Study Designs in Epidemiology
|
|
|
- Claud Griffin
- 6 years ago
- Views:
Transcription
1 Study Designs in Epidemiology Dr. Sireen Alkhaldi, BDS, MPH, DrPH First semester 2017/ 2018 Department of Family and Community Medicine School of Medicine/ The University of Jordan
2 Epidemiologic Study Design Study design is the arrangement of conditions for the collection and analysis of data to provide the most accurate answer to a question in the most economical way.
3 Types of Epidemiologic Study Designs I. Based on objective/focus/research question: 1. Descriptive studies Describe: who, when, where & how many 2. Analytic studies Analyze: How and why
4 Types of Epidemiological Study Designs II. Based on the role of the investigator 1. Observational studies The investigator observes nature No intervention 2. Intervention/Experimental studies Investigator intervenes He has a control over the situation
5 Types of Epidemiological Study Designs III. Based on timing : 1. One-time (one-spot) studies Conducted at a point in time An individual is observed at once 2. Longitudinal (Follow-up) studies Conducted in a period of time Individuals are followed over a period of time
6 Types of Epidemiological Study Designs IV. Based on direction of follow-up/data collection: 1. Prospective Conducted forward in time 2. Retrospective Conducted backward in time
7 Timeframe of Studies Prospective Study looks forward, looks to the future, examines future events, follows a condition, concern or disease into the future Study begins here time
8 Timeframe of Studies Retrospective Study to look back, looks back in time to study events that have already occurred Study begins here time
9 Types of Epidemiological Study Designs V. Based on type of data they generate 1. Qualitative studies Generate contextual data Also called exploratory studies 2. Quantitative studies Generate numerical data Also called explanatory studies
10 Types of Epidemiological Study Designs VII. Standard classification 1. Cross-sectional studies 2. Case-control studies 3. Cohort studies 4. Experimental studies
11 Types of Epidemiological Study Designs Descriptive studies describe occurrence of outcome Analytic studies describe association between exposure and outcome
12 Scientific Research
13 Basic Research Study Designs in Epidemiology Study design is the arrangement of conditions for the collection and analysis of data to provide the most accurate answer to a question in the most economical way.
14 Descriptive Analytic Case report RCT Cohort study Case series Ecologic study Experimental Case-Control study Cross-sectional study Observational Epidemiologic Study Designs
15 Descriptive Studies (generating hypothesis) Descriptive studies are usually the first phase of an epidemiological investigation. These studies are concerned with observing the distribution of disease or health related characteristics in human populations. Such studies basically ask the questions of what, who, where, and when.
16 Research Hypothesis A hypothesis is a supposition, arrived at from observation or reflection. It can be accepted or rejected using the techniques of analytical epidemiology. A hypothesis should specify the following: 1. The population. 2. The specific cause being considered. 3. Expected outcome disease. 4. Time response relationship (expectation). 5. Be understandable, measurable and testable.
17
18 CHARACTERISTICS EXAMINED IN DESCRIPTIVE STUDIES TIME PLACE PERSON Year, Season Climatic zones Age, Birth order Month, Week Day, Hour of onset Duration Country, region Urban/ rural / Local community Towns, Cities, Institutions Sex, Family size Marital State, Height, Weight Occupation, Social status, Education, Blood pressure, Blood cholesterol, Personal habits
19 Case Reports Detailed presentation of a single case or handful of cases Generally report a new or unique finding e.g. previous undescribed disease e.g. unexpected link between diseases e.g. unexpected new therapeutic effect e.g. adverse events
20 Case Series Experience of a group of patients with a similar diagnosis Cases may be identified from a single or multiple sources Generally report on new/unique condition May be the only realistic design for rare disorders
21 Case Series Advantages Useful for hypothesis generation Informative for very rare diseases with few established risk factors Disadvantages Cannot study cause and effect relationships Cannot assess disease frequency
22 Ecological Studies (correlation study) The ecologic study is a hypothesis generating study. Usually using group-level data, it examines if two factors are correlated with each other. It involves the collection of events over a defined population base and by the use of denominator data to determine rates. It results in Ecological Fallacy: Failure in reasoning that arises when an inference is made about an individual based on aggregate data for a group.
23 Basic Question in Analytic Epidemiology Are exposure and disease linked? Exposure Disease
24 Basic Questions in Analytic Epidemiology To prevent and control diseases What is the exposure? Who are the exposed? What are the potential health effects? Generate a hypothesis about the relationship between exposure and effect, and then test this hypothesis. Study designs.. direct how this whole investigation is conducted.
25 Analytical Studies (testing hypothesis) Observational Studies Cross-sectional Case-control Cohort Experimental Studies Randomized controlled clinical trials Community trials
26 Observational Studies non-experimental study designs Observational because there is no individual intervention Treatment and exposures occur in a non-controlled environment Individuals can be observed prospectively, retrospectively, or currently
27
28 Cross-sectional studies An observational design that surveys exposures and disease status at a single point in time (a cross-section of the population) time Study only exists at this point in time
29 Cross-sectional Design Study population No Disease Disease factor present factor absent factor present factor absent time Study only exists at this point in time
30 Cross-sectional Studies Are the simplest form of observational studies. Often used to study conditions that are relatively frequent with long duration of expression (nonfatal, chronic conditions) It measures prevalence, not incidence of disease Example: community surveys Not suitable for studying rare or highly fatal diseases or a disease with short duration of expression.
31 Cross-sectional Advantages of cross-sectional studies Less time consuming Less expensive Provides more information Describes the population well Generates hypothesis Cross-sectional study provides a snap-shot or a photograph of a population at a certain point in time. 31
32 Cross-sectional studies Disadvantages Weakest observational design, (it measures prevalence, not incidence of disease). Prevalent cases are survivors The temporal sequence of exposure and effect may be difficult or impossible to determine. Usually don t know when disease occurred Rare events a problem. Quickly emerging diseases a problem. Least useful in establishing causation.
33 Case-Control Studies An observational design comparing exposures in disease cases vs. healthy controls from the same population. exposure data collected retrospectively. most feasible design where disease outcomes are rare. This is the first approach to test causal hypothesis. Definition of a case is crucial to a case control study.
34 SELECTION OF CONTROLS The controls must be free from the disease under study. They must be similar to the cases as possible, except for the absence of the disease under study. Selection of an appropriate control group is an important pre requisite, because we will be making comparison with controls..
35 Case- Control Design factor present factor absent factor present factor absent past Cases (disease) Controls (no disease) present Study population time Study begins here
36 Case-Control Study Strengths: 1) Less expensive and less time consuming 2) Efficient for studying rare diseases 3) Allows the study of several different aetiological factors for one disease. 4) No attrition problems. 5) Ethical problems minimal (no risk to participants)
37 Case-Control Study Limitations 1. Inappropriate when disease outcome for a specific exposure is not known at start of study. 2. Selection of an appropriate control group may be difficult. 3. Inefficient for evaluation of rare exposure 4. Difficult to establish temporal sequence 5. Determining exposure will often rely on memory, leading to bias. 6. We cannot measure incidence,& can only estimate the relative risk.
