Influence of melatonin and oestradiol on the opioidergic regulation of LH and prolactin release in pony mares
|
|
|
- Gwendoline Robinson
- 6 years ago
- Views:
Transcription
1 Influence of melatonin and oestradiol on the opioidergic regulation of LH and prolactin release in pony mares C Aurich, J Lange, H-O Hoppen and J E Aurich 'Institut für Reproduktionsmedizin, 2Klinik für Geburtshilfe und Gynäkologie des Rindes and 3 Abteilung für Endokrinologie, Tierärztliche Hochschule, Bünteweg 15, D Hannover, Germany (Requests for offprints should be addressed to C Aurich, Institut für Reproduktionsmedizin, Tierärztliche Hochschule, Bünteweg 15, D Hannover, Germany} Abstract The aim of this study was to investigate the influence of oestradiol, melatonin and season on the opioid regulation oflh and prolactin release. Effects of the opioid antagonist naloxone (0\m=.\5mg/kg) on LH and prolactin secretion were determined in ovariectomized pony mares. In experiment 1, mares in January (n=6) were pretreated with oestradiol benzoate (5 \g=m\g/kg)for 20 days. In experiment 2, beginning in May, mares (n=7) received melatonin (15 mg) for 15 days and subsequently a combination of melatonin plus oestradiol for 20 days. In experiment 3, beginning in May, mares (n=6) were pretreated with oestradiol for 30 days, left untreated for 12 days and then given melatonin for 35 days. In all experiments the animals were injected with the opioid antagonist naloxone and saline on 2 consecutive days prior to treatment. In experiment 1, animals received naloxone and saline on days 10 and 11 and 20 and 21 following oestradiol treatment. In experiment 2, naloxone and saline were administered on days 15 and 16 following melatonin treatment and on days 10 and 11 and 20 and 21 of melatonin plus oestradiol treatment. In experiment 3, the animals received naloxone and saline on days 10 and 11, 20 and 21 and 30 and 31 of oestradiol treatment, prior to melatonin treatment and on days 15 and 16, 25 and 26 and 35 and 36 following melatonin. In January (experiment 1), naloxone evoked a significant (P<0\m=.\05)LH release at all times, however the LH increment in response to naloxone increased during oestradiol pretreatment (P<0\m=.\05)During the breeding season (experiments 2 and 3), naloxone induced a significant (P<0\m=.\05)increase in plasma pretreated LH concentrations when mares had not been with oestradiol or melatonin and after oestradiol pretreatment. Basal LH concentrations and the LH increment in response to naloxone increased significantly (P<0\m=.\05)during the 30-day oestradiol pretreatment. Melatonin decreased the naloxone-induced LH release and the LH release in response to naloxone and saline no longer differed after 25 and 35 days of melatonin pretreatment. When melatonin was given together with oestradiol for 20 days, again a significant (P<0\m=.\05)LH release in response to naloxone occurred. Prolactin release was significantly (P<0\m=.\05)increased by naloxone when mares had been pretreated with only melatonin. The opioid antagonist did not affect prolactin release in mares that had not been pretreated or received oestradiol either alone or in combination with melatonin. In conclusion, in long\x=req-\ term ovariectomized mares, opioids inhibit LH secretion inhibition of independent from ovarian factors. This opioid LH secretion is enhanced by oestradiol and reduced by melatonin. Although short-term melatonin treatment in\x=req-\ activates the opioid regulation of LH release, a prolonged influence of melatonin as occurs in winter does not prevent activation of the opioid system. This indicates that effects of melatonin on the opioid regulation of LH release change with time. An opioid inhibition of prolactin secretion is activated by melatonin given for 15\p=n-\35days but is lost under the prolonged melatonin signal in winter. Journal of Endocrinology (1997) 154, 241\p=n-\248 influence of a short-day Introduction Reproductive activity in the horse changes throughout the year and the physiological breeding season lasts from approximately March to October. The reduction in ovar ian function during the winter months is primarily caused by a decrease in gonadotrophic stimulation. In female and male horses, endogenous opioids participate in the regu lation of luteinizing hormone (LH) release as inhibitory neuromodulators (Behrens et al. 1993, Aurich et al. 1994!, > 1995). In mares, the opioid antagonist naloxone causes an increase in plasma LH concentrations during the luteal but not the follicular phase of the cycle (Behrens et al. 1993) and during the anovulatory season in winter (Aurich et al. 1994a). It has therefore been suggested that opioidergic mechanisms inhibit LH release during seasonal anoestrus and that transition into the breeding season coincides with a switch from a continuous opioidergic
2 inhibition to an intermittent, cycle-dependent blockade of LH release (Aurich et al. 1994a). In cyclic mares, the opioidergic inhibition of LH secretion is most likely activated by a sequence of oestrogen and progesteronedominated steroid environments, characteristic for the oestrous cycle but, in ovariectomized mares, could also be stimulated by oestradiol alone (Aurich et al. 1995). In contrast to the breeding season, the opioidergic systems regulating LH release in seasonally anovulatory mares do not depend on progesterone. In these animals, the opioid ergic inhibition of LH secretion might be activated by continuous exposure to low oestrogen concentrations or independent from ovarian factors (Aurich et al ). Besides regulating LH release, opioids inhibit prolactin secretion in stallions (Aurich et al. 1994fr) and in ovariec tomized, steroid-treated mares (Aurich et al. 1995). Plasma prolactin concentrations in the horse are low in winter and increase during the breeding season (Worthy et at. 1987). Circannual changes in prolactin release, as in other species, are largely regulated by photoperiod (Johnson 1987). Photoperiodic stimuli are transmitted into changes in melatonin release from the pineal gland and melatonin directly or indirectly inhibits gonadotrophin-releasing hormone (GnRH) synthesis (Strauss et al. 1979). Effects of melatonin on plasma prolactin concentrations have not yet been investigated in the equine species. We have therefore investigated effects of oestradiol, melatonin and season on the opioidergic regulation of LH and prolactin release in ovariectomized mares. Materials and Methods Animals The animals used in this study (n=7) were non-lactating Shetland pony mares, aged between 4 and 12 years (7-6 ± 2-7 years, ± s.d.) and weighing ±8-0 kg ( ± s.d.). Ponies were kept in outdoor paddocks and fed hay and mineral supplements. Water was freely available. The mares had been ovariectomized by laparotomy using standard surgical procedures between 4 and 6 months before the first experiment. Experimental design The animals were used for three different experiments carried out over a 19-month period. Experiment 1 was performed in January and experiments 2 and 3 between May and July during 2 consecutive years. In experiment 1, mares (n=6) were first treated i.v. with naloxone (0-5 mg/kg body weight) and saline on 2 con secutive days to determine the effect of naloxone on LH and prolactin secretion in non steroid-treated, ovari ectomized mares outside the breeding season. Half of the mares were given naloxone on day 1 and saline on day 2 and the other half were treated in opposite order. Starting on day 2 after naloxone or saline experiments, mares received oestradiol benzoate (5 µg/kg body weight, s.c.) for 20 days once daily. On days 10 and 11 and days 20 and 21, the animals were again injected with naloxone and saline respectively. Oestradiol was always given in the afternoon and thus after naloxone or saline experiments. In experiment 2, mares (n=7) were treated with naloxone and saline on days 1 and 2 as described for experiment 1. After the naloxone/saline experiment on day 2, mares received melatonin (15 mg) for 15 days. Melatonin was given orally at 1530 h approximately 4 h before dusk. This treatment has been shown to advance the daily rise in plasma melatonin concentrations and thus imitates a short-day photoperiodic signal (Guillaume & Palmer 1991). Naloxone and saline experiments were repeated after 15 and 16 days. The mares were then given melatonin (15 mg orally) plus oestradiol benzoate (5 µg/kg body weight, s.c.) for 20 days and on days 10 and 11 and days 20 and 21 received naloxone and saline. One animal, for reasons not related to the experiment, had to be excluded from the study after day 15 of the combined melatonin plus oestradiol treatment. In experiment 3, mares (n=6) received naloxone and saline on 2 consecutive days to determine opioidergic effects on LH and prolactin secretion in non-pretreated, ovariectomized mares during the breeding season. Starting on day 2, mares received oestradiol benzoate (5 µg/kg body weight, s.c.) for 30 days once daily. Oestradiol was always injected after naloxone or saline experiments. On days 10 and 11, days 20 and 21 and days 30 and 31 after the start of oestradiol pretreatment, the animals received naloxone and saline as described for days 1 and 2. They were then left untreated for 12 days and subsequently pretreated with melatonin (15 nig orally) for 35 days. The LH and prolactin response to naloxone and saline was determined on the 2 days immediately before melatonin treatment, on days 15 and 16, days 25 and 26 and days 35 and 36 after the start of melatonin pretreatment with half of the mares receiving naloxone on the first day and saline on the second day and the other half treated in opposite order. Experimental procedures For each naloxone or saline injection an indwelling cath eter was placed in one jugular vein 30 min before the first blood sample was taken. Blood samples for determination of LH and prolactin were then collected at 15-min intervals for 3 h. After 60 min sampling, naloxone or 10 ml saline were injected via the catheter into the jugular vein. Blood was collected into heparinized polystyrol tubes. Samples were centrifuged immediately for 20 min at 1000 g and plasma was frozen at 20 C until hormone analysis. Naloxone (Sigma, Deisenhofen, Germany) was freshly dissolved in 10 ml saline and filter sterilized before
3 injection. Oestradiol benzoate (Sigma) was dissolved in corn oil at a concentration of 1 g/1 and administered s.c. at 24-h intervals. Melatonin (Sigma) was dissolved in ethanol at a concentration of 30-0 g/1 and 0-5 ml solution were given on 5 g sugar cubes. After evaporation of ethanol at room temperature, the cubes were stored at 20 C. The ponies were fed one melatonin-containing sugar cube per day. Hormone analysis Plasma concentrations of LH and prolactin were measured by RIA as described previously. The assay for LH utilized equine LH (UCB, Braine L'Alleud, Belgium) as standard and for iodination and an antibody raised in rabbits against equine LH (A 543, UCB). Intra- and interassay coef ficients of variation (CV) were 5-8 and 18-1% respectively; the minimal detectable concentration was 0-5 µg/l and cross-reactivity of the antibody with follicle-stimulating hormone was <2-8% (Behrens et al. 1993). The RIA for prolactin was performed with an antiserum raised in rabbits against equine prolactin (AFP ; Dr A F Parlow, Harbor-UCLA Medical Center, Torrance, CA, USA). Intra- and interassay CV values were 4-1 and 10-3% respectively; the minimal detectable concentration was 0-25 µg/l and cross-reactivity of the antibody with equine growth hormone (AFP 7424C, Dr A F Parlow) was <0-l% (Aurich et al. 1995). Statistical analysis Prolactin and LH release in individual mares was calcu lated as the area under the curve (AUC, µ /1/15 min) for the time period from 0 to 120 min after injection of naloxone or saline. The unit takes into account the 15-min sampling interval. The pretreatment baseline was sub tracted from the value for each post-treatment sample. For LH, the baseline was calculated as the mean of five values before naloxone or saline injections and for prolactin as the mean of the last three values before naloxone or saline injections. The first two samples were excluded for calculating basal prolactin concentrations in order to mini mize effects of a possible stress-induced prolactin release in association with i.v. catheterization. Statistical comparisons were made with the SPSS/PC+ statistics package (Norusis 1988). Because no assumption was made about the distri bution of data, non parametric tests were used. Naloxone and respective saline experiments were compared by Wilcoxon matched pairs ranked sum test. Basal hormone concentrations in the same animals at different times and naloxone-induced LH and prolactin release (AUC) after different oestradiol and melatonin pretreatments were compared by Friedman's two-way ANOVA, taking into account the sequential nature of data. For comparisons of basal hormone concentrations, values from control experiments were used. Data given are means ± s.e.m. Results Oestradiol As can be expected from the experimental design, plasma oestradiol concentrations were affected by the oestradiol pretreatment. Oestradiol concentrations were significantly with oestradiol increased when mares had been pretreated benzoate, whereas melatonin treatment did not cause any changes (see Table 1). LH In January (experiment 1), basal LH was not significantly affected by the oestradiol pretreatment (see Table 1). Naloxone induced a significant ( <0 5) release of LH both before and following 10 and 20 days of pretreatment with oestradiol, when the AUC was compared with the respective saline experiment (see Fig. la). The release of LH in response to naloxone (AUC), was significantly ( <0 5) higher when mares had been given oestradiol for 10 and 20 days than without steroid pretreatment. After 10 days of oestradiol pretreatment, maximal LH concen trations in response to naloxone were reached within 15 min (naloxone 32-4 ±11-7 vs saline 22-1 ± 10-1 µg/l) and alter 2(1 days of oestradiol pretreatment within 30 min (naloxone 50-5 ± 28-0 vs saline 32-5 ± 17-3 ng/1). In May (experiment 2), basal plasma LH concentrations were not affected by melatonin pretreatment for 15 days but increased significantly (P<001) following the com bined oestradiol plus melatonin pretreatment (see Table 1). Naloxone induced a significant (P<0-05) LH release, when mares had neither been pretreated with melatonin nor with oestradiol. After oral melatonin pretreatment for 15 days and after subsequent combined melatonin and oestradiol pretreatment for 10 days, naloxone did not significantly increase LH release. However, when the combined treatment was continued for an additional 10 days, naloxone injection was again followed by a signifi cant (P<0-05) LH release. The LH increment in response to naloxone (AUC) was significantly higher when mares had been pretreated with melatonin for 15 days followed by melatonin plus oestradiol benzoate for 20 days than in mares not pretreated ( <0 01; see Fig. 2d). Maximal LH concentrations in mares not pretreated with melatonin and oestradiol were 19-0 ± 3-3 and 14-9 ± 3-1 µg/l respect ively, 30 min after injection of naloxone and saline. After the combined 20-day melatonin plus oestradiol pre treatment maximal LH concentrations 15 min after naloxone injection were 47-4 ± 4 µg/l, compared with 37-4 ± 4-4 µg/l in saline experiments. In experiment 3, during the physiological breeding season of the horse, basal LH concentrations increased continuously during the 30-day oestradiol pretreatment (P<0-001). In contrast, melatonin pretreatment decreased basal LH concentrations when compared with the
4 Table 1 Basal LH, prolactin and oestradiol concentrations n plasma of mares before injections of naloxone and saline. Significant differences n LH, prolactin and oestradiol concentrations respectively, within Individual experiments are indicated in parentheses (ANOVA). ''^Significant differences between experiments when LH, prolactin and oestradiol concentrations respectively, before any melatonin or oestradiol treatment was initiated, were compared among experiments (ANOVA) Experiment No pretreatment Oestradiol for 10 days Oestradiol for 20 days No pretreatment Melatonin for 1 5 days Melatonin for 25 days + oestradiol for 10 days Melatonin for 35 days + oestradiol for 20 days No pretreatment Oestradiol for 10 days Oestradiol for 20 days Oestradiol for 30 days No pretreatment Melatonin for 1 5 days Melatonin for 25 days Melatonin for 35 days LH (µ8/ ) 9-9 ± 1-3a 20-1 ± ± ±3-2b ± ± ±3-3 (P<0-01) 15-0±2-4b 39-1 ± ± ± (P<0-001) 30-3 ± 6-0b 23-5 ± ± ±3-l (P<0-01) Prolactin (µß/ ) 2-8 ± ± ± ±0-3a 1-0 ± ± ±0-1 (P<0-05) 5-0 ± 0-9b 6-6 ± ± ± ± ± ± ±0-7 (P<0-05) Oestradiol (pmol/l) 29-0 ± ± ± 4-0 (P<0-01) 36-3 ± ± ± ±9-5 (P<0-05) 27-2 ± ± ± ± 7-3 (P<0-05) 13-6 ± ± ± ±5-1 Values within columns with different superscripts are significantly (P<0-05) different. 300-, (a) < 3 " ra o ri 10-0 /» 10dE2 20dE2-10dE2 20dE2 Figure 1 (a) LH and (b) prolactin release calculated as area under the curve (AUC; ug/l/1 5 min) for the time period from 0 to 120 min after administration of naloxone (0-5 mg/kg; solid bars) or saline (open bars) in ovariectomized mares (ovx, n=6-7) during the nonbreeding season ± oestradiol benzoate (E2/ 5 µ /kg s.c.) pretreatment for 10 or 20 days. Values are means ± s.e.m. 'Significant difference between naloxone and respective saline experiments (P<0-05). pre-melatonin period ( <0 01; see Table 1). Injection of naloxone on day 1 and 2 (no pretreatment) resulted in a significant (P<0-05) release of LH, calculated as AUC. Mean maximal plasma LH concentrations 15 min after of naloxone and saline were 13-7 ±2-1 and injection 15-0 ±2-1 pg/1 respectively. Oestradiol pretreatment for 10, 20 and 30 days increased the naloxone-induced LH increment (P<0-05; see Fig. 2b). When mares had been pretreated with oestradiol for 30 days, LH concentrations 15 min after injection of naloxone and saline were ± 21-9 and 95-3 ± 15-7 µg/l respectively. In con trast, melatonin pretreatment markedly reduced the LH response to naloxone. A significant difference between naloxone and saline experiments was found after 15 days but was no longer present after 25 and 35 days of melatonin pretreatment (see Fig. 2c).
5 75 (a) 50 (d) 50- i* * I. 40 < ^ 1 20 J! o a. 10- o- ^à^acm -25- ovx 15dM 25dM+E2 35d M+E dM 25dM+E2 35d M+E2 300 (b) 50- ir! 200- I" 100- I * 40 O Ü 30 c V 20 jo o rx 10- aill ovx 10dE2 20dE2 30d E dE2 20dE2 30d E2 75- (c) 50 (0 8< Jcâie H - 20 Q. 10- III -25- ovx 15dM 25dM 35d M dM 25dM 35d M Figure 2 (a-c) LH and (d-f) prolactin release calculated as area under the curve (AUC; µg/l/15 min) for the time period from 0 to 120 min after administration of naloxone (0-5 mg/kg; solid bars) or saline (open bars) in ovariectomized mares (ovx, n=6-7) during the breeding season ± pretreatment over different time periods with (b, e) oestradiol benzoate (E2, 5 µg/kg s.c), (c, f) melatonin (M; 15 mg) or (a, d) E2+M. Values are means ± s.e.m. 'Significant difference between naloxone and respective saline experiments (P<005). Prolactin Basal prolactin concentrations were not affected by oestra diol pretreatment (experiments 1 and 3) but were signifi cantly (P<0-05) reduced by either melatonin (experiment 2) or melatonin combined with oestradiol (experiment 1; see Table 1). In experiment 1 (January), naloxone did not cause any changes in plasma prolactin concentrations, irrespective of any oestradiol pretreatment (see Fig. lb). In experiment 2,
6 during the breeding season, naloxone failed to induce prolactin release in mares not pretreated with melatonin or oestradiol. After oral melatonin pretreatment for 15 days, naloxone caused a significant ( <0 5) prolactin release, compared with AUC values from respective saline exper iments. Prolactin concentrations 30 min after naloxone or saline injections were 2-1 ±0-6 and 1-2 ±0-4 pg/1 respectively. No naloxone-inducible prolactin secretion was found after the combined melatonin and oestradiol pretreatment for 10 and 20 days (see Fig. 2d). In exper iment 3, injection of naloxone on days 1 and 2 (no pretreatment) again did not significantly change prolactin release. Oestradiol pretreatment for 10, 20 and 30 days had no effect on the prolactin response to naloxone (see Fig. 2e). After melatonin pretreatment for 15, 25 and 35 days, naloxone evoked a significant prolactin secretion. Although the naloxone-induced increment in prolactin release tended to increase during the melatonin pretreat ment, no significant difference was found. Prolactin con centrations 15 min after naloxone and saline injections were 3-9 ± 0-6 and 2-7 ± 0-4 pg/1 after 15 days, 5-5 ± 1-4 and 2-2 ± 0-5 pg/1 after 25 days and 5-7 ±1-3 and 23 ± 0-6 pg/1 respectively, after 35 days of melatonin pretreatment (see Fig. 2/). Discussion In this study, effects of oestradiol and melatonin on the opioidergic regulation of LH and prolactin release were investigated during the breeding and non-breeding season of the horse. In May, oestradiol pretreatment caused an increase in basal LH concentrations, reflecting a stimula tory effect of oestradiol on LH secretion previously described (Garcia & Ginther 1978, Baldwin et al. 1991, Thompson et al. 1991). This stimulation is exerted primar ily at the pituitary level (Baldwin et al. 1991). Melatonin pretreatment for up to 35 days beginning in May did not affect LH but decreased prolactin concentrations. In the horse, as in sheep (Kennaway et al. 1982, Lincoln & Ebling 1985) melatonin therefore is able to inhibit prolactin secretion. Before oestradiol pretreatment, LH concentrations in January were significantly lower than corresponding values in May, which is in agreement with the seasonal variation in LH release described previously (Turner et al. 1979). Animals in our study had been ovariectomized several months earlier. As has been suggested for sheep (Goodman et al. 1982, Robinson et al. 1985), seasonal changes in LH release in the horse seem to be regulated at least in part independently from ovarian factors. In experiment 3, LH concentrations before melatonin pretreatment tended to be higher than before oestradiol pretreatment. Although plasma oestradiol concentrations were not markedly different, the 12-day interval between oestradiol and melatonin pretreatments might have been too short, and residual, LH-stimulatory oestrogen effects before the melatonin treatment cannot be ruled out. Basal prolactin concentrations in experiment 3 were lower than in exper iment 2 although the time of year and experimental procedures were identical. Because experiment 3 was performed 12 months later than experiment 2, the differ ence may be linked to long-term changes in prolactin release after ovariectomy. In ovariectomized mares not pretreated with oestradiol or melatonin, naloxone caused a significant LH release, irrespective of the time of the year. This indicates, that in long-term ovariectomized pony mares, opioids inhibit LH secretion and is in contrast to results from short-term ovariectomized mares (Aurich et al. 1995). Therefore, an inhibitory opioidergic tone on LH release exists in ovaryintact mares (Behrens et al. 1993), is lost after ovariectomy (Aurich et al. 1995) but can be restored after prolonged absence of the ovaries. This is in agreement with results from ewes (Schillo et al. 1985) and female rabbits (Younglai et al. 1988), where a steroid-independent opioidergic inhibition of LH release exists under certain conditions. Melatonin pretreatment during the markedly reduced the opioidergic breeding season inhibition of LH secre tion. While LH concentrations still tended to increase in response to naloxone after 15 days of melatonin pretreat ment, no naloxone-reversible, i.e. opioidergic, inhibition was found after 25 and 35 days of melatonin. It is therefore unlikely that a prolonged period of daily melatonin secre tion at the end of the breeding season in autumn activates the opioidergic inhibition of LH release found in seasonally anovulatory mares (Aurich et al. 199Ad). In contrast, in non oestradiol-pretreated mares in winter, exposed to a natural short-day melatonin signal for several -weeks, an opioid ergic inhibition of LH release was present. Although short-term, i.e. 15- to 35-day, melatonin pretreatment inactivates the opioidergic regulation of LH release, the natural, prolonged influence of a short-day melatonin signal does not prevent reactivation of the opioidergic systems. The opioidergic regulation of LH release there fore seems to be influenced not only by short-term melatonin effects but also by the photoperiodic history of the animal. The exogenous melatonin signal given during the breeding season may also not be totally equivalent the endogenous profile to observed under natural winter days. The melatonin treatment used in our study has been shown to elevate plasma melatonin concentrations for several hours until after the onset of endogenous melatonin release at dusk (Guillaume & Palmer 1991). Because melatonin was always given at the same hour, gradual changes in the period of daily melatonin release as seen in winter were not imitated. Differences in the of LH release between melatonin- opioidergic regulation pretreated mares in summer and non-pretreated mares in winter may reflect in part a different pattern of melatonin signals.
7 In winter as well as during the breeding season, an opioidergic inhibition of LH release was present under the influence of exogenous oestradiol and the LH increment in response to naloxone increased with the duration of oestradiol treatment. This indicates that the opioidergic inhibition of GnRH and thus LH secretion can be enhanced by oestrogens. An oestradiol-dependent acti vation of LH-inhibitory opioidergic systems does exist also in short-term ovariectomized mares, pretreated for 8 days with oestradiol (Aurich et al. 1995). However, in that study, mares were investigated only in the breeding season and plasma oestradiol concentrations resembled values found in ovary-intact mares during the follicular phase of the cycle. Oestradiol concentrations in the present study were lower and comparable to those physiologically seen in mares with reduced ovarian activity outside the breed ing season (Aurich et al. 1994a). The opioidergic inhibition oflh release in seasonally anovulatory mares (Aurich et al. 1994a) therefore is in part steroid-independent enhanced by low oestradiol concentrations. Oestrogens but can be may be part of a negative feed-back loop acting on GnRH and thus LH secretion in non-breeding season mares as has been found also in sheep (Karsch et al. 1993). In the horse, these feedback mechanisms seem to involve opioidergic neuronal pathways. When melatonin and oestradiol were given combined, the inactivating effect of melatonin mechanisms was alone on the LH regulatory opioidergic reversed by oestradiol and an inhibitory opioidergic tone reactivated. An oestrogen-induced, opioidergic inhibition of GnRH secretion does not exclude stimulatory effects of oestradiol on LH release at the pituitary gland, explaining increased pre-naloxone LH concentrations in plasma of mares pretreated with oestradiol. Prolactin release is in part under opioidergic control but its regulation is different from the mechanisms regulating LH release. No naloxone-induced changes in prolactin secretion were found in non-pretreated, ovariectomized mares, irrespective of the season. Melatonin pretreatment during the breeding season induced an opioidergic inhi bition of prolactin secretion and the amount of prolactin secreted in response to the opioid antagonist increased with the duration of melatonin pretreatment. Although melatonin given for days activated an opioidergic inhibition of prolactin release, the secretion of this hor mone was not under opioidergic control in mares in winter, exposed for a prolonged time period to an endog enous short-day melatonin signal. This suggests that al though a short-day melatonin signal initially activates an opioidergic inhibition of prolactin release, activity of the opioidergic systems changes with time. Opioids therefore could participate in a reduction in prolactin secretion in autumn but do not inhibit prolactin release throughout the non-breeding season. As in sheep, a prolonged inhibitory effect of short daylength on prolactin secretion could also be mediated through non-opioidergic mechanisms directly at the pituitary gland (Lincoln & Clarke 1994, 1995, Houghton et al. 1995) and such mechanisms would mask an opioidergic inhibition at the hypothalamic level. In conclusion, opioidergic neuronal systems, involved in the regulation of LH and prolactin secretion in the mare, interact with both melatonin and oestradiol. However, although seasonal variations in prolactin and LH release in the horse occur in parallel (Turner et al. 1979, Evans et al. 1991), the opioidergic regulation of these hormones is activated differentially. Given during the breeding melatonin inactivates the opioidergic inhibition season, of LH secretion but a prolonged influence of melatonin as in winter does not prevent reactivation of opioidergic systems. Irrespective of the season, the naloxone-reversible inhibition of LH release is enhanced by oestradiol, suggesting that the opioidergic regulation of LH release in seasonally anovulatory mares might in part be activated by low oestradiol concentrations. Oestradiol is also able to counteract the effects of melatonin on the LH regulatory opioidergic pathways. An opioidergic inhibition of prolactin release can be activated by melatonin but is lost under the prolonged influence of an endogenous short-day melatonin signal. Acknowledgements The authors are grateful to the Mehl-Miilhens Foundation for financial support, the Dorothea Erxleben Program of the State of Niedersachsen for a research fellowship to Dr C Aurich and to Dr A F Parlow, Harbor-UCLA Medical Center, Torrance, CA, USA, for reagents used in the prolactin RIA. References Aurich C, Schlote S, Hoppen H-O, Klug E, Hoppe H & Aurich JH 1994(3 Effects of the opioid antagonist naloxone on release of luteinizing hormone in mares during the anovulatory season. Journal of Endocrinology Aurich C, Sieme H, Hoppe H & Schlote S 1994b Involvement of of LH and testosterone release endogenous opioids in the regulation in the male horse. Journal of Reproduction and Fertility Aurich C, Daels PF, Ball BA & Aurich JE 1995 Effects of gonadal steroids on the opioid regulation of LH and prolactin release in ovariectomized pony mares. Journal of Endocrinology Baldwin DM, Roser JF, Muyan M, Lasley & Dybdal 1991 Direct effects of free and conjugated steroids on GnRH stimulated LH release in cultured equine anterior pituitary cells. Journal of Reproduction and Fertility Suppl Behrens C, Aurich JE, Klug E, Naumann & Hoppen H-O 1993 Inhibition of gonadotrophin release in mares during the luteal phase of the oestrous cycle by endogenous opioids. Journal of Reproduction and Fertility Evans MJ, Alexander SL, Irvine CHG, Livesay JH & Donald RA 1991 In vitro and in vivo studies of equine prolactin secretion throughout the year. Journal of Reproduction and Fertility Suppl and season on Garcia MC & Ginther OJ 1978 Effects of ovariectomy plasma luteinizing hormone in mares. Endocrinology
8 Goodman RL, Bittman EL, Foster DL & Karsch FJ 1982 Alterations in the control of luteinizing hormone pulse frequency underlie the seasonal variation in estradiol negative feedback in the ewe. Biology of Reproduction Guillaume D & Palmer E 1991 Effect of oral melatonin on the date of the first ovulation after ovarian inactivity in mares under artificial photoperiod. Journal of Reproduction and Fertility Suppl Houghton DC, Young IR & McMillen IC 1995 Response of prolactin to different photoperiods after surgical disconnection of the hypothalamus and pituitary in sheep fetuses. Journal of Reproduction and Fertility Johnson AL 1987 Seasonal and photoperiod-induced changes in serum prolactin and pituitary responsiveness to thyrotropin releasing hormone in the mare. Proceedings of the Society for Experimental Biology and Medicine Karsch FJ, Dahl GE, Evans NP, Manning JM, Mayfield KP, Moenter SM & Foster DL 1993 Seasonal changes in gonadotrophin-releasing hormone secretion in the ewe: alteration in response to the negative feedback action of estradiol. Biology of Reproduction Kennaway DJ, Gilmore TA & Seamark RF 1982 Effect of melatonin implants on the circadian rhythm of plasma melatonin and prolactin in sheep. Endocrinology Lincoln GA & Ebling FJP 1985 Effect of constant-release implants of melatonin on seasonal cycles in reproduction, prolactin secretion and moulting in rams. Journal of Reproduction and Fertility Lincoln GA & Clarke IJ 1994 Photoperiodically induced cycles in the secretion of prolactin in hypothalamo-pituitary disconnected rams: evidence for translation of the melatonin signal in the pituitary gland. Journal of Neuroendocrinology Lincoln GA & Clarke IJ 1995 Evidence that melatonin acts at the mechanism to pituitary gland through a dopamine-independent mediate effects of daylength on the secretion of prolactin in the ram. Journal of Neuroendocrinology Norusis MJ 1988 SPSS/PC+ for the IBM PC/XT/AT and PS2. SPSS Ine, Chicago. Robinson JE, Radford HM & Karsch FJ 1985 Seasonal changes in pulsatile luteinizing hormone (LH) secretion in the ewe, relationship of frequency of LH pulses to day length and response to estradiol negative feedback. Biology of Reproduction Schillo KK, Kuehl D & Jackson GL 1985 Do endogenous opioid peptides mediate the effects of photoperiod on release of LH and prolactin in ovariectomized ewes? Biology of Reproduction Strauss SS, Chen CL, Kalra SP & Sharp DC 1979 Localization of gonadotropin releasing honnone in the hypothalamus of ovariectomized pony mares. Journal of Reproduction and Fertility Suppl Thompson DL, Johnson L, Garza F, St George L, Rabb MH, Barry BE Sc French DD 1991 Relationships among LH, FSH and prolactin secretion, storage and response to secretagogue and hypothalamic GnRH content in ovariectomized pony mares administered testosterone, dihydrotestosterone, estradiol, progesterone, dexamethasone or follicular fluid. Domestic Animal Endocrinology Turner DD, Garcia MC & Ginther OJ 1979 Follicular and gonadotropic changes throughout the year in pony mares. American Journal of Veterinary Research Worthy K, Colquhoun K, Escreet R, Dunlop M, Renton JP & Douglas TA 1987 Plasma prolactin concentrations in non-pregnant mares at different times of the year and in relation to events in the cycle. Journal of Reproduction and Fertility Suppl Younglai EV, Wilkinson M & Thompson 1988 Effects of ovariectomy and estradiol replacement on naloxone induced LH secretion in the female rabbit. Journal of Steroid Biochemist^ Received 4 December 1995 Revised manuscript received 25 November 1996 Accepted 27 February 1997
Growth of small follicles and concentrations of FSH during the equine oestrous cycle
 Growth of small follicles and concentrations of FSH during the equine oestrous cycle O. J. Ginther and D. R. Bergfelt Department of Animal Health and Biomedicai Sciences, Veterinary Science Building, University
Growth of small follicles and concentrations of FSH during the equine oestrous cycle O. J. Ginther and D. R. Bergfelt Department of Animal Health and Biomedicai Sciences, Veterinary Science Building, University
Concentrations of Circulating Gonadotropins During. Various Reproductive States in Mares
 BIOLOGY OF REPRODUCTION, 744-75 (19) Concentrations of Circulating Gonadotropins During Various Reproductive States in Mares KURT F. MILLER, S. L. BERG, D. C. SHARP and. J. GINTHER Department of Veterinary
BIOLOGY OF REPRODUCTION, 744-75 (19) Concentrations of Circulating Gonadotropins During Various Reproductive States in Mares KURT F. MILLER, S. L. BERG, D. C. SHARP and. J. GINTHER Department of Veterinary
Proceedings of the Annual Resort Symposium of the American Association of Equine Practitioners AAEP
 www.ivis.org Proceedings of the Annual Resort Symposium of the American Association of Equine Practitioners AAEP Jan. 19-21, 2006 Rome, Italy www.ivis.org Reprinted in the IVIS website with the permission
www.ivis.org Proceedings of the Annual Resort Symposium of the American Association of Equine Practitioners AAEP Jan. 19-21, 2006 Rome, Italy www.ivis.org Reprinted in the IVIS website with the permission
Inhibition of gonadotrophin release in mares during the luteal phase of the oestrous cycle by endogenous opioids
 Inhibition of gonadotrophin release in mares during the luteal phase of the oestrous cycle by endogenous opioids C. Behrens, J. E. Aurich, E. Klug, H. Naumann and H-O. Hoppen 1 Klinik für Geburtshilfe
Inhibition of gonadotrophin release in mares during the luteal phase of the oestrous cycle by endogenous opioids C. Behrens, J. E. Aurich, E. Klug, H. Naumann and H-O. Hoppen 1 Klinik für Geburtshilfe
during the ensuing pregnancy in mares
 Effect of GnRH treatment during the anovulatory season on multiple ovulation rate and on follicular development during the ensuing pregnancy in mares O. J. Ginther and D. R. Bergfelt University of Wisconsin-Madison,
Effect of GnRH treatment during the anovulatory season on multiple ovulation rate and on follicular development during the ensuing pregnancy in mares O. J. Ginther and D. R. Bergfelt University of Wisconsin-Madison,
Changes in LH pulse frequency and amplitude in intact
 Changes in LH pulse frequency and amplitude in intact mares during the transition into the breeding season B. P. Fitzgerald, K. J. Affleck, S. P. Barrows, W. L. Murdoch, K. B. Barker and R. G. Loy Department
Changes in LH pulse frequency and amplitude in intact mares during the transition into the breeding season B. P. Fitzgerald, K. J. Affleck, S. P. Barrows, W. L. Murdoch, K. B. Barker and R. G. Loy Department
Changes in Patterns of Luteinizing Hormone Secretion Before and After the First Ovulation in the Postpartum Mare
 BIOLOGY OF REPRODUCTION 33, 316-33 (1985) Changes in Patterns of Luteinizing Hormone Secretion Before and After the First Ovulation in the Postpartum Mare BARRY P. FiTZGERALD, 3 HELEN I ANSON, SANDRAJ.
BIOLOGY OF REPRODUCTION 33, 316-33 (1985) Changes in Patterns of Luteinizing Hormone Secretion Before and After the First Ovulation in the Postpartum Mare BARRY P. FiTZGERALD, 3 HELEN I ANSON, SANDRAJ.
Treatment 3 Days After Ovulation In Mares
 Luteal Regression And Follicle Development Following Prostaglandin-F 2α Treatment 3 Days After Ovulation In Mares D.R. Bergfelt a, R.A. Pierson b, and O.J. Ginther a a University of Wisconsin, Madison,
Luteal Regression And Follicle Development Following Prostaglandin-F 2α Treatment 3 Days After Ovulation In Mares D.R. Bergfelt a, R.A. Pierson b, and O.J. Ginther a a University of Wisconsin, Madison,
Modulation of luteinizing hormone and folliclestimulating
 University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Faculty Papers and Publications in Animal Science Animal Science Department 1992 Modulation of luteinizing hormone and folliclestimulating
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Faculty Papers and Publications in Animal Science Animal Science Department 1992 Modulation of luteinizing hormone and folliclestimulating
Follicle profile and plasma gonadotropin concentration in pubertal female ponies
 Brazilian Journal of Medical and Biological Research (0) 37: 913-9 Puberty in female pony ISSN 00-79X 913 Follicle profile and plasma gonadotropin concentration in pubertal female ponies Departamento de
Brazilian Journal of Medical and Biological Research (0) 37: 913-9 Puberty in female pony ISSN 00-79X 913 Follicle profile and plasma gonadotropin concentration in pubertal female ponies Departamento de
Seasonality in mares
 Ž. Animal Reproduction Science 60 61 2000 245 262 www.elsevier.comrlocateranireprosci Seasonality in mares P. Nagy ), D. Guillaume, P. Daels Department of Physiology of Reproduction in Domestic Mammals,
Ž. Animal Reproduction Science 60 61 2000 245 262 www.elsevier.comrlocateranireprosci Seasonality in mares P. Nagy ), D. Guillaume, P. Daels Department of Physiology of Reproduction in Domestic Mammals,
REPRODUCTION & GENETICS. Hormones
 REPRODUCTION & GENETICS Hormones http://www.youtube.com/watch?v=np0wfu_mgzo Objectives 2 Define what hormones are; Compare and contrast the male and female hormones; Explain what each hormone in the mail
REPRODUCTION & GENETICS Hormones http://www.youtube.com/watch?v=np0wfu_mgzo Objectives 2 Define what hormones are; Compare and contrast the male and female hormones; Explain what each hormone in the mail
Sow Reproduction and Seasonal Infertility. Darlington Pig Discussion Group 13 th March 2014 Richard Bull
 Sow Reproduction and Seasonal Infertility Darlington Pig Discussion Group 13 th March 2014 Richard Bull Richard Bull Taurus Concepts Ltd Sow Reproduction Endogenous Hormones Gland Hormone Function Hypothalamus
Sow Reproduction and Seasonal Infertility Darlington Pig Discussion Group 13 th March 2014 Richard Bull Richard Bull Taurus Concepts Ltd Sow Reproduction Endogenous Hormones Gland Hormone Function Hypothalamus
Plasma LH and FSH concentrations in prepubertal beef heifers before and in response to repeated injections of low doses of Gn-RH
 Plasma LH and FSH concentrations in prepubertal beef heifers before and in response to repeated injections of low doses of Gn-RH B. J. McLeod, W. Haresign, A. R. Peters and G. E. Lamming A.R.C. Research
Plasma LH and FSH concentrations in prepubertal beef heifers before and in response to repeated injections of low doses of Gn-RH B. J. McLeod, W. Haresign, A. R. Peters and G. E. Lamming A.R.C. Research
GONADOTROPHIN (LUTEINISING)- RELEASING HORMONE AND ANALOGUES (GnRH OR LHRH)
 GONADOTROPHIN (LUTEINISING)- RELEASING HORMONE AND ANALOGUES (GnRH OR LHRH) Naturally occurring hormone, produced by the hypothalamus and transferred to the anterior pituitary gland in the hypophyseal
GONADOTROPHIN (LUTEINISING)- RELEASING HORMONE AND ANALOGUES (GnRH OR LHRH) Naturally occurring hormone, produced by the hypothalamus and transferred to the anterior pituitary gland in the hypophyseal
The effects of growth hormone or melatonin on the reproductive axis of stallions
 Louisiana State University LSU Digital Commons LSU Master's Theses Graduate School 2002 The effects of growth hormone or melatonin on the reproductive axis of stallions William Andrew Storer Louisiana
Louisiana State University LSU Digital Commons LSU Master's Theses Graduate School 2002 The effects of growth hormone or melatonin on the reproductive axis of stallions William Andrew Storer Louisiana
Effect of naloxone on the plasma levels of LH, FSH, prolactin and testosterone in Beetal bucks
 Small Ruminant Research 37 (2000) 51±55 Effect of naloxone on the plasma levels of LH, FSH, prolactin and testosterone in Beetal bucks Baljeet Singh, V.D. Dixit, Pardeep Singh *, G.C. Georgie, V.P. Dixit
Small Ruminant Research 37 (2000) 51±55 Effect of naloxone on the plasma levels of LH, FSH, prolactin and testosterone in Beetal bucks Baljeet Singh, V.D. Dixit, Pardeep Singh *, G.C. Georgie, V.P. Dixit
(Received 9th January 1974)
 RELEASE OF LH AND FSH IN THE NORMAL INTACT RAM BY SYNTHETIC LH-RF AND THE EFFECT OF PRETREATMENT WITH TESTOSTERONE PROPIONATE C. R. N. HOPKINSON, H. C. PANT and R. J. FITZPATRICK Department of Veterinary
RELEASE OF LH AND FSH IN THE NORMAL INTACT RAM BY SYNTHETIC LH-RF AND THE EFFECT OF PRETREATMENT WITH TESTOSTERONE PROPIONATE C. R. N. HOPKINSON, H. C. PANT and R. J. FITZPATRICK Department of Veterinary
Changes in FSH and the pulsatile secretion of LH during the delay in oestrus induced by treatment of ewes
 Changes in FSH and the pulsatile secretion of LH during the delay in oestrus induced by treatment of ewes with bovine follicular fluid A. S. McNeilly M.R.C. Reproductive Biology Unit, University of Edinburgh
Changes in FSH and the pulsatile secretion of LH during the delay in oestrus induced by treatment of ewes with bovine follicular fluid A. S. McNeilly M.R.C. Reproductive Biology Unit, University of Edinburgh
OPIOID INHIBITION OF THE PULSATILE LUTEINIZING HORMONE RELEASE AS ASSESSED BY NALOXONE TREATMENT IN THE LACTATING RABBIT
 Reproduction OPIOID INHIBITION OF THE PULSATILE LUTEINIZING HORMONE RELEASE AS ASSESSED BY NALOXONE TREATMENT IN THE LACTATING RABBIT Marongiu M.L. 1 *, Gulinati A. 2 1 Department of Scienze Zootecniche,
Reproduction OPIOID INHIBITION OF THE PULSATILE LUTEINIZING HORMONE RELEASE AS ASSESSED BY NALOXONE TREATMENT IN THE LACTATING RABBIT Marongiu M.L. 1 *, Gulinati A. 2 1 Department of Scienze Zootecniche,
KISSPEPTIN AND GNIH CONTROL OF GNRH IN FEMALE MAMMALS
 KISSPEPTIN AND GNIH CONTROL OF GNRH IN FEMALE MAMMALS M.J. Zamiri Department of Animal Science, College of Agriculture, Shiraz University, Shiraz, Iran mjzamiri@gmail.com Introduction Since the discovery
KISSPEPTIN AND GNIH CONTROL OF GNRH IN FEMALE MAMMALS M.J. Zamiri Department of Animal Science, College of Agriculture, Shiraz University, Shiraz, Iran mjzamiri@gmail.com Introduction Since the discovery
Low Plasma Estradiol is Required for the Expression of Daily Increase in Plasma Gonadotropins in the Lactating Golden Hamster (Mesocricetus auratus)
 Journal of Reproduction and Development, Vol. 43, No. 2, 1997 Low Plasma Estradiol is Required for the Expression of Daily Increase in Plasma Gonadotropins in the Lactating Golden Hamster (Mesocricetus
Journal of Reproduction and Development, Vol. 43, No. 2, 1997 Low Plasma Estradiol is Required for the Expression of Daily Increase in Plasma Gonadotropins in the Lactating Golden Hamster (Mesocricetus
FSH immediately after the end of bff treatment at luteolysis, and they remained above control levels for
 Changes in the secretion of LH pulses, FSH and prolactin during the preovulatory phase of the oestrous cycle of the ewe and the influence of treatment with bovine follicular fluid during the luteal phase
Changes in the secretion of LH pulses, FSH and prolactin during the preovulatory phase of the oestrous cycle of the ewe and the influence of treatment with bovine follicular fluid during the luteal phase
injection. golden hamsters, and also established that pentobarbitone blockade of (Received 18 August 1969)
 J. Phy8iol. (1970), 206, pp. 471-479 471 With 1 text-figure Printed in Great Britain INFLUENCE OF OESTROGEN ON THYROID FUNCTION IN THE EWE BY IAN R. FALCONER From the Department of Applied Biochemistry
J. Phy8iol. (1970), 206, pp. 471-479 471 With 1 text-figure Printed in Great Britain INFLUENCE OF OESTROGEN ON THYROID FUNCTION IN THE EWE BY IAN R. FALCONER From the Department of Applied Biochemistry
Role of thyroid hormones. in the neuroendocrine control of seasonal.. reproduction in red deer hinds
 Role of thyroid hormones in the neuroendocrine control of seasonal. reproduction in red deer hinds A thesis submitted in partial fulfilment of the requirements for the Degree of Doctor of Philosophy at
Role of thyroid hormones in the neuroendocrine control of seasonal. reproduction in red deer hinds A thesis submitted in partial fulfilment of the requirements for the Degree of Doctor of Philosophy at
Differences in the plasma concentrations of FSH and LH in ovariectomized Booroola FF and ++ ewes
 Differences in the plasma concentrations of FSH and LH in ovariectomized Booroola FF and ++ ewes K. P. McNatty, M. Fisher, F. Collins, N. L. Hudson, D. A. Heath, K. Ball and K. M. Henderson Wallaceville
Differences in the plasma concentrations of FSH and LH in ovariectomized Booroola FF and ++ ewes K. P. McNatty, M. Fisher, F. Collins, N. L. Hudson, D. A. Heath, K. Ball and K. M. Henderson Wallaceville
Reproductive FSH. Analyte Information
 Reproductive FSH Analyte Information 1 Follicle-stimulating hormone Introduction Follicle-stimulating hormone (FSH, also known as follitropin) is a glycoprotein hormone secreted by the anterior pituitary
Reproductive FSH Analyte Information 1 Follicle-stimulating hormone Introduction Follicle-stimulating hormone (FSH, also known as follitropin) is a glycoprotein hormone secreted by the anterior pituitary
Central Progesterone Involvement in Estrogen- Induced Prolactin and Luteinizing Hormone Secretion Surges in Female Rats
 Southern Illinois University Carbondale OpenSIUC Honors Theses University Honors Program 5-10-2014 Central Progesterone Involvement in Estrogen- Induced Prolactin and Luteinizing Hormone Secretion Surges
Southern Illinois University Carbondale OpenSIUC Honors Theses University Honors Program 5-10-2014 Central Progesterone Involvement in Estrogen- Induced Prolactin and Luteinizing Hormone Secretion Surges
Copyright is owned by the Author of the thesis. Permission is given for a copy to be downloaded by an individual for the purpose of research and
 Copyright is owned by the Author of the thesis. Permission is given for a copy to be downloaded by an individual for the purpose of research and private study only. The thesis may not be reproduced elsewhere
Copyright is owned by the Author of the thesis. Permission is given for a copy to be downloaded by an individual for the purpose of research and private study only. The thesis may not be reproduced elsewhere
BIOLOGY - CLUTCH CH.45 - ENDOCRINE SYSTEM.
 !! www.clutchprep.com Chemical signals allow cells to communicate with each other Pheromones chemical signals released to the environment to communicate with other organisms Autocrine signaling self-signaling,
!! www.clutchprep.com Chemical signals allow cells to communicate with each other Pheromones chemical signals released to the environment to communicate with other organisms Autocrine signaling self-signaling,
Animal and Veterinary Science Department University of Idaho. REGULATION OF REPRODUCTION AVS 222 (Instructor: Dr. Amin Ahmadzadeh) Chapter 5
 Animal and Veterinary Science Department University of Idaho REGULATION OF REPRODUCTION AVS 222 (Instructor: Dr. Amin Ahmadzadeh) Chapter 5 I. DEFINITIONS A. Endocrine Gland B. Hormone Chemical messenger
Animal and Veterinary Science Department University of Idaho REGULATION OF REPRODUCTION AVS 222 (Instructor: Dr. Amin Ahmadzadeh) Chapter 5 I. DEFINITIONS A. Endocrine Gland B. Hormone Chemical messenger
Reproductive Hormones
 Reproductive Hormones Male gonads: testes produce male sex cells! sperm Female gonads: ovaries produce female sex cells! ovum The union of male and female sex cells during fertilization produces a zygote
Reproductive Hormones Male gonads: testes produce male sex cells! sperm Female gonads: ovaries produce female sex cells! ovum The union of male and female sex cells during fertilization produces a zygote
Cholecystokinin antagonist, proglumide, stimulates growth hormone release in the rat
 J. Biosci., Vol. 15, Number 1, March 1990, pp. 17 21. Printed in India. Cholecystokinin antagonist, proglumide, stimulates growth hormone release in the rat E. VIJAYAN* and S. M. McCANN Department of Physiology,
J. Biosci., Vol. 15, Number 1, March 1990, pp. 17 21. Printed in India. Cholecystokinin antagonist, proglumide, stimulates growth hormone release in the rat E. VIJAYAN* and S. M. McCANN Department of Physiology,
The reproductive lifespan
 The reproductive lifespan Reproductive potential Ovarian cycles Pregnancy Lactation Male Female Puberty Menopause Age Menstruation is an external indicator of ovarian events controlled by the hypothalamicpituitary
The reproductive lifespan Reproductive potential Ovarian cycles Pregnancy Lactation Male Female Puberty Menopause Age Menstruation is an external indicator of ovarian events controlled by the hypothalamicpituitary
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion
 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion A.S. Leaflet R1362 Acacia A. Alcivar, graduate research assistant,
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion A.S. Leaflet R1362 Acacia A. Alcivar, graduate research assistant,
/97/$03.00/0 Vol. 138, No. 12
 0013-7227/97/$03.00/0 Vol. 138, No. 12 Endocrinology Printed in U.S.A. Copyright 1997 by The Endocrine Society Estradiol Requirements for Induction and Maintenance of the Gonadotropin-Releasing Hormone
0013-7227/97/$03.00/0 Vol. 138, No. 12 Endocrinology Printed in U.S.A. Copyright 1997 by The Endocrine Society Estradiol Requirements for Induction and Maintenance of the Gonadotropin-Releasing Hormone
Chapter 14 Reproduction Review Assignment
 Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
The reproductive system
 The reproductive system THE OVARIAN CYCLE HORMONAL REGULATION OF OOGENSIS AND OVULATION hypothalamic-pituitary-ovary axis Overview of the structures of the endocrine system Principal functions of the
The reproductive system THE OVARIAN CYCLE HORMONAL REGULATION OF OOGENSIS AND OVULATION hypothalamic-pituitary-ovary axis Overview of the structures of the endocrine system Principal functions of the
Mechanisms of precocious puberty induced in male rats by
 Mechanisms of precocious puberty induced in male rats by pituitary grafts R. Aguilar, C. Bellido, J. E. S\l=a'\nchez-Criadoand E. Aguilar Department of Physiology, Faculty of Medicine, Cordoba University,
Mechanisms of precocious puberty induced in male rats by pituitary grafts R. Aguilar, C. Bellido, J. E. S\l=a'\nchez-Criadoand E. Aguilar Department of Physiology, Faculty of Medicine, Cordoba University,
Fertility Diagnostics
 Fertility Diagnostics Fertility hormones measured on PATHFAST For internal use only Diagnostics PATHFAST Chemiluminescence-immuno-analyzer 1 Content: page 1. Fertility hormones - general aspects 1.1 Reproductive
Fertility Diagnostics Fertility hormones measured on PATHFAST For internal use only Diagnostics PATHFAST Chemiluminescence-immuno-analyzer 1 Content: page 1. Fertility hormones - general aspects 1.1 Reproductive
LH (Horse) ELISA Kit. Catalog Number KA assays Version: 01. Intended for research use only.
 LH (Horse) ELISA Kit Catalog Number KA2302 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
LH (Horse) ELISA Kit Catalog Number KA2302 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH.
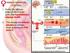 1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
Endocrine Glands. Endocrine glands
 ENDOCRINOLOGY Endocrine Glands Endocrine glands Produce substances called hormones. Ductless glands, i.e., they release hormones directly into the bloodstream Hormones only act at their target tissue where
ENDOCRINOLOGY Endocrine Glands Endocrine glands Produce substances called hormones. Ductless glands, i.e., they release hormones directly into the bloodstream Hormones only act at their target tissue where
MULTIPLE CHOICE: match the term(s) or description with the appropriate letter of the structure.
 Chapter 27 Exam Due NLT Thursday, July 31, 2015 Name MULTIPLE CHOICE: match the term(s) or description with the appropriate letter of the structure. Figure 27.1 Using Figure 27.1, match the following:
Chapter 27 Exam Due NLT Thursday, July 31, 2015 Name MULTIPLE CHOICE: match the term(s) or description with the appropriate letter of the structure. Figure 27.1 Using Figure 27.1, match the following:
Endocrine Glands: Hormone-secreting organs are called endocrine glands
 University of Jordan Department of Physiology and Biochemistry Nursing students, Academic year 2017/2018. ******************************************************************* Ref: Principles of Anatomy
University of Jordan Department of Physiology and Biochemistry Nursing students, Academic year 2017/2018. ******************************************************************* Ref: Principles of Anatomy
Page 1. A wide variety of ovarian abnormalities are encountered in clinical practice
 A wide variety of ovarian abnormalities are encountered in clinical practice Common Problems Anovulatory follicles Persistent anovulatory follicles Hemorrhagic/Luteinized follicles Persistent corpus luteum
A wide variety of ovarian abnormalities are encountered in clinical practice Common Problems Anovulatory follicles Persistent anovulatory follicles Hemorrhagic/Luteinized follicles Persistent corpus luteum
Pu Isatile Administration of Gonadotropin- Releasing Hormone Advances Ovulation in Cycling Mares
 BIOLOGY OF REPRODUCTION 35, 1123-1130 (1986) Pu Isatile Administration of Gonadotropin- Releasing Hormone Advances Ovulation in Cycling Mares A. L. JOHNSON2 Department of Animal Sciences Rutgers, The State
BIOLOGY OF REPRODUCTION 35, 1123-1130 (1986) Pu Isatile Administration of Gonadotropin- Releasing Hormone Advances Ovulation in Cycling Mares A. L. JOHNSON2 Department of Animal Sciences Rutgers, The State
Two important cells in female are the theca cells and the granulose cells. Granulosa cells are affected by the two gonadotropin hormones; FSH and LH.
 1 UGS physiology sheet #13 lecture 3 Dr.Saleem Khresha. Now we will start discussing the female reproductive system Ovarian Steroids Two important cells in female are the theca cells and the granulose
1 UGS physiology sheet #13 lecture 3 Dr.Saleem Khresha. Now we will start discussing the female reproductive system Ovarian Steroids Two important cells in female are the theca cells and the granulose
Hormonal Control of Human Reproduction
 Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
Modulation by photoperiod of gonadotrophin secretion in intact and castrated Djungarian hamsters
 Modulation by photoperiod of gonadotrophin secretion in intact and castrated Djungarian hamsters S. M. Simpson, B. K. Follett and D. H. Ellis A.R.C. Research Group on Photoperiodism & Reproduction, Department
Modulation by photoperiod of gonadotrophin secretion in intact and castrated Djungarian hamsters S. M. Simpson, B. K. Follett and D. H. Ellis A.R.C. Research Group on Photoperiodism & Reproduction, Department
LH (Canine) ELISA Kit
 LH (Canine) ELISA Kit Catalog Number KA2292 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
LH (Canine) ELISA Kit Catalog Number KA2292 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion
 Beef Research Report, 1996 Animal Science Research Reports 1997 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion Acacia
Beef Research Report, 1996 Animal Science Research Reports 1997 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion Acacia
Effect of ovariectomy at two periods of the year on LH and FSH basal concentrations and pituitary response to LHRH in the brown hare (Lepus europaeus)
 Effect of ovariectomy at two periods of the year on LH and FSH basal concentrations and pituitary response to LHRH in the brown hare (Lepus europaeus) M. Caillol, M. Mondain-Monval, M. Meunier and A. S.
Effect of ovariectomy at two periods of the year on LH and FSH basal concentrations and pituitary response to LHRH in the brown hare (Lepus europaeus) M. Caillol, M. Mondain-Monval, M. Meunier and A. S.
Managing the Mare for Optimal Fertility
 REVIEW Managing the Mare for Optimal Fertility C.H.G. IRVINE 1 * and Susan L. ALEXANDER 2 1 Animal & Veterinary Sciences Group, Lincoln University, Canterbury, and 2 Department of Endocrinology, Christchurch
REVIEW Managing the Mare for Optimal Fertility C.H.G. IRVINE 1 * and Susan L. ALEXANDER 2 1 Animal & Veterinary Sciences Group, Lincoln University, Canterbury, and 2 Department of Endocrinology, Christchurch
CY Tse, AMK Chow, SCS Chan. Introduction
 Effects of an extended-interval dosing regimen of triptorelin depot on the hormonal profile of patients with endometriosis: prospective observational study CY Tse, AMK Chow, SCS Chan Objective. To evaluate
Effects of an extended-interval dosing regimen of triptorelin depot on the hormonal profile of patients with endometriosis: prospective observational study CY Tse, AMK Chow, SCS Chan Objective. To evaluate
Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to Equine Chorionic Gonadotropin
 Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to quine Chorionic Gonadotropin S. L. Pulley, L. D. Wallace, H. I. Mellieon, and J. S. Stevenson
Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to quine Chorionic Gonadotropin S. L. Pulley, L. D. Wallace, H. I. Mellieon, and J. S. Stevenson
Proceedings of the 59th Annual Convention of the American Association of Equine Practitioners - AAEP -
 http://www.ivis.org Proceedings of the 59th Annual Convention of the American Association of Equine Practitioners - AAEP - December 7-11, 2013 Nashville, TN, USA Next Meeting : Dec. 6-10, 2014 - Salt Lake
http://www.ivis.org Proceedings of the 59th Annual Convention of the American Association of Equine Practitioners - AAEP - December 7-11, 2013 Nashville, TN, USA Next Meeting : Dec. 6-10, 2014 - Salt Lake
Systemic concentrations of hormones during the development of follicular waves in mares and women: a comparative study
 REPRODUCTION RESEARCH Systemic concentrations of hormones during the development of follicular waves in mares and women: a comparative study O J Ginther 1,, M A Beg, E L Gastal, M O Gastal, A R Baerwald
REPRODUCTION RESEARCH Systemic concentrations of hormones during the development of follicular waves in mares and women: a comparative study O J Ginther 1,, M A Beg, E L Gastal, M O Gastal, A R Baerwald
REPRODUCCIÓN. La idea fija. Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
Course: Animal Production. Instructor: Ms. Hutchinson. Objectives: After completing this unit of instruction, students will be able to:
 Course: Animal Production Unit Title: Hormones TEKS: 130.3 (C)(6)(A) Instructor: Ms. Hutchinson Objectives: After completing this unit of instruction, students will be able to: A. Define what hormones
Course: Animal Production Unit Title: Hormones TEKS: 130.3 (C)(6)(A) Instructor: Ms. Hutchinson Objectives: After completing this unit of instruction, students will be able to: A. Define what hormones
Concentrations of Luteinizing Hormone and Ovulatory Responses in Dairy Cows Before Timed Artificial Insemination
 Concentrations of Luteinizing Hormone and Ovulatory Responses in Dairy Cows Before Timed Artificial Insemination S. L. Pulley, D. H. Keisler, S. L. Hill, and J. S. Stevenson Summary The objective of this
Concentrations of Luteinizing Hormone and Ovulatory Responses in Dairy Cows Before Timed Artificial Insemination S. L. Pulley, D. H. Keisler, S. L. Hill, and J. S. Stevenson Summary The objective of this
Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle?
 Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle? Introduction: The menstrual cycle (changes within the uterus) is an approximately 28-day cycle that
Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle? Introduction: The menstrual cycle (changes within the uterus) is an approximately 28-day cycle that
Reproductive System (Hormone Function) Physiology Department Medical School, University of Sumatera Utara
 Reproductive System (Hormone Function) Physiology Department Medical School, University of Sumatera Utara 1 Endocrine Control: Three Levels of Integration Hormones of the hypothalamic-anterior pituitary
Reproductive System (Hormone Function) Physiology Department Medical School, University of Sumatera Utara 1 Endocrine Control: Three Levels of Integration Hormones of the hypothalamic-anterior pituitary
A.J. TILBROOK, 2 ' 3 D.M. DE KRETSER, 4 and I.J. CLARKE 5 ABSTRACT
 BIOLOGY OF REPRODUCTION 49, 779-788 (1993) Human Recombinant Inhibin A Suppresses Plasma Follicle-Stimulating Hormone to Intact Levels But Has No Effect on Luteinizing Hormone in Castrated Rams' A.J. TILBROOK,
BIOLOGY OF REPRODUCTION 49, 779-788 (1993) Human Recombinant Inhibin A Suppresses Plasma Follicle-Stimulating Hormone to Intact Levels But Has No Effect on Luteinizing Hormone in Castrated Rams' A.J. TILBROOK,
REPRODUCTIVE ENDOCRINOLOGY OF THE MALE
 Reproductive Biotechnologies Andrology I REPRODUCTIVE ENDOCRINOLOGY OF THE MALE Prof. Alberto Contri REPRODUCTIVE ENDOCRINOLOGY OF THE MALE SPERMATOGENESIS AND REPRODUCTIVE BEHAVIOR RELATED TO THE ACTIVITY
Reproductive Biotechnologies Andrology I REPRODUCTIVE ENDOCRINOLOGY OF THE MALE Prof. Alberto Contri REPRODUCTIVE ENDOCRINOLOGY OF THE MALE SPERMATOGENESIS AND REPRODUCTIVE BEHAVIOR RELATED TO THE ACTIVITY
Fukushima-ku, Osaka. Synopsis. and LH release by investigating the effects of exogenous estrogen on the progesteroneinduced
 Further Studies on the Causal Relationship between the Secretion of Estrogen and the Release of Luteinizing Hormone in the Rat FUMIHIKO KOBAYASHI, KATSUMI HARA AND TAMOTSU MIYAKE Shionogi Research Laboratory,
Further Studies on the Causal Relationship between the Secretion of Estrogen and the Release of Luteinizing Hormone in the Rat FUMIHIKO KOBAYASHI, KATSUMI HARA AND TAMOTSU MIYAKE Shionogi Research Laboratory,
Female Reproductive System. Lesson 10
 Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
Endocrine part one. Presented by Dr. Mohammad Saadeh The requirements for the Clinical Chemistry Philadelphia University Faculty of pharmacy
 Endocrine part one Presented by Dr. Mohammad Saadeh The requirements for the Clinical Chemistry Philadelphia University Faculty of pharmacy HORMONES Hormones are chemicals released by a cell or a gland
Endocrine part one Presented by Dr. Mohammad Saadeh The requirements for the Clinical Chemistry Philadelphia University Faculty of pharmacy HORMONES Hormones are chemicals released by a cell or a gland
DEVELOPMENTAL CHANGES IN GONADOTROPltf RELEASING HORMONE NEURONS IN THE BRAIN OF THE FEMALE RABBIT (oryctolagus cuniculus)
 DEVELOPMENTAL CHANGES IN GONADOTROPltf RELEASING HORMONE NEURONS IN THE BRAIN OF THE FEMALE RABBIT (oryctolagus cuniculus) By WARREN G. FOSTER, B.Sc., M.Sc. A Thesis Submitted to the School of Graduate
DEVELOPMENTAL CHANGES IN GONADOTROPltf RELEASING HORMONE NEURONS IN THE BRAIN OF THE FEMALE RABBIT (oryctolagus cuniculus) By WARREN G. FOSTER, B.Sc., M.Sc. A Thesis Submitted to the School of Graduate
9.4 Regulating the Reproductive System
 9.4 Regulating the Reproductive System The Reproductive System to unite a single reproductive cell from a female with a single reproductive cell from a male Both male and female reproductive systems include
9.4 Regulating the Reproductive System The Reproductive System to unite a single reproductive cell from a female with a single reproductive cell from a male Both male and female reproductive systems include
Testosterone and progesterone in peripheral plasma during the oestrous cycle of the mare
 Testosterone and progesterone in peripheral plasma during the oestrous cycle of the mare P. Silberzahn, D. Quincey, C. Rosier and P. Leymarie ^Laboratoire de physico-chimie biologique, U.E.R. des Sciences
Testosterone and progesterone in peripheral plasma during the oestrous cycle of the mare P. Silberzahn, D. Quincey, C. Rosier and P. Leymarie ^Laboratoire de physico-chimie biologique, U.E.R. des Sciences
Time / days. Explain how the release of FSH is controlled by negative feedback.
 1. The graph shows the changes in concentration of the hormones responsible for controlling the menstrual cycle. A Hormone concentration Oestrogen B C 0 14 28 Time / days WD Phillips and TJ Chilton A Level
1. The graph shows the changes in concentration of the hormones responsible for controlling the menstrual cycle. A Hormone concentration Oestrogen B C 0 14 28 Time / days WD Phillips and TJ Chilton A Level
CHANGES IN LEVELS OF FOLLICLE STIMULATING HORMONE AND LUTEINIZING HORMONE IN THE BOVINE PITUITARY GLAND AT OVULATION
 CHANGES IN LEVELS OF FOLLICLE STIMULATING HORMONE AND LUTEINIZING HORMONE IN THE BOVINE PITUITARY GLAND AT OVULATION A. M. RAKHA and H. A. ROBERTSON The Division of Agricultural Biochemistry, Department
CHANGES IN LEVELS OF FOLLICLE STIMULATING HORMONE AND LUTEINIZING HORMONE IN THE BOVINE PITUITARY GLAND AT OVULATION A. M. RAKHA and H. A. ROBERTSON The Division of Agricultural Biochemistry, Department
NROSCI/BIOSC 1070 and MSNBIO 2070 September 11, 2017 Control Mechanisms 2: Endocrine Control
 NROSCI/BIOSC 1070 and MSNBIO 2070 September 11, 2017 Control Mechanisms 2: Endocrine Control Hormones are chemical messengers that are secreted into the blood by endocrine cells or specialized neurons.
NROSCI/BIOSC 1070 and MSNBIO 2070 September 11, 2017 Control Mechanisms 2: Endocrine Control Hormones are chemical messengers that are secreted into the blood by endocrine cells or specialized neurons.
A. K. EYONG AND V. B. BRAIDE
 GLOBAL JOURNAL OF MEDICAL SCIENCES VOL 8, NO. 1&2, 2009: 23-30 COPYRIGHT BACHUDO SCIENCE CO. LTD PRINTED IN NIGERIA. ISSN 1596-2911 www.globaljournalseries.com; Email: info@globaljournalseries.com EFFECTS
GLOBAL JOURNAL OF MEDICAL SCIENCES VOL 8, NO. 1&2, 2009: 23-30 COPYRIGHT BACHUDO SCIENCE CO. LTD PRINTED IN NIGERIA. ISSN 1596-2911 www.globaljournalseries.com; Email: info@globaljournalseries.com EFFECTS
GONADAL FUNCTION: An Overview
 GONADAL FUNCTION: An Overview University of PNG School of Medicine & Health Sciences Division of Basic Medical Sciences Clinical Biochemistry BMLS III & BDS IV VJ Temple 1 What are the Steroid hormones?
GONADAL FUNCTION: An Overview University of PNG School of Medicine & Health Sciences Division of Basic Medical Sciences Clinical Biochemistry BMLS III & BDS IV VJ Temple 1 What are the Steroid hormones?
Gonadotropin-Releasing Hormone Treatment Induces Follicular Growth and Ovulation in Seasonally Anestrous Mares'
 BIOLOGY OF REPRODUCTION 36,1199-1206 (1987) Gonadotropin-Releasing Hormone Treatment Induces Follicular Growth and Ovulation in Seasonally Anestrous Mares' A. L. JOHNSON Department of Animal Sciences Rutgers,
BIOLOGY OF REPRODUCTION 36,1199-1206 (1987) Gonadotropin-Releasing Hormone Treatment Induces Follicular Growth and Ovulation in Seasonally Anestrous Mares' A. L. JOHNSON Department of Animal Sciences Rutgers,
Evaluation of BioRelease P4 LA 300 in the Mare. Mobile Veterinary Practice, Amarillo, Tx, Abraham Equine, Mendota Ranch, Canadian,Tx
 Evaluation of BioRelease P4 LA 300 in the Mare P. J. Burns 1, C. Morrow 2 & J. Abraham 3 1 Burns BioSolutions, Lexington KY 40515, (www.burnsbiosolutions.com) 2 Mobile Veterinary Practice, Amarillo, Tx,
Evaluation of BioRelease P4 LA 300 in the Mare P. J. Burns 1, C. Morrow 2 & J. Abraham 3 1 Burns BioSolutions, Lexington KY 40515, (www.burnsbiosolutions.com) 2 Mobile Veterinary Practice, Amarillo, Tx,
injected daily were {Received 7th August 1972) Summary. Groups of rats ovariectomized 5 weeks previously
 FFCTS OF XOGNOUS OSTRADIOL AND PROGSTRON ON SRUM LVLS OF PROSTAGLANDIN F AND LUTINIZING HORMON IN CHRONICALLY OVARICTOMIZD RATS S. K. SAKSNA, R. STL and M. J. K. HARPR Worcester Foundation for xperimental
FFCTS OF XOGNOUS OSTRADIOL AND PROGSTRON ON SRUM LVLS OF PROSTAGLANDIN F AND LUTINIZING HORMON IN CHRONICALLY OVARICTOMIZD RATS S. K. SAKSNA, R. STL and M. J. K. HARPR Worcester Foundation for xperimental
Available online at Theriogenology xxx (2009) xxx xxx
 Available online at www.sciencedirect.com 1 2 3 4 5 6 7 8 9 10 11 The effect of hormone treatments (hcg and cloprostenol) and season on the incidence of hemorrhagic anovulatory follicles in the mare: A
Available online at www.sciencedirect.com 1 2 3 4 5 6 7 8 9 10 11 The effect of hormone treatments (hcg and cloprostenol) and season on the incidence of hemorrhagic anovulatory follicles in the mare: A
Reproductive physiology
 Reproductive physiology Sex hormones: Androgens Estrogens Gestagens Learning objectives 86 (also 90) Sex Genetic sex Gonadal sex Phenotypic sex XY - XX chromosomes testes - ovaries external features Tha
Reproductive physiology Sex hormones: Androgens Estrogens Gestagens Learning objectives 86 (also 90) Sex Genetic sex Gonadal sex Phenotypic sex XY - XX chromosomes testes - ovaries external features Tha
Neuroendocrine Regulation of the Estrous Cycle and Seasonal Breeding in the Ewe
 BIOLOGY OF REPRODUCTION 20, 74-85 (1979) Neuroendocrine Regulation of the Estrous Cycle and Seasonal Breeding in the Ewe SANDRA J. LEGAN and FRED J. KARSCH INTRODUCTION One of the most intriguing aspects
BIOLOGY OF REPRODUCTION 20, 74-85 (1979) Neuroendocrine Regulation of the Estrous Cycle and Seasonal Breeding in the Ewe SANDRA J. LEGAN and FRED J. KARSCH INTRODUCTION One of the most intriguing aspects
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following hormones controls the release of anterior pituitary gonadotropins? A) LH
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following hormones controls the release of anterior pituitary gonadotropins? A) LH
10.7 The Reproductive Hormones
 10.7 The Reproductive Hormones December 10, 2013. Website survey?? QUESTION: Who is more complicated: men or women? The Female Reproductive System ovaries: produce gametes (eggs) produce estrogen (steroid
10.7 The Reproductive Hormones December 10, 2013. Website survey?? QUESTION: Who is more complicated: men or women? The Female Reproductive System ovaries: produce gametes (eggs) produce estrogen (steroid
Seasonal influence on equine follicle dynamics
 Seasonal influence on equine follicle dynamics O. J. Ginther 1,3, E. L. Gastal 2, M. O. Gastal 2, M. A. Beg 1 Anim. Reprod., v.,1 n.1, p.31-44, Oct./.Dec. 04 Eutheria Foundation, Cross Plains, Wisconsin
Seasonal influence on equine follicle dynamics O. J. Ginther 1,3, E. L. Gastal 2, M. O. Gastal 2, M. A. Beg 1 Anim. Reprod., v.,1 n.1, p.31-44, Oct./.Dec. 04 Eutheria Foundation, Cross Plains, Wisconsin
LH (Rodent) ELISA Kit
 LH (Rodent) ELISA Kit Catalog Number KA2332 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
LH (Rodent) ELISA Kit Catalog Number KA2332 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
Advanced Non-Cycling Program. Health
 Advanced Non-Cycling Program Health Why Treat Non-Cycling Cows? Treating cows that have not been detected in oestrus ( non-cycling ) prior to the planned start of mating with DIB-Synch provides a return
Advanced Non-Cycling Program Health Why Treat Non-Cycling Cows? Treating cows that have not been detected in oestrus ( non-cycling ) prior to the planned start of mating with DIB-Synch provides a return
FOLLICLE-STIMULATING HORMONE CONTENT OF THE PITUITARY GLAND BEFORE IMPLANTATION IN THE MOUSE AND RAT
 FOLLICLE-STIMULATING HORMONE CONTENT OF THE PITUITARY GLAND BEFORE IMPLANTATION IN THE MOUSE AND RAT B. M. BINDON Department of Veterinary Physiology, University of Sydney, Sydney 2006, Australia (Received
FOLLICLE-STIMULATING HORMONE CONTENT OF THE PITUITARY GLAND BEFORE IMPLANTATION IN THE MOUSE AND RAT B. M. BINDON Department of Veterinary Physiology, University of Sydney, Sydney 2006, Australia (Received
HORMONES & REPRODUCTION OUTLINE
 1 HORMONES & REPRODUCTION Dr. Steinmetz OUTLINE 2 The Endocrine System Sexual Reproduction Hormonal Role in Sexual Differentiation Gender Differences and Gender Identity Characterizing Complex Behaviors
1 HORMONES & REPRODUCTION Dr. Steinmetz OUTLINE 2 The Endocrine System Sexual Reproduction Hormonal Role in Sexual Differentiation Gender Differences and Gender Identity Characterizing Complex Behaviors
THE REPRODUCTIVE neuroendocrine axis of many
 0013-7227/03/$15.00/0 Endocrinology 144(4):1426 1434 Printed in U.S.A. Copyright 2003 by The Endocrine Society doi: 10.1210/en.2002-220965 Prenatal Programming of Reproductive Neuroendocrine Function:
0013-7227/03/$15.00/0 Endocrinology 144(4):1426 1434 Printed in U.S.A. Copyright 2003 by The Endocrine Society doi: 10.1210/en.2002-220965 Prenatal Programming of Reproductive Neuroendocrine Function:
Stage 4 - Ovarian Cancer Symptoms
 WELCOME Stage 4 - Ovarian Cancer Symptoms University of Baghdad College of Nursing Department of Basic Medical Sciences Overview of Anatomy and Physioloy II Second Year Students Asaad Ismail Ahmad,
WELCOME Stage 4 - Ovarian Cancer Symptoms University of Baghdad College of Nursing Department of Basic Medical Sciences Overview of Anatomy and Physioloy II Second Year Students Asaad Ismail Ahmad,
SUMMARY. at metoestrus and dioestrus, and could not be demonstrated at oestrus. to the
 A PRIMING EFFECT OF LUTEINIZING HORMONE RELEASING FACTOR ON THE ANTERIOR PITUITARY GLAND IN THE FEMALE RAT M. S. AIYER, SHARON A. CHIAPPA and G. FINK Department of Human Anatomy, South Parks Road, Oxford,
A PRIMING EFFECT OF LUTEINIZING HORMONE RELEASING FACTOR ON THE ANTERIOR PITUITARY GLAND IN THE FEMALE RAT M. S. AIYER, SHARON A. CHIAPPA and G. FINK Department of Human Anatomy, South Parks Road, Oxford,
Development of an estradiol-dopamine anagonist protocol for inducing ovulation in seasonally anovulatory mares
 Louisiana State University LSU Digital Commons LSU Doctoral Dissertations Graduate School 2012 Development of an estradiol-dopamine anagonist protocol for inducing ovulation in seasonally anovulatory mares
Louisiana State University LSU Digital Commons LSU Doctoral Dissertations Graduate School 2012 Development of an estradiol-dopamine anagonist protocol for inducing ovulation in seasonally anovulatory mares
SISTEMA REPRODUCTOR (LA IDEA FIJA) Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
Concentrations of luteinizing hormone and ovulatory responses in dairy cows before timed artificial insemination
 Kansas Agricultural Experiment Station Research Reports Volume 0 Issue Dairy Research (98-0) Article 8 0 Concentrations of luteinizing hormone and ovulatory responses in dairy cows before timed artificial
Kansas Agricultural Experiment Station Research Reports Volume 0 Issue Dairy Research (98-0) Article 8 0 Concentrations of luteinizing hormone and ovulatory responses in dairy cows before timed artificial
Rhythm Plus- Comprehensive Female Hormone Profile
 Rhythm Plus- Comprehensive Female Hormone Profile Patient: SAMPLE REPORT DOB: Sex: F Order Number: K00000 Completed: Received: Collected: SAMPLE REPORT Sample # Progesterone (pg/ml) Hormone Results Oestradiol
Rhythm Plus- Comprehensive Female Hormone Profile Patient: SAMPLE REPORT DOB: Sex: F Order Number: K00000 Completed: Received: Collected: SAMPLE REPORT Sample # Progesterone (pg/ml) Hormone Results Oestradiol
Circulating Levels of Melatonin following Its Oral Administration or Subcutaneous Injection in Sheep and Goats
 Aust. J. Bioi. Sci. 33, 1980, 349-53 Circulating Levels of Melatonin following Its Oral Administration or Subcutaneous Injection in Sheep and Goats D. J. Kennaway and R. F. Seamark Department of Obstetrics
Aust. J. Bioi. Sci. 33, 1980, 349-53 Circulating Levels of Melatonin following Its Oral Administration or Subcutaneous Injection in Sheep and Goats D. J. Kennaway and R. F. Seamark Department of Obstetrics
Article Artikel. F E van Niekerk a and C H van Niekerk a
 Article Artikel The effect of dietary protein on reproduction in the mare. III. Ovarian and uterine changes during the anovulatory, transitional and ovulatory periods in the non-pregnant mare F E van Niekerk
Article Artikel The effect of dietary protein on reproduction in the mare. III. Ovarian and uterine changes during the anovulatory, transitional and ovulatory periods in the non-pregnant mare F E van Niekerk
CASE 41. What is the pathophysiologic cause of her amenorrhea? Which cells in the ovary secrete estrogen?
 CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
Effect of a Single Injection of Human Chorionic Gonadotropin (hcg) on Testicular Hormones and Gonadotropins in the Thoroughbred Stallion
 NOTE Effect of a Single Injection of Human Chorionic Gonadotropin (hcg) on Testicular Hormones and Gonadotropins in the Thoroughbred Stallion Nobuo TSUNODA 1, Qiang WENG 2, 3, Shunichi NAGATA 4, Hiroyuki
NOTE Effect of a Single Injection of Human Chorionic Gonadotropin (hcg) on Testicular Hormones and Gonadotropins in the Thoroughbred Stallion Nobuo TSUNODA 1, Qiang WENG 2, 3, Shunichi NAGATA 4, Hiroyuki
