Production of Volatile Compounds from Irradiated Oil Emulsion Containing Amino Acids or Proteins C. JO AND D.U. AHN
|
|
|
- Loraine Sherman
- 6 years ago
- Views:
Transcription
1 JFS: Food Chemistry and Toxicology Production of Volatile Compounds from Irradiated Oil Emulsion Containing Amino Acids or Proteins C. JO AND D.U. AHN ABSTRACT: Oil emulsions containing amino acids, glutathione, bovine serum albumin, gelatin, or myofibrillar proteins were prepared. The emulsions were irradiated at 0, 2.5, 5.0, or 10.0 kgy absorbed doses and analyzed for volatile compounds. Irradiation increased the production of aldehydes (for example, hexanal, heptanal, octanal, and nonanal) indicating that lipid oxidation of oil emulsion was accelerated by irradiation. Irradiation produced, by radiolytic degradations, new volatile compounds from oil emulsions containing leucine, valine, isoleucine, phenylalanine, methionine, or cysteine. This indicated that radiolysis of protein may play an important role in offodor generation of irradiated meat. Key Words: volatile compounds, irradiation, off-odor, radiolytic degradation, lipid oxidation Introduction IRRADIATION IS AMONG THE BEST METHODS FOR CONTROLLING pathogenic bacteria in raw meat, and low dose (< 7 kgy) irradiation is permitted for use in poultry and red meats to control pathogenic bacteria (Olson 1998). However, irradiation produces a characteristic aroma as well as other significant alterations in meat flavor that significantly and negatively affect consumer acceptance. Huber and others (1953) reported that meat sterilized by irradiation developed a characteristic odor, which has been described as metallic, sulfide, wet dog, wet grain, or burnt. These investigators assumed that the off-odor was the result of free-radical oxidation initiated by the irradiation process. Merritt and others (1978) postulated that carbonyls are formed in irradiated meats due to the reactions of hydrocarbon radicals with molecular oxygen, which follows the same pathway as normal lipid oxidation. Hansen and others (1987) reported that the amount of octane, 1-octene, hexanal, and nonane in irradiated chicken increased with the irradiation dose, but the volatile compounds were not unique products of irradiation. Gas chromatographic separation and odor evaluation of volatile compounds indicated that most sulfur and carbonyl compounds had low odor thresholds and were considered important to irradiation odor (Angellini and others 1975). Batzer and Doty (1955) found that methyl mercaptan and hydrogen sulfide were important to irradiation odor, and the precursors of the undesirable odor compounds in irradiated meat were sulfur-containing compounds that were water-soluble. These results suggest that sulfur-containing compounds could be the major volatile components responsible for irradiation odor in meat. Ahn and others (1999b) suggested that the volatile compounds responsible for off-odor in irradiated meat are produced by the impact of radiation on protein and lipid molecules and are distinctly different from those of lipid oxidation. Patterson and Stevenson (1995) studied odorous volatile compounds in irradiated chicken meat and found that dimethyltrisulfide is the most potent off-odor compound, followed by cis-3- and trans-6-nonenals, oct-1-en-3-one, and bis(methylthio-) methane. They also provided evidence to support the concept that the changes detected after irradiation are distinctly different from those of warmed-over flavor in oxidized meat. Heath and others (1990) reported that irradiating uncooked chicken breast and leg at 2 or 3 kgy produced a hot fat, burned oil, or burned feathers odor that remained after the thighs were cooked. Hashim and others (1995) reported that irradiating uncooked chicken breast and thigh produced a characteristic bloody and sweet aroma that remained after the thighs were cooked but was not detectable after the breasts were cooked. Ahn and others (1999b) reported that panelists detected irradiation odor from irradiated pork loin and described it as barbecued corn-like. However, the panelists showed no preference to the odor. Although previous evidence has indicated that radiolytic products of protein components were more important than lipid oxidation in the production of irradiation odor in meat, the precise mechanisms by which off-odor is produced are unknown. Substantial differences between the radiation chemistry of food components in model systems and complex food systems exist, but the differences are mostly quantitative rather than qualitative. This fact indicates that off-odor production by irradiation is likely influenced not only by meat composition but also by potential chemical interactions among volatile components. However, this cannot be established without pursuing mechanistic studies using both model and meat systems. Radiolysis and lipid oxidation are the 2 major possible reaction mechanisms by which volatile compounds could be generated from meat. However, the irradiation dose-dependent generation of hydroxyl radicals and its mechanism on the off-odor production and the initiation of lipid oxidation in biological systems are more suggestive than conclusive. Also, the exact nature and identity of the volatile compounds that contribute to irradiationinduced off-odor are not known. Since animal tissues are composed of 3 major components, proteins, lipids, and water, volatile compounds from model systems prepared with the basic elements of meat will allow us to define the substrates and elucidate the mechanisms of off-odor production in irradiated meat. The objectives of this research were to elucidate the distinction between volatile compounds previously implicated for oxidized flavor and irradiation odor and to understand the mecha- 612 JOURNAL OF FOOD SCIENCE Vol. 65, No. 4, Institute of Food Technologists
2 nisms of off-odor production in irradiated meat at the chemical basis. Results and Discussion IRRADIATED OIL EMULSIONS PRODUCED LARGER NUMBERS OF volatile compounds than nonirradiated controls regardless of compounds added to the oil emulsions (Tables 1 to 4). More volatile compounds than those listed in the tables were produced by irradiation, but the amounts of some volatile compounds were negligible or inconsistent. Chloroform was produced from all samples, but the amount was not influenced by irradiation dose. Previous reports (Kanatt and others 1998; Jo and others 1999; Jo and Ahn 2000) indicated that irradiation increased lipid oxidation as measured by 2-thiobarbituric acid-reactive substances (TBARS) and/or carbonyl content. Jo and others (1999) reported that lipid oxidation-dependent volatile compounds, such as aldehydes, ketones, and alcohols, were not influenced by irradiation at d 0 but were increased during 7-d storage in aerobic conditions. Several aldehydes quantified and listed in Tables 1 to 4 were produced only in irradiated samples. The amounts of 1- heptene and 1-nonene produced by irradiation were dose dependent as reported previously (Ahn and others 1999a; Jo and others 1999). However, 1-heptene was produced only in the irradiated oil emulsion containing no amino acid, isoleucine, valine, or glutathione (Tables 1 and 4) because of differences in the column and method used. 1,1-Oxybis ethane was 1 of the major compounds produced from irradiated oil emulsion containing aliphatic, hydroxyl, basic side-chain group amino acids, and myofibrillar protein (Tables 1 to 4). The 2-methyl-1-propene, 2,2,6,6- tetramethyl, and 2,2,4-trimethyl-1- were found in almost all samples, but the effect of irradiation was not significant (p > 0.05). Isobutyraldehyde and 3-methylbutanal were produced in irradiated oil emulsion containing leucine, and the amount of 3- methylbutanal increased in a dose-dependent manner up to 10 kgy (Table 5). The irradiation dose-dependent production of 2- methylbutanal from oil emulsion containing isoleucine suggests deamination and decarboxylation of isoleucine by irradiation. Diehl (1995) explained that deamination and decarboxylation are the primary reactions of free radicals by irradiation in aliphatic amino acid. He also indicated that deamination plays a greater role than decarboxylation in irradiated alanine. Valine, which has a similar structure to leucine, produced isobutyraldehyde, as leucine did, but the production rate was much higher than that of leucine, indicating that deamination and decarboxylation were major reactions of radiolysis (Table 5). Among the amino acids with aromatic side chains, phenylalanine produced benzaldehyde and benzene acetaldehyde upon irradiation. Phenylalanine readily reacts with the transient species of water radiolysis and induces hydroxylation of the aromatic ring as the principal reaction (Diehl 1995). Therefore, o-, m-, and p-tyrosine can be formed by irradiation, and