Clinical. Clostridium Difficile: Standard Operating Procedure. Document Control Summary. Contents
|
|
|
- Margery King
- 5 years ago
- Views:
Transcription
1 Clinical Clostridium Difficile: Standard Operating Procedure Document Control Summary Status: Version: Author/Title: Owner/Title: Approved by: Ratified: Related Trust Strategy and/or Strategic Aims Implementation Date: Review Date: Key Words: Associated Policy or Standard Operating Procedures Replacement. Replaces: Clostridium Difficile Policy v3.1 v1.0 Date: November 2015 Judy Carr - Lead Infection Prevention and Control Nurse Kenny Laing - Deputy Director of Nursing Policy and Procedures Committee Date: 17/12/15 Policy and Procedures Committee Date: 17/12/15 Provide high quality services, built on best known practice and evaluated through clear process and outcome measures December 2015 December 2018 Isolation. C Diff Infection Prevention Control and Decontamination Policy Hand Decontamination SOP Standard Precautions and personal Protective Equipment SOP Decontamination of Reusable Medical Devices Policy SOP Isolation Policy SOP Guidelines for Glove usage in Clinical Practice SOP Mental Capacity Act Antibiotic Policy Contents 1. Introduction Purpose Scope Definitions Clinical Features Diagnosis Laboratory confirmation Risk Factors Transmission... 6
2 10. Surveillance Patient Management (see Associate documents 2 & 3) Medical Management of C. difficile Visits to other departments Discharge of patients Outbreaks of C. difficile Cadaver Death certification Control of Antibiotic Usage Monitoring Compliance References Change Control Amendment History Version Dates Amendments 1. Introduction C. difficile is a bacterium of the family Clostridium (the family also includes the bacteria that cause tetanus, botulism, and gas gangrene). It is an anaerobic bacterium (i.e. it does not grow in the presence of oxygen) and produces spores that can survive for a long time in the environment. It is found in the large intestine in approximately 5% of the population. It can cause illness when the balance of the normal gut flora is disturbed by the frequent use of certain antibiotics, e.g. cephalosporins. The antibiotics disturb the balance of bacteria in the large bowel which enables the C. difficile bacteria to proliferate. Once established the bacteria produces toxins, which are responsible for the diarrhoea and which damage the cells lining the bowel. It affects predominately the colon and may result in a wide spectrum of disease ranging in severity from trivial diarrhoea, through moderately severe disease with abdominal pain, diarrhoea and systemic upset to life -threatening pseudomembranous colitis, toxic megacolon and paralytic ileus. The spores produced easily colonise the environment and medical equipment and can also be spread via contact and the faecal-oral route. The prevention and control of this organism is increasingly problematic in UK healthcare. New strains are emerging which are more virulent than previous ones; these have been identified locally. Page 2 of 11
3 The average increased length of stay of a patient with CDI is 21 days and the cost estimated at over 4000 per case. C. difficile has the potential to cause large outbreaks in healthcare settings if not managed appropriately, i.e. via standard precautions and isolation. If a patient has diarrhoea (Bristol Stool Chart types 5-7, Associate document 1) that is not clearly attributable to an underlying condition (e.g. inflammatory colitis, overflow) or therapy (e.g. laxatives, enteral feeding) then it is necessary to determine if this is due to CDI. Ideally isolate patient in a single room collect a specimen and seek advice from the Infection Prevention and Control (IPC) Team. 2. Purpose Patients have a right to be protected from preventable infections and healthcare staff have a duty to safeguard the well-being of their patients. The Health Act 2008 requires all NHS bodies to have in place appropriate core policies of which Clostridium difficile is one. The purpose of this SOP is to provide guidance about the management and control of Clostridium Difficile (C.difficile) within the healthcare setting. 3. Scope This document applies to all South Staffordshire and Shropshire employees and all those visiting the Trusts premises such as contractors, agency/bank/locum staff, students and volunteers 4. Definitions Clostridium difficile Infection (CDI) 1 episode of diarrhoea, defined as either a stool loose enough to take the shape of a container used to sample it or as Bristol Stool Chart types 5-7 (Associate document 1), that is not attributable to another cause, including medicines and that occurs at the same time as a positive toxin assay (with or without a positive C. difficile culture) and/or endoscopic evidence of pseudomembranous colitis. (DH 2009b) A period of increased incidence (PII) of CDI: 2 or more new cases (occurring >48 hours post admission, not relapses) in a 28 day period in a ward. An outbreak of CDI: 2 or more cases caused by the same strain related in time and place over a defined period that is based on the date of onset of the 1st case (DH 2009b) Diarrhoea - defined by the passage of liquid or watery stools three or more times a day. Bristol Stool Chart - type 5 or 7. Colonisation - A long term relationship in which a micro-organism lives on (or in) a host, without any adverse reaction by the host to its presence Infection - The outcome of an interaction between a host and a micro-organism in which the host reacts in an observable way. The evidence is usually a clinical infection. 5. Clinical Features Usually associated with the prior use of antibiotics Page 3 of 11
4 Explosive, watery, foul smelling diarrhoea. Abdominal pain/tenderness Fever Predominantly affects the colon, but terminal ileum can also be affected High white blood cell count generally associated with more severe disease Mild CDI Moderate CDI Severe CDI Lifethreatening CDI is not associated with a raised WCC; it is typically associated with <3 stools of types 5 7 on the Bristol Stool Chart per day. is associated with a raised WCC that is <15 109/L; it is typically associated with 3 5 stools per day. is associated with a WCC >15 109/L, or an acute rising serum creatinine (i.e. >50% increase above baseline), or a temperature of >38.5 C, or evidence of severe colitis (abdominal or radiological signs). The number of stools may be a less reliable indicator of severity. includes hypotension, partial or complete ileus or toxic megacolon, or CT evidence of severe disease. 6. Diagnosis This should be based on clinical assessment of CDI, symptoms and risk factors. As speed of diagnosis is important and to ensure the efficient use of isolation facilities, clinicians should, in line with the SIGHT protocol (Associate document 7), ensure that stool specimens are sent for toxin testing as soon as infective diarrhoea is suspected. (DH 2009). All patients with suspected or confirmed CDI must be commenced on the Clostridium difficile Care Pathway immediately (Associate document 3) along with the Bristol Stool Chart (Associate document 1). When sending stool specimens to the laboratory, the form should request C. difficile investigation and state any recent antibiotic history, frequency of diarrhoea, stool type e.g. Type 5-7, date of onset of diarrhoea. The diarrhoea sample should comply with Type 5-7 of the Bristol Stool Form Scale or the sample must take on the shape of the container and ideally be at least ¼ filled (to indicate the patient has diarrhoea).; otherwise the sample will not be tested for C. difficile. 7. Laboratory Confirmation C. difficile infection (CDI) is a clinical diagnosis, but laboratory confirmation should be used to guide therapy and management as there are many causes for healthcare associated diarrhoea. Patients who are otherwise healthy may have carriage of C. difficile. If these patients develop diarrhoea for other causes the C. difficile may be detected as a bystander. For this reason laboratory diagnostics differentiate between detecting toxin that indicates disease and detecting organism by culture or PCR that may represent carriage. Page 4 of 11
5 Testing methods are as follows: 1) Stool samples are screened with a GDH test; this detects the C. difficile-specific enzyme glutamate dehydrogenase antigen (GDH). If the GDH test is negative the stool is negative and there is no evidence of C. difficile. 2) If the GDH is positive then a toxin test is performed. If this is positive then there is evidence of C. difficile infection. These patients require treatment for CDI and isolation to prevent cross infection. If the toxin test is negative then a PCR (Polymerase Chain Reaction) is performed 3) If the toxin test is negative (and the GDH test is positive) a PCR is performed. If the PCR is negative the stool is negative and there is no evidence of C. difficile. If the PCR is positive there is evidence of potential carriage (or potential excretor ). This means though the diarrhoea may not have been caused by C. difficile there is an infection control risk and these patients should be isolated to prevent cross-infections though they may not require specific therapy. This means there are 3 possible laboratory results. Results Interpretation Action GDH, EIA or (NAAT) -ve C. difficile negative No evidence of CDI look for other causes of diarrhoea GDH, EIA or (NAAT) +ve C. difficile infection Treat CDI, infection control tox +ve GDH, EIA or (NAAT) +ve/ tox ve PCR +ve GDH, EIA or (NAAT) +ve/ tox ve PCR ve Potential C. difficile carriage/( potential excretor ) measures No evidence of CDI look for other causes of diarrhoea. Do not necessarily need to treat CDI, but take infection control measures. C. difficile negative No evidence of CDI look for other causes of diarrhoea 8. Risk Factors Elderly (over 65 years) Long length of stay in healthcare settings Recent use of antibiotics especially broad spectrum e.g. cephalosporins, which are harmful to normal gut flora Recent surgery, especially gastro-intestinal surgery Serious underlying disease/illness Immunocompromising conditions Prolonged use of proton pump inhibitors Poor cleaning or infection control practise Page 5 of 11
