Beyond the intention-to treat effect: Per-protocol effects in randomized trials
|
|
|
- Charleen Sheena Freeman
- 5 years ago
- Views:
Transcription
1 Beyond the intention-to treat effect: Per-protocol effects in randomized trials Miguel Hernán DEPARTMENTS OF EPIDEMIOLOGY AND BIOSTATISTICS
2 Intention-to-treat analysis (estimator) estimates intention-to-treat effect (estimand) Intention-to-treat effect The effect of being assigned to a treatment strategy, regardless of treatment received, in a particular setting Intention-to-treat effects are agnostic about postrandomization decisions Changes in studied treatment: discontinuation, switching Use of concomitant therapies prohibited by the study protocol etc. Hernán - Beyond the intention-to-treat 2
3 Demystifying intention-to-treat effects: Not necessarily preserve the null Consider a non-blinded trial The ITT effect may not be null even if treatment has a null effect on the outcome Patients and doctors may just alter their behavior in ways that affect their outcome Most pragmatic trials are not blinded Hernán - Beyond the intention-to-treat 3
4 Demystifying intention-to-treat effects: Not necessarily biased towards the null When the treatment effect is not monotonic not in the same direction for all individuals Trial of active treatment vs placebo 30% of the individuals assigned to treatment did not adhere to treatment direction of the effect in adherers opposite to that in non-adherers An ITT analysis may misleadingly indicate a beneficial effect of the less efficacious treatment Hernán - Beyond the intention-to-treat 4
5 Demystifying intention-to-treat effects: Not necessarily biased towards the null Even if the treatment effect is monotonic Trial of 2 active treatments with differential adherence due to a mild, easily palliated side effect An ITT analysis may misleadingly indicate a beneficial effect of the less efficacious treatment Many pragmatic trials are head-to-head trials Hernán - Beyond the intention-to-treat 5
6 Demystifying intention-to-treat effects: Bias towards the null is often undesirable Safety trials Non-inferiority trials In these trials, a conservative ITT analysis is statistical malpractice A trial designed to quantify harm and whose protocol foresees only an ITT analysis could be referred to as a randomized cynical trial Many pragmatic trials are for safety, non-inferiority Hernán - Beyond the intention-to-treat 6
7 Demystifying intention-to-treat effects: Not necessarily a measure of effectiveness Degree of adherence outside the trial may change drastically after doctors and patients learn of the trial s findings Actual effectiveness in the community may differ from ITT effect estimate from trial Hernán - Beyond the intention-to-treat 7
8 Demystifying intention-to-treat effects: Not of primary interest for doctors and patients For example, a couple trying to decide whether to use a contraceptive method would want to know the effectiveness of the method when used as indicated not the estimated effectiveness in a population in which, say, 40% of couples failed to use the method properly That is, not the ITT effect Pragmatic trials are designed to guide clinical decisions by patients and doctors Hernán - Beyond the intention-to-treat 8
9 Need a complement to the ITT effect: An effect measure (an estimand) not affected by the degree of adherence usable in safety, noninferiority trials clinically relevant, patient-centered Per-protocol effect: the effect of implementing the treatment strategies as described in the protocol Hernán - Beyond the intention-to-treat 9
10 A big difference between ITT effect and per-protocol effect We have a universally accepted way of estimating ITT effects ITT analysis Almost uncontroversial We don t have a universally accepted way of estimating per-protocol effects There are many types of per-protocol analysis Including the commonly used, unadjusted, naïve perprotocol analysis Hernán - Beyond the intention-to-treat 10
11 Intention-to-treat effect Analysis plan Simple Compare outcome distribution between group assigned to different strategies Regardless of whether individuals actually followed the strategies Often overlooked problem: ITT analysis cannot be conducted if there are losses to follow-up Potential selection bias due to informative censoring Hernán - Beyond the intention-to-treat 11
12 Intention-to-treat effect Analysis plan Estimating ITT effect requires adjustment for selection bias due to loss to follow-up Adjustment for baseline and post-baseline covariates Little et al, NEJM 2012 In fact, intention-to-treat effect is more precisely defined as the effect of being assigned to a strategy, regardless of strategy received, while staying under follow-up throughout the study Hernán - Beyond the intention-to-treat 12
13 Per-protocol effect Analysis plan Not so simple Treatment decisions after baseline are not randomized Potential post-randomization confounding and selection bias Example In a statins trial, statin use after baseline may depend on post-baseline cholesterol levels; dropout may depend on side effects and prognosis Hernán - Beyond the intention-to-treat 13
14 Per-protocol effect Analysis plan Estimating the per-protocol effect requires adjustment for confounding Adjustment for baseline and post-baseline covariates In addition to adjustment for selection bias same as for ITT effects Hernán - Beyond the intention-to-treat 14
15 Effects (estimands) vs. analyses (estimators) The elephant in the room Typical ITT and per-protocol analyses adjust for neither pre- nor post-randomization variables Potentially biased estimates of ITT and per protocol effects This is a problem for all randomized trials because treatment choices and participation decisions after baseline are not randomly assigned But especially for pragmatic trials with lots of room for non-adherence and loss to follow-up Hernán - Beyond the intention-to-treat 15
16 A pragmatic randomized trial is a follow-up study with baseline randomization Analysis methods to adjust for post-baseline confounding and selection bias are the same methods used for observational follow-up studies Adjustment for post-randomization (time-varying) variables require special techniques Inverse probability (IP) weighting, g-formula, etc Developed by Robins et al since 1986 Instrumental variable estimation Hernán - Beyond the intention-to-treat 16
17 Case study Hormone therapy and breast cancer Question What is the effect of postmenopausal hormone therapy on risk of breast cancer in postmenopausal women? Data A Women s Health Initiative randomized trial ~16,000 postmenopausal U.S. women Toh et al. Epidemiology 2010; 21: Hernán - Beyond the intention-to-treat 17
18 Effect of hormone therapy, what effect? Effect of assignment to hormone therapy under the study s conditions? Intention-to-treat effect Effect of hormone therapy use as instructed by the study s protocol? Per-protocol effect BOTH They answer different questions Hernán - Beyond the intention-to-treat 18
