NDA /S-001. ALDARA [al dar a] (imiquimod) Cream, 5% For Dermatologic Use Only - Not for Ophthalmic Use.
|
|
|
- Ralf Boyd
- 5 years ago
- Views:
Transcription
1 ALDARA [al dar a] (imiquimod) Cream, 5% For Dermatologic Use Only - Not for Ophthalmic Use. DESCRIPTION Aldara is the brand name for imiquimod which is an immune response modifier. Each gram of the 5% cream contains 50 mg of imiquimod in an off-white oil-in-water vanishing cream base consisting of isostearic acid, cetyl alcohol, stearyl alcohol, white petrolatum, polysorbate 60, sorbitan monostearate, glycerin, xanthan gum, purified water, benzyl alcohol, methylparaben, and propylparaben. Chemically, imiquimod is 1-(2-methylpropyl)-1H-imidazo[4,5-c]quinolin- 4-amine. Imiquimod has a molecular formula of C 14 H 16 N 4 and a molecular weight of Its structural formula is: NH 2 N N N CH 3 CH 3 3
2 CLINICAL PHARMACOLOGY Pharmacodynamics Imiquimod has no direct antiviral activity in cell culture. A study in 22 patients with genital/perianal warts comparing imiquimod and vehicle shows that imiquimod induces mrna encoding cytokines including interferon-α at the treatment site. In addition HPVL1 mrna and HPV DNA are significantly decreased following treatment. However, the clinical relevance of these findings is unknown. Pharmacokinetics Percutaneous absorption of [ 14 C] imiquimod was minimal in a study involving 6 healthy subjects treated with a single topical application (5 mg) of [ 14 C] imiquimod cream formulation. No radioactivity was detected in the serum (lower limit of quantitation: 1 ng/ml) and < 0.9% of the radiolabelled dose was excreted in the urine and feces following topical application. 8 CLINICAL STUDIES In a double-blind, placebo-controlled clinical trial, 209 otherwise healthy patients 18 years of age and older with genital/perianal warts were treated with Aldara 5% cream or vehicle control 3X/week for a maximum of 16 weeks. The median baseline wart area was 69 mm 2 (range 8 to 5525 mm 2 ). Patient accountability is shown in the figure below. 4
3 1004-IMIQ Patient Accountability Enrolled N=209 Imiquimod 5% N = 109 Vehicle N = 100 Completed 16 Weeks of Treatment Not Clear N = 36 Withdrew N = 19 Completed 16 Weeks of Treatment Not Clear N = 62 Withdrew N = 27 Clear N = 54 Clear N = 11 Completed 12 Weeks of Followup* Remained Clear N = 39 Completed 12 Weeks of Follow-up* Remained Clear N = 9 * The other patients were either lost to follow-up or experienced recurrences. Data on complete clearance are listed in the table below. The median time to complete wart clearance was 10 weeks. 5
4 CLEARANCE - STUDY 1004 Treatment Patients with Complete Clearance of Warts Patients Without Follow-up Patients with Warts Remaining at Week 16 Overall imiquimod 5% 50% 17% 33% (n=109) vehicle (n=100) 11% 27% 62% Females imiquimod 5% 72% 11% 17% (n=46) vehicle (n=40) 20% 33% 48% Males imiquimod 5% 33% 22% 44% (n=63) vehicle (n=60) 5% 23% 72% INDICATIONS AND USAGE Aldara 5% cream is indicated for the treatment of external genital and perianal warts/condyloma acuminata in adults. CONTRAINDICATIONS None known WARNINGS Aldara cream has not been evaluated for the treatment of urethral, intra-vaginal, cervical, rectal, or intra-anal human papilloma viral disease and is not recommended for these conditions. 6
5 PRECAUTIONS General Local skin reactions such as erythema, erosion, excoriation/flaking, and edema are common. Should severe local skin reaction occur, the cream should be removed by washing the treatment area with mild soap and water. Treatment with Aldara cream can be resumed after the skin reaction has subsided. There is no clinical experience with Aldara cream therapy immediately following the treatment of genital/perianal warts with other cutaneously applied drugs; therefore, Aldara cream administration is not recommended until genital/perianal tissue is healed from any previous drug or surgical treatment. Aldara has the potential to exacerbate inflammatory conditions of the skin. Information for Patients Patients using Aldara 5% cream should receive the following information and instructions. The effect of Aldara 5% cream on the transmission of genital/perianal warts is unknown. Aldara 5% cream may weaken condoms and vaginal diaphragms. Therefore, concurrent use is not recommended. 1. This medication is to be used as directed by a physician. It is for external use only. Eye contact should be avoided. 2. The treatment area should not be bandaged or otherwise covered or wrapped as to be occlusive. 3. Sexual (genital, anal, oral) contact should be avoided while the cream is on the skin. 4. It is recommended that 6-10 hours following Aldara 5% cream application the treatment area be washed with mild soap and water. 5. It is common for patients to experience local skin reactions such as erythema, erosion, excoriation/flaking, and edema at the site of application or surrounding areas. Most skin reactions are mild to moderate. Severe skin reactions can occur and should be reported promptly to the prescribing physician. 6. Uncircumcised males treating warts under the foreskin should retract the foreskin and clean the area daily. 7. Patients should be aware that new warts may develop during therapy, as Aldara is not a cure. 7
6 Carcinogenicity, Mutagenesis, and Impairment of Fertility Rodent carcinogenicity data are not available. Imiquimod was without effect in a series of eight different mutagenicity assays including Ames, mouse lymphoma, CHO chromosome aberration, human lymphocyte chromosome aberration, SHE cell transformation, rat and hamster bone marrow cytogenetics, and mouse dominant lethal test. Daily oral administration of imiquimod to rats, at doses up to 8 times the recommended human dose on a mg/m 2 basis throughout mating, gestation, parturition and lactation, demonstrated no impairment of reproduction. Pregnancy Pregnancy Category B: There are no adequate and well-controlled studies in pregnant women. Imiquimod was not found to be teratogenic in rat or rabbit teratology studies. In rats at a high maternally toxic dose (28 times human dose on a mg/m 2 basis), reduced pup weights and delayed ossification were observed. In developmental studies with offspring of pregnant rats treated with imiquimod (8 times human dose), no adverse effects were demonstrated. Nursing Mothers It is not known whether topically applied imiquimod is excreted in breast milk. Pediatric Use Safety and efficacy in patients below the age of 18 years have not been established. ADVERSE REACTIONS 8
