Using Diagnostic Tools for Validation and Verification of an Allergen Control Program June 2016, by The Acheson Group, TAG
|
|
|
- Milton Johnston
- 5 years ago
- Views:
Transcription
1 Using Diagnostic Tools for Validation and Verification of an Allergen Control Program June 2016, by The Acheson Group, TAG This paper is sponsored by Neogen
2 Table of Contents What You ll Learn From this Paper... 3 The Role of Testing... 3 Allergen Control Preventive Measures... 4 Methods... 4 Test and Hold... 4 Regulatory Requirements and Actions... 5 USDA... 5 FDA... 5 Labeling FALCPA... 5 Preventive Controls... 5 Current Good Manufacturing Practices... 6 Industry Requirements: GFSI... 7 Summary... 9 Our recommendations:... 9 About Our Paper Sponsor Neogen About The Acheson Group (TAG) For more information on The Acheson Group, TAG, LLC Lindsay Nix; Lindsay.Nix@AchesonGroup.com; (406) //achesongroup.com/ For more information on Neogen contact: Gerry Broski; gbroski@neogen.com; x 2361 Foodsafety.neogen.com The Acheson Group - Validation and Verification of an Allergen Control Program
3 What You ll Learn From this Paper This paper is the second in a series focused on testing. The first paper provided an overview of raw material, finished product, and environmental testing, with a focus on allergen testing. This paper will walk through how an allergen control program can play a role in assuring there are no undeclared allergens in food products. This paper is designed to: Provide the basic background information for o Raw material and finished product testing for undeclared allergens o Food contact surface testing to verify they are free of residual allergens Explain how the FDA final Preventive Controls Rule for Human Food addresses allergen controls Illustrate how FDA has updated the sanitation of food and non-food contact surfaces to emphasize allergen control Explain how allergens are addressed within GFSI schemes Describe the considerations when determining the best way to include testing in your food safety program At the end of this paper, we trust you will have a better understanding of how allergen testing can be used in the food industry, the factors influencing testing both product and food contact surfaces, how the proposed regulations have focused on the need to control allergens, and the benefits allergen testing provides. The Role of Testing The same variables that were discussed in the white paper Food Safety Testing and FSMA, GFSI, and Brand Protection should be considered for allergen testing as well. The specific reasons may be different but the importance of testing for verifying that controls are working and reducing the likelihood of undeclared allergens is the same. Similarly, when it comes to allergen testing the main types of testing that occur in a food environment are: Raw material testing o Verifies product specifications e.g., gluten-free o Ensures the supplier is executing its allergen control program Finished product testing o Confirms the formulation is accurate o Verifies cleaning and the controls to minimize cross-contact are effective Product contact o Provides verification of sanitation standard operating procedures using visual screening and rapid allergen screening tests. The Acheson Group - Validation and Verification of an Allergen Control Program
4 Allergen Preventive Control Measures Every company has the goal of protecting its consumers and its reputation. To ensure they protect allergenic consumers, companies are required to label products to be truthful and not misleading, including representing all allergens present (FALCPA 2004). The ingredient declaration is the source for the consumer to review if the product contains any allergenic substances that could be harmful. Increased vigilance for allergen control throughout the supply chain could benefit the food industry as a whole. One way to increase confidence in the supply chain is through allergen testing, both of food products and in the environment. Testing may help reduce the incidence of undeclared allergens. As reported in the FDA s report: The Reportable Food Registry: A Five Year Overview of Targeting Inspection Resources and Identifying Patterns of Adulteration: September 8, 2009-September 7, 2014, undeclared food allergens represent 47% of reportable incidences. This report is showing that, in Year 5, not only did undeclared food allergens trigger almost half of the RFR reports for the year, but also that most of the reports were associated with bakeries. Overall, dairy produced the greatest number of primary reports (24) up by nearly 150% over Year 4 (10), with half of these concerning undeclared allergens. Bakery ran a close second with 23 primary reports. This report highlights the need to enact allergen control preventive measures. Testing can play a role in this. Methods Allergen diagnostic tests can provide either a qualitative (pass/fail at a designated level) or quantitative result. Quantitative tests are most often used during the validation process and qualitative tests, often called screening tests, are used most often for verification and routine monitoring. The quantitative tests provide a numerical value to gauge cleaning and other intervention effectiveness during the validation process. The qualitative tests are often employed as a component of a preoperative inspection to determine whether to proceed with production or as a postoperative inspection to determine if sanitation efforts have been effective in removing allergens to targeted levels. The most popular and accepted diagnostic methodology for allergen detection is an enzyme-linked, immunosorbent assay, or ELISA. This technology utilizes an antibody raised against the allergenic material, most often a protein and referred to as an antigen, to capture the material from a sample. The antibody is typically linked to a second, much larger particle, often colloidal gold that enables its detection either visually through the formation of a test line on a lateral flow device or a color change in a micro well. The advantage of the ELISA method lies with its ability to detect the specific allergen of concern (its specificity) in sufficiently low concentrations (its sensitivity) to prevent harmful reactions by allergic or intolerant consumers. Test and Hold Most production facilities utilize allergen screening tests as either a post-operations check to verify cleaning efficacy or as a pre-operative check to determine that the production surfaces have been cleaned appropriately to begin production. Since these assays typically return a result in less than ten minutes, there is normally no test and hold issue. The exception can come from facilities that send out their allergen testing to commercial or corporate laboratories and then await the results before releasing product. The Acheson Group - Validation and Verification of an Allergen Control Program
5 Regulatory Requirements and Actions USDA USDA s Food Safety and Inspection Service (FSIS) has recently issued Notice 29-13, Targeted Verification of Product Formulation and Labeling for the Eight Most Common ( Big 8 ) Food Allergens. This is a notice to Inspection Plant Personnel to ensure that allergens are labeled accordingly. The IPP will accomplish the verification through review of the records and observations of the allergen control program. FDA FALCPA Currently, the Food Allergen Labeling and Consumer Protection Act (FALCPA 2004), requires food manufacturers to have risk mitigation steps in place to minimize the risk of accidental cross-contact between non-allergenic products and allergens (e.g., egg, fish and shell fish, milk, peanuts, soy, tree nuts and wheat). Food processors can voluntarily make use of allergen precautionary language such as, manufactured in the same facility as peanuts. While this statement is informative to the consumer, this type of statement cannot be used in lieu of good manufacturing practices. Therefore it is important to have a system in place to make sure that labels accurately indicate if an allergen is present. Labeling The objective of any allergen control program is to ensure that the product label accurately represents the ingredients present in the product so that consumers can make an informed decision. A significant number of allergen related recalls occur when product is mislabeled and has the potential to be contaminated with allergenic ingredients not listed on the label. One of the most important elements of an allergen control plan, (ACP), therefore, is control and coordination of all elements of the manufacturing process including a check that the proper labels are applied. This labeling requirement is also important for ingredients received from suppliers. Care must be taken to ensure that suppliers follow the same rigorous ACP standards as are required internally. The final location where product labeling is critical is in the storage areas of the facility. Allergenic material should be labeled and stored in sealed containers in designated, segregated locations within the storage facility and a plan must be in place to handle spills or other environmental contamination events. Preventive Controls The Food Safety Modernization Act (FSMA) is maintaining the elements of the FALCPA and, within the context of the final preventive controls rule for human food, it is shifting the responsibility from the final finished packaging form and the labeling to prevention and proposing to require an allergen control program with specific elements that will address cleaning and sanitation, cross-contact, and labeling (section (c) (2)). Specifically, it is proposing to require appropriate sanitation controls are in place and effective for undeclared allergens (section (c) (3). The final Preventive Controls rule for Human Food identifies the major elements of an effective allergen control program. It also places strong ownership and accountability by the facility for an effective allergen The Acheson Group - Validation and Verification of an Allergen Control Program