19 Methodological challenges for per protocol effect Time-varying treatment Women may not adhere to their assigned treatment (hormone therapy or placebo) Time-varying confounders Use of hormone therapy depends on age, BMI, symptoms may be affected by prior treatment Also better to estimate absolute risks Appropriately adjusted survival curves Not only hazard ratios Hernán - Beyond the intention-to-treat 19
20 Methodological approach to estimate per protocol effect Estimate IP weights to adjust for time-varying confounding Need data on post-randomization variables Estimate IP weighted hazards model to estimate Hazard ratios Survival (or cumulative incidence) Compare survival curves for continuous treatment vs. no treatment Standardize curves to baseline variables Hernán - Beyond the intention-to-treat 20
21 Hazard ratio of breast cancer Hormone therapy vs. placebo Intention to treat effect estimate 1.25 (1.01, 1.54) Per protocol effect estimate 1.68 (1.24 to 2.28) Suppose you are a woman considering initiation of hormone therapy and who plans to take it as instructed by your doctor Which hazard ratio do you want? Hernán - Beyond the intention-to-treat 21
22 Validity of per-protocol effect estimates Relies on adjustment for post-randomization confounding and selection bias via the same analytic methods and under the same untestable assumptions that we usually reserve for observational studies Hernán - Beyond the intention-to-treat 22
23 Review: Classification of treatment strategies according to their time course Point interventions Intervention occurs at a single time Examples: one-dose vaccination, short-lived traumatic event, surgery Sustained strategies Interventions occur at several times Examples: medical treatments, lifestyle, environmental exposures Hernán - Beyond the intention-to-treat 23
24 Choice of statistical adjustment method depends on type of strategies Comparison of strategies involving point interventions only All methods work if all confounders are measured or the instrumental variable conditions hold Comparison of sustained strategies Generally only g-methods work Developed by Robins and collaborators since 1986 Hernán - Beyond the intention-to-treat 24
25 Per-protocol effect is generally a contrast of sustained (dynamic) treatment strategies Not a comparison of continuous treatment A vs. continuous treatment B But a comparison of strategies of the sort start taking A, continue taking A until toxicity arises, then switch to B Implications for definition of per-protocol effect definition of adherence data collection requirements: need post-randomization data on treatment adherence and (time-varying) confounders Hernán - Beyond the intention-to-treat 25
26 Conclusions (I) There are good reasons for ITT analyses to remain the primary analyses of many randomized trials Also good reasons for appropriately adjusted perprotocol analyses as an integral component of randomized trial analysis especially relevant to patients and clinicians can also be used by modelers and healthcare planners to estimate an upper bound of the impact of changes in recommendations Hernán - Beyond the intention-to-treat 26
27 Conclusions (II) The validity of per-protocol effects requires Explicit definition of per-protocol effect and adherence A priori specification of the statistical plan for the perprotocol analysis High-quality data on adherence and prognostic factors Appropriate adjustment methods These requirements necessitate changes in the way we design and conduct trials Hernán - Beyond the intention-to-treat 27
28 Thank you (more on This work is supported by PCORI ME Additional readings Hernán MA, Hernández-Díaz S. Beyond the intention to treat in comparative effectiveness research. Clinical Trials 2012; 9(1): Hernán MA, Hernández-Díaz S, Robins JM. Randomized trials analyzed like observational studies. Annals of Internal Medicine 2013; 159(8): Toh S, Hernán MA. Causal inference from longitudinal studies with baseline randomization. International Journal of Biostatistics 2008; 4(1): Article 22 Hernán - Beyond the intention-to-treat 28
Generalizing the right question, which is?
 Generalizing RCT results to broader populations IOM Workshop Washington, DC, April 25, 2013 Generalizing the right question, which is? Miguel A. Hernán Harvard School of Public Health Observational studies
Generalizing RCT results to broader populations IOM Workshop Washington, DC, April 25, 2013 Generalizing the right question, which is? Miguel A. Hernán Harvard School of Public Health Observational studies
Per-Protocol Analyses of Pragmatic Trials
 The new england journal of medicine Statistics in Medicine Per-Protocol Analyses of Pragmatic Trials Miguel A. Hernán, M.D., Dr.P.H., and James M. Robins, M.D. Pragmatic trials are designed to address
The new england journal of medicine Statistics in Medicine Per-Protocol Analyses of Pragmatic Trials Miguel A. Hernán, M.D., Dr.P.H., and James M. Robins, M.D. Pragmatic trials are designed to address
ICH E9(R1) Technical Document. Estimands and Sensitivity Analysis in Clinical Trials STEP I TECHNICAL DOCUMENT TABLE OF CONTENTS
 ICH E9(R1) Technical Document Estimands and Sensitivity Analysis in Clinical Trials STEP I TECHNICAL DOCUMENT TABLE OF CONTENTS A.1. Purpose and Scope A.2. A Framework to Align Planning, Design, Conduct,
ICH E9(R1) Technical Document Estimands and Sensitivity Analysis in Clinical Trials STEP I TECHNICAL DOCUMENT TABLE OF CONTENTS A.1. Purpose and Scope A.2. A Framework to Align Planning, Design, Conduct,
Treatment changes in cancer clinical trials: design and analysis
 Treatment changes in cancer clinical trials: design and analysis Ian White Statistical methods and designs in clinical oncology Paris, 9 th November 2017 Plan 1. Treatment changes
Treatment changes in cancer clinical trials: design and analysis Ian White Statistical methods and designs in clinical oncology Paris, 9 th November 2017 Plan 1. Treatment changes
A (Constructive/Provocative) Critique of the ICH E9 Addendum
 A (Constructive/Provocative) Critique of the ICH E9 Addendum Daniel Scharfstein Johns Hopkins University dscharf@jhu.edu April 18, 2018 1 / 28 Disclosures Regularly consult with pharmaceutical and device
A (Constructive/Provocative) Critique of the ICH E9 Addendum Daniel Scharfstein Johns Hopkins University dscharf@jhu.edu April 18, 2018 1 / 28 Disclosures Regularly consult with pharmaceutical and device
Estimands and Sensitivity Analysis in Clinical Trials E9(R1)
 INTERNATIONAL CONCIL FOR HARMONISATION OF TECHNICAL REQUIREMENTS FOR PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED GUIDELINE Estimands and Sensitivity Analysis in Clinical Trials E9(R1) Current Step 2 version
INTERNATIONAL CONCIL FOR HARMONISATION OF TECHNICAL REQUIREMENTS FOR PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED GUIDELINE Estimands and Sensitivity Analysis in Clinical Trials E9(R1) Current Step 2 version
Estimands in klinischen Studien: was ist das und geht mich das was an?