7 In controlled clinical trials, the most frequently reported adverse reactions were those of local skin and application site reactions; some patients also reported systemic reactions. These reactions were usually mild to moderate in intensity; however, severe reactions were reported with 3X/week application. These reactions were more frequent and more intense with daily application than with 3X/week application. Overall, in the 3X/week application clinical studies, 1.2% (4/327) of the patients discontinued due to local skin/application site reactions. The incidence and severity of local skin reactions during controlled clinical trials are shown in the following table. 3X/WEEK APPLICATION WART SITE REACTION AS ASSESSED BY INVESTIGATOR MILD/MODERATE SEVERE FEMALES MALES FEMALES MALES 5% Imiquimo d N=114 Vehicl e N=99 5% Imiquim od N=156 Vehicl e N=157 5% Imiquim od N=114 Vehicl e N=99 5% Imiquim od N=156 Vehic le N=15 7 Erythema 61% 21% 54% 22% 4% 0% 4% 0% Erosion 30% 8% 29% 6% 1% 0% 1% 0% Excoriatio n/ Flaking 18% 8% 25% 8% 0% 0% 1% 0% Edema 17% 5% 12% 1% 1% 0% 0% 0% Induration 5% 2% 7% 2% 0% 0% 0% 0% Ulceration 5% 1% 4% 1% 3% 0% 0% 0% Scabbing 4% 0% 13% 3% 0% 0% 0% 0% Vesicles 3% 0% 2% 0% 0% 0% 0% 0% Remote site skin reactions were also reported in female and male patients treated 3X/week with imiquimod 5% cream. The severe remote site skin reactions reported for females were erythema (3%), ulceration (2%), and edema (1%); and for males, erosion (2%), and erythema, edema, induration, and excoriation/flaking (each 1%). 9
8 Adverse events judged to be probably or possibly related to Aldara reported by more than 5% of patients are listed below; also included are soreness, influenza-like symptoms and myalgia. 10
9 3X/WEEK APPLICATION FEMALES MALES 5% 5% Imiquimod Vehicle Imiquimod Vehicle (n=117) (n=103) (n=156) (n=158) APPLICATION SITE DISORDERS: APPLICATION SITE REACTIONS Wart Site: Itching 32% 20% 22% 10% Burning 26% 12% 9% 5% Pain 8% 2% 2% 1% Soreness 3% 0% 0% 1% FUNGAL INFECTION a 11% 3% 2% 1% SYSTEMIC REACTIONS: Headache 4% 3% 5% 2% Influenza-like symptoms 3% 2% 1% 0% Myalgia 1% 0% 1% 1% *Incidences reported without regard to causality with Aldara. Adverse events judged to be possibly or probably related to Aldara and reported by more than 1% of patients include: Application Site Disorders: Wart Site Reactions (burning, hypopigmentation, irritation, itching, pain, rash, sensitivity, soreness, stinging, tenderness); Remote Site Reactions (bleeding, burning, itching, pain, tenderness, tinea cruris); Body as a Whole: fatigue, fever, influenza-like symptoms; Central and Peripheral Nervous System Disorders: headache; Gastro- Intestinal System Disorders: diarrhea; Musculo-Skeletal System Disorders: myalgia
10 OVERDOSAGE Overdosage of Aldara 5% cream in humans is unlikely due to minimal percutaneous absorption. Animal studies reveal a rabbit dermal lethal imiquimod dose of greater than 1600 mg/m 2. Persistent topical overdosing of Aldara 5% cream could result in severe local skin reactions. The most clinically serious adverse event reported following multiple oral imiquimod doses of >200 mg was hypotension which resolved following oral or intravenous fluid administration. DOSAGE AND ADMINISTRATION Aldara cream is to be applied 3 times per week, prior to normal sleeping hours, and left on the skin for 6-10 hours. Following the treatment period cream should be removed by washing the treated area with mild soap and water. Examples of 3 times per week application schedules are: Monday, Wednesday, Friday; or Tuesday, Thursday, Saturday application prior to sleeping hours. Aldara treatment should continue until there is total clearance of the genital/perianal warts or for a maximum of 16 weeks. Local skin reactions (erythema) at the treatment site are common. A rest period of several days may be taken if required by the patient s discomfort or severity of the local skin reaction. Treatment may resume once the reaction subsides. Non-occlusive dressings such as cotton gauze or cotton underwear may be used in the management of skin reactions. The technique for proper dose administration should be demonstrated by the prescriber to maximize the benefit of Aldara therapy. Handwashing before and after cream application is recommended. Aldara 5% cream is packaged in single-use packets which contain sufficient cream to cover a wart area of up to 20 cm 2 ; use of excessive amounts of cream should be avoided. Patients should be instructed to apply Aldara cream to external genital/perianal warts. A thin layer is applied to the wart area and rubbed in until the cream is no longer visible. The application site is not to be occluded. HOW SUPPLIED Aldara (imiquimod) cream, 5%, is supplied in single-use packets which contain 250 mg of the cream. Available as: box of 12 packets NDC , and box of 30 packets NDC Do not store above 30 o C (86 o F). Avoid freezing. Caution: Federal Law prohibis dispensing without prescription. 12
11 1
12 This is a representation of an electronic record that was signed electronically and this page is the manifestation of the electronic signature /s/ Jonathan Wilkin 12/8/01 04:51:26 PM
PRODUCT MONOGRAPH APO-IMIQUIMOD. Imiquimod Cream, 5% w/w. 250 mg single-dose packet. Immune Response Modifier
 PRODUCT MONOGRAPH APO-IMIQUIMOD Imiquimod Cream, 5% w/w 50 mg single-dose packet Immune Response Modifier APOTEX INC. DATE OF REVISION: 50 Signet Drive February 8, 0 Toronto, Ontario M9L T9 Control No.
PRODUCT MONOGRAPH APO-IMIQUIMOD Imiquimod Cream, 5% w/w 50 mg single-dose packet Immune Response Modifier APOTEX INC. DATE OF REVISION: 50 Signet Drive February 8, 0 Toronto, Ontario M9L T9 Control No.
DOSAGE FORMS AND STRENGTHS Cream: Each gram contains 10 mg of ozenoxacin (1%) (3).
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use XEPI TM safely and effectively. See full prescribing information for XEPI TM. XEPI TM (ozenoxacin)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use XEPI TM safely and effectively. See full prescribing information for XEPI TM. XEPI TM (ozenoxacin)
DOVONEX (calcipotriene) Cream, 0.005% Rx only FOR TOPICAL DERMATOLOGIC USE ONLY. Not for Ophthalmic, Oral or Intravaginal Use.
