Ensuring quality, safety and efficacy of vaccines
|
|
|
- Harry Randall
- 6 years ago
- Views:
Transcription
1 EDCTP Third Annual Forum 2006 Partnership and African Leadership: Challenges & Opportunities Ensuring quality, safety and efficacy of vaccines Vaccine regulatory issues in African countries Lahouari Belgharbi, Immunization, Vaccines & Biological (IVB) World Health Organization (WHO) World Health Organization
2 Outline of the presentation 1. Issues and challenges met by regulatory systems in Africa 2. WHO policy to ensure quality, safety and efficacy of vaccines 3. Activities planned and implemented to address the above issues 4. Monitoring, progress and impact on vaccine regulatory systems
3 Issues & challenges met by regulatory systems in Africa : Scientific issues Limited expertise in new vaccines science (Combo, DNA, adjuvant, preservative, vector, etc..) Huge viral diversity (HIV, Rotavirus) & complex geographic distribution Weak registration & licensing process when dealing with biologicals Inadequate immune response to natural infection Immune correlates of protection difficult to establish Vaccine response is not always better than natural infection Lack of appropriate models indicating that human trials are key to develop research Definition of efficacy and end points are key in clinical trial protocol
4 Issues & challenges met by regulatory systems in Africa Regulatory issues No/weak regulations or not consistent with international standards (lot release, GMP, GCP, GLP, etc) Overlap of roles of parties (NRA & Ethics) Limited knowledge of foreign sponsored regulations Absence of provision for expedite reviews Regulation/Guidelines in place require updating (article 58, ECBS guidelines) No inspection of clinical trials sites Lack of procedures to assess, authorize and monitor CTs Limited/no Pharmacovigilance & laboratory capacity Managerial issues Limited human resources for regulation Lack of funding to develop NRA oversight No or limited local manufacturing capacity Limited exchange or sharing regulatory information to guide decision making Sponsors and manufacturers influence decision making that is not consistent with international standards
5 FUNCTIONNAL NRA THAT HAVE CAPACITY TO REGULATE VACCINES, AFRICA, July 2006 Tunisia Morocco Algeria Egypt Senegal Ghana Nigeria Ethiopia Uganda Functionnal NRA potential to become functionnal Zimbabwe Limited or weak vaccine regulatory system South Africa
6 Assured quality source of vaccine procured through WHO prequalification, industrialized countries 6 developing countries 24 manufacturers Australia Belgium Canada Denmark France Germany Hungary Italy Japan Rep.Korea United Kingdom USA Switzerland Sweden 13% population Brazil Bulgaria Cuba India Indonesia Senegal 24% population 65 pre-qualified vaccines used in 112 countries 53% total population
7 Main source of vaccines used for national immunization programmes in Africa, 2006 Producing Only Y.Fever is produced, all other vaccines are procured through UNICEF 2 % Procuring Source of of vaccines, 2006 Number Number of of countries countries Total = 46 countries Total = 46 countries UN UN agency agency Procuring Procuring Producing Producing World Health Organization, HTP/V&B/ATT. LBelgharbi World Health Organization, HTP/V&B/ATT. LBelgharbi UN agency % Using WHO Prequalification scheme % 15 out 19 countries are procuring From country that have functional NRA 92% 92% countries used vaccines of of assured quality
8 2.WHO policy to ensure quality, safety and efficacy of vaccines
9 World Health Organization s Goals Ensure that 100% 100% of vaccines used in all national immunization programmes are of assured quality Definition of Assured quality vaccines for a vaccine producing country National Regulatory Authority (NRA) independent from vaccine manufacturer & or procurement system NRA fully functional (system + 6 regulatory functions implemented) No unresolved reported problem with vaccine Guided by Experts Committee on Standardization of Biologicals (ECBS) recommendations on safety, efficacy and quality issued in WHO Technical Report Series (TSR)
10 Applicants Dossier (manufacturer (manufacturer or or distributor) distributor) Vaccine regulatory process Pre-marketing phase Licensing/Registration= evaluation process Post Marketing phase Market distribution Product Evaluation Dossier testing Laboratory access Lot Lot release testing Post Post marketing marketing AEFI AEFI surveillance surveillance Quality Licensing facility facility Regulatory inspections Inspections Safety Safety Efficacy Authorization clinical trials trials (Ethical (Ethical review review process, process, compliance compliance against against GLP,GMP,GCP) GLP,GMP,GCP) Marketing Authorization (M.A.) (M.A.) monitoring
11 Applicants Dossier (manufacturer (manufacturer or or distributor) distributor) Pre-marketing phase Licensing/Registration= evaluation process Product Evaluation Dossier Function 1 Marketing testing Autorization & Activities Quality licensing Licensing facility facility Safety Safety Efficacy Vaccine regulatory process Function 4 Laboratory access access Regulatory Function 5 inspections Regulatory inspections Function 6:Supervision Authorization clinical of clinical trials trials trials (Ethical (Ethical review review process, process, compliance compliance against against GLP,GMP,GCP) GLP,GMP,GCP) Post Marketing phase Market distribution Function 2 Function 3 Postmarketing Lot Lot release testing Marketing Authorization (M.A.) (M.A.) Post Post marketing marketing AEFI surveillance surveillance Inspections monitoring
12 Regulatory functions Regulatory functions Regulatory system Marketing Autorization & Licensing activities Postmarketing: AEFI Lot release Laboratory access Regulatory inspections Authorization & monitoring of CTs National Regulatory Functions recommended for vaccine development Source of vaccines UN agency Procure Produce Functions undertaken in producing Countries with functional NRA in countries that conduct clinical trials CTs : Clinical trials, UN: United Nations, AEFI: Adverse Events Following Immunization
13 The Global Training Network (GTN) trained 1200 staff from 100 Global resource to raise technical skills in the field of biological countries since 1996 & was supported by WB, JICA, regulation: the Global Training Network since 1996 DFID,AusAid,WHO,EU,IDB,ADB,UNICEF,WHO + EDCTP 7-Health Canada Canada - NRA 1-Instituto Finlay -Producer 3-CECMED -NRA Cuba Courses 1 Regulatory inspections 2 - Quality Control Methods 3 - Laboratory Quality Systems 4 vaccine regulation) 5 - Animal Husbandry 6 - Short Course in DTP 7 - Lot Release and Lab Access 8 Post marketing surveillance/aefi 9 - Licensing (for procuring countries) - U.S.A. - Producer 2,3-NIBSC UK - NCL 7- AFSSAPS France - NRA 4.Senegal 8- MOH Tunisia - MoH 5-CECAL/Fiocruz Brazil - Producer 2,5,6-NVI Netherlands - Producer 8-University of Cape Town South Africa -NRA Contract Work India 10.Benin 1o.Ethiopia 8-MoH Sri Lanka -MoH 2-Biken Japan - Producer 2-BioFarma Indonesia -Producer Participants 1.Korea 4-TGA Australia - NRA 1. NRA Staff with Government Plan 2. Staff of Manufacturer with NRA & Strategic Plan 3. NRA Staff and EPI Staff from vaccine procuring countries (for AEFI course only)
14 4.Activities planned and implemented
15 By end of 2010 : 37 out of 46 COUNTRIES WILL HAVE DEVELOPED APPROPRIATE CRITICAL REGULATORY FUNCTIONS 2006: 6 countries 2007: 6 countries 2008: 8 countries 2009: 8 countries 2010: 9 countries
16 Activities recommended to strengthen vaccine regulatory systems in Africa Sensitization of of country Stakeholders Development of of Institutional Development Plan Plan (IDP) Joint inspection of of Clinical trials sites sites Networks & Forum to to Exchange and and share Regulatory information Development regulation & procedures NRA assessment & Follow up up visits to to monitor impact Training on on critical regulatory functions Joint review of of clinical trials applications Building centre Excellence & roster of regulatory/scientific experts