6 9. Transmission C. difficile is easily spread from patient to patient as spores, shed in the faeces, which can survive in the environment on any surface for a long period of time. These spores are resistant to heat, alcohol and acids in the stomach. C. difficile can be spread via the hands of healthcare workers, if they have touched contaminated surfaces or from direct contact with an infected patient 10. Surveillance Confirmation of Clostridium difficile infection is via the laboratory detection of Clostridium difficile Toxins A and or B in stool samples. Do not retest for C. difficile toxin positive cases if patients are still symptomatic within a period of 28 days unless symptoms resolve and then recur and there is a need to confirm recurrent Clostridium difficile illness More than one test per patient may be required if the first test is negative but where there is a strong clinical suspicion of CDI, retest a second sample 24 hours later. Further tests might be necessary in light of clinical evidence. Generally it is not advisable to test children under the age of 2 years in whom toxigenic strains of C. difficile and toxins A and B may be present in the absence of symptoms. Sudden increases in the number and/or severity of cases detected in a ward or across several units within a hospital legitimate reasons for typing requests. However, this is best undertaken in a planned way, following discussion with the relevant microbiology laboratory &/or IPCT. 11. Patient Management (see Associate documents 2 & 3) The tables in the appendices provide an overview of the management of C. difficile positive patients in the inpatient setting. The guidance on general management, record keeping, treatment, hygiene, patient placement and cleaning should be followed carefully to prevent harm and minimise the risk of spread. The Care Pathway and Fluid/Stool chart provided in Associate documents 3 & 4 are to be used in the inpatient setting. Robust infection prevention and control practices are crucial in reducing the incidence of CDI, especially Hand washing with soap and water rather than using alcohol gel The use of gloves and aprons Isolation Decontamination Minimising all unnecessary patient movement Remember wash your hands after any contact with the patient or their environment. Page 6 of 11
7 12. Medical Management of C. difficile The patient must be medically assessed. Clinicians must consider CDI as a diagnosis in its own right, grading each case for severity, treating accordingly and ensuring the ongoing monitoring of bowel function using the Bristol Stool Chart (If the medical officer requires more information this can be sought from the Microbiologist or Infection Prevention and control Team). Key principles of treatment are: Early treatment is important and should be commenced as soon as the diagnosis is confirmed Medications which alter the gut flora, including aperients, enemas and antacids must be stopped. All antibiotics that are clearly not required and may be contributing to the symptoms should be stopped, as should other drugs that might cause diarrhoea Anti-motility agents such as loperamide must not be prescribed in acute CDI. It is unclear whether proton pump inhibitors (PPIs) are a risk factor in relation to CDI; their use should be reviewed, especially in a patient who experiences recurrent CDI (DOH2009) Fluid management and maintaining electrolyte balance are essential (Associate document 4) Nutrition is very important and early consideration should be given to NG feeding. a low albumin level is associated with more severe disease Treatment must be given for the full course, even if symptoms resolve before the course is completed. (Refer to Treatment Algorithms Associate document 5) Patients to be monitored daily for frequency and severity of diarrhoea using the Bristol Stool Chart (Associate document 1). CDI should be managed as a diagnosis in its own right, with each patient reviewed daily regarding fluid resuscitation, electrolyte replacement and nutritional review. Monitor daily for signs of increasing severity of disease, with early referral to the local acute Trusts as patients may deteriorate very rapidly. Treat according to severity See below Mild and moderate CDI Severe CDI Lifethreatening CDI Oral metronidazole mg tds for days. Oral vancomycin 125 mg qds for days. In severe CDI cases not responding to oral vancomycin 125 mg qds, high-dosage oral vancomycin (up to 500 mg qds, if necessary administered via a nasogastric tube) +/- intravenous (iv) metronidazole 500 mg tds is recommended. The addition of oral rifampicin (300 mg bd) or iv immunoglobulin (400 mg/kg) may also be considered. Oral vancomycin up to 500 mg qds for days via nasogastric tube or rectal installation plus iv metronidazole 500 mg tds. Such patients should be closely monitored, with specialist surgical input, and should have their blood lactate measured. Colectomy should be considered, especially if caecal dilatation is >10 cm. Colectomy is best performed before blood lactate rises >5 mmol/l, when survival is extremely poor. Page 7 of 11
8 If diarrhoea persists despite 20 days treatment but the patient is stable and the daily number of type 5 7 motions has decreased, the WCC is normal, and there is no abdominal pain or distension, the persistent diarrhoea may be due to post-infective irritable bowel syndrome. The patient may be treated with an anti-motility agent such as loperamide 2 mg prn (instead of metronidazole or vancomycin). The patient should be closely observed for evidence of a therapeutic response and to ensure there is no evidence of colonic dilatation. For first recurrence, repeat the same antibiotic used to treat the initial episode (unless the first episode was treated with metronidazole and the recurrence is severe CDI, in which case vancomycin should be used). For subsequent recurrences, use vancomycin 125 mg qds. Consider the alternatives listed in the treatment algorithm in Associate document Visits to other departments Visits to other departments should be kept to a minimum Visits that are clinically necessary, for investigation and treatment, prior arrangements should be made and the following principles adhered to: Infected patients should be seen at the end of the working session and only sent for when the department is ready to deal with them. Patients should not be left waiting areas with other patients. Staff should adhere to strict infection control procedures during the investigation or treatment Disposable equipment should be used whenever possible; non disposable equipment must be thoroughly cleaned and decontaminated after use. The patient should return directly to their ward/unit immediately following the procedure. 14. Discharge of patients Patients should only be discharged to residential settings when their stool has returned to normal and the risk of relapse assessed. Patients can be discharged to their own home if they still have diarrhoea but are improving. The patients GP and the residential home, where relevant, should be informed that the patient has been C. difficile positive during their inpatient stay, so that future antibiotic therapy is appropriate (See Associate document 6). Where patients are discharged to their own home the possibility of symptoms recurring should be discussed with the patient and/or carer. If patients have a history of relapse/reinfection a supply of pads, gloves and aprons should be given to the carer to prevent cross infection. A Transfer/Discharge Checklist should be completed and sent to the patient s GP or to the receiving hospital ward (See Associate document 6). Page 8 of 11
9 15. Outbreaks of C. difficile The Infection control and Prevention Team will: adhere to the following definitions for use in identifying and managing incidents of CDI Single Case C. difficile infection A period of increased incidence (PII) of CDI An outbreak of C. difficile infection One episode of diarrhoea, defined either as stool loose enough to take the shape of a container used to sample it or as Bristol Stool Chart types 5 7 (Associate document 1), that is not attributable to any other cause, including medicines (Associate document 2), and that occurs at the same time as a positive toxin assay (with or without a positive C.difficile culture) and/or endoscopic evidence of pseudomembranous colitis (PMC). Two or more new cases (occurring >48 hours post admission, not relapses) in a 28-day period on a ward Two or more cases caused by the same strain related in time and place over a defined period that is based on the date of onset of the first case. The following actions will be taken if a period of increased incidence of CDI or an outbreak is identified on a ward 1. Ward staff, urgently inform the clinical director, matron, ward manager and directorate manager. 2. IPC team, conduct a weekly C. difficile ward audit using the Department of Health s C. difficile High Impact Intervention (HII) tool. The audit should continue until the weekly score is >90% in three consecutive weeks and there have been no further >48 hours cases of CDI on the ward during that period. Feed back the audit results to the matron or ward manager. 3. The antibiotic pharmacist, Carry out a weekly antibiotic review in the ward (using local tools); 4. Clean the whole ward with chlorine-containing agent until no further symptomatic patients are present on the ward. Emphasise that each bed space needs to be cleaned separately with separate cloths. 5. Use the HPA Clostridium difficile Ribotyping Network for England (CDRNE) or Centre for Infections to undertake PCR (polymerase chain reaction) ribotyping of all isolates from patients in the ward. 6. The ICT should carry out an automatic review of ward PIIs each week. 7. An incident meeting should be held as determined by the size and rate of growth of the PII by assessment of the situation by the DIPC and/or the duty microbiologist with the clinical director and consultants, depending on the number of cases. The Trust will report any outbreak as a serious untoward incident (SUIs) to the relevant organisations and subject them to root cause analysis. Page 9 of 11
