Chemistry Thermodynamics Neatly answer all questions completely for credit. Show all work.
|
|
|
- Meagan Spencer
- 5 years ago
- Views:
Transcription
1 Teacher Notes Time: 90 minutes (plus 30 minutes for teacher preparation) Student Difficult: moderate Purpose: Design and model processes, materials, and equipment that might be used to build a device that measures the calories of heat energy available in foods. Overview: Process Skills: Analyzing data Experimenting Applying concepts Identifying patterns Calculating Inferring Collecting data Interpreting Communicating Organizing data Lab Preparation: Prior to the lab, you will need to collect and organize the materials students will need to build a calorimeter. Other readily available materials may be included or substituted for those in the materials list. Notes on Technique Review with students the use of a balance to measure the mass of each sample. Also review the correct method of reading the water level in a graduated cylinder. Immediately after igniting the food sample, students should place the large can around the cork and then quickly but carefully put the small can in place above the flame. If the food flame goes out, the sample should be removed along with its holder, from the calorimeter, reignited, and the apparatus quickly reassembled. If possible, the food samples should be burned until only ashes remain. Reaction Energy 1 Thermochemistry
2 Tips and Tricks This lab works best with groups of 3 4 students. Once the calorimeter is built and before any samples are burned, you might want to have students rehearse the assembly process. The faster they are able to get the burning food sample under the can of water, the more accurate their results will be; less heat will be lost to the surroundings. Igniting the nut samples may take longer than the other foods. Some foods may be more difficult to hold in place than others. In these cases, use a small binder or alligator clips (with a little tube to attach to wire) to hold food. Modeling clay can be a good alternative to cork. Checkpoints 1) By the end of the first class period, have students turn in a detailed one-page plan/procedure for approval. 2) During the second class period, have students revise procedures according to your comments and begin the procedure. 3) After the first data collection, have students turn in a copy of their data so that you can ensure they are collecting data correctly. 4) Following completion of the experiment, have students turn in their procedure, data, and Analysis and Conclusions answers. Students labs should be evaluated on lab technique, quality and clarity of observations, and the explanation of observations and conclusions. Sample Procedure (Procedures will vary). A calorimeter was made from two metal cans, one smaller so it could nest inside the larger can. The top and bottom of the larger can were removed and holes were made in the rim near the bottom to allow oxygen needed for combustion enter. Two small holes were punched in opposite sides of the smaller can about ¼ of the way from the top. A wooden dowel was passed through the holes to support the smaller can above the burning food sample. Using the cork, needle, and pliers, a setup for mounting the food sample for burning was devised. A measured amount of water was added to the smaller can. Both the volume and temperature of the water were recorded. A food sample was massed on the balance and them mounted on the needle. The sample was ignited, and as soon as it caught fire, the large can was placed over the cork and the smaller can with its support was positioned above the flame. When the sample was completely burned, the smaller can was carefully removed. The water was stirred and the temperature of the water was observed and recorded. The burnt food sample was allowed to cool, carefully removed from the needle, and massed. Its mass was recorded. Three trials were made with samples of the same food to ensure consistency. Then the procedure was repeated for three other food samples. Reaction Energy 2 Thermochemistry
3 Background Background information is provided for students. If time allows, interested students can do online research to find out about calorimeter designs. Answers: Analysis 1. Answers will depend on the food choices. 2. Students should find that foods containing fats and oils have higher calorie content. 3. Answers will vary according to the food choices. Students should find that foods with high fat and oil content burned more quickly. 4. Answers will vary. Conclusions 1. Students should find that their heat transfer measurements were higher for foods that are rated as higher in calories. 2. Students should recognize that some heat energy is lost to the air. Students may suggest that a calorimeter design with better insulation would produce more accurate measurements. Students may suggest that a water jacket surrounding the burning sample would collect more of the heat transfer. 3. Students should recognize the relationship between measurements of high calories by the calorimeter and more energy available to the human body. 4. Students should find that their higher measurements were comparable to higher calorie ratings of foods. They should account for the differences by identifying inefficiencies in the calorimeter design. For high-fat foods that don t burn (oddly, some popular snack chips do not burn) students should suggest reasons why. Reaction Energy 3 Thermochemistry
4 Thermodynamics In this lab, you will design, construct, and test your own device to determine the amount of heat energy in snack foods. Purpose: Design and model processes, materials, and equipment that might be used to build a device that measures the calories of heat energy available in foods. Engineering Design Process Identify a Need Conduct Research Brainstorm Solutions Select a Solution Design a Solution What s the problem? What have others done? What are the constraints? How can you solve the problem? Brainstorm multiple ideas. Which ideas best meet the need? Which ideas meet all your constraints? Choose the best one. Describe or draw a diagram of the steps you will take to solve the problem. Make a list of what you will need to implement your solution. Follow your plan. Test and Evaluate Test it out! Analyze your results. Were they what you expected? Think about what your results say about your plan. Redesign to Improve Communicate What changes could you make that would give you better results? Make your design even better. Test it out! Share your results with others throughout the design process and get their feedback. Report on your final design. Prior Knowledge Temperature, Thermochemistry, Heat capacity, Energy transfer, Specific heat, Chemical reaction, Calorimetry Reaction Energy 4 Thermochemistry
5 Possible materials may include, but are not limited to: alligator clips balance can opener cork distilled water gloves, heat resistant graduated cylinder graph paper hammer knife matches metal cans (1 large, 1 small) metal file modeling clay nail needle pencil pliers porcelain tile, large and flat stirrer thermometer tongs wire wood dowel snack foods (a variety, such as, cracker, marshmallow, popcorn, pretzel, potato chip, roasted peanut, cashew or other whole nut) Background Have you ever heard the question, Do you eat to live or live to eat? For most people, the answer is both! Food contains energy that the human body uses to function and survive. That energy, released in the form of heat as food is digested, is transferred to all parts of the body. The unit used for measuring the transfer of heat is the calorie. One calorie (cal) is the energy required to raise the temperature of 1 gram (or 1 ml) of water 1 degree Celsius. The energy content of food is also measured in calories. However, these calories are actually large Calories, or kilocalories (Kcal). A kilocalorie is equivalent to 1,000 calories, or 1,000 times the heat energy of 1 calorie. The number of calories in a particular quantity of a packaged food is often listed on the packaging. Our bodies crave fats because fats come packed with energy. When your body takes in food, it burns that food in much the same way a log burns in a fireplace. So, the more calories your body takes in, the more time it will take for it to burn them off. Think of a big and a small log. Which one will take longer to burn? The calorimeter measures the amount of energy in foods by burning them in the most isolated conditions. This ensures that the maximum amount of heat gets transferred from the burning food so to determine how much energy there is in it. Reaction Energy 5 Thermochemistry
