Rapid appraisal of the literature: Identifying study biases
|
|
|
- Duane Gervase Barber
- 6 years ago
- Views:
Transcription
1 Rapid appraisal of the literature: Identifying study biases Rita Popat, PhD Clinical Assistant Professor Division of Epidemiology Stanford University School of Medicine August 7, 2007
2 What is critical appraisal? Balanced assessment of benefits and strengths of research against its flaws and weaknesses Assessment of research process and results To be undertaken by all health professionals as part of their work
3 Why should we critically appraise? Published research is not always valid we cannot take conclusions for granted, even if the article is published in a peer-reviewed journal. Published research is not always relevant the abstract may indicate relevance but you will need to read the complete article to judge its applicability to your own practice/circumstances. To improve clinical effectiveness, we need a systematic framework to interpret research, rather than relying on a haphazard or casual approach. 3
4 Key Steps To Effective Critical Appraisal 1. Are the results valid? Focus of today s lecture 2. What are the results? 3. How will these results help me work with my patients? 4
5 Outline Quick review of study designs What is validity? Identifying study biases that can threaten internal validity 5
6 Observational vs. Experimental Studies Experimental studies the investigator tries to control the environment in which the hypothesis is tested (the randomized, double-blind clinical trial is the gold standard) Observational studies the population is observed without any interference by the investigator 6
7 Why Observational Studies? Cheaper Faster Can examine long-term effects Hypothesis-generating Sometimes, experimental studies are not ethical (e.g., randomizing subjects to smoke) Sometimes, experimental studies are not possible examples randomizing subjects to gestational diabetes studying natural progression of a disease studying long term effects of drugs 7
8 THE LANCET Vol 359 January 5, 2002
9 Cohort Studies Target population Exposed (E+) Disease-free cohort Not Exposed (E-) TIME Disease (I E+ ) Disease-free Disease (I E- ) Disease-free 9
10 Measure of association in cohort studies: Relative Risk (RR) Exposure + Outcome + a Outcome - b a + b Exposure - c d c + d RR = a c a + c + b d = I I E E + Incidence (probability) of outcome among exposed Incidence (probability)of outcome among unexposed 10
11 Active Inactive T2DM + T2DM RR = = = 0.73 Interpretation: Active women are 27% less likely to develop T2DM compared to inactive women Energy expenditure was at least 1000 kcal/wk. 11
12 Cohort Studies: Advantages & Disadvantages Advantages Allows you to measure true rates and risks of disease for the exposed and the unexposed groups. Temporality is correct Can be used to study multiple outcomes. Prevents bias in the ascertainment of exposure that may occur after a person develops a disease. Disadvantages Can be lengthy and costly! Loss to follow-up is a problem (especially if nonrandom). 12
13 Case-Control Studies Target population Cases (outcome +) Controls (outcome -) Exposed in past Not exposed Exposed in the past Not Exposed 13
14 Measure of Association in case-control studies: Odds Ratio (OR) Disease (D+) Cases No disease(d-) Controls Exposure (E+) a b No exposure(e-) c d a+c b+d OR = Odds of exposure among cases P( E D) Odds of exposure among controls = P( E D) P( E D ) P( E D ) = a c b d a+ c a+ c b + d b + d = ad bc 14
15 Cases Controls NSAIDS NSAIDS OR = ( 561 )( 74 ( 71 )( 971 ) ) = 0.60 Interpretation: NSAIDs use is associated with a 40% reduction in the risk of colon cancer 15
16 Case-Control Studies: Advantages & Disadvantages Advantages: Cheap and fast Great for rare diseases Disadvantages: Exposure estimates are subject to recall bias (those with the disease are searching for reasons why they got sick and may be more likely to report an exposure) interviewer bias (interviewer may prompt a positive response in cases). Temporality is a problem (did exposure cause disease or disease cause exposure?) 16
17 Intervention Studies Target population Intervention group Disease-free cohort No intervention group TIME Disease Disease-free Disease Disease-free 17
18 Eligible participants Randomized Standard lifestyle recommendations Intensive Metformin Placebo Lifestyle (n = 1079) (n = 1073) (n = 1082) 18
19 DPP trial: Primary Outcome - Diabetes 19
20 Intervention Studies: Advantages & Disadvantages Advantages: Allows randomization (controls for confounding) Allows double-blind assessment (controls bias) Disadvantages: Can be lengthy and costly! Loss to follow-up is a problem (especially if nonrandom). Ethical limitations 20
21 Causation in human health & disease Association does not prove causation If a putative risk factor and the occurrence of an outcome are strongly associated with each other it does not provide evidence that the risk factor causes the disease, only implies that it is correlated with outcome Non-causal explanations may cause a spurious association study biases (measurement error, selection bias, confounding, sampling error) 21
22 Study validity INTERNAL VALIDITY Do we believe the results? EXTERNAL VALIDITY Can the results be applied to the target population i.e., beyond the subjects in the study? 22
23 Threats to Internal validity: Non-causal explanations due to study biases Confounding Selection bias Misclassification bias (measurement error) 23
24 Confounding Confounding is the defined as a distortion of an exposure-outcome association brought about by the association of another factor with both the outcome and exposure exposure outcome Confounding factor 24
25 JAMA. 2004;292: Research question - Is physical activity associated with risk of T2DM? Physical activity T2DM + BMI + Potential confounder 25
26 Why worry about confounding? Spurious association Exaggerate an association (over-estimate) Attenuate an association (under-estimate) Obscure an association 26
27 Methods for controlling confounding Design phase Randomization Matching Restriction Analysis phase: statistical adjustment for confounders 27
28 Controlling confounding by randomization
29 Identifying potential confounders Subset of Table 1 from JAMA. 2004;292:
30 Statistical adjustment for confounding Crude HR =2.9/3.9 = 0.74 Is BMI a confounder of the relationship b/w physical activity and T2DM? Compare the crude hazard ratio (HR) to the adjusted HR. Weinstein et al. JAMA 292:
31 Statistical adjustment for confounding Weinstein et al. JAMA 292: % rule for identifying confounders: (Crude HR - Adjusted HR) X % Crude HR
32 True or false A randomized clinical trial design cannot be affected by bias due to confounding? Answer: False (if randomization is not done appropriately, then can introduce bias due to confounding) 32
33 Identifying bias due to confounding in a RCT Check the randomization procedure in the methods section (e.g., blocked randomization schemes when sample size is small) Check Table 1 see if groups are balanced If not, how was it handled? Was intention to treat analysis used? 33
34 JAMA. 2006;296:
35 JAMA. 2006;296:
36 Selection bias A form of sampling bias due to systematic differences between those who are selected for study (or agree to participate) and those who are not selected (or refuse to participate). 36
37 Selection bias Improper selection of cases or controls in a casecontrol study Subjects lost to follow-up varies according to both the exposure of interest and the outcome (e.g., in prospective cohort studies and clinical trials) Can affect any study design, although case-control studies more prone to selection bias Selection bias can cause either overestimates or underestimates of the true associations between the exposure and disease in the underlying population 37
38 Identifying Selection bias What are the response rates? Was follow-up complete? Ideally should have follow-up for at least 80% of the initial sample/cohort Does drop out differ in the groups being compared (e.g., treatment and placebo groups)? 38
39 Selection bias example: In this case-control study, response rate was ~80% in cases and ~60% in controls. Could there be selection bias especially among controls? 39
40 Selection bias example: Scenario: NSAID users in the base population were more likely to participate in this study than were non-nsaid users p 1 p 2 So observed proportion of NSAID users among controls (92.9%) is greater than true proportion of exposure Hence observed odds ratio of 0.54 is an overestimate (i.e., true odds ratio is greater than 0.54) [Note that Odds ratio = p 1 * (1- p 2 ) / p 2 *(1- p 1 )] 40
