Anti-Migraine Agents
|
|
|
- Lesley Boone
- 6 years ago
- Views:
Transcription
1 DRUG POLICY BENEFIT APPLICATION Anti-Migraine Agents Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations or exceptions may apply. Benefits may vary based on contract, and individual member benefits must be verified. Wellmark determines medical necessity only if the benefit exists and no contract exclusions are applicable. This medical policy may not apply to FEP. Benefits are determined by the Federal Employee Program. DESCRIPTION The intent of the Anti-Migraine agents criteria is to ensure appropriate therapy selection according to the Food and Drug Administration (FDA)-approved product labeling and/or clinical guidelines and/or clinical trials. The criteria will encourage the use of preferred generic anti-migraine agents first and ensure quantity limits are not exceeded based on FDA-approved maximum daily dosing and product packaging. The anti-migraine agents in this policy are all part of the 5-HT1 Serotonin Receptor Agonist class of drugs, also known as the Triptans class. Triptans are believed to effect migraine relief by binding to serotonin (5- hydroxy-tryptamine) receptors in the brain, where they act to induce vasoconstriction of extracerebral blood vessels and also reduce neurogenic inflammation. POLICY PRIOR AUTHORIZATION I. Frova (frovatriptan) and Relpax (eletriptan) may be considered medically necessary when the following criteria is met: The patient has had an inadequate treatment response after at least a 30 day trial of or is intolerant to at least THREE of the following preferred generic triptan medications unless the patient is currently receiving a positive therapeutic outcome on the requested medications through health insurance (excludes obtainment as samples or via manufacturer s patient assistance programs): o naratriptan (Amerge) o (Imitrex) o rizatriptan (Maxalt) o zomitriptan (Zomig) o (Imitrex) nasal spray o (Imitrex) subcutaneous injection Approval will be for lifetime. If additional quantities are requested then post-limit prior authorization criteria must be met. II. Zomig (zolmitriptan) nasal spray may be considered medically necessary when the following criteria is met: Patient has a diagnosis of migraine (with or without aura) Wellmark Blue Cross and Blue Shield is an independent licensee of the Blue Cross and Blue Shield Association. 1
2 intolerant to at least ONE of the following preferred oral generic triptan medications unless the patient is currently receiving a positive therapeutic outcome on the requested medication through health insurance (excludes obtainment as samples or via manufacturer s patient assistance programs): naratriptan (Amerge) (Imitrex) rizatriptan (Maxalt) intolerant to Zolmitriptan (Zomig) oral intolerant to (Imitrex) nasal spray unless the patient is currently receiving a positive therapeutic outcome on the requested medication through health insurance (excludes obtainment as samples or via manufacturer s patient assistance programs): OR The patient has a diagnosis of cluster headache intolerant to BOTH of the following unless the patient is currently receiving a positive therapeutic outcome on the requested medications through health insurance (excludes obtainment as samples or via manufacturer s patient assistance programs): (Imitrex) nasal spray (Imitrex) subcutaneous injection Approval will be for lifetime. If additional quantities are requested then post-limit prior authorization criteria must be met. III. Alsuma and Sumavel Dosepro may be considered medically necessary when all of the following criteria are met: Patient has a diagnosis of migraine (with or without aura) intolerant to at least TWO of the following generic oral triptan medications unless the patient is currently receiving a positive therapeutic outcome on the requested medications through health insurance (excludes obtainment as samples or via manufacturer s patient assistance programs): naratriptan (Amerge) (Imitrex) rizatriptan (Maxalt) zomitriptan (Zomig) intolerant to BOTH of the following: (Imitrex) nasal spray (Imitrex) subcutaneous injection OR The patient has a diagnosis of cluster headache intolerant to BOTH of the following unless the patient is currently receiving a positive therapeutic outcome on the requested medications through health insurance (excludes obtainment as samples or via manufacturer s patient assistance programs):: (Imitrex) nasal spray (Imitrex) subcutaneous injection Wellmark Blue Cross and Blue Shield is an independent licensee of the Blue Cross and Blue Shield Association. 2
3 Approval will be for lifetime. If additional quantities are requested then post-limit prior authorization criteria must be met. IV. Treximet ( + naproxen) may be considered medically necessary when ALL of the following criteria are met: Patient is 12 years of age or older Patient has a diagnosis of migraine headaches (with or without aura) Patient must try and fail a therapeutic trial of and naproxen as separate products taken at the same time. Treatment failure cannot be caused by a lack of compliance to therapy or the unwillingness to take and naproxen separately. Approval will be for lifetime. V. Prior authorization is not required for Imitrex, (excluding Alsuma and Sumavel), Amerge, naratriptan, Axert, almotriptan, Maxalt, Maxalt MLT, rizatriptan, Zomig, Zomig ZMT or zolmitriptan for quantities less than or equal to the Standard Benefit Allowance (see Quantity Limit Chart below). POST-LIMIT PRIOR AUTHORIZATION I. Additional quantities of Imitrex, (excluding Treximet), Alsuma, Amerge, naratriptan, Axert, almotriptan, Frova, frovatriptan, Maxalt, Maxalt MLT, rizatriptan, Relpax, eletriptan, Sumavel, Zomig, Zomig ZMT or zolmitriptan will be considered medically necessary if the following criteria is met: The patient does not have confirmed or suspected cardiovascular or cerebrovascular disease, or uncontrolled hypertension The patient is not treating more than eight headaches per month with a 5-HT1 Serotonin Receptor Agonist AND The patient has a diagnosis of migraine headache o The patient is currently using migraine prophylactic therapy or unable to take migraine prophylactic therapies due to inadequate response, intolerance, or contraindication [Note: examples of prophylactic therapy are divalproex sodium, topiramate, valproate sodium, metoprolol, propranolol, timolol, atenolol, nadolol, amitriptyline, venlafaxine.] o Medication is being prescribed by a neurologist or in consultation with a headache specialist o Medication overuse headache has been considered and ruled out OR The patient has a diagnosis of cluster headache o Medication is being prescribed by a neurologist or in consultation with a headache specialist o The request is for Alsuma, Imitrex () Injection, Imitrex () nasal spray, Sumavel DosePro, or Zomig Nasal Spray Approval will be for lifetime. Medications on this policy are considered not medically necessary for patients who do not meet the criteria set forth above. Quantity limits apply. Wellmark Blue Cross and Blue Shield is an independent licensee of the Blue Cross and Blue Shield Association. 3
