Executive Summary: 2016 Interventional Pain and Large Joint Services Guideline Updates
|
|
|
- Emily Stone
- 5 years ago
- Views:
Transcription
1 Executive Summary: 2016 Interventional Pain and Large Joint Services Guideline Updates On December 4, 2014, CareCore National and MedSolutions announced a merger that brought together two of the leading medical benefits management (MBM) companies into evicore healthcare. This combination allowed for two complementary businesses to jointly develop a broader portfolio of innovative and efficient solutions that improve quality of care and reduce medical costs. As a result, evicore healthcare is better positioned to enhance our health plan partners ability to control cost growth and improve health outcomes by ensuring their members receive appropriate, evidence-based care. At the outset of the merger, CareCore and MedSolutions each had legacy sets of proprietary and nationally recognized Interventional Pain and Large Joint Services guidelines. evicore has taken advantage of this overlap by merging their respective strengths into one set of harmonized guidelines. The results of that extensive harmonization process resulted in the forthcoming condition based patient/member centric guidelines. The publication of one set of guidelines improves the ability to maintain the highest quality, evidence-based clinical guidelines. The evicore guidelines undergo a formal review annually and are based upon major national and international association and society guidelines and criteria, peer-reviewed literature, major treatises and, input from health plans, practicing academic and community-based physicians. This executive summary represents the results of the most recent annual revision process. Presented in this executive summary are the material changes were made to the evicore Interventional Pain Management and Large Joint Services guidelines. "Material changes" are defined as changes that could greatly impact the approval or denial rat of specific, common requests. (Note: In the following summary, all new language for 2016 is presented in blue font. Language that has been removed for 2016 is crossed through). CMM 200: Epidural Steroid Injections CMM 201: Facet Joint Injections CMM 207: Epidural Adhesiolysis CMM 208: Radiofrequency Ablation CMM 210: Implantable Intrathecal Drug Delivery Systems CMM 311: Knee Arthroplasty The effective date of these changes is targeted for August 19,
2 CMM 200: Epidural Steroid Injections The definition of Radiculopathy was revised for further clarification. Radiculopathy definition for 2015: Radiculopathy must be documented by physical examination and should be corroborated with imaging studies and/or electrodiagnostic testing if performed. In cases with clearly evident radicular symptoms and correlating neurological findings on examination, imaging studies and/or electrodiagnostic testing is not necessary to clinically document a radiculopathy. The presence of leg pain or arm pain and possible findings on an advanced diagnostic imaging study in and of itself does not substantiate the diagnosis of radiculopathy. There must also be clinical evidence as described above. Radiculopathy definition for 2016: Radiculopathy, for the purpose of this policy, is defined as the presence of severe, disabling pain, dysaethesia(s) or paraesthesia(s) reported by the individual in a specified dermatomal distribution of an involved named spinal root(s) and ONE or MORE of the following: Loss of strength of specific named muscle(s) or myotomal distribution(s) demonstrated on detailed neurologic examination (within the prior 3 months) concordant with nerve root compression of the involved named spinal nerve root(s) Altered sensation to light touch, pressure, pin prick or temperature demonstrated on a detailed neurologic examination (within the prior 3 months) in the sensory distribution concordant with nerve root compression of the involved named spinal nerve root(s) Diminished, absent or asymmetric reflex(es) within the prior 3 months concordant with nerve root compression of the involved named spinal nerve root(s) Either of the following: o A concordant radiologist s interpretation of an advanced diagnostic imaging study (MRI or CT) of the spine demonstrating compression of the involved named spinal nerve root(s) (Performed within the prior 12 months) o Electrodiagnostic studies (EMG/NCV s) diagnostic of nerve root compression of the involved named spinal nerve root(s). (Performed within the prior 12 months). 2
3 CMM 201: Facet Joint Injections CMM 201: Diagnostic Facet Injections The guidelines were updated to allow for a second diagnostic facet joint injection/medial branch block, as follows: One diagnostic facet joint injection/medial branch block is considered medically necessary to determine whether chronic neck or back pain is of facet joint origin when ALL of the following criteria are met: Pain is exacerbated by extension and rotation, or is associated with lumbar rigidity Pain has persisted despite appropriate conservative treatment (e.g., nonsteroidal anti-inflammatory drugs (NSAIDs, exercise) Clinical findings and imaging studies suggest no other obvious cause of the pain (e.g., spinal stenosis, disc degeneration or herniation, infection, tumor, fracture). When there is greater than 80% pain relief from a single diagnostic facet joint injection/medial branch block, there is sufficient evidence of facet pathology and a second confirmatory block is unnecessary. A second diagnostic facet joint injection/medial branch block is medically necessary when the first diagnostic facet joint injection/medial branch block is positive. A second diagnostic facet joint injection/medial branch block is medically necessary no sooner than one week following the initial diagnostic facet joint injection/medial branch block Positive diagnostic medial branch block or facet joint injection using either a local anesthetic or a local anesthetic combined with corticosteroid as evidenced by either of the following: A beneficial clinical response to an intra-articular facet injection or medial branch block performed with a local anesthetic with greater than 80% pain relief reported for 50% of the duration of the effect of the local anesthetic used A beneficial clinical response to an intra-articular facet joint injection or medial branch block performed with a local anesthetic and a corticosteroid with at least a 50% reduction in pain for at least two (2) weeks. If the first diagnostic facet joint injection/medial branch block is not positive, then a second diagnostic facet joint injection/medial branch block is considered not medically necessary 3
4 CMM 201: References Added 1. Boswell MV, Manchikanti L, Kaye AD, et al. A best-evidence systematic appraisal of the 2. diagnostic accuracy and utility of facet (zygapophysial) joint injections in chronic spinal pain. Pain Physician 2015;18:E497-E Civelek E, Cansever T, Kabatas S, et al. Comparison of effectiveness of facet joint injection and radiofrequency denervation in chronic low back pain. Turk Neurosurg. 2012;22: Fuchs S, Erbe T, Fischer HL, Tibesku CO. Intraarticular hyaluronic acid versus glucocorticoidinjections for nonradicular pain in the lumbar spine. J Vasc Interv Radiol. 2005; 16: Lakemeier S, LInd M, Schultz W, Fuchs-Winkelmann S, Timmesfeld N, Foelsch C, Peterlein 6. CD. A comparison of intraarticular lumbar facet joint steroid injections and lumbar facet joint radiofrequency denervation in the treatment of low back pain: A randomized, controlled, doubleblind trial. Anesth Analg. 2013;117: Manchikanti L, Abdi S, Atluri S, et al. An update of comprehensive evidence-based guidelines for interventional techniques of chronic spinal pain: Part II: Guidance and recommendations. Pain Physician. 2013;16:S49-S Manchikanti L, Kaye AD, Boswell MV, et al. A systematic review and best evidence synthesis of the effectiveness of therapeutic facet joint interventions in managing chronic spinal pain. Pain Physician. 2015; 18:E535-E Manchikanti L, Singh V, Falco FJE, Cash KA, Pampati V. Evaluation of lumbar facet joint nerve blocks in managing chronic low back pain: A randomized, double-blind, controlled trial with a 2-year follow-up. Int J Med Sci. 2010;7: Manchikanti L, Singh V, Falco FJE, Cash KA, Fellows B. Comparative outcomes of a 2-year 11. follow-up of cervical medial branch blocks in management of chronic neck pain: A randomized, double-blind controlled trial. Pain Physician. 2010;13: Manchikanti L, Pampati V, Bakhit C, et al. Effectiveness of lumbar facet joint nerve blocks in chronic low back pain: A randomized clinical trial. Pain Physician. 2001;4: Manchikanti L, Singh V, Falco FJE, Cash KA, Pampati V, Fellows B. The role of thoracic medial branch locks in managing chronic mid and upper back pain: A randomized, double-blind, active control trial with a 2-year followup. Anesthesiol Res Pract. 2012;2012: Manchukonda R, Manchikanti K, Cash K, et al. Facet joint pain in chronic spinal pain: an evaluation of prevalence and false-positive rate of diagnostic blocks. J Spinal Disord Tech. 2007;20(7): Park SC, Kim KH. Effect of adding cervical facet joint injections in a multimodal treatment program for longstanding cervical myofascial pain syndrome with referral pain patterns of cervical facet joint syndrome. J Anesth. 2012;26: Ribeiro LH, Furtado RN, Konai MS, et al. Effect of facet joint injection versus systemic steroids in low back pain: A randomized controlled trial. Spine (Phila Pa 1976). 2013;38: Schneider G, Jull G, Smith A, Emery C, Faris P, Cook C, Frizzell B, Salo P. Derivation of a clinical decision guide in the diagnosis of cervical facet joint pain. Arch Phys Med Rehabil, 2014; 95(9): Yun DH, Kim HS, Yoo SD, Kim DH, Chon JM, Choi SH, Hwang DG, Jung PK. Efficacy of ultrasonographyguided injections in patients with facet syndrome of the low lumbar spine. Ann Rehabil Med. 2012;36:
5 CMM 207: Epidural Adhesiolysis CMM 207: Epidural Adhesiolysis Indications Previously, Epidural Adhesiolysis was approvable for limited clinical scenarios. evicore now considers this procedure as experimental, investigational or unproven as a treatment for back pain: There is insufficient scientific evidence to support the use of epidural adhesiolysis, performed by catheter or endoscopically, as a treatment for back pain. It is considered experimental, investigational or unproven. CMM 207: Epidural Adhesiolysis References Added 1. Choi E, Nahm FS, Lee PB. Evaluation of prognostic predictors of percutaneous Adhesiolysis using a Racz catheter for post lumbar surgery syndrome or spinal stenosis. Pain Physician. 2013; 16:E531-E Gerdesmeyer L, Wagenpfeil S, Birkenmaier C, et al. Percutaneous epidural lysis of adhesions in chronic lumbar radicular pain: A randomized double-blind placebo controlled trial. Pain Physician. 2013; 16: Helm II S, Benyamin RM, Chopra P, Deer TR, Justiz R. Percutaneous adhesiolysis in the 4. management of chronic low back pain in post lumbar surgery syndrome and spinal stenosis: A systematic review. Pain Physician. 2012; 15:E435-E Helm II S, Benyamin RM, Falco FJE. Refinement in evidence synthesis of percutaneousadhesiolysis. Pain Physician. 2013; 16: Helm II S, Hayek SM, Colson J, et al. Spinal endoscopic adhesiolysis in post lumbar surgerysyndrome: An update of the assessment of the evidence. Pain Physician. 2013; 16:SE125-SE Kim SH, Choi WJ, Suh JH, et al. Effects of transforaminal balloon treatment in patients with lumbar foraminal stenosis: A randomized, controlled, double-blind trial. Pain Physician. 2013; 16: Koh WU, Choi SS, Park WY, et al. Transforaminal hypertonic saline for the treatment of lumbar lateral canal stenosis: A double-blinded, randomized, active-control trial. Pain Physician. 2013; Manchikanti L, Abdi S, Atluri S, et al. An update of comprehensive evidence-based guidelines for interventional techniques of chronic spinal pain: Part II: Guidance and recommendations. Pain Physician. 2013;16:S49-S Manchikanti L, Cash KA, McManus CD, Pampati V. Assessment of effectiveness of 11. percutaneous adhesiolysis in managing chronic low back pain secondary to lumbar central spinal canal stenosis. Int J Med Sci. 2013; 10: Manchikanti L, Manchikanti KN, Gharibo CG, Kaye AD. Efficacy of percutaneous Adhesiolysis in the treatment of lumbar post surgery syndrome. Anesth Pain Med. 2015; in press. 13. Manchikanti L, Singh V, Cash KA, Pampati V, Datta S. Assessment of effectiveness of percutaneous adhesiolysis and caudal epidural injections in managing lumbar post surgery syndrome: A 2-year follow-up of randomized, controlled trial. J Pain Res. 2012; 5:
6 CMM 208: Radiofrequency Ablation CMM 208: Conservative Care The duration of conservative care was updated from 4 weeks of conservative care to 3 months. Failure of at least 4 weeks three (3) months of conservative care (e.g., exercise, physical methods including physical therapy, chiropractic care, NSAID s and/or analgesics) To align with the changes made to CMM 201: Facet Joint Injections, CMM 208: Radiofrequency Joint Ablation/Denervation was updated to state that radiofrequency ablation (RFA) of facet mediated pain is considered medically necessary if two sequential diagnostic facet joint injections/medial branch blocks are positive; if the two injections/blocks are not positive, then RFA is considered not medically necessary. Radiofrequency ablation of facet mediated pain is considered medically necessary if two sequential diagnostic facet joint injections/medial branch blocks are positive If the two sequential diagnostic facet joint injections/medial branch blocks are not positive, radiofrequency ablation of facet mediated pain is considered not medically necessary When an injection or block is considered positive, a second (confirmatory) block is not medically necessary to perform a radiofrequency joint denervation/ablation. CMM 208: References Added 1. Boswell MV, Manchikanti L, Kaye AD, et al. A best-evidence systematic appraisal of the 2. diagnostic accuracy and utility of facet (zygapophysial) joint injections in chronic spinal pain. Pain Physician. 2015;18:E497-E Civelek E, Cansever T, Kabatas S, et al. Comparison of effectiveness of facet joint injection and radiofrequency denervation in chronic low back pain. Turk Neurosurg. 2012; 22: Cohen SP, Williams KA, Kurihara C, et al. Multicenter, randomized, comparative costeffectiveness study comparing 0, 1, and 2 diagnostic medial branch (facet joint nerve) block treatment paradigms before lumbar facet radiofrequency denervation. Anesthesiology. 2010; 113: Dobrogowski J, Wrzosek A, Wordliczek J. Radiofrequency denervation with or without addition of pentoxifylline or methylprednisolone for chronic lumbar zygapophysial joint pain. Pharmacol Rep. 2005;57: Joo YC, Park JY, Kim KH. Comparison of alcohol ablation with repeated thermal radiofrequency ablation in medial branch neurotomy for the treatment of recurrent thoracolumbar facet joint pain. J Anesth. 2013; 27: Lakemeier S, Lind M, Schultz W, et al. A comparison of intraarticular lumbar facet joint steroid injections and lumbar facet joint radiofrequency denervation in the treatment of low back pain: A randomized, controlled, double-blind trial. Anesth Analg. 2013;117: Lord S, Barnsley L, Wallis B, McDonald G, Bogduk N. Percutaneous radio-frequency neurotomy for chronic cervical zygapophyseal-joint pain. N Engl J Med. 1996; 335: MacVicar J, Borowczyk JM, MacVicar AM, Loughnan BM, Bogduk N. Cervical medial branch radiofrequency neurotomy in New Zealand. Pain Med. 2012;13: Manchikanti L, Abdi S, Atluri S, et al. An update of comprehensive evidence-based guidelines for interventional techniques of chronic spinal pain: Part II: Guidance and recommendations. Pain Physician. 2013;16:S49-S Manchikanti L, Kaye AD, Boswell MV, et al. A systematic review and best evidence synthesis of the effectiveness of therapeutic facet joint interventions in managing chronic spinal pain. Pain Physician. 2015;18:E535-E Moon JY, Lee PB, Kim YC, Choi SP, Sim WS. An alternative distal approach for the lumbar 13. medial branch radiofrequency denervation: A prospective randomized comparative study. Anesth Analg. 2013;116: Sapir DA, Gorup JM. Radiofrequency medial branch neurotomy in litigant and non-litigant 15. patients with cervical whiplash. Spine (Phila Pa 1976). 2001;26:E268-E Speldewinde GC. Outcomes of percutaneous zygapophysial and sacroiliac joint neurotomy in a community setting. Pain Med. 2011;12:
7 CMM 210: Implantable Intrathecal Drug Delivery Systems CMM 210: Life Expectancy Criterion Previously, the criteria for trial with percutaneous intrathecal or epidural drug delivery system included the requirement that the patient have a life expectancy of greater than three (3) months. This criterion was removed from the 2016 guidelines for Implantable Intrathecal Drug Delivery Systems. CMM 210: Additional Indications Two additional indications were added to the guidelines for 2016, as follows: A trial with a percutaneous intrathecal drug delivery system for severe, refractory spasticity or chronic intractable dystonia is considered medically necessary when there is failure, contraindication or intolerance to at least a six-week trial of oral antispasmodic drugs and physical therapy. A trial with a percutaneous intrathecal or epidural drug delivery system for cancerrelated pain is considered medically necessary when there is failure, intolerance, or contraindication to noninvasive methods of pain control, including systemic opioids. 7
8 CMM 311: Knee Arthroplasty CMM 311: CPT Codes The following CPT codes were added to the Knee Arthoplasty guidelines: Removal of prosthesis, including total knee prosthesis, methylmethacrylate with or without insertion of spacer, knee Arthrodesis, knee, any technique CMM 311: Age and BMI Previously, patients had to be at least 50 years of age and have a Body Mass Index (BMI) of less than 40 to be considered for a partial knee arthroplasty. Age and BMI requirements were removed from these guidelines for CMM 311: Not Medically Necessary Criteria Three indications were added to the not medically necessary criteria for Partial (unicompartmental) knee arthroplasty, as follows: Partial (unicompartmental) knee arthroplasty is considered not medically necessary when any of the following criteria is present: Severe Grade III or IV patellofemoral joint arthritis (when unicompartmental arthroplasty to be performed is medial or lateral) Prior high tibial osteotomy Tibial or femoral shaft deformity Radiographic evidence of medial or lateral subluxation Flexion contracture greater than 15º Varus deformity greater than 15º or a valgus deformity greater than 20º Inflammatory arthropathy Active local or systemic infection Severe loss of musculature, neuromuscular compromise or vascular deficiency in the affected limb, rendering the procedure unjustifiable Osteoporosis or other osseous abnormalities which would make the likelihood of a poor outcome more probable Severe lack of collateral ligament integrity leading to joint instability. CMM 311: Simultaneous Bilateral Total Knee Replacement The following language was removed from the knee arthroplasty guidelines: Based on the increased risk of serious complications (cardiac complications, pulmonary complications and mortality) simultaneous bilateral total knee replacement may be considered not medically necessary. 8
9 CMM 311: References Added/Updated 1. American Academy of Orthopaedic Surgeons (AAOS). AAOS clinical guideline on osteoarthritis of the knee, 2nd edition Available at: 2. Berend KR, Lombardi AV Jr, Adams JB. Obesity, young age, patellofemoral disease, and anterior knee pain: identifying the unicondylar arthroplasty patient in the United States. Orthopedics, 2007; 30(5, suppl); Meneghini M. Revision Total Knee Arthroplasty. In Glassman AH, Lachiewicz PF, Tanzer M. eds. Orthopaedic Knowledge Update: Hip and Knee Reconstruction 4 th edition, 2011, American Academy of Orthopaedic Surgeons, Rosemont, IL. 4. Newman MT, Lonner JH, Ries M. Unicompartmental, patellofemoral, and bicompartmental arthroplasty. In Glassman AH, Lachiewicz PF, Tanzer M. eds. Orthopaedic Knowledge Update: Hip and Knee Reconstruction 4 th edition, 2011, American Academy of Orthopaedic Surgeons, Rosemont, IL. 5. Skou ST, Roos EM, Laursen MB, Rathleff MS et al. A randomized, controlled trial of total knee replacement.new England Journal of Medicine, 2015; 373(17):