41 Some strategies for minimizing selection bias Careful enumeration and thorough attempts at recruiting all cases within the source population High standards for methods of control selection (population-based ideal) Minimizing non-response and refusals Minimizing loss to follow-up (in cohort and RCTs) 41
42 Information bias (aka measurement error) Misclassification of outcome Misclassification of exposure status 42
43 Information bias (measurement error) Imperfect definitions of study variables (outcome or predictors) or flawed data collection procedures Erroneous classification of outcome exposure 43
44 Information bias misclassification Gold-standard Outcome Outcome + - Study Outcome + Outcome - a TP c FN b FP d TN a+b c+d a+c b+d Sensitivity = a / (a+c) Specificity = d / (b+d) 44
45 Misclassification of the outcome Exposure + (treatment) Outcome + Outcome - a b Exposure (placebo) c d Non-differential misclassification occurs when the degree of misclassification of outcome is independent of exposure status Tends to bias the association toward the null Occurs when the sensitivity and specificity of the classification of outcome are same for exposed and non-exposed groups but less than 100% 45
46 Misclassification of the outcome Exposure + (treatment) Outcome + Outcome - a b Exposure (placebo) c d Differential misclassification occurs when the degree of misclassification differs between the groups being compared. May bias the association either toward or away from the null hypothesis Occurs when the sensitivity and specificity of the classification of outcome differ for exposed and nonexposed groups 46
47 Misclassification of the outcome: NSAIDs and colon cancer example Cases Controls NSAIDS NSAIDS Most likely scenario in this study Is misclassification of outcome likely to be nondifferential or differential with respect to exposure? 47
48 JAMA. 2006;296: Study Measures The primary measures were the Medical Outcomes Study 36- Item Short- Form Health Survey (SF-36) bodily pain and physical function scales and the American Academy of Orthopaedic Surgeons MODEMS version of the Oswestry Disability Index (ODI). Secondary measures included patient self-reported improvement, work status, and satisfaction with current symptoms and with care. Symptom severity was measured by the Sciatica Bothersomeness Index (range, 0-24; higher scores represent worse symptoms). Is misclassification of outcome likely to be nondifferential or differential with respect to exposure?
49
50 Misclassification of the exposure Cases (outcome +) Controls (outcome -) Exposure + a b Exposure - c d Non-differential misclassification occurs when the degree of misclassification of exposure is independent of outcome/disease status Tends to bias the association toward the null Occurs when the sensitivity and specificity of the classification of exposure are same for those with and without the outcome but less than 100% 50
51 Misclassification of the outcome Cases (outcome +) Controls (outcome -) Exposure + a b Exposure - c d Differential misclassification occurs when the degree of misclassification differs between the groups being compared. May bias the association either toward or away from the null hypothesis Occurs when the sensitivity and specificity of the classification of exposure differ for those with and without the outcome 51
52 Misclassification of the exposure: NSAIDs and colon cancer example Cases Controls NSAIDS NSAIDS Non-differential: poor recall in cases and controls Differential: cases recall NSAID use better than controls? Is misclassification of exposure likely to be nondifferential or differential with respect to outcome? 52
53 Some strategies for minimizing misclassification bias Test reliability and validity of instruments used to determine outcome and exposure Similar methods for determining outcome and exposure in all study subjects Train interviewers, blind interviewers to outcome status and study hypothesis ( interviewer bias) Blind subjects to study hypothesis ( recall bias) Use incident cases in case-control studies, not prevalent ( recall bias) Try to use objective measures (e.g., pharmacy records vs. self-report use of medications) 53
54 Summary : Study designs and biases Threats to internal validity Case-control Cohort RCT Confounding Generally present Generally present Not likely (due to randomization) Selection bias Likely (e.g., when low response rates) May occur due to differential loss to follow up May occur due to differential loss to follow-up Misclassification of exposure More likely to be differential Generally nondifferential Not likely; if exists then nondifferential (e.g., drop-in/drop out) Misclassification of outcome Most likely nondifferential Most likely nondifferential Most likely nondifferential 54
55 Once you are satisfied the study findings are valid You can now ask whether the association causal? Evaluate positive features of causation Temporality Strength of the association Dose-response relationship Consistency of findings Biologic plausibility 55
56 Best Experiment/RCT Prospective cohort Retrospective cohort Cost and ease Case-control Proof of cause Correlation/x-section Case series Case report Best 56
57 Critical appraisal tools Assist with systematic critique of published research papers Several tools available One of my favorite and look for link to CASP Appraisal Tools 57
58 Critical appraisal of an article about therapy or prevention Primary guides: Was the assignment of patients to treatment groups randomized? Were all patients who entered the trial properly accounted for at the conclusion? Was follow up complete? Were patients analyzed in the groups to which they were randomized, that is, was an intention to treat analysis used? 58
59 Critical appraisal of an article about therapy or prevention Secondary guides: Were patients, health workers and study personnel blinded to treatment? Does the study provide evidence that blinding was effective? Were the groups similar at the start of the trial? Was there an adequate Table 1? If not, were adjustments made for differences? Aside from the experimental intervention, were the groups treated equally? 59
60 Critical appraisal of an article about therapy or prevention What were the results? How large was the treatment effect? How precise was the estimate of the treatment effect? Were confidence intervals given? Will the results help me with my patients? Will discuss these aspects on August 9 th, 2007! 60
What is a case control study? Tarani Chandola Social Statistics University of Manchester
 What is a case control study? Tarani Chandola Social Statistics University of Manchester Imagine. It is 1950 You suspect an association b/w smoking and lung cancer How would you show this? Cases Find cases
What is a case control study? Tarani Chandola Social Statistics University of Manchester Imagine. It is 1950 You suspect an association b/w smoking and lung cancer How would you show this? Cases Find cases
Epidemiological study design. Paul Pharoah Department of Public Health and Primary Care
 Epidemiological study design Paul Pharoah Department of Public Health and Primary Care Molecules What/why? Organelles Cells Tissues Organs Clinical medicine Individuals Public health medicine Populations
Epidemiological study design Paul Pharoah Department of Public Health and Primary Care Molecules What/why? Organelles Cells Tissues Organs Clinical medicine Individuals Public health medicine Populations
Observational Study Designs. Review. Today. Measures of disease occurrence. Cohort Studies
 Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
UNDERSTANDING EPIDEMIOLOGICAL STUDIES. Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA
 UNDERSTANDING EPIDEMIOLOGICAL STUDIES Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA Study design in epidemiological research: Summary Observational
UNDERSTANDING EPIDEMIOLOGICAL STUDIES Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA Study design in epidemiological research: Summary Observational
Purpose. Study Designs. Objectives. Observational Studies. Analytic Studies
 Purpose Study Designs H.S. Teitelbaum, DO, PhD, MPH, FAOCOPM AOCOPM Annual Meeting Introduce notions of study design Clarify common terminology used with description and interpretation of information collected
Purpose Study Designs H.S. Teitelbaum, DO, PhD, MPH, FAOCOPM AOCOPM Annual Meeting Introduce notions of study design Clarify common terminology used with description and interpretation of information collected