10 16. Cadaver Infection control precautions for handling deceased patients are the same as those used when the patient is alive. Faecal soiling around the cadaver should be cleaned first with detergent and then with a chlorine- containing cleaning agent. Plastic body bags are not necessary, but may be used as part of general practice in accordance with standard precautions for all patients. There is negligible risk to undertakers provided that standard infection control precautions are used. 17. Death certification If a patient with CDI dies, the death certificate should state whether CDI was part of the sequence of events leading directly to death or whether it was the underlying cause of death. If either case applies CDI should be mentioned in Part 1 of the certificate. B If CDI was not part of the sequence of events leading directly to death but contributed in some way to it, this should be mentioned in Part 2. If a doctor is in doubt about the circumstances of death when writing the certificate, they should consult with the trust s multidisciplinary clinical review team for CDI. B Doctors have a legal duty to mention CDI on a death certificate if it was part of the sequence of events directly leading to death or contributed in some way. 18. Control of Antibiotic Usage Avoidance of unnecessary antibiotic use. The use of narrow spectrum antibiotics whenever the causative pathogen is known. Review of "blind" empirical antibiotic therapy as soon as the causative pathogen has been identified Avoidance, wherever possible, of the use of antibiotic "cocktails". Regular chart review to ensure that antibiotics are discontinued as soon as possible. 19. Monitoring Compliance This policy will be reviewed three yearly or earlier in light of new national guidance or other significant change in circumstances. Compliance with this policy will be monitored through the mechanisms detailed in the table below. Where compliance is deemed to be insufficient and the assurance provided is limited then remedial actions will be drawn together through an action plan. This progress against the action plan will be monitored at the specified committee/group. The results of the annual audit will be escalated to the appropriate committee/group where appropriate. Page 10 of 11
11 20. References Centre for Disease Control (2005) Clostridium Difficile: Information for Healthcare Providers. Updated July. Clostridium Standards Group. (2003) Report to the Department of Health. DH Publications. Department of Health (2006) A Simple Guide to Clostridium Difficile. DH Publications. Healthcare Commission (2005) Management, Prevention and Surveillance of Clostridium Difficile. December. Johnson, S. & Gerding, DN. (2004) Clostridium Difficile. (in) Mayhall, CG. ed. Hospital Epidemiology and Infection Control. 3 rd ed. Lippincott Williams & Wilkins. Lincolnshire Care Pathway Partnership. March Mallett. J, Bailey. C, ed (2004) Manual of Clinical Nursing Procedures. 6 th edition. Royal Marsden NHS Trust. Poxton, IR. (2005) Clostridium Difficile Issues Explored. The Clinical Services Journal. November. Department of Health (2009).Clostridium Difficile Infection: How to deal with the problem. London. Department of Health. Department of Health (2008) The Health and Social Care Act Code of Practice for the NHS on the prevention and control of Healthcare associated infections and related guidance. London. Department of Health Department of Health (2012) Updated guidance on the diagnosis and reporting of Clostridium Page 11 of 11
GUIDELINE FOR THE MANAGEMENT OF ANTIBIOTIC- ASSOCIATED DIARRHOEA IN ADULTS
 GUIDELINE FOR THE MANAGEMENT OF ANTIBIOTIC- ASSOCIATED DIARRHOEA IN ADULTS Version 3.0 Date ratified May 2008 Review date May 2010 Ratified by NUH Antibiotic Guidelines Committee NUH Drugs and Therapeutics
GUIDELINE FOR THE MANAGEMENT OF ANTIBIOTIC- ASSOCIATED DIARRHOEA IN ADULTS Version 3.0 Date ratified May 2008 Review date May 2010 Ratified by NUH Antibiotic Guidelines Committee NUH Drugs and Therapeutics
Clostridium difficile
 Clostridium difficile Care Homes IPC Study Day Sue Barber Infection Prevention & Control Lead AV & Chiltern CCG s Clostridium difficile A spore forming Bacterium. Difficult to grow in the laboratory hence
Clostridium difficile Care Homes IPC Study Day Sue Barber Infection Prevention & Control Lead AV & Chiltern CCG s Clostridium difficile A spore forming Bacterium. Difficult to grow in the laboratory hence
Lifting the lid on a difficile problem part 2 (Clinical) Evidence Based Practice. Problem in evolution (1) Problem in evolution (1) Interventions (2)
 Lifting the lid on a difficile problem part (Clinical) Dr Philip T Mannion Consultant Microbiologist, Rhyl Evidence Based Practice Antibiotic prescribing guidance Isolation policy Hand hygiene (soap and
Lifting the lid on a difficile problem part (Clinical) Dr Philip T Mannion Consultant Microbiologist, Rhyl Evidence Based Practice Antibiotic prescribing guidance Isolation policy Hand hygiene (soap and
ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE
 ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE The diagnosis of CDI should be based on a combination of clinical and laboratory findings. A case definition for the usual
ENGLISH FOR PROFESSIONAL PURPOSES UNIT 3 HOW TO DEAL WITH CLOSTRIDIUM DIFFICILE The diagnosis of CDI should be based on a combination of clinical and laboratory findings. A case definition for the usual
Clostridium difficile Infection: Diagnosis and Management
 Clostridium difficile Infection: Diagnosis and Management Brian Viviano D.O. Case study 42 year old female with history of essential hypertension and COPD presents to ED complaining of 24 hours of intractable,
Clostridium difficile Infection: Diagnosis and Management Brian Viviano D.O. Case study 42 year old female with history of essential hypertension and COPD presents to ED complaining of 24 hours of intractable,
Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review
 Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review October 18, 2010 James Kahn and Carolyn Kenney, MSIV Overview Burden of disease associated
Diagnosis, Management, and Prevention of Clostridium difficile infection in Long-Term Care Facilities: A Review October 18, 2010 James Kahn and Carolyn Kenney, MSIV Overview Burden of disease associated
Clostridium difficile Infection (CDI) Management Guideline
 Clostridium difficile Infection (CDI) Management Guideline Do not test all patients with loose or watery stools for CDI o CDI is responsible for
Clostridium difficile Infection (CDI) Management Guideline Do not test all patients with loose or watery stools for CDI o CDI is responsible for
The incubation period is unknown. However; the onset of clinical disease is typically 5-10 days after initiation of antimicrobial treatment.
 C. DIFFICILE Case definition CONFIRMED CASE A patient is defined as a case if they are one year of age or older AND have one of the following requirements: A laboratory confirmation of a positive toxin
C. DIFFICILE Case definition CONFIRMED CASE A patient is defined as a case if they are one year of age or older AND have one of the following requirements: A laboratory confirmation of a positive toxin
Clostridium difficile infection (CDI) Week 52 (Ending 30/12/2017)
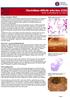 Clostridium difficile infection (CDI) Week 52 (Ending 30/12/2017) What is Clostridium difficile? Clostridium difficile is a Gram-positive anaerobic spore forming bacillus. It is ubiquitous in nature and
Clostridium difficile infection (CDI) Week 52 (Ending 30/12/2017) What is Clostridium difficile? Clostridium difficile is a Gram-positive anaerobic spore forming bacillus. It is ubiquitous in nature and
Prevention of Healthcare- Associated Gastrointestinal Infections Michael A. Borg and Rodianne Abela
 Chapter 19 Prevention of Healthcare- Associated Gastrointestinal Infections Michael A. Borg and Rodianne Abela Key Points Noroviruses are the commonest cause of healthcare-associated gastroenteritis. Isolation
Chapter 19 Prevention of Healthcare- Associated Gastrointestinal Infections Michael A. Borg and Rodianne Abela Key Points Noroviruses are the commonest cause of healthcare-associated gastroenteritis. Isolation
Stony Brook Adult Clostridium difficile Management Guidelines. Discontinue all unnecessary antibiotics
 Stony Brook Adult Clostridium difficile Management Guidelines Summary: Use of the C Diff Infection (CDI) PowerPlan (Adult) Required Patient with clinical findings suggestive of Clostridium difficile infection
Stony Brook Adult Clostridium difficile Management Guidelines Summary: Use of the C Diff Infection (CDI) PowerPlan (Adult) Required Patient with clinical findings suggestive of Clostridium difficile infection
9/18/2018. Clostridium Difficile: Updates on Diagnosis and Treatment. Clostridium difficile Infection (CDI) Clostridium difficile Infection (CDI)
 Clostridium Difficile: Updates on Diagnosis and Treatment Elizabeth Hudson, DO, MPH 9/25/18 Antibiotic-associated diarrhea and colitis were well established soon after widespread use of antibiotics In
Clostridium Difficile: Updates on Diagnosis and Treatment Elizabeth Hudson, DO, MPH 9/25/18 Antibiotic-associated diarrhea and colitis were well established soon after widespread use of antibiotics In
Infection Prevention & Control Core Skills Level 2
 Infection Prevention & Control Core Skills Level 2 Learning outcomes Risk assessment of patients Critical examination of the situation MRSA, CDT & CPE Ongoing challenges future-proofing infection control
Infection Prevention & Control Core Skills Level 2 Learning outcomes Risk assessment of patients Critical examination of the situation MRSA, CDT & CPE Ongoing challenges future-proofing infection control
Guidance for obtaining faecal specimens from patients with diarrhoea (Background information)
 Guidance for obtaining faecal specimens from patients with diarrhoea (Background information) Version 1.0 Date of Issue: January 2009 Review Date: January 2010 Page 1 of 11 Contents 1. Introduction...
