Cell Processes: Osmosis
|
|
|
- Lynn Bradford
- 6 years ago
- Views:
Transcription
1 Osmosis May 2015 page 1 Cell Processes: Osmosis URL: Learning Outcomes After completing this module you should be able to: Determine that osmolarity of a solution Use the two compartment model to determine which way water will flow through a selectively permeable membrane, and the rate of flow of water Osmosis is a special kind of diffusion First we told you that diffusion through a membrane was just a special case of diffusion -- we got you to assume that there were only two compartments of interest (left and right), which simplified the maths a lot. Now, we're going to tell you that osmosis is a special case of diffusion through the membrane. And it's the membrane that makes it special. Recall that membranes vary in terms of their permeability. Some might be like the proverbial brick wall, while others might be 'as leaky as a sieve'. Imagine a membrane with gaps* just big enough to let water through, but too small to let anything else across. How is this a special case of diffusion through a membrane? Basically, what happens is that water can diffuse through the membrane, but nothing else in the water -- salt, sugar, or other solute can. Instead of general permeability of the membrane, what we care about now is permeability to water, or P water. Continuous
2 Osmosis May 2015 page 2 Discrete *more properly, permeability of the membrane has to do with selective gateways and polarity and so on, but imagining gaps in the membrane will work too. Putting the "Os" into "Osmolarity" We need to make one other change. So far we've used the molarity (moles of solute per litre) of solutions, like sugar water, as our way of measuring concentration. In truth, the measure of concentration that we need is not molarity, but osmolarity. Osmolarity is defined by osmoles of solute per litre (kind of like moles, but with an OS on the front). The abbreviation, which we'll use a lot, is OsM. So what is an osmole? As you may know, when you dissolve salt in water, it dissociates into Na + and Cl -, in other words, into two pieces. If 1 mole of molecules dissolves into two "pieces" it creates 2 osmoles. If we started with 1 mole of MgCl 2, which dissolves into 3 "pieces", we would get 3 osmoles. Sugar is an organic rather than ionic compound, so it does not dissociate, and 1 mole of sugar dissolves into 1 osmole. So when you are working out osmolarity the first thing you will need to determine is whether or not the salt in question dissociates into smaller ions. When we're talking about diffusion across a membrane (including osmosis), it doesn't actually matter that salt ions are smaller than sugar molecules, or that their molecular weight is less, or even that the ions are charged while the sugar molecules are electrically neutral. The only thing that actually matters is how many "pieces" the salt dissociates into. For example for a salt "molecule*" that dissociates into two ions, when you dissolve it into water, you will get TWO ions rather than one, and this is what matters for diffusion. Essentially the salt molecule provides twice as many "bits" of dissolved stuff as the sugar molecule, so the osmolarity is also doubled. If I dissolve 2 moles of table salt (NaCl) in 1 litre of water, what osmolarity is produced? One molecule of salt makes 2 "bits" when dissolved 2 moles of salt will make 4 moles of "bits" Answer: 4 moles of "bits" / 1L of water = 4 OsM
3 Osmosis May 2015 page 3 If I dissolve 4 moles of MgCl 2 in 2 L of water what will the osmolarity be? MgCl 2 dissolves into Mg Cl - or 3 bits osmolarity is "bits" / litre Answer: 4 moles MgCl2 = 12 moles of "bits" 12 moles / 2L = 6 OsM *Yes, I know that salt is an ionic compound and comes in the of crystalline lattice. However, it is easier to think in terms of a salt "molecule" Calculating osmolarity If you know molarity: If you need to figure out the osmolarity of a solution and you're given the molar concentration, its pretty simple, just multiply the molarity by however many "pieces" the solute dissociates into. What is the osmolarity of a 2.6 M solution of table salt (NaCl)? Salt dissolves into 2 "bits". Read the section above again! Answer: 2.6 M * 2 = 5.2 OsM If you know the amount of solute: What if you just know the amount of a solute that was added? For example: what is the approximate osmolarity of the sugar in a can of Red Bull? According to the can which just happens to be sitting next to me, there are 27 g of sugar in 250 ml. The sugar in Red Bull is a mixture of glucose and sucrose (a disaccharide made up of one molecule of glucose and one molecule of fructose bonded together). The molecular weight of sucrose is 342. For simplicity, however we will assume here that all the sugar is in the of glucose. First, grams to moles: 27g * 1 mole/180g = 0.15 moles Finally, we know that sugar does not break apart, so 1 M = 1 OsM. Now that everything is in the right units, we just need to divide moles by litres to get the osmolarity: 0.15 moles / 0.25 litres = 0.60 OsM.
4 Osmosis May 2015 page 4 So the osmolarity of Red Bull is 0.60 OsM, whereas the normal osmolarity of plasma is OsM. What is the osmolarity of a glass of skim milk? (13g of lactose in 236 ml) Lactose is a kind of sugar and has a molecular weight of 342g per mole. 13g lactose * 1 mole/342g = moles. Lactose does not dissolve into smaller "pieces". Answer: moles / litres = 0.16 OsM Think about that next time you have a Red Bull for breakfast! It can take many solutes to make osmolarity On the last screen, we calculated the osmolarity of the sugar in Red Bull to be 0.60 OsM. That is not, however, the same thing as the osmolarity of Red Bull. The reason is that sugar is not the only thing dissolved in that can. For example, there are also 35mg of salt. To calculate the total osmolarity, we also need to take the salt into account (as well as a bunch of other minor players like color and flavor molecules, but we'll stick with the salt for now). In order to figure out how many moles of salt we have, we need to know the molecular weight. One mole of sodium weighs 23g, and one mole of chlorine weighs 35.4g. When the sodium and chlorine are combined in a single molecule, one mole of salt weighs = 58.4g. So, converting from mg to moles: 35.4 mg * 1 g / 1000 mg * 1 mole / 58.4g = moles And we know that salt dissociates into one sodium ion and one chlorine ion (Na + and Cl - ), making TWO moles of ions. Or, undissolved moles --> moles of dissolved ions And, remembering that there are 0.25L the osmolarity associated with the salt is: osmoles / 0.25 liters = OsM To get the total osmolarity of the Red Bull, we add up the two osmolarities associated with each ingredient: total osmolarity = 0.60 OsM OsM = OsM. Obviously we could keep going down the ingredient list, but probably you've got the idea by now.