38 Cohort Study Subjects are selected by exposure and followed to see development of disease Cohort study is known by a variety of names: prospective study, longitudinal study, incidence study & forward looking study.
39 Cohort Study Is an observational design comparing individuals with a known risk factor or exposure with others without the risk factor or exposure. Looking for a difference in the risk (incidence) of a disease over time. Best observational design. Data usually collected prospectively (some retrospective).
40 Cohort Study Indications: When there is a good evidence of an association between exposure & disease. When exposure is rare, but incidence is high among the exposed. When attrition of the study population can be minimized (due to long follow-up period). When ample funds are available.
41 Cohort Design Study population free of disease present Factor present Factor absent disease no disease disease no disease future time Study begins here
42 Prospective Cohort study Exposed Outcome Measure exposure Baseline Study begins here Non-exposed time Outcome
43 Advantages of cohort studies 1. Valuable when exposure is rare 2. Examines multiple outcomes of a single exposures 3. Temporal relationship is known 4. Allow direct measurement of risk 5. Minimize bias in ascertainment of exposure Exposure status determined before disease detection (avoid information bias). Subjects selected before disease detection (avoid selection bias).
44 Limitations of Cohort Study 1. Expensive 2. Time-consuming 3. Inefficient for rare diseases or diseases with long latency 4. Loss to follow-up is a problem
45 Experimental Studies In an experiment, we are interested in the consequences of some treatment on some outcome. Types of experimental studies: 1. Clinical trial: on patients in clinical settings. 2. Field trial: on healthy people in the field. 3. Community trial: on the community as a whole.
46 Experimental Design (Intervention studies) Individuals are allocated in to treatment and control groups by the investigator. If properly done, experimental studies can produce high quality data. Thy are the gold standard study design.
47 Experimental Studies The quality of Gold standard in experimental studies can be achieved through: Randomization Blinding Placebo
48 Randomization: random allocation of study subjects in to treatment & control groups. Avoids bias & confounding, and increases confidence in the results. Blinding: Denying information on treatment / control status (single, double or triple blinding). This helps to avoid observation bias. Placebo: an inert material indistinguishable from active treatment. Used to avoid Placebo effect: tendency to report favourable response regardless of physiological efficacy. (Placebo is used as blinding procedure )
49 RCT (Randomized Controlled Trial) An experimental design with subjects randomly assigned to treatment and comparison groups. Clinical trials are the most well known experimental design. The ultimate design in testing causal hypotheses.
50 RANDOMIZATION Study population Intervention Control outcome no outcome outcome no outcome baseline future time Study begins here (baseline point)
51 Randomized Controlled Trials (RCTs) Randomized Controlled Trials (RCTs) The gold standard of research designs Provides most convincing evidence of relationship between exposure and effect Trials of hormone replacement therapy in menopausal women found no protection for heart disease, contradicting findings of prior observational studies.
52 Randomized Controlled Trials Disadvantages of RCTs: Very expensive Not appropriate to answer certain types of questions: It may be unethical, for example, to assign persons to certain treatment or comparison groups.
Research Methodology Workshop. Study Type
 Research Methodology Workshop Study Type What type of question? Frequency: how common is it? Aetiology: What caused this? Intervention: Does this intervention work? Diagnosis or test evaluation: How accurate
Research Methodology Workshop Study Type What type of question? Frequency: how common is it? Aetiology: What caused this? Intervention: Does this intervention work? Diagnosis or test evaluation: How accurate
Handout 1: Introduction to the Research Process and Study Design STAT 335 Fall 2016
 DESIGNING OBSERVATIONAL STUDIES As we have discussed, for the purpose of establishing cause-and-effect relationships, observational studies have a distinct disadvantage in comparison to randomized comparative
DESIGNING OBSERVATIONAL STUDIES As we have discussed, for the purpose of establishing cause-and-effect relationships, observational studies have a distinct disadvantage in comparison to randomized comparative
Design strategies in quantitative research an introduction
 Design strategies in quantitative research an introduction Key messages Research design can be broadly divided in to descriptive and analytical. Descriptive studies look at the individual, location and
Design strategies in quantitative research an introduction Key messages Research design can be broadly divided in to descriptive and analytical. Descriptive studies look at the individual, location and
CONCEPTUALIZING A RESEARCH DESIGN
 CONCEPTUALIZING A RESEARCH DESIGN Detty Nurdiati Dept of Obstetrics & Gynecology Fac of Medicine, Universitas Gadjah Mada Yogyakarta, Indonesia Conceptualizing a Research Design The Research Design The
CONCEPTUALIZING A RESEARCH DESIGN Detty Nurdiati Dept of Obstetrics & Gynecology Fac of Medicine, Universitas Gadjah Mada Yogyakarta, Indonesia Conceptualizing a Research Design The Research Design The
Overview of Study Designs
 Overview of Study Designs Kyoungmi Kim, Ph.D. July 13 & 20, 2016 This seminar is jointly supported by the following NIH-funded centers: We are video recording this seminar so please hold questions until
Overview of Study Designs Kyoungmi Kim, Ph.D. July 13 & 20, 2016 This seminar is jointly supported by the following NIH-funded centers: We are video recording this seminar so please hold questions until
Epidemiological study design. Paul Pharoah Department of Public Health and Primary Care
 Epidemiological study design Paul Pharoah Department of Public Health and Primary Care Molecules What/why? Organelles Cells Tissues Organs Clinical medicine Individuals Public health medicine Populations
Epidemiological study design Paul Pharoah Department of Public Health and Primary Care Molecules What/why? Organelles Cells Tissues Organs Clinical medicine Individuals Public health medicine Populations
Epidemiologic Methods and Counting Infections: The Basics of Surveillance
 Epidemiologic Methods and Counting Infections: The Basics of Surveillance Ebbing Lautenbach, MD, MPH, MSCE University of Pennsylvania School of Medicine Nothing to disclose PENN Outline Definitions / Historical
Epidemiologic Methods and Counting Infections: The Basics of Surveillance Ebbing Lautenbach, MD, MPH, MSCE University of Pennsylvania School of Medicine Nothing to disclose PENN Outline Definitions / Historical
Overview of Study Designs in Clinical Research
 Overview of Study Designs in Clinical Research Systematic Reviews (SR), Meta-Analysis Best Evidence / Evidence Guidelines + Evidence Summaries Randomized, controlled trials (RCT) Clinical trials, Cohort
Overview of Study Designs in Clinical Research Systematic Reviews (SR), Meta-Analysis Best Evidence / Evidence Guidelines + Evidence Summaries Randomized, controlled trials (RCT) Clinical trials, Cohort
Trial Designs. Professor Peter Cameron
 Trial Designs Professor Peter Cameron OVERVIEW Review of Observational methods Principles of experimental design applied to observational studies Population Selection Looking for bias Inference Analysis
Trial Designs Professor Peter Cameron OVERVIEW Review of Observational methods Principles of experimental design applied to observational studies Population Selection Looking for bias Inference Analysis
Penelitian IKM/Epidemiologi. Contact: Blog: suyatno.blog.undip.ac.id Hp/Telp: /
 Penelitian IKM/Epidemiologi Oleh : Suyatno, Ir. MKes Contact: E-mail: suyatnofkmundip@gmail.com Blog: suyatno.blog.undip.ac.id Hp/Telp: 08122815730 / 024-70251915 Epidemiologic Study Designs I. Experimental
Penelitian IKM/Epidemiologi Oleh : Suyatno, Ir. MKes Contact: E-mail: suyatnofkmundip@gmail.com Blog: suyatno.blog.undip.ac.id Hp/Telp: 08122815730 / 024-70251915 Epidemiologic Study Designs I. Experimental
Types of Biomedical Research
 INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency Family Medicine Section of
INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency Family Medicine Section of
12/26/2013. Types of Biomedical Research. Clinical Research. 7Steps to do research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH S T A T I S T I C
 Types of Biomedical Research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency
Types of Biomedical Research INTRODUCTION & MEASUREMENT IN CLINICAL RESEARCH Sakda Arj Ong Vallipakorn, MD MSIT, MA (Information Science) Pediatrics, Pediatric Cardiology Emergency Medicine, Ped Emergency
Answer keys for Assignment 4: Measures of disease frequency
 Answer keys for Assignment 4: Measures of disease frequency (The correct answer is underlined in bold text) 1. This statistic is used to estimate the risk of acquiring a disease. It may be measured as
Answer keys for Assignment 4: Measures of disease frequency (The correct answer is underlined in bold text) 1. This statistic is used to estimate the risk of acquiring a disease. It may be measured as
INTRODUCTION TO EPIDEMIOLOGICAL STUDY DESIGNS PHUNLERD PIYARAJ, MD., MHS., PHD.