 DOVONEX (calcipotriene) Cream, 0.005% Rx only FOR TOPICAL DERMATOLOGIC USE ONLY. Not for Ophthalmic, Oral or Intravaginal Use. DESCRIPTION Dovonex (calcipotriene) Cream, 0.005% contains calcipotriene monohydrate,
DOVONEX (calcipotriene) Cream, 0.005% Rx only FOR TOPICAL DERMATOLOGIC USE ONLY. Not for Ophthalmic, Oral or Intravaginal Use. DESCRIPTION Dovonex (calcipotriene) Cream, 0.005% contains calcipotriene monohydrate,
PRODUCT INFORMATION WARTEC SOLUTION
 PRODUCT INFORMATION WARTEC SOLUTION NAME OF THE MEDICINE WARTEC Solution contains podophyllotoxin as the active ingredient. CAS number: 518-28-5 DESCRIPTION WARTEC Solution is a blue solution for topical
PRODUCT INFORMATION WARTEC SOLUTION NAME OF THE MEDICINE WARTEC Solution contains podophyllotoxin as the active ingredient. CAS number: 518-28-5 DESCRIPTION WARTEC Solution is a blue solution for topical
Ultravate (halobetasol propionate) Cream, 0.05% (halobetasol propionate) Ointment, 0.05% For Dermatological Use Only. Not for Ophthalmic Use.
 Ultravate (halobetasol propionate) Cream, 0.05% (halobetasol propionate) Ointment, 0.05% For Dermatological Use Only. Not for Ophthalmic Use. Rx only DESCRIPTION Ultravate contains halobetasol propionate,
Ultravate (halobetasol propionate) Cream, 0.05% (halobetasol propionate) Ointment, 0.05% For Dermatological Use Only. Not for Ophthalmic Use. Rx only DESCRIPTION Ultravate contains halobetasol propionate,
LUZU (luliconazole) Cream, 1% for topical use Initial U.S. Approval: 2013
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUZU safely and effectively. See full prescribing information for LUZU. LUZU (luliconazole) Cream,
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUZU safely and effectively. See full prescribing information for LUZU. LUZU (luliconazole) Cream,
Product Information ALDARA TM
 Product Information ALDARA TM NAME OF THE MEDICINE is 1-(2-methylpropyl)-1H-imidazo [4,5-c]quinolin-4-amine. It is an odourless, white to off-white crystalline solid. has a molecular formula of C 14 H
Product Information ALDARA TM NAME OF THE MEDICINE is 1-(2-methylpropyl)-1H-imidazo [4,5-c]quinolin-4-amine. It is an odourless, white to off-white crystalline solid. has a molecular formula of C 14 H
The Ointment is back!
 Now Available for Plaque Psoriasis The Ointment is back! Bioequivalent to Dovonex * Ointment Before prescribing, please see attached full Prescribing Information. The same Delivers the reliability you
Now Available for Plaque Psoriasis The Ointment is back! Bioequivalent to Dovonex * Ointment Before prescribing, please see attached full Prescribing Information. The same Delivers the reliability you
For topical use only. Not for oral, ophthalmic, or intravaginal use.
 DESOXIMETASONE Ointment USP, 0.25% For topical use only. Not for oral, ophthalmic, or intravaginal use. Rx only DESCRIPTION Desoximetasone ointment USP, 0.25% contains the active synthetic corticosteroid
DESOXIMETASONE Ointment USP, 0.25% For topical use only. Not for oral, ophthalmic, or intravaginal use. Rx only DESCRIPTION Desoximetasone ointment USP, 0.25% contains the active synthetic corticosteroid
LOPROX CREAM (ciclopirox) 0.77% FOR DERMATOLOGIC USE ONLY. NOT FOR USE IN EYES. Rx Only
 LOPROX CREAM (ciclopirox) 0.77% FOR DERMATOLOGIC USE ONLY. NOT FOR USE IN EYES. Rx Only DESCRIPTION LOPROX Cream (ciclopirox) 0.77% is for topical use. Each gram of LOPROX Cream contains 7.70 mg of ciclopirox
LOPROX CREAM (ciclopirox) 0.77% FOR DERMATOLOGIC USE ONLY. NOT FOR USE IN EYES. Rx Only DESCRIPTION LOPROX Cream (ciclopirox) 0.77% is for topical use. Each gram of LOPROX Cream contains 7.70 mg of ciclopirox
Cordran Cream and Cordran Ointment Flurandrenolide, USP
 Cordran Cream and Cordran Ointment Flurandrenolide, USP DESCRIPTION Cordran (flurandrenolide, USP) is a potent corticosteroid intended for topical use. Flurandrenolide occurs as white to off-white, fluffy,
Cordran Cream and Cordran Ointment Flurandrenolide, USP DESCRIPTION Cordran (flurandrenolide, USP) is a potent corticosteroid intended for topical use. Flurandrenolide occurs as white to off-white, fluffy,
Apo-Imiquimod imiquimod 5% w/w cream
 New Zealand Consumer Medicine Information What is in this leaflet Apo-Imiquimod imiquimod 5% w/w cream Please read this leaflet carefully before you start using Apo-Imiqumod. This leaflet answers some
New Zealand Consumer Medicine Information What is in this leaflet Apo-Imiquimod imiquimod 5% w/w cream Please read this leaflet carefully before you start using Apo-Imiqumod. This leaflet answers some
Rx only FOR TOPICAL DERMATOLOGIC USE ONLY, NOT FOR OPHTHALMIC, ORAL, OR INTRAVAGINAL USE
 STZC:R1 ERTACZO TM (SERTACONAZOLE NITRATE) CREAM, 2% Rx only FOR TOPICAL DERMATOLOGIC USE ONLY, NOT FOR OPHTHALMIC, ORAL, OR INTRAVAGINAL USE DESCRIPTION: ERTACZO TM (sertaconazole nitrate) Cream, 2%,
STZC:R1 ERTACZO TM (SERTACONAZOLE NITRATE) CREAM, 2% Rx only FOR TOPICAL DERMATOLOGIC USE ONLY, NOT FOR OPHTHALMIC, ORAL, OR INTRAVAGINAL USE DESCRIPTION: ERTACZO TM (sertaconazole nitrate) Cream, 2%,
FULL PRESCRIBING INFORMATION
 IGLIGTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use DESNATE Gel safely and effectively. See full prescribing information for DESNATE Gel. DESNATE (desonide)
IGLIGTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use DESNATE Gel safely and effectively. See full prescribing information for DESNATE Gel. DESNATE (desonide)
Product Information Australia APO-IMIQUIMOD CREAM. NAME OF THE MEDICINE Imiquimod. 1-(2-methylpropyl)-1H-imidazo[4,5-c]quinolin-4-amine