Confounding and Bias
 28 th International Conference on Pharmacoepidemiology and Therapeutic Risk Management Barcelona, Spain August 22, 2012 Confounding and Bias Tobias Gerhard, PhD Assistant Professor, Ernest Mario School
28 th International Conference on Pharmacoepidemiology and Therapeutic Risk Management Barcelona, Spain August 22, 2012 Confounding and Bias Tobias Gerhard, PhD Assistant Professor, Ernest Mario School
EBM, Study Design and Numbers. David Frankfurter, MD Professor OB/GYN The George Washington University
 EBM, Study Design and Numbers David Frankfurter, MD Professor OB/GYN The George Washington University 1978? 1978 Best Picture of 1978 The Deer Hunter Universal Studios The 50th Academy Awards were held
EBM, Study Design and Numbers David Frankfurter, MD Professor OB/GYN The George Washington University 1978? 1978 Best Picture of 1978 The Deer Hunter Universal Studios The 50th Academy Awards were held
Study design. Chapter 64. Research Methodology S.B. MARTINS, A. ZIN, W. ZIN
 Chapter 64 Study design S.B. MARTINS, A. ZIN, W. ZIN Research Methodology Scientific methodology comprises a set of rules and procedures to investigate the phenomena of interest. These rules and procedures
Chapter 64 Study design S.B. MARTINS, A. ZIN, W. ZIN Research Methodology Scientific methodology comprises a set of rules and procedures to investigate the phenomena of interest. These rules and procedures
Health Studies 315. Clinical Epidemiology: Evidence of Risk and Harm
 Health Studies 315 Clinical Epidemiology: Evidence of Risk and Harm 1 Patients encounter (possibly) risky exposures Alcohol during pregnancy (fetal risk) Electromagnetic fields (cancer risk) Vasectomy
Health Studies 315 Clinical Epidemiology: Evidence of Risk and Harm 1 Patients encounter (possibly) risky exposures Alcohol during pregnancy (fetal risk) Electromagnetic fields (cancer risk) Vasectomy
University of Wollongong. Research Online. Australian Health Services Research Institute
 University of Wollongong Research Online Australian Health Services Research Institute Faculty of Business 2011 Measurement of error Janet E. Sansoni University of Wollongong, jans@uow.edu.au Publication
University of Wollongong Research Online Australian Health Services Research Institute Faculty of Business 2011 Measurement of error Janet E. Sansoni University of Wollongong, jans@uow.edu.au Publication
Anssi Auvinen University of Tampere STUK Radiation and Nuclear Safety Authority International Agency for Research on Cancer
 Sources of uncertainty in epidemiological studies Anssi Auvinen University of Tampere STUK Radiation and Nuclear Safety Authority International Agency for Research on Cancer Error and bias Concepts not
Sources of uncertainty in epidemiological studies Anssi Auvinen University of Tampere STUK Radiation and Nuclear Safety Authority International Agency for Research on Cancer Error and bias Concepts not
Observational Medical Studies. HRP 261 1/13/ am
 Observational Medical Studies HRP 261 1/13/03 10-11 11 am To Drink or Not to Drink? Volume 348:163-164 January 9, 2003 Ira J. Goldberg, M.D. A number of epidemiologic studies have found an association
Observational Medical Studies HRP 261 1/13/03 10-11 11 am To Drink or Not to Drink? Volume 348:163-164 January 9, 2003 Ira J. Goldberg, M.D. A number of epidemiologic studies have found an association
Types of Biomedical Research
 INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency Family Medicine Section of
INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency Family Medicine Section of
An Introduction to Epidemiology
 An Introduction to Epidemiology Wei Liu, MPH Biostatistics Core Pennington Biomedical Research Center Baton Rouge, LA Last edited: January, 14 th, 2014 TABLE OF CONTENTS Introduction.................................................................
An Introduction to Epidemiology Wei Liu, MPH Biostatistics Core Pennington Biomedical Research Center Baton Rouge, LA Last edited: January, 14 th, 2014 TABLE OF CONTENTS Introduction.................................................................
Bias. Zuber D. Mulla
 Bias Zuber D. Mulla Explanations when you Observe or Don t Observe an Association Truth Chance Bias Confounding From Epidemiology in Medicine (Hennekens & Buring) Bias When you detect an association or
Bias Zuber D. Mulla Explanations when you Observe or Don t Observe an Association Truth Chance Bias Confounding From Epidemiology in Medicine (Hennekens & Buring) Bias When you detect an association or
Epidemiologic Methods and Counting Infections: The Basics of Surveillance
 Epidemiologic Methods and Counting Infections: The Basics of Surveillance Ebbing Lautenbach, MD, MPH, MSCE University of Pennsylvania School of Medicine Nothing to disclose PENN Outline Definitions / Historical
Epidemiologic Methods and Counting Infections: The Basics of Surveillance Ebbing Lautenbach, MD, MPH, MSCE University of Pennsylvania School of Medicine Nothing to disclose PENN Outline Definitions / Historical
The role of Randomized Controlled Trials
 The role of Randomized Controlled Trials Dr. Georgia Salanti Lecturer in Epidemiology University of Ioannina School of Medicine Outline Understanding study designs and the role of confounding Observational
The role of Randomized Controlled Trials Dr. Georgia Salanti Lecturer in Epidemiology University of Ioannina School of Medicine Outline Understanding study designs and the role of confounding Observational
Epidemiology & Evidence Based Medicine. Patrick Linden & Matthew McCall
 Epidemiology & Evidence Based Medicine Patrick Linden & Matthew McCall Areas Covered in this Talk Bradford Hill Criteria Bias and confounding Study designs, advantages and disadvantages Basic definitions
Epidemiology & Evidence Based Medicine Patrick Linden & Matthew McCall Areas Covered in this Talk Bradford Hill Criteria Bias and confounding Study designs, advantages and disadvantages Basic definitions
3. Factors such as race, age, sex, and a person s physiological state are all considered determinants of disease. a. True
 / False 1. Epidemiology is the basic science of public health. LEARNING OBJECTIVES: CNIA.BOYL.17.2.1 - Define epidemiology. 2. Within the field of epidemiology, the term distribution refers to the relationship
/ False 1. Epidemiology is the basic science of public health. LEARNING OBJECTIVES: CNIA.BOYL.17.2.1 - Define epidemiology. 2. Within the field of epidemiology, the term distribution refers to the relationship
Clinical Research Design and Conduction
 Clinical Research Design and Conduction Study Design M Mohsen Ibrahim, MD Cardiac Department Cairo University Algorithm for classification of types of clinical research Grimes DA and Schulz KF. Lancet
Clinical Research Design and Conduction Study Design M Mohsen Ibrahim, MD Cardiac Department Cairo University Algorithm for classification of types of clinical research Grimes DA and Schulz KF. Lancet
12/26/2013. Types of Biomedical Research. Clinical Research. 7Steps to do research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH S T A T I S T I C
 Types of Biomedical Research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency
Types of Biomedical Research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency
Challenges of Observational and Retrospective Studies
 Challenges of Observational and Retrospective Studies Kyoungmi Kim, Ph.D. March 8, 2017 This seminar is jointly supported by the following NIH-funded centers: Background There are several methods in which
Challenges of Observational and Retrospective Studies Kyoungmi Kim, Ph.D. March 8, 2017 This seminar is jointly supported by the following NIH-funded centers: Background There are several methods in which
Incorporating Clinical Information into the Label
 ECC Population Health Group LLC expertise-driven consulting in global maternal-child health & pharmacoepidemiology Incorporating Clinical Information into the Label Labels without Categories: A Workshop
ECC Population Health Group LLC expertise-driven consulting in global maternal-child health & pharmacoepidemiology Incorporating Clinical Information into the Label Labels without Categories: A Workshop
Chapter 2. Epidemiological and Toxicological Studies
 Chapter 2 Epidemiological and Toxicological Studies Introduction Epidemiological and toxicological studies seek to determine the cause of a particular illness or disease. Etiology is the study of causes.