 Estimands in klinischen Studien: was ist das und geht mich das was an? CTU Lecture 03.04.2018 Sven Trelle Mention > ICH E9(R1) Statistical Principles for Clinical Trials "Addendum to focus on statistical
Estimands in klinischen Studien: was ist das und geht mich das was an? CTU Lecture 03.04.2018 Sven Trelle Mention > ICH E9(R1) Statistical Principles for Clinical Trials "Addendum to focus on statistical
Beyond Controlling for Confounding: Design Strategies to Avoid Selection Bias and Improve Efficiency in Observational Studies
 September 27, 2018 Beyond Controlling for Confounding: Design Strategies to Avoid Selection Bias and Improve Efficiency in Observational Studies A Case Study of Screening Colonoscopy Our Team Xabier Garcia
September 27, 2018 Beyond Controlling for Confounding: Design Strategies to Avoid Selection Bias and Improve Efficiency in Observational Studies A Case Study of Screening Colonoscopy Our Team Xabier Garcia
Transmission to CHMP July Adoption by CHMP for release for consultation 20 July Start of consultation 31 August 2017
 1 2 3 30 August 2017 EMA/CHMP/ICH/436221/2017 Committee for Human Medicinal Products 4 ICH E9 (R1) addendum on estimands and sensitivity 5 analysis in clinical trials to the guideline on statistical 6
1 2 3 30 August 2017 EMA/CHMP/ICH/436221/2017 Committee for Human Medicinal Products 4 ICH E9 (R1) addendum on estimands and sensitivity 5 analysis in clinical trials to the guideline on statistical 6
Randomized Experiments with Noncompliance. David Madigan
 Randomized Experiments with Noncompliance David Madigan Introduction Noncompliance is an important problem in randomized experiments involving humans Includes e.g. switching subjects to standard therapy
Randomized Experiments with Noncompliance David Madigan Introduction Noncompliance is an important problem in randomized experiments involving humans Includes e.g. switching subjects to standard therapy
Estimands. EFPIA webinar Rob Hemmings, Frank Bretz October 2017
 Estimands EFPIA webinar Rob Hemmings, Frank Bretz October 2017 Why estimands? To explain which treatment effect is described to prescribers and other stakeholders. To align objectives with (design and)
Estimands EFPIA webinar Rob Hemmings, Frank Bretz October 2017 Why estimands? To explain which treatment effect is described to prescribers and other stakeholders. To align objectives with (design and)
Complier Average Causal Effect (CACE)
 Complier Average Causal Effect (CACE) Booil Jo Stanford University Methodological Advancement Meeting Innovative Directions in Estimating Impact Office of Planning, Research & Evaluation Administration
Complier Average Causal Effect (CACE) Booil Jo Stanford University Methodological Advancement Meeting Innovative Directions in Estimating Impact Office of Planning, Research & Evaluation Administration
In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials
 In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials are comparative, large scale studies that typically
In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials are comparative, large scale studies that typically
Estimands, Missing Data and Sensitivity Analysis: some overview remarks. Roderick Little
 Estimands, Missing Data and Sensitivity Analysis: some overview remarks Roderick Little NRC Panel s Charge To prepare a report with recommendations that would be useful for USFDA's development of guidance
Estimands, Missing Data and Sensitivity Analysis: some overview remarks Roderick Little NRC Panel s Charge To prepare a report with recommendations that would be useful for USFDA's development of guidance
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO CONSIDER ON MISSING DATA
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 15 November 2001 CPMP/EWP/1776/99 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 15 November 2001 CPMP/EWP/1776/99 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO
EU regulatory concerns about missing data: Issues of interpretation and considerations for addressing the problem. Prof. Dr.
 EU regulatory concerns about missing data: Issues of interpretation and considerations for addressing the problem Prof. Dr. Karl Broich Disclaimer No conflicts of interest Views expressed in this presentation
EU regulatory concerns about missing data: Issues of interpretation and considerations for addressing the problem Prof. Dr. Karl Broich Disclaimer No conflicts of interest Views expressed in this presentation
Strategies for handling missing data in randomised trials
 Strategies for handling missing data in randomised trials NIHR statistical meeting London, 13th February 2012 Ian White MRC Biostatistics Unit, Cambridge, UK Plan 1. Why do missing data matter? 2. Popular
Strategies for handling missing data in randomised trials NIHR statistical meeting London, 13th February 2012 Ian White MRC Biostatistics Unit, Cambridge, UK Plan 1. Why do missing data matter? 2. Popular
State of the art pharmacoepidemiological study designs for post-approval risk assessment
 State of the art pharmacoepidemiological study designs for post-approval risk assessment Cardiac Safety Research Consortium Think Tank Round Table Meeting Thursday, March 6, 2014 Jennifer L. Lund, PhD
State of the art pharmacoepidemiological study designs for post-approval risk assessment Cardiac Safety Research Consortium Think Tank Round Table Meeting Thursday, March 6, 2014 Jennifer L. Lund, PhD
The Logic of Causal Inference. Uwe Siebert, MD, MPH, MSc, ScD
 The Logic of Causal Inference Uwe Siebert, MD, MPH, MSc, ScD (1) Dept. of Public Health, Health Services Research and Health Technology Assessment, UMIT - University for Health Sciences, Medical Informatics
The Logic of Causal Inference Uwe Siebert, MD, MPH, MSc, ScD (1) Dept. of Public Health, Health Services Research and Health Technology Assessment, UMIT - University for Health Sciences, Medical Informatics
How the ICH E9 addendum around estimands may impact our clinical trials
 Danish Society for Biopharmaceutical Statistics Copenhagen October 26, 2017 How the ICH E9 addendum around estimands may impact our clinical trials Frank Bretz (Novartis) ICH E9(R1) Expert Working Group
Danish Society for Biopharmaceutical Statistics Copenhagen October 26, 2017 How the ICH E9 addendum around estimands may impact our clinical trials Frank Bretz (Novartis) ICH E9(R1) Expert Working Group
Comparison And Application Of Methods To Address Confounding By Indication In Non- Randomized Clinical Studies
 University of Massachusetts Amherst ScholarWorks@UMass Amherst Masters Theses 1911 - February 2014 Dissertations and Theses 2013 Comparison And Application Of Methods To Address Confounding By Indication
University of Massachusetts Amherst ScholarWorks@UMass Amherst Masters Theses 1911 - February 2014 Dissertations and Theses 2013 Comparison And Application Of Methods To Address Confounding By Indication
Statistical, clinical and ethical considerations when minimizing confounding for overall survival in cancer immunotherapy trials
 Statistical, clinical and ethical considerations when minimizing confounding for overall survival in cancer immunotherapy trials Dominik Heinzmann, PhD Global Development Team Leader HER2 Associate Director
Statistical, clinical and ethical considerations when minimizing confounding for overall survival in cancer immunotherapy trials Dominik Heinzmann, PhD Global Development Team Leader HER2 Associate Director
Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis
 EFSA/EBTC Colloquium, 25 October 2017 Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis Julian Higgins University of Bristol 1 Introduction to concepts Standard
EFSA/EBTC Colloquium, 25 October 2017 Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis Julian Higgins University of Bristol 1 Introduction to concepts Standard
Non-inferiority trials and switch from non-inferiority to superiority. D Costagliola U 943 INSERM and UPMC Paris 06
 Non-inferiority trials and switch from non-inferiority to superiority D Costagliola U 943 INSERM and UPMC Paris 06 References l ICH E 9 et E10 l Points to consider on biostatistical methodological issues
Non-inferiority trials and switch from non-inferiority to superiority D Costagliola U 943 INSERM and UPMC Paris 06 References l ICH E 9 et E10 l Points to consider on biostatistical methodological issues
Estimands and Their Role in Clinical Trials
 Estimands and Their Role in Clinical Trials Frank Bretz (Novartis) EFSPI/PSI UK September 28, 2015 Acknowledgements ICH E9(R1) Expert Working Group M Akacha, D Ohlssen, H Schmidli, G Rosenkranz (Novartis)
Estimands and Their Role in Clinical Trials Frank Bretz (Novartis) EFSPI/PSI UK September 28, 2015 Acknowledgements ICH E9(R1) Expert Working Group M Akacha, D Ohlssen, H Schmidli, G Rosenkranz (Novartis)
GRADE. Grading of Recommendations Assessment, Development and Evaluation. British Association of Dermatologists April 2014
 GRADE Grading of Recommendations Assessment, Development and Evaluation British Association of Dermatologists April 2014 Previous grading system Level of evidence Strength of recommendation Level of evidence
GRADE Grading of Recommendations Assessment, Development and Evaluation British Association of Dermatologists April 2014 Previous grading system Level of evidence Strength of recommendation Level of evidence
The ROBINS-I tool is reproduced from riskofbias.info with the permission of the authors. The tool should not be modified for use.