5 Osmosis May 2015 page 5 What is the osmolarity of seawater given the following: Solute g / L molecular weight Cl Na Mg S To convert grams to moles, multiply grams by 1 / molecular weight. Add all the molarity from first hint. Answer: 19 * 1/ * 1/ *1/ * 1/32 = = 1.08 OsM = 1080 mosm Which way will water flow? Why does all this matter? Water has a tendency to move across a membrane from a lower osmolarity to a higher osmolarity. In other words, from the dilute side to the concentrated side. The outcome is that the dilute side loses water and becomes more concentrated, and the concentrated side gains water to become more dilute, so the two sides become more similar. In the figure osmolarity is 3 times higher on the right, but the red molecules are trapped over there. Instead, the water can move, so it goes TOWARDS the highosmolarity side. That makes the left side more concentrated and the right side less concentrated In the figure below, there are 3 solutions (0.2M, 0.5M, and 0.7M) separated by membranes. Your job is to figure out which direction(s) water is moving:
6 Osmosis May 2015 page 6 The first thing to be careful of is to calculate the osmolarities before you start. Since MgCl 2 dissociates into 3 pieces, its osmolarity is triple, i.e., 0.6 OsM. Sugar does not dissociate, so it stays at 0.5 OsM. The third solution contains both sets of solutes, so its osmolarity is = 1.1 OsM. Then remember that water goes from low to high osmolarity, from weak to concentrated solutions. You can think of it as water "trying" to equalise the concentrations. So water flows from the middle compartment to either side, since both have higher osmolarity. and how fast? We determined which way water flows, but shouldn't it go faster to the right (toward the very concentrated solution) than to the left (a solution that is barely different in concentration)? In fact this is true, and it is directly reflected in the equation for the 2-compartment model: Continuous Discrete
7 Osmosis May 2015 page 7 We don't know either the permeability (P water) or the area of the membrane (A). But this doesn't really matter if we just want to know how the rates of flow compare. We do know how the concentration differences compare (C right - C left ). Between the first and second compartments, the difference in osmolarity is only 0.1 OsM. Between the second and third compartments, the difference in osmolarity is 0.6 OsM. So water should flow 6 times faster to the right than to the left. Of course this state of affairs only lasts for a split second. As soon as water starts to flow, the concentration differences change, and the relative rates of flow change until equilibrium is reached. If you are using the continuous equation, the adjustments will be made immediately. If you are using the discrete version, the adjustment will only be made after Δt time has passed. What else can the equations tell us? Continuous Discrete Here are a few last questions on interpreting the equations: What will the rate of water flow be if permeability is zero? Permeability is P water. Zero times anything is zero. Answer: No matter how high the difference in osmolarity, no osmosis will occur if the permeability is 0. For example, osmosis will not work through a brick wall
8 Osmosis May 2015 page 8 When will the rate of water flow be the highest? the product of three quantities is maximized when each quantity is maximized Answer: Assuming that area (A) is constant, you need the permeability (P) to be as high as possible. You also need the difference in osmolarity (Cright Cleft) to be as high as possible for example, one side pure water with OsM = 0 and the other side with as high osmolarity as possible. What happens if the osmolarity of the two solutions is the same? If the osmolarities are the same, what about the concentrations? Answer: If the two sides have the same concentrations, then (Cright Cleft) = 0 and NO net osmosis will take place, no matter how large or leaky your membrane is. None of these was really rocket science. The point is that the equation for rate of water flow is simply a alisation of what our intuition tells us should happen. The equation puts intuition into mathematical language! Where does it all end? So, a quick recap. We can figure out which way water is flowing. Also, using the iteration technique at the end of the section on diffusion through a membrane, we can figure out how much water is flowing at each time step (Δ t). However, there is still one more property of the system that we can figure out -- what happens at the end of that slower and slower process -- in other words, what are the equilibrium concentrations, and how much water is on either side of the membrane. We can do this without endlessly pushing buttons on a calculator. Let's use sugar as an example. Say we started with 0.2 moles of sugar in 1 litre of water on one side, and 0.8 moles of sugar in 1 litre of water on the other side and the two compartments are the same size. The water is free to move back and forth across the membrane, but the sugar's not going anywhere, and no extra sugar or water can move in or out of the 2-compartment system. So at the end of the day,
9 Osmosis May 2015 page 9 there will still be 0.2 moles of sugar on one side and 0.8 on the other side, and there will still be 2 litres of water, and the concentrations on either side will be equal, BUT the water will not be equally distributed. How can you find the final water levels using intuition? The final water level on the 0.8 mole side must be 4 times higher than on the 0.2 mole side. The total water level has to add up to 2 litres Answer: 1.6 L+ 0.4 L fulfills these conditions How can you find the final water levels using al equations? the final molarities must be equal, so 0.2/W 1 = 0.8/W 2, or 0.2 W 2 = 0.8 W 1,or W 2 = 4 W 1 The total amount of water is 2 L so W 1 + W 2 = 2 Answer: Substitute 4 W1 for W2, so W1 + 4 W1 = 2 5 W1 = 2 W1 = 0.4 L W2 = = 1.6 L Drink your water Let s apply what we have learnt above to the situation where a person drinks 1.5 litres of water. The osmolarity of bodily fluids is approximately 0.3 OsM (or 300 mosm). The total fluid volume of a normal person is about 45 litres, of which 30 litres is inside cells (Intracellular volume) and 15 litres is outside the cells (Extracellular volume which includes plasma). We can use these figures to determine the total number of solutes present in each compartment. Osmolarity (osmoles/l H 2O) x volume (Litres) = total solutes (osmoles) Below is a table of the osmolarities associated with the intracellular and extracellular compartments before taking the drink:
10 Osmosis May 2015 page 10 Before drinking Volume osmolarity Total solutes in compartment Total body 45 litres 0.3 OsM 0.3 x 45 = 13.5 osmoles intracellular 30 litres 0.3 OsM 0.3 x 30 = 9.0 osmoles extracellular 15 litres 0.3 OsM 0.3 x 15 = 4.5 osmoles After drinking 1.5 litre of water, the water will be absorbed by the gastrointestinal tract and will enter the plasma and extracellular fluid and will equilibrate with the intracellular compartment. As the solutes that are present inside the cell cannot leave the cell, equilibration will only involve the movement or diffusion of water (Osmosis). Let s look at what happens to the whole body first: After drinking Total solutes volume osmolarity Total body 13.5 osmoles 46.5 litres = 0.29 The new osmolarity is 0.29 and not 0.3OsM. Water will move to equilibrate the intra- and extra- cellular compartments to this NEW osmolarity. To determine the movement of water we work back from the osmolarity. After drinking Total solutes volume osmolarity intracellular 9.0 osmoles = 31.0 litres 0.29 extracellular 4.5 osmoles = 15.5 litres 0.29 So we can see that because the solutes do not move out of the cells the 1.5 litres of water will distribute so that 1 litre stays in the extracellular space and 0.5 litre diffuses across the cell membrane to the intracelullar compartment. Sharks and alligators and goldfish, oh my! So what, if anything, does all of this tell us about life-as-we-know-it? Here is one small example. Below is a table of osmolarities associated with environments and organisms: seawater: 1 OsM shark: OsM freshwater: OsM goldfish: OsM alligator: OsM mammal: OsM So a shark is a little "saltier" than its environment, and a freshwater fish is a lot saltier (inside) than the water around it. Why? Sharks do not generally "drink". Instead they get freshwater from the seawater around them. Because their insides are a little saltier than the water around them, freshwater flows INTO their cells from their surroundings. The opposite of what would happen to you or me, who would get pickled if we spent too long in the ocean.
11 Osmosis May 2015 page 11 On the other hand, mammals are a lot saltier than their environment. But when you drink water, your body fluids become more diluted, so water flows INTO your cells. That's why everyone wants you to drink 8-10 glasses a day of water. And by the same token, if you put a shark into freshwater, the results would NOT be pretty. (The difference in OsM between shark and freshwater is 1.074, vs for goldfish and freshwater.) Summary Osmosis is a special case of diffusion in which only water moves across a membrane from dilute side (higher water concentration) to concentrated side (lower water concentration). For osmosis, the measure of concentration changes from molarity to osmolarity. Osmolarity is defined by osmoles of solute per litre of solution e.g. 1 mol/l solution of KCl has molarity (M) of 1, but its osmolarity (OsM) is 2 because KCl creates 2 osmoles by dissociating into K + and Cl - in water. However, a 1M solution of glucose has an osmolarity of 1 OsM because glucose is not an ionic compound and it does not dissociate into smaller pieces in the presence of water. The osmolarity of a solution is given by adding the osmoles of all the solutes present in the solution. During osmosis, the rate of flow of water across the membrane depends on number of factors including permeability of membrane to water, the area of membrane and the concentration difference between the two sides. These factors are summed up in the equation: Flow = P water * A * ΔC (ΔC = C right C left) So, if ΔC is doubled flow of water would double and so on. Eventually equilibrium is reached when concentrations on both sides are the same however, under equilibrium conditions, the amount of water on both sides will not be equal. Learning Outcomes You should now be able to: Determine the osmolarity of a solution. Determine which way water will flow through a selectively permeable membrane, and find the rate of flow of water using a two-compartment model.