 INTRODUCTION TO EPIDEMIOLOGICAL STUDY DESIGNS PHUNLERD PIYARAJ, MD., MHS., PHD. 1 OBJECTIVES By the end of this section, you will be able to: Provide a definition of epidemiology Describe the major types
INTRODUCTION TO EPIDEMIOLOGICAL STUDY DESIGNS PHUNLERD PIYARAJ, MD., MHS., PHD. 1 OBJECTIVES By the end of this section, you will be able to: Provide a definition of epidemiology Describe the major types
Critical Appraisal of Research Protocol
 Critical Appraisal of Research Protocol Moazzam Ali MD, PhD, MPH Department of Reproductive Health and Research World Health Organization Geneva, Switzerland GFMER November 27, 2017 1 Today's presentation
Critical Appraisal of Research Protocol Moazzam Ali MD, PhD, MPH Department of Reproductive Health and Research World Health Organization Geneva, Switzerland GFMER November 27, 2017 1 Today's presentation
Study design. Chapter 64. Research Methodology S.B. MARTINS, A. ZIN, W. ZIN
 Chapter 64 Study design S.B. MARTINS, A. ZIN, W. ZIN Research Methodology Scientific methodology comprises a set of rules and procedures to investigate the phenomena of interest. These rules and procedures
Chapter 64 Study design S.B. MARTINS, A. ZIN, W. ZIN Research Methodology Scientific methodology comprises a set of rules and procedures to investigate the phenomena of interest. These rules and procedures
Clinical problems and choice of study designs
 Evidence Based Dentistry Clinical problems and choice of study designs Asbjørn Jokstad University of Oslo, Norway Nov 21 2001 1 Manipulation with intervention Yes Experimental study No Non-experimental
Evidence Based Dentistry Clinical problems and choice of study designs Asbjørn Jokstad University of Oslo, Norway Nov 21 2001 1 Manipulation with intervention Yes Experimental study No Non-experimental
Asian Journal of Research in Biological and Pharmaceutical Sciences Journal home page:
 Review Article ISSN: 2349 4492 Asian Journal of Research in Biological and Pharmaceutical Sciences Journal home page: www.ajrbps.com REVIEW ON EPIDEMIOLOGICAL STUDY DESIGNS V.J. Divya *1, A. Vikneswari
Review Article ISSN: 2349 4492 Asian Journal of Research in Biological and Pharmaceutical Sciences Journal home page: www.ajrbps.com REVIEW ON EPIDEMIOLOGICAL STUDY DESIGNS V.J. Divya *1, A. Vikneswari
Study Designs in Epidemiology
 Study Designs in Epidemiology Headlines Epidemiological research Classification of designs Qualitative methods Quantitative methods Choice of design Epidemiological Research Lab research: applies knowledge
Study Designs in Epidemiology Headlines Epidemiological research Classification of designs Qualitative methods Quantitative methods Choice of design Epidemiological Research Lab research: applies knowledge
Study Design. Svetlana Yampolskaya, Ph.D. Summer 2013
 Study Design Svetlana Yampolskaya, Ph.D. Summer 2013 Study Design Single point in time Cross-Sectional Research Multiple points in time Study only exists in this point in time Longitudinal Research Study
Study Design Svetlana Yampolskaya, Ph.D. Summer 2013 Study Design Single point in time Cross-Sectional Research Multiple points in time Study only exists in this point in time Longitudinal Research Study
Gathering. Useful Data. Chapter 3. Copyright 2004 Brooks/Cole, a division of Thomson Learning, Inc.
