Current Infectious Disease Screening for the Live Organ Donor
|
|
|
- Horace Armstrong
- 6 years ago
- Views:
Transcription
1 2013 Public Health and Safety Guidelines for reducing HIV, HBV and HCV Transmission Though Organ Transplantation: Implications for Counseling and Disclosure Dianne LaPointe Rudow DNP, ANP-BC, CCTC Director of The Zweig Family Center for Living Donation Associate Professor of Health Evidence and Policy Recanati Miller Transplantation Institute Mount Sinai Medical Center New York, New York
2 Objectives Discuss what the new Public Health and Safety Guidelines for reducing HIV, HBV and HCV Transmission Though Organ Transplantation mean for the donor evaluation Describe the elements of PHS screening Identify what and when testing needs to be disclosed Describe best strategies for donor counseling on PHS guidelines
3 Current Infectious Disease Screening for the Live Organ Donor CMV (Cytomegalovirus) Antibody EBV (Epstein Barr Virus) Antibody HIV 1,2 (Human Immunodeficiency Virus) antibody testing HepBsAg (Hepatitis B surface antigen) HepBcAB (Hepatitis B core antibody) HepBsAB (Hepatitis B surface antibody) recommend removal HCV (Hepatitis C Virus) antibody testing Syphilis testing TB screening Testing for diseases prevalent in endemic areas( Chagas, Strongyloides, West Nile) Infectious disease risk assessment Should be at least as comprehensive as screening of a deceased donor Utilize FDA approved tests when available NAT testing considered
4 What are the PHS Guidelines? On June 19,2013 the PHS Guidelines for reducing HIV,HBV and HCV through organ transplantation was released by HRSA, CDC It supersedes the 1994 PHS Guideline for preventing HIV transmission through transplant ( Old CDC High risk criteria) Expands old guidelines to include HBV and HCV Uses updated factors known to be associated with increased likelihood of transmitting infection Limits focus to organs and blood vessel conduits recovered for transplant not human cell and tissue products Related to adult and pediatric donors, living and deceased as well as transplant candidates and recipients Does NOT assess infection beyond HIV, HBV, and HCV Goal: to provide Guidance to OPO personnel, transplant centers ( MDs, RNs, administrators, lab personnel,) and those developing and implementing infection control programs Input was sought from Public and private health professionals over a few years
5 Methodology and Reporting PHS Guideline for Reducing Human Immunodeficiency Virus, Hepatitis B Virus, and Hepatitis C Virus Transmission Through Organ Transplantation rg/issueopen.cfm?articleid=2975
6 Areas of Questions for Systematic Review
7 Areas of focus for the Guidelines Risk factors for recent infection Risk assessment ( screening) of live (LD) and deceased donors (DD) Testing of LD and DD Informed consent discussions with transplant candidates Testing of recipients pre and post transplant Collection and storage of donor and recipient specimens Tracking and Reporting of HIV,HBV,HCV transmissions Recommendations for further study
8 Behavioral and non behavioral risk factors varying degree of risk
9 Screening Guidelines 1994 Guidelines 2013 Guideline Donors who meet one or more of the following 11 criteria should be identified as being at increased risk for recent HIV, HBV, and HCV infections. Each factor listed reflects increased risk of all 3 pathogens as Behavior/History Exclusionary Criteria an aggregate, as there is overlap of associated risk, even though each factor does not convey risk from all pathogens equally. The first six factors address sexual contact; the definition of had sex refers to any method of sexual contact, including vaginal, anal and oral contact: #5- Persons who have had sex in the preceding 12 months with any People who have had sex with a person known or suspected to have HIV, person described in items 1-4 or with a person known or suspected to HBV, or HCV infections in the preceding 12 months have HIV infection #1- Men who have had sex with another man in the preceding 5 years MSM in the preceding 12 months #5- Persons who have had sex in the preceding 12 months with any person described in items 1-4 or with a person known or suspected to have HIV infection #4- Men and women who have engaged in sex in exchange for money or drugs in the preceding 5 years #5- Persons who have had sex in the preceding 12 months with any person described in items 1-4 or with a person known or suspected to have HIV infection #5- Persons who have had sex in the preceding 12 months with any person described in items 1-4 or with a person known or suspected to have HIV infection Specific Exclusionary Criteria for Ped Donors #2- Children born to mothers with HIV infection or mothers who meet the behavioral or laboratory exclusionary criteria for adult donors (regardless of HIV status) should not be accepted as donors unless HIV infection can be definitely excluded in the child as follows: Children >18 months of age who are born to mothers with or at risk for HIV infection, who have not been breastfed within the last 12 months, and whose HIV Ab tests, physical exam, and review of medical records don not indicate evidence of HIV infection can be accepted as donors Women who have had sex with a man with a history of MSM behavior in the preceding 12 months People who have had sex in exchange for money or drugs in the preceding 12 months People who have had sex with a person who had sex in exchange for money or drugs in the preceding 12 months People who have had sex with a person that has injected drugs by IV, IM, or subq route for nonmedical reasons in the preceding 12 months A child who is <18 months of age and born to a mother known to be infected with, or at increased risk for HIV, HBV, or HCV infections
10 Screening Guidelines Specific Exclusionary Criteria for Ped Donors #3- Children <18 months of age who are born to mothers with or at risk for HIV infection or who have been breastfed within the past 12 months should not be accepted as donors regardless of their HIV status A child who has been breastfed within the preceding 12 months and the mother is known to be infected with, or at increased risk for HIV infection (Recommendation 4) #2- Persons who report nonmedical IV, IM, or subq injection of drugs in the preceding 5 years #7- Inmates of correctional systems. (This exclusion is to address issues such as difficulties with informed consent and increased prevalence of HIV in this population.) Laboratory and Other Medical Exclusionary Criteria #3- Persons whose history, physical exam, medical records, or autopsy reports reveal other evidence of HIV infection or high-risk behavior, such as a diagnosis of AIDS, unexplained weight loss, night sweats, blue or purple spots on the skin or mucous membranes typical of Kaposi s Sarcoma, unexplained lymphadenopathy lasting > 1 month, unexplained temperature >100.5 F (38.6 C) for >10 days, unexplained persistent cough, MSM contact, sexually transmitted diseases, or needle tracks or other signs of parenteral drug abuse. NEW #3-Persons with hemophilia or related clotting disorders who have received human-derived clotting factor concentrates #6- Person who have been exposed in the preceding 12 months to known or suspected HIV-infected blood through percutaneous inoculation or through contact with an open wound, nonintact skin, or mucous membrane. Laboratory and Other Medical Exclusionary Criteria #1- Persons who cannot be tested for HIV infection because of refusal, inadequate blood samples (e.g. hemodilution that could result in a false negative test), or any other reasons #2- Persons with a repeatedly reactive screening assay for HIV-1 or HIV-2 Ab regardless of the result of supplemental assays. People who have injected drugs by IV, IM, or subq route for nonmedical reasons in the preceding 12 months People who have been in lockup, jail, prison, or a juvenile correctional facility for more than 72 hours in the preceding 12 months People who have been newly diagnosed with or have been treated for syphilis, gonorrhea, Chlamydia, or genital ulcers in the preceding 12 months Donors who meet the following criterion should be identified as being at increased risk for recent HCV infection only: People who have been on hemodialysis in the preceding 12 months Not addressed specifically in 2013 med-soc criteria Not addressed specifically in 2013 med-soc criteria Hemodilution specifically addressed in Recommendation 5b