10 CMM 312: Knee Surgery (Arthroscopic and Open) CPT Codes The following CPT Codes were added to CMM 312: Arthroscopy, knee, surgical; with meniscectomy (medial OR lateral, including any meniscal shaving) including debridement/shaving of articular cartilage (chondroplasty), same or separate compartment(s), when f d Arthroscopy, knee, surgical; with meniscus repair (medial OR lateral) Arthroscopy, knee, surgical; with meniscus repair (medial AND lateral) Arthroscopy, knee, surgical; drilling for osteochondritis dissecans with bone grafting, with or without internal fixation (including debridement of base of l i ) Arthroscopy, knee, surgical; drilling for intact osteochondritis dissecans lesion Arthroscopy, knee, surgical; drilling for intact osteochondritis dissecans lesion with internal fixation Arthroscopically aided anterior cruciate ligament repair/augmentation or t ti Arthroscopically aided posterior cruciate ligament repair/augmentation or reconstruction. CMM 312: Anterior Cruciate Ligament Reconstruction Guidelines were added for Anterior Cruciate Ligament Reconstruction: Allograft Knee Ligament Reconstruction - Knee ligament reconstruction (i.e., anterior cruciate) using allograft tissue is considered medically necessary for the treatment of ligament injury (e.g., rupture, laxity) when ANY of the following conditions is met: Previous reconstruction has failed and requires revision Surgical reconstruction requires the use of multiple ligament transfers Individual has a medical condition (e.g., anatomic anomaly, prior knee injury or prior knee surgery) that precludes the use of autograft tissue. Knee ligament reconstruction (i.e., anterior cruciate) using allograft tissue for ANY other indication not listed above is considered not medically necessary. Anterior cruciate ligament reconstruction with allograft (see above for allograft specific criteria) or autograft is considered medically necessary when ALL the following criteria have been met: Severe, disabling pain and a documented loss of knee function which interferes with the ability to carry out age appropriate activities of daily living and/or demands of employment Knee instability which is noted as giving way weakness, or buckling 10
11 MRI, Arthroscopy, or Arthrogram demonstrates a tear/disruption or significant laxity of the anterior cruciate ligament Positive Lachman s Test ANY of the following abnormal physical examination findings: o Positive Anterior Drawer Test o Positive Pivot Shift Test o Positive KT arthrometer (>3.5 mm =+1, >5-7 mm = +2, >7 mm =+3) Failure of non-surgical management for at least three (3) months in duration. Anterior cruciate ligament reconstruction with allograft (see above for allograft specific criteria) or autograft is considered medically necessary in an acute injury setting where hemathrosis, effusion, and joint instability have been documented. This may include ANY of the following: A confirmed ACL tear and a repairable meniscus tear Need to return to high demand activities that require cutting, pivoting, and/or agility activities in which ACL insufficiency may predispose to further instability episodes, that may result in new articular or meniscal cartilage injuries Concomitant ligament injuries (i.e., multiligamentous knee injury) that require reconstruction to provide stability. CMM 312: Posterior Cruciate Ligament Reconstruction Guidelines were added for Posterior Cruciate Ligament Reconstruction: Allograft Knee Ligament Reconstruction - Knee ligament reconstruction (i.e., posterior cruciate) using allograft tissue is considered medically necessary for the treatment of ligament injury (e.g., rupture, laxity) when ANY of the following conditions is met: Previous reconstruction has failed and requires revision Surgical reconstruction requires the use of multiple ligament transfers Individual has a medical condition (e.g., anatomic anomaly, prior knee injury or prior knee surgery) that precludes the use of autograft tissue. Knee ligament reconstruction (i.e., posterior cruciate) using allograft tissue for ANY other indication not listed above is considered not medically necessary. Posterior cruciate ligament reconstruction with allograft (see above for allograft specific criteria) or autograft is considered medically necessary when ALL the following criteria have been met: Severe, disabling pain and a documented loss of knee function to an extent which interferes with the ability to carry out the age appropriate activities of daily living and/or demands of employment Individual has undergone an MRI or Arthroscopy or Arthrogram which demonstrates a tear/disruption or significant laxity of the posterior cruciate ligament; 11
12 Individual demonstrates Positive Posterior Drawer Sign and/or positive Tibial Drop Back Test and/or Quadriceps Active Test either of the following abnormal physical examination findings: o Eight (8) millimeters or more of increased posterior translation on stress radiographs o Positive KT-1000 arthrometer (>7.6 mm of increased posterior translation) Failure of non-surgical care for at least three (3) months in duration Posterior cruciate ligament reconstruction with allograft (see above for allograft specific criteria) or autograft is considered medically necessary in an acute injury setting where hemathrosis, effusion and joint instability have been documented. This may include instances where there are concomitant ligament injuries (i.e., multiligamentous knee) that require reconstruction. CMM 312: Medial Collateral/Lateral Collateral Ligament Repair/Reconstruction Guidelines were added for Medial Collateral/Lateral Collateral Ligament Repair/Reconstruction: Allograft Knee Ligament Reconstruction - Knee ligament reconstruction (i.e., medial collateral, lateral collateral) using allograft tissue is considered medically necessary for the treatment of ligament injury (e.g., rupture, laxity) when ANY of the following conditions is met: Previous reconstruction has failed and requires revision Surgical reconstruction requires the use of multiple ligament transfers Individual has a medical condition (e.g., anatomic anomaly, prior knee injury or prior knee surgery) that precludes the use of autograft tissue. Knee ligament reconstruction (i.e., medial collateral, lateral collateral) using allograft tissue for ANY other indication not listed above is considered not medically necessary. Medial collateral/lateral collateral ligament repair with allograft (see above for allograft specific criteria) or autograft is considered medically necessary when ALL of the following criteria have been met: Severe, disabling pain Loss of knee function which interferes with the ability to carry out age appropriate activities of daily living and/or demands of employment Individual reports knee instability which is noted as giving way weakness or buckling MRI or other diagnostic study demonstrates a tear/disruption of the medial or lateral collateral ligament Positive Valgus Stress Test (Medial), or Varus Stress Test (Lateral) Failure of non-surgical management for at least six (6) weeks duration. Medial collateral or lateral collateral ligament repair/reconstruction with allograft or 12
13 autograft is considered medically necessary in an acute injury setting where total disruption of the ligament (i.e., multi-ligamentous knee injury) is documented on MRI examination and effusion and joint instability have been documented on physical examination. CMM 312: References Added 1. Crawford DC, Safran MR. Osteochondritis Dissecans of the knee. J Am Acad Orthop Surg, 2006; 14: Sherman SL, Garrity J, Bauer K, Cook J et al. Fresh osteochondral allograft transplantation for the knee: current concepts. J Am Acad Orthop Surg 2014 Feb; Vol. 22 (2), pp Graf K, Sekiya J, Wojitys E. Long-term results after combined medial meniscal allograft transplantation and anterior cruciate ligament reconstruction: Minimum 8.5-year follow-up study. Arthroscopy. 2004:20(2): Linko E, Harilainen A, Malmivaara A, Seitsalo S. Surgical versus conservative interventions for anterior cruciate ligament ruptures in adults. Cochrane Database Syst Rev Apr 18;(2):CD Peterson R, Shelton W, Bomboy A. Allograft versus autograft patellar tendon anterior cruciate ligament reconstruction: A 5-year follow-up. Arthroscopy. 2001;17(1): Sekiya J, Giffin J, Irrgang J, et al. Clinical outcomes after combined meniscal allograft transplantation and anterior cruciate ligament reconstruction. Am J Sports Med. 2003;31(6): Graf K, Sekiya J, Wojitys E. Long-term results after combined medial meniscal allograft transplantation and anterior cruciate ligament reconstruction: Minimum 8.5-year follow-up study. Arthroscopy. 2004:20(2): Biau D, Tournoux C, Katsahian Set al. Bone-patellar tendon-bone autografts versus hamstring autografts for reconstruction of anterior cruciate ligament: meta-analysis. BMJ. 2006;332(7548):
14 CMM 313: Hip Arthroplasty CMM 313: Age and BMI Previously, patients had to be at least 50 years of age and have a Body Mass Index (BMI) of less than 40 to be considered for partial and total hip arthroplasty. Age and BMI requirements were removed from these guidelines for CMM 313: Tonnis Grade 3 For Partial Hip Arthroplasty, Tonnis Grade 3 was added as a criterion: Partial hip arthroplasty is considered medically necessary when all of the following criteria have been met: Tonnis Grade 3 osteoarthritis History of chronic severe, disabling pain for at least six (6) months in duration Loss of hip function secondary to osteoarthritis which interferes with the ability to carry out age-appropriate activities of daily living and/or their demands of employment Failure of non-surgical management (e.g., ice, relative rest/activity modification, weight loss, bracing, medications [e.g., anti-inflammatories], injections [steroid] and/or physical therapy) for at least three months. CMM 313: References Added 1. Shimmin A, Beaule PE, Campbell P. Metal-on-metal hip resurfacing arthroplasty. J Bone Joint Surg Am, 2008; 90(3): Parker M, Gurusamy K, Azegami S. Arthroplasties (with and without bone cement) for proximal femoral fractures in adults. Cochrane Database Syst Rev. 2010, Issue 6. Art. No.: CD DOI: / CD pub4. 14
15 CMM 314: Additional Criteria CMM 314: Hip Arthroscopy This policy was reorganized into two sections: 1) indications for non-arthroscopic hip surgery and 2) indications for arthroscopic hip surgery. Additional criteria were added as follows: Non-arthroscopic hip surgery, Hip surgery either arthroscopic or open surgery is considered medically necessary for ANY of the following clinical situations: o Individual has experienced an acute fracture of the hip (femoral or acetabular) o Individual has a mal-union of a previous fracture o Individual has experienced an acute or post traumatic injury in which there is a correlation between examination and diagnostic imaging findings confirming a condition which is reasonably suspected of producing the individual s severe pain and limitation in function o Individual with persistent hip pain or dysfunction of a non-traumatic etiology for at least three (3) months in duration (e.g., avascular necrosis, loose bodies, dysplasia) o Tumor or infection o Femoroacetabular Impingement (FAI) Syndrome, including labral tear or synovial biopsy when an individual has ALL of the following criteria: Positive impingement sign (i.e., sudden pain on 90 degree hip flexion with adduction and internal rotation or extension and external rotation) Moderate to severe hip pain that is worsened by flexion activities (e.g. squatting or prolonged sitting) that significantly limits activities Unresponsive to at least 3 months of physician-directed non-surgical care Radiographic confirmation of FAI (e.g., pistol-grip deformity, alpha angle greater than 50 degrees, coxa profunda, and/or acetabular retroversion) Documented closure of the proximal femoral physis Documented absence of ALL of the following: Tönnis grade 2 osteoarthritis (i.e., small cysts in femoral head or acetabulum, increasing narrowing of joint space, moderate loss of sphericity of femoral head) Tönnis grade 3 osteoarthritis (i.e., large cysts, severe narrowing or obliteration of joint space, severe deformity of femoral head, avascular necrosis Joint space is less than 2 mm wide anywhere along the sourcil Arthroscopic hip surgery is considered medical necessary for ANY of the following clinical situations: o Femoroacetabular Impingement (FAI) Syndrome when an individual has ALL of the following criteria: Positive impingement sign (i.e., sudden pain on 90 degree hip flexion with adduction and internal rotation or extension and external rotation) Moderate to severe hip pain that is worsened by flexion activities (e.g. squatting or prolonged sitting) that significantly limits activities 15