6 Safety Chemistry Thermodynamics While doing this activity, observe the following safety precautions. Always wear safety goggles and a lab apron to protect your eyes and clothing. If you get a piece of burning food on your skin, flush the affected area in a sink immediately, and alert your teacher. Avoid touching sharp edges of metal apparatus. When handling metal apparatus, be careful and conscious of your movements, and avoid cutting your fingers on any unfinished edges. Call your teacher in the event of an accident. Working with open flames is dangerous, and you should take every precaution not to allow burning matches or food near anything flammable. Always work on a nonflammable surface. Procedure 1) Form a hypothesis about what materials or methods you can use to build a device and measure the heat energy of various snack foods. 2) Come up with a plan. Develop and conduct an experiment to determine what methods and materials can be used to measure the heat transfer between food samples and water. Limit the number of trials and foods you choose for your experiment to those that can be completed during the time your teacher has allotted for this lab. Consult with your teacher to make sure that the conditions you have chosen are appropriate. 3) Write out a procedure for your experiment. As you plan the procedure, make the following decisions: a. Decide what methods and/or materials you will use to build your device. b. Decide how you will measure the heat energy transfer. c. Select the materials and technology that you will need for your experiment from those that your teacher has provided. d. Decide the kind of data you will collect and develop a method for recording the data. e. Decide how you will determine that your measurements are as valid as you can make them. f. Decide what safety procedures are necessary. 4) Have your teacher approve your plans. 5) Obtain your materials and set up any apparatus you will need. 6) Take appropriate safety precautions. 7) Make objective observations. 8) Collect data and organize them into appropriate tables and/or graphs. Be certain that the graphs and tables are properly constructed and labeled. Compare the amounts of calories in different foods and identify which foods contain the most energy. 9) Share your results with other teams. Elicit their feedback on your design. 10) Improve your design and test it. Reaction Energy 6 Thermochemistry
7 Name: Class: Date: Chemistry Thermodynamics Cleanup and Disposal Clean all apparatus and your lab station. Return equipment to its proper place. Dispose of foods and ashes in the containers designated by your teacher. Wash your hands thoroughly after all work is finished and before you leave the lab. Analysis 1) Summarizing Data Summarize your findings and observations, including an analysis of any data tables or graphs that you created. 2) Identifying Relationships For each food you tested, describe the heat energy content in relation to other foods. Why do different foods produce different results? 3) Analyzing Methods Did the foods require the same time to burn and did that affect the amount of heat transferred? Did all of the foods burn completely? Reaction Energy 7 Thermochemistry
8 Name: Class: Date: Chemistry Thermodynamics 4) Describing Events Share your results with your classmates. Were your results similar to those of your classmates? Conclusions 1) Drawing Conclusions What conclusions can you draw about the data you collected? What additional conclusions can you draw from class results? 2) Evaluating Models Was your device an accurate device for measuring the calories in food? Was any heat lost? Suggest ways that you could improve the design. Reaction Energy 8 Thermochemistry
9 Name: Class: Date: Chemistry Thermodynamics 3) Compare and Contrast How does the burning of food in this experiment compare to the use of food by the body? 4) Evaluate Results How do your results compare to the calorie ratings listed on the packages of the food you tested? What could have influenced any differences? Reaction Energy 9 Thermochemistry
Food as Fuel. Investigative Lab 78. Name Class Date. Measuring the Chemical Energy Stored in Food
 Name Class Date Food as Fuel Measuring the Chemical Energy Stored in Food Investigative Lab 78 Question How can you measure the calorie content of a peanut? Lab Overview In this investigation, you will
Name Class Date Food as Fuel Measuring the Chemical Energy Stored in Food Investigative Lab 78 Question How can you measure the calorie content of a peanut? Lab Overview In this investigation, you will
FOOD CALORIMETRY OVERVIEW & PURPOSE
 FOOD CALORIMETRY OVERVIEW & PURPOSE How much energy is released when your body burns food? Have you ever looked at the nutrition label on a package of food or a soda can? It s here that you can find Calories
FOOD CALORIMETRY OVERVIEW & PURPOSE How much energy is released when your body burns food? Have you ever looked at the nutrition label on a package of food or a soda can? It s here that you can find Calories
Lab 7: Heat and Calorimetry
 Concepts to explore: Gain applicable knowledge about calories Compare the calorie content of food samples Introduction Have you ever roasted marshmallows over a campfire? You have probably heard that foods
Concepts to explore: Gain applicable knowledge about calories Compare the calorie content of food samples Introduction Have you ever roasted marshmallows over a campfire? You have probably heard that foods
Experimental Procedure
 1 of 10 9/12/2018, 1:26 PM https://www.sciencebuddies.org/science-fair-projects/project-ideas/foodsci_p012/cooking-food-science/food-calorimeter (http://www.sciencebuddies.org/science-fairprojects/project-ideas/foodsci_p012/cooking-food-science/food-calorimeter)
1 of 10 9/12/2018, 1:26 PM https://www.sciencebuddies.org/science-fair-projects/project-ideas/foodsci_p012/cooking-food-science/food-calorimeter (http://www.sciencebuddies.org/science-fairprojects/project-ideas/foodsci_p012/cooking-food-science/food-calorimeter)
1. Calculate the energy content in peanuts using measurements involving the bomb calorimeter.
 Food Explorations Lab: STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will work in groups and with your teacher to determine the potential energy of a peanut through the use of
Food Explorations Lab: STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will work in groups and with your teacher to determine the potential energy of a peanut through the use of
The Great Peanut Problem
 The Great Peanut Problem Calorimetry SCIENTIFIC Introduction All human activity requires burning food for energy. How much energy is released when food burns in the body? How is the calorie content of
The Great Peanut Problem Calorimetry SCIENTIFIC Introduction All human activity requires burning food for energy. How much energy is released when food burns in the body? How is the calorie content of
Sweet Calories: Energy Content of Marshmallows Lab
 Honors Chemistry Name: Date: Pd: Partner(s): Sweet Calories: Energy Content of Marshmallows Lab Introduction: All human activity requires burning food for energy. How much energy is released when food
Honors Chemistry Name: Date: Pd: Partner(s): Sweet Calories: Energy Content of Marshmallows Lab Introduction: All human activity requires burning food for energy. How much energy is released when food
Energy Content of Foods
 Skills Practice Lab Energy Content of Foods PROBEWARE LAB You are a lab technician working for NASA. Recently you were given the job of deciding what type of foods should be included in the next space
Skills Practice Lab Energy Content of Foods PROBEWARE LAB You are a lab technician working for NASA. Recently you were given the job of deciding what type of foods should be included in the next space
The Energy in Food. Energy (J/g) = (final temperature start temperature) x mass of water (g) x 4.2 (J per o C) Mass of food burned (g)
 The Energy in Food NAME DATE Food supplies energy for all animals without it we could not live. The quantity of energy stored in food is of great interest to humans. The energy your body needs for running,
The Energy in Food NAME DATE Food supplies energy for all animals without it we could not live. The quantity of energy stored in food is of great interest to humans. The energy your body needs for running,
Energy Found in a Cheeto and a Marshmallow
 Energy Found in a Cheeto and a Marshmallow Class: SCH 4U Date Lab Completed: Mon. Oct. 17, 2016 Teacher. Ms. G. Warner Name: Sarah Smith Partner: Chester Field Introduction: The energy that is found in
Energy Found in a Cheeto and a Marshmallow Class: SCH 4U Date Lab Completed: Mon. Oct. 17, 2016 Teacher. Ms. G. Warner Name: Sarah Smith Partner: Chester Field Introduction: The energy that is found in
LEARN * DREAM * AWAKEN* DISCOVER * ENLIGHTEN * INVESTIGATE * QUESTION * EXPLORE
 Physical Science CHEMISTRY () S C I E N C E C H E M I S T R Y This Enrichment4You e-guide provides a brief overview of energy in food. In this e-guide you will: *Learn Basic Information About Calculating
Physical Science CHEMISTRY () S C I E N C E C H E M I S T R Y This Enrichment4You e-guide provides a brief overview of energy in food. In this e-guide you will: *Learn Basic Information About Calculating
ENZYME CONCENTRATIONS AND ENZYME ACTIVITY: PLANNING SHEET
 Activity 2.11 Student Sheet ENZYME CONCENTRATIONS AND ENZYME ACTIVITY: PLANNING SHEET To investigate how enzyme concentration can affect the initial rate of reaction. Wear eye protection, lab coats and
Activity 2.11 Student Sheet ENZYME CONCENTRATIONS AND ENZYME ACTIVITY: PLANNING SHEET To investigate how enzyme concentration can affect the initial rate of reaction. Wear eye protection, lab coats and
The Power Within a Peanut: Tremendous Energy Potential
 Lesson Plan for Grades 9-12 Length of Lesson: 1 hr 30 min Authored by: Mohammad Kamyab Javanmardi for UT Environmental Science Institute Date created: [02/05/2015] Subject area/course: Chemistry (energy
Lesson Plan for Grades 9-12 Length of Lesson: 1 hr 30 min Authored by: Mohammad Kamyab Javanmardi for UT Environmental Science Institute Date created: [02/05/2015] Subject area/course: Chemistry (energy
How do scientists design experiments? This activity will introduce you to the concept of variables.
