PUBLICATIONS SYSTEM CHANGE TRANSMITTAL FOR TRICARE POLICY MANUAL (TPM), AUGUST 2002
|
|
|
- Hollie Andrews
- 5 years ago
- Views:
Transcription
1 OFFICE OF THE ASSISTANT SECRETARY OF DEFENSE HEALTH AFFAIRS EAST CENTRETECH PARKWAY AURORA, COLORADO TAICARE MANAGEMENT ACTIVITY MB&RB CHANGE M MAY 13,2010 PUBLICATIONS SYSTEM CHANGE TRANSMITTAL FOR TRICARE POLICY MANUAL (TPM), AUGUST 2002 The TRICARE Management Activity has authorized the following addition(s)/revision(s). CHANGE TITLE: CODING AND CLARIFICATION UPDATES DECEMBER 2009 CONREQ: PAGE CHANGE(S): See page 2. SUMMARY OF CHANGE(S): See page 3 EFFECTIVE AND IMPLEMENTATION DATE: Upon direction of the Contracting Officer. This change is made in conjunction with Aug 2002 TOM, Change No. 97, Aug 2002 TRM, Change No. 115, and Aug 2002 TSM, Change No. 80. Reimbursement Branch ATTACHMENT(S): DISTRIBUTION: 15 PAGE(S) M WHEN PRESCRIBED ACTION HAS BEEN TAKEN, FILE THIS TRANSMITTAL WITH BASIC DOCUMENT.
2 CHANGE M MAY 13, 2010 REMOVE PAGE(S) INSERT PAGE(S) CHAPTER 4 Section 20.1, pages 1 through 4 Section 20.1, pages 1 through 4 CHAPTER 6 Table of Contents, page i Table of Contents, page i Section 3.1, pages 1 and 2 Section 3.1, pages 1 and 2 CHAPTER 7 Section 16.2, page 1 Section 16.2, page 1 CHAPTER 8 Section 9.1, pages 3 through 5 Section 9.1, pages 3 through 5 CHAPTER 10 Section 5.1, pages 3 and 4 Section 5.1, pages 3 and 4 Section 6.1, page 1 Section 6.1, pages 1 and 2 2
3 CHANGE M MAY 13, 2010 SUMMARY OF CHANGES CHAPTER 4 1. Section Added CPT code range Paragraph II.S. deleted CPT code. Paragraph II.U. added exclusion: Spinal cord and deep brain neurostimulation in the treatment of chronic intractable headache or migraine pain is unproven. CHAPTER 6 2. Table of Contents. Deleted And Counseling from Subject of Section Section 3.1. Deleted And Counseling from Subject of section. Deleted CPT Procedure Code paragraph (original paragraph I.) and renumbered subsequent paragraphs. Added clarification that genetic counseling must be provided by a TRICARE authorized provider and be submitted with appropriate Evaluation and Management (E&M) codes. Paragraph III.B. Added an exclusion for medical genetics and genetic counseling services, each 30 minutes face-to-face with patient/family (CPT code 96040). CHAPTER 7 4. Section Paragraph III. removed by a clinical psychologist. Deleted Exclusion B. Patient meeting criteria for a psychiatric diagnosis. CHAPTER 8 5. Section 9.1. Clarified that legend vitamins may be cost-shared only when used as a specific treatment of a medical condition. Prenatal vitamins requiring a prescription in the United States may be cost-shared for prenatal care only. CHAPTER Section 5.1. Clarified eligibility for TAMP eligibles who were also eligible for Operation Noble Eagle/Operation Enduring Freedom Demonstration benefits. 7. Section 6.1. Clarified eligibility for TRICARE for Life. 3
4
5 SURGERY CHAPTER 4 SECTION 20.1 NERVOUS SYSTEM ISSUE DATE: August 26, 1985 AUTHORITY: 32 CFR 199.4(c)(2) and (c)(3) I. CPT 1 PROCEDURE CODES , , , , , , , , 64730, 64732, , 95961, 95962, , 95978, II. DESCRIPTION A. The nervous system consists of the central and peripheral nervous systems. The central is comprised of the brain and spinal cord and the peripheral includes all the other neural elements. The nervous system is the organ system which along with the endocrine system, correlates the adjustments and reactions of an organism to internal and environmental conditions. B. Therapeutic embolization is a type of procedure that is commonly performed by interventional radiologist to occlude blood vessels. A microcatheter or balloon is threaded into a vein, or artery for the purposes of embolization, blocking a pathologic vascular channel. C. Stereotactic implantation of depth electrodes is an invasive procedure in which needle-like electrodes are implanted through burr holes in the skull into the depths of specific brain areas to localize a seizure focus in patients who are candidates for surgery or to implant a brain stimulator in the thalamus to control tremors. D. Psychosurgery is brain surgery directed at destroying normal and healthy brain tissue in order to relieve mental and psychic symptoms that other treatment modalities such as drug therapy and psychotherapy have been ineffectual in treating, for the purpose of changing or controlling behavior. E. The Guglielmi Detachable Coil (GDC) is an extremely fine wire made from platinum, one of the softest metals, at the end of a longer stainless steel wire. In a controlled manner, the surgeon uses a micro-catheter to thread each coil through blood vessels to the aneurysm site. Application of a very-low-voltage electric current detaches and releases the coil into the aneurysm. Once in place, the GDC coils fill the aneurysm, isolating it from circulation to 1 CPT only 2006 American Medical Association (or such other date of publication of CPT). All Rights Reserved. 1
6 CHAPTER 4, SECTION 20.1 NERVOUS SYSTEM reduce the likelihood or rupture and hemorrhagic stroke. By applying a low voltage direct current to a stainless steel wire at the base of the coil, the platinum coil is detached. This applied current not only detaches the coil but also promotes electrothrombosis within the aneurysm. III. POLICY A. Services and supplies required in the diagnosis and treatment of illness or injury involving the nervous system are covered. B. Therapeutic embolization (CPT 2 procedure code 61624) may be covered for the following indications. The list of indications is not all inclusive. Other indications are covered when documented by reliable evidence as safe, effective and comparable or superior to standard care (proven). 1. Cerebral Arteriovenous Malformations (AVMs). 2. Vein of Galen Aneurysm. 3. Inoperable or High-Risk Intracranial Aneurysms. 4. Dural Arteriovenous Fistulas. 5. Meningioma. C. Implantation of depth electrodes is covered. Implantation of a U.S. Food and Drug Administration (FDA) approved vagus nerve stimulator as adjunctive therapy in reducing the frequency of seizures in adults and adolescents over 12 years of age, which are refractory to anti-epileptic medication is covered. Battery replacement is also covered. D. Spinal cord and deep brain stimulation are covered in the treatment of chronic intractable pain. Coverage includes: 1. The accessories necessary for the effective functioning of the covered device. 2. Repair, adjustment, replacement and removal of the covered device and associated surgical costs. E. The GDC may be cost-shared for embolizing unruptured intracranial aneurysms that, because of their morphology, their location, or the patient s general medical condition, are considered by the treating neurosurgical team to be: 1. Very high risk for management by traditional operative techniques; or 2. Inoperable; or 2 CPT only 2006 American Medical Association (or such other date of publication of CPT). All Rights Reserved. 2
7 CHAPTER 4, SECTION 20.1 NERVOUS SYSTEM 3. For embolizing other vascular malformation such as AVMs and arteriovenous fistulae of the neurovasculature, to include arterial and venous embolizations in the peripheral vasculature. IV. EXCLUSIONS A. N-butyl-2-cyanoacrylate (Histacryl Bleu ), iodinated poppy seed oils (e.g., Ethiodol ), and absorbable gelatin sponges are not FDA approved. B. Transcutaneous, percutaneous, functional dorsal column electrical stimulation in the treatment of multiple sclerosis or other motor function disorders is unproven. C. Deep brain neurostimulation in the treatment of insomnia, depression, anxiety, and substance abuse is unproven. D. Psychosurgery is not in accordance with accepted professional medical standards and is not covered. E. Endovascular GDC treatment of wide-necked aneurysms and rupture is unproven. F. Cerebellar stimulators/pacemakers for the treatment of neurological disorders are unproven. G. Dorsal Root Entry Zone (DREZ) thermocoagulation or microcoagulation neurosurgical procedure is unproven. H. Epidural steroid injections for thoracic pain are unproven. I. Extraoperative electrocortiography for stimulation and recording in order to determine electrical thresholds of neurons as an indicator of seizure focus is unproven. J. Neuromuscular Electrical Stimulation (NMES) for the treatment of denervated muscles is unproven. K. Stereotactic cingulotomy is unproven. L. Sacral nerve neurostimulator (CPT 3 procedure codes 64561, 64581, 64585, and 64590). See Chapter 4, Section 14.1 for coverage policy for the urinary system and the Sacral Nerve Root Stimulation (SNS). M. Laminoplasty, cervical with decompression of the spinal cord, two or more vertebral segments with reconstruction of the posterior bony elements (CPT 3 procedure codes and 63051). N. Balloon angioplasty, intracranial, percutaneous (CPT 3 procedure code 61630) is unproven. Effective January 1, CPT only 2006 American Medical Association (or such other date of publication of CPT). All Rights Reserved. 3 C-106, September 25, 2009