6 control program to eliminate the likelihood of an undeclared allergen. The Preventive Controls rule does not explicitly require testing for allergens as a mandatory part of the allergen control program. It does however identify where testing may play a role in verification and validation. For example, if a company is manufacturing a cheese filled cracker and a peanut butter filled cracker on the same production line, it would be useful to perform allergen testing on the contact surfaces. This will provide confidence in the SSOPs, demonstrating that the cleaning processes are effective at removing the allergens. For example, if a new product is being formulated to contain eggs, and there are no eggs used on that line today, there are a number of steps that could be considered to ensure allergen control. But looking at this through the lenses of allergen cross-contact cleaning validation and verification, scheduling the line time and effective cleaning and sanitation are key. Since no other products contain egg, then this product should be run last (albeit considering other allergens on the line as well) and the cleaning and sanitation may easily be validated by performing allergen-specific quantitative testing on the different equipment surface types to ensure the procedures are effective. This can then be verified by qualitative allergen tests. The FDA is not requiring validation for allergen controls in the way that validation is required for process controls. However, if there are ways to validate that cross-contact is avoided, for example, by doing a study where the amount of an allergen can be quantified after a specified sanitation process is employed, this is recommended. Current Good Manufacturing Practices While most people equate preventive controls with the new requirements to control specific hazards, the FDA final rule also updates current good manufacturing practices, and one of the most notable updates of cgmps relates to allergen control, with 23 changes to clearly distinguish requirements directed to allergen cross-contact. The basis for these is to prevent a failure to: properly clean and sanitize control movement of products, traffic and allergenic dust/aerosols properly store ingredients, works-in-process, and rework develop and maintain accurate and not misleading labels This increased focus on GMPs for allergens sends a strong signal to the food industry that the FDA is serious. What s more, the revisions are changing from recommendations to requirements. Industry members can protect their brands by developing and maintaining an allergen control program to ensure there is a significantly reduced possibility of an undeclared allergen. So where do you start? The best place to start is to know the risk, performing a risk assessment of the ingredients. The risk assessment includes an evaluation of the inherent risk (e.g., is the product an allergen or contain an allergenic component?). Secondly, consider the supplier. Examples of some areas to review can be the history of specification compliance, third party audit performance, have they had any recalls/ withdrawals, do they manufacture other products with allergens, etc. Once the risk is assessed it is time to determine how to best mitigate that potential risk. Mitigation strategies can include auditing the suppliers, and requiring testing of the ingredient to be reported on the certificate of analysis. This can assist in understanding the risk coming into the facility. The Acheson Group - Validation and Verification of an Allergen Control Program
7 The risk assessment and mitigation does not stop at receiving. Next, you must know the facility and production. There are a number of activities that can be controlled to reduce the risk of unintentional allergen cross-contact. For example if using an allergen or a product containing an allergen that generates dust or a liquid that can aerosolize, the air handling should be considered. This topic is addressed in the cgmp update on plant and grounds, section Also consider if there are overlapping traffic patterns or movement of goods that could move these potential allergens around. Scheduling of production plays a role as well to minimize the potential for cross-contact by scheduling least to most allergens, (section (c)). Once these risks have been assessed they will need effective mitigation. Cleaning and sanitation are key components of an allergen control program. There are a number of areas to consider for effective allergen cleaning. If the product scheduling is such that the most allergen contain products are produced last, cleaning must ensure that the potential for the start-up of the least allergen containing products are free from the risk of cross-contact. This is true of all the utensils that are used on that production line or in that area as well, as well as employees hands (section (c (2))). In addition to visual inspection, testing the different product surfaces that are exposed to the allergens can initially validate the SSOPs. Testing can also be used at set frequencies to continue to verify the removal of all the target allergens from those surfaces. These set frequencies can be determined and included as a part of the Master Sanitation Schedule or as part of your food safety plan. Practices should be reconsidered, for example, when a formula changes, or when a new product is added to that production line. Other allergen cleaning considerations include how to effectively and demonstrably remove dust and aerosols near the production line processing allergens or from traffic moving this around the facility. One item that may be overlooked is cleaning the cleaning equipment. As stated earlier, validated and verified sanitation as part of an overall allergen control program is key. And any testing information that is applicable to managing these hazards plays a strong role in the verification of the overall program. It can help provide the confidence that the allergen hazards are under control. Industry Requirements: GFSI Increasingly, GFSI has become a major force within much of the food industry. GFSI set forth a guidance document outlining expectations of food safety management systems and different audit schemes have been benchmarked against the GFSI standard. Every GFSI scheme has a section that identifies the requirements for allergen management. SQF has published an allergen guidance document to further define what their specific GFSI scheme requires for allergen control. This document goes into very specific details on validation and verification. The expectation is that a manufacturing facility must document that their procedures will significantly reduce the likelihood of an unintentional allergen being present in a finished product. This requirement for validation is more than just a finished product test revealing that the allergen of concern is not present. The sanitation validation must include The Acheson Group - Validation and Verification of an Allergen Control Program
8 testing evidence, where a test is available, that the specific allergen of concern has been removed or reduced to an acceptable level from contact surfaces. SQF has listed accepted methods such as ELISA and lateral flow. Another GFSI benchmarked scheme, BRC, also has detailed allergen control requirements. Similar to SQF the cleaning methods in a facility must be validated and verified to ensure they remove or reduce the allergen to acceptable levels. Another scheme, FSSC 22000, does not mention anything specifically about validation or verification but the allergen requirement states products shall be protected from unintended allergens. SQF, BRC, and FSSC note testing as an option to verify the conformity of raw materials, and allergens could be considered as part of the specification. FSSC also mentions testing in the context of verification planning, noting that testing can be used to verify the overall effect of prerequisite programs, operational PRPs and elements within the HACCP plan, and generally to make sure that hazard levels are within identified acceptable levels. The Acheson Group - Validation and Verification of an Allergen Control Program
9 Summary Because there is no cure for food allergies the food industry must protect its consumers and brands with an effective allergen control program. The ultimate goal of an allergen control program is to provide truthful and not misleading product labels with accurate allergen information for consumers. In order to do this, manufacturers must know if allergens are present in the products, including the possibility of inadvertent inclusion due to cross-contact. Many facilities handle both allergen containing and non-allergen containing products, and must be vigilant about practices within the facility to limit transfer. While finished product testing can verify that a system is under control, it is reactive and in some cases testing is impractical, either because of food production (workflow) issues, detection issues related processing effects on the allergen, or because it is difficult to gain statistically significant results. Preventing allergen cross-contact is preferred, and one mechanism to do this is through thorough cleaning and sanitation. Testing can be used as a tool to define and verify thorough. Today, both qualitative and quantitative tests are available for many of the major allergens. With FDA s increased focus on cross-contact of allergens (both as part of preventive controls and cgmps), and the detailed requirements of several GFSI schemes, facilities would be wise to carefully assess how they are currently controlling allergens. Our Recommendations: The allergen control plan is intended to provide systematic structure to prevent cross-contact, develop and maintain accurate and truthful product labels, and provide safe products to consumers. The elements to build a strong allergen control plan include: assessing allergens in the facility when performing the hazard assessment assessing the adequacy of sanitation o Validation and verification through product contact testing including frequencies of such activities What s important is that the methods used and the frequency of implementation provide confidence the risks are mitigated. Additional elements to control allergens include good hygienic practices, product scheduling, traffic control, and labeling. In addition to all the preventive measures, finished product testing can provide further confidence in the effectiveness of the allergen control program. The Acheson Group - Validation and Verification of an Allergen Control Program