 1.6 (page 1) Science Projects For ALL Students Variables How do scientists design experiments? This activity will introduce you to the concept of variables. Scientists test their ideas by conducting experiments.
1.6 (page 1) Science Projects For ALL Students Variables How do scientists design experiments? This activity will introduce you to the concept of variables. Scientists test their ideas by conducting experiments.
Cut out these cards and match them up to make correct sentences. Arrange the sentences in a sensible order, and stick them into your book.
 7Ia-1 Energy from food Cut out these cards and match them up to make correct sentences. Arrange the sentences in a sensible order, and stick them into your book. explain why different people need different
7Ia-1 Energy from food Cut out these cards and match them up to make correct sentences. Arrange the sentences in a sensible order, and stick them into your book. explain why different people need different
Running head: CALORIMETRY TO COMPARE ENERGY CONTENT OF FOODS 1. Using Calorimetry as an Experimental Means to Compare the Energy Content of Two Foods
 Running head: CALORIMETRY TO COMPARE ENERGY CONTENT OF FOODS 1 Using Calorimetry as an Experimental Means to Compare the Energy Content of Two Foods Emily Morales Providence Extension Program Mason Campus
Running head: CALORIMETRY TO COMPARE ENERGY CONTENT OF FOODS 1 Using Calorimetry as an Experimental Means to Compare the Energy Content of Two Foods Emily Morales Providence Extension Program Mason Campus
Determining Caloric Content of Food. Sudha Madhugiri, Ph.D. Collin College Department of Chemistry
 Determining Caloric Content of Food Sudha Madhugiri, Ph.D. Collin College Department of Chemistry We all know that food gives us energy. Some foods are tastier but are not so healthy. Some foods help you
Determining Caloric Content of Food Sudha Madhugiri, Ph.D. Collin College Department of Chemistry We all know that food gives us energy. Some foods are tastier but are not so healthy. Some foods help you
! Which nut has the most. stored energy? By: Andrea Rendon. Chemistry. April, 18th, 2014
 Which nut has the most stored energy? By: Andrea Rendon Chemistry April, 18th, 2014 Which nut, walnuts, pecans, or almonds have the most stored energy? A nut is a fruit composed of a hard shell and seed.
Which nut has the most stored energy? By: Andrea Rendon Chemistry April, 18th, 2014 Which nut, walnuts, pecans, or almonds have the most stored energy? A nut is a fruit composed of a hard shell and seed.
Lab #1 Laboratory Safety and Equipment
 Page1 Lab #1 and Equipment Objectives: Identify the locations of important safety information and equipment. Understand how to properly use safety equipment in the laboratory. Understand the importance
Page1 Lab #1 and Equipment Objectives: Identify the locations of important safety information and equipment. Understand how to properly use safety equipment in the laboratory. Understand the importance
Laboratory 10: Energy in Chemical Reactions
 Name: Partners name(s): Lecture Notes 1. How is heat measured? Laboratory 10: Energy in Chemical Reactions Stamp: 2. How are heat and temperature different? 3. What is a calorie? 4. What is heat of solution?
Name: Partners name(s): Lecture Notes 1. How is heat measured? Laboratory 10: Energy in Chemical Reactions Stamp: 2. How are heat and temperature different? 3. What is a calorie? 4. What is heat of solution?
Safety In the Science Lab
 Lab Safety: Everyone Is Responsible! Safety In the Science Lab Rules and Symbols Ms. A. Mormando's Class 1 Safety First Science is a hands-on laboratory class. You will be doing many laboratory activities,
Lab Safety: Everyone Is Responsible! Safety In the Science Lab Rules and Symbols Ms. A. Mormando's Class 1 Safety First Science is a hands-on laboratory class. You will be doing many laboratory activities,
Determination of Vitamin C in Fruit Juices
 Microscale Determination of Vitamin C in Fruit Juices Vitamin C is an important nutrient in the human diet. It is essential for preventing the disease called scurvy and for maintaining good health. Vitamin
Microscale Determination of Vitamin C in Fruit Juices Vitamin C is an important nutrient in the human diet. It is essential for preventing the disease called scurvy and for maintaining good health. Vitamin
Hydrolysis and Dehydration Synthesis
 Hydrolysis and Dehydration Synthesis Topic Ball-and-stick models can demonstrate hydrolysis and dehydration synthesis in carbohydrates. Introduction Living things are made up of both inorganic and organic
Hydrolysis and Dehydration Synthesis Topic Ball-and-stick models can demonstrate hydrolysis and dehydration synthesis in carbohydrates. Introduction Living things are made up of both inorganic and organic
Materials and Equipment
 Lab Group Members Date: Teacher/Period: Problem: Hypothesis: IV: Variables That Will not Change Control Changes DV: Materials and Equipment Safety Issues Sketch of Experimental Setup BLM 89 Lab BLM 90
Lab Group Members Date: Teacher/Period: Problem: Hypothesis: IV: Variables That Will not Change Control Changes DV: Materials and Equipment Safety Issues Sketch of Experimental Setup BLM 89 Lab BLM 90
Potato Calories. Science Fact or Fiction? Instructions
 Instructions Materials: [1] What does 200 calories look like? Food labels showing calorie content Scales Plates Examples of different foods Data sheet showing weight of 200 calories per food (attached)
Instructions Materials: [1] What does 200 calories look like? Food labels showing calorie content Scales Plates Examples of different foods Data sheet showing weight of 200 calories per food (attached)
Problem: What would happen to enzyme activity if enzymes are placed outside their normal conditions? Hypothesis:
 Name: Date: Period: Honors Biology: Enzyme Lab Background information What would happen to your cells if they made a poisonous chemical? You might think that they would die. In fact, your cells are always
Name: Date: Period: Honors Biology: Enzyme Lab Background information What would happen to your cells if they made a poisonous chemical? You might think that they would die. In fact, your cells are always
Observing Respiration
 Chapter 9 Cellular Respiration Design an Experiment Observing Respiration Introduction Cellular respiration occurs in all living things. During this process, animals take in oxygen and release carbon dioxide
Chapter 9 Cellular Respiration Design an Experiment Observing Respiration Introduction Cellular respiration occurs in all living things. During this process, animals take in oxygen and release carbon dioxide
Energy in the Food We Eat
 Energy in the Food We Eat Name: You know that the energy that keeps your brain and body going comes from the food you eat. Your digestive system and the cells in your body break down the food and gradually