16 Unresponsive to at least 3 months of physician-directed non-surgical care Radiographic confirmation of FAI (e.g., pistol-grip deformity, alpha angle greater than 50 degrees, coxa profunda, and/or acetabular retroversion) Documented closure of the proximal femoral physis Documented absence of ALL of the following: Tönnis grade 2 osteoarthritis (i.e., small cysts in femoral head or acetabulum, increasing narrowing of joint space, moderate loss of sphericity of femoral head) Tönnis grade 3 osteoarthritis (i.e., large cysts, severe narrowing or obliteration of joint space, severe deformity of femoral head, avascular necrosis Joint space is less than 2 mm wide anywhere along the sourcil o In conjunction with a periacetabular osteotomy o Labral pathology when an individual has ALL of the following criteria: Mechanical symptoms of the hip catching, locking or giving way An advanced diagnostic imaging study confirming labral pathology amenable to surgical management o Synovial biopsy o Irrigation and debridement of an intra-articular joint space infection o Removal of an ossific or osteochondral loose body confirmed radiographically o Arthroscopic hip surgery is considered experimental and investigational for all other indications. CMM 314: References Added 1. Gupta A, Redmond JM, Hammarstedt JE, Lindner D, Stake CE, Domb BG. Does obesity affect outcomes after hip arthroscopy? A cohort analysis. J Bone Joint Surg Am. 2015;97(1):
CLINICAL GUIDELINES. CMM-201 ~ Facet Joint Injections. Version 19.0 Effective August 11, 2017
 CLINICAL GUIDELINES CMM-201 ~ Facet Joint Injections Version 19.0 Effective August 11, 2017 evicore healthcare Clinical Decision Support Tool Diagnostic Strategies:This tool addresses common symptoms and
CLINICAL GUIDELINES CMM-201 ~ Facet Joint Injections Version 19.0 Effective August 11, 2017 evicore healthcare Clinical Decision Support Tool Diagnostic Strategies:This tool addresses common symptoms and
FACET JOINT INJECTIONS Version 18.0; Effective
 evicore healthcare. Clinical Decision Support Tool Diagnostic Strategies This tool addresses common symptoms and symptom complexes. Imaging requests for patients with atypical symptoms or clinical presentations
evicore healthcare. Clinical Decision Support Tool Diagnostic Strategies This tool addresses common symptoms and symptom complexes. Imaging requests for patients with atypical symptoms or clinical presentations
Cigna Medical Coverage Policies Musculoskeletal Facet Joint Injections/Medial Branch Blocks
 Cigna Medical Coverage Policies Musculoskeletal January 15, 2018 Instructions for use The following coverage policy applies to health benefit plans administered by Cigna. Coverage policies are intended
Cigna Medical Coverage Policies Musculoskeletal January 15, 2018 Instructions for use The following coverage policy applies to health benefit plans administered by Cigna. Coverage policies are intended
CLINICAL GUIDELINES. CMM-207: Epidural Adhesiolysis. Version Effective February 15, 2019
 CLINICAL GUIDELINES CMM-207: Version 1.0.2019 Effective February 15, 2019 Clinical guidelines for medical necessity review of speech therapy services. CMM-207: CMM-207.1: Definition 3 CMM-207.2: General
CLINICAL GUIDELINES CMM-207: Version 1.0.2019 Effective February 15, 2019 Clinical guidelines for medical necessity review of speech therapy services. CMM-207: CMM-207.1: Definition 3 CMM-207.2: General
CLINICAL GUIDELINES. CMM-201: Facet Joint Injections/Medial Branch Blocks. Version Effective October 22, 2018
 CLINICAL GUIDELINES CMM-201: Facet Joint Injections/Medial Branch Blocks Version 20.0.2018 Effective October 22, 2018 Clinical guidelines for medical necessity review of speech therapy services. CMM-201:
CLINICAL GUIDELINES CMM-201: Facet Joint Injections/Medial Branch Blocks Version 20.0.2018 Effective October 22, 2018 Clinical guidelines for medical necessity review of speech therapy services. CMM-201:
Cigna Medical Coverage Policies Musculoskeletal Facet Joint Injections/Medial Branch Blocks
 Cigna Medical Coverage Policies Musculoskeletal January 15, 2019 Instructions for use The following coverage policy applies to health benefit plans administered by Cigna. Coverage policies are intended
Cigna Medical Coverage Policies Musculoskeletal January 15, 2019 Instructions for use The following coverage policy applies to health benefit plans administered by Cigna. Coverage policies are intended
Cigna Medical Coverage Policies Musculoskeletal Radiofrequency Joint Ablation/Denervation
 Cigna Medical Coverage Policies Musculoskeletal Radiofrequency Joint Ablation/Denervation Effective January 1, 2016 Instructions for use The following coverage policy applies to health benefit plans administered
Cigna Medical Coverage Policies Musculoskeletal Radiofrequency Joint Ablation/Denervation Effective January 1, 2016 Instructions for use The following coverage policy applies to health benefit plans administered
evicore MSK joint surgery procedures requiring prior authorization
 evicore MSK joint surgery procedures requiring prior authorization Moda Health Commercial Group and Individual Members* Updated 1/30/2018 *Check EBT to verify member enrollment in evicore program Radiology
evicore MSK joint surgery procedures requiring prior authorization Moda Health Commercial Group and Individual Members* Updated 1/30/2018 *Check EBT to verify member enrollment in evicore program Radiology
FEP Medical Policy Manual
 FEP Medical Policy Manual Effective Date: April 15, 2018 Related Policies: 6.01.23 Diagnosis and Treatment of Sacroiliac Joint Pain 7.01.120 Facet Arthroplasty Facet Joint Denervation Description Percutaneous
FEP Medical Policy Manual Effective Date: April 15, 2018 Related Policies: 6.01.23 Diagnosis and Treatment of Sacroiliac Joint Pain 7.01.120 Facet Arthroplasty Facet Joint Denervation Description Percutaneous
Cigna Medical Coverage Policies Musculoskeletal Knee Arthroplasty Total and Partial
 Cigna Medical Coverage Policies Musculoskeletal Knee Arthroplasty Total and Partial Effective January 25, 2017 Instructions for use The following coverage policy applies to health benefit plans administered
Cigna Medical Coverage Policies Musculoskeletal Knee Arthroplasty Total and Partial Effective January 25, 2017 Instructions for use The following coverage policy applies to health benefit plans administered
DESCRIPTION OF PROCEDURE/SERVICE/PHARMACEUTICAL
 Subject: Percutaneous Epidural Adhesiolysis for Chronic Low Back Pain (RACZ Procedure) Original Effective Date: 10/12/15 Policy Number: MCP-257 Revision Date(s): Review Date: 12/16/15, 12/14/16, 6/22/17
Subject: Percutaneous Epidural Adhesiolysis for Chronic Low Back Pain (RACZ Procedure) Original Effective Date: 10/12/15 Policy Number: MCP-257 Revision Date(s): Review Date: 12/16/15, 12/14/16, 6/22/17
Cigna Medical Coverage Policies Musculoskeletal Knee Arthroplasty Total and Partial
 Cigna Medical Coverage Policies Musculoskeletal Knee Arthroplasty Total and Partial Effective February 1, 2016 Instructions for use The following coverage policy applies to health benefit plans administered
Cigna Medical Coverage Policies Musculoskeletal Knee Arthroplasty Total and Partial Effective February 1, 2016 Instructions for use The following coverage policy applies to health benefit plans administered
Hip, Knee and Shoulder Surgery
 Hip, Knee and Shoulder Surgery Policy Number: MM.06.030 Lines of Business: HMO; PPO; QUEST Integration; Medicare Advantage Section: Surgery Place(s) of Service: Outpatient; Inpatient Original Effective
Hip, Knee and Shoulder Surgery Policy Number: MM.06.030 Lines of Business: HMO; PPO; QUEST Integration; Medicare Advantage Section: Surgery Place(s) of Service: Outpatient; Inpatient Original Effective
MEDICAL POLICY STATEMENT INDIANA MEDICAID Original Issue Date Next Annual Review Effective Date 11/01/ /01/ /17/2017
 MEDICAL POLICY STATEMENT INDIANA MEDICAID Original Issue Date Next Annual Review Effective Date 11/01/2017 11/01/2018 12/17/2017 Policy Name Policy Number Policy Type MEDICAL Administrative Pharmacy Reimbursement
MEDICAL POLICY STATEMENT INDIANA MEDICAID Original Issue Date Next Annual Review Effective Date 11/01/2017 11/01/2018 12/17/2017 Policy Name Policy Number Policy Type MEDICAL Administrative Pharmacy Reimbursement
Guideline Number: NIA_CG_301 Last Revised Date: March 2018 Responsible Department: Clinical Operations
 Magellan Healthcare Clinical guidelines PARAVERTEBRAL FACET JOINT INJECTIONS OR BLOCKS (no U/S) CPT Codes: Cervical Thoracic Region: 64490 (+ 64491, +64492) Lumbar Sacral Region: 64493 (+64494, +64495)
Magellan Healthcare Clinical guidelines PARAVERTEBRAL FACET JOINT INJECTIONS OR BLOCKS (no U/S) CPT Codes: Cervical Thoracic Region: 64490 (+ 64491, +64492) Lumbar Sacral Region: 64493 (+64494, +64495)
Facet Joint Denervation. Populations Interventions Comparators Outcomes Individuals: With suspected facet joint pain
 Protocol Facet Joint Denervation (701116) Medical Benefit Effective Date: 01/01/16 Next Review Date: 11/18 Preauthorization Yes Review Dates: 09/09, 09/10, 07/11, 07/12, 05/13, 05/14, 01/15, 11/15, 11/16,
Protocol Facet Joint Denervation (701116) Medical Benefit Effective Date: 01/01/16 Next Review Date: 11/18 Preauthorization Yes Review Dates: 09/09, 09/10, 07/11, 07/12, 05/13, 05/14, 01/15, 11/15, 11/16,
MEDICAL POLICY SUBJECT: RADIOFREQUENCY FACET DENERVATION
 MEDICAL POLICY PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied.