11 Assessment of Live Donors 1. Have you had sex with a person known or suspected to have HIV, HBV, or HCV infection in the preceding 12 months? Yes No 2. Are you a Man who has had sex with men (MSM) in the preceding 12 months? Yes No 3. Are you a woman who has had sex with a man with a history of MSM behavior in the preceding 12 months? Yes No 4. Have you sex in exchange for money or drugs in the preceding 12 months? Yes No 5. Have you had sex with a person who had sex in exchange for money or drugs in the preceding 12 months? Yes No 6. Have you had sex with a person who injected drugs by intravenous, intramuscular, or subcutaneous route for nonmedical reasons in the preceding 12 months? Yes No 7. Have injected drugs by intravenous, intramuscular, or subcutaneous route for nonmedical reasons in the preceding 12 months? Yes No 8. Have you been in lockup, jail, prison, or a juvenile correctional facility for more than 72 consecutive hours in the preceding 12 months? Yes No 9. Have you been newly diagnosed with, or have been treated for, syphilis, gonorrhea, Chlamydia, or genital ulcers in the preceding 12 months? Yes No
12 Individual Programmatic Approach to Assessment The donor evaluation is invasive on multiple levels PHS increased risk screening is part of the psychosocial evaluation PHS increased risk screening is part of the medical evaluation Developing a relationship with the donor is critical for a truthful bi-directional interaction Documentation in the medical record is required and audited by program surveyors Keeping records confidential is important Approach varies Define SEX Interview Questionnaires and discussion Signed document attesting to low risk Method to consent to disclose behavior Risk assessment determines testing
13 Recommended Testing for Donor
14 Recommended Testing for Recipient of PHS Increased Risk Donor
15 Implication for Policy Changes PHS document are guidelines The Final Rule notes that the OPTN Board of Directors is responsible for developing policies that are consistent with recommendations of the Centers for Disease Control and Prevention (CDC) to test potential organ donors and following transplant recipients to prevent the spread of infectious disease. Currently, policy requirements don t fully align with the new Guideline s recommendations OPTN Task force (Living Donor Committee,OPO Committee, Operations and Safety Committee, four professional societies and DTAC) developed proposal out for public comment to modify some existing policy language and also create new policies to reflect recommendations outlined in PHS document. Goal: to promote transplant patient safety, increase capacity to identify patient safety issues, and ultimately, increase the number of transplants by improving donor screening requirements.
16 OPTN: policy addresses: All potential LDs should be informed of the donor evaluation process, including the review of medical and behavioral history, physical examination, and laboratory tests to identify the presence of infectious agents or medical conditions that could be transmitted by organ transplantation. LD should be interviewed in a confidential manner about behaviors that may have increased the potential donor s probability of having HIV, HBV, or HCV infection. LDs with behaviors associated with an increased risk of acquiring HIV, HBV, or HCV identified during evaluation should receive individualized counseling on specific strategies to prevent exposure to these viruses during the time period prior to surgery. All living potential donors should be tested for HIV, HBV, and HCV as close as possible to the date of the organ recovery operation, but at least within the 28-day time period prior to surgery. When organs from HBV or HCV infected donors will be used, the transplant team care should have an informed consent discussion with the transplant candidate, or medical decision maker, prior to transplantation regarding the risks related to disease transmission. (This includes HBV core positive) Post transplant testing of recipients should be conducted when the live donor is identified as being at increased risk for HCV infection When a living LD recovery center receives information before organ recovery that a potential LD is infected with HIV, HBV, or HCV, the living donor recovery center should notify the transplant center intended to receive the organ, public health authorities,the OPTN and the LD LD should be given the option to OPT out prior to disclosure to recipient and OPTN Role for Physicians, Nurse, Psychosocial Personnel and the ILDA
17 Implications for Donor Education, Testing and Counseling Donors need to be assessed for increased risk Difficult to discuss Honesty Interventions to minimize future risk Testing Multiple visits False positive results Testing errors Disclosure if you can perform NAT testing must you disclose? Does recipient rights take precedence over donor Relationship What impact will disclosure have on the donor recipient relationship Is there ever a time when disclosure is more risky that to disclose Options available to preserve relationships Will this result in less donors willing to donate
18 Case Studies 26 year old male whose father is in need of a kidney transplant. He is medically cleared. Social history; Single homosexual male in a monogamous relationship. He has not disclosed to his parents his sexual orientation Are you a Man who has had sex with men (MSM) in the preceding 12 months? Yes Implications: Testing Counseling Disclosure Abstinence 37 year old female who wants to donate to her sister. Medically cleared for donation Social history; married for 12 years has two children works as a secretary Spouse is a salesman who travels two weeks out of the month, their relationship has been strained at this time. Team notes spouse to be quite flirtatious. PHS Questions 1-9 No Implications: Testing Counseling Disclosure Abstinence
19 Resource available on OPTN website
20 Summary PHS Guidelines are available to assist transplant programs to determine who is at increased risk for HIV,HBV,HCV Risk assessment and testing of the donor is critical for donor safety and reduction of transmission of disease Live donor teams need to be mindful of the confidentiality and disclosure issues that can affect the donor should they be determine to be at increased risk
21
Hot Topics in Tissue Safety
 Hot Topics in Tissue Safety 37 th Annual AATB Meeting National Harbor, Maryland 03 October 2013 Scott A. Brubaker, CTBS Chief Policy Officer PHS Guideline for Reducing Human Immunodeficiency Virus, Hepatitis
Hot Topics in Tissue Safety 37 th Annual AATB Meeting National Harbor, Maryland 03 October 2013 Scott A. Brubaker, CTBS Chief Policy Officer PHS Guideline for Reducing Human Immunodeficiency Virus, Hepatitis
Patient Name: MRN: DOB: Treatment Location:
 Page 1 of 5 I. TO (Required) This Section is required to be completed by all patients who undergo kidney transplant surgery. I hereby consent to and authorize Dr. and his/her assistant(s), including supervised
Page 1 of 5 I. TO (Required) This Section is required to be completed by all patients who undergo kidney transplant surgery. I hereby consent to and authorize Dr. and his/her assistant(s), including supervised
Name of Recipient: Recipient s DOB (if known) Relationship to Recipient: (Example: mother, father, sister, brother, friend, etc)
 The Christ Hospital Health Network DONOR REGISTRATION INFORMATION Phone: 513-585-2493 Fax: 513-585-0433 (Please be advised donor information is needed ONLY to register donor in the Christ Hospital system.
The Christ Hospital Health Network DONOR REGISTRATION INFORMATION Phone: 513-585-2493 Fax: 513-585-0433 (Please be advised donor information is needed ONLY to register donor in the Christ Hospital system.
