ASSESSMENT OF IMMUNOLOGICAL THERAPY WITH INFLIXIMAB AND ETANERCEPT IN THE TREATMENT OF PSORIASIS ARTHROPATHY
|
|
|
- Thomas Evan Banks
- 5 years ago
- Views:
Transcription
1 Vol XIV, Number 2, June 2010 Pages Copyright reserved 2010 ORIGINAL PAPERS ASSESSMENT OF IMMUNOLOGICAL THERAPY WITH INFLIXIMAB AND ETANERCEPT IN THE TREATMENT OF PSORIASIS ARTHROPATHY Liana Suciu, Carmen Cristescu, Mirela Voicu, Maria Suciu, B. Bumbăcilă University of Medicine and Pharmacy Timisoara, Faculty of Pharmacy, Department of Pharmacology and Clinical Pharmacy Abstract. Psoriasis arthritis is a seronegative chronic inflammatory arthropathy, which occurs in patients with psoriasis and is accompanied by characteristic symptoms. Shortcomings of conventional, frequent and sometimes severe side effects induced by classical immunomodulatory substances (methotrexate, cyclosporine, leflunomide), lack of ethyo-pathogenic theories has been leading to research on new therapeutic perspectives in psoriasis, including for psoriasis arthropathy. Pathogenesis of psoriasis has highlighted a number of changes related to keratinocytes, T lymphocytes, dermal fibroblasts and a number of genes encoding several types of proteins involved in proliferation of keratinocytes. The aim of the present study was to highlight the effectiveness of with biological agents (infliximab and etanercept), assessed by improvement of PASI score, and to assess the incidence of adverse effects detected in patients during the study group. Patients enrolled in the study were selected by eligibility criteria and the final number was 14, from which eight patients were treated with infliximab and six were treated with etanercept in Dermatology Clinic from Timisoara. In the infliximab - treated group to whom the drug was administered iv 5mg/kgc for two hours at weeks 0, 2, 6 and 8 weeks thereafter until week 24, a rate of 62.5% of cases showed a 75% improvement in index PASI, in 12 weeks. Evaluation performed at week 24 showed improvement of symptoms (objective by index steps) to 87.5% of patients, thus proving efficacy. Administration over 24 weeks resulted in adverse effects: myalgia and fever in 32% of patients, the increase of transaminases in 25% of patients. No patient treated with infliximab did not show de novo appearance of antibodies antiadn DC. The group of patients treated with Etanercept, administered in doses 25mg/twice a week, for 24 weeks, were obtained the following results: after 12 treatment PASI index improved to 50% of cases, after 24 weeks the percentage of patients which occurred improvement was Adverse effects in this group were represented by: transient leukopenia and thrombocytopenia (one case), autoimmune events (one case) and local reactions as redness and induration at the site of administration, delivered spontaneously (two patients). This study has shown considerable clinical effects in psoriasis arthropathy, but it is not considered a first-line because of the incidence of adverse events which has to be monitored and interpreted in the context of each patient's clinical condition on such and not least because of the extremely high cost of the treatment. Keywords: psoriasis arthropathy,, biological agents Introduction Psoriasis arthropathy is a chronic seronegative inf lammator y arthritis Liana Suciu University of Medicine and Pharmacy 2 Piata Eftimie Murgu Str., Timişoara lianads@yahoo.com which occurs in patients with psoriasis and is accompanied by characteristic symptoms. The onset is insidious for the articular manifestations in most patients. The onset of psoriasis lesions precedes in most cases the articular manifestation, only about 10% of the cutaneous manifestations appear simultaneously with those of the joint [1,2]. The joint, in addition to events occurring in the skin and nails of patients with psoriasis, presents features XIV, Vol.14, Number. 2/
2 Immunological in the treatment of psoriasis arthropathy that suggest the diagnosis of psoriatic arthritis: a) it relatively frequently involves isolated distal interphalangeal joints of the upper fingers and lower limbs, with asymmetric oligoartropathy; b) axial affliction of the spine and sacroiliac joint; c) the presence of osteolysis. Psoriasis arthropathy is considered less aggressive than rheumatoid arthritis. However, disease progression is variable from destructive forms, mild to mutilating forms, leading to severe bone loss and change fingers. Presently, it is known that 40-50% of patients have distorting arthritis; in a cohort study from Toronto, Gladman observed that 11-19% of patients have increased disability and mortality rates than those of the general population [1,6]. The axial form of psoriatic arthritis develops slowly, with limited repercussions on the backbone, in contrast to radiological changes which increase over time. Among the complications seen in patients with psoriatic arthritis we can distinguish: i) psoriatic enteropathy described by Sushter M. (1967) which manifests itself by malabsorption syndrome and small bowel villous atrophy; ii) renal amyloidosis; iii) heart failure, atrioventricular block. Currently, it is considered that psoriasis is an immune-mediated disease; its pathogenesis highlights a number of changes related to keratinocytes, T lymphocytes, endothelial cells, dermal fibroblasts. The main pathological changes of psoriasis are: rapid proliferation and differentiation of keratinocytes and abnormal and inflammatory infiltrate in the dermis, epidermis and the synovial [1,3]. Shortcomings of conventional and frequent and sometimes severe side effects are induced by conventional systemic (methotrexate, cyclosporine, leflunomide); the lack of etiopathogenic theories have led to research on new therapeutic perspectives in psoriasis, including arthropathy psoriasis. A number of studies have demonstrated the role of T lymphocytes in the initiation and maintenance of abnormal proliferation and differentiation process of keratinocytes. The interaction between activated T lymphocytes and keratinocytes is achieved mainly through proinflammatory cytokines: interferongamma, TNF-alpha. Other molecules involved in this process are: E selectins and HLA-DR molecules. On the activated T lymphocytes surface, there were identified markers of the type CD2 + CD3 +, CD5 +, CD28 +, CLA +, CD25 (IL-2 receptor), CD27. Cytokine production has the effect of keratinocyte proliferation, neutrophil migration, angiogenesis and increased expression of adhesion molecules. Experimental models demonstrated that epithelial hyperplasia is dependent in particular of TNF-alpha, a cytokine secreted by keratinocytes. Keratinocytes have a basal secretion of TNF-alpha, but in the epithelial hyperplasia processes, TNFalpha is significantly elevated both in the serum and on local level. For these reasons it sought to block the activity of this cytokine and reduced epithelial hyperplasia [4,5,7]. Infliximab is a chimera monoclonal antibody that is a human immunoglobulin constant region and murine variable region. Infliximab is able to pair both serum TNF alpha and the transmembrane forms of TNF-alpha. By this coupling, TNF-alpha cannot exercise its proinflammatory activity. Infliximab is currently used to treat Crohn s disease, rheumatoid arthritis and moderate to severe forms of psoriasis and psoriatic arthritis. Prolonged administration of infliximab has led to several adverse effects, including myalgia, arthralgia, pyrexia, rash, increased transaminases, headache, vertigo, infections. A special reaction is given by the occurrence of autoimmune biological changes: anti-dna antibodies in patients treated with infliximab. The clinical significance of these antibodies is not known. The risk of a malignant tumor, by blocking TNF-alpha is an important element; it seems to be increased, but this is hard to prove in clinical trials [2,4]. Etanercept is a recombinant protein containing the p75 TNF receptor and the Fc portion of human IgG1 immunoglobulin. This molecule binds competitively to TNF alpha and TNF beta, preventing them from interacting with their membrane receptors and therefore, from exercising their proinflammatory effects. Etanercept has proven to be effective in spondylitis and medium and severe forms of psoriasis. In this case there were reported autoimmune phenomena and theoretical risk of developing cancer by inhibiting the TNF [5,6]. The aim of the study is to assess: the effectiveness of with these biological agents and incidence of adverse effects arising from administering the two drugs. Materials and methods In this retrospective study the information was extracted from the files of patients treated as outpatient in the Dermatology Clinic of Timişoara. 118