![Product Information Australia APO-IMIQUIMOD CREAM. NAME OF THE MEDICINE Imiquimod. 1-(2-methylpropyl)-1H-imidazo[4,5-c]quinolin-4-amine Product Information Australia APO-IMIQUIMOD CREAM. NAME OF THE MEDICINE Imiquimod. 1-(2-methylpropyl)-1H-imidazo[4,5-c]quinolin-4-amine](/thumbs/83/88391058.jpg) APO-IMIQUIMOD CREAM NAME OF THE MEDICINE Imiquimod. Chemical Name: 1-(2-methylpropyl)-1H-imidazo[4,5-c]quinolin-4-amine Structural Formula: Molecular Formula: C 14 H 16 N 4 Molecular Weight: 240.3 CAS
APO-IMIQUIMOD CREAM NAME OF THE MEDICINE Imiquimod. Chemical Name: 1-(2-methylpropyl)-1H-imidazo[4,5-c]quinolin-4-amine Structural Formula: Molecular Formula: C 14 H 16 N 4 Molecular Weight: 240.3 CAS
The chemical name of acyclovir, USP is 2-amino-1,9-dihydro-9-[(2-hydroxyethoxy)methyl]-6Hpurin-6-one; it has the following structural formula:
![The chemical name of acyclovir, USP is 2-amino-1,9-dihydro-9-[(2-hydroxyethoxy)methyl]-6Hpurin-6-one; it has the following structural formula: The chemical name of acyclovir, USP is 2-amino-1,9-dihydro-9-[(2-hydroxyethoxy)methyl]-6Hpurin-6-one; it has the following structural formula:](/thumbs/74/71271167.jpg) Acyclovir Ointment, USP 5% DESCRIPTION Acyclovir, USP, is a synthetic nucleoside analogue active against herpes viruses. Acyclovir ointment, USP 5% is a formulation for topical administration. Each gram
Acyclovir Ointment, USP 5% DESCRIPTION Acyclovir, USP, is a synthetic nucleoside analogue active against herpes viruses. Acyclovir ointment, USP 5% is a formulation for topical administration. Each gram
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT ALDARA 5% cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains 12.5 mg of imiquimod in 250 mg cream (5 %).
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT ALDARA 5% cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains 12.5 mg of imiquimod in 250 mg cream (5 %).
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory ZODERM E CREAM. Oxiconazole Nitrate Cream
 For the use only of Registered Medical Practitioners or a Hospital or a Laboratory ZODERM E CREAM Oxiconazole Nitrate Cream QUALITATIVE AND QUANTITATIVE COMPOSITION ZODERM E CREAM contains: Oxiconazole
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory ZODERM E CREAM Oxiconazole Nitrate Cream QUALITATIVE AND QUANTITATIVE COMPOSITION ZODERM E CREAM contains: Oxiconazole
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT ALDARA 5% cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains 12.5 mg of imiquimod in 250 mg cream (5 %).
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT ALDARA 5% cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains 12.5 mg of imiquimod in 250 mg cream (5 %).
(imiquimod) Cream, 3.75% w/w
 PRODUCT MONOGRAPH Pr VYLOMA TM (imiquimod) Cream, 3.75% w/w Immune response modifier Manufactured for: Valeant Canada LP 2150 St-Elzéar Blvd., West Laval, Quebec H7L 4A8 Canada Date of Preparation: May
PRODUCT MONOGRAPH Pr VYLOMA TM (imiquimod) Cream, 3.75% w/w Immune response modifier Manufactured for: Valeant Canada LP 2150 St-Elzéar Blvd., West Laval, Quebec H7L 4A8 Canada Date of Preparation: May
HYDROCORTISONE OINTMENT USP,
 HYDROCORTISONE- hydrocortisone ointment E. Fougera & Co. a division of Fougera Pharmaceuticals Inc. ---------- HYDROCORTISONE OINTMENT USP, 1% Rx only For External Us e Only Not For Ophthalmic Us e DESCRIPTION:
HYDROCORTISONE- hydrocortisone ointment E. Fougera & Co. a division of Fougera Pharmaceuticals Inc. ---------- HYDROCORTISONE OINTMENT USP, 1% Rx only For External Us e Only Not For Ophthalmic Us e DESCRIPTION:
Gynofort (butoconazole nitrate) Vaginal Cream, 2.0%
 Gynofort (butoconazole nitrate) Vaginal Cream, 2.0% Rx Only Decription Butoconazole Nitrate Vaginal Cream, 2.0% contains butoconazole nitrate 2%, an imidazole derivative with antifungal activity. Its chemical
Gynofort (butoconazole nitrate) Vaginal Cream, 2.0% Rx Only Decription Butoconazole Nitrate Vaginal Cream, 2.0% contains butoconazole nitrate 2%, an imidazole derivative with antifungal activity. Its chemical
HIGHLIGHTS OF PRESCRIBING INFORMATION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use epinastine hydrochloride ophthalmic solution safely and effectively. See full prescribing information
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use epinastine hydrochloride ophthalmic solution safely and effectively. See full prescribing information
Revised: 06/2013. Page 1
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use EXTINA Foam safely and effectively. See full prescribing information for EXTINA Foam. EXTINA (ketoconazole)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use EXTINA Foam safely and effectively. See full prescribing information for EXTINA Foam. EXTINA (ketoconazole)
Trifluridine Ophthalmic Solution, 1% Sterile
 Trifluridine Ophthalmic Solution, 1% Sterile DESCRIPTION Trifluridine (also known as trifluorothymidine, F 3 TdR,F 3 T), is an antiviral drug for topical treatment of epithelial keratitis caused by herpes
Trifluridine Ophthalmic Solution, 1% Sterile DESCRIPTION Trifluridine (also known as trifluorothymidine, F 3 TdR,F 3 T), is an antiviral drug for topical treatment of epithelial keratitis caused by herpes
Chemically, clobetasol propionate is 21-chloro-9-fluoro,11β,17-dihydroxy-16βmethylpregna-1,4-diene-3,20-dione
 Clobetasol Propionate Cream USP, 0.05% FOR TOPICAL DERMATOLOGIC USE ONLY NOT FOR OPHTHALMIC, ORAL, OR INTRAVAGINAL USE. DESCRIPTION Clobetasol propionate cream USP, 0.05% contain the active compound clobetasol
Clobetasol Propionate Cream USP, 0.05% FOR TOPICAL DERMATOLOGIC USE ONLY NOT FOR OPHTHALMIC, ORAL, OR INTRAVAGINAL USE. DESCRIPTION Clobetasol propionate cream USP, 0.05% contain the active compound clobetasol
VIROPTIC Ophthalmic Solution, 1% Sterile (trifluridine ophthalmic solution)
 VIROPTIC Ophthalmic Solution, 1% Sterile (trifluridine ophthalmic solution) PRODUCT OVERVIEW: VIROPTIC SOLUTION DESCRIPTION VIROPTIC is the brand name for trifluridine (also known as trifluorothymidine,
