Obstructive sleep apnea (OSA) is a common disorder,
|
|
|
- Benjamin Stone
- 5 years ago
- Views:
Transcription
1 Original Research Upper Airway Stimulation for Obstructive Sleep Apnea: Patient- Reported Outcomes after 48 Months of Follow-up Otolaryngology Head and Neck Surgery 1 7 Ó American Academy of Otolaryngology Head and Neck Surgery Foundation 2017 Reprints and permission: sagepub.com/journalspermissions.nav DOI: / M. Boyd Gillespie, MD, MSc 1, Ryan J. Soose, MD 2, B. Tucker Woodson, MD 3, Kingman P. Strohl, MD 4, Joachim T. Maurer, MD 5, Nico de Vries, MD 6, David L. Steward, MD 7, Jonathan Z. Baskin, MD 4, M. Safwan Badr, MD 8, Ho-sheng Lin, MD 8, Tapan A. Padhya, MD 9, Sam Mickelson, MD 10, W. McDowell Anderson, MD 9, Olivier M. Vanderveken, MD 11, and Patrick J. Strollo Jr, MD 2 ; on Behalf of the STAR Trial Investigators Sponsorships or competing interests that may be relevant to content are disclosed at the end of this article. Abstract Objective. To assess patient-based outcomes of participants in a large cohort study the STAR trial (Stimulation Therapy for Apnea Reduction) 48 months after implantation with an upper airway stimulation system for moderate to severe obstructive sleep apnea. Study Design. A multicenter prospective cohort study. Setting. Industry-supported multicenter academic and clinical setting. Subjects. Participants (n = 91) at 48 months from a cohort of 126 implanted participants. Methods. A total of 126 participants received an implanted upper airway stimulation system in a prospective phase III trial. Patient-reported outcomes at 48 months, including Epworth Sleepiness Scale (ESS), Functional Outcomes of Sleep Questionnaire (FOSQ), and snoring level, were compared with preimplantation baseline. Results. A total of 91 subjects completed the 48-month visit. Daytime sleepiness as measured by ESS was significantly reduced (P =.01), and sleep-related quality of life as measured by FOSQ significantly improved (P =.01) when compared with baseline. Soft to no snoring was reported by 85% of bed partners. Two patients required additional surgery without complication for lead malfunction. Conclusion. Upper airway stimulation maintained a sustained benefit on patient-reported outcomes (ESS, FOSQ, snoring) at 48 months in select patients with moderate to severe obstructive sleep apnea. Keywords obstructive sleep apnea, cranial nerve, hypoglossal nerve, sleep, device, implant, long term, clinical, apnea hypopnea index, sleep, quality of life Received September 14, 2016; revised January 4, 2017; accepted January 11, Obstructive sleep apnea (OSA) is a common disorder, with a prevalence of 5% to 10% of the adult US population. OSA is associated with daytime sleepiness, snoring, poor sleep quality, and an increased risk of cardiovascular disease and motor vehicle accidents. 1,2 Continuous positive airway pressure (CPAP) is the recognized first-line therapy for the majority of patients with OSA; however, many patients reject CPAP therapy due to 1 University of Tennessee, Memphis, Tennessee, USA 2 School of Medicine, University of Pittsburgh, Pittsburgh, PA, USA 3 Medical College of Wisconsin, Milwaukee, Wisconsin, USA 4 Case Western Reserve University, Cleveland, Ohio, USA 5 University Hospital Mannheim, Mannheim, Germany 6 OLVG Hospital and ACTA, Amsterdam, Netherlands 7 University of Cincinnati Medical Center, Cincinnati, Ohio, USA 8 Wayne State University, Detroit, Michigan, USA 9 University of South Florida College of Medicine, Tampa, Florida, USA 10 Advanced ENT, Atlanta, Georgia, USA 11 Antwerp University Hospital, Edegem, Belgium This article was presented at the 2016 AAO-HNSF Annual Meeting & OTO EXPO; September 18-21, 2016; San Diego, California. Corresponding Author: M. Boyd Gillespie, MD, MSc, Department of Otolaryngology-HNS, 910 Madison Avenue, Suite 408, Memphis, TN 38163, USA. mgilles8@uthsc.edu
2 2 Otolaryngology Head and Neck Surgery discomfort, air leaks, failure to improve, nasal blockage, and/or claustrophobia. Low adherence rates of 30% to 60% over time limit the effectiveness of CPAP therapy and result in a significant number of untreated or undertreated patients with symptomatic OSA. 3,4 Upper airway surgery is an option for patients with symptomatic OSA who are unwilling or unable to adhere to CPAP therapy. Traditionally, upper airway surgery has consisted of a variety of procedures that either remove redundant upper airway tissues or statically reposition tissues to enlarge the upper airway lumen. These procedures are often painful with a prolonged recovery period. In addition, traditional surgical approaches frequently fail to address the underlying increased collapsibility of the airway that exists in OSA patients. 5,6 Initial studies of hypoglossal nerve stimulation in animal models of OSA showed significant reduction in airway collapsibility. 7,8 Early feasibility trials in humans of several hypoglossal nerve stimulation devices demonstrated significant reduction in apnea-hypopnea index (AHI) as well as improvements in sleep-related quality of life These encouraging findings led to a large multicenter phase III study of an upper airway stimulation (UAS) system the STAR trial (Stimulation Therapy for Apnea Reduction). The UAS system is a fully implantable system that consists of a stimulation lead attached to the distal hypoglossal nerve, an intercostal sensing lead to detect the respiratory cycle, and a pulse generator on the anterior chest wall inferior to the clavicle. The device is activated by the patient prior to sleep with a handheld remote. The STAR trial demonstrated a significant reduction in the median AHI at 12 months, from 29.3 to 9.0, with twothirds (66%) of the implanted participants considered successful responders to therapy by previously published criteria of surgical success (AHI decrease 50% and overall AHI \20). 12 Given the results of the STAR trial, the US Food and Drug Administration approved upper airway stimulation in 2014 for use in select patients who meet rigorous inclusion criteria, consisting of moderate to severe OSA (AHI, 20-65), failure of CPAP therapy, body mass index (BMI) 32 kg/m 2, and absence of complete circumferential palatal collapse on drug-induced sleep endoscopy. The present work is based on a multicenter longitudinal observational study of the original 126 subjects implanted with a UAS system as part of the STAR trial. The STAR trial protocol required follow-up polysomnograms (PSGs) at 12 and 18 months postimplantation as well as an optional PSG at 36 months, the results of which have been reported. 13 In addition, the protocol called for regularly scheduled office follow-up every 6 months for up to 5 years after implantation to evaluate validated secondary outcome measures of clinical effectiveness namely, the Epworth Sleepiness Scale (ESS), the Functional Outcomes of Sleep Questionnaire (FOSQ), and the Subject and Bed-Partner Snoring Scale and to assess for potential adverse events related to the device. This study presents the results of these secondary outcome measures 48 months following implantation. Methods Study Subjects The STAR trial multicenter cohort included adults with a history of moderate to severe OSA and intolerance or inadequate adherence to CPAP. The trial was approved by the institutional review board (United States) and medical ethics committee (Europe) in each participating center. An independent Clinical Events Committee and a Data Safety Monitoring Board provided review and adjudication of safety data. Key study exclusion criteria included BMI.32 kg/m 2,neuromuscular disease including hypoglossal nerve palsy or injury, severe cardiopulmonary disorders, active psychiatric disease, and comorbid nonrespiratory sleep disorders that would confound functional sleep-related assessments. Participants who met inclusion/exclusion criteria underwent 3 screening tests: an inlaboratory attended PSG, a surgical consultation visit, and a drug-induced sleep endoscopy. Participants were excluded after the PSG for an AHI \20 or.50 events per hour of sleep, for central and/or mixed apnea index.25%oftheahi,orfora nonsupine AHI \10. Participants were excluded if pronounced anatomic abnormalities would prevent effective use of the device (eg, tonsil size, 3 or 4). Drug-induced sleep endoscopy assessed site and pattern of upper airway collapse under sedation (eg, propofol and/or midazolam) and excluded any participants with observed complete concentric collapse at the level of the velopharynx. Study Procedures A total of 126 qualified participants who met preimplant screening criteria underwent device implantation with the Inspire UAS system (Inspire Medical Systems, Maple Grove, Minnesota). Details of the surgical technique are described in a prior publication. 12 The device was activated 1 month after the implant procedure. During the first month of at-home use, participants were encouraged to use the device to acclimate to the sensation and gradually increase the stimulation over a predetermined range to improve snoring and daytime sleepiness. Between 2 and 6 months, 1 in-laboratory PSG titration studies were conducted to determine more precisely the optimal range of stimulation for maximal AHI reduction. Additional titration studies were performed for some participants after 6 months based on previous titration results and participant feedback. Study Data Results have been reported on the primary study outcomes of AHI and oxygen desaturation index based on scheduled follow-up PSGs collected at 12 and 18 months per protocol, with an additional optional PSG at 36 months. The present study focuses on the self-reported patient secondary outcomes collected every 6 months through a total of 48 months to date. Secondary outcome measures include subjective sleepiness and sleep-related quality of life with the validated ESS 14 and the FOSQ. 15 Clinical variables including BMI, neck circumference, tongue function, speech, swallowing, and
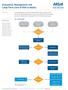
3 Gillespie et al 3 Figure 1. CONSORT diagram. PSG, polysomnography; RCT, randomized controlled trial. daily device use by self-report were measured at scheduled visits to assess for any changes over the course of the study. Subjective report of snoring was collected from participants and bed partners reports with a categorical scale (no snoring, soft snoring, loud snoring, very intense snoring, or bed partner leaves room). All reported adverse events were reviewed and coded by the Clinical Events Committee. Serious adverse events were defined as any events that led to death, life-threatening illness, permanent impairment, or new or prolonged hospitalization. Adverse events were categorized as procedure related if related to the surgical procedure or device related if secondary to use of the device after therapy activation. Adverse events could also be judged as not fitting in either category. Statistical Analysis Ordinal scale data for secondary outcomes (ESS, FOSQ) were tested and found to conform to parametric testing parameters. The paired t test was used to evaluate the difference between study baseline and 48 months at the 5% significance level. Adverse events are reported with descriptive statistics. Results A total of 95 subjects (75%) of the original 126-patient cohort showed for the 48-month follow-up (Figure 1); however, 4 subjects had incomplete data, leaving 91 (73%) for data analysis. At 48 months, 3 subjects in the original cohort had died; 3 had undergone elective explantation of the UAS system; and 25 subjects were lost to follow-up. The cause of death of the 3 subjects has been reported and included a sudden daytime death in a 48-year-old woman with a family history of sudden cardiac death, a 55-year-old man who died from a likely cardiac arrest complicated by a fall down a flight of stairs, and a 64-year-old man who was a victim of a homicide. 13 Three subjects underwent uneventful explantation of the UAS device, including 1 therapy nonresponder who requested removal before moving away from his study site, 1 therapy responder who developed a septic sternoclavicular joint adjacent to the device, and 1 Table 1. Comparison of Patients Who Completed the 48-Month Visit with Those Lost to Follow-up. a 48-mo Visit Measures Completed Not Completed P Value Patients, n Male, n Age, y BMI, kg/m AHI Baseline mo Baseline ESS FOSQ mo nightly use, % Abbreviations: AHI, apnea-hypopnea index; BMI, body mass index; ESS, Epworth Sleepiness Scale; FOSQ, Functional Outcomes of Sleep Questionnaire. a Values presented as mean 6 SD unless noted otherwise. therapy responder who suffered from prolonged insomnia complicated by psychological issues. Of the 25 subjects lost to follow-up, 15 missed the 48- month visit; 5 exited the study; and 5 were from 3 study sites that were subsequently closed. The 5 patients who exited the study decided to leave due to relocation (1 patient), inability/unwillingness to adhere to the study follow-up schedule (2 patients), and unavailability (ie, study personnel were unable to contact patient after numerous attempts; 2 patients). Three study sites were closed owing to loss of principal investigator (1 site with 3 patients) and inability of study site to continue participation (2 sites with 1 patient each). In comparing the 95 patients who completed the 48-month follow-up to the 25 who missed the visit (Table 1), there was no discernable difference between the groups with regard to therapy response at 12 months and reported nightly use at 36 months. Subjects lost to follow-up