3 Liana Suciu et al Initially, the group consisted of 80 patients receiving conventional systemic and topical with methotrexate, retinoids, cyclosporine, pimecrolimus. Of these, 14 patients corresponded to one of the following criteria and were included in the study: have developed or are at risk to develop toxicity from therapies used, so the standard could not be used; have become intolerant to standard therapies; became non-responsive to with methotrexate, acitretin or their combination (clinical response <50% in terms of improving the PASI score); have a disease that requires repeated hospitalizations (more than three hospitalizations per year); present comorbidities which excluded systemic. Based on clinical and biological evaluation, the following patients were excluded from the study group: patients with active infections: sepsis, an active tuberculosis, opportunistic infections; heart failure patients, class NYHA III/IV; concomitant administration of live vaccines with germ; pregnancy/lactation; patients under the age of 17 years; patients with malignancy or premalignant stage of disease; patients with demyelination; patients with a hypersensitivity to one of the components of the product. From the group of 14 patients, 8 were treated with infliximab and six were treated with etanercept over a period of 24 weeks. Effectiveness evaluation was conducted by analyzing changes in the PASI index (psoriasis area and severity index); although it is the most common score, it presents limitations. PASI score was assessed for each group initially, when the patient was included in the study and then at weeks 12 and 24. Laboratory tests were conducted bimonthly (blood count, liver tests) and clinical consultations were held monthly. Infliximab was administrated to 8 patients, tree female and five male, with a mean age of 49 years (30-75). Infliximab was administered at a dose of 5 mg/kgc, by intravenous infusion over 2-3 hours at weeks 0, 2, 6 and then again as one injection every eight weeks. In the group treated with etanercept there were three females and three males, with a mean age of 56 years (40-80). Etanercept was administered at doses of 25 mg subcutaneously twice weekly for 24 weeks. Results and discussions It can be noticed from table no. I that the group of patients treated with infliximab achieved an improvement of PASI score with 75% after 12 weeks of treatment in 62.5% of the cases, percentage that increased to 87.5% at week 24 of treatment. The incidence of adverse events was as follows: upper respiratory tract infection: 2 patients; headache: 2 patients; increase in serum transaminases: 2 patients; fatigue: 2 patients; myocardial infarction: one patient. No patients treated with infliximab showed the de novo appearance of antibodies Anti dsdna. In the group treated with infliximab, the average PASI score was 59 ± After 12 No. Patient Name Age (years) Sex Initial PASI Score after 12 after 24 Final variation of (%) 1 M.I. 50 F ,7 2 N.O. 75 M ,9 3 P.C. 42 F ,5 4 B.I. 39 M ,1 5 C.C. 50 F ,1 6 G.I. 48 M ,4 7 A.M. 60 M ,7 8 T.F. 30 M ,3 Avg±SD 49,2 ± 13,6 59 ± 13,1 26,1 ± 16,8 15,7 ± 5,4 73 ± 8,6 Table I. Characteristics of patients treated with infliximab XIV, Vol.14, Number. 2/
4 Immunological in the treatment of psoriasis arthropathy Figure 1. Evolution of PASI score in the group of patients treated with infliximab treatment, PASI average score decreased significantly (26.1 ± 16.8, p = 0.003, non-parametric test Mann-Whitney). In week 24 of treatment, the scores decreased compared to the PASI score at 12 weeks, but without reaching statistical significance (15.75 ± 5.4, p = 0.2, non-parametric test Mann-Whitney). The overall average PASI score improved with 73% from baseline (p = , non-parametric test Mann-Whitney) (figure 1). It should be noted that occasionally, antiinflammatory treatments have been used, according to symptoms. A patient with psoriasis, skin and nail lesions that presented a score of 80 at the beginning of NAPS nail treatment showed a very good recovery of the nail blade at 12 weeks of treatment. The group treated with etanercept achieved a score improvement of 75% after 12 treatment in 50% of cases, and the score reached this value in 66.66% of patients at 24 weeks. Side effects were the following: gastrointestinal disorders: two patients; infections: one patient; psychiatric symptoms: one patient; Figure 2. Evolution of PASI score in the group of patients treated with etanercept autoimmune manifestations: one patient; local reactions at the injection administration of etanercept which was reversible: two patients; transient leukopenia and thrombocytopenia: one patient. In the group treated with etanercept, the mean PASI score was 50.8 ± After 12 treatment, the PASI average score decreased significantly (24 ± 13.8, p = 0.005, t-test for independent values). In week 24 of treatment, the PASI score decreased compared with the PASI score at 12 weeks, but without reaching statistical significance (16 ± 5.3, p = 0.21,t-test). There were no statistically significant differences detected in terms of therapeutic response between the two groups: in the group treated with infliximab, the average PASI score improved by 73% and in the group treated with etanercept, the average PASI score improved by 67.4 % (p = 0.27, non-parametric test Mann-Whitney). Side effects were present in both groups of patients, but it can be noticed that the group treated with etanercept has a greater incidence of them. It can be mentioned that only one patient presented No. Patient Name Age (years) Sex Initial PASI Score after 12 weeks of after 24 Final variation of (%) 1 N.C 58 M ,1 2 V.N. 69 M ,7 3 S.O. 80 F ,1 4 A.S. 40 M L.N. 50 F ,6 6 A.D. 40 F Avg±SD 56,1 ± 16,1 50,8 ± 12,1 24 ± 13,8 16 ± 5,3 67,4± 13,2 Table II. Characteristics of patients treated with etanercept 120
5 Liana Suciu et al an autoimmune manifestation and there hasn t been evidence of any malignant process. Conclusions This study has shown considerable clinical effects in psoriasis arthropathy by PASI assessment score. However, the biological cannot be considered as a first-line because of the incidence of side effects. It should be monitored and interpreted in the clinical context of each patient. Because the number of studied patients was not very large and because the treatment protocols provided a period of no longer then 24 weeks without a break, the patients were monitored and there were no serious adverse effects revealed which could be connected with the treatment. The initiation of immunological in psoriasis involves assessing the balance of benefit/risk/ cost for each patient, so that currently the number of patients receiving such is quite limited. References 1. Guidelines of care for the management of psoriasis and psoriatic arthritis, The American Academy of Dermatology, Baker BS, Fry Lionel. Immunotargeting: A biological approach to the treatment of psoriasis; Drug Discovery today:therapeutic Strategies, Zaragoza V, Perez A, Sanchez JL, et al. Long-term safety and efficacy of Etanercept in the treatment of psoriasis, ACTAS Dermo-Sifiliograficas, Alecu M, Coman G, Costache I, Agraviloaie A. Terapia imunologică în psoriasis, DermatoVenerol (Bucureşti),53: Kormeili T, Lowe NJ, Yamauchi PS. Psoriasis: immunopathogenesis and evolving immunomodulators and systemic therapies. US experiences. Br.J.Dermatol. 2004; 151: Smith CH, Anstey AV, Barker JN, Burden AD, Chalmers RJ, Chandler D, et al. British Association of Dermatologists guidelines for use of biological interventions in psoriasis 2005, Br. J. Dermatol. 2005; 153: Mehlis SL, Gordon KB. The Immunology of psoriasis and biologic immuno. J. Am. Acad. Dermatol. 2003; 49:S XIV, Vol.14, Number. 2/
THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS
 Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 5 (54) No. 2-2012 THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS Mădălina FRÎNCU 1 Abstract: Biological
Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 5 (54) No. 2-2012 THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS Mădălina FRÎNCU 1 Abstract: Biological
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8
 London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y.