4 Brand Name Generic Name QUANTITY LIMIT CHART Dose Max Dose Standard Quantity Limit Post-Limit PA Quantity Limit (up to) 12 injections/30 Alsuma 6mg/0.5ml 12mg/24hr Amerge & Tablet: 1mg, naratriptan 5mg/24hr. 12 tablets/30 18 tablets/30 naratriptan 2.5mg Axert & Tablet: 6.25mg, almotriptan 25mg/24hr. 12 tablets/30 18 tablets/30 almotriptan 12.5mg Frova frovatriptan Tablet: 2.5mg 7.5mg/24hr. 18 tablets/30 27 tablets/30 Imitrex & Imitrex & injectable Imitrex & nasal spray Maxalt & rizatriptan Maxalt MLT & rizatriptan ODT Relpax Sumavel Dosepro Treximet rizatriptan eletriptan needle-free device + naproxen Tablet: 25mg, 50mg, 100mg Injection: 4mg/0.5ml, 6mg/0.5ml Nasal spray: 5mg 200mg/24hr. 12 tablets/30 12mg/24hr. 12 injections/30 18 tablets/30 27 injections/30 40mg/24hr. 24 bottles/30 36 bottles/30 20mg 12 bottles/30 18 bottles/30 Tablet: 5mg,10mg Oral disintegrating 30mg/24hr. 18 tablets/30 27 tablets/30 tablet: 5mg,10mg Tablet: 20mg, 40mg Injection: 4mg/0.5ml 6mg/0.5ml 80mg/24hr. 12 tablets/30 12mg/24hr. 12 injections/30 18 tablets/30 27 injections/30 85mg/500mg 2 tab/24 hr. 9 tablets/30 N/A Zomig & zolmitriptan Zomig ZMT & zolmitriptan ODT zolmitriptan Tablet: 2.5mg,5mg Oral disintegrating tablet: 2.5mg,5mg 10mg/24hr. 12 tablets/30 18 tablets/30 Zomig nasal spray zolmitriptan Nasal spray: 2.5, 5mg/spray 10mg/24hr 12 bottles/30 18 bottles/30 CLINICAL RATIONALE The intent of the criteria is to encourage the use of preferred generic anti-migraine agents first and provide coverage consistent with product labeling, FDA guidance, standards of medical practice, evidence-based drug information, and/or published guidelines. Alsuma, Amerge, Axert, Frova, Imitrex, Maxalt/Maxalt-MLT, Relpax, Sumavel DosePro, Treximet, and Zomig/Zomig-ZMT are indicated for the acute treatment of migraine attacks with or without aura. Axert and Treximet are indicated for the acute treatment of migraine headache pain with or without aura in adolescents ages 12 to 17 years. Alsuma, Imitrex Injection, and Wellmark Blue Cross and Blue Shield is an independent licensee of the Blue Cross and Blue Shield Association. 4
5 Sumavel DosePro are indicated for the acute treatment of cluster headache episodes. Additionally, Imitrex Nasal Spray and Zomig Nasal Spray have a level A recommendation for the treatment of cluster headaches per American Headache Society treatment guidelines. Triptan therapy is contraindicated in patients with confirmed or suspected cardiovascular disease (e.g., ischemic or vasospastic coronary artery disease) or cerebrovascular disease (e.g., stroke or transient ischemic attacks), and in patients with uncontrolled hypertension. Frequent use of acute migraine drugs (e.g. ergotamine, triptans, opioids, or combination of these drugs for 10 or more per month) may lead to exacerbation of headache (medication overuse headache). To decrease the risk of medication-overuse headache ( rebound headache or drug-induced headache ) many experts limit acute therapy to two headache per week on a regular basis. Therefore, the prescriber must have considered and ruled out the diagnosis of medication overuse headache; and, the limits are set at a quantity sufficient to treat 8 headaches per month and may allow for up to 9 headache per month up to maximum recommended daily dosing, contingent on package dispensing requirements. Since manufacturer package sizes may vary, it is the discretion of the dispensing pharmacy to fill quantities per package size up to these quantity limits. In such cases the filling limit and day supply may be less than what is indicated in the Post-limit PA Quantity Limit column in the Quantity Limit chart. For prevention of migraine headache, the American Academy of Neurology and the American Headache Society 2012 guideline update recommendations state that the following medications are established as effective and should be offered for migraine prevention: β-adrenergic blocking agents, metoprolol, propranolol, timolol; and antiepileptic drugs (AEDs), divalproex sodium, topiramate, sodium valproate. Additionally the following medications are probably effective: antidepressants, amitriptyline, venlafaxine; and β-adrenergic blocking agents, atenolol, nadolol and should be considered for migraine prevention. Efficacy and safety of individual agents, even within the same class of drugs, may vary among patients therefore, if the patient fails one preventive medication, others should be tried as failure of one agent does not preclude success with another one. Therefore, patients with migraine headache must be currently taking or had inadequate response, intolerance, or contraindication to prophylactic therapies. Cluster headache is a most painful form of primary headache lasting 15 to 180 minutes, occurring from once every other day to eight times per day, and associated with one or more of various ipsilateral symptoms (conjunctival injection, lacrimation, nasal congestion, rhinorrhea, forehead and facial sweating, miosis, ptosis or eyelid edema). Preventative treatment of cluster headache aims to decrease the incidence of attacks while acute therapy is directed at alleviating the symptoms of an individual attack when it occurs. Given the severity, fast onset and short time to peak intensity of this type of headache, treatment should be rapid and effective. The treatments of choice for acute cluster headache attacks are oxygen, and intranasal or subcutaneous or intranasal zolmitriptan, or a combination of both. Alsuma, Imitrex injection, Imitrex Nasal Spray, Sumavel DosePro and Zomig Nasal Spray will also be considered for patients with cluster headache who require quantities in excess of the initial limit. PROCEDURES AND BILLING CODES To report provider services, use appropriate CPT* codes, Alpha Numeric (HCPCS level 2) codes, Revenue codes, and/or ICD diagnostic codes. Code(s), if applicable. REFERENCES Wellmark Blue Cross and Blue Shield is an independent licensee of the Blue Cross and Blue Shield Association. 5
6 Alsuma [package insert]. Columbia, MD: Meridian Medical Technologies, Inc.; April Amerge [package insert]. Research Triangle Park, NC: GlaxoSmithKline; October Axert [package insert]. Titusville, NJ: Ortho-McNeil Pharmaceutical, Inc.; August Frova [package insert]. Chadds Ford, PA: Endo Pharmaceuticals Inc.; October Imitrex Injection [package insert]. Research Triangle Park, NC: GlaxoSmithKline; October Imitrex Nasal Spray [package insert]. Research Triangle Park, NC: GlaxoSmithKline; November Imitrex Tablets [package insert]. Research Triangle Park, NC: GlaxoSmithKline; November Maxalt and Maxalt-MLT [package insert]. Whitehouse Station, NJ: Merck & Co., Inc; January Relpax [package insert]. New York, NY: Roerig, Division of Pfizer, Inc.; March Sumavel DosePro [package insert]. San Diego, CA: Zogenix, Inc.; February Treximet [package insert]. Research Triangle Park, NC: GlaxoSmithKline; May Zomig and Zomig-ZMT [package insert]. Hayward, CA: Impax Pharmaceuticals Inc.; September Zomig Nasal Spray [package insert]. Hayward, CA: Impax Pharmaceuticals Inc.; September AHFS DI (Adult and Pediatric) [database online]. Hudson, OH: Lexi-Comp, Inc.; [available with subscription]. Accessed May Micromedex Solutions [database online]. Greenwood Village, CO: Truven Health Analytics Inc. Updated periodically. [available with subscription]. Accessed May Beithon J, Gallenberg M, Johnson K, et al. Institute for Clinical Systems Improvement. Diagnosis and Treatment of Headache. Updated January Accessed May Silberstein S, Holland S, Freitag F, et al. Evidence-Based Guideline Update: Pharmacologic Treatment for Episodic Migraine Prevention in Adults: Report of the Quality and the American Headache Society Standards Subcommittee of the American Academy of Neurology. Neurology 2012;78; Lewis D, Ashwal S, Hershey A, et al. Practice Parameter: Pharmacological treatment of migraine headache