VIROPTIC Ophthalmic Solution, 1% Sterile (trifluridine ophthalmic solution) PRODUCT OVERVIEW: VIROPTIC SOLUTION DESCRIPTION VIROPTIC is the brand name for trifluridine (also known as trifluorothymidine,
EXTERNAL ANOGENITAL WARTS
 EXTERNAL ANOGENITAL WARTS Whats new HPV vaccination is now available to MSM under the age of 45 years attending Sexual Health Services. This vaccination is recommended even if a prior/current history of
EXTERNAL ANOGENITAL WARTS Whats new HPV vaccination is now available to MSM under the age of 45 years attending Sexual Health Services. This vaccination is recommended even if a prior/current history of
3 DOSAGE FORMS AND STRENGTHS
 PATADAY- olopatadine hydrochloride solution/ drops Alcon Laboratories, Inc. ---------- HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PATADAY safely
PATADAY- olopatadine hydrochloride solution/ drops Alcon Laboratories, Inc. ---------- HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PATADAY safely
Reference ID:
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ENDARI TM safely and effectively. See full prescribing information for ENDARI. ENDARI (L-glutamine
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ENDARI TM safely and effectively. See full prescribing information for ENDARI. ENDARI (L-glutamine
CORTISPORIN Cream (neomycin and polymyxin B sulfates and hydrocortisone acetate cream, USP)
 CORTISPORIN Cream (neomycin and polymyxin B sulfates and hydrocortisone acetate cream, USP) DESCRIPTION CORTISPORIN Cream (neomycin and polymyxin B sulfates and hydrocortisone acetate cream, USP) is a
CORTISPORIN Cream (neomycin and polymyxin B sulfates and hydrocortisone acetate cream, USP) DESCRIPTION CORTISPORIN Cream (neomycin and polymyxin B sulfates and hydrocortisone acetate cream, USP) is a
SUMMARY OF THE PRODUCT CHARACTERISTICS
 SUMMARY OF THE PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Zoviduo 50 mg/g and 10 mg/g cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 gram cream contains 50 mg aciclovir and 10 mg hydrocortisone.
SUMMARY OF THE PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Zoviduo 50 mg/g and 10 mg/g cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 gram cream contains 50 mg aciclovir and 10 mg hydrocortisone.
ESKATA TM (hydrogen peroxide) topical solution Initial U.S. Approval: 2017
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ESKATA TM safely and effectively. See full prescribing information for ESKATA TM. ESKATA TM (hydrogen
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ESKATA TM safely and effectively. See full prescribing information for ESKATA TM. ESKATA TM (hydrogen
ESKATA (hydrogen peroxide) topical solution Initial U.S. Approval: 2017
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ESKATA safely and effectively. See full prescribing information for ESKATA. ESKATA (hydrogen peroxide)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ESKATA safely and effectively. See full prescribing information for ESKATA. ESKATA (hydrogen peroxide)
Reference ID:
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use FINACEA Gel safely and effectively. See full prescribing information for FINACEA Gel. FINACEA (azelaic
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use FINACEA Gel safely and effectively. See full prescribing information for FINACEA Gel. FINACEA (azelaic
2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 NAME OF THE MEDICINAL PRODUCT Skinoren 20 % cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 g Skinoren cream contains 200 mg (20 %) azelaic acid. For the full list of excipients, see section 6.1 List
NAME OF THE MEDICINAL PRODUCT Skinoren 20 % cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 g Skinoren cream contains 200 mg (20 %) azelaic acid. For the full list of excipients, see section 6.1 List
FLUOCINOLONE ACETONIDE-
 FLUOCINOLONE ACETONIDE- fluocinolone acetonide cream E. & Co. a division of Pharmaceuticals Inc. ---------- FLUOCINOLONE ACETONIDE CREAM, USP 0.01%, 0.025% For Topical Us e Only. Not for Ophthalmic Us
FLUOCINOLONE ACETONIDE- fluocinolone acetonide cream E. & Co. a division of Pharmaceuticals Inc. ---------- FLUOCINOLONE ACETONIDE CREAM, USP 0.01%, 0.025% For Topical Us e Only. Not for Ophthalmic Us
PRODUCT INFORMATION H 2
 PRODUCT IFORMATIO ZOVIRAX COLD SORE CREAM APPROVED AME: Aciclovir COMPOSITIO: Aciclovir 5% w/w. DESCRIPTIO: Aciclovir is a synthetic acyclic purine nucleoside analogue. Its chemical name is 9-((2-hydroxyethoxy)methyl)guanine.
PRODUCT IFORMATIO ZOVIRAX COLD SORE CREAM APPROVED AME: Aciclovir COMPOSITIO: Aciclovir 5% w/w. DESCRIPTIO: Aciclovir is a synthetic acyclic purine nucleoside analogue. Its chemical name is 9-((2-hydroxyethoxy)methyl)guanine.
Each gram of the ointment contains 0.25 mg Fluocinolone Acetonide in a base containing White Petrolatum.
 FLUOCINOLONE ACETONIDE- fluocinolone acetonide ointment E. Fougera & Co. a division of Fougera Pharmaceuticals Inc. ---------- FLUOCINOLONE ACETONIDE OINTMENT, USP, 0.025% For Topical Us e Only. Not for
FLUOCINOLONE ACETONIDE- fluocinolone acetonide ointment E. Fougera & Co. a division of Fougera Pharmaceuticals Inc. ---------- FLUOCINOLONE ACETONIDE OINTMENT, USP, 0.025% For Topical Us e Only. Not for
DERMATOP Ointment (prednicarbate ointment) 0.1% FOR DERMATOLOGIC USE ONLY. NOT FOR USE IN EYES.
 DERMATP intment (prednicarbate ointment) 0.1% FR DERMATLGIC USE NLY. NT FR USE IN EYES. DESCRIPTIN DERMATP intment (prednicarbate ointment) 0.1% contains the non-halogenated prednisolone derivative prednicarbate.
DERMATP intment (prednicarbate ointment) 0.1% FR DERMATLGIC USE NLY. NT FR USE IN EYES. DESCRIPTIN DERMATP intment (prednicarbate ointment) 0.1% contains the non-halogenated prednisolone derivative prednicarbate.