Chapter 2 Epidemiological and Toxicological Studies Introduction Epidemiological and toxicological studies seek to determine the cause of a particular illness or disease. Etiology is the study of causes.
Bias. Sam Bracebridge
 Bias Sam Bracebridge Bias Errors in epidemiological measurements Based on the slides of Sam Bracebridge By the end of the lecture fellows will be able to Define bias Identify different types of bias Explain
Bias Sam Bracebridge Bias Errors in epidemiological measurements Based on the slides of Sam Bracebridge By the end of the lecture fellows will be able to Define bias Identify different types of bias Explain
EPIDEMIOLOGY-BIOSTATISTICS EXAM Midterm 2004 PRINT YOUR LEGAL NAME:
 EPIDEMIOLOGY-BIOSTATISTICS EXAM Midterm 2004 PRINT YOUR LEGAL NAME: Instructions: This exam is 20% of your course grade. The maximum number of points for the course is 1,000; hence, this exam is worth
EPIDEMIOLOGY-BIOSTATISTICS EXAM Midterm 2004 PRINT YOUR LEGAL NAME: Instructions: This exam is 20% of your course grade. The maximum number of points for the course is 1,000; hence, this exam is worth
HARM. Definition modified from the IHI definition of Harm by the QUEST Harm Workgroup
 Indonesia Clinical epidemiology and Evidence Based Medicine (ICE(ICE-EBM) HARM Jarir At Thobari (FK UGM) Efficacious Medical intervention Harm HARM Unintended physical injury resulting from or contributed
Indonesia Clinical epidemiology and Evidence Based Medicine (ICE(ICE-EBM) HARM Jarir At Thobari (FK UGM) Efficacious Medical intervention Harm HARM Unintended physical injury resulting from or contributed
Epidemiologic Study Designs. (RCTs)
 Epidemiologic Study Designs Epidemiologic Study Designs Experimental (RCTs) Observational Analytical Descriptive Case-Control Cohort + cross-sectional & ecologic Epidemiologic Study Designs Descriptive
Epidemiologic Study Designs Epidemiologic Study Designs Experimental (RCTs) Observational Analytical Descriptive Case-Control Cohort + cross-sectional & ecologic Epidemiologic Study Designs Descriptive
ADENIYI MOFOLUWAKE MPH APPLIED EPIDEMIOLOGY WEEK 5 CASE STUDY ASSIGNMENT APRIL
 ADENIYI MOFOLUWAKE MPH 510 - APPLIED EPIDEMIOLOGY WEEK 5 CASE STUDY ASSIGNMENT APRIL 4 2013 Question 1: What makes the first study a case-control study? The first case study is a case-control study because
ADENIYI MOFOLUWAKE MPH 510 - APPLIED EPIDEMIOLOGY WEEK 5 CASE STUDY ASSIGNMENT APRIL 4 2013 Question 1: What makes the first study a case-control study? The first case study is a case-control study because
How to use this appraisal tool: Three broad issues need to be considered when appraising a case control study:
 CASP Checklist: 11 questions to help you make sense of a Case Control Study How to use this appraisal tool: Three broad issues need to be considered when appraising a case control study: Are the results
CASP Checklist: 11 questions to help you make sense of a Case Control Study How to use this appraisal tool: Three broad issues need to be considered when appraising a case control study: Are the results
Bias. A systematic error (caused by the investigator or the subjects) that causes an incorrect (overor under-) estimate of an association.
 Bias A systematic error (caused by the investigator or the subjects) that causes an incorrect (overor under-) estimate of an association. Here, random error is small, but systematic errors have led to
Bias A systematic error (caused by the investigator or the subjects) that causes an incorrect (overor under-) estimate of an association. Here, random error is small, but systematic errors have led to
STUDY DESIGNS WHICH ONE IS BEST?
 STUDY DESIGNS WHICH ONE IS BEST? David Nunan, PhD Research Fellow and Tutor Nuffield Department of Primary Care Health Sciences and Oxford Centre for Evidence Based Medicine University of Oxford Exercise
STUDY DESIGNS WHICH ONE IS BEST? David Nunan, PhD Research Fellow and Tutor Nuffield Department of Primary Care Health Sciences and Oxford Centre for Evidence Based Medicine University of Oxford Exercise
11 questions to help you make sense of a case control study
 Critical Appraisal Skills Programme (CASP) making sense of evidence 11 questions to help you make sense of a case control study How to use this appraisal tool Three broad issues need to be considered when
Critical Appraisal Skills Programme (CASP) making sense of evidence 11 questions to help you make sense of a case control study How to use this appraisal tool Three broad issues need to be considered when
CRITICAL APPRAISAL OF MEDICAL LITERATURE. Samuel Iff ISPM Bern
 CRITICAL APPRAISAL OF MEDICAL LITERATURE Samuel Iff ISPM Bern siff@ispm.unibe.ch Contents Study designs Asking good questions Pitfalls in clinical studies How to assess validity (RCT) Conclusion Step-by-step
CRITICAL APPRAISAL OF MEDICAL LITERATURE Samuel Iff ISPM Bern siff@ispm.unibe.ch Contents Study designs Asking good questions Pitfalls in clinical studies How to assess validity (RCT) Conclusion Step-by-step
Research Methodology Workshop. Study Type
 Research Methodology Workshop Study Type What type of question? Frequency: how common is it? Aetiology: What caused this? Intervention: Does this intervention work? Diagnosis or test evaluation: How accurate
Research Methodology Workshop Study Type What type of question? Frequency: how common is it? Aetiology: What caused this? Intervention: Does this intervention work? Diagnosis or test evaluation: How accurate
Evidence-Based Medicine Journal Club. A Primer in Statistics, Study Design, and Epidemiology. August, 2013
 Evidence-Based Medicine Journal Club A Primer in Statistics, Study Design, and Epidemiology August, 2013 Rationale for EBM Conscientious, explicit, and judicious use Beyond clinical experience and physiologic
Evidence-Based Medicine Journal Club A Primer in Statistics, Study Design, and Epidemiology August, 2013 Rationale for EBM Conscientious, explicit, and judicious use Beyond clinical experience and physiologic
Clinical problems and choice of study designs
 Evidence Based Dentistry Clinical problems and choice of study designs Asbjørn Jokstad University of Oslo, Norway Nov 21 2001 1 Manipulation with intervention Yes Experimental study No Non-experimental