8 CHAPTER 4, SECTION 20.1 NERVOUS SYSTEM O. Transcatheter placement of intravascular stent(s) intracranial, (e.g., atherosclerotic stenosis) including angioplasty, if performed (CPT 4 procedure code 61635) is unproven. Effective January 1, P. Balloon dilation o f intracranial vasospasm, initial vessel (CPT 4 procedure code 61640) each additional vessel in same family (CPT 4 procedure code 61641) or different vascular family (CPT 4 procedure code 61642) is unproven. Effective January 1, Q. Sphenopalatine ganglion block (CPT 4 procedure code 64505) for the treatment of chronic migraine headaches and neck pain is unproven. R. Radiofrequency ablation (percutaneous radiofrequency facet denervation, percutaneous facet coagulation, percutaneous radiofrequency neurotomy, radiofrequency facet rhizotomy, radiofrequency articular rhizolysis) (CPT 4 procedure codes 64622, 64623, 64626, 64627) for the treatment of chronic spinal pain is unproven. Pulsed radiofrequency ablation for spinal pain is unproven. S. Implantation of Occipital Nerve Stimulator for the treatment of chronic intractable migraine headache is unproven. T. Cryoablation of Occipital Nerve (CPT 4 procedure code 64640) for the treatment of chronic intractable headache is unproven. U. Spinal cord and deep brain neurostimulation in the treatment of chronic intractable headache or migraine pain is unproven. V. EFFECTIVE DATES A. January 1, 1989, for PAVM. B. April 1, 1994, for therapeutic embolization for treatment of meningioma. C. July 14, 1997, for GDC. D. The date of FDA approval of the embolization device for all other embolization procedures. E. June 1, 2004, for Magnetoencephalography. - END - 4 CPT only 2006 American Medical Association (or such other date of publication of CPT). All Rights Reserved. 4
9 CHAPTER 6 PATHOLOGY AND LABORATORY SECTION SUBJECT 1.1 General 2.1 Transfusion Services For Whole Blood, Blood Components And Blood Derivatives 3.1 Diagnostic Genetic Testing i
10
11 PATHOLOGY AND LABORATORY CHAPTER 6 SECTION 3.1 DIAGNOSTIC GENETIC TESTING ISSUE DATE: March 10, 2000 AUTHORITY: 32 CFR 199.4(a)(1)(i) I. DESCRIPTION Genetic testing intended to be confirmatory of a clinical diagnosis which is already suspected based on the patient s symptoms. II. POLICY A. Diagnostic genetic testing when medically proven and appropriate and when the results of the test will influence the medical management of the individual is a TRICARE benefit. B. The following diagnostic tests are covered. This is not an all inclusive list, but provides examples of covered diagnostic tests. 1. Chromosome analysis (to include karyotyping and/or high resolution chromosome analysis) in some cases of habitual abortion or infertility. 2. Testing for Marfan Syndrome and chromosome analysis (to include karyotyping and/or high resolution chromosome analysis) of children. Common indications for chromosome analysis in children to include ambiguity of external genitalia, small-forgestational age infants, multiple anomalies and failure to thrive. 3. Other medically necessary genetic diagnostic tests. C. Services should be billed using the appropriate Evaluation and Management (E&M) codes. III. EXCLUSIONS A. Routine genetic testing that does not influence the beneficiary s medical management. 1
12 CHAPTER 6, SECTION 3.1 DIAGNOSTIC GENETIC TESTING B. CPT 1 procedure code medical genetics and genetic counseling services, each 30 minutes face-to-face with patient/family. - END - 1 CPT only 2006 American Medical Association (or such other date of publication of CPT). All Rights Reserved. 2
13 MEDICINE CHAPTER 7 SECTION 16.2 HEALTH AND BEHAVIOR ASSESSMENT/INTERVENTION ISSUE DATE: March 11, 2002 AUTHORITY: 32 CFR 199.4(a)(1) I. CPT 1 PROCEDURE CODE RANGE II. DESCRIPTION Health and behavior assessment procedures are used to identify the psychological, behavioral, emotional, cognitive, and social factors important to the prevention, treatment, or management of physical health problems. The focus of the assessment is not on mental health but on the biopsychosocial factors important to physical health problems and treatment. III. POLICY Health and behavior assessment performed in conjunction with the medical or surgical treatment of a covered illness or injury is covered. IV. EXCLUSION Family evaluation and assessment (without the patient present) (CPT 1 procedure code 96155). - END - 1 CPT only 2006 American Medical Association (or such other date of publication of CPT). All Rights Reserved. 1
14
15 CHAPTER 8, SECTION 9.1 PHARMACY BENEFITS PROGRAM a. The drug is approved for marketing by the U.S. Food and Drug Administration; b. The drug is prescribed by a provider, acting within the scope of his/her license, for its labeled indication; and c. The drug is furnished by a provider in accordance with all applicable state laws and licensing requirements. 2. Off-label use. Drugs may be cost-shared for off-label uses when determined, by the contractor with responsibility for the venue distributing the drugs, that reliable evidence demonstrates such usage is safe and effective. As presented in order of relative weight in 32 CFR 199.2, reliable evidence means: a. Well controlled studies of clinical meaningful endpoints, published in refereed medical literature. b. Published formal technology assessments. c. Published reports of national professional medical associations. d. Published national medical policy organizations. e. Published reports of national expert opinion organizations. 3. Drugs grandfathered by the Federal Food, Drug and Cosmetic Act of 1938 may be cost-shared as if FDA approved. 4. Insulin and related supplies may be cost-shared for diabetic patients, regardless of whether or not a prescription is required under state law. 5. Orphan Drugs. Pharmaceutical agents with FDA orphan drug designation and marketing approval may be cost-shared when used in the treatment of a rare disease or condition. For the purpose of the pharmacy benefits program, TRICARE adopts the FDA definition of the term rare disease or condition. 6. Legend vitamins may be cost-shared only when used as a specific treatment of a medical condition. In addition, prenatal vitamins that require a prescription in the United States may be cost-shared. Prenatal vitamins requiring a prescription are covered for prenatal care only. 7. Some drugs require prior authorization. For these drugs, prior authorization request forms and criteria, in addition to other formulary information, are available at: or D. Eligibility 1. Uniformed Service members who are on active duty; 3
16 CHAPTER 8, SECTION 9.1 PHARMACY BENEFITS PROGRAM 2. All beneficiaries authorized TRICARE benefits per 32 CFR 199.3; 3. Medicare eligible beneficiaries: a. Pursuant to Section 711 of the FY 2001 National Defense Authorization Act, Medicare Eligible beneficiaries based on age, whose TRICARE eligibility is determined by 10 U.S.C. Section 1086, are eligible for Medicare Part A and, except as provided in paragraph III.B. below, are enrolled in Medicare Part B, are eligible for the TRICARE pharmacy benefits program, effective April 1, b. Individuals, who before April 1, 2001, have attained the age of 65 and who are not enrolled in Medicare Part B are eligible for the TRICARE Senior Pharmacy Program; and 4. Overseas TRICARE beneficiaries listed in DEERS (with APO or FPO address) are eligible for the TRICARE Mail Order Program. For these beneficiaries a prescription is required for a U.S. Food and Drug Administration approved prescription drug from an authorized provider who is licensed to practice in the United States. E. Reimbursement 1. The prescription drug claims for eligible beneficiaries will be reimbursed in accordance with the applicable reimbursement sections of the TRICARE Reimbursement Manual. Beneficiaries shall pay a co-pay in accordance with Chapter 12, Section 11.1 or TRICARE Reimbursement Manual, Chapter 2, Addendum A as appropriate. All deductibles and co-pays apply towards the catastrophic cap. 2. Beneficiary appeal rights are governed by 32 CFR III. EXCLUSIONS A. Drugs prescribed or furnished by a member of the patient s immediate family. B. Drugs, including compounded preparations, that are available over the counter. C. Group C Designation. Investigational drugs with FDA Group C designation have reproducible efficacy in one or more specific tumor types. Such a drug has altered or is likely to alter the pattern of treatment of the disease and can be safely administered by properly trained physicians without specialized supportive care facilities. TRICARE may not costshare use of Group C designated drugs because authorization for Group C distribution for a specific indication is not equivalent to formal FDA approval for that indication. Medical care related to the use of Group C designated drugs may be cost-shared only when the care would have been provided in the absence of the use of the Group C designated drug. D. Orphan drugs without marketing approval, but which are made available on a compassionate use basis, may not be cost-shared. E. Treatment Investigational New Drugs (IND). Under the FDA treatment IND (investigational new drug) regulations enacted in 1987, drugs that are in controlled clinical trials can be provided outside those trials to treat patients with serious or immediately life- 4