in children and adolescents: Report of the American Academy of Neurology Quality Standards Subcommittee and the Practice Committee of the Child Neurology Society. Neurology 2004;63; Law S, Derry S, and Moore RA. Triptans for acute cluster headache (Review). The Cochrane Collaboration; Cochrane Database of Systematic Reviews 2010; Issue 4. Francis G, Becker W, Pringsheim T. Acute and Preventive Pharmacologic Treatment of Cluster Headache. Neurology August 3, : Van Vliet, J, Bahra, A, Martin V, et al. Intranasal Sumatriptan in Cluster Headache, Randomized Placebo-Controlled Double-Blind Study. Neurology 2003;60: Rapoport A, Mathew N, Silberstein S. et al. Zolmitriptan Nasal Spray in the Acute Treatment of Cluster Headache, A double-blind study. Neurology 2007;69: Elizabeth Cittadini, MD; Arne May, MD; Andreas Straube, Effectiveness of Intranasal Zolmitriptan in Acute Cluster Headache, A Randomized, Placebo-Controlled, Double-blind Crossover Study. ArchNeurol.2006;63. *Some content reprinted from CVSHealth POLICY HISTORY Policy #: Policy Creation: April 2006 Reviewed: July 2017 Revised: July 2017 Current Effective Date: November 30, 2017 Wellmark Blue Cross and Blue Shield is an independent licensee of the Blue Cross and Blue Shield Association. 6
Pharmacy Medical Necessity Guidelines: Migraine Medications
 Pharmacy Medical Necessity Guidelines: Effective: September 18, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
Pharmacy Medical Necessity Guidelines: Effective: September 18, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
Triptan Quantity Limit
 *- Florida Healthy Kids Triptan Quantity Limit Override(s) Quantity Limit Approval Duration 1 year Oral Tablets Axert (almotriptan) tablets Relpax (eletriptan) tablets 6 tablets (6.25 mg) 12 tablets (12.5
*- Florida Healthy Kids Triptan Quantity Limit Override(s) Quantity Limit Approval Duration 1 year Oral Tablets Axert (almotriptan) tablets Relpax (eletriptan) tablets 6 tablets (6.25 mg) 12 tablets (12.5
Page: 1 of 5. Sumatriptan Tablets and Nasal Spray (Imitrex) / sumatriptan and naproxen sodium (Treximet tablets)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 0 Subject: Sumatriptan (Imitrex / Treximet) Page: 1 of 5 Last Review Date: September 12, 2014 Sumatriptan
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 0 Subject: Sumatriptan (Imitrex / Treximet) Page: 1 of 5 Last Review Date: September 12, 2014 Sumatriptan
Clinical Policy: Triptans Reference Number: CP.CPA.217 Effective Date: Last Review Date: Line of Business: Commercial
 Clinical Policy: Reference Number: CP.CPA.217 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Reference Number: CP.CPA.217 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Triptans Reference Number: CP.HNMC.217 Effective Date: Last Review Date: Line of Business: Medicaid Medi-Cal
 Clinical Policy: Reference Number: CP.HNMC.217 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: Reference Number: CP.HNMC.217 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this policy for important
Maxalt. Maxalt / Maxalt-MLT (rizatriptan) Description. Section: Prescription Drugs Effective Date: April 1, 2016
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Maxalt Page: 1 of 5 Last Review Date: March 18, 2016 Maxalt Description Maxalt / Maxalt-MLT (rizatriptan)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Maxalt Page: 1 of 5 Last Review Date: March 18, 2016 Maxalt Description Maxalt / Maxalt-MLT (rizatriptan)
Sumatriptan Injection (Imitrex / Alsuma / Sumavel / Zembrace)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.11 Subject: Sumatriptan Injection Page: 1 of 6 Last Review Date: March 16, 2018 Sumatriptan Injection
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.11 Subject: Sumatriptan Injection Page: 1 of 6 Last Review Date: March 16, 2018 Sumatriptan Injection
Sumatriptan Injection (Imitrex / Alsuma / Sumavel / Zembrace)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.11 Subject: Sumatriptan Injection Page: 1 of 6 Last Review Date: March 17, 2017 Sumatriptan Injection
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.11 Subject: Sumatriptan Injection Page: 1 of 6 Last Review Date: March 17, 2017 Sumatriptan Injection
Sumatriptan Tablets, Nasal Spray (Imitrex), Nasal Powder (Onzetra Xsail), sumatriptan and naproxen sodium (Treximet tablets)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 05.70.10 Subject: Sumatriptan Page: 1 of 5 Last Review Date: December 2, 2016 Sumatriptan Description Sumatriptan
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 05.70.10 Subject: Sumatriptan Page: 1 of 5 Last Review Date: December 2, 2016 Sumatriptan Description Sumatriptan
Triptans Quantity Limit Program Summary
 Triptans Quantity Limit Program Summary FDA APPROVED INDICATIONS AND DOSAGE 1-13,14,23,24 Agents Amerge (naratriptan) 1, 2.5 tablets Axert (almotriptan) 6.25, 12.5 tablets migraine attacks with/without
Triptans Quantity Limit Program Summary FDA APPROVED INDICATIONS AND DOSAGE 1-13,14,23,24 Agents Amerge (naratriptan) 1, 2.5 tablets Axert (almotriptan) 6.25, 12.5 tablets migraine attacks with/without
Drug Therapy Guidelines
 Drug Therapy Guidelines Applicable Medical Benefit Effective: 5/1/18 Pharmacy- Formulary 1 x Next Review: 3/19 Pharmacy- Formulary 2 x Date of Origin: 8/29/06 Triptans: almotriptan, Amerge, Axert, Frova,
Drug Therapy Guidelines Applicable Medical Benefit Effective: 5/1/18 Pharmacy- Formulary 1 x Next Review: 3/19 Pharmacy- Formulary 2 x Date of Origin: 8/29/06 Triptans: almotriptan, Amerge, Axert, Frova,
Migranal Nasal Spray. Migranal Nasal Spray (dihydroergotamine) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: June 22, 2017 Migranal Nasal Spray
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: June 22, 2017 Migranal Nasal Spray
UTILIZATION MANAGEMENT CRITERIA
 SUMATRIPTAN (ALSUMA, IMITREX, SUMAVEL DOSEPRO, ZECUITY ) UTILIZATION MANAGEMENT CRITERIA DRUG CLASS: BRAND (generic) NAME: Serotonin 5-HT1 receptor agonists Imitrex (sumatriptan), Alsuma (sumatriptan),
SUMATRIPTAN (ALSUMA, IMITREX, SUMAVEL DOSEPRO, ZECUITY ) UTILIZATION MANAGEMENT CRITERIA DRUG CLASS: BRAND (generic) NAME: Serotonin 5-HT1 receptor agonists Imitrex (sumatriptan), Alsuma (sumatriptan),
Clinical Policy: Triptans Reference Number: CP.CPA.217 Effective Date: Last Review Date: Line of Business: Commercial
 Clinical Policy: Reference Number: CP.CPA.217 Effective Date: 11.16.16 Last Review Date: 11.18 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Reference Number: CP.CPA.217 Effective Date: 11.16.16 Last Review Date: 11.18 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Sumatriptan Reference Number: CP.CPA.260 Effective Date: Last Review Date: Line of Business: Commercial
 Clinical Policy: Reference Number: CP.CPA.260 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Reference Number: CP.CPA.260 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Page: 1 of 6. Aimovig (erenumab-aooe) injection, Ajovy (fremanezumab-vfrm) injection, Emgality (galcanezumab-gnim)
 Page: 1 of 6 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Last Review Date: November 30, 2018 Description Aimovig (erenumab-aooe) injection, Ajovy (fremanezumab-vfrm)
Page: 1 of 6 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Last Review Date: November 30, 2018 Description Aimovig (erenumab-aooe) injection, Ajovy (fremanezumab-vfrm)
Regulatory Status FDA approved indication: Migranal Nasal Spray is indicated for the acute treatment of migraine headaches with or without aura (1).