 Gathering Chapter 3 Useful Data Copyright 2004 Brooks/Cole, a division of Thomson Learning, Inc. Principal Idea: The knowledge of how the data were generated is one of the key ingredients for translating
Gathering Chapter 3 Useful Data Copyright 2004 Brooks/Cole, a division of Thomson Learning, Inc. Principal Idea: The knowledge of how the data were generated is one of the key ingredients for translating
Choosing the right study design
 Choosing the right study design Main types of study design Randomised controlled trial (RCT) Cohort study Case-control study Cross-sectional study Case series/case note review Expert opinion BEST QUALITY
Choosing the right study design Main types of study design Randomised controlled trial (RCT) Cohort study Case-control study Cross-sectional study Case series/case note review Expert opinion BEST QUALITY
Chapter 02. Basic Research Methodology
 Chapter 02 Basic Research Methodology Definition RESEARCH Research is a quest for knowledge through diligent search or investigation or experimentation aimed at the discovery and interpretation of new
Chapter 02 Basic Research Methodology Definition RESEARCH Research is a quest for knowledge through diligent search or investigation or experimentation aimed at the discovery and interpretation of new
observational studies Descriptive studies
 form one stage within this broader sequence, which begins with laboratory studies using animal models, thence to human testing: Phase I: The new drug or treatment is tested in a small group of people for
form one stage within this broader sequence, which begins with laboratory studies using animal models, thence to human testing: Phase I: The new drug or treatment is tested in a small group of people for
STUDY DESIGN. Jerrilyn A. Cambron, DC, PhD Department of Research. RE6002: Week 2. National University of Health Sciences
 STUDY DESIGN Jerrilyn A. Cambron, DC, PhD Department of Research RE6002: Week 2 Let s look at the big picture Research Descriptive Analytical Qualitative study: Interview Cross sectional: Case study Observational
STUDY DESIGN Jerrilyn A. Cambron, DC, PhD Department of Research RE6002: Week 2 Let s look at the big picture Research Descriptive Analytical Qualitative study: Interview Cross sectional: Case study Observational
Lecture 4: Research Approaches
 Lecture 4: Research Approaches Lecture Objectives Theories in research Research design approaches ú Experimental vs. non-experimental ú Cross-sectional and longitudinal ú Descriptive approaches How to
Lecture 4: Research Approaches Lecture Objectives Theories in research Research design approaches ú Experimental vs. non-experimental ú Cross-sectional and longitudinal ú Descriptive approaches How to
Epidemiologic Study Designs. (RCTs)
 Epidemiologic Study Designs Epidemiologic Study Designs Experimental (RCTs) Observational Analytical Descriptive Case-Control Cohort + cross-sectional & ecologic Epidemiologic Study Designs Descriptive
Epidemiologic Study Designs Epidemiologic Study Designs Experimental (RCTs) Observational Analytical Descriptive Case-Control Cohort + cross-sectional & ecologic Epidemiologic Study Designs Descriptive
Module 5. The Epidemiological Basis of Randomised Controlled Trials. Landon Myer School of Public Health & Family Medicine, University of Cape Town
 Module 5 The Epidemiological Basis of Randomised Controlled Trials Landon Myer School of Public Health & Family Medicine, University of Cape Town Introduction The Randomised Controlled Trial (RCT) is the
Module 5 The Epidemiological Basis of Randomised Controlled Trials Landon Myer School of Public Health & Family Medicine, University of Cape Town Introduction The Randomised Controlled Trial (RCT) is the
Purpose. Study Designs. Objectives. Observational Studies. Analytic Studies
 Purpose Study Designs H.S. Teitelbaum, DO, PhD, MPH, FAOCOPM AOCOPM Annual Meeting Introduce notions of study design Clarify common terminology used with description and interpretation of information collected
Purpose Study Designs H.S. Teitelbaum, DO, PhD, MPH, FAOCOPM AOCOPM Annual Meeting Introduce notions of study design Clarify common terminology used with description and interpretation of information collected
STUDY DESIGNS WHICH ONE IS BEST?
 STUDY DESIGNS WHICH ONE IS BEST? David Nunan, PhD Research Fellow and Tutor Nuffield Department of Primary Care Health Sciences and Oxford Centre for Evidence Based Medicine University of Oxford Exercise
STUDY DESIGNS WHICH ONE IS BEST? David Nunan, PhD Research Fellow and Tutor Nuffield Department of Primary Care Health Sciences and Oxford Centre for Evidence Based Medicine University of Oxford Exercise
Basic Epidemiology: Study Types. Learning Objectives. The Five Ws of Epidemiology. At the end of this presentation, participants will be able to:
 Basic Epidemiology: Study Types John Kobayashi, MD, MPH August 12, 2009 Learning Objectives At the end of this presentation, participants will be able to: Define some key concepts in epidemiology Describe
Basic Epidemiology: Study Types John Kobayashi, MD, MPH August 12, 2009 Learning Objectives At the end of this presentation, participants will be able to: Define some key concepts in epidemiology Describe
UNDERSTANDING EPIDEMIOLOGICAL STUDIES. Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA
 UNDERSTANDING EPIDEMIOLOGICAL STUDIES Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA Study design in epidemiological research: Summary Observational
UNDERSTANDING EPIDEMIOLOGICAL STUDIES Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA Study design in epidemiological research: Summary Observational
TEST YOURSELF: Multiple Choice 1 QUESTIONS
 1. In the investigation of an epidemic the most appropriate measure to describe the frequency of occurrence of illness is the: A. Prevalence B. Incidence rate C. Case-fatality rate D. Attack rate E. Mortality
1. In the investigation of an epidemic the most appropriate measure to describe the frequency of occurrence of illness is the: A. Prevalence B. Incidence rate C. Case-fatality rate D. Attack rate E. Mortality
Epidemiology: Overview of Key Concepts and Study Design. Polly Marchbanks
 Epidemiology: Overview of Key Concepts and Study Design Polly Marchbanks Lecture Outline (1) Key epidemiologic concepts - Definition - What epi is not - What epi is - Process of epi research Lecture Outline
Epidemiology: Overview of Key Concepts and Study Design Polly Marchbanks Lecture Outline (1) Key epidemiologic concepts - Definition - What epi is not - What epi is - Process of epi research Lecture Outline
Issues to Consider in the Design of Randomized Controlled Trials
 Issues to Consider in the Design of Randomized Controlled Trials Jay Wilkinson, MD, MPH Professor of Pediatrics & Epidemiology Miller School of Medicine Seminar Purpose To facilitate an interactive discussion
Issues to Consider in the Design of Randomized Controlled Trials Jay Wilkinson, MD, MPH Professor of Pediatrics & Epidemiology Miller School of Medicine Seminar Purpose To facilitate an interactive discussion
Appraising the Literature Overview of Study Designs
 Chapter 5 Appraising the Literature Overview of Study Designs Barbara M. Sullivan, PhD Department of Research, NUHS Jerrilyn A. Cambron, PhD, DC Department of Researach, NUHS EBP@NUHS Ch 5 - Overview of
Chapter 5 Appraising the Literature Overview of Study Designs Barbara M. Sullivan, PhD Department of Research, NUHS Jerrilyn A. Cambron, PhD, DC Department of Researach, NUHS EBP@NUHS Ch 5 - Overview of
Clinical Research Scientific Writing. K. A. Koram NMIMR
 Clinical Research Scientific Writing K. A. Koram NMIMR Clinical Research Branch of medical science that determines the safety and effectiveness of medications, devices, diagnostic products and treatment
Clinical Research Scientific Writing K. A. Koram NMIMR Clinical Research Branch of medical science that determines the safety and effectiveness of medications, devices, diagnostic products and treatment
Epidemiology overview
 Epidemiology overview Riris Andono Ahmad 1 Public Health Approach Surveillance: What is the problem? Problem Risk Factor Identification: What is the cause? Intervention Evaluation: What works? Implementation:
Epidemiology overview Riris Andono Ahmad 1 Public Health Approach Surveillance: What is the problem? Problem Risk Factor Identification: What is the cause? Intervention Evaluation: What works? Implementation:
Protocol Development: The Guiding Light of Any Clinical Study
 Protocol Development: The Guiding Light of Any Clinical Study Susan G. Fisher, Ph.D. Chair, Department of Clinical Sciences 1 Introduction Importance/ relevance/ gaps in knowledge Specific purpose of the
Protocol Development: The Guiding Light of Any Clinical Study Susan G. Fisher, Ph.D. Chair, Department of Clinical Sciences 1 Introduction Importance/ relevance/ gaps in knowledge Specific purpose of the
Lecture 5 Conducting Interviews and Focus Groups
 Lecture 5 Conducting Interviews and Focus Groups Talking to participants enables in-depth information about the experiences of health and illness; and of factors that influence health and illness behaviour
Lecture 5 Conducting Interviews and Focus Groups Talking to participants enables in-depth information about the experiences of health and illness; and of factors that influence health and illness behaviour
Study Designs. chapter. W hy. L earning O bjectives
 holbox/shutterstock holbox/shutterstock chapter 2 Study Designs W hen and W hy Key Questions How do you know when the results of a study are credible? What makes a good study? How do you decide which kind
holbox/shutterstock holbox/shutterstock chapter 2 Study Designs W hen and W hy Key Questions How do you know when the results of a study are credible? What makes a good study? How do you decide which kind
Research in Real-World Settings: PCORI s Model for Comparative Clinical Effectiveness Research
 Research in Real-World Settings: PCORI s Model for Comparative Clinical Effectiveness Research David Hickam, MD, MPH Director, Clinical Effectiveness & Decision Science Program April 10, 2017 Welcome!
