal conditions conform to the
Slide 1. Slide 2. Slide 3. Learning Outcomes. Acid base terminology ARTERIAL BLOOD GAS INTERPRETATION
 Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
Acid-Base Balance Dr. Gary Mumaugh
 Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Hypo/Hypernatremia. Stuart L. Goldstein MD. Director, Center for Acute Care Nephrology Cincinnati Children s Hospital
 Hypo/Hypernatremia Stuart L. Goldstein MD Director, Center for Acute Care Nephrology Cincinnati Children s Hospital Objectives Understand Fluid cellular shifts Understand maintenance fluid and calculations
Hypo/Hypernatremia Stuart L. Goldstein MD Director, Center for Acute Care Nephrology Cincinnati Children s Hospital Objectives Understand Fluid cellular shifts Understand maintenance fluid and calculations
fluids were subjected to analysis. C. Gravimetric estimation of serum water:
 THE DISPLACEMENT OF SERUM WATER BY THE LIPIDS OF HYPERLIPEMIC SERUM. A NEW METHOD FOR THE RAPID DETERMINATION OF SERUM WATER' By MARGARET J. ALBRINK, PAULINE M. HALD, EVELYN B. MAN, AND JOHN P. PETERS
THE DISPLACEMENT OF SERUM WATER BY THE LIPIDS OF HYPERLIPEMIC SERUM. A NEW METHOD FOR THE RAPID DETERMINATION OF SERUM WATER' By MARGARET J. ALBRINK, PAULINE M. HALD, EVELYN B. MAN, AND JOHN P. PETERS
Renal Physiology Part II. Bio 219 Napa Valley College Dr. Adam Ross
 Renal Physiology Part II Bio 219 Napa Valley College Dr. Adam Ross Fluid and Electrolyte balance As we know from our previous studies: Water and ions need to be balanced in order to maintain proper homeostatic
Renal Physiology Part II Bio 219 Napa Valley College Dr. Adam Ross Fluid and Electrolyte balance As we know from our previous studies: Water and ions need to be balanced in order to maintain proper homeostatic
Excretion Chapter 29. The Mammalian Excretory System consists of. The Kidney. The Nephron: the basic unit of the kidney.
 Excretion Chapter 29 The Mammalian Excretory System consists of The Kidney 1. Vertebrate kidneys perform A. Ion balance B. Osmotic balance C. Blood pressure D. ph balance E. Excretion F. Hormone production
Excretion Chapter 29 The Mammalian Excretory System consists of The Kidney 1. Vertebrate kidneys perform A. Ion balance B. Osmotic balance C. Blood pressure D. ph balance E. Excretion F. Hormone production
Cellular Physiology. Body Fluids: 1) Water: (universal solvent) Body water varies based on of age, sex, mass, and body composition
 Membrane Physiology Body Fluids: 1) Water: (universal solvent) Body water varies based on of age, sex, mass, and body composition H 2 O ~ 73% body weight Low body fat; Low bone mass H 2 O ( ) ~ 60% body
Membrane Physiology Body Fluids: 1) Water: (universal solvent) Body water varies based on of age, sex, mass, and body composition H 2 O ~ 73% body weight Low body fat; Low bone mass H 2 O ( ) ~ 60% body
Transport across the cell membrane
 Transport across the cell membrane Learning objectives Body compartments ECF and ICF Constituents Lipid Bilayer: Barrier to water and water-soluble substances ions glucose H 2 O urea CO 2 O 2 N 2 halothane
Transport across the cell membrane Learning objectives Body compartments ECF and ICF Constituents Lipid Bilayer: Barrier to water and water-soluble substances ions glucose H 2 O urea CO 2 O 2 N 2 halothane
The Urinary System PART B
 15 The Urinary System PART B PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Urinary Bladder Smooth,
15 The Urinary System PART B PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Urinary Bladder Smooth,
Water in the Body. Page Water = ~50 to 70% of body weight, depending on body fat % (higher body fat=lower water content)
 Water in the Body Page 373-375 Water = ~50 to 70% of body weight, depending on body fat % (higher body fat=lower water content) Major fluid compartments: Intracellular fluid: cytoplasm of cells is an aqueous
Water in the Body Page 373-375 Water = ~50 to 70% of body weight, depending on body fat % (higher body fat=lower water content) Major fluid compartments: Intracellular fluid: cytoplasm of cells is an aqueous
Objectives Body Fluids Electrolytes The Kidney and formation of urine
 Objectives Body Fluids Outline the functions of water in the body. State how water content varies with age and sex. Differentiate between intracellular and extra-cellular fluid. Explain how water moves
Objectives Body Fluids Outline the functions of water in the body. State how water content varies with age and sex. Differentiate between intracellular and extra-cellular fluid. Explain how water moves
Acid-Base Balance * OpenStax
 OpenStax-CNX module: m46409 1 Acid-Base Balance * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will be
OpenStax-CNX module: m46409 1 Acid-Base Balance * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will be
Acid - base equilibrium
 Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
BCH 450 Biochemistry of Specialized Tissues
 BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
Fluid, Electrolyte and Acid-Base Disorders
 Fluid, Electrolyte and Acid-Base Disorders Alluru S. Reddi Fluid, Electrolyte and Acid-Base Disorders Clinical Evaluation and Management 1 3 Alluru S. Reddi Department of Medicine Division of Nephrology
Fluid, Electrolyte and Acid-Base Disorders Alluru S. Reddi Fluid, Electrolyte and Acid-Base Disorders Clinical Evaluation and Management 1 3 Alluru S. Reddi Department of Medicine Division of Nephrology
Urinary System and Excretion. Bio105 Lecture 20 Chapter 16
 Urinary System and Excretion Bio105 Lecture 20 Chapter 16 1 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of the urinary system
Urinary System and Excretion Bio105 Lecture 20 Chapter 16 1 Outline Urinary System I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of the urinary system
Done By: Lulu Al-Obaid - Abdulrahman Al-Rashed Reviewed By: Mohammed Jameel Khulood Al-Raddadi
 Done By: Lulu Al-Obaid - Abdulrahman Al-Rashed Reviewed By: Mohammed Jameel Khulood Al-Raddadi At the end of this lecture student should be able to describe: The loop of Henle is referred to as countercurrent
Done By: Lulu Al-Obaid - Abdulrahman Al-Rashed Reviewed By: Mohammed Jameel Khulood Al-Raddadi At the end of this lecture student should be able to describe: The loop of Henle is referred to as countercurrent
Technical University of Mombasa Faculty of Applied and Health Sciences
 Technical University of Mombasa Faculty of Applied and Health Sciences DEPARTMENT OF MEDICAL SCIENCES UNIVERSITY EXAMINATION FOR THE DEGREE OF BACHELOR OF MEDICAL LABORATORY SCIENCES BMLS 12S -Regular
Technical University of Mombasa Faculty of Applied and Health Sciences DEPARTMENT OF MEDICAL SCIENCES UNIVERSITY EXAMINATION FOR THE DEGREE OF BACHELOR OF MEDICAL LABORATORY SCIENCES BMLS 12S -Regular