Evidence Based Dentistry Clinical problems and choice of study designs Asbjørn Jokstad University of Oslo, Norway Nov 21 2001 1 Manipulation with intervention Yes Experimental study No Non-experimental
Penelitian IKM/Epidemiologi. Contact: Blog: suyatno.blog.undip.ac.id Hp/Telp: /
 Penelitian IKM/Epidemiologi Oleh : Suyatno, Ir. MKes Contact: E-mail: suyatnofkmundip@gmail.com Blog: suyatno.blog.undip.ac.id Hp/Telp: 08122815730 / 024-70251915 Epidemiologic Study Designs I. Experimental
Penelitian IKM/Epidemiologi Oleh : Suyatno, Ir. MKes Contact: E-mail: suyatnofkmundip@gmail.com Blog: suyatno.blog.undip.ac.id Hp/Telp: 08122815730 / 024-70251915 Epidemiologic Study Designs I. Experimental
GLOSSARY OF GENERAL TERMS
 GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
CRITICAL APPRAISAL SKILLS PROGRAMME Making sense of evidence about clinical effectiveness. 11 questions to help you make sense of case control study
 CRITICAL APPRAISAL SKILLS PROGRAMME Making sense of evidence about clinical effectiveness 11 questions to help you make sense of case control study General comments Three broad issues need to be considered
CRITICAL APPRAISAL SKILLS PROGRAMME Making sense of evidence about clinical effectiveness 11 questions to help you make sense of case control study General comments Three broad issues need to be considered
Choosing the right study design
 Choosing the right study design Main types of study design Randomised controlled trial (RCT) Cohort study Case-control study Cross-sectional study Case series/case note review Expert opinion BEST QUALITY
Choosing the right study design Main types of study design Randomised controlled trial (RCT) Cohort study Case-control study Cross-sectional study Case series/case note review Expert opinion BEST QUALITY
Trial Designs. Professor Peter Cameron
 Trial Designs Professor Peter Cameron OVERVIEW Review of Observational methods Principles of experimental design applied to observational studies Population Selection Looking for bias Inference Analysis
Trial Designs Professor Peter Cameron OVERVIEW Review of Observational methods Principles of experimental design applied to observational studies Population Selection Looking for bias Inference Analysis
Tool to Assess Risk of Bias in Cohort Studies
 Tool to Assess Risk of Bias in Cohort Studies Contributed by the CLARITY Group at McMaster University 1. Was selection of exposed and non-exposed cohorts drawn from the same population? Exposed and unexposed
Tool to Assess Risk of Bias in Cohort Studies Contributed by the CLARITY Group at McMaster University 1. Was selection of exposed and non-exposed cohorts drawn from the same population? Exposed and unexposed
Bias and confounding. Mads Kamper-Jørgensen, associate professor, Section of Social Medicine
 Bias and confounding Mads Kamper-Jørgensen, associate professor, maka@sund.ku.dk PhD-course in Epidemiology l 7 February 2017 l Slide number 1 The world according to an epidemiologist Exposure Outcome
Bias and confounding Mads Kamper-Jørgensen, associate professor, maka@sund.ku.dk PhD-course in Epidemiology l 7 February 2017 l Slide number 1 The world according to an epidemiologist Exposure Outcome
Epidemiology: Overview of Key Concepts and Study Design. Polly Marchbanks
 Epidemiology: Overview of Key Concepts and Study Design Polly Marchbanks Lecture Outline (1) Key epidemiologic concepts - Definition - What epi is not - What epi is - Process of epi research Lecture Outline
Epidemiology: Overview of Key Concepts and Study Design Polly Marchbanks Lecture Outline (1) Key epidemiologic concepts - Definition - What epi is not - What epi is - Process of epi research Lecture Outline
Lecture 5 Conducting Interviews and Focus Groups
 Lecture 5 Conducting Interviews and Focus Groups Talking to participants enables in-depth information about the experiences of health and illness; and of factors that influence health and illness behaviour
Lecture 5 Conducting Interviews and Focus Groups Talking to participants enables in-depth information about the experiences of health and illness; and of factors that influence health and illness behaviour
CONCEPTUALIZING A RESEARCH DESIGN
 CONCEPTUALIZING A RESEARCH DESIGN Detty Nurdiati Dept of Obstetrics & Gynecology Fac of Medicine, Universitas Gadjah Mada Yogyakarta, Indonesia Conceptualizing a Research Design The Research Design The
CONCEPTUALIZING A RESEARCH DESIGN Detty Nurdiati Dept of Obstetrics & Gynecology Fac of Medicine, Universitas Gadjah Mada Yogyakarta, Indonesia Conceptualizing a Research Design The Research Design The
Welcome to this series focused on sources of bias in epidemiologic studies. In this first module, I will provide a general overview of bias.
 Welcome to this series focused on sources of bias in epidemiologic studies. In this first module, I will provide a general overview of bias. In the second module, we will focus on selection bias and in
Welcome to this series focused on sources of bias in epidemiologic studies. In this first module, I will provide a general overview of bias. In the second module, we will focus on selection bias and in
NUHS Evidence Based Practice I Journal Club. Date:
 Topic NUHS Evidence Based Practice I Journal Club Team Members: Date: Featured Research Article: Vancouver format example: Weinstein JN, Tosteson TD, Lurie JD, Tosteson AN, Hanscom B, Skinner JS, Abdu
Topic NUHS Evidence Based Practice I Journal Club Team Members: Date: Featured Research Article: Vancouver format example: Weinstein JN, Tosteson TD, Lurie JD, Tosteson AN, Hanscom B, Skinner JS, Abdu
Confounding and Interaction
 Confounding and Interaction Why did you do clinical research? To find a better diagnosis tool To determine risk factor of disease To identify prognosis factor To evaluate effectiveness of therapy To decide
Confounding and Interaction Why did you do clinical research? To find a better diagnosis tool To determine risk factor of disease To identify prognosis factor To evaluate effectiveness of therapy To decide
The comparison or control group may be allocated a placebo intervention, an alternative real intervention or no intervention at all.