Lab 4: Osmosis and Diffusion
 Page 4.1 Lab 4: Osmosis and Diffusion Cells need to obtain water and other particles from the fluids that surround them. Water and other particles also move out of cells. Osmosis (for water) and diffusion
Page 4.1 Lab 4: Osmosis and Diffusion Cells need to obtain water and other particles from the fluids that surround them. Water and other particles also move out of cells. Osmosis (for water) and diffusion
How would you prepare 455 grams of an aqueous solution that is 6.50% sodium sulfate by mass?
 62 How would you prepare 455 grams of an aqueous solution that is 6.50% sodium sulfate by mass? Start a concentration calculation by writing the definition of the unit(s) you're using! We know everything
62 How would you prepare 455 grams of an aqueous solution that is 6.50% sodium sulfate by mass? Start a concentration calculation by writing the definition of the unit(s) you're using! We know everything
Osmoregulation and Osmotic Balance
 OpenStax-CNX module: m44808 1 Osmoregulation and Osmotic Balance OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this
OpenStax-CNX module: m44808 1 Osmoregulation and Osmotic Balance OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this
Membrane Transport II (Osmosis) Linda S. Costanzo, Ph.D.
 Membrane Transport II (Osmosis) Linda S. Costanzo, Ph.D. OBJECTIVES: 1. Be able to define and calculate osmolarity 2. Describe osmosis across a semipermeable membrane and the volume changes that will occur
Membrane Transport II (Osmosis) Linda S. Costanzo, Ph.D. OBJECTIVES: 1. Be able to define and calculate osmolarity 2. Describe osmosis across a semipermeable membrane and the volume changes that will occur
INVESTIGATION : Determining Osmolarity of Plant Tissue
 INVESTIGATION : Determining Osmolarity of Plant Tissue AP Biology This lab investigation has two main components. In the first component, you will learn about the osmolarity of plant tissues and the property
INVESTIGATION : Determining Osmolarity of Plant Tissue AP Biology This lab investigation has two main components. In the first component, you will learn about the osmolarity of plant tissues and the property
The table indicates how changing the variable listed alone will alter diffusion rate.
 Rate of Diffusion (flux) Concentration gradient substance x surface area of membrane x lipid solubility = Distance (thickness of membrane) x molecular weight Table 3-1: Factors Influencing the Rate of
Rate of Diffusion (flux) Concentration gradient substance x surface area of membrane x lipid solubility = Distance (thickness of membrane) x molecular weight Table 3-1: Factors Influencing the Rate of
Diffusion & Osmosis - Exercise 4
 Diffusion & Osmosis - Exercise 4 Objectives -Define: Solvent, Solute, and Solution -Define: Diffusion, Selectively permeable membrane, Osmosis, and Dialysis -Understand rule of thumb: Concentration will
Diffusion & Osmosis - Exercise 4 Objectives -Define: Solvent, Solute, and Solution -Define: Diffusion, Selectively permeable membrane, Osmosis, and Dialysis -Understand rule of thumb: Concentration will
LAB 4: OSMOSIS AND DIFFUSION
 Page 4.1 LAB 4: OSMOSIS AND DIFFUSION Cells need to obtain water and other particles from the fluids that surround them. Water and other particles also move out of cells. Osmosis (for water) and diffusion
Page 4.1 LAB 4: OSMOSIS AND DIFFUSION Cells need to obtain water and other particles from the fluids that surround them. Water and other particles also move out of cells. Osmosis (for water) and diffusion
 Taking care of business Go to this page and enter room SJ123: http://tinyurl.com/physclicker Take 2 minutes to complete this survey: http://tinyurl.com/physdis Online quiz this weekend: Released Thursday
Taking care of business Go to this page and enter room SJ123: http://tinyurl.com/physclicker Take 2 minutes to complete this survey: http://tinyurl.com/physdis Online quiz this weekend: Released Thursday
WATER AND SOLUTE MOVEMENT THROUGH RED BLOOD CELLS
 WATER AND SOLUTE MOVEMENT THROUGH RED BLOOD CELLS Purpose This exercise is designed to demonstrate the properties of cellular membranes and the movement of water and solutes across them. In this lab, you
WATER AND SOLUTE MOVEMENT THROUGH RED BLOOD CELLS Purpose This exercise is designed to demonstrate the properties of cellular membranes and the movement of water and solutes across them. In this lab, you
Cell Membranes: Diffusion and Osmosis
 STO-112 Cell Membranes: Diffusion and Osmosis Part 1: Diffusion Diffusion is a process by which molecules move into or out of cells. To diffuse into or out of a cell, molecules must pass through the cell
STO-112 Cell Membranes: Diffusion and Osmosis Part 1: Diffusion Diffusion is a process by which molecules move into or out of cells. To diffuse into or out of a cell, molecules must pass through the cell
Biology. Membranes.
 1 Biology Membranes 2015 10 28 www.njctl.org 2 Vocabulary active transport carrier protein channel protein concentration gradient diffusion enzymatic activity facilitated diffusion fluid mosaic hypertonic
1 Biology Membranes 2015 10 28 www.njctl.org 2 Vocabulary active transport carrier protein channel protein concentration gradient diffusion enzymatic activity facilitated diffusion fluid mosaic hypertonic
Passive Transport. Does not expend cellular energy for the movement to take place. Ex-rolling down a hill
 Passive Transport Fluid Mosaic Model Passive Transport Does not expend cellular energy for the movement to take place Ex-rolling down a hill Parts of a Solution Solute: what gets dissolved Solvent: What
Passive Transport Fluid Mosaic Model Passive Transport Does not expend cellular energy for the movement to take place Ex-rolling down a hill Parts of a Solution Solute: what gets dissolved Solvent: What
Enzymes Help Us Digest Food 1
 Enzymes Help Us Digest Food 1 Introduction to Sugars and Enzymes The food you eat contains many different types of molecules, including two types of sugar molecules: monosaccharides and disaccharides.
Enzymes Help Us Digest Food 1 Introduction to Sugars and Enzymes The food you eat contains many different types of molecules, including two types of sugar molecules: monosaccharides and disaccharides.
LAB 04 Diffusion and Osmosis
 LAB 04 Diffusion and Osmosis Objectives: Describe the physical mechanisms of diffusion and osmosis. Understand the relationship between surface area and rate of diffusion. Describe how molar concentration
LAB 04 Diffusion and Osmosis Objectives: Describe the physical mechanisms of diffusion and osmosis. Understand the relationship between surface area and rate of diffusion. Describe how molar concentration
BIOLOGY 1101 LAB 1: OSMOSIS & DIFFUSION. READING: Please read pages & in your text prior to lab.
 BIOLOGY 1101 LAB 1: OSMOSIS & DIFFUSION READING: Please read pages 27-31 & 83-86 in your text prior to lab. INTRODUCTION: All living things depend on water. A water molecule is made up of an oxygen atom
BIOLOGY 1101 LAB 1: OSMOSIS & DIFFUSION READING: Please read pages 27-31 & 83-86 in your text prior to lab. INTRODUCTION: All living things depend on water. A water molecule is made up of an oxygen atom
To understand osmosis, we must focus on the behavior of the solvent, not the solute.