4 4 Otolaryngology Head and Neck Surgery Table 2. Comparison of Baseline Characteristics of Initial STAR Cohort and Those Completing 12- and 48-Month Follow-up. a Baseline Characteristics Preimplant (N = 126) 12 mo (n = 124) 48 mo (n = 91) Age, y BMI, kg/m AHI, events/h Abbreviation: AHI, apnea-hypopnea index; BMI, body mass index; STAR, Stimulation Therapy for Apnea Reduction. a Values presented as mean 6 SD. Figure 3. Epworth Sleepiness Scale results over time: normal level of daytime sleepiness 10. Results in mean and standard deviation. Figure 2. Functional Outcome of Sleep (FOSQ) results over time: normal sleep-related quality of life Results in mean and standard deviation. had a trend toward younger age and worse sleep-related quality of life at baseline. Complete data from 91 of the active 95 subjects at 48- month follow-up show that they did not differ significantly from the original study cohort with regard to baseline variables of age, BMI, or AHI (Table 2). With regard to secondary patient-based outcomes, 89 subjects had a median FOSQ score of 18.6 (Q1, 16.2; Q3, 19.6), which demonstrates significantly improved sleep-related quality of life, on average, when compared with baseline ( vs , P =.01).This finding shows that mean levels are significantly improved over baseline, although lower (17.9) than fully normal values (Figure 2). ESS scores (n = 89) at 48 months have a median value of 6 (Q1, 4; Q3, 10) and significantly less daytime sleepiness, on average, when compared with baseline ( vs , P =.01). The average Epworth scores are therefore within the range of normal (10) and likewise stable over time (Figure 3). Snoring improvement was also consistent over time, with 91% of subjects (n = 89) and 85% of bed partners (n = 92) reporting soft or no snoring at 48 months versus only 22% of subjects and 17% of bed partners at baseline (Figure 4). A total of 81% of subjects (75 of 93) self-reported nightly use of the device at 48 months, which remained unchanged since 24-month follow-up (Figure 5). Three patients died within the first 36 months from the events described above. These deaths were fully adjudicated by a data safety monitoring committee and determined to be unrelated to the implant procedure or device. Throughout 48 months of follow-up, there have been a total of 5 serious adverse events (5 of 126, 4.0%). Three patients underwent elective explantation as described above, and 2 required subsequent surgery between 36 and 48 months to replace malfunctioning device components (1 sensing lead due to insulation breach and 1 stimulation lead and implantable pulse generator to reposition the electrode location to improve therapy response). The revision operations were without complications or significant sequelae. Since the 36- month follow-up, nonserious adverse events continue to decrease (Table 3) with fewer cases of discomfort from electrical stimulation or tongue abrasion. There is ongoing intermittent need to adjust stimulation levels to improve function (3 patients) or check the function and reset the parameters of the patient-controlled handheld remote (9 patients). Discussion OSA is a prevalent disorder in the adult population that increases the risk of cardiovascular morbidity and motor vehicle accidents. 1,2 Clinically, OSA patients present with poor sleep quality, loud bothersome snoring, and excessive daytime sleepiness. Upper airway surgery is an option for patients with symptomatic OSA who are unable or unwilling to adhere to CPAP therapy. OSA operations, however, are often only partially effective, are painful, and require prolonged recovery times. The limited effectiveness of sleep surgery may be due to the focus on reducing soft tissue obstruction while failing to address the underlying increase in airway collapsibility caused by reduced neuromuscular tone, which is thought to be the primary pathophysiologic basis for OSA. 5,6 The present study is an ongoing multicenter longitudinal observational study of the original STAR trial subjects 48 months after implantation with an UAS device. A previously published study showed that the primary outcome AHI remained low on average (mean, 6.2) at 36 months. 13 Patient-based outcomes at 36 months demonstrated ongoing
5 Gillespie et al 5 Figure 4. Change in self-reported (A) and bed partner reported (B) snoring intensity over time. Figure 5. Patient-reported nightly use of upper airway stimulation therapy over time. improvement of sleep-related quality of life and daytime sleepiness levels on average, as well as low levels of snoring severity. This study confirms the durability of response in these secondary patient-based outcomes at 48 months, which are essentially unchanged from 36-month follow-up. Four years after implantation, patients have symptoms that are significantly improved over baseline, including normal levels of daytime sleepiness, near normal levels of sleeprelated quality of life, and improved snoring. Although the present results cannot be confirmed by a 48-month PSG, which was not part of the study protocol, the results suggest ongoing subjective effectiveness of therapy with regard to bothersome symptoms of OSA that are clinically relevant to patients. Whereas median FOSQ scores show that greater than half of patients have achieved normal levels of sleep-related quality of life, the mean FOSQ scores, though improved over baseline, remain below the normal range, indicating a level of ongoing symptoms among some patients. A recent case report described a STAR trial participant who had ongoing symptoms of snoring and poor sleep despite a significant reduction in AHI from 43 to 12 after implantation of the UAS system. 16 The patient was successfully fitted with an oral appliance to augment the UAS therapy, with subsequent resolution of symptoms and AHI reduction to 2. This case report highlights that OSA is a chronic disorder that requires ongoing follow-up and adjustment of therapy over time to optimize patient symptoms. The 48-month data additionally confirms the previously noted high level of therapy adherence, with 81% of subjects self-reporting nightly use of the device. After self-reported nightly use of 86% at 1 year, there was a decrease to 81% at year 2, which has been stable to year 4. These data compare well to CPAP, which is the recognized standard-ofcare therapy for moderate to severe OSA. Currently, the Centers of Medicare and Medicaid Services requires documentation of CPAP use a minimum of.4 hours a night for.70% of nights to provide coverage for CPAP service. The.4 hours/night is considered minimal, however, as most clinicians stress that patients should wear CPAP all night, if possible. In a prospective trial of CPAP use among.1000 patients, only 40% of patients were adherent to the minimum.4 hours for.70% of nights after 6 months of follow-up. 17 The recently reported SAVE trial (Sleep Apnea Cardiovascular Endpoints) found that patients with a history of moderate to severe OSA and cardiovascular disease randomized to CPAP had no reduction in cardiovascular deaths when compared with non-cpap controls. 18 One explanation for the negative finding was that the mean duration of CPAP adherence was only 3.3 hours per night in the CPAP group. Although there are potential inaccuracies of selfreported use, it remains notable that 81% of subjects who were previously noncompliant with CPAP now use the UAS system nightly 4 years after implantation. Discrepancy between self-reported CPAP use and objectively measured use by smart card report has been noted, with 60% of CPAP patients reporting nightly use but only 46% meeting criteria for regular use (4 hours night on.70% of nights). 19 Therefore, it is possible that patients who use a device nightly still fall below recommended standards of use. Although previous evidence suggests that some patients experience modest reductions in systolic and diastolic blood pressure with regular use of UAS therapy, there is currently no evidence to demonstrate a reduction in cardiac events
6 6 Otolaryngology Head and Neck Surgery Table 3. Nonserious Adverse Events over 48 Months of STAR Trial. Events, n Adverse Events 0-12 mo mo mo mo Total Participants with Event, % a (n) Procedure-related nonserious adverse events Postoperative discomfort related to incisions (37) Postoperative discomfort independent of incisions (34) Temporary tongue weakness (23) Intubation effects (15) Headache (8) Other postoperative symptoms (14) Mild infection (1) Device-related nonserious adverse events Discomfort due to electrical stimulation (73) Tongue abrasion (33) Dry mouth (16) Mechanical pain associated with presence of the device (12) Temporary internal device usability or functionality complaint (20) Temporary external device usability or functionality complaint (30) Other acute symptoms (30) Mild infection (1) Abbreviation: STAR, Stimulation Therapy for Apnea Reduction. a Percentage based on original cohort of implanted participants, N = 126. with UAS therapy. 20 Since the STAR trial, modified UAS devices have undergone a software update that allows objective measurement of weekly device usage; however, this update was not available in this cohort of patients. In addition to the ongoing improvement in patient symptoms, the study found a low rate of long-term side effects and device-related adverse events. Two patients required reoperation between 36 and 48 months for lead-related failure; therefore, ongoing assessment is needed to get a better estimate of the life expectancy of device components. A total of 25 nonserious adverse events were recorded between 36 and 48 months, which was down from 48 reported events between 24 and 36 months and consistent with an observed decreasing trend overall. This decrease is mainly attributable to reduced reports of tongue discomfort with electrical stimulation, which is likely due to therapy acclimation as well as ongoing programming adjustments to improve comfort. It is possible that patients with 48-month follow-up have fewer side effects and complaints than the cohort lost to follow-up. The main study limitation was the increased number of patients lost to follow-up at 48 months compared with 36 months (25 vs 4). Factors that influence adherence to follow-up include individual patient characteristics, social support, medical staff characteristics, and research study design. 21 The trend of older age for those who completed follow-up versus those lost at 48 months is consistent with other trials that have noted poorer follow-up in younger cohorts, perhaps due to increased demands of work-life balance among younger subjects. 22 With regard to medical staff, loss of a principal investigator and study site support accounted for 20% of follow-up loss at 48 months. This trial, like many other multiyear trials, is experiencing greater loss of follow-up after 3 years. 23 Conclusions UAS therapy demonstrates stable, long-term improvement in patient-reported symptoms of sleepiness, sleep-related quality of life, and snoring among patients with moderate to severe OSA who meet selection criteria. The therapy has high rates of adherence as compared with CPAP, with acceptable rates of adverse events. Ongoing follow-up is required to determine the natural product life of the device components. Author Contributions M. Boyd Gillespie, date collection, interpretation, and writing; Ryan J. Soose, date collection, interpretation, and writing; B. Tucker Woodson, data collection, analysis, interpretation and writing; Kingman P. Strohl, date collection, interpretation, and revising; Joachim T. Maurer, date collection, interpretation, and writing; Nico de Vries, date collection, interpretation, and revising; David L. Steward, date collection, interpretation, and revising; Jonathan Z. Baskin, date collection, interpretation, and revising; M. Safwan Badr, date collection, interpretation, and revising; Ho-sheng Lin, date collection, interpretation, and revising; Tapan A. Padhya, date collection, interpretation, and revising; Sam Mickelson, date collection, interpretation, and revising; W. McDowell Anderson, date collection, interpretation, and revising; Olivier M. Vanderveken, data collection, interpretation, and manuscript revision; Patrick J. Strollo Jr, datecol- lection, interpretation, and revising.