10 About Our Paper Sponsor Neogen Neogen is a leader in developing and marketing diagnostic test kits that provide food safety solutions. Its products are unsurpassed in terms of ease of use, convenience and speed. Neogen s comprehensive line of rapid food safety diagnostics includes: Food Allergens Neogen offers food allergen test kits to detect almond, egg, gliadin (gluten), hazelnut, crustacea, total milk, mustard, peanut, sesame, shellfish, soy, and walnut residues. Neogen provides allergen test kits in multiple formats for validation, verification and finished product and environmental, (contact surface), testing. Foodborne Pathogens Neogen provides multiple different pathogen detection systems. Neogen s revolutionary new ANSR system is the quickest and easiest testing method to definitively detect pathogen DNA in food and environmental samples providing results in as little as 10 minutes. Reveal 2.0 tests for pathogens are fast and easy immunological lateral flow devices, which can be used at nearly any facility. General Microbiology Neogen s Soleris system rapid detects microbial contamination by monitoring the color differences produced by changing ph or CO 2. Using Soleris, time to results for general microbiological assays are reduced by up to 60%. Neogen offers NEO-GRID and ISO-GRID disposable filter membrane system and culture media for rapid and easy enumeration of bacteria in liquids. Natural Toxins (including mycotoxins) Neogen s new Reveal Q+ fully quantitative lateral flow test for mycotoxins are quick and easy, with results in just minutes. Agri-Screen and Veratox mycotoxin tests kits provide screening and quantitative ELISA-based test for aflatoxin, DON, fumonisin, zearalenone, ochratoxin, and T-2/HT-2. Sanitation Neogen s AccuPoint Advanced sanitation monitoring system quickly and easily measures ATP collected from food contact surfaces or liquids as a verification of sanitation programs. DataManager software allows for complete data traceability and data analysis. About The Acheson Group, TAG The Acheson Group (TAG) brings clients practical food safety solutions. Food companies today worry about operational efficiency and risk control, brand reputation and complying with current and new regulations. TAG brings a unique suite of services that provides deep industry expertise and a global perspective focused on assessing and managing operational risk, reputational risk and regulatory risk. Our team is comprised of noted industry leaders with global experience in food safety and defense. Our clients span farm to fork and include technology companies looking to assist all sectors of the food industry. The Acheson Group - Validation and Verification of an Allergen Control Program
Food Allergen Environmental Monitoring Guide
 Food Allergen Environmental Monitoring Guide A EUROFINS WHITE PAPER OCTOBER 2012 This document by Eurofins is licensed under a Creative Commons Attribution 3.0 Unported License. A COMPREHENSIVE GUIDE TO
Food Allergen Environmental Monitoring Guide A EUROFINS WHITE PAPER OCTOBER 2012 This document by Eurofins is licensed under a Creative Commons Attribution 3.0 Unported License. A COMPREHENSIVE GUIDE TO
Allergens Assessing risk and mapping your facility
 Allergens Assessing risk and mapping your facility Evaluate and mitigate your allergen risks Allergen mapping is a part of allergen risk assessment and is an effective tool to identify and track allergens
Allergens Assessing risk and mapping your facility Evaluate and mitigate your allergen risks Allergen mapping is a part of allergen risk assessment and is an effective tool to identify and track allergens
TIP SHEET 17 ALLERGEN MANAGEMENT APPLICABLE CODE ELEMENT(S) LEARNING OBJECTIVES O CONTROL AND PREVENT THE SOURCES OF ALLERGENS O 2.8.
 ALLERGEN MANAGEMENT Food allergies affect a small but growing proportion of the population. They are most common in children, although some can be life-long, and can cause mild to severe and sometimes
ALLERGEN MANAGEMENT Food allergies affect a small but growing proportion of the population. They are most common in children, although some can be life-long, and can cause mild to severe and sometimes
Allergen Control in the Food Plant
 Allergen Control in the Food Plant Presented by: Melissa Calicchia, M.S., Senior Technical Director Food Safety Solutions, Inc. Food Microbiological Laboratories, Inc. Presented to: Southern California
Allergen Control in the Food Plant Presented by: Melissa Calicchia, M.S., Senior Technical Director Food Safety Solutions, Inc. Food Microbiological Laboratories, Inc. Presented to: Southern California
This whitepaper will cover the suggested methodology to be employed when developing a PCP plan. This includes how to develop your plan in accordance
 This whitepaper will cover the suggested methodology to be employed when developing a PCP plan. This includes how to develop your plan in accordance with FSMA s Final Rule for Preventive Controls for Human
This whitepaper will cover the suggested methodology to be employed when developing a PCP plan. This includes how to develop your plan in accordance with FSMA s Final Rule for Preventive Controls for Human
Reducing the risk of allergen contamination in the factory
 Reducing the risk of allergen contamination in the factory Barbara Hirst RSSL Sponsored by Reactions To Food ADVERSE REACTIONS TO FOOD GENERIC May occur in anyone who consumes sufficient quantity of the
Reducing the risk of allergen contamination in the factory Barbara Hirst RSSL Sponsored by Reactions To Food ADVERSE REACTIONS TO FOOD GENERIC May occur in anyone who consumes sufficient quantity of the
ALLERGENS. Many different foods can cause an allergic reaction, however; the majority of food allergies are caused by: Peanuts, Soybeans, Milk, Eggs,
 ALLERGENS People can eat most food safely. But, imagine what it would be like if eating a peanut butter sandwich or drinking a glass of milk left you vomiting, gasping for breath, and furiously scratching
ALLERGENS People can eat most food safely. But, imagine what it would be like if eating a peanut butter sandwich or drinking a glass of milk left you vomiting, gasping for breath, and furiously scratching
Precautionary Allergen Labelling (PAL) and the effects for the fruit and vegetable processing industry
 PROFEL.2018.006 07 March 2018 Precautionary Allergen Labelling (PAL) and the effects for the fruit and vegetable processing industry Introduction The Dutch delegation raised the issue of non-homogeneous
PROFEL.2018.006 07 March 2018 Precautionary Allergen Labelling (PAL) and the effects for the fruit and vegetable processing industry Introduction The Dutch delegation raised the issue of non-homogeneous
Benchmarking Allergen Management to GFSI Requirements
 Benchmarking Allergen Management to GFSI Requirements Bill McBride, Foodlink Management Services, Australia representing GFSI & SQF Global Food Safety Initiative (GFSI) The Global Food Safety Initiative
Benchmarking Allergen Management to GFSI Requirements Bill McBride, Foodlink Management Services, Australia representing GFSI & SQF Global Food Safety Initiative (GFSI) The Global Food Safety Initiative
Allergy Inspection Guide (4/01)
 Allergy Inspection Guide (4/01) GUIDANCE ON INSPECTIONS OF FIRMS PRODUCING FOOD PRODUCTS SUSCEPTIBLE TO CONTAMINATION WITH ALLERGENIC INGREDIENTS This guidance is reference material for investigators and
Allergy Inspection Guide (4/01) GUIDANCE ON INSPECTIONS OF FIRMS PRODUCING FOOD PRODUCTS SUSCEPTIBLE TO CONTAMINATION WITH ALLERGENIC INGREDIENTS This guidance is reference material for investigators and
Code of Practice Version 2 Management of Food Allergens
 Code of Practice Version 2 Management of Food Allergens Contents Page No. 1. Introduction & Objective 3 2. Risk Assessment 4 3. Allergen Management 8 Appendix 1 10 Appendix 2 11 Appendix 3 12 Appendix
Code of Practice Version 2 Management of Food Allergens Contents Page No. 1. Introduction & Objective 3 2. Risk Assessment 4 3. Allergen Management 8 Appendix 1 10 Appendix 2 11 Appendix 3 12 Appendix
Cleaning and Allergens
 Introduction The prevalence of people with life-threatening food allergies is increasing. It is now believed as many as 1-2% of adults and 5-8% of children are affected. Avoidance of allergen-containing
Introduction The prevalence of people with life-threatening food allergies is increasing. It is now believed as many as 1-2% of adults and 5-8% of children are affected. Avoidance of allergen-containing
July 27-30, th Annual Meeting of the Poultry Science Association Louisville, Kentucky. Presented at:
 July 27-30, 2015 104 th Annual Meeting of the Poultry Science Association Louisville, Kentucky Presented at: From Egg to Plate The Influence of Gut Health Symposium Presentations distributed by DuPont
July 27-30, 2015 104 th Annual Meeting of the Poultry Science Association Louisville, Kentucky Presented at: From Egg to Plate The Influence of Gut Health Symposium Presentations distributed by DuPont
FDA Food Safety Modernization Act (FSMA) January 4, 2011
 FDA Food Safety Modernization Act (FSMA) January 4, 2011 FDA Food Safety Modernization Act (FSMA) The Most Sweeping Reform of the U.S. Food Safety Laws in More than 70 Years FDA Food Safety Modernization
FDA Food Safety Modernization Act (FSMA) January 4, 2011 FDA Food Safety Modernization Act (FSMA) The Most Sweeping Reform of the U.S. Food Safety Laws in More than 70 Years FDA Food Safety Modernization
FOOD SERVICES FOOD ALLERGENS: ANALYTICAL RISK ASSESSMENT
 FOOD SERVICES FOOD ALLERGENS: ANALYTICAL RISK ASSESSMENT FOOD ALLERGENS Accurate and reliable testing to protect consumers Each year, millions of people have allergic reactions to food. Although most food
FOOD SERVICES FOOD ALLERGENS: ANALYTICAL RISK ASSESSMENT FOOD ALLERGENS Accurate and reliable testing to protect consumers Each year, millions of people have allergic reactions to food. Although most food
Lab Services Sample Testing Information
 Commitment to Excellence Today s food industry is growing rapidly as it tries to address the global food challenge. With this growth comes an increase in demand for diagnostic testing. Food producers are
Commitment to Excellence Today s food industry is growing rapidly as it tries to address the global food challenge. With this growth comes an increase in demand for diagnostic testing. Food producers are
SUPPLIER INGREDIENT INFORMATION. 50 lb bag
 SUPPLIER INGREDIENT INFORMATION CHOLMONDLEY'S ENGLISH MUFFIN LOAF BASE BRAND: ORTH 3206354-00 Cholmondley's English Muffin Loaf Base is a pre-blended, fully prepared, dry mix base for using in bread applications.
SUPPLIER INGREDIENT INFORMATION CHOLMONDLEY'S ENGLISH MUFFIN LOAF BASE BRAND: ORTH 3206354-00 Cholmondley's English Muffin Loaf Base is a pre-blended, fully prepared, dry mix base for using in bread applications.
Potential Hazards in Food; Ensuring the Concept of Tayyab in Food. Mian N. Riaz, Ph.D
 Potential Hazards in Food; Ensuring the Concept of Tayyab in Food Mian N. Riaz, Ph.D Director - Food Protein R & D Center; Texas A&M University, Texas, USA E-mail: mnriaz@tamu.edu Abstract: Halal means
Potential Hazards in Food; Ensuring the Concept of Tayyab in Food Mian N. Riaz, Ph.D Director - Food Protein R & D Center; Texas A&M University, Texas, USA E-mail: mnriaz@tamu.edu Abstract: Halal means
Food Commissaries under FSMA and the US FDA model Food Code
 Food Commissaries under FSMA and the US FDA model Food Code Introduction A food commissary is a facility or operation that procures and/or produces foods intended for distribution. A retail or foodservice
Food Commissaries under FSMA and the US FDA model Food Code Introduction A food commissary is a facility or operation that procures and/or produces foods intended for distribution. A retail or foodservice
ARE YOU READY FOR RETAIL?
 ARE YOU READY FOR RETAIL? JAY SHIRODKER AGENDA Current landscape of food safety oversight What is changing for Manufacturers, Retail, Consumers Drivers of changes How is retail industry impacted Food Safety
ARE YOU READY FOR RETAIL? JAY SHIRODKER AGENDA Current landscape of food safety oversight What is changing for Manufacturers, Retail, Consumers Drivers of changes How is retail industry impacted Food Safety
FDA RELEASES FOOD CURRENT GOOD MANUFACTURING PRACTICE MODERNIZATION REPORT
 November 17, 2005 FDA RELEASES FOOD CURRENT GOOD MANUFACTURING PRACTICE MODERNIZATION REPORT Executive Summary On November 2, 2005, FDA issued the Food CGMP Modernization - A Focus on Food Safety report.
November 17, 2005 FDA RELEASES FOOD CURRENT GOOD MANUFACTURING PRACTICE MODERNIZATION REPORT Executive Summary On November 2, 2005, FDA issued the Food CGMP Modernization - A Focus on Food Safety report.
Allergen Management, Supporting Your Business
 Allergen Management, Supporting Your Business Ruth Bell, AF Associates International Food Safety and Quality Management Specialists office@af-associates.co.uk www.af-associates.co.uk Ruth Bell MSc, FIFST
Allergen Management, Supporting Your Business Ruth Bell, AF Associates International Food Safety and Quality Management Specialists office@af-associates.co.uk www.af-associates.co.uk Ruth Bell MSc, FIFST
AKA Good Manufacturing Practice (GMP) Certification Program
 AKA Good Manufacturing Practice (GMP) Certification Program Preamble The American Kratom Association (AKA) is establishing this program to assure the safety and integrity of kratom dietary supplements
AKA Good Manufacturing Practice (GMP) Certification Program Preamble The American Kratom Association (AKA) is establishing this program to assure the safety and integrity of kratom dietary supplements
QUALITY SYSTEMS MANUAL. Allergen Management
 Page: 1 of 9 PURPOSE: To ensure all precautions are taken to prevent product contamination by products that are determined as allergens. RESPONSIBILITY: The Department Manager is responsible for ensuring
Page: 1 of 9 PURPOSE: To ensure all precautions are taken to prevent product contamination by products that are determined as allergens. RESPONSIBILITY: The Department Manager is responsible for ensuring
Final Rule for Preventive Controls for Animal Food
 Final Rule for Preventive Controls for Animal Food http://www.fda.gov/fsma THE FUTURE IS NOW 1 Background Current Good Manufacturing Practice, Hazard Analysis, and Risk-Based Preventive Controls for Food
Final Rule for Preventive Controls for Animal Food http://www.fda.gov/fsma THE FUTURE IS NOW 1 Background Current Good Manufacturing Practice, Hazard Analysis, and Risk-Based Preventive Controls for Food
Food Safety Modernization Act (FSMA) Overview
 Food Safety Modernization Act (FSMA) Overview Achyut Adhikari Assistant Professor & Extension Food Safety Specialist School of Nutrition and Food Sciences Louisiana State University AgCenter Why Food Safety?
Food Safety Modernization Act (FSMA) Overview Achyut Adhikari Assistant Professor & Extension Food Safety Specialist School of Nutrition and Food Sciences Louisiana State University AgCenter Why Food Safety?
