SECTION 13.0 SPECIMENS FOR LABORATORY EXAMINATION
|
|
|
- James Little
- 5 years ago
- Views:
Transcription
1 SECTION 13.0 SPECIMENS FOR LABORATORY EXAMINATION Introduction Collection of Specimens for Microbiological Examination: Safe Handling of Specimens Packaging & Transportation of Specimens Specimen Rejection Table 1: The Importance of Data on Request Forms Developed by Patricia Coughlan, Máire Flynn, Liz Forde & Niamh McDonnell In consultation with Brendan O Reilly Chief Medical Scientist, CUH & Colm Power Senior Medical Scientist KGH, Date developed August 2012 Approved by Cork and Kerry Infection Prevention and Control Committee Kerry Infection Prevention and Control Committee Reference number IPCG 13/ 2012 Revision number 0 Revision date 2015 or sooner if new evidence becomes available Responsibility for Infection Prevention and Control Nurses review HSE South (Cork &Kerry) Page 1 of 6
2 Introduction Collection and transport of specimens is the responsibility of the Doctor or Environmental Health Officer requesting the test. To obtain the best information from a laboratory the following general principles apply to any specimen for transport and should be observed. Collection of Specimens for Microbiological Examination: Collect specimen at the appropriate time e.g. prior to the commencement of or administration of antimicrobial therapy when possible. Collect specimen with as little contamination as possible to ensure that the sample will be representative of the infective site. Collect an adequate amount of specimen. Inadequate amounts of specimen may yield false-negative results. Use sterile equipment and aseptic technique to collect specimens to prevent introduction of microorganisms during invasive procedures. Label laboratory approved containers with: patient identification such as patient name, identification number/address and date of birth, specimen details and date specimen was taken,e.g. the site the swab was taken from; whether MSU or CSU. The lids should be screwed on tightly. Label laboratory request form with Patient name, identification number, Location (ward or GP) address, Date of birth, Sex, Requesting clinician, Site of swab or specimen type i.e whether CSU or MSU Date of specimen collection. State clearly the test required. The patients clinical symptoms, reasons for taking the specimen and whether antibiotic therapy is in progress or not, should also be included. List antibiotics and duration if commenced, as this enables laboratory staff to streamline cultures and sensitivities and speeds up reporting. (See table 1). Please refer to Section 11.4 Wound Care for criteria for taking a wound swab. Note: Separate forms should be sent for different specimen types e.g. one form for a urine specimen and another for a wound specimen etc. HSE South (Cork &Kerry) Page 2 of 6
3 Safe Handling of Specimens The container with specimen should be placed in an individual transparent plastic transport bag as soon as it has been labelled. The transport bag must be sealed. Specimen bags must not be sealed with pins, staples, metal clips etc. and must not be used more than once Any specimen may contain potentially pathogenic organisms. To avoid presenting a hazard to anyone in contact with the specimens, it is most important to avoid contamination of the outside of specimen containers and to ensure that they are securely closed and safely handled. Leaking specimens are a serious biohazard risk to persons transporting the specimen and laboratory staff. Blood stained forms should never be sent to the laboratory. Furthermore, if a specimen leaks, contaminating organisms may get into the container and a false result ensue. To ensure safety: Adhere to Standard Precautions for taking and processing of all specimens regardless of the patients infectious status, known or unknown. Specimen containers are put in plastic bags with a self-sealing seal. The request form is kept separate from the specimen. This system provides protection for all staff from any accidental spillage and prevents contamination of the accompanying request form. A self-sealing plastic compartment with request form attached is the type used by the Microbiology Department. Whilst awaiting transport, specimens should be stored appropriately and securely, away from patient and visitor areas, and from food and medicines. For microbiological investigation if processing is delayed, refrigeration is preferable to storage at ambient temperature, with the following exceptions: o Blood cultures incubate the bottles at C. o CSF hold specimen at room temperature. Check the individual specimen requirements to determine if refrigeration is advisable as some organisms are temperature sensitive. There are specific collection requirements for some tests; where doubt exists, the appropriate laboratory should be consulted. Packaging & Transportation of Specimens Postal service Specimens sent to the laboratory e.g. from GP surgeries should be packaged by the sender, in appropriate specimen containers and packaging, and should be properly labeled. Any package that is not packed correctly or marked as directed may result in delay/ rejection by An Post. The following triple packaging system must be obeyed: 1. The specimen must be enclosed in a primary receptacle. The sender must ensure that the container is the appropriate one for the purpose, that the specimen HSE South (Cork &Kerry) Page 3 of 6
4 container is properly closed and that it is not externally contaminated by the contents. 2. The primary receptacle must be placed in a secondary watertight receptacle. 3. An outer packaging of adequate strength for its capacity mass and intended use with at least one surface having a minimum dimension of 100 X 100mm must be used to send the sample. Absorbent material must be placed between the primary and secondary receptacle. The UN 3373 diamond shaped symbol must be displayed on the external surface of the outer packaging and clearly marked Biological Substance, Category B adjacent to the symbol. The outer package must show name and address of destination of sample name and address of sender who to contact in case of loss, damage or leakage. Courier, van or taxi (i.e. by means other than by An Post) Specimens sent to the laboratory must be packaged correctly according to guidelines for sending of samples through the normal post. Alternatively, special transport boxes may be used. The transport box must be made of smooth impervious material such as plastic or metal, which can easily be cleaned and disinfected. must be secured with a fastenable lid. must retain liquid in the event of leakage of a specimen. must clearly labeled with the UN 3373 diamond shaped mark and the proper shipping name Biological substance, Category B for new type transportation boxes and Diagnostic Specimen-Fragile with Care, for the older transportation boxes. must be labeled to identify the sender of the package, including relevant telephone numbers to be contacted in case of loss, damage or leakage. Where further advice on packaging for transportation of specimens is sought the laboratory to which the specimen is being sent should be contacted. Specimen Rejection Unacceptable specimens may result in test cancellation or delay. Common causes of such action include: The samples are received in a hazardous condition. The sample identification on either the request form or the sample bottle is incomplete. The sample is unlabelled or incorrectly labelled. The sample is received with incorrect patient preparation. The sample has been collected under inappropriate conditions. HSE South (Cork &Kerry) Page 4 of 6