Risky Business Preventing Disease Transmission From Donor Organs. Peter Chin-Hong, MD UCSF September 2012
 Risky Business Preventing Disease Transmission From Donor Organs Peter Chin-Hong, MD UCSF September 2012 Case 1: Something rare? 54 yo WM with HBV/HCV/HCC Day 5: Fever to 102.4, mild frontal HA since time
Risky Business Preventing Disease Transmission From Donor Organs Peter Chin-Hong, MD UCSF September 2012 Case 1: Something rare? 54 yo WM with HBV/HCV/HCC Day 5: Fever to 102.4, mild frontal HA since time
MONTEFIORE MEDICAL CENTER TRANSPLANT PROGRAM LIVING DONOR EVALUATION FORM History Questionnaire
 MONTEFIORE MEDICAL CENTER TRANSPLANT PROGRAM LIVING DONOR EVALUATION FORM History Questionnaire Donor s Name: Today s Date: Social Security #: Date of Birth Age Sex Address: Telephone #: (home) (work)
MONTEFIORE MEDICAL CENTER TRANSPLANT PROGRAM LIVING DONOR EVALUATION FORM History Questionnaire Donor s Name: Today s Date: Social Security #: Date of Birth Age Sex Address: Telephone #: (home) (work)
Increased Risk Donors for Organ Transplantation
 A Tool Kit to Assist Transplant Programs in the Use of Increased Risk Donors for Organ Transplantation February 2016 Table of Contents Table of Contents... 2 1.0 Increased Risk Donor Tool Kit... 3 1.1
A Tool Kit to Assist Transplant Programs in the Use of Increased Risk Donors for Organ Transplantation February 2016 Table of Contents Table of Contents... 2 1.0 Increased Risk Donor Tool Kit... 3 1.1
Directed Semen Donor Eligibility Requirements
 Directed Semen Donor Eligibility Requirements Directed (known) reproductive donors using the University Andrology Sperm Banking Program must follow FDA requirements to prevent the transmission of relevant
Directed Semen Donor Eligibility Requirements Directed (known) reproductive donors using the University Andrology Sperm Banking Program must follow FDA requirements to prevent the transmission of relevant
Infectious Disease Considerations in Organ Donation. Nikole A Neidlinger MD FACS Chief Medical Officer
 Infectious Disease Considerations in Organ Donation Nikole A Neidlinger MD FACS Chief Medical Officer Screening Donors and Infectious Disease considerations: Deceased Donor Screening Donors at increased
Infectious Disease Considerations in Organ Donation Nikole A Neidlinger MD FACS Chief Medical Officer Screening Donors and Infectious Disease considerations: Deceased Donor Screening Donors at increased
HEALTH SCREENING QUESTIONNAIRE. Work-up. Donor ID EdgeCell #:
 HEALTH SCREENING QUESTIONNAIRE Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca CT
HEALTH SCREENING QUESTIONNAIRE Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca CT
RISK OF DISEASE TRANSMISSION FROM ORGAN DONORS PATIENT INFORMATION GUIDE
 Receiving an organ transplant carries many risks, including the risk of getting a disease from the June 2017 donor. This is true for every organ we transplant. BC Transplant makes every effort to minimize
Receiving an organ transplant carries many risks, including the risk of getting a disease from the June 2017 donor. This is true for every organ we transplant. BC Transplant makes every effort to minimize
The DRAI (Donors >12 yrs old) versus Requirements
 Q# or Pg # Pg 1 Question or section as it appears on the DRAI questionnaire (Donors >12 yrs old) 5-7-12 Donor Name, Person Interviewed (name, relationship), Contact Information (phone, address, city, state,
Q# or Pg # Pg 1 Question or section as it appears on the DRAI questionnaire (Donors >12 yrs old) 5-7-12 Donor Name, Person Interviewed (name, relationship), Contact Information (phone, address, city, state,
[Submitted Electronically]
![[Submitted Electronically] [Submitted Electronically]](/thumbs/82/85246007.jpg) [Submitted Electronically] Dr. Matthew J. Kuehnert, Director Office of Blood, Organ, and Other Tissue Safety Division of Healthcare Quality Promotion National Center for Emerging and Zoonotic Infectious
[Submitted Electronically] Dr. Matthew J. Kuehnert, Director Office of Blood, Organ, and Other Tissue Safety Division of Healthcare Quality Promotion National Center for Emerging and Zoonotic Infectious
HEALTH SCREENING QUESTIONNAIRE CT Work-up. Donor ID EdgeCell #:
 HEALTH SCREENING QUESTIONNAIRE CT Work-up Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca
HEALTH SCREENING QUESTIONNAIRE CT Work-up Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca
Ask at Least Annually. Ask Older Adults. Have you been sexually active in the last year? Have you ever been sexually active?
 Essential Sexual Health Questions to Ask Adults Ask all of your adult patients the sexual health questions on this card. They will help you assess the patient s level of sexual risk and determine whether
Essential Sexual Health Questions to Ask Adults Ask all of your adult patients the sexual health questions on this card. They will help you assess the patient s level of sexual risk and determine whether
Improving Utilization of Organs from Increased Risk Donors. Matthew J. Kuehnert, MD CDC
 Improving Utilization of Organs from Increased Risk Donors Matthew J. Kuehnert, MD CDC Conflict of Interest Disclosure I have no relevant financial relationships to disclose. 2016 AST Improving Utilization
Improving Utilization of Organs from Increased Risk Donors Matthew J. Kuehnert, MD CDC Conflict of Interest Disclosure I have no relevant financial relationships to disclose. 2016 AST Improving Utilization
2017 CST-Astellas Canadian Transplant Fellows Symposium. Optimizing use of organs from Increased Risk Donors
 2017 CST-Astellas Canadian Transplant Fellows Symposium Optimizing use of organs from Increased Risk Donors Atual Humar, MD Atul Humar is a Professor in the Department of Medicine, University of Toronto.
2017 CST-Astellas Canadian Transplant Fellows Symposium Optimizing use of organs from Increased Risk Donors Atual Humar, MD Atul Humar is a Professor in the Department of Medicine, University of Toronto.
Achieving Consensus on Increased Risk Donors to Improve Access to Organ Transplantation Executive Summary
 Achieving Consensus on Increased Risk Donors to Improve Access to Organ Transplantation Executive Summary Introduction To address the current shortage of organs for transplantation, (114,651 candidates
Achieving Consensus on Increased Risk Donors to Improve Access to Organ Transplantation Executive Summary Introduction To address the current shortage of organs for transplantation, (114,651 candidates
Are Your Patients Well Prepared for Kidney Transplant?
 Are Your Patients Well Prepared for Kidney Transplant? Venkatesh Kumar Ariyamuthu MD FASN Disclosures National Kidney Foundation Scientific Advisory Board 78 year old Hispanic female ESRD secondary to
Are Your Patients Well Prepared for Kidney Transplant? Venkatesh Kumar Ariyamuthu MD FASN Disclosures National Kidney Foundation Scientific Advisory Board 78 year old Hispanic female ESRD secondary to
DHQ v2.0, February 2016 Table Detailing Changes from v1.3 v1.3 Revised language in v2.0 Rationale Question 1: Are you
 DHQ v2.0, February 2016 Table Detailing s from v1.3 v1.3 Revised language in v2.0 Rationale Question 1: Are you Q 1, No Feeling healthy and well today? Question 2: Are you Q 2, No change Currently taking
DHQ v2.0, February 2016 Table Detailing s from v1.3 v1.3 Revised language in v2.0 Rationale Question 1: Are you Q 1, No Feeling healthy and well today? Question 2: Are you Q 2, No change Currently taking
Hepatitis C Best Practice Guidelines For Local Health Departments
 Hepatitis C Best Practice Guidelines For Local Health Departments LHDs are responsible for investigating and reporting all physician reported cases of acute hepatitis C (HCV). For clients known to have
Hepatitis C Best Practice Guidelines For Local Health Departments LHDs are responsible for investigating and reporting all physician reported cases of acute hepatitis C (HCV). For clients known to have
Increased Risk Donors. November 1, 2018 BC Kidney Days. Jagbir Gill MD MPH Associate Professor of Medicine UBC
 Increased Risk Donors November 1, 2018 BC Kidney Days Jagbir Gill MD MPH Associate Professor of Medicine UBC Median dialysis exposure prior to DDKT Case scenarios Which donors should we accept kidneys
Increased Risk Donors November 1, 2018 BC Kidney Days Jagbir Gill MD MPH Associate Professor of Medicine UBC Median dialysis exposure prior to DDKT Case scenarios Which donors should we accept kidneys
Question: 1. Are you currently taking an antibiotic?