subsequent oxidation converts these to various isomers of dihydroxyphenylalanine. Simic (1983) explained that the reactions of electron adducts to aromatic residues would give hydrogenated derivatives, thus benzene rings can be converted to cyclohexadiene derivatives. The determination of o-tyrosine is 1 of the methods used to detect irradiated food (Meier and others 1996). Other aromatic amino acids (tyrosine, tryptophane, and histidine) were also known as irradiation-sensitive, but no characteristic volatile compounds were found in this study. Sulfur-containing amino acids (cysteine, cystine, and methionine) react with free radicals more easily than aliphatic amino acids (Simic 1983). Electron reaction with sulfhydryl derivatives leads to formation of H 2 S and is of considerable concern in Table 1 Major volatile compounds found in irradiated oil emulsion containing no amino acids or amino acids with aliphatic groups None Leu Ile Val Gly Ala Pro 1,1-Oxybis ethane 2-Propanone* Hexane Isobutyraldehyde* 1-Heptene 3-Methylbutanal* 2-Methylbutanal* Chloroform Hexanal* Heptanal* Octanal* Nonanal* 2-Methyl-1-propene 2,2,6,6-Tetramethyl- 2,2,4-Trimethyl-1- Abbreviations: Leu = Leucine; Ile = iosleucine; Val = valine; Gly = glycine, Ala = alanine; Pro = proline Table 2 Major volatile compounds found in irradiated oil emulsions containing amino acids with basic, acidic, or amide side-chain groups His Arg Lys Asp Glu Asn Gln 1,1-Oxybis ethane Chloroform Hexanal* Nonanal* 2-Methyl-1-propene 2,2,6,6-Tetramethyl- 2,2,4-Trimethyl-1- Abbreviations: His = histidine; Arg = arginine; Lys = lysine; Asp = aspartic acid; Glu = glutamic acid; Asn = asparagine; Gln = glutamine Table 3 Major volatile compounds found in irradiated oil emulsion containing amino acids with aromatic or those with hydroxyl sidechain groups Tyr Trp Phe Thr Ser Volatile compounds N I N I N I N I N I 1,1-Oxybis ethane Propanal* 2-Propanone* Chloroform Benzaldehyde Benzeneacetaldehyde Hexanal* Nonanal* 2-Methyl-1-propene 2,2,6,6-Tetramethyl 2,2,4-Trimethyl-1- Abbreviations: Tyr = tyrosine; Trp = tryptophane; Phe = phenylalanine; Thr = threonine; Ser = serine irradiation technology due to its unpleasant odor (Simic 1983). However, H 2 S was not detected in this study because the molecular scan range used was 46.1 to 550 to eliminate CO 2 peak. The volatile compounds detected in oil emulsion containing cysteine were carbon oxide disulfide and carbon disulfide (Table 5). Simic Food Chemistry and Toxicology Vol. 65, No. 4, 2000 JOURNAL OF FOOD SCIENCE 613
3 Volatile Compounds of Irradiated Oil Emulsion... Table 4 Major volatile compounds found in irradiated oil emulsions containing sulfur-containing amino acids, glutathione (GSH), bovine serum albumin (BSA), gelatin, or myofibrillar protein (MFP) Met Cys Cystine GSH BSA Gelatin MFP Pentane Carbon oxide disulfide Methanethiol 1,1-Oxybis ethane 3-Methylbutanal* 2-Methylbutanal* Methylbenzene Carbon disulfide* 2-Propenal* 1-Heptene Chloroform Dimethyldisulfide* Benzaldehyde* Hexanal* Ethylbenzene* Dimethyltrisulfide 3-Methylthiopropanal* Nonanal* 2-Methyl-1-propene 2,2,6,6-Tetramethyl- 2,2,4-Trimethyl-1- Abbreviations: Met = methionine; Cys = cysteine; GSH = glutathione; BSA = bovine serum albumin; MFP = myofibrillar protein (1983) reported that aqueous electrons (e - aq ) generated by irradiation exclusively attack -SH and not the -NH 3+ group of cysteine, and induce -radiolysis. It was also assumed that cysteine underwent dimerization reaction to form cystine as suggested by others (Simic 1983; Diehl 1995). In contrast to cysteine, cystine produced carbon disulfide from nonirradiated oil emulsion, but carbon disulfide was not found in irradiated oil emulsions (Table 5). The carbon oxide sulfide was also produced from the irradiated oil emulsion containing glutathione due probably to cysteine in the structure. The amount of carbon oxide sulfide from glutathione was much smaller than that from cysteine, and probably the cysteine present as part of a peptide is less susceptible to free-radical reaction (Diehl 1995). Four sulfur-containing compounds were produced by irradiation from oil emulsion containing methionine (Table 5). These compounds have been suggested as the main off-odor generators (Schweigert and others 1954; Patterson and Stevenson 1995). This indicated that the major source of off-odor production in irradiated meat could be sulfur-containing amino acids via the radiolytic degradation of their side chains. Interestingly, 2-propenal, which generates unpleasant odor, was also produced by irradiation in a dose-dependent manner. Removal of -SCH 3 moiety from methionine followed by deamination and decarboxylation could have produced 2-propenal. Since the dimethylthiogroup (-C-S-C-) has a low reactivity with aqueous electrons (e - aq, Simic 1983), methionine is more likely to react with hydroxyl radicals from water radiolysis. Bovine serum albumin (BSA), gelatin, and myofibrillar protein produced many benzene-containing compounds, but irradiation at the 2.5-kGy dose could not create these products except for benzaldehyde from BSA (Table 5). The rigid spatial structure of protein molecules could have protected amino acid side chains from radiolytic degradation, in contrast to the effect on free amino acids. Diehl (1995) reported that radicals formed by Table 5 The relative amounts of characteristic volatile compounds in irradiated oil emulsions containing amino acids or proteins Irradiation dose (kgy) Amino acid Volatile or proteins compounds SEM 1 Leucine Isoleucine total ion counts 10 3 Isobutyraldehyde* 0 b 197 b 243 b 1110 a 55 3-Methylbutanal* 0 d 9000 c b a 2573 Butanal* 0 b 77 ab 166 a 160 a 37 2-Methylbutanal* 0 d c b a 1390 Valine Isobutyraldehyde* 0 d 5700 c b a 520 Phenylalanine Benzaldehyde* 0 d 224 c 284 b 390 a 17 Benzene acetaldehyde 0 d 1770 c 4060 b 8000 a 755 Methionine Methanethiol 0 d 3560 c 6000 b a Propenal* 0 c 389 c 2470 b 6240 a 222 Dimethyldisulfide* 0 d 7030 c b a 645 Dimethyltrisulfide 0 d 33 c 273 b 432 a 11 3-Methylthiopropanal* 0 c 1280 bc 3320 b 9370 a 668 Cysteine Carbonoxide disulfide 0 b 0 b 19 b 145 a 13 Carbon disulfide* 0 d 384 c 1060 b 1530 a 101 Cystine Carbon disulfide* 742 a 0 b 0 b 0 b 28 Glutathione Carbonoxide disulfide 0 b 0 b 69 b 261 a 40 Bovine serum albumin Methylbenzene 0 b 0 b 130 a 137 a 21 Ethylbenzene* 0 b 0 b 132 a 148 a 23 Benzaldehyde* 0 c 42 b 98 a 117 a 13 Gelatin Benzaldehyde* 0 b 0 b 152 ab 275 a 44 Myofibrillar proteins Methylbenzene 0 b 0 b 210 a 227 a 37 Ethylbenzene* 0 b 0 b 214 a 199 a 39 a-d Means with different superscripts in the same row differ significantly (p < 0.05). 1 Standard errors of the mean among different irradiation doses (n = 16) irradiation can be held in position and be recombined. Therefore, radiation damage to certain amino acids in proteins is quite limited, and the proteins tested may not be affected at low-dose irradiation ( 2.5 kgy). WHO (1994) reported that little changes in amino-acid composition were observed at doses below 50 kgy. Bachman and others (1973) reported that free-radical yield measured by electron spin resonance (ESR) spectroscopy was increased by irradiation, but there were no changes in organoleptic characteristics and chemical composition in gelatin irradiated at the 5-kGy dose. Oil emulsion containing myofibrillar protein produced 3-methylbutanal and 2-methylbutanal, but the amounts of these compounds were not influenced by irradiation doses between 0 and 10 kgy. Ahn and others (1998) and Shahidi and Pegg (1994) reported that hexanal is the most sensitive indicator for lipid oxidation and flavor deterioration in meat products. Our results showed that irradiation significantly increased the amount of hexanal in oil emulsion containing different compounds (Table 6). Apparently, lipid oxidation was accelerated by irradiation. Irradiationinduced oxidative chemical changes were dose-dependent, and the presence of oxygen had a significant effect on the rate of lipid oxidation. Other aldehydes such as propanal, heptanal, octanal, and nonanal were also produced but the trends were similar to that of the hexanal (data not shown, p < 0.05.) Ang and Lyon (1990) reported that hexanal and pentanal had a very strong correlation with TBARS, and off-odor related to lipid oxidation in meat. 614 JOURNAL OF FOOD SCIENCE Vol. 65, No. 4, 2000