Managing Allergens within a manufacturing context. Helen Brown CampdenBRI Chipping Campden
 Managing Allergens within a manufacturing context Helen Brown CampdenBRI Chipping Campden Food Allergens An unusual food safety issue Harmless natural substances however In allergic individuals cause reactions,
Managing Allergens within a manufacturing context Helen Brown CampdenBRI Chipping Campden Food Allergens An unusual food safety issue Harmless natural substances however In allergic individuals cause reactions,
FSMA & The Dietary Supplements Industry
 FSMA & The Dietary Supplements Industry Overview FDA Food Safety Modernization Act (FSMA) Preventive Controls for Human Food (PCHF) Foreign Supplier Verification Programs (FSVP) Produce Verification activities
FSMA & The Dietary Supplements Industry Overview FDA Food Safety Modernization Act (FSMA) Preventive Controls for Human Food (PCHF) Foreign Supplier Verification Programs (FSVP) Produce Verification activities
Laboratories & Consulting Group
 Rapid Pathogen Screening E.coli O157 EHEC Non-O157 STEC Standard Microbiology Testing Total, Aerobic, & Anaerobic Plate Count Generic E. coli and Coliforms Staphylococcus aureus Yeast & Mold Microbial
Rapid Pathogen Screening E.coli O157 EHEC Non-O157 STEC Standard Microbiology Testing Total, Aerobic, & Anaerobic Plate Count Generic E. coli and Coliforms Staphylococcus aureus Yeast & Mold Microbial
Alignment of FSMA with Existing Food Safety Programs International Citrus & Beverage Conference
 Alignment of FSMA with Existing Food Safety Programs International Citrus & Beverage Conference Donald Kautter US Food and Drug Administration Center for Food Safety and Applied Nutrition Office of Food
Alignment of FSMA with Existing Food Safety Programs International Citrus & Beverage Conference Donald Kautter US Food and Drug Administration Center for Food Safety and Applied Nutrition Office of Food
Chapter 2 Keeping Food Safe
 Chapter 2 Keeping Food Safe Copyright 2011 by the National Restaurant Association Educational Foundation (NRAEF) and published by Pearson Education, Inc. All rights reserved. 2.1 Introduction to Food Safety
Chapter 2 Keeping Food Safe Copyright 2011 by the National Restaurant Association Educational Foundation (NRAEF) and published by Pearson Education, Inc. All rights reserved. 2.1 Introduction to Food Safety
Allergen Management The View and Experiences of DFSV
 Allergen Management The View and Experiences of DFSV FAMS2017 Rob Antonic Food Safety Manager Overview DFSV and our Key Functions. Regulatory obligations of manufacturers. What is an allergen management
Allergen Management The View and Experiences of DFSV FAMS2017 Rob Antonic Food Safety Manager Overview DFSV and our Key Functions. Regulatory obligations of manufacturers. What is an allergen management
CSM Bakery Solutions 1912 Montreal Road Tucker GA
 CSM Bakery Solutions 1912 Montreal Road Tucker GA 30084 770-938-3823 PRODUCT DATA SHEET www.csmbakerysolutions.com CIB 300 CHOCOLATE ICING BASE MATERIAL CODES Article number CSM article number 10179662
CSM Bakery Solutions 1912 Montreal Road Tucker GA 30084 770-938-3823 PRODUCT DATA SHEET www.csmbakerysolutions.com CIB 300 CHOCOLATE ICING BASE MATERIAL CODES Article number CSM article number 10179662
Availability of FSIS Compliance Guidelines for Allergens and. Ingredients of Public Health Concern: Identification, Prevention
 This document is scheduled to be published in the Federal Register on 11/16/2015 and available online at http://federalregister.gov/a/2015-28935, and on FDsys.gov Billing Code 3410-DM-P DEPARTMENT OF AGRICULTURE
This document is scheduled to be published in the Federal Register on 11/16/2015 and available online at http://federalregister.gov/a/2015-28935, and on FDsys.gov Billing Code 3410-DM-P DEPARTMENT OF AGRICULTURE
Allergen and Gluten Free Options Use the following steps to determine if you should pursue the allergen/gluten free market
 Allergen and Gluten Free Options Use the following steps to determine if you should pursue the allergen/gluten free market Step 1: Watch the video, A Food Manufacturer s Guide to Allergens & Gluten. If
Allergen and Gluten Free Options Use the following steps to determine if you should pursue the allergen/gluten free market Step 1: Watch the video, A Food Manufacturer s Guide to Allergens & Gluten. If
Why all the Attention to Food Safety?
 Why all the Attention to Food Safety? IFST London April 23, 2015 By Gale Prince, CFS SAGE Food Safety LLC Presentation Agenda Historical Trends in food safety issues Detecting food safety issues Consumer
Why all the Attention to Food Safety? IFST London April 23, 2015 By Gale Prince, CFS SAGE Food Safety LLC Presentation Agenda Historical Trends in food safety issues Detecting food safety issues Consumer
GMA Industry Handbook for Safe Processing of Nuts Building on Salmonella Guidance for Low-Moisture Foods
 GMA Industry Handbook for Safe Processing of Nuts Building on Salmonella Guidance for Low-Moisture Foods NPSA Annual Meeting Memphis, Tennessee September 16-18, 2010 Glenn Black, Ph.D. Director of Science
GMA Industry Handbook for Safe Processing of Nuts Building on Salmonella Guidance for Low-Moisture Foods NPSA Annual Meeting Memphis, Tennessee September 16-18, 2010 Glenn Black, Ph.D. Director of Science
Coles Supermarkets Customer Focused Allergen Management & Labelling. Neil McSkimming Policy & Legislation Manager - Food Coles Quality Team
 Coles Supermarkets Customer Focused Allergen Management & Labelling Neil McSkimming Policy & Legislation Manager - Food Coles Quality Team September 2008 1 Coles a customer focused retailer 750 + supermarkets
Coles Supermarkets Customer Focused Allergen Management & Labelling Neil McSkimming Policy & Legislation Manager - Food Coles Quality Team September 2008 1 Coles a customer focused retailer 750 + supermarkets
Cleaning Verification Tools. Joe Cardamone, General Manager Cell Biosciences Pty Ltd
 Cleaning Verification Tools Joe Cardamone, General Manager Cell Biosciences Pty Ltd Welcome and Introduction Why is Cleaning and Cleaning Verification important? Risk Food Standards Code Definitions Tools
Cleaning Verification Tools Joe Cardamone, General Manager Cell Biosciences Pty Ltd Welcome and Introduction Why is Cleaning and Cleaning Verification important? Risk Food Standards Code Definitions Tools
# 2371 SOUR CREAM AND CHIVES SAVORY
 Product Overview: Dry, free flowing powder produced with a uniform slightly tart cream and onion flavor. Maltodextrin based product able to maintain quality and consistency of color and flavor. Product
Product Overview: Dry, free flowing powder produced with a uniform slightly tart cream and onion flavor. Maltodextrin based product able to maintain quality and consistency of color and flavor. Product
Allergen Cleaning, Verification & Validation
 Allergen Cleaning, Verification & Validation Dan Schmitz Director of Operations Commercial Food Sanitation An Intralox company Strategic consulting Training and education Providing durable solutions to
Allergen Cleaning, Verification & Validation Dan Schmitz Director of Operations Commercial Food Sanitation An Intralox company Strategic consulting Training and education Providing durable solutions to
Ingredient Statement: Dextrose, Natural & Artificial Flavor, FD&C Red #40 (E129), FD&C Red #3 (E127).
 Product Overview: Dry, free flowing granular powder produced with a red color and uniform strawberry flavor and aroma. Strawberry Concentrate Corn Treat Mix is a dextrose base product able to maintain
Product Overview: Dry, free flowing granular powder produced with a red color and uniform strawberry flavor and aroma. Strawberry Concentrate Corn Treat Mix is a dextrose base product able to maintain
FSMA: Implementation and Impact on Global food Industry and role of FSPCA in Preventive Controls Education and Outreach
 FSMA: Implementation and Impact on Global food Industry and role of FSPCA in Preventive Controls Education and Outreach 56th Annual Tanner Lecture Chicago section IFT Purnendu C. Vasavada Professor Emeritus,
FSMA: Implementation and Impact on Global food Industry and role of FSPCA in Preventive Controls Education and Outreach 56th Annual Tanner Lecture Chicago section IFT Purnendu C. Vasavada Professor Emeritus,
Final Rules Preventive Controls for Human Food Preventive Controls for Animal Food
 Final Rules Preventive Controls for Human Food Preventive Controls for Animal Food http://www.fda.gov/fsma THE FUTURE IS NOW 1 Current Good Manufacturing Practice, Hazard Analysis, and Risk-Based Preventive
Final Rules Preventive Controls for Human Food Preventive Controls for Animal Food http://www.fda.gov/fsma THE FUTURE IS NOW 1 Current Good Manufacturing Practice, Hazard Analysis, and Risk-Based Preventive
An Introduction to Modern Food Safety & Quality Management
 An Introduction to Modern Food Safety & Quality Management Natalie Ohanessian Food Happy Consulting Produced by An Introduction to Modern Food Safety & Quality Management Natalie Ohanessian, CFS, PCQI
An Introduction to Modern Food Safety & Quality Management Natalie Ohanessian Food Happy Consulting Produced by An Introduction to Modern Food Safety & Quality Management Natalie Ohanessian, CFS, PCQI
Product Name and Number: 2710 Sunny Pop Popcorn Document #: Revision Date:10/15/2014 Revision #: 1 Revision Reason: New form Reviser: JS
 Product Overview: Sunny Pop Popcorn is a great tasting popcorn that is made with three simple ingredients and meets or exceeds school nutritional guidelines. Product Physical Properties Flavor / Odor (sensory)
Product Overview: Sunny Pop Popcorn is a great tasting popcorn that is made with three simple ingredients and meets or exceeds school nutritional guidelines. Product Physical Properties Flavor / Odor (sensory)
Allergen Environmental Monitoring
 Allergen Environmental Monitoring Douglas L. Marshall, Ph.D., CFS Chief Scientific Officer Eurofins Microbiology Laboratories douglasmarshall@eurofinsus.com Texas Association for Food Protection Annual
Allergen Environmental Monitoring Douglas L. Marshall, Ph.D., CFS Chief Scientific Officer Eurofins Microbiology Laboratories douglasmarshall@eurofinsus.com Texas Association for Food Protection Annual
Product Information Sheet
 Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Food Purchasing & Receiving Review
 Food Purchasing & Receiving Review Food Purchasing and Receiving 1. The first step in maintaining the safety of the food you prepare and serve, is to ensure that the food is safe as it enters your establishment.
Food Purchasing & Receiving Review Food Purchasing and Receiving 1. The first step in maintaining the safety of the food you prepare and serve, is to ensure that the food is safe as it enters your establishment.
FOOD SERVICES FOOD SAFETY: LIMITS OF CONTAMINATION
 FOOD SERVICES FOOD SAFETY: LIMITS OF CONTAMINATION FOOD CONTAMINATION Food manufacturers need to ensure that their customers are protected Consumers are becoming increasingly concerned about the safety
FOOD SERVICES FOOD SAFETY: LIMITS OF CONTAMINATION FOOD CONTAMINATION Food manufacturers need to ensure that their customers are protected Consumers are becoming increasingly concerned about the safety
Food Safety Modernization Act - Impacts on the Grain and Feed Industry
 Food Safety Modernization Act - Impacts on the Grain and Feed Industry NGFA 117th Annual Meeting and Convention March 17, 2013 San Francisco, California David Fairfield, NGFA Vice President of Feed Services
Food Safety Modernization Act - Impacts on the Grain and Feed Industry NGFA 117th Annual Meeting and Convention March 17, 2013 San Francisco, California David Fairfield, NGFA Vice President of Feed Services
EXPORTING TO THE USA. Andy Kerridge Primority Ltd
 EXPORTING TO THE USA Andy Kerridge Primority Ltd ak@primority.com F.S.M.A. The US FDA's Food Safety Modernization Act is the most exhaustive reform of US food safety laws in >70 years. Signed into law
EXPORTING TO THE USA Andy Kerridge Primority Ltd ak@primority.com F.S.M.A. The US FDA's Food Safety Modernization Act is the most exhaustive reform of US food safety laws in >70 years. Signed into law
Product Information Sheet REZISTA HV
 Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Product Information Sheet REZISTA DF
 Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Best Practices for Food Allergen Validation & Verification
 Best Practices for Food Allergen Validation & Verification CONTENTS Food Production Allergen Validation and Verification of the Environment...2 Cleaning to a Validated Standard...2 Migrating from Validation
Best Practices for Food Allergen Validation & Verification CONTENTS Food Production Allergen Validation and Verification of the Environment...2 Cleaning to a Validated Standard...2 Migrating from Validation
Product Information Sheet CLARIA Elite
 Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Product Information Sheet
 Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Proposed FDA Food Safety Modernization Act:
 Proposed FDA Food Safety Modernization Act: Its impact on distillers production and sales Paula Emberland Business Analyst History of the FDA 1862 Beginning of Bureau of Chemistry 1927 Bureau of Chemistry
Proposed FDA Food Safety Modernization Act: Its impact on distillers production and sales Paula Emberland Business Analyst History of the FDA 1862 Beginning of Bureau of Chemistry 1927 Bureau of Chemistry
HALAL FOOD AND SAFETY COMPLIANCE AGROASIA 2011 HALAL INDUSTRY RESEARCH CENTRE
 HALAL FOOD AND SAFETY COMPLIANCE AGROASIA 2011 July 2010 Page 1 Revolutionizing Assistance in the disposal Industry of in Prime a Shariah Liquid Compliant Emulsion way Industries! Halal Industry Research
HALAL FOOD AND SAFETY COMPLIANCE AGROASIA 2011 July 2010 Page 1 Revolutionizing Assistance in the disposal Industry of in Prime a Shariah Liquid Compliant Emulsion way Industries! Halal Industry Research
Food ingredients Suppliers management
 Food ingredients Suppliers management Introduction This paper reviews the process of ingredient supplier approval and selection. UK food manufacturing sector relies on suppliers to guarantee the safety
Food ingredients Suppliers management Introduction This paper reviews the process of ingredient supplier approval and selection. UK food manufacturing sector relies on suppliers to guarantee the safety
Product Information Sheet SODA-LO EXTRA FINE IODIZED
 Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Re: National Bioengineered Food Disclosure Standard; Proposed Rule; Request for Comments, 83 Fed. Reg (May 4, 2018), Docket No.
 VIA ELECTRONIC SUBMISSION July 3, 2018 Dockets Management Staff (HFA-305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Re: National Bioengineered Food Disclosure Standard;
VIA ELECTRONIC SUBMISSION July 3, 2018 Dockets Management Staff (HFA-305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Re: National Bioengineered Food Disclosure Standard;
POLICY & PROCEDURE FOOD HYGIENE POLICY DATE OF ADOPTION Related Documents: The Professional Food Handlers Guide
 Responsible Department Link POLICY & PROCEDURE Original Date of Adoption 07.06.16 Current Date of Adoption 07.06.16 Audit Committee Review Date - Date of Review 07.06.18 TITLE FOOD HYGIENE POLICY Related
Responsible Department Link POLICY & PROCEDURE Original Date of Adoption 07.06.16 Current Date of Adoption 07.06.16 Audit Committee Review Date - Date of Review 07.06.18 TITLE FOOD HYGIENE POLICY Related
Food Safety and Inspection Service:
 1 Overview and Update on Current FSIS Labeling Activities Jeff Canavan, Deputy Director Labeling and Program Delivery Staff, FSIS February 5, 2018 2 Mission in Action We are the public health agency in
1 Overview and Update on Current FSIS Labeling Activities Jeff Canavan, Deputy Director Labeling and Program Delivery Staff, FSIS February 5, 2018 2 Mission in Action We are the public health agency in
Plant Based Foods Association Certified Plant Based Claim Certification Program
 Plant Based Foods Association Certified Plant Based Claim Certification Program January 2, 2019 Version 1, Issue 2 Contents 1. Background 1.1 Purpose 1.2 Scope 2. Terms and Definitions 3. Certification
Plant Based Foods Association Certified Plant Based Claim Certification Program January 2, 2019 Version 1, Issue 2 Contents 1. Background 1.1 Purpose 1.2 Scope 2. Terms and Definitions 3. Certification
LLERGEN NFORMATION MANUAL & AUDITOR GUIDELINES
 A I LLERGEN NFORMATION MANUAL & AUDITOR GUIDELINES AIB International 1213 Bakers Way PO Box 3999 Manhattan, KS 66505-3999 785-537-4750 800-633-5137 Fax 785-537-0106 www.aibinternational.com ALLERGEN INFORMATION
A I LLERGEN NFORMATION MANUAL & AUDITOR GUIDELINES AIB International 1213 Bakers Way PO Box 3999 Manhattan, KS 66505-3999 785-537-4750 800-633-5137 Fax 785-537-0106 www.aibinternational.com ALLERGEN INFORMATION
PT. INTEGRAL MULIA CIPTA
 PT. INTEGRAL MULIA CIPTA Food Allergen Control Policy Doc No FSTL/10/01 Objective, Purpose & Scope Effective programs must be in place to: Evaluate, identify and control food allergens and materials which
PT. INTEGRAL MULIA CIPTA Food Allergen Control Policy Doc No FSTL/10/01 Objective, Purpose & Scope Effective programs must be in place to: Evaluate, identify and control food allergens and materials which
Product Information Sheet TASTEVA Stevia Sweetener
 Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Food Recalls: May 25, 2016 GREENBERG TRAURIG, LLP ATTORNEYS AT LAW Greenberg Traurig, LLP. All rights reserved.