5 Supplies of Request forms and specimen containers: Request forms and sample / specimen containers are issued from Hospital Stores/Laboratory. Order supplies in advance to facilitate timely delivery. References & Bibliography CUH Pathology User Handbook Revision Department of Health (2007) Transport of Infectious Substance Best Practice Guidance for Microbiology Laboratories. HSE South (Cork &Kerry) Page 5 of 6
6 Table 1: The Importance of Data on Request Forms Data Rationale Patient s name & identification Used for filing purposes or to distinguish between people of the same name. number Date & time of Different organisms survive for varying periods. Delay in specimen processing specimen may influence interpretation of results. collection Location (Ward or GP address) This allows the report to be returned to the ward/gp with minimum delay. Patient s date of The lab will investigate for different pathogens in birth: accordance with the patient s age; e.g., eye swab from a neonate would require detailed investigation for Neisseria gonorrhoeae. Some antibiotics would be contraindicated in certain age groups. Relevant Info Cellulitis, pyrexia, pus, discharge, treatment, wound infection, provisional diagnosis, chemotherapy etc. Specimen details Be as specific as possible to help lab in distinguishing including site: pathogens from normal flora e.g. Staph aureus may be considered normal flora in the nose but would be reported as a possible pathogen in other sites. A swab taken from an operative site following a clean operation (e.g. knee replacement), any organism would require investigation whereas a swab from a chronic leg ulcer will yield many potentially pathogenic organisms, which may be colonising the area. Antibiotic therapy: State if a patient is on antibiotics so that laboratory results can be correctly interpreted. Routine sensitivity testing may not always include the particular antibiotic that the patient is currently receiving, unless this is stated on the form. Antibiotic therapy should be based on clinical signs of infection and not solely on laboratory results. Doctor requesting Enables lab staff to obtain further information or investigation communicate important results quickly to the doctor concerned i.e. consultant or doctor requesting the test. Investigation requested: The form should state clearly what the specimen is and where it has been taken from with the correct request. N.B. A viral swab cannot be used for general bacteriological analysis. Viral swabs are available from the microbiology laboratory directly. HSE South (Cork &Kerry) Page 6 of 6
Community Infection Prevention and Control Guidance for Health and Social Care. Specimen Collection
 Community Infection Prevention and Control Guidance for Health and Social Care Specimen Collection Version 1.01 May 2015 Harrogate and District NHS Foundation Trust Specimen Collection May 2015 Version
Community Infection Prevention and Control Guidance for Health and Social Care Specimen Collection Version 1.01 May 2015 Harrogate and District NHS Foundation Trust Specimen Collection May 2015 Version
POLICY DOCUMENT. Specimen Transportation Policy
 POLICY DOCUMENT Specimen Transportation Policy Owner: DGM - Pathology Date Created: 01/10/2004 The following fields are updated automatically: Version No: 2.0 Status: Approved Next Review Date: 22/01/2013
POLICY DOCUMENT Specimen Transportation Policy Owner: DGM - Pathology Date Created: 01/10/2004 The following fields are updated automatically: Version No: 2.0 Status: Approved Next Review Date: 22/01/2013
Collection and Transportation of Clinical Specimens
 Collection and Transportation of Clinical Specimens IDK II Faculty of Nursing Universitas Indonesia PDF Created with deskpdf PDF Writer - Trial :: http://www.docudesk.com The laboratory diagnosis of an
Collection and Transportation of Clinical Specimens IDK II Faculty of Nursing Universitas Indonesia PDF Created with deskpdf PDF Writer - Trial :: http://www.docudesk.com The laboratory diagnosis of an
Taking nose, throat and skin swabs. Clinical Skills
 Clinical Skills Taking nose, throat and skin swabs Carole Loveridge (Lecturer in Women s Health) Aims and Objectives Aims and Objectives The aim of this module is to facilitate learning regarding the purpose
Clinical Skills Taking nose, throat and skin swabs Carole Loveridge (Lecturer in Women s Health) Aims and Objectives Aims and Objectives The aim of this module is to facilitate learning regarding the purpose
SUBJECT: Management of Human Body Fluids/Waste (Bloodborne Pathogens)
 Page 1 of 6 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 PURPOSE To establish uniform procedures for the safe management of human body fluids
Page 1 of 6 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 PURPOSE To establish uniform procedures for the safe management of human body fluids
Manchester Cytology Centre Synovial Fluid Analysis Service User Manual 2017/2018
 Page 1 of 12 Author: Sarah Ferris Directorate of Laboratory Medicine Manchester Cytology Centre Synovial Fluid Analysis Service User Manual 2017/2018 Page 2 of 12 Author: Sarah Ferris Table of contents
Page 1 of 12 Author: Sarah Ferris Directorate of Laboratory Medicine Manchester Cytology Centre Synovial Fluid Analysis Service User Manual 2017/2018 Page 2 of 12 Author: Sarah Ferris Table of contents
Best Practice Guidance 12_Version 1.2. Title: Collection of Biological Samples
 This document is for guidance on the collection of biological samples that have been granted ethical approval under CUREC procedures. Certain research involving the taking of samples that consist of, or
This document is for guidance on the collection of biological samples that have been granted ethical approval under CUREC procedures. Certain research involving the taking of samples that consist of, or
Guidance Document Infectious Substances
 Guidance Document Infectious Substances Note: 1. The following Guidance Document was developed by the ICAO DGP. The original ICAO document reflects references to the ICAO Technical Instructions these have
Guidance Document Infectious Substances Note: 1. The following Guidance Document was developed by the ICAO DGP. The original ICAO document reflects references to the ICAO Technical Instructions these have
General Guidelines for Sample Submissions
 HISTORY General Guidelines for Sample Submissions (See the Client Handbook for specific test requirements.) The history of the animal is very important, including treatments, vaccinations and feeding,
HISTORY General Guidelines for Sample Submissions (See the Client Handbook for specific test requirements.) The history of the animal is very important, including treatments, vaccinations and feeding,
CLINICAL LABORATORY SERVICES
 13. SPECIMEN COLLECTION AND HANDLING GUIDELINES 13.1. General guidelines For microbiology investigations the prompt and accurate isolation of infecting agents is directly influenced by the quality of the
13. SPECIMEN COLLECTION AND HANDLING GUIDELINES 13.1. General guidelines For microbiology investigations the prompt and accurate isolation of infecting agents is directly influenced by the quality of the
Patient Preparation Unique patient preparation requirements are listed under each test in the Test Directory.