 GCC CHM 130LL Osmosis and Dialysis Purpose: The purpose of this experiment is to observe the closely related phenomena of osmosis and diffusion as it relates to dialysis. It is hoped that you will be able
GCC CHM 130LL Osmosis and Dialysis Purpose: The purpose of this experiment is to observe the closely related phenomena of osmosis and diffusion as it relates to dialysis. It is hoped that you will be able
Chapter 4 Reading Guide
 Chapter 4 Reading Guide 1. How many covalent bonds does carbon (C) form? 2. What is the chemical formula for glucose? 3. List the major dietary monosaccharides and disaccharides. What are the components
Chapter 4 Reading Guide 1. How many covalent bonds does carbon (C) form? 2. What is the chemical formula for glucose? 3. List the major dietary monosaccharides and disaccharides. What are the components
Membrane Transport. Anatomy 36 Unit 1
 Membrane Transport Anatomy 36 Unit 1 Membrane Transport Cell membranes are selectively permeable Some solutes can freely diffuse across the membrane Some solutes have to be selectively moved across the
Membrane Transport Anatomy 36 Unit 1 Membrane Transport Cell membranes are selectively permeable Some solutes can freely diffuse across the membrane Some solutes have to be selectively moved across the
Ch 3 Membrane Transports
 Ch 3 Membrane Transports what's so dynamic about cell membranes? living things get nutrients and energy from the envrionment this is true of the entire organism and each cell this requires transport in/out
Ch 3 Membrane Transports what's so dynamic about cell membranes? living things get nutrients and energy from the envrionment this is true of the entire organism and each cell this requires transport in/out
Applications of Freezing Point Osmometry
 Applications of Freezing Point Osmometry Table of Contents Chapter 1 Introduction and Basic Principles 1 Chapter 2 Biological Applications 3 2.1 Range of, and reason for, abnormal serum values 5 2.2 Osmolality
Applications of Freezing Point Osmometry Table of Contents Chapter 1 Introduction and Basic Principles 1 Chapter 2 Biological Applications 3 2.1 Range of, and reason for, abnormal serum values 5 2.2 Osmolality
Diffusion and Osmosis
 Diffusion and Osmosis OBJECTIVES: 1. To explore how different molecules move by diffusion and osmosis through semi-permeable membranes. 2. To understand how concentration affects the movement of substances
Diffusion and Osmosis OBJECTIVES: 1. To explore how different molecules move by diffusion and osmosis through semi-permeable membranes. 2. To understand how concentration affects the movement of substances
Explain how the structure of the plasma membrane allows material to move through. Explain the processes of Passive Transport and Active Transport.
 Objectives: Explain how the structure of the plasma membrane allows material to move through. Explain the processes of Passive Transport and Active Transport. Predict whether active or passive transport
Objectives: Explain how the structure of the plasma membrane allows material to move through. Explain the processes of Passive Transport and Active Transport. Predict whether active or passive transport
Cell Processes. Chapter 3. Learning Target 3/15/16. l I can. l 1)Explain the difference between diffusion and osmosis.
 Chapter 3 Cell Processes Learning Target l I can. l 1)Explain the difference between diffusion and osmosis. l 2)Predict the movement of particles into and out of a cell. 1 Section 3.2 MOVING CELLULAR MATERIAL
Chapter 3 Cell Processes Learning Target l I can. l 1)Explain the difference between diffusion and osmosis. l 2)Predict the movement of particles into and out of a cell. 1 Section 3.2 MOVING CELLULAR MATERIAL
Keywords (reading p ) Ammonia toxicity Urea Uric acid Osmoconformer Osmoregulator Passive transport Facilitated diffusion Active transport
 Controlling the Internal Environment II: Salt and water balance Keywords (reading p. 936-949) Ammonia toxicity Urea Uric acid Osmoconformer Osmoregulator Passive transport Facilitated diffusion Active
Controlling the Internal Environment II: Salt and water balance Keywords (reading p. 936-949) Ammonia toxicity Urea Uric acid Osmoconformer Osmoregulator Passive transport Facilitated diffusion Active
Transport through membranes
 Transport through membranes Membrane transport refers to solute and solvent transfer across both cell membranes, epithelial and capillary membranes. Biological membranes are composed of phospholipids stabilised
Transport through membranes Membrane transport refers to solute and solvent transfer across both cell membranes, epithelial and capillary membranes. Biological membranes are composed of phospholipids stabilised
CHAPTER. Movement Across Plasma Membrane. Chapter 6 Outline. Diffusion Osmosis. Membrane Potential Cell Signaling
 CHAPTER 6 Interaction Between Cells and the Extracellular Environment Chapter 6 Outline Extracellular Environment Diffusion Osmosis Carrier-Mediated Carrier Mediated Transport Membrane Potential Cell Signaling
CHAPTER 6 Interaction Between Cells and the Extracellular Environment Chapter 6 Outline Extracellular Environment Diffusion Osmosis Carrier-Mediated Carrier Mediated Transport Membrane Potential Cell Signaling
AP Biology Lab 1c Water Potential
 Page 1 of 9 AP Biology Lab 1c Water Potential In this part of the exercise you will use potato cores placed in different molar concentrations of sucrose in order to determine the water potential of potato
Page 1 of 9 AP Biology Lab 1c Water Potential In this part of the exercise you will use potato cores placed in different molar concentrations of sucrose in order to determine the water potential of potato
The cell membrane can be compared to a sieve/colander. When would you use a colander? What does a cell membrane and a colander have in common?
 The cell membrane can be compared to a sieve/colander. When would you use a colander? What does a cell membrane and a colander have in common? All cells are surrounded by a cell membrane Cell membranes
The cell membrane can be compared to a sieve/colander. When would you use a colander? What does a cell membrane and a colander have in common? All cells are surrounded by a cell membrane Cell membranes
What kind of things must pass into and out of cells?? Be careful not to go too fast.
 1. A membrane s molecular organization results in selective permeability What kind of things must pass into and out of cells?? Be careful not to go too fast. Permeability of a molecule through a membrane
1. A membrane s molecular organization results in selective permeability What kind of things must pass into and out of cells?? Be careful not to go too fast. Permeability of a molecule through a membrane
In the Name of God, the Most Merciful, the Most Compassionate. Movement of substances across the plasma membrane
 *Quick Revision: In the Name of God, the Most Merciful, the Most Compassionate Movement of substances across the plasma membrane Passive transport (doesn't require metabolic energy) Active transport (requires
*Quick Revision: In the Name of God, the Most Merciful, the Most Compassionate Movement of substances across the plasma membrane Passive transport (doesn't require metabolic energy) Active transport (requires
Razi Kittaneh & Tamer Barakat. Bayan Abusheikha. Faisal Mohammed
 3 Razi Kittaneh & Tamer Barakat Bayan Abusheikha Faisal Mohammed Transport and Osmolality In the last lecture we briefly talked about Transport, there are 2 types of transport: 1) Passive Transport 2)
3 Razi Kittaneh & Tamer Barakat Bayan Abusheikha Faisal Mohammed Transport and Osmolality In the last lecture we briefly talked about Transport, there are 2 types of transport: 1) Passive Transport 2)
What kind of things must pass into and out of cells?? Be careful not to go too fast.
 1. A membrane s molecular organization results in selective permeability What kind of things must pass into and out of cells?? Be careful not to go too fast. Permeability of a molecule through a membrane
1. A membrane s molecular organization results in selective permeability What kind of things must pass into and out of cells?? Be careful not to go too fast. Permeability of a molecule through a membrane
Diffusion, Osmosis and Active Transport
 Diffusion, Osmosis and Active Transport Part A: Diffusion A living cell interacts constantly with the environmental medium that surrounds it. The plasma membrane surrounding a cell is a living, selectively
Diffusion, Osmosis and Active Transport Part A: Diffusion A living cell interacts constantly with the environmental medium that surrounds it. The plasma membrane surrounding a cell is a living, selectively
Transport Across the Cell Membrane 11/5/07
 11/5/07 "The difference between the internal and external chemical composition of a cell represents a degree of order, that can be maintained only by a barrier to free movement into and out of the cell.