4 Irradiation produced significant amounts of volatile compounds from oil emulsion containing amino acids, indicating that the radiolytic products of amino acids could be important for off-odor production in irradiated meat. However, the responses of singular amino acid and proteins to irradiation and those of oil emulsion and meat systems would be different. Therefore, this study should be considered as a step to determine the sources of volatiles and the mechanisms of off-odor production in irradiated meat. Conclusions THE EVIDENCE SUGGESTS THAT THE RADIOLYTIC PRODUCTS OF meat components, such as proteins, lipids, amides, amines, vitamins and so on, are the major sources of off-odor volatiles in irradiated meat. Irradiation produced significant amount of volatile compounds from oil emulsion containing sulfur-containing amino acids and those with aliphatic side-chains. Volatile compounds produced by lipid oxidation during and after irradiation in the presence of oxygen also could contribute to the development of off-odor in irradiated sample. Materials and Methods Table 6 The relative amount of hexanal produced in irradiated oil emulsions containing different amino acids, glutathione, bovine serum albumin, and gelatin Irradiation dose (kgy) Amino acids or proteins SEM 1 total ion counts 10 3 None 0 d 484 c 1088 b 1680 a Isoleucine 0 b 0 b 110 b 411 a 31.3 Threonine 282 b 715 a 758 a 918 a 82.1 Serine 0 c 517 b 590 b 917 a 34.1 Histidine 0 b 1078 a 944 a 813 a Lysine 0 d 565 c 675 b 915 a 26.7 Aspartic acid 0 c 863 b 821 b 1236 a 37.9 Glutamic acid 0 c 674 b 724 b 857 a 21.9 Asparagine 143 c 732 b 678 b 986 a 74.9 Glutamine 0 d 584 c 739 b 968 a 38.8 Tyrosine 225 b 802 ab 1185 a 1343 a Methionine 0 d 190 c 303 b 458 a 18.2 Cystine 0 d 744 c 926 b 1053 a 28.7 Glutathione 0 b 349 b 1101 a 1512 a Bovine serum albumin 583 b 1358 a 1231 a 1703 a Gelatin 501 b 225 b 675 b 1176 a a-d Means with different superscripts in the same row differ significantly (p < 0.05). 1Standard errors of the mean among different irradiation doses (n = 16) a 4 C refrigerator and analyzed for volatile compounds. Food Chemistry and Toxicology Sample preparation Oil emulsions were prepared by blending soybean oil (2 ml, Preferred Products, Inc., Eden Prairie, Minn., U.S.A.), cholesterol (5% of soybean oil, w/v), and Triton X-100 (50 L) with 200 ml maleate buffer (50 mm, ph 5.8). Amino acid (50 mm), glutathione (25 mm), or protein was added to the buffer before blending with a Waring blender for 2 min at high speed. Amino acids used included those with the aliphatic side-chain group (leucine, isoleucine, valine, glycine, alanine, and proline), the hydroxyl side-chain group (threonine and serine), the basic side-chain group (histidine, arginine, and lysine), the acidic side-chain group (aspartate and glutamate), the amide side-chain group (glutamine and asparagine), the aromatic side-chain group (tyrosine, tryptophane, and phenylalanine), and the sulfur-containing side-chain groups (methionine, cysteine, and cystine). Proteins used included bovine serum albumin (BSA, 5%, w/v), gelatin (2%, w/v), and myofibrillar proteins (MFP, 20%, w/v). Myofibrillar proteins were obtained using the following procedure: Ground pork muscle was homogenized with a solution [100 mm KCl, 20 mm potassium phosphate (ph 6.8), 2 mm MgCl 2, 2 mm EGTA, and 1 mm NaN 3 (standard salt solution)] for 10 s in a Waring blender and centrifuged at 1500 g for 10 min. The sediment was suspended in 8 volumes (v/w) of standard salt solution and homogenized in the blender for another 10 s. The homogenate was filtered through a household nylon net strainer, and the filtrate was centrifuged at 1500 g for 10 min. The sediment was resuspended in 8 volumes (v/w) of 100 mm KCl, homogenized for 10 s in the blender, and centrifuged at 1500 g. The sediment was used as a myofibrillar protein for this study. A 10-mL portion of oil emulsion was transferred to 20-mL sample vials, irradiated at the 0, 2.5, 5.0, or 10.0 kgy absorbed dose using an Electron Beam irradiator (Circe IIIR Thomson CSF Linac, St. Aubin, France) with a 10 MeV energy level, a 10 kw power level, and an average 86 kgy/min dose rate after storage overnight in a 4 C cooler. The irradiation process was conducted at room temperature with single-layer display and single-sided dosage. Oil emulsion with no amino acid was prepared as a control. Samples were stored in Volatile compounds analysis A Precept II and a Purge-and-Trap concentrator 3000 (Tekmar-Dohrmann, Cincinnati, Ohio, U.S.A.) were used to purge and trap volatile compounds as described by Ahn and others (1998) with modifications. A gas chromatograph (GC; Model 6890, Hewlett Packard Co., Wilmington, Del., U.S.A.) equipped with a mass selective detector (MSD; Model 5973, Hewlett Packard Co.) was used to identify and quantify the volatile compounds. Each sample (2 ml) was transferred to a 40-mL sample vial, and headspace was flushed with helium gas (99.999% purity) for 5 s to minimize oxidation during the waiting period before analysis. Samples were purged with helium (40 ml/min) for 14 min at 40 C. Volatile compounds were trapped using a Tenax/silica/charcoal column (Tekmar-Dohrmann) and desorbed for 1 min at 220 C. The temperature of transfer lines was maintained at 135 C. A modified column was used to improve separation of volatile compounds. An HP-Wax column (7 m, 250 m i.d., 0.25 m nominal) was combined with an HP-5 column (30 m, 250 m i.d., 0.25 m nominal) using a Glass Press-fit Connector (Hewlett Packard Co.) A split inlet (split ratio, 49:1, inlet temperature 180 C) was used to inject volatile compounds into the column, and a ramped oven temperature was used (7 C for 2.5 min, increased to 25 C/min, to 105 C/min, to 200 C/min, and held for 0.33 min). Liquid nitrogen was used to cool the oven below the ambient temperature. Helium was the carrier gas at a constant flow of 1.2 ml/min. The ionization potential of mass spectrum was 70 ev; the scanned mass range was 46.1 to 550, and the scan velocity was 2.94 scan/sec. The identification of volatile compounds was achieved by comparing mass spectral data with those of the Wiley library (Hewlett Packard Co.), and some compounds were verified using external standards. Standards were added to sample vials containing 2 ml of oil emulsion and run with the same method used for oil emulsions with amino acids or proteins. Carbon oxide sulfide, 2,3-dimethyldisulfide, ethylbenzene, isobutyraldehyde were purchased from Aldrich (Milwaukee, Wisc., U.S.A.), and 2-methylbutanal, 3-methylbutanal, 2- propenal, 2-propanone, hexanal, heptanal, octanal, and nonanal were obtained from Chromatography Research Supplies Inc. (Addison, Ill., U.S.A.) The peak area (total ion counts 10 3 ) Vol. 65, No. 4, 2000 JOURNAL OF FOOD SCIENCE 615
5 Volatile Compounds of Irradiated Oil Emulsion... was reported as the amount of volatile compounds released. The peak area less than 20,000 was discarded and was considered as no production. Statistical analysis Analysis of variance was conducted to determine any difference in volatile profiles and volatile contents by using the SAS program (SAS 1989). Four replications were conducted, and significant level used was p < Mean values were compared by Student-Newman-Keul s multiple range test (Steel and Torrie 1980), and mean values and standard errors of the mean (SEM) were reported. References Ahn DU, Olson DG, Lee JI, Jo C, Chen X, Wu C Packaging and irradiation effects on lipid oxidation and volatiles in pork patties. J Food Sci 63: Ahn DU, Olson DG, Jo C, Love J, Jin SK. 1999a. Volatiles production and lipid oxidation on irradiated cooked sausage as related to packaging and storage. J Food Sci 64: Ahn DU, Jo C, Olson DG. 1999b. Analysis of volatile components and the sensory characteristics of irradiated raw pork. Meat Sci 54: Ang CYW, Lyon BG Evaluation of warmed-over flavor during chill storage of cooked broiler breast, thigh and skin by chemical, instrumental and sensory methods. J Food Sci 55: Angellini P, Merritt C Jr, Mendelshon JM, King FJ Effect of irradiation on volatile constituents of stored haddock flesh. J Food Sci 40: Bachman S, Galant S, Gasyna Z, Witkowski S, Zegota H Effects of ionizing radiation on gelatin in the solid state. In: Improvement of Food Quality by Irradiation. Proc. Series, Int. Atomic Energy Agency, Vienna. p Batzer OF, Doty DM Nature of undesirable odors formed by gamma-irradiation of beef. J Agri Food Chem 3: Diehl JF Chemical effects of ionizing radiation. In: Diehl JF, editor. Safety of Irradiated Foods. 