Research in Real-World Settings: PCORI s Model for Comparative Clinical Effectiveness Research David Hickam, MD, MPH Director, Clinical Effectiveness & Decision Science Program April 10, 2017 Welcome!
Department of OUTCOMES RESEARCH
 Department of OUTCOMES RESEARCH Clinical Research Design Sources of Error Types of Clinical Research Randomized Trials! Daniel I. Sessler, M.D. Professor and Chair Department of OUTCOMES RESEARCH The Cleveland
Department of OUTCOMES RESEARCH Clinical Research Design Sources of Error Types of Clinical Research Randomized Trials! Daniel I. Sessler, M.D. Professor and Chair Department of OUTCOMES RESEARCH The Cleveland
RANDOMIZED CLINICAL TRIALS. Study Designs. Randomized Clinical Trials (RCT) ASSIGN EXPOSURE Follow up Check for OUTCOME. Experimental studies
 RANDOMIZED CLINICAL TRIALS Study Designs Observational studies Analytic studies Descriptive studies Randomized Controlled trials Case control Cohort Cross sectional Randomized Clinical Trials (RCT) Assign
RANDOMIZED CLINICAL TRIALS Study Designs Observational studies Analytic studies Descriptive studies Randomized Controlled trials Case control Cohort Cross sectional Randomized Clinical Trials (RCT) Assign
3. Factors such as race, age, sex, and a person s physiological state are all considered determinants of disease. a. True
 / False 1. Epidemiology is the basic science of public health. LEARNING OBJECTIVES: CNIA.BOYL.17.2.1 - Define epidemiology. 2. Within the field of epidemiology, the term distribution refers to the relationship
/ False 1. Epidemiology is the basic science of public health. LEARNING OBJECTIVES: CNIA.BOYL.17.2.1 - Define epidemiology. 2. Within the field of epidemiology, the term distribution refers to the relationship
GLOSSARY OF GENERAL TERMS
 GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials
 In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials are comparative, large scale studies that typically
In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials are comparative, large scale studies that typically
Anatomy of Medical Studies: Part I. Emily D Agostino Epidemiology and Biostatistics CUNY School of Public Health at Hunter College
 Anatomy of Medical Studies: Part I!! Emily D Agostino Epidemiology and Biostatistics CUNY School of Public Health at Hunter College I. Standard format of a Medical Study Abstract (quick overview: what
Anatomy of Medical Studies: Part I!! Emily D Agostino Epidemiology and Biostatistics CUNY School of Public Health at Hunter College I. Standard format of a Medical Study Abstract (quick overview: what
Intervention vs. Observational Trials:
 Intervention vs. Observational Trials: A Brief Introduction Introduction Just like an architect translates a vision for a building into blueprints, so an investigator translates their research idea into
Intervention vs. Observational Trials: A Brief Introduction Introduction Just like an architect translates a vision for a building into blueprints, so an investigator translates their research idea into
M6728. Goals. Depression Scores. Research Designs
 M6728 Research Designs Goals Match appropriate research designs to the study purpose and questions Differentiate between experimental and non-experimental studies Discuss epidemiologic designs Evaluate
M6728 Research Designs Goals Match appropriate research designs to the study purpose and questions Differentiate between experimental and non-experimental studies Discuss epidemiologic designs Evaluate
Study Designs CHAPTER 2. Jones & Bartlett Learning, LLC NOT FOR SALE OR DISTRIBUTION. Jones & Bartlett Learning, LL NOT FOR SALE OR DISTRIBUT
 CHAPTER 2 Study Designs servational studies, we generally observe a phenomenon, whereas LEARNING OBJECTIVES in randomized studies, we intervene and measure a response. By the end of this chapter, Jones
CHAPTER 2 Study Designs servational studies, we generally observe a phenomenon, whereas LEARNING OBJECTIVES in randomized studies, we intervene and measure a response. By the end of this chapter, Jones
Cohort studies. Training Course in Sexual and Reproductive Health Research Geneva Nguyen Thi My Huong, MD PhD WHO/RHR/SIS
 Cohort studies Training Course in Sexual and Reproductive Health Research Geneva 2012 Nguyen Thi My Huong, MD PhD WHO/RHR/SIS OUTLINE Overview Definitions Study design Basic measures Advantages and disadvantages
Cohort studies Training Course in Sexual and Reproductive Health Research Geneva 2012 Nguyen Thi My Huong, MD PhD WHO/RHR/SIS OUTLINE Overview Definitions Study design Basic measures Advantages and disadvantages
Evidence-Based Medicine Journal Club. A Primer in Statistics, Study Design, and Epidemiology. August, 2013
 Evidence-Based Medicine Journal Club A Primer in Statistics, Study Design, and Epidemiology August, 2013 Rationale for EBM Conscientious, explicit, and judicious use Beyond clinical experience and physiologic
Evidence-Based Medicine Journal Club A Primer in Statistics, Study Design, and Epidemiology August, 2013 Rationale for EBM Conscientious, explicit, and judicious use Beyond clinical experience and physiologic
HARM. Definition modified from the IHI definition of Harm by the QUEST Harm Workgroup
 Indonesia Clinical epidemiology and Evidence Based Medicine (ICE(ICE-EBM) HARM Jarir At Thobari (FK UGM) Efficacious Medical intervention Harm HARM Unintended physical injury resulting from or contributed
Indonesia Clinical epidemiology and Evidence Based Medicine (ICE(ICE-EBM) HARM Jarir At Thobari (FK UGM) Efficacious Medical intervention Harm HARM Unintended physical injury resulting from or contributed
The comparison or control group may be allocated a placebo intervention, an alternative real intervention or no intervention at all.