7 Gillespie et al 7 Disclosures Competing interests: M. Boyd Gillespie, Inspire Medical Systems study investigator, consultant, honoraria; ImThera study investigator; Medtronic consultant; Olympus study investigator; Ryan J. Soose, Inspire Medical Systems study investigator, consultant; B. Tucker Woodson, Inspire Medical Systems study investigator, consultant; Medtronic consultant, royalty; Siesta Medical royalty; Cryosa consultant; Kingman P. Strohl, Inspire Medical Systems study investigator, consultant; Joachim T. Maurer, Inspire Medical Systems study investigator, consultant, surgical trainer, invited speaker; GlaxoSmithKline, Weinmann, Olympus, ResMed, Neuwirth, Medtronic, and Heinen & Löwenstein, personal fees outside the submitted work; Nyxoah consultant; ReVent, ImThera invited speaker; Nico de Vries, Inspire Medical Systems study investigator, consultant; Philips, Olympus consultant; Night Balance/ReVent medical advisor, shareholder, funding from company; Patent devices, systems, and methods for monitoring sleep position; David L. Steward, Inspire Medical Systems study investigator, consultant; Jonathan Z. Baskin, Inspire Medical Systems study investigator; M. Safwan Badr, Inspire Medical Systems study investigator, consultant; Ho-sheng Lin, Inspire Medical Systems study investigator, consultant; Intuitive Surgical consultant; Chechpoint Surgical consultant; Tapan A. Padhya, Inspire Medical Systems study investigator, consultant; Sam Mickelson, Inspire Medical Systems study investigator, consultant; ImThera Medical research funding; W. McDowell Anderson, Inspire Medical Systems study investigator; Olivier M. Vanderveken, Inspire Medical study investigator; SomnoMed research grant; Nyxoah research grant, consultant; ReVent research grant; Philips Electronics BV consultant; Patrick J. Strollo Jr, Inspire Medical Systems study investigator, consultant; ResMed scientific advisory board, research grant; Philips Respironics research grant; Itamar consultant. Sponsorships: Inspire Medical Systems, study design and conduct; collection of the data; approval of the manuscript. Funding source: Inspire Medical Systems, study design and conduct; collection of the data; approval of the manuscript. References 1. Young T, Palta M, Dempsey J, et al. Burden of sleep apnea: rationale, design, and major findings of the Wisconsin Sleep Cohort Study. WMJ. 2009;108: Shahar E, Whitney CW, Redline S, et al. Sleep-disordered breathing and cardiovascular disease: cross-sectional results of the Sleep Heart Health Study. Am J Respir Crit Care Med. 2001;163: Engleman HM, Wild MR. Improving CPAP use by patients with the sleep apnoea/hypopnea syndrome (SAHS). Sleep Med Rev. 2003;7: Weaver TE, Sawyer AM. Adherence to continuous positive airway pressure treatment for obstructive sleep apnoea: implications for future interventions. Indian J Med Res. 2010;131: McGinley BM, Schwartz AR, Schneider H, et al. Upper airway neuromuscular compensation during sleep is defective in obstructive sleep apnea. J Appl Physiol. 2008;105: Patil SP, Schneider H, Marx JJ, et al. Neuromechanical control of upper airway patency during sleep. J Appl Physiol. 2007; 102: Yoo PB, Durand DM. Effects of selective hypoglossal nerve stimulation on canine upper airway mechanics. J Appl Physiol. 2005;99: Bishara H, Odeh M, Schnall RP, et al. Electrically-activated dilator muscles reduce pharyngeal resistance in anaesthetized dogs with upper airway obstruction. EurRespirJ. 1995;8: Van de Heyning PH, Badr MS, Baskin JZ, et al. Implanted upper airway stimulation device for obstructive sleep apnea. Laryngoscope. 2012;122: Kezirian EJ, Goding GS, Malhotra A, et al. Hypoglossal nerve stimulation improves obstructive sleep apnea: 12 month outcomes. J Sleep Res. 2014;23: Friedman M, Jacobowitz O, Hwang MS, et al. Targeted hypoglossal nerve stimulation for the treatment of obstructive sleep apnea: six-month results. Laryngoscope. 2016;126: Strollo PJ, Soose RJ, Maurer JT, et al. Upper airway stimulation for obstructive sleep apnea. N Engl J Med. 2014;370: Woodson BT, Soose RJ, Gillespie MB, et al. Three-year outcomes of cranial nerve stimulation for obstructive sleep apnea: the STAR trial. Otolaryngol Head Neck Surg. 2016;154: Johns MW. A new method for measuring daytime sleepiness: the Epworth Sleepiness Scale. Sleep. 1991;14: Weaver TE, Laizner AM, Evans LK, et al. An instrument to measure functional status outcomes for disorders of excessive sleepiness. Sleep. 1997;20: Lee JJ, Sahu N, Rogers R, Soose RJ. Severe obstructive sleep apnea treated with combination hypoglossal nerve stimulation and oral appliance therapy. J Dental Sleep Med. 2015;2: Kushida CA, Nichols DA, Holmes TH, et al. Effects of continuous positive airway pressure on neurocognitive function in obstructive sleep apnea patients: the Apnea Positive Pressure Long-term Efficacy Study (APPLES). Sleep. 2012;35: McEvoy RD, Antic NA, Heeley E, et al. CPAP for prevention of cardiovascular events in obstructive sleep apnea. N Engl J Med. 2016;375: Kribbs NB, Pack AI, Kline LR, et al. Objective measurement patterns of nasal CPAP use by patients with obstructive sleep apnea. Am Rev Repir Dis. 1993;147: Woodson BT, Gillespie MB, Soose RJ, et al. Randomized controlled withdrawal study of upper airway stimulation on OSA: short- and long-term effect. Otolaryngol Head Neck Surg. 2014;151: Wu X, Lin H. Patient adherence and follow-up in clinical research: a systematic review of measurements, associated factors, and intervention strategies. J Clin Res Ophthalmol. 2015;2: Silveira MPT, Guttier MC, Moreira LB, et al. Predictors of non-adherence to clinical follow-up among patients participating in a randomized trial of pharmaceutical care intervention in HIV-positive adults in southern Brazil. AIDS Behav. 2014;18: S85-S Somerson JS, Bartush KC, Shroff JB, et al. Loss of follow-up in orthopaedic clinical trials: a systematic review. Int Orthop. 2016;40:
Upper Airway Stimulation for Obstructive Sleep Apnea: 5-Year Outcomes
 Original Research Upper Airway Stimulation for Obstructive Sleep Apnea: 5-Year Outcomes Otolaryngology Head and Neck Surgery 1 9 Ó American Academy of Otolaryngology Head and Neck Surgery Foundation 2018
Original Research Upper Airway Stimulation for Obstructive Sleep Apnea: 5-Year Outcomes Otolaryngology Head and Neck Surgery 1 9 Ó American Academy of Otolaryngology Head and Neck Surgery Foundation 2018
Upper Airway Stimulation for Obstructive Sleep Apnea
 Upper Airway Stimulation for Obstructive Sleep Apnea Background, Mechanism and Clinical Data Overview Seth Hollen RPSGT 21 May 2016 1 Conflicts of Interest Therapy Support Specialist, Inspire Medical Systems
Upper Airway Stimulation for Obstructive Sleep Apnea Background, Mechanism and Clinical Data Overview Seth Hollen RPSGT 21 May 2016 1 Conflicts of Interest Therapy Support Specialist, Inspire Medical Systems
Inspire Therapy for Sleep Apnea
 Inspire Therapy for Sleep Apnea Patient Guide Giving You the FREEDOM TO SLEEP Like Everyone Else Take Comfort. Inspire therapy can help. Inspire therapy is a breakthrough implantable treatment option for
Inspire Therapy for Sleep Apnea Patient Guide Giving You the FREEDOM TO SLEEP Like Everyone Else Take Comfort. Inspire therapy can help. Inspire therapy is a breakthrough implantable treatment option for
Upper Airway Muscle Stimulation for Obstructive Sleep Apnea
 Upper Airway Muscle Stimulation for Obstructive Sleep Apnea M. Safwan Badr, MD, MBA Chair, Department of Medicine, Wayne State University School of Medicine. Staff Physician, John D. Dingell VA Medical
Upper Airway Muscle Stimulation for Obstructive Sleep Apnea M. Safwan Badr, MD, MBA Chair, Department of Medicine, Wayne State University School of Medicine. Staff Physician, John D. Dingell VA Medical
Comparing Upper Airway Stimulation to Expansion Sphincter Pharyngoplasty: A Single University Experience
 771395AORXXX10.1177/0003489418771395Annals of Otology, Rhinology & LaryngologyHuntley et al research-article2018 Original Article Comparing Upper Airway Stimulation to Expansion Sphincter Pharyngoplasty:
771395AORXXX10.1177/0003489418771395Annals of Otology, Rhinology & LaryngologyHuntley et al research-article2018 Original Article Comparing Upper Airway Stimulation to Expansion Sphincter Pharyngoplasty:
Microsurgical Anatomy of the Terminal Hypoglossal Nerve Relevant for Neurostimulation in Obstructive Sleep Apnea.
