Forward Looking Statements
|
|
|
- Alexandra Francis
- 5 years ago
- Views:
Transcription
1
2 Forward Looking Statements The matters discussed in this presentation that are not historical facts are "forwardlooking statements." Forward-looking statements include, but are not limited to, statements containing the words "believes," "anticipates," "intends," "expects," "projects" and words of similar import. Readers are cautioned not to place undue reliance on these forward-looking statements, which are based on the information available to management at this time and which speak only as of the date of this presentation. The Company undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise. These forward-looking statements involve known and unknown risks, uncertainties and other factors that may cause actual results, performance or achievements of the Company or its industry to be materially different from any future results, performance or achievements expressed or implied by such forward-looking statements. All forward-looking statements should be evaluated with the understanding of their inherent uncertainty. While the Company believes the information contained herein is reliable, the Company makes no representations or warranties regarding the accuracy or completeness of this information. In addition, any investment in the Company is subject to numerous risks. Investors must be able to afford the loss of their entire investment. Any such representations and warranties and further discussion of risk factors would be made solely in formal agreements executed by the Company with its investors.
3 2013 Insolvent & near bankruptcy No ongoing operations Lost dronabinol license Deficient in SEC reporting Approx. $3M market cap Today Non-bankruptcy reorganization New capital raised Re-gained dronabinol license Current in SEC reporting Approx. $25M market cap Newly organized research program Phase 2B dronabinol clinical trial to be completed mid-2016 Phase 2A ampakine clinical trial to begin 3Q, pending financing Committed management team and board of directors 3
4 Arnold Lippa Jeff Margolis Chairman, CEO VP, Sec/Treas, Director Robert Weingarten CFO, Director Richard Purcell Senior VP R& D John Greer Chairman, Scientific Advisory Board Prof & Dir. Neuroscience Ctr., U. Alberta Katie MacFarlane Director, CCO Agile Therapeutics James Sapirstein Director, CEO ContraVir Pharm 4
5 Ampakines Positive allosteric modulators of AMPA glutamate receptors Positive effects for treatment of drug-induced respiratory depression in Phase 2A studies Positive effects for treatment of central sleep apnea in Phase 2A study Positive effects for treatment of ADHD in Phase 2A study Cannabinoids Dronabinol (Δ9-THC) is a generic FDA-approved drug Positive Phase 2A data for treatment of obstructive sleep apnea Phase 2B clinical trial in progress Use patent for the treatment of sleep related breathing disorders Positive preclinical results in Pompe disease, neonatal apnea and spinal disorders 5
6 Cortex is a leader in the discovery and development of innovative pharmaceuticals for the treatment of breathing disorders Drug-induced respiratory depression (RD) - ampakines Acute use surgical anesthesia with propofol Semi-acute use post-surgical pain management with opiates Chronic use in conjunction with opiates Sleep Apnea Obstructive sleep apnea (OSA) dronabinol Central sleep apnea (CSA) - ampakines Orphan Diseases Pompe & Perinatal Apnea Strong IP protection for compounds and uses Over $5 million in NIH grants supporting drug development 6
7 Cortex is a leader in the discovery and development of innovative pharmaceuticals for the treatment of breathing disorders Drug-Induced Respiratory Depression (RD) Patients treated with anesthetic, analgesic and/or sedatives in association with surgery (12 million/yr) or medical procedures (17 million/yr) are at risk for RD, which can be lethal (cardio-respiratory arrest) or require intubation & longer hospital stay Obstructive Sleep Apnea 18 million patients in the US CPAP is very problematic due to high non-compliance Associated morbidity (hypertension, heart disease) and mortality issues Central Sleep Apnea Affects > 5 million heart failure patients in the US with high morbidity and mortality Approx 40% of heart failure patients have CSA, which promotes heart disease progression and increases the risk of mortality. No current therapy for treatment of CSA in CHF 7
8 AMPA Receptors Mediate Synaptic Transmission in the Brain Glutamate is the major excitatory neurotransmitter in the CNS AMPAKINE Fast excitatory transmission is mediated by AMPA-type glutamate receptors Ampakines are positive, allosteric modulators of the AMPA-type glutamate receptor Prolong and strengthen synaptic transmission Presynaptic Glutamate AMPA NMDA Excitation Postsynaptic + AMPAKINE
9 AMPAKINES Novel Treatment for Respiratory Depression Initial research conducted by Dr. J. Greer, U. Alberta Ren et al, Anesthesiology. 110: , 2009 Neurons in this brainstem region control inspiratory breathing rhythm PreBotC neurons use AMPA receptors for signaling Opiates and other depressants mediate their inhibitory effects on breathing at this site AMPA-PAMs normalize breathing by enhancing firing of PreBotC respiratory rhythm neurons
10 Most frequent lethal side effect of opiate use is respiratory depression (RD) In-patient, post-surgical opiate use (~12M patients/year) increases risk for RD RD also occurs during anesthetic procedures (e.g. colonoscopy) with propofol (20 MM procedures/year) Large market potential in excess of $1 Billion/year in the US Unmet Need : Therapeutic drug treatment that can counter and reduce respiratory depression without interfering with analgesia or anesthesia Short-term studies that can be conducted rapidly and inexpensively 10
11 Stage of Development Completed Phase 1 in healthy volunteers and Phase 2a in central sleep apnea Ready for Phase 2A studies in opiate- and propofol-induced respiratory depression and central sleep apnea Targeted Indication Prevention of propofol-induced respiratory depression during surgery Post-surgical prevention of opiate-induced respiratory depression Intellectual Property Protected by an issued Composition-of-Matter Patent (expires 2028), filed worldwide; a method-of-use patent (expires 2030) Strong Preclinical Pharmacology Broad-spectrum reversal & prevention of drug-induced respiratory depression 11
12 CX1739 injection Fentanyl Fentanyl Prolonged opiate-induced respiratory depression leads to lethality CX1739 reverses opiate-induced respiratory depression and prevents lethality 12
13 Experimental Design: Administer a lethal dose of propofol to rats Inject CX1739 within 1 minute Propofol Propofol + CX
14 Two clinical studies were run in normal, healthy volunteers with CX717, an earlier Ampakine Moderate respiratory depression was induced experimentally by infusion of the opioid, Alfentanil Respiratory and analgesia end-points were measured Oral CX717 prevented the onset of respiratory depression without impacting the pain-relieving properties of the opioid 14
15 Breathing Rate NORMAL RD 8 BPM Breaths/min ** NS Placebo CX717 ** P < 0.01 NS - Not Significant 0 Baseline +Alfentanil +Naloxone Alfentanil reduced breathing rate & produced respiratory depression CX717 maintains respiratory rate in the presence of Alfentanil Data are expressed as the basal respiratory rate. N= 15 and 16 per group. CX717 dose is 1500mg. 15
16 CX717 Maintains the Analgesic Properties of Opioids Analgesia 2.0 Delivery of a electrical stimulation to finger Analgesia Pain Sensitivity NS Placebo CX Baseline +Alfentanil +Naloxone Alfentanil reduced the pain sensitivity (produced analgesia) Analgesia was unaffected by CX717 Data are expressed as the pain sensitivity, normalized to the Baseline measurement. N = 15 and 16 per group. CX717 dose is 1500mg. 16
17 Autosomal recessive metabolic disorder caused by an accumulation of lysosomal glycogen due to a deficiency of alpha glucosidase Damages muscle and nerve cells throughout the body Respiratory failure is the most common cause of death Replacement enzyme therapy 17
18 evie Effects of Ampakine on Minute Volume * * p<.05, compared to vehicle ** p<.05, compared to mutant Elmallah et al, in press 18
19 Mechanism of Action A Positive Allosteric Modulator of AMPA receptors (AMPA-PAM) Water-soluble for injectable dosage forms Stage of Development Injectable routes being studied in animal models of respiratory depression Supported by SBIR contract Targeted Indication Injectable rescue treatment for opiate and propofol-induced respiratory depression Intellectual Property Protected by an issued Composition-of-Matter Patent (expires 2028), filed worldwide; a method-of-use patent (expires 2030) Strong Preclinical pharmacology package 19
20 Sleep Apnea Repetitive episodes of airflow cessation (apnea) or reduction (hypopnea) for more than 10s during sleep Three types: Obstructive, Central & Mixed The Sleep Apnea Market is Large 18 million U.S. adults with moderate or severe sleep apnea Market potential for sleep apnea is $3-9 Billion/Year Current Treatments CPAP device Surgery; dental devices Clear Market Need Poor compliance with CPAP No drug treatment available
21 Obstructive sleep apnea (OSA) involves a decrease or complete halt in airflow despite an ongoing effort to breathe during sleep Occurs when the muscles relax during sleep Soft tissue in back of throat collapses and obstructs upper airway Affects 18 MM adults in the U.S.; no current drug treatment available Significant morbidity due to stroke, hypertension, heart failure, diabetes, and other cardiovascular diseases Normal Breathing Snoring OSA 21
22 Disease State Scope of the Problem in the US Es5mated US Prevalence Annual Es5mated Cost to Society Annual Indicated Drug Therapy Expenditures OSA MM $75.0 Billion $ 0 Asthma 6, MM $18.3 Billion $13.5 Billion Hypertension MM $73.4 Billion $48.5 Billion Diabetes 11, MM $174 Billion $20.6 Billion 1 Obstructive sleep apnea and sleep. National Sleep Foundation Web site. 2 Manufacturer Recommendations 3 Qualitative Market Research, Physician / Patient interviews, CPAP Supply USA, 5 American Sleep Apnea Association, Asthma & Allergy Foundation of America 7 Espicom Business Intelligence's New Drug Futures, Burt, V., et al., Hypertension, Lloyd-Jones, D., et al., Circulation 119(3):e21-181, Acmite Market Intelligence, Arrowhead, Gloabal Diabetes Market, American Diabetes Assoc.,
23 Works as an air splint to keep upper airway open during sleep 30% of patients prescribed CPAP never initiate treatment when prescribed a machine Over 50% of patients stop using CPAP in the first year of use ; may only wear 3-4h/ night 23
24 Mechanism of Action Dronabinol is (Δ-9)THC, a cannabinoid agonist Commercial Status FDA approved for the treatment of anorexia in AIDS patients and nausea and vomiting in cancer patients undergoing chemotherapy Schedule III drug available by prescription, low risk of addiction Stage of Development Phase 2A data demonstrates statistically significant improvement in OSA Phase 2B study in OSA is in progress with completion in mid-2016 Intellectual Property Issued method-of-use patent in the US for the use of Dronabinol for treating OSA (expires 2025) & pending patents on modified release formulations Funding NIH funded $5MM grant for Phase 2B study in OSA 24
25 Randomized, double-blind, placebo-controlled dose escalation study in 22 patients with OSA Randomized to Placebo (N=5) or Dronabinol (N=17) for 21 days 2.5, 5 and 10 mg/night studied with weekly dose escalation Overnight polysomnogram (PSG) at baseline, and after 7, 14 and 21 days of treatment Efficacy tests: Apnea-Hypopnea Index (AHI) used to assess OSA efficacy Stanford Sleepiness Scale (SSS) used to measure daytime sleepiness 25
26 40% AHI Mean % Change from Baseline to End of Treatment Reduction in Sleep Apnea 30% 20% 10% 0% -10% -20% -30% Placebo Dronabinol P< % 26
27 Apnea Suppression as a Function of Dose and Time mg 5 mg 10 mg AHI versus Baseline st Half 2nd Half -20 n= 17 n= 13 n= 8 The plasma half-life of dronabinol is 2 4 hours. 27
28 Phase 2B study in progress at University of Illinois 120 subjects (40/group, 6 wks dosing) Doses: Placebo, 2.5 mg, 10 mg qd Study costs funded by NIH Grant for $5 MM Top line data by mid
29 Issued Method-of-Use patent for dronabinol and OSA Expires in 2025 Potential proprietary dosage and pulse-dose formulation to provide efficacy over entire night Pending patent applications US government maintains close oversight on off-label dronabinol sales due to its Schedule III status Generics and medical marijuana are not covered for this use by third party payers Off-label prescriptions unlikely 29
30 Compound Indication Status Estimated Start Date Estimated Completion Dronabinol Obstructive Sleep Apnea Phase IIB started mid-2016 CX1739 CX1739/ CX717 CX1942 Opiate-induced RD Phase IIA 2Q2015 3Q2015 Propofol-induced RD Phase IIA 3Q2015 4Q2015 Pompe Disease Phase IIA 4Q2015 2Q2016 Injectable for RD Pre-clinical studies 3Q2014 2Q2015
RespireRx Pharmaceuticals Inc. Executives Presenting at the International Cannabinoid Derived Pharmaceuticals Summit
 RespireRx Pharmaceuticals Inc. Executives Presenting at the International Cannabinoid Derived Pharmaceuticals Summit December 11 12, 2018 Workshops, December 10, 2018 The Revere Hotel 200 Stuart St., Boston,
RespireRx Pharmaceuticals Inc. Executives Presenting at the International Cannabinoid Derived Pharmaceuticals Summit December 11 12, 2018 Workshops, December 10, 2018 The Revere Hotel 200 Stuart St., Boston,
Avenue Therapeutics, Inc. August 2016
 Avenue Therapeutics, Inc. August 2016 Forward Looking Statements Statements in this presentation that are not descriptions of historical facts are forward looking statements within the meaning of the safe
Avenue Therapeutics, Inc. August 2016 Forward Looking Statements Statements in this presentation that are not descriptions of historical facts are forward looking statements within the meaning of the safe
James S. Manuso PhD President, CEO, Vice-Chairman. RespireRX Pharmaceuticals Inc. (OTCQB: RSPI)
 CEOCFO Magazine ceocfointerviews.com All rights reserved! Issue: February 10, 2017 Dronabinol for the Treatment of Obstructive Sleep Apnea and Ampakines for Treating Central Sleep Apnea, Drug-Induced Respiratory
CEOCFO Magazine ceocfointerviews.com All rights reserved! Issue: February 10, 2017 Dronabinol for the Treatment of Obstructive Sleep Apnea and Ampakines for Treating Central Sleep Apnea, Drug-Induced Respiratory
Avenue Therapeutics, Inc. September 2016
 Avenue Therapeutics, Inc. September 2016 Forward Looking Statements Statements in this presentation that are not descriptions of historical facts are forward looking statements within the meaning of the
Avenue Therapeutics, Inc. September 2016 Forward Looking Statements Statements in this presentation that are not descriptions of historical facts are forward looking statements within the meaning of the
Avenue Therapeutics, Inc. May 2017
 Avenue Therapeutics, Inc. May 2017 Forward Looking Statements Statements in this presentation that are not descriptions of historical facts are forward looking statements within the meaning of the safe
Avenue Therapeutics, Inc. May 2017 Forward Looking Statements Statements in this presentation that are not descriptions of historical facts are forward looking statements within the meaning of the safe
Memory Pharmaceuticals Establishes Plans for Clinical Program for MEM 3454 in Schizophrenia. -Broadens Roche Nicotinic Alpha-7 Alliance-
 Memory Pharmaceuticals Establishes Plans for Clinical Program for MEM 3454 in Schizophrenia -Broadens Roche Nicotinic Alpha-7 Alliance- -Secures Additional $5M Debt Financing to Support Phase 2a Trial-
Memory Pharmaceuticals Establishes Plans for Clinical Program for MEM 3454 in Schizophrenia -Broadens Roche Nicotinic Alpha-7 Alliance- -Secures Additional $5M Debt Financing to Support Phase 2a Trial-
AVENUE THERAPEUTICS, INC. NASDAQ: ATXI JANUARY 2018
 AVENUE THERAPEUTICS, INC. NASDAQ: ATXI JANUARY 2018 Forward Looking Statements Statements in this presentation that are not descriptions of historical facts are forward-looking statements within the meaning
AVENUE THERAPEUTICS, INC. NASDAQ: ATXI JANUARY 2018 Forward Looking Statements Statements in this presentation that are not descriptions of historical facts are forward-looking statements within the meaning
August 7, Q Financial Results
 August 7, 2018 Q2 2018 Financial Results 1 Agenda Today s Speakers Paul Cox, Senior Director, Investor Relations Jeff Jonas, M.D., Chief Executive Officer Steve Kanes, M.D., Ph.D., Chief Medical Officer
August 7, 2018 Q2 2018 Financial Results 1 Agenda Today s Speakers Paul Cox, Senior Director, Investor Relations Jeff Jonas, M.D., Chief Executive Officer Steve Kanes, M.D., Ph.D., Chief Medical Officer
SPECIALTY MEDICAL CANNABIS COMPANY
 SPECIALTY MEDICAL CANNABIS COMPANY TSX Venture: RVV OTCQB: RVVTF Corporate Presentation - August 2018 ReviveThera.com Revive Therapeutics Ltd. Office: 905-605-5535 E-mail: info@revivethera.com Forward
SPECIALTY MEDICAL CANNABIS COMPANY TSX Venture: RVV OTCQB: RVVTF Corporate Presentation - August 2018 ReviveThera.com Revive Therapeutics Ltd. Office: 905-605-5535 E-mail: info@revivethera.com Forward
Dr Alireza Yarahmadi and Dr Arvind Perathur Mercy Medical Center - Winter Retreat Des Moines February 2012
 Dr Alireza Yarahmadi and Dr Arvind Perathur Mercy Medical Center - Winter Retreat Des Moines February 2012 Why screen of OSA prior to surgery? What factors increase the risk? When due to anticipate problems?
Dr Alireza Yarahmadi and Dr Arvind Perathur Mercy Medical Center - Winter Retreat Des Moines February 2012 Why screen of OSA prior to surgery? What factors increase the risk? When due to anticipate problems?
Oventus: Innovators in Sleep Apnoea Treatment Investor lunch presentation Tattersall s Club, Brisbane
 Oventus: Innovators in Sleep Apnoea Treatment Investor lunch presentation Tattersall s Club, Brisbane February 2018 Twitter: @OventusLtd About Oventus (ASX: OVN) Oventus is commercialising its Airway Technology
Oventus: Innovators in Sleep Apnoea Treatment Investor lunch presentation Tattersall s Club, Brisbane February 2018 Twitter: @OventusLtd About Oventus (ASX: OVN) Oventus is commercialising its Airway Technology
Forward Looking Information
 Vital Signs, Inc. Forward Looking Information Except for historical information discussed, the statements made today are forward-looking statements that involve risks and uncertainties. Investors are cautioned
Vital Signs, Inc. Forward Looking Information Except for historical information discussed, the statements made today are forward-looking statements that involve risks and uncertainties. Investors are cautioned
AXIM Biotechnologies Reports Year End 2017 Results
 March 19, 2018 AXIM Biotechnologies Reports Year End 2017 Results NEW YORK, March 19, 2018 (GLOBE NEWSWIRE) -- AXIM Biotechnologies, Inc. (AXIM Biotech) (OTC:AXIM), a world leader in cannabinoid research
March 19, 2018 AXIM Biotechnologies Reports Year End 2017 Results NEW YORK, March 19, 2018 (GLOBE NEWSWIRE) -- AXIM Biotechnologies, Inc. (AXIM Biotech) (OTC:AXIM), a world leader in cannabinoid research
COMPANY PRESENTATION JULY Bob Bechard Executive Vice-President Corporate Development & Licensing
 COMPANY PRESENTATION JULY 2018 Bob Bechard Executive Vice-President Corporate Development & Licensing Forward Looking Statements Some statements in this release may contain forward-looking information.
COMPANY PRESENTATION JULY 2018 Bob Bechard Executive Vice-President Corporate Development & Licensing Forward Looking Statements Some statements in this release may contain forward-looking information.
ROBERT C. PRITCHARD DIRECTOR MICHAEL O. FOSTER ASSISTANT DIR. SLEEP APNEA
 ROBERT C. PRITCHARD DIRECTOR MICHAEL O. FOSTER ASSISTANT DIR. SLEEP APNEA A Person is physically qualified to drive a motor vehicle if that person; -(5) has no established medical history or clinical diagnosis
ROBERT C. PRITCHARD DIRECTOR MICHAEL O. FOSTER ASSISTANT DIR. SLEEP APNEA A Person is physically qualified to drive a motor vehicle if that person; -(5) has no established medical history or clinical diagnosis
About VirtuOx. Was marketed exclusively by Phillips Healthcare division, Respironics for 3 years
 About VirtuOx VirtuOx, Inc. assists physicians and Durable Medical Equipment (DME)( companies diagnose respiratory diseases and qualify patients for home respiratory equipment under the guidelines of CMS
About VirtuOx VirtuOx, Inc. assists physicians and Durable Medical Equipment (DME)( companies diagnose respiratory diseases and qualify patients for home respiratory equipment under the guidelines of CMS
Corporate Presentation. October 2017
 AEPP Breakthrough technology for women s cancers Corporate Presentation October 2017 Oncolix 2017 1 Safe Harbor Certain of the statements and the projections set forth in this presentation constitute forward-looking
AEPP Breakthrough technology for women s cancers Corporate Presentation October 2017 Oncolix 2017 1 Safe Harbor Certain of the statements and the projections set forth in this presentation constitute forward-looking
For personal use only
 ASX Release Oventus presentation for AGM of Thorney Technologies Ltd Brisbane, Australia 24 th November 2017: Oventus Medical Ltd (ASX: OVN) is pleased to release a copy of the presentation that Founder
ASX Release Oventus presentation for AGM of Thorney Technologies Ltd Brisbane, Australia 24 th November 2017: Oventus Medical Ltd (ASX: OVN) is pleased to release a copy of the presentation that Founder
MEDICAL CANNABIS COMPANY
 MEDICAL CANNABIS COMPANY TSX Venture: RVV OTCQB: RVVTF Corporate Presentation - June 2018 ReviveThera.com Revive Therapeutics Ltd. Office: 905-605-5535 E-mail: info@revivethera.com Forward looking statements
MEDICAL CANNABIS COMPANY TSX Venture: RVV OTCQB: RVVTF Corporate Presentation - June 2018 ReviveThera.com Revive Therapeutics Ltd. Office: 905-605-5535 E-mail: info@revivethera.com Forward looking statements
ASX Investor Presentation
 ASX Investor Presentation Sydney, Australia Dr Peter Neustadt Executive Chairman and CEO November 2014 Copyright and registered trademark notice ASX Investor Presentation Page 0 Who we are SomnoMed Limited
ASX Investor Presentation Sydney, Australia Dr Peter Neustadt Executive Chairman and CEO November 2014 Copyright and registered trademark notice ASX Investor Presentation Page 0 Who we are SomnoMed Limited
Itamar Medical. Investor Presentation August 2018
 Itamar Medical Investor Presentation August 2018 Disclaimer Itamar Medical Ltd. (the "Company") is furnishing this presentation and any information given during this presentation, solely for the consideration
Itamar Medical Investor Presentation August 2018 Disclaimer Itamar Medical Ltd. (the "Company") is furnishing this presentation and any information given during this presentation, solely for the consideration
November 2, Q Financial Results
 November 2, 2017 Q3 2017 Financial Results Agenda Today s Speakers Paul Cox, Senior Director, Investor Relations Jeff Jonas, M.D., Chief Executive Officer Steve Kanes, M.D., Ph.D., Chief Medical Officer
November 2, 2017 Q3 2017 Financial Results Agenda Today s Speakers Paul Cox, Senior Director, Investor Relations Jeff Jonas, M.D., Chief Executive Officer Steve Kanes, M.D., Ph.D., Chief Medical Officer
David Zaccardelli, PharmD Chief Executive Officer. J.P. Morgan 37 th Annual Healthcare Conference January 8, 2019
 David Zaccardelli, PharmD Chief Executive Officer J.P. Morgan 37 th Annual Healthcare Conference January 8, 2019 Disclaimer Certain information contained in this presentation relates to or is based on
David Zaccardelli, PharmD Chief Executive Officer J.P. Morgan 37 th Annual Healthcare Conference January 8, 2019 Disclaimer Certain information contained in this presentation relates to or is based on
Theravance Announces Positive Results from Phase 1 and Phase 2 Clinical Studies with TD-1211 in Development for Opioid-Induced Constipation
 Theravance Announces Positive Results from Phase 1 and Phase 2 Clinical Studies with TD-1211 in Development for Opioid-Induced Constipation TD-1211 Achieves Primary and Secondary Endpoints SOUTH SAN FRANCISCO,
Theravance Announces Positive Results from Phase 1 and Phase 2 Clinical Studies with TD-1211 in Development for Opioid-Induced Constipation TD-1211 Achieves Primary and Secondary Endpoints SOUTH SAN FRANCISCO,
DARA Reports Year-End 2012 Financial Results
 April 1, 2013 DARA Reports Year-End 2012 Financial Results Company Provides a Commercial Portfolio and Development Pipeline Business Update RALEIGH, NC -- (MARKETWIRE) -- 04/01/13 -- DARA BioSciences,
April 1, 2013 DARA Reports Year-End 2012 Financial Results Company Provides a Commercial Portfolio and Development Pipeline Business Update RALEIGH, NC -- (MARKETWIRE) -- 04/01/13 -- DARA BioSciences,
N a s d a q : I N S Y
 N a s d a q : I N S Y 0 Safe Harbor Statement This presentation contains both historical information and forward-looking statements. Forward-looking statements are based on management's current expectations
N a s d a q : I N S Y 0 Safe Harbor Statement This presentation contains both historical information and forward-looking statements. Forward-looking statements are based on management's current expectations
A New, Clinically Proven Sleep Apnea Therapy for people unable to use CPAP.
 A New, Clinically Proven Sleep Apnea Therapy for people unable to use CPAP. Take Heart. If You Have OSA, You re Not Alone. Like you, more than 18 million Americans are estimated to have Obstructive Sleep
A New, Clinically Proven Sleep Apnea Therapy for people unable to use CPAP. Take Heart. If You Have OSA, You re Not Alone. Like you, more than 18 million Americans are estimated to have Obstructive Sleep
Investors Update: Abuse-Deterrent d-amphetamine Immediate Release (ADAIR) for ADHD
 Investors Update: Abuse-Deterrent d-amphetamine Immediate Release (ADAIR) for ADHD February 7, 2017 1 Forward-Looking Statements This presentation contains forward-looking statements within the meaning
Investors Update: Abuse-Deterrent d-amphetamine Immediate Release (ADAIR) for ADHD February 7, 2017 1 Forward-Looking Statements This presentation contains forward-looking statements within the meaning
Annual Stockholder Meeting May 30, confidently live life with ease
 Annual Stockholder Meeting May 30, 2018 confidently live life with ease Forward-Looking Statements This presentation contains certain forward-looking statements that involve risks and uncertainties that
Annual Stockholder Meeting May 30, 2018 confidently live life with ease Forward-Looking Statements This presentation contains certain forward-looking statements that involve risks and uncertainties that
Investor presentation. Bioshares Biotech Summit July 2017
 Investor presentation Bioshares Biotech Summit 2017 22 July 2017 1 Disclaimer Some of the information in this presentation may refer to Dimerix Limited ( Dimerix or the Company ) based on information available
Investor presentation Bioshares Biotech Summit 2017 22 July 2017 1 Disclaimer Some of the information in this presentation may refer to Dimerix Limited ( Dimerix or the Company ) based on information available
SPECIALTY MEDICAL CANNABIS COMPANY
 SPECIALTY MEDICAL CANNABIS COMPANY TSX Venture: RVV OTCQB: RVVTF Corporate Presentation - August 2018 ReviveThera.com Revive Therapeutics Ltd. Office: 905-605-5535 E-mail: info@revivethera.com Forward
SPECIALTY MEDICAL CANNABIS COMPANY TSX Venture: RVV OTCQB: RVVTF Corporate Presentation - August 2018 ReviveThera.com Revive Therapeutics Ltd. Office: 905-605-5535 E-mail: info@revivethera.com Forward
NASDAQ: ZGNX. Company Presentation. October 2017
 NASDAQ: ZGNX Company Presentation October 2017 2 Forward Looking Statement Zogenix cautions you that statements included in this presentation that are not a description of historical facts are forward-looking
NASDAQ: ZGNX Company Presentation October 2017 2 Forward Looking Statement Zogenix cautions you that statements included in this presentation that are not a description of historical facts are forward-looking
Committed to Transforming the Treatment Paradigm for Migraine Prevention
 Committed to Transforming the Treatment Paradigm for Migraine Prevention 36th Annual J.P. Morgan Healthcare Conference January 8, 2018 Forward-Looking Statements This presentation and the accompanying
Committed to Transforming the Treatment Paradigm for Migraine Prevention 36th Annual J.P. Morgan Healthcare Conference January 8, 2018 Forward-Looking Statements This presentation and the accompanying
Cowen Investor Conference March confidently live life with ease
 Cowen Investor Conference March 2018 confidently live life with ease Forward-Looking Statements This presentation contains certain forward-looking statements that involve risks and uncertainties that could
Cowen Investor Conference March 2018 confidently live life with ease Forward-Looking Statements This presentation contains certain forward-looking statements that involve risks and uncertainties that could
Index SLEEP MEDICINE CLINICS. Note: Page numbers of article titles are in boldface type.
 549 SLEEP MEDICINE CLINICS Sleep Med Clin 1 (2007) 549 553 Note: Page numbers of article titles are in boldface type. A Abdominal motion, in assessment of sleep-related breathing disorders, 452 454 Adherence,
549 SLEEP MEDICINE CLINICS Sleep Med Clin 1 (2007) 549 553 Note: Page numbers of article titles are in boldface type. A Abdominal motion, in assessment of sleep-related breathing disorders, 452 454 Adherence,
Dental Sleep Medicine Basics
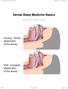 Dental Sleep Medicine Basics Written By: Patrick Tessier 2018 www.tap.wiki/ Page 1 of 8 INTRODUCTION Here are some basic aspect of Dental Sleep Medicine. This viewpoint is from an industry participant,
Dental Sleep Medicine Basics Written By: Patrick Tessier 2018 www.tap.wiki/ Page 1 of 8 INTRODUCTION Here are some basic aspect of Dental Sleep Medicine. This viewpoint is from an industry participant,
AVENUE THERAPEUTICS, INC. NASDAQ: ATXI NOV 2018
 AVENUE THERAPEUTICS, INC. NASDAQ: ATXI NOV 2018 Forward Looking Statements Statements in this presentation that are not descriptions of historical facts are forward-looking statements within the meaning
AVENUE THERAPEUTICS, INC. NASDAQ: ATXI NOV 2018 Forward Looking Statements Statements in this presentation that are not descriptions of historical facts are forward-looking statements within the meaning
Postpartum depression is the most common medical complication of childbirth, estimated to affect approximately 400,000 women annually in the U.S.
 Sage Therapeutics Announces FDA Approval of ZULRESSO (brexanolone) Injection, the First and Only Treatment Specifically Indicated for Postpartum Depression March 19, 2019 Approval based on results from
Sage Therapeutics Announces FDA Approval of ZULRESSO (brexanolone) Injection, the First and Only Treatment Specifically Indicated for Postpartum Depression March 19, 2019 Approval based on results from
SPHERIX ANNOUNCES FIRST QUARTER 2010 FINANCIAL RESULTS
 SPHERIX Investor Relations Phone: (301) 897-2564 Email: info@spherix.com SPHERIX ANNOUNCES FIRST QUARTER 2010 FINANCIAL RESULTS BETHESDA, MD, May 21, 2010 - Spherix Incorporated (NASDAQ CM: SPEX), an innovator
SPHERIX Investor Relations Phone: (301) 897-2564 Email: info@spherix.com SPHERIX ANNOUNCES FIRST QUARTER 2010 FINANCIAL RESULTS BETHESDA, MD, May 21, 2010 - Spherix Incorporated (NASDAQ CM: SPEX), an innovator
Tamsulosin Hydrochloride 0.4 mg Capsule
 Tamsulosin Hydrochloride 0.4 mg Capsule, Tamsulosin Hydrochloride 0.4 mg Capsule India, Tamsulosin Hydrochloride 0.4 mg Capsule manufacturers India, side effects Tamsulosin Hydrochloride 0.4 mg Capsule
Tamsulosin Hydrochloride 0.4 mg Capsule, Tamsulosin Hydrochloride 0.4 mg Capsule India, Tamsulosin Hydrochloride 0.4 mg Capsule manufacturers India, side effects Tamsulosin Hydrochloride 0.4 mg Capsule
Calliditas Therapeutics Q2 Report Webcast August 16, 2018, 10:00 Presenters: Renée Aguiar-Lucander, CEO Fredrik Johansson, CFO
 Calliditas Therapeutics Q2 Report 2018 Webcast August 16, 2018, 10:00 Presenters: Renée Aguiar-Lucander, CEO Fredrik Johansson, CFO Disclaimer Important information This presentation may contain certain
Calliditas Therapeutics Q2 Report 2018 Webcast August 16, 2018, 10:00 Presenters: Renée Aguiar-Lucander, CEO Fredrik Johansson, CFO Disclaimer Important information This presentation may contain certain
Ganaxolone as a Treatment for Drug-Resistant Epilepsy in Children
 Ganaxolone as a Treatment for Drug-Resistant Epilepsy in Children ANTIEPILEPTIC DRUG and DEVICE TRIALS XIII May 13-15, 2015 Turnberry Isle Miami Hotel Gail M. Farfel, PhD Chief Development & Regulatory
Ganaxolone as a Treatment for Drug-Resistant Epilepsy in Children ANTIEPILEPTIC DRUG and DEVICE TRIALS XIII May 13-15, 2015 Turnberry Isle Miami Hotel Gail M. Farfel, PhD Chief Development & Regulatory
Innovation In Ophthalmics
 Innovation In Ophthalmics Ophthalmic Innovation Summit @ AAO 2018 October 25, 2018 Mark Iwicki Chairman & CEO, Kala Pharmaceuticals Disclaimers and Notices This presentation contains forward-looking statements
Innovation In Ophthalmics Ophthalmic Innovation Summit @ AAO 2018 October 25, 2018 Mark Iwicki Chairman & CEO, Kala Pharmaceuticals Disclaimers and Notices This presentation contains forward-looking statements
Positive Airway Pressure and Oral Devices for the Treatment of Obstructive Sleep Apnea
 Positive Airway Pressure and Oral Devices for the Treatment of Obstructive Sleep Apnea Policy Number: Original Effective Date: MM.01.009 11/01/2009 Line(s) of Business: Current Effective Date: HMO; PPO
Positive Airway Pressure and Oral Devices for the Treatment of Obstructive Sleep Apnea Policy Number: Original Effective Date: MM.01.009 11/01/2009 Line(s) of Business: Current Effective Date: HMO; PPO
PATENCY-1 Top-Line Results
 PATENCY-1 Top-Line Results December 13, 2016 2016 Proteon Therapeutics, Inc. Cautionary Note Regarding Forward-Looking Statements This presentation contains statements that are, or may be deemed to be,
PATENCY-1 Top-Line Results December 13, 2016 2016 Proteon Therapeutics, Inc. Cautionary Note Regarding Forward-Looking Statements This presentation contains statements that are, or may be deemed to be,
TSX Venture: RVV OTCQB: RVVTF
 TSX Venture: RVV OTCQB: RVVTF The image part with relationship ID rid3 was not found in the file. 2017 Annual General and Special Meeting of Shareholders December 19, 2017 Revive Therapeutics Ltd. Office:
TSX Venture: RVV OTCQB: RVVTF The image part with relationship ID rid3 was not found in the file. 2017 Annual General and Special Meeting of Shareholders December 19, 2017 Revive Therapeutics Ltd. Office:
For personal use only CHANGING THE WAY THE WORLD BREATHES
 CHANGING THE WAY THE WORLD BREATHES - MAY 2016 NASAL RESPIRATORY TECHNOLOGY COMMERICAL IN CONFIDENCE WWW..GLOBAL WWW..GLOBAL Disclaimer This document contains certain forward-looking statements, relating
CHANGING THE WAY THE WORLD BREATHES - MAY 2016 NASAL RESPIRATORY TECHNOLOGY COMMERICAL IN CONFIDENCE WWW..GLOBAL WWW..GLOBAL Disclaimer This document contains certain forward-looking statements, relating
POLICY All patients will be assessed for risk factors associated with OSA prior to any surgical procedures.
 Revised Date: Page: 1 of 7 SCOPE All Pre-Admission Testing (PAT) and Same Day Surgery (SDS) nurses at HRMC. PURPOSE The purpose of this policy is to provide guidelines for identifying surgical patients
Revised Date: Page: 1 of 7 SCOPE All Pre-Admission Testing (PAT) and Same Day Surgery (SDS) nurses at HRMC. PURPOSE The purpose of this policy is to provide guidelines for identifying surgical patients
Financial Presentation
 Financial Presentation Certain of the statements that will be made during this meeting, as well as statements contained in our 2006 Annual Report to Shareholders and Annual Report on Form 10-K filed with
Financial Presentation Certain of the statements that will be made during this meeting, as well as statements contained in our 2006 Annual Report to Shareholders and Annual Report on Form 10-K filed with
in China Shanghai Office Beijing Office (+86) (+86)
 SLEEP Apnea in China Guide 2018-2019 Shanghai Office (+86) 21 2426 6400 Beijing Office (+86) 010 6464 0611 www.pacificprime.cn Follow us on WeChat t A comprehensive overview of sleep apnea Perhaps you
SLEEP Apnea in China Guide 2018-2019 Shanghai Office (+86) 21 2426 6400 Beijing Office (+86) 010 6464 0611 www.pacificprime.cn Follow us on WeChat t A comprehensive overview of sleep apnea Perhaps you
Itamar Medical. December Investors Presentation.
 Itamar Medical December 2017. Investors Presentation. Disclaimer Itamar Medical Ltd. (the "Company") is furnishing this presentation and any information given during this presentation, solely for the consideration
Itamar Medical December 2017. Investors Presentation. Disclaimer Itamar Medical Ltd. (the "Company") is furnishing this presentation and any information given during this presentation, solely for the consideration
Pierre Legault CEO June 2, 2014
 April 2012 Pierre Legault CEO June 2, 2014 Forward Looking Statements This presentation includes statements that are, or may be deemed, forward-looking statements. In some cases, these forward-looking
April 2012 Pierre Legault CEO June 2, 2014 Forward Looking Statements This presentation includes statements that are, or may be deemed, forward-looking statements. In some cases, these forward-looking
OWC PHARMACEUTICALS RESEARCH OTCQB:OWCP
 OWC PHARMACEUTICALS RESEARCH OTCQB:OWCP Disclaimer This document details business information of OWC Pharmaceuticals Research Corp and/or our wholly-owned Israeli subsidiary, One World Cannabis Ltd. (collectively,
OWC PHARMACEUTICALS RESEARCH OTCQB:OWCP Disclaimer This document details business information of OWC Pharmaceuticals Research Corp and/or our wholly-owned Israeli subsidiary, One World Cannabis Ltd. (collectively,
UPDATES IN SLEEP APNEA:
 UPDATES IN SLEEP APNEA: CPAP,CPAP COMPLIANCE, & ALTERNATIVES MICHELLE ZETOONY, DO, FCCP, FACOI BOARD CERTIFIED PULMONARY, CRITICAL CARE, SLEEP & INTERNAL MEDICINE CLEARWATER, FL DISCLOSURE I have no conflicts
UPDATES IN SLEEP APNEA: CPAP,CPAP COMPLIANCE, & ALTERNATIVES MICHELLE ZETOONY, DO, FCCP, FACOI BOARD CERTIFIED PULMONARY, CRITICAL CARE, SLEEP & INTERNAL MEDICINE CLEARWATER, FL DISCLOSURE I have no conflicts
ADAPTIMMUNE INVESTOR PRESENTATION. August 2016
 ADAPTIMMUNE INVESTOR PRESENTATION August 2016 DISCLAIMER This presentation contains forward-looking statements, as that term is defined under the Private Securities Litigation Reform Act of 1995 (PSLRA),
ADAPTIMMUNE INVESTOR PRESENTATION August 2016 DISCLAIMER This presentation contains forward-looking statements, as that term is defined under the Private Securities Litigation Reform Act of 1995 (PSLRA),
Developing Xanamem for Alzheimer s Dementia
 Developing Xanamem for Alzheimer s Dementia Dr. Bill Ketelbey CEO, Actinogen Medical Investor Presentation March 2015 Forward Statements This presentation has been prepared by Actinogen Limited. ( Actinogen
Developing Xanamem for Alzheimer s Dementia Dr. Bill Ketelbey CEO, Actinogen Medical Investor Presentation March 2015 Forward Statements This presentation has been prepared by Actinogen Limited. ( Actinogen
Quality ID #278: Sleep Apnea: Positive Airway Pressure Therapy Prescribed National Quality Strategy Domain: Effective Clinical Care
 Quality ID #278: Sleep Apnea: Positive Airway Pressure Therapy Prescribed National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process
Quality ID #278: Sleep Apnea: Positive Airway Pressure Therapy Prescribed National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process
Corporate Presentation August 6, 2015
 Corporate Presentation August 6, 2015 Creating the Next Generation of CNS Drugs Forward-Looking Statement This presentation contains forward-looking statements. These statements relate to future events
Corporate Presentation August 6, 2015 Creating the Next Generation of CNS Drugs Forward-Looking Statement This presentation contains forward-looking statements. These statements relate to future events
Investor Presentation
 ASX:THC B u i l d i n g a M e d i c i n a l C a n n a b i s B u s i n e s s Investor Presentation November 2017 DISCLAIMER The material in this presentation (material) is not and does not constitute an
ASX:THC B u i l d i n g a M e d i c i n a l C a n n a b i s B u s i n e s s Investor Presentation November 2017 DISCLAIMER The material in this presentation (material) is not and does not constitute an
Acorda Acquisition of Civitas Therapeutics. September 24, 2014
 Acorda Acquisition of Civitas Therapeutics September 24, 2014 Forward Looking Statement This presentation includes forward-looking statements within the meaning of the Private Securities Litigation Reform
Acorda Acquisition of Civitas Therapeutics September 24, 2014 Forward Looking Statement This presentation includes forward-looking statements within the meaning of the Private Securities Litigation Reform
AM-125 : Intranasal Betahistine
 AM-125 : Intranasal Betahistine February 3, 2017 NASDAQ: EARS Forward-looking Statements This presentation and the accompanying oral commentary contain forward-looking statements that involve substantial
AM-125 : Intranasal Betahistine February 3, 2017 NASDAQ: EARS Forward-looking Statements This presentation and the accompanying oral commentary contain forward-looking statements that involve substantial
Verona Pharma Announces Positive Top-Line Data from Phase 2a Clinical Trial in COPD with RPL554 Dosed in Addition to Tiotropium (Spiriva )
 Press release 7 September 2017 Verona Pharma Announces Positive Top-Line Data from Phase 2a Clinical Trial in COPD with RPL554 Dosed in Addition to Tiotropium (Spiriva ) Achieved significant and clinically
Press release 7 September 2017 Verona Pharma Announces Positive Top-Line Data from Phase 2a Clinical Trial in COPD with RPL554 Dosed in Addition to Tiotropium (Spiriva ) Achieved significant and clinically
Galvus US NDA Approvable - Overview
 Galvus US NDA Approvable - Overview Galvus NDA filed with FDA Jan 2006 PDUFA action date extended by 3 months until February 26th 2007 Significant new clinical data added to NDA - Approximately 1000 patient
Galvus US NDA Approvable - Overview Galvus NDA filed with FDA Jan 2006 PDUFA action date extended by 3 months until February 26th 2007 Significant new clinical data added to NDA - Approximately 1000 patient
Committed to Transforming the Treatment Paradigm for Migraine Prevention
 June 14, 2018 Committed to Transforming the Treatment Paradigm for Migraine Prevention September 6, 2018 Forward-Looking Statements This presentation and the accompanying commentary contains certain forward-looking
June 14, 2018 Committed to Transforming the Treatment Paradigm for Migraine Prevention September 6, 2018 Forward-Looking Statements This presentation and the accompanying commentary contains certain forward-looking
Bobby W. Sandage, Jr., PhD President & Chief Executive Officer. Lazard Capital Markets 8 th Annual Healthcare Conference
 Bobby W. Sandage, Jr., PhD President & Chief Executive Officer Lazard Capital Markets 8 th Annual Healthcare Conference Statements in this presentation that are not descriptions of historical facts are
Bobby W. Sandage, Jr., PhD President & Chief Executive Officer Lazard Capital Markets 8 th Annual Healthcare Conference Statements in this presentation that are not descriptions of historical facts are
Positive Airway Pressure and Oral Devices for the Treatment of Obstructive Sleep Apnea
 Positive Airway Pressure and Oral Devices for the Treatment of Obstructive Sleep Apnea Policy Number: Original Effective Date: MM.01.009 11/01/2009 Line(s) of Business: Current Effective Date: HMO; PPO;
Positive Airway Pressure and Oral Devices for the Treatment of Obstructive Sleep Apnea Policy Number: Original Effective Date: MM.01.009 11/01/2009 Line(s) of Business: Current Effective Date: HMO; PPO;
Axsome Therapeutics, Inc. AXS-05 R&D Day CNS Pipeline Overview
 AXS-05 R&D Day April 24, 2018 Axsome Therapeutics, Inc. AXS-05 R&D Day CNS Pipeline Overview Herriot Tabuteau, MD Chief Executive Officer Axsome Therapeutics, Inc. New York City 3 Forward-Looking Statements
AXS-05 R&D Day April 24, 2018 Axsome Therapeutics, Inc. AXS-05 R&D Day CNS Pipeline Overview Herriot Tabuteau, MD Chief Executive Officer Axsome Therapeutics, Inc. New York City 3 Forward-Looking Statements
Dynavax Corporate Presentation
 Dynavax Corporate Presentation Forward-Looking Statements This presentation contains forward-looking statements, including statements regarding our HEPLISAV-B TM regulatory submissions, product profile,
Dynavax Corporate Presentation Forward-Looking Statements This presentation contains forward-looking statements, including statements regarding our HEPLISAV-B TM regulatory submissions, product profile,
TOPIC: Continuing Coverage of CPAP Machines and Supplies for the Treatment of Obstructive Sleep Apnea
 These documents are not used to determine benefits or reimbursement. Please reference the appropriate certificate or contract for benefit information. BLUE CROSS BLUE SHIELD of MI MEDICAL POLICY Enterprise:
These documents are not used to determine benefits or reimbursement. Please reference the appropriate certificate or contract for benefit information. BLUE CROSS BLUE SHIELD of MI MEDICAL POLICY Enterprise:
PHYSICIAN COMPETENCY FOR ADULT DEEP SEDATION (Ages 14 and older)
 Name Score PHYSICIAN COMPETENCY FOR ADULT DEEP SEDATION (Ages 14 and older) 1. Pre-procedure evaluation for moderate sedation should involve all of the following EXCEPT: a) Airway Exam b) Anesthetic history
Name Score PHYSICIAN COMPETENCY FOR ADULT DEEP SEDATION (Ages 14 and older) 1. Pre-procedure evaluation for moderate sedation should involve all of the following EXCEPT: a) Airway Exam b) Anesthetic history
Dear, Respectfully, United Sleep Centers SLEEP STUDY DATE: FEBUARY 26, 2015 AT OUR DOWNEY CENTER TIME: 10PM, PLEASE ARRIVE ON TIME
 Sleep Study Instructions SLEEP STUDY DATE: FEBUARY 26, 2015 AT OUR DOWNEY CENTER TIME: 10PM, PLEASE ARRIVE ON TIME ADDRESS: 11411 BROOKSHIRE AVE SUITE 505 DOWNEY, CA Dear, Thank you for choosing us as
Sleep Study Instructions SLEEP STUDY DATE: FEBUARY 26, 2015 AT OUR DOWNEY CENTER TIME: 10PM, PLEASE ARRIVE ON TIME ADDRESS: 11411 BROOKSHIRE AVE SUITE 505 DOWNEY, CA Dear, Thank you for choosing us as
2019 COLLECTION TYPE: MIPS CLINICAL QUALITY MEASURES (CQMS) MEASURE TYPE: Process
 Quality ID #277: Sleep Apnea: Severity Assessment at Initial Diagnosis National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Management of Chronic Conditions 2019 COLLECTION
Quality ID #277: Sleep Apnea: Severity Assessment at Initial Diagnosis National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Management of Chronic Conditions 2019 COLLECTION
Strengthening our global leadership in treatment of addiction. Morgan Stanley Global Healthcare Conference September 13 th and 14 th 2018
 Strengthening our global leadership in treatment of addiction Morgan Stanley Global Healthcare Conference September 13 th and 14 th 2018 Forward Looking Statements This presentation contains certain statements
Strengthening our global leadership in treatment of addiction Morgan Stanley Global Healthcare Conference September 13 th and 14 th 2018 Forward Looking Statements This presentation contains certain statements
The ResMed Story John Brydon
 The ResMed Story John Brydon jbrydon@help-factory.com.au www.help-factory.com.au Copyright reserved John Brydon 2015-2016 1 Lecture Content ResMed Today - Overview Obstructive Sleep Apnea (OSA) CPAP Treatment
The ResMed Story John Brydon jbrydon@help-factory.com.au www.help-factory.com.au Copyright reserved John Brydon 2015-2016 1 Lecture Content ResMed Today - Overview Obstructive Sleep Apnea (OSA) CPAP Treatment
ASCEND Phase 2 Trial of AXS-05 in MDD Topline Results Conference Call
 NASDAQ: AXSM ASCEND Phase 2 Trial of in MDD Topline Results Conference Call January 7, 2019 Overview in MDD ASCEND Phase 2 Trial Topline Results Introduction Mark Jacobson, Senior Vice President, Operations
NASDAQ: AXSM ASCEND Phase 2 Trial of in MDD Topline Results Conference Call January 7, 2019 Overview in MDD ASCEND Phase 2 Trial Topline Results Introduction Mark Jacobson, Senior Vice President, Operations
Equity Research. Industry Update. Key Takeaways The Impact of The Opioid Epidemic on The Pain Management Landscape
 Member FINRA and SIPC Equity Research November 17, 2017 Healthcare Industry Update Key Takeaways The Impact of The Opioid Epidemic on The Pain Management Landscape Conclusions We hosted a key opinion leader
Member FINRA and SIPC Equity Research November 17, 2017 Healthcare Industry Update Key Takeaways The Impact of The Opioid Epidemic on The Pain Management Landscape Conclusions We hosted a key opinion leader
INVESTOR PRESENTATION
 INVESTOR PRESENTATION JUNE 2016 OTCQB:CANN ABOUT General Cannabis Corporation (GCC) is the all-in-one resource for the highest quality service providers available to the regulated Cannabis Industry. We
INVESTOR PRESENTATION JUNE 2016 OTCQB:CANN ABOUT General Cannabis Corporation (GCC) is the all-in-one resource for the highest quality service providers available to the regulated Cannabis Industry. We
Novartis announces Phase III STRIVE data published in NEJM demonstrating significant and sustained efficacy of erenumab in migraine prevention
 Novartis International AG Novartis Global Communications CH-4002 Basel Switzerland http://www.novartis.com MEDIA RELEASE COMMUNIQUE AUX MEDIAS MEDIENMITTEILUNG Novartis announces Phase III STRIVE data
Novartis International AG Novartis Global Communications CH-4002 Basel Switzerland http://www.novartis.com MEDIA RELEASE COMMUNIQUE AUX MEDIAS MEDIENMITTEILUNG Novartis announces Phase III STRIVE data
Press Release. RedHill Biopharma Announces Enrollment of Last Patient in the BEKINDA Phase II Study for IBS-D
 Press Release RedHill Biopharma Announces Enrollment of Last Patient in the BEKINDA Phase II Study for IBS-D Top-line results are expected in the third quarter of 2017 The randomized, double-blind, placebo-controlled
Press Release RedHill Biopharma Announces Enrollment of Last Patient in the BEKINDA Phase II Study for IBS-D Top-line results are expected in the third quarter of 2017 The randomized, double-blind, placebo-controlled
Synergy Pharmaceuticals TRULANCE (Plecanatide) Receives U.S. FDA Approval for the Treatment of Adults with Chronic Idiopathic Constipation
 January 19, 2017 Synergy Pharmaceuticals TRULANCE (Plecanatide) Receives U.S. FDA Approval for the Treatment of Adults with Chronic Idiopathic Constipation NEW YORK--(BUSINESS WIRE)-- Synergy Pharmaceuticals
January 19, 2017 Synergy Pharmaceuticals TRULANCE (Plecanatide) Receives U.S. FDA Approval for the Treatment of Adults with Chronic Idiopathic Constipation NEW YORK--(BUSINESS WIRE)-- Synergy Pharmaceuticals
Obstructive Sleep Apnea Syndrome. Common sleep disorder causes high blood pressure and heart attacks
 Obstructive Sleep Apnea Syndrome Common sleep disorder causes high blood pressure and heart attacks Message: Sleep apnea is very common. It is estimated that 158 million people worldwide suffer from sleep
Obstructive Sleep Apnea Syndrome Common sleep disorder causes high blood pressure and heart attacks Message: Sleep apnea is very common. It is estimated that 158 million people worldwide suffer from sleep
Use of Technology in the Assessment of Type 2 Diabetes and Sleep Apnea
 Use of Technology in the Assessment of Type 2 Diabetes and Sleep Apnea Eileen R. Chasens, PhD Associate Professor University of Pittsburgh September 3, 2014 Disclosure I do not own a Smart Phone, I have
Use of Technology in the Assessment of Type 2 Diabetes and Sleep Apnea Eileen R. Chasens, PhD Associate Professor University of Pittsburgh September 3, 2014 Disclosure I do not own a Smart Phone, I have
Treatment of visual impairment due to macular edema secondary to branch retinal vein occlusion (BRVO):
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Treatment of visual impairment due to macular edema secondary to branch retinal
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Treatment of visual impairment due to macular edema secondary to branch retinal
Aviragen Therapeutics, Inc. and Vaxart Inc. Joint Conference Call UNLOCKING THE FULL POTENTIAL OF ORAL VACCINES
 Aviragen Therapeutics, Inc. and Vaxart Inc. Joint Conference Call UNLOCKING THE FULL POTENTIAL OF ORAL VACCINES October 30, 2017 Safe Harbor This presentation contains forward-looking statements about
Aviragen Therapeutics, Inc. and Vaxart Inc. Joint Conference Call UNLOCKING THE FULL POTENTIAL OF ORAL VACCINES October 30, 2017 Safe Harbor This presentation contains forward-looking statements about
Inspire. therapy for sleep apnea. Giving you the freedom to sleep like everyone else
 Inspire therapy for sleep apnea Giving you the freedom to sleep like everyone else Take Comfort. Take Action. Inspire therapy can help. Here are some reasons people like you have chosen Inspire therapy
Inspire therapy for sleep apnea Giving you the freedom to sleep like everyone else Take Comfort. Take Action. Inspire therapy can help. Here are some reasons people like you have chosen Inspire therapy
Oireachtas Joint Committee on Health and Children 7 th February 2013
 Page1 The (ISAT) is a voluntary, non-profit making, non-political advocacy and patient support group for sufferers (and their families) of Sleep Apnoea. ISAT was formed in 2000, following a patient meeting
Page1 The (ISAT) is a voluntary, non-profit making, non-political advocacy and patient support group for sufferers (and their families) of Sleep Apnoea. ISAT was formed in 2000, following a patient meeting
THN. Sleep Therapy Study. ImThera. Information for Participants. Caution: Investigational device. Limited by United States law to investigational use.
 THN Sleep Therapy Study Information for Participants Caution: Investigational device. Limited by United States law to investigational use. ImThera Obstructive sleep apnea (OSA) is a very serious condition.
THN Sleep Therapy Study Information for Participants Caution: Investigational device. Limited by United States law to investigational use. ImThera Obstructive sleep apnea (OSA) is a very serious condition.
Oncology Therapeutics without Compromise APRIL 2011
 Oncology Therapeutics without Compromise APRIL 2011 Forward Looking Statements This presentation contains forward-looking statements that involve substantial risks and uncertainties, including among other
Oncology Therapeutics without Compromise APRIL 2011 Forward Looking Statements This presentation contains forward-looking statements that involve substantial risks and uncertainties, including among other
February 23, Q4 and Year-End 2016 Financial Results
 February 23, 2017 Q4 and Year-End 2016 Financial Results 2 RETHINKING CNS Agenda Today s Speakers Paul Cox, Senior Director, Investor Relations Jeff Jonas, M.D., Chief Executive Officer Jim Doherty, Ph.D.,
February 23, 2017 Q4 and Year-End 2016 Financial Results 2 RETHINKING CNS Agenda Today s Speakers Paul Cox, Senior Director, Investor Relations Jeff Jonas, M.D., Chief Executive Officer Jim Doherty, Ph.D.,
Scientific Discoveries Allow Development of Targeted Therapeutics for Individuals with Autism Spectrum Disorders
 Scientific Discoveries Allow Development of Targeted Therapeutics for Individuals with Autism Spectrum Disorders Randall L. Carpenter, M.D. Co-Founder, President and CEO August 9, 2012 Research Supported
Scientific Discoveries Allow Development of Targeted Therapeutics for Individuals with Autism Spectrum Disorders Randall L. Carpenter, M.D. Co-Founder, President and CEO August 9, 2012 Research Supported
Medical Affairs Policy
 Medical Affairs Policy Service: Sleep Disorder Treatment: Positive Airway Pressure Devices and Oral Appliances (CPAP, BPAP, BiPAP, BiPAP ST, BiPAP with backup, BiPAP -Auto SV, VPAP, VPAP Adapt, VPAP adapt
Medical Affairs Policy Service: Sleep Disorder Treatment: Positive Airway Pressure Devices and Oral Appliances (CPAP, BPAP, BiPAP, BiPAP ST, BiPAP with backup, BiPAP -Auto SV, VPAP, VPAP Adapt, VPAP adapt
Revolutionizing the Treatment of Cancer
 Revolutionizing the Treatment of Cancer September 2013 Safe Harbor Statement The statements that follow (including projections and business trends) are forward looking statements. Rexahn's actual results
Revolutionizing the Treatment of Cancer September 2013 Safe Harbor Statement The statements that follow (including projections and business trends) are forward looking statements. Rexahn's actual results
COMPANY PRESENTATION. Bernard Fortier, CEO
 COMPANY PRESENTATION Bernard Fortier, CEO Forward Looking Statements Some statements in this release may contain forward-looking information. All statements, other than of historical fact, that address
COMPANY PRESENTATION Bernard Fortier, CEO Forward Looking Statements Some statements in this release may contain forward-looking information. All statements, other than of historical fact, that address
STUDY 1 PHASE 3 TOP-LINE RESULTS. September 2017
 STUDY 1 PHASE 3 TOP-LINE RESULTS September 2017 Forward Looking Statement Zogenix cautions you that statements included in this presentation that are not a description of historical facts are forward-looking
STUDY 1 PHASE 3 TOP-LINE RESULTS September 2017 Forward Looking Statement Zogenix cautions you that statements included in this presentation that are not a description of historical facts are forward-looking
Merrill Lynch's Global Pharmaceutical, Biotechnology, and Medical Device Conference. February 7, 2007
 Merrill Lynch's Global Pharmaceutical, Biotechnology, and Medical Device Conference February 7, 2007 Information related to forward-looking statements This presentation includes forward-looking statements
Merrill Lynch's Global Pharmaceutical, Biotechnology, and Medical Device Conference February 7, 2007 Information related to forward-looking statements This presentation includes forward-looking statements
(Nasdaq: VIVE) Advancing the Science of Women s Intimate Health. December 2018
 (Nasdaq: VIVE) Advancing the Science of Women s Intimate Health December 2018 Safe Harbor Statement All statements in this presentation that are not based on historical fact are forward looking statements.
(Nasdaq: VIVE) Advancing the Science of Women s Intimate Health December 2018 Safe Harbor Statement All statements in this presentation that are not based on historical fact are forward looking statements.
2016 Physician Quality Reporting System Data Collection Form: Sleep Apnea (for patients aged 18 and older)
 2016 Physician Quality Reporting System Data Collection Form: Sleep Apnea (for patients aged 18 and older) IMPORTANT: Any measure with a 0% performance rate (100% for inverse measures) is not considered
2016 Physician Quality Reporting System Data Collection Form: Sleep Apnea (for patients aged 18 and older) IMPORTANT: Any measure with a 0% performance rate (100% for inverse measures) is not considered
For personal use only
 Australian Securities Exchange Limited Companies Announcements Office SYDNEY 4 April, 2012 New Australian Drug Developer Lists on ASX Key points Growth-focused Australian drug and therapeutic development
Australian Securities Exchange Limited Companies Announcements Office SYDNEY 4 April, 2012 New Australian Drug Developer Lists on ASX Key points Growth-focused Australian drug and therapeutic development
Sleep Disorders and the Metabolic Syndrome
 Sleep Disorders and the Metabolic Syndrome Tom V. Cloward, M.D. Intermountain Sleep Disorders Center LDS Hospital Objectives Describe how sleep disorders impact your daily medical practice Don Don t do
Sleep Disorders and the Metabolic Syndrome Tom V. Cloward, M.D. Intermountain Sleep Disorders Center LDS Hospital Objectives Describe how sleep disorders impact your daily medical practice Don Don t do