11/5/07 "The difference between the internal and external chemical composition of a cell represents a degree of order, that can be maintained only by a barrier to free movement into and out of the cell.
Big. Cellular Processes: Idea. Energy and Communication DIFFUSION AND OSMOSIS. What causes my plants to wilt if I forget to water them?
 Big Cellular Processes: Idea 2 Energy and Communication INVESTIGATION 4 DIFFUSION AND OSMOSIS What causes my plants to wilt if I forget to water them? BACKGROUND Cells must move materials through membranes
Big Cellular Processes: Idea 2 Energy and Communication INVESTIGATION 4 DIFFUSION AND OSMOSIS What causes my plants to wilt if I forget to water them? BACKGROUND Cells must move materials through membranes
Concept 7.1: Cellular membranes are fluid mosaics of lipids and proteins
 Concept 7.1: Cellular membranes are fluid mosaics of lipids and proteins Lipids: Non-polar substances such as fat that contain C, H, O. Phospholipids: Lipid with phosphate group, very abundant in plasma
Concept 7.1: Cellular membranes are fluid mosaics of lipids and proteins Lipids: Non-polar substances such as fat that contain C, H, O. Phospholipids: Lipid with phosphate group, very abundant in plasma
Diffusion, Osmosis and Active Transport
 Diffusion, Osmosis and Active Transport 1 of 25 Boardworks Ltd 2012 2 of 25 Boardworks Ltd 2012 Substances spread out by diffusion 3 of 25 Boardworks Ltd 2012 Particles in solutions and in gases move around
Diffusion, Osmosis and Active Transport 1 of 25 Boardworks Ltd 2012 2 of 25 Boardworks Ltd 2012 Substances spread out by diffusion 3 of 25 Boardworks Ltd 2012 Particles in solutions and in gases move around
EH1008 Biomolecules. Inorganic & Organic Chemistry. Water. Lecture 2: Inorganic and organic chemistry.
 EH1008 Biomolecules Lecture 2: Inorganic and organic chemistry limian.zheng@ucc.ie 1 Inorganic & Organic Chemistry Inorganic Chemistry: generally, substances that do not contain carbon Inorganic molecules:
EH1008 Biomolecules Lecture 2: Inorganic and organic chemistry limian.zheng@ucc.ie 1 Inorganic & Organic Chemistry Inorganic Chemistry: generally, substances that do not contain carbon Inorganic molecules:
Name Date Block. Lactase Lab. (Adapted from Lactase Investigation Philadelphia Public Schools and Enzymes Help Us Digest Food from Bryn Mawr)
 Name Date Block Lactase Lab (Adapted from Lactase Investigation Philadelphia Public Schools and Enzymes Help Us Digest Food from Bryn Mawr) BACKGROUND INFORMATION: The food we eat contains many different
Name Date Block Lactase Lab (Adapted from Lactase Investigation Philadelphia Public Schools and Enzymes Help Us Digest Food from Bryn Mawr) BACKGROUND INFORMATION: The food we eat contains many different
MITOCW watch?v=by shzyi7q
 MITOCW watch?v=by shzyi7q The following content is provided under a Creative Commons license. Your support will help MIT OpenCourseWare continue to offer high quality educational resources for free. To
MITOCW watch?v=by shzyi7q The following content is provided under a Creative Commons license. Your support will help MIT OpenCourseWare continue to offer high quality educational resources for free. To
Experiment 6: STANDARDIZATION OF A BASE; MASS PERCENT OF AN ACID
 Experiment 6: STANDARDIZATION OF A BASE; MASS PERCENT OF AN ACID Introduction The reaction of an acid and a base to form a salt and water is known as neutralization. In this experiment; potassium acid
Experiment 6: STANDARDIZATION OF A BASE; MASS PERCENT OF AN ACID Introduction The reaction of an acid and a base to form a salt and water is known as neutralization. In this experiment; potassium acid
DIFFUSON AND OSMOSIS INTRODUCTION diffusion concentration gradient. net osmosis water potential active transport
 DIFFUSON AND OSMOSIS NAME DATE INTRODUCTION The life of a cell is dependent on efficiently moving material into and out of the cell across the cell membrane. Raw materials such as oxygen and sugars needed
DIFFUSON AND OSMOSIS NAME DATE INTRODUCTION The life of a cell is dependent on efficiently moving material into and out of the cell across the cell membrane. Raw materials such as oxygen and sugars needed
Unit 1 Matter & Energy for Life
 Unit 1 Matter & Energy for Life Chapter 2 Interaction of Cell Structure Biology 2201 Primary Membrane Function: Homeostasis Conditions in the cell must remain more or less constant under many different
Unit 1 Matter & Energy for Life Chapter 2 Interaction of Cell Structure Biology 2201 Primary Membrane Function: Homeostasis Conditions in the cell must remain more or less constant under many different
Body Fluid Compartments
 Yıldırım Beyazıt University Faculty of Medicine Department of Physiology Body Fluid Compartments Dr. Sinan Canan Body fluid balance 1 Body fluid compartments 2 Water distribution Tissue % Water Blood 83,0
Yıldırım Beyazıt University Faculty of Medicine Department of Physiology Body Fluid Compartments Dr. Sinan Canan Body fluid balance 1 Body fluid compartments 2 Water distribution Tissue % Water Blood 83,0
Osmoregulation_and_Excretion_p2.notebook June 01, 2017
 What water balance problems face organisms? Animals balance water gain and loss in one of two ways: 1. Osmoconformers are isotonic to their surroundings so they do not lose or gain water they usually live
What water balance problems face organisms? Animals balance water gain and loss in one of two ways: 1. Osmoconformers are isotonic to their surroundings so they do not lose or gain water they usually live
Carbohydrate Structure and Function Introduction
 Carbohydrate Structure and Function Introduction Transcript Hello and welcome to our Institute of Nutritional Endocrinology's presentation on Macronutrients. Today we are going to be talking about Carbohydrates,
Carbohydrate Structure and Function Introduction Transcript Hello and welcome to our Institute of Nutritional Endocrinology's presentation on Macronutrients. Today we are going to be talking about Carbohydrates,
Biochemical Concepts. Section 4.6 The Chemistry of Water. Pre-View 4.6. A Covalent Polar Molecule
 Biochemical Concepts Section 4.6 The Chemistry of Water Pre-View 4.6 Polar molecule a molecule that has a partial positive charge on one end and a partial negative charge on the other end Hydrogen bond
Biochemical Concepts Section 4.6 The Chemistry of Water Pre-View 4.6 Polar molecule a molecule that has a partial positive charge on one end and a partial negative charge on the other end Hydrogen bond
Osmosis Practice Problems. Good practice for test-taking strategy, too.
 Osmosis Practice Problems Good practice for test-taking strategy, too. #1 If you soak your hands in dishwater, you may notice that your skin absorbs water and swells into wrinkles. This is because your
Osmosis Practice Problems Good practice for test-taking strategy, too. #1 If you soak your hands in dishwater, you may notice that your skin absorbs water and swells into wrinkles. This is because your
Interactions Between Cells and the Extracellular Environment
 Chapter 6 Interactions Between Cells and the Extracellular Environment Et Extracellular lll environment Includes all parts of the body outside of cells Cells receive nourishment Cells release waste Cells
Chapter 6 Interactions Between Cells and the Extracellular Environment Et Extracellular lll environment Includes all parts of the body outside of cells Cells receive nourishment Cells release waste Cells
Cell Transport. Movement of molecules
 Cell Transport Movement of molecules TEKS Students will investigate and explain cellular processes, including homeostasis and transport of molecules Homeostasis The maintaining of a stable body system
Cell Transport Movement of molecules TEKS Students will investigate and explain cellular processes, including homeostasis and transport of molecules Homeostasis The maintaining of a stable body system
Unit 1 Matter & Energy for Life
 Unit 1 Matter & Energy for Life Chapter 2 Interaction of Cell Structures Biology 2201 Primary Membrane Function: Homeostasis Section 2.2 Conditions in the cell must remain more or less constant under many
Unit 1 Matter & Energy for Life Chapter 2 Interaction of Cell Structures Biology 2201 Primary Membrane Function: Homeostasis Section 2.2 Conditions in the cell must remain more or less constant under many
Warm Up 12/06/2018. In a Solution of Salt Water, which substance acts as the Solvent and which substance acts as the Solute?