2 nd Ed. New York: Marcel Dekker. p Hansen TJ, Chen GC, Shieh JJ Volatiles in skin of low-dose irradiated fresh chicken. J Food Sci 52: Hashim IB, Resurreccion AVA, MaWatters KH Descriptive sensory analysis of irradiated frozen or refrigerated chicken. J Food Sci 60: Heath JL, Owens SL. Tesch S, Hannah KW Effect of high-energy electron irradiation of chicken on thiobarbituric acid values, shear values, odor, and cook yield. Poultry Sci 69: Huber W, Brasch A, Waly A Effect of processing conditions on organoleptic changes in foodstuffs sterilized with high-intensity electrons. Food Tech 7: Jo C, Lee JI, Ahn DU Lipid oxidation, color changes, and volatiles production in irradiated pork sausage with different fat content and packaging during storage. Meat Sci 51: Jo C, Ahn DU Volatiles production and oxidative changes in irradiated pork sausage with different fatty acid composition and packaging during storage. J Food Sci. 65: Kanatt SR, Paul P, DÕSouza SF, Thomas P Lipid peroxidation in chicken meat during chilled storage as affected by antioxidants combined with low-dose gamma irradiation. J Food Sci 63: Meier W, Hediger H, Artho A Determination of o-tyrosine in shrimp, fish, mussels, frog legs, and egg white. In: McMurray CH, Stewart EM, Gray R, Pearce J, editors. Detection Method for Irradiated Food. Cambridge: The Royal Soc. Chem. P Merritt C Jr., Angelini P, Graham RA Effect of radiation parameters on the formation of radiolysis products in meat and meat substances. J Agri Food Chem 26: Olson DG Irradiated food. Food Tech 52: Patterson RLS, Stevenson MH Irradiation-induced off-odor in chicken and its possible control. Br Poultry Sci 36: SAS SAS User s Guide. Cary, N.C.: Statistical Analysis Systems Institute. Schweigert BS, Doty DM, Niven CF Jr A summary of studies on the irradiation of meats. In: Radiation Sterilization: Review of the Literature in Selected Fields. U.S. Department of Commerce, Office of Technical Service. p Shahidi F, Pegg RB Hexanal as an indicator of the flavor deterioration of meat and meat products. Am Chem Soc Symp Ser 558: Simic MG Radiation chemistry of water-soluble food components. In: Josephson ES, Peterson MS, editors. Preservation of Food by Ionizing Radiation. Vol. II. Boca Raton, Fla.: CRC Press. p Steel RGD, Torrie JH Principles and Procedures of Statistics; A Biochemical Approach. 2 nd Ed. New York: McGraw-Hill Book Co. p WHO Chemistry of food irradiation. In: Safety and Nutritional Adequacy of Irradiated Food. Geneva: World Health Organization. p MS received 7/21/99; revised 1/5/00; accepted 4/10/00. Journal paper No. J of the Iowa Agriculture and Home Economics Experiment Station, Ames, Iowa. Project No. 3322, supported by the National Pork Producers Council and Iowa State University Research Grants. Authors are with the Department of Animal Science, Iowa State University, Ames, Iowa , U.S.A. Direct correspondence to D.U. Ahn ( duahn@iastate.edu). 616 JOURNAL OF FOOD SCIENCE Vol. 65, No. 4, 2000
Production of Off-Odor Volatiles from Liposome-Containing Amino Acid Homopolymers by Irradiation D.U. AHN AND E.J. LEE
 JFS: Production of Off-Odor Volatiles from Liposome-Containing Amino Acid Homopolymers by Irradiation D.U. AHN AND E.J. LEE ABSTRACT: Irradiation not only generated many new volatiles but also destroyed
JFS: Production of Off-Odor Volatiles from Liposome-Containing Amino Acid Homopolymers by Irradiation D.U. AHN AND E.J. LEE ABSTRACT: Irradiation not only generated many new volatiles but also destroyed
Production of Volatiles from Amino Acid Homopolymers by Irradiation D.U. AHN
 JFS: Production of Volatiles from Amino Acid Homopolymers by Irradiation D.U. AHN ABSTRACT: Amino acid homopolymers were used to determine production of radiolytic volatiles by irradiation. Many new volatiles
JFS: Production of Volatiles from Amino Acid Homopolymers by Irradiation D.U. AHN ABSTRACT: Amino acid homopolymers were used to determine production of radiolytic volatiles by irradiation. Many new volatiles
Fat Content Influences the Color, Lipid Oxidation and Volatiles of Irradiated Ground Beef
 AS 656 ASL R2497 2010 Fat Content Influences the Color, Lipid Oxidation and Volatiles of Irradiated Ground Beef Hesham Ismail Iowa State University Eun Joo Lee Iowa State University Kyung Yuk Ko Iowa State
AS 656 ASL R2497 2010 Fat Content Influences the Color, Lipid Oxidation and Volatiles of Irradiated Ground Beef Hesham Ismail Iowa State University Eun Joo Lee Iowa State University Kyung Yuk Ko Iowa State
Use of antioxidants to reduce lipid oxidation and off-odor volatiles of irradiated pork homogenates and patties
 Meat Science 63 (2003) 1 8 www.elsevier.com/locate/meatsci Use of antioxidants to reduce lipid oxidation and off-odor volatiles of irradiated pork homogenates and patties K.C. Nam, D.U. Ahn* Department
Meat Science 63 (2003) 1 8 www.elsevier.com/locate/meatsci Use of antioxidants to reduce lipid oxidation and off-odor volatiles of irradiated pork homogenates and patties K.C. Nam, D.U. Ahn* Department
Molecular Biology. general transfer: occurs normally in cells. special transfer: occurs only in the laboratory in specific conditions.
 Chapter 9: Proteins Molecular Biology replication general transfer: occurs normally in cells transcription special transfer: occurs only in the laboratory in specific conditions translation unknown transfer:
Chapter 9: Proteins Molecular Biology replication general transfer: occurs normally in cells transcription special transfer: occurs only in the laboratory in specific conditions translation unknown transfer:
Biomolecules: amino acids
 Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
PROCESSING AND PRODUCTS. Double-Packaging Is Effective in Reducing Lipid Oxidation and Off-Odor Volatiles of Irradiated Raw Turkey Meat 1
 PROCESSING AND PRODUCTS Double-Packaging Is Effective in Reducing Lipid Oxidation and Off-Odor Volatiles of Raw Turkey Meat 1 K. C. Nam and D. U. Ahn 2 Department of Animal Science, Iowa State University,
PROCESSING AND PRODUCTS Double-Packaging Is Effective in Reducing Lipid Oxidation and Off-Odor Volatiles of Raw Turkey Meat 1 K. C. Nam and D. U. Ahn 2 Department of Animal Science, Iowa State University,
Page 8/6: The cell. Where to start: Proteins (control a cell) (start/end products)
 Page 8/6: The cell Where to start: Proteins (control a cell) (start/end products) Page 11/10: Structural hierarchy Proteins Phenotype of organism 3 Dimensional structure Function by interaction THE PROTEIN
Page 8/6: The cell Where to start: Proteins (control a cell) (start/end products) Page 11/10: Structural hierarchy Proteins Phenotype of organism 3 Dimensional structure Function by interaction THE PROTEIN
Combination of aerobic and vacuum packaging to control lipid oxidation and off-odor volatiles of irradiated raw turkey breast
 Meat Science 63 (2003) 389 395 www.elsevier.com/locate/meatsci Combination of aerobic and vacuum packaging to control lipid oxidation and off-odor volatiles of irradiated raw turkey breast K.C. Nam, D.U.
Meat Science 63 (2003) 389 395 www.elsevier.com/locate/meatsci Combination of aerobic and vacuum packaging to control lipid oxidation and off-odor volatiles of irradiated raw turkey breast K.C. Nam, D.U.
Volatile production in irradiated normal, pale soft exudative (PSE) and dark rm dry (DFD) pork under di erent packaging and storage conditions $
 Meat Science 57 (2001) 419±426 www.elsevier.com/locate/meatsci Volatile production in irradiated normal, pale soft exudative (PSE) and dark rm dry (DFD) pork under di erent packaging and storage conditions
Meat Science 57 (2001) 419±426 www.elsevier.com/locate/meatsci Volatile production in irradiated normal, pale soft exudative (PSE) and dark rm dry (DFD) pork under di erent packaging and storage conditions
Chemical Nature of the Amino Acids. Table of a-amino Acids Found in Proteins
 Chemical Nature of the Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. There are 20 a- amino acids that are relevant to the make-up of mammalian proteins (see below). Several
Chemical Nature of the Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. There are 20 a- amino acids that are relevant to the make-up of mammalian proteins (see below). Several
Reactions and amino acids structure & properties
 Lecture 2: Reactions and amino acids structure & properties Dr. Sameh Sarray Hlaoui Common Functional Groups Common Biochemical Reactions AH + B A + BH Oxidation-Reduction A-H + B-OH + energy ª A-B + H
Lecture 2: Reactions and amino acids structure & properties Dr. Sameh Sarray Hlaoui Common Functional Groups Common Biochemical Reactions AH + B A + BH Oxidation-Reduction A-H + B-OH + energy ª A-B + H
9/6/2011. Amino Acids. C α. Nonpolar, aliphatic R groups
 Amino Acids Side chains (R groups) vary in: size shape charge hydrogen-bonding capacity hydrophobic character chemical reactivity C α Nonpolar, aliphatic R groups Glycine (Gly, G) Alanine (Ala, A) Valine
Amino Acids Side chains (R groups) vary in: size shape charge hydrogen-bonding capacity hydrophobic character chemical reactivity C α Nonpolar, aliphatic R groups Glycine (Gly, G) Alanine (Ala, A) Valine
1. Describe the relationship of dietary protein and the health of major body systems.
 Food Explorations Lab I: The Building Blocks STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will be constructing animal and plant proteins using beads to represent the amino acids.
Food Explorations Lab I: The Building Blocks STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will be constructing animal and plant proteins using beads to represent the amino acids.