17 ACTIVITIES TO STRENGTHEN VACCINE REGULATORY SYSTEMS IN AFRICA Planning to address gaps Regulatory pathway Surveillance AEFI GTN Training Capacity building Fundraising Advocacy Activities 1 Activities 2... Activities conducted 1.Fund raising plan 2. Three NRA planning workshops countries with IDP* for 28 countries 3. Meeting of Developing Countries vaccines regulatory network (DCVRN) 4.Joint review of CTs applications for MeningoA + Workshop on regulatory procedures for clinical evaluation of vaccines+forum for the evaluation of clinical data of rotavirus vaccines for registration purposes 5. Sensitization/advocacy workshop for all country stakeholders 6. GTN training provided to AFR countries on vaccine regulation Status Completed Completed Completed Completed 1 out 3 completed 9 out of 28 countries completed * Institutional Development Plan (IDP) 7. GTN Training provided to AFR countries on PMS/AEFI 8. GTN training provided to AFR countries on clinical evaluation 15 out of 28 completed 6 out of 28 countries completed
18 NRA PLANNING WORKSHOPS CONDUCTED TO DEVELOP INSTIUTIONAL DEVCELOPMENT PLAN (IDP) FOR 28 COUNTRIES IN 2005 Funded by WHO & AAVP Addis Abeba,, Jan.2005 Ouagadougou, May 2005 Gaborone,Dec.2005
19 AFR : Regional initiatives Objectives & expected outcome 1. Planning NRA activities through development of Institutional Development Plan: completed in 28 countries 2. Promote communication among NRAs and raise awareness on regulatory changes and challenges: DCVRN input, AVAREF, Joint review, Joint inspection of CTs, Sensitization workshops (Uganda) 3. Promote communication between sponsors/regulators/ethics committees/ research centres to determine specific needs for different types of vaccines: same as point 2 4. Provide expert support to assess suitability of clinical data for registration : DCVRN, AVAREF, training on clinical evaluation 5. Facilitate capacity building activities and availability of expertise for regulatory review of clinical trial applications and monitoring of clinical trials: NRA assessment, follow up visits, meeting of regulators and training on relevant regulatory functions. joint review of CTAs, joint inspection, AVAREF 6. Plan and organise training on relevant regulatory functions that are critical for African NRAs: Clinical evaluation, PMS/AEFI and regulation. DCVRN on regulatory inspections of CTs
20 WHO/EDCTP activities planned & conducted to support the development of an harmonized regulatory framework in Africa, PLANNING PHASE: Jan, May and Dec.: 3 NRA planning workshops for 28 countries (Addis, Ouagadougou & Gaborone): Main outcome: 28 Institutional Development Plans (IDP) to implement harmonized/common regulatory framework to ensure quality, safety, efficacy of vaccines and relevant clinical trials conducted in Africa. Countries involved: Ghana, The Gambia, Ethiopia, Kenya, Uganda, Senegal, Mali, Centrafrican Republic, Togo, Guinea, Chad, Benin, Cameroon, Niger, Rwanda, Angola, Botswana, DRC, Malawi, Namibia, Rwanda, South Africa, Tanzania, Zambia & Zimbabwe, EDCTP. WHO funding 2. March: training course on authorisation/approval of clinical trials (Pretoria, South Africa) Main outcome: Providing knowledge to regulators and vaccine experts about principle of vaccine clinical evaluation relevant to authorisation and monitoring of clinical trials Countries involved: Ghana, the Gambia, Uganda, Kenya, Nigeria & Ethiopia. WHO funding
21 WHO/EDCTP activities planned & conducted to support the development of an harmonized regulatory framework in Africa, PLANNING PHASE: Sept.2005: Workshop on Regulatory Procedures for clinical evaluation of Vaccines (Addis, Ethiopia) 1. Main outcome: Templates procedures for submission/review clinical trials applications and integration of activities & importation/release of clinical batches 2. Countries involved: Botswana, Ghana, Cameroon, Ethiopia, The Gambia, Uganda, Kenya, Mali, Nigeria, Senegal, Tanzania, Zambia, South Africa. WHO funding 4. Dec.2005: Regulatory forum on clinical evaluation of rotavirus vaccines (Botswana 12/05) 1. Main outcome: a) Presentation & discussions of scientific information on issues that may affect the efficacy and safety of rotavirus vaccines. B) allow countries to make a final decision with regards to registration of rotavirus vaccines 2. Countries involved:botswana, Ghana, Gambia, Zimbabwe, Malawi, Cote d'ivoire, South Africa, Zambia, and Cameroon: WHO funding
22 WHO/EDCTP activities planned & conducted to support the development of an harmonized regulatory framework in Africa, IMPLEMENTATION PHASE Jan-May: EDCTP/WHO agreement developed & signed 1. Main outcome: Support of Euros for 18 months. (June 2006-Dec.2007) 2. Countries involved: Ghana, Uganda, Tanzania, Nigeria, Malawi, The Gambia, Mozambique, Rwanda, Gabon, Mali, Burkina Faso, Kenya, Ethiopia, Zambia, Cote D'Ivoire, EDCTP funding 2. June: Joint review of CTA of Conjugate Meningitis A vaccine (Banjul, The Gambia) Main outcome: Review of clinical trials application for phase II Conj:Menin.A vaccine by Mali & the Gambia national regulatory & vaccine experts on relevant gaps/missing information concerning Men.A vaccine Clinical trials. Country involved: The Gambia, Mali, Ghana, Senegal.- WHO funding 3. July & August: Country workshop in Uganda & Senegal to sensitize all stakeholder for implementation of IDP recommendations: Main outcome: Updated IDP and coordination plan to involved all stakeholders in follow up implementation of recommendations Country involved: Senegal & Uganda WHO funding
23 WHO/EDCTP activities planned & conducted to support the development of an harmonized regulatory framework in Africa, IMPLEMENTATION PHASE : Sept.: AFRO Vaccine Regulatory Forum AVAREF (Accra, Ghana, Sept) EDCTP funding completed 2. Development & translation of training material for the Benin (French) and Ethiopian (English) courses on authorisation & monitoring of clinical trials completed Jan.2007: Joint inspection of CT of Conjugate meningo A vaccine - EDCTP funding 2. March 2007: Workshop on regulatory inspections of clinical trials (tentative) - EDCTP funding 3. April & Oct.: AVAREF meeting 4. Sept.2005: Training for 10 countries (french speaking) on authorisation & monitoring of CTs: EDCTP funding 5. Feb & June 2007: Training for 20 countries (english speaking) on authorisation & monitoring of CTs (Addis,Ehtiopia) EDCTP funding 6. Jan-Dec: Follow up IDP and monitoring activities in 5 countries
24 4. Progress and impact on vaccine regulatory systems
25 Country Status: 0 NRA assessment conducted as of 1998 Country status : NRA assessment conducted & planned NRA assessment conducted Not yet conducted To be completed in 2006 however NRA prescreening already completed The boundaries and names shown and the designations used on this map do not imply the expression of any opinion whatsoever on the part of the World Health Organization concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. Dotted lines on maps represent approximate border lines for which there may not yet be full agreement.
26 Country Status: 68 NRA assessments conducted (Oct.1998-Dec 2005) Country status : NRA assessment conducted & planned NRA assessment conducted Not yet conducted To be completed in 2006 however NRA prescreening already completed The boundaries and names shown and the designations used on this map do not imply the expression of any opinion whatsoever on the part of the World Health Organization concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. Dotted lines on maps represent approximate border lines for which there may not yet be full agreement.
27 220 Regulatory experts recruited (Oct.1998 April 2006) NRA regulatory experts The boundaries and names shown and the designations used on this map do not imply the expression of any opinion whatsoever on the part of the World Health Organization concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. Dotted lines on maps represent approximate border lines for which there may not yet be full agreement.