MEDICAL POLICY PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied.
d EFFECTIVE DATE: POLICY LAST UPDATED:
 Medical Coverage Policy Epidural Injections for Pain Management d EFFECTIVE DATE: 04 01 2018 POLICY LAST UPDATED: 03 20 2018 OVERVIEW Epidural injections are generally performed to treat pain arising from
Medical Coverage Policy Epidural Injections for Pain Management d EFFECTIVE DATE: 04 01 2018 POLICY LAST UPDATED: 03 20 2018 OVERVIEW Epidural injections are generally performed to treat pain arising from
ACL Athletic Career. ACL Rupture - Warning Features Intensive pain Immediate swelling Locking Feel a Pop Dead leg Cannot continue to play
 FIMS Ambassador Tour to Eastern Europe, 2004 Belgrade, Serbia Montenegro Acute Knee Injuries - Controversies and Challenges Professor KM Chan OBE, JP President of FIMS Belgrade ACL Athletic Career ACL
FIMS Ambassador Tour to Eastern Europe, 2004 Belgrade, Serbia Montenegro Acute Knee Injuries - Controversies and Challenges Professor KM Chan OBE, JP President of FIMS Belgrade ACL Athletic Career ACL
MSK Covered Services. Musculoskeletal: Joint Metal-on-metal total hip resurfacing, including acetabular and femoral components
 CPT CODE S2118 MSK Covered Services Musculoskeletal: Joint Metal-on-metal total hip resurfacing, including acetabular and femoral components 23000 Removal of subdeltoid calcareous deposits, open 23020
CPT CODE S2118 MSK Covered Services Musculoskeletal: Joint Metal-on-metal total hip resurfacing, including acetabular and femoral components 23000 Removal of subdeltoid calcareous deposits, open 23020
Original Date: December 2015 Page 1 of 8 FOR CMS (MEDICARE) MEMBERS ONLY
 National Imaging Associates, Inc. Clinical guidelines TOTAL JOINT ARTHROPLASTY -Total Hip Arthroplasty -Total Knee Arthroplasty -Replacement/Revision Hip or Knee Arthroplasty CPT4 Codes: Please refer to
National Imaging Associates, Inc. Clinical guidelines TOTAL JOINT ARTHROPLASTY -Total Hip Arthroplasty -Total Knee Arthroplasty -Replacement/Revision Hip or Knee Arthroplasty CPT4 Codes: Please refer to
Evaluation and Management of Knee Pain. Michael Cassat, MD University of Arkansas for Medical Sciences
 Evaluation and Management of Knee Pain Michael Cassat, MD University of Arkansas for Medical Sciences Disclosure I have no actual or potential conflict of interest in relation to this program/presentation.
Evaluation and Management of Knee Pain Michael Cassat, MD University of Arkansas for Medical Sciences Disclosure I have no actual or potential conflict of interest in relation to this program/presentation.
5/31/2013. Disclosures. Lumbar Facet Joint Pain: Evidence. I have nothing to disclose
 Disclosures : Evidence I have nothing to disclose David J. Lee, MD Professor Pain Management Center Department of Anesthesia Facet Joint Pain Prevalence Spinal pain 54-80% lifetime 80-90% resolve in 6
Disclosures : Evidence I have nothing to disclose David J. Lee, MD Professor Pain Management Center Department of Anesthesia Facet Joint Pain Prevalence Spinal pain 54-80% lifetime 80-90% resolve in 6
2/5/2019. Facet Joint Pain. Biomechanics
 Facet Arthropathy as a Pain Source Evaluation and Management Shelby Spine Jan 31 st Feb 2 nd, 2019 Kushagra Verma MD, MS Adult and Pediatric Scoliosis And Spine Deformity Beach Orthopaedics Specialty Institute
Facet Arthropathy as a Pain Source Evaluation and Management Shelby Spine Jan 31 st Feb 2 nd, 2019 Kushagra Verma MD, MS Adult and Pediatric Scoliosis And Spine Deformity Beach Orthopaedics Specialty Institute
W. Dilworth Cannon, M.D. Professor of Clinical Orthopaedic Surgery University of California San Francisco
 Knee Pain And Injuries In Adults W. Dilworth Cannon, M.D. Professor of Clinical Orthopaedic Surgery University of California San Francisco Pain Control Overview Narcotics rarely necessary after 1 st 1-2
Knee Pain And Injuries In Adults W. Dilworth Cannon, M.D. Professor of Clinical Orthopaedic Surgery University of California San Francisco Pain Control Overview Narcotics rarely necessary after 1 st 1-2
Priorities Forum Statement GUIDANCE
 Priorities Forum Statement Number 21 Subject Knee Arthroscopy including arthroscopic knee washouts Date of decision November 2016 Date refreshed March 2017 Date of review November 2018 Osteoarthritis of
Priorities Forum Statement Number 21 Subject Knee Arthroscopy including arthroscopic knee washouts Date of decision November 2016 Date refreshed March 2017 Date of review November 2018 Osteoarthritis of
Medical Practice for Sports Injuries and Disorders of the Knee
 Sports-Related Injuries and Disorders Medical Practice for Sports Injuries and Disorders of the Knee JMAJ 48(1): 20 24, 2005 Hirotsugu MURATSU*, Masahiro KUROSAKA**, Tetsuji YAMAMOTO***, and Shinichi YOSHIDA****
Sports-Related Injuries and Disorders Medical Practice for Sports Injuries and Disorders of the Knee JMAJ 48(1): 20 24, 2005 Hirotsugu MURATSU*, Masahiro KUROSAKA**, Tetsuji YAMAMOTO***, and Shinichi YOSHIDA****
To: Manuel Suarez, M.D. Medical Director Neighborhood Health Plan Management Department 5757 Plaza Dr. Cyprus, CA Mailstop: CA
 To: Manuel Suarez, M.D. Medical Director Neighborhood Health Plan Management Department 5757 Plaza Dr. Cyprus, CA 30630 Mailstop: CA124-01290 Subject: Denial of Cervical Spinal Injection Procedures Dear
To: Manuel Suarez, M.D. Medical Director Neighborhood Health Plan Management Department 5757 Plaza Dr. Cyprus, CA 30630 Mailstop: CA124-01290 Subject: Denial of Cervical Spinal Injection Procedures Dear
MEDICAL POLICY MEDICAL POLICY DETAILS POLICY STATEMENT. Page: 1 of 5
 Page: 1 of 5 MEDICAL POLICY MEDICAL POLICY DETAILS Medical Policy Title RADIOFREQUENCY JOINT ABLATION/DENERVATION Policy Number 7.01.42 Category Technology Assessment Effective Date 10/18/01 Revised Date
Page: 1 of 5 MEDICAL POLICY MEDICAL POLICY DETAILS Medical Policy Title RADIOFREQUENCY JOINT ABLATION/DENERVATION Policy Number 7.01.42 Category Technology Assessment Effective Date 10/18/01 Revised Date
MEDICAL POLICY EFFECTIVE DATE: 08/15/13 REVISED DATE: 07/17/14 SUBJECT: SPINAL INJECTIONS (EPIDURAL AND FACET INJECTIONS) FOR PAIN MANAGEMENT
 MEDICAL POLICY SUBJECT: SPINAL INJECTIONS (EPIDURAL AND PAGE: 1 OF: 7 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases,
MEDICAL POLICY SUBJECT: SPINAL INJECTIONS (EPIDURAL AND PAGE: 1 OF: 7 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases,
For Commercial products, please refer to the following policy: Preauthorization via Web-Based Tool for Procedures
 Medical Coverage Policy Total Joint Arthroplasty Hip and Knee EFFECTIVE DATE: 08/01/2017 POLICY LAST UPDATED: 06/06/2017 OVERVIEW Joint replacement surgery, also known as arthroplasty, has proved to be
Medical Coverage Policy Total Joint Arthroplasty Hip and Knee EFFECTIVE DATE: 08/01/2017 POLICY LAST UPDATED: 06/06/2017 OVERVIEW Joint replacement surgery, also known as arthroplasty, has proved to be
Re: Treatment Modalities for Facetogenic Pain, Policy #141
 March 30, 2017 Blue Cross and Blue Shield of Alabama Attn: Health Management - Medical Policy P.O. Box 995 Birmingham, AL 35298-0001 Fax: 205-220-0878 Re: Treatment Modalities for Facetogenic Pain, Policy
March 30, 2017 Blue Cross and Blue Shield of Alabama Attn: Health Management - Medical Policy P.O. Box 995 Birmingham, AL 35298-0001 Fax: 205-220-0878 Re: Treatment Modalities for Facetogenic Pain, Policy
Orthopedic Coding Changes for 2012
 Orthopedic Coding Changes for Lynn M. Anderanin, CPC,CPC-I, COSC Vertebroplasty 22520- Percutaneous vertebroplasty, 1 vertebral body, unilateral or bilateral injection; thoracic 22520- Percutaneous vertebroplasty,
Orthopedic Coding Changes for Lynn M. Anderanin, CPC,CPC-I, COSC Vertebroplasty 22520- Percutaneous vertebroplasty, 1 vertebral body, unilateral or bilateral injection; thoracic 22520- Percutaneous vertebroplasty,
Spinal and Trigger Point Injections
 Spinal and Trigger Point Injections I. Policy University Health Alliance (UHA) will reimburse for nonsurgical interventional treatment for subacute and chronic spinal pain when determined to be medically
Spinal and Trigger Point Injections I. Policy University Health Alliance (UHA) will reimburse for nonsurgical interventional treatment for subacute and chronic spinal pain when determined to be medically
Lysis of Epidural Adhesions
 Medical Policy Manual Surgery, Policy No. 94 Lysis of Epidural Adhesions Next Review: September 2019 Last Review: October 2018 Effective: November 1, 2018 IMPORTANT REMINDER Medical Policies are developed
Medical Policy Manual Surgery, Policy No. 94 Lysis of Epidural Adhesions Next Review: September 2019 Last Review: October 2018 Effective: November 1, 2018 IMPORTANT REMINDER Medical Policies are developed
MEDICAL POLICY SUBJECT: RADIOFREQUENCY JOINT ABLATION / DENERVATION
 MEDICAL POLICY SUBJECT: RADIOFREQUENCY JOINT PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
MEDICAL POLICY SUBJECT: RADIOFREQUENCY JOINT PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
Nonsurgical Interventional Treatments for Spinal Pain Management
 Nonsurgical Interventional Treatments for Spinal Pain Management I. Policy University Health Alliance (UHA) will reimburse for nonsurgical interventional treatment for subacute and chronic spinal pain
Nonsurgical Interventional Treatments for Spinal Pain Management I. Policy University Health Alliance (UHA) will reimburse for nonsurgical interventional treatment for subacute and chronic spinal pain
Overview Ligament Injuries. Anatomy. Epidemiology Very commonly injured joint. ACL Injury 20/06/2016. Meniscus Tears. Patellofemoral Problems
 Overview Ligament Injuries Meniscus Tears Pankaj Sharma MBBS, FRCS (Tr & Orth) Consultant Orthopaedic Surgeon Manchester Royal Infirmary Patellofemoral Problems Knee Examination Anatomy Epidemiology Very
Overview Ligament Injuries Meniscus Tears Pankaj Sharma MBBS, FRCS (Tr & Orth) Consultant Orthopaedic Surgeon Manchester Royal Infirmary Patellofemoral Problems Knee Examination Anatomy Epidemiology Very
Mr Aslam Mohammed FRCS, FRCS (Orth) Consultant Orthopaedic Surgeon Specialising in Lower Limb Arthroplasty and Sports Injury
 Mr Aslam Mohammed FRCS, FRCS (Orth) Consultant Orthopaedic Surgeon Specialising in Lower Limb Arthroplasty and Sports Injury I qualified from the Welsh National School of Medicine in Cardiff in 1984. I
Mr Aslam Mohammed FRCS, FRCS (Orth) Consultant Orthopaedic Surgeon Specialising in Lower Limb Arthroplasty and Sports Injury I qualified from the Welsh National School of Medicine in Cardiff in 1984. I
Clinical Policy: Lysis of Epidural Lesions Reference Number: CP.MP.116
 Clinical Policy: Reference Number: CP.MP.116 Effective Date: 07/16 Last Review Date: 07/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.116 Effective Date: 07/16 Last Review Date: 07/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Contents SECTION 1: GENERAL TRAUMA AND RECONSTRUCTIVE HIP SURGERY
 SECTION 1: GENERAL TRAUMA AND RECONSTRUCTIVE HIP SURGERY 1. Acetabular and Pelvic Fractures...3 2. Acetabular Orientation (Total Hips)...6 3. Acetabular Osteotomy...7 4. Achilles Tendon Ruptures...9 5.