 Table A. The Risk Of Bias In Non-romized Studies of Interventions (ROBINS-I) I) assessment tool The ROBINS-I tool is reproduced from riskofbias.info with the permission of the auths. The tool should not
Table A. The Risk Of Bias In Non-romized Studies of Interventions (ROBINS-I) I) assessment tool The ROBINS-I tool is reproduced from riskofbias.info with the permission of the auths. The tool should not
GRADE. Grading of Recommendations Assessment, Development and Evaluation. British Association of Dermatologists April 2018
 GRADE Grading of Recommendations Assessment, Development and Evaluation British Association of Dermatologists April 2018 Previous grading system Level of evidence Strength of recommendation Level of evidence
GRADE Grading of Recommendations Assessment, Development and Evaluation British Association of Dermatologists April 2018 Previous grading system Level of evidence Strength of recommendation Level of evidence
ICH E9 (R1) Addendum on Estimands and Sensitivity Analysis
 ICH E9 (R1) Addendum on Estimands and Sensitivity Analysis Rob Hemmings Mouna Akacha MHRA + ICH E9 (R1) Expert Working Group Novartis 1 Disclaimer (I) Not a statistical topic This impacts every clinical
ICH E9 (R1) Addendum on Estimands and Sensitivity Analysis Rob Hemmings Mouna Akacha MHRA + ICH E9 (R1) Expert Working Group Novartis 1 Disclaimer (I) Not a statistical topic This impacts every clinical
Declaration of interests. Register-based research on safety and effectiveness opportunities and challenges 08/04/2018
 Register-based research on safety and effectiveness opportunities and challenges Morten Andersen Department of Drug Design And Pharmacology Declaration of interests Participate(d) in research projects
Register-based research on safety and effectiveness opportunities and challenges Morten Andersen Department of Drug Design And Pharmacology Declaration of interests Participate(d) in research projects
GLOSSARY OF GENERAL TERMS
 GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
The RoB 2.0 tool (individually randomized, cross-over trials)
 The RoB 2.0 tool (individually randomized, cross-over trials) Study design Randomized parallel group trial Cluster-randomized trial Randomized cross-over or other matched design Specify which outcome is
The RoB 2.0 tool (individually randomized, cross-over trials) Study design Randomized parallel group trial Cluster-randomized trial Randomized cross-over or other matched design Specify which outcome is
Evidence Based Medicine
 Course Goals Goals 1. Understand basic concepts of evidence based medicine (EBM) and how EBM facilitates optimal patient care. 2. Develop a basic understanding of how clinical research studies are designed
Course Goals Goals 1. Understand basic concepts of evidence based medicine (EBM) and how EBM facilitates optimal patient care. 2. Develop a basic understanding of how clinical research studies are designed
Fundamental Clinical Trial Design
 Design, Monitoring, and Analysis of Clinical Trials Session 1 Overview and Introduction Overview Scott S. Emerson, M.D., Ph.D. Professor of Biostatistics, University of Washington February 17-19, 2003
Design, Monitoring, and Analysis of Clinical Trials Session 1 Overview and Introduction Overview Scott S. Emerson, M.D., Ph.D. Professor of Biostatistics, University of Washington February 17-19, 2003
Real-world data in pragmatic trials
 Real-world data in pragmatic trials Harold C. Sox,MD The Patient-Centered Outcomes Research Institute Washington, DC Presenter Disclosure Information In compliance with the accrediting board policies,
Real-world data in pragmatic trials Harold C. Sox,MD The Patient-Centered Outcomes Research Institute Washington, DC Presenter Disclosure Information In compliance with the accrediting board policies,
Module 5. The Epidemiological Basis of Randomised Controlled Trials. Landon Myer School of Public Health & Family Medicine, University of Cape Town
 Module 5 The Epidemiological Basis of Randomised Controlled Trials Landon Myer School of Public Health & Family Medicine, University of Cape Town Introduction The Randomised Controlled Trial (RCT) is the
Module 5 The Epidemiological Basis of Randomised Controlled Trials Landon Myer School of Public Health & Family Medicine, University of Cape Town Introduction The Randomised Controlled Trial (RCT) is the
Missing data in clinical trials: a data interpretation problem with statistical solutions?
 INTERVIEW SERIES Clin. Invest. (2012) 2(1), 11 17 Missing data in clinical trials: a data interpretation problem with statistical solutions? Robert Hemmings and David Wright speak to Laura Harvey, Assistant
INTERVIEW SERIES Clin. Invest. (2012) 2(1), 11 17 Missing data in clinical trials: a data interpretation problem with statistical solutions? Robert Hemmings and David Wright speak to Laura Harvey, Assistant
Methodological aspects of non-inferiority and equivalence trials
 Methodological aspects of non-inferiority and equivalence trials Department of Clinical Epidemiology Leiden University Medical Center Capita Selecta 09-12-2014 1 Why RCTs Early 1900: Evidence based on
Methodological aspects of non-inferiority and equivalence trials Department of Clinical Epidemiology Leiden University Medical Center Capita Selecta 09-12-2014 1 Why RCTs Early 1900: Evidence based on
Accounting for treatment switching/discontinuation in comparative effectiveness studies
 Global Medical Affairs RWE and Digital Accounting for treatment switching/discontinuation in comparative effectiveness studies Melvin Skip Olson, PhD Global Head, Real World Data Strategy and Innovation
Global Medical Affairs RWE and Digital Accounting for treatment switching/discontinuation in comparative effectiveness studies Melvin Skip Olson, PhD Global Head, Real World Data Strategy and Innovation
Supplement 2. Use of Directed Acyclic Graphs (DAGs)
 Supplement 2. Use of Directed Acyclic Graphs (DAGs) Abstract This supplement describes how counterfactual theory is used to define causal effects and the conditions in which observed data can be used to
Supplement 2. Use of Directed Acyclic Graphs (DAGs) Abstract This supplement describes how counterfactual theory is used to define causal effects and the conditions in which observed data can be used to
Estimands for time to event endpoints in oncology and beyond
 Estimands for time to event endpoints in oncology and beyond Kaspar Rufibach, Hans Ulrich Burger Methods, Collaboration & Outreach Group (MCO) Department of Biostatistics, Roche Basel Context and problem
Estimands for time to event endpoints in oncology and beyond Kaspar Rufibach, Hans Ulrich Burger Methods, Collaboration & Outreach Group (MCO) Department of Biostatistics, Roche Basel Context and problem
Data and Safety Monitoring in Pragmatic Clinical Trials. Susan S. Ellenberg, PhD Greg Simon, MD, MPH Jeremy Sugarman, MD, MPH, MA
 Data and Safety Monitoring in Pragmatic Clinical Trials Susan S. Ellenberg, PhD Greg Simon, MD, MPH Jeremy Sugarman, MD, MPH, MA Overview The need for DSMBs (Jeremy Sugarman) Special considerations for
Data and Safety Monitoring in Pragmatic Clinical Trials Susan S. Ellenberg, PhD Greg Simon, MD, MPH Jeremy Sugarman, MD, MPH, MA Overview The need for DSMBs (Jeremy Sugarman) Special considerations for
The role of Randomized Controlled Trials
 The role of Randomized Controlled Trials Dr. Georgia Salanti Lecturer in Epidemiology University of Ioannina School of Medicine Outline Understanding study designs and the role of confounding Observational
The role of Randomized Controlled Trials Dr. Georgia Salanti Lecturer in Epidemiology University of Ioannina School of Medicine Outline Understanding study designs and the role of confounding Observational
95% 2.5% 2.5% +2SD 95% of data will 95% be within of data will 1.96 be within standard deviations 1.96 of sample mean
 Efficient Clinical Trials John H. Powers, MD Senior Medical Scientist SAIC in support of Clinical Collaborative Research Branch NIAID/NIH Introduction Definitions what is a small clinical trial? Review
Efficient Clinical Trials John H. Powers, MD Senior Medical Scientist SAIC in support of Clinical Collaborative Research Branch NIAID/NIH Introduction Definitions what is a small clinical trial? Review
Confounding by indication developments in matching, and instrumental variable methods. Richard Grieve London School of Hygiene and Tropical Medicine
 Confounding by indication developments in matching, and instrumental variable methods Richard Grieve London School of Hygiene and Tropical Medicine 1 Outline 1. Causal inference and confounding 2. Genetic
Confounding by indication developments in matching, and instrumental variable methods Richard Grieve London School of Hygiene and Tropical Medicine 1 Outline 1. Causal inference and confounding 2. Genetic
Brief introduction to instrumental variables. IV Workshop, Bristol, Miguel A. Hernán Department of Epidemiology Harvard School of Public Health
 Brief introduction to instrumental variables IV Workshop, Bristol, 2008 Miguel A. Hernán Department of Epidemiology Harvard School of Public Health Goal: To consistently estimate the average causal effect
Brief introduction to instrumental variables IV Workshop, Bristol, 2008 Miguel A. Hernán Department of Epidemiology Harvard School of Public Health Goal: To consistently estimate the average causal effect
The (mis)use of treatment switching adjustment methods in health technology assessment busting some myths!