17 CHAPTER 8, SECTION 9.1 PHARMACY BENEFITS PROGRAM threatening diseases for which no comparable or satisfactory alternate therapy exists. TRICARE may not cost-share treatment INDs because they have not received FDA marketing approval. However, medical care related to the use of treatment INDs may be costshared when the patient s medical condition warrants their administration and the care is provided in accordance with generally accepted standards of medical practice. F. Irinotecan (Camptosar ) for treatment of metastatic esophageal cancer is unproven. IV. EFFECTIVE DATES A. Labeled uses: the date of FDA approval for the specific indication. B. Off-labeled uses: the date that reliable evidence establishes the safety and efficacy of the drug for that specific use. C. Orphan drugs: the date of FDA marketing approval. - END - 5
18
19 CHAPTER 10, SECTION 5.1 TRANSITIONAL ASSISTANCE MANAGEMENT PROGRAM (TAMP) processed by the contractor using the same rules and cost-shares that apply to active duty dependents. E. Change in eligibility status of a beneficiary during an inpatient hospital stay (see the TRICARE Reimbursement Manual (TRM), Chapter 6, Section 2). F. In cases involving the existence of Other Health Insurance (OHI) for dependents and/or sponsors, treat as double coverage as required by the TRICARE Reimbursement Manual. G. TRICARE Prime: 1. Enrollment in Prime. TAMP eligibles may enroll or re-enroll in TRICARE Prime. 2. Effective Date of Enrollment in TRICARE Prime is as follows: a. TAMP eligibles (including the former active duty member) who were enrolled in Prime immediately prior to their change in status may continue their enrollment in TRICARE Prime with no break in coverage. A reenrollment application must be completed prior to the TAMP expiration period in order to continue with TRICARE Prime. The effective date shall be the date the sponsor separated from active duty as the intent is to ensure that Prime coverage is seamless. See Chapter 10, Section 2.1 for further information on the effective date of enrollment. b. TAMP eligibles who were not enrolled in Prime (including TRICARE Prime Remote (TPR) and TRICARE Prime Remote Active Duty Family Member (TPRADFM)) immediately prior to their change in status may choose to enroll in TRICARE Prime while receiving TAMP coverage but such enrollment is subject to the twentieth of the month rule. That is, if an application for an initial enrollment is received after the twentieth day of the month, Prime enrollment will begin on the first day of the second month after the month in which the application was received by the contractor. See Chapter 10, Section 2.1 for further information on the effective date of enrollment. c. TAMP eligibles whose sponsor is called to active duty. (1) TAMP eligible family members who were enrolled in Prime immediately prior to their sponsor s change in status to active duty may continue their reenrollment in TRICARE Prime with no break in coverage if they reenroll in TRICARE Prime within 30 days of the return to active duty status. If reenrollment is accomplished within 30 days of the return to active duty status, the reenrollment will be retroactive to the date of the change in status from TAMP to active duty. If reenrollment is not accomplished within 30 days of the return to active duty status, the twentieth of the month rule will apply. (2) TAMP eligible family members not enrolled in Prime immediately prior to reactivation (i.e., return to active duty) may choose to enroll in Prime but such initial enrollment is subject to the twentieth of the month rule. That is, if an application for an initial enrollment in Prime is received after the twentieth of the month, Prime enrollment will begin on the first day of the second month after the month in which the application was received by 3 C-120, March 19, 2010
20 CHAPTER 10, SECTION 5.1 TRANSITIONAL ASSISTANCE MANAGEMENT PROGRAM (TAMP) the contractor. See Chapter 10, Section 2.1 for further information on effective date of initial enrollments and reenrollments. (3) For information on the effective dates of enrollments for Active Duty Service Member (ADSM) see the TRICARE Operations Manual (TOM), Chapter 6, Section 1. d. While the TPR and TPRADFM are not available to TAMP eligibles, these programs are considered a Prime-like benefit and enrollment or reenrollment in Prime shall be available to them as stated above. H. The Continued Health Care Benefit Program (CHCBP) may be available to members (and their dependents) after the expiration of TAMP entitlement. The CHCBP is a program that requires enrollment and the payment of quarterly premiums. Application for CHCBP must occur within 60 days of loss of TAMP eligibility. See Chapter 10, Section 4.1 for further information. I. Dental Coverage 1. Dental benefits for TAMP-eligibles are limited to space available care in the Dental Treatment Facility (DTF). 2. The TRICARE Dental Program (TDP) is a voluntary dental insurance program that is available to Active Duty Family Members (ADFMs), Selected Reserve and Individual Ready Reserve members, and their eligible family members. The TDP is not part of the benefits offered under TAMP. Sponsors who were enrolled in the TDP prior to being activated, who then return to Reserve status, may be eligible to re-enroll in the TDP. J. Demonstrations. 1. TAMP eligibles who were also eligible for the benefits of the Operation Noble Eagle/Operation Enduring Freedom Demonstration shall retain those benefits during their TAMP eligibility if the Demonstration is still active. See the TOM, Chapter 20, Section 4. The demonstration was effective for claims for services provided on or after September 14, 2001, and before November 1, The provisions of the demonstration were made permanent by section 704 and 705 of the National Defense Authorization Act (NDAA) for Fiscal Year See Chapter 10, Section TAMP eligibles with a DEERS indicator B for Bosnia shall retain the same special demonstration benefits available to them while on active duty during their TAMP eligibility. - END - 4
21 ELIGIBILITY AND ENROLLMENT CHAPTER 10 SECTION 6.1 TRICARE FOR LIFE ISSUE DATE: September 25, 2001 AUTHORITY: 10 USC 1086(d) I. DESCRIPTION Pursuant to Section 712 of the Fiscal Year (FY) 2001 National Defense Authorization Act (NDAA), Medicare eligible beneficiaries based on age, whose TRICARE eligibility is determined by 10 United States Code (USC) Section 1086, are eligible for Medicare Part A, and are enrolled in Medicare Part B, are eligible for the TRICARE benefit effective October 1, Beneficiaries under age 65 who are also Medicare eligible, are also eligible for TRICARE For Life (TFL) (see the TRICARE Operations Manual (TOM), Chapter 22, Section 1, paragraphs 2.3. and 2.4.). II. POLICY A. Introduction: Section 712 extends TRICARE eligibility to persons who would otherwise have lost their TRICARE eligibility due to attainment of entitlement to hospital insurance benefits under Part A of Medicare based on age. In order for these individuals to retain their TRICARE eligibility, they must be enrolled in the supplementary medical insurance program under Part B of Medicare. In general, in the case of medical or dental care provided to these individuals for which payment may be made under both Medicare and TRICARE, Medicare is the primary payer and TRICARE will normally pay the actual out-of-pocket costs incurred by the person. B. Eligibility. The contractors shall determine from the Defense Enrollment Eligibility Reporting System (DEERS) if the individual is eligible for the TRICARE benefit. C. Under certain conditions TFL beneficiaries may enroll in TRICARE Prime (see the TOM, Chapter 6, Section 1, paragraph 8.4. through ). D. Claims will be reimbursed with the applicable reimbursement sections of the TRICARE Policy, Reimbursement, and Operations manuals. E. Appeal rights are covered in the TOM, Chapter 13. 1
22 CHAPTER 10, SECTION 6.1 TRICARE FOR LIFE F. The contractor shall educate beneficiaries about this benefit as identified by the government. - END - 2
Chapter 4 Section 20.1
 Surgery Chapter 4 Section 20.1 Issue Date: August 29, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) Copyright: CPT only 2006 American Medical Association (or such other date of publication of CPT). All
Surgery Chapter 4 Section 20.1 Issue Date: August 29, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) Copyright: CPT only 2006 American Medical Association (or such other date of publication of CPT). All
Chapter 4 Section 20.1
 Surgery Chapter 4 Section 20.1 Issue Date: August 29, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 61000-61626, 61680-62264, 62268-62284, 62290-63048, 63055-64484, 64505-64595,
Surgery Chapter 4 Section 20.1 Issue Date: August 29, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 61000-61626, 61680-62264, 62268-62284, 62290-63048, 63055-64484, 64505-64595,
Chapter 4 Section 20.1
 Surgery Chapter 4 Section 20.1 Issue Date: August 29, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) Copyright: CPT only 2006 American Medical Association (or such other date of publication of CPT). All