 UvA-DARE (Digital Academic Repository) Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y. Link to publication Citation for published version (APA): Goedkoop, A.
UvA-DARE (Digital Academic Repository) Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y. Link to publication Citation for published version (APA): Goedkoop, A.
A. Kopchev, S.Monov, D. Kyurkchiev, I.Ivanova, T. Georgiev (UMHAT St. Ivan Rilski, Medical University - Sofia, Bulgaria)
 International Journal of Pharmaceutical Science Invention ISSN (Online): 2319 6718, ISSN (Print): 2319 670X Volume 6 Issue 7 July 2017 PP. 08-12 Vascular endothelial growth factor (VEGF), cartilage oligomeric
International Journal of Pharmaceutical Science Invention ISSN (Online): 2319 6718, ISSN (Print): 2319 670X Volume 6 Issue 7 July 2017 PP. 08-12 Vascular endothelial growth factor (VEGF), cartilage oligomeric
1. Background: Infliximab is administered parenterally; therefore, it is not covered under retail pharmacy benefits.
 Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
PACKAGE INSERT. Each vial of the REVELLEX product contains 100 mg of infliximab. After reconstitution, each vial
 PACKAGE INSERT SCHEDULING STATUS S4 PROPRIETARY NAME AND DOSAGE FORM REVELLEX 100 mg COMPOSITION Each vial of the REVELLEX product contains 100 mg of infliximab. After reconstitution, each vial of REVELLEX
PACKAGE INSERT SCHEDULING STATUS S4 PROPRIETARY NAME AND DOSAGE FORM REVELLEX 100 mg COMPOSITION Each vial of the REVELLEX product contains 100 mg of infliximab. After reconstitution, each vial of REVELLEX
Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs)
 Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs) January 2010 This technology summary is based on information available at the time of research
Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs) January 2010 This technology summary is based on information available at the time of research
Announcing HUMIRA. Psoriasis Starter Package
 Announcing HUMIRA (adalimumab) Psoriasis Starter Package HUMIRA is indicated for the treatment of adult patients with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy
Announcing HUMIRA (adalimumab) Psoriasis Starter Package HUMIRA is indicated for the treatment of adult patients with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy
Comparison of Various Biological Agents in the Treatment of Psoriasis
 VOL.11NO.5 NO.9 MAY SEPTEMBER 2006 2006 Comparison of Various Biological Agents in the Treatment of Psoriasis MBBS(HK), FRCP, FHKCP, FHKAM(Med) Yaumatei Dermatology Clinic, Social Hygiene Service. This
VOL.11NO.5 NO.9 MAY SEPTEMBER 2006 2006 Comparison of Various Biological Agents in the Treatment of Psoriasis MBBS(HK), FRCP, FHKCP, FHKAM(Med) Yaumatei Dermatology Clinic, Social Hygiene Service. This
ustekinumab (Stelara )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
LOCALLY AVAILABLE BIOLOGIC AGENTS IN THE TREATMENT OF PSORIATIC ARTHRITIS
 Locally Available Biologic Agents in the Treatment of Psoriatic Arthritis 253 Phil. J. Internal Medicine, 47: 253-259, Nov.-Dec., 2009 LOCALLY AVAILABLE BIOLOGIC AGENTS IN THE TREATMENT OF PSORIATIC ARTHRITIS
Locally Available Biologic Agents in the Treatment of Psoriatic Arthritis 253 Phil. J. Internal Medicine, 47: 253-259, Nov.-Dec., 2009 LOCALLY AVAILABLE BIOLOGIC AGENTS IN THE TREATMENT OF PSORIATIC ARTHRITIS
Rheumatoid arthritis
 Rheumatoid arthritis 1 Definition Rheumatoid arthritis is one of the most common inflammatory disorders affecting the population worldwide. It is a systemic inflammatory disease which affects not only
Rheumatoid arthritis 1 Definition Rheumatoid arthritis is one of the most common inflammatory disorders affecting the population worldwide. It is a systemic inflammatory disease which affects not only
The role of current biologic therapies in psoriasis
 : An Update on and IL-17 Inhibitors Joanna Dong, BA; Gary Goldenberg, MD PRACTICE POINTS The newest biologics for treatment of moderate to severe plaque psoriasis are and IL-17 inhibitors with unprecedented
: An Update on and IL-17 Inhibitors Joanna Dong, BA; Gary Goldenberg, MD PRACTICE POINTS The newest biologics for treatment of moderate to severe plaque psoriasis are and IL-17 inhibitors with unprecedented
ETANERCEPT Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW)
 Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL 18830 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL 18830 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Subject: Remicade (Page 1 of 5)
 Subject: Remicade (Page 1 of 5) Objective: I. To ensure that Health Share/Tuality Health Alliance (THA) has a process by which the appropriate utilization of Remicade (Infliximab) for members whose diagnosis
Subject: Remicade (Page 1 of 5) Objective: I. To ensure that Health Share/Tuality Health Alliance (THA) has a process by which the appropriate utilization of Remicade (Infliximab) for members whose diagnosis
Anti-TNF biologic agents Dr Lluís Puig
 Department of Dermatology Hospital de la Santa Creu i Sant Pau IPC NOVARTIS PSORIASIS PRECEPTORSHIP Anti-TNF biologic agents Dr Lluís Puig Barcelona, July 9th-10th, 2013 Anti-TNF therapy in the pathophysiology
Department of Dermatology Hospital de la Santa Creu i Sant Pau IPC NOVARTIS PSORIASIS PRECEPTORSHIP Anti-TNF biologic agents Dr Lluís Puig Barcelona, July 9th-10th, 2013 Anti-TNF therapy in the pathophysiology
TRANSPARENCY COMMITTEE OPINION. 26 April 2006
 TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
The New and Emerging Agents: Dermatology
 Psoriasis Treatment 2013: What is New and on the Horizon? Neil J Korman, MD, PhD Professor of Dermatology University Hospitals Case Medical Center Clinical Director Murdough Family Center for Psoriasis
Psoriasis Treatment 2013: What is New and on the Horizon? Neil J Korman, MD, PhD Professor of Dermatology University Hospitals Case Medical Center Clinical Director Murdough Family Center for Psoriasis
Antirheumatic drugs. Rheumatic Arthritis (RA)
 Antirheumatic drugs Rheumatic Arthritis (RA) Disease Modifying Antirheumatic drugs (DMARDs) DMARDs are used in the treatment of rheumatic arthritis RA and have been shown to slow the course of the disease,
Antirheumatic drugs Rheumatic Arthritis (RA) Disease Modifying Antirheumatic drugs (DMARDs) DMARDs are used in the treatment of rheumatic arthritis RA and have been shown to slow the course of the disease,
Etanercept for Treatment of Hidradenitis
 Home Search Browse Resources Help What's New About Purpose Etanercept for Treatment of Hidradenitis This study is currently recruiting patients. Sponsors and Collaborators: University of Pennsylvania Amgen
Home Search Browse Resources Help What's New About Purpose Etanercept for Treatment of Hidradenitis This study is currently recruiting patients. Sponsors and Collaborators: University of Pennsylvania Amgen
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 13 May 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
3. Has the patient shown improvement in signs and symptoms of the disease? Y N
 Pharmacy Prior Authorization MERC CARE (MEDICAID) Renflexis (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE (MEDICAID) Renflexis (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
STELARA (ustekinumab)
 STELARA (ustekinumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
STELARA (ustekinumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
DRAFT. Remission rates, calculated using observed case (OC) analyses were as follows: Year 1 Year 2 Year 3 Year 4 All patients 62.
 DRAFT New Efficacy Data Shows Cimzia (certolizumab pegol) Provides Long-Term Remission of Moderate to Severe Crohn s Disease Regardless of Prior Anti-TNF Exposure, According to Data Presented at DDW Oral
DRAFT New Efficacy Data Shows Cimzia (certolizumab pegol) Provides Long-Term Remission of Moderate to Severe Crohn s Disease Regardless of Prior Anti-TNF Exposure, According to Data Presented at DDW Oral
What is Enbrel? Key features
 What is Enbrel? Enbrel (also known by its generic name etanercept) is a biologic medication approved in April 2004 by the U.S. Food and Drug Administration (FDA) for the treatment of moderate to severe
What is Enbrel? Enbrel (also known by its generic name etanercept) is a biologic medication approved in April 2004 by the U.S. Food and Drug Administration (FDA) for the treatment of moderate to severe
Clinical Policy: Ustekinumab (Stelara) Reference Number: ERX.SPMN.167
 Clinical Policy: (Stelara) Reference Number: ERX.SPMN.167 Effective Date: 10/16 Last Review Date: 12/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Stelara) Reference Number: ERX.SPMN.167 Effective Date: 10/16 Last Review Date: 12/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Proposed Health Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Secukinumab for treating ankylosing spondylitis after inadequate response to non-steroidal anti-inflammatory drugs
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Secukinumab for treating ankylosing spondylitis after inadequate response to non-steroidal anti-inflammatory drugs
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Psoriasis, Incidence, Quality of Life, Psoriatic Arthritis, Prevalence
 1.0 Abstract Title Prevalence and Incidence of Articular Symptoms and Signs Related to Psoriatic Arthritis in Patients with Psoriasis Severe or Moderate with Adalimumab Treatment (TOGETHER). Keywords Psoriasis,
1.0 Abstract Title Prevalence and Incidence of Articular Symptoms and Signs Related to Psoriatic Arthritis in Patients with Psoriasis Severe or Moderate with Adalimumab Treatment (TOGETHER). Keywords Psoriasis,
Horizon Scanning Centre November Secukinumab for active and progressive psoriatic arthritis. SUMMARY NIHR HSC ID: 5330
 Horizon Scanning Centre November 2012 Secukinumab for active and progressive psoriatic arthritis. SUMMARY NIHR HSC ID: 5330 Secukinumab is a high-affinity fully human monoclonal antibody that antagonises
Horizon Scanning Centre November 2012 Secukinumab for active and progressive psoriatic arthritis. SUMMARY NIHR HSC ID: 5330 Secukinumab is a high-affinity fully human monoclonal antibody that antagonises
Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65
 Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
MMS Pharmacology Lecture 2. Antirheumatic drugs. Dr Sura Al Zoubi
 MMS Pharmacology Lecture 2 Antirheumatic drugs Dr Sura Al Zoubi Revision Rheumatoid Arthritis Definition (RA): is the most common systemic inflammatory disease characterized by symmetrical inflammation
MMS Pharmacology Lecture 2 Antirheumatic drugs Dr Sura Al Zoubi Revision Rheumatoid Arthritis Definition (RA): is the most common systemic inflammatory disease characterized by symmetrical inflammation
Jurnal Medical Aradean (Arad Medical Journal) Vol. XVIII, issue 1, 2015, pp Vasile Goldis University Press (www.jmedar.
 Vol. XVIII, issue 1, 15, pp. 4-45 15 Vasile Goldis University Press (www.jmedar.ro) DEMOGRAPHIC AND CLINICAL PARAMETERS EVALUATION FOR PATIRNTS WITH PSORIATIC VERSUS RHEUMATOID ARTHRITIS Camelia Ciacli
Vol. XVIII, issue 1, 15, pp. 4-45 15 Vasile Goldis University Press (www.jmedar.ro) DEMOGRAPHIC AND CLINICAL PARAMETERS EVALUATION FOR PATIRNTS WITH PSORIATIC VERSUS RHEUMATOID ARTHRITIS Camelia Ciacli
The implication of an immunologic
 Evidence-Based Review of Biologic Therapy for Psoriasis: Infliximab, Etanercept, Adalimumab, Efalizumab, and Alefacept Jeffrey M. Weinberg, MD; Robin Buchholz, MD; Noah Scheinfeld, MD DERMATOLOGY ABSTRACT
Evidence-Based Review of Biologic Therapy for Psoriasis: Infliximab, Etanercept, Adalimumab, Efalizumab, and Alefacept Jeffrey M. Weinberg, MD; Robin Buchholz, MD; Noah Scheinfeld, MD DERMATOLOGY ABSTRACT
SCIENTIFIC DISCUSSION. London, 27 April 2006 Product name: HUMIRA/TRUDEXA Procedure number: EMEA/H/C/ /II/26
 SCIENTIFIC DISCUSSION London, 27 April 2006 Product name: HUMIRA/TRUDEXA Procedure number: EMEA/H/C/481-482/II/26 3.1. Introduction Adalimumab is a recombinant human immunoglobulin (IgG 1 ) monoclonal
SCIENTIFIC DISCUSSION London, 27 April 2006 Product name: HUMIRA/TRUDEXA Procedure number: EMEA/H/C/481-482/II/26 3.1. Introduction Adalimumab is a recombinant human immunoglobulin (IgG 1 ) monoclonal
Certolizumab pegol (Cimzia) for psoriatic arthritis second line
 Certolizumab pegol (Cimzia) for psoriatic arthritis second line This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Certolizumab pegol (Cimzia) for psoriatic arthritis second line This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Ontario Public Drug Programs. Inflectra (infliximab) Frequently Asked Questions
 Ontario Public Drug Programs Inflectra (infliximab) Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Effective February 25 2016, Inflectra (infliximab) will be added
Ontario Public Drug Programs Inflectra (infliximab) Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Effective February 25 2016, Inflectra (infliximab) will be added
Eli Lilly and Company
 Eli Lilly and Company Ixekizumab Phase 2 Psoriasis Data Investment Community Discussion April 10, 2012 Safe Harbor Provision This presentation contains forward-looking statements that are based on management's
Eli Lilly and Company Ixekizumab Phase 2 Psoriasis Data Investment Community Discussion April 10, 2012 Safe Harbor Provision This presentation contains forward-looking statements that are based on management's
Cimzia shows duration of response up to week 26 in moderate to severe Crohn s patients who failed the intravenous infusion treatment infliximab.
 C3073-0409 CIMZIA (certolizumab pegol) PROVIDES LONG-TERM REMISSION AND RESPONSE RATES IN INFLIXIMAB-REFRACTORY CROHN S PATIENTS Cimzia shows duration of response up to week 26 in moderate to severe Crohn
C3073-0409 CIMZIA (certolizumab pegol) PROVIDES LONG-TERM REMISSION AND RESPONSE RATES IN INFLIXIMAB-REFRACTORY CROHN S PATIENTS Cimzia shows duration of response up to week 26 in moderate to severe Crohn
Pharmacy Management Drug Policy
 SUBJECT: Cimzia (Certolizumab pegol) - for Ankylosing Spondylitis, Crohn s Disease, Psoriatic Arthritis and Rheumatoid Arthritis POLICY NUMBER: PHARMACY-07 EFFECTIVE DATE: 5/2009 LAST REVIEW DATE: 6/13/2018
SUBJECT: Cimzia (Certolizumab pegol) - for Ankylosing Spondylitis, Crohn s Disease, Psoriatic Arthritis and Rheumatoid Arthritis POLICY NUMBER: PHARMACY-07 EFFECTIVE DATE: 5/2009 LAST REVIEW DATE: 6/13/2018
Immunosuppressants. Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia
 Immunosuppressants Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Immunosuppressive Agents Very useful in minimizing the occurrence of exaggerated or inappropriate
Immunosuppressants Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Immunosuppressive Agents Very useful in minimizing the occurrence of exaggerated or inappropriate
Amjevita (adalimumab-atto)
 *- Florida Healthy Kids Amjevita (adalimumab-atto) Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2 #* ^ prefilled
*- Florida Healthy Kids Amjevita (adalimumab-atto) Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2 #* ^ prefilled
Biologic Therapies for Psoriasis. A Systematic Review
 Biologic Therapies for Psoriasis. A Systematic Review WOLF-HENNING BOEHNCKE, JÖRG PRINZ, and ALICE B. GOTTLIEB ABSTRACT. Alefacept, efalizumab, etanercept, and infliximab are currently approved for the
Biologic Therapies for Psoriasis. A Systematic Review WOLF-HENNING BOEHNCKE, JÖRG PRINZ, and ALICE B. GOTTLIEB ABSTRACT. Alefacept, efalizumab, etanercept, and infliximab are currently approved for the
Kevzara (sarilumab) NEW PRODUCT SLIDESHOW
 Kevzara (sarilumab) NEW PRODUCT SLIDESHOW Introduction Brand name: Kevzara Generic name: Sarilumab Pharmacological class: Interleukin-6 antagonist Strength and Formulation: 150mg/1.14mL, 200mg/1.14mL;
Kevzara (sarilumab) NEW PRODUCT SLIDESHOW Introduction Brand name: Kevzara Generic name: Sarilumab Pharmacological class: Interleukin-6 antagonist Strength and Formulation: 150mg/1.14mL, 200mg/1.14mL;
Clinical Policy: Apremilast (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17
 Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
How the Innate Immune System Profiles Pathogens
 How the Innate Immune System Profiles Pathogens Receptors on macrophages, neutrophils, dendritic cells for bacteria and viruses Broad specificity - Two main groups of bacteria: gram positive, gram-negative
How the Innate Immune System Profiles Pathogens Receptors on macrophages, neutrophils, dendritic cells for bacteria and viruses Broad specificity - Two main groups of bacteria: gram positive, gram-negative
SCIENTIFIC DISCUSSION
 European Medicines Agency London, 20 September 2007 Product name: Remicade Procedure number: EMEA/H/C/240/II/95 SCIENTIFIC DISCUSSION 7 Westferry Circus, Canary Wharf, London, E14 4HB, UK Tel. (44-20)
European Medicines Agency London, 20 September 2007 Product name: Remicade Procedure number: EMEA/H/C/240/II/95 SCIENTIFIC DISCUSSION 7 Westferry Circus, Canary Wharf, London, E14 4HB, UK Tel. (44-20)
Appendix 1: Frequently Asked Questions
 Appendix 1: Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Effective February 25 2016, Inflectra (infliximab) will be added to the Ontario Drug Benefit (ODB) Formulary
Appendix 1: Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Effective February 25 2016, Inflectra (infliximab) will be added to the Ontario Drug Benefit (ODB) Formulary
Technology appraisal guidance Published: 4 June 2015 nice.org.uk/guidance/ta340
 Ustekinumab for treating active psoriatic arthritis Technology appraisal guidance Published: 4 June 2015 nice.org.uk/guidance/ta340 NICE 2017. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Ustekinumab for treating active psoriatic arthritis Technology appraisal guidance Published: 4 June 2015 nice.org.uk/guidance/ta340 NICE 2017. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Scottish Medicines Consortium
 Scottish Medicines Consortium ustekinumab, 45mg solution for injection (Stelara ) No. (572/09) Janssen-Cilag Ltd 15 January 2010 The Scottish Medicines Consortium (SMC) has completed its assessment of
Scottish Medicines Consortium ustekinumab, 45mg solution for injection (Stelara ) No. (572/09) Janssen-Cilag Ltd 15 January 2010 The Scottish Medicines Consortium (SMC) has completed its assessment of
2. Does the patient have a diagnosis of ulcerative colitis or Crohn s? Y N
 Pharmacy Prior Authorization AETA BETTER HEALTH LOUISIAA (MEDICAID) Remicade (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Pharmacy Prior Authorization AETA BETTER HEALTH LOUISIAA (MEDICAID) Remicade (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Patient #1. Rheumatoid Arthritis. Rheumatoid Arthritis. 45 y/o female Morning stiffness in her joints >1 hour
 Patient #1 Rheumatoid Arthritis Essentials For The Family Medicine Physician 45 y/o female Morning stiffness in her joints >1 hour Hands, Wrists, Knees, Ankles, Feet Polyarticular, symmetrical swelling
Patient #1 Rheumatoid Arthritis Essentials For The Family Medicine Physician 45 y/o female Morning stiffness in her joints >1 hour Hands, Wrists, Knees, Ankles, Feet Polyarticular, symmetrical swelling
INFLIXIMAB Remicade (infliximab), Inflectra (infliximab-dyyb), Renflexis (infliximab-abda)
 RATIONALE FOR INCLUSION IN PA PROGRAM Background Remicade, Renflexis and Inflectra are tumor necrosis factor (TNFα) blockers. Tumor necrosis factor is an endogenous protein that regulates a number of physiologic
RATIONALE FOR INCLUSION IN PA PROGRAM Background Remicade, Renflexis and Inflectra are tumor necrosis factor (TNFα) blockers. Tumor necrosis factor is an endogenous protein that regulates a number of physiologic
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Types of osteoarthritis
 ARTHRITIS Osteoarthritis is a degenerative joint disease is the most common joint disorder. It is a frequent part of aging and is an important cause of physical disability in persons older than 65 years
ARTHRITIS Osteoarthritis is a degenerative joint disease is the most common joint disorder. It is a frequent part of aging and is an important cause of physical disability in persons older than 65 years
HELPING YOU AND YOUR PATIENTS TALK OPENLY ABOUT MODERATELY TO SEVERELY ACTIVE RA
 SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
Research Article Clinical Outcome of a Novel Anti-CD6 Biologic Itolizumab in Patients of Psoriasis with Comorbid Conditions
 Dermatology Research and Practice Volume 2, Article ID 13326, 4 pages http://dx.doi.org/.1155/2/13326 Research Article Clinical Outcome of a Novel Anti-CD6 Biologic Itolizumab in Patients of Psoriasis
Dermatology Research and Practice Volume 2, Article ID 13326, 4 pages http://dx.doi.org/.1155/2/13326 Research Article Clinical Outcome of a Novel Anti-CD6 Biologic Itolizumab in Patients of Psoriasis
Clinical Policy: Secukinumab (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17
 Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Proposal relating to the funding of TNF inhibitors (Humira and Enbrel) and gabapentin (Neurontin)
 14 July 2015 Proposal relating to the funding of TNF inhibitors (Humira and Enbrel) and gabapentin (Neurontin) PHARMAC is seeking feedback on a proposal relating to the funding of the TNF-inhibitor medicines
14 July 2015 Proposal relating to the funding of TNF inhibitors (Humira and Enbrel) and gabapentin (Neurontin) PHARMAC is seeking feedback on a proposal relating to the funding of the TNF-inhibitor medicines
Clinical Policy: Apremilast (Otezla) Reference Number: CP.PHAR.245 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Revision Log See Important Reminder at the end of this policy for important
Psoriasis management. A/Prof Amanda Oakley Dermatologist, Waikato
 Psoriasis management A/Prof Amanda Oakley Dermatologist, Waikato AbbVie Breakfast Session, 14 June 2014 Disclosure This breakfast session is sponsored by Abbvie Autoimmune skin disorders Psoriasis Eczema
Psoriasis management A/Prof Amanda Oakley Dermatologist, Waikato AbbVie Breakfast Session, 14 June 2014 Disclosure This breakfast session is sponsored by Abbvie Autoimmune skin disorders Psoriasis Eczema
What prescribers need to know
 HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
Clinical Policy: Ustekinumab (Stelara) Reference Number: ERX.SPA.01 Effective Date:
 Clinical Policy: (Stelara) Reference Number: ERX.SPA.01 Effective Date: 04.01.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Stelara) Reference Number: ERX.SPA.01 Effective Date: 04.01.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Otezla (apremilast) Page 1 of 7 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Otezla (apremilast) Prime Therapeutics will review Prior Authorization requests Prior
Otezla (apremilast) Page 1 of 7 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Otezla (apremilast) Prime Therapeutics will review Prior Authorization requests Prior
Health Technology Appraisal: etanercept, infliximab and adalimumab for the treatment of psoriatic arthritis (rev 104, 125)
 Health Technology Appraisal: etanercept, infliximab and adalimumab for the treatment of psoriatic arthritis (rev 104, 125) Personal view of etanercept, infliximab and adalimumab for psoriatic arthritis
Health Technology Appraisal: etanercept, infliximab and adalimumab for the treatment of psoriatic arthritis (rev 104, 125) Personal view of etanercept, infliximab and adalimumab for psoriatic arthritis
Cimzia (certolizumab pegol) Data Showed Broad and Rapid Relief From Burden of Symptoms In Rheumatoid Arthritis Patients
 Cimzia (certolizumab pegol) Data Showed Broad and Rapid Relief From Burden of Symptoms In Rheumatoid Arthritis Patients Rapid, sustained and clinically meaningful improvement in wideranging patient-reported
Cimzia (certolizumab pegol) Data Showed Broad and Rapid Relief From Burden of Symptoms In Rheumatoid Arthritis Patients Rapid, sustained and clinically meaningful improvement in wideranging patient-reported
Elements of Successful PBS Applications. Barbara Radulski RN. Copyright
 Elements of Successful PBS Applications Barbara Radulski RN PBS Requirements April 1 2006 THE RULES PBS Requirements 18 years and over Psoriasis x 6 months Failed to achieve an adequate response to 3 systemic
Elements of Successful PBS Applications Barbara Radulski RN PBS Requirements April 1 2006 THE RULES PBS Requirements 18 years and over Psoriasis x 6 months Failed to achieve an adequate response to 3 systemic
Pediatric Use: Safety and effectiveness of Ustekinumab (STELARA ) in pediatric patients have not been evaluated.
 Original Issue Date (Created): January 1, 2010 Most Recent Review Date (Revised): January 28, 2014 Effective Date: April 1, 2014 I. POLICY Preauthorization Requirements for Ustekinumab (STELARA ) Note:
Original Issue Date (Created): January 1, 2010 Most Recent Review Date (Revised): January 28, 2014 Effective Date: April 1, 2014 I. POLICY Preauthorization Requirements for Ustekinumab (STELARA ) Note:
3. Does the patient have a diagnosis of rheumatoid arthritis (RA) with moderate to high disease activity?
 Pharmacy Prior Authorization MERC CARE PLA (MEDICAID) Enbrel (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE PLA (MEDICAID) Enbrel (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
NEW ZEALAND DATA SHEET
 1 PRODUCT NAME COSENTYX 150 mg Powder for Injection COSENTYX 150 mg/ml Solution for Injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredient: Secukinumab Chemical name: Recombinant human
1 PRODUCT NAME COSENTYX 150 mg Powder for Injection COSENTYX 150 mg/ml Solution for Injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredient: Secukinumab Chemical name: Recombinant human
Xeljanz (tofacitinib), Xeljanz XR (tofacitinib extended-release)
 Market DC Xeljanz (tofacitinib), Xeljanz XR (tofacitinib extended-release) Override(s) Prior Authorization Quantity Limit Medications Xeljanz (tofacitinib) Approval Duration 1 year Quantity Limit May be
Market DC Xeljanz (tofacitinib), Xeljanz XR (tofacitinib extended-release) Override(s) Prior Authorization Quantity Limit Medications Xeljanz (tofacitinib) Approval Duration 1 year Quantity Limit May be
Local Natalizumab Treatment Protocol
 Local Natalizumab Treatment Protocol 1. New medicine name: Natalizumab 300mg concentrate for solution for infusion (Natalizumab ) 2. Licensed indication(s): Natalizumab is indicated for single disease
Local Natalizumab Treatment Protocol 1. New medicine name: Natalizumab 300mg concentrate for solution for infusion (Natalizumab ) 2. Licensed indication(s): Natalizumab is indicated for single disease
Incorporating Biologics Into Your Practice
 Incorporating Biologics Into Your Practice Jeffrey M. Sobell MD Tufts University School of Medicine SkinCare Physicians Ora Clinical Research Disclosure Of Relationships With Industry Amgen AbbVie Celgene
Incorporating Biologics Into Your Practice Jeffrey M. Sobell MD Tufts University School of Medicine SkinCare Physicians Ora Clinical Research Disclosure Of Relationships With Industry Amgen AbbVie Celgene
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET SIMPONI Solution for Injection in a pre-filled syringe Solution for Injection in a pre-filled pen, SmartJect NAME OF MEDICINE SIMPONI Solution for Injection in a pre-filled syringe
NEW ZEALAND DATA SHEET SIMPONI Solution for Injection in a pre-filled syringe Solution for Injection in a pre-filled pen, SmartJect NAME OF MEDICINE SIMPONI Solution for Injection in a pre-filled syringe
MAF Shalaby Prof. Rheumatology Al Azhar University, Cairo, Egypt.
 MAF Shalaby Prof. Rheumatology Al Azhar University, Cairo, Egypt. AUTOIMMUNE DISEASE RA SLE VASCULITIS RELAPSING POLYCHONDRITIS SS DM/PM SJOGREN S SYNDROME RHEUMATOID ARTHRITIS Classically immune mediated
MAF Shalaby Prof. Rheumatology Al Azhar University, Cairo, Egypt. AUTOIMMUNE DISEASE RA SLE VASCULITIS RELAPSING POLYCHONDRITIS SS DM/PM SJOGREN S SYNDROME RHEUMATOID ARTHRITIS Classically immune mediated
Prior Authorization Conditions for Approval of Enbrel (etanercept) Website Form Submit request via: Fax
 Prior Authorization Conditions for Approval of Enbrel (etanercept) Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Enbrel (etanercept) require a prior
Prior Authorization Conditions for Approval of Enbrel (etanercept) Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Enbrel (etanercept) require a prior
Cimzia (certolizumab pegol)
 DRUG POLICY BENEFIT APPLICATION Cimzia (certolizumab pegol) Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations
DRUG POLICY BENEFIT APPLICATION Cimzia (certolizumab pegol) Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations
PHARMACY POLICY STATEMENT Ohio Medicaid
 DRUG NAME BILLING CODE BENEFIT TYPE SITE OF SERVICE ALLOWED COVERAGE REQUIREMENTS LIST OF DIAGNOSES CONSIDERED NOT MEDICALLY NECESSARY PHARMACY POLICY STATEMENT Ohio Medicaid Enbrel (etanercept) Must use
DRUG NAME BILLING CODE BENEFIT TYPE SITE OF SERVICE ALLOWED COVERAGE REQUIREMENTS LIST OF DIAGNOSES CONSIDERED NOT MEDICALLY NECESSARY PHARMACY POLICY STATEMENT Ohio Medicaid Enbrel (etanercept) Must use
Psoriasis. Jessica Kaffenberger, M.D. Assistant Professor of Dermatology Division of Dermatology The Ohio State University Wexner Medical Center
 Psoriasis Jessica Kaffenberger, M.D. Assistant Professor of Dermatology Division of Dermatology The Ohio State University Wexner Medical Center Learning objectives Recognize the different types of psoriasis
Psoriasis Jessica Kaffenberger, M.D. Assistant Professor of Dermatology Division of Dermatology The Ohio State University Wexner Medical Center Learning objectives Recognize the different types of psoriasis
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
Regulatory Status FDA-approved indication: Orencia is a selective T cell co-stimulation modulator indicated for: (1)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Orencia Page: 1 of 9 Last Review Date: September 20, 2018 Orencia Description Orencia (abatacept)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Orencia Page: 1 of 9 Last Review Date: September 20, 2018 Orencia Description Orencia (abatacept)
Criteria Inclusion criteria Exclusion criteria. despite treatment with csdmards, NSAIDs, and/or previous anti-tnf therapy and/or
 Supplementary Material Table S1 Eligibility criteria (PICOS) for the SLR Criteria Inclusion criteria Exclusion criteria Population Adults (aged 18 years) with active PsA despite treatment with csdmards,
Supplementary Material Table S1 Eligibility criteria (PICOS) for the SLR Criteria Inclusion criteria Exclusion criteria Population Adults (aged 18 years) with active PsA despite treatment with csdmards,
CIMZIA (certolizumab pegol)
 Pre - PA Allowance None Prior-Approval Requirements Age Diagnoses 18 years of age or older Patient must have ONE of the following: 1. Moderate to severe Crohn s Disease (CD) a. Inadequate response, intolerance