 1. RANDOMISED CONTROLLED TRIALS (Treatment studies) (Relevant JAMA User s Guides, Numbers IIA & B: references (3,4) Introduction: The most valid study design for assessing the effectiveness (both the benefits
1. RANDOMISED CONTROLLED TRIALS (Treatment studies) (Relevant JAMA User s Guides, Numbers IIA & B: references (3,4) Introduction: The most valid study design for assessing the effectiveness (both the benefits
4/10/2018. Choosing a study design to answer a specific research question. Importance of study design. Types of study design. Types of study design
 Choosing a study design to answer a specific research question Importance of study design Will determine how you collect, analyse and interpret your data Helps you decide what resources you need Impact
Choosing a study design to answer a specific research question Importance of study design Will determine how you collect, analyse and interpret your data Helps you decide what resources you need Impact
Chapter 13 Summary Experiments and Observational Studies
 Chapter 13 Summary Experiments and Observational Studies What have we learned? We can recognize sample surveys, observational studies, and randomized comparative experiments. o These methods collect data
Chapter 13 Summary Experiments and Observational Studies What have we learned? We can recognize sample surveys, observational studies, and randomized comparative experiments. o These methods collect data
Some interpretational issues connected with observational studies
 Some interpretational issues connected with observational studies D.R. Cox Nuffield College, Oxford, UK and Nanny Wermuth Chalmers/Gothenburg University, Gothenburg, Sweden ABSTRACT After some general
Some interpretational issues connected with observational studies D.R. Cox Nuffield College, Oxford, UK and Nanny Wermuth Chalmers/Gothenburg University, Gothenburg, Sweden ABSTRACT After some general
Clinical Research Design and Conduction
 Clinical Research Design and Conduction Study Design M Mohsen Ibrahim, MD Cardiac Department Cairo University Algorithm for classification of types of clinical research Grimes DA and Schulz KF. Lancet
Clinical Research Design and Conduction Study Design M Mohsen Ibrahim, MD Cardiac Department Cairo University Algorithm for classification of types of clinical research Grimes DA and Schulz KF. Lancet
Systematic Reviews and Meta- Analysis in Kidney Transplantation
 Systematic Reviews and Meta- Analysis in Kidney Transplantation Greg Knoll MD MSc Associate Professor of Medicine Medical Director, Kidney Transplantation University of Ottawa and The Ottawa Hospital KRESCENT
Systematic Reviews and Meta- Analysis in Kidney Transplantation Greg Knoll MD MSc Associate Professor of Medicine Medical Director, Kidney Transplantation University of Ottawa and The Ottawa Hospital KRESCENT
Mixed Methods Study Design
 1 Mixed Methods Study Design Kurt C. Stange, MD, PhD Professor of Family Medicine, Epidemiology & Biostatistics, Oncology and Sociology Case Western Reserve University 1. Approaches 1, 2 a. Qualitative
1 Mixed Methods Study Design Kurt C. Stange, MD, PhD Professor of Family Medicine, Epidemiology & Biostatistics, Oncology and Sociology Case Western Reserve University 1. Approaches 1, 2 a. Qualitative
Chapter 13. Experiments and Observational Studies. Copyright 2012, 2008, 2005 Pearson Education, Inc.
 Chapter 13 Experiments and Observational Studies Copyright 2012, 2008, 2005 Pearson Education, Inc. Observational Studies In an observational study, researchers don t assign choices; they simply observe
Chapter 13 Experiments and Observational Studies Copyright 2012, 2008, 2005 Pearson Education, Inc. Observational Studies In an observational study, researchers don t assign choices; they simply observe
CRITICAL APPRAISAL OF MEDICAL LITERATURE. Samuel Iff ISPM Bern
 CRITICAL APPRAISAL OF MEDICAL LITERATURE Samuel Iff ISPM Bern siff@ispm.unibe.ch Contents Study designs Asking good questions Pitfalls in clinical studies How to assess validity (RCT) Conclusion Step-by-step
CRITICAL APPRAISAL OF MEDICAL LITERATURE Samuel Iff ISPM Bern siff@ispm.unibe.ch Contents Study designs Asking good questions Pitfalls in clinical studies How to assess validity (RCT) Conclusion Step-by-step
Design of Experiments & Introduction to Research
 Design of Experiments & Introduction to Research 1 Design of Experiments Introduction to Research Definition and Purpose Scientific Method Research Project Paradigm Structure of a Research Project Types
Design of Experiments & Introduction to Research 1 Design of Experiments Introduction to Research Definition and Purpose Scientific Method Research Project Paradigm Structure of a Research Project Types
Is There An Association?
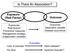 Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
The following are questions that students had difficulty with on the first three exams.
 The following are questions that students had difficulty with on the first three exams. Exam 1 1. A measure has construct validity if it: a) really measures what it is supposed to measure b) appears, on
The following are questions that students had difficulty with on the first three exams. Exam 1 1. A measure has construct validity if it: a) really measures what it is supposed to measure b) appears, on
Study Design STUDY DESIGN CASE SERIES AND CROSS-SECTIONAL STUDY DESIGN
 STUDY DESIGN CASE SERIES AND CROSS-SECTIONAL Daniel E. Ford, MD, MPH Vice Dean for Clinical Investigation Johns Hopkins School of Medicine Introduction to Clinical Research July 15, 2014 STUDY DESIGN Provides
STUDY DESIGN CASE SERIES AND CROSS-SECTIONAL Daniel E. Ford, MD, MPH Vice Dean for Clinical Investigation Johns Hopkins School of Medicine Introduction to Clinical Research July 15, 2014 STUDY DESIGN Provides
Process of Designing & Implementing a Research Project
 Research Question, Hypothesis, Variables Dr. R.M. Pandey Prof & Head Department of Biostatistics A.I.I.M.S., New Delhi rmpandey@yahoo.com Process of Designing & Implementing a Research Project 2 HYPOTHESIS
Research Question, Hypothesis, Variables Dr. R.M. Pandey Prof & Head Department of Biostatistics A.I.I.M.S., New Delhi rmpandey@yahoo.com Process of Designing & Implementing a Research Project 2 HYPOTHESIS
Gender Identity and Expression. Week 8
 Gender Identity and Expression Week 8 1 Objectives 1. Describe research methods for studying gender identity and expression as they relate to health 2. Identify some key health disparities in relation
Gender Identity and Expression Week 8 1 Objectives 1. Describe research methods for studying gender identity and expression as they relate to health 2. Identify some key health disparities in relation
Study Designs. Randomized Clinical Trials (RCT) RCT: Example 1. RCT: Two by Two Table. Outcome. Exposure. Yes a b No c d
 Study Designs Randomized Clinical Trials (RCT) Assign Exposure Randomly Experimental studies Analytic studies Observational studies Descriptive studies POPULATION Population People without the outcome
Study Designs Randomized Clinical Trials (RCT) Assign Exposure Randomly Experimental studies Analytic studies Observational studies Descriptive studies POPULATION Population People without the outcome
Chapter 11: Experiments and Observational Studies p 318
 Chapter 11: Experiments and Observational Studies p 318 Observation vs Experiment An observational study observes individuals and measures variables of interest but does not attempt to influence the response.