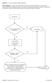 Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Hepatitis STARS Program. Geri Brown, M.D. Associate Professor Department of Internal Medicine October 4, 2003
 Hepatitis 2003 STARS Program Geri Brown, M.D. Associate Professor Department of Internal Medicine October 4, 2003 Outline n Hepatitis A Epidemiology and screening Transmission n Hepatitis B Epidemiology
Hepatitis 2003 STARS Program Geri Brown, M.D. Associate Professor Department of Internal Medicine October 4, 2003 Outline n Hepatitis A Epidemiology and screening Transmission n Hepatitis B Epidemiology
Donor Risk Assessment Interview (Donor >12 yrs old)
 Your logo Donor Risk Assessment Interview (Donor >12 yrs old) Your address Donor Name: First Middle Last Person Interviewed: Name Relationship Contact Information: ( ) Phone Address City State Zip The
Your logo Donor Risk Assessment Interview (Donor >12 yrs old) Your address Donor Name: First Middle Last Person Interviewed: Name Relationship Contact Information: ( ) Phone Address City State Zip The
Practice Steps for Implementation of Guidelines Recommendations The guideline recommendations are shown schematically -
 ASK SCREEN Test for HIV and STI Practice Steps for Implementation of Guidelines Recommendations The guideline recommendations are shown schematically - Routinely obtain a thorough sexual history from all
ASK SCREEN Test for HIV and STI Practice Steps for Implementation of Guidelines Recommendations The guideline recommendations are shown schematically - Routinely obtain a thorough sexual history from all
December 20, Dear Dr. Frieden:
 December 20, 2011 The Honorable Thomas Frieden, MD, MPH Director, Centers for Disease Control and Prevention 1600 Clifton Road, NE. Mailstop A-07 Atlanta, GA 30329 Ref: [Docket No. CDC-2011-0011]; Public
December 20, 2011 The Honorable Thomas Frieden, MD, MPH Director, Centers for Disease Control and Prevention 1600 Clifton Road, NE. Mailstop A-07 Atlanta, GA 30329 Ref: [Docket No. CDC-2011-0011]; Public
PHSKC HIV Testing Survey: Knowledge, Attitudes and Practices
 PHSKC HIV Testing Survey: Knowledge, Attitudes and Practices Page One This anonymous survey is intended to collect information about HIV testing attitudes and practices. Results will be used by Public
PHSKC HIV Testing Survey: Knowledge, Attitudes and Practices Page One This anonymous survey is intended to collect information about HIV testing attitudes and practices. Results will be used by Public
ALASKA NATIVE MEDICAL CENTER SEXUALLY TRANSMITTED DISEASE SCREENING AND TREATMENT GUIDELINES
 ALASKA NATIVE MEDICAL CENTER SEXUALLY TRANSMITTED DISEASE SCREENING AND TREATMENT GUIDELINES A. Screening Page Chlamydia and Gonorrhea 1 HIV 1 Syphilis 1 Genital Herpes 2 Hepatitis A 2 Hepatitis B 2 Hepatitis
ALASKA NATIVE MEDICAL CENTER SEXUALLY TRANSMITTED DISEASE SCREENING AND TREATMENT GUIDELINES A. Screening Page Chlamydia and Gonorrhea 1 HIV 1 Syphilis 1 Genital Herpes 2 Hepatitis A 2 Hepatitis B 2 Hepatitis
Uniform Donor Risk Assessment Interview (Donor >12 yrs old)
 Your logo Uniform Donor Risk Assessment Interview (Donor >12 yrs old) Your address Donor Name: First Middle Last Person Interviewed: Name Relationship Contact Information: ( ) Phone Address City State
Your logo Uniform Donor Risk Assessment Interview (Donor >12 yrs old) Your address Donor Name: First Middle Last Person Interviewed: Name Relationship Contact Information: ( ) Phone Address City State
Ad Hoc Disease Transmission Advisory Committee Report
 Ad Hoc Disease Transmission Advisory Committee Report OPTN/UNOS Board of Directors Meeting Dr. Michael Green, MD, MPH, Chair Dr. Daniel Kaul, MD, Vice Chair June 23-24, 2014 PHS Guideline for Reducing
Ad Hoc Disease Transmission Advisory Committee Report OPTN/UNOS Board of Directors Meeting Dr. Michael Green, MD, MPH, Chair Dr. Daniel Kaul, MD, Vice Chair June 23-24, 2014 PHS Guideline for Reducing
ABO: Tissue#:
 Donor Name: Place of Interview: - Date-Time Interviewed: --/--/ --:-- Person Conducting Interview and Completing Form: - Person Interviewed: - Relationship to Potential Donor: - Address: - Phone: - Phone
Donor Name: Place of Interview: - Date-Time Interviewed: --/--/ --:-- Person Conducting Interview and Completing Form: - Person Interviewed: - Relationship to Potential Donor: - Address: - Phone: - Phone
Uniform DRAI - Donor >12 yrs old Requirements Crosswalk
 Q# or Pg # Pg 1 Question or section as it appears on the Uniform DRAI - Donor >12 yrs old 9-10-14 Donor Name, Person Interviewed (name, relationship), Contact Information (phone, address, city, state,
Q# or Pg # Pg 1 Question or section as it appears on the Uniform DRAI - Donor >12 yrs old 9-10-14 Donor Name, Person Interviewed (name, relationship), Contact Information (phone, address, city, state,
FORM TITLE: Risk Behavior Assessment. 1. Are you a male who has had sex with another male in the preceding five years?
 RELESE DTE: 12/07/2012 EFFECTIVE DTE: 12/13/2012 1. re you a male who has had sex with another male in the preceding five years? 2. Have you injected drugs for non-medical reason in the preceding five
RELESE DTE: 12/07/2012 EFFECTIVE DTE: 12/13/2012 1. re you a male who has had sex with another male in the preceding five years? 2. Have you injected drugs for non-medical reason in the preceding five
HIV AIDS and Other Infectious Diseases
 HIV AIDS and Other Infectious Diseases Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Chapter 1 - Introduction Despite the availability of a vaccine since
HIV AIDS and Other Infectious Diseases Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Chapter 1 - Introduction Despite the availability of a vaccine since
Billing and Coding for HIV Services
 Billing and Coding for HIV Services Financial Disclosure This speaker does not have any financial relationships with commercial entities to disclose. This speaker will not discuss any off-label use or
Billing and Coding for HIV Services Financial Disclosure This speaker does not have any financial relationships with commercial entities to disclose. This speaker will not discuss any off-label use or
Risk of Other Donor-Derived Infections (nonhiv, nonhcv) Daniel Kaul MD Associate Professor University of Michigan
 Risk of Other Donor-Derived Infections (nonhiv, nonhcv) Daniel Kaul MD Associate Professor University of Michigan Conflict of Interest Disclosure I have no relevant financial relationships to disclose
Risk of Other Donor-Derived Infections (nonhiv, nonhcv) Daniel Kaul MD Associate Professor University of Michigan Conflict of Interest Disclosure I have no relevant financial relationships to disclose
Question: 1. Are you currently taking an antibiotic?