Energy in the Food We Eat Name: You know that the energy that keeps your brain and body going comes from the food you eat. Your digestive system and the cells in your body break down the food and gradually
Lesson 8: Effect of temperature on an oyster s heart rate
 Lesson 8: Effect of temperature on an oyster s heart rate Focus Question: How does temperature affect the heart rate of an oyster? Purpose: In this lab based activity, students investigate the effect of
Lesson 8: Effect of temperature on an oyster s heart rate Focus Question: How does temperature affect the heart rate of an oyster? Purpose: In this lab based activity, students investigate the effect of
MAKE SMART INJECTION CHOICES INJECTION PROVIDERS GUIDE FOR SAFE INJECTIONS
 MAKE SMART INJECTION CHOICES INJECTION PROVIDERS GUIDE FOR SAFE INJECTIONS MAKE SMART INJECTION CHOICES WHO recommends syringes with re-use prevention (RUP) features for all injections. RUP syringes with
MAKE SMART INJECTION CHOICES INJECTION PROVIDERS GUIDE FOR SAFE INJECTIONS MAKE SMART INJECTION CHOICES WHO recommends syringes with re-use prevention (RUP) features for all injections. RUP syringes with
SENDING SECRET MESSAGES (1 Hour)
 SENDING SECRET MESSAGES (1 Hour) Addresses NGSS Level of Difficulty: 3 Grade Range: K-2 OVERVIEW In this activity, students explore how sound waves travel through various materials. They will build a sound
SENDING SECRET MESSAGES (1 Hour) Addresses NGSS Level of Difficulty: 3 Grade Range: K-2 OVERVIEW In this activity, students explore how sound waves travel through various materials. They will build a sound
Stored. Introduction: Name. process by. of usable. respiration. stored energy. allowing us to. the. that can be will indirectly.
 The Heat is On The Enery Stored in Food Introduction: Plants utilize sunliht durin photosynthesis too convert carbon dioxide and water into lucose (suar) and oxyen. This lucose has enery stored in its
The Heat is On The Enery Stored in Food Introduction: Plants utilize sunliht durin photosynthesis too convert carbon dioxide and water into lucose (suar) and oxyen. This lucose has enery stored in its
Marathon Snack Lab. Key Terms
 Lesson 3.4 Marathon Snack Lab Name Date Period calorie calorimetry Key Terms Engage Imagine that you are in a long race. Your body is rapidly depleting its energy reserves. You need energy. You grab a
Lesson 3.4 Marathon Snack Lab Name Date Period calorie calorimetry Key Terms Engage Imagine that you are in a long race. Your body is rapidly depleting its energy reserves. You need energy. You grab a
Experimental Procedure
 1 of 7 9/12/2018, 2:19 PM https://www.sciencebuddies.org/science-fair-projects/project-ideas/humbio_p043/human-biology-health/iron-rich-foods (http://www.sciencebuddies.org/science-fair-projects /project-ideas/humbio_p043/human-biology-health/iron-rich-foods)
1 of 7 9/12/2018, 2:19 PM https://www.sciencebuddies.org/science-fair-projects/project-ideas/humbio_p043/human-biology-health/iron-rich-foods (http://www.sciencebuddies.org/science-fair-projects /project-ideas/humbio_p043/human-biology-health/iron-rich-foods)
Exploding Corn: Student Pages DIFFERENCES BEWEEN MASS AND VOLUME CHANGES WITH POPCORN
 EXPERIMENT 2: Exploding Corn: Student Pages DIFFERENCES BEWEEN MASS AND VOLUME CHANGES WITH POPCORN BACKGROUND According to The Popcorn Board, Americans today consume 17 billion quarts of popped popcorn
EXPERIMENT 2: Exploding Corn: Student Pages DIFFERENCES BEWEEN MASS AND VOLUME CHANGES WITH POPCORN BACKGROUND According to The Popcorn Board, Americans today consume 17 billion quarts of popped popcorn
The Effect of Hydrogen Peroxide Concentration (substrate) on the Activity of the Enzyme Catalase
 The Effect of Hydrogen Peroxide Concentration (substrate) on the Activity of the Enzyme Catalase Exercise adapted from: Allot, A. & Mindorff, D. (2007). Biology Course Companion. Oxford: Oxford University
The Effect of Hydrogen Peroxide Concentration (substrate) on the Activity of the Enzyme Catalase Exercise adapted from: Allot, A. & Mindorff, D. (2007). Biology Course Companion. Oxford: Oxford University
Burning Snacks in Chem Lab Michael Hayes Hopewell High School Hopewell City Public Schools
 Science Science CH.1, CH.3, CH.5 Laboratories Experiential Learning This lesson uses graphing calculators and word processing software to facilitate student use of technology to determine the energy content
Science Science CH.1, CH.3, CH.5 Laboratories Experiential Learning This lesson uses graphing calculators and word processing software to facilitate student use of technology to determine the energy content
Stored Mechanical Gravitational Electrical. measured as. Forms of Energy
 Thermodynamics is the branch of science that studies the transformation of energy from one form to another. Thermochemistry specifically studies heat changes that accompany chemical reactions and phase
Thermodynamics is the branch of science that studies the transformation of energy from one form to another. Thermochemistry specifically studies heat changes that accompany chemical reactions and phase
Unit 5L.4: Food. Know that humans require food as an energy source. Know that a balanced diet must contain proteins, fats,
 Unit 5L.4: as an energy source Balanced diet Requirements for different lifestyles Science skills: Observing Classifying By the end of this unit you should: Know that humans require food as an energy source.
Unit 5L.4: as an energy source Balanced diet Requirements for different lifestyles Science skills: Observing Classifying By the end of this unit you should: Know that humans require food as an energy source.
Organic Compounds in the Foods
 Organic Compounds in the Foods Purpose: This lab activity will help you understand the chemical composition (i.e., carbohydrates, proteins, and fats) of the foods that you eat. Materials we will be using:
Organic Compounds in the Foods Purpose: This lab activity will help you understand the chemical composition (i.e., carbohydrates, proteins, and fats) of the foods that you eat. Materials we will be using:
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education
 UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education *4373149320* CO-ORDINATED SCIENCES 0654/63 Paper 6 Alternative to Practical May/June 2012 1 hour
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education *4373149320* CO-ORDINATED SCIENCES 0654/63 Paper 6 Alternative to Practical May/June 2012 1 hour
MATERIAL SAFETY DATA SHEET
 Page 1 of 6 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Material Identity Product Name: Citric Acid, Anhydrous, Powder Chemical Formula: C6H8O7 Product Code: CH-77929A General or Generic ID: Not available.
Page 1 of 6 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Material Identity Product Name: Citric Acid, Anhydrous, Powder Chemical Formula: C6H8O7 Product Code: CH-77929A General or Generic ID: Not available.