Summary of Product Characteristics
 Summary of Product Characteristics 1. NAME OF THE MEDICINAL PRODUCT Terbinafine Dermapharm, 10 mg/g, cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One gram of cream contains 10 mg of terbinafine hydrochloride.
Summary of Product Characteristics 1. NAME OF THE MEDICINAL PRODUCT Terbinafine Dermapharm, 10 mg/g, cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One gram of cream contains 10 mg of terbinafine hydrochloride.
50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate).
 DUPISOR Composition Gel 50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate). Action Calcipotriol is a non-steroidal antipsoriatic agent, derived from vitamin D. Calcipotriol
DUPISOR Composition Gel 50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate). Action Calcipotriol is a non-steroidal antipsoriatic agent, derived from vitamin D. Calcipotriol
Core Safety Profile. Pharmaceutical form(s)/strength: solution 1%, spray 1%, cream 1%, gel 1% SK/H/PSUR/0005/001 Date of FAR:
 Core Safety Profile Active substance: Terbinafine Pharmaceutical form(s)/strength: solution 1%, spray 1%, cream 1%, gel 1% P-RMS: SK/H/PSUR/0005/001 Date of FAR: 08.06.2012 4.3 Contraindications Hypersensitivity
Core Safety Profile Active substance: Terbinafine Pharmaceutical form(s)/strength: solution 1%, spray 1%, cream 1%, gel 1% P-RMS: SK/H/PSUR/0005/001 Date of FAR: 08.06.2012 4.3 Contraindications Hypersensitivity
Viroptic (trifluridine) solution [Monarch Pharmaceuticals, Inc.]
![Viroptic (trifluridine) solution [Monarch Pharmaceuticals, Inc.] Viroptic (trifluridine) solution [Monarch Pharmaceuticals, Inc.]](/thumbs/94/118693790.jpg) Viroptic (trifluridine) solution [Monarch Pharmaceuticals, Inc.] Description VIROPTIC is the brand name for trifluridine (also known as trifluorothymidine, F3TdR,F3T), an antiviral drug for topical treatment
Viroptic (trifluridine) solution [Monarch Pharmaceuticals, Inc.] Description VIROPTIC is the brand name for trifluridine (also known as trifluorothymidine, F3TdR,F3T), an antiviral drug for topical treatment
ALDARA 5% Cream PACKAGE LEAFLET: INFORMATION FOR THE USER. What is in this leaflet? 1. WHAT ALDARA CREAM IS AND WHAT IT IS USED FOR
 PACKAGE LEAFLET: INFORMATION FOR THE USER ALDARA 5% Cream IMIQUIMOD This leaflet is a copy of the Summary of Product Characteristics and Patient Information Leaflet for a medicine, which outlines the conditions
PACKAGE LEAFLET: INFORMATION FOR THE USER ALDARA 5% Cream IMIQUIMOD This leaflet is a copy of the Summary of Product Characteristics and Patient Information Leaflet for a medicine, which outlines the conditions
CLINICAL PHARMACOLOGY
 ELOCON brand of mometasone furoate ointment Ointment 0.1% For Dermatologic Use Only Not for Ophthalmic Use DESCRIPTION ELOCON (mometasone furoate ointment) Ointment, 0.1%, contains mometasone furoate,
ELOCON brand of mometasone furoate ointment Ointment 0.1% For Dermatologic Use Only Not for Ophthalmic Use DESCRIPTION ELOCON (mometasone furoate ointment) Ointment, 0.1%, contains mometasone furoate,
CONTRAINDICATIONS None. (4)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use FINACEA FOAM safely and effectively. See full prescribing information for FINACEA FOAM. FINACEA (azelaic
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use FINACEA FOAM safely and effectively. See full prescribing information for FINACEA FOAM. FINACEA (azelaic
Prescribing Information. Taro-Clobetasol. Taro-Clobetasol
 Prescribing Information Pr Taro-Clobetasol Clobetasol Propionate Cream USP, 0.05% w/w Pr Taro-Clobetasol Clobetasol Propionate Ointment USP, 0.05% w/w Therapeutic Classification Topical corticosteroid
Prescribing Information Pr Taro-Clobetasol Clobetasol Propionate Cream USP, 0.05% w/w Pr Taro-Clobetasol Clobetasol Propionate Ointment USP, 0.05% w/w Therapeutic Classification Topical corticosteroid
Provide relief with Topicort 0.05% 1,2. For your patients with corticosteroid-responsive dermatoses... IMPORTANT SAFETY INFORMATION
 For your patients with corticosteroid-responsive dermatoses... SPREAD THE Provide relief with Topicort 0.05% 1,2 Group C molecule 3 Low allergenic potential 4-6 Available in cream and ointment 3,7 Topicort
For your patients with corticosteroid-responsive dermatoses... SPREAD THE Provide relief with Topicort 0.05% 1,2 Group C molecule 3 Low allergenic potential 4-6 Available in cream and ointment 3,7 Topicort
CONTRAINDICATIONS None.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PAZEO safely and effectively. See full prescribing information for PAZEO. PAZEO (olopatadine hydrochloride
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PAZEO safely and effectively. See full prescribing information for PAZEO. PAZEO (olopatadine hydrochloride
What you need to know about EGW
 For the treatment of external genital and perianal warts (EGW) What you need to know about EGW Indication VEREGEN is indicated for the topical treatment of external genital and perianal warts (Condylomata
For the treatment of external genital and perianal warts (EGW) What you need to know about EGW Indication VEREGEN is indicated for the topical treatment of external genital and perianal warts (Condylomata
Immodium / loprarmide
 Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
Chemical structure of calcipotriol
 PRODUCT INFORMATION DAIVONEX CREAM AUST R 57354 Calcipotriol 50 microgram/g NAME OF THE MEDICINE: CALCIPOTRIOL DESCRIPTION Calcipotriol is a white or almost white crystalline substance. It is a vitamin
PRODUCT INFORMATION DAIVONEX CREAM AUST R 57354 Calcipotriol 50 microgram/g NAME OF THE MEDICINE: CALCIPOTRIOL DESCRIPTION Calcipotriol is a white or almost white crystalline substance. It is a vitamin
For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory. Light Liquid Paraffin and White Soft Paraffin Cream
 For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory OILATUM CREAM / KIDS CREAM Light Liquid Paraffin and White Soft Paraffin Cream QUALITATIVE AND QUANTITATIVE COMPOSITION
For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory OILATUM CREAM / KIDS CREAM Light Liquid Paraffin and White Soft Paraffin Cream QUALITATIVE AND QUANTITATIVE COMPOSITION
CORTISPORIN Ointment (neomycin and polymyxin B sulfates, bacitracin zinc, and. hydrocortisone ointment, USP)
 CORTISPORIN Ointment (neomycin and polymyxin B sulfates, bacitracin zinc, and hydrocortisone ointment, USP) DESCRIPTION CORTISPORIN Ointment (neomycin and polymyxin B sulfates, bacitracin zinc, and hydrocortisone
CORTISPORIN Ointment (neomycin and polymyxin B sulfates, bacitracin zinc, and hydrocortisone ointment, USP) DESCRIPTION CORTISPORIN Ointment (neomycin and polymyxin B sulfates, bacitracin zinc, and hydrocortisone
SUCRALFATE TABLETS, USP
 1234567890 10 210002 SUCRALFATE TABLETS, USP DESCRIPTION Sucralfate is an -D-glucopyranoside, -D-fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex. It has the following structural formula:
1234567890 10 210002 SUCRALFATE TABLETS, USP DESCRIPTION Sucralfate is an -D-glucopyranoside, -D-fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex. It has the following structural formula:
DIPROLENE AF (augmented betamethasone dipropionate) Cream, 0.05% for topical use Initial U.S. Approval: 1983
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DIPROLENE AF Cream safely and effectively. See full prescribing information for DIPROLENE AF Cream.