SECTION 1: GENERAL TRAUMA AND RECONSTRUCTIVE HIP SURGERY 1. Acetabular and Pelvic Fractures...3 2. Acetabular Orientation (Total Hips)...6 3. Acetabular Osteotomy...7 4. Achilles Tendon Ruptures...9 5.
See Policy CPT/HCPCS CODE section below for any prior authorization requirements
 Effective Date: 7/1/2018 Section: MED Policy No: 123 Medical Officer 7/1/18 Date Technology Assessment Committee Approved Date: 10/10; 12/15 Medical Policy Committee Approved Date: 8/94; 7/96; 8/97; 4/98;
Effective Date: 7/1/2018 Section: MED Policy No: 123 Medical Officer 7/1/18 Date Technology Assessment Committee Approved Date: 10/10; 12/15 Medical Policy Committee Approved Date: 8/94; 7/96; 8/97; 4/98;
FAI syndrome with or without labral tear.
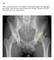 Case This 16-year-old female, soccer athlete was treated for pain in the right groin previously. Now has acute onset of pain in the left hip. The pain was in the groin that was worse with activities. Diagnosis
Case This 16-year-old female, soccer athlete was treated for pain in the right groin previously. Now has acute onset of pain in the left hip. The pain was in the groin that was worse with activities. Diagnosis
Clinical Policy: Selective Nerve Root Blocks and Transforaminal Epidural Steroid Injections
 Clinical Policy: Selective Nerve Root Blocks and Transforaminal Epidural Steroid Injections Reference Number: PA.CP.MP.165 Effective Date: 09/18 Last Review Date: 09/18 Coding Implications Revision Log
Clinical Policy: Selective Nerve Root Blocks and Transforaminal Epidural Steroid Injections Reference Number: PA.CP.MP.165 Effective Date: 09/18 Last Review Date: 09/18 Coding Implications Revision Log
Shoulder Subacromial Decompression. 15 CPT & Coding Issues for Orthopedic & Spine ASC Facilities. 15 CPT & Coding Issues for Orthopedics and Spine
 Orthopedics and Spine 12th Annual Orthopedic, Spine & Pain Management- Driven ASC The Future of Spine Conference by Becker s ASC Review & Becker s Spine Review Speaker Stephanie Ellis, R.N., CPC Ellis
Orthopedics and Spine 12th Annual Orthopedic, Spine & Pain Management- Driven ASC The Future of Spine Conference by Becker s ASC Review & Becker s Spine Review Speaker Stephanie Ellis, R.N., CPC Ellis
Humber. Arthroscopy Knee
 Humber Arthroscopy Knee Intervention Diagnostic & Therapeutic Arthroscopy Knee OPCS Codes W85 Therapeutic endoscopic operations on cavity of knee joint W851 Endoscopic removal of loose body from knee joint
Humber Arthroscopy Knee Intervention Diagnostic & Therapeutic Arthroscopy Knee OPCS Codes W85 Therapeutic endoscopic operations on cavity of knee joint W851 Endoscopic removal of loose body from knee joint
Vijay Singh, M.D Roosevelt Rd., Niagara, WI 54151
 Publications and Book Chapters Vijay Singh, M.D. 1601 Roosevelt Rd., Niagara, WI 54151 PUBLICATIONS Jul-Aug 2012 Jul-Aug 2012 Jul-Aug 2012 Jul-Aug 2012 Jul-Aug 2012 July 2012 July 2012 May-Jun 2012 May-Jun
Publications and Book Chapters Vijay Singh, M.D. 1601 Roosevelt Rd., Niagara, WI 54151 PUBLICATIONS Jul-Aug 2012 Jul-Aug 2012 Jul-Aug 2012 Jul-Aug 2012 Jul-Aug 2012 July 2012 July 2012 May-Jun 2012 May-Jun
MEDICAL POLICY EFFECTIVE DATE: 08/15/13 REVISED DATE: 07/17/14, 06/18/15, 05/25/16, 05/18/17
 MEDICAL POLICY SUBJECT: SPINAL INJECTIONS (EPIDURAL AND PAGE: 1 OF: 10 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product,
MEDICAL POLICY SUBJECT: SPINAL INJECTIONS (EPIDURAL AND PAGE: 1 OF: 10 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product,
Surgical Treatment for Sacroiliac Joint Pain
 Surgical Treatment for Sacroiliac Joint Pain Policy Number: 6.01.23 Last Review: 5/2014 Origination: 5/2013 Next Review: 5/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
Surgical Treatment for Sacroiliac Joint Pain Policy Number: 6.01.23 Last Review: 5/2014 Origination: 5/2013 Next Review: 5/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
Evaluation and Treatment of Knee Arthritis Classification of Knee Arthritis Osteoarthritis Osteoarthritis Osteoarthritis of Knee
 1 2 Evaluation and Treatment of Knee Arthritis John Zebrack, MD Reno Orthopaedic Clinic Classification of Knee Arthritis Non-inflammatory Osteoarthritis Primary Secondary Post-traumatic, dysplasia, neuropathic,
1 2 Evaluation and Treatment of Knee Arthritis John Zebrack, MD Reno Orthopaedic Clinic Classification of Knee Arthritis Non-inflammatory Osteoarthritis Primary Secondary Post-traumatic, dysplasia, neuropathic,
I have nothing to disclose
 Management of Common Knee Disorders: What You Knee d to Know UCSF Essentials of Women s Health July 8, 2015 Carlin Senter, M.D. I have nothing to disclose Learning objectives: in 1 hour you will be able
Management of Common Knee Disorders: What You Knee d to Know UCSF Essentials of Women s Health July 8, 2015 Carlin Senter, M.D. I have nothing to disclose Learning objectives: in 1 hour you will be able
Osteoarthritis. Dr Anthony Feher. With special thanks to Dr. Tim Williams and Dr. Bhatia for allowing me to use some of their slides
 Osteoarthritis Dr Anthony Feher With special thanks to Dr. Tim Williams and Dr. Bhatia for allowing me to use some of their slides No Financial Disclosures Number one chronic disability in the United States
Osteoarthritis Dr Anthony Feher With special thanks to Dr. Tim Williams and Dr. Bhatia for allowing me to use some of their slides No Financial Disclosures Number one chronic disability in the United States
SHOULDER ARTHROPLASTY (TOTAL, HEMI, REVERSE)/ARTHRODESIS
 evicore healthcare. Clinical Decision Support Tool Diagnostic Strategies This tool addresses common symptoms and symptom complexes. Imaging requests for patients with atypical symptoms or clinical presentations
evicore healthcare. Clinical Decision Support Tool Diagnostic Strategies This tool addresses common symptoms and symptom complexes. Imaging requests for patients with atypical symptoms or clinical presentations
Clinical Policy Title: Spine pain facet joint injections
 Clinical Policy Title: Spine pain facet joint injections Clinical Policy Number: 03.02.07 Effective Date: April 1, 2016 Initial Review Date: June 16, 2013 Most Recent Review Date: January 11, 2018 Next
Clinical Policy Title: Spine pain facet joint injections Clinical Policy Number: 03.02.07 Effective Date: April 1, 2016 Initial Review Date: June 16, 2013 Most Recent Review Date: January 11, 2018 Next
EPIDURAL STEROID AND FACET INJECTIONS FOR SPINAL PAIN
 EPIDURAL STEROID AND FACET INJECTIONS FOR SPINAL PAIN UnitedHealthcare Oxford Clinical Policy Policy Number: PAIN 019.21 T2 Effective Date: October 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE...
EPIDURAL STEROID AND FACET INJECTIONS FOR SPINAL PAIN UnitedHealthcare Oxford Clinical Policy Policy Number: PAIN 019.21 T2 Effective Date: October 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE...