A. Background for Trainer: B. What OSHA Requires: Bloodborne Pathogens. Lesson Plan 6080a
 Lesson Plan 6080a This training session outline is designed to follow the accompanying booklet, OSHA s Bloodborne Pathogens Standard. The booklet reviews what employees who are potentially exposed to the
Lesson Plan 6080a This training session outline is designed to follow the accompanying booklet, OSHA s Bloodborne Pathogens Standard. The booklet reviews what employees who are potentially exposed to the
Chemistry of food and FOOD GRAINS Build a simple calorimeter. Regina Zibuck
 Chemistry of food and FOOD GRAINS Build a simple calorimeter Regina Zibuck rzibuck@wayne.edu Build and use a calorimeter (15 %) include calibration data in the 5 sheets. Written test (85 %) (Students are
Chemistry of food and FOOD GRAINS Build a simple calorimeter Regina Zibuck rzibuck@wayne.edu Build and use a calorimeter (15 %) include calibration data in the 5 sheets. Written test (85 %) (Students are
Lab-Specific Standard Operating Procedure (LSOP)- Picric Acid (2,4,6-Trinitrophenol) and Solutions Containing Picric Acid
 Office of the Vice President for Research Environmental Health and Safety Policies, Programs and Procedures Lab-Specific Standard Operating Procedure (LSOP)- Picric Acid (2,4,6-Trinitrophenol) and Solutions
Office of the Vice President for Research Environmental Health and Safety Policies, Programs and Procedures Lab-Specific Standard Operating Procedure (LSOP)- Picric Acid (2,4,6-Trinitrophenol) and Solutions
LAB: DIFFUSION ACROSS A SELECTIVELY PERMEABLE MEMBRANE
 LAB: DIFFUSION ACROSS A SELECTIVELY PERMEABLE MEMBRANE NAME: PERIOD: DATE: Building Background Knowledge: 1) SELECTIVELY PERMEABLE MEMBRANE: Every cell is surrounded by a selectively permeable membrane
LAB: DIFFUSION ACROSS A SELECTIVELY PERMEABLE MEMBRANE NAME: PERIOD: DATE: Building Background Knowledge: 1) SELECTIVELY PERMEABLE MEMBRANE: Every cell is surrounded by a selectively permeable membrane
Marathon Snack Alternate Assignment. Key Terms
 Lesson 3.4 Marathon Snack Alternate Assignment Name Date Period calorie calorimetry Key Terms Engage Imagine that you are in a long race. Your body is rapidly depleting its energy reserves. You need energy.
Lesson 3.4 Marathon Snack Alternate Assignment Name Date Period calorie calorimetry Key Terms Engage Imagine that you are in a long race. Your body is rapidly depleting its energy reserves. You need energy.
Entry Deadline: October 14, 2014 Each team participating will need to call the Extension office at to enter.
 2014 Palo Pinto County 4-H Food Challenge Sunday, October 19, 2014 2:00 p.m. Registration/Box Check will Begin 2:30 p.m. Contest to Begin Palo Pinto County Extension Office Entry Deadline: October 14,
2014 Palo Pinto County 4-H Food Challenge Sunday, October 19, 2014 2:00 p.m. Registration/Box Check will Begin 2:30 p.m. Contest to Begin Palo Pinto County Extension Office Entry Deadline: October 14,
Experimental Procedure
 1 of 6 10/3/2018, 2:39 PM https://www.sciencebuddies.org/science-fair-projects/project-ideas/biomed_p012/medical-biotechnology/stopping-fungus-with-antifungal-medicines (http://www.sciencebuddies.org/science-fair-projects/project-ideas/biomed_p012/medical-biotechnology/stopping-fungus-with-antifungal-medicines)
1 of 6 10/3/2018, 2:39 PM https://www.sciencebuddies.org/science-fair-projects/project-ideas/biomed_p012/medical-biotechnology/stopping-fungus-with-antifungal-medicines (http://www.sciencebuddies.org/science-fair-projects/project-ideas/biomed_p012/medical-biotechnology/stopping-fungus-with-antifungal-medicines)
Design a Cell. Investigative Lab 6. Comparing the Effects of Cell Shape on Diffusion Rate. Name Class Date
 Design a Cell Comparing the Effects of Cell Shape on Diffusion Rate Question What cell shapes are the most efficient at bringing in substances by diffusion? Lab Overview In this investigation you will
Design a Cell Comparing the Effects of Cell Shape on Diffusion Rate Question What cell shapes are the most efficient at bringing in substances by diffusion? Lab Overview In this investigation you will
Nutrition, Digestive System & Enzyme Review Packet
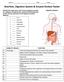 Name: Date: Class: Nutrition, Digestive System & Enzyme Review Packet Identify the major parts of the human digestive system on a diagram. Using the key, match the following parts to their correct structure
Name: Date: Class: Nutrition, Digestive System & Enzyme Review Packet Identify the major parts of the human digestive system on a diagram. Using the key, match the following parts to their correct structure
Name: Date Block Selective Permeability
 LAB Name: Date Block Selective Permeability OBJECTIVES: Observe the selective permeability of an artificial membrane. Observe diffusion of substances across an artificial membrane. Devise a model for the
LAB Name: Date Block Selective Permeability OBJECTIVES: Observe the selective permeability of an artificial membrane. Observe diffusion of substances across an artificial membrane. Devise a model for the
Material Safety Data Sheet
 Material Safety Data Sheet Conforms to ISO 11014:2009 VENTOL CLEANER 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY Product name : VENTOL CLEANER Use/description of product : VENTOL
Material Safety Data Sheet Conforms to ISO 11014:2009 VENTOL CLEANER 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY Product name : VENTOL CLEANER Use/description of product : VENTOL
SCIENCE (REVISED SYLLABUS) HIGHER LEVEL
 J.37 PRE-JUNIOR CERTIFICATE EXAMINATION, 2010 SCIENCE (REVISED SYLLABUS) HIGHER LEVEL TIME : 2 HOURS INSTRUCTIONS 1. Write your name, school name and teacher s name in the boxes provided on this page.
J.37 PRE-JUNIOR CERTIFICATE EXAMINATION, 2010 SCIENCE (REVISED SYLLABUS) HIGHER LEVEL TIME : 2 HOURS INSTRUCTIONS 1. Write your name, school name and teacher s name in the boxes provided on this page.
MATERIAL SAFETY DATA SHEET
 MATERIAL SAFETY DATA SHEET ELAN CHEMICAL COMPANY, INC Date Printed: October 23, 2007 Date Updated: August 26, 2005 1. Product /Company Identification Product Name: NATURAL ACETIC ACID Manufacturer: Elan
MATERIAL SAFETY DATA SHEET ELAN CHEMICAL COMPANY, INC Date Printed: October 23, 2007 Date Updated: August 26, 2005 1. Product /Company Identification Product Name: NATURAL ACETIC ACID Manufacturer: Elan
2018 Version. Introduction to Science Junior Science
 2018 Version Introduction to Science Junior Science 1 What is Science? Science is both a collection of knowledge and the process for building that knowledge. Science asks questions about the natural world
2018 Version Introduction to Science Junior Science 1 What is Science? Science is both a collection of knowledge and the process for building that knowledge. Science asks questions about the natural world
Molecular Gastronomy Lab Lesson Plan: Spherification and Olive Oil Powder For an advanced high school Chemistry class
 Molecular Gastronomy Lab Lesson Plan: Spherification and Olive Oil Powder For an advanced high school Chemistry class Topic and Purpose: Exploring transformations from one molecular state to another through
Molecular Gastronomy Lab Lesson Plan: Spherification and Olive Oil Powder For an advanced high school Chemistry class Topic and Purpose: Exploring transformations from one molecular state to another through
CITY OF WAUPACA, WISCONSIN WATER DEPARTMENT WATER SERVICE LATERAL THAWING RULES AND PROCEDURES (prepared February 24, 2015)
 CITY OF WAUPACA, WISCONSIN WATER DEPARTMENT WATER SERVICE LATERAL THAWING RULES AND PROCEDURES (prepared February 24, 2015) Pursuant to Wisconsin Public Service Commission 185.88 rules on Frozen Laterals:
CITY OF WAUPACA, WISCONSIN WATER DEPARTMENT WATER SERVICE LATERAL THAWING RULES AND PROCEDURES (prepared February 24, 2015) Pursuant to Wisconsin Public Service Commission 185.88 rules on Frozen Laterals:
Year 8 Assessment. Autumn Term Remember!