 Specimen Collection Lab results are only as good as the specimen provided. Patient preparation, venipuncture technique, specimen handling, and transportation can all affect the quantity of results. Health
Specimen Collection Lab results are only as good as the specimen provided. Patient preparation, venipuncture technique, specimen handling, and transportation can all affect the quantity of results. Health
DEPARTMENT OF HEALTH RESEARCH INSTITUTE FOR TROPICAL MEDICINE. SPECIMEN MANAGEMENT: Collection, Packaging and Transport of EVD Specimens
 DEPARTMENT OF HEALTH RESEARCH INSTITUTE FOR TROPICAL MEDICINE SPECIMEN MANAGEMENT: Collection, Packaging and Transport of EVD Specimens OUTLINE Introduction Specimen collection Guidelines Timing of Collection
DEPARTMENT OF HEALTH RESEARCH INSTITUTE FOR TROPICAL MEDICINE SPECIMEN MANAGEMENT: Collection, Packaging and Transport of EVD Specimens OUTLINE Introduction Specimen collection Guidelines Timing of Collection
Manchester Cytology Centre Synovial Fluid Analysis Service User Manual 2016
 Page 1 of 13 Directorate of Laboratory Medicine Manchester Cytology Centre Synovial Fluid Analysis Service User Manual 2016 Page 2 of 13 Table of contents Table of contents... 2 1. About us... 3 1.1 Opening
Page 1 of 13 Directorate of Laboratory Medicine Manchester Cytology Centre Synovial Fluid Analysis Service User Manual 2016 Page 2 of 13 Table of contents Table of contents... 2 1. About us... 3 1.1 Opening
APPENDIX E COLLECTION AND SUBMISSION OF SPECIMENS FOR TB TESTING
 APPENDIX E COLLECTION AND SUBMISSION OF SPECIMENS FOR TB TESTING For additional information about the collection, transport or submission of specimens for AFB examination, contact BC Public Health & Microbiology
APPENDIX E COLLECTION AND SUBMISSION OF SPECIMENS FOR TB TESTING For additional information about the collection, transport or submission of specimens for AFB examination, contact BC Public Health & Microbiology
Monitoring the Health of Transplanted Organs DONOR GENOTYPING MANUAL
 Monitoring the Health of Transplanted Organs DONOR GENOTYPING MANUAL 10101 Innovation Drive. Suite 600. Wauwatosa, WI 53226 1-888-214-3151 MKT-008 v3.0 MKT-008 v3.0 TABLE OF CONTENTS I Intended Use...
Monitoring the Health of Transplanted Organs DONOR GENOTYPING MANUAL 10101 Innovation Drive. Suite 600. Wauwatosa, WI 53226 1-888-214-3151 MKT-008 v3.0 MKT-008 v3.0 TABLE OF CONTENTS I Intended Use...
BLOOD CULTURE POLICY FOR PAEDIATRICS
 BLOOD CULTURE POLICY FOR PAEDIATRICS 1. INTRODUCTION Blood culture to detect bacteraemia is an important investigation with major implications for the diagnosis of patients with infection and the selection
BLOOD CULTURE POLICY FOR PAEDIATRICS 1. INTRODUCTION Blood culture to detect bacteraemia is an important investigation with major implications for the diagnosis of patients with infection and the selection
HANDLING GENERAL INFORMATION TEST REQUESTS TYPES OF TESTS TEST REQUEST FORM
 SECTION 3: SAMPLE COLLECTION & HANDLING GENERAL INFORMATION SAMPLE INTRODUCTION COLLECTION & HANDLING GENERAL results. INFORMATION The quality of laboratory test results depends greatly on the quality
SECTION 3: SAMPLE COLLECTION & HANDLING GENERAL INFORMATION SAMPLE INTRODUCTION COLLECTION & HANDLING GENERAL results. INFORMATION The quality of laboratory test results depends greatly on the quality
Specimen Collection. Urine Vacutainers. Laboratory Services September 2018
 1 Specimen Collection Urine Vacutainers Laboratory Services September 2018 Agenda Before you begin Collection Labeling Packaging & Transport Resources Click on the links above to be taken directly to that
1 Specimen Collection Urine Vacutainers Laboratory Services September 2018 Agenda Before you begin Collection Labeling Packaging & Transport Resources Click on the links above to be taken directly to that
PEN USER MANUAL Byetta 5 micrograms solution for injection in pre-filled pen (exenatide)
 PEN USER MANUAL Byetta 5 micrograms solution for injection in pre-filled pen (exenatide) Section 1. WHAT YOU NEED TO KNOW ABOUT YOUR BYETTA PEN Read this section completely before you begin. Then, move
PEN USER MANUAL Byetta 5 micrograms solution for injection in pre-filled pen (exenatide) Section 1. WHAT YOU NEED TO KNOW ABOUT YOUR BYETTA PEN Read this section completely before you begin. Then, move
ADMINISTRATIVE SERVICES MANUAL
 1 of 10 Purpose Scope University of Alaska Anchorage departments will develop plans and procedures to limit occupational exposure to blood and other potentially infectious materials (PIM) in compliance
1 of 10 Purpose Scope University of Alaska Anchorage departments will develop plans and procedures to limit occupational exposure to blood and other potentially infectious materials (PIM) in compliance
SAMPLE. Collecting a faeces specimen
 7. Perform steps 10 16 of the common steps (see pp. 39 43). Evidence base: PHE (2014a) To ensure that: the patient is safe and comfortable. the specimen has been correctly collected and documented in the
7. Perform steps 10 16 of the common steps (see pp. 39 43). Evidence base: PHE (2014a) To ensure that: the patient is safe and comfortable. the specimen has been correctly collected and documented in the
Biosafety & Biosecurity in the Laboratory. Based on the OIE Terrestrial Manual for Diagnostic Tests and Vaccines
 Biosafety & Biosecurity in the Laboratory Based on the OIE Terrestrial Manual for Diagnostic Tests and Vaccines Keeping people and animals safe Keeping samples safe Definitions Biosafety: The measures
Biosafety & Biosecurity in the Laboratory Based on the OIE Terrestrial Manual for Diagnostic Tests and Vaccines Keeping people and animals safe Keeping samples safe Definitions Biosafety: The measures
CHEMOTHERAPY NETWORK GROUP POLICY FOR ADMINISTRATION OF CYTOTOXIC CHEMOTHERAPY
 CHEMOTHERAPY NETWORK GROUP POLICY FOR ADMINISTRATION OF CYTOTOXIC CHEMOTHERAPY Version 4.0 March 2016 Review date March 2018 Introduction It is the purpose of this policy to provide clear guidelines that
CHEMOTHERAPY NETWORK GROUP POLICY FOR ADMINISTRATION OF CYTOTOXIC CHEMOTHERAPY Version 4.0 March 2016 Review date March 2018 Introduction It is the purpose of this policy to provide clear guidelines that
4. Laboratory Diagnosis of Viral Haemorrhagic Fever Viruses
 4. Laboratory Diagnosis of Viral Haemorrhagic Fever Viruses 4.1 Introduction Early infection with VHF is characterised by a short non-specific viral prodrome with an extensive differential diagnosis. The
4. Laboratory Diagnosis of Viral Haemorrhagic Fever Viruses 4.1 Introduction Early infection with VHF is characterised by a short non-specific viral prodrome with an extensive differential diagnosis. The
Policy Title: Clinical Asepsis Policy Policy Number :19. Effective Date: 6/10/2013 Review Date: 6/10/2016
 Policy Title: Clinical Asepsis Policy Policy Number :19 6.4.9. Take/send instruments and handpieces to the decontamination/sterilization area. 6.4.10. Remove and dispose of the disposable gown (if used)