 Neuromodulation. 05 Dec;8(8):7-8. doi: 0./ner.347. Epub 05 Sep 6. Microsurgical Anatomy of the Terminal Hypoglossal Nerve Relevant for Neurostimulation in Obstructive Sleep Apnea. Bassiri Gharb B, Tadisina
Neuromodulation. 05 Dec;8(8):7-8. doi: 0./ner.347. Epub 05 Sep 6. Microsurgical Anatomy of the Terminal Hypoglossal Nerve Relevant for Neurostimulation in Obstructive Sleep Apnea. Bassiri Gharb B, Tadisina
Inspire. therapy for sleep apnea. Giving you the freedom to sleep like everyone else
 Inspire therapy for sleep apnea Giving you the freedom to sleep like everyone else Take Comfort. Take Action. Inspire therapy can help. Here are some reasons people like you have chosen Inspire therapy
Inspire therapy for sleep apnea Giving you the freedom to sleep like everyone else Take Comfort. Take Action. Inspire therapy can help. Here are some reasons people like you have chosen Inspire therapy
Upper Airway Stimulation for Obstructive Sleep Apnea: Results from the ADHERE Registry
 Original Research Upper Airway Stimulation for Obstructive Sleep Apnea: Results from the ADHERE Registry Otolaryngology Head and Neck Surgery 1 7 Ó American Academy of Otolaryngology Head and Neck Surgery
Original Research Upper Airway Stimulation for Obstructive Sleep Apnea: Results from the ADHERE Registry Otolaryngology Head and Neck Surgery 1 7 Ó American Academy of Otolaryngology Head and Neck Surgery
Inspire Therapy for Obstructive Sleep Apnea. Clinical Data Update
 Inspire Therapy for Obstructive Sleep Apnea For OSA patients unable to tolerate or get consistent benefit from CPAP Clinical Data Update Summary of published long-term 3-year outcomes data Treatment Goals
Inspire Therapy for Obstructive Sleep Apnea For OSA patients unable to tolerate or get consistent benefit from CPAP Clinical Data Update Summary of published long-term 3-year outcomes data Treatment Goals
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of hypoglossal nerve stimulation for moderate to severe obstructive sleep apnoea
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of hypoglossal nerve stimulation for moderate to severe obstructive sleep apnoea
A New, Clinically Proven Sleep Apnea Therapy for people unable to use CPAP.
 A New, Clinically Proven Sleep Apnea Therapy for people unable to use CPAP. Take Heart. If You Have OSA, You re Not Alone. Like you, more than 18 million Americans are estimated to have Obstructive Sleep
A New, Clinically Proven Sleep Apnea Therapy for people unable to use CPAP. Take Heart. If You Have OSA, You re Not Alone. Like you, more than 18 million Americans are estimated to have Obstructive Sleep
Inspire Therapy for Sleep Apnea
 Inspire Therapy for Sleep Apnea Patient Guide Giving You the FREEDOM TO SLEEP Like Everyone Else Take Comfort. Inspire therapy can help. Inspire therapy is a breakthrough implantable treatment option for
Inspire Therapy for Sleep Apnea Patient Guide Giving You the FREEDOM TO SLEEP Like Everyone Else Take Comfort. Inspire therapy can help. Inspire therapy is a breakthrough implantable treatment option for
CLINICAL MEDICAL POLICY
 Policy Name: Policy Number: Responsible Department(s): CLINICAL MEDICAL POLICY Hypoglossal Nerve Stimulation in the Treatment of Obstructive Sleep Apnea MP-079-MD-DE Medical Management Provider Notice
Policy Name: Policy Number: Responsible Department(s): CLINICAL MEDICAL POLICY Hypoglossal Nerve Stimulation in the Treatment of Obstructive Sleep Apnea MP-079-MD-DE Medical Management Provider Notice
Inspire Therapy for Sleep Apnea
 Inspire Therapy for Sleep Apnea Take Heart. If you have OSA, you re not alone. More than 18 million Americans are estimated to have Obstructive Sleep Apnea (OSA). OSA occurs when the tongue and other soft
Inspire Therapy for Sleep Apnea Take Heart. If you have OSA, you re not alone. More than 18 million Americans are estimated to have Obstructive Sleep Apnea (OSA). OSA occurs when the tongue and other soft
Selective upper airway stimulation for obstructive sleep apnea: a single center clinical experience
 DOI 10.1007/s00405-016-4297-6 MISCELLANEOUS Selective upper airway stimulation for obstructive sleep apnea: a single center clinical experience Clemens Heiser 1 Andreas Knopf 1 Murat Bas 1 Constanze Gahleitner
DOI 10.1007/s00405-016-4297-6 MISCELLANEOUS Selective upper airway stimulation for obstructive sleep apnea: a single center clinical experience Clemens Heiser 1 Andreas Knopf 1 Murat Bas 1 Constanze Gahleitner
Hypoglossal Nerve Stimulation: Effective Longterm Therapy for Obstructive Sleep Apnea
 AANA Journal Course Hypoglossal Nerve Stimulation: Effective Longterm Therapy for Obstructive Sleep Apnea Karen J. Maresch, MSN, CRNA Obstructive sleep apnea (OSA) is a worldwide health problem. Historically,
AANA Journal Course Hypoglossal Nerve Stimulation: Effective Longterm Therapy for Obstructive Sleep Apnea Karen J. Maresch, MSN, CRNA Obstructive sleep apnea (OSA) is a worldwide health problem. Historically,
Neuromuscular Stimulation for Sleep Apnea. Overview 1/24/2013. Disclosures. Midline Glossectomy. Palate + Hypopharyngeal Surgery: BMI
 Neuromuscular Stimulation for Sleep Apnea Disclosures The following personal financial relationships with commercial interests relevant to this presentation: Eric J. Kezirian, MD, MPH Director, Division
Neuromuscular Stimulation for Sleep Apnea Disclosures The following personal financial relationships with commercial interests relevant to this presentation: Eric J. Kezirian, MD, MPH Director, Division
Obstructive sleep apnea (OSA) is a common disorder
 Original Research Sleep Medicine and Surgery Drug-Induced Sedation Endoscopy in the Evaluation of OSA Patients with Incomplete Oral Appliance Therapy Response Otolaryngology Head and Neck Surgery 2015,
Original Research Sleep Medicine and Surgery Drug-Induced Sedation Endoscopy in the Evaluation of OSA Patients with Incomplete Oral Appliance Therapy Response Otolaryngology Head and Neck Surgery 2015,
11/19/2012 ก! " Varies 5-86% in men 2-57% in women. Thailand 26.4% (Neruntarut et al, Sleep Breath (2011) 15: )
 Snoring ก Respiratory sound generated in the upper airway during sleep that typically occurs during inspiration but may occur during expiration ICSD-2, 2005..... ก ก! Prevalence of snoring Varies 5-86%
Snoring ก Respiratory sound generated in the upper airway during sleep that typically occurs during inspiration but may occur during expiration ICSD-2, 2005..... ก ก! Prevalence of snoring Varies 5-86%
OBSTRUCTIVE SLEEP APNEA and WORK Treatment Update
 OBSTRUCTIVE SLEEP APNEA and WORK Treatment Update David Claman, MD Professor of Medicine Director, UCSF Sleep Disorders Center 415-885-7886 Disclosures: None Chronic Sleep Deprivation (0 v 4 v 6 v 8 hrs)
OBSTRUCTIVE SLEEP APNEA and WORK Treatment Update David Claman, MD Professor of Medicine Director, UCSF Sleep Disorders Center 415-885-7886 Disclosures: None Chronic Sleep Deprivation (0 v 4 v 6 v 8 hrs)
Tolerance of Positive Airway Pressure following Site-Specific Surgery of Upper Airway
 34 The Open Sleep Journal, 2008, 1, 34-39 Open Access Tolerance of Positive Airway Pressure following Site-Specific Surgery of Upper Airway Ho-Sheng Lin *,#,1,2, Roger Toma #,2, Cara Glavin 2, Mark Toma
34 The Open Sleep Journal, 2008, 1, 34-39 Open Access Tolerance of Positive Airway Pressure following Site-Specific Surgery of Upper Airway Ho-Sheng Lin *,#,1,2, Roger Toma #,2, Cara Glavin 2, Mark Toma
Upper Airway Stimulation for Obstructive Sleep Apnea: Self-Reported Outcomes at 24 Months
 pii: jc-00477-14 http://dx.doi.org/10.5664/jcsm.5390 SCIENTIFIC INVESTIGATIONS Upper Airway Stimulation for Obstructive Sleep Apnea: Self-Reported Outcomes at 24 Months Ryan J. Soose, MD 1 ; B. Tucker
pii: jc-00477-14 http://dx.doi.org/10.5664/jcsm.5390 SCIENTIFIC INVESTIGATIONS Upper Airway Stimulation for Obstructive Sleep Apnea: Self-Reported Outcomes at 24 Months Ryan J. Soose, MD 1 ; B. Tucker
Inspire Therapy for Sleep Apnea
 Inspire Therapy for Sleep Apnea Patient Guide Giving You the FREEDOM TO SLEEP Like Everyone Else Take Comfort. Inspire therapy can help. Inspire therapy is a breakthrough implantable treatment option for
Inspire Therapy for Sleep Apnea Patient Guide Giving You the FREEDOM TO SLEEP Like Everyone Else Take Comfort. Inspire therapy can help. Inspire therapy is a breakthrough implantable treatment option for
11/13/2017. Jeremy Tabak MD, FAASM Medical Director Baptist Hospital Sleep Lab Medical Director Baptist Sleep Lab at Galloway
 Jeremy Tabak MD, FAASM Medical Director Baptist Hospital Sleep Lab Medical Director Baptist Sleep Lab at Galloway HypnoLaus study: OSA effect on mortality US Preventive Services Task Force recommendations
Jeremy Tabak MD, FAASM Medical Director Baptist Hospital Sleep Lab Medical Director Baptist Sleep Lab at Galloway HypnoLaus study: OSA effect on mortality US Preventive Services Task Force recommendations
THN. Sleep Therapy Study. ImThera. Information for Participants. Caution: Investigational device. Limited by United States law to investigational use.