 Remember! Each section of questions gets harder as you go through (higher level questions at the end of each section). Try all questions. Write down an idea even if you re not sure you might get a mark!
Remember! Each section of questions gets harder as you go through (higher level questions at the end of each section). Try all questions. Write down an idea even if you re not sure you might get a mark!
Introduction to Science Junior Science. Easy to read Version
 Introduction to Science Junior Science Easy to read Version 1 1a What is Science? Science is both a collection of knowledge and the process for building that knowledge. Science asks questions about the
Introduction to Science Junior Science Easy to read Version 1 1a What is Science? Science is both a collection of knowledge and the process for building that knowledge. Science asks questions about the
MARY PACX ARTHRITIS PROGRAM hpdrl of Ihr Va~ocowtr (oaslal Utalth Authority
 - 2 MARY PACX ARTHRITIS PROGRAM hpdrl of Ihr Va~ocowtr (oaslal Utalth Authority OCCUPATIONAL THERAPY METHOD OF MAKING THE POLYETHYLENE WORKING SPLINT FOR THE WRIST Equipment / Supplies Required: plaster
- 2 MARY PACX ARTHRITIS PROGRAM hpdrl of Ihr Va~ocowtr (oaslal Utalth Authority OCCUPATIONAL THERAPY METHOD OF MAKING THE POLYETHYLENE WORKING SPLINT FOR THE WRIST Equipment / Supplies Required: plaster
Identification of Organic Compounds Lab
 Identification of Organic Compounds Lab Introduction All organic compounds contain the element carbon (C). Organic compounds usually also contain oxygen (O) or hydrogen (H) or both. They may also contain
Identification of Organic Compounds Lab Introduction All organic compounds contain the element carbon (C). Organic compounds usually also contain oxygen (O) or hydrogen (H) or both. They may also contain
Core practical 1: Investigate how enzyme concentration affects the initial rate of an enzyme-controlled reaction
 Core practical 1 Student sheet Core practical 1: Investigate how enzyme concentration affects the initial Objectives To be able to measure the initial rate of enzyme activity To understand why measuring
Core practical 1 Student sheet Core practical 1: Investigate how enzyme concentration affects the initial Objectives To be able to measure the initial rate of enzyme activity To understand why measuring
Organic Molecule Composition of Milk: Lab Investigation
 Name: Organic Molecule Composition of Milk: Lab Investigation Introduction & Background Milk & milk products have been a major food source from earliest recorded history. Milk is a natural, nutritionally
Name: Organic Molecule Composition of Milk: Lab Investigation Introduction & Background Milk & milk products have been a major food source from earliest recorded history. Milk is a natural, nutritionally
SAFETY DATA SHEET DUNLOP DISPERSION ADHESIVES AND GROUTS
 SAFETY DATA SHEET DUNLOP DISPERSION ADHESIVES AND GROUTS 1. IDENTIFICATION OF THE PREPARATION PRODUCT NAMES: PRODUCT DESCRIPTION: APPLICATIONS: MANUFACTURER: Dunlop CTA, Dunlop Fix and Grout, Dunlop Waterproof
SAFETY DATA SHEET DUNLOP DISPERSION ADHESIVES AND GROUTS 1. IDENTIFICATION OF THE PREPARATION PRODUCT NAMES: PRODUCT DESCRIPTION: APPLICATIONS: MANUFACTURER: Dunlop CTA, Dunlop Fix and Grout, Dunlop Waterproof
Directions for the Creation and Use of GHS Labels
 Directions for the Creation and Use of GHS Labels Globally Harmonized System (GHS) labels must have the following elements: 1. Product Name or Identifier 2. Pictograms (See Final Page) 3. Signal Word (Danger
Directions for the Creation and Use of GHS Labels Globally Harmonized System (GHS) labels must have the following elements: 1. Product Name or Identifier 2. Pictograms (See Final Page) 3. Signal Word (Danger
 You should ALWAYS store persulfates: The Right Chemistry When Handled Right In a cool, dry area with temperatures below 77ºF (25ºC) for best stability. In a well-ventilated space. Away from incompatible
You should ALWAYS store persulfates: The Right Chemistry When Handled Right In a cool, dry area with temperatures below 77ºF (25ºC) for best stability. In a well-ventilated space. Away from incompatible
May 13, 2017 Data Back Ban of Artificial Trans Fats
 EDUCATOR GUIDE May 13, 2017 Data Back Ban of Artificial Trans Fats Teacher Guide: Greasy Spots Class time: 30-50 minutes. Purpose: Students can use three different types of assays to test a variety of
EDUCATOR GUIDE May 13, 2017 Data Back Ban of Artificial Trans Fats Teacher Guide: Greasy Spots Class time: 30-50 minutes. Purpose: Students can use three different types of assays to test a variety of
LAB: DIFFUSION ACROSS A SELECTIVELY PERMEABLE MEMBRANE
 LAB: DIFFUSION ACROSS A SELECTIVELY PERMEABLE MEMBRANE NAME: PERIOD: DATE: Building Background Knowledge: 1) SELECTIVELY PERMEABLE MEMBRANE: Every cell is surrounded by a selectively permeable membrane
LAB: DIFFUSION ACROSS A SELECTIVELY PERMEABLE MEMBRANE NAME: PERIOD: DATE: Building Background Knowledge: 1) SELECTIVELY PERMEABLE MEMBRANE: Every cell is surrounded by a selectively permeable membrane
RISK ASSESMENT AND PERSONAL PROTECTIVE EQUIPMENTS
 RISK ASSESMENT AND PERSONAL PROTECTIVE EQUIPMENTS Ahmet ERSOY Occupational Health and Safety Expert Ankara Personal Protection and PPE OHS aims to protect the health, safety and welfare of people at work.
RISK ASSESMENT AND PERSONAL PROTECTIVE EQUIPMENTS Ahmet ERSOY Occupational Health and Safety Expert Ankara Personal Protection and PPE OHS aims to protect the health, safety and welfare of people at work.