Policy Title: Clinical Asepsis Policy Policy Number :19 6.4.9. Take/send instruments and handpieces to the decontamination/sterilization area. 6.4.10. Remove and dispose of the disposable gown (if used)
Specimens, Collection, handling and transportation SOP
 Clinical Specimens, Collection, handling and transportation SOP Document Control Summary Status: Version: Author/Owner: Approved by: Ratified: Related Trust Strategy and/or Strategic Aims Implementation
Clinical Specimens, Collection, handling and transportation SOP Document Control Summary Status: Version: Author/Owner: Approved by: Ratified: Related Trust Strategy and/or Strategic Aims Implementation
National Dairy Development Board
 1 P a g e National Dairy Development Board Standard Operating Procedures (SOPs) for Vaccination 2 P a g e SOP for vaccine storage Storage The vaccine should be stored in a refrigerator between +4 to +
1 P a g e National Dairy Development Board Standard Operating Procedures (SOPs) for Vaccination 2 P a g e SOP for vaccine storage Storage The vaccine should be stored in a refrigerator between +4 to +
SECTION 10.4 LEGIONELLA
 SECTION 10.4 LEGIONELLA Introduction Legionella in Healthcare Settings Legionnaires Disease Appendix 10.4.1 Management of Nebulisers Developed by Patricia Coughlan, Maire Flynn, Liz Forde, Niamh Mc Donnell
SECTION 10.4 LEGIONELLA Introduction Legionella in Healthcare Settings Legionnaires Disease Appendix 10.4.1 Management of Nebulisers Developed by Patricia Coughlan, Maire Flynn, Liz Forde, Niamh Mc Donnell
Rapid-VIDITEST. Influenza A
 Rapid-VIDITEST Influenza A (One step Influenza A Card test for the detection of Influenza type A antigen from human nasopharyngeal specimens (swab, nasopharyngeal wash and aspirate). Instruction manual
Rapid-VIDITEST Influenza A (One step Influenza A Card test for the detection of Influenza type A antigen from human nasopharyngeal specimens (swab, nasopharyngeal wash and aspirate). Instruction manual
Section 10.5 Varicella
 Section 10.5 Varicella Chickenpox Introduction Transmission Signs and Symptoms Complications Diagnosis Treatment Infection Prevention and Control Precautions for Residents with Chickenpox Additional considerations
Section 10.5 Varicella Chickenpox Introduction Transmission Signs and Symptoms Complications Diagnosis Treatment Infection Prevention and Control Precautions for Residents with Chickenpox Additional considerations
Safety Committee Prototypical Safety Program Manual
 1 Bloodborne Pathogens Exposure Control Plan Policy The Department Bloodborne Pathogens Exposure Control Plan is designed to comply with the requirements of the OSHA Bloodborne Pathogens Standard, 29 CFR
1 Bloodborne Pathogens Exposure Control Plan Policy The Department Bloodborne Pathogens Exposure Control Plan is designed to comply with the requirements of the OSHA Bloodborne Pathogens Standard, 29 CFR
Helicobacter pylori Antigen Test
 Helicobacter pylori Antigen Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-HP(s) Specimen: Fecal Specimen * Please read the instructions carefully before use INTENDED USE Helicobacter
Helicobacter pylori Antigen Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-HP(s) Specimen: Fecal Specimen * Please read the instructions carefully before use INTENDED USE Helicobacter
Influenza-Associated Pediatric Mortality rev Jan 2018
 rev Jan 2018 Infectious Agent Influenza A, B or C virus BASIC EPIDEMIOLOGY Transmission Transmission occurs via droplet spread. After a person infected with influenza coughs, sneezes, or talks, influenza
rev Jan 2018 Infectious Agent Influenza A, B or C virus BASIC EPIDEMIOLOGY Transmission Transmission occurs via droplet spread. After a person infected with influenza coughs, sneezes, or talks, influenza
INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust)
 A member of: Association of UK University Hospitals INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust) IPC17 SINGLE USE AND SINGLE PATIENT USE MEDICAL
A member of: Association of UK University Hospitals INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust) IPC17 SINGLE USE AND SINGLE PATIENT USE MEDICAL
Management of Outbreaks Care Homes IPC Study Day
 Management of Outbreaks Care Homes IPC Study Day Sue Barber Infection Prevention & Control Lead AV & Chiltern CCG s Diarrhoea and/or vomiting May be bacterial or viral May be non-infectious in origin but
Management of Outbreaks Care Homes IPC Study Day Sue Barber Infection Prevention & Control Lead AV & Chiltern CCG s Diarrhoea and/or vomiting May be bacterial or viral May be non-infectious in origin but
Lab Alert REMINDER: BioFire- FilmArray Respiratory Panel
 Department of Pathology Laboratory Alert November 11, 2015 Lab Alert REMINDER: BioFire- FilmArray Respiratory Panel The Molecular Pathology and Microbiology Sections of the Pathology Laboratory is continuing
Department of Pathology Laboratory Alert November 11, 2015 Lab Alert REMINDER: BioFire- FilmArray Respiratory Panel The Molecular Pathology and Microbiology Sections of the Pathology Laboratory is continuing
Chapter 11 PREVENTING INFECTION. Elsevier items and derived items 2010 by Mosby, Inc. an affiliate of Elsevier Inc. All rights reserved
 Chapter 11 PREVENTING INFECTION Infection is a major safety and health hazard. The health team follows certain practices and procedures to protect patients, residents, visitors, and staff from infection.
Chapter 11 PREVENTING INFECTION Infection is a major safety and health hazard. The health team follows certain practices and procedures to protect patients, residents, visitors, and staff from infection.
Chlamydia Rapid Screen Test (RAP-2858) RUO in the USA. Revised 28 Jul 2006
 INDICATION For the rapid detection of Chlamydia Trachomatis antigens in swab specimens. For in vitro diagnostic use only, except in the United States where it is intended for Research Use Only. SUMMARY
INDICATION For the rapid detection of Chlamydia Trachomatis antigens in swab specimens. For in vitro diagnostic use only, except in the United States where it is intended for Research Use Only. SUMMARY
Information for Health Care Workers
 Information for Health Care Workers This Bulletin highlights precautions to be taken by health care workers who may be exposed to blood and body fluids. In general, workers should minimize direct contact
Information for Health Care Workers This Bulletin highlights precautions to be taken by health care workers who may be exposed to blood and body fluids. In general, workers should minimize direct contact
TRANSCRIPT for Lantus SoloSTAR pen injection for your patients
 TRANSCRIPT for Lantus SoloSTAR pen injection for your patients SUPER: Prescription Lantus is a long-acting insulin used to treat adults with type 2 diabetes and adults and pediatric patients (children
TRANSCRIPT for Lantus SoloSTAR pen injection for your patients SUPER: Prescription Lantus is a long-acting insulin used to treat adults with type 2 diabetes and adults and pediatric patients (children
Chapter 12. Preventing Infection. Elsevier items and derived items 2014, 2010 by Mosby, an imprint of Elsevier Inc. All rights reserved.