28 CHANGES DOCUMENTED TO IMPROVE REGULATORY OVERSIGHT OF VACCINES Burkina Faso Ethiopia (planned) Senegal (planned) The Gambia (ongoing) Mali (ongoing) Ghana (ongoing) 1. Plan planned and implemented for all countries involved 2. Training planned and conducted for all countries involved 3. Template procedures to evaluate CTs applications 4. Amended regulation to Involve NRA in evaluation of CTs 5. Clarification of roles/responsibilities to authorize CTs 6. Focal point & training requested for staff 7. Guidelines discussed, amended for endorsement by MoH 8. Coordination among NRA/Ethics Committee to authorise CTs 9. 1 st African Vaccine regulators forum (Ghana, Accra, Sept.2006) Nigeria (ongoing) South Africa (expert's input)
29 1 st AVAREF meeting, Sept.2006 African Vaccine Regulators Forum Participants: 19 Countries, NRA and Ethics committee Experienced NRAs: EMEA & US FDA Product sponsors: GSK,MVPPATH, WRAIR, US NIH Themes : selected disease of importance : HIV, malaria, Menigo A & Rotavirus, regulators: Funded by: WHO, EDCTP; MVPATH and AAVP Issues: Low funding for NRAs, conflict of interest because of limiting resources, lack of confidence in IRBs, separate institutions, access epidemiological data, laboratory capacity, ADR investigation, information sharing network Recommendations: Need to expand and sustain capacity building in vaccine trials oversight Joint review of clinical trial application should be expanded Strong interest in conducting joint inspection of clinical trials sites Increase training opportunities to develop regulatory capacity Develop guidance for clarification roles of NRA & ethics committees Pharmacovigilance provision re article 58 should be flexible for implementation Develop clinical trials case definition of efficacy for malaria vaccines Secretariat 's forum is hosted in WHO/AFRO
30 Next steps Plan for the 2 nd phase and identify resources to sustain WHO/EDCTP initiative (initial phase granted Euros to be completed by Dec.2007) Support in priority coordination and monitoring of initiative (forum, networking, African experts in regional coordination mechanism/institutions) Support training, coordinate curriculum development and expand to all relevant institutions (NRA, Ethics, Research, Pharmacovigilance centre) Expand networking among key networking international leader and country partners Use and expand IT to advocate, publish best practices, experience and develop online training
31 Information sources for regulators (access upon request, used as as space to to share information with all all country participants and partners (eg.edctp,, UNICEF, NRAs, NCLs, Ethics, etc)
Evaluating the acceptability of vaccine and vaccination programmes: an individual and public health perspective
 The regulatory process Evaluating the acceptability of vaccine and vaccination programmes: an individual and public health perspective Annecy, Les Pensières July 7-9, 2008 Dr. Nora Dellepiane, Scientist
The regulatory process Evaluating the acceptability of vaccine and vaccination programmes: an individual and public health perspective Annecy, Les Pensières July 7-9, 2008 Dr. Nora Dellepiane, Scientist
Procedure for Expedited Review of imported pre-qualified vaccines for use in national immunization programmes
 Procedure for Expedited Review of imported pre-qualified vaccines for use in national immunization programmes Dr Nora Dellepiane/Dr Anil Kumar Chawla WHO/HQ-Geneva, Switzerland 1 Expedited review procedure
Procedure for Expedited Review of imported pre-qualified vaccines for use in national immunization programmes Dr Nora Dellepiane/Dr Anil Kumar Chawla WHO/HQ-Geneva, Switzerland 1 Expedited review procedure
Quality assurance for essential medicines and health products: moving towards an harmonized approach
 Quality assurance for essential medicines and health products: moving towards an harmonized approach Prequalification of vaccines David Wood QSS/EMP/WHO 12 December 2012 A growing portion of vaccines procured
Quality assurance for essential medicines and health products: moving towards an harmonized approach Prequalification of vaccines David Wood QSS/EMP/WHO 12 December 2012 A growing portion of vaccines procured
Regulatory Capacity Building and Developing Countries: CBER Perspective
 Regulatory Capacity Building and Developing Countries: CBER Perspective Karen Midthun, M.D. Director Center for Biologics Evaluation and Research US Food and Drug Administration IOM Meeting Strengthening
Regulatory Capacity Building and Developing Countries: CBER Perspective Karen Midthun, M.D. Director Center for Biologics Evaluation and Research US Food and Drug Administration IOM Meeting Strengthening
IMMUNIZATION VACCINE DEVELOPMENT
 IMMUNIZATION VACCINE DEVELOPMENT MONTHLY IMMUNIZATION AND POLIO UPDATE IN THE AFRICAN REGION January February 2017 (Vol 5 issue N 1) District data completeness and coverage of DTP3 containing vaccine per
IMMUNIZATION VACCINE DEVELOPMENT MONTHLY IMMUNIZATION AND POLIO UPDATE IN THE AFRICAN REGION January February 2017 (Vol 5 issue N 1) District data completeness and coverage of DTP3 containing vaccine per
REGIONAL ALLIANCE FOR NATIONAL REGULATORY AUTHORITIES FOR VACCINES IN THE WESTERN PACIFIC. second edition
 REGIONAL ALLIANCE FOR NATIONAL REGULATORY AUTHORITIES FOR VACCINES IN THE WESTERN PACIFIC second edition World Health Organization 2014 The designations employed and the presentation of the material in
REGIONAL ALLIANCE FOR NATIONAL REGULATORY AUTHORITIES FOR VACCINES IN THE WESTERN PACIFIC second edition World Health Organization 2014 The designations employed and the presentation of the material in
ASLM Building laboratory capacity in Africa in a sustainable way
 ASLM Building laboratory capacity in Africa in a sustainable way Tsehaynesh Messele, PhD ASLM, Chief Executive Officer Annual AMDS meeting September 29-30, 2014 Geneva Laboratory capacity gaps in Africa
ASLM Building laboratory capacity in Africa in a sustainable way Tsehaynesh Messele, PhD ASLM, Chief Executive Officer Annual AMDS meeting September 29-30, 2014 Geneva Laboratory capacity gaps in Africa
511,000 (57% new cases) ~50,000 ~30,000
 Latest global TB estimates - 2007 (Updated Mar 2009) All forms of TB Greatest number of cases in Asia; greatest rates per capita in Africa Multidrug-resistant TB (MDR-TB) Estimated number of cases 9.27
Latest global TB estimates - 2007 (Updated Mar 2009) All forms of TB Greatest number of cases in Asia; greatest rates per capita in Africa Multidrug-resistant TB (MDR-TB) Estimated number of cases 9.27
IMMUNIZATION VACCINES & EMERGENCIES
 No report No data No data IMMUNIZATION VACCINES & EMERGENCIES ROUTINE IMMUNIZATION PERFORMANCE IN THE AFRICAN REGION October 2013 issue Number of children vaccinated with the 3 rd dose of DTPcontaining
No report No data No data IMMUNIZATION VACCINES & EMERGENCIES ROUTINE IMMUNIZATION PERFORMANCE IN THE AFRICAN REGION October 2013 issue Number of children vaccinated with the 3 rd dose of DTPcontaining
Ouagadougou Declaration
 Ouagadougou Declaration on Primary Health Care and Health Systems in Africa: Achieving Better Health for Africa in the New Millennium A declaration by the Members States of the WHO African Region 30 April
Ouagadougou Declaration on Primary Health Care and Health Systems in Africa: Achieving Better Health for Africa in the New Millennium A declaration by the Members States of the WHO African Region 30 April
AIDS in Africa. An Update. Basil Reekie
 AIDS in Africa An Update Basil Reekie Contents General Statistics The trend of HIV in Africa Ugandan experience UNAIDS 2006 Latest African Statistics by Country HIV Intervention Light at the end of the
AIDS in Africa An Update Basil Reekie Contents General Statistics The trend of HIV in Africa Ugandan experience UNAIDS 2006 Latest African Statistics by Country HIV Intervention Light at the end of the
( A JICA-IRRI-PhilRice Initiative) Presented by Noel Magor, Head Unit Impact Acceleration and Training Center, IRRI
 ( A JICA-IRRI-PhilRice Initiative) Presented by Noel Magor, Head Unit Impact Acceleration and Training Center, IRRI Long partnership between; IRRI & PhilRice IRRI & JICA PhilRice & JICA The Season Long
( A JICA-IRRI-PhilRice Initiative) Presented by Noel Magor, Head Unit Impact Acceleration and Training Center, IRRI Long partnership between; IRRI & PhilRice IRRI & JICA PhilRice & JICA The Season Long