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: November 30, 2018 Migranal Nasal Spray
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: November 30, 2018 Migranal Nasal Spray
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 CGRP Page 1 of 8 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
CGRP Page 1 of 8 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
Zomig. Zomig / Zomig-ZMT (zolmitriptan) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.22 Subject: Zomig Page: 1 of 5 Last Review Date: March 16, 2018 Zomig Description Zomig / Zomig-ZMT
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.22 Subject: Zomig Page: 1 of 5 Last Review Date: March 16, 2018 Zomig Description Zomig / Zomig-ZMT
Sumatriptan Tablets, Nasal Spray (Imitrex), Nasal Powder (Onzetra Xsail), sumatriptan and naproxen sodium (Treximet tablets)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 05.70.10 Subject: Sumatriptan Page: 1 of 6 Last Review Date: March 16, 2018 Sumatriptan Description Sumatriptan
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 05.70.10 Subject: Sumatriptan Page: 1 of 6 Last Review Date: March 16, 2018 Sumatriptan Description Sumatriptan
Sumatriptan Tablets, Nasal Spray (Imitrex), Nasal Powder (Onzetra Xsail), sumatriptan and naproxen sodium (Treximet tablets)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 0 Subject: Sumatriptan Page: 1 of 6 Last Review Date: November 30, 2018 Sumatriptan Description Sumatriptan
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 0 Subject: Sumatriptan Page: 1 of 6 Last Review Date: November 30, 2018 Sumatriptan Description Sumatriptan
Zomig. Zomig / Zomig-ZMT (zolmitriptan) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.22 Subject: Zomig Page: 1 of 5 Last Review Date: November 30, 2018 Zomig Description Zomig / Zomig-ZMT
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.22 Subject: Zomig Page: 1 of 5 Last Review Date: November 30, 2018 Zomig Description Zomig / Zomig-ZMT
Clinical Policy: Sumatriptan Reference Number: CP.HNMC.260 Effective Date: Last Review Date: Line of Business: Medicaid Medi-Cal
 Clinical Policy: Reference Number: CP.HNMC.260 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: Reference Number: CP.HNMC.260 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this policy for important
Abortive Agents. Available Strengths. Formulary Limits. Tablet: 5mg, 10mg ODT: 5mg, 10 mg 25mg, 50mg, 100mg. 5mg/act, 20mg/act
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Migraine Therapy P&T DATE: 9/12/2017 CLASS: Neurological Disorders REVIEW HISTORY 12/16, 9/15, 2/15, 2/10, 5/07 LOB: MCL
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Migraine Therapy P&T DATE: 9/12/2017 CLASS: Neurological Disorders REVIEW HISTORY 12/16, 9/15, 2/15, 2/10, 5/07 LOB: MCL
SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection
 SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit
SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 CGRP Page 1 of 13 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
CGRP Page 1 of 13 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
ADVANCES IN MIGRAINE MANAGEMENT
 ADVANCES IN MIGRAINE MANAGEMENT Joanna Girard Katzman, M.D.MSPH Assistant Professor, Dept. of Neurology Project ECHO, Chronic Pain Program University of New Mexico Outline Migraine throughout the decades
ADVANCES IN MIGRAINE MANAGEMENT Joanna Girard Katzman, M.D.MSPH Assistant Professor, Dept. of Neurology Project ECHO, Chronic Pain Program University of New Mexico Outline Migraine throughout the decades
ONZETRA XSAIL (sumatriptan) nasal powder
 ONZETRA XSAIL (sumatriptan) nasal powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
ONZETRA XSAIL (sumatriptan) nasal powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
Literature Scan: Triptans
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Disclosures. Triptans for Kids 5/16/13
 5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
Neuromuscular Blocking Agents
 Neuromuscular Blocking Agents DRUG POLICY This Prior Authorization request will be reviewed for medical necessity only. Benefits are subject to the terms and conditions of the patient s contract. Please
Neuromuscular Blocking Agents DRUG POLICY This Prior Authorization request will be reviewed for medical necessity only. Benefits are subject to the terms and conditions of the patient s contract. Please
MIGRAINE UPDATE. Objectives & Disclosures. Learn techniques used to diagnose headaches. Become familiar with medications used for headache treatment.
 MIGRAINE UPDATE Karen L. Bremer, MD November 16, 2018 Objectives & Disclosures Learn techniques used to diagnose headaches. Become familiar with medications used for headache treatment. Disclosure: I am
MIGRAINE UPDATE Karen L. Bremer, MD November 16, 2018 Objectives & Disclosures Learn techniques used to diagnose headaches. Become familiar with medications used for headache treatment. Disclosure: I am
Commercial Formulary and Utilization Management Program Updates for January 2017 Producer Communication #782 Issued November 10, 2016
 Commercial and Utilization Management Program Updates for January 2017 Producer Communication #782 Issued November 10, 2016 Message The Capital BlueCross Pharmacy & Therapeutics (P&T) Committee, consisting
Commercial and Utilization Management Program Updates for January 2017 Producer Communication #782 Issued November 10, 2016 Message The Capital BlueCross Pharmacy & Therapeutics (P&T) Committee, consisting
Immediate Release Opioid Analgesics (Brand and Generic): Acute Pain Duration Limit with MME Limit and Post Limit Policy
 BENEFIT APPLICATION DRUG POLICY Immediate Release Opioid Analgesics (Brand and Generic): Acute Pain Duration Limit with MME Limit and Post Limit Policy Benefit determinations are based on the applicable
BENEFIT APPLICATION DRUG POLICY Immediate Release Opioid Analgesics (Brand and Generic): Acute Pain Duration Limit with MME Limit and Post Limit Policy Benefit determinations are based on the applicable
Acetaminophen/Aspirin/Ibuprofen Containing Immediate Release Opioid Analgesics: Quantity Limit Policy
 DRUG POLICY Acetaminophen/Aspirin/Ibuprofen Containing Immediate Release Opioid Analgesics: Quantity Limit Policy BENEFIT APPLICATION Benefit determinations are based on the applicable contract language
DRUG POLICY Acetaminophen/Aspirin/Ibuprofen Containing Immediate Release Opioid Analgesics: Quantity Limit Policy BENEFIT APPLICATION Benefit determinations are based on the applicable contract language
Case Presentation. Case Presentation. Case Presentation. Truths about Headaches (2017) Most headaches were muscle-tension headaches
 Agenda Case presentation Migraine Morphology Primary and Premonitory Phase Secondary Headache Aura Headache Primer on Pain Medication Overuse Headache Case Presentation RT is a 25 year old woman with daily
Agenda Case presentation Migraine Morphology Primary and Premonitory Phase Secondary Headache Aura Headache Primer on Pain Medication Overuse Headache Case Presentation RT is a 25 year old woman with daily