 Warm Up 12/06/2018 In a Solution of Salt Water, which substance acts as the Solvent and which substance acts as the Solute? Cell Membrane and Cell Transport How Nutrients move in and Wastes Move Out of
Warm Up 12/06/2018 In a Solution of Salt Water, which substance acts as the Solvent and which substance acts as the Solute? Cell Membrane and Cell Transport How Nutrients move in and Wastes Move Out of
Measuring Osmotic Potential
 Measuring Osmotic Potential INTRODUCTION All cells require essential materials to ensure their survival. Chemical, physical, and biological processes are used to move these materials inside of cells. Similar
Measuring Osmotic Potential INTRODUCTION All cells require essential materials to ensure their survival. Chemical, physical, and biological processes are used to move these materials inside of cells. Similar
FIGURE A. The phosphate end of the molecule is polar (charged) and hydrophilic (attracted to water).
 PLASMA MEMBRANE 1. The plasma membrane is the outermost part of a cell. 2. The main component of the plasma membrane is phospholipids. FIGURE 2.18 A. The phosphate end of the molecule is polar (charged)
PLASMA MEMBRANE 1. The plasma membrane is the outermost part of a cell. 2. The main component of the plasma membrane is phospholipids. FIGURE 2.18 A. The phosphate end of the molecule is polar (charged)
ROADMAP FREEDOM FROM STUTTERING. Copyright 2017 Life Quality, Inc. All Rights Reserved
 ROADMAP TO FREEDOM FROM STUTTERING Copyright 2017 Life Quality, Inc. All Rights Reserved Contents Why freedom from stuttering 3 What is it?.. 4 The path.. 5 Three massive mistakes on the way...6 Automated
ROADMAP TO FREEDOM FROM STUTTERING Copyright 2017 Life Quality, Inc. All Rights Reserved Contents Why freedom from stuttering 3 What is it?.. 4 The path.. 5 Three massive mistakes on the way...6 Automated
Membrane Dynamics. The body is mostly water. Figure 5.1a Body Fluid Compartments. Figure 5.1c Body Fluid Compartments 2016 Pearson Education, Inc.
 Figure 5.1a Body Fluid Compartments Membrane Dynamics The body is mostly water Figure 5.1c Body Fluid Compartments 1 Osmotic Equilibrium Chemical Disequilibrium ØWater moves freely between the ECF and
Figure 5.1a Body Fluid Compartments Membrane Dynamics The body is mostly water Figure 5.1c Body Fluid Compartments 1 Osmotic Equilibrium Chemical Disequilibrium ØWater moves freely between the ECF and
Passive and Active transport across a cell membrane REVIEW MEMBRANE TRANSPORT
 Passive and Active transport across a cell membrane REVIEW MEMBRANE TRANSPORT Cell (plasma) membrane Thin, flexible barrier Membranes also organize the interior of a cell. Cell organelles are defined by
Passive and Active transport across a cell membrane REVIEW MEMBRANE TRANSPORT Cell (plasma) membrane Thin, flexible barrier Membranes also organize the interior of a cell. Cell organelles are defined by
Cell Membranes. Q: What components of the cell membrane are in a mosaic pattern?
 Cell Membranes The cell / plasma membrane is. Selective in that it allows things in and some things out of the cell. Recall that phospholipids have hydrophobic and hydrophilic. The term to describe this
Cell Membranes The cell / plasma membrane is. Selective in that it allows things in and some things out of the cell. Recall that phospholipids have hydrophobic and hydrophilic. The term to describe this
Chapter 4 Cell Membrane Transport
 Chapter 4 Cell Membrane Transport Plasma Membrane Review o Functions Separate ICF / ECF Allow exchange of materials between ICF / ECF such as obtaining O2 and nutrients and getting rid of waste products
Chapter 4 Cell Membrane Transport Plasma Membrane Review o Functions Separate ICF / ECF Allow exchange of materials between ICF / ECF such as obtaining O2 and nutrients and getting rid of waste products
Macromolecule modeling lab
 Macromolecule modeling lab Goal: Use ball and stick models to build some of the macromolecules that make up cells also see how complex and large these macromolecules can be Directions to read before starting:
Macromolecule modeling lab Goal: Use ball and stick models to build some of the macromolecules that make up cells also see how complex and large these macromolecules can be Directions to read before starting:
= only some molecules can get in or out of the cell. allow substances (other than lipids) in and out
 Name: Cell Membrane and Cell Transport Notes I. Cell Membrane (cells need an inside and outside) a. separate cell from its environment b. cell membrane is the boundary c. cell membrane controls what gets
Name: Cell Membrane and Cell Transport Notes I. Cell Membrane (cells need an inside and outside) a. separate cell from its environment b. cell membrane is the boundary c. cell membrane controls what gets
A molecule that can pass though the cell membrane.
 Cell Membrane All cells and most organelles are surrounded by a protective barrier known as the cell membrane, aka the plasma membrane. Section of a cell membrane It is mostly made of phospholipids (fats).
Cell Membrane All cells and most organelles are surrounded by a protective barrier known as the cell membrane, aka the plasma membrane. Section of a cell membrane It is mostly made of phospholipids (fats).
Cellular Physiology. Body Fluids: 1) Water: (universal solvent) Body water varies based on of age, sex, mass, and body composition
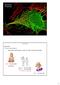 Membrane Physiology Body Fluids: 1) Water: (universal solvent) Body water varies based on of age, sex, mass, and body composition H 2 O ~ 73% body weight Low body fat; Low bone mass H 2 O ( ) ~ 60% body
Membrane Physiology Body Fluids: 1) Water: (universal solvent) Body water varies based on of age, sex, mass, and body composition H 2 O ~ 73% body weight Low body fat; Low bone mass H 2 O ( ) ~ 60% body
Cellular Transport Worksheet
 Cellular Transport Worksheet Name Section A: Cell Membrane Structure 1. Label the cell membrane diagram. You ll need to draw lines to some of the structures. **Draw cholesterol molecules in the membrane.**
Cellular Transport Worksheet Name Section A: Cell Membrane Structure 1. Label the cell membrane diagram. You ll need to draw lines to some of the structures. **Draw cholesterol molecules in the membrane.**
Ch. 7 Diffusion, Osmosis, and Movement across a Membrane
 Ch. 7 Diffusion, Osmosis, and Movement across a Membrane Diffusion Spontaneous movement of particles from an area of high concentration to an area of low concentration Does not require energy (exergonic)
Ch. 7 Diffusion, Osmosis, and Movement across a Membrane Diffusion Spontaneous movement of particles from an area of high concentration to an area of low concentration Does not require energy (exergonic)
Biology. Slide 1 / 74. Slide 2 / 74. Slide 3 / 74. Membranes. Vocabulary
 Slide 1 / 74 Slide 2 / 74 iology Membranes 2015-10-28 www.njctl.org Vocabulary Slide 3 / 74 active transport carrier protein channel protein concentration gradient diffusion enzymatic activity facilitated
Slide 1 / 74 Slide 2 / 74 iology Membranes 2015-10-28 www.njctl.org Vocabulary Slide 3 / 74 active transport carrier protein channel protein concentration gradient diffusion enzymatic activity facilitated
Food labels made easy
 Food labels made easy 1 Food labels made easy Healthy eating is important for everyone, whether you ve got diabetes or not. That means eating more wholegrains, beans, peas, lentils, dhal, nuts, fish, fruit
Food labels made easy 1 Food labels made easy Healthy eating is important for everyone, whether you ve got diabetes or not. That means eating more wholegrains, beans, peas, lentils, dhal, nuts, fish, fruit
LESSON Exploring: Salt in Our Lives Introduce how salt affects living and non-living things.