Update on Meningococcal A Vaccine Development and Introduction
 Update on Meningococcal A Vaccine Development and Introduction WHO Product Development for Vaccines Advisory Committee Meeting (PD-VAC) @ Geneva, 7-9 September 2015 Dr Marie-Pierre Preziosi, WHO Initiative
Update on Meningococcal A Vaccine Development and Introduction WHO Product Development for Vaccines Advisory Committee Meeting (PD-VAC) @ Geneva, 7-9 September 2015 Dr Marie-Pierre Preziosi, WHO Initiative
THE CARE WE PROMISE FACTS AND FIGURES 2017
 THE CARE WE PROMISE FACTS AND FIGURES 2017 2 SOS CHILDREN S VILLAGES INTERNATIONAL WHERE WE WORK Facts and Figures 2017 205 58 79 families and transit 31 Foster homes 162 8 3 173 214 2 115 159 136 148
THE CARE WE PROMISE FACTS AND FIGURES 2017 2 SOS CHILDREN S VILLAGES INTERNATIONAL WHERE WE WORK Facts and Figures 2017 205 58 79 families and transit 31 Foster homes 162 8 3 173 214 2 115 159 136 148
IMMUNIZATION & VACCINE PREVENTABLE DISEASES
 IMMUNIZATION & VACCINE PREVENTABLE DISEASES MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION July-August 2015 (Vol 3, issue N 6 ) Special issue on WHO/UNICEF Estimates of National Immunization Coverage
IMMUNIZATION & VACCINE PREVENTABLE DISEASES MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION July-August 2015 (Vol 3, issue N 6 ) Special issue on WHO/UNICEF Estimates of National Immunization Coverage
Current State of Global HIV Care Continua. Reuben Granich 1, Somya Gupta 1, Irene Hall 2, John Aberle-Grasse 2, Shannon Hader 2, Jonathan Mermin 2
 Current State of Global HIV Care Continua Reuben Granich 1, Somya Gupta 1, Irene Hall 2, John Aberle-Grasse 2, Shannon Hader 2, Jonathan Mermin 2 1) International Association of Providers of AIDS Care
Current State of Global HIV Care Continua Reuben Granich 1, Somya Gupta 1, Irene Hall 2, John Aberle-Grasse 2, Shannon Hader 2, Jonathan Mermin 2 1) International Association of Providers of AIDS Care
Procedure for the expedited review of imported prequalified vaccines with view to granting a marketing authorization
 Procedure for the expedited review of imported prequalified vaccines with view to granting a marketing authorization Prequalification Stakeholders Meeting Geneva, CICG 4 and 5 April 2011 Dr. Nora Dellepiane
Procedure for the expedited review of imported prequalified vaccines with view to granting a marketing authorization Prequalification Stakeholders Meeting Geneva, CICG 4 and 5 April 2011 Dr. Nora Dellepiane
PROGRESS REPORT ON THE ROAD MAP FOR ACCELERATING THE ATTAINMENT OF THE MILLENNIUM DEVELOPMENT GOALS RELATED TO MATERNAL AND NEWBORN HEALTH IN AFRICA
 5 July 2011 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-first session Yamoussoukro, Côte d Ivoire, 29 August 2 September 2011 Provisional agenda item 17.1 PROGRESS REPORT ON THE ROAD MAP FOR
5 July 2011 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-first session Yamoussoukro, Côte d Ivoire, 29 August 2 September 2011 Provisional agenda item 17.1 PROGRESS REPORT ON THE ROAD MAP FOR
Collaborative procedure for licensing Prequalified vaccines
 Collaborative procedure for licensing Prequalified vaccines DCVMN meeting Shanghai, China March 2015 World Health Organization, EMP/RHT/PQT rodriguezhernandezc@who.int meekd@who.int Strategic priorities
Collaborative procedure for licensing Prequalified vaccines DCVMN meeting Shanghai, China March 2015 World Health Organization, EMP/RHT/PQT rodriguezhernandezc@who.int meekd@who.int Strategic priorities
IMMUNIZATION VACCINE DEVELOPMENT
 IMMUNIZATION VACCINE DEVELOPMENT MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION July-August 2016 (Vol 4, issue N 5) Special issue on 2015 WHO/UNICEF Estimates of National Immunization Coverage (WUENIC)
IMMUNIZATION VACCINE DEVELOPMENT MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION July-August 2016 (Vol 4, issue N 5) Special issue on 2015 WHO/UNICEF Estimates of National Immunization Coverage (WUENIC)
IMMUNIZATION VACCINE DEVELOPMENT
 IMMUNIZATION VACCINE DEVELOPMENT MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION November December 2016 (Vol 4, issue N 7) District data completeness and coverage of DTP3 containing vaccine per country
IMMUNIZATION VACCINE DEVELOPMENT MONTHLY IMMUNIZATION UPDATE IN THE AFRICAN REGION November December 2016 (Vol 4, issue N 7) District data completeness and coverage of DTP3 containing vaccine per country
Medicines Regulation, Regulatory Harmonization, Global Initiatives. Tonya Villafana, PhD, MPH Senior Health Specialist, World Bank
 Medicines Regulation, Regulatory Harmonization, Global Initiatives Tonya Villafana, PhD, MPH Senior Health Specialist, World Bank September, 13, 2012 Presentation Outline The importance of regulation Attributes
Medicines Regulation, Regulatory Harmonization, Global Initiatives Tonya Villafana, PhD, MPH Senior Health Specialist, World Bank September, 13, 2012 Presentation Outline The importance of regulation Attributes
1) SO1: We would like to suggest that the indicator used to measure vaccine hesitancy be DTP 1 to measles first dose dropout.
 To SAGE Secretariat, WHO Dear Professor Helen Rees, Dear Dr. Jean Marie Okwo-Bele, On behalf of the Civil Society Constituency of the GAVI Alliance, we would like to thank SAGE and its members for the
To SAGE Secretariat, WHO Dear Professor Helen Rees, Dear Dr. Jean Marie Okwo-Bele, On behalf of the Civil Society Constituency of the GAVI Alliance, we would like to thank SAGE and its members for the
Expert Group Meeting on the Regional Report for the African Gender and Development Index
 Expert Group Meeting on the Regional Report for the African Gender and Development Index 9-10 October 2017 United Nations Conference Centre, Addis Ababa, Ethiopia Aide Memoire July 2017 I. Background and
Expert Group Meeting on the Regional Report for the African Gender and Development Index 9-10 October 2017 United Nations Conference Centre, Addis Ababa, Ethiopia Aide Memoire July 2017 I. Background and
ASLM Anti-microbial Resistance in Africa and Global Health Security. 9TH INTEREST WORKSHOP May 2015 Harare
 ASLM Anti-microbial Resistance in Africa and Global Health Security 9TH INTEREST WORKSHOP 2015 8 May 2015 Harare Recent Jim O Neil Report commissioned by the UK government predicts dire consequences of
ASLM Anti-microbial Resistance in Africa and Global Health Security 9TH INTEREST WORKSHOP 2015 8 May 2015 Harare Recent Jim O Neil Report commissioned by the UK government predicts dire consequences of
Status Report on WSS MDG Roadmaps and Country Status Overviews WSP Africa
 Status Report on WSS MDG Roadmaps and Country Status Overviews WSP Africa Ede Ijjasz Global Manager Water and Sanitation Program World Water Forum, Mexico, March 18, 2006 Outline 1. The Water Supply and
Status Report on WSS MDG Roadmaps and Country Status Overviews WSP Africa Ede Ijjasz Global Manager Water and Sanitation Program World Water Forum, Mexico, March 18, 2006 Outline 1. The Water Supply and
Malaria Funding. Richard W. Steketee MACEPA, PATH. April World Malaria Day 2010, Seattle WA
 Malaria Funding Richard W. Steketee MACEPA, PATH April World Malaria Day 2010, Seattle WA Malaria Funding Is there a plan? Is there money? Where does the money come from? Is the money moving efficiently?
Malaria Funding Richard W. Steketee MACEPA, PATH April World Malaria Day 2010, Seattle WA Malaria Funding Is there a plan? Is there money? Where does the money come from? Is the money moving efficiently?
Sourcing of ARVs & HIV diagnostics. Procurement for Impact P4i
 Sourcing of ARVs & HIV diagnostics Procurement for Impact P4i AMDS ANNUAL STAKEHOLDERS AND PARTNERS MEETING 29 September 2014 Global Fund: Procurement for Impact: P4i Our objective was straightforward:
Sourcing of ARVs & HIV diagnostics Procurement for Impact P4i AMDS ANNUAL STAKEHOLDERS AND PARTNERS MEETING 29 September 2014 Global Fund: Procurement for Impact: P4i Our objective was straightforward:
Vaccine assessment for prequalification
 Vaccine assessment for prequalification Briefing on Vaccine Prequalification for manufacturers DCVMN meeting, Kunming, China, 13 March 2016 Olivier Lapujade World Health Organization, EMP/RHT/PQT lapujadeo@who.int
Vaccine assessment for prequalification Briefing on Vaccine Prequalification for manufacturers DCVMN meeting, Kunming, China, 13 March 2016 Olivier Lapujade World Health Organization, EMP/RHT/PQT lapujadeo@who.int
What is this document and who is it for?