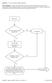 Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Public Health, Infections and Transplantation
 Public Health, Infections and Transplantation Dr Kerry Chant Chief Health Officer & Deputy Director General Population and Public Health NSW Ministry of Health May2014 Public Health Infection and Transplantation
Public Health, Infections and Transplantation Dr Kerry Chant Chief Health Officer & Deputy Director General Population and Public Health NSW Ministry of Health May2014 Public Health Infection and Transplantation
Sexually Transmi/ed Diseases
 Sexually Transmi/ed Diseases Chapter Fourteen 2013 McGraw-Hill Higher Education. All rights reserved. Also known as sexually transmitted infections The Major STDs (STIs) HIV/AIDS Chlamydia Gonorrhea Human
Sexually Transmi/ed Diseases Chapter Fourteen 2013 McGraw-Hill Higher Education. All rights reserved. Also known as sexually transmitted infections The Major STDs (STIs) HIV/AIDS Chlamydia Gonorrhea Human
COMMUNICABLE DISEASE REPORT
 June 2011 Department of Health and Community Services Government of Newfoundland and Labrador COMMUNICABLE DISEASE REPORT Reporting Sexually Transmitted and Bloodborne Infections All laboratory-confirmed
June 2011 Department of Health and Community Services Government of Newfoundland and Labrador COMMUNICABLE DISEASE REPORT Reporting Sexually Transmitted and Bloodborne Infections All laboratory-confirmed
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Tested pathogen Donor test/ technique
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Tested pathogen Donor test/ technique
Organ Procurement and Transplantation Network
 OPTN Organ Procurement and Transplantation Network POLICIES This document provides the policy language approved by the OPTN/UNOS Board at its meeting in June 2015 as part of the Operations and Safety Committee
OPTN Organ Procurement and Transplantation Network POLICIES This document provides the policy language approved by the OPTN/UNOS Board at its meeting in June 2015 as part of the Operations and Safety Committee
IMPORTANT HEALTH INFORMATION
 IMPORTANT HEALTH INFORMATION SU-6523MI Page 1 of 8 Table of Contents Page What is an HIV test?..........................................1 Will the HIV test tell me if I have AIDS?............................1
IMPORTANT HEALTH INFORMATION SU-6523MI Page 1 of 8 Table of Contents Page What is an HIV test?..........................................1 Will the HIV test tell me if I have AIDS?............................1
TRANSMISSIBLE SPONGIFORM ENCEPHALOPATHIES ADVISORY COMMITTEE MEETING October2010. Issue Summary
 TRANSMISSIBLE SPONGIFORM ENCEPHALOPATHIES ADVISORY COMMITTEE MEETING 28-29October2010 Issue Summary Informational Topic: FDA s Geographic Donor Deferral Policy to Reduce the Possible Risk of Transmission
TRANSMISSIBLE SPONGIFORM ENCEPHALOPATHIES ADVISORY COMMITTEE MEETING 28-29October2010 Issue Summary Informational Topic: FDA s Geographic Donor Deferral Policy to Reduce the Possible Risk of Transmission
DHQ Flowcharts v2.0 eff. February 2016
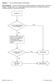 Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
SUBJECT: Hepatitis C Virus (HCV) Counseling/Education, Testing, Referral, and Partner Notification
 SUBJECT: Hepatitis C Virus (HCV) Counseling/Education, Testing, Referral, and Partner Notification Hepatitis C virus (HCV) infection is the most common chronic bloodborne infection in the United States.
SUBJECT: Hepatitis C Virus (HCV) Counseling/Education, Testing, Referral, and Partner Notification Hepatitis C virus (HCV) infection is the most common chronic bloodborne infection in the United States.
BAY-ARENAC BEHAVIORAL HEALTH AUTHORITY POLICIES AND PROCEDURES MANUAL
 Page: 1 of 8 Policy Bay-Arenac Behavioral Health Authority (BABHA) is committed to high standards of employee and consumer safety practices. This standard will include the prevention, surveillance, identification
Page: 1 of 8 Policy Bay-Arenac Behavioral Health Authority (BABHA) is committed to high standards of employee and consumer safety practices. This standard will include the prevention, surveillance, identification
Hepatitis Case Investigation
 * indicates required fields Does patient also have: Hepatitis Case Investigation West Virginia Electronic Disease Surveillance System Division of Surveillance and Disease Control Infectious Disease Epidemiology
* indicates required fields Does patient also have: Hepatitis Case Investigation West Virginia Electronic Disease Surveillance System Division of Surveillance and Disease Control Infectious Disease Epidemiology
No Wrong Door Integrative Screener 1.0 Feedback Sheet 01/14/15
 Name: Derek S. ID: Date: DOB: 07/10/95 Age: 19 Agency: 01/14/15 Demographic Information Age (in years): Country of Origin: Primary Language: Length of Time Living in US (if not born in US): Sex Assigned
Name: Derek S. ID: Date: DOB: 07/10/95 Age: 19 Agency: 01/14/15 Demographic Information Age (in years): Country of Origin: Primary Language: Length of Time Living in US (if not born in US): Sex Assigned
HEPATITIS C, ACUTE CRUDE DATA. Number of Cases 5 Annual Incidence a LA County 0.05 California b 0.10 United States b 0.68 Age at Diagnosis Mean 38
 2016 Annual Morbidity Report HEPATITIS C, ACUTE a Rates calculated based on less than 19 cases or events are considered unreliable b Calculated from: CDC. Notice to Readers: Final 2016 Reports of Nationally
2016 Annual Morbidity Report HEPATITIS C, ACUTE a Rates calculated based on less than 19 cases or events are considered unreliable b Calculated from: CDC. Notice to Readers: Final 2016 Reports of Nationally
CORD BLOOD TRANSPLANTATION STUDY CBU EXCLUSION AND QUARANTINE RELEASE FORM
 (Any answer of Yes indicates exclusion) 1. Informed Consent and Maternal/Infant Demographics a. Lack of confirmation of signed and witnessed informed consent 1 Yes 2 No Is the following information missing
(Any answer of Yes indicates exclusion) 1. Informed Consent and Maternal/Infant Demographics a. Lack of confirmation of signed and witnessed informed consent 1 Yes 2 No Is the following information missing
New Brunswick Report on Sexually Transmitted and Blood Borne Infections, 2016
 New Brunswick Report on Sexually Transmitted and Blood Borne Infections, 6 Table of Contents. Introduction.... Methodology... 3. Data Limitations.... Definitions used... 3 5. Overview of STBBI epidemiology
New Brunswick Report on Sexually Transmitted and Blood Borne Infections, 6 Table of Contents. Introduction.... Methodology... 3. Data Limitations.... Definitions used... 3 5. Overview of STBBI epidemiology
2.0 MINIMUM PROCUREMENT STANDARDS FOR AN ORGAN PROCUREMENT ORGANIZATION (OPO)
 2.0 MINIMUM PROCUREMENT STANDARDS FOR AN ORGAN PROCUREMENT ORGANIZATION (OPO) In order to maximize the gift of donation and optimize recipient outcomes and safety, the Organ Procurement Organization (OPO)
2.0 MINIMUM PROCUREMENT STANDARDS FOR AN ORGAN PROCUREMENT ORGANIZATION (OPO) In order to maximize the gift of donation and optimize recipient outcomes and safety, the Organ Procurement Organization (OPO)
patients with blood borne viruses Controlled Document Number: Version Number: 4 Controlled Document Sponsor: Controlled Document Lead:
 CONTROLLED DOCUMENT Procedure for the management of patients with blood borne viruses CATEGORY: CLASSIFICATION: PURPOSE Controlled Document Number: Version Number: 4 Controlled Document Sponsor: Controlled
CONTROLLED DOCUMENT Procedure for the management of patients with blood borne viruses CATEGORY: CLASSIFICATION: PURPOSE Controlled Document Number: Version Number: 4 Controlled Document Sponsor: Controlled
keyword: hepatitis Hepatitis
 www.bpac.org.nz keyword: hepatitis Hepatitis Key reviewers: Dr Susan Taylor, Microbiologist, Diagnostic Medlab, Auckland Dr Tim Blackmore, Infectious Diseases Physician and Microbiologist, Wellington Hospital,
www.bpac.org.nz keyword: hepatitis Hepatitis Key reviewers: Dr Susan Taylor, Microbiologist, Diagnostic Medlab, Auckland Dr Tim Blackmore, Infectious Diseases Physician and Microbiologist, Wellington Hospital,
10/17/2015. Chapter 55. Care of the Patient with HIV/AIDS. History of HIV. HIV Modes of Transmission
 Chapter 55 Care of the Patient with HIV/AIDS All items and derived items 2015, 2011, 2006 by Mosby, Inc., an imprint of Elsevier Inc. All rights reserved. History of HIV Remains somewhat obscure The earlier
Chapter 55 Care of the Patient with HIV/AIDS All items and derived items 2015, 2011, 2006 by Mosby, Inc., an imprint of Elsevier Inc. All rights reserved. History of HIV Remains somewhat obscure The earlier
Bloodborne Pathogens Exposure Procedure
 Bloodborne Pathogens Exposure Procedure Background: Bloodborne pathogens are infectious microorganisms present in blood that can cause disease in humans. These pathogens include, but are not limited to,
Bloodborne Pathogens Exposure Procedure Background: Bloodborne pathogens are infectious microorganisms present in blood that can cause disease in humans. These pathogens include, but are not limited to,
Hepatitis C January 26, 2018
 Hepatitis C January 26, 2018 Case Investigation Guidelines Contents A. Purpose...2 B. Case Definitions...2 a. Acute Hepatitis C (2016...2 b. Chronic Hepatitis C (2016)...3 c. Perinatal Hepatitis C (2017
Hepatitis C January 26, 2018 Case Investigation Guidelines Contents A. Purpose...2 B. Case Definitions...2 a. Acute Hepatitis C (2016...2 b. Chronic Hepatitis C (2016)...3 c. Perinatal Hepatitis C (2017
EXPOSURE (HIV/HEPATITIS) BLOOD & BODY FLUIDS
 Page(s): 1 of 11 PURPOSE To set a standardized procedure to ensure that employees are evaluated in a consistent and timely manner.. POLICY A. The treatment of Team Member exposure to bloodborne pathogens
Page(s): 1 of 11 PURPOSE To set a standardized procedure to ensure that employees are evaluated in a consistent and timely manner.. POLICY A. The treatment of Team Member exposure to bloodborne pathogens
Welcome to Your Reading Assignment
 Welcome to Your Reading Assignment This workbook contains four reading assignments. It is filled with easy-to-read articles you can use to help keep yourself and those you care about safe. After each reading
Welcome to Your Reading Assignment This workbook contains four reading assignments. It is filled with easy-to-read articles you can use to help keep yourself and those you care about safe. After each reading
NEOMED ACADEMIC POLICY
 (A) PURPOSE The purpose of this Policy is to delineate the management of incidents of exposure to blood-borne pathogens that occur to students while they are in the educational setting. (B) SCOPE This
(A) PURPOSE The purpose of this Policy is to delineate the management of incidents of exposure to blood-borne pathogens that occur to students while they are in the educational setting. (B) SCOPE This
DHMH Activities toward Implementing Requirements of Md. Code Ann., Health-General , Hepatitis C Prevention and Control within Maryland
 DHMH Activities toward Implementing Requirements of Md. Code Ann., Health-General 18-1001, Hepatitis C Prevention and Control within Maryland Submitted by: Maryland Department of Health and Mental Hygiene
DHMH Activities toward Implementing Requirements of Md. Code Ann., Health-General 18-1001, Hepatitis C Prevention and Control within Maryland Submitted by: Maryland Department of Health and Mental Hygiene
BC Enhanced Hepatitis Strain Surveillance Project Hepatitis B. Acute HBV Questionnaire
 BC Enhanced Hepatitis Strain Surveillance Project Hepatitis B Informed Consent: Please read to individual and answer any questions There are about 60,000 people with hepatitis B in BC. We want to find
BC Enhanced Hepatitis Strain Surveillance Project Hepatitis B Informed Consent: Please read to individual and answer any questions There are about 60,000 people with hepatitis B in BC. We want to find
Background. that NAT screening could significantly reduce the rate of undetected HCV for all donor risk strata.
 American Journal of Transplantation 2011; 11: 1201 1208 Wiley Periodicals Inc. C 2011 The Authors Journal compilation C 2011 The American Society of Transplantation and the American Society of Transplant
American Journal of Transplantation 2011; 11: 1201 1208 Wiley Periodicals Inc. C 2011 The Authors Journal compilation C 2011 The American Society of Transplantation and the American Society of Transplant
Hepatitis B and C Overview, Outbreaks, and Recommendations. Viral Hepatitis Language. Types of Viral Hepatitis 7/1/2013
 Hepatitis B and C Overview, Outbreaks, and Recommendations Elizabeth Lawlor, MS Healthy Kansans living in safe and sustainable environments. Viral Hepatitis Language Acute infection is when the infection
Hepatitis B and C Overview, Outbreaks, and Recommendations Elizabeth Lawlor, MS Healthy Kansans living in safe and sustainable environments. Viral Hepatitis Language Acute infection is when the infection
An evaluation of the STD profiles and safe sex practices of a sample of swingers
 An evaluation of the STD profiles and safe sex practices of a sample of swingers Presented by: Edward M. Fernandes, Ph.D. Assistant Professor of Psychology Barton College, North Carolina George Gaither,
An evaluation of the STD profiles and safe sex practices of a sample of swingers Presented by: Edward M. Fernandes, Ph.D. Assistant Professor of Psychology Barton College, North Carolina George Gaither,
TB in Corrections Phoenix, Arizona
 TB in Corrections Phoenix, Arizona March 24, 2011 Contact Investigation in the Correctional Setting Jessica Quintero, BAAS March 24, 2011 Jessica Quintero, BAAS has the following disclosures to make: No
TB in Corrections Phoenix, Arizona March 24, 2011 Contact Investigation in the Correctional Setting Jessica Quintero, BAAS March 24, 2011 Jessica Quintero, BAAS has the following disclosures to make: No
STI & HIV PRE-TEST ANSWER KEY
 Name: STI & HIV PRE-TEST ANSWER KEY 1. You can catch a STI or HIV from door knobs, toilet seats, or FALSE drinking fountains. STIs are transmitted sexually, requiring sexual contact. Some STIs can be transmitted
Name: STI & HIV PRE-TEST ANSWER KEY 1. You can catch a STI or HIV from door knobs, toilet seats, or FALSE drinking fountains. STIs are transmitted sexually, requiring sexual contact. Some STIs can be transmitted
Confirmed (Laboratory Tests) Serum positive for IgM anti-hbc or, hepatitis B surface antigen (HbsAg).