Surgery Chapter 4 Section 20.1 Issue Date: August 29, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) Copyright: CPT only 2006 American Medical Association (or such other date of publication of CPT). All
PUBLICATIONS SYSTEM CHANGE TRANSMITTAL FOR TRICARE POLICY MANUAL (TPM), AUGUST 2002
 OFFICE OFTHE ASSISTANT SECRETARY OF DEFENSE HEALTH AFFAIRS 16401 EAST CENTAETECH PARKWAY AURORA, COLORADO 80011 9066 TRICARE MANAGEMENT ACTIVITY MB&RB CHANGE 163 6010.54-M AUGUST 10, 2012 PUBLICATIONS
OFFICE OFTHE ASSISTANT SECRETARY OF DEFENSE HEALTH AFFAIRS 16401 EAST CENTAETECH PARKWAY AURORA, COLORADO 80011 9066 TRICARE MANAGEMENT ACTIVITY MB&RB CHANGE 163 6010.54-M AUGUST 10, 2012 PUBLICATIONS
Chapter 18 Section 2. EXPIRED - Department Of Defense (DoD) Cancer Prevention And Treatment Clinical Trials Demonstration
 s And Pilot Projects Chapter 18 Section 2 EXPIRED - Department Of Defense (DoD) Cancer Prevention And Treatment Clinical Trials 1.0 PURPOSE The purpose of this demonstration is to improve TRICARE-eligible
s And Pilot Projects Chapter 18 Section 2 EXPIRED - Department Of Defense (DoD) Cancer Prevention And Treatment Clinical Trials 1.0 PURPOSE The purpose of this demonstration is to improve TRICARE-eligible
See Policy CPT/HCPCS CODE section below for any prior authorization requirements
 Effective Date: 1/1/2019 Section: SUR Policy No: 395 1/1/19 Medical Policy Committee Approved Date: 8/17; 2/18; 12/18 Medical Officer Date APPLIES TO: Medicare Only See Policy CPT/HCPCS CODE section below
Effective Date: 1/1/2019 Section: SUR Policy No: 395 1/1/19 Medical Policy Committee Approved Date: 8/17; 2/18; 12/18 Medical Officer Date APPLIES TO: Medicare Only See Policy CPT/HCPCS CODE section below
Highmark Medical Policy Bulletin
 Highmark Medical Policy Bulletin Section: Miscellaneous Number: Z 7 Topic: Electrical Nerve Stimulation Effective Date: March 3, 2003 Issued Date: November 1, 2003 Date Last Reviewed: 12/2002 General Policy
Highmark Medical Policy Bulletin Section: Miscellaneous Number: Z 7 Topic: Electrical Nerve Stimulation Effective Date: March 3, 2003 Issued Date: November 1, 2003 Date Last Reviewed: 12/2002 General Policy
Surgical Privileges Form: "Neurosurgery" Clinical Privileges Request. Requested (To be completed by the applicant) Not Recommended (For committee use)
 Surgical Form: Clinical Request "Neurosurgery" Applicant s Name:. License No. (If Any):... Date:... Scope of Practice:. Facility:.. Place of Work:. the applicant) CATEGORY I: Core : 1. Interpretation of
Surgical Form: Clinical Request "Neurosurgery" Applicant s Name:. License No. (If Any):... Date:... Scope of Practice:. Facility:.. Place of Work:. the applicant) CATEGORY I: Core : 1. Interpretation of
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: spinal_cord_stimulation 3/1980 10/2017 10/2018 10/2017 Description of Procedure or Service Spinal cord stimulation
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: spinal_cord_stimulation 3/1980 10/2017 10/2018 10/2017 Description of Procedure or Service Spinal cord stimulation
PERCUTANEOUS FACET JOINT DENERVATION
 Status Active Medical and Behavioral Health Policy Section: Surgery Policy Number: IV-95 Effective Date: 10/22/2014 Blue Cross and Blue Shield of Minnesota medical policies do not imply that members should
Status Active Medical and Behavioral Health Policy Section: Surgery Policy Number: IV-95 Effective Date: 10/22/2014 Blue Cross and Blue Shield of Minnesota medical policies do not imply that members should
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: facet_joint_denervation 6/2009 4/2017 4/2018 4/2017 Description of Procedure or Service Facet joint denervation
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: facet_joint_denervation 6/2009 4/2017 4/2018 4/2017 Description of Procedure or Service Facet joint denervation
National Imaging Associates, Inc. Clinical guidelines FACET JOINT INJECTIONS, MEDIAL BRANCH BLOCKS, AND FACET JOINT RADIOFREQUENCY NEUROTOMY
 National Imaging Associates, Inc. Clinical guidelines FACET JOINT INJECTIONS, MEDIAL BRANCH BLOCKS, AND FACET JOINT RADIOFREQUENCY NEUROTOMY CPT Codes: Refer to pages 5 and 6 LCD ID Number: L35936 J K
National Imaging Associates, Inc. Clinical guidelines FACET JOINT INJECTIONS, MEDIAL BRANCH BLOCKS, AND FACET JOINT RADIOFREQUENCY NEUROTOMY CPT Codes: Refer to pages 5 and 6 LCD ID Number: L35936 J K
CHAPTER 7 SECTION 24.1 PHASE I, PHASE II, AND PHASE III CANCER CLINICAL TRIALS TRICARE POLICY MANUAL M, AUGUST 1, 2002 MEDICINE
 MEDICINE CHAPTER 7 SECTION 24.1 ISSUE DATE: AUTHORITY: 32 CFR 199.4(e)(26) I. DESCRIPTION The Department of Defense (DoD) Cancer Prevention and Treatment Clinical Trials Demonstration was conducted from
MEDICINE CHAPTER 7 SECTION 24.1 ISSUE DATE: AUTHORITY: 32 CFR 199.4(e)(26) I. DESCRIPTION The Department of Defense (DoD) Cancer Prevention and Treatment Clinical Trials Demonstration was conducted from
2019 ABBOTT REIMBURSEMENT GUIDE CMS Physician Fee Schedule
 ABBOTT REIMBURSEMENT GUIDE CMS Physician Fee Schedule This document and the information contained herein is for general information purposes only and is not intended and does not constitute legal, reimbursement,
ABBOTT REIMBURSEMENT GUIDE CMS Physician Fee Schedule This document and the information contained herein is for general information purposes only and is not intended and does not constitute legal, reimbursement,
2018 Cerebrovascular Reimbursement Coding Fact Sheet
 The information contained in this document is provided for informational purposes only and represents no statement, promise, or guarantee by Cordis Corporation concerning levels of reimbursement, payment,
The information contained in this document is provided for informational purposes only and represents no statement, promise, or guarantee by Cordis Corporation concerning levels of reimbursement, payment,
AXIUM DETACHABLE COILS (FAMILY) CODING AND REIMBURSEMENT GUIDE
 AXIUM DETACHABLE COILS (FAMILY) CODING AND REIMBURSEMENT GUIDE Axium Detachable Coils (Family) Axium detachable coils consist of a platinum embolization coil attached to a delivery pusher. When activated,
AXIUM DETACHABLE COILS (FAMILY) CODING AND REIMBURSEMENT GUIDE Axium Detachable Coils (Family) Axium detachable coils consist of a platinum embolization coil attached to a delivery pusher. When activated,
CARD/MAIL/PRE-APPROVAL/PREFERRED RIDER FOR PRESCRIPTION DRUG [INSURANCE] [Policy]holder: Group Policy No: Effective Date:
![CARD/MAIL/PRE-APPROVAL/PREFERRED RIDER FOR PRESCRIPTION DRUG [INSURANCE] [Policy]holder: Group Policy No: Effective Date: CARD/MAIL/PRE-APPROVAL/PREFERRED RIDER FOR PRESCRIPTION DRUG [INSURANCE] [Policy]holder: Group Policy No: Effective Date:](/thumbs/79/80269988.jpg) RIDER FOR PRESCRIPTION DRUG [INSURANCE] [Policy]holder: Group Policy No: Effective Date: CARD/MAIL/PRE-APPROVAL/PREFERRED The Prescription Drug Coverage under this Rider [replaces] [supplements] the Prescription
RIDER FOR PRESCRIPTION DRUG [INSURANCE] [Policy]holder: Group Policy No: Effective Date: CARD/MAIL/PRE-APPROVAL/PREFERRED The Prescription Drug Coverage under this Rider [replaces] [supplements] the Prescription
No An act relating to health insurance coverage for early childhood developmental disorders, including autism spectrum disorders. (S.
 No. 158. An act relating to health insurance coverage for early childhood developmental disorders, including autism spectrum disorders. (S.223) It is hereby enacted by the General Assembly of the State
No. 158. An act relating to health insurance coverage for early childhood developmental disorders, including autism spectrum disorders. (S.223) It is hereby enacted by the General Assembly of the State
32 CFR (a)(4), (a)(6)(iii), and (a)(6)(iv)
 CHAPTER 15 SECTION 1 ISSUE DATE: November 6, 2007 AUTHORITY: 32 CFR 199.14(a)(4), (a)(6)(iii), and (a)(6)(iv) I. APPLICABILITY This policy is mandatory for the reimbursement of services provided either
CHAPTER 15 SECTION 1 ISSUE DATE: November 6, 2007 AUTHORITY: 32 CFR 199.14(a)(4), (a)(6)(iii), and (a)(6)(iv) I. APPLICABILITY This policy is mandatory for the reimbursement of services provided either
HF10 THERAPY 2018 Ambulatory Surgery Center Reimbursement and Coding Reference Guide
 HF10 therapy, delivered by the Nevro Senza System, is the high-frequency spinal cord stimulation technology operated at 10,000 Hz designed to aid in the management of chronic intractable pain of the trunk
HF10 therapy, delivered by the Nevro Senza System, is the high-frequency spinal cord stimulation technology operated at 10,000 Hz designed to aid in the management of chronic intractable pain of the trunk
Neurostimulator Devices and Supplies
 Neurostimulator Devices and Supplies Chapter.1 Enrollment..................................................................... -2.2 Benefits, Limitations, and Authorization Requirements...........................
Neurostimulator Devices and Supplies Chapter.1 Enrollment..................................................................... -2.2 Benefits, Limitations, and Authorization Requirements...........................