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DIPROLENE AF Cream safely and effectively. See full prescribing information for DIPROLENE AF Cream.
1 INDICATIONS AND USAGE. 1.1 Limitation of Use FULL PRESCRIBING INFORMATION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use TRIFERIC safely and effectively. See full prescribing information for TRIFERIC. TRIFERIC (ferric
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use TRIFERIC safely and effectively. See full prescribing information for TRIFERIC. TRIFERIC (ferric
Dr A Anzarut, MSc, CIP, MD, FRCSC
 Dr A Anzarut, MSc, CIP, MD, FRCSC Plastic and Cosmetic Surgery ASSH Fellowship trained hand surgeon 201 2763 Beverly Street, Duncan, British Columbia Tel: (250) 597 2064 Fax: (250) 597-1297 PATIENT INFORMATION
Dr A Anzarut, MSc, CIP, MD, FRCSC Plastic and Cosmetic Surgery ASSH Fellowship trained hand surgeon 201 2763 Beverly Street, Duncan, British Columbia Tel: (250) 597 2064 Fax: (250) 597-1297 PATIENT INFORMATION
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Fungasil 10 mg/g cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One gram of cream contains 10 mg of terbinafine hydrochloride. Excipients
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Fungasil 10 mg/g cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One gram of cream contains 10 mg of terbinafine hydrochloride. Excipients
DOSAGE AND ADMINISTRATION Apply a thin layer of FINACEA Gel twice daily to affected area(s).
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use FINACEA Gel safely and effectively. See full prescribing information for FINACEA Gel. FINACEA (azelaic
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use FINACEA Gel safely and effectively. See full prescribing information for FINACEA Gel. FINACEA (azelaic
Reference ID:
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use TRIFERIC safely and effectively. See full prescribing information for TRIFERIC. TRIFERIC (ferric
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use TRIFERIC safely and effectively. See full prescribing information for TRIFERIC. TRIFERIC (ferric
HIGHLIGHTS OF PRESCRIBING INFORMATION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use VERDESO Foam safely and effectively. See full prescribing information for VERDESO Foam. VERDESO (desonide)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use VERDESO Foam safely and effectively. See full prescribing information for VERDESO Foam. VERDESO (desonide)
ADVERSE REACTIONS The most common (>10%) adverse reactions are hypercalcemia, nausea, and diarrhea. (6.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOSLYRA safely and effectively. See full prescribing information for PHOSLYRA. PHOSLYRA (calcium
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOSLYRA safely and effectively. See full prescribing information for PHOSLYRA. PHOSLYRA (calcium
NEW ZEALAND DATA SHEET PEVARYL 1% w/w Foaming Solution
 1 PRODUCT NAME PEVARYL 1% w/w foaming solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Econazole nitrate 1.0% w/w (as econazole base) Excipients with known effect: Benzyl alcohol For a full list of
1 PRODUCT NAME PEVARYL 1% w/w foaming solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Econazole nitrate 1.0% w/w (as econazole base) Excipients with known effect: Benzyl alcohol For a full list of
NEOSPORIN Ophthalmic Solution Sterile (neomycin and polymyxin B sulfates and gramicidin ophthalmic solution, USP)
 NEOSPORIN Ophthalmic Solution Sterile (neomycin and polymyxin B sulfates and gramicidin ophthalmic solution, USP) NEOSPORIN SOLUTION DESCRIPTION NEOSPORIN Ophthalmic Solution (neomycin and polymyxin B
NEOSPORIN Ophthalmic Solution Sterile (neomycin and polymyxin B sulfates and gramicidin ophthalmic solution, USP) NEOSPORIN SOLUTION DESCRIPTION NEOSPORIN Ophthalmic Solution (neomycin and polymyxin B
Fluocinolone Acetonide 0.01% Topical Oil (Scalp Oil)
 Fluocinolone Acetonide 0.01% Topical Oil (Scalp Oil) For Topical Use Only- Not for Oral, Ophthalmic, or Intravaginal Use DESCRIPTION Fluocinolone Acetonide 0.01% Topical Oil contains fluocinolone acetonide
Fluocinolone Acetonide 0.01% Topical Oil (Scalp Oil) For Topical Use Only- Not for Oral, Ophthalmic, or Intravaginal Use DESCRIPTION Fluocinolone Acetonide 0.01% Topical Oil contains fluocinolone acetonide
0BCore Safety Profile. Pharmaceutical form(s)/strength: Cream 1% DK/H/PSUR/0009/005 Date of FAR:
 0BCore Safety Profile Active substance: Pimecrolimus Pharmaceutical form(s)/strength: Cream 1% P-RMS: DK/H/PSUR/0009/005 Date of FAR: 06.06.2013 4.3 Contraindications Hypersensitivity to pimecrolimus,
0BCore Safety Profile Active substance: Pimecrolimus Pharmaceutical form(s)/strength: Cream 1% P-RMS: DK/H/PSUR/0009/005 Date of FAR: 06.06.2013 4.3 Contraindications Hypersensitivity to pimecrolimus,
There s comfort in the familiar
 When treating corticosteroid-responsive dermatoses... [ Be sure to write Psorcon (diflorasone) Cream ] Diflorasone Diacetate Cream, 0.05%, 60 g $ 0 Benefits to the healthcare professional: There s comfort
When treating corticosteroid-responsive dermatoses... [ Be sure to write Psorcon (diflorasone) Cream ] Diflorasone Diacetate Cream, 0.05%, 60 g $ 0 Benefits to the healthcare professional: There s comfort
Panretin (alitretinoin) gel 0.1% (For topical use only) Rx only DESCRIPTION
 Panretin (alitretinoin) gel 0.1% (For topical use only) Rx only DESCRIPTION Panretin gel 0.1% contains alitretinoin and is intended for topical application only. The chemical name is 9-cis-retinoic acid
Panretin (alitretinoin) gel 0.1% (For topical use only) Rx only DESCRIPTION Panretin gel 0.1% contains alitretinoin and is intended for topical application only. The chemical name is 9-cis-retinoic acid
WARNINGS AND PRECAUTIONS
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use XOLEGEL Gel safely and effectively. See full prescribing information for XOLEGEL Gel. XOLEGEL (ketoconazole)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use XOLEGEL Gel safely and effectively. See full prescribing information for XOLEGEL Gel. XOLEGEL (ketoconazole)
Revised: 03/2018. *Sections or subsections omitted from the full prescribing information are not listed.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DIPROLENE Ointment safely and effectively. See full prescribing information for DIPROLENE Ointment.