Mr Simon Jennings BSc, MB BS, FRCS, Dip Sports Med FRCS (Trauma & Orthopaedics)
 Mr Simon Jennings BSc, MB BS, FRCS, Dip Sports Med FRCS (Trauma & Orthopaedics) Consultant Orthopaedic Surgeon Northwick Park Hospital 107 Harley Street RSM 16 th September 2010 Orthopaedic Surgeon Knee
Mr Simon Jennings BSc, MB BS, FRCS, Dip Sports Med FRCS (Trauma & Orthopaedics) Consultant Orthopaedic Surgeon Northwick Park Hospital 107 Harley Street RSM 16 th September 2010 Orthopaedic Surgeon Knee
Cigna Medical Coverage Policies Musculoskeletal Shoulder Arthroplasty (Total, Hemi, Reverse)/Arthrodesis
 Cigna Medical Coverage Policies Musculoskeletal Shoulder Arthroplasty (Total, Hemi, Reverse)/Arthrodesis Effective January 1, 2016 Instructions for use The following coverage policy applies to health benefit
Cigna Medical Coverage Policies Musculoskeletal Shoulder Arthroplasty (Total, Hemi, Reverse)/Arthrodesis Effective January 1, 2016 Instructions for use The following coverage policy applies to health benefit
Dupuytrens contracture
 OA Wrist Ganglion/Cysts Dupuytrens contracture Carpal Tunnel Syndrome Carpal Tunnel pathway For advice on management of CTS please follow link to Map of Medicine Trigger Finger Trigger finger pathway For
OA Wrist Ganglion/Cysts Dupuytrens contracture Carpal Tunnel Syndrome Carpal Tunnel pathway For advice on management of CTS please follow link to Map of Medicine Trigger Finger Trigger finger pathway For
SOFT TISSUE INJURIES OF THE KNEE: Primary Care and Orthopaedic Management
 SOFT TISSUE INJURIES OF THE KNEE: Primary Care and Orthopaedic Management Gauguin Gamboa Australia has always been a nation where emphasis on health and fitness has resulted in an active population engaged
SOFT TISSUE INJURIES OF THE KNEE: Primary Care and Orthopaedic Management Gauguin Gamboa Australia has always been a nation where emphasis on health and fitness has resulted in an active population engaged
Human ACL reconstruction
 Human ACL reconstruction current state of the art Rudolph Geesink MD PhD Maastricht The Netherlands Human or canine ACL repair...!? ACL anatomy... right knees! ACL double bundles... ACL double or triple
Human ACL reconstruction current state of the art Rudolph Geesink MD PhD Maastricht The Netherlands Human or canine ACL repair...!? ACL anatomy... right knees! ACL double bundles... ACL double or triple
KNEE ARTHROPLASTY- TOTAL AND PARTIAL
 evicore healthcare. Clinical Decision Support Tool Diagnostic Strategies This tool addresses common symptoms and symptom complexes. Imaging requests for patients with atypical symptoms or clinical presentations
evicore healthcare. Clinical Decision Support Tool Diagnostic Strategies This tool addresses common symptoms and symptom complexes. Imaging requests for patients with atypical symptoms or clinical presentations
Phillip Weidner, DO, DABA, BCPM May 20th, 2014
 Phillip Weidner, DO, DABA, BCPM May 20th, 2014 Introduction Comprehensive Pain Management Center Interventional therapies Epidural steroid injections Sacroiliac interventions Facet joint disease Who to
Phillip Weidner, DO, DABA, BCPM May 20th, 2014 Introduction Comprehensive Pain Management Center Interventional therapies Epidural steroid injections Sacroiliac interventions Facet joint disease Who to
MEDICAL POLICY SUBJECT: SPINAL INJECTIONS (EPIDURAL AND FACET INJECTIONS) FOR PAIN MANAGEMENT
 MEDICAL POLICY SUBJECT: SPINAL INJECTIONS (EPIDURAL AND PAGE: 1 OF: 10 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product
MEDICAL POLICY SUBJECT: SPINAL INJECTIONS (EPIDURAL AND PAGE: 1 OF: 10 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product
ACL AND PCL INJURIES OF THE KNEE JOINT
 ACL AND PCL INJURIES OF THE KNEE JOINT Dr.KN Subramanian M.Ch Orth., FRCS (Tr & Orth), CCT Orth(UK) Consultant Orthopaedic Surgeon, Special interest: Orthopaedic Sports Injury, Shoulder and Knee Surgery,
ACL AND PCL INJURIES OF THE KNEE JOINT Dr.KN Subramanian M.Ch Orth., FRCS (Tr & Orth), CCT Orth(UK) Consultant Orthopaedic Surgeon, Special interest: Orthopaedic Sports Injury, Shoulder and Knee Surgery,
Interventional Pain Management
 Origination: 5/21/08 Revised: 10/02/17 Annual Review: 11/02/17 Purpose: To provide interventional pain management clinical coordination criteria for the Medical Department staff to reference when making
Origination: 5/21/08 Revised: 10/02/17 Annual Review: 11/02/17 Purpose: To provide interventional pain management clinical coordination criteria for the Medical Department staff to reference when making
Bone Bangalore
 Dr Suresh Annamalai MBBS, MRCS(Edn), FRCS( Tr & Orth)(Edn), FEBOT(European Board), Young Hip and Knee Fellowship(Harrogate, UK) HOD & Consultant Arthroplasty and Arthroscopic Surgeon Manipal Hospital,
Dr Suresh Annamalai MBBS, MRCS(Edn), FRCS( Tr & Orth)(Edn), FEBOT(European Board), Young Hip and Knee Fellowship(Harrogate, UK) HOD & Consultant Arthroplasty and Arthroscopic Surgeon Manipal Hospital,
On Field Assessment and Management of Acute Knee Injuries: A Physiotherapist s Perspective
 On Field Assessment and Management of Acute Knee Injuries: A Physiotherapist s Perspective Jessica Condliffe Physiotherapist / Clinic Manager TBI Health Wellington Presentation Outline Knee anatomy review
On Field Assessment and Management of Acute Knee Injuries: A Physiotherapist s Perspective Jessica Condliffe Physiotherapist / Clinic Manager TBI Health Wellington Presentation Outline Knee anatomy review
Anterior Cruciate Ligament Surgery
 Anatomy Anterior Cruciate Ligament Surgery Roger Ostrander, MD Andrews Institute Anatomy Anatomy Function Primary restraint to anterior tibial translation Secondary restraint to internal tibial rotation
Anatomy Anterior Cruciate Ligament Surgery Roger Ostrander, MD Andrews Institute Anatomy Anatomy Function Primary restraint to anterior tibial translation Secondary restraint to internal tibial rotation
Re: Occipital Neuralgia and Headache Treatment, Policy Number: 2018T0080Y
 May 10, 2018 United Healthcare Medical Policy Department 9500 Bren Road East Minnetonka, MN 55343 via Email: mpq@uhc.com Re: Occipital Neuralgia and Headache Treatment, Policy Number: 2018T0080Y To Whom
May 10, 2018 United Healthcare Medical Policy Department 9500 Bren Road East Minnetonka, MN 55343 via Email: mpq@uhc.com Re: Occipital Neuralgia and Headache Treatment, Policy Number: 2018T0080Y To Whom
Ideal Candidate for Cartilage Restoration. Large or Complex Lesions
 Complex Biological Knee Reconstruction: Bipolar, Multifocal Lesions and Osteoarthritis William Bugbee, MD Attending Physician, Scripps Clinic 18 th International Sports Medicine Fellow s Conference Ideal
Complex Biological Knee Reconstruction: Bipolar, Multifocal Lesions and Osteoarthritis William Bugbee, MD Attending Physician, Scripps Clinic 18 th International Sports Medicine Fellow s Conference Ideal
Oh My Aching Knee. Oh My Aching Knee WHO AM I? 10/15/2012. Jan Pieter Hommen, MD Orthopedic Surgeon Sports Medicine Arthroscopy Joint Replacements
 Oh My Aching Knee Jan Pieter Hommen, MD Orthopedic Surgeon Sports Medicine Arthroscopy Joint Replacements Oh My Aching Knee Jan Pieter Hommen, MD Orthopedic Surgeon Sports Medicine Arthroscopy Joint Replacements
Oh My Aching Knee Jan Pieter Hommen, MD Orthopedic Surgeon Sports Medicine Arthroscopy Joint Replacements Oh My Aching Knee Jan Pieter Hommen, MD Orthopedic Surgeon Sports Medicine Arthroscopy Joint Replacements
Clinical Policy: Caudal or Interlaminar Epidural Steroid Injections
 Clinical Policy: Reference Number: PA.CP.MP.164 Effective Date: 09/18 Last Review Date: 09/18 Coding Implications Revision Log Description Epidural steroid injections have been used for pain control in
Clinical Policy: Reference Number: PA.CP.MP.164 Effective Date: 09/18 Last Review Date: 09/18 Coding Implications Revision Log Description Epidural steroid injections have been used for pain control in
Corporate Medical Policy
 Corporate Medical Policy Epidural Steroid Injections for Back Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: epidural_steroid_injections_for_back_pain 2/2016 4/2017 4/2018
Corporate Medical Policy Epidural Steroid Injections for Back Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: epidural_steroid_injections_for_back_pain 2/2016 4/2017 4/2018
October 1999, Supplement 1 Volume 15 Number 7
 October 1999, Supplement 1 Volume 15 Number 7
October 1999, Supplement 1 Volume 15 Number 7
Knee Dislocation: Spectrum of Injury, Evolution of Treatment & Modern Outcomes
 Knee Dislocation: Spectrum of Injury, Evolution of Treatment & Modern Outcomes William M Weiss, MD MSc FRCSC Orthopedic Surgery & Rehabilitation Sports Medicine, Arthroscopy & Extremity Reconstruction
Knee Dislocation: Spectrum of Injury, Evolution of Treatment & Modern Outcomes William M Weiss, MD MSc FRCSC Orthopedic Surgery & Rehabilitation Sports Medicine, Arthroscopy & Extremity Reconstruction
Additions: lumbar spine/spondy. spondy. panners? Elbow dislocation?
 Additions: lumbar spine/spondy spondy panners? Elbow dislocation? 16 y.o.. female swimmer has had right shoulder pain for the past 6 months. Ultrasound treatment and rest have failed to provide relief.
Additions: lumbar spine/spondy spondy panners? Elbow dislocation? 16 y.o.. female swimmer has had right shoulder pain for the past 6 months. Ultrasound treatment and rest have failed to provide relief.