Guidance for obtaining faecal specimens from patients with diarrhoea (Background information) Version 1.0 Date of Issue: January 2009 Review Date: January 2010 Page 1 of 11 Contents 1. Introduction...
EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE
 EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click
EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click
Case 1. Which of the following would be next appropriate investigation/s regarding the pts diarrhoea?
 Case 1 21 yr old HIV +ve, Cd4-100 HAART naïve Profuse diarrhoea for 3/52. Stool MC&S ve Which of the following would be next appropriate investigation/s regarding the pts diarrhoea? Repeat stool MC&S Stool
Case 1 21 yr old HIV +ve, Cd4-100 HAART naïve Profuse diarrhoea for 3/52. Stool MC&S ve Which of the following would be next appropriate investigation/s regarding the pts diarrhoea? Repeat stool MC&S Stool
Safe Patient Care Keeping our Residents Safe
 Safe Patient Care Keeping our Residents Safe 2016 Diarrhoea & Vomiting Infection Prevention & Control in Residential Care Setting Patricia Coughlan, Infection Prevention Control Nurse, HSE Disability Services
Safe Patient Care Keeping our Residents Safe 2016 Diarrhoea & Vomiting Infection Prevention & Control in Residential Care Setting Patricia Coughlan, Infection Prevention Control Nurse, HSE Disability Services
Guidelines on the Prevention, Detection, And Management of Clostridium difficile Infection
 Guidelines on the Prevention, Detection, And Management of Clostridium difficile Infection 1 CLINICAL GUIDELINES CHECKLIST Name of guidelines Guidelines on the Prevention, Detection, and Management of
Guidelines on the Prevention, Detection, And Management of Clostridium difficile Infection 1 CLINICAL GUIDELINES CHECKLIST Name of guidelines Guidelines on the Prevention, Detection, and Management of
What is C difficile? (Clostridium difficile) Patient information leaflet
 What is C difficile? (Clostridium difficile) Patient information leaflet What is C difficile? C difficile is short for Clostridium difficile, a rod-shaped bug that lives in the bowel of less than 5% of
What is C difficile? (Clostridium difficile) Patient information leaflet What is C difficile? C difficile is short for Clostridium difficile, a rod-shaped bug that lives in the bowel of less than 5% of
Clostridium difficile Essential information
 Clostridium difficile Essential information Clostridium difficile Origins Clostridium difficile (C. diff) is a Gram positive, spore forming, anaerobic bacterium with a rod structure. It was first identified
Clostridium difficile Essential information Clostridium difficile Origins Clostridium difficile (C. diff) is a Gram positive, spore forming, anaerobic bacterium with a rod structure. It was first identified
Division of GIM Lecture Series Case Presentation David A. Erickson, M.D October 9th, 2013
 Division of GIM Lecture Series Case Presentation David A. Erickson, M.D October 9th, 2013 Financial Disclosures No financial disclosures Objectives Review a case of recurrent Clostridium difficile infection
Division of GIM Lecture Series Case Presentation David A. Erickson, M.D October 9th, 2013 Financial Disclosures No financial disclosures Objectives Review a case of recurrent Clostridium difficile infection
ABSTRACT PURPOSE METHODS
 ABSTRACT PURPOSE The purpose of this study was to characterize the CDI population at this institution according to known risk factors and to examine the effect of appropriate evidence-based treatment selection
ABSTRACT PURPOSE The purpose of this study was to characterize the CDI population at this institution according to known risk factors and to examine the effect of appropriate evidence-based treatment selection
Clostridium difficile
 Clostridium difficile Infection Control Team Patient Information Leaflet What is Clostridium difficile? Clostridium difficile (sometimes called C. diff) is a type of bacteria. They live in the intestine
Clostridium difficile Infection Control Team Patient Information Leaflet What is Clostridium difficile? Clostridium difficile (sometimes called C. diff) is a type of bacteria. They live in the intestine
more intense treatments are needed to get rid of the infection.
 What Is Clostridium Difficile (C. Diff)? Clostridium difficile, or C. diff for short, is an infection from a bacterium that can grow in your intestines and cause bad GI symptoms. The main risk of getting
What Is Clostridium Difficile (C. Diff)? Clostridium difficile, or C. diff for short, is an infection from a bacterium that can grow in your intestines and cause bad GI symptoms. The main risk of getting
Clostridium Difficile Infection in Adults Treatment and Prevention
 Clostridium Difficile Infection in Adults Treatment and Prevention Definition: Clostridium Difficile colonizes the human intestinal tract after the normal gut flora has been altered by antibiotic therapy
Clostridium Difficile Infection in Adults Treatment and Prevention Definition: Clostridium Difficile colonizes the human intestinal tract after the normal gut flora has been altered by antibiotic therapy
Procedure for the Prevention Control and Management of Clostridium difficile Infection in Care Settings in Shetland
 Procedure for the Prevention Control and Management of Clostridium difficile Infection in Care Settings in Shetland Adapted from: Model Infection Control Policies (Transmission Based), HPS ICT September
Procedure for the Prevention Control and Management of Clostridium difficile Infection in Care Settings in Shetland Adapted from: Model Infection Control Policies (Transmission Based), HPS ICT September
Annex C: - CDI What s the diff? 4 th Annual Outbreak Management Workshop September 19, 2013 Naideen Bailey & Grace Volkening
 Annex C: - CDI What s the diff? 4 th Annual Outbreak Management Workshop September 19, 2013 Naideen Bailey & Grace Volkening There s an updated Annex C Annex C is an extension to the PIDAC Infection Prevention
Annex C: - CDI What s the diff? 4 th Annual Outbreak Management Workshop September 19, 2013 Naideen Bailey & Grace Volkening There s an updated Annex C Annex C is an extension to the PIDAC Infection Prevention
Guidance on Prevention and Control of Clostridium difficile Infection (CDI) in health and social care settings in Scotland.
 Guidance on Prevention and Control of Clostridium difficile Infection (CDI) in health and social care settings in Scotland. Scottish Health Protection Network Scottish Guidance No 6 2017 edition. September
Guidance on Prevention and Control of Clostridium difficile Infection (CDI) in health and social care settings in Scotland. Scottish Health Protection Network Scottish Guidance No 6 2017 edition. September
Latex and Occupational Dermatitis Policy Incorporating Glove Selection
 Latex and Occupational Dermatitis Policy Incorporating Glove Selection DOCUMENT CONTROL: Version: 3 Ratified by: Risk Management Sub Group Date ratified: 17 July 2013 Name of originator/author: Health
Latex and Occupational Dermatitis Policy Incorporating Glove Selection DOCUMENT CONTROL: Version: 3 Ratified by: Risk Management Sub Group Date ratified: 17 July 2013 Name of originator/author: Health
Protocol for the Scottish Surveillance Programme for Clostridium difficile infection.
 National Services Scotland Protocol for the Scottish Surveillance Programme for Clostridium difficile infection. User manual. Version 4.0 Revised January 2017 Health Protection Scotland is a division of
National Services Scotland Protocol for the Scottish Surveillance Programme for Clostridium difficile infection. User manual. Version 4.0 Revised January 2017 Health Protection Scotland is a division of
The Epidemiology of Clostridium difficile DANIEL SAMAN, DRPH, MPH RESEARCH SCIENTIST ESSENTIA INSTITUTE OF RURAL HEALTH
 The Epidemiology of Clostridium difficile DANIEL SAMAN, DRPH, MPH RESEARCH SCIENTIST ESSENTIA INSTITUTE OF RURAL HEALTH Some history first Clostridium difficile, a spore-forming gram-positive (i.e., thick
The Epidemiology of Clostridium difficile DANIEL SAMAN, DRPH, MPH RESEARCH SCIENTIST ESSENTIA INSTITUTE OF RURAL HEALTH Some history first Clostridium difficile, a spore-forming gram-positive (i.e., thick
Journey to Decreasing Clostridium Difficile and the Unexpected Twist. Jackie Morton, Infection Prevention Cortney Swiggart, Medication Safety Officer
 Journey to Decreasing Clostridium Difficile and the Unexpected Twist Jackie Morton, Infection Prevention Cortney Swiggart, Medication Safety Officer 4/13/2018 Objectives Discuss the organism and clinical
Journey to Decreasing Clostridium Difficile and the Unexpected Twist Jackie Morton, Infection Prevention Cortney Swiggart, Medication Safety Officer 4/13/2018 Objectives Discuss the organism and clinical
This SOP applies to all staff employed by NHS Greater Glasgow & Clyde and locum staff on fixed term contracts and volunteer staff.