CHM333 LECTURE 6: 1/25/12 SPRING 2012 Professor Christine Hrycyna AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID:
 AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID: - The R group side chains on amino acids are VERY important. o Determine the properties of the amino acid itself o Determine
AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID: - The R group side chains on amino acids are VERY important. o Determine the properties of the amino acid itself o Determine
Lipid oxidation and volatile production in irradiated raw pork batters prepared with commercial soybean oil containing vitamin E
 Radiation Physics and Chemistry 61 (2001) 41 47 Lipid oxidation and volatile production in irradiated raw pork batters prepared with commercial soybean oil containing vitamin E Cheorun Jo a, Dong Uk Ahn
Radiation Physics and Chemistry 61 (2001) 41 47 Lipid oxidation and volatile production in irradiated raw pork batters prepared with commercial soybean oil containing vitamin E Cheorun Jo a, Dong Uk Ahn
Effect of Dietary Vitamin E and Irradiation on Lipid Oxidation, Color, and Volatiles of Fresh and Previously Frozen Turkey Breast Patties
 Animal Industry Report AS 650 ASL R1855 2004 Effect of Dietary Vitamin E and Irradiation on Lipid Oxidation, Color, and Volatiles of Fresh and Previously Frozen Turkey Breast Patties K. C. Nam B. R. Min
Animal Industry Report AS 650 ASL R1855 2004 Effect of Dietary Vitamin E and Irradiation on Lipid Oxidation, Color, and Volatiles of Fresh and Previously Frozen Turkey Breast Patties K. C. Nam B. R. Min
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
(65 pts.) 27. (10 pts.) 28. (15 pts.) 29. (10 pts.) TOTAL (100 points) Moorpark College Chemistry 11 Spring Instructor: Professor Gopal
 Moorpark College Chemistry 11 Spring 2012 Instructor: Professor Gopal Examination # 5: Section Five May 1, 2012 Name: (print) GOOD LUCK! Directions: Make sure your examination contains TWELVE total pages
Moorpark College Chemistry 11 Spring 2012 Instructor: Professor Gopal Examination # 5: Section Five May 1, 2012 Name: (print) GOOD LUCK! Directions: Make sure your examination contains TWELVE total pages
Amino acids-incorporated nanoflowers with an
 Amino acids-incorporated nanoflowers with an intrinsic peroxidase-like activity Zhuo-Fu Wu 1,2,+, Zhi Wang 1,+, Ye Zhang 3, Ya-Li Ma 3, Cheng-Yan He 4, Heng Li 1, Lei Chen 1, Qi-Sheng Huo 3, Lei Wang 1,*
Amino acids-incorporated nanoflowers with an intrinsic peroxidase-like activity Zhuo-Fu Wu 1,2,+, Zhi Wang 1,+, Ye Zhang 3, Ya-Li Ma 3, Cheng-Yan He 4, Heng Li 1, Lei Chen 1, Qi-Sheng Huo 3, Lei Wang 1,*
LAB#23: Biochemical Evidence of Evolution Name: Period Date :
 LAB#23: Biochemical Evidence of Name: Period Date : Laboratory Experience #23 Bridge Worth 80 Lab Minutes If two organisms have similar portions of DNA (genes), these organisms will probably make similar
LAB#23: Biochemical Evidence of Name: Period Date : Laboratory Experience #23 Bridge Worth 80 Lab Minutes If two organisms have similar portions of DNA (genes), these organisms will probably make similar
1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids
 Amino acids 1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids 5-To understand amino acids synthesis Amino
Amino acids 1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids 5-To understand amino acids synthesis Amino
Effects of oleoresin tocopherol combinations on lipid oxidation, off-odor, and color of irradiated raw and cooked pork patties
 Meat Science 75 (2007) 61 70 MEAT SCIENCE www.elsevier.com/locate/meatsci Effects of oleoresin tocopherol combinations on lipid oxidation, off-odor, and color of irradiated raw and cooked pork patties
Meat Science 75 (2007) 61 70 MEAT SCIENCE www.elsevier.com/locate/meatsci Effects of oleoresin tocopherol combinations on lipid oxidation, off-odor, and color of irradiated raw and cooked pork patties
Effect of irradiation on the quality of turkey ham during storage
 Meat Science 66 (2003) 63 68 www.elsevier.com/locate/meatsci Effect of irradiation on the quality of turkey ham during storage M.J. Zhu a, E.J. Lee a, A. Mendonca b, D.U. Ahn a, * a Department of Animal
Meat Science 66 (2003) 63 68 www.elsevier.com/locate/meatsci Effect of irradiation on the quality of turkey ham during storage M.J. Zhu a, E.J. Lee a, A. Mendonca b, D.U. Ahn a, * a Department of Animal
Amino Acids. Amino Acids. Fundamentals. While their name implies that amino acids are compounds that contain an NH. 3 and CO NH 3
 Fundamentals While their name implies that amino acids are compounds that contain an 2 group and a 2 group, these groups are actually present as 3 and 2 respectively. They are classified as α, β, γ, etc..
Fundamentals While their name implies that amino acids are compounds that contain an 2 group and a 2 group, these groups are actually present as 3 and 2 respectively. They are classified as α, β, γ, etc..
Lipids: diverse group of hydrophobic molecules
 Lipids: diverse group of hydrophobic molecules Lipids only macromolecules that do not form polymers li3le or no affinity for water hydrophobic consist mostly of hydrocarbons nonpolar covalent bonds fats
Lipids: diverse group of hydrophobic molecules Lipids only macromolecules that do not form polymers li3le or no affinity for water hydrophobic consist mostly of hydrocarbons nonpolar covalent bonds fats
Proteins are sometimes only produced in one cell type or cell compartment (brain has 15,000 expressed proteins, gut has 2,000).
 Lecture 2: Principles of Protein Structure: Amino Acids Why study proteins? Proteins underpin every aspect of biological activity and therefore are targets for drug design and medicinal therapy, and in
Lecture 2: Principles of Protein Structure: Amino Acids Why study proteins? Proteins underpin every aspect of biological activity and therefore are targets for drug design and medicinal therapy, and in
PROCESSING AND PRODUCTS
 PROCESSING AND PRODUCTS Use of Double Packaging and Antioxidant Combinations to Improve Color, Lipid Oxidation, and Volatiles of Raw and Cooked Turkey Breast Patties 1 K. C. Nam and D. U. Ahn 2 Department
PROCESSING AND PRODUCTS Use of Double Packaging and Antioxidant Combinations to Improve Color, Lipid Oxidation, and Volatiles of Raw and Cooked Turkey Breast Patties 1 K. C. Nam and D. U. Ahn 2 Department
Effects of Diet, Packaging and Irradiation on Protein Oxidation, Lipid Oxidation of Raw Broiler Thigh Meat
 AS 659 ASL R26 203 Effects of Diet, Packaging and Irradiation on Protein Oxidation, Lipid Oxidation of Raw Broiler Thigh Meat Shan Xiao Iowa State University Wan Gang Zhang Iowa State University Eun Joo
AS 659 ASL R26 203 Effects of Diet, Packaging and Irradiation on Protein Oxidation, Lipid Oxidation of Raw Broiler Thigh Meat Shan Xiao Iowa State University Wan Gang Zhang Iowa State University Eun Joo
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 ANSWERS For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 ANSWERS For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the
Chapter 3: Amino Acids and Peptides
 Chapter 3: Amino Acids and Peptides BINF 6101/8101, Spring 2018 Outline 1. Overall amino acid structure 2. Amino acid stereochemistry 3. Amino acid sidechain structure & classification 4. Non-standard
Chapter 3: Amino Acids and Peptides BINF 6101/8101, Spring 2018 Outline 1. Overall amino acid structure 2. Amino acid stereochemistry 3. Amino acid sidechain structure & classification 4. Non-standard
Biomolecules Amino Acids & Protein Chemistry
 Biochemistry Department Date: 17/9/ 2017 Biomolecules Amino Acids & Protein Chemistry Prof.Dr./ FAYDA Elazazy Professor of Biochemistry and Molecular Biology Intended Learning Outcomes ILOs By the end
Biochemistry Department Date: 17/9/ 2017 Biomolecules Amino Acids & Protein Chemistry Prof.Dr./ FAYDA Elazazy Professor of Biochemistry and Molecular Biology Intended Learning Outcomes ILOs By the end
Effect of Irradiating Shell Eggs on Quality Attributes and Functional Properties of Yolk and White
 Animal Industry Report AS 654 ASL R2263 2008 Effect of Irradiating Shell Eggs on Quality Attributes and Functional Properties of Yolk and White Byung R. Min Ki C. Nam Eun J. Lee Kyung Y. Ko Darrell W.
Animal Industry Report AS 654 ASL R2263 2008 Effect of Irradiating Shell Eggs on Quality Attributes and Functional Properties of Yolk and White Byung R. Min Ki C. Nam Eun J. Lee Kyung Y. Ko Darrell W.