 Measles and Rubella Initiative s Standard Operating Procedures for Accessing Support for Measles and Rubella Supplementary Immunization Activities During 2016 In the context of measles and rubella elimination
Measles and Rubella Initiative s Standard Operating Procedures for Accessing Support for Measles and Rubella Supplementary Immunization Activities During 2016 In the context of measles and rubella elimination
TFI Proceedings, Recommendations and implications for 2005
 January 2005 N 053 TFI 2004 - Proceedings, Recommendations and implications for 2005 The 12th meeting of the Task Force on Immunization (TFI) in Africa and the 11th meeting of the Africa Regional Inter-
January 2005 N 053 TFI 2004 - Proceedings, Recommendations and implications for 2005 The 12th meeting of the Task Force on Immunization (TFI) in Africa and the 11th meeting of the Africa Regional Inter-
Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005
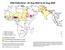 Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005 Wild virus type 1 Wild virus type 3 Wild virus type 1 & 3 Endemic countries Re-established transmission countries Case or outbreak following importation *Excludes
Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005 Wild virus type 1 Wild virus type 3 Wild virus type 1 & 3 Endemic countries Re-established transmission countries Case or outbreak following importation *Excludes
JOINT TB AND HIV PROGRAMMING
 JOINT TB AND HIV PROGRAMMING Haileyesus Getahun, WHO. On behalf of the Global Fund Interagency TB and HIV Working Group (Global Fund, PEPFAR, Stop TB Partnership, UNAIDS, WHO) I was admitted in a hospital
JOINT TB AND HIV PROGRAMMING Haileyesus Getahun, WHO. On behalf of the Global Fund Interagency TB and HIV Working Group (Global Fund, PEPFAR, Stop TB Partnership, UNAIDS, WHO) I was admitted in a hospital
Market Updates: Routine Vaccine Introductions IPV. Vaccine Industry Consultation October 2018 UNICEF Supply Division
 Market Updates: Routine Vaccine Introductions IPV Vaccine Industry Consultation October 2018 UNICEF Supply Division Outline Review of rapid introduction of IPV Overview of the current situation for 2018
Market Updates: Routine Vaccine Introductions IPV Vaccine Industry Consultation October 2018 UNICEF Supply Division Outline Review of rapid introduction of IPV Overview of the current situation for 2018
History, implementation and impact of MenA conjugate on disease burden in Africa
 History, implementation and impact of MenA conjugate on disease burden in Africa First Regional Meningococcal Symposium, 19-20 March 2012 Buenos Aires, Argentina Co-hosted by the Sabin Vaccine Institute
History, implementation and impact of MenA conjugate on disease burden in Africa First Regional Meningococcal Symposium, 19-20 March 2012 Buenos Aires, Argentina Co-hosted by the Sabin Vaccine Institute
Presentation by Dr Philippe Douste-Blazy. Chair of UNITAID Special Representative of the UN SG for Innovative Financing for Development
 Presentation by Dr Philippe Douste-Blazy Chair of UNITAID Special Representative of the UN SG for Innovative Financing for Development The Millennium Goals : Hope for Humanity 2000 UN Millennium Summit
Presentation by Dr Philippe Douste-Blazy Chair of UNITAID Special Representative of the UN SG for Innovative Financing for Development The Millennium Goals : Hope for Humanity 2000 UN Millennium Summit
IMMUNIZATION VACCINE DEVELOPMENT
 IMMUNIZATION VACCINE DEVELOPMENT IMMUNIZATION AND POLIO UPDATE IN THE AFRICAN REGION July August 2017 (Vol 5 issue N 4) Special issue featuring WHO/UNICEF Estimates of National Immunization Coverage (WUENIC
IMMUNIZATION VACCINE DEVELOPMENT IMMUNIZATION AND POLIO UPDATE IN THE AFRICAN REGION July August 2017 (Vol 5 issue N 4) Special issue featuring WHO/UNICEF Estimates of National Immunization Coverage (WUENIC
Objectives of Rotavirus Surveillance
 Objectives of Rotavirus Surveillance Contribute data to estimate the burden of rotavirus diarrhea in children < 5 children Document circulating rotavirus strains in the AFR region Support awareness and
Objectives of Rotavirus Surveillance Contribute data to estimate the burden of rotavirus diarrhea in children < 5 children Document circulating rotavirus strains in the AFR region Support awareness and
The MenAfrivac Experience A Successful Approach. Dr Bernard FRITZELL BFL conseils France
 The MenAfrivac Experience A Successful Approach Dr Bernard FRITZELL BFL conseils France 1 2 1996 Ministers of Health and Interior from 16 African countries recognized epidemic meningitis as a high priority
The MenAfrivac Experience A Successful Approach Dr Bernard FRITZELL BFL conseils France 1 2 1996 Ministers of Health and Interior from 16 African countries recognized epidemic meningitis as a high priority
Aboubacar Kampo Chief of Health UNICEF Nigeria
 Aboubacar Kampo Chief of Health UNICEF Nigeria Many thanks to UNICEF colleagues in Supply Division-Copenhagen and NY for contributing to this presentation Thirty-five countries are responsible for 98%
Aboubacar Kampo Chief of Health UNICEF Nigeria Many thanks to UNICEF colleagues in Supply Division-Copenhagen and NY for contributing to this presentation Thirty-five countries are responsible for 98%
PROGRESS REPORT ON CHILD SURVIVAL: A STRATEGY FOR THE AFRICAN REGION. Information Document CONTENTS
 29 June 2009 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-ninth session Kigali, Republic of Rwanda, 31 August 4 September 2009 Provisional agenda item 9.2 PROGRESS REPORT ON CHILD SURVIVAL: A
29 June 2009 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-ninth session Kigali, Republic of Rwanda, 31 August 4 September 2009 Provisional agenda item 9.2 PROGRESS REPORT ON CHILD SURVIVAL: A
PROGRESS REPORT ON DECADE OF TRADITIONAL MEDICINE IN THE AFRICAN REGION. Progress Report. CONTENTS Paragraphs BACKGROUND PROGRESS MADE...
 5 July 2011 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-first session Yamoussoukro, Côte d Ivoire, 29 August 2 September 2011 Provisional agenda item 17.2 PROGRESS REPORT ON DECADE OF TRADITIONAL
5 July 2011 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-first session Yamoussoukro, Côte d Ivoire, 29 August 2 September 2011 Provisional agenda item 17.2 PROGRESS REPORT ON DECADE OF TRADITIONAL
AFRICA. The continent of All challenges
 AFRICA The continent of All challenges Part of Resources and burden of the disease in Africa Africa=25% Rest of the World=75% Africa=0.6% Rest of the World=99.4% 0% 20% 40% 60% 80% 100% 57 Critical shortage
AFRICA The continent of All challenges Part of Resources and burden of the disease in Africa Africa=25% Rest of the World=75% Africa=0.6% Rest of the World=99.4% 0% 20% 40% 60% 80% 100% 57 Critical shortage
CONTENTS. Paragraphs I. BACKGROUND II. PROGRESS REPORT ON THE AFRICAN REGIONAL IMMUNIZATION STRATEGIC PLAN
 23 September 2013 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-third session Brazzaville, Republic of Congo, 2 6 September, 2013 Agenda item 14 IMMUNIZATION IN THE AFRICAN REGION: PROGRESS REPORT
23 September 2013 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-third session Brazzaville, Republic of Congo, 2 6 September, 2013 Agenda item 14 IMMUNIZATION IN THE AFRICAN REGION: PROGRESS REPORT
Excellence and Originality from Necessity: Palliative Care in Africa. Dr Emmanuel Luyirika Executive Director, African Palliative Care Association
 Excellence and Originality from Necessity: Palliative Care in Africa Dr Emmanuel Luyirika Executive Director, African Palliative Care Association Summary of the presentation 1. Introduction 2. Background
Excellence and Originality from Necessity: Palliative Care in Africa Dr Emmanuel Luyirika Executive Director, African Palliative Care Association Summary of the presentation 1. Introduction 2. Background
Comments on this bulletin are welcome. Please to Dr. Mary Agócs
 Global Invasive Bacterial Vaccine Preventable Diseases (IB-VPD) Information and Surveillance Bulletin Reporting Period: January through December 2010 Volume 4: October 2011 The World Health Organization
Global Invasive Bacterial Vaccine Preventable Diseases (IB-VPD) Information and Surveillance Bulletin Reporting Period: January through December 2010 Volume 4: October 2011 The World Health Organization
Pneumococcal Conjugate Vaccine: Current Supply & Demand Outlook. UNICEF Supply Division