Reimbursement Guide Zenith Fenestrated AAA Endovascular Graft
 MEDICAL Reimbursement Guide Zenith Fenestrated AAA Endovascular Graft Disclaimer: The information provided herein reflects Cook s understanding of the procedure(s) and/or device(s) from sources that may
MEDICAL Reimbursement Guide Zenith Fenestrated AAA Endovascular Graft Disclaimer: The information provided herein reflects Cook s understanding of the procedure(s) and/or device(s) from sources that may
Corporate Medical Policy
 Corporate Medical Policy Endovascular Therapies for Extracranial Vertebral Artery Disease File Name: Origination: Last CAP Review: Next CAP Review: Last Review: endovascular_therapies_for_extracranial_vertebral_artery_disease
Corporate Medical Policy Endovascular Therapies for Extracranial Vertebral Artery Disease File Name: Origination: Last CAP Review: Next CAP Review: Last Review: endovascular_therapies_for_extracranial_vertebral_artery_disease
CY2015 Hospital Outpatient: Endovascular Procedure APCs and Complexity Adjustments
 CY2015 Hospital Outpatient: Endovascular Procedure APCs Complexity Adjustments Comprehensive Ambulatory Payment Classifications (c-apcs) CMS finalized the implementation of 25 Comprehensive APC to further
CY2015 Hospital Outpatient: Endovascular Procedure APCs Complexity Adjustments Comprehensive Ambulatory Payment Classifications (c-apcs) CMS finalized the implementation of 25 Comprehensive APC to further
All Indiana Health Coverage Programs Hospitals, Ambulatory Surgical Centers, Physicians, and Durable Medical Equipment Providers
 P R O V I D E R B U L L E T I N B T 2 0 0 0 3 2 S E P T E M B E R 8, 2 0 0 0 To: Subject: All Indiana Health Coverage Programs Hospitals, Ambulatory Surgical Centers, Physicians, and Durable Medical Equipment
P R O V I D E R B U L L E T I N B T 2 0 0 0 3 2 S E P T E M B E R 8, 2 0 0 0 To: Subject: All Indiana Health Coverage Programs Hospitals, Ambulatory Surgical Centers, Physicians, and Durable Medical Equipment
Medical Review Criteria Implantable Neurostimulators
 Medical Review Criteria Implantable Neurostimulators Subject: Implantable Neurostimulators Effective Date: April 14, 2017 Authorization: Prior authorization is required for covered implantable stimulators
Medical Review Criteria Implantable Neurostimulators Subject: Implantable Neurostimulators Effective Date: April 14, 2017 Authorization: Prior authorization is required for covered implantable stimulators
Inspire Medical Systems. Physician Billing Guide
 Inspire Medical Systems Physician Billing Guide 2019 Inspire Medical Systems Physician Billing Guide This Physician Billing Guide was developed to help providers correctly bill for Inspire Upper Airway
Inspire Medical Systems Physician Billing Guide 2019 Inspire Medical Systems Physician Billing Guide This Physician Billing Guide was developed to help providers correctly bill for Inspire Upper Airway
Department of Neurological Surgery
 Department of Neurological Surgery CAT 1 A Basic Privileges: Patient management, including H & Ps and diagnostic and therapeutic treatments, procedures and interventions, Requiring a level of training
Department of Neurological Surgery CAT 1 A Basic Privileges: Patient management, including H & Ps and diagnostic and therapeutic treatments, procedures and interventions, Requiring a level of training
CY2017 Hospital Outpatient: Vascular Procedure APCs and Complexity Adjustments
 CY2017 Hospital Outpatient: Vascular Procedure APCs and Complexity Adjustments Comprehensive Ambulatory Payment Classifications (c-apcs) In CY2015 and in an effort to help pay providers for quality, not
CY2017 Hospital Outpatient: Vascular Procedure APCs and Complexity Adjustments Comprehensive Ambulatory Payment Classifications (c-apcs) In CY2015 and in an effort to help pay providers for quality, not
CHAPTER 1 Section 11.1, pages 1 and 2 Section 11.1, pages 1 and 2
 CHANGE 11 6010.60-M OCTOBER 27, 2017 REMOVE PAGE(S) INSERT PAGE(S) CHAPTER 1 Section 11.1, pages 1 and 2 Section 11.1, pages 1 and 2 CHAPTER 4 Section 20.1, pages 3 through 5 Section 20.1, pages 3 through
CHANGE 11 6010.60-M OCTOBER 27, 2017 REMOVE PAGE(S) INSERT PAGE(S) CHAPTER 1 Section 11.1, pages 1 and 2 Section 11.1, pages 1 and 2 CHAPTER 4 Section 20.1, pages 3 through 5 Section 20.1, pages 3 through
Chapter 18 Section 15. Department Of Defense (DoD) Applied Behavior Analysis (ABA) Pilot For Non-Active Duty Family Members (NADFMs)
 Demonstrations And Pilot Projects Chapter 18 Section 15 Department Of Defense (DoD) Applied Behavior Analysis (ABA) Pilot 1.0 PURPOSE Under authority of 10 United States Code (USC) 1092, TRICARE will continue
Demonstrations And Pilot Projects Chapter 18 Section 15 Department Of Defense (DoD) Applied Behavior Analysis (ABA) Pilot 1.0 PURPOSE Under authority of 10 United States Code (USC) 1092, TRICARE will continue
Ultrasound and Fluoroscopic Paravertebral Facet Joint Injections
 Policy Number FAC06222011RP Ultrasound and Fluoroscopic Approved By UnitedHealthcare Medicare Committee Current Approval Date 06/25/2014 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable
Policy Number FAC06222011RP Ultrasound and Fluoroscopic Approved By UnitedHealthcare Medicare Committee Current Approval Date 06/25/2014 IMPORTANT NOTE ABOUT THIS REIMBURSEMENT POLICY This policy is applicable
Nevro Reimbursement Support
 HF10 therapy, delivered by the Nevro Senza System, is the high-frequency spinal cord stimulation technology operated at 10,000 Hz designed to aid in the management of chronic intractable pain of the trunk
HF10 therapy, delivered by the Nevro Senza System, is the high-frequency spinal cord stimulation technology operated at 10,000 Hz designed to aid in the management of chronic intractable pain of the trunk
ADDITIONS. The following codes have been added.
 ADDITIONS The following codes have been added. 99446 Interprofessional telephone/internet assessment and management service provided by treating/requesting physician or other qualified health care professional;
ADDITIONS The following codes have been added. 99446 Interprofessional telephone/internet assessment and management service provided by treating/requesting physician or other qualified health care professional;
2016 HF10 THERAPY REIMBURSEMENT REFERENCE GUIDE
 206 HF0 THERAPY REIMBURSEMENT REFERENCE GUIDE HF0 therapy, delivered by the Nevro Senza System, is a new high-frequency spinal cord stimulation technology designed to aid in the management of chronic intractable
206 HF0 THERAPY REIMBURSEMENT REFERENCE GUIDE HF0 therapy, delivered by the Nevro Senza System, is a new high-frequency spinal cord stimulation technology designed to aid in the management of chronic intractable
Sample page. POWER UP YOUR CODING with Optum360, your trusted coding partner for 32 years. Visit optum360coding.com.
 2018 Complete Guide for Interventional Radiology An in-depth guide to interventional radiology coding, billing, and reimbursement for facilities and physicians POWER UP YOUR CODING with Optum360, your
2018 Complete Guide for Interventional Radiology An in-depth guide to interventional radiology coding, billing, and reimbursement for facilities and physicians POWER UP YOUR CODING with Optum360, your
Quality Metrics. Stroke Related Procedure Outcomes
 Quality Metrics Stroke Related Procedure Outcomes Below is a description of some of the stroke-related procedures performed at St. Dominic Hospital in Jackson, with quality information on the complication
Quality Metrics Stroke Related Procedure Outcomes Below is a description of some of the stroke-related procedures performed at St. Dominic Hospital in Jackson, with quality information on the complication
TRICARE Dental Options
 APRIL 2018 TRICARE Dental Options TRICARE dental options for you and your family This fact sheet provides information about the TRICARE Active Duty Dental Program (ADDP), the TRICARE Dental Program (TDP)
APRIL 2018 TRICARE Dental Options TRICARE dental options for you and your family This fact sheet provides information about the TRICARE Active Duty Dental Program (ADDP), the TRICARE Dental Program (TDP)
Corporate Medical Policy
 Corporate Medical Policy Epidural Steroid Injections for Back Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: epidural_steroid_injections_for_back_pain 2/2016 4/2017 4/2018
Corporate Medical Policy Epidural Steroid Injections for Back Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: epidural_steroid_injections_for_back_pain 2/2016 4/2017 4/2018
$250 (Deductible does not apply to Tier 1 and Tier 2) $500 (Deductible does not apply to Tier 1 and Tier 2)
 Benefit Summary Outpatient Prescription Drug Illinois 5/50/100/250 Plan 455 Your Co-payment and/or Co-insurance is determined by the tier to which the Prescription Drug List (PDL) Management Committee
Benefit Summary Outpatient Prescription Drug Illinois 5/50/100/250 Plan 455 Your Co-payment and/or Co-insurance is determined by the tier to which the Prescription Drug List (PDL) Management Committee
Detailed Summary of the Proposed Rule for the Hospital Outpatient Prospective Payment System
 Detailed Summary of the Proposed Rule for the Hospital Outpatient Prospective Payment System The Centers for Medicare and Medicaid Services (CMS) released its proposed rule for calendar year (CY) 2017
Detailed Summary of the Proposed Rule for the Hospital Outpatient Prospective Payment System The Centers for Medicare and Medicaid Services (CMS) released its proposed rule for calendar year (CY) 2017
Ultrasound Reimbursement Information for Anesthesiology 1
 GE Healthcare Ultrasound Reimbursement Information for Anesthesiology 1 January, 2009 www.gehealthcare.com/reimbursement This overview addresses coding, coverage, and for ultrasound guidance with continuous
GE Healthcare Ultrasound Reimbursement Information for Anesthesiology 1 January, 2009 www.gehealthcare.com/reimbursement This overview addresses coding, coverage, and for ultrasound guidance with continuous
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Implanted Peripheral Nerve Stimulator (PNS) for Pain Control Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Implanted Peripheral Nerve Stimulator (PNS)
Implanted Peripheral Nerve Stimulator (PNS) for Pain Control Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Implanted Peripheral Nerve Stimulator (PNS)
Chapter 4 Section 9.1
 Surgery Chapter 4 Section 9.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 33010-33130, 33140, 33141, 33361-33369, 33200-37186, 37195-37785, 92950-93272,
Surgery Chapter 4 Section 9.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 33010-33130, 33140, 33141, 33361-33369, 33200-37186, 37195-37785, 92950-93272,
2012 CPT Coding Update AANS/CNS Joint Section on Disorders of the Spine and Peripheral Nerves
 2012 CPT Coding Update AANS/CNS Joint Section on Disorders of the Spine and Peripheral Nerves Joseph S. Cheng, M.D., M.S. Associate Professor of Neurological Surgery, Orthopedic Surgery, and Rehabilitation
2012 CPT Coding Update AANS/CNS Joint Section on Disorders of the Spine and Peripheral Nerves Joseph S. Cheng, M.D., M.S. Associate Professor of Neurological Surgery, Orthopedic Surgery, and Rehabilitation
H F 1 0 T H E R A P Y R E I M B U R S E M E N T R E F E R E N C E G U I D E
 HF10 therapy, delivered by the Nevro Senza System, is the high-frequency spinal cord stimulation technology designed to aid in the management of chronic intractable pain of the trunk/limbs without paresthesia.