 Chapter 12 Preventing Infection Infection Infection is a major safety and health hazard. The health team follows certain practices and procedures to protect patients, residents, visitors, and staff from
Chapter 12 Preventing Infection Infection Infection is a major safety and health hazard. The health team follows certain practices and procedures to protect patients, residents, visitors, and staff from
Urinary Catheters. Prevalence of Infections
 Urinary Catheterisation Urinary catheterisation is defined as an intervention to enable the emptying of the bladder by insertion of a catheter. Catheters can be short term less than 28 days or long term
Urinary Catheterisation Urinary catheterisation is defined as an intervention to enable the emptying of the bladder by insertion of a catheter. Catheters can be short term less than 28 days or long term
Collection of Dried Blood Spots from Infants for Diagnosis of HIV by DNA PCR. MOH Regional Trainings March 2013
 Collection of Dried Blood Spots from Infants for Diagnosis of HIV by DNA MOH Regional Trainings March 2013 Outline Introduce DBS testing Rationale Testing algorithm Sample collection, storage and transportation
Collection of Dried Blood Spots from Infants for Diagnosis of HIV by DNA MOH Regional Trainings March 2013 Outline Introduce DBS testing Rationale Testing algorithm Sample collection, storage and transportation
Infection Control Precautions during the Clinical Management of Injecting Drug Users with Possible, Probable or Confirmed Anthrax
 Infection Control Precautions during the Clinical Management of Injecting Drug Users with Possible, Probable or Confirmed Anthrax (Adapted from guidance developed by Health Protection Scotland and HPA
Infection Control Precautions during the Clinical Management of Injecting Drug Users with Possible, Probable or Confirmed Anthrax (Adapted from guidance developed by Health Protection Scotland and HPA
Rapid-VIDITEST. Influenza A+B
 Rapid-VIDITEST Influenza A+B (One step Influenza A+B blister Test for the detection of Influenza type A and type B from nasal swabs, nasal wash or nasal aspirate specimens). Instruction manual Producer:
Rapid-VIDITEST Influenza A+B (One step Influenza A+B blister Test for the detection of Influenza type A and type B from nasal swabs, nasal wash or nasal aspirate specimens). Instruction manual Producer:
5. Use of antibiotics, which disturbs balance of normal flora. 6. Poor nutritional status.
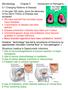 Microbiology Chapter 5 Introduction to Pathogens 5:1 Changing Patterns of Disease In the past 100 years, since the discovery of the Germ Theory of Disease was accepted: We have learned that microbes cause
Microbiology Chapter 5 Introduction to Pathogens 5:1 Changing Patterns of Disease In the past 100 years, since the discovery of the Germ Theory of Disease was accepted: We have learned that microbes cause
Health technology description. What is an evidence note. Key points. Literature search
 Health technology description In response to an enquiry from the Scottish Dental Clinical Effectiveness Programme Number 66 April 2017 After dental instruments have been sterilised unwrapped and are dry,
Health technology description In response to an enquiry from the Scottish Dental Clinical Effectiveness Programme Number 66 April 2017 After dental instruments have been sterilised unwrapped and are dry,
Bloodborne Pathogens 29 CFR
 Bloodborne Pathogens 29 CFR 1910.1030 Revised OSHA Bloodborne Pathogens Compliance Directive (CPL2-2.44D) Could You Contract a Disease at Work? Administering first aid? Cleaning the restrooms? Using a
Bloodborne Pathogens 29 CFR 1910.1030 Revised OSHA Bloodborne Pathogens Compliance Directive (CPL2-2.44D) Could You Contract a Disease at Work? Administering first aid? Cleaning the restrooms? Using a
Providence Medford Medical Center Pathology Department
 Providence Medford Medical Center Pathology Department Anatomic pathology services including histology, cytology and autopsies are offered through Providence Medford Medical Center Pathology Department.
Providence Medford Medical Center Pathology Department Anatomic pathology services including histology, cytology and autopsies are offered through Providence Medford Medical Center Pathology Department.
Control of Substances Hazardous to Health (COSHH) Regulations 2002
 Control of Substances Hazardous to Health (COSHH) Regulations 2002 1. Purpose This document provides guidance to staff on how to comply with the COSHH Regulations 2002 (as amended) and in particular informs
Control of Substances Hazardous to Health (COSHH) Regulations 2002 1. Purpose This document provides guidance to staff on how to comply with the COSHH Regulations 2002 (as amended) and in particular informs
Chapter 13. Preventing Infection. Copyright 2019 by Elsevier, Inc. All rights reserved.
 Chapter 13 Preventing Infection Copyright 2019 by Elsevier, Inc. All rights reserved. Lesson 13.1 Define the key terms and key abbreviations in this chapter. Identify what microbes need to live and grow.
Chapter 13 Preventing Infection Copyright 2019 by Elsevier, Inc. All rights reserved. Lesson 13.1 Define the key terms and key abbreviations in this chapter. Identify what microbes need to live and grow.