ABORTIVE AGENTS. Average cost per 30 days. Form Limits SEROTONIN AGONISTS $ $ Reserved for treatment failure to either Sumatriptan PA; QL
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Migraine Therapy P&T DATE: 12/11/2018 CLASS: Neurological Disorders REVIEW HISTORY 9/17, 12/16, 9/15, 2/15, 2/10, LOB: MCL
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Migraine Therapy P&T DATE: 12/11/2018 CLASS: Neurological Disorders REVIEW HISTORY 9/17, 12/16, 9/15, 2/15, 2/10, LOB: MCL
Acute Migraine Treatment in Adults
 PL Detail-Document #301104 This PL Detail-Document gives subscribers additional insight related the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER November 2014 Acute Migraine Treatment
PL Detail-Document #301104 This PL Detail-Document gives subscribers additional insight related the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER November 2014 Acute Migraine Treatment
TREXIMET UTILIZATION MANAGEMENT CRITERIA
 TREXIMET UTILIZATION MANAGEMENT CRITERIA DRUG CLASS: BRAND (generic) NAME: 5HT1 agonists Treximet (sumatriptan/naproxen sodium) 85 mg / 500 mg tablet FDA INDICATIONS: Sumatriptan/naproxen is indicated
TREXIMET UTILIZATION MANAGEMENT CRITERIA DRUG CLASS: BRAND (generic) NAME: 5HT1 agonists Treximet (sumatriptan/naproxen sodium) 85 mg / 500 mg tablet FDA INDICATIONS: Sumatriptan/naproxen is indicated
MEASURE #3: PREVENTIVE MIGRAINE MEDICATION PRESCRIBED Headache
 MEASURE #3: PREVENTIVE MIGRAINE MEDICATION PRESCRIBED Headache Measure Description Percentage of patients age 18 years old and older diagnosed with migraine headache whose migraine frequency is 4 migraine
MEASURE #3: PREVENTIVE MIGRAINE MEDICATION PRESCRIBED Headache Measure Description Percentage of patients age 18 years old and older diagnosed with migraine headache whose migraine frequency is 4 migraine
MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache
 MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache Measure Description Percentage of patients age 12 years and older with a diagnosis of migraine who were prescribed a guideline recommended
MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache Measure Description Percentage of patients age 12 years and older with a diagnosis of migraine who were prescribed a guideline recommended
IMITREX UTILIZATION MANAGEMENT CRITERIA
 IMITREX UTILIZATION MANAGEMENT CRITERIA DRUG CLASS: 5HT1 agonists BRAND NAME: Imitrex 4 mg injection kit or refill (Generic) (sumatriptan) 6 mg injection kit or refill Injection vial (1 dose) 5 mg nasal
IMITREX UTILIZATION MANAGEMENT CRITERIA DRUG CLASS: 5HT1 agonists BRAND NAME: Imitrex 4 mg injection kit or refill (Generic) (sumatriptan) 6 mg injection kit or refill Injection vial (1 dose) 5 mg nasal
Faculty Disclosure. Karen L. Bremer, MD. Dr. Bremer has listed no financial interest/arrangement that would be considered a conflict of interest.
 Faculty Disclosure Karen L. Bremer, MD Dr. Bremer has listed no financial interest/arrangement that would be considered a conflict of interest. HEADACHE UPDATE Karen L. Bremer, MD November 10, 2017 karen.bremer@creighton.edu
Faculty Disclosure Karen L. Bremer, MD Dr. Bremer has listed no financial interest/arrangement that would be considered a conflict of interest. HEADACHE UPDATE Karen L. Bremer, MD November 10, 2017 karen.bremer@creighton.edu
Short Clinical Guidelines: Headache, Key Points for Diagnosis and Treatment
 Clinical Highlights 1. Headache is diagnosed by history and physical examination with limited need for imaging or laboratory tests. 2. Warning signs of possible disorder other than primary headache are:
Clinical Highlights 1. Headache is diagnosed by history and physical examination with limited need for imaging or laboratory tests. 2. Warning signs of possible disorder other than primary headache are:
Management of headache
 Management of headache TJ Steiner Imperial College London Based on European principles of management of common headache disorders TJ Steiner, K Paemeleire, R Jensen, D Valade, L Savi, MJA Lainez, H-C Diener,
Management of headache TJ Steiner Imperial College London Based on European principles of management of common headache disorders TJ Steiner, K Paemeleire, R Jensen, D Valade, L Savi, MJA Lainez, H-C Diener,
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Aimovig) Reference Number: CP.PHAR.128 Effective Date: 07.10.18 Last Review Date: 08.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Aimovig) Reference Number: CP.PHAR.128 Effective Date: 07.10.18 Last Review Date: 08.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Adult & Pediatric Patients. Stanford Health Care, Division Pain Medicine
 Acute Treatment Strategies in Adult & Pediatric Patients Theresa Mallick Searle, MS, RN BC, ANP BC Disclosures Speakers Bureau: Allergan, Depomed Acute Treatment Strategies in Adult & Pediatric Patients
Acute Treatment Strategies in Adult & Pediatric Patients Theresa Mallick Searle, MS, RN BC, ANP BC Disclosures Speakers Bureau: Allergan, Depomed Acute Treatment Strategies in Adult & Pediatric Patients
PHARMACY COVERAGE GUIDELINES ORIGINAL EFFECTIVE DATE: 5/18/17 SECTION: DRUGS LAST REVIEW DATE: 5/17/18 LAST CRITERIA REVISION DATE: ARCHIVE DATE:
 ALLZITAL (butalbital and acetaminophen) 25 mg/325 mg oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
ALLZITAL (butalbital and acetaminophen) 25 mg/325 mg oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
Allzital (butalbital-acetaminophen), butalbital-aspirin-caffeine, butalbitalaspirin-caffeine-codeine,
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Butalbital Analgesics Page: 1 of 6 Last Review Date: March 16, 2018 Butalbital Analgesics Description
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Butalbital Analgesics Page: 1 of 6 Last Review Date: March 16, 2018 Butalbital Analgesics Description
Migraine, Tension, and Cluster Headache: Primary Care for Primary Headaches. Jonathon M. Firnhaber, MD, FAAFP
 Migraine, Tension, and Cluster Headache: Primary Care for Primary Headaches Jonathon M. Firnhaber, MD, FAAFP Learning objectives 1. Utilize evidence-based strategies to diagnose patients presenting with
Migraine, Tension, and Cluster Headache: Primary Care for Primary Headaches Jonathon M. Firnhaber, MD, FAAFP Learning objectives 1. Utilize evidence-based strategies to diagnose patients presenting with
Botox (onabotulinumtoxina) Dysport (abobotulinumtoxina) Xeomin (incobotulinumtoxina) Myobloc (rimabotulinumtoxinb)
 Botox (onabotulinumtoxina) Dysport (abobotulinumtoxina) Xeomin (incobotulinumtoxina) Myobloc (rimabotulinumtoxinb) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage Original Effective Date:
Botox (onabotulinumtoxina) Dysport (abobotulinumtoxina) Xeomin (incobotulinumtoxina) Myobloc (rimabotulinumtoxinb) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage Original Effective Date:
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Ajovy) Reference Number: CP.PHAR.## Effective Date: 10.30.18 Last Review Date: 02.19 Line of Business: Commercial, TBD HIM*, Medicaid Revision Log See Important Reminder at the end of
Clinical Policy: (Ajovy) Reference Number: CP.PHAR.## Effective Date: 10.30.18 Last Review Date: 02.19 Line of Business: Commercial, TBD HIM*, Medicaid Revision Log See Important Reminder at the end of
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: occipital_nerve_stimulation 8/2010 5/2017 5/2018 5/2017 Description of Procedure or Service Occipital nerve
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: occipital_nerve_stimulation 8/2010 5/2017 5/2018 5/2017 Description of Procedure or Service Occipital nerve
Pharmacy Updates Summary
 All of the following changes were reviewed and approved by the SFHP Pharmacy & Therapeutics (P&T) Committee on 4/16/2014 Effective date: 5/15/2014 Therapeutic Classes reviewed: ADHD Ophthalmic antihistamines
All of the following changes were reviewed and approved by the SFHP Pharmacy & Therapeutics (P&T) Committee on 4/16/2014 Effective date: 5/15/2014 Therapeutic Classes reviewed: ADHD Ophthalmic antihistamines
Calcitonin Gene Related Peptide Receptor Inhibitors Prior Authorization Criteria:
 Request for Prior Authorization for Calcitonin Gene Related Peptide Receptor Inhibitors Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Calcitonin Gene
Request for Prior Authorization for Calcitonin Gene Related Peptide Receptor Inhibitors Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Calcitonin Gene
Subject: CGRP Inhibitors
 ARCHIVED (NOT ACTIVE RETIRED) Archived: 08/01/18 09-J2000-98 Original Effective Date: 06/15/18 Reviewed: 05/09/18 Revised: 08/01/18 Next Review: ARCHIVED (NOT ACTIVE RETIRED) Subject: CGRP Inhibitors THIS
ARCHIVED (NOT ACTIVE RETIRED) Archived: 08/01/18 09-J2000-98 Original Effective Date: 06/15/18 Reviewed: 05/09/18 Revised: 08/01/18 Next Review: ARCHIVED (NOT ACTIVE RETIRED) Subject: CGRP Inhibitors THIS
Clinical Learning Days November 10, 2017
 Migraine Clinical Learning Days November 10, 2017 Alyssa Lettich. MD Neurosciences Institute/Neurosciences Clinical Program Medical Director Headache Disclosures: none Learning Objectives: At the conclusion
Migraine Clinical Learning Days November 10, 2017 Alyssa Lettich. MD Neurosciences Institute/Neurosciences Clinical Program Medical Director Headache Disclosures: none Learning Objectives: At the conclusion
Headache A Practical Approach
 Headache A Practical Approach Integrated Pain Symposium December 1, 2017 Alyssa Lettich. MD Neurosciences Institute/Neurosciences Clinical Program Medical Director Headache and Pain Development Teams Disclosures:
Headache A Practical Approach Integrated Pain Symposium December 1, 2017 Alyssa Lettich. MD Neurosciences Institute/Neurosciences Clinical Program Medical Director Headache and Pain Development Teams Disclosures:
OH, MY ACHING HEAD! I HAVE NO DISCLOSURES OR CONFLICTS OF INTERESTS TO DECLARE MANAGING HEADACHE IN THE OUTPATIENT SETTING SECONDARY HEADACHES
 1 JUSTIN A. OSSMAN, MD CHATTANOOGA FAMILY MEDICINE UPDATE OH, MY ACHING HEAD! MANAGING HEADACHE IN THE OUTPATIENT SETTING 2 I HAVE NO DISCLOSURES OR CONFLICTS OF INTERESTS TO DECLARE OBJECTIVES International
1 JUSTIN A. OSSMAN, MD CHATTANOOGA FAMILY MEDICINE UPDATE OH, MY ACHING HEAD! MANAGING HEADACHE IN THE OUTPATIENT SETTING 2 I HAVE NO DISCLOSURES OR CONFLICTS OF INTERESTS TO DECLARE OBJECTIVES International
Formulary Medical Necessity Program
 BENEFIT APPLICATION Formulary Medical Necessity Program DRUG POLICY Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations
BENEFIT APPLICATION Formulary Medical Necessity Program DRUG POLICY Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations
Headache Questionnaire
 Date: All Headache Patients We would appreciate your cooperation in filling out this form. In our evaluation of headache, your history is typically our most valuable tool for diagnosis and subsequent treatment.
Date: All Headache Patients We would appreciate your cooperation in filling out this form. In our evaluation of headache, your history is typically our most valuable tool for diagnosis and subsequent treatment.
BELBUCA (buprenorphine buccal film)
 RATIONALE FOR INCLUSION IN PA PROGRAM Background Belbuca is indicated for the management of chronic pain severe enough to require daily, aroundthe-clock, long-acting opioid treatment for which alternative
RATIONALE FOR INCLUSION IN PA PROGRAM Background Belbuca is indicated for the management of chronic pain severe enough to require daily, aroundthe-clock, long-acting opioid treatment for which alternative
HARVARD PILGRIM HEALTH CARE RECOMMENDED MEDICATION REQUEST GUIDELINES
 Generic Brand HICL GCN Exception/Other ONABOTULINUMTOXINA BOTOX 04867 BRAND BOTOX COSMETIC ABOBOTULINUMTOXINA DYSPORT 36477 RIMABOTULINUMTOXINB MYOBLOC 21869 INCOBOTULINUMTOXINA XEOMIN 36687 Please use
Generic Brand HICL GCN Exception/Other ONABOTULINUMTOXINA BOTOX 04867 BRAND BOTOX COSMETIC ABOBOTULINUMTOXINA DYSPORT 36477 RIMABOTULINUMTOXINB MYOBLOC 21869 INCOBOTULINUMTOXINA XEOMIN 36687 Please use
Pharmacy Policy Bulletin
 Pharmacy Policy Bulletin Title: Policy #: Age Edits Rx.01.2 Application of pharmacy policy is determined by benefits and contracts. Benefits may vary based on product line, group, or contract. Some medications
Pharmacy Policy Bulletin Title: Policy #: Age Edits Rx.01.2 Application of pharmacy policy is determined by benefits and contracts. Benefits may vary based on product line, group, or contract. Some medications
STEP THERAPY CRITERIA
 DRUG CLASS PRODUCTS) BRAND NAME (BRAND ONLY) (generic) STEP THERAPY CRITERIA ATYPICAL ANTIPSYCHOTICS (BRAND ONLY ABILIFY (AL TABLET & AL SOLUTION ONLY) (aripiprazole) FANAPT (BRAND ONLY) (iloperidone)
DRUG CLASS PRODUCTS) BRAND NAME (BRAND ONLY) (generic) STEP THERAPY CRITERIA ATYPICAL ANTIPSYCHOTICS (BRAND ONLY ABILIFY (AL TABLET & AL SOLUTION ONLY) (aripiprazole) FANAPT (BRAND ONLY) (iloperidone)
Outpatient Headache Care Guideline
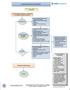 1 Outpatient Care Guideline Inclusion criteria: children > 3 yrs with headaches Is urgent emergency department, neuroimaging, or Neurology consultation indicated? Referral to ED if: New severe headache
1 Outpatient Care Guideline Inclusion criteria: children > 3 yrs with headaches Is urgent emergency department, neuroimaging, or Neurology consultation indicated? Referral to ED if: New severe headache
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Emgality) Reference Number: CP.PHAR.## Effective Date: 11.13.18 Last Review Date: 02.19 Line of Business: Commercial, TBD HIM*, Medicaid Revision Log See Important Reminder at the end
Clinical Policy: (Emgality) Reference Number: CP.PHAR.## Effective Date: 11.13.18 Last Review Date: 02.19 Line of Business: Commercial, TBD HIM*, Medicaid Revision Log See Important Reminder at the end
ARxCH. Annual Review of Changes in Healthcare. Calcitonin Gene-Related Peptide Receptors and the Prevention of Migraines. Abstract