Behnam Hosseini, Mohammad Hossein Ataei, Sirous Momenzadeh, Reza Jalili Khoshnood, Davood Ommi
 Original Article The comparison between steroid and hypertonic saline 10% with steroid in transforaminal epidural injection in patients with unilateral foraminal stenosis Behnam Hosseini, Mohammad Hossein
Original Article The comparison between steroid and hypertonic saline 10% with steroid in transforaminal epidural injection in patients with unilateral foraminal stenosis Behnam Hosseini, Mohammad Hossein
Adult Reconstruction Hip Education Tracks
 Adult Reconstruction Hip Education Tracks Adult Reconstruction Hip Track for the Specialist - HIP1 ICL 281 A Case-based Approach to High Risk Total Hip - When Do I Do Something Differently? ICL 241 The
Adult Reconstruction Hip Education Tracks Adult Reconstruction Hip Track for the Specialist - HIP1 ICL 281 A Case-based Approach to High Risk Total Hip - When Do I Do Something Differently? ICL 241 The
Labral Tears/FAI. Andrew Parker, MD
 Labral Tears/FAI Andrew Parker, MD Athletic Hip Injuries Incidence of hip injuries has increased dramatically over the last decade In part due to better recognition with improved imaging and arthroscopy,
Labral Tears/FAI Andrew Parker, MD Athletic Hip Injuries Incidence of hip injuries has increased dramatically over the last decade In part due to better recognition with improved imaging and arthroscopy,
Treatment of meniscal lesions and isolated lesions of the anterior cruciate ligament of the knee in adults
 QUICK REFERENCE GUIDE Treatment of meniscal s and isolated s of the anterior cruciate ligament of the knee in adults June 2008 AIM OF THE GUIDELINES To encourage good practices in the areas of meniscal
QUICK REFERENCE GUIDE Treatment of meniscal s and isolated s of the anterior cruciate ligament of the knee in adults June 2008 AIM OF THE GUIDELINES To encourage good practices in the areas of meniscal
Medical Affairs Policy
 Medical Affairs Policy Service: Back Pain Procedures-Epidural Injection (Caudal Epidural, Selective Nerve Root Block, Interlaminar, Transforaminal, Translaminar Epidural Injection) PUM 250-0015-1706 Medical
Medical Affairs Policy Service: Back Pain Procedures-Epidural Injection (Caudal Epidural, Selective Nerve Root Block, Interlaminar, Transforaminal, Translaminar Epidural Injection) PUM 250-0015-1706 Medical
CMM-201~Facet Joint Injections- Medical Branch Blocks
 MedSolutions, Inc. Clinical Decision Support Tool Diagnostic Strategies This tool addresses common symptoms and symptom complexes. Requests for patients with atypical symptoms or clinical presentations
MedSolutions, Inc. Clinical Decision Support Tool Diagnostic Strategies This tool addresses common symptoms and symptom complexes. Requests for patients with atypical symptoms or clinical presentations
Management of lumbar zygapophysial (facet) joint pain
 Submit a Manuscript: http://www.wjgnet.com/esps/ Help Desk: http://www.wjgnet.com/esps/helpdesk.aspx DOI: 10.5312/wjo.v7.i5.315 World J Orthop 2016 May 18; 7(5): 315-337 ISSN 2218-5836 (online) 2016 Baishideng
Submit a Manuscript: http://www.wjgnet.com/esps/ Help Desk: http://www.wjgnet.com/esps/helpdesk.aspx DOI: 10.5312/wjo.v7.i5.315 World J Orthop 2016 May 18; 7(5): 315-337 ISSN 2218-5836 (online) 2016 Baishideng
INTERVENTIONAL TREATMENT FOR PAIN OF SPINAL ORIGIN
 INTERVENTIONAL TREATMENT FOR PAIN OF SPINAL ORIGIN Dr John MacVicar, Medical Director Southern Rehab, Christchurch, NZ AOCPRM Nov 23 rd 2018 INTERVENTIONAL TREATMENT FOR PAIN OF SPINAL ORIGIN An evidence-based
INTERVENTIONAL TREATMENT FOR PAIN OF SPINAL ORIGIN Dr John MacVicar, Medical Director Southern Rehab, Christchurch, NZ AOCPRM Nov 23 rd 2018 INTERVENTIONAL TREATMENT FOR PAIN OF SPINAL ORIGIN An evidence-based
SOFT TISSUE KNEE INJURIES
 SOFT TISSUE KNEE INJURIES Soft tissue injuries of the knee commonly occur in all sports or in any activity that requires sudden changes in activity or movement. The knee is a complex joint and any injury
SOFT TISSUE KNEE INJURIES Soft tissue injuries of the knee commonly occur in all sports or in any activity that requires sudden changes in activity or movement. The knee is a complex joint and any injury
Knee Case Studies. You might KNEED to know some of this stuff
 Knee Case Studies You might KNEED to know some of this stuff Mark Mildren, MD Specializing in Adult Reconstruction Slocum Center for Orthopedics & Sports Medicine 25 th Annual Orthopedic & Sports Medicine
Knee Case Studies You might KNEED to know some of this stuff Mark Mildren, MD Specializing in Adult Reconstruction Slocum Center for Orthopedics & Sports Medicine 25 th Annual Orthopedic & Sports Medicine
RN(EC) ENC(C) GNC(C) MN ACNP *** MECHANISM OF INJURY.. MOST IMPORTANT *** - Useful in determining mechanism of injury / overuse
 HISTORY *** MECHANISM OF INJURY.. MOST IMPORTANT *** Age of patient Sport / Occupation - Certain conditions are more prevalent in particular age groups (Osgood Schlaters in youth / Degenerative Joint Disease
HISTORY *** MECHANISM OF INJURY.. MOST IMPORTANT *** Age of patient Sport / Occupation - Certain conditions are more prevalent in particular age groups (Osgood Schlaters in youth / Degenerative Joint Disease
Degenerative arthritis of Hip Bone Bangalore. Prof Sharath Rao Head, Dept. of Orthopaedics KMC Manipal
 Degenerative arthritis of Hip Prof Sharath Rao Head, Dept. of Orthopaedics KMC Manipal Hip joint Classical Synovial joint Biomechanics of hip Force coincides with trabecular pattern Hip joint Acetabulum
Degenerative arthritis of Hip Prof Sharath Rao Head, Dept. of Orthopaedics KMC Manipal Hip joint Classical Synovial joint Biomechanics of hip Force coincides with trabecular pattern Hip joint Acetabulum
Rehabilitation Guidelines for Anterior Cruciate Ligament (ACL) Reconstruction
 Rehabilitation Guidelines for Anterior Cruciate Ligament (ACL) Reconstruction The knee is the body's largest joint, and the place where the femur, tibia, and patella meet to form a hinge-like joint. These
Rehabilitation Guidelines for Anterior Cruciate Ligament (ACL) Reconstruction The knee is the body's largest joint, and the place where the femur, tibia, and patella meet to form a hinge-like joint. These
Index. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Adductor strain, 625 Agility-training lateral hurdles, 689 Ankle sprain, and lateral ligament reconstruction, complications of, 704 705
Index Note: Page numbers of article titles are in boldface type. A Adductor strain, 625 Agility-training lateral hurdles, 689 Ankle sprain, and lateral ligament reconstruction, complications of, 704 705
Discussion Points 10/17/16. Spine Pain is Ubiquitous. Interventional Pain Management
 Interventional Pain Management Blake Christensen, D.O. Fellowship Trained Interventional Pain Management Board Eligible in Anesthesiology and Interventional Pain Management Oklahoma Interventional Pain
Interventional Pain Management Blake Christensen, D.O. Fellowship Trained Interventional Pain Management Board Eligible in Anesthesiology and Interventional Pain Management Oklahoma Interventional Pain
Post test for O&P 2 Hrs CE. The Exam
 Post test for O&P 2 Hrs CE The Exam This examination is taken in "open book" format. That means you are free to answer the questions after research or discussion with your fellow workers. We feel this
Post test for O&P 2 Hrs CE The Exam This examination is taken in "open book" format. That means you are free to answer the questions after research or discussion with your fellow workers. We feel this
Kevin S. Ladin, M.D. Board Certified in Physical Medicine & Rehabilitation ABMS Subspecialty Board Certified in Pain Medicine
 Kevin S. Ladin, M.D. Board Certified in Physical Medicine & Rehabilitation ABMS Subspecialty Board Certified in Pain Medicine kslmd@cox.net EBM in Pain Medicine Evidence-based medicine (EBM) is a form
Kevin S. Ladin, M.D. Board Certified in Physical Medicine & Rehabilitation ABMS Subspecialty Board Certified in Pain Medicine kslmd@cox.net EBM in Pain Medicine Evidence-based medicine (EBM) is a form
Hip Injuries & Arthroscopy in Athletes
 Hip Injuries & Arthroscopy in Athletes John P Salvo, MD Sports Medicine Rothman Institute Philadelphia, PA EATA Annual Meeting January, 2011 Hip Injuries & Arthroscopy in Anatomy History Physical Exam
Hip Injuries & Arthroscopy in Athletes John P Salvo, MD Sports Medicine Rothman Institute Philadelphia, PA EATA Annual Meeting January, 2011 Hip Injuries & Arthroscopy in Anatomy History Physical Exam
No Disclosures. Topics. Pediatric ACL Tears
 Knee Injuries in Skeletally Immature Athletes No Disclosures Zachary Stinson, M.D. 2 Topics ACL Tears and Tibial Eminence Fractures Meniscus Injuries Discoid Meniscus Osteochondritis Dessicans Patellar
Knee Injuries in Skeletally Immature Athletes No Disclosures Zachary Stinson, M.D. 2 Topics ACL Tears and Tibial Eminence Fractures Meniscus Injuries Discoid Meniscus Osteochondritis Dessicans Patellar
Lysis of Epidural Adhesions
 Lysis of Epidural Adhesions Policy Number: 8.01.18 Last Review: 6/2018 Origination: 8/2007 Next Review: 6/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for lysis
Lysis of Epidural Adhesions Policy Number: 8.01.18 Last Review: 6/2018 Origination: 8/2007 Next Review: 6/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for lysis
CLINICS IN SPORTS MEDICINE
 Clin Sports Med 25 (2006) 365 369 CLINICS IN SPORTS MEDICINE A Acetabular labrum, tears of, hip arthroscopy in, 264 Acetabular rim, trimming of, and labral repair, new method for, 293 297 Acetabulum, femoral
Clin Sports Med 25 (2006) 365 369 CLINICS IN SPORTS MEDICINE A Acetabular labrum, tears of, hip arthroscopy in, 264 Acetabular rim, trimming of, and labral repair, new method for, 293 297 Acetabulum, femoral
The condition occurs when the proximal femur repeatedly comes into contact with the native acetabular rim during normal hip range of motion.
 RIM SYNDROME [femoroacetabular impingement] It has been suggested to be a preosteoarthritic mechanism. The condition occurs when the proximal femur repeatedly comes into contact with the native acetabular
RIM SYNDROME [femoroacetabular impingement] It has been suggested to be a preosteoarthritic mechanism. The condition occurs when the proximal femur repeatedly comes into contact with the native acetabular
NEUROABLATION FOR TREATMENT OF CHRONIC PAIN
 NEUROABLATION FOR TREATMENT OF CHRONIC PAIN Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical
NEUROABLATION FOR TREATMENT OF CHRONIC PAIN Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical
