Pocket Adaptor Kit FOR SPINAL CORD STIMULATION. Tip Cards
|
|
|
- Anna Clarke
- 5 years ago
- Views:
Transcription
1 Pocket Adaptor Kit FOR SPINAL CORD STIMULATION Tip Cards model (1 4) model (2 4)
2 The Pocket Adaptor Tip Cards are to be used for educational purposes only. Please refer to the Pocket Adaptor Kit Implant Manual for complete instructions, warnings and cautions. Pocket Adaptor Model (1 4) and Model (2 4) are intended to be implanted with a new replacement 16-electrode neurostimulator in the same pocket used for the explanted Note: Implanting in the same neurostimulator pocket allows for a single-incision procedure. The RestoreUltra (Model 37712) neurostimulator is shown on the following procedure cards. The following neurostimulators are also compatible with the Pocket Adaptors although the orientation of the sockets on the header block is different: Socket II (back) Electrodes 8-15 Socket I (front) Electrodes 0-7 RestoreAdvanced (Model 37713) PrimeAdvanced (Model 37702) Restore (Model 37711) RestorePrime (Model 37701) Compatible Quadripolar Extensions Quadripolar extension model number Model (1 4) Model (2 4) 7471 a 7472 a LZ b a The Models 7471 and 7472 extensions are bifurcated on the proximal end. b The Model 7498 extension is bifurcated on the distal end. Warning: Evaluate the suitability of a Pocket Adaptor for patients with an implanted Model 7498 extension. The extension is bifurcated on the distal end, connecting 2 leads in parallel. If one lead should break, an impedance measurement may yield a result in the normal range because of the presence of the second lead. Thus, open circuits may be difficult to detect, which can increase the potential for risks related to MRI.
3 Procedure #1 2 4 Pocket Adaptor with 2 extensions or a bifurcated extension (8 combined electrodes) Caution: Only use a 2 4 Pocket Adaptor for systems using the Model 7471 or the Model 7472 bifurcated extension. Using two 1 4 Pocket Adaptors with these bifurcated extensions will cause electrode numbering to be mismatched when programming and cause some neurostimulator features to be inoperable. Retract the explanted neurostimulator set screws. 2 4 Pocket Adaptor with 2 extensions or a bifurcated extension Remove only Extension 2 from Socket II (back) of the explanted Insert Extension 2 into Connector Port 2 (bottom) of the adaptor. Remove Extension 1 from Socket I (front) of the explanted Insert Extension 1 into Connector Port 1 (top) of the adaptor. Tighten all adaptor setscrews.
4 Procedure #1 2 4 Pocket Adaptor with 2 extensions or a bifurcated extension (8 combined electrodes) 2 4 Pocket Adaptor with 2 extensions or a bifurcated extension Socket I (top) Electrodes 0-7 Slowly advance the adaptor connection into Socket I (top) of the Insert the plug into Socket II (bottom) of the Tighten all neurostimulator setscrews. SKIN NEUROSTIMULATOR ADAPTOR COILED WIRES Place the neurostimulator, adaptor, and coiled wires (placed behind the adaptor) into the subcutaneous pocket. Note: Ensure there are no sharp bends in the wires and the wires are not between the devices. Program the lead and electrode configuration (see Programming tab) and perform a system integrity check to ensure that lead configuration is set up appropriately and implanted leads and extensions are functional.
5 Procedure #2 1 4 Pocket Adaptor with 1 extension (4 electrodes) Retract the explanted neurostimulator set screws. Remove the extension from the Insert the extension into the adaptor. 1 4 Pocket Adaptor with 1 extension Tighten the adaptor setscrews. Socket I (top) Electrodes 0-3 Slowly advance the adaptor connection into Socket I (top) of the Insert the plug into Socket II (bottom) of the
6 Procedure #2 Pocket Placement Procedure 1 4 Pocket Adaptor with 1 extension To (4 electrodes) be used with all pocket adaptor procedures Tighten all neurostimulator setscrews. COILED WIRE Place the neurostimulator, adaptor, and coiled wires (placed behind the adaptor) into the subcutaneous pocket. 1 4 Pocket Adaptor with 1 extension SKIN NEUROSTIMULATOR ADAPTOR Note: Ensure there are no sharp bends in the wires and the wires are not between the devices. Program the lead and electrode configuration (see Programming tab) and perform a system integrity check to ensure that lead configuration is set up appropriately and implanted leads and extensions are functional.
7 Procedure #3 Two 1 4 Pocket Adaptors with 2 extensions (4 electrodes each) Caution: Only use a 2 4 Pocket Adaptor for systems using the Model 7471 or the Model 7472 bifurcated extension. Using two 1 4 Pocket Adaptors with these bifurcated extensions will cause electrode numbering to be mismatched when programming and cause some neurostimulator features to be inoperable. Retract the explanted neurostimulator set screws. Remove only Extension 2 from Socket II (back) of the explanted Insert Extension 2 into one of the adaptors. Socket II (bottom) Slowly advance the first adaptor connection into Socket II (bottom) of the new Remove Extension 1 from Socket I (front) of the explanted Two 1 4 Pocket Adaptors with 2 extensions Insert Extension 1 into the other adaptor.
8 Procedure #3 Two 1 4 Pocket Adaptors with 2 extensions (4 electrodes each) Socket I (top) Advance the second adaptor connection into Socket I (top) of the new Tighten all adaptor setscrews (both adaptors). Tighten all neurostimulator setscrews. Two 1x4 Pocket Adaptors with 2 extensions SKIN NEUROSTIMULATOR ADAPTORS COILED WIRES Place the neurostimulator, adaptors, and coiled wires (placed behind the adaptors) into the subcutaneous pocket. Note: Ensure there are no sharp bends in the wires and the wires are not between the devices. Program the lead and electrode configuration (see Programming tab) and perform a system integrity check to ensure that lead configuration is set up appropriately and implanted leads and extensions are functional.