HF10 therapy, delivered by the Nevro Senza System, is the high-frequency spinal cord stimulation technology designed to aid in the management of chronic intractable pain of the trunk/limbs without paresthesia.
2014 Deleted CPT Codes
 2014 Deleted CPT Codes Surgery 13150 - Repair, complex, eyelids, nose, ears and/or lips; 1.0 cm or less 19102 - Biopsy of breast; percutaneous, needle core, using imaging guidance 19103 - Biopsy of breast;
2014 Deleted CPT Codes Surgery 13150 - Repair, complex, eyelids, nose, ears and/or lips; 1.0 cm or less 19102 - Biopsy of breast; percutaneous, needle core, using imaging guidance 19103 - Biopsy of breast;
CODING SHEETS CHRONIC INTRACTABLE PAIN MANAGEMENT. Effective January 1, 2009 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE
 CODING SHEETS CHRONIC INTRACTABLE PAIN MANAGEMENT Effective January 1, 2009 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE Phone: 800-609-1108 Email: codmanpump@aol.com Fax: 303-703-1572
CODING SHEETS CHRONIC INTRACTABLE PAIN MANAGEMENT Effective January 1, 2009 CODMAN 3000 NEUROMODULATION AND ONCOLOGY REIMBURSEMENT HOTLINE Phone: 800-609-1108 Email: codmanpump@aol.com Fax: 303-703-1572
CHAPTER 1 SECTION 10.1 TRICARE STANDARD - CLINICAL PREVENTIVE SERVICES TRICARE/CHAMPUS POLICY MANUAL M DEC 1998 MEDICAL SERVICES
 TRICARE/CHAMPUS POLICY MANUAL 6010.47-M DEC 1998 MEDICAL SERVICES CHAPTER 1 SECTION 10.1 Issue Date: April 19, 1983 Authority: 32 CFR 199.4(e)(3)(ii) and (g)(37) I. PROCEDURE CODES 45300, 45330, 45355,
TRICARE/CHAMPUS POLICY MANUAL 6010.47-M DEC 1998 MEDICAL SERVICES CHAPTER 1 SECTION 10.1 Issue Date: April 19, 1983 Authority: 32 CFR 199.4(e)(3)(ii) and (g)(37) I. PROCEDURE CODES 45300, 45330, 45355,
Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association
 Implanted Peripheral Nerve Stimulator (PNS) Page 1 of 6 Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association Title: Implanted Peripheral Nerve Stimulator (PNS) for Pain
Implanted Peripheral Nerve Stimulator (PNS) Page 1 of 6 Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association Title: Implanted Peripheral Nerve Stimulator (PNS) for Pain
POLICY AND PROCEDURE
 PAGE: Page 1 of 8 SCOPE: This policy applies to any provider furnishing services represented by Category III CPT codes. PURPOSE & IMPORTANT REMINDER: This policy is current at the time of publication.
PAGE: Page 1 of 8 SCOPE: This policy applies to any provider furnishing services represented by Category III CPT codes. PURPOSE & IMPORTANT REMINDER: This policy is current at the time of publication.
Pipeline Embolization Device
 Pipeline Embolization Device The power to redefine aneurysm treatment. REDEFINE The Pipeline device redefines treatment for large or giant wide-necked aneurysms by reconstructing the parent artery and
Pipeline Embolization Device The power to redefine aneurysm treatment. REDEFINE The Pipeline device redefines treatment for large or giant wide-necked aneurysms by reconstructing the parent artery and
Questions and Answers on 2009 H1N1 Vaccine Financing
 Questions and Answers on 2009 H1N1 Vaccine Financing General Financing Questions Considerations of financing distinguish between those related to the vaccine itself, the ancillary supplies needed to administer
Questions and Answers on 2009 H1N1 Vaccine Financing General Financing Questions Considerations of financing distinguish between those related to the vaccine itself, the ancillary supplies needed to administer
Neurostimulators and Neuromuscular
 Neurostimulators and Neuromuscular Stimulators Chapter.1 Enrollment..................................................................... -2.2 Benefits, Limitations, and Authorization Requirements...........................
Neurostimulators and Neuromuscular Stimulators Chapter.1 Enrollment..................................................................... -2.2 Benefits, Limitations, and Authorization Requirements...........................
Neuromuscular Electrical Stimulator (NMES) Corporate Medical Policy
 Neuromuscular Electrical Stimulator (NMES) Corporate Medical Policy File name: Neuromuscular Electrical Stimulator (NMES) File Code: UM.NS.04 Origination: 05/01/2007 Last Review: 06/2018 Next Review: 06/2019
Neuromuscular Electrical Stimulator (NMES) Corporate Medical Policy File name: Neuromuscular Electrical Stimulator (NMES) File Code: UM.NS.04 Origination: 05/01/2007 Last Review: 06/2018 Next Review: 06/2019
Chapter 4 Section 9.1
 Surgery Chapter 4 Section 9.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 33010-33130, 33140, 33141, 33200-37186, 37195-37785, 92950-93272, 93303-93581,
Surgery Chapter 4 Section 9.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 33010-33130, 33140, 33141, 33200-37186, 37195-37785, 92950-93272, 93303-93581,
tens_(transcutaneous_electrical_nerve_stimulator) 7/ / / /2014 This policy is NOT effective until January 13, 2015
 Corporate Medical Policy TENS (Transcutaneous Electrical Nerve Stimulator) File Name: Origination: Last CAP Review: Next CAP Review: Last Review: tens_(transcutaneous_electrical_nerve_stimulator) 7/1982
Corporate Medical Policy TENS (Transcutaneous Electrical Nerve Stimulator) File Name: Origination: Last CAP Review: Next CAP Review: Last Review: tens_(transcutaneous_electrical_nerve_stimulator) 7/1982
Neuroform Microdelivery Stent System
 Neuroform Microdelivery Stent System Patient Information Booklet TABLE OF CONTENTS What is the Purpose of This Booklet?...2 What is a Wide Neck, Intracranial Aneurysm?...2 What is the Treatment for Wide
Neuroform Microdelivery Stent System Patient Information Booklet TABLE OF CONTENTS What is the Purpose of This Booklet?...2 What is a Wide Neck, Intracranial Aneurysm?...2 What is the Treatment for Wide
Vascular Plug Procedures 2014 CODING AND PAYMENT REFERENCE GUIDE ST. JUDE MEDICAL - CARDIOVASCULAR DIVISION
 Vascular Plug Procedures 2014 CODING AND PAYMENT REFERENCE GUIDE ST. JUDE MEDICAL - CARDIOVASCULAR DIVISION IMPORTANT: St. Jude Medical provides this reference guide for general information purposes only
Vascular Plug Procedures 2014 CODING AND PAYMENT REFERENCE GUIDE ST. JUDE MEDICAL - CARDIOVASCULAR DIVISION IMPORTANT: St. Jude Medical provides this reference guide for general information purposes only
Introducing a New Treatment Method for Brain Aneurysms
 Pipeline Embolization Device Introducing a New Treatment Method for Brain Aneurysms UNDERSTANDING ANEURYSMS What is a brain aneurysm? An aneurysm is an outpouching in an artery caused by weakness in the
Pipeline Embolization Device Introducing a New Treatment Method for Brain Aneurysms UNDERSTANDING ANEURYSMS What is a brain aneurysm? An aneurysm is an outpouching in an artery caused by weakness in the
Diagnostic and interventional venous procedures (lower extremity)
 2017 Coding and Medicare payment guide Diagnostic and interventional venous procedures (lower extremity) All coding, coverage, billing and payment information provided herein by Philips Volcano is gathered
2017 Coding and Medicare payment guide Diagnostic and interventional venous procedures (lower extremity) All coding, coverage, billing and payment information provided herein by Philips Volcano is gathered
Chapter 15 Section 1
 Chapter 15 Section 1 Issue Date: November 6, 2007 Authority: 32 CFR 199.14(a)(3) and (a)(6)(ii) 1.0 APPLICABILITY This policy is mandatory for the reimbursement of services provided either by network or
Chapter 15 Section 1 Issue Date: November 6, 2007 Authority: 32 CFR 199.14(a)(3) and (a)(6)(ii) 1.0 APPLICABILITY This policy is mandatory for the reimbursement of services provided either by network or
2 016 HF10 THERAPY HOSPITAL OUTPATIENT DEPARTMENT AND AMBULATORY SURGERY CENTER REIMBURSEMENT REFERENCE GUIDE
 HF10 therapy, delivered by the Nevro Senza System, is a new high-frequency spinal cord stimulation technology designed to aid in the management of chronic intractable pain of the trunk/limbs, including
HF10 therapy, delivered by the Nevro Senza System, is a new high-frequency spinal cord stimulation technology designed to aid in the management of chronic intractable pain of the trunk/limbs, including