CS612 - Algorithms in Bioinformatics
 Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Chemistry 121 Winter 17
 Chemistry 121 Winter 17 Introduction to Organic Chemistry and Biochemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State) E-mail: upali@latech.edu Office: 311 Carson Taylor Hall ; Phone: 318-257-4941;
Chemistry 121 Winter 17 Introduction to Organic Chemistry and Biochemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State) E-mail: upali@latech.edu Office: 311 Carson Taylor Hall ; Phone: 318-257-4941;
Macromolecules of Life -3 Amino Acids & Proteins
 Macromolecules of Life -3 Amino Acids & Proteins Shu-Ping Lin, Ph.D. Institute of Biomedical Engineering E-mail: splin@dragon.nchu.edu.tw Website: http://web.nchu.edu.tw/pweb/users/splin/ Amino Acids Proteins
Macromolecules of Life -3 Amino Acids & Proteins Shu-Ping Lin, Ph.D. Institute of Biomedical Engineering E-mail: splin@dragon.nchu.edu.tw Website: http://web.nchu.edu.tw/pweb/users/splin/ Amino Acids Proteins
Quality Characteristics of Irradiated Ready-to-Eat Breast Rolls from Turkeys Fed Conjugated Linoleic Acid 1
 Quality Characteristics of Irradiated Ready-to-Eat Breast Rolls from Turkeys Fed Conjugated Linoleic Acid 1 M. Du,* D. U. Ahn,*,2 A. F. Mendonca, and I. V. Wesley *Department of Animal Science, and Department
Quality Characteristics of Irradiated Ready-to-Eat Breast Rolls from Turkeys Fed Conjugated Linoleic Acid 1 M. Du,* D. U. Ahn,*,2 A. F. Mendonca, and I. V. Wesley *Department of Animal Science, and Department
PROCESSING AND PRODUCTS
 PROCESSING AND PRODUCTS Profiles and Lipid Oxidation of Irradiated Cooked Chicken Meat from Laying Hens Fed Diets Containing Conjugated Linoleic Acid 1 M. Du, D. U. Ahn, 2 K. C. Nam, and J. L. Sell Department
PROCESSING AND PRODUCTS Profiles and Lipid Oxidation of Irradiated Cooked Chicken Meat from Laying Hens Fed Diets Containing Conjugated Linoleic Acid 1 M. Du, D. U. Ahn, 2 K. C. Nam, and J. L. Sell Department
Effect of Antioxidants on Consumer Acceptance of Irradiated Turkey Meat E.J. LEE, J. LOVE, AND D.U. AHN
 JFS: Effect of Antioxidants on Consumer Acceptance of Irradiated Turkey Meat E.J. LEE, J. LOVE, AND D.U. AHN ABSTRACT: Antioxidants had no effect on the production of sulfur compounds, color change, and
JFS: Effect of Antioxidants on Consumer Acceptance of Irradiated Turkey Meat E.J. LEE, J. LOVE, AND D.U. AHN ABSTRACT: Antioxidants had no effect on the production of sulfur compounds, color change, and
Animal Industry Report
 Animal Industry Report AS 650 ASL R1861 2004 Use of Double Packaging and Antioxidant Combinations to Improve Color, Lipid Oxidation, and Volatiles of Raw and Cooked Turkey Breast Patties K. C. Nam Iowa
Animal Industry Report AS 650 ASL R1861 2004 Use of Double Packaging and Antioxidant Combinations to Improve Color, Lipid Oxidation, and Volatiles of Raw and Cooked Turkey Breast Patties K. C. Nam Iowa
Objective: You will be able to explain how the subcomponents of
 Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
Introduction to Protein Structure Collection
 Introduction to Protein Structure Collection Teaching Points This collection is designed to introduce students to the concepts of protein structure and biochemistry. Different activities guide students
Introduction to Protein Structure Collection Teaching Points This collection is designed to introduce students to the concepts of protein structure and biochemistry. Different activities guide students
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A
 Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Packaging and Irradiation Effects on Lipid Oxidation and Volatiles in Cooked Pork Patties
 Packaging and Irradiation Effects on Lipid Oxidation and Volatiles in Cooked Pork Patties D. U. Ahn, professor, D. G. Olson, professor, C. Jo, graduate student, X. Chen, graduate student, J. Wu, graduate
Packaging and Irradiation Effects on Lipid Oxidation and Volatiles in Cooked Pork Patties D. U. Ahn, professor, D. G. Olson, professor, C. Jo, graduate student, X. Chen, graduate student, J. Wu, graduate
Identification of free amino acids in several crude extracts of two legumes
 1 2 Identification of free amino acids in several crude extracts of two legumes using Thin Layer Chromatography 3 Authors 4 5 6 7 8 9 Taghread Hudaib Key words 10 11 12 13 14 15 16 17 18 19 20 Amino acids;
1 2 Identification of free amino acids in several crude extracts of two legumes using Thin Layer Chromatography 3 Authors 4 5 6 7 8 9 Taghread Hudaib Key words 10 11 12 13 14 15 16 17 18 19 20 Amino acids;
Effect of Double-packaging and Acid Combination on the Quality of Irradiated Raw Turkey Patties K.C. NAM AND D.U. AHN
 JFS: Effect of Double-packaging and Acid Combination on the Quality of Raw Turkey Patties K.C. NAM AND D.U. AHN ABSTRACT: Effects of double-packageing (combinational use of vacuum and aerobic packaging
JFS: Effect of Double-packaging and Acid Combination on the Quality of Raw Turkey Patties K.C. NAM AND D.U. AHN ABSTRACT: Effects of double-packageing (combinational use of vacuum and aerobic packaging
(30 pts.) 16. (24 pts.) 17. (20 pts.) 18. (16 pts.) 19. (5 pts.) 20. (5 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
Food Irradiation. Dose Rate Effect
 Food Irradiation Dose Rate Effect Dose Rate Effect in Food Irradiation Aim of all food processing methods is to minimize damage to the nutrients while maximizing damage to microoganisms Codex Alimentarius
Food Irradiation Dose Rate Effect Dose Rate Effect in Food Irradiation Aim of all food processing methods is to minimize damage to the nutrients while maximizing damage to microoganisms Codex Alimentarius
Moorpark College Chemistry 11 Fall Instructor: Professor Gopal. Examination # 5: Section Five May 7, Name: (print)
 Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
Copyright 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings
 Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version]
![Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version] Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version]](/thumbs/84/90818314.jpg) Earth/matriX: SCIENCE TODAY Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version] By Charles William Johnson Earth/matriX Editions P.O.
Earth/matriX: SCIENCE TODAY Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version] By Charles William Johnson Earth/matriX Editions P.O.
JFS: Food Chemistry and Toxicology. Food Chemistry and Toxicology. Introduction Consumers and regulatory agencies are well aware of the dangers
 JFS: Prevention of Pinking, Off-odor, and Lipid Oxidation in Pork Loin Using Double Packaging K.C. NAM, B.R. MIN, S.C. LEE, J. CORDRAY, AND D.U. AHN ABSTRACT: Lipid oxidation, color, volatiles, and sensory
JFS: Prevention of Pinking, Off-odor, and Lipid Oxidation in Pork Loin Using Double Packaging K.C. NAM, B.R. MIN, S.C. LEE, J. CORDRAY, AND D.U. AHN ABSTRACT: Lipid oxidation, color, volatiles, and sensory
number Done by Corrected by Doctor Dr.Diala
 number 32 Done by Mousa Salah Corrected by Bahaa Najjar Doctor Dr.Diala 1 P a g e In the last lecture we talked about the common processes between all amino acids which are: transamination, deamination,
number 32 Done by Mousa Salah Corrected by Bahaa Najjar Doctor Dr.Diala 1 P a g e In the last lecture we talked about the common processes between all amino acids which are: transamination, deamination,
Maha AbuAjamieh. Tamara Wahbeh. Mamoon Ahram
 12 Maha AbuAjamieh Tamara Wahbeh Mamoon Ahram - - Go to this sheet s last page for definitions of the words with an asterisk above them (*) - You should memorise the 3-letter abbreviations, of all the
12 Maha AbuAjamieh Tamara Wahbeh Mamoon Ahram - - Go to this sheet s last page for definitions of the words with an asterisk above them (*) - You should memorise the 3-letter abbreviations, of all the
If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out.