 LESSON Exploring: Salt in Our Lives Introduce how salt affects living and non-living things. What We Are Hoping For: Learning Goals Abiotic and Biotic Factors Water Human Impact o A, B Learning Goals:
LESSON Exploring: Salt in Our Lives Introduce how salt affects living and non-living things. What We Are Hoping For: Learning Goals Abiotic and Biotic Factors Water Human Impact o A, B Learning Goals:
Electrolytes Solution
 Electrolytes Solution Substances that are not dissociated in solution are called nonelectrolytes, and those with varying degrees of dissociation are called electrolytes. Urea and dextrose are examples
Electrolytes Solution Substances that are not dissociated in solution are called nonelectrolytes, and those with varying degrees of dissociation are called electrolytes. Urea and dextrose are examples
SAFETY GOGGLES MUST BE WORN AT ALL TIMES!
 SUGAR ME UP, DUDE! PURPOSE: To determine the density of five sugar reference solutions To determine the density of your two beverages, using the references as a guide To graph density versus percent sucrose
SUGAR ME UP, DUDE! PURPOSE: To determine the density of five sugar reference solutions To determine the density of your two beverages, using the references as a guide To graph density versus percent sucrose
1.14. Passive Transport
 Passive Transport 1.14 Simple Diffusion Cell s are selectively permeable only certain substances are able to pass through them. As mentioned in section 1.2, cell s are largely composed of a phospholipid
Passive Transport 1.14 Simple Diffusion Cell s are selectively permeable only certain substances are able to pass through them. As mentioned in section 1.2, cell s are largely composed of a phospholipid
Section 4 Decision-making
 Decision-making : Decision-making Summary Conversations about treatments Participants were asked to describe the conversation that they had with the clinician about treatment at diagnosis. The most common
Decision-making : Decision-making Summary Conversations about treatments Participants were asked to describe the conversation that they had with the clinician about treatment at diagnosis. The most common
Each cell has its own border, which separates the cell from its surroundings and also determines what comes in and what goes out.
 7.3 Cell Transport Wednesday, December 26, 2012 10:02 AM Vocabulary: Diffusion: process in which cells become specialized in structure and function Facilitated diffusion: process of diffusion in which
7.3 Cell Transport Wednesday, December 26, 2012 10:02 AM Vocabulary: Diffusion: process in which cells become specialized in structure and function Facilitated diffusion: process of diffusion in which
Making An Exercise Plan
 Making An Exercise Plan The purpose of an Exercise Rx (prescription) is to provide a guide for your workouts that 1. is specifically designed to help you meet your fitness goals, 2. is controlled by how
Making An Exercise Plan The purpose of an Exercise Rx (prescription) is to provide a guide for your workouts that 1. is specifically designed to help you meet your fitness goals, 2. is controlled by how
Diffusion, osmosis, transport mechanisms 43
 Diffusion, osmosis, transport mechanisms 43 DIFFUSION, OSMOSIS AND TRANSPORT MECHANISMS The cell membrane is a biological membrane that separates the interior of all cells from the outside environment
Diffusion, osmosis, transport mechanisms 43 DIFFUSION, OSMOSIS AND TRANSPORT MECHANISMS The cell membrane is a biological membrane that separates the interior of all cells from the outside environment
BIOLOGY 12 - Cell Membrane and Cell Wall Function: Chapter Notes
 BIOLOGY 12 - Cell Membrane and Cell Wall Function: Chapter Notes The cell membrane is the gateway into the cell, and must allow needed things such as nutrients into the cell without letting them escape.
BIOLOGY 12 - Cell Membrane and Cell Wall Function: Chapter Notes The cell membrane is the gateway into the cell, and must allow needed things such as nutrients into the cell without letting them escape.
Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic
 Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic compounds. What are inorganic molecules? Molecules that CANNOT
Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic compounds. What are inorganic molecules? Molecules that CANNOT
MITOCW watch?v=nx76xs_4fre
 MITOCW watch?v=nx76xs_4fre The following content is provided under a Creative Commons license. Your support will help MIT OpenCourseWare continue to offer high quality educational resources for free. To
MITOCW watch?v=nx76xs_4fre The following content is provided under a Creative Commons license. Your support will help MIT OpenCourseWare continue to offer high quality educational resources for free. To
FDA/CFSAN: Guidance on How to Understand a...e the Nutrition Facts Panel on Food Labels
 U.S. Food and Drug Administration Center for Food Safety and Applied Nutrition June 2000 Guidance on How to Understand and Use the Nutrition Facts Panel on Food Labels People look at food labels for different
U.S. Food and Drug Administration Center for Food Safety and Applied Nutrition June 2000 Guidance on How to Understand and Use the Nutrition Facts Panel on Food Labels People look at food labels for different
Exemplar for Internal Achievement Standard. Biology Level 3
 Exemplar for Internal Achievement Standard Biology Level 3 This exemplar supports assessment against: Achievement Standard 91604 Demonstrate understanding of how an animal maintains a stable internal environment
Exemplar for Internal Achievement Standard Biology Level 3 This exemplar supports assessment against: Achievement Standard 91604 Demonstrate understanding of how an animal maintains a stable internal environment
BIOL 305L Spring 2019 Laboratory Six
 Please print Full name clearly: BIOL 305L Spring 2019 Laboratory Six Osmosis in potato and carrot samples Introduction Osmosis is the movement of water molecules through a selectively permeable membrane
Please print Full name clearly: BIOL 305L Spring 2019 Laboratory Six Osmosis in potato and carrot samples Introduction Osmosis is the movement of water molecules through a selectively permeable membrane
MS Learn Online Feature Presentation MS and Your Emotions, part two Deborah Miller, PhD. Tracey>> Welcome to MS Learn Online, I m Tracey Kimball.