CMS Rulings. Department of Health and Human Services. Centers for Medicare & Medicaid Services. Ruling No Date: May 3, 2005
 CMS Rulings Department of Health and Human Services Centers for Medicare & Medicaid Services Ruling No. 05-01 Date: May 3, 2005 CMS Rulings are decisions of the Administrator that serve as precedent final
CMS Rulings Department of Health and Human Services Centers for Medicare & Medicaid Services Ruling No. 05-01 Date: May 3, 2005 CMS Rulings are decisions of the Administrator that serve as precedent final
Case Log Mapping Update: April 2018 Review Committee for Neurological Surgery
 Case Log Mapping Update: April 2018 Review Committee for Neurological Surgery The Review Committee has made the following changes to the CPT code mappings: The following previously untracked CPT codes
Case Log Mapping Update: April 2018 Review Committee for Neurological Surgery The Review Committee has made the following changes to the CPT code mappings: The following previously untracked CPT codes
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: vagus_nerve_stimulation 6/1998 5/2017 5/2018 5/2017 Description of Procedure or Service Stimulation of the
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: vagus_nerve_stimulation 6/1998 5/2017 5/2018 5/2017 Description of Procedure or Service Stimulation of the
CPT 2015: Prepare Your Coding Practice For New Codes As Technology Makes An Advance
 2015 Radiology Coding Survival Guide Section X : 2015 Coding Updates CPT 2015: Prepare Your Coding Practice For New Codes As Technology Makes An Advance Watch for changes in Vertebral fracture assessment,
2015 Radiology Coding Survival Guide Section X : 2015 Coding Updates CPT 2015: Prepare Your Coding Practice For New Codes As Technology Makes An Advance Watch for changes in Vertebral fracture assessment,
TRICARE Dental Options
 JANUARY 2019 TRICARE Dental Options TRICARE dental options for you and your family This fact sheet provides information about the TRICARE Active Duty Dental Program (ADDP), the TRICARE Dental Program (TDP),
JANUARY 2019 TRICARE Dental Options TRICARE dental options for you and your family This fact sheet provides information about the TRICARE Active Duty Dental Program (ADDP), the TRICARE Dental Program (TDP),
UNMH Neurosurgery Clinical Privileges
 All new applicants must meet the following requirements as approved by the UNMH Board of Trustees effective: 02/20/2015 INSTRUCTIONS Applicant: Check off the "Requested" box for each privilege requested.
All new applicants must meet the following requirements as approved by the UNMH Board of Trustees effective: 02/20/2015 INSTRUCTIONS Applicant: Check off the "Requested" box for each privilege requested.
Coding of Procedures in Interventional Nephrology Produced in collaboration with:
 Coding of Procedures in Interventional Nephrology 2013 Produced in collaboration with: Introduction Proper coding of interventional procedures is a difficult and daunting, but essential task. Each procedure
Coding of Procedures in Interventional Nephrology 2013 Produced in collaboration with: Introduction Proper coding of interventional procedures is a difficult and daunting, but essential task. Each procedure
2009 Pain Coding Update and Pain Industry Business Trends
 2009 Pain Coding Update and Pain Industry Business Trends Linda Van Horn, MBA June 13, 2009 2009 Pain Coding Update and Pain Industry Trends Agenda 2009 CPT Coding Updates Pay For Incentives ICD-10 American
2009 Pain Coding Update and Pain Industry Business Trends Linda Van Horn, MBA June 13, 2009 2009 Pain Coding Update and Pain Industry Trends Agenda 2009 CPT Coding Updates Pay For Incentives ICD-10 American
Inspire Medical Systems. Hospital Billing Guide
 Inspire Medical Systems Hospital Billing Guide Inspire Medical Systems Hospital Billing Guide This Hospital Billing Guide was developed to help centers correctly bill for Inspire Upper Airway Stimulation
Inspire Medical Systems Hospital Billing Guide Inspire Medical Systems Hospital Billing Guide This Hospital Billing Guide was developed to help centers correctly bill for Inspire Upper Airway Stimulation
Basic Standards for. Fellowship Training in. Acute and Chronic Pain Management. in Anesthesiology
 Basic Standards for Fellowship Training in Acute and Chronic Pain Management in Anesthesiology American Osteopathic Association and American Osteopathic College of Anesthesiologists BOT, 7/1995 BOT, 11/2002
Basic Standards for Fellowship Training in Acute and Chronic Pain Management in Anesthesiology American Osteopathic Association and American Osteopathic College of Anesthesiologists BOT, 7/1995 BOT, 11/2002
2018 HEMODIALYSIS CATHETERS CODING AND REIMBURSEMENT GUIDE
 2018 HEMODIALYSIS CATHETERS CODING AND REIMBURSEMENT GUIDE Contents Overview of Central Venous Access s for Hemodialysis 2 Procedures Using Hemodialysis s 2 Physician Reimbursement for Hemodialysis s 3
2018 HEMODIALYSIS CATHETERS CODING AND REIMBURSEMENT GUIDE Contents Overview of Central Venous Access s for Hemodialysis 2 Procedures Using Hemodialysis s 2 Physician Reimbursement for Hemodialysis s 3
Corporate Medical Policy
 Corporate Medical Policy Axial Lumbosacral Interbody Fusion File Name: Origination: Last CAP Review: Next CAP Review: Last Review: axial_lumbosacral_interbody_fusion 6/2009 10/2017 10/2018 10/2017 Description
Corporate Medical Policy Axial Lumbosacral Interbody Fusion File Name: Origination: Last CAP Review: Next CAP Review: Last Review: axial_lumbosacral_interbody_fusion 6/2009 10/2017 10/2018 10/2017 Description
2013 Coding Changes. Diagnostic Radiology. Nuclear Medicine
 2013 Coding Changes The principal coding changes affecting Radiologists in 2013 occur in the Interventional Radiology Section of the AMA/CPT Manual. As in the past, we continue to see the Relative Update
2013 Coding Changes The principal coding changes affecting Radiologists in 2013 occur in the Interventional Radiology Section of the AMA/CPT Manual. As in the past, we continue to see the Relative Update
1105 two (2) vertebrae... 1, add on per additional vertebra
 SPINE STAGE OPERATIONS Staged operations shall be paid at 100% for the first stage and 85% for the second stage. Where the second stage pays a higher fee 100% shall be paid and the first stage shall be
SPINE STAGE OPERATIONS Staged operations shall be paid at 100% for the first stage and 85% for the second stage. Where the second stage pays a higher fee 100% shall be paid and the first stage shall be
Family Planning Eligibility Program
 INDIANA HEALTH COVERAGE PROGRAMS PROVIDER REFERENCE M ODULE Family Planning Eligibility Program L I B R A R Y R E F E R E N C E N U M B E R : P R O M O D 0 0 0 5 3 P U B L I S H E D : N O V E M B E R 2
INDIANA HEALTH COVERAGE PROGRAMS PROVIDER REFERENCE M ODULE Family Planning Eligibility Program L I B R A R Y R E F E R E N C E N U M B E R : P R O M O D 0 0 0 5 3 P U B L I S H E D : N O V E M B E R 2
July 5, Dear Parents:
 Dear Parents: July 5, 2013 We were recently made aware of some changes regarding autism benefits for retirees under your insurance through TRICARE-Prime. Please see the excerpt below that was released
Dear Parents: July 5, 2013 We were recently made aware of some changes regarding autism benefits for retirees under your insurance through TRICARE-Prime. Please see the excerpt below that was released
Reimbursement Information for Diagnostic Ultrasound and Ultrasound-guided Vascular Procedures 1
 GE Healthcare Reimbursement Information for Diagnostic Ultrasound and Ultrasound-guided Vascular Procedures 1 January, 2013 www.gehealthcare.com/reimbursement This overview addresses coding, coverage,
GE Healthcare Reimbursement Information for Diagnostic Ultrasound and Ultrasound-guided Vascular Procedures 1 January, 2013 www.gehealthcare.com/reimbursement This overview addresses coding, coverage,
Neuro-Vascular Intervention AAPC Regional Conference Springfield, MA
 Neuro-Vascular Intervention AAPC Regional Conference Springfield, MA October 8, 2010 1 Presented by: David Zielske, MD,CIRCC, CPC H, CCC, CCS, RCC General Recommendations for Physician Dictations State