 Page 1 of 8 SOP Objective To ensure that Healthcare Workers (HCWs) are aware of the actions and precautions necessary to minimise the risk of outbreaks and the importance of diagnosing patients clinical
Page 1 of 8 SOP Objective To ensure that Healthcare Workers (HCWs) are aware of the actions and precautions necessary to minimise the risk of outbreaks and the importance of diagnosing patients clinical
A Pharmacist Perspective
 Leveraging Technology to Reduce CDI A Pharmacist Perspective Ed Eiland, Pharm.D., MBA, BCPS (AQ-ID) Clinical Practice and Business Supervisor Huntsville Hospital System Huntsville Hospital 881 licensed
Leveraging Technology to Reduce CDI A Pharmacist Perspective Ed Eiland, Pharm.D., MBA, BCPS (AQ-ID) Clinical Practice and Business Supervisor Huntsville Hospital System Huntsville Hospital 881 licensed
Management of Outbreaks Care Homes IPC Study Day
 Management of Outbreaks Care Homes IPC Study Day Sue Barber Infection Prevention & Control Lead AV & Chiltern CCG s Diarrhoea and/or vomiting May be bacterial or viral May be non-infectious in origin but
Management of Outbreaks Care Homes IPC Study Day Sue Barber Infection Prevention & Control Lead AV & Chiltern CCG s Diarrhoea and/or vomiting May be bacterial or viral May be non-infectious in origin but
Information for Primary Care: Managing patients who require assessment for Ebola virus disease Updated 17 Oct 2014
 Information for Primary Care: Managing patients who require assessment for Ebola virus This guidance is aimed at clinical staff undertaking direct patient care in primary care, including GP surgeries,
Information for Primary Care: Managing patients who require assessment for Ebola virus This guidance is aimed at clinical staff undertaking direct patient care in primary care, including GP surgeries,
March 3, To: Hospitals, Long Term Care Facilities, and Local Health Departments
 March 3, 2010 To: Hospitals, Long Term Care Facilities, and Local Health Departments From: NYSDOH Bureau of Healthcare Associated Infections HEALTH ADVISORY: GUIDANCE FOR PREVENTION AND CONTROL OF HEALTHCARE
March 3, 2010 To: Hospitals, Long Term Care Facilities, and Local Health Departments From: NYSDOH Bureau of Healthcare Associated Infections HEALTH ADVISORY: GUIDANCE FOR PREVENTION AND CONTROL OF HEALTHCARE
This SOP applies to all staff employed by NHS Greater Glasgow & Clyde and locum staff on fixed term contracts and volunteer staff.
 Page 1 of 9 Review SOP Objective To ensure that Healthcare Workers (HCWs) are aware of the actions and precautions necessary to minimise the risk of outbreaks and the importance of diagnosing patients
Page 1 of 9 Review SOP Objective To ensure that Healthcare Workers (HCWs) are aware of the actions and precautions necessary to minimise the risk of outbreaks and the importance of diagnosing patients
CLOSTRIDIUM DIFFICILE
 Other useful information sources NHS Choices: www.nhs.uk NHS 111 (for urgent medical help that is not a 999 emergency) Public Health England: www.phe.gov.uk National Patient Safety Agency - www.npsa.nhs.uk/cleanyourhands
Other useful information sources NHS Choices: www.nhs.uk NHS 111 (for urgent medical help that is not a 999 emergency) Public Health England: www.phe.gov.uk National Patient Safety Agency - www.npsa.nhs.uk/cleanyourhands
Policy Objective. This policy applies to all staff employed by NHS Greater Glasgow & Clyde and locum staff on fixed term contracts.
 1 of 9 Policy Objective To ensure that Healthcare Workers are aware of the actions and precautions necessary to minimise the risk of outbreaks and the importance of diagnosing patients clinical conditions
1 of 9 Policy Objective To ensure that Healthcare Workers are aware of the actions and precautions necessary to minimise the risk of outbreaks and the importance of diagnosing patients clinical conditions
C. Difficile Testing Protocol
 C. Difficile Testing Protocol Caroline Donovan, RN, BSN, ONC- Infection Control Practitioner Abegail Pangan, RN, MSN, CIC- Infection Control Practitioner U.S. NEWS & WORLD REPORT 2017 2018 RANKINGS Acute
C. Difficile Testing Protocol Caroline Donovan, RN, BSN, ONC- Infection Control Practitioner Abegail Pangan, RN, MSN, CIC- Infection Control Practitioner U.S. NEWS & WORLD REPORT 2017 2018 RANKINGS Acute
Clostridium difficile (C. difficile or C. diff)
 What is Clostridium difficile (C. difficile)? Clostridium difficile is a bacterium (germ) that can be found in the gut of about 3 in 100 people. Where there are small numbers of the bacterium it does not
What is Clostridium difficile (C. difficile)? Clostridium difficile is a bacterium (germ) that can be found in the gut of about 3 in 100 people. Where there are small numbers of the bacterium it does not
Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Contact Name and Job Title (author)
 Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Contact ame and Job Title (author) Directorate & Speciality Date of submission December 2015 Date on which guideline
Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Contact ame and Job Title (author) Directorate & Speciality Date of submission December 2015 Date on which guideline
VARICELLA ZOSTER (CHICKENPOX/SHINGLES) INFECTION CONTROL PROCEDURE
 Reference Number: UHB 076 Version Number: 2 Date of Next Review: 23 June 2018 Previous Trust/LHB Reference Number: IPCD Policy No 8 T/45 VARICELLA ZOSTER (CHICKENPOX/SHINGLES) INFECTION CONTROL PROCEDURE
Reference Number: UHB 076 Version Number: 2 Date of Next Review: 23 June 2018 Previous Trust/LHB Reference Number: IPCD Policy No 8 T/45 VARICELLA ZOSTER (CHICKENPOX/SHINGLES) INFECTION CONTROL PROCEDURE
1. INTRODUCTION. 1.1 Standard Precautions
 1. INTRODUCTION 1.1 Standard Precautions Standard precautions, originally known as universal precautions, are essential components in preventing the transmission of infectious diseases in the healthcare
1. INTRODUCTION 1.1 Standard Precautions Standard precautions, originally known as universal precautions, are essential components in preventing the transmission of infectious diseases in the healthcare
INCREASED INCIDENT /OUTBREAK OF DIARRHOEA AND/OR VOMITING
 INCREASED INCIDENT /OUTBREAK OF DIARRHOEA AND/OR VOMITING Documentation to support the management of an increased incident or outbreak of Diarrhoea and/or Vomiting including Norovirus 1 Contents Page Introduction
INCREASED INCIDENT /OUTBREAK OF DIARRHOEA AND/OR VOMITING Documentation to support the management of an increased incident or outbreak of Diarrhoea and/or Vomiting including Norovirus 1 Contents Page Introduction
Questions and answers about the laboratory diagnosis of Clostridium difficile infection (CDI)
 Questions and answers about the laboratory diagnosis of Clostridium difficile infection (CDI) The NHS Centre for Evidence based Purchasing (CEP) has published the results of an evaluation of the performance
Questions and answers about the laboratory diagnosis of Clostridium difficile infection (CDI) The NHS Centre for Evidence based Purchasing (CEP) has published the results of an evaluation of the performance
Infection control in Aged Residential Care Facilities. Dr Sally Roberts Clinical Advisor for IP&C Service, ADHB
 Infection control in Aged Residential Care Facilities Dr Sally Roberts Clinical Advisor for IP&C Service, ADHB Background Endemic infections Epidemic infections Managing outbreaks Administrative measures
Infection control in Aged Residential Care Facilities Dr Sally Roberts Clinical Advisor for IP&C Service, ADHB Background Endemic infections Epidemic infections Managing outbreaks Administrative measures
Prevention and Control of Healthcare-Associated Norovirus
 Purpose: Audience: Policy: To prevent healthcare-associated norovirus infections in patients, employees, contract workers, volunteers, visitors and students and to control and eradicate norovirus infections
Purpose: Audience: Policy: To prevent healthcare-associated norovirus infections in patients, employees, contract workers, volunteers, visitors and students and to control and eradicate norovirus infections
Deafblind Scotland Infection Control Policy
 Deafblind Scotland vision A society in which deafblind people have the permanent support and recognition necessary to be equal citizens Deafblind Scotland Infection Control Policy What do we mean by Infection
Deafblind Scotland vision A society in which deafblind people have the permanent support and recognition necessary to be equal citizens Deafblind Scotland Infection Control Policy What do we mean by Infection
SOP Objective To provide Healthcare Workers (HCWs) with details of the precautions necessary to minimise the risk of RSV cross-infection.