 Calcitonin Gene-Related Peptide Receptors and the Prevention of Migraines Brian Schuler, PharmD Candidate 2018 1 1 University of Findlay College of Pharmacy Abstract Migraines are the third most prevalent
Calcitonin Gene-Related Peptide Receptors and the Prevention of Migraines Brian Schuler, PharmD Candidate 2018 1 1 University of Findlay College of Pharmacy Abstract Migraines are the third most prevalent
Ergotamine/Dihydroergotamine Products
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
90 dosage units per 90 days OR. Extended-release Formulations Ultram ER 90 dosage units per 90 days OR
 Pre - PA Allowance 12 years of age or older Quantity Immediate-release Formulation Ultracet 720 dosage units per 90 days OR Ultram 720 dosage units per 90 days Extended-release Formulations Ultram ER 90
Pre - PA Allowance 12 years of age or older Quantity Immediate-release Formulation Ultracet 720 dosage units per 90 days OR Ultram 720 dosage units per 90 days Extended-release Formulations Ultram ER 90
Xartemis XR (oxycodone / acetaminophen extended release)
 RATIONALE FOR INCLUSION IN PA PROGRAM Background Xartemis XR is a combination of oxycodone and acetaminophen in a dosage formulation to deliver both immediate pain relief, in less than an hour, and extended-release
RATIONALE FOR INCLUSION IN PA PROGRAM Background Xartemis XR is a combination of oxycodone and acetaminophen in a dosage formulation to deliver both immediate pain relief, in less than an hour, and extended-release
Headache. Section 1. Migraine headache. Clinical presentation
 Section 1 Headache Migraine headache 1 Clinical presentation It is important to recognize just how significant a problem migraine headache is. It has been estimated that migraine affects 11% of the United
Section 1 Headache Migraine headache 1 Clinical presentation It is important to recognize just how significant a problem migraine headache is. It has been estimated that migraine affects 11% of the United
HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in
 HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in Anesthesia and Neurology Harvard Medical School Limited time
HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in Anesthesia and Neurology Harvard Medical School Limited time
Drug Class Review Triptans
 Drug Class Review Triptans April 2013 Last Report: Update #4 (June 2009) The purpose of reports is to make available information regarding the comparative clinical effectiveness and harms of different
Drug Class Review Triptans April 2013 Last Report: Update #4 (June 2009) The purpose of reports is to make available information regarding the comparative clinical effectiveness and harms of different
Drug Class Review Triptans
 Drug Class Review Triptans Preliminary Update Scan #3 February 2014 Last Report: Update #4 (June 2009) The purpose of reports is to make available information regarding the comparative clinical effectiveness
Drug Class Review Triptans Preliminary Update Scan #3 February 2014 Last Report: Update #4 (June 2009) The purpose of reports is to make available information regarding the comparative clinical effectiveness
Update on Diagnosis and Management of Migraines
 Update on Diagnosis and Management of Migraines Joel J. Heidelbaugh, MD, FAAFP, FACG Clinical Professor Departments of Family Medicine and Urology University of Michigan Learning Objectives To distinguish
Update on Diagnosis and Management of Migraines Joel J. Heidelbaugh, MD, FAAFP, FACG Clinical Professor Departments of Family Medicine and Urology University of Michigan Learning Objectives To distinguish
10/17/2017 CHRONIC MIGRAINES BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES PATIENT CASE EPIDEMIOLOGY EPIDEMIOLOGY
 BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES JENNIFER SHIN, PHARMD PGY2 AMBULATORY CARE PHARMACY RESIDENT COMMUNITYCARE HEALTH CENTERS PHARMACOTHERAPY ROUNDS OCTOBER 20, 2017
BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES JENNIFER SHIN, PHARMD PGY2 AMBULATORY CARE PHARMACY RESIDENT COMMUNITYCARE HEALTH CENTERS PHARMACOTHERAPY ROUNDS OCTOBER 20, 2017
RATIONALE FOR INCLUSION IN PA PROGRAM
 RATIONALE FOR INCLUSION IN PA PROGRAM Background Tramadol is a centrally acting synthetic opioid analgesic used to treat moderate to moderately severe chronic pain in adults. Along from analgesia, tramadol
RATIONALE FOR INCLUSION IN PA PROGRAM Background Tramadol is a centrally acting synthetic opioid analgesic used to treat moderate to moderately severe chronic pain in adults. Along from analgesia, tramadol
Imitrex for ocular migraines
 Imitrex for ocular migraines Information about ocular, optical, and ophthalmic Migraine, accurate diagnoses, and the importance of correct diagnostic and classification terminology. 14-10-2015 1 Answer
Imitrex for ocular migraines Information about ocular, optical, and ophthalmic Migraine, accurate diagnoses, and the importance of correct diagnostic and classification terminology. 14-10-2015 1 Answer
MORPHINE IR DRUG CLASS Morphine IR, Dilaudid IR (hydromorphone), Opana IR (oxymorphone)
 Pre - PA Allowance Tablets & Suppositories Morphine sulfate tablets Morphine sulfate suppositories Oxymorphone tablets Hydromorphone tablets Hydromorphone suppositories 360 tablets per 90 days OR 360 suppositories
Pre - PA Allowance Tablets & Suppositories Morphine sulfate tablets Morphine sulfate suppositories Oxymorphone tablets Hydromorphone tablets Hydromorphone suppositories 360 tablets per 90 days OR 360 suppositories
Pre - PA Allowance. Prior-Approval Requirements LEVORPHANOL TARTRATE. None
 Pre - PA Allowance None Prior-Approval Requirements Prior authorization is not required if prescribed by an oncologist and/or the member has paid pharmacy claims for an oncology medication(s) in the past
Pre - PA Allowance None Prior-Approval Requirements Prior authorization is not required if prescribed by an oncologist and/or the member has paid pharmacy claims for an oncology medication(s) in the past
Medication For Migraine Chart: Table 1: Acute Treatment when the attack begins
 Medication For Migraine Chart: Table 1: Acute Treatment when the attack begins Page a Analgesics (painkillers) Non-steroidal antiinflammatory drugs (NSAIDs) Prescription required Brand Name Formulation
Medication For Migraine Chart: Table 1: Acute Treatment when the attack begins Page a Analgesics (painkillers) Non-steroidal antiinflammatory drugs (NSAIDs) Prescription required Brand Name Formulation
Duragesic Patch (fentanyl patch) Prior authorization is not required if prescribed by an oncologist
 Pre - PA Allowance Quantity 30 patches every 90 days Prior-Approval Requirements Prior authorization is not required if prescribed by an oncologist Age 2 years of age or older Diagnosis Patient must have
Pre - PA Allowance Quantity 30 patches every 90 days Prior-Approval Requirements Prior authorization is not required if prescribed by an oncologist Age 2 years of age or older Diagnosis Patient must have
HYSINGLA ER (hydrocodone bitartrate) Prior authorization is not required if prescribed by an oncologist.