 THN Sleep Therapy Study Information for Participants Caution: Investigational device. Limited by United States law to investigational use. ImThera Obstructive sleep apnea (OSA) is a very serious condition.
THN Sleep Therapy Study Information for Participants Caution: Investigational device. Limited by United States law to investigational use. ImThera Obstructive sleep apnea (OSA) is a very serious condition.
RESEARCH PACKET DENTAL SLEEP MEDICINE
 RESEARCH PACKET DENTAL SLEEP MEDICINE American Academy of Dental Sleep Medicine Dental Sleep Medicine Research Packet Page 1 Table of Contents Research: Oral Appliance Therapy vs. Continuous Positive Airway
RESEARCH PACKET DENTAL SLEEP MEDICINE American Academy of Dental Sleep Medicine Dental Sleep Medicine Research Packet Page 1 Table of Contents Research: Oral Appliance Therapy vs. Continuous Positive Airway
FEP Medical Policy Manual
 FEP Medical Policy Manual Effective Date: January 15, 2018 Related Policies: 2.01.18 Diagnosis and Medical Management of Obstructive Sleep Apnea Surgical Treatment of Snoring and Obstructive Sleep Apnea
FEP Medical Policy Manual Effective Date: January 15, 2018 Related Policies: 2.01.18 Diagnosis and Medical Management of Obstructive Sleep Apnea Surgical Treatment of Snoring and Obstructive Sleep Apnea
Upper-Airway Stimulation for Obstructive Sleep Apnea
 original article Upper-Airway Stimulation for Obstructive Sleep Apnea Patrick J. Strollo, Jr., M.D., Ryan J. Soose, M.D., Joachim T. Maurer, M.D., Nico de Vries, M.D., Jason Cornelius, M.D., Oleg Froymovich,
original article Upper-Airway Stimulation for Obstructive Sleep Apnea Patrick J. Strollo, Jr., M.D., Ryan J. Soose, M.D., Joachim T. Maurer, M.D., Nico de Vries, M.D., Jason Cornelius, M.D., Oleg Froymovich,
Sleep Apnea Treatment Options, A Year After ASV
 Sleep Apnea Treatment Options, A Year After ASV Nancy Collop, MD, FAASM Emory University Associated Professional Sleep Societies, LLC 1 Conflict of Interest Disclosures for Speakers x 1. I do not have
Sleep Apnea Treatment Options, A Year After ASV Nancy Collop, MD, FAASM Emory University Associated Professional Sleep Societies, LLC 1 Conflict of Interest Disclosures for Speakers x 1. I do not have
Management Issues in Hypoglossal Stimulation for OSA. A Sleep Medicine view of surgery
 Management Issues in Hypoglossal Stimulation for OSA Kingman P Strohl M.D. Professor of Medicine, Physiology & Biophysicis, and Oncology Center for sleep Disorders Reseach Case Western Reserve University,
Management Issues in Hypoglossal Stimulation for OSA Kingman P Strohl M.D. Professor of Medicine, Physiology & Biophysicis, and Oncology Center for sleep Disorders Reseach Case Western Reserve University,
Sleep Medicine. Paul Fredrickson, MD Director. Mayo Sleep Center Jacksonville, Florida.
 Sleep Medicine Paul Fredrickson, MD Director Mayo Sleep Center Jacksonville, Florida Fredrickson.Paul@mayo.edu DISCLOSURES No relevant conflicts to report. Obstructive Sleep Apnea The most common sleep
Sleep Medicine Paul Fredrickson, MD Director Mayo Sleep Center Jacksonville, Florida Fredrickson.Paul@mayo.edu DISCLOSURES No relevant conflicts to report. Obstructive Sleep Apnea The most common sleep
Diagnostic Accuracy of the Multivariable Apnea Prediction (MAP) Index as a Screening Tool for Obstructive Sleep Apnea
 Original Article Diagnostic Accuracy of the Multivariable Apnea Prediction (MAP) Index as a Screening Tool for Obstructive Sleep Apnea Ahmad Khajeh-Mehrizi 1,2 and Omid Aminian 1 1. Occupational Sleep
Original Article Diagnostic Accuracy of the Multivariable Apnea Prediction (MAP) Index as a Screening Tool for Obstructive Sleep Apnea Ahmad Khajeh-Mehrizi 1,2 and Omid Aminian 1 1. Occupational Sleep
Management of OSA. saurabh maji
 Management of OSA saurabh maji INTRODUCTION Obstructive sleep apnea is a major public health problem Prevalence of OSAS in INDIA is 2.4% to 4.96% in men and 1% to 2 % in women In the rest of the world
Management of OSA saurabh maji INTRODUCTION Obstructive sleep apnea is a major public health problem Prevalence of OSAS in INDIA is 2.4% to 4.96% in men and 1% to 2 % in women In the rest of the world
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: December 2016 Effective Date: January 15, 2017 Related Policies 2.01.18 Diagnosis and Medical Management of Obstructive Sleep Apnea Syndrome Surgical Treatment of
FEP Medical Policy Manual Last Review: December 2016 Effective Date: January 15, 2017 Related Policies 2.01.18 Diagnosis and Medical Management of Obstructive Sleep Apnea Syndrome Surgical Treatment of
Evaluation, Management and Long-Term Care of OSA in Adults
 Evaluation, Management and Long-Term Care of OSA in Adults AUGUST 2015 Providing diagnostic tools and therapies that are evidence-based is a key part of a successful sleep practice. This resource outlines
Evaluation, Management and Long-Term Care of OSA in Adults AUGUST 2015 Providing diagnostic tools and therapies that are evidence-based is a key part of a successful sleep practice. This resource outlines
Effect of upper-airway stimulation for obstructive sleep apnoea on airway dimensions
 ORIGINAL ARTICLE SLEEP Effect of upper-airway stimulation for obstructive sleep apnoea on airway dimensions Faiza Safiruddin 1, Olivier M. Vanderveken 2,3, Nico de Vries 1,2,3, Joachim T. Maurer 4, Kent
ORIGINAL ARTICLE SLEEP Effect of upper-airway stimulation for obstructive sleep apnoea on airway dimensions Faiza Safiruddin 1, Olivier M. Vanderveken 2,3, Nico de Vries 1,2,3, Joachim T. Maurer 4, Kent
Christopher D. Turnbull 1,2, Daniel J. Bratton 3, Sonya E. Craig 1, Malcolm Kohler 3, John R. Stradling 1,2. Original Article
 Original Article In patients with minimally symptomatic OSA can baseline characteristics and early patterns of CPAP usage predict those who are likely to be longer-term users of CPAP Christopher D. Turnbull
Original Article In patients with minimally symptomatic OSA can baseline characteristics and early patterns of CPAP usage predict those who are likely to be longer-term users of CPAP Christopher D. Turnbull
Outcome Measures in OSA Defining Our Treatment Goal. Defining common outcome metrics in OSA Al-Shawwa Sleep Med Rev 2008
 Outcome Measures in OSA Defining Our Treatment Goal Disclosures Apnicure Minor stock holder sleep apnea device Siesta Medical Minor stock holder sleep apnea device Patent Pending 61/624,105 Sinus diagnostics
Outcome Measures in OSA Defining Our Treatment Goal Disclosures Apnicure Minor stock holder sleep apnea device Siesta Medical Minor stock holder sleep apnea device Patent Pending 61/624,105 Sinus diagnostics
Corporate Medical Policy
 Corporate Medical Policy PALATOPHARYNGOPLASTY/UVULOPALATOPHARYGOPLASTY Description of Procedure or Service Palatopharyngoplasty refers to several surgical approaches for management of the upper airway,
Corporate Medical Policy PALATOPHARYNGOPLASTY/UVULOPALATOPHARYGOPLASTY Description of Procedure or Service Palatopharyngoplasty refers to several surgical approaches for management of the upper airway,
Management of OSA in the Acute Care Environment. Robert S. Campbell, RRT FAARC HRC, Philips Healthcare May, 2018
 Management of OSA in the Acute Care Environment Robert S. Campbell, RRT FAARC HRC, Philips Healthcare May, 2018 1 Learning Objectives Upon completion, the participant should be able to: Understand pathology
Management of OSA in the Acute Care Environment Robert S. Campbell, RRT FAARC HRC, Philips Healthcare May, 2018 1 Learning Objectives Upon completion, the participant should be able to: Understand pathology
Polysomnography (PSG) (Sleep Studies), Sleep Center
 Policy Number: 1036 Policy History Approve Date: 07/09/2015 Effective Date: 07/09/2015 Preauthorization All Plans Benefit plans vary in coverage and some plans may not provide coverage for certain service(s)
Policy Number: 1036 Policy History Approve Date: 07/09/2015 Effective Date: 07/09/2015 Preauthorization All Plans Benefit plans vary in coverage and some plans may not provide coverage for certain service(s)
Effect of two types of mandibular advancement splints on snoring and obstructive sleep apnoea
 European Journal of Orthodontics 20 (1998) 293 297 1998 European Orthodontic Society Effect of two types of mandibular advancement splints on snoring and obstructive sleep apnoea J. Lamont*, D. R. Baldwin**,
European Journal of Orthodontics 20 (1998) 293 297 1998 European Orthodontic Society Effect of two types of mandibular advancement splints on snoring and obstructive sleep apnoea J. Lamont*, D. R. Baldwin**,
Hypoglossal Nerve Stimulator Surgery for treatment of OSA. Disclosures. Hypoglossal Nerve Stimulation 11/9/2016
 Hypoglossal Nerve Stimulator Surgery for treatment of OSA olando Molina MD South Florida ENT Associates A Disclosures I do not have any relevant financial disclosures at this time. Hypoglossal Nerve Stimulation