Neuro-Vascular Intervention AAPC Regional Conference Springfield, MA October 8, 2010 1 Presented by: David Zielske, MD,CIRCC, CPC H, CCC, CCS, RCC General Recommendations for Physician Dictations State
Reimbursement Information for Diagnostic Ultrasound and Ultrasound-guided Procedures Commonly Performed by Otolaryngologists
 GE Healthcare Reimbursement Information for Diagnostic Ultrasound and Ultrasound-guided Procedures Commonly Performed by Otolaryngologists 1 January, 2013 www.gehealthcare.com/reimbursement imagination
GE Healthcare Reimbursement Information for Diagnostic Ultrasound and Ultrasound-guided Procedures Commonly Performed by Otolaryngologists 1 January, 2013 www.gehealthcare.com/reimbursement imagination
ABBOTT CODING GUIDE CHRONIC PAIN. Effective January 1, 2019 INTRO SPINAL CORD STIMULATION (SCS) RADIOFREQUENCY ABLATION (RFA)
 ABBOTT CODING GUIDE CHRONIC PAIN Effective January 1, 2019 CHRONIC PAIN Effective January 1, 2019 Introduction The Chronic Pain Coding Guide is intended to provide reference material related to general
ABBOTT CODING GUIDE CHRONIC PAIN Effective January 1, 2019 CHRONIC PAIN Effective January 1, 2019 Introduction The Chronic Pain Coding Guide is intended to provide reference material related to general
2018 Endovascular Reimbursement Coding Fact Sheet
 The information contained in this document is provided for informational purposes only and represents no statement, promise, or guarantee by Cordis Corporation concerning levels of reimbursement, payment,
The information contained in this document is provided for informational purposes only and represents no statement, promise, or guarantee by Cordis Corporation concerning levels of reimbursement, payment,
Regence Enliven Dental Plan Highlights for Groups /1/2018
 Plan Features This plan is based and includes preventive and diagnostic services, as well as restorative and major services. Orthodontia is included for all ages. This plan features an Exclusive Provider
Plan Features This plan is based and includes preventive and diagnostic services, as well as restorative and major services. Orthodontia is included for all ages. This plan features an Exclusive Provider
CHAPTER 1 Section 3.1, page 5 Section 3.1, page 5
 CHANGE 193 6010.57-M NOVEMBER 20, 2017 REMOVE PAGE(S) INSERT PAGE(S) CHAPTER 1 Section 3.1, page 5 Section 3.1, page 5 CHAPTER 4 Section 6.1, pages 1 through 4 Section 6.1, pages 1 through 4 Section 15.1,
CHANGE 193 6010.57-M NOVEMBER 20, 2017 REMOVE PAGE(S) INSERT PAGE(S) CHAPTER 1 Section 3.1, page 5 Section 3.1, page 5 CHAPTER 4 Section 6.1, pages 1 through 4 Section 6.1, pages 1 through 4 Section 15.1,
Arterial Map of the Thorax, Abdomen and Pelvis 2017 Edition
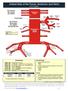 Arterial Map of the Thorax, Abdomen and Pelvis Angiography 75605 (-26) Aortography, thoracic 75625 (-26) Aortography, abdominal by serialography 75630 (-26) Aortography, abdominal + bilat iliofemoral 75705
Arterial Map of the Thorax, Abdomen and Pelvis Angiography 75605 (-26) Aortography, thoracic 75625 (-26) Aortography, abdominal by serialography 75630 (-26) Aortography, abdominal + bilat iliofemoral 75705
Chapter 4 Section 9.1
 Surgery Chapter 4 Section 9.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 33010-33130, 33140, 33141, 33361-33369, 33200-37186, 37195-37785, 92950-93272,
Surgery Chapter 4 Section 9.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 33010-33130, 33140, 33141, 33361-33369, 33200-37186, 37195-37785, 92950-93272,
Codes for Back and Spinal Procedures
 20930 Allograft for spine surgery only; morselized 20931 Allograft for spine surgery only; structural 20936 Autograft for spine surgery only (includes harvesting the graft); local (eg, ribs, spinous process,
20930 Allograft for spine surgery only; morselized 20931 Allograft for spine surgery only; structural 20936 Autograft for spine surgery only (includes harvesting the graft); local (eg, ribs, spinous process,
Neurosurgical decision making in structural lesions causing stroke. Dr Rakesh Ranjan MS, MCh, Dip NB (Neurosurgery)
 Neurosurgical decision making in structural lesions causing stroke Dr Rakesh Ranjan MS, MCh, Dip NB (Neurosurgery) Subarachnoid Hemorrhage Every year, an estimated 30,000 people in the United States experience
Neurosurgical decision making in structural lesions causing stroke Dr Rakesh Ranjan MS, MCh, Dip NB (Neurosurgery) Subarachnoid Hemorrhage Every year, an estimated 30,000 people in the United States experience
2017 Cardiology Survival Guide
 2017 Cardiology Survival Guide Chapter 2: Angioplasty/Atherectomy/Stent The term angioplasty literally means "blood vessel repair." During an angioplasty procedure, the physician inserts a catheter, with
2017 Cardiology Survival Guide Chapter 2: Angioplasty/Atherectomy/Stent The term angioplasty literally means "blood vessel repair." During an angioplasty procedure, the physician inserts a catheter, with
Non-voluntary dental (2-9) Florida
 Non-voluntary dental (2-9) Option 1 DMO Option 2 Freedom-of-Choice Monthly selection between DMO and PPO Max Option 3 Freedom-of-Choice Monthly selection between DMO and PPO Option 4 PPO Max Copay 64 Copay
Non-voluntary dental (2-9) Option 1 DMO Option 2 Freedom-of-Choice Monthly selection between DMO and PPO Max Option 3 Freedom-of-Choice Monthly selection between DMO and PPO Option 4 PPO Max Copay 64 Copay
Report to the Senate Armed Services Committee
 Report to the Senate Armed Services Committee The Department of Defense Comprehensive Autism Care Demonstration REPORT ON EFFORTS BEING CONDUCTED BY THE DEPARTMENT OF DEFENSE ON APPLIED BEHAVIOR ANALYSIS
Report to the Senate Armed Services Committee The Department of Defense Comprehensive Autism Care Demonstration REPORT ON EFFORTS BEING CONDUCTED BY THE DEPARTMENT OF DEFENSE ON APPLIED BEHAVIOR ANALYSIS
2015 Facility and Physician Billing Guide Heart Valve Technologies
 2015 Facility and Physician Billing Guide Heart Valve Technologies PHYSICIAN BILLING CODES Clinicians use Current Procedural Terminology (CPT 1 ) codes to bill for procedures and services. Each CPT code
2015 Facility and Physician Billing Guide Heart Valve Technologies PHYSICIAN BILLING CODES Clinicians use Current Procedural Terminology (CPT 1 ) codes to bill for procedures and services. Each CPT code
Clinical Policy: Cochlear Implant Replacements Reference Number: CP.MP.14
 Clinical Policy: Reference Number: CP.MP.14 Effective Date: 02/09 Last Review Date: 09/17 Revision Log Coding Implications See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.14 Effective Date: 02/09 Last Review Date: 09/17 Revision Log Coding Implications See Important Reminder at the end of this policy for important regulatory and
Procedures commonly seen at Vanderbilt Medical Center PACU s: Cervical, thoracic, lumbar, and sacral spine surgeries. Goes to 6N
 Procedures commonly seen at Vanderbilt Medical Center PACU s: Cervical, thoracic, lumbar, and sacral spine surgeries Goes to 6N Burr holes and Craniotomies for hemorrhage, tumors, trauma, debulking Goes
Procedures commonly seen at Vanderbilt Medical Center PACU s: Cervical, thoracic, lumbar, and sacral spine surgeries Goes to 6N Burr holes and Craniotomies for hemorrhage, tumors, trauma, debulking Goes
OFFICE OF THE ASSISTANT SECRETARY OF DEFENSE HEAL TH AFFAIRS EASTCENTRETE H PARKWAY AURORA, CO ~9066
 OFFICE OF THE ASSISTANT SECRETARY OF DEFENSE HEAL TH AFFAIRS 16401 EASTCENTRETE H PARKWAY AURORA, CO 80011 ~9066 DEFENSE HEALTH GENC HPOB CHANGE154 6010.56-M NOVEMBER 18, 2015 PUBLICATIONS SYSTEM CHANGE
OFFICE OF THE ASSISTANT SECRETARY OF DEFENSE HEAL TH AFFAIRS 16401 EASTCENTRETE H PARKWAY AURORA, CO 80011 ~9066 DEFENSE HEALTH GENC HPOB CHANGE154 6010.56-M NOVEMBER 18, 2015 PUBLICATIONS SYSTEM CHANGE
Complete Guide for Interventional Radiology
 2015 Complete Guide for Interventional Radiology Contents Introduction... 1 CPT Codes and Descriptions...1 Procedure Codes...2 Chapter 1: The Basics... 5 APC Basics Why Is This Important?...5 CCI Edits
2015 Complete Guide for Interventional Radiology Contents Introduction... 1 CPT Codes and Descriptions...1 Procedure Codes...2 Chapter 1: The Basics... 5 APC Basics Why Is This Important?...5 CCI Edits