 Sign In Forgot Password Register username username password password Sign In If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki
Sign In Forgot Password Register username username password password Sign In If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki
2. Ionization Sources 3. Mass Analyzers 4. Tandem Mass Spectrometry
 Dr. Sanjeeva Srivastava 1. Fundamental of Mass Spectrometry Role of MS and basic concepts 2. Ionization Sources 3. Mass Analyzers 4. Tandem Mass Spectrometry 2 1 MS basic concepts Mass spectrometry - technique
Dr. Sanjeeva Srivastava 1. Fundamental of Mass Spectrometry Role of MS and basic concepts 2. Ionization Sources 3. Mass Analyzers 4. Tandem Mass Spectrometry 2 1 MS basic concepts Mass spectrometry - technique
Properties of amino acids in proteins
 Properties of amino acids in proteins one of the primary roles of DNA (but far from the only one!!!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids
Properties of amino acids in proteins one of the primary roles of DNA (but far from the only one!!!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids
Effect of Garlic, Onion, and their Combination on the Quality and Sensory Characteristics of Irradiated Raw Ground Beef
 Animal Industry Report AS 659 ASL R2765 2013 Effect of Garlic, Onion, and their Combination on the Quality and Sensory Characteristics of Irradiated Raw Ground Beef Han Sul Yang Eun Joo Lee Sunhee Moon
Animal Industry Report AS 659 ASL R2765 2013 Effect of Garlic, Onion, and their Combination on the Quality and Sensory Characteristics of Irradiated Raw Ground Beef Han Sul Yang Eun Joo Lee Sunhee Moon
The Structure and Function of Macromolecules
 The Structure and Function of Macromolecules Macromolecules are polymers Polymer long molecule consisting of many similar building blocks. Monomer the small building block molecules. Carbohydrates, proteins
The Structure and Function of Macromolecules Macromolecules are polymers Polymer long molecule consisting of many similar building blocks. Monomer the small building block molecules. Carbohydrates, proteins
1. (38 pts.) 2. (25 pts.) 3. (15 pts.) 4. (12 pts.) 5. (10 pts.) Bonus (12 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2010 Instructor: Professor Torres Examination #5: Section Five May 4, 2010 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total pages
Moorpark College Chemistry 11 Spring 2010 Instructor: Professor Torres Examination #5: Section Five May 4, 2010 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total pages
استاذ الكيمياءالحيوية
 قسم الكيمياء الحيوية د.دولت على سالمه استاذ الكيمياءالحيوية ٢٠١٥-٢٠١٤ الرمز الكودي : ٥١٢ المحاضرة األولى ١ Content : Definition of proteins Definition of amino acids Definition of peptide bond General
قسم الكيمياء الحيوية د.دولت على سالمه استاذ الكيمياءالحيوية ٢٠١٥-٢٠١٤ الرمز الكودي : ٥١٢ المحاضرة األولى ١ Content : Definition of proteins Definition of amino acids Definition of peptide bond General
Gentilucci, Amino Acids, Peptides, and Proteins. Peptides and proteins are polymers of amino acids linked together by amide bonds CH 3
 Amino Acids Peptides and proteins are polymers of amino acids linked together by amide bonds Aliphatic Side-Chain Amino Acids - - H CH glycine alanine 3 proline valine CH CH 3 - leucine - isoleucine CH
Amino Acids Peptides and proteins are polymers of amino acids linked together by amide bonds Aliphatic Side-Chain Amino Acids - - H CH glycine alanine 3 proline valine CH CH 3 - leucine - isoleucine CH
Amino Acids. Lecture 4: Margaret A. Daugherty. Fall Swiss-prot database: How many proteins? From where?
 Lecture 4: Amino Acids Margaret A. Daugherty Fall 2004 Swiss-prot database: How many proteins? From where? 1986 Use http://us.expasy.org to get to swiss-prot database Proteins are the workhorses of the
Lecture 4: Amino Acids Margaret A. Daugherty Fall 2004 Swiss-prot database: How many proteins? From where? 1986 Use http://us.expasy.org to get to swiss-prot database Proteins are the workhorses of the
Classification of amino acids: -
 Page 1 of 8 P roteinogenic amino acids, also known as standard, normal or primary amino acids are 20 amino acids that are incorporated in proteins and that are coded in the standard genetic code (subunit
Page 1 of 8 P roteinogenic amino acids, also known as standard, normal or primary amino acids are 20 amino acids that are incorporated in proteins and that are coded in the standard genetic code (subunit
Introduction to Peptide Sequencing
 Introduction to Peptide equencing Quadrupole Ion Traps tructural Biophysics Course December 3, 2014 12/8/14 Introduction to Peptide equencing - athan Yates 1 Why are ion traps used to sequence peptides?
Introduction to Peptide equencing Quadrupole Ion Traps tructural Biophysics Course December 3, 2014 12/8/14 Introduction to Peptide equencing - athan Yates 1 Why are ion traps used to sequence peptides?
Proteins consist in whole or large part of amino acids. Simple proteins consist only of amino acids.
 Today we begin our discussion of the structure and properties of proteins. Proteins consist in whole or large part of amino acids. Simple proteins consist only of amino acids. Conjugated proteins contain
Today we begin our discussion of the structure and properties of proteins. Proteins consist in whole or large part of amino acids. Simple proteins consist only of amino acids. Conjugated proteins contain
The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5
 Key Concepts: The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5 Proteins include a diversity of structures, resulting in a wide range of functions Proteins Enzymatic s
Key Concepts: The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5 Proteins include a diversity of structures, resulting in a wide range of functions Proteins Enzymatic s
9/16/15. Properties of Water. Benefits of Water. More properties of water
 Properties of Water Solid/Liquid Density Water is densest at 4⁰C Ice floats Allows life under the ice Hydrogen bond Ice Hydrogen bonds are stable Liquid water Hydrogen bonds break and re-form Benefits
Properties of Water Solid/Liquid Density Water is densest at 4⁰C Ice floats Allows life under the ice Hydrogen bond Ice Hydrogen bonds are stable Liquid water Hydrogen bonds break and re-form Benefits
AP Bio. Protiens Chapter 5 1
 Concept.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 0% of the dry mass of most cells Protein functions include structural support, storage, transport,
Concept.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 0% of the dry mass of most cells Protein functions include structural support, storage, transport,
CHAPTER 21: Amino Acids, Proteins, & Enzymes. General, Organic, & Biological Chemistry Janice Gorzynski Smith
 CHAPTER 21: Amino Acids, Proteins, & Enzymes General, Organic, & Biological Chemistry Janice Gorzynski Smith CHAPTER 21: Amino Acids, Proteins, Enzymes Learning Objectives: q The 20 common, naturally occurring
CHAPTER 21: Amino Acids, Proteins, & Enzymes General, Organic, & Biological Chemistry Janice Gorzynski Smith CHAPTER 21: Amino Acids, Proteins, Enzymes Learning Objectives: q The 20 common, naturally occurring
Effect of Muscle Type, Packaging, and Irradiation on Lipid Oxidation, Volatile Production, and Color in Raw Pork Patties
 Swine Research Report, 1997 Animal Science Research Reports 1998 Effect of Muscle Type, Packaging, and Irradiation on Lipid Oxidation, Volatile Production, and Color in Raw Pork Patties Dong U. Ahn Iowa
Swine Research Report, 1997 Animal Science Research Reports 1998 Effect of Muscle Type, Packaging, and Irradiation on Lipid Oxidation, Volatile Production, and Color in Raw Pork Patties Dong U. Ahn Iowa
Volatile Sulfur Compounds in Foods as a Result of Ionizing Radiation
 Animal Science Publications Animal Science 8-2011 Volatile Sulfur Compounds in Foods as a Result of Ionizing Radiation Xuetong Fan United States Department of Agrilculture Eun Joo Lee Iowa State University
Animal Science Publications Animal Science 8-2011 Volatile Sulfur Compounds in Foods as a Result of Ionizing Radiation Xuetong Fan United States Department of Agrilculture Eun Joo Lee Iowa State University
Amino acids. You are required to know and identify the 20 amino acids : their names, 3 letter abbreviations and their structures.
 Amino acids You are required to know and identify the 20 amino acids : their names, 3 letter abbreviations and their structures. If you wanna make any classification in the world, you have to find what
Amino acids You are required to know and identify the 20 amino acids : their names, 3 letter abbreviations and their structures. If you wanna make any classification in the world, you have to find what
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges
 Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 9. AMINO ACIDS, PEPTIDES AND
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 9. AMINO ACIDS, PEPTIDES AND
A Chemical Look at Proteins: Workhorses of the Cell
 A Chemical Look at Proteins: Workhorses of the Cell A A Life ciences 1a Lecture otes et 4 pring 2006 Prof. Daniel Kahne Life requires chemistry 2 amino acid monomer and it is proteins that make the chemistry
A Chemical Look at Proteins: Workhorses of the Cell A A Life ciences 1a Lecture otes et 4 pring 2006 Prof. Daniel Kahne Life requires chemistry 2 amino acid monomer and it is proteins that make the chemistry
Date: EXERCISE 4. Figure 1. Amino acid structure.