 Pneumococcal Conjugate Vaccine: Current Supply & Demand Outlook UNICEF Supply Division Update: October 2013 0 Pneumococcal Conjugate Vaccine (PCV) Supply & Demand Outlook October 2013 Update Key updates
Pneumococcal Conjugate Vaccine: Current Supply & Demand Outlook UNICEF Supply Division Update: October 2013 0 Pneumococcal Conjugate Vaccine (PCV) Supply & Demand Outlook October 2013 Update Key updates
Prioritizing Emergency Polio Eradication Activities
 Prioritizing Emergency Polio Eradication Activities Managing the Financing Gap the other half of the Emergency Hamid Jafari GPEI Financing 2012-13: Budget = $2.23 b - Confirmed contributions = $1.14 b
Prioritizing Emergency Polio Eradication Activities Managing the Financing Gap the other half of the Emergency Hamid Jafari GPEI Financing 2012-13: Budget = $2.23 b - Confirmed contributions = $1.14 b
Vaccine Development in the Developing World; past, present and future: SEAR Perspective
 Speech by Dr Samlee Plianbangchang Regional Director, WHO South-East Asia Vaccine Development in the Developing World; past, present and future: SEAR Perspective 15 June 2010 Bangkok, Thailand Vaccine
Speech by Dr Samlee Plianbangchang Regional Director, WHO South-East Asia Vaccine Development in the Developing World; past, present and future: SEAR Perspective 15 June 2010 Bangkok, Thailand Vaccine
CANCER OF THE CERVIX IN THE AFRICAN REGION: CURRENT SITUATION AND WAY FORWARD
 23 June 2010 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: FRENCH Sixtieth session Malabo, Equatorial Guinea, 30 August 3 September 2010 Provisional agenda item 7.4 CANCER OF THE CERVIX IN THE AFRICAN REGION:
23 June 2010 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: FRENCH Sixtieth session Malabo, Equatorial Guinea, 30 August 3 September 2010 Provisional agenda item 7.4 CANCER OF THE CERVIX IN THE AFRICAN REGION:
WHO VACCINE SUPPLY & QUALITY SUPPORT FOR NATIONAL IMMUNIZATION PROGRAMMES
 WHO VACCINE SUPPLY & QUALITY SUPPORT FOR NATIONAL IMMUNIZATION PROGRAMMES Immunization and vaccine development in the last decade Since the Millennium Summit in 2000, immunization substantial international
WHO VACCINE SUPPLY & QUALITY SUPPORT FOR NATIONAL IMMUNIZATION PROGRAMMES Immunization and vaccine development in the last decade Since the Millennium Summit in 2000, immunization substantial international
POLIOMYELITIS ERADICATION: PROGRESS REPORT. Information Document CONTENTS BACKGROUND PROGRESS MADE NEXT STEPS... 12
 5 August 9 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-ninth session Kigali, Republic of Rwanda, August 4 September 9 POLIOMYELITIS ERADICATION: PROGRESS REPORT Information Document CONTENTS
5 August 9 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Fifty-ninth session Kigali, Republic of Rwanda, August 4 September 9 POLIOMYELITIS ERADICATION: PROGRESS REPORT Information Document CONTENTS
Rotavirus Vaccine: Supply & Demand Update. UNICEF Supply Division
 Rotavirus Vaccine: & Demand Update UNICEF Division July 0 Rotavirus Vaccine (RV): & Demand Update July This update provides new information on countries and their scheduled RV introductions, forecasted
Rotavirus Vaccine: & Demand Update UNICEF Division July 0 Rotavirus Vaccine (RV): & Demand Update July This update provides new information on countries and their scheduled RV introductions, forecasted
Global EHS Resource Center
 Global EHS Resource Center Understand environmental and workplace safety requirements that affect your global operations. 800.372.1033 bna.com/gelw Global EHS Resource Center This comprehensive research
Global EHS Resource Center Understand environmental and workplace safety requirements that affect your global operations. 800.372.1033 bna.com/gelw Global EHS Resource Center This comprehensive research
CEO Board report Seth Berkley MD CEO
 CEO Board report Seth Berkley MD CEO Dhaka, Bangladesh, 16 17 November 2011 Bangladesh, November 2011 Meningitis Incidence Children < 12 months Dhaka Shishu / Shishu Shahsta Catchment Area 2008 2011 1800
CEO Board report Seth Berkley MD CEO Dhaka, Bangladesh, 16 17 November 2011 Bangladesh, November 2011 Meningitis Incidence Children < 12 months Dhaka Shishu / Shishu Shahsta Catchment Area 2008 2011 1800
Outline. World Health Organization 14 July, Strengthening for capacity to regulate influenza vaccines
 Strengthening for capacity to regulate influenza vaccines Outline WHO reorganization and Regulatory Systems Strengthening (RSS) Team WHO Regulatory Systems Strengthening Department of Essential Medicines
Strengthening for capacity to regulate influenza vaccines Outline WHO reorganization and Regulatory Systems Strengthening (RSS) Team WHO Regulatory Systems Strengthening Department of Essential Medicines
Vaccine Production and Regulation
 Vaccine Production and Regulation Dr Houda LANGAR Essential Vaccines and Biologicals Policy Department of Health Systems & Services Development Capacity Development Workshop on Health System Development
Vaccine Production and Regulation Dr Houda LANGAR Essential Vaccines and Biologicals Policy Department of Health Systems & Services Development Capacity Development Workshop on Health System Development
Health systems and HIV: advocacy. Interagency Coalition on AIDS and Development
 Health systems and HIV: Priorities for civil society advocacy Michelle Munro Interagency Coalition on AIDS and Development 1 Overview GTAG, civil society and health systems advocacy Health systems and
Health systems and HIV: Priorities for civil society advocacy Michelle Munro Interagency Coalition on AIDS and Development 1 Overview GTAG, civil society and health systems advocacy Health systems and
The Promise of Introducing Rubella Containing Vaccines on the Impact of Rubella and Measles Control
 The Promise of Introducing Rubella Containing Vaccines on the Impact of Rubella and Measles Control GAVI Partners Forum 4 6 December 2012, Dar es Salaam, Tanzania Maya van den Ent, MPH, UNICEF Peter Strebel,
The Promise of Introducing Rubella Containing Vaccines on the Impact of Rubella and Measles Control GAVI Partners Forum 4 6 December 2012, Dar es Salaam, Tanzania Maya van den Ent, MPH, UNICEF Peter Strebel,
Annex 2 A. Regional profile: West Africa
 Annex 2 A. Regional profile: West Africa 355 million people at risk for malaria in 215 297 million at high risk A. Parasite prevalence, 215 Funding for malaria increased from US$ 233 million to US$ 262
Annex 2 A. Regional profile: West Africa 355 million people at risk for malaria in 215 297 million at high risk A. Parasite prevalence, 215 Funding for malaria increased from US$ 233 million to US$ 262
HIV DIAGNOSTIC TESTS IN LOW- AND MIDDLE-INCOME COUNTRIES: FORECASTS OF GLOBAL DEMAND FOR
 HIV DIAGNOSTIC TESTS IN LOW- AND MIDDLE-INCOME COUNTRIES: FORECASTS OF GLOBAL DEMAND FOR 2015 2020 Presented by Vincent Habiyambere (WHO), Biyi Adesina (Avenir Health) and Meghan Wareham (CHAI) on behalf
HIV DIAGNOSTIC TESTS IN LOW- AND MIDDLE-INCOME COUNTRIES: FORECASTS OF GLOBAL DEMAND FOR 2015 2020 Presented by Vincent Habiyambere (WHO), Biyi Adesina (Avenir Health) and Meghan Wareham (CHAI) on behalf
Quality Assurance Policy. for the. Procurement of HIV Point-of-Care Technology. under the UNITAID Grant
 Quality Assurance Policy for the Procurement of HIV Point-of-Care Technology under the UNITAID Grant UNICEF Supply Division Quality Assurance Centre and Health Technology Centre Revisions Version Date
Quality Assurance Policy for the Procurement of HIV Point-of-Care Technology under the UNITAID Grant UNICEF Supply Division Quality Assurance Centre and Health Technology Centre Revisions Version Date
FP2020 Expert Advisory Community Webinar
 FP2020 Expert Advisory Community Webinar Oct. 24, 2017 www.familyplanning2020.org #FP2020Progress @FP2020Global Facebook.com/familyplanning2020 AGENDA Welcome Key Updates: 2017 Family Planning Summit to
FP2020 Expert Advisory Community Webinar Oct. 24, 2017 www.familyplanning2020.org #FP2020Progress @FP2020Global Facebook.com/familyplanning2020 AGENDA Welcome Key Updates: 2017 Family Planning Summit to
2. Treatment coverage: 3. Quality of care: 1. Access to diagnostic services:
 The theme for World TB Day 2014 is Reach the missed 3 million. Every year 3 million people who fall ill with TB are missed by health systems and do not always get the TB services that they need and deserve.
The theme for World TB Day 2014 is Reach the missed 3 million. Every year 3 million people who fall ill with TB are missed by health systems and do not always get the TB services that they need and deserve.