 The (mis)use of treatment switching adjustment methods in health technology assessment busting some myths! ISPOR Barcelona, 2018 Dr Nicholas Latimer (n.latimer@sheffield.ac.uk) Reader in Health Economics,
The (mis)use of treatment switching adjustment methods in health technology assessment busting some myths! ISPOR Barcelona, 2018 Dr Nicholas Latimer (n.latimer@sheffield.ac.uk) Reader in Health Economics,
Inverse Probability of Censoring Weighting for Selective Crossover in Oncology Clinical Trials.
 Paper SP02 Inverse Probability of Censoring Weighting for Selective Crossover in Oncology Clinical Trials. José Luis Jiménez-Moro (PharmaMar, Madrid, Spain) Javier Gómez (PharmaMar, Madrid, Spain) ABSTRACT
Paper SP02 Inverse Probability of Censoring Weighting for Selective Crossover in Oncology Clinical Trials. José Luis Jiménez-Moro (PharmaMar, Madrid, Spain) Javier Gómez (PharmaMar, Madrid, Spain) ABSTRACT
Critical Appraisal Series
 Definition for therapeutic study Terms Definitions Study design section Observational descriptive studies Observational analytical studies Experimental studies Pragmatic trial Cluster trial Researcher
Definition for therapeutic study Terms Definitions Study design section Observational descriptive studies Observational analytical studies Experimental studies Pragmatic trial Cluster trial Researcher
COMMENTARY: DATA ANALYSIS METHODS AND THE RELIABILITY OF ANALYTIC EPIDEMIOLOGIC RESEARCH. Ross L. Prentice. Fred Hutchinson Cancer Research Center
 COMMENTARY: DATA ANALYSIS METHODS AND THE RELIABILITY OF ANALYTIC EPIDEMIOLOGIC RESEARCH Ross L. Prentice Fred Hutchinson Cancer Research Center 1100 Fairview Avenue North, M3-A410, POB 19024, Seattle,
COMMENTARY: DATA ANALYSIS METHODS AND THE RELIABILITY OF ANALYTIC EPIDEMIOLOGIC RESEARCH Ross L. Prentice Fred Hutchinson Cancer Research Center 1100 Fairview Avenue North, M3-A410, POB 19024, Seattle,
Experimental Design. Terminology. Chusak Okascharoen, MD, PhD September 19 th, Experimental study Clinical trial Randomized controlled trial
 Experimental Design Chusak Okascharoen, MD, PhD September 19 th, 2016 Terminology Experimental study Clinical trial Randomized controlled trial 1 PHASES OF CLINICAL TRIALS Phase I: First-time-in-man studies
Experimental Design Chusak Okascharoen, MD, PhD September 19 th, 2016 Terminology Experimental study Clinical trial Randomized controlled trial 1 PHASES OF CLINICAL TRIALS Phase I: First-time-in-man studies
Title:Bounding the Per-Protocol Effect in Randomized Trials: An Application to Colorectal Cancer Screening
 Author's response to reviews Title:Bounding the Per-Protocol Effect in Randomized Trials: An Application to Colorectal Cancer Screening Authors: Sonja A Swanson (sswanson@hsph.harvard.edu) Oyvind Holme
Author's response to reviews Title:Bounding the Per-Protocol Effect in Randomized Trials: An Application to Colorectal Cancer Screening Authors: Sonja A Swanson (sswanson@hsph.harvard.edu) Oyvind Holme
AVOIDING BIAS AND RANDOM ERROR IN DATA ANALYSIS
 AVOIDING BIAS AND RANDOM ERROR IN DATA ANALYSIS Susan S. Ellenberg, Ph.D. Perelman School of Medicine University of Pennsylvania FDA Clinical Investigator Course Silver Spring, MD November 14, 2018 OVERVIEW
AVOIDING BIAS AND RANDOM ERROR IN DATA ANALYSIS Susan S. Ellenberg, Ph.D. Perelman School of Medicine University of Pennsylvania FDA Clinical Investigator Course Silver Spring, MD November 14, 2018 OVERVIEW
DRAFT (Final) Concept Paper On choosing appropriate estimands and defining sensitivity analyses in confirmatory clinical trials
 DRAFT (Final) Concept Paper On choosing appropriate estimands and defining sensitivity analyses in confirmatory clinical trials EFSPI Comments Page General Priority (H/M/L) Comment The concept to develop
DRAFT (Final) Concept Paper On choosing appropriate estimands and defining sensitivity analyses in confirmatory clinical trials EFSPI Comments Page General Priority (H/M/L) Comment The concept to develop
A Framework for Patient-Centered Outcomes Research
 A Framework for Patient-Centered Outcomes Research David H. Hickam, MD, MPH Director of Research Methodology, PCORI Baltimore, MD August 9, 2016 Session Faculty Disclosures David H. Hickam, MD, MPH No
A Framework for Patient-Centered Outcomes Research David H. Hickam, MD, MPH Director of Research Methodology, PCORI Baltimore, MD August 9, 2016 Session Faculty Disclosures David H. Hickam, MD, MPH No
Glossary of Practical Epidemiology Concepts
 Glossary of Practical Epidemiology Concepts - 2009 Adapted from the McMaster EBCP Workshop 2003, McMaster University, Hamilton, Ont. Note that open access to the much of the materials used in the Epi-546
Glossary of Practical Epidemiology Concepts - 2009 Adapted from the McMaster EBCP Workshop 2003, McMaster University, Hamilton, Ont. Note that open access to the much of the materials used in the Epi-546
Is There An Association?