 1. RANDOMISED CONTROLLED TRIALS (Treatment studies) (Relevant JAMA User s Guides, Numbers IIA & B: references (3,4) Introduction: The most valid study design for assessing the effectiveness (both the benefits
1. RANDOMISED CONTROLLED TRIALS (Treatment studies) (Relevant JAMA User s Guides, Numbers IIA & B: references (3,4) Introduction: The most valid study design for assessing the effectiveness (both the benefits
Evaluating Scientific Journal Articles. Tufts CTSI s Mission & Purpose. Tufts Clinical and Translational Science Institute
 Tufts Clinical and Translational Science Institute Lori Lyn Price, MAS Biostatistics, Epidemiology, and Research Design (BERD) Tufts Clinical and Translational Science Institute (CTSI) Tufts CTSI s Mission
Tufts Clinical and Translational Science Institute Lori Lyn Price, MAS Biostatistics, Epidemiology, and Research Design (BERD) Tufts Clinical and Translational Science Institute (CTSI) Tufts CTSI s Mission
Glossary of Practical Epidemiology Concepts
 Glossary of Practical Epidemiology Concepts - 2009 Adapted from the McMaster EBCP Workshop 2003, McMaster University, Hamilton, Ont. Note that open access to the much of the materials used in the Epi-546
Glossary of Practical Epidemiology Concepts - 2009 Adapted from the McMaster EBCP Workshop 2003, McMaster University, Hamilton, Ont. Note that open access to the much of the materials used in the Epi-546
INTERNAL VALIDITY, BIAS AND CONFOUNDING
 OCW Epidemiology and Biostatistics, 2010 J. Forrester, PhD Tufts University School of Medicine October 6, 2010 INTERNAL VALIDITY, BIAS AND CONFOUNDING Learning objectives for this session: 1) Understand
OCW Epidemiology and Biostatistics, 2010 J. Forrester, PhD Tufts University School of Medicine October 6, 2010 INTERNAL VALIDITY, BIAS AND CONFOUNDING Learning objectives for this session: 1) Understand
Evidence- and Value-based Solutions for Health Care Clinical Improvement Consults, Content Development, Training & Seminars, Tools
 Definition Key Points Key Problems Bias Choice Lack of Control Chance Observational Study Defined Epidemiological study in which observations are made, but investigators do not control the exposure or
Definition Key Points Key Problems Bias Choice Lack of Control Chance Observational Study Defined Epidemiological study in which observations are made, but investigators do not control the exposure or
Mixed Methods Study Design
 1 Mixed Methods Study Design Kurt C. Stange, MD, PhD Professor of Family Medicine, Epidemiology & Biostatistics, Oncology and Sociology Case Western Reserve University 1. Approaches 1, 2 a. Qualitative
1 Mixed Methods Study Design Kurt C. Stange, MD, PhD Professor of Family Medicine, Epidemiology & Biostatistics, Oncology and Sociology Case Western Reserve University 1. Approaches 1, 2 a. Qualitative
Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis
 EFSA/EBTC Colloquium, 25 October 2017 Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis Julian Higgins University of Bristol 1 Introduction to concepts Standard
EFSA/EBTC Colloquium, 25 October 2017 Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis Julian Higgins University of Bristol 1 Introduction to concepts Standard
INTRODUCTION TO EPIDEMIOLOGICAL STUDY DESIGNS PHUNLERD PIYARAJ, MD., MHS., PHD.
 INTRODUCTION TO EPIDEMIOLOGICAL STUDY DESIGNS PHUNLERD PIYARAJ, MD., MHS., PHD. 1 OBJECTIVES By the end of this section, you will be able to: Provide a definition of epidemiology Describe the major types
INTRODUCTION TO EPIDEMIOLOGICAL STUDY DESIGNS PHUNLERD PIYARAJ, MD., MHS., PHD. 1 OBJECTIVES By the end of this section, you will be able to: Provide a definition of epidemiology Describe the major types
Data that can be classified as belonging to a distinct number of categories >>result in categorical responses. And this includes:
 This sheets starts from slide #83 to the end ofslide #4. If u read this sheet you don`t have to return back to the slides at all, they are included here. Categorical Data (Qualitative data): Data that
This sheets starts from slide #83 to the end ofslide #4. If u read this sheet you don`t have to return back to the slides at all, they are included here. Categorical Data (Qualitative data): Data that
Objectives. Distinguish between primary and secondary studies. Discuss ways to assess methodological quality. Review limitations of all studies.
 Study Design Objectives 2 Distinguish between primary and secondary studies. Discuss ways to assess methodological quality. Review limitations of all studies. Key Terms 3 Evidence based medicine Primary
Study Design Objectives 2 Distinguish between primary and secondary studies. Discuss ways to assess methodological quality. Review limitations of all studies. Key Terms 3 Evidence based medicine Primary
Mobile Phones & Cancer How does epidemiology investigate this?
 Mobile Phones & Cancer How does epidemiology investigate this? Anthony Swerdlow ICNIRP MEMBER/ CHAIR OF THE ICNIRP STANDING COMMITTEE ON EPIDEMIOLOGY INSTITUTE OF CANCER RESEARCH There are now >5 billion
Mobile Phones & Cancer How does epidemiology investigate this? Anthony Swerdlow ICNIRP MEMBER/ CHAIR OF THE ICNIRP STANDING COMMITTEE ON EPIDEMIOLOGY INSTITUTE OF CANCER RESEARCH There are now >5 billion
Case-control studies. Hans Wolff. Service d épidémiologie clinique Département de médecine communautaire. WHO- Postgraduate course 2007 CC studies
 Case-control studies Hans Wolff Service d épidémiologie clinique Département de médecine communautaire Hans.Wolff@hcuge.ch Outline Case-control study Relation to cohort study Selection of controls Sampling
Case-control studies Hans Wolff Service d épidémiologie clinique Département de médecine communautaire Hans.Wolff@hcuge.ch Outline Case-control study Relation to cohort study Selection of controls Sampling
Module 2: Fundamentals of Epidemiology Issues of Interpretation. Slide 1: Introduction. Slide 2: Acknowledgements. Slide 3: Presentation Objectives
 Slide 1: Introduction Issues of of Interpretation in in Epidemiologic Studies Developed through APTR Initiative to Enhance Prevention and Population Health Developed Education through in collaboration
Slide 1: Introduction Issues of of Interpretation in in Epidemiologic Studies Developed through APTR Initiative to Enhance Prevention and Population Health Developed Education through in collaboration
Epidemiologic study designs
 Epidemiologic study designs and critical appraisal of scientific papers Rolf H.H. Groenwold, MD, PhD Bio sketch MD, PhD in epidemiology Associate professor of Epidemiology at UMC Utrecht Research focus
Epidemiologic study designs and critical appraisal of scientific papers Rolf H.H. Groenwold, MD, PhD Bio sketch MD, PhD in epidemiology Associate professor of Epidemiology at UMC Utrecht Research focus
Main objective of Epidemiology. Statistical Inference. Statistical Inference: Example. Statistical Inference: Example
 Main objective of Epidemiology Inference to a population Example: Treatment of hypertension: Research question (hypothesis): Is treatment A better than treatment B for patients with hypertension? Study
Main objective of Epidemiology Inference to a population Example: Treatment of hypertension: Research question (hypothesis): Is treatment A better than treatment B for patients with hypertension? Study
Gender Identity and Expression. Week 8
 Gender Identity and Expression Week 8 1 Objectives 1. Describe research methods for studying gender identity and expression as they relate to health 2. Identify some key health disparities in relation
Gender Identity and Expression Week 8 1 Objectives 1. Describe research methods for studying gender identity and expression as they relate to health 2. Identify some key health disparities in relation
HOW TO CRITIQUE AN ARTICLE ON THERAPY
 HOW TO CRITIQUE AN ARTICLE ON THERAPY DR. BRUCE F. D.C., M.P.H.* Abstract: The ability to critique the literature and source out relevant information to answer a clinical question is a skill that is being
HOW TO CRITIQUE AN ARTICLE ON THERAPY DR. BRUCE F. D.C., M.P.H.* Abstract: The ability to critique the literature and source out relevant information to answer a clinical question is a skill that is being
PLS 506 Mark T. Imperial, Ph.D. Lecture Notes: Reliability & Validity
 PLS 506 Mark T. Imperial, Ph.D. Lecture Notes: Reliability & Validity Measurement & Variables - Initial step is to conceptualize and clarify the concepts embedded in a hypothesis or research question with