EBOLA SITUATION REPORT
 4 MARCH 205 CORRIGENDUM CASES/ DEATHS (data up to March 205) Guinea Liberia Sierra Leone Mali Nigeria Senegal Spain United Kingdom United States of America Total 8 6 20 8 0 0 0 4 329 229 47 3546 9249 9807
4 MARCH 205 CORRIGENDUM CASES/ DEATHS (data up to March 205) Guinea Liberia Sierra Leone Mali Nigeria Senegal Spain United Kingdom United States of America Total 8 6 20 8 0 0 0 4 329 229 47 3546 9249 9807
Funding for AIDS: The World Bank s Role. Yolanda Tayler, WB Bi-regional Workshop for the Procurement of ARVs Phnom Penh, Cambodia
 Funding for AIDS: The World Bank s Role Yolanda Tayler, WB Bi-regional Workshop for the Procurement of ARVs Phnom Penh, Cambodia Outline New resources needs estimates Bridging the gap Global overview of
Funding for AIDS: The World Bank s Role Yolanda Tayler, WB Bi-regional Workshop for the Procurement of ARVs Phnom Penh, Cambodia Outline New resources needs estimates Bridging the gap Global overview of
EBOLA SITUATION REPORT
 09 SEPTEMBER 2015 SUMMARY There were 2 confirmed cases of Ebola virus disease (EVD) reported in the week to 6 September: 1 in Guinea and 1 in Sierra Leone. Overall case incidence has remained stable at
09 SEPTEMBER 2015 SUMMARY There were 2 confirmed cases of Ebola virus disease (EVD) reported in the week to 6 September: 1 in Guinea and 1 in Sierra Leone. Overall case incidence has remained stable at
ANNEX Page. AFR/RC61/11 4 July 2011 ORIGINAL: ENGLISH REGIONAL COMMITTEE FOR AFRICA
 4 July 2011 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-first session Yamoussoukro, Côte d Ivoire, 29 August 2 September 2011 Provisional agenda item 16 PROGRESS REPORT ON POLIOMYELITIS ERADICATION
4 July 2011 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-first session Yamoussoukro, Côte d Ivoire, 29 August 2 September 2011 Provisional agenda item 16 PROGRESS REPORT ON POLIOMYELITIS ERADICATION
Lessons learned from the IeDEA West Africa Collaboration
 Lessons learned from the IeDEA West Africa Collaboration François DABIS with the contribution of Didier Koumavi EKOUEVI INSERM U-897, Bordeaux, France, Programme PACCI, ANRS site, Abidjan, Côte d Ivoire
Lessons learned from the IeDEA West Africa Collaboration François DABIS with the contribution of Didier Koumavi EKOUEVI INSERM U-897, Bordeaux, France, Programme PACCI, ANRS site, Abidjan, Côte d Ivoire
ENHANCING THE ROLE OF TRADITIONAL MEDICINE IN HEALTH SYSTEMS: A STRATEGY FOR THE AFRICAN REGION. Report of the Secretariat EXECUTIVE SUMMARY
 3 September 2013 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-third session Brazzaville, Republic of Congo, 2 6 September 2013 Agenda item 10 ENHANCING THE ROLE OF TRADITIONAL MEDICINE IN HEALTH
3 September 2013 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-third session Brazzaville, Republic of Congo, 2 6 September 2013 Agenda item 10 ENHANCING THE ROLE OF TRADITIONAL MEDICINE IN HEALTH
Global Fund Results Fact Sheet Mid-2011
 Global Fund Results Fact Sheet Mid-2011 This fact sheet outlines some of the common questions and answers regarding results reported by Global Fund-supported programs, including the principles and approach
Global Fund Results Fact Sheet Mid-2011 This fact sheet outlines some of the common questions and answers regarding results reported by Global Fund-supported programs, including the principles and approach
Comparative Analyses of Adolescent Nutrition Indicators
 Comparative Analyses of Adolescent Nutrition Indicators Rukundo K. Benedict, PhD The DHS Program Stakeholders Consultation on Adolescent Girls Nutrition: Evidence, Guidance, and Gaps October 30 31, 2017
Comparative Analyses of Adolescent Nutrition Indicators Rukundo K. Benedict, PhD The DHS Program Stakeholders Consultation on Adolescent Girls Nutrition: Evidence, Guidance, and Gaps October 30 31, 2017
EBOLA SITUATION REPORT
 25 FEBRUARY 205 CASES/ DEATHS (data up to 22 February 205) Guinea Liberia Sierra Leone Mali Nigeria Senegal Spain United Kingdom United States of America Total 8 6 20 8 0 0 0 4 355 209 4037 346 9238 9604
25 FEBRUARY 205 CASES/ DEATHS (data up to 22 February 205) Guinea Liberia Sierra Leone Mali Nigeria Senegal Spain United Kingdom United States of America Total 8 6 20 8 0 0 0 4 355 209 4037 346 9238 9604
THE WHO FRAMEWORK CONVENTION ON TOBACCO CONTROL: 10 YEARS OF IMPLEMENTATION IN THE AFRICAN REGION
 THE WHO FRAMEWORK CONVENTION ON TOBACCO CONTROL: 10 YEARS OF IMPLEMENTATION IN THE AFRICAN REGION AFRO Library Cataloguing-in-Publication Data The WHO Framework Convention on Tobacco Control: 10 years
THE WHO FRAMEWORK CONVENTION ON TOBACCO CONTROL: 10 YEARS OF IMPLEMENTATION IN THE AFRICAN REGION AFRO Library Cataloguing-in-Publication Data The WHO Framework Convention on Tobacco Control: 10 years
Eligibility List 2018
 The Global Fund s 2017-2022 strategy and allocation-based approach enables strategic investment to accelerate the end of HIV/AIDS, tuberculosis and malaria and build resilient and sustainable systems for
The Global Fund s 2017-2022 strategy and allocation-based approach enables strategic investment to accelerate the end of HIV/AIDS, tuberculosis and malaria and build resilient and sustainable systems for
Global Fund ARV Fact Sheet 1 st June, 2009
 Global Fund ARV Fact Sheet 1 st June, 2009 This fact sheet outlines the principles and approach in determining the number of people on antiretroviral drugs (ARVs) for HIV/AIDS treatment, with a breakdown
Global Fund ARV Fact Sheet 1 st June, 2009 This fact sheet outlines the principles and approach in determining the number of people on antiretroviral drugs (ARVs) for HIV/AIDS treatment, with a breakdown
O c t o b e r 1 0,
 STUDENT VOICES FROM THE FIELD: WORLD HEALTH ORGANIZATION GENEVA, SWITZERLAND O c t o b e r 1 0, 2 0 1 6 M o l l y P e z z u l o University at Albany School of Public Health MPH Epidemiology Candidate 17
STUDENT VOICES FROM THE FIELD: WORLD HEALTH ORGANIZATION GENEVA, SWITZERLAND O c t o b e r 1 0, 2 0 1 6 M o l l y P e z z u l o University at Albany School of Public Health MPH Epidemiology Candidate 17
Global Update of the 2010 GIVS Goals
 Global Update of the 2010 GIVS Goals Global Immunization Meeting New York, 17-19 Feb 2009 JM Okwo-Bele, WHO P. Salama, UNICEF Projected Changes in Under-5 yr mortality due to VPDs M illio n s 3.0 2.5 2.0
Global Update of the 2010 GIVS Goals Global Immunization Meeting New York, 17-19 Feb 2009 JM Okwo-Bele, WHO P. Salama, UNICEF Projected Changes in Under-5 yr mortality due to VPDs M illio n s 3.0 2.5 2.0
EBOLA SITUATION REPORT
 EBOLA SITUATION REPORT 11 FEBRUARY 2015 CASES/ DEATHS (data up to 8 February 2015) Guinea Liberia Sierra Leone Mali Nigeria Senegal Spain United Kingdom United States of America Total 8 6 20 8 1 0 1 0
EBOLA SITUATION REPORT 11 FEBRUARY 2015 CASES/ DEATHS (data up to 8 February 2015) Guinea Liberia Sierra Leone Mali Nigeria Senegal Spain United Kingdom United States of America Total 8 6 20 8 1 0 1 0
MEASLES ELIMINATION BY 2020: A STRATEGY FOR THE AFRICAN REGION. Report of the Secretariat. Executive Summary
 16 June 2011 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-first session Yamoussoukro, Côte d Ivoire, 29 August 2 September 2011 Provisional agenda item 13 MEASLES ELIMINATION BY 2020: A STRATEGY
16 June 2011 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-first session Yamoussoukro, Côte d Ivoire, 29 August 2 September 2011 Provisional agenda item 13 MEASLES ELIMINATION BY 2020: A STRATEGY
GABON. Neglected tropical disease treatment report profile for mass treatment of NTDs
 GABON Neglected tropical disease treatment report 2017 1 2017 profile for mass treatment of NTDs NEGLECTED TROPICAL DISEASES Neglected tropical diseases (NTDs) are a group of preventable and treatable
GABON Neglected tropical disease treatment report 2017 1 2017 profile for mass treatment of NTDs NEGLECTED TROPICAL DISEASES Neglected tropical diseases (NTDs) are a group of preventable and treatable
SGCEP SCIE 1121 Environmental Science Spring 2012 Section Steve Thompson:
 SGCEP SCIE 1121 Environmental Science Spring 2012 Section 20531 Steve Thompson: steventhompson@sgc.edu http://www.bioinfo4u.net/ 1 First, a brief diversion... Into... how to do better on the next exam,
SGCEP SCIE 1121 Environmental Science Spring 2012 Section 20531 Steve Thompson: steventhompson@sgc.edu http://www.bioinfo4u.net/ 1 First, a brief diversion... Into... how to do better on the next exam,
Diagnostic Procurement The Clinton Foundation HIV/AIDS Initiative