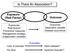 Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
Welcome to this third module in a three-part series focused on epidemiologic measures of association and impact.
 Welcome to this third module in a three-part series focused on epidemiologic measures of association and impact. 1 This three-part series focuses on the estimation of the association between exposures
Welcome to this third module in a three-part series focused on epidemiologic measures of association and impact. 1 This three-part series focuses on the estimation of the association between exposures
Adjusting the Crossover Effect in Overall Survival Analysis Using a Rank Preserving Structural Failure Time Model: The Case of Sunitinib GIST Trial
 Adjusting the Crossover Effect in Overall Survival Analysis Using a Rank Preserving Structural Failure Time Model: The Case of Sunitinib GIST Trial Xin Huang 1 and Qiang (Casey) Xu 2 1 Pfizer Oncology
Adjusting the Crossover Effect in Overall Survival Analysis Using a Rank Preserving Structural Failure Time Model: The Case of Sunitinib GIST Trial Xin Huang 1 and Qiang (Casey) Xu 2 1 Pfizer Oncology
Structural Approach to Bias in Meta-analyses
 Original Article Received 26 July 2011, Revised 22 November 2011, Accepted 12 December 2011 Published online 2 February 2012 in Wiley Online Library (wileyonlinelibrary.com) DOI: 10.1002/jrsm.52 Structural
Original Article Received 26 July 2011, Revised 22 November 2011, Accepted 12 December 2011 Published online 2 February 2012 in Wiley Online Library (wileyonlinelibrary.com) DOI: 10.1002/jrsm.52 Structural
MATH& 146 Lesson 6. Section 1.5 Experiments
 MATH& 146 Lesson 6 Section 1.5 Experiments 1 Experiments Studies where the researchers assign treatments to cases are called experiments. When this assignment includes randomization (such as coin flips)
MATH& 146 Lesson 6 Section 1.5 Experiments 1 Experiments Studies where the researchers assign treatments to cases are called experiments. When this assignment includes randomization (such as coin flips)
EBM: Therapy. Thunyarat Anothaisintawee, M.D., Ph.D. Department of Family Medicine, Ramathibodi Hospital, Mahidol University
 EBM: Therapy Thunyarat Anothaisintawee, M.D., Ph.D. Department of Family Medicine, Ramathibodi Hospital, Mahidol University How to appraise therapy literature Are the result valid? What are the result?
EBM: Therapy Thunyarat Anothaisintawee, M.D., Ph.D. Department of Family Medicine, Ramathibodi Hospital, Mahidol University How to appraise therapy literature Are the result valid? What are the result?
Study Design. Study design. Patrick Breheny. January 23. Patrick Breheny Introduction to Biostatistics (171:161) 1/34
 Study design Patrick Breheny January 23 Patrick Breheny Introduction to Biostatistics (171:161) 1/34 in the ideal world In an ideal world, We have a list of everyone in the population of interest We randomly
Study design Patrick Breheny January 23 Patrick Breheny Introduction to Biostatistics (171:161) 1/34 in the ideal world In an ideal world, We have a list of everyone in the population of interest We randomly
Estimating the impact of community-level interventions: The SEARCH Trial and HIV Prevention in Sub-Saharan Africa
 University of Massachusetts Amherst From the SelectedWorks of Laura B. Balzer June, 2012 Estimating the impact of community-level interventions: and HIV Prevention in Sub-Saharan Africa, University of
University of Massachusetts Amherst From the SelectedWorks of Laura B. Balzer June, 2012 Estimating the impact of community-level interventions: and HIV Prevention in Sub-Saharan Africa, University of
CHL 5225 H Advanced Statistical Methods for Clinical Trials. CHL 5225 H The Language of Clinical Trials
 CHL 5225 H Advanced Statistical Methods for Clinical Trials Two sources for course material 1. Electronic blackboard required readings 2. www.andywillan.com/chl5225h code of conduct course outline schedule
CHL 5225 H Advanced Statistical Methods for Clinical Trials Two sources for course material 1. Electronic blackboard required readings 2. www.andywillan.com/chl5225h code of conduct course outline schedule
Careful with Causal Inference
 Careful with Causal Inference Richard Emsley Professor of Medical Statistics and Trials Methodology Department of Biostatistics & Health Informatics Institute of Psychiatry, Psychology & Neuroscience Correlation
Careful with Causal Inference Richard Emsley Professor of Medical Statistics and Trials Methodology Department of Biostatistics & Health Informatics Institute of Psychiatry, Psychology & Neuroscience Correlation
Biostatistics 3. Developed by Pfizer. March 2018
 BROUGHT TO YOU BY Biostatistics 3 Developed by Pfizer March 2018 This learning module is intended for UK healthcare professionals only. Job bag: PP-GEP-GBR-0986 Date of preparation March 2018. Agenda I.
BROUGHT TO YOU BY Biostatistics 3 Developed by Pfizer March 2018 This learning module is intended for UK healthcare professionals only. Job bag: PP-GEP-GBR-0986 Date of preparation March 2018. Agenda I.
Estimands in oncology
 Estimands in oncology Steven Teerenstra 1,2 1 Radboud university Nijmegen medical center, NL 2 Statistical assessor at Medicines Evaluation Board (MEB), NL Disclaimer 2 Views expressed my own, based on
Estimands in oncology Steven Teerenstra 1,2 1 Radboud university Nijmegen medical center, NL 2 Statistical assessor at Medicines Evaluation Board (MEB), NL Disclaimer 2 Views expressed my own, based on
Collecting Data Example: Does aspirin prevent heart attacks?
 Collecting Data In an experiment, the researcher controls or manipulates the environment of the individuals. The intent of most experiments is to study the effect of changes in the explanatory variable
Collecting Data In an experiment, the researcher controls or manipulates the environment of the individuals. The intent of most experiments is to study the effect of changes in the explanatory variable
Variables Research involves trying to determine the relationship between two or more variables.
 1 2 Research Methodology Week 4 Characteristics of Observations 1. Most important know what is being observed. 2. Assign behaviors to categories. 3. Know how to Measure. 4. Degree of Observer inference.
1 2 Research Methodology Week 4 Characteristics of Observations 1. Most important know what is being observed. 2. Assign behaviors to categories. 3. Know how to Measure. 4. Degree of Observer inference.