 Hepatitis B Hepatitis B is a liver disease that results from infection with the Hepatitis B virus. It can range in severity from a mild illness lasting a few weeks to a serious, lifelong illness. Hepatitis
Hepatitis B Hepatitis B is a liver disease that results from infection with the Hepatitis B virus. It can range in severity from a mild illness lasting a few weeks to a serious, lifelong illness. Hepatitis
ANNUAL MORBIDITY REPORT
 DUTCHESS COUNTY DEPARTMENT OF HEALTH S ANNUAL MORBIDITY REPORT Marcus J. Molinaro, Dutchess County Executive Kari Reiber, MD, Acting Commissioner of Health Volume 8, Issue FIRST CONGENITAL SYPHILIS CASE
DUTCHESS COUNTY DEPARTMENT OF HEALTH S ANNUAL MORBIDITY REPORT Marcus J. Molinaro, Dutchess County Executive Kari Reiber, MD, Acting Commissioner of Health Volume 8, Issue FIRST CONGENITAL SYPHILIS CASE
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding on national level More stringent - recommended on national level Not legy binding and not recommended on
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding on national level More stringent - recommended on national level Not legy binding and not recommended on
Non-reproductive tissues and cells
 Colour key Tested pathogen VIRAL Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recommended on national level Not
Colour key Tested pathogen VIRAL Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recommended on national level Not
HIV, STI AND OTHER BLOOD- BORNE DISEASES. Kolářová M., EPI Autumn 2015
 HIV, STI AND OTHER BLOOD- BORNE DISEASES Kolářová M., EPI Autumn 2015 Chlamydia infection Genital warts Gonorrhoea Hepatitis B Hepatitis C HIV infection and AIDS Human papillomavirus infection Sexually
HIV, STI AND OTHER BLOOD- BORNE DISEASES Kolářová M., EPI Autumn 2015 Chlamydia infection Genital warts Gonorrhoea Hepatitis B Hepatitis C HIV infection and AIDS Human papillomavirus infection Sexually
Appendix B Complications, Treatment, and Prevention of STIs
 From Management of Men s Reproductive Health Problems 2003 EngenderHealth Appendix B Complications, Treatment, and Prevention of STIs EngenderHealth Men s Reproductive Health Problems B.1 Complications,
From Management of Men s Reproductive Health Problems 2003 EngenderHealth Appendix B Complications, Treatment, and Prevention of STIs EngenderHealth Men s Reproductive Health Problems B.1 Complications,
How does HBV affect the liver?
 Hepatitis B Why is the liver important? Your liver is a vital organ that performs many essential functions. It s the largest solid organ in the body and is located under your rib cage on the upper right
Hepatitis B Why is the liver important? Your liver is a vital organ that performs many essential functions. It s the largest solid organ in the body and is located under your rib cage on the upper right
Viral Hepatitis Diagnosis and Management
 Viral Hepatitis Diagnosis and Management CLINICAL BACKGROUND Viral hepatitis is a relatively common disease (25 per 100,000 individuals in the United States) caused by a diverse group of hepatotropic agents
Viral Hepatitis Diagnosis and Management CLINICAL BACKGROUND Viral hepatitis is a relatively common disease (25 per 100,000 individuals in the United States) caused by a diverse group of hepatotropic agents
43. Guidelines on Needle stick Injury
 43. Guidelines on Needle stick Injury The following information is abstracted from the South African Department of Health guidelines entitled: Management of Occupational Exposure to the Human Immunodeficiency
43. Guidelines on Needle stick Injury The following information is abstracted from the South African Department of Health guidelines entitled: Management of Occupational Exposure to the Human Immunodeficiency
EMPLOYEE HEALTH TABLE OF CONTENTS
 EMPLOYEE HEALTH TABLE OF CONTENTS PHYSICAL ASSESSMENT.2 REPORTING EMPLOYEE INFECTIONS...3 SUMMARY OF IMPORTANT RECOMMENDATIONS AND WORK RESTRICTIONS FOR PERSONNEL WITH INFECTIOUS DISEASES.4 HEPATITIS B
EMPLOYEE HEALTH TABLE OF CONTENTS PHYSICAL ASSESSMENT.2 REPORTING EMPLOYEE INFECTIONS...3 SUMMARY OF IMPORTANT RECOMMENDATIONS AND WORK RESTRICTIONS FOR PERSONNEL WITH INFECTIOUS DISEASES.4 HEPATITIS B
Hepatitis C Virus (HCV)
 Clinical Practice Guidelines Hepatitis C Virus (HCV) OBJECTIVE The purpose is to guide the appropriate diagnosis and management of Hepatitis C Virus (HCV). GUIDELINE These are only guidelines, and are
Clinical Practice Guidelines Hepatitis C Virus (HCV) OBJECTIVE The purpose is to guide the appropriate diagnosis and management of Hepatitis C Virus (HCV). GUIDELINE These are only guidelines, and are
iphis Case ID #: Reported Date: Reporting Source: Diagnosing Health Unit: Branch Office: Outbreak Number:
 Case Investigation Form: Hepatitis C Legend iphis system mandatory Required Case Information (add dates as YYYY-MM-DD) Case s Last Name: Case s First Name: Middle Name: Birth Date: Male Gender: Female
Case Investigation Form: Hepatitis C Legend iphis system mandatory Required Case Information (add dates as YYYY-MM-DD) Case s Last Name: Case s First Name: Middle Name: Birth Date: Male Gender: Female
San Francisco Department of Public Health Program Collaboration and Service Integration Surveillance Baseline Assessment
 Background and Purpose San Francisco Department of Public Health This syndemics assessment is the first step in developing a sustainable system of primary prevention and clinical care in San Francisco
Background and Purpose San Francisco Department of Public Health This syndemics assessment is the first step in developing a sustainable system of primary prevention and clinical care in San Francisco
MSM DEFERRAL POLICY ISSUE DOCUMENT: MSM DEFERRAL ISSUE BACKGROUND
 MSM DEFERRAL POLICY ISSUE DOCUMENT: MSM DEFERRAL ISSUE Countries around the world are reviewing the policy of deferring from blood donation men who have had sex with men (MSM). Both Canadian Blood Services
MSM DEFERRAL POLICY ISSUE DOCUMENT: MSM DEFERRAL ISSUE Countries around the world are reviewing the policy of deferring from blood donation men who have had sex with men (MSM). Both Canadian Blood Services
Jackie Williams BBV/Sexual Health Trainer
 Jackie Williams BBV/Sexual Health Trainer HEPATITIS The Basics What does your liver do? FUNCTIONS n storage of substances - glycogen, iron and vitamins n disposal of metabolic wastes - urea and bile n
Jackie Williams BBV/Sexual Health Trainer HEPATITIS The Basics What does your liver do? FUNCTIONS n storage of substances - glycogen, iron and vitamins n disposal of metabolic wastes - urea and bile n
To provide the guidelines for the management of healthcare workers who have had an occupational exposure to blood and/or body fluids.
 TITLE/DESCRIPTION: MANAGEMENT OF OCCUPATIONAL EXPOSURE TO HBV, HCV, and HIV INDEX NUMBER: EFFECTIVE DATE: APPLIES TO: ISSUING AUTHORITY: 01/01/2009 01/01/2013 All GCC Countries GULF COOPERATION COUNCIL
TITLE/DESCRIPTION: MANAGEMENT OF OCCUPATIONAL EXPOSURE TO HBV, HCV, and HIV INDEX NUMBER: EFFECTIVE DATE: APPLIES TO: ISSUING AUTHORITY: 01/01/2009 01/01/2013 All GCC Countries GULF COOPERATION COUNCIL
Knowledge, attitude and practices among health care workers on needle-stick injuries
 Kathmandu University Medical Journal (2003) Vol. 1,. 2, 91-94 Original Article Knowledge, attitude and practices among health care workers on needle-stick injuries Gurubacharya DL 1, KC Mathura 2, Karki
Kathmandu University Medical Journal (2003) Vol. 1,. 2, 91-94 Original Article Knowledge, attitude and practices among health care workers on needle-stick injuries Gurubacharya DL 1, KC Mathura 2, Karki
Making Your Blood Donation Safe
 BLOOD DONOR EDUCATIONAL MATERIALS Making Your Blood Donation Safe Pete Lawson Burned in a wildfire, LifeStream blood donations saved his life. WWW.LSTREAM.ORG NEW! Important Information on Iron! Recent
BLOOD DONOR EDUCATIONAL MATERIALS Making Your Blood Donation Safe Pete Lawson Burned in a wildfire, LifeStream blood donations saved his life. WWW.LSTREAM.ORG NEW! Important Information on Iron! Recent
Note: Staff who work in case management programs should attend the AIDS Institute training, "Addressing Prevention in HIV Case Management.