INVESTIGATING BREATHING: SHEET A
 Activity 7.13a Student Sheet INVESTIGATING BREATHING: SHEET A Purpose To investigate tidal volume, vital capacity, rate of breathing, respiratory minute ventilation and oxygen consumption. To interpret
Activity 7.13a Student Sheet INVESTIGATING BREATHING: SHEET A Purpose To investigate tidal volume, vital capacity, rate of breathing, respiratory minute ventilation and oxygen consumption. To interpret
Core practical 4: Investigate the effect of sucrose concentration on pollen tube growth
 Core practical 4 Teacher sheet Core practical 4: Investigate the effect of sucrose concentration on Objectives Observe the growth of pollen grains Develop the skills of planning investigations and carrying
Core practical 4 Teacher sheet Core practical 4: Investigate the effect of sucrose concentration on Objectives Observe the growth of pollen grains Develop the skills of planning investigations and carrying
Who took Kaleb s ipod? -- An organic compound mystery
 Who took Kaleb s ipod? -- An organic compound mystery Dr. Jennifer Doherty, Dr. Ingrid Waldron and Dr. Lori Spindler, Department of Biology, University of Pennsylvania, copyright 2009 Adapted from Identity
Who took Kaleb s ipod? -- An organic compound mystery Dr. Jennifer Doherty, Dr. Ingrid Waldron and Dr. Lori Spindler, Department of Biology, University of Pennsylvania, copyright 2009 Adapted from Identity
Safety Data Sheet. Professional Dental Topical 5% Sodium Fluoride Varnish for dental use
 1. IDENTIFICATION OF THE MATERIAL AND SUPPLIER Product Use: Manufacturer Name: Address: Fluorilaq TM Professional Dental Topical 5% Sodium Fluoride Varnish for dental use Pascal International, Inc. 2929
1. IDENTIFICATION OF THE MATERIAL AND SUPPLIER Product Use: Manufacturer Name: Address: Fluorilaq TM Professional Dental Topical 5% Sodium Fluoride Varnish for dental use Pascal International, Inc. 2929
TRACKS Lesson Plan. Drinks and Calcium Rethink Your Drink Special Needs Students Any Grade
 TRACKS Lesson Plan Drinks and Calcium Rethink Your Drink Special Needs Students Any Grade I. Nutrition Education Goal & Objective: Goal 1: Students will comprehend concepts consistent with USDA guidance
TRACKS Lesson Plan Drinks and Calcium Rethink Your Drink Special Needs Students Any Grade I. Nutrition Education Goal & Objective: Goal 1: Students will comprehend concepts consistent with USDA guidance
PARTICIPANT HANDOUT. Module 4: Tips for Reducing Exposure to Bloodborne and Other Infectious Diseases While Caring for Clients
 Caring for Yourself While Caring for Others Module 4: Tips for Reducing Exposure to Bloodborne and Other Infectious Diseases While Caring for Clients PARTICIPANT HANDOUT DEPARTMENT OF HEALTH AND HUMAN
Caring for Yourself While Caring for Others Module 4: Tips for Reducing Exposure to Bloodborne and Other Infectious Diseases While Caring for Clients PARTICIPANT HANDOUT DEPARTMENT OF HEALTH AND HUMAN
Glove Use Considerations
 Glove Use Considerations One of the first lines of defense against laboratory hazards is protective gloves, so it pays to choose your gloves wisely and seek advice if necessary. In a lab setting our hands
Glove Use Considerations One of the first lines of defense against laboratory hazards is protective gloves, so it pays to choose your gloves wisely and seek advice if necessary. In a lab setting our hands
Pulpdent Corporation Revision Date: May 1, 2017 Safety Data Sheet
 1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Etch-All 10% Phosphoric Acid Etching Gel 1.2 1.2.2 1.2.3 Application / Use SIC Use Category 1.3 Manufacturer Pulpdent
1.0 Commercial Product Name and Supplier 1.1 Commercial product name / designation Etch-All 10% Phosphoric Acid Etching Gel 1.2 1.2.2 1.2.3 Application / Use SIC Use Category 1.3 Manufacturer Pulpdent
BIO 12 UNIT 04: The Cell Membrane BCLN Rev. July, 2015
 Project 1: Osmosis Lab Name: Potential Credits: /50 Project Goals: to gain and demonstrate a better understanding of osmosis Instructions Please read through the Unit 4 Lessons, paying particular attention
Project 1: Osmosis Lab Name: Potential Credits: /50 Project Goals: to gain and demonstrate a better understanding of osmosis Instructions Please read through the Unit 4 Lessons, paying particular attention
TOPIC NFL PLAY 60 Kids Day Live Virtual Field Trip
 EDUCATOR COMPANION GUIDE TOPIC NFL PLAY 60 Kids Day Live Virtual Field Trip KEY LEARNING OBJECTIVES In the following activities students will: Model the functioning of the heart. Describe how the heart
EDUCATOR COMPANION GUIDE TOPIC NFL PLAY 60 Kids Day Live Virtual Field Trip KEY LEARNING OBJECTIVES In the following activities students will: Model the functioning of the heart. Describe how the heart
Who took Jerell s ipod? An organic compound mystery 1
 Who took Jerell s ipod? An organic compound mystery 1 Jerell is a 10 th grade student who works at McDonald s on the weekends. While on break, Jerell was studying for his biology test and listening to
Who took Jerell s ipod? An organic compound mystery 1 Jerell is a 10 th grade student who works at McDonald s on the weekends. While on break, Jerell was studying for his biology test and listening to
Safety Data Sheet CITRA FORCE
 Safety Data Sheet CITRA FORCE Section 1 - Chemical Product and Company Identification Supplier s Information: Superco Specialty Products 25041 Anza Drive Valencia, CA 91355 (661) 775-8877 Section 2 - Hazard
Safety Data Sheet CITRA FORCE Section 1 - Chemical Product and Company Identification Supplier s Information: Superco Specialty Products 25041 Anza Drive Valencia, CA 91355 (661) 775-8877 Section 2 - Hazard
The OSHA Standard. The OSHA Standard. The OSHA Standard
 Bloodborne Pathogen Training 1 What are Bloodborne Pathogens? We already have learned that pathogens are infectious agents that can cause us to get sick like viruses or bacteria or germs or parasites.
Bloodborne Pathogen Training 1 What are Bloodborne Pathogens? We already have learned that pathogens are infectious agents that can cause us to get sick like viruses or bacteria or germs or parasites.