 Student s name: Date: Points: Assistant s signature: Index numer: /6 EXERISE 4 AMIN AIDS AND PRTEINS. Amino acids are structural units (monomers) of proteins. There are 20 different amino acids coded for
Student s name: Date: Points: Assistant s signature: Index numer: /6 EXERISE 4 AMIN AIDS AND PRTEINS. Amino acids are structural units (monomers) of proteins. There are 20 different amino acids coded for
PROTEINS. Building blocks, structure and function. Aim: You will have a clear picture of protein construction and their general properties
 PROTEINS Building blocks, structure and function Aim: You will have a clear picture of protein construction and their general properties Reading materials: Compendium in Biochemistry, page 13-49. Microbiology,
PROTEINS Building blocks, structure and function Aim: You will have a clear picture of protein construction and their general properties Reading materials: Compendium in Biochemistry, page 13-49. Microbiology,
Moorpark College Chemistry 11 Fall Instructor: Professor Gopal. Examination #5: Section Five December 7, Name: (print) Section:
 Moorpark College Chemistry 11 Fall 2011 Instructor: Professor Gopal Examination #5: Section Five December 7, 2011 Name: (print) Section: alkene < alkyne < amine < alcohol < ketone < aldehyde < amide
Moorpark College Chemistry 11 Fall 2011 Instructor: Professor Gopal Examination #5: Section Five December 7, 2011 Name: (print) Section: alkene < alkyne < amine < alcohol < ketone < aldehyde < amide
Human Biochemistry Option B
 Human Biochemistry Option B A look ahead... Your body has many functions to perform every day: Structural support, genetic information, communication, energy supply, metabolism Right now, thousands of
Human Biochemistry Option B A look ahead... Your body has many functions to perform every day: Structural support, genetic information, communication, energy supply, metabolism Right now, thousands of
Cells N5 Homework book
 1 Cells N5 Homework book 2 Homework 1 3 4 5 Homework2 Cell Ultrastructure and Membrane 1. Name and give the function of the numbered organelles in the cell below: A E B D C 2. Name 3 structures you might
1 Cells N5 Homework book 2 Homework 1 3 4 5 Homework2 Cell Ultrastructure and Membrane 1. Name and give the function of the numbered organelles in the cell below: A E B D C 2. Name 3 structures you might
Methionine (Met or M)
 Fig. 5-17 Nonpolar Fig. 5-17a Nonpolar Glycine (Gly or G) Alanine (Ala or A) Valine (Val or V) Leucine (Leu or L) Isoleucine (Ile or I) Methionine (Met or M) Phenylalanine (Phe or F) Polar Trypotphan (Trp
Fig. 5-17 Nonpolar Fig. 5-17a Nonpolar Glycine (Gly or G) Alanine (Ala or A) Valine (Val or V) Leucine (Leu or L) Isoleucine (Ile or I) Methionine (Met or M) Phenylalanine (Phe or F) Polar Trypotphan (Trp
3. AMINO ACID AND PEPTIDES
 3. AMINO ACID AND PEPTIDES 3.1 Amino Acids and Peptides General structure - Only 20 amino-acids are found in proteins - Amino group and carboxyl group - α-carbon and side chain group 3.1 Amino Acids and
3. AMINO ACID AND PEPTIDES 3.1 Amino Acids and Peptides General structure - Only 20 amino-acids are found in proteins - Amino group and carboxyl group - α-carbon and side chain group 3.1 Amino Acids and
Catabolism of Carbon skeletons of Amino acids. Amino acid metabolism
 Catabolism of Carbon skeletons of Amino acids Amino acid metabolism Carbon skeleton Carbon Skeleton a carbon skeleton is the internal structure of organic molecules. Carbon Arrangements The arrangement
Catabolism of Carbon skeletons of Amino acids Amino acid metabolism Carbon skeleton Carbon Skeleton a carbon skeleton is the internal structure of organic molecules. Carbon Arrangements The arrangement
PROCESSING AND PRODUCTS
 PROCESSING AND PRODUCTS Effects of Dietary Vitamin E Supplementation on Lipid Oxidation and Volatiles Content of Irradiated, Cooked Turkey Meat Patties with Different Packaging 1 D. U. AHN,2 J. L. SELL,
PROCESSING AND PRODUCTS Effects of Dietary Vitamin E Supplementation on Lipid Oxidation and Volatiles Content of Irradiated, Cooked Turkey Meat Patties with Different Packaging 1 D. U. AHN,2 J. L. SELL,
Biochemistry - I. Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I
 Biochemistry - I Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I Hello, welcome to the course Biochemistry 1 conducted by me Dr. S Dasgupta,
Biochemistry - I Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I Hello, welcome to the course Biochemistry 1 conducted by me Dr. S Dasgupta,
Sprint. Revolutionary technology for the rapid, safe and direct determination of protein
 Sprint Rapid Protein Analyzer Revolutionary technology for the rapid, safe and direct determination of protein Benefits Its simple to use Fast analysis of all types of food No hazardous chemicals Safer
Sprint Rapid Protein Analyzer Revolutionary technology for the rapid, safe and direct determination of protein Benefits Its simple to use Fast analysis of all types of food No hazardous chemicals Safer
Macromolecules Structure and Function
 Macromolecules Structure and Function Within cells, small organic molecules (monomers) are joined together to form larger molecules (polymers). Macromolecules are large molecules composed of thousands
Macromolecules Structure and Function Within cells, small organic molecules (monomers) are joined together to form larger molecules (polymers). Macromolecules are large molecules composed of thousands
2. Which of the following amino acids is most likely to be found on the outer surface of a properly folded protein?
 Name: WHITE Student Number: Answer the following questions on the computer scoring sheet. 1 mark each 1. Which of the following amino acids would have the highest relative mobility R f in normal thin layer
Name: WHITE Student Number: Answer the following questions on the computer scoring sheet. 1 mark each 1. Which of the following amino acids would have the highest relative mobility R f in normal thin layer
Introduction to proteins and protein structure
 Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Lecture 3: 8/24. CHAPTER 3 Amino Acids
 Lecture 3: 8/24 CHAPTER 3 Amino Acids 1 Chapter 3 Outline 2 Amino Acid Are Biomolecules and their Atoms Can Be Visualized by Two Different Ways 1) Fischer projections: Two dimensional representation of
Lecture 3: 8/24 CHAPTER 3 Amino Acids 1 Chapter 3 Outline 2 Amino Acid Are Biomolecules and their Atoms Can Be Visualized by Two Different Ways 1) Fischer projections: Two dimensional representation of
Practice Problems 3. a. What is the name of the bond formed between two amino acids? Are these bonds free to rotate?
 Life Sciences 1a Practice Problems 3 1. Draw the oligopeptide for Ala-Phe-Gly-Thr-Asp. You do not need to indicate the stereochemistry of the sidechains. Denote with arrows the bonds formed between the
Life Sciences 1a Practice Problems 3 1. Draw the oligopeptide for Ala-Phe-Gly-Thr-Asp. You do not need to indicate the stereochemistry of the sidechains. Denote with arrows the bonds formed between the
Four Classes of Biological Macromolecules. Biological Macromolecules. Lipids
 Biological Macromolecules Much larger than other par4cles found in cells Made up of smaller subunits Found in all cells Great diversity of func4ons Four Classes of Biological Macromolecules Lipids Polysaccharides
Biological Macromolecules Much larger than other par4cles found in cells Made up of smaller subunits Found in all cells Great diversity of func4ons Four Classes of Biological Macromolecules Lipids Polysaccharides
Lecture 4. Grouping Amino Acid 7/1/10. Proteins. Amino Acids. Where Are Proteins Located. Nonpolar Amino Acids
 Proteins Lecture 4 Proteins - Composition of Proteins (Amino Acids) Chapter 21 ection 1-6! Proteins are compounds of high molar mass consisting almost entirely of amino acid chain(s)! Molar masses range
Proteins Lecture 4 Proteins - Composition of Proteins (Amino Acids) Chapter 21 ection 1-6! Proteins are compounds of high molar mass consisting almost entirely of amino acid chain(s)! Molar masses range
EFFECTS OF AMINO ACID SUBSTITUTIONS FOR WHEY PROTEIN CONCENTRATE ON WEANLING PIG PERFORMANCE. Authors: J. Chung, S.D. Carter and J.C.
 EFFECTS OF AMINO ACID SUBSTITUTIONS FOR WHEY PROTEIN CONCENTRATE ON WEANLING PIG PERFORMANCE 1999 Animal Science Research Report Authors: Story in Brief Pages 266-272 J. Chung, S.D. Carter and J.C. Whisenhunt
EFFECTS OF AMINO ACID SUBSTITUTIONS FOR WHEY PROTEIN CONCENTRATE ON WEANLING PIG PERFORMANCE 1999 Animal Science Research Report Authors: Story in Brief Pages 266-272 J. Chung, S.D. Carter and J.C. Whisenhunt
Amino Acids. Review I: Protein Structure. Amino Acids: Structures. Amino Acids (contd.) Rajan Munshi
 Review I: Protein Structure Rajan Munshi BBSI @ Pitt 2005 Department of Computational Biology University of Pittsburgh School of Medicine May 24, 2005 Amino Acids Building blocks of proteins 20 amino acids
Review I: Protein Structure Rajan Munshi BBSI @ Pitt 2005 Department of Computational Biology University of Pittsburgh School of Medicine May 24, 2005 Amino Acids Building blocks of proteins 20 amino acids
AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy
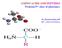 AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy Dr. Basima Sadiq Jaff PhD. Clinical Biochemistry MEDICAL AND BIOLOGICAL IMPORTANCE 1. building blocks of proteins. Some amino acids are found in
AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy Dr. Basima Sadiq Jaff PhD. Clinical Biochemistry MEDICAL AND BIOLOGICAL IMPORTANCE 1. building blocks of proteins. Some amino acids are found in
Short polymer. Dehydration removes a water molecule, forming a new bond. Longer polymer (a) Dehydration reaction in the synthesis of a polymer
 HO 1 2 3 H HO H Short polymer Dehydration removes a water molecule, forming a new bond Unlinked monomer H 2 O HO 1 2 3 4 H Longer polymer (a) Dehydration reaction in the synthesis of a polymer HO 1 2 3
HO 1 2 3 H HO H Short polymer Dehydration removes a water molecule, forming a new bond Unlinked monomer H 2 O HO 1 2 3 4 H Longer polymer (a) Dehydration reaction in the synthesis of a polymer HO 1 2 3
Metabolism of amino acids. Vladimíra Kvasnicová
 Metabolism of amino acids Vladimíra Kvasnicová Classification of proteinogenic AAs -metabolic point of view 1) biosynthesis in a human body nonessential (are synthesized) essential (must be present in
Metabolism of amino acids Vladimíra Kvasnicová Classification of proteinogenic AAs -metabolic point of view 1) biosynthesis in a human body nonessential (are synthesized) essential (must be present in
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and
 This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