 Pre - PA Allowance None Prior authorization is not required if prescribed by an oncologist. Prior-Approval Requirements Age 18 years of age or older Diagnosis Patient must have the following: 1. Pain,
Pre - PA Allowance None Prior authorization is not required if prescribed by an oncologist. Prior-Approval Requirements Age 18 years of age or older Diagnosis Patient must have the following: 1. Pain,
Reviews/Evaluations. Medication-Overuse Headache
 Reviews/Evaluations Medication-Overuse Headache Population-based studies suggest that the prevalence of chronic daily headache is 3-5%(1,2). Drug-induced headaches are the most common cause of chronic
Reviews/Evaluations Medication-Overuse Headache Population-based studies suggest that the prevalence of chronic daily headache is 3-5%(1,2). Drug-induced headaches are the most common cause of chronic
OXYCODONE IR (oxycodone)
 RATIONALE FOR INCLUSION IN PA PROGRAM Background Oxycodone hydrochloride, a pure opioid agonist, is used in the treatment of moderate to severe pain (1-2). The precise mechanism of action is unknown; however,
RATIONALE FOR INCLUSION IN PA PROGRAM Background Oxycodone hydrochloride, a pure opioid agonist, is used in the treatment of moderate to severe pain (1-2). The precise mechanism of action is unknown; however,
Botox (onabotulinumtoxina) Dysport (abobotulinumtoxina) Xeomin (incobotulinumtoxina) Myobloc (rimabotulinumtoxinb)
 Botox (onabotulinumtoxina) Dysport (abobotulinumtoxina) Xeomin (incobotulinumtoxina) Myobloc (rimabotulinumtoxinb) Line(s) of Business: HMO; PPO; QUEST Integration Medicare Advantage Original Effective
Botox (onabotulinumtoxina) Dysport (abobotulinumtoxina) Xeomin (incobotulinumtoxina) Myobloc (rimabotulinumtoxinb) Line(s) of Business: HMO; PPO; QUEST Integration Medicare Advantage Original Effective
Nucynta IR. Nucynta IR (tapentadol immediate-release) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Nucynta IR Page: 1 of 9 Last Review Date: December 8, 2017 Nucynta IR Description Nucynta IR (tapentadol
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Nucynta IR Page: 1 of 9 Last Review Date: December 8, 2017 Nucynta IR Description Nucynta IR (tapentadol
How do we treat migraine? New SIGN Guidelines
 How do we treat migraine? New SIGN Guidelines Managing your migraine Migraine Trust, Edinburgh 2018 Callum Duncan Consultant Neurologist Aberdeen Royal Infirmary Chair SIGN Guideline 155 Premonitory Mood
How do we treat migraine? New SIGN Guidelines Managing your migraine Migraine Trust, Edinburgh 2018 Callum Duncan Consultant Neurologist Aberdeen Royal Infirmary Chair SIGN Guideline 155 Premonitory Mood
Overuse of barbiturate and opioid containing medications for primary headache disorders Description
 Measure Title Overuse of barbiturate and opioid containing medications for primary headache disorders Description Percentage of s age 12 years and older with a diagnosis of primary headache who were prescribed
Measure Title Overuse of barbiturate and opioid containing medications for primary headache disorders Description Percentage of s age 12 years and older with a diagnosis of primary headache who were prescribed
SASKATCHEWAN FORMULARY COMMITTEE BULLETIN UPDATE TO THE 57th EDITION OF THE SASKATCHEWAN FORMULARY
 Saskatchewan Health Drug Plan and Extended Benefits Branch April 2008 Bulletin #114 ISSN 0708-3246 SASKATCHEWAN FORMULARY COMMITTEE BULLETIN UPDATE TO THE 57th EDITION OF THE SASKATCHEWAN FORMULARY The
Saskatchewan Health Drug Plan and Extended Benefits Branch April 2008 Bulletin #114 ISSN 0708-3246 SASKATCHEWAN FORMULARY COMMITTEE BULLETIN UPDATE TO THE 57th EDITION OF THE SASKATCHEWAN FORMULARY The
Headache. Headache Case. Migraine Headache. Eric Kraus, MD. Types» Without aura (common)» With aura (classic)
 Headache Eric Kraus, MD Headache Case This 23 year-old female has headaches behind the right eye that cause her to lay down in a quiet room. They have a throbbing quality and she may vomit. Migraine Headache
Headache Eric Kraus, MD Headache Case This 23 year-old female has headaches behind the right eye that cause her to lay down in a quiet room. They have a throbbing quality and she may vomit. Migraine Headache
[If yes, no further questions.]
![[If yes, no further questions.] [If yes, no further questions.]](/thumbs/94/118728940.jpg) 05/30/2014 Prior Authorization AETA BETTER HEALTH OF ILLIOIS MEDICAID Botox (IL88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and
05/30/2014 Prior Authorization AETA BETTER HEALTH OF ILLIOIS MEDICAID Botox (IL88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and
NBPDP FORMULARY UPDATE
 Bulletin #864 June 20, 2013 NBPDP FORMULARY UPDATE This update to the New Brunswick Prescription Drug Program (NBPDP) Formulary is effective June 20, 2013. Included in this bulletin: Regular Benefit Additions
Bulletin #864 June 20, 2013 NBPDP FORMULARY UPDATE This update to the New Brunswick Prescription Drug Program (NBPDP) Formulary is effective June 20, 2013. Included in this bulletin: Regular Benefit Additions
Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D.
 Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D. Objectives Present patient case Review epidemiology/pathophysiology
Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D. Objectives Present patient case Review epidemiology/pathophysiology
RATIONALE FOR INCLUSION IN PA PROGRAM
 RATIONALE FOR INCLUSION IN PA PROGRAM Background hydromorphone (Exalgo, Dilaudid) and oxymorphone (Opana and Opana ER) are Schedule II narcotics prescribed to treat moderate to severe pain. Morphine produces
RATIONALE FOR INCLUSION IN PA PROGRAM Background hydromorphone (Exalgo, Dilaudid) and oxymorphone (Opana and Opana ER) are Schedule II narcotics prescribed to treat moderate to severe pain. Morphine produces
RATIONALE FOR INCLUSION IN PA PROGRAM
 RATIONALE FOR INCLUSION IN PA PROGRAM Background Methadone hydrochloride is a long-acting opioid agonist at mu-opioid receptors that is used to manage pain that requires long-term, daily opioid treatment
RATIONALE FOR INCLUSION IN PA PROGRAM Background Methadone hydrochloride is a long-acting opioid agonist at mu-opioid receptors that is used to manage pain that requires long-term, daily opioid treatment
QUANTITY LIMIT CRITERIA
 DRUG CLASS INHALATION BRAND NAME (generic) QUANTITY LIMIT CRITERIA SHORT ACTING BETA2-ADRENERGIC AGONIST ORAL (albuterol inhalation solution) PROAIR HFA PROAIR RESPICLICK PROVENTIL HFA VENTOLIN HFA XOPENEX
DRUG CLASS INHALATION BRAND NAME (generic) QUANTITY LIMIT CRITERIA SHORT ACTING BETA2-ADRENERGIC AGONIST ORAL (albuterol inhalation solution) PROAIR HFA PROAIR RESPICLICK PROVENTIL HFA VENTOLIN HFA XOPENEX