PLS 506 Mark T. Imperial, Ph.D. Lecture Notes: Reliability & Validity Measurement & Variables - Initial step is to conceptualize and clarify the concepts embedded in a hypothesis or research question with
Department of OUTCOMES RESEARCH
 Department of OUTCOMES RESEARCH Clinical Research Design Sources of Error Types of Clinical Research Randomized Trials! Daniel I. Sessler, M.D. Professor and Chair Department of OUTCOMES RESEARCH The Cleveland
Department of OUTCOMES RESEARCH Clinical Research Design Sources of Error Types of Clinical Research Randomized Trials! Daniel I. Sessler, M.D. Professor and Chair Department of OUTCOMES RESEARCH The Cleveland
Appraising the Literature Overview of Study Designs
 Chapter 5 Appraising the Literature Overview of Study Designs Barbara M. Sullivan, PhD Department of Research, NUHS Jerrilyn A. Cambron, PhD, DC Department of Researach, NUHS EBP@NUHS Ch 5 - Overview of
Chapter 5 Appraising the Literature Overview of Study Designs Barbara M. Sullivan, PhD Department of Research, NUHS Jerrilyn A. Cambron, PhD, DC Department of Researach, NUHS EBP@NUHS Ch 5 - Overview of
Updates in Therapeutics 2015: The Pharmacotherapy Preparatory Review & Recertification Course
 Updates in Therapeutics 2015: The Pharmacotherapy Preparatory Review & Recertification Course Study Designs: Fundamentals and Interpretation Kevin M. Sowinski, Pharm.D., FCCP Purdue University, College
Updates in Therapeutics 2015: The Pharmacotherapy Preparatory Review & Recertification Course Study Designs: Fundamentals and Interpretation Kevin M. Sowinski, Pharm.D., FCCP Purdue University, College
First author. Title: Journal
 First author Title: Journal Year: Volume: Issue: First page: Last Page: 1. Type of study Controlled trial without randomization Section B Observational cohort study Section B 2. Type of report Full paper
First author Title: Journal Year: Volume: Issue: First page: Last Page: 1. Type of study Controlled trial without randomization Section B Observational cohort study Section B 2. Type of report Full paper
Applied Analysis of Variance and Experimental Design. Lukas Meier, Seminar für Statistik
 Applied Analysis of Variance and Experimental Design Lukas Meier, Seminar für Statistik About Me Studied mathematics at ETH. Worked at the statistical consulting service and did a PhD in statistics (at
Applied Analysis of Variance and Experimental Design Lukas Meier, Seminar für Statistik About Me Studied mathematics at ETH. Worked at the statistical consulting service and did a PhD in statistics (at
Clinical research in AKI Timing of initiation of dialysis in AKI
 Clinical research in AKI Timing of initiation of dialysis in AKI Josée Bouchard, MD Krescent Workshop December 10 th, 2011 1 Acute kidney injury in ICU 15 25% of critically ill patients experience AKI
Clinical research in AKI Timing of initiation of dialysis in AKI Josée Bouchard, MD Krescent Workshop December 10 th, 2011 1 Acute kidney injury in ICU 15 25% of critically ill patients experience AKI
Controlling Bias & Confounding
 Controlling Bias & Confounding Chihaya Koriyama August 5 th, 2015 QUESTIONS FOR BIAS Key concepts Bias Should be minimized at the designing stage. Random errors We can do nothing at Is the nature the of
Controlling Bias & Confounding Chihaya Koriyama August 5 th, 2015 QUESTIONS FOR BIAS Key concepts Bias Should be minimized at the designing stage. Random errors We can do nothing at Is the nature the of
exposure/intervention
 Kathleen A. Kennedy, MD, MPH University of Texas Medical School at Houston Medical School Most clinical research evaluates an association between exposure/intervention outcome. 1 Does the investigator
Kathleen A. Kennedy, MD, MPH University of Texas Medical School at Houston Medical School Most clinical research evaluates an association between exposure/intervention outcome. 1 Does the investigator
Biases in clinical research. Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University
 Biases in clinical research Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University Learning objectives Describe the threats to causal inferences in clinical studies Understand the role of
Biases in clinical research Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University Learning objectives Describe the threats to causal inferences in clinical studies Understand the role of
CHECK-LISTS AND Tools DR F. R E Z A E I DR E. G H A D E R I K U R D I S TA N U N I V E R S I T Y O F M E D I C A L S C I E N C E S
 CHECK-LISTS AND Tools DR F. R E Z A E I DR E. G H A D E R I K U R D I S TA N U N I V E R S I T Y O F M E D I C A L S C I E N C E S What is critical appraisal? Critical appraisal is the assessment of evidence
CHECK-LISTS AND Tools DR F. R E Z A E I DR E. G H A D E R I K U R D I S TA N U N I V E R S I T Y O F M E D I C A L S C I E N C E S What is critical appraisal? Critical appraisal is the assessment of evidence
Causal Association : Cause To Effect. Dr. Akhilesh Bhargava MD, DHA, PGDHRM Prof. Community Medicine & Director-SIHFW, Jaipur
 Causal Association : Cause To Effect Dr. MD, DHA, PGDHRM Prof. Community Medicine & Director-SIHFW, Jaipur Measure of Association- Concepts If more disease occurs in a group that smokes compared to the
Causal Association : Cause To Effect Dr. MD, DHA, PGDHRM Prof. Community Medicine & Director-SIHFW, Jaipur Measure of Association- Concepts If more disease occurs in a group that smokes compared to the
Protocol Development: The Guiding Light of Any Clinical Study
 Protocol Development: The Guiding Light of Any Clinical Study Susan G. Fisher, Ph.D. Chair, Department of Clinical Sciences 1 Introduction Importance/ relevance/ gaps in knowledge Specific purpose of the
Protocol Development: The Guiding Light of Any Clinical Study Susan G. Fisher, Ph.D. Chair, Department of Clinical Sciences 1 Introduction Importance/ relevance/ gaps in knowledge Specific purpose of the
Clinical Epidemiology I: Deciding on Appropriate Therapy
 Clinical Epidemiology I: Deciding on Appropriate Therapy 1 Clinical Scenario [UG2B] 65 yo man controlled HTN 6-mo Hx cardioversion-resistant afib Benefit vs risk of long-term anticoagulation:? prevent
Clinical Epidemiology I: Deciding on Appropriate Therapy 1 Clinical Scenario [UG2B] 65 yo man controlled HTN 6-mo Hx cardioversion-resistant afib Benefit vs risk of long-term anticoagulation:? prevent
Measures of Association
 Measures of Association Lakkana Thaikruea M.D., M.S., Ph.D. Community Medicine Department, Faculty of Medicine, Chiang Mai University, Thailand Introduction One of epidemiological studies goal is to determine
Measures of Association Lakkana Thaikruea M.D., M.S., Ph.D. Community Medicine Department, Faculty of Medicine, Chiang Mai University, Thailand Introduction One of epidemiological studies goal is to determine
Overview of Study Designs in Clinical Research
 Overview of Study Designs in Clinical Research Systematic Reviews (SR), Meta-Analysis Best Evidence / Evidence Guidelines + Evidence Summaries Randomized, controlled trials (RCT) Clinical trials, Cohort
Overview of Study Designs in Clinical Research Systematic Reviews (SR), Meta-Analysis Best Evidence / Evidence Guidelines + Evidence Summaries Randomized, controlled trials (RCT) Clinical trials, Cohort
MCQ Course in Pediatrics Al Yamamah Hospital June Dr M A Maleque Molla, FRCP, FRCPCH
 MCQ Course in Pediatrics Al Yamamah Hospital 10-11 June Dr M A Maleque Molla, FRCP, FRCPCH Q1. Following statements are true in the steps of evidence based medicine except ; a) Convert the need for information
MCQ Course in Pediatrics Al Yamamah Hospital 10-11 June Dr M A Maleque Molla, FRCP, FRCPCH Q1. Following statements are true in the steps of evidence based medicine except ; a) Convert the need for information
Cigarette Smoking and Lung Cancer
 Centers for Disease Control and Prevention Epidemiology Program Office Case Studies in Applied Epidemiology No. 731-703 Cigarette Smoking and Lung Cancer Learning Objectives After completing this case
Centers for Disease Control and Prevention Epidemiology Program Office Case Studies in Applied Epidemiology No. 731-703 Cigarette Smoking and Lung Cancer Learning Objectives After completing this case
In the 1700s patients in charity hospitals sometimes slept two or more to a bed, regardless of diagnosis.