 Addressing Prevention with HIV Positive Clients This one-day training will prepare participants to help people living with HIV to avoid sexual and substance use behaviors that can result in transmitting
Addressing Prevention with HIV Positive Clients This one-day training will prepare participants to help people living with HIV to avoid sexual and substance use behaviors that can result in transmitting
POLICY ON BLOOD AND BODY FLUID EXPOSURE (NEEDLE STICK)
 POLICY ON BLOOD AND BODY FLUID EXPOSURE (NEEDLE STICK) Dated: January 1, 2018 Supersedes: Blood and Body Fluid Exposure (Needle stick) dated July 1, 2015 I. POLICY It is the policy of New York Medical
POLICY ON BLOOD AND BODY FLUID EXPOSURE (NEEDLE STICK) Dated: January 1, 2018 Supersedes: Blood and Body Fluid Exposure (Needle stick) dated July 1, 2015 I. POLICY It is the policy of New York Medical
Frequently Asked Questions about HIV/AIDS: Transmission and Prevention How is HIV passed from one person to another?
 Frequently Asked Questions about HIV/AIDS: Transmission and Prevention How is HIV passed from one person to another? HIV transmission can occur when blood, semen (including pre-seminal fluid or "pre-cum"),
Frequently Asked Questions about HIV/AIDS: Transmission and Prevention How is HIV passed from one person to another? HIV transmission can occur when blood, semen (including pre-seminal fluid or "pre-cum"),
Chapter 20. Preview. Bellringer Key Ideas Risks of Teen Sexual Activity Teen Pregnancy Abstinence Eliminates the Risks of Teen Sexual Activity
 Section 1 What Are the Risks? Preview Bellringer Key Ideas Risks of Teen Sexual Activity Teen Pregnancy Abstinence Eliminates the Risks of Teen Sexual Activity Section 1 What Are the Risks? Bellringer
Section 1 What Are the Risks? Preview Bellringer Key Ideas Risks of Teen Sexual Activity Teen Pregnancy Abstinence Eliminates the Risks of Teen Sexual Activity Section 1 What Are the Risks? Bellringer
User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire. Table of Contents
 User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire Table of Contents Purpose Introduction Capture Questions Methods of Administration DHQ Materials - Structure and
User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire Table of Contents Purpose Introduction Capture Questions Methods of Administration DHQ Materials - Structure and
Looking at NY: Our rate of chlamydia is higher than the US as a whole; we rank 13th among all states.
 1 In this presentation, we will discuss: What are the most common STDs in NYS What are the most common STDs Can I get gonorrhea from oral sex How do you know if you/or your partner has an STD? Does getting
1 In this presentation, we will discuss: What are the most common STDs in NYS What are the most common STDs Can I get gonorrhea from oral sex How do you know if you/or your partner has an STD? Does getting
The Alphabet Soup of Viral Hepatitis Testing
 The Alphabet Soup of Viral Hepatitis Testing August 18, 2011 Patricia Slev, PhD, DABCC Medical Director, Serologic Hepatitis and Retrovirus Laboratory, ARUP Laboratories Assistant Professor of Pathology,
The Alphabet Soup of Viral Hepatitis Testing August 18, 2011 Patricia Slev, PhD, DABCC Medical Director, Serologic Hepatitis and Retrovirus Laboratory, ARUP Laboratories Assistant Professor of Pathology,
Breastfeeding Center Milk Connection Questionnaire and Release for Donor
 Breastfeeding Center Milk Connection Questionnaire and Release for Donor All of us at The Breastfeeding Center are deeply grateful to you for your important gift to infants in our community. Your donated
Breastfeeding Center Milk Connection Questionnaire and Release for Donor All of us at The Breastfeeding Center are deeply grateful to you for your important gift to infants in our community. Your donated
The new German strategy on HIV, Hepatitis B, C and STI, an integrated approach. Ines Perea Ministry of Health, Germany
 The new German strategy on HIV, Hepatitis B, C and STI, an integrated approach Ines Perea Ministry of Health, Germany Reasons for a new strategy in 2016 New international agreements (SDG s) Renewed political
The new German strategy on HIV, Hepatitis B, C and STI, an integrated approach Ines Perea Ministry of Health, Germany Reasons for a new strategy in 2016 New international agreements (SDG s) Renewed political
Hepatitis B Virus and the Opioid Crisis
 Hepatitis B Virus and the Opioid Crisis Alice Asher, RN, Ph.D. Epidemiology and Surveillance Branch Division of Viral Hepatitis National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention Centers
Hepatitis B Virus and the Opioid Crisis Alice Asher, RN, Ph.D. Epidemiology and Surveillance Branch Division of Viral Hepatitis National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention Centers
Chapter 7 Reproductive Tract Infections and HIV/AIDS
 Chapter 7 Reproductive Tract Infections and HIV/AIDS Introduction Reproductive Tract Infections (RTIs) Infections caused by a variety of organisms that affect upper and/or lower reproductive tracts Most
Chapter 7 Reproductive Tract Infections and HIV/AIDS Introduction Reproductive Tract Infections (RTIs) Infections caused by a variety of organisms that affect upper and/or lower reproductive tracts Most
Race (Check one): White Black Asian American Indian/Eskimo/ALEU Hawaiian Native/Pacific Islander Other
 Please mail or fax to: Dallas Transplant Institute Pre-Transplant Group 1420 Viceroy Drive Dallas, TX 75235 Fax: (214) 366-6088 Donor Name: SS#: Date of birth: Age: Sex: Male Female Address: City/State/Zip
Please mail or fax to: Dallas Transplant Institute Pre-Transplant Group 1420 Viceroy Drive Dallas, TX 75235 Fax: (214) 366-6088 Donor Name: SS#: Date of birth: Age: Sex: Male Female Address: City/State/Zip
Non-reproductive tissues and cells
 Ministry of Health: Institute for Transplantation and Biomedicine / Colour key VIRAL HIV 1 and HIV 2 Hepatitis B Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More
Ministry of Health: Institute for Transplantation and Biomedicine / Colour key VIRAL HIV 1 and HIV 2 Hepatitis B Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More
Sexually Transmitted Infections. Naluce Manuela Morris, MPH, CHES
 Sexually Transmitted Infections Naluce Manuela Morris, MPH, CHES Handshake Activity Handshake Activity Mrs. or Mr. X Mrs. or Mr. Y Carry this glove but do not put it on Put this glove on before shaking
Sexually Transmitted Infections Naluce Manuela Morris, MPH, CHES Handshake Activity Handshake Activity Mrs. or Mr. X Mrs. or Mr. Y Carry this glove but do not put it on Put this glove on before shaking