Yorkhill Laboratories Policy for the Packaging and Transport of Biological Samples (sent from laboratories) H&S/Doc/07. Yorkhill Hospitals
 Management Procedure Packaging and Transport of This document together with Standard Operating Procedures, instruction manuals, etc, forms part of, and is representative of the Quality Management System
Management Procedure Packaging and Transport of This document together with Standard Operating Procedures, instruction manuals, etc, forms part of, and is representative of the Quality Management System
My Doctor Says I Need to Inject Insulin In Special Sites... Which Ones Should I Use? Getting Started. Site Selection
 My Doctor Says I Need to Inject Insulin In Special Sites... Which Ones Should I Use? Getting Started Site Selection You need to know where to inject insulin so that your injections will be easier, safer
My Doctor Says I Need to Inject Insulin In Special Sites... Which Ones Should I Use? Getting Started Site Selection You need to know where to inject insulin so that your injections will be easier, safer
The most current laboratory testing information can be obtained at
 About Adventist Lab Partners Reference Laboratory Adventist Lab Partners (ALPs) Reference Laboratory is affiliated with Adventist Health System. Adventist Lab Partners has been a hospital-based laboratory
About Adventist Lab Partners Reference Laboratory Adventist Lab Partners (ALPs) Reference Laboratory is affiliated with Adventist Health System. Adventist Lab Partners has been a hospital-based laboratory
Area Laboratory Service
 THIS IS A CONTROLLED DOCUMENT. DO NOT COPY Area Laboratory Service Organisational Unit: Title: Ref No: Version No: Date Issued: Author: Authorised by: Review date: Location of this Copy : Microbiology
THIS IS A CONTROLLED DOCUMENT. DO NOT COPY Area Laboratory Service Organisational Unit: Title: Ref No: Version No: Date Issued: Author: Authorised by: Review date: Location of this Copy : Microbiology
SAFETY & HEALTH REQUIREMENTS FOR USE OF MATERIALS DERIVED FROM NHPS
 Introduction SAFETY & HEALTH REQUIREMENTS FOR USE OF MATERIALS DERIVED FROM NHPS 1. Experimentation with NHP-derived samples (cell cultures, tissues, body fluids or other wastes) may expose NUS staff and
Introduction SAFETY & HEALTH REQUIREMENTS FOR USE OF MATERIALS DERIVED FROM NHPS 1. Experimentation with NHP-derived samples (cell cultures, tissues, body fluids or other wastes) may expose NUS staff and
LABORATORY ALERT: Ebola Virus Disease (EVD)
 County of Santa Clara Public Health Department Public Health Laboratory 2220 Moorpark Avenue, 2 nd Floor San José, CA 95128 408.885.4272 DATE: August 21, 2014 LABORATORY ALERT: Ebola Virus Disease (EVD)
County of Santa Clara Public Health Department Public Health Laboratory 2220 Moorpark Avenue, 2 nd Floor San José, CA 95128 408.885.4272 DATE: August 21, 2014 LABORATORY ALERT: Ebola Virus Disease (EVD)
University Hospital Limerick Laboratory User Manual Histology File Name: MP-L-HIS-USERMAN Edition No.: 04 Date of Issue: 7 th October 2015 HISTOLOGY
 HISTOLOGY Contents 1. Index: Test Repertoire - Histology... 2 2. Laboratory Details... 2 3. Background Information... 3 4. Quality Assurance... 3 5. Specimen Collection, Handling and Storage... 3 6. Specimen
HISTOLOGY Contents 1. Index: Test Repertoire - Histology... 2 2. Laboratory Details... 2 3. Background Information... 3 4. Quality Assurance... 3 5. Specimen Collection, Handling and Storage... 3 6. Specimen
Gabriel L. Hamer, PhD DEPARTMENT OF ENTOMOLOGY. Minnie Belle Heep 2475 TAMU College Station, TX
 DEPARTMENT OF ENTOMOLOGY To: Texas Municipalities From: Gabriel L. Hamer, PhD, Clinical Assistant Professor, Department of Entomology Date: 17 April 2013 Subject: Dead bird surveillance for West Nile virus
DEPARTMENT OF ENTOMOLOGY To: Texas Municipalities From: Gabriel L. Hamer, PhD, Clinical Assistant Professor, Department of Entomology Date: 17 April 2013 Subject: Dead bird surveillance for West Nile virus
Supervisors, Department Heads and Principals will:
 The Greater Victoria School District is committed to each student s success in learning within a responsive and safe environment. REGULATION 4213 UNIVERSAL PRECAUTIONS Preamble Universal Precautions are
The Greater Victoria School District is committed to each student s success in learning within a responsive and safe environment. REGULATION 4213 UNIVERSAL PRECAUTIONS Preamble Universal Precautions are
Diphtheria rev Jan 2018
 rev Jan 2018 Infectious Agent Toxin-producing strains of Corynebacterium diphtheriae BASIC EPIDEMIOLOGY Transmission Direct person-to-person transmission by intimate respiratory and physical contact. Cutaneous
rev Jan 2018 Infectious Agent Toxin-producing strains of Corynebacterium diphtheriae BASIC EPIDEMIOLOGY Transmission Direct person-to-person transmission by intimate respiratory and physical contact. Cutaneous
Deafblind Scotland Infection Control Policy
 Deafblind Scotland vision A society in which deafblind people have the permanent support and recognition necessary to be equal citizens Deafblind Scotland Infection Control Policy What do we mean by Infection
Deafblind Scotland vision A society in which deafblind people have the permanent support and recognition necessary to be equal citizens Deafblind Scotland Infection Control Policy What do we mean by Infection
GYNAECOLOGICAL CYTOLOGY
 The laboratory runs a tour of the department for Practice Nurses on request, as part of their training, explaining the procedures and requirements for Gynae LBC. RECOMMENDED PROTOCOL FOR SMEAR TAKERS Only
The laboratory runs a tour of the department for Practice Nurses on request, as part of their training, explaining the procedures and requirements for Gynae LBC. RECOMMENDED PROTOCOL FOR SMEAR TAKERS Only
INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 4074 First edition 2002-02-15 Natural latex rubber condoms Requirements and test methods Préservatifs masculins en latex de caoutchouc naturel Exigences et méthodes d'essai Reference
INTERNATIONAL STANDARD ISO 4074 First edition 2002-02-15 Natural latex rubber condoms Requirements and test methods Préservatifs masculins en latex de caoutchouc naturel Exigences et méthodes d'essai Reference
Norovirus in Long Term Care Facilities Outbreak Checklist
 Norovirus in Long Term Care Facilities Outbreak Checklist Norovirus Description The typical symptoms are nausea, vomiting, fever, abdominal cramps, and watery non-bloody diarrhea. The usual incubation
Norovirus in Long Term Care Facilities Outbreak Checklist Norovirus Description The typical symptoms are nausea, vomiting, fever, abdominal cramps, and watery non-bloody diarrhea. The usual incubation
JOB A IDS for collection, storage and transport of specimens for laboratory confirmation of Middle East respiratory syndrome coronavirus (MERS-CoV)
 JOB A IDS for collection, storage and transport of specimens for laboratory confirmation of Middle East respiratory syndrome coronavirus (MERS-CoV) Integrated Disease Surveillance Programme Disease Prevention
JOB A IDS for collection, storage and transport of specimens for laboratory confirmation of Middle East respiratory syndrome coronavirus (MERS-CoV) Integrated Disease Surveillance Programme Disease Prevention
QUALITY SYSTEMS MANUAL. Allergen Management
 Page: 1 of 9 PURPOSE: To ensure all precautions are taken to prevent product contamination by products that are determined as allergens. RESPONSIBILITY: The Department Manager is responsible for ensuring
Page: 1 of 9 PURPOSE: To ensure all precautions are taken to prevent product contamination by products that are determined as allergens. RESPONSIBILITY: The Department Manager is responsible for ensuring
SECTION 10.2 NOROVIRUS (WINTER VOMITING DISEASE)
 SECTION 10.2 NOROVIRUS (WINTER VOMITING DISEASE) What is? What are the Symptoms? Spread of Infection How Infectious is the Virus? Who is at Risk of Contracting? How is treated? Prevention of Spread Why
SECTION 10.2 NOROVIRUS (WINTER VOMITING DISEASE) What is? What are the Symptoms? Spread of Infection How Infectious is the Virus? Who is at Risk of Contracting? How is treated? Prevention of Spread Why
Rapid-VIDITEST. Strep A Card. One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture.