Sampling Controlled experiments Summary. Study design. Patrick Breheny. January 22. Patrick Breheny Introduction to Biostatistics (BIOS 4120) 1/34
 Sampling Study design Patrick Breheny January 22 Patrick Breheny to Biostatistics (BIOS 4120) 1/34 Sampling Sampling in the ideal world The 1936 Presidential Election Pharmaceutical trials and children
Sampling Study design Patrick Breheny January 22 Patrick Breheny to Biostatistics (BIOS 4120) 1/34 Sampling Sampling in the ideal world The 1936 Presidential Election Pharmaceutical trials and children
Issues to Consider in the Design of Randomized Controlled Trials
 Issues to Consider in the Design of Randomized Controlled Trials Jay Wilkinson, MD, MPH Professor of Pediatrics & Epidemiology Miller School of Medicine Seminar Purpose To facilitate an interactive discussion
Issues to Consider in the Design of Randomized Controlled Trials Jay Wilkinson, MD, MPH Professor of Pediatrics & Epidemiology Miller School of Medicine Seminar Purpose To facilitate an interactive discussion
16:35 17:20 Alexander Luedtke (Fred Hutchinson Cancer Research Center)
 Conference on Causal Inference in Longitudinal Studies September 21-23, 2017 Columbia University Thursday, September 21, 2017: tutorial 14:15 15:00 Miguel Hernan (Harvard University) 15:00 15:45 Miguel
Conference on Causal Inference in Longitudinal Studies September 21-23, 2017 Columbia University Thursday, September 21, 2017: tutorial 14:15 15:00 Miguel Hernan (Harvard University) 15:00 15:45 Miguel
Comparisons of Dynamic Treatment Regimes using Observational Data
 Comparisons of Dynamic Treatment Regimes using Observational Data Bryan Blette University of North Carolina at Chapel Hill 4/19/18 Blette (UNC) BIOS 740 Final Presentation 4/19/18 1 / 15 Overview 1 Motivation
Comparisons of Dynamic Treatment Regimes using Observational Data Bryan Blette University of North Carolina at Chapel Hill 4/19/18 Blette (UNC) BIOS 740 Final Presentation 4/19/18 1 / 15 Overview 1 Motivation
Supplementary Online Content
 Supplementary Online Content Holme Ø, Løberg M, Kalager M, et al. Effect of flexible sigmoidoscopy screening on colorectal cancer incidence and mortality: a randomized clinical trial. JAMA. doi:10.1001/jama.2014.8266
Supplementary Online Content Holme Ø, Løberg M, Kalager M, et al. Effect of flexible sigmoidoscopy screening on colorectal cancer incidence and mortality: a randomized clinical trial. JAMA. doi:10.1001/jama.2014.8266
Discontinuation and restarting in patients on statin treatment: prospective open cohort study using a primary care database
 open access Discontinuation and restarting in patients on statin treatment: prospective open cohort study using a primary care database Yana Vinogradova, 1 Carol Coupland, 1 Peter Brindle, 2,3 Julia Hippisley-Cox
open access Discontinuation and restarting in patients on statin treatment: prospective open cohort study using a primary care database Yana Vinogradova, 1 Carol Coupland, 1 Peter Brindle, 2,3 Julia Hippisley-Cox
Right Answers, Wrong Ques2ons. Ralph I Horwitz
 Right Answers, Wrong Ques2ons Ralph I Horwitz Disclosures Employed by GlaxoSmithKline Views expressed reflect mine alone and not those of GSK Right Answer, Wrong Ques2on A young couple moves into an apartment
Right Answers, Wrong Ques2ons Ralph I Horwitz Disclosures Employed by GlaxoSmithKline Views expressed reflect mine alone and not those of GSK Right Answer, Wrong Ques2on A young couple moves into an apartment
Research Methodology. Characteristics of Observations. Variables 10/18/2016. Week Most important know what is being observed.
 Research Methodology 1 Characteristics of Observations 1. Most important know what is being observed. 2. Assign behaviors to categories. 3. Know how to Measure. 4. Degree of Observer inference. 2 Variables
Research Methodology 1 Characteristics of Observations 1. Most important know what is being observed. 2. Assign behaviors to categories. 3. Know how to Measure. 4. Degree of Observer inference. 2 Variables
Everolimus for the Treatment of Advanced Renal Cell Carcinoma (arcc ) - Additional Analysis Using RPSFT
 for the Treatment of Advanced Renal Cell Carcinoma (arcc ) - Additional Analysis Using RPSFT Thank you very much for the opportunity to submit additional analysis in relation to the above technology appraisal
for the Treatment of Advanced Renal Cell Carcinoma (arcc ) - Additional Analysis Using RPSFT Thank you very much for the opportunity to submit additional analysis in relation to the above technology appraisal
Comparative Effectiveness Research Using Observational Data: Active Comparators to Emulate Target Trials with Inactive Comparators
 Comparative Effectiveness Research Using Observational Data: Active Comparators to Emulate Target Trials with Inactive Comparators The Harvard community has made this article openly available. Please share
Comparative Effectiveness Research Using Observational Data: Active Comparators to Emulate Target Trials with Inactive Comparators The Harvard community has made this article openly available. Please share
Estimating Direct Effects of New HIV Prevention Methods. Focus: the MIRA Trial
 Estimating Direct Effects of New HIV Prevention Methods. Focus: the MIRA Trial Michael Rosenblum UCSF: Helen Cheng, Nancy Padian, Steve Shiboski, Ariane van der Straten Berkeley: Nicholas P. Jewell, Mark
Estimating Direct Effects of New HIV Prevention Methods. Focus: the MIRA Trial Michael Rosenblum UCSF: Helen Cheng, Nancy Padian, Steve Shiboski, Ariane van der Straten Berkeley: Nicholas P. Jewell, Mark
Epidemiological study design. Paul Pharoah Department of Public Health and Primary Care
 Epidemiological study design Paul Pharoah Department of Public Health and Primary Care Molecules What/why? Organelles Cells Tissues Organs Clinical medicine Individuals Public health medicine Populations
Epidemiological study design Paul Pharoah Department of Public Health and Primary Care Molecules What/why? Organelles Cells Tissues Organs Clinical medicine Individuals Public health medicine Populations
Study Designs in Epidemiology
 Study Designs in Epidemiology Dr. Sireen Alkhaldi, BDS, MPH, DrPH First semester 2017/ 2018 Department of Family and Community Medicine School of Medicine/ The University of Jordan Epidemiologic Study
Study Designs in Epidemiology Dr. Sireen Alkhaldi, BDS, MPH, DrPH First semester 2017/ 2018 Department of Family and Community Medicine School of Medicine/ The University of Jordan Epidemiologic Study
Adjusting overall survival for treatment switch
 Adjusting overall survival for treatment switch Recommendations of a crossinstitutional statistical working group Claire Watkins Director and Consultant Statistician, Clarostat Consulting Ltd Cytel East
Adjusting overall survival for treatment switch Recommendations of a crossinstitutional statistical working group Claire Watkins Director and Consultant Statistician, Clarostat Consulting Ltd Cytel East
Delfini Evidence Tool Kit
 General 1. Who is sponsoring and funding the study? What are the affiliations of the authors? Study Design Assessment Internal Validity Assessment Considerations: This can be helpful information and is
General 1. Who is sponsoring and funding the study? What are the affiliations of the authors? Study Design Assessment Internal Validity Assessment Considerations: This can be helpful information and is
Sensitivity Analysis in Observational Research: Introducing the E-value
 Sensitivity Analysis in Observational Research: Introducing the E-value Tyler J. VanderWeele Harvard T.H. Chan School of Public Health Departments of Epidemiology and Biostatistics 1 Plan of Presentation
Sensitivity Analysis in Observational Research: Introducing the E-value Tyler J. VanderWeele Harvard T.H. Chan School of Public Health Departments of Epidemiology and Biostatistics 1 Plan of Presentation
About Reading Scientific Studies
 About Reading Scientific Studies TABLE OF CONTENTS About Reading Scientific Studies... 1 Why are these skills important?... 1 Create a Checklist... 1 Introduction... 1 Abstract... 1 Background... 2 Methods...