 Page 1 MS Learn Online Feature Presentation MS and Your Emotions, part two Deborah Miller, PhD Tracey>> Welcome to MS Learn Online, I m Tracey Kimball. Tom>> and I m Tom Kimball. In the first installment
Page 1 MS Learn Online Feature Presentation MS and Your Emotions, part two Deborah Miller, PhD Tracey>> Welcome to MS Learn Online, I m Tracey Kimball. Tom>> and I m Tom Kimball. In the first installment
Cells are the smallest units of life CH 3
 Cells are the smallest units of life CH 3 The Cell Theory 1. All living things are composed of one or more cells. 2. Cells are the basic units of structure and function. 3. Cells are produced only from
Cells are the smallest units of life CH 3 The Cell Theory 1. All living things are composed of one or more cells. 2. Cells are the basic units of structure and function. 3. Cells are produced only from
Hockey Nutrition Tips
 Hockey Nutrition Tips 6 Classes of Nutrients Essential for Top Performance 1. Carbohydrates 2. Fat 3. Protein 4. Vitamins 5. Minerals 6. Water Carbohydrates: are a source of energy that can be either simple
Hockey Nutrition Tips 6 Classes of Nutrients Essential for Top Performance 1. Carbohydrates 2. Fat 3. Protein 4. Vitamins 5. Minerals 6. Water Carbohydrates: are a source of energy that can be either simple
Biology Cell Unit Homework Packet #3
 Biology Cell Unit Homework Packet #3 Name DUE: Hour HW #5 Egg Demo Drawings Analysis HW #6 Elodea Drawings lab Analysis HW #7 Cell Questions Membrane and Transport HW #8 Questions / 5 possible points Homework
Biology Cell Unit Homework Packet #3 Name DUE: Hour HW #5 Egg Demo Drawings Analysis HW #6 Elodea Drawings lab Analysis HW #7 Cell Questions Membrane and Transport HW #8 Questions / 5 possible points Homework
Passive Transport: Practice Problems PAP BIOLOGY
 Passive Transport: Practice Problems PAP BIOLOGY #1 Draw a diagram where the cell has low concentration of salt molecules and the environment it is in has a high concentration of salt molecules in a water
Passive Transport: Practice Problems PAP BIOLOGY #1 Draw a diagram where the cell has low concentration of salt molecules and the environment it is in has a high concentration of salt molecules in a water
7.3 Cell Boundaries. Regents Biology. Originally prepared by Kim B. Foglia. Revised and adapted by Nhan A. Pham
 7.3 Cell Boundaries Originally prepared by Kim B. Foglia. Revised and adapted by Nhan A. Pham Don t look at your reading guide/textbook or talk with each other yet! Write down one thing you know/remember
7.3 Cell Boundaries Originally prepared by Kim B. Foglia. Revised and adapted by Nhan A. Pham Don t look at your reading guide/textbook or talk with each other yet! Write down one thing you know/remember
BIOLOGY 12 - Cell Membrane and Cell Wall Function: Chapter Notes
 BIOLOGY 12 - Cell Membrane and Cell Wall Function: Chapter Notes The cell membrane is the gateway into the cell, and must allow needed things such as nutrients into the cell without letting them escape.
BIOLOGY 12 - Cell Membrane and Cell Wall Function: Chapter Notes The cell membrane is the gateway into the cell, and must allow needed things such as nutrients into the cell without letting them escape.
Unit 3: Cellular Processes. 1. SEPARTION & PROTECTION: the contents of the cell from the. 2. TRANSPORT: the transport of in and out of the cell
 Unit 3: Cellular Processes Name: Aim #14 Cell Membrane: How does the cell membrane function to maintain homeostasis? Date: _ I. The Cell Membrane: What is it? Also known as A thin structure that acts as
Unit 3: Cellular Processes Name: Aim #14 Cell Membrane: How does the cell membrane function to maintain homeostasis? Date: _ I. The Cell Membrane: What is it? Also known as A thin structure that acts as
BIOLOGY 12 - Cell Membrane and Cell Wall Function: Chapter Notes
 BIOLOGY 12 - Cell Membrane and Cell Wall Function: Chapter Notes The cell membrane is the gateway into the cell, and must allow needed things such as nutrients into the cell without letting them escape.
BIOLOGY 12 - Cell Membrane and Cell Wall Function: Chapter Notes The cell membrane is the gateway into the cell, and must allow needed things such as nutrients into the cell without letting them escape.
8.8b Osmosis Project. Grade 8 Activity Plan
 8.8b Osmosis Project Grade 8 Activity Plan Reviews and Updates 2 8.8b Osmosis Project Objectives: 1. To demonstrate osmosis and the permeability of the cell membrane. 2. Use plant cells to demonstrate
8.8b Osmosis Project Grade 8 Activity Plan Reviews and Updates 2 8.8b Osmosis Project Objectives: 1. To demonstrate osmosis and the permeability of the cell membrane. 2. Use plant cells to demonstrate
Transport of Solutes and Water
 Transport of Solutes and Water Across cell membranes 1. Simple and Facilitated diffusion. 2. Active transport. 3. Osmosis. Simple diffusion Simple diffusion - the red particles are moving from an area
Transport of Solutes and Water Across cell membranes 1. Simple and Facilitated diffusion. 2. Active transport. 3. Osmosis. Simple diffusion Simple diffusion - the red particles are moving from an area
11 - Blood Tests (Completed 05/29/18) Transcript by Rev.com. Page 1 of 5
 Megan: Hey everyone, I'm Megan Ramos, and I'm here today to answer your questions. Today's question involves lab tests, and what blood tests you should ask your doctor for. You can find a list of recommended
Megan: Hey everyone, I'm Megan Ramos, and I'm here today to answer your questions. Today's question involves lab tests, and what blood tests you should ask your doctor for. You can find a list of recommended
TRANSPORT ACROSS MEMBRANES
 Unit 2: Cells, Membranes and Signaling TRANSPORT ACROSS MEMBRANES Chapter 5 Hillis Textbook TYPES OF TRANSPORT ACROSS THE CELL (PLASMA) MEMBRANE: What do you remember? Complete the chart with what you
Unit 2: Cells, Membranes and Signaling TRANSPORT ACROSS MEMBRANES Chapter 5 Hillis Textbook TYPES OF TRANSPORT ACROSS THE CELL (PLASMA) MEMBRANE: What do you remember? Complete the chart with what you
VIDEO_ Sarah Hallberg - Interview (San Diego 2017)
 VIDEO_ Sarah Hallberg - Interview (San Diego 2017) Dr. Andreas Eenfeldt: If you are a doctor and you want to help your patients with type 2 diabetes to reverse their disease, how do you do it? You can
VIDEO_ Sarah Hallberg - Interview (San Diego 2017) Dr. Andreas Eenfeldt: If you are a doctor and you want to help your patients with type 2 diabetes to reverse their disease, how do you do it? You can
The Transport of Materials Across Cell Membranes
 The Transport of Materials Across Cell Membranes EK 2.B.1.b. LO 2.10 The Plasma Membrane 2 EK 2.B.1.b. LO 2.10 The Plasma Membrane The cell membrane is said to be semi permeable or selectively permeable
The Transport of Materials Across Cell Membranes EK 2.B.1.b. LO 2.10 The Plasma Membrane 2 EK 2.B.1.b. LO 2.10 The Plasma Membrane The cell membrane is said to be semi permeable or selectively permeable
The Kidney & Its Role in Maintaining Homeostasis. A lesson on the importance of,, &
 The Kidney & Its Role in Maintaining Homeostasis A lesson on the importance of,, & How Does the Body Maintain Homeostasis? Every cell produces metabolic waste. Product of cellular respiration: CO 2 Another
The Kidney & Its Role in Maintaining Homeostasis A lesson on the importance of,, & How Does the Body Maintain Homeostasis? Every cell produces metabolic waste. Product of cellular respiration: CO 2 Another
Chapter 7 Cell Structure and Function. Chapter 7, Section 3 Cell Boundaries and Transport
 Chapter 7 Cell Structure and Function Chapter 7, Section 3 Cell Boundaries and Transport 1 7.3 A. Cell Membrane Is Described Three Ways: 1. Selectively Permeable Membrane: Limits what enters and exits
Chapter 7 Cell Structure and Function Chapter 7, Section 3 Cell Boundaries and Transport 1 7.3 A. Cell Membrane Is Described Three Ways: 1. Selectively Permeable Membrane: Limits what enters and exits
Multiple Choice Review- Membranes & Enzymes
 Multiple Choice Review- Membranes & Enzymes 1. Cell membranes are and regulate the materials moving into and out of the cell, in order to maintain equilibrium. a. completely permeable b. ionically permeable
Multiple Choice Review- Membranes & Enzymes 1. Cell membranes are and regulate the materials moving into and out of the cell, in order to maintain equilibrium. a. completely permeable b. ionically permeable