 Control Case In the 1700s patients in charity hospitals sometimes slept two or more to a bed, regardless of diagnosis. This depicts a patient who finds himself lying with a corpse (definitely a case ).
Control Case In the 1700s patients in charity hospitals sometimes slept two or more to a bed, regardless of diagnosis. This depicts a patient who finds himself lying with a corpse (definitely a case ).
12 CANCER Epidemiology Methodological considerations
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 12 CANCER 12.1 Epidemiology 12.1.1 Methodological
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 12 CANCER 12.1 Epidemiology 12.1.1 Methodological
Beyond Controlling for Confounding: Design Strategies to Avoid Selection Bias and Improve Efficiency in Observational Studies
 September 27, 2018 Beyond Controlling for Confounding: Design Strategies to Avoid Selection Bias and Improve Efficiency in Observational Studies A Case Study of Screening Colonoscopy Our Team Xabier Garcia
September 27, 2018 Beyond Controlling for Confounding: Design Strategies to Avoid Selection Bias and Improve Efficiency in Observational Studies A Case Study of Screening Colonoscopy Our Team Xabier Garcia
Study Design. Svetlana Yampolskaya, Ph.D. Summer 2013
 Study Design Svetlana Yampolskaya, Ph.D. Summer 2013 Study Design Single point in time Cross-Sectional Research Multiple points in time Study only exists in this point in time Longitudinal Research Study
Study Design Svetlana Yampolskaya, Ph.D. Summer 2013 Study Design Single point in time Cross-Sectional Research Multiple points in time Study only exists in this point in time Longitudinal Research Study
GATE: Graphic Appraisal Tool for Epidemiology picture, 2 formulas & 3 acronyms
 GATE: Graphic Appraisal Tool for Epidemiology 1991-2015 1 picture, 2 formulas & 3 acronyms 1 GATE: Graphic Appraisal Tool for Epidemiology Graphic Architectural Tool for Epidemiology Graphic Approach To
GATE: Graphic Appraisal Tool for Epidemiology 1991-2015 1 picture, 2 formulas & 3 acronyms 1 GATE: Graphic Appraisal Tool for Epidemiology Graphic Architectural Tool for Epidemiology Graphic Approach To
Confounding Bias: Stratification
 OUTLINE: Confounding- cont. Generalizability Reproducibility Effect modification Confounding Bias: Stratification Example 1: Association between place of residence & Chronic bronchitis Residence Chronic
OUTLINE: Confounding- cont. Generalizability Reproducibility Effect modification Confounding Bias: Stratification Example 1: Association between place of residence & Chronic bronchitis Residence Chronic
Critical Appraisal Series
 Definition for therapeutic study Terms Definitions Study design section Observational descriptive studies Observational analytical studies Experimental studies Pragmatic trial Cluster trial Researcher
Definition for therapeutic study Terms Definitions Study design section Observational descriptive studies Observational analytical studies Experimental studies Pragmatic trial Cluster trial Researcher
Lecture Slides. Elementary Statistics Eleventh Edition. by Mario F. Triola. and the Triola Statistics Series 1.1-1
 Lecture Slides Elementary Statistics Eleventh Edition and the Triola Statistics Series by Mario F. Triola 1.1-1 Chapter 1 Introduction to Statistics 1-1 Review and Preview 1-2 Statistical Thinking 1-3
Lecture Slides Elementary Statistics Eleventh Edition and the Triola Statistics Series by Mario F. Triola 1.1-1 Chapter 1 Introduction to Statistics 1-1 Review and Preview 1-2 Statistical Thinking 1-3
Journal Club Overview
 Journal Club Overview What is a Journal Club? Overview of the critical appraisal for primary research study designs Or, what are we doing now? What is a Journal Club? An educational meeting in which a
Journal Club Overview What is a Journal Club? Overview of the critical appraisal for primary research study designs Or, what are we doing now? What is a Journal Club? An educational meeting in which a
Causation Assessment and Methodology
 Causation Assessment and Methodology Kurt T. Hegmann, MD, MPH University of Utah Content Attestation I, Kurt Hegmann, MD hereby declare that the content for this activity, including any presentation of
Causation Assessment and Methodology Kurt T. Hegmann, MD, MPH University of Utah Content Attestation I, Kurt Hegmann, MD hereby declare that the content for this activity, including any presentation of
observational studies Descriptive studies
 form one stage within this broader sequence, which begins with laboratory studies using animal models, thence to human testing: Phase I: The new drug or treatment is tested in a small group of people for
form one stage within this broader sequence, which begins with laboratory studies using animal models, thence to human testing: Phase I: The new drug or treatment is tested in a small group of people for
Teaching critical appraisal of randomised controlled trials
 Teaching critical appraisal of randomised controlled trials Dr Kamal R. Mahtani BSc PhD MBBS PGDip MRCGP Deputy Director Centre for Evidence Based Medicine University of Oxford November 2014 1 objectives
Teaching critical appraisal of randomised controlled trials Dr Kamal R. Mahtani BSc PhD MBBS PGDip MRCGP Deputy Director Centre for Evidence Based Medicine University of Oxford November 2014 1 objectives
Interpretation of Epidemiologic Studies
 Interpretation of Epidemiologic Studies Paolo Boffetta Mount Sinai School of Medicine, New York, USA International Prevention Research Institute, Lyon, France Outline Introduction to epidemiology Issues
Interpretation of Epidemiologic Studies Paolo Boffetta Mount Sinai School of Medicine, New York, USA International Prevention Research Institute, Lyon, France Outline Introduction to epidemiology Issues
Fixed Effect Combining
 Meta-Analysis Workshop (part 2) Michael LaValley December 12 th 2014 Villanova University Fixed Effect Combining Each study i provides an effect size estimate d i of the population value For the inverse
Meta-Analysis Workshop (part 2) Michael LaValley December 12 th 2014 Villanova University Fixed Effect Combining Each study i provides an effect size estimate d i of the population value For the inverse
Is There An Association?
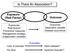 Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