About Reading Scientific Studies TABLE OF CONTENTS About Reading Scientific Studies... 1 Why are these skills important?... 1 Create a Checklist... 1 Introduction... 1 Abstract... 1 Background... 2 Methods...
Clinical Epidemiology I: Deciding on Appropriate Therapy
 Clinical Epidemiology I: Deciding on Appropriate Therapy 1 Clinical Scenario [UG2B] 65 yo man controlled HTN 6-mo Hx cardioversion-resistant afib Benefit vs risk of long-term anticoagulation:? prevent
Clinical Epidemiology I: Deciding on Appropriate Therapy 1 Clinical Scenario [UG2B] 65 yo man controlled HTN 6-mo Hx cardioversion-resistant afib Benefit vs risk of long-term anticoagulation:? prevent
A NEW TRIAL DESIGN FULLY INTEGRATING BIOMARKER INFORMATION FOR THE EVALUATION OF TREATMENT-EFFECT MECHANISMS IN PERSONALISED MEDICINE
 A NEW TRIAL DESIGN FULLY INTEGRATING BIOMARKER INFORMATION FOR THE EVALUATION OF TREATMENT-EFFECT MECHANISMS IN PERSONALISED MEDICINE Dr Richard Emsley Centre for Biostatistics, Institute of Population
A NEW TRIAL DESIGN FULLY INTEGRATING BIOMARKER INFORMATION FOR THE EVALUATION OF TREATMENT-EFFECT MECHANISMS IN PERSONALISED MEDICINE Dr Richard Emsley Centre for Biostatistics, Institute of Population
Systematic Reviews and Meta- Analysis in Kidney Transplantation
 Systematic Reviews and Meta- Analysis in Kidney Transplantation Greg Knoll MD MSc Associate Professor of Medicine Medical Director, Kidney Transplantation University of Ottawa and The Ottawa Hospital KRESCENT
Systematic Reviews and Meta- Analysis in Kidney Transplantation Greg Knoll MD MSc Associate Professor of Medicine Medical Director, Kidney Transplantation University of Ottawa and The Ottawa Hospital KRESCENT
PhD Course in Biostatistics
 PhD Course in Biostatistics Univ.-Prof. DI Dr. Andrea Berghold Institute for Medical Informatics, Statistics and Documentation Medical University of Graz andrea.berghold@medunigraz.at Content Introduction
PhD Course in Biostatistics Univ.-Prof. DI Dr. Andrea Berghold Institute for Medical Informatics, Statistics and Documentation Medical University of Graz andrea.berghold@medunigraz.at Content Introduction
Evidence Based Practice
 Evidence Based Practice RCS 6740 7/26/04 Evidence Based Practice: Definitions Evidence based practice is the integration of the best available external clinical evidence from systematic research with individual
Evidence Based Practice RCS 6740 7/26/04 Evidence Based Practice: Definitions Evidence based practice is the integration of the best available external clinical evidence from systematic research with individual
Noninferiority Clinical Trials
 Noninferiority Clinical Trials Scott Evans Harvard University Brasilia, Brazil May 7, 2010 Special Obrigado Valdair Pinto, M.D. Interfarma Outline Rationale and Examples Design Assumptions Constancy Assay
Noninferiority Clinical Trials Scott Evans Harvard University Brasilia, Brazil May 7, 2010 Special Obrigado Valdair Pinto, M.D. Interfarma Outline Rationale and Examples Design Assumptions Constancy Assay
Research in Real-World Settings: PCORI s Model for Comparative Clinical Effectiveness Research
 Research in Real-World Settings: PCORI s Model for Comparative Clinical Effectiveness Research David Hickam, MD, MPH Director, Clinical Effectiveness & Decision Science Program April 10, 2017 Welcome!
Research in Real-World Settings: PCORI s Model for Comparative Clinical Effectiveness Research David Hickam, MD, MPH Director, Clinical Effectiveness & Decision Science Program April 10, 2017 Welcome!
Using Statistical Principles to Implement FDA Guidance on Cardiovascular Risk Assessment for Diabetes Drugs
 Using Statistical Principles to Implement FDA Guidance on Cardiovascular Risk Assessment for Diabetes Drugs David Manner, Brenda Crowe and Linda Shurzinske BASS XVI November 9-13, 2009 Acknowledgements
Using Statistical Principles to Implement FDA Guidance on Cardiovascular Risk Assessment for Diabetes Drugs David Manner, Brenda Crowe and Linda Shurzinske BASS XVI November 9-13, 2009 Acknowledgements
Understanding noninferiority trials
 Review article http://dx.doi.org/10.3345/kjp.2012.55.11.403 Korean J Pediatr 2012;55(11):403-407 eissn 1738-1061 pissn 2092-7258 Understanding noninferiority trials Seokyung Hahn, PhD Department of Medicine,
Review article http://dx.doi.org/10.3345/kjp.2012.55.11.403 Korean J Pediatr 2012;55(11):403-407 eissn 1738-1061 pissn 2092-7258 Understanding noninferiority trials Seokyung Hahn, PhD Department of Medicine,
Causal versus Casual Inference
 ASA Biopharmaceutical Section Workshop Washington, DC 13 Sep 2018 Causal versus Casual Inference What Happens When I Take This Medication? Stephen J. Ruberg, PhD President Analytix Thinking, LLC AnalytixThinking@gmail.com
ASA Biopharmaceutical Section Workshop Washington, DC 13 Sep 2018 Causal versus Casual Inference What Happens When I Take This Medication? Stephen J. Ruberg, PhD President Analytix Thinking, LLC AnalytixThinking@gmail.com
Instrumental Variables Estimation: An Introduction
 Instrumental Variables Estimation: An Introduction Susan L. Ettner, Ph.D. Professor Division of General Internal Medicine and Health Services Research, UCLA The Problem The Problem Suppose you wish to
Instrumental Variables Estimation: An Introduction Susan L. Ettner, Ph.D. Professor Division of General Internal Medicine and Health Services Research, UCLA The Problem The Problem Suppose you wish to
C2 Training: August 2010
 C2 Training: August 2010 Introduction to meta-analysis The Campbell Collaboration www.campbellcollaboration.org Pooled effect sizes Average across studies Calculated using inverse variance weights Studies
C2 Training: August 2010 Introduction to meta-analysis The Campbell Collaboration www.campbellcollaboration.org Pooled effect sizes Average across studies Calculated using inverse variance weights Studies
Assessing methods for dealing with treatment switching in clinical trials: A follow-up simulation study
 Article Assessing methods for dealing with treatment switching in clinical trials: A follow-up simulation study Statistical Methods in Medical Research 2018, Vol. 27(3) 765 784! The Author(s) 2016 Reprints
Article Assessing methods for dealing with treatment switching in clinical trials: A follow-up simulation study Statistical Methods in Medical Research 2018, Vol. 27(3) 765 784! The Author(s) 2016 Reprints