Hypoglossal Nerve Stimulator Surgery for treatment of OSA olando Molina MD South Florida ENT Associates A Disclosures I do not have any relevant financial disclosures at this time. Hypoglossal Nerve Stimulation
GOALS. Obstructive Sleep Apnea and Cardiovascular Disease (OVERVIEW) FINANCIAL DISCLOSURE 2/1/2017
 Obstructive Sleep Apnea and Cardiovascular Disease (OVERVIEW) 19th Annual Topics in Cardiovascular Care Steven Khov, DO, FAAP Pulmonary Associates of Lancaster, Ltd February 3, 2017 skhov2@lghealth.org
Obstructive Sleep Apnea and Cardiovascular Disease (OVERVIEW) 19th Annual Topics in Cardiovascular Care Steven Khov, DO, FAAP Pulmonary Associates of Lancaster, Ltd February 3, 2017 skhov2@lghealth.org
Surgical Options for the Successful Treatment of Obstructive Sleep Apnea
 Surgical Options for the Successful Treatment of Obstructive Sleep Apnea Benjamin J. Teitelbaum, MD, FACS Otolaryngology Head and Neck Surgery Saint Agnes Medical Center Fresno, California Terms Apnea
Surgical Options for the Successful Treatment of Obstructive Sleep Apnea Benjamin J. Teitelbaum, MD, FACS Otolaryngology Head and Neck Surgery Saint Agnes Medical Center Fresno, California Terms Apnea
Underdiagnosis of Sleep Apnea Syndrome in U.S. Communities
 ORIGINAL ARTICLE Underdiagnosis of Sleep Apnea Syndrome in U.S. Communities Vishesh Kapur, M.D., 1 Kingman P. Strohl, M.D., 2 Susan Redline, M.D., M.P.H., 3 Conrad Iber, M.D., 4 George O Connor, M.D.,
ORIGINAL ARTICLE Underdiagnosis of Sleep Apnea Syndrome in U.S. Communities Vishesh Kapur, M.D., 1 Kingman P. Strohl, M.D., 2 Susan Redline, M.D., M.P.H., 3 Conrad Iber, M.D., 4 George O Connor, M.D.,
Goals Session Putting it all Together
 Goals Session Putting it all Together Adherence to therapy Case presentations Treatment issues Charles Atwood, MD, FCCP, FAASM University of Pittsburgh Disclosures Advisory - Carecore National; Philips;
Goals Session Putting it all Together Adherence to therapy Case presentations Treatment issues Charles Atwood, MD, FCCP, FAASM University of Pittsburgh Disclosures Advisory - Carecore National; Philips;
Tired of being tired?
 Tired of being tired? Narval CC MRD ResMed.com/Narval Sleepiness and snoring are possible symptoms of sleep apnea. Did you know that one in every four adults has some form of sleep disordered-breathing
Tired of being tired? Narval CC MRD ResMed.com/Narval Sleepiness and snoring are possible symptoms of sleep apnea. Did you know that one in every four adults has some form of sleep disordered-breathing
Edward M. Weaver, MD, MPH. University of Washington VA Puget Sound
 What is the Role of Soft Palate Surgery in OSA? Edward M. Weaver, MD, MPH University of Washington Harborview Medical Center VA Puget Sound Question: Should we do UPPP? Answer: Yes Role of Palate Surgery
What is the Role of Soft Palate Surgery in OSA? Edward M. Weaver, MD, MPH University of Washington Harborview Medical Center VA Puget Sound Question: Should we do UPPP? Answer: Yes Role of Palate Surgery
What is the Role of Soft Palate Surgery in OSA?
 What is the Role of Soft Palate Surgery in OSA? Edward M. Weaver, MD, MPH Seattle VA Medical Center University of Washington Harborview Medical Center Acknowledgments This material is the result of work
What is the Role of Soft Palate Surgery in OSA? Edward M. Weaver, MD, MPH Seattle VA Medical Center University of Washington Harborview Medical Center Acknowledgments This material is the result of work
Outcomes of Upper Airway Surgery in Obstructive Sleep Apnea
 Original Research Outcomes of Upper Airway Surgery in Obstructive Sleep Apnea Hadiseh Hosseiny 1, Nafiseh Naeimabadi 1, Arezu Najafi 1 *, Reihaneh Heidari 1, Khosro Sadeghniiat-Haghighi 1 1. Occupational
Original Research Outcomes of Upper Airway Surgery in Obstructive Sleep Apnea Hadiseh Hosseiny 1, Nafiseh Naeimabadi 1, Arezu Najafi 1 *, Reihaneh Heidari 1, Khosro Sadeghniiat-Haghighi 1 1. Occupational
Sonographic evaluation of tongue motions during upper airway stimulation for obstructive sleep apnea a pilotstudy
 DOI 10.1007/s11325-016-1383-3 SLEEP BREATHING PHYSIOLOGY AND DISORDERS ORIGINAL ARTICLE Sonographic evaluation of tongue motions during upper airway stimulation for obstructive sleep apnea a pilotstudy
DOI 10.1007/s11325-016-1383-3 SLEEP BREATHING PHYSIOLOGY AND DISORDERS ORIGINAL ARTICLE Sonographic evaluation of tongue motions during upper airway stimulation for obstructive sleep apnea a pilotstudy
OBSTRUCTIVE SLEEP APNEA and WORK Treatment Update
 OBSTRUCTIVE SLEEP APNEA and WORK Treatment Update David Claman, MD Professor of Medicine Director, UCSF Sleep Disorders Center 415-885-7886 Disclosres: None Chronic Sleep Deprivation (0 v 4 v 6 v 8 hrs)
OBSTRUCTIVE SLEEP APNEA and WORK Treatment Update David Claman, MD Professor of Medicine Director, UCSF Sleep Disorders Center 415-885-7886 Disclosres: None Chronic Sleep Deprivation (0 v 4 v 6 v 8 hrs)
CPAP The Treatment of Choice for Patients with OSA
 CPAP The Treatment of Choice for Patients with OSA Samuel T. Kuna, M.D. Department of Medicine Center for Sleep and Respiratory Neurobiology University of Pennsylvania Pulmonary, Critical Care & Sleep
CPAP The Treatment of Choice for Patients with OSA Samuel T. Kuna, M.D. Department of Medicine Center for Sleep and Respiratory Neurobiology University of Pennsylvania Pulmonary, Critical Care & Sleep
International Journal of Scientific & Engineering Research Volume 9, Issue 1, January ISSN
 International Journal of Scientific & Engineering Research Volume 9, Issue 1, January-2018 342 The difference of sleep quality between 2-channel ambulatory monitor and diagnostic polysomnography Tengchin
International Journal of Scientific & Engineering Research Volume 9, Issue 1, January-2018 342 The difference of sleep quality between 2-channel ambulatory monitor and diagnostic polysomnography Tengchin
Prefabricated Oral Appliances for Obstructive Sleep Apnea
 Medical Policy Manual Allied Health, Policy No. 36 Prefabricated Oral Appliances for Obstructive Sleep Apnea Next Review: May 2019 Last Review: April 2018 Effective: May 1, 2018 IMPORTANT REMINDER Medical
Medical Policy Manual Allied Health, Policy No. 36 Prefabricated Oral Appliances for Obstructive Sleep Apnea Next Review: May 2019 Last Review: April 2018 Effective: May 1, 2018 IMPORTANT REMINDER Medical
Sleep Disordered Breathing
 Sleep Disordered Breathing SDB SDB Is an Umbrella Term for Many Disorders characterized by a lack of drive to breathe Results n repetitive pauses in breathing with no effort Occurs for a minimum of 10
Sleep Disordered Breathing SDB SDB Is an Umbrella Term for Many Disorders characterized by a lack of drive to breathe Results n repetitive pauses in breathing with no effort Occurs for a minimum of 10
Oral Appliances and their Clinical Applications
 Oral Appliances and their Clinical Applications Peter Cistulli MBBS, PhD, MBA, FRACP Professor of Respiratory Medicine & Head of Discipline of Sleep Medicine University of Sydney Director, Centre for Sleep
Oral Appliances and their Clinical Applications Peter Cistulli MBBS, PhD, MBA, FRACP Professor of Respiratory Medicine & Head of Discipline of Sleep Medicine University of Sydney Director, Centre for Sleep
Learning Objectives. And it s getting worse. The Big Picture. Dr. Roger Roubal
 Learning Objectives How to screen for sleep apnea; questions to ask your patients Industry treatment guidelines; when to consider an oral appliance vs. a CPAP What goals/thresholds to set for successful
Learning Objectives How to screen for sleep apnea; questions to ask your patients Industry treatment guidelines; when to consider an oral appliance vs. a CPAP What goals/thresholds to set for successful
Post-approval upper airway stimulation predictors of treatment effectiveness in the ADHERE registry
 ORIGINAL ARTICLE SLEEP Post-approval upper airway stimulation predictors of treatment effectiveness in the ADHERE registry Clemens Heiser 1, Armin Steffen 2, Maurits Boon 3, Benedikt Hofauer 1, Karl Doghramji
ORIGINAL ARTICLE SLEEP Post-approval upper airway stimulation predictors of treatment effectiveness in the ADHERE registry Clemens Heiser 1, Armin Steffen 2, Maurits Boon 3, Benedikt Hofauer 1, Karl Doghramji
A Deadly Combination: Central Sleep Apnea & Heart Failure
 A Deadly Combination: Central Sleep Apnea & Heart Failure Sanjaya Gupta, MD FACC FHRS Ohio State University Symposium May 10 th, 2018 Disclosures Boston Scientific: fellowship support, speaking honoraria
A Deadly Combination: Central Sleep Apnea & Heart Failure Sanjaya Gupta, MD FACC FHRS Ohio State University Symposium May 10 th, 2018 Disclosures Boston Scientific: fellowship support, speaking honoraria
BTS sleep Course. Module 10 Therapies I: Mechanical Intervention Devices (Prepared by Debby Nicoll and Debbie Smith)