Pre - PA Allowance None Prior-Approval Requirements Age Diagnoses 18 years of age or older Patient must have ONE of the following: 1. Moderate to severe Crohn s Disease (CD) a. Inadequate response, intolerance
Monitoring Patients on Biologic Therapies Murlidhar Rajagopalan, MBBS, MD
 Monitoring Patients on Biologic Therapies Murlidhar Rajagopalan, MBBS, MD Biologics available for psoriasis 1) tumor necrosis factor alpha (TNF-α) inhibitors, 2) interleukin (IL)-12/23 inhibitors, and
Monitoring Patients on Biologic Therapies Murlidhar Rajagopalan, MBBS, MD Biologics available for psoriasis 1) tumor necrosis factor alpha (TNF-α) inhibitors, 2) interleukin (IL)-12/23 inhibitors, and
Primary Results Citation 2
 Table S1. Adalimumab clinical trials 1 ClinicalTrials.gov Rheumatoid Arthritis 3 NCT00195663 Breedveld FC, Weisman MH, Kavanaugh AF, et al. The PREMIER study. A multicenter, randomized, double-blind clinical
Table S1. Adalimumab clinical trials 1 ClinicalTrials.gov Rheumatoid Arthritis 3 NCT00195663 Breedveld FC, Weisman MH, Kavanaugh AF, et al. The PREMIER study. A multicenter, randomized, double-blind clinical
What is your diagnosis? a. Lymphocytic colitis. b. Collagenous colitis. c. Common variable immunodeficiency (CVID) associated colitis
 Case History A 24 year old male presented with fatigue, fever, watery diarrhea, and a cough with sputum production for the past three weeks. His past medical history was significant for recurrent bouts
Case History A 24 year old male presented with fatigue, fever, watery diarrhea, and a cough with sputum production for the past three weeks. His past medical history was significant for recurrent bouts
UvA-DARE (Digital Academic Repository) Innovative therapies and new targets in psoriasis de Groot, M. Link to publication
 UvA-DARE (Digital Academic Repository) Innovative therapies and new targets in psoriasis de Groot, M. Link to publication Citation for published version (APA): de Groot, M. (2011). Innovative therapies
UvA-DARE (Digital Academic Repository) Innovative therapies and new targets in psoriasis de Groot, M. Link to publication Citation for published version (APA): de Groot, M. (2011). Innovative therapies
Remicade. Remicade (infliximab), Inflectra (infliximab-dyyb) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Remicade Page: 1 of 9 Last Review Date: June 22, 2017 Remicade Description Remicade (infliximab),
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Remicade Page: 1 of 9 Last Review Date: June 22, 2017 Remicade Description Remicade (infliximab),
Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS.
 Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Clinical Policy: Secukinumab (Cosentyx) Reference Number: ERX.SPA.165 Effective Date:
 Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
EXAMINING THE CRUCIAL COALITION BETWEEN DERMATOLOGY AND RHEUMATOLOGY IN PSORIATIC ARTHRITIS
 EXAMINING THE CRUCIAL COALITION BETWEEN DERMATOLOGY AND RHEUMATOLOGY IN PSORIATIC ARTHRITIS ACTIVITY 1: EARLY COLLABORATION IN THE TREATMENT OF PSA Key Slides COMMON COMORBIDITIES OF PSORIATIC DISEASE
EXAMINING THE CRUCIAL COALITION BETWEEN DERMATOLOGY AND RHEUMATOLOGY IN PSORIATIC ARTHRITIS ACTIVITY 1: EARLY COLLABORATION IN THE TREATMENT OF PSA Key Slides COMMON COMORBIDITIES OF PSORIATIC DISEASE
IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients
 IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients with 4 weeks of treatment in a Phase 2a trial A. B. Kimball 1, J. Krueger 2, T. Sullivan
IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients with 4 weeks of treatment in a Phase 2a trial A. B. Kimball 1, J. Krueger 2, T. Sullivan
MOLECULAR IMMUNOLOGY Manipulation of immune response Autoimmune diseases & the pathogenic mechanism
 MOLECULAR IMMUNOLOGY Manipulation of immune response Autoimmune diseases & the pathogenic mechanism SCHMAIEL SHIRDEL CONTENT 2 Introduction Autoimmune diseases Classification Involved components Autoimmune
MOLECULAR IMMUNOLOGY Manipulation of immune response Autoimmune diseases & the pathogenic mechanism SCHMAIEL SHIRDEL CONTENT 2 Introduction Autoimmune diseases Classification Involved components Autoimmune
An otherwise healthy 12-year-old
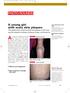 1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
ADALIMUMAB Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW)
 Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Spondyloarthropathies: Disease Perception Limits Market
 Spondyloarthropathies: Disease Perception Limits Market Psoriatic arthritis and ankylosing spondylitis form part of the group of diseases known as the spondyloarthropathies. Psoriatic arthritis is a form
Spondyloarthropathies: Disease Perception Limits Market Psoriatic arthritis and ankylosing spondylitis form part of the group of diseases known as the spondyloarthropathies. Psoriatic arthritis is a form
Details of UNCOVER-2 and UNCOVER-3 Study Results Published in The Lancet
 June 10, 2015 Eli Lilly and Company Lilly Corporate Center Indianapolis, Indiana 46285 U.S.A. +1.317.276.2000 www.lilly.com For Release: Refer to: Immediately Tim Coulom; tim.coulom@lilly.com; 317-771-2241
June 10, 2015 Eli Lilly and Company Lilly Corporate Center Indianapolis, Indiana 46285 U.S.A. +1.317.276.2000 www.lilly.com For Release: Refer to: Immediately Tim Coulom; tim.coulom@lilly.com; 317-771-2241
Prothena Corporation plc
 Prothena Corporation plc PRX003 Investor Update: Phase 2 Development Strategy September 29, 2016 Agenda Dr. Gene Kinney, Chief Operating Officer Introduction Dr. Ken Flanagan, Senior Scientist Th17 and
Prothena Corporation plc PRX003 Investor Update: Phase 2 Development Strategy September 29, 2016 Agenda Dr. Gene Kinney, Chief Operating Officer Introduction Dr. Ken Flanagan, Senior Scientist Th17 and
Rituximab (weekly) for Primary Cutaneous B cell Lymphoma
 Rituximab (weekly) for Primary Cutaneous B cell Lymphoma Indication: Palliative therapy for Low grade Primary Cutaneous B cell Lymphoma (Primary cutaneous Follicle centre cell Lymphoma and Primary cutaneous
Rituximab (weekly) for Primary Cutaneous B cell Lymphoma Indication: Palliative therapy for Low grade Primary Cutaneous B cell Lymphoma (Primary cutaneous Follicle centre cell Lymphoma and Primary cutaneous