 Diagnostic Procurement The Clinton Foundation HIV/AIDS Initiative 28 October 2008 Geneva 1 CHAI Work in Diagnostics - Context Diagnostic Test Agreements - In 2002 when CHAI was formed, the price of many
Diagnostic Procurement The Clinton Foundation HIV/AIDS Initiative 28 October 2008 Geneva 1 CHAI Work in Diagnostics - Context Diagnostic Test Agreements - In 2002 when CHAI was formed, the price of many
Expert Group Meeting on Strategies for Creating Urban Youth Employment: Solutions for Urban Youth in Africa
 Expert Group Meeting on Strategies for Creating Urban Youth Employment: Solutions for Urban Youth in Africa Measurement/indicators of youth employment Gora Mboup Global Urban Observatory (GUO) UN-HABITAT
Expert Group Meeting on Strategies for Creating Urban Youth Employment: Solutions for Urban Youth in Africa Measurement/indicators of youth employment Gora Mboup Global Urban Observatory (GUO) UN-HABITAT
Update from GAVI Aurelia Nguyen
 Update from GAVI Aurelia Nguyen (Copenhagen, Denmark, 27 June 2012) GAVI vaccine support Currently supported vaccines: pentavalent, pneumococcal, rotavirus, meningitis A, human papillomavirus (HPV), rubella,
Update from GAVI Aurelia Nguyen (Copenhagen, Denmark, 27 June 2012) GAVI vaccine support Currently supported vaccines: pentavalent, pneumococcal, rotavirus, meningitis A, human papillomavirus (HPV), rubella,
HIBA ABDALRAHIM Capsca Focal Point Public Health Authority
 HIBA ABDALRAHIM Capsca Focal Point Public Health Authority Introduction Definition Symptom Transmission Global situation Local situation Control Content Introduction Yellow fever (YF) is a mosquito-borne
HIBA ABDALRAHIM Capsca Focal Point Public Health Authority Introduction Definition Symptom Transmission Global situation Local situation Control Content Introduction Yellow fever (YF) is a mosquito-borne
Regional Consultation on Nutrition and HIV/AIDS in French Speaking Countries in Africa Region
 Regional Consultation on Nutrition and HIV/AIDS in French Speaking Countries in Africa Region Evidence, lessons and recommendations for action Ouagadougou, Burkina Faso 17-20 November 2008 CONCEPT PAPER
Regional Consultation on Nutrition and HIV/AIDS in French Speaking Countries in Africa Region Evidence, lessons and recommendations for action Ouagadougou, Burkina Faso 17-20 November 2008 CONCEPT PAPER
FRAMEWORK FOR IMPLEMENTING THE GLOBAL STRATEGY TO ELIMINATE YELLOW FEVER EPIDEMICS (EYE), IN THE AFRICAN REGION. Report of the Secretariat
 13 June 2017 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-seventh session Victoria Falls, Republic of Zimbabwe, 28 August 1 September 20177 Provisional agenda item 11 FRAMEWORK FOR IMPLEMENTING
13 June 2017 REGIONAL COMMITTEE FOR AFRICA ORIGINAL: ENGLISH Sixty-seventh session Victoria Falls, Republic of Zimbabwe, 28 August 1 September 20177 Provisional agenda item 11 FRAMEWORK FOR IMPLEMENTING
Stakeholders meeting on Malaria
 Plans and progress towards EDCTP2 Stakeholders meeting on Malaria 19 September 2013 Austrian Ministry of Science and Research First district of Vienna, Freyung 3, Vienna, Austria. Michael Makanga, MD PhD.
Plans and progress towards EDCTP2 Stakeholders meeting on Malaria 19 September 2013 Austrian Ministry of Science and Research First district of Vienna, Freyung 3, Vienna, Austria. Michael Makanga, MD PhD.
Vaccines: (inter)national regulation and quality in resource limited settings
 Vaccines: (inter)national regulation and quality in resource limited settings Raffaella Ravinetto QUAMED Research & Networking Institute of Tropical Medicine Antwerp 12th July 2017 Based on a presentation
Vaccines: (inter)national regulation and quality in resource limited settings Raffaella Ravinetto QUAMED Research & Networking Institute of Tropical Medicine Antwerp 12th July 2017 Based on a presentation
Global Measles and Rubella Update. April 2018
 Global Measles and Rubella Update April 218 Measles Number of Reported Measles by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug Sep Oct
Global Measles and Rubella Update April 218 Measles Number of Reported Measles by WHO Regions 218 Region Member States* Suspected cases Measles cases Clin Epi Lab Jan Feb Mar Apr May Jun Jul Aug Sep Oct
Overview of the PAHO strategy and activities to strengthen National Regulatory Authorities in the Americas
 Overview of the PAHO strategy and activities to strengthen National Regulatory Authorities in the Americas Ma. de los Angeles Cortes Regional Advisor on Vaccines and Biologicals HSS/MT Annecy, France March
Overview of the PAHO strategy and activities to strengthen National Regulatory Authorities in the Americas Ma. de los Angeles Cortes Regional Advisor on Vaccines and Biologicals HSS/MT Annecy, France March
Ending the AIDS Epidemic in Adolescents
 Ending the AIDS Epidemic in Adolescents Eastern and Southern Africa Regional Update on the ALL IN Overview 13 October 2015 AIDS-related deaths has declined for all age groups Except adolescents! Eastern
Ending the AIDS Epidemic in Adolescents Eastern and Southern Africa Regional Update on the ALL IN Overview 13 October 2015 AIDS-related deaths has declined for all age groups Except adolescents! Eastern
UNAIDS 2013 AIDS by the numbers
 UNAIDS 2013 AIDS by the numbers 33 % decrease in new HIV infections since 2001 29 % decrease in AIDS-related deaths (adults and children) since 2005 52 % decrease in new HIV infections in children since
UNAIDS 2013 AIDS by the numbers 33 % decrease in new HIV infections since 2001 29 % decrease in AIDS-related deaths (adults and children) since 2005 52 % decrease in new HIV infections in children since
Copyright 2011 Joint United Nations Programme on HIV/AIDS (UNAIDS) All rights reserved ISBN
 UNAIDS DATA TABLES 2011 Copyright 2011 Joint United Nations Programme on HIV/AIDS (UNAIDS) All rights reserved ISBN 978-92-9173-945-5 UNAIDS / JC2225E The designations employed and the presentation of
UNAIDS DATA TABLES 2011 Copyright 2011 Joint United Nations Programme on HIV/AIDS (UNAIDS) All rights reserved ISBN 978-92-9173-945-5 UNAIDS / JC2225E The designations employed and the presentation of
Influenza Surveillance In the WHO African Region
 Vol. 2 N 52, Updated on 2 January 207 Contents Highlights ackground Methodology Review of 206 influenza virus circulation Seasonal patterns of influenza transmission Way forward Conclusion Editor Dr Ibramima
Vol. 2 N 52, Updated on 2 January 207 Contents Highlights ackground Methodology Review of 206 influenza virus circulation Seasonal patterns of influenza transmission Way forward Conclusion Editor Dr Ibramima
Update on PMTCT. African Health Profession Regulatory Collaborative for Nurses and Midwives. Johannesburg, Republic of South Africa, June 18-22, 2012
 PMTCT Update Update on PMTCT Margarett Davis, MD, MPH Chief, Maternal and Child Health Branch Division of Global HIV/AIDS Centers for Disease Control and Prevention (CDC) African Health Profession Regulatory
PMTCT Update Update on PMTCT Margarett Davis, MD, MPH Chief, Maternal and Child Health Branch Division of Global HIV/AIDS Centers for Disease Control and Prevention (CDC) African Health Profession Regulatory
What Really Works for Raising African Women Leaders in Global Health
 What Really Works for Raising African Women Leaders in Global Health DR. AMA POKUAA FENNY 2018 EXTREME AFFORDABILITY CONFERENCE JULY 9, 2018 ENSIGN COLLEGE OF PUBLIC HEALTH, KPONG, GHANA Outline Goals
What Really Works for Raising African Women Leaders in Global Health DR. AMA POKUAA FENNY 2018 EXTREME AFFORDABILITY CONFERENCE JULY 9, 2018 ENSIGN COLLEGE OF PUBLIC HEALTH, KPONG, GHANA Outline Goals
WHO/PQT Updates on WHO Prequalification Priorities and Regulatory Systems Strengthening work
 WHO/PQT Updates on WHO Prequalification Priorities and Regulatory Systems Strengthening work Dr Drew Meek Prequalification Team Regulation of Medicines and other Health Technologies Essential Medicines
WHO/PQT Updates on WHO Prequalification Priorities and Regulatory Systems Strengthening work Dr Drew Meek Prequalification Team Regulation of Medicines and other Health Technologies Essential Medicines
Plan of Presentation
 CONTINENTAL CONFERENCE ON MATERNAL, INFANT AND CHILD HEALTH IN AFRICA Plan of Presentation Regional Frameworks on SRH Maputo Plan of Action Five-Year Review of MPoA What is CARMMA? Why CARMMA? What is
CONTINENTAL CONFERENCE ON MATERNAL, INFANT AND CHILD HEALTH IN AFRICA Plan of Presentation Regional Frameworks on SRH Maputo Plan of Action Five-Year Review of MPoA What is CARMMA? Why CARMMA? What is