 Page 1 of 11 SOP Objective To provide Healthcare Workers (HCWs) with details of the precautions necessary to minimise the risk of RSV cross-infection. This SOP applies to all staff employed by NHS Greater
Page 1 of 11 SOP Objective To provide Healthcare Workers (HCWs) with details of the precautions necessary to minimise the risk of RSV cross-infection. This SOP applies to all staff employed by NHS Greater
Clostridium difficile infection surveillance: Applying the case definition
 Clostridium difficile infection surveillance: Applying the case definition PICNet Conference March 3 rd 2016 Presented by: Tara Leigh Donovan, MSc Managing Consultant (Former Epidemiologist) 1 Disclaimer
Clostridium difficile infection surveillance: Applying the case definition PICNet Conference March 3 rd 2016 Presented by: Tara Leigh Donovan, MSc Managing Consultant (Former Epidemiologist) 1 Disclaimer
Infection Control Precautions during the Clinical Management of Injecting Drug Users with Possible, Probable or Confirmed Anthrax
 Infection Control Precautions during the Clinical Management of Injecting Drug Users with Possible, Probable or Confirmed Anthrax (Adapted from guidance developed by Health Protection Scotland and HPA
Infection Control Precautions during the Clinical Management of Injecting Drug Users with Possible, Probable or Confirmed Anthrax (Adapted from guidance developed by Health Protection Scotland and HPA
NHS Suffolk Drug & Therapeutics Committee New Medicine Report (Adopted by the CCG until review and further notice)
 NHS Suffolk Drug & Therapeutics Committee New Medicine Report (Adopted by the CCG until review and further notice) This drug has been reviewed because it is a product that may be prescribed in primary
NHS Suffolk Drug & Therapeutics Committee New Medicine Report (Adopted by the CCG until review and further notice) This drug has been reviewed because it is a product that may be prescribed in primary
Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister
 Li StarFish S.r.l. Via Cavour, 35-20063 Cernusco S/N (MI), Italy Tel. +39-02-92150794 - Fax. +39-02-92157285 info@listarfish.it -www.listarfish.it Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister One
Li StarFish S.r.l. Via Cavour, 35-20063 Cernusco S/N (MI), Italy Tel. +39-02-92150794 - Fax. +39-02-92157285 info@listarfish.it -www.listarfish.it Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister One
6/14/2012. Welcome! PRESENTATION OUTLINE CLOSTRIDIUM DIFFICILE PREVENTION. Teaming Up to Prevent Infections! 1) Impact. 2) Testing Recommendations
 CLOSTRIDIUM DIFFICILE PREVENTION Beth Goodall, RN, BSN Board Certified in Infection Prevention and Control DCH Health System Epidemiology Director Welcome! Teaming Up to Prevent Infections! CLOSTRIDIUM
CLOSTRIDIUM DIFFICILE PREVENTION Beth Goodall, RN, BSN Board Certified in Infection Prevention and Control DCH Health System Epidemiology Director Welcome! Teaming Up to Prevent Infections! CLOSTRIDIUM
DISCLOSURE Relevant relationships with commercial entities Wyeth (received advisory board & speaker honoraria) Potential for conflicts of interest wit
 GASTROENTERITIS DISCLOSURE Relevant relationships with commercial entities Wyeth (received advisory board & speaker honoraria) Potential for conflicts of interest within this presentation fidaxomicin (which
GASTROENTERITIS DISCLOSURE Relevant relationships with commercial entities Wyeth (received advisory board & speaker honoraria) Potential for conflicts of interest within this presentation fidaxomicin (which
ACG Clinical Guideline: Diagnosis, Treatment, and Prevention of Clostridium difficile Infections
 ACG Clinical Guideline: Diagnosis, Treatment, and Prevention of Clostridium difficile Infections Christina M. Surawicz, MD 1, Lawrence J. Brandt, MD 2, David G. Binion, MD 3, Ashwin N. Ananthakrishnan,
ACG Clinical Guideline: Diagnosis, Treatment, and Prevention of Clostridium difficile Infections Christina M. Surawicz, MD 1, Lawrence J. Brandt, MD 2, David G. Binion, MD 3, Ashwin N. Ananthakrishnan,
Viral or Suspected Viral Gastroenteritis Outbreaks
 Viral or Suspected Viral Gastroenteritis Outbreaks Information for Directors and Staff of Early Childhood Education and Care Services Introduction Gastroenteritis outbreaks in early childhood education
Viral or Suspected Viral Gastroenteritis Outbreaks Information for Directors and Staff of Early Childhood Education and Care Services Introduction Gastroenteritis outbreaks in early childhood education
CLOSTRIDIUM DIFICILE. Negin N Blattman Infectious Diseases Phoenix VA Healthcare System
 CLOSTRIDIUM DIFICILE Negin N Blattman Infectious Diseases Phoenix VA Healthcare System ANTIBIOTIC ASSOCIATED DIARRHEA 1978: C diff first identified 1989-1992: Four large outbreaks in the US caused by J
CLOSTRIDIUM DIFICILE Negin N Blattman Infectious Diseases Phoenix VA Healthcare System ANTIBIOTIC ASSOCIATED DIARRHEA 1978: C diff first identified 1989-1992: Four large outbreaks in the US caused by J
Influenza Outbreak Control Measure Trigger Tool for Care Homes
 Influenza Outbreak Control Measure Trigger Tool for Care Homes To be used on instruction of your Health Protection Teams (HPT) The control measures in this tool are in addition to Standard Infection Control
Influenza Outbreak Control Measure Trigger Tool for Care Homes To be used on instruction of your Health Protection Teams (HPT) The control measures in this tool are in addition to Standard Infection Control
Factsheet LINACLOTIDE (Constella ) Irritable Bowel Syndrome constipation predominant (IBS-C)
 North Central London Joint Formulary Committee Factsheet LINACLOTIDE (Constella ) Irritable Bowel Syndrome constipation predominant (IBS-C) Start date: September 2018 Review date: September 2021 Document
North Central London Joint Formulary Committee Factsheet LINACLOTIDE (Constella ) Irritable Bowel Syndrome constipation predominant (IBS-C) Start date: September 2018 Review date: September 2021 Document
Clostridium Difficile Associated Disease. Edmund Krasinski, Jr., D.O., F.A.C.G. Southwest Conference on Medicine 2011
 Clostridium Difficile Associated Disease Edmund Krasinski, Jr., D.O., F.A.C.G. Southwest Conference on Medicine 2011 Introduction Which of the following is more common in community hospitals in the Southeast
Clostridium Difficile Associated Disease Edmund Krasinski, Jr., D.O., F.A.C.G. Southwest Conference on Medicine 2011 Introduction Which of the following is more common in community hospitals in the Southeast
Algorithms & Information Sheets
 Minor Ailment Scheme Algorithms & Information Sheets Acute Diarrhoea Diarrhoea is an increased frequency, fluidity or volume of the bowel movements with the passage of soft and watery stools as compared
Minor Ailment Scheme Algorithms & Information Sheets Acute Diarrhoea Diarrhoea is an increased frequency, fluidity or volume of the bowel movements with the passage of soft and watery stools as compared
Care Home Template. Guidelines for the Management of outbreaks of Norovirus
 Care Home Template Guidelines for the Management of outbreaks of Norovirus Title: Procedural Document Type: Reference: Version: Ratified by: Date ratified: Freedom of Information: Name of originator/author:
Care Home Template Guidelines for the Management of outbreaks of Norovirus Title: Procedural Document Type: Reference: Version: Ratified by: Date ratified: Freedom of Information: Name of originator/author:
Clostridium difficile Infection Antibiotics Guidelines. Contents
 Clostridium difficile Infection Antibiotics Guidelines Classification: Clinical Guideline Lead Author: Antibiotic Steering Committee Additional author(s): Authors Division: DCSS & Tertiary Medicine Unique
Clostridium difficile Infection Antibiotics Guidelines Classification: Clinical Guideline Lead Author: Antibiotic Steering Committee Additional author(s): Authors Division: DCSS & Tertiary Medicine Unique
STANDARD OPERATING PROCEDURE (SOP) CHICKENPOX [VARICELLA ZOSTER VIRUS (VZV)]
![STANDARD OPERATING PROCEDURE (SOP) CHICKENPOX [VARICELLA ZOSTER VIRUS (VZV)] STANDARD OPERATING PROCEDURE (SOP) CHICKENPOX [VARICELLA ZOSTER VIRUS (VZV)]](/thumbs/85/92218605.jpg) Page 1 of 9 SOP Objective To ensure that patients with chickenpox (Varicella Zoster Virus) are cared for appropriately and actions are taken to minimise the risk of cross-infection. This SOP applies to
Page 1 of 9 SOP Objective To ensure that patients with chickenpox (Varicella Zoster Virus) are cared for appropriately and actions are taken to minimise the risk of cross-infection. This SOP applies to
CDI The Impact. Disclosures. Acknowledgments. Objectives and Agenda. What s in the Name? 11/14/2012. Lets Talk Numbers
 Disclosures No conflict of interest to declare Acknowledgments Objectives and Agenda Society for Healthcare Epidemiology of America (SHEA) and the Infectious Diseases Society of America (IDSA) Guidelines
Disclosures No conflict of interest to declare Acknowledgments Objectives and Agenda Society for Healthcare Epidemiology of America (SHEA) and the Infectious Diseases Society of America (IDSA) Guidelines
ESCHERICHIA COLI. Pet Health Council Escherichia coli Updated November
 ESCHERICHIA COLI Introduction Escherichia coli 0157:H7 (E. coli 0157) is a cause of food borne illness. Infection may result in sickness and diarrhoea, which can often be severe and with blood. Complications
ESCHERICHIA COLI Introduction Escherichia coli 0157:H7 (E. coli 0157) is a cause of food borne illness. Infection may result in sickness and diarrhoea, which can often be severe and with blood. Complications
Amy-Jo Hooley Specialist Clinical Pharmacist
 Gut Decontamination Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Contact Name and Job Title (author) Directorate & Speciality Guideline for the Safe Administration
Gut Decontamination Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Contact Name and Job Title (author) Directorate & Speciality Guideline for the Safe Administration
JMSCR Vol 05 Issue 07 Page July 2017
 www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i7.15 Rapid Diagnosis of Toxigenic Clostridium
www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i7.15 Rapid Diagnosis of Toxigenic Clostridium
C Difficile - The Ultimate Challenge: Controlling the Spread
 C Difficile - The Ultimate Challenge: Controlling the Spread Linda R. Greene, RN, MPS, CIC Manager of Infection Prevention Highland Hospital Rochester, NY University of Rochester Medical Center linda_greene@urmc.rochester.edu
C Difficile - The Ultimate Challenge: Controlling the Spread Linda R. Greene, RN, MPS, CIC Manager of Infection Prevention Highland Hospital Rochester, NY University of Rochester Medical Center linda_greene@urmc.rochester.edu
Understanding C. diff. atomalliance.org/cdifftraining
 Understanding C. diff atomalliance.org/cdifftraining This booklet is a printed guide of the online educational resource atomalliance.org/cdifftraining Target Audience Understanding C. diff This educational
Understanding C. diff atomalliance.org/cdifftraining This booklet is a printed guide of the online educational resource atomalliance.org/cdifftraining Target Audience Understanding C. diff This educational
Infection Prevention and Control (IPC)
 Infection Prevention and Control (IPC) Standard Operating Procedure for CHICKENPOX (VARICELLA ZOSTER VIRUS) in a healthcare setting WARNING This document is uncontrolled when printed. Check local intranet
Infection Prevention and Control (IPC) Standard Operating Procedure for CHICKENPOX (VARICELLA ZOSTER VIRUS) in a healthcare setting WARNING This document is uncontrolled when printed. Check local intranet
VIRAL GASTROENTERITIS (NOROVIRUS) INFECTION CONTROL IN UNIVERSITY HEALTH BOARD HOSPITALS PROCEDURE
 Reference Number: UHB 075 Version Number: 4 Date of Next Review: 1 st Feb 2021 Previous Trust/LHB Reference Number: T142 VIRAL GASTROENTERITIS (NOROVIRUS) INFECTION CONTROL IN UNIVERSITY HEALTH BOARD HOSPITALS
Reference Number: UHB 075 Version Number: 4 Date of Next Review: 1 st Feb 2021 Previous Trust/LHB Reference Number: T142 VIRAL GASTROENTERITIS (NOROVIRUS) INFECTION CONTROL IN UNIVERSITY HEALTH BOARD HOSPITALS
Issue Notes This guidance replaces all similar guidance issued by the former organisations. KEY POINTS
 Infection Prevention and Control Practice Guidance Note Hand Hygiene and the use of Gloves V03 Date issued Issue 1 May 15 Issue 2 Oct 17 Author/Designation Responsible Officer / Designation Planned review
Infection Prevention and Control Practice Guidance Note Hand Hygiene and the use of Gloves V03 Date issued Issue 1 May 15 Issue 2 Oct 17 Author/Designation Responsible Officer / Designation Planned review
Outbreak Management Supplementary Resource. Residential Care Facilities
 Outbreak Management Supplementary Resource Residential Care Facilities The Ministry of Health have published guidelines on the management of norovirus outbreaks, which should be used as a reference document
Outbreak Management Supplementary Resource Residential Care Facilities The Ministry of Health have published guidelines on the management of norovirus outbreaks, which should be used as a reference document
Guidelines for the Control of a Suspected or Confirmed Outbreak of Viral Gastroenteritis (Norovirus) in an Assisted Living Facility or Nursing Home
 Guidelines for the Control of a Suspected or Confirmed Outbreak of Viral Gastroenteritis (Norovirus) in an Assisted Living Facility or Nursing Home The following is a summary of guidelines developed to
Guidelines for the Control of a Suspected or Confirmed Outbreak of Viral Gastroenteritis (Norovirus) in an Assisted Living Facility or Nursing Home The following is a summary of guidelines developed to
Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate
 Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate Objectives Summarize the changing epidemiology and demographics of patients at risk for Clostridium
Clostridium Difficile Infection: Applying New Treatment Guidelines and Strategies to Reduce Recurrence Rate Objectives Summarize the changing epidemiology and demographics of patients at risk for Clostridium
Tuberculosis Procedure ICPr016. Table of Contents
 Tuberculosis Procedure ICPr016 Table of Contents Tuberculosis Procedure ICPr016... 1 What is Tuberculosis?... 2 Any required definitions/explanations... 2 NHFT... 2 Tuberculosis (TB)... 3 Latent TB...
Tuberculosis Procedure ICPr016 Table of Contents Tuberculosis Procedure ICPr016... 1 What is Tuberculosis?... 2 Any required definitions/explanations... 2 NHFT... 2 Tuberculosis (TB)... 3 Latent TB...