TWISTING STAIR STEPPER WITH BANDS NO. 068 USER MANUAL
 TWISTING STAIR STEPPER WITH BANDS NO. 068 USER MANUAL IMPORTANT: Read all instructions carefully before using this product. Retain owner s manual for future reference. For customer service, please contact:
TWISTING STAIR STEPPER WITH BANDS NO. 068 USER MANUAL IMPORTANT: Read all instructions carefully before using this product. Retain owner s manual for future reference. For customer service, please contact:
Biology Partnership (A Teacher Quality Grant) Lesson Plan Construction Form
 Biology Partnership (A Teacher Quality Grant) Lesson Plan Construction Form Identifying Information: (Group Members and Schools, Title of Lesson, Length in Minutes, Course Level) Group Members: Marilyn
Biology Partnership (A Teacher Quality Grant) Lesson Plan Construction Form Identifying Information: (Group Members and Schools, Title of Lesson, Length in Minutes, Course Level) Group Members: Marilyn
The Hydrogen Peroxide Breakdown
 Biology The Hydrogen Peroxide Breakdown Examining Factors That Affect the Reaction Rate of Enzymes MATERIALS AND RESOURCES EACH GROUP aprons 2 beakers, 50 ml calculator, graphing forceps goggles graduated
Biology The Hydrogen Peroxide Breakdown Examining Factors That Affect the Reaction Rate of Enzymes MATERIALS AND RESOURCES EACH GROUP aprons 2 beakers, 50 ml calculator, graphing forceps goggles graduated
Food Is Fuel - Student Edition (Human Biology)
 Food Is Fuel - Student Edition (Human Biology) The Program in Human Biology, Stanford Univ- ersity, (HumBio) CK Editor Say Thanks to the Authors Click http://www.ck.org/saythanks (No sign in required)
Food Is Fuel - Student Edition (Human Biology) The Program in Human Biology, Stanford Univ- ersity, (HumBio) CK Editor Say Thanks to the Authors Click http://www.ck.org/saythanks (No sign in required)
Georgia Performance Standards Framework for Biology 9-12
 The following instructional plan is part of a GaDOE collection of Unit Frameworks, Performance Tasks, examples of Student Work, and Teacher Commentary. Many more GaDOE approved instructional plans are
The following instructional plan is part of a GaDOE collection of Unit Frameworks, Performance Tasks, examples of Student Work, and Teacher Commentary. Many more GaDOE approved instructional plans are
Investigating Osmosis By Amy Dewees,Jenkintown.High School and Dr. Ingrid Waldron, Department of Biology, University of Pennsylvania, 20091
 Investigating Osmosis By Amy Dewees,Jenkintown.High School and Dr. Ingrid Waldron, Department of Biology, University of Pennsylvania, 20091 What is diffusion? What does it mean to say that a membrane is
Investigating Osmosis By Amy Dewees,Jenkintown.High School and Dr. Ingrid Waldron, Department of Biology, University of Pennsylvania, 20091 What is diffusion? What does it mean to say that a membrane is
POPCORN MATH ACTIVITY BOOK. Group # Popcorn Type
 POPCORN MATH ACTIVITY BOOK Group # Popcorn Type TABLE OF CONTENTS Activity Sheet for L.E. #1 - Getting Started...3-4 Activity Sheet for L.E. #2 - Popped vs. Unpopped...5-6 Activity Sheet for L.E. #3 -
POPCORN MATH ACTIVITY BOOK Group # Popcorn Type TABLE OF CONTENTS Activity Sheet for L.E. #1 - Getting Started...3-4 Activity Sheet for L.E. #2 - Popped vs. Unpopped...5-6 Activity Sheet for L.E. #3 -
Lab #4: Nutrition & Assays for Detecting Biological Molecules - Introduction
 Lab #4: Nutrition & Assays for Detecting Biological Molecules - Introduction Most biological molecules fall into one of four varieties: proteins, carbohydrates, lipids and nucleic acids. These are sometimes
Lab #4: Nutrition & Assays for Detecting Biological Molecules - Introduction Most biological molecules fall into one of four varieties: proteins, carbohydrates, lipids and nucleic acids. These are sometimes
OBSERVING MITOSIS. Purpose. Preparing the cells. Interpreting what you see on your cell preparation. Salters-Nuffield Advanced Biology Resources
 Activity 3.9 Student Sheet OBSERVING MITOSIS Purpose To prepare some slides of actively dividing plant tissue. To observe the stages of the cell cycle in living tissue. To determine the duration of the
Activity 3.9 Student Sheet OBSERVING MITOSIS Purpose To prepare some slides of actively dividing plant tissue. To observe the stages of the cell cycle in living tissue. To determine the duration of the
Acting in an Emergency (Video- Acting in an emergency and preventing disease transmission)
 CHAPTER 2 Acting in an Emergency (Video- Acting in an emergency and preventing disease transmission) Lesson Objectives 1. Explain how bloodborne pathogens may be transmitted from an infected person to
CHAPTER 2 Acting in an Emergency (Video- Acting in an emergency and preventing disease transmission) Lesson Objectives 1. Explain how bloodborne pathogens may be transmitted from an infected person to
You should wear eye protection throughout this practical.
 Practical 8 - Broad bean root tip squash This practical focuses on setting up and manipulating apparatus and making and recording observations. Further skills can be developed using additional information
Practical 8 - Broad bean root tip squash This practical focuses on setting up and manipulating apparatus and making and recording observations. Further skills can be developed using additional information
Safety Data Sheet Product Name: Heparin / PF4 Serum Panel - For In Vitro Diagnostic Use Only
 Section 1 Identification Product Name Catalog Numbers Heparin / PF4 Assayed Antibody Serum Panel 4036027, 2 Member QC Panel 4036028, Multi-Member Qualification Panel (6 member) Company Identification AKERS
Section 1 Identification Product Name Catalog Numbers Heparin / PF4 Assayed Antibody Serum Panel 4036027, 2 Member QC Panel 4036028, Multi-Member Qualification Panel (6 member) Company Identification AKERS
Naval Support Activity Monterey / Naval Postgraduate School
 Background: Bloodborne pathogens are viruses present in human blood and body fluids that can cause disease in humans. Diseases like the hepatitis B virus (HBV), human immunodeficiency virus (HIV) and others
Background: Bloodborne pathogens are viruses present in human blood and body fluids that can cause disease in humans. Diseases like the hepatitis B virus (HBV), human immunodeficiency virus (HIV) and others
Material Safety Data Sheet SunClean Concrete Tile Cleaner
 1. Production Description Product Name: Concrete Tile Cleaner Application of the substance: Cleaner Acid Based Manufacturer/Supplier: Sun Clean Cleaning Ind. LLC P.O. Box 41475, SHARJAH UAE Tel: +971 (6)
1. Production Description Product Name: Concrete Tile Cleaner Application of the substance: Cleaner Acid Based Manufacturer/Supplier: Sun Clean Cleaning Ind. LLC P.O. Box 41475, SHARJAH UAE Tel: +971 (6)
MATERIAL SAFETY DATA SHEET
 Product Name: Catalog Number: 1003 Company: Sedia HIV-1 LAg-Avidity EIA for Dried Blood Spots Sedia Biosciences Corp. 4900 NE 122 nd Ave. Email: Phone: +1-503-459-4159 Fax: +1-503-459-4168 customerservice@sediabio.com
Product Name: Catalog Number: 1003 Company: Sedia HIV-1 LAg-Avidity EIA for Dried Blood Spots Sedia Biosciences Corp. 4900 NE 122 nd Ave. Email: Phone: +1-503-459-4159 Fax: +1-503-459-4168 customerservice@sediabio.com
SAFETY DATA SHEET BWT-27
 1. Product and Manufacturer Identification Product Name: Manufacturer: Apollo Technologies, Inc. 31441 Santa Margarita Parkway, Suite A-219 Rancho Santa Margarita, Ca. 92688 For more information call:
1. Product and Manufacturer Identification Product Name: Manufacturer: Apollo Technologies, Inc. 31441 Santa Margarita Parkway, Suite A-219 Rancho Santa Margarita, Ca. 92688 For more information call:
Patient/Carer instructions for the administration of Subcutaneous Cytarabine
 Patient/Carer instructions for the administration of Subcutaneous Cytarabine This document covers the following information: What cytarabine is What subcutaneous means What happens if you decide to inject
Patient/Carer instructions for the administration of Subcutaneous Cytarabine This document covers the following information: What cytarabine is What subcutaneous means What happens if you decide to inject