 Rapid-VIDITEST Strep A Card One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec,
Rapid-VIDITEST Strep A Card One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec,
Human Influenza A (Swine Flu) Rapid test
 Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Infectious Material. hazmat.dot.gov/regs/rules.htm iata.org/dangerousgoods
 Infectious Material Most medical specimens sent to an esoteric laboratory such as Mayo Medical Laboratories are flown on passenger aircraft or are carried by common air carrier such as FedEx. The International
Infectious Material Most medical specimens sent to an esoteric laboratory such as Mayo Medical Laboratories are flown on passenger aircraft or are carried by common air carrier such as FedEx. The International
Bloodborne Pathogens Training
 Bloodborne Pathogens Training OSHA S Bloodborne Pathogen Standard 29CFR 1910.1030 Employers must: Develop an Exposure Control Plan (ECP) that details their Bloodborne Pathogens (BBP) Program Provide employees
Bloodborne Pathogens Training OSHA S Bloodborne Pathogen Standard 29CFR 1910.1030 Employers must: Develop an Exposure Control Plan (ECP) that details their Bloodborne Pathogens (BBP) Program Provide employees
Biospecimen Adult Urine Procedures. Event: Pre-Preg, PV1, PV2, Birth, 6M, 12M, 36M, and 60M
 Biospecimen Adult Urine Procedures Event: Pre-Preg, PV1, PV2, Birth, 6M, 12M, 36M, and 60M Domain: Type of Document: Biospecimen Standard Operating Procedures This page intentionally left blank. BIO Adult
Biospecimen Adult Urine Procedures Event: Pre-Preg, PV1, PV2, Birth, 6M, 12M, 36M, and 60M Domain: Type of Document: Biospecimen Standard Operating Procedures This page intentionally left blank. BIO Adult
Fibre wedge with Mesh
 Fibre wedge with Mesh MANUFACTURED BY SIMPLE STUFF WORKS ASSOCIATES PLEASE NOTE It is unsafe to assemble and use the equipment without fully reading the instructions 1. User Assistance If you are experiencing
Fibre wedge with Mesh MANUFACTURED BY SIMPLE STUFF WORKS ASSOCIATES PLEASE NOTE It is unsafe to assemble and use the equipment without fully reading the instructions 1. User Assistance If you are experiencing
MICROBIOLOGICAL TESTING IN PICU
 MICROBIOLOGICAL TESTING IN PICU This is a guideline for the taking of microbiological samples in PICU to diagnose or exclude infection. The diagnosis of infection requires: Ruling out non-infectious causes
MICROBIOLOGICAL TESTING IN PICU This is a guideline for the taking of microbiological samples in PICU to diagnose or exclude infection. The diagnosis of infection requires: Ruling out non-infectious causes
Bloodborne Pathogens. Montclair Kimberley Academy 1
 Bloodborne Pathogens Montclair Kimberley Academy 1 Introduction! Approximately 5.6 million workers in health care and other facilities are at risk of exposure to bloodborne pathogens such as human immunodeficiency
Bloodborne Pathogens Montclair Kimberley Academy 1 Introduction! Approximately 5.6 million workers in health care and other facilities are at risk of exposure to bloodborne pathogens such as human immunodeficiency
BLOODBORNEPATHOGENS. CAP Safety Meetings. Revision: CAP Safety Meetings [Bloodborne Pathogens]
![BLOODBORNEPATHOGENS. CAP Safety Meetings. Revision: CAP Safety Meetings [Bloodborne Pathogens] BLOODBORNEPATHOGENS. CAP Safety Meetings. Revision: CAP Safety Meetings [Bloodborne Pathogens]](/thumbs/73/68341667.jpg) BLOODBORNEPATHOGENS CAP Safety Meetings Revision: 10-2011 2011 Copyright - PEC/Premier Safety Management, Inc. All Rights Reserved Revision: [10-2011] 1 THEBLOODBORNEPATHOGENSSTANDARD The Bloodborne Pathogens
BLOODBORNEPATHOGENS CAP Safety Meetings Revision: 10-2011 2011 Copyright - PEC/Premier Safety Management, Inc. All Rights Reserved Revision: [10-2011] 1 THEBLOODBORNEPATHOGENSSTANDARD The Bloodborne Pathogens
Emergency medicine. Immobilisation. Immobilisation. Quality since 1903
 Immobilisation Immobilisation The immobilisation of emergency patients is becoming increasingly important for the fire brigade and the rescue services. Many different systems are used depending on the
Immobilisation Immobilisation The immobilisation of emergency patients is becoming increasingly important for the fire brigade and the rescue services. Many different systems are used depending on the
Bloodborne Pathogens LVHN s Annual Safety Course
 Slide 1 Bloodborne Pathogens LVHN s Annual Safety Course Nucleus Medical Media (2010). Blood Carries Nutrients. Smart Imagebase. Lehigh Valley Health Network cares about your safety especially about protecting
Slide 1 Bloodborne Pathogens LVHN s Annual Safety Course Nucleus Medical Media (2010). Blood Carries Nutrients. Smart Imagebase. Lehigh Valley Health Network cares about your safety especially about protecting
For use only with INSTRUCTIONS FOR USE
 TM For use only with INSTRUCTIONS FOR USE IMPORTANT NOTICE: Please read this safety information first. 1. Follistim Pen is a precision device. It is very important that you read and follow all directions
TM For use only with INSTRUCTIONS FOR USE IMPORTANT NOTICE: Please read this safety information first. 1. Follistim Pen is a precision device. It is very important that you read and follow all directions
FOR INFECTION TO OCCUR: Bloodborne Pathogens are viral diseases that can infect a person if they are exposed Hepatitis B Hepatitis C HIV
 OCCUPATIONAL EXPOSURE TO BLOODBORNE PATHOGENS Bloodborne Pathogens are viral diseases that can infect a person if they are exposed Hepatitis B Hepatitis C HIV FOR INFECTION TO OCCUR: A germ Bloodborne
OCCUPATIONAL EXPOSURE TO BLOODBORNE PATHOGENS Bloodborne Pathogens are viral diseases that can infect a person if they are exposed Hepatitis B Hepatitis C HIV FOR INFECTION TO OCCUR: A germ Bloodborne
Line Listing and Specimen Collection. Module 5
 Line Listing and Specimen Collection Module 5 Learning Outcomes By the end of this module you will be able to: Explain the role of the line list in outbreak management. Identify when cases should be added.
Line Listing and Specimen Collection Module 5 Learning Outcomes By the end of this module you will be able to: Explain the role of the line list in outbreak management. Identify when cases should be added.