 Food Recalls: 2016 Greenberg Traurig, LLP. Attorneys at Law. All rights reserved. Greenberg Traurig is a trademark and trade name of Greenberg Traurig, LLP and Greenberg Traurig, P.A. This presentation
Food Recalls: 2016 Greenberg Traurig, LLP. Attorneys at Law. All rights reserved. Greenberg Traurig is a trademark and trade name of Greenberg Traurig, LLP and Greenberg Traurig, P.A. This presentation
Product Information Sheet CLARIA Bliss
 Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Beverages. Ensure safe and high quality goods
 Beverages Ensure safe and high quality goods Sensitive test systems to analyse your products and equipment on various parameters Microbiology/hygiene Constituents Product adulteration Vitamins Allergens
Beverages Ensure safe and high quality goods Sensitive test systems to analyse your products and equipment on various parameters Microbiology/hygiene Constituents Product adulteration Vitamins Allergens
Why Should the Malting and Brewing Industry Be Concerned About Food Safety? Part 1
 MBAA TQ vol. 53, no. 1 2016 pp. 34 38 Why Should the Malting and Brewing Industry Be Concerned About Food Safety? Part 1 Tatiana A. Lorca Ecolab, Inc., St. Paul, MN, U.S.A. How many of those reading remember
MBAA TQ vol. 53, no. 1 2016 pp. 34 38 Why Should the Malting and Brewing Industry Be Concerned About Food Safety? Part 1 Tatiana A. Lorca Ecolab, Inc., St. Paul, MN, U.S.A. How many of those reading remember
The Implications for Analysis of the VSEP Reviewed VITAL Grid
 The Implications for Analysis of the VSEP Reviewed VITAL Grid VITAL 2.0 it s a sensitive issue! AOAC Meeting October 2012 Las Vegas Rob Sherlock Technical Manager DTS FACTA VITAL (Voluntary Incidental
The Implications for Analysis of the VSEP Reviewed VITAL Grid VITAL 2.0 it s a sensitive issue! AOAC Meeting October 2012 Las Vegas Rob Sherlock Technical Manager DTS FACTA VITAL (Voluntary Incidental
The FDA Food Safety Modernization Act: The Key New Requirements
 June 15, 2011 The FDA Food Safety Modernization Act: The Key New Requirements Executive Summary INSTITUTE FOR FOOD LAWS & REGULATIONS Michigan State University 140 G.M. Trout Building East Lansing, MI
June 15, 2011 The FDA Food Safety Modernization Act: The Key New Requirements Executive Summary INSTITUTE FOR FOOD LAWS & REGULATIONS Michigan State University 140 G.M. Trout Building East Lansing, MI
The Oratory School Association Food Allergen Policy
 The Oratory School Association Food Allergen Policy Policy Aim The aim is to ensure that the food provided by catering services within the Oratory School Association is stored, handled, prepared and served
The Oratory School Association Food Allergen Policy Policy Aim The aim is to ensure that the food provided by catering services within the Oratory School Association is stored, handled, prepared and served
Product Information Sheet SODA-LO EXTRA FINE
 Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Contents: 1. Supplier Information 2. Emergency Contacts 3. Product Information 4. Regulatory Status 5. Ingredient Statement 6. Quality Documents 7. Kosher/Halal Status 8. Allergen Status 9. Sulfur Dioxide
Declaration for Enzymes for use in Oenological Practices
 Declaration for Enzymes for use in Oenological Practices 25 March 2015 Food Fit for Human Consumption We hereby confirm that all raw materials used in the production of aforementioned products have a purity
Declaration for Enzymes for use in Oenological Practices 25 March 2015 Food Fit for Human Consumption We hereby confirm that all raw materials used in the production of aforementioned products have a purity
OVERVIEW OF FOOD SAFETY REGULATION IN THE UNITED STATES. Presented By William C. Balek International Sanitary Supply Association March 30, 2001
 OVERVIEW OF FOOD SAFETY REGULATION IN THE UNITED STATES Presented By William C. Balek International Sanitary Supply Association March 30, 2001 I INTRODUCTION The regulation of food safety in the United
OVERVIEW OF FOOD SAFETY REGULATION IN THE UNITED STATES Presented By William C. Balek International Sanitary Supply Association March 30, 2001 I INTRODUCTION The regulation of food safety in the United
Environmental, Health and Safety
 Environmental, Health and Safety Codes of Practice The Environmental, Health and Safety (EHS) Codes of Practice set forth Zimmer EHS requirements for our business functions and facilities worldwide. In
Environmental, Health and Safety Codes of Practice The Environmental, Health and Safety (EHS) Codes of Practice set forth Zimmer EHS requirements for our business functions and facilities worldwide. In
Thresholds - VITAL Concept. Allergen analysis what should we consider when moving towards allergen thresholds? Pauline Titchener Product Manager
 Allergen analysis what should we consider when moving towards allergen thresholds? Pauline Titchener Product Manager Thresholds - VITAL Concept Utilises Action Levels concept Action Levels are determined
Allergen analysis what should we consider when moving towards allergen thresholds? Pauline Titchener Product Manager Thresholds - VITAL Concept Utilises Action Levels concept Action Levels are determined
FDA and USDA Food Safety Regulations 101: Preventing Adulteration and Contamination
 Food Law and Regulation Boot Camp November 15-16, 2016 FDA and USDA Food Safety Regulations 101: Preventing Adulteration and Contamination Justin J. Prochnow Shareholder, Greenberg Traurig Mollie Kober
Food Law and Regulation Boot Camp November 15-16, 2016 FDA and USDA Food Safety Regulations 101: Preventing Adulteration and Contamination Justin J. Prochnow Shareholder, Greenberg Traurig Mollie Kober
cgmp (21 CFR 111) Regulation and Compliance Overview
 cgmp (21 CFR 111) Regulation and Compliance Overview Neogen Effective Compliance Seminar September 23, 2014 Michael McGuffin President, American Herbal Products Association mmcguffin@ahpa.org Regulation
cgmp (21 CFR 111) Regulation and Compliance Overview Neogen Effective Compliance Seminar September 23, 2014 Michael McGuffin President, American Herbal Products Association mmcguffin@ahpa.org Regulation
Food Labelling in Canada
 Food Labelling in Canada Food Safety and Quality Summit, New Delhi Nitin Verma, Counsellor Agriculture & Regulatory Specialist Canadian Food Inspection Agency (CFIA) December 5, 2017 2017 Her Majesty the
Food Labelling in Canada Food Safety and Quality Summit, New Delhi Nitin Verma, Counsellor Agriculture & Regulatory Specialist Canadian Food Inspection Agency (CFIA) December 5, 2017 2017 Her Majesty the
Animal Products Notice
 Animal Products Notice Labelling Requirements for Exports of Dairy Based Infant Formula Products and Formulated Supplementary Food for Young Children 18 December 2014 An animal products notice issued under
Animal Products Notice Labelling Requirements for Exports of Dairy Based Infant Formula Products and Formulated Supplementary Food for Young Children 18 December 2014 An animal products notice issued under
IIT & CII - FACE Certified Food Professional Course. Webinar Subject : Allergen Management. Tutor : Dr K.N Shashikanth Date: October 7 th 2013
 IIT & CII - FACE Certified Food Professional Course Webinar Subject : Allergen Management Tutor : Dr K.N Shashikanth Date: October 7 th 2013 1 Allergen Management Allergen are substances, usually proteins
IIT & CII - FACE Certified Food Professional Course Webinar Subject : Allergen Management Tutor : Dr K.N Shashikanth Date: October 7 th 2013 1 Allergen Management Allergen are substances, usually proteins
7475 West Main St. Milwaukee, WI 53214
 Product Description A dough conditioner that helps in the machinability of the dough and improves the volume of the finished products. Regulatory Product is produced from materials that are wholesome and
Product Description A dough conditioner that helps in the machinability of the dough and improves the volume of the finished products. Regulatory Product is produced from materials that are wholesome and
Preventive Controls for Human Foods & Preventive Controls Qualified Individuals
 Preventive Controls for Human Foods & Preventive Controls Qualified Individuals MATTHEW TAYLOR TEXAS A&M UNIVERSITY USDA NIFA AWARD NUMBER 2015-70020-24397 Food Safety Modernization Act I thank the President
Preventive Controls for Human Foods & Preventive Controls Qualified Individuals MATTHEW TAYLOR TEXAS A&M UNIVERSITY USDA NIFA AWARD NUMBER 2015-70020-24397 Food Safety Modernization Act I thank the President
Meeting Consumers Expectations
 Meeting Consumers Expectations February 19, 2008 Savannah, GA Gale Prince gale@galeprince.com Health Benefits Nutrition 64% of us are overweight 30% are obese 20% are on a diet Consumers are making the
Meeting Consumers Expectations February 19, 2008 Savannah, GA Gale Prince gale@galeprince.com Health Benefits Nutrition 64% of us are overweight 30% are obese 20% are on a diet Consumers are making the
Issue Date: 4/26/18 Supersedes Version Dated: 11/20/17 Page 1 of 5
 Issue Date: 4/26/18 Supersedes Version Dated: 11/20/17 Page 1 of 5 Product Information Ingredients Organic Certification Country of Origin Gluten-Free Allergen-Free Production Organic white rice flour
Issue Date: 4/26/18 Supersedes Version Dated: 11/20/17 Page 1 of 5 Product Information Ingredients Organic Certification Country of Origin Gluten-Free Allergen-Free Production Organic white rice flour
Control of Listeria monocytogenes in Ready-to-Eat Foods: Draft Guidance
 Control of Listeria monocytogenes in Ready-to-Eat Foods: Draft Guidance Federal Register / Vol. 82, No. 10 / Tuesday, January 17, 2017 Mickey Parish Senior Science Advisor FDA CFSAN mickey.parish@fda.hhs.gov
Control of Listeria monocytogenes in Ready-to-Eat Foods: Draft Guidance Federal Register / Vol. 82, No. 10 / Tuesday, January 17, 2017 Mickey Parish Senior Science Advisor FDA CFSAN mickey.parish@fda.hhs.gov
Minimizing the Risk of Cross- Contamination
 Minimizing the Risk of Cross- Contamination Rob Middendorf Business Development Director Booth #2746 What We Do Outline What is Cross Contamination? Why is it a risk? How to Prevent/Reduce? Hygienic Design
Minimizing the Risk of Cross- Contamination Rob Middendorf Business Development Director Booth #2746 What We Do Outline What is Cross Contamination? Why is it a risk? How to Prevent/Reduce? Hygienic Design
ILSI Workshop Romer Labs Food Allergen Testing solutions
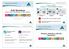 Romer Labs Experts in food allergen testing ILSI Workshop Romer Labs Food Allergen Testing solutions 8/10 AgraQuant Plus ELISA kits 22 AgraQuant ELISA kits 19 AgraStrip LFD kits Bangkok April 6 2015 Lukas
Romer Labs Experts in food allergen testing ILSI Workshop Romer Labs Food Allergen Testing solutions 8/10 AgraQuant Plus ELISA kits 22 AgraQuant ELISA kits 19 AgraStrip LFD kits Bangkok April 6 2015 Lukas
REVISED CODEX GENERAL STANDARD FOR IRRADIATED FOODS CODEX STAN , REV
 Irradiated foods Page 1 of 10 1. SCOPE REVISED CODEX GENERAL STANDARD FOR IRRADIATED FOODS CODEX STAN 106-1983, REV.1-2003 This standard applies to foods processed by ionizing radiation that is used in
Irradiated foods Page 1 of 10 1. SCOPE REVISED CODEX GENERAL STANDARD FOR IRRADIATED FOODS CODEX STAN 106-1983, REV.1-2003 This standard applies to foods processed by ionizing radiation that is used in
THE NON PENICILLIN BETA LACTAM DRUG CROSS CONTAMINATION PREVENTION; USFDA PERSPECTIVE
 THE NON PENICILLIN BETA LACTAM DRUG CROSS CONTAMINATION PREVENTION; USFDA PERSPECTIVE An overview by Sarah Vugigi, M. Pharm, Elys Chemical Industries Ltd, Nairobi, Kenya INTRODUCTION This guidance describes
THE NON PENICILLIN BETA LACTAM DRUG CROSS CONTAMINATION PREVENTION; USFDA PERSPECTIVE An overview by Sarah Vugigi, M. Pharm, Elys Chemical Industries Ltd, Nairobi, Kenya INTRODUCTION This guidance describes
Issue Date: 7/12/18 Supersedes Version Dated: 8/1/16 Page 1 of 5
 Issue Date: 7/12/18 Supersedes Version Dated: 8/1/16 Page 1 of 5 Product Information Ingredients Organic Certification Country of Origin Gluten-Free Allergen-Free Production Organic rice flour, organic
Issue Date: 7/12/18 Supersedes Version Dated: 8/1/16 Page 1 of 5 Product Information Ingredients Organic Certification Country of Origin Gluten-Free Allergen-Free Production Organic rice flour, organic
GROCERY MANUFACTURERS ASSOCIATION. Science Forum. Connecting Sound Science and Responsible Solutions
 GROCERY MANUFACTURERS ASSOCIATION Science Forum Connecting Sound Science and Responsible Solutions FSMA Implementation Michael M. Landa Director, Center for Food Safety and Applied Nutrition Food and Drug
GROCERY MANUFACTURERS ASSOCIATION Science Forum Connecting Sound Science and Responsible Solutions FSMA Implementation Michael M. Landa Director, Center for Food Safety and Applied Nutrition Food and Drug
Allergen management it s all about the consumer safety
 Allergen management it s all about the consumer safety Kirsten Grinter President, Allergen Bureau The Allergen Bureau Ltd ACN 162 786 389 Discussion Who is the Allergen Bureau The VITAL Program & PAL VITAL
Allergen management it s all about the consumer safety Kirsten Grinter President, Allergen Bureau The Allergen Bureau Ltd ACN 162 786 389 Discussion Who is the Allergen Bureau The VITAL Program & PAL VITAL
HAZARD ANALYSIS CRITICAL CONTROL POINT (HACCP) SYSTEM
 The HACCP Inspection/Food Audit HAZARD ANALYSIS CRITICAL CONTROL POINT (HACCP) SYSTEM The HACCP system was originally designed by the National Aeronautics and Space Administration (NASA) and is used by
The HACCP Inspection/Food Audit HAZARD ANALYSIS CRITICAL CONTROL POINT (HACCP) SYSTEM The HACCP system was originally designed by the National Aeronautics and Space Administration (NASA) and is used by
Planning For The FDA s 'Deeming Rule' For E- Cigarettes
 Law360, New York (September 21, 2015, 3:39 PM ET) -- The 2009 Family Smoking Prevention and Tobacco Control Act (TCA)[1] gave the U.S. Food and Drug Administration the authority to oversee the manufacture,
Law360, New York (September 21, 2015, 3:39 PM ET) -- The 2009 Family Smoking Prevention and Tobacco Control Act (TCA)[1] gave the U.S. Food and Drug Administration the authority to oversee the manufacture,
CFSAN Update Michael M. Landa
 CFSAN Update Comments by Michael M. Landa Director, Center for Food Safety and Applied Nutrition JIFSAN Fall Advisory Council Meeting November 14, 2013 1 CFSAN Plan for Program Priorities (2013 2014) Reduce
CFSAN Update Comments by Michael M. Landa Director, Center for Food Safety and Applied Nutrition JIFSAN Fall Advisory Council Meeting November 14, 2013 1 CFSAN Plan for Program Priorities (2013 2014) Reduce