CONTROL OF VIRAL GASTROENTERITIS OUTBREAKS IN CALIFORNIA LONG-TERM CARE FACILITIES
 CONTROL OF VIRAL GASTROENTERITIS OUTBREAKS IN CALIFORNIA LONG-TERM CARE FACILITIES California Department of Health Services Division of Communicable Disease Control In Conjunction with Licensing and Certification
CONTROL OF VIRAL GASTROENTERITIS OUTBREAKS IN CALIFORNIA LONG-TERM CARE FACILITIES California Department of Health Services Division of Communicable Disease Control In Conjunction with Licensing and Certification
Clostridium difficile Infection (CDI) Guideline Update:
 Clostridium difficile Infection (CDI) Guideline Update: Understanding the Data Behind the Recommendations Erik R. Dubberke, MD, MSPH A Webinar for HealthTrust Members Professor of Medicine September 24,
Clostridium difficile Infection (CDI) Guideline Update: Understanding the Data Behind the Recommendations Erik R. Dubberke, MD, MSPH A Webinar for HealthTrust Members Professor of Medicine September 24,
SMT19969: A Selective Therapy for C. difficile Infection
 SMT19969: A Selective Therapy for C. difficile Infection One Bug, One Drug 25 th September 2012 SMT19969: A Selective Therapy for CDI SMT19969 is a novel antibiotic for the specific treatment of Clostridium
SMT19969: A Selective Therapy for C. difficile Infection One Bug, One Drug 25 th September 2012 SMT19969: A Selective Therapy for CDI SMT19969 is a novel antibiotic for the specific treatment of Clostridium
Chickenpox Procedure. (IPC Policy Manual)
 Chickenpox Procedure (IPC Policy Manual) DOCUMENT CONTROL: Version: 1 Ratified by: Clinical Policy Approval Group Date ratified: 3 July 2018 Name of originator/author: Senior Clinical Nurse Specialist
Chickenpox Procedure (IPC Policy Manual) DOCUMENT CONTROL: Version: 1 Ratified by: Clinical Policy Approval Group Date ratified: 3 July 2018 Name of originator/author: Senior Clinical Nurse Specialist
L. Clifford McDonald, MD. Senior Advisor for Science and Integrity September 16, 2015
 Controversies and Current Issues in Diagnosis, Surveillance, and Treatment of Clostridium difficile infeciton L. Clifford McDonald, MD Senior Advisor for Science and Integrity September 16, 2015 Division
Controversies and Current Issues in Diagnosis, Surveillance, and Treatment of Clostridium difficile infeciton L. Clifford McDonald, MD Senior Advisor for Science and Integrity September 16, 2015 Division
Clostridium difficile Infection (CDI)
 18.09.10 월요집담회 Clostridium difficile Infection (CDI) R4 송주혜 Clostridium difficile infection (CDI) Anaerobic gram (+), spore-forming, toxin(tcda&tcdb)-producing bacillus Transmitted among humans through
18.09.10 월요집담회 Clostridium difficile Infection (CDI) R4 송주혜 Clostridium difficile infection (CDI) Anaerobic gram (+), spore-forming, toxin(tcda&tcdb)-producing bacillus Transmitted among humans through
INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust)
 A member of: Association of UK University Hospitals INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust) IPC17 SINGLE USE AND SINGLE PATIENT USE MEDICAL
A member of: Association of UK University Hospitals INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust) IPC17 SINGLE USE AND SINGLE PATIENT USE MEDICAL
Fecal microbiota transplantation: Breaking the chain of recurrent C. difficile infection
 Fecal microbiota transplantation: Breaking the chain of recurrent C. difficile infection Issue Date: June 2013 Vol. 8 No. 6 Author: Amy Marinski, MSN, RN, CCRN, CNL More than 3 million new cases of Clostridium
Fecal microbiota transplantation: Breaking the chain of recurrent C. difficile infection Issue Date: June 2013 Vol. 8 No. 6 Author: Amy Marinski, MSN, RN, CCRN, CNL More than 3 million new cases of Clostridium
Falls The Assessment, Prevention and Management of Patient Falls (Adult Services) 1.34
 SECTION: 1 PATIENT CARE Including Physical Healthcare POLICY /PROCEDURE: 1.34 NATURE AND SCOPE: SUBJECT (Title): POLICY AND PROCEDURE - TRUST WIDE FALLS: THE ASSESSMENT, PREVENTION AND MANAGEMENT OF PATIENT
SECTION: 1 PATIENT CARE Including Physical Healthcare POLICY /PROCEDURE: 1.34 NATURE AND SCOPE: SUBJECT (Title): POLICY AND PROCEDURE - TRUST WIDE FALLS: THE ASSESSMENT, PREVENTION AND MANAGEMENT OF PATIENT
CHEMOTHERAPY NETWORK GROUP POLICY FOR ADMINISTRATION OF CYTOTOXIC CHEMOTHERAPY
 CHEMOTHERAPY NETWORK GROUP POLICY FOR ADMINISTRATION OF CYTOTOXIC CHEMOTHERAPY Version 4.0 March 2016 Review date March 2018 Introduction It is the purpose of this policy to provide clear guidelines that
CHEMOTHERAPY NETWORK GROUP POLICY FOR ADMINISTRATION OF CYTOTOXIC CHEMOTHERAPY Version 4.0 March 2016 Review date March 2018 Introduction It is the purpose of this policy to provide clear guidelines that
There are four important ways in which frontline workers can work together with Peel Public Health to prevent illness due to outbreaks.
 Chapter 4 Outbreaks What is an outbreak? Preparing for an outbreak Identifying an outbreak Outbreak identification process Managing and controlling an outbreak 11 steps Outbreak control measures Outbreak
Chapter 4 Outbreaks What is an outbreak? Preparing for an outbreak Identifying an outbreak Outbreak identification process Managing and controlling an outbreak 11 steps Outbreak control measures Outbreak
Improving Prevention and Control of Infection Quarter 2 Report: April 2009 September 2009
 Improving Prevention and Control of Infection Quarter 2 Report: April 2009 September 2009 1. Introduction This Quarter 2 updates the Health Board on infection prevention and control issues within the BCUHB.
Improving Prevention and Control of Infection Quarter 2 Report: April 2009 September 2009 1. Introduction This Quarter 2 updates the Health Board on infection prevention and control issues within the BCUHB.
Communicable Disease Policy
 Communicable Disease Policy Gastroenteritis (Diarrhoea and Vomiting), including Norovirus: Is easily spread and may lead to an outbreak (two or more cases) Outbreaks can be due to food poisoning or the
Communicable Disease Policy Gastroenteritis (Diarrhoea and Vomiting), including Norovirus: Is easily spread and may lead to an outbreak (two or more cases) Outbreaks can be due to food poisoning or the
PROCEDURE FOR BLOOD GLUCOSE MONITORING
 PROCEDURE FOR BLOOD GLUCOSE MONITORING First Issued Issue Version Two Purpose of Issue/Description of Change Planned Review Date To promote safe and effective blood glucose monitoring using Trust equipment
PROCEDURE FOR BLOOD GLUCOSE MONITORING First Issued Issue Version Two Purpose of Issue/Description of Change Planned Review Date To promote safe and effective blood glucose monitoring using Trust equipment
Hand Hygiene: Preventing avoidable harm in our care
 Hand Hygiene: Preventing avoidable harm in our care Hand Hygiene Training Presentation for Healthcare Workers in Community and Primary Care National HSE HCAI AMR Clinical Programme 2017 What we will cover
Hand Hygiene: Preventing avoidable harm in our care Hand Hygiene Training Presentation for Healthcare Workers in Community and Primary Care National HSE HCAI AMR Clinical Programme 2017 What we will cover
HEALTHCARE- ASSOCIATED CLOSTRIDIUM DIFFICILE INFECTIONS IN CANADIAN ACUTE- CARE HOSPITALS
 HEALTHCARE- ASSOCIATED CLOSTRIDIUM DIFFICILE INFECTIONS IN CANADIAN ACUTE- CARE HOSPITALS SURVEILLANCE REPORT JANUARY 1 st, 2007 TO DECEMBER 31 st, 2012 TO PROMOTE AND PROTECT THE HEALTH OF CANADIANS THROUGH
HEALTHCARE- ASSOCIATED CLOSTRIDIUM DIFFICILE INFECTIONS IN CANADIAN ACUTE- CARE HOSPITALS SURVEILLANCE REPORT JANUARY 1 st, 2007 TO DECEMBER 31 st, 2012 TO PROMOTE AND PROTECT THE HEALTH OF CANADIANS THROUGH
Alert Organisms Group A Streptococcus (GAS) and Invasive Group A Streptococcus (igas)
 Infection Prevention and Control Assurance Standard Operating Procedure 25 (IPC SOP 25) Alert Organisms Group A Streptococcus (GAS) and Invasive Group A Streptococcus (igas) Why we have a procedure? To
Infection Prevention and Control Assurance Standard Operating Procedure 25 (IPC SOP 25) Alert Organisms Group A Streptococcus (GAS) and Invasive Group A Streptococcus (igas) Why we have a procedure? To
Management of Gastroenteritis Outbreaks. Approval Signature: Date of Approval: March 4, 2010 Review Date: March 2013
 Personal Care Home/Long Term Care Facility Infection Prevention and Control Program Operational Directive Management of Gastroenteritis Outbreaks Approval Signature: Date of Approval: March 4, 2010 Review
Personal Care Home/Long Term Care Facility Infection Prevention and Control Program Operational Directive Management of Gastroenteritis Outbreaks Approval Signature: Date of Approval: March 4, 2010 Review
INFECTION PREVENTION AND CONTROL
 INFECTION PREVENTION AND CONTROL Extended guidelines and resource pack for general practice. For use alongside the IPC Core Policy Documents This guideline has been provided by OCCG as an example of best
INFECTION PREVENTION AND CONTROL Extended guidelines and resource pack for general practice. For use alongside the IPC Core Policy Documents This guideline has been provided by OCCG as an example of best
Staff Immunisation Policy
 Policy No: IC05 Version: 8.0 Name of Policy: Staff Immunisation Policy Effective From: 18/08/2015 Date Ratified 15/07/2015 Ratified Infection Prevention & Control Committee Review Date 01/07/2017 Sponsor
Policy No: IC05 Version: 8.0 Name of Policy: Staff Immunisation Policy Effective From: 18/08/2015 Date Ratified 15/07/2015 Ratified Infection Prevention & Control Committee Review Date 01/07/2017 Sponsor