Chapter 11: Experiments and Observational Studies p 318 Observation vs Experiment An observational study observes individuals and measures variables of interest but does not attempt to influence the response.
Review. Chapter 5. Common Language. Ch 3: samples. Ch 4: real world sample surveys. Experiments, Good and Bad
 Review Ch 3: samples Sampling terminology Proportions Margin of error Ch 4: real world sample surveys Questions to ask about a study Errors in sample surveys Concerns about survey questions Probability
Review Ch 3: samples Sampling terminology Proportions Margin of error Ch 4: real world sample surveys Questions to ask about a study Errors in sample surveys Concerns about survey questions Probability
Critical Appraisal Series
 Definition for therapeutic study Terms Definitions Study design section Observational descriptive studies Observational analytical studies Experimental studies Pragmatic trial Cluster trial Researcher
Definition for therapeutic study Terms Definitions Study design section Observational descriptive studies Observational analytical studies Experimental studies Pragmatic trial Cluster trial Researcher
I. Identifying the question Define Research Hypothesis and Questions
 Term Paper I. Identifying the question What is the question? (What are my hypotheses?) Is it possible to answer the question with statistics? Is the data obtainable? (birth weight, socio economic, drugs,
Term Paper I. Identifying the question What is the question? (What are my hypotheses?) Is it possible to answer the question with statistics? Is the data obtainable? (birth weight, socio economic, drugs,
Cohort studies. Training course in research methodology and research protocol development Geneva Nguyen Thi My Huong, MD PhD WHO/RHR/SIS
 Cohort studies Training course in research methodology and research protocol development Geneva 2018 Nguyen Thi My Huong, MD PhD WHO/RHR/SIS OUTLINE Overview Definitions Study design Basic measures Advantages
Cohort studies Training course in research methodology and research protocol development Geneva 2018 Nguyen Thi My Huong, MD PhD WHO/RHR/SIS OUTLINE Overview Definitions Study design Basic measures Advantages
Common Statistical Issues in Biomedical Research
 Common Statistical Issues in Biomedical Research Howard Cabral, Ph.D., M.P.H. Boston University CTSI Boston University School of Public Health Department of Biostatistics May 15, 2013 1 Overview of Basic
Common Statistical Issues in Biomedical Research Howard Cabral, Ph.D., M.P.H. Boston University CTSI Boston University School of Public Health Department of Biostatistics May 15, 2013 1 Overview of Basic
Randomized Controlled Trials Shortcomings & Alternatives icbt Leiden Twitter: CAAL 1
 Randomized Controlled Trials Shortcomings & Alternatives icbt Leiden 2017 Casper Albers University of Groningen www.casperalbers.nl c.j.albers@rug.nl Twitter: CAAL 1 Overview 1. Short introduction 2. What
Randomized Controlled Trials Shortcomings & Alternatives icbt Leiden 2017 Casper Albers University of Groningen www.casperalbers.nl c.j.albers@rug.nl Twitter: CAAL 1 Overview 1. Short introduction 2. What
Research Methods: Operational Studies in Tuberculosis. Introduction to Epidemiology. Epidemiology. Epidemiology. The Public Health Cycle
 Research Methods: Operational Studies in Tuberculosis Introduction to Epidemiology Michael Lauzardo, MD Principal Investigator, Southeastern National Tuberculosis Center Assistant Professor, Division of
Research Methods: Operational Studies in Tuberculosis Introduction to Epidemiology Michael Lauzardo, MD Principal Investigator, Southeastern National Tuberculosis Center Assistant Professor, Division of
CHECK-LISTS AND Tools DR F. R E Z A E I DR E. G H A D E R I K U R D I S TA N U N I V E R S I T Y O F M E D I C A L S C I E N C E S
 CHECK-LISTS AND Tools DR F. R E Z A E I DR E. G H A D E R I K U R D I S TA N U N I V E R S I T Y O F M E D I C A L S C I E N C E S What is critical appraisal? Critical appraisal is the assessment of evidence
CHECK-LISTS AND Tools DR F. R E Z A E I DR E. G H A D E R I K U R D I S TA N U N I V E R S I T Y O F M E D I C A L S C I E N C E S What is critical appraisal? Critical appraisal is the assessment of evidence
Causation Assessment and Methodology
 Causation Assessment and Methodology Kurt T. Hegmann, MD, MPH University of Utah Content Attestation I, Kurt Hegmann, MD hereby declare that the content for this activity, including any presentation of
Causation Assessment and Methodology Kurt T. Hegmann, MD, MPH University of Utah Content Attestation I, Kurt Hegmann, MD hereby declare that the content for this activity, including any presentation of
Observational the researcher collects information on attributes or measurements of interest but DOES NOT influence the results.
 Distinction between Observational and Experimental Studies Observational the researcher collects information on attributes or measurements of interest but DOES NOT influence the results. Experimental the
Distinction between Observational and Experimental Studies Observational the researcher collects information on attributes or measurements of interest but DOES NOT influence the results. Experimental the
Beyond the intention-to treat effect: Per-protocol effects in randomized trials
 Beyond the intention-to treat effect: Per-protocol effects in randomized trials Miguel Hernán DEPARTMENTS OF EPIDEMIOLOGY AND BIOSTATISTICS Intention-to-treat analysis (estimator) estimates intention-to-treat
Beyond the intention-to treat effect: Per-protocol effects in randomized trials Miguel Hernán DEPARTMENTS OF EPIDEMIOLOGY AND BIOSTATISTICS Intention-to-treat analysis (estimator) estimates intention-to-treat
UNIT I SAMPLING AND EXPERIMENTATION: PLANNING AND CONDUCTING A STUDY (Chapter 4)
 UNIT I SAMPLING AND EXPERIMENTATION: PLANNING AND CONDUCTING A STUDY (Chapter 4) A DATA COLLECTION (Overview) When researchers want to make conclusions/inferences about an entire population, they often
UNIT I SAMPLING AND EXPERIMENTATION: PLANNING AND CONDUCTING A STUDY (Chapter 4) A DATA COLLECTION (Overview) When researchers want to make conclusions/inferences about an entire population, they often
EBM, Study Design and Numbers. David Frankfurter, MD Professor OB/GYN The George Washington University
 EBM, Study Design and Numbers David Frankfurter, MD Professor OB/GYN The George Washington University 1978? 1978 Best Picture of 1978 The Deer Hunter Universal Studios The 50th Academy Awards were held
EBM, Study Design and Numbers David Frankfurter, MD Professor OB/GYN The George Washington University 1978? 1978 Best Picture of 1978 The Deer Hunter Universal Studios The 50th Academy Awards were held
CRITICAL EVALUATION OF BIOMEDICAL LITERATURE
 Chapter 9 CRITICAL EVALUATION OF BIOMEDICAL LITERATURE M.G.Rajanandh, Department of Pharmacy Practice, SRM College of Pharmacy, SRM University. INTRODUCTION Reviewing the Biomedical Literature poses a