9 Programming Pocket Adaptor One 2 4 One 1 4 Two 1 4s Neurostimulator socket and corresponding electrodes If Socket I is used, the default electrode numbering is as follows and requires no renumbering: Lead I: 0-3 Lead II: 4-7 If Socket II is used, the electrodes require renumbering to the following: Lead I: 8-11 Lead II: If Socket I is used, the default electrode numbering is 0-3 and requires no renumbering. If Socket II is used, the electrodes require renumbering to Socket I default electrode numbering for Lead I is 0-3 and requires no renumbering. Socket II electrodes for Lead II require renumbering to Lead Configuration a, b a This is the configuration to select in the Lead Configuration screen of the N Vision clinician programmer. b If additional leads are added to the system, the lead configuration will be different from what is given in this column. Programming
10 NEUROSTIMULATION SYSTEMS FOR PAIN THERAPY Brief Summary: Product manuals must be reviewed prior to use for detailed disclosure. Indications : Implantable neurostimulation systems A Medtronic implantable neurostimulation system is indicated for spinal cord stimulation (SCS) system as an aid in the management of chronic, intractable pain of the trunk and/or limbs including unilateral or bilateral pain associated with the following conditions: Failed Back Syndrome (FBS) or low back syndrome or failed back Radicular pain syndrome or radiculopathies resulting in pain secondary to FBS or herniated disk Postlaminectomy pain Multiple back operations Unsuccessful disk surgery Degenerative Disk Disease (DDD)/herniated disk pain refractory to conservative and surgical interventions Peripheral causalgia Epidural fibrosis Arachnoiditis or lumbar adhesive arachnoiditis Complex Regional Pain Syndrome (CRPS), Reflex Sympathetic Dystrophy (RSD), or causalgia Contraindications: Diathermy Do not use shortwave diathermy, microwave or therapeutic ultrasound diathermy (all now referred to as diathermy) on patients implanted with a neurostimulation system. Energy from diathermy can be transferred through the implanted system and cause tissue damage at the locations of the implanted electrodes, resulting in severe injury or death. Warnings: Sources of strong electromagnetic interference (eg, defibrillation, diathermy, electrocautery, MRI, RF ablation, and therapeutic ultrasound) can interact with the neurostimulation system, resulting in serious patient injury or death. These and other sources of EMI can also result in system damage, operational changes to the neurostimulator or unexpected changes in stimulation. Rupture or piercing of the neurostimulator can result in severe burns. An implanted cardiac device (eg, pacemaker, defibrillator) may damage a neurostimulator, and the electrical pulses from the neurostimulator may result in an inappropriate response of the cardiac device. Precautions: The safety and effectiveness of this therapy has not been established for pediatric use (patients under the age of 18), pregnancy, unborn fetus, or delivery. Patients should be detoxified from narcotics prior to lead placement. Clinicians and patients should follow programming guidelines and precautions provided in product manuals. Patients should avoid activities that may put undue stress on the implanted neurostimulation system components. Patients should not scuba dive below 10 meters of water or enter hyperbaric chambers above 2.0 atmosphere absolute (ATA). Electromagnetic interference, postural changes, and other activities may cause shocking or jolting. Adverse Events: Adverse events may include: undesirable change in stimulation described by some patients as uncomfortable, jolting or shocking; hematoma, epidural hemorrhage, paralysis, seroma, CSF leakage, infection, erosion, allergic response, hardware malfunction or migration, pain at implant site, loss of pain relief, chest wall stimulation, and surgical risks. For further information, please call Medtronic at and/or consult Medtronic s website at USA Rx Only Rev United States of America Medtronic Neuromodulation 710 Medtronic Parkway Minneapolis, MN USA Tel Toll-free UC EN NI9017 Medtronic, Inc All Rights Reserved. Printed in USA.
TAKE CONTROL OF YOUR FUTURE
 NEUROSTIMULATION SYSTEMS FOR PAIN THERAPY Brief Summary: Product manuals must be reviewed prior to use for detailed disclosure. Indications Implantable neurostimulation systems - A Medtronic implantable
NEUROSTIMULATION SYSTEMS FOR PAIN THERAPY Brief Summary: Product manuals must be reviewed prior to use for detailed disclosure. Indications Implantable neurostimulation systems - A Medtronic implantable
NEW JERSEY PAINCONSULTANTS OFFERS PATIENTS FIRST IMPLANTABLE NEUROSTIMULATION SYSTEM WITH MOTION SENSOR TECHNOLOGY FOR THE MANAGEMENT OF CHRONIC PAIN
 Contact: Barbara Carey Practice Manager New Jersey Pain Consultants 175 Morristown Rd Basking Ridge, NJ 07920 (908) 630-0175 NEW JERSEY PAINCONSULTANTS OFFERS PATIENTS FIRST IMPLANTABLE NEUROSTIMULATION
Contact: Barbara Carey Practice Manager New Jersey Pain Consultants 175 Morristown Rd Basking Ridge, NJ 07920 (908) 630-0175 NEW JERSEY PAINCONSULTANTS OFFERS PATIENTS FIRST IMPLANTABLE NEUROSTIMULATION
USING YOUR INTELLIS IMPLANTABLE SPINAL CORD STIMULATION SYSTEM
 USING YOUR INTELLIS IMPLANTABLE SPINAL CORD STIMULATION SYSTEM This guide provides basic resources and information to help you use your Medtronic Intellis Spinal Cord Stimulation system. Note that the
USING YOUR INTELLIS IMPLANTABLE SPINAL CORD STIMULATION SYSTEM This guide provides basic resources and information to help you use your Medtronic Intellis Spinal Cord Stimulation system. Note that the
Medtronic, Inc.
 Recharging System Quick Guide for Chronic pain www.medtronic.com Medtronic, Inc. 710 Medtronic Parkway Minneapolis, MN 55432-5604 USA Tel: 763-505-5000 Toll-free: 1-800-328-0810 UC201002746a EN 2013 Medtronic,
Recharging System Quick Guide for Chronic pain www.medtronic.com Medtronic, Inc. 710 Medtronic Parkway Minneapolis, MN 55432-5604 USA Tel: 763-505-5000 Toll-free: 1-800-328-0810 UC201002746a EN 2013 Medtronic,
Out of pain. Into relief.
 Spinal Cord Stimulation for Chronic Pain USING YOUR TRIAL STIMULATION SYSTEM Out of pain. Into relief. SCREENING TRIAL GUIDE AND PATIENT DIARY You are taking an important step in having a trial for spinal
Spinal Cord Stimulation for Chronic Pain USING YOUR TRIAL STIMULATION SYSTEM Out of pain. Into relief. SCREENING TRIAL GUIDE AND PATIENT DIARY You are taking an important step in having a trial for spinal
SINGAPORE GUIDED LUMBAR PROCEDURES WORKSHOP ULTRASOUND AND FLUOROSCOPIC OCTOBER 29-30, 2016 ACADEMIA, SINGAPORE GENERAL HOSPITAL PRESENTED BY:
 ULTRASOUND AND FLUOROSCOPIC GUIDED LUMBAR PROCEDURES WORKSHOP ACADEMIA, SINGAPORE GENERAL HOSPITAL SINGAPORE OCTOBER 29-30, 2016 CORPORATE SUPPORT: PRESENTED BY: Ultrasound and Fluoroscopic Guided Lumbar
ULTRASOUND AND FLUOROSCOPIC GUIDED LUMBAR PROCEDURES WORKSHOP ACADEMIA, SINGAPORE GENERAL HOSPITAL SINGAPORE OCTOBER 29-30, 2016 CORPORATE SUPPORT: PRESENTED BY: Ultrasound and Fluoroscopic Guided Lumbar
DOCTOR DISCUSSION GUIDE
 DOCTOR DISCUSSION GUIDE BE PREPARED For the best outcome from a visit with your doctor, it s important to be prepared. The more completely and clearly you describe the pain you re experiencing, the easier
DOCTOR DISCUSSION GUIDE BE PREPARED For the best outcome from a visit with your doctor, it s important to be prepared. The more completely and clearly you describe the pain you re experiencing, the easier
USING YOUR IMPLANTED SPINAL CORD STIMULATION SYSTEM
 USING YOUR IMPLANTED SPINAL CORD STIMULATION SYSTEM OUT OF PAIN. INTO RELIEF. USING THIS GUIDE This guide provides basic information to help you use your Medtronic spinal cord stimulation system. It also
USING YOUR IMPLANTED SPINAL CORD STIMULATION SYSTEM OUT OF PAIN. INTO RELIEF. USING THIS GUIDE This guide provides basic information to help you use your Medtronic spinal cord stimulation system. It also
Neurostimulation Systems
 Neurostimulation Systems Expanding the Array of Pain Control Solutions PROVEN EFFECTIVE FOR SIMPLE PAIN ITREL 3 SYSTEM For simple neuropathic pain Totally implantable for enhanced quality of life Easy
Neurostimulation Systems Expanding the Array of Pain Control Solutions PROVEN EFFECTIVE FOR SIMPLE PAIN ITREL 3 SYSTEM For simple neuropathic pain Totally implantable for enhanced quality of life Easy
LIVE LIFE WITH LESS PAIN
 LIVE LIFE WITH LESS PAIN Medtronic Pain Therapies Medtronic Inc. 710 Medtronic Pkwy. Minneapolis, MN 55432 USA Tel. 1-763-505-5000 medtronic.com 2017 Medtronic. UC201502755c EN Living with pain can be
LIVE LIFE WITH LESS PAIN Medtronic Pain Therapies Medtronic Inc. 710 Medtronic Pkwy. Minneapolis, MN 55432 USA Tel. 1-763-505-5000 medtronic.com 2017 Medtronic. UC201502755c EN Living with pain can be
PATIENT CONTROLLER ST. JUDE MEDICAL UNDERSTANDING IMPORTANT FEATURES ON YOUR
 UNDERSTANDING IMPORTANT FEATURES ON YOUR ST. JUDE MEDICAL PATIENT CONTROLLER THINGS TO KNOW BEFORE AND AFTER AN MRI OR SURGICAL PROCEDURE WHEN YOU HAVE A ST. JUDE MEDICAL PROCLAIM NEUROSTIMULATION SYSTEM
UNDERSTANDING IMPORTANT FEATURES ON YOUR ST. JUDE MEDICAL PATIENT CONTROLLER THINGS TO KNOW BEFORE AND AFTER AN MRI OR SURGICAL PROCEDURE WHEN YOU HAVE A ST. JUDE MEDICAL PROCLAIM NEUROSTIMULATION SYSTEM
PARKINSON S SYMPTOM TRACKER
 PARKINSON S SYMPTOM You can help your doctor make good treatment decisions by tracking your symptoms. A well-kept Symptom Tracker provides a clear picture of when you are taking your medications, when
PARKINSON S SYMPTOM You can help your doctor make good treatment decisions by tracking your symptoms. A well-kept Symptom Tracker provides a clear picture of when you are taking your medications, when
Proven pain therapy. Stimulating results.
 Proven pain therapy. Stimulating results. Living with chronic pain can be difficult. We re here to help. We understand the physical and mental suffering associated with chronic pain. Not being able to
Proven pain therapy. Stimulating results. Living with chronic pain can be difficult. We re here to help. We understand the physical and mental suffering associated with chronic pain. Not being able to
DRG THERAPY FOR CHRONIC PAIN ACCURATE CLINICAL STUDY FACT SHEET FOR PATIENTS
 ACCURATE CLINICAL STUDY DRG THERAPY FOR CHRONIC PAIN FACT SHEET FOR PATIENTS It was the kind of pain where you couldn t push through it now I am back to living life again and not having any pain. Jenifer,
ACCURATE CLINICAL STUDY DRG THERAPY FOR CHRONIC PAIN FACT SHEET FOR PATIENTS It was the kind of pain where you couldn t push through it now I am back to living life again and not having any pain. Jenifer,
Take control of your pain therapy. Getting started with Algovita SCS.
 Take control of your pain therapy. Getting started with Algovita SCS. The two-step approach to spinal cord stimulation therapy. You ve talked with your doctor. You ve discussed it with your family. You
Take control of your pain therapy. Getting started with Algovita SCS. The two-step approach to spinal cord stimulation therapy. You ve talked with your doctor. You ve discussed it with your family. You
PATIENT PROGRAMMER 97740
 PATIENT PROGRAMMER 97740 Pain therapy user manual for neurostimulation system models 37022, 37701, 37702, 37703, 37704, 37711, 37712, 37713, 37714, 97702, 97712, 97713, 97714! USA Rx only 2013 Medtronic,
PATIENT PROGRAMMER 97740 Pain therapy user manual for neurostimulation system models 37022, 37701, 37702, 37703, 37704, 37711, 37712, 37713, 37714, 97702, 97712, 97713, 97714! USA Rx only 2013 Medtronic,
Surgical Lead Implantation Guide
 Surgical Lead Implantation Guide Neurostimulation Therapy for Chronic Pain Innovating for life. Medtronic, PrimeAdvanced, Restore, RestoreAdvanced, RestoreUltra, and Specify are registered trademarks of
Surgical Lead Implantation Guide Neurostimulation Therapy for Chronic Pain Innovating for life. Medtronic, PrimeAdvanced, Restore, RestoreAdvanced, RestoreUltra, and Specify are registered trademarks of
St. Jude Medical 8-Channel Adapter. Clinician's Manual
 St. Jude Medical 8-Channel Adapter Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. For U.S. California Only: Proposition 65, a State of California
St. Jude Medical 8-Channel Adapter Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. For U.S. California Only: Proposition 65, a State of California
AT THE FOREFRONT OF MENDING THE BRAIN
 AT THE FOREFRONT OF MENDING THE BRAIN Abbott's neuromodulation portfolio fuels earnings growth while offering lifechanging treatment Many people who are dealt with a diagnosis of Parkinson's disease or
AT THE FOREFRONT OF MENDING THE BRAIN Abbott's neuromodulation portfolio fuels earnings growth while offering lifechanging treatment Many people who are dealt with a diagnosis of Parkinson's disease or
InterStim Therapy. symptom diary
 InterStim Therapy symptom diary Symptom DIARY Begin date: End date: Baseline Test assessment Follow-up visit 2 Your doctor and Medtronic, maker of InterStim Therapy, know that finding the right treatment
InterStim Therapy symptom diary Symptom DIARY Begin date: End date: Baseline Test assessment Follow-up visit 2 Your doctor and Medtronic, maker of InterStim Therapy, know that finding the right treatment
Algovita. MRI Procedure Guidelines. Spinal Cord Stimulation System. 201x
 Algovita Spinal Cord Stimulation System MRI Procedure Guidelines Read this manual before performing an MRI scan on a patient implanted with an Algovita Spinal Cord Stimulation System. ONLY 201x Algovita
Algovita Spinal Cord Stimulation System MRI Procedure Guidelines Read this manual before performing an MRI scan on a patient implanted with an Algovita Spinal Cord Stimulation System. ONLY 201x Algovita
Read this manual before performing an MRI scan on a patient implanted with an Algovita Spinal Cord Stimulation System.
 Algovita Spinal Cord Stimulation System MRI Procedure Guidelines Read this manual before performing an MRI scan on a patient implanted with an Algovita Spinal Cord Stimulation System. ONLY 0300-000175-001
Algovita Spinal Cord Stimulation System MRI Procedure Guidelines Read this manual before performing an MRI scan on a patient implanted with an Algovita Spinal Cord Stimulation System. ONLY 0300-000175-001
DRG STIMULATION FOR CHRONIC PAIN FOCUS ON YOUR LIFE NOT YOUR PAIN
 FOCUS ON YOUR LIFE NOT YOUR PAIN DRG STIMULATION FOR CHRONIC PAIN Focus on your friendships, not on your pain. FOCUS ON YOUR LIFE, NOT ON YOUR PAIN. Chronic pain is personal, and it can be difficult to
FOCUS ON YOUR LIFE NOT YOUR PAIN DRG STIMULATION FOR CHRONIC PAIN Focus on your friendships, not on your pain. FOCUS ON YOUR LIFE, NOT ON YOUR PAIN. Chronic pain is personal, and it can be difficult to
DRG STIMULATION INTERVENTIONAL PAIN CENTER
 DRG STIMULATION Shawn M. Sills, MD at Touchstone Interventional Pain Center has been selected by St Jude Medical to be first in Southern Oregon delivering this new FDA approved therapy to patients. You
DRG STIMULATION Shawn M. Sills, MD at Touchstone Interventional Pain Center has been selected by St Jude Medical to be first in Southern Oregon delivering this new FDA approved therapy to patients. You
Deep Brain Stimulation (DBS) For patients with tremor
 Patient Education Deep Brain Stimulation (DBS) For patients with tremor Please read this handout before reading and signing the form Special Consent for Procedural Treatment. This handout describes how
Patient Education Deep Brain Stimulation (DBS) For patients with tremor Please read this handout before reading and signing the form Special Consent for Procedural Treatment. This handout describes how
MEDTRONIC CARELINK NETWORK FOR PACEMAKERS. Comparison between the Medtronic CareLink Network for Pacemakers and Transtelephonic Monitoring
 MEDTRONIC CARELINK NETWORK FOR PACEMAKERS Comparison between the Medtronic CareLink Network for Pacemakers and Transtelephonic Monitoring Transtelephonic Monitoring Transmission What can you determine
MEDTRONIC CARELINK NETWORK FOR PACEMAKERS Comparison between the Medtronic CareLink Network for Pacemakers and Transtelephonic Monitoring Transtelephonic Monitoring Transmission What can you determine
Activa RC and Activa PC
 Activa RC and Activa PC neurostimulators for deep brain stimulation From the company with the most experience in DBS The Activa Family Innovative programming platform More choices for patients With the
Activa RC and Activa PC neurostimulators for deep brain stimulation From the company with the most experience in DBS The Activa Family Innovative programming platform More choices for patients With the
Model 5392 EPG Temporary Pacer
 Model 5392 EPG Temporary Pacer Compatible Components Reference Card 5392 Surgical Cables 5487 Disposable, short 5487L Disposable, long 5832S Reusable, small clip 5833S 5833SL Disposable, small clip, short
Model 5392 EPG Temporary Pacer Compatible Components Reference Card 5392 Surgical Cables 5487 Disposable, short 5487L Disposable, long 5832S Reusable, small clip 5833S 5833SL Disposable, small clip, short
Percutaneous Lead Kit. Models 3143, 3146, 3149, 3153, 3156, 3159, 3183, 3186, Clinician's Manual
 Percutaneous Lead Kit Models 3143, 3146, 3149, 3153, 3156, 3159, 3183, 3186, 3189 Clinician's Manual Unless otherwise noted, indicates that the name is a trademark of, or licensed to, St. Jude Medical
Percutaneous Lead Kit Models 3143, 3146, 3149, 3153, 3156, 3159, 3183, 3186, 3189 Clinician's Manual Unless otherwise noted, indicates that the name is a trademark of, or licensed to, St. Jude Medical
8/28/18. Anesthetic considerations for patients with implanted devices for treating chronic pain and more. Objectives:
 Anesthetic considerations for patients with implanted devices for treating chronic pain and more Alaa Abd-Elsayed, MD, MPH Medical Director, UW Pain Services Medical Director, Pain Clinic Section Head,
Anesthetic considerations for patients with implanted devices for treating chronic pain and more Alaa Abd-Elsayed, MD, MPH Medical Director, UW Pain Services Medical Director, Pain Clinic Section Head,
Sacral Nerve Stimulation (SNS) System Implantation
 Patient & Family Guide 2016 Sacral Nerve Stimulation (SNS) System Implantation www.nshealth.ca Sacral Nerve Stimulation (SNS) System Implantation SNS system implantation involves placing a lead ( leed,
Patient & Family Guide 2016 Sacral Nerve Stimulation (SNS) System Implantation www.nshealth.ca Sacral Nerve Stimulation (SNS) System Implantation SNS system implantation involves placing a lead ( leed,
Paddle Lead Kit. Models 3214, 3219, 3224, 3228, 3240, 3243, 3244, 3245, 3246, 3262, 3266, 3268, 3283, 3286, Clinician's Manual
 Paddle Lead Kit Models 3214, 3219, 3224, 3228, 3240, 3243, 3244, 3245, 3246, 3262, 3266, 3268, 3283, 3286, 3288 Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the
Paddle Lead Kit Models 3214, 3219, 3224, 3228, 3240, 3243, 3244, 3245, 3246, 3262, 3266, 3268, 3283, 3286, 3288 Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the
Discussion Points 10/17/16. Spine Pain is Ubiquitous. Interventional Pain Management
 Interventional Pain Management Blake Christensen, D.O. Fellowship Trained Interventional Pain Management Board Eligible in Anesthesiology and Interventional Pain Management Oklahoma Interventional Pain
Interventional Pain Management Blake Christensen, D.O. Fellowship Trained Interventional Pain Management Board Eligible in Anesthesiology and Interventional Pain Management Oklahoma Interventional Pain
DBS Extension Kit for Deep Brain Stimulation
 DBS Extension Kit for Deep Brain Stimulation 7482 Implant manual Rx Only 26 mm 30 mm Ø2.8mm 10-110 cm 3.8 mm Model 7482 Extension (All dimensions are approximate.) iii The following are trademarks of
DBS Extension Kit for Deep Brain Stimulation 7482 Implant manual Rx Only 26 mm 30 mm Ø2.8mm 10-110 cm 3.8 mm Model 7482 Extension (All dimensions are approximate.) iii The following are trademarks of
PAIN INTER- DRG THERAPY FOR CHRONIC PAIN RUPTED LIFE TRANSFORMED
 PAIN INTER- RUPTED DRG THERAPY FOR CHRONIC PAIN LIFE TRANSFORMED A DIFFERENT TYPE OF PAIN CALLS FOR A DIFFERENT APPROACH. Chronic pain* is a common condition, affecting almost one in five adults globally.
PAIN INTER- RUPTED DRG THERAPY FOR CHRONIC PAIN LIFE TRANSFORMED A DIFFERENT TYPE OF PAIN CALLS FOR A DIFFERENT APPROACH. Chronic pain* is a common condition, affecting almost one in five adults globally.
FREEDOM SPINAL CORD STIMULATION SYSTEM WAVECREST PROGRAMMER USER MANUAL
 FREEDOM SPINAL CORD STIMULATION SYSTEM WAVECREST PROGRAMMER USER MANUAL Caution: Federal law restricts this device to sale by or on the order of a physician. WAVECREST PROGRAMMING SOFTWARE VERSION 2.0
FREEDOM SPINAL CORD STIMULATION SYSTEM WAVECREST PROGRAMMER USER MANUAL Caution: Federal law restricts this device to sale by or on the order of a physician. WAVECREST PROGRAMMING SOFTWARE VERSION 2.0
Patient Information FOR NEUROSTIMULATION
 Patient Information FOR NEUROSTIMULATION Every 5 seconds somewhere in the world another person s life is improved by a Medtronic product. At Medtronic, with over 30 years of collaboratively working with
Patient Information FOR NEUROSTIMULATION Every 5 seconds somewhere in the world another person s life is improved by a Medtronic product. At Medtronic, with over 30 years of collaboratively working with
Lead and Extension Insertion Tool. Model Clinician's Manual
 Lead and Extension Insertion Tool Model 1803 Clinician's Manual Unless otherwise noted, indicates that the name is a trademark of, or licensed to, St. Jude Medical or one of its subsidiaries. ST. JUDE
Lead and Extension Insertion Tool Model 1803 Clinician's Manual Unless otherwise noted, indicates that the name is a trademark of, or licensed to, St. Jude Medical or one of its subsidiaries. ST. JUDE
BEFORE AND AFTER YOUR MRI SCAN. For patients with St. Jude Medical neurostimulation systems for chronic pain
 BEFORE AND AFTER YOUR MRI SCAN For patients with St. Jude Medical neurostimulation systems for chronic pain Preparing for your MRI Depending on what type of neurostimulation system you have, you may be
BEFORE AND AFTER YOUR MRI SCAN For patients with St. Jude Medical neurostimulation systems for chronic pain Preparing for your MRI Depending on what type of neurostimulation system you have, you may be
Implantable Pulse Generator. Prodigy IPG Prodigy MRI IPG. Clinician's Manual
 Implantable Pulse Generator Prodigy IPG Prodigy MRI IPG Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. Unless otherwise noted, indicates
Implantable Pulse Generator Prodigy IPG Prodigy MRI IPG Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. Unless otherwise noted, indicates
EN/197877_001/ccs 1/29/01 2:18 PM Page 1. Itrel. EZ Model 7434A Patient Programmer. User Manual Rx Only
 9900213EN/197877_001/ccs 1/29/01 2:18 PM Page 1 Itrel EZ Model 7434A Patient Programmer User Manual Rx Only 9V 9900213EN/197877_001/ccs 1/29/01 2:18 PM Page 2 Explanation of Symbols on Products and Packaging
9900213EN/197877_001/ccs 1/29/01 2:18 PM Page 1 Itrel EZ Model 7434A Patient Programmer User Manual Rx Only 9V 9900213EN/197877_001/ccs 1/29/01 2:18 PM Page 2 Explanation of Symbols on Products and Packaging
Chronic pain Neurostimulation What is neurostimulation and how does it work? Neurostimulation is a proven, effective, reversible treatment
 Chronic pain Neurostimulation What is neurostimulation and how does it work? Neurostimulation is a proven, effective, reversible treatment alternative when other pain treatments provide unsatisfactory
Chronic pain Neurostimulation What is neurostimulation and how does it work? Neurostimulation is a proven, effective, reversible treatment alternative when other pain treatments provide unsatisfactory
Q & A: How to Safely Scan Patients with a SureScan MRI Pacemaker
 Q & A: How to Safely Scan Patients with a SureScan MRI Pacemaker Patient Scheduling and Screening Q: Is there any special (other than regular MRI consent) consent form that should be signed by the patient?
Q & A: How to Safely Scan Patients with a SureScan MRI Pacemaker Patient Scheduling and Screening Q: Is there any special (other than regular MRI consent) consent form that should be signed by the patient?
DRG STIMULATION FOR CHRONIC PAIN FOCUS ON YOUR LIFE NOT YOUR PAIN
 FOCUS ON YOUR LIFE NOT YOUR PAIN DRG STIMULATION FOR CHRONIC PAIN Focus on your friendships, not on your pain. FOCUS ON YOUR LIFE, NOT ON YOUR PAIN. Chronic pain is personal, and it can be difficult to
FOCUS ON YOUR LIFE NOT YOUR PAIN DRG STIMULATION FOR CHRONIC PAIN Focus on your friendships, not on your pain. FOCUS ON YOUR LIFE, NOT ON YOUR PAIN. Chronic pain is personal, and it can be difficult to
Prodigy Neurostimulation System Programming and Reference. Models 3855, Clinician's Manual
 Prodigy Neurostimulation System Programming and Reference Models 3855, 3856 Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. Unless otherwise
Prodigy Neurostimulation System Programming and Reference Models 3855, 3856 Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. Unless otherwise
AWAY Model # RTLAGF-900 REV
 AWAY Model # RTLAGF-900 REV2.4.9.15 Contents User manual English 2 General Description 2 Intended Use 3 Included in this Package 4 Instructions for Use 7 Application Duration 7 Electrode Placement 8 Tips
AWAY Model # RTLAGF-900 REV2.4.9.15 Contents User manual English 2 General Description 2 Intended Use 3 Included in this Package 4 Instructions for Use 7 Application Duration 7 Electrode Placement 8 Tips
ONLY. Physician Implant Manual
 ONLY Physician Implant Manual NEVRO CORP. All questions or concerns about Nevro Corp. products should be forwarded to: Nevro Corp. 1800 Bridge Parkway Redwood City, CA 94065, USA Tel: +1.650.251.0005 Fax:
ONLY Physician Implant Manual NEVRO CORP. All questions or concerns about Nevro Corp. products should be forwarded to: Nevro Corp. 1800 Bridge Parkway Redwood City, CA 94065, USA Tel: +1.650.251.0005 Fax:
AspireSR. Essential information about your new device
 AspireSR Essential information about your new device FAQs for Patients Your doctor recommends that you receive the latest VNS Therapy device. This brochure will give you more information on the differences
AspireSR Essential information about your new device FAQs for Patients Your doctor recommends that you receive the latest VNS Therapy device. This brochure will give you more information on the differences
Parkinson s disease, Essential Tremor and primary dystonia
 Parkinson s disease, Essential Tremor and primary dystonia What is Deep Brain Stimulation and how does it work? Deep Brain Stimulation (DBS) uses one or two surgically implanted medical devices, similar
Parkinson s disease, Essential Tremor and primary dystonia What is Deep Brain Stimulation and how does it work? Deep Brain Stimulation (DBS) uses one or two surgically implanted medical devices, similar
Dr. Guy Buchanan MBBS, FANZCA, FFPMANZCA Specialist Pain Medicine Physician & Anaesthetist Associate - Victoria Pain Specialists
 anagement of Patients with Implante Dr. Guy Buchanan MBBS, FANZCA, FFPMANZCA Specialist Pain Medicine Physician & Anaesthetist Associate - Victoria Pain Specialists overview brief introduction to the
anagement of Patients with Implante Dr. Guy Buchanan MBBS, FANZCA, FFPMANZCA Specialist Pain Medicine Physician & Anaesthetist Associate - Victoria Pain Specialists overview brief introduction to the
2014 AAPM 56 th Annual Meeting
 2014 AAPM 56 th Annual Meeting SAM Diagnostic Radiology MR Safety - Deep Brain Stimulator and Other Neurostimulators Yunhong Shu, Ph.D. Mayo Clinic, Rochester, MN Outline Background of Neurostimulator
2014 AAPM 56 th Annual Meeting SAM Diagnostic Radiology MR Safety - Deep Brain Stimulator and Other Neurostimulators Yunhong Shu, Ph.D. Mayo Clinic, Rochester, MN Outline Background of Neurostimulator
Clinician's Manual. Paddle Lead Kit
 Paddle Lead Kit Models 3214, 3218, 3219, 3220, 3224, 3227, 3228, 3229, 3240, 3243, 3244, 3245, 3246, 3262, 3266, 3268, 3283, 3286, 3288 Clinician's Manual Unless otherwise noted, indicates that the name
Paddle Lead Kit Models 3214, 3218, 3219, 3220, 3224, 3227, 3228, 3229, 3240, 3243, 3244, 3245, 3246, 3262, 3266, 3268, 3283, 3286, 3288 Clinician's Manual Unless otherwise noted, indicates that the name
UNLOCK THE ANSWER. The Reveal LINQ TM System lets your physician learn about your heart while you live your life.
 UNLOCK THE ANSWER The Reveal LINQ TM System lets your physician learn about your heart while you live your life. WHY LONG-TERM MONITORING MAY BE RIGHT FOR YOU Accurate information is the key to better
UNLOCK THE ANSWER The Reveal LINQ TM System lets your physician learn about your heart while you live your life. WHY LONG-TERM MONITORING MAY BE RIGHT FOR YOU Accurate information is the key to better
Precision Montage MRI 16 Contact Implantable Pulse Generator
 Precision Montage MRI 16 Contact Implantable Pulse Generator Directions for Use CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 91053657-02 REV
Precision Montage MRI 16 Contact Implantable Pulse Generator Directions for Use CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 91053657-02 REV
PAIN LIFE INTER- RUPTED TRANSFORMED BURSTDR STIMULATION FOR CHRONIC PAIN
 PAIN INTER- RUPTED BURSTDR STIMULATION FOR CHRONIC PAIN LIFE TRANSFORMED p Sam B, 18 months after implant and able to keep things shipshape on the waters of San Francisco Bay. IMAGINE YOUR LIFE TRANSFORMED
PAIN INTER- RUPTED BURSTDR STIMULATION FOR CHRONIC PAIN LIFE TRANSFORMED p Sam B, 18 months after implant and able to keep things shipshape on the waters of San Francisco Bay. IMAGINE YOUR LIFE TRANSFORMED
IS THERE A LINK BETWEEN ATRIAL FIBRILLATION AND MY STROKE? Finding answers about cryptogenic stroke
 IS THERE A LINK BETWEEN ATRIAL FIBRILLATION AND MY STROKE? Finding answers about cryptogenic stroke UNDERSTANDING WHAT CAUSED YOUR STROKE IS VERY IMPORTANT A stroke happens when a blood vessel in the brain
IS THERE A LINK BETWEEN ATRIAL FIBRILLATION AND MY STROKE? Finding answers about cryptogenic stroke UNDERSTANDING WHAT CAUSED YOUR STROKE IS VERY IMPORTANT A stroke happens when a blood vessel in the brain
PACING AT THE BUNDLE OF HIS
 PACING AT THE BUNDLE OF HIS Cardiac Rhythm Heart Failure Coding Corner Disclaimer While Medtronic offers opinions about how to code when using our technology according to its FDA-approved labeling, these
PACING AT THE BUNDLE OF HIS Cardiac Rhythm Heart Failure Coding Corner Disclaimer While Medtronic offers opinions about how to code when using our technology according to its FDA-approved labeling, these
The VNS Therapy Magnet
 The VNS Therapy Magnet A unique benefit of VNS Therapy For patients with epilepsy treated with VNS Therapy What is the Patient Magnet? Use of the magnet is not necessary to receive VNS Therapy. The magnet
The VNS Therapy Magnet A unique benefit of VNS Therapy For patients with epilepsy treated with VNS Therapy What is the Patient Magnet? Use of the magnet is not necessary to receive VNS Therapy. The magnet
Physician Implant Manual
 Physician Implant Manual CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 9055521-001 Rev A Physician Implant Manual Copyright 2006 by Advanced
Physician Implant Manual CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 9055521-001 Rev A Physician Implant Manual Copyright 2006 by Advanced
Proclaim Implantable Pulse Generator. Models 3660, 3662, 3665, Clinician's Manual
 Proclaim Implantable Pulse Generator Models 3660, 3662, 3665, 3667 Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. Unless otherwise noted,
Proclaim Implantable Pulse Generator Models 3660, 3662, 3665, 3667 Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. Unless otherwise noted,
MRI GUIDELINES FOR INSPIRE THERAPY
 MRI GUIDELINES FOR INSPIRE THERAPY Clinician s Manual ONLY The following is a trademark of Inspire Medical Systems, Inc.: Inspire Introduction Read the information in this manual prior to conducting an
MRI GUIDELINES FOR INSPIRE THERAPY Clinician s Manual ONLY The following is a trademark of Inspire Medical Systems, Inc.: Inspire Introduction Read the information in this manual prior to conducting an
MRI GUIDELINES FOR INSPIRE THERAPY
 MRI GUIDELINES FOR INSPIRE THERAPY Clinician s Manual The following is a trademark of Inspire Medical Systems, Inc.: Inspire This product and/or the use of this product in a method may be covered by one
MRI GUIDELINES FOR INSPIRE THERAPY Clinician s Manual The following is a trademark of Inspire Medical Systems, Inc.: Inspire This product and/or the use of this product in a method may be covered by one
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: spinal_cord_stimulation 3/1980 10/2017 10/2018 10/2017 Description of Procedure or Service Spinal cord stimulation
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: spinal_cord_stimulation 3/1980 10/2017 10/2018 10/2017 Description of Procedure or Service Spinal cord stimulation
ICD-9-CM Diagnosis Code options
 ICD-9-CM Diagnosis Code options Diagnosis codes are used by both physicians and facilities to document the indication for the procedure. Intrathecal drug delivery is directed at managing chronic, intractable
ICD-9-CM Diagnosis Code options Diagnosis codes are used by both physicians and facilities to document the indication for the procedure. Intrathecal drug delivery is directed at managing chronic, intractable
WHAT CAN I DO ABOUT ATRIAL FIBRILLATION? Finding answers about atrial fibrillation
 WHAT CAN I DO ABOUT ATRIAL FIBRILLATION? Finding answers about atrial fibrillation DEBBIE S STORY With a full schedule of entertaining friends, dancing, working as a nurse and babysitting her granddog,
WHAT CAN I DO ABOUT ATRIAL FIBRILLATION? Finding answers about atrial fibrillation DEBBIE S STORY With a full schedule of entertaining friends, dancing, working as a nurse and babysitting her granddog,
Surgical Leads. Directions for Use. CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician.
 Surgical Leads Directions for Use CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 91078747-08 Content: 92190987 REV A Guarantees Boston Scientific
Surgical Leads Directions for Use CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 91078747-08 Content: 92190987 REV A Guarantees Boston Scientific
Tell the doctor who prescribed your MRI scan that you have an implanted Medtronic neurostimulation system.
 Before Your I For patients with Medtronic neurostimulation systems for chronic pain The following materials are intended to be used by implant physicians with patients who are to receive Medtronic neurostimulation
Before Your I For patients with Medtronic neurostimulation systems for chronic pain The following materials are intended to be used by implant physicians with patients who are to receive Medtronic neurostimulation
FREEDOM SPINAL CORD STIMULATION SYSTEM WAVECREST PROGRAMMER USER MANUAL
 FREEDOM SPINAL CORD STIMULATION SYSTEM WAVECREST PROGRAMMER USER MANUAL Caution: Federal law restricts this device to sale by or on the order of a physician. WAVECREST APPLICATION SOFTWARE VERSION 2.0
FREEDOM SPINAL CORD STIMULATION SYSTEM WAVECREST PROGRAMMER USER MANUAL Caution: Federal law restricts this device to sale by or on the order of a physician. WAVECREST APPLICATION SOFTWARE VERSION 2.0
PACEMAKERS ARE NOT JUST FOR THE HEART! Ab Siadati MD
 PACEMAKERS ARE NOT JUST FOR THE HEART! Ab Siadati MD WHAT IS DEEP BRAIN STIMULATION? WHY SHOULD YOU CONSIDER DBS SURGERY FOR YOUR PATIENTS? HOW DOES DBS WORK? DBS electrical stimulation overrides abnormal
PACEMAKERS ARE NOT JUST FOR THE HEART! Ab Siadati MD WHAT IS DEEP BRAIN STIMULATION? WHY SHOULD YOU CONSIDER DBS SURGERY FOR YOUR PATIENTS? HOW DOES DBS WORK? DBS electrical stimulation overrides abnormal
SYSTEM IMPLANT MANUAL
 SYSTEM IMPLANT MANUAL Inspire II Implantable Pulse Generator Model 3024 Stimulation Lead Model 4063 Sensing Lead Model 4323 Rx Only Table of Contents Explanation of Symbols on Product or Package Labeling
SYSTEM IMPLANT MANUAL Inspire II Implantable Pulse Generator Model 3024 Stimulation Lead Model 4063 Sensing Lead Model 4323 Rx Only Table of Contents Explanation of Symbols on Product or Package Labeling
Electrodes (for TransAeris System)
 Surgical Procedures for Implantation of TransLoc Electrodes (for TransAeris System) 1 R L 1 TransAeris System TransLoc Electrode Kit 3 3 TransAeris Patient Kit (FrictionLoc) TransAeris Patient Kit (TransAeris
Surgical Procedures for Implantation of TransLoc Electrodes (for TransAeris System) 1 R L 1 TransAeris System TransLoc Electrode Kit 3 3 TransAeris Patient Kit (FrictionLoc) TransAeris Patient Kit (TransAeris
Medtronic DBS Therapy Implanted neurostimulators
 Medtronic, Activa, Soletra, and Kinetra are registered trademarks of Medtronic, Inc. DBS is a trademark of Medtronic, Inc. Medtronic DBS Therapy Implanted neurostimulators Information for prescribers c
Medtronic, Activa, Soletra, and Kinetra are registered trademarks of Medtronic, Inc. DBS is a trademark of Medtronic, Inc. Medtronic DBS Therapy Implanted neurostimulators Information for prescribers c
Algovita Spinal Cord Stimulation System
 Algovita Spinal Cord Stimulation System Stimulator Models 2408 and 2412 For IPG replacement Implant Manual for Stimulator IPG implant.indb 1 Algovita is a trademark of QIG Group, LLC CoreGuard is a trademark
Algovita Spinal Cord Stimulation System Stimulator Models 2408 and 2412 For IPG replacement Implant Manual for Stimulator IPG implant.indb 1 Algovita is a trademark of QIG Group, LLC CoreGuard is a trademark
Instead of feeling pain, the patient will experience tingling.
 Spinal Cord Stimulation or SCS is a technique involving electrical stimulation of a precise part of the spinal cord. A very low energy current is used. This essentially shuts down the pain signals from
Spinal Cord Stimulation or SCS is a technique involving electrical stimulation of a precise part of the spinal cord. A very low energy current is used. This essentially shuts down the pain signals from
Your guidebook for getting results with Medtronic Bladder Control Therapy Delivered by the NURO System TAKE BACK CONTROL.
 Your guidebook for getting results with Medtronic Bladder Control Therapy Delivered by the NURO System TAKE BACK CONTROL. RESET. RESTORE. * REGAIN CONTROL. Congratulations! You ve decided to try a therapy
Your guidebook for getting results with Medtronic Bladder Control Therapy Delivered by the NURO System TAKE BACK CONTROL. RESET. RESTORE. * REGAIN CONTROL. Congratulations! You ve decided to try a therapy
See Policy CPT/HCPCS CODE section below for any prior authorization requirements
 Effective Date: 1/1/2019 Section: SUR Policy No: 395 1/1/19 Medical Policy Committee Approved Date: 8/17; 2/18; 12/18 Medical Officer Date APPLIES TO: Medicare Only See Policy CPT/HCPCS CODE section below
Effective Date: 1/1/2019 Section: SUR Policy No: 395 1/1/19 Medical Policy Committee Approved Date: 8/17; 2/18; 12/18 Medical Officer Date APPLIES TO: Medicare Only See Policy CPT/HCPCS CODE section below
Neurostimulation. Spinal cord stimulators use electrodes placed in the space surrounding the spinal cord (epidural space).
 Neurostimulation.305145613.pdf 1 8/11/12 11:32 PM Neurostimulation Neurostimulation, also called electrostimulation, uses low-level electrical impulses to interfere with pain signals or block them from
Neurostimulation.305145613.pdf 1 8/11/12 11:32 PM Neurostimulation Neurostimulation, also called electrostimulation, uses low-level electrical impulses to interfere with pain signals or block them from
Advanced ICD Concepts
 1 4 2 5 7 3 6 8 Advanced ICD Concepts This presentation is provided with the understanding that the slide content must not be altered in any manner as the content is subject to FDA regulations. This presentation
1 4 2 5 7 3 6 8 Advanced ICD Concepts This presentation is provided with the understanding that the slide content must not be altered in any manner as the content is subject to FDA regulations. This presentation
RNS System User Manual
 2018 NeuroPace, Inc. Rev Date: 2018-06 DN 1017337 Rev 2 This Manual supports: ii RNS Neurostimulator: model RNS-300M with firmware version 7.0 NeuroPace Cortical Strip Lead: models CL-315-10, CL-325-10,
2018 NeuroPace, Inc. Rev Date: 2018-06 DN 1017337 Rev 2 This Manual supports: ii RNS Neurostimulator: model RNS-300M with firmware version 7.0 NeuroPace Cortical Strip Lead: models CL-315-10, CL-325-10,
MRI Procedure Information. For St. Jude Medical MR Conditional Neurostimulation Systems. Clinician's Manual
 MRI Procedure Information For St. Jude Medical MR Conditional Neurostimulation Systems Clinician's Manual Unless otherwise noted, indicates that the name is a trademark of, or licensed to, St. Jude Medical
MRI Procedure Information For St. Jude Medical MR Conditional Neurostimulation Systems Clinician's Manual Unless otherwise noted, indicates that the name is a trademark of, or licensed to, St. Jude Medical
WHY AM I FAINTING? Finding answers about unexplained fainting
 WHY AM I FAINTING? Finding answers about unexplained fainting PERSONAL STORIES For about 15 years, Lloyd s friends and family thought he was clumsy. Each spring, he seemed to have an unusual accident.
WHY AM I FAINTING? Finding answers about unexplained fainting PERSONAL STORIES For about 15 years, Lloyd s friends and family thought he was clumsy. Each spring, he seemed to have an unusual accident.
PNS FOR BACK PAIN IMPROVING PATIENT OUTCOMES
 PNS FOR BACK PAIN IMPROVING PATIENT OUTCOMES New Level-1 evidence The SubQStim Study The Focus SM workflow A recommended and standardised approach for applying PNS based on clinical data BACK PAIN REDUCE
PNS FOR BACK PAIN IMPROVING PATIENT OUTCOMES New Level-1 evidence The SubQStim Study The Focus SM workflow A recommended and standardised approach for applying PNS based on clinical data BACK PAIN REDUCE
1.5 Tesla and 3 Tesla Magnetic Resonance Imaging (MRI) Guidelines for the Senza System (IPG1000 and IPG1500)
 1.5 Tesla and 3 Tesla Magnetic Resonance Imaging (MRI) Guidelines for the Senza System (IPG1000 and IPG1500) ONLY NEVRO CORP. All questions or concerns about Nevro products should be forwarded to: Nevro
1.5 Tesla and 3 Tesla Magnetic Resonance Imaging (MRI) Guidelines for the Senza System (IPG1000 and IPG1500) ONLY NEVRO CORP. All questions or concerns about Nevro products should be forwarded to: Nevro
Percutaneous Leads. Directions for Use Content: REV A
 Percutaneous Leads Directions for Use CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 91078744-07 Content: 92190387 REV A Guarantees Boston Scientific
Percutaneous Leads Directions for Use CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 91078744-07 Content: 92190387 REV A Guarantees Boston Scientific
Call Medtronic at 1 (800) to verify the patient s current implanted system
 MRI SURESCAN SYSTEMS Patient Scanning Process Transvenous Implantable Cardiac Systems PATIENT PRESCREENING SureScan Pacing, Defibrillation, and CRT (CRT-D and CRT-P) Systems Verification Verify that patient
MRI SURESCAN SYSTEMS Patient Scanning Process Transvenous Implantable Cardiac Systems PATIENT PRESCREENING SureScan Pacing, Defibrillation, and CRT (CRT-D and CRT-P) Systems Verification Verify that patient
RestoreSensor Implant manual. Multi-program rechargeable neurostimulator. ! USA Rx only
 RestoreSensor 37714 Multi-program rechargeable neurostimulator Implant manual! USA Rx only 2010 Explanation of symbols on product or package labeling Refer to the appropriate product for symbols that
RestoreSensor 37714 Multi-program rechargeable neurostimulator Implant manual! USA Rx only 2010 Explanation of symbols on product or package labeling Refer to the appropriate product for symbols that
CARDIAC DEVICE MR-CONDITIONAL PRODUCT SUMMARY CHART
 CARDIAC DEVICE MR-CONDITIONAL PRODUCT SUMMARY CHART December 2015 This chart encompasses all Medtronic cardiac devices FDA-Approved as MR Conditional and included in the MRI SureScan portfolio. If a device
CARDIAC DEVICE MR-CONDITIONAL PRODUCT SUMMARY CHART December 2015 This chart encompasses all Medtronic cardiac devices FDA-Approved as MR Conditional and included in the MRI SureScan portfolio. If a device
MRI Guidelines for VNS Therapy
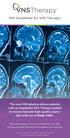 I Guidelines for VNS Therapy The new I labeling allows patients with an implanted VNS Therapy System to receive fast and high quality scans also with use of Body Coils*. I can be safely performed on patients
I Guidelines for VNS Therapy The new I labeling allows patients with an implanted VNS Therapy System to receive fast and high quality scans also with use of Body Coils*. I can be safely performed on patients
Sacral Nerve Stimulation. Patient Information Leaflet
 Sacral Nerve Stimulation Patient Information Leaflet Sacral Nerve Stimulation Sacral nerve stimulation (SNS) is a treatment option, initially developed for people suffering with bladder control problems
Sacral Nerve Stimulation Patient Information Leaflet Sacral Nerve Stimulation Sacral nerve stimulation (SNS) is a treatment option, initially developed for people suffering with bladder control problems
ImageReady MRI Head Only Guidelines for Spectra WaveWriter Spinal Cord Stimulator System
 ImageReady MRI Head Only Guidelines for Spectra WaveWriter Spinal Cord Stimulator System CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 91171762-01
ImageReady MRI Head Only Guidelines for Spectra WaveWriter Spinal Cord Stimulator System CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 91171762-01
MEET MICRA. Micra TM ACTUAL SIZE. Transcatheter Pacing System
 MEET MICRA ACTUAL SIZE Micra TM Transcatheter Pacing System MEET MICRA The world s smallest pacemaker 1 MINIATURIZED. 93% smaller than modern-day pacemakers 6 Completely self contained within the heart,
MEET MICRA ACTUAL SIZE Micra TM Transcatheter Pacing System MEET MICRA The world s smallest pacemaker 1 MINIATURIZED. 93% smaller than modern-day pacemakers 6 Completely self contained within the heart,
1.5 Tesla and 3 Tesla Magnetic Resonance Imaging (MRI) Guidelines for the Senza System (IPG1000 and IPG1500)
 1.5 Tesla and 3 Tesla Magnetic Resonance Imaging (MRI) Guidelines for the Senza System (IPG1000 and IPG1500). ONLY NEVRO CORP. All questions or concerns about Nevro products should be forwarded to: Nevro
1.5 Tesla and 3 Tesla Magnetic Resonance Imaging (MRI) Guidelines for the Senza System (IPG1000 and IPG1500). ONLY NEVRO CORP. All questions or concerns about Nevro products should be forwarded to: Nevro
MRI Procedure Information. For St. Jude Medical MR Conditional Neurostimulation Systems. Clinician's Manual
 MRI Procedure Information For St. Jude Medical MR Conditional Neurostimulation Systems Clinician's Manual Unless otherwise noted, indicates that the name is a trademark of, or licensed to, St. Jude Medical
MRI Procedure Information For St. Jude Medical MR Conditional Neurostimulation Systems Clinician's Manual Unless otherwise noted, indicates that the name is a trademark of, or licensed to, St. Jude Medical
Epidural Steroid Injection
 Epidural Steroid Injection Epidural steroid injections (ESI) are performed to place anti-inflammatory medication (steroid) and local anesthetic in the epidural space to target irritated nerves and relieve
Epidural Steroid Injection Epidural steroid injections (ESI) are performed to place anti-inflammatory medication (steroid) and local anesthetic in the epidural space to target irritated nerves and relieve
Implantation Procedure
 VNS Therapy System For Healthcare Professionals October 2010 Worldwide Version Caution: U.S. federal law restricts this device to sale by or on the order of a physician. 2011 0344 Note: This is one part
VNS Therapy System For Healthcare Professionals October 2010 Worldwide Version Caution: U.S. federal law restricts this device to sale by or on the order of a physician. 2011 0344 Note: This is one part
COMMONLY BILLED CODES SPINAL CORD STIMULATION FOR CHRONIC PAIN-TRUNK AND/OR LIMBS
 COMMONLY BILLED CODES SPINAL CORD STIMULATION FOR CHRONIC PAIN-TRUNK AND/OR LIMBS EFFECTIVE JANUARY 2018 1 Medtronic provides this information for your convenience only. It does not constitute legal advice
COMMONLY BILLED CODES SPINAL CORD STIMULATION FOR CHRONIC PAIN-TRUNK AND/OR LIMBS EFFECTIVE JANUARY 2018 1 Medtronic provides this information for your convenience only. It does not constitute legal advice
PATIENT PROGRAMMER 37642
 PATIENT PROGRAMMER 37642 Medtronic DBS Therapy user manual Activa PC Model 37601 Activa RC Model 37612 Activa SC Model 37602 Activa SC Model 37603! USA Rx only 2008 Medtronic, Activa, and SoftStart/Stop
PATIENT PROGRAMMER 37642 Medtronic DBS Therapy user manual Activa PC Model 37601 Activa RC Model 37612 Activa SC Model 37602 Activa SC Model 37603! USA Rx only 2008 Medtronic, Activa, and SoftStart/Stop
WEARABLE ANTENNA ASSEMBLY USER MANUAL
 WEARABLE ANTENNA ASSEMBLY USER MANUAL Caution: Federal law restricts this device to sale by or on the order of a physician. WEARABLE ANTENNA ASSEMBLY KIT (PDBT-915-2K) WEARABLE ANTENNA ASSEMBLY (PDBT-915-2A)
WEARABLE ANTENNA ASSEMBLY USER MANUAL Caution: Federal law restricts this device to sale by or on the order of a physician. WEARABLE ANTENNA ASSEMBLY KIT (PDBT-915-2K) WEARABLE ANTENNA ASSEMBLY (PDBT-915-2A)
BRAVO. ph Monitoring System. A patient-friendly test for heartburn
 BRAVO ph Monitoring System A patient-friendly test for heartburn Bravo ph Monitoring System Why Test for Heartburn? Do you have a great deal of indigestion or a burning sensation in the center of your
BRAVO ph Monitoring System A patient-friendly test for heartburn Bravo ph Monitoring System Why Test for Heartburn? Do you have a great deal of indigestion or a burning sensation in the center of your
SYNCHROMED ISOMED Implantable infusion systems
 Medtronic, SynchroMed, and IsoMed are registered trademarks of Medtronic, Inc. Lioresal is a registered trademark of Novartis Pharma, AG. SYNCHROMED ISOMED Implantable infusion systems Information for
Medtronic, SynchroMed, and IsoMed are registered trademarks of Medtronic, Inc. Lioresal is a registered trademark of Novartis Pharma, AG. SYNCHROMED ISOMED Implantable infusion systems Information for