ENG MYCO WELL D- ONE REV. 1.UN 29/09/2016 REF. MS01283 REF. MS01321 (COMPLETE KIT)
 ENG MYCO WELL D- ONE MYCO WELL D-ONE System for the presumptive identification and antimicrobial susceptibility test of urogenital mycoplasmas, Gardnerella vaginalis, Trichomonas vaginalis, Candida albicans
ENG MYCO WELL D- ONE MYCO WELL D-ONE System for the presumptive identification and antimicrobial susceptibility test of urogenital mycoplasmas, Gardnerella vaginalis, Trichomonas vaginalis, Candida albicans
Teaching Intermittent Catheterisation. Liz Croxon Clinical Facilitator National Rehabilitation Hospital Dunlaoghaire
 Teaching Intermittent Catheterisation Liz Croxon Clinical Facilitator National Rehabilitation Hospital Dunlaoghaire Aim of Workshop To provide knowledge of the principles and practises of teaching a client
Teaching Intermittent Catheterisation Liz Croxon Clinical Facilitator National Rehabilitation Hospital Dunlaoghaire Aim of Workshop To provide knowledge of the principles and practises of teaching a client
4. RED AND BLUE TOP COLLECTION SWABS WILL NO LONGER BE SUPPLIED FOR BACTERIAL CULTURE
 Laboratory Testing Update Specimen Transport Changes dated 12-24-14 SEH Laboratory is making significant changes to specimen transports! The Laboratory is introducing new testing platforms along with working
Laboratory Testing Update Specimen Transport Changes dated 12-24-14 SEH Laboratory is making significant changes to specimen transports! The Laboratory is introducing new testing platforms along with working
AMBULANCE DECONTAMINATION GUIDELINES SUSPECTED INFLUENZA PATIENT
 AMBULANCE DECONTAMINATION GUIDELINES SUSPECTED INFLUENZA PATIENT Reprinted with the Permission of John Hill, President Iowa EMS Association Following are general guidelines for cleaning or maintaining
AMBULANCE DECONTAMINATION GUIDELINES SUSPECTED INFLUENZA PATIENT Reprinted with the Permission of John Hill, President Iowa EMS Association Following are general guidelines for cleaning or maintaining
Hazardous Drug Safety
 Annual Compliance Education This course contains annual compliance education necessary to meet compliance and regulatory requirements. Instructions: To receive credit for completion: 1. Read the content
Annual Compliance Education This course contains annual compliance education necessary to meet compliance and regulatory requirements. Instructions: To receive credit for completion: 1. Read the content
Product Training & Certification
 Product Training & Certification Oratect Oral Fluid Drug Screen Device For Professional Point of Care Use Oratect Training Page 1 of 12 MKT-045 (C) Oratect Oral Fluid Drug Screen Device Training and Certification
Product Training & Certification Oratect Oral Fluid Drug Screen Device For Professional Point of Care Use Oratect Training Page 1 of 12 MKT-045 (C) Oratect Oral Fluid Drug Screen Device Training and Certification
Scottish E. coli O157/Shiga toxin-producing E. coli Reference Laboratory
 Scottish E. coli O157/Shiga toxin-producing E. coli Reference Laboratory 1 Authors: SERL68:Version L. Allison 6 Issue date & M. 27/09/2017 Hanson This is an electronic document Authors: L. Allison which
Scottish E. coli O157/Shiga toxin-producing E. coli Reference Laboratory 1 Authors: SERL68:Version L. Allison 6 Issue date & M. 27/09/2017 Hanson This is an electronic document Authors: L. Allison which
June 4, Page 1 of 5 POLICY STATEMENT
 POLICY STATEMENT This policy has been written to inform all staff, clients, relatives and other visitors to the homes of service users about the risks associated with MRSA, AIDS and HIV hazards in the
POLICY STATEMENT This policy has been written to inform all staff, clients, relatives and other visitors to the homes of service users about the risks associated with MRSA, AIDS and HIV hazards in the
Intrathecal. By the end of this chapter, you will be able to: Introduction. Chapter 20
 Chapter 20 Intrathecal Preparations By the end of this chapter, you will be able to: l Describe the specific risks with intrathecal cytotoxic products l List five checks that need to be carried out on
Chapter 20 Intrathecal Preparations By the end of this chapter, you will be able to: l Describe the specific risks with intrathecal cytotoxic products l List five checks that need to be carried out on
Irish Mycobacteria Reference Laboratory St James s Hospital
 Edition No. 09 IMRL Doc. No.: LP-IMRL-0042 Author:: Margaret Fitzgibbon Date: 20.02.17 Authorised By: Emma Roycroft Issue Date: 24.02.17 St James s Hospital User Manual IMRL User Manual Page 1 of 28 THE
Edition No. 09 IMRL Doc. No.: LP-IMRL-0042 Author:: Margaret Fitzgibbon Date: 20.02.17 Authorised By: Emma Roycroft Issue Date: 24.02.17 St James s Hospital User Manual IMRL User Manual Page 1 of 28 THE
Microbiology Collection
 Microbiology Collection Notify the Microbiology Laboratory at 920-738-6317 if any of the following etiological agents are suspected to be present in the specimens sent to the laboratory. Bacillius anthracis
Microbiology Collection Notify the Microbiology Laboratory at 920-738-6317 if any of the following etiological agents are suspected to be present in the specimens sent to the laboratory. Bacillius anthracis
SAMPLE PROCEDURE , 07/11
 SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
Rapid-VIDITEST Swine Flu
 Rapid-VIDITEST Swine Flu One Step Influenza type A Antigen Card test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz
Rapid-VIDITEST Swine Flu One Step Influenza type A Antigen Card test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz
Rapid-VIDITEST. Strep A Blister. One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture.
 Rapid-VIDITEST Strep A Blister One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II
Rapid-VIDITEST Strep A Blister One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II
Respiratory Protection for Exposures to the Influenza A (H1N1) Virus. Frequently Asked Questions (FAQs)
 3M Occupational Health and 3M Center Environmental Safety Division St. Paul, MN 55144-1000 651 733 1110 Respiratory Protection for Exposures to the Influenza A (H1N1) Virus Frequently Asked Questions (FAQs)
3M Occupational Health and 3M Center Environmental Safety Division St. Paul, MN 55144-1000 651 733 1110 Respiratory Protection for Exposures to the Influenza A (H1N1) Virus Frequently Asked Questions (FAQs)
Safety Data Sheet Product Name: Heparin / PF4 Serum Panel - For In Vitro Diagnostic Use Only
 Section 1 Identification Product Name Catalog Numbers Heparin / PF4 Assayed Antibody Serum Panel 4036027, 2 Member QC Panel 4036028, Multi-Member Qualification Panel (6 member) Company Identification AKERS
Section 1 Identification Product Name Catalog Numbers Heparin / PF4 Assayed Antibody Serum Panel 4036027, 2 Member QC Panel 4036028, Multi-Member Qualification Panel (6 member) Company Identification AKERS
CHLAMYDIA/GC AMPLIFIED RNA ASSAY
 Lab Dept: Test Name: Microbiology/Virology CHLAMYDIA/GC AMPLIFIED RNA ASSAY General Information Lab Order Codes: CGRNA Synonyms: CPT Codes: Test Includes: Chlamydia trachomatis/neisseria gonorrhoeae by
Lab Dept: Test Name: Microbiology/Virology CHLAMYDIA/GC AMPLIFIED RNA ASSAY General Information Lab Order Codes: CGRNA Synonyms: CPT Codes: Test Includes: Chlamydia trachomatis/neisseria gonorrhoeae by