 BTS sleep Course Module 10 Therapies I: Mechanical Intervention Devices (Prepared by Debby Nicoll and Debbie Smith) S1: Overview of OSA Definition History Prevalence Pathophysiology Causes Consequences
BTS sleep Course Module 10 Therapies I: Mechanical Intervention Devices (Prepared by Debby Nicoll and Debbie Smith) S1: Overview of OSA Definition History Prevalence Pathophysiology Causes Consequences
WHAT YOU NEED TO KNOW ABOUT SLEEP APNEA
 WHAT YOU NEED TO KNOW ABOUT SLEEP APNEA Wayne Driscoll Clinical Education Specialist 2 SLEEP APNEA IN THE NEWS Carrie Fisher died from sleep apnea, other factors, coroner says USA Today NJ Transit engineer
WHAT YOU NEED TO KNOW ABOUT SLEEP APNEA Wayne Driscoll Clinical Education Specialist 2 SLEEP APNEA IN THE NEWS Carrie Fisher died from sleep apnea, other factors, coroner says USA Today NJ Transit engineer
Polysomnography for Obstructive Sleep Apnea Should Include Arousal-Based Scoring: An American Academy of Sleep Medicine Position Statement
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 Polysomnography for Obstructive Sleep Apnea Should Include Arousal-Based Scoring: An American Academy of Sleep Medicine Position Statement Raman K. Malhotra,
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 Polysomnography for Obstructive Sleep Apnea Should Include Arousal-Based Scoring: An American Academy of Sleep Medicine Position Statement Raman K. Malhotra,
Clinical Trials in OSA
 24th ANNUAL ADVANCES IN SLEEP APNEA AND SNORING February 16-17, 2018 Grand Hyatt on Union Square San Francisco, California Clinical Trials in OSA Samuel T. Kuna, MD Department of Medicine Center for Sleep
24th ANNUAL ADVANCES IN SLEEP APNEA AND SNORING February 16-17, 2018 Grand Hyatt on Union Square San Francisco, California Clinical Trials in OSA Samuel T. Kuna, MD Department of Medicine Center for Sleep
Outline. Major variables contributing to airway patency/collapse. OSA- Definition
 Outline Alicia Gruber Kalamas, MD Associate Clinical Professor of Anesthesia & Perioperative Care University of California, San Francisco September 2011 Definition Pathophysiology Patient Risk Factors
Outline Alicia Gruber Kalamas, MD Associate Clinical Professor of Anesthesia & Perioperative Care University of California, San Francisco September 2011 Definition Pathophysiology Patient Risk Factors
Overview 1/20/2014. Disclosures. Update on Hypopharyngeal and Base of Tongue Management in OSA
 Update on Hypopharyngeal and Base of Management in OSA Disclosures The following personal financial relationships with commercial interests relevant to this presentation: Eric J. Kezirian, MD, MPH Professor
Update on Hypopharyngeal and Base of Management in OSA Disclosures The following personal financial relationships with commercial interests relevant to this presentation: Eric J. Kezirian, MD, MPH Professor
Sleep endoscopy with artificial induction of sleep and somnography in 385 snorers
 Sleep endoscopy with artificial induction of sleep and somnography in 385 snorers N.S. HESSEL, D.M. LAMAN, V.C.P.J. VAN AMMERS, N. DE VRIES, H. VAN DUIJN SINT LUCAS ANDREAS ZIEKENHUIS, LOCATIE SINT LUCAS,
Sleep endoscopy with artificial induction of sleep and somnography in 385 snorers N.S. HESSEL, D.M. LAMAN, V.C.P.J. VAN AMMERS, N. DE VRIES, H. VAN DUIJN SINT LUCAS ANDREAS ZIEKENHUIS, LOCATIE SINT LUCAS,
A new beginning in therapy for women
 A new beginning in therapy for women OSA in women Tailored solutions for Her AutoSet for Her algorithm ResMed.com Women and OSA OSA has traditionally been considered to be a male disease. However, recent
A new beginning in therapy for women OSA in women Tailored solutions for Her AutoSet for Her algorithm ResMed.com Women and OSA OSA has traditionally been considered to be a male disease. However, recent
Snoring and Its Outcomes
 Disclosures None Snoring and Its Outcomes Jolie Chang, MD Otolaryngology, Head and Neck Surgery University of California, San Francisco February 14, 2014 Otolaryngology Head Outline Snoring and OSA Acoustics
Disclosures None Snoring and Its Outcomes Jolie Chang, MD Otolaryngology, Head and Neck Surgery University of California, San Francisco February 14, 2014 Otolaryngology Head Outline Snoring and OSA Acoustics
Surgical Treatment of Snoring and Obstructive Sleep Apnea Syndrome
 Surgical Treatment of Snoring and Obstructive Sleep Apnea Syndrome Policy Number: 7.01.101 Last Review: 8/2018 Origination: 8/2005 Next Review: 8/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue
Surgical Treatment of Snoring and Obstructive Sleep Apnea Syndrome Policy Number: 7.01.101 Last Review: 8/2018 Origination: 8/2005 Next Review: 8/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue
Surgical Treatment of OSA. Han-Soo Bae, MD Monroe Ear Nose and Throat Associates May 5, 2017
 Surgical Treatment of OSA Han-Soo Bae, MD Monroe Ear Nose and Throat Associates May 5, 2017 Disclosure None Treatment of OSA PAP Oral appliance Surgery OSA and Mortality Surgical Treatment of OSA Surgery
Surgical Treatment of OSA Han-Soo Bae, MD Monroe Ear Nose and Throat Associates May 5, 2017 Disclosure None Treatment of OSA PAP Oral appliance Surgery OSA and Mortality Surgical Treatment of OSA Surgery
18/06/2009 NZ Respiratory & Sleep Institute
 Sleep Disorders in Primary Care - a personal view 18/06/2009 Andrew G Veale NZ Respiratory & Sleep Institute Abnormal Sleep Disorders of the initiation & maintenance of sleep (DIMS) Insomnia 1 o or 2 o
Sleep Disorders in Primary Care - a personal view 18/06/2009 Andrew G Veale NZ Respiratory & Sleep Institute Abnormal Sleep Disorders of the initiation & maintenance of sleep (DIMS) Insomnia 1 o or 2 o
Obstructive sleep apnoea How to identify?
 Obstructive sleep apnoea How to identify? Walter McNicholas MD Newman Professor in Medicine, St. Vincent s University Hospital, University College Dublin, Ireland. Potential conflict of interest None Obstructive
Obstructive sleep apnoea How to identify? Walter McNicholas MD Newman Professor in Medicine, St. Vincent s University Hospital, University College Dublin, Ireland. Potential conflict of interest None Obstructive
Clinical Policy Title: Uvulopalatopharyngoplasty
 Clinical Policy Title: Uvulopalatopharyngoplasty Clinical Policy Number: 10.03.05 Effective Date: October 1, 2015 Initial Review Date: June 17, 2015 Most Recent Review Date: July 20, 2017 Next Review Date:
Clinical Policy Title: Uvulopalatopharyngoplasty Clinical Policy Number: 10.03.05 Effective Date: October 1, 2015 Initial Review Date: June 17, 2015 Most Recent Review Date: July 20, 2017 Next Review Date:
Oral Appliances and their Clinical Indications in OSA
 Oral Appliances and their Clinical Indications in OSA Disclosures Andrew Chan, MB BS, PhD, FRACP, FCCP Staff Specialist, Department of Respiratory and Sleep Medicine, Royal North Shore Hospital, University
Oral Appliances and their Clinical Indications in OSA Disclosures Andrew Chan, MB BS, PhD, FRACP, FCCP Staff Specialist, Department of Respiratory and Sleep Medicine, Royal North Shore Hospital, University
Upper Airway Stimulation for Obstructive Sleep Apnea: The Surgical Learning Curve
 The Laryngoscope VC 2015 The American Laryngological, Rhinological and Otological Society, Inc. Upper Airway Stimulation for Obstructive Sleep Apnea: The Surgical Learning Curve Alexander W. Murphey, MD;
The Laryngoscope VC 2015 The American Laryngological, Rhinological and Otological Society, Inc. Upper Airway Stimulation for Obstructive Sleep Apnea: The Surgical Learning Curve Alexander W. Murphey, MD;
DOWNLOAD OR READ : TREATMENT FOR SNORING PROBLEMS PDF EBOOK EPUB MOBI
 DOWNLOAD OR READ : TREATMENT FOR SNORING PROBLEMS PDF EBOOK EPUB MOBI Page 1 Page 2 treatment for snoring problems treatment for snoring problems pdf treatment for snoring problems problem, presenting
DOWNLOAD OR READ : TREATMENT FOR SNORING PROBLEMS PDF EBOOK EPUB MOBI Page 1 Page 2 treatment for snoring problems treatment for snoring problems pdf treatment for snoring problems problem, presenting
Inspire Medical Systems. Physician Billing Guide
 Inspire Medical Systems Physician Billing Guide 2019 Inspire Medical Systems Physician Billing Guide This Physician Billing Guide was developed to help providers correctly bill for Inspire Upper Airway
Inspire Medical Systems Physician Billing Guide 2019 Inspire Medical Systems Physician Billing Guide This Physician Billing Guide was developed to help providers correctly bill for Inspire Upper Airway
Positive Airway Pressure and Oral Devices for the Treatment of Obstructive Sleep Apnea
 Positive Airway Pressure and Oral Devices for the Treatment of Obstructive Sleep Apnea Policy Number: Original Effective Date: MM.01.009 11/01/2009 Line(s) of Business: Current Effective Date: HMO; PPO;
Positive Airway Pressure and Oral Devices for the Treatment of Obstructive Sleep Apnea Policy Number: Original Effective Date: MM.01.009 11/01/2009 Line(s) of Business: Current Effective Date: HMO; PPO;
Kaniethapriya A.S, Ganesh Prasad S.