Chapter 9 CRITICAL EVALUATION OF BIOMEDICAL LITERATURE M.G.Rajanandh, Department of Pharmacy Practice, SRM College of Pharmacy, SRM University. INTRODUCTION Reviewing the Biomedical Literature poses a
Goal: To become familiar with the methods that researchers use to investigate aspects of causation and methods of treatment
 Key Dates TU Mar 28 Unit 18 Loss of control drinking in alcoholics (on course website); Marlatt assignment TH Mar 30 Unit 19; Term Paper Step 2 TU Apr 4 Begin Biological Perspectives, Unit IIIA and 20;
Key Dates TU Mar 28 Unit 18 Loss of control drinking in alcoholics (on course website); Marlatt assignment TH Mar 30 Unit 19; Term Paper Step 2 TU Apr 4 Begin Biological Perspectives, Unit IIIA and 20;
Goal: To become familiar with the methods that researchers use to investigate aspects of causation and methods of treatment
 Goal: To become familiar with the methods that researchers use to investigate aspects of causation and methods of treatment Scientific Study of Causation and Treatment Methods for studying causation Case
Goal: To become familiar with the methods that researchers use to investigate aspects of causation and methods of treatment Scientific Study of Causation and Treatment Methods for studying causation Case
Module 4 Introduction
 Module 4 Introduction Recall the Big Picture: We begin a statistical investigation with a research question. The investigation proceeds with the following steps: Produce Data: Determine what to measure,
Module 4 Introduction Recall the Big Picture: We begin a statistical investigation with a research question. The investigation proceeds with the following steps: Produce Data: Determine what to measure,
AS 3.12, Externally assessed, 4 credits Experiments and Observational Studies
 AS 3.12, Externally assessed, 4 credits Experiments and Observational Studies Do drugs have an adverse effect on teenage brains? Does listening to music help students study? What information would you
AS 3.12, Externally assessed, 4 credits Experiments and Observational Studies Do drugs have an adverse effect on teenage brains? Does listening to music help students study? What information would you
Focused Research Questions and Study Designs
 Focused Research Questions and Study Designs PAROS Literature Review Workshop 11-12 April 2011, Khoo Teck Puat Hospital, Singapore Dianne Bautista, PhD Singapore Clinical Research Institute 11-12 April
Focused Research Questions and Study Designs PAROS Literature Review Workshop 11-12 April 2011, Khoo Teck Puat Hospital, Singapore Dianne Bautista, PhD Singapore Clinical Research Institute 11-12 April
1. Draft checklist for judging on quality of animal studies (Van der Worp et al., 2010)
 Appendix C Quality appraisal tools (QATs) 1. Draft checklist for judging on quality of animal studies (Van der Worp et al., 2010) 2. NICE Checklist for qualitative studies (2012) 3. The Critical Appraisal
Appendix C Quality appraisal tools (QATs) 1. Draft checklist for judging on quality of animal studies (Van der Worp et al., 2010) 2. NICE Checklist for qualitative studies (2012) 3. The Critical Appraisal
Observational Study Designs. Review. Today. Measures of disease occurrence. Cohort Studies
 Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
The Research Enterprise in Psychology Chapter 2
 The Research Enterprise in Psychology Chapter 2 This multimedia product and its contents are protected under copyright law. The following are prohibited by law: any public performance or display, including
The Research Enterprise in Psychology Chapter 2 This multimedia product and its contents are protected under copyright law. The following are prohibited by law: any public performance or display, including
Epidemiologic Methods I & II Epidem 201AB Winter & Spring 2002
 DETAILED COURSE OUTLINE Epidemiologic Methods I & II Epidem 201AB Winter & Spring 2002 Hal Morgenstern, Ph.D. Department of Epidemiology UCLA School of Public Health Page 1 I. THE NATURE OF EPIDEMIOLOGIC
DETAILED COURSE OUTLINE Epidemiologic Methods I & II Epidem 201AB Winter & Spring 2002 Hal Morgenstern, Ph.D. Department of Epidemiology UCLA School of Public Health Page 1 I. THE NATURE OF EPIDEMIOLOGIC
A to Z OF RESEARCH METHODS AND TERMS APPLICABLE WITHIN SOCIAL SCIENCE RESEARCH
 A to Z OF RESEARCH METHODS AND TERMS APPLICABLE WITHIN SOCIAL SCIENCE RESEARCH QUANTATIVE RESEARCH METHODS Are concerned with trying to quantify things; they ask questions such as how long, how many or
A to Z OF RESEARCH METHODS AND TERMS APPLICABLE WITHIN SOCIAL SCIENCE RESEARCH QUANTATIVE RESEARCH METHODS Are concerned with trying to quantify things; they ask questions such as how long, how many or
Designed Experiments have developed their own terminology. The individuals in an experiment are often called subjects.
 When we wish to show a causal relationship between our explanatory variable and the response variable, a well designed experiment provides the best option. Here, we will discuss a few basic concepts and
When we wish to show a causal relationship between our explanatory variable and the response variable, a well designed experiment provides the best option. Here, we will discuss a few basic concepts and
Rapid appraisal of the literature: Identifying study biases
 Rapid appraisal of the literature: Identifying study biases Rita Popat, PhD Clinical Assistant Professor Division of Epidemiology Stanford University School of Medicine August 7, 2007 What is critical
Rapid appraisal of the literature: Identifying study biases Rita Popat, PhD Clinical Assistant Professor Division of Epidemiology Stanford University School of Medicine August 7, 2007 What is critical
Checklist for appraisal of study relevance (child sex offenses)
 Appendix 3 Evaluation protocols. [posted as supplied by author] Checklist for appraisal of study relevance (child sex offenses) First author, year, reference number Relevance Yes No Cannot answer Not applicable
Appendix 3 Evaluation protocols. [posted as supplied by author] Checklist for appraisal of study relevance (child sex offenses) First author, year, reference number Relevance Yes No Cannot answer Not applicable
Quantitative Methods. Lonnie Berger. Research Training Policy Practice
 Quantitative Methods Lonnie Berger Research Training Policy Practice Defining Quantitative and Qualitative Research Quantitative methods: systematic empirical investigation of observable phenomena via
Quantitative Methods Lonnie Berger Research Training Policy Practice Defining Quantitative and Qualitative Research Quantitative methods: systematic empirical investigation of observable phenomena via
2017 DISEASE DETECTIVES (B,C) KAREN LANCOUR National Bio Rules Committee Chairman
 2017 DISEASE DETECTIVES (B,C) KAREN LANCOUR National Bio Rules Committee Chairman Event Rules 2017 DISCLAIMER This presentation was prepared using draft rules. There may be some changes in the final copy
2017 DISEASE DETECTIVES (B,C) KAREN LANCOUR National Bio Rules Committee Chairman Event Rules 2017 DISCLAIMER This presentation was prepared using draft rules. There may be some changes in the final copy