 Original Article A Comparative Study of Effects of Oropharyngeal Exercises and Continuous Positive Airway Pressure (CPAP) on Parameters of Sleep on Moderate Obstructive Sleep Apnea Syndrome (OSAS) Patients
Original Article A Comparative Study of Effects of Oropharyngeal Exercises and Continuous Positive Airway Pressure (CPAP) on Parameters of Sleep on Moderate Obstructive Sleep Apnea Syndrome (OSAS) Patients
What is Oral Appliance Therapy?
 You may have been diagnosed by your physician as required treatment for sleep-disordered breathing (snoring and/or obstructive sleep apnea). This condition may pose serious health risks since it disrupts
You may have been diagnosed by your physician as required treatment for sleep-disordered breathing (snoring and/or obstructive sleep apnea). This condition may pose serious health risks since it disrupts
Advances in the Evaluation and Management of Adult OSA
 Advances in the Evaluation and Management of Adult OSA Michelle Cao, DO Clinical Associate Professor Pulmonary, Critical Care, and Sleep Medicine Stanford University Conflict of Interest Disclosure I declare
Advances in the Evaluation and Management of Adult OSA Michelle Cao, DO Clinical Associate Professor Pulmonary, Critical Care, and Sleep Medicine Stanford University Conflict of Interest Disclosure I declare
in China Shanghai Office Beijing Office (+86) (+86)
 SLEEP Apnea in China Guide 2018-2019 Shanghai Office (+86) 21 2426 6400 Beijing Office (+86) 010 6464 0611 www.pacificprime.cn Follow us on WeChat t A comprehensive overview of sleep apnea Perhaps you
SLEEP Apnea in China Guide 2018-2019 Shanghai Office (+86) 21 2426 6400 Beijing Office (+86) 010 6464 0611 www.pacificprime.cn Follow us on WeChat t A comprehensive overview of sleep apnea Perhaps you
Positive Airway Pressure and Oral Devices for the Treatment of Obstructive Sleep Apnea
 Positive Airway Pressure and Oral Devices for the Treatment of Obstructive Sleep Apnea Policy Number: Original Effective Date: MM.01.009 11/01/2009 Line(s) of Business: Current Effective Date: HMO; PPO
Positive Airway Pressure and Oral Devices for the Treatment of Obstructive Sleep Apnea Policy Number: Original Effective Date: MM.01.009 11/01/2009 Line(s) of Business: Current Effective Date: HMO; PPO
Effect of Three Different Mandibular Advancement Devices and Two Different Bite Techniques on the Resultant Sleep Metrics
 Effect of Three Different Mandibular Advancement Devices and Two Different Bite Techniques on the Resultant Sleep Metrics Shamshudin (Sam) Kherani, DDS, FAGD, MICCMO Background It has been documented in
Effect of Three Different Mandibular Advancement Devices and Two Different Bite Techniques on the Resultant Sleep Metrics Shamshudin (Sam) Kherani, DDS, FAGD, MICCMO Background It has been documented in
IEHP considers the treatment of obstructive sleep apnea (OSA) medically necessary according to the criteria outlined below:
 : Positive Airway Pressure, Oral Appliances, and Surgical Interventions Policy: Obstructive sleep apnea (OSA) is characterized by an interruption of breathing during sleep most commonly due to extra or
: Positive Airway Pressure, Oral Appliances, and Surgical Interventions Policy: Obstructive sleep apnea (OSA) is characterized by an interruption of breathing during sleep most commonly due to extra or
In-Patient Sleep Testing/Management Boaz Markewitz, MD
 In-Patient Sleep Testing/Management Boaz Markewitz, MD Objectives: Discuss inpatient sleep programs and if they provide a benefit to patients and sleep centers Identify things needed to be considered when
In-Patient Sleep Testing/Management Boaz Markewitz, MD Objectives: Discuss inpatient sleep programs and if they provide a benefit to patients and sleep centers Identify things needed to be considered when
OSA - Obstructive sleep apnoea What you need to know if you think you might have OSA
 OSA - Obstructive sleep apnoea What you need to know if you think you might have OSA Obstructive sleep apnoea, or OSA, is a breathing problem that happens when you sleep. It can affect anyone men, women
OSA - Obstructive sleep apnoea What you need to know if you think you might have OSA Obstructive sleep apnoea, or OSA, is a breathing problem that happens when you sleep. It can affect anyone men, women
Precision Sleep Medicine
 Precision Sleep Medicine Picking Winners Improves Outcomes and Avoids Side-Effects North American Dental Sleep Medicine Conference February 17-18, 2017 Clearwater Beach, FL John E. Remmers, MD Conflict
Precision Sleep Medicine Picking Winners Improves Outcomes and Avoids Side-Effects North American Dental Sleep Medicine Conference February 17-18, 2017 Clearwater Beach, FL John E. Remmers, MD Conflict
NON-INVASIVE VENTILATION MADE RIDICULOUSLY SIMPLE
 NON-INVASIVE VENTILATION MADE RIDICULOUSLY SIMPLE Jennifer Newitt, MD 3 rd year Pulmonary/Critical Care Fellow Mentor: Patrick Strollo Jr, MD Myth or Fact?!? Myth or Fact?!? Treatment for Obstructive
NON-INVASIVE VENTILATION MADE RIDICULOUSLY SIMPLE Jennifer Newitt, MD 3 rd year Pulmonary/Critical Care Fellow Mentor: Patrick Strollo Jr, MD Myth or Fact?!? Myth or Fact?!? Treatment for Obstructive
Contemporary Snoring Management
 Contemporary Snoring Management Eric J. Kezirian, MD, MPH Director, Division of Sleep Surgery Otolaryngology Head and Neck Surgery University of California, San Francisco ekezirian@ohns.ucsf.edu Sleepsurgery.ucsf.edu
Contemporary Snoring Management Eric J. Kezirian, MD, MPH Director, Division of Sleep Surgery Otolaryngology Head and Neck Surgery University of California, San Francisco ekezirian@ohns.ucsf.edu Sleepsurgery.ucsf.edu
PATIENT MANUAL. Inspire IV Generator Model Rx Only
 PATIENT MANUAL Inspire IV Generator Model 3028 Rx Only The following is a registered trademark of Inspire Medical Systems, Inc.: Inspire Table of Contents Glossary v 1. Introduction 1 About This Manual
PATIENT MANUAL Inspire IV Generator Model 3028 Rx Only The following is a registered trademark of Inspire Medical Systems, Inc.: Inspire Table of Contents Glossary v 1. Introduction 1 About This Manual
MP Surgical Treatment of Snoring and Obstructive Sleep Apnea Syndrome
 Medical Policy MP 7.01.101 BCBSA Ref. Policy: 7.01.101 Last Review: 09/19/2018 Effective Date: 09/19/2018 Section: Surgery End Date: 03/19/2019 Related Policies 9.01.502 Experimental / Investigational
Medical Policy MP 7.01.101 BCBSA Ref. Policy: 7.01.101 Last Review: 09/19/2018 Effective Date: 09/19/2018 Section: Surgery End Date: 03/19/2019 Related Policies 9.01.502 Experimental / Investigational
Provent Therapy for Obstructive Sleep Apnea: Impact of Nasal Obstruction
 The Laryngoscope VC 2015 The American Laryngological, Rhinological and Otological Society, Inc. Provent Therapy for Obstructive Sleep Apnea: Impact of Nasal Obstruction Michael Friedman, MD; Michelle S
The Laryngoscope VC 2015 The American Laryngological, Rhinological and Otological Society, Inc. Provent Therapy for Obstructive Sleep Apnea: Impact of Nasal Obstruction Michael Friedman, MD; Michelle S
DECLARATION OF CONFLICT OF INTEREST
 DECLARATION OF CONFLICT OF INTEREST Obstructive sleep apnoea How to identify? Walter McNicholas MD Newman Professor in Medicine, St. Vincent s University Hospital, University College Dublin, Ireland. Potential
DECLARATION OF CONFLICT OF INTEREST Obstructive sleep apnoea How to identify? Walter McNicholas MD Newman Professor in Medicine, St. Vincent s University Hospital, University College Dublin, Ireland. Potential
JMSCR Vol 05 Issue 01 Page January 2017
 www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i1.161 Risk of Failure of Adenotonsillectomy
www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i1.161 Risk of Failure of Adenotonsillectomy
Snoring: What Works?
 Snoring: What Works? Eric J. Kezirian, MD, MPH Director, Division of Sleep Surgery Otolaryngology Head and Neck Surgery University of California, San Francisco ekezirian@ohns.ucsf.edu Sleepsurgery.ucsf.edu
Snoring: What Works? Eric J. Kezirian, MD, MPH Director, Division of Sleep Surgery Otolaryngology Head and Neck Surgery University of California, San Francisco ekezirian@ohns.ucsf.edu Sleepsurgery.ucsf.edu
Mandibular Advancement Device vs CPAP in the Treatment of Obstructive Sleep Apnea: Are they Equally Effective in Short Term Health Outcomes?
 NoSnores Mandibular Advancement Device (mouthguard) for Snoring & Sleep Apnea CLINICAL REFERENCES J Clin Sleep Med. 2013 Sep;9(9):971-2. doi: 10.5664/jcsm.3008. Mandibular Advancement Device vs CPAP in
NoSnores Mandibular Advancement Device (mouthguard) for Snoring & Sleep Apnea CLINICAL REFERENCES J Clin Sleep Med. 2013 Sep;9(9):971-2. doi: 10.5664/jcsm.3008. Mandibular Advancement Device vs CPAP in