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DIPROLENE Ointment safely and effectively. See full prescribing information for DIPROLENE Ointment.
INDICATIONS ACULAR 0,4% ophthalmic solution is indicated for the reduction of ocular pain and burning/stinging following corneal refractive surgery.
 Page 1 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME (and dosage form) ACULAR 0,4% COMPOSITION ACULAR 0,4% ophthalmic solution contains: Ketorolac tromethamine: 4 mg/ml Preservative: Benzalkonium chloride
Page 1 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME (and dosage form) ACULAR 0,4% COMPOSITION ACULAR 0,4% ophthalmic solution contains: Ketorolac tromethamine: 4 mg/ml Preservative: Benzalkonium chloride
ELOCON (mometasone furoate) Ointment, 0.1% for topical use Initial U.S. Approval: 1987
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ELOCON Ointment safely and effectively. See full prescribing information for ELOCON Ointment. ELOCON
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ELOCON Ointment safely and effectively. See full prescribing information for ELOCON Ointment. ELOCON
Chemical Name: 4H-Pyrano[3,2-g]quinoline-2,8-dicarboxylic acid, 9-ethyl-6,9-dihydro-4,6-dioxo-10-propyl-, disodium salt.
![Chemical Name: 4H-Pyrano[3,2-g]quinoline-2,8-dicarboxylic acid, 9-ethyl-6,9-dihydro-4,6-dioxo-10-propyl-, disodium salt. Chemical Name: 4H-Pyrano[3,2-g]quinoline-2,8-dicarboxylic acid, 9-ethyl-6,9-dihydro-4,6-dioxo-10-propyl-, disodium salt.](/thumbs/72/68057993.jpg) ALCRIL (nedocromil sodium ophthalmic solution) 2% sterile DESCRIPTIN ALCRIL (nedocromil sodium ophthalmic solution) 2% is a clear, yellow, sterile solution for topical ophthalmic use. Nedocromil sodium
ALCRIL (nedocromil sodium ophthalmic solution) 2% sterile DESCRIPTIN ALCRIL (nedocromil sodium ophthalmic solution) 2% is a clear, yellow, sterile solution for topical ophthalmic use. Nedocromil sodium
AUSTRALIAN PRODUCT INFORMATION RESOLVE PLUS 1.0 (MICONAZOLE NITRATE, HYDROCORTISONE) CREAM
 AUSTRALIAN PRODUCT INFORMATION RESOLVE PLUS 1.0 (MICONAZOLE NITRATE, HYDROCORTISONE) CREAM 1 NAME OF THE MEDICINE Miconazole nitrate, Hydrocortisone. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Miconazole
AUSTRALIAN PRODUCT INFORMATION RESOLVE PLUS 1.0 (MICONAZOLE NITRATE, HYDROCORTISONE) CREAM 1 NAME OF THE MEDICINE Miconazole nitrate, Hydrocortisone. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Miconazole
The no t-so-simple cutaneous wart
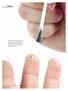 Salicylic acid is a commonly used over-the-counter treatment for nongenital warts. Clearance rates of up to 75% have been reported with salicylic acid. istockphoto 10 Clinical practice guide Dermatology
Salicylic acid is a commonly used over-the-counter treatment for nongenital warts. Clearance rates of up to 75% have been reported with salicylic acid. istockphoto 10 Clinical practice guide Dermatology
QBREXZA (glycopyrronium) cloth, 2.4%, for topical use Initial U.S. Approval: 2018
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use QBREXZA safely and effectively. See full prescribing information for QBREXZA. QBREXZA (glycopyrronium)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use QBREXZA safely and effectively. See full prescribing information for QBREXZA. QBREXZA (glycopyrronium)
CUTIVATE (fluticasone propionate) Lotion PharmaDerm, A division of Nycomed US Inc. Rx Only
 Page 3 CUTIVATE (fluticasone propionate) Lotion PharmaDerm, A division of Nycomed US Inc. Rx Only FOR TOPICAL USE ONLY. NOT FOR OPHTHALMIC, ORAL, OR INTRAVAGINAL USE. DESCRIPTION CUTIVATE (fluticasone
Page 3 CUTIVATE (fluticasone propionate) Lotion PharmaDerm, A division of Nycomed US Inc. Rx Only FOR TOPICAL USE ONLY. NOT FOR OPHTHALMIC, ORAL, OR INTRAVAGINAL USE. DESCRIPTION CUTIVATE (fluticasone
1. NAME OF THE MEDICINAL PRODUCT. Lamisil 1% cutaneous solution. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 1. NAME OF THE MEDICINAL PRODUCT Lamisil 1% cutaneous solution. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active substance: 10 mg terbinafine hydrochloride per 1 g solution (1% w/w). Excipient(s) with
1. NAME OF THE MEDICINAL PRODUCT Lamisil 1% cutaneous solution. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active substance: 10 mg terbinafine hydrochloride per 1 g solution (1% w/w). Excipient(s) with
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Nizoral 20 mg/g Cream Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Ketoconazole 2% w/w (each gram of cream contains 20mg). Excipients; propylene
1 NAME OF THE MEDICINAL PRODUCT Nizoral 20 mg/g Cream Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Ketoconazole 2% w/w (each gram of cream contains 20mg). Excipients; propylene
University Health Services at CMU STI Awareness Month specials for students:
 University Health Services at CMU STI Awareness Month specials for students: -Free condoms during April (10 per student) -Free walk-in rapid HIV testing April 22-27 (no appointment needed) -STI Screening
University Health Services at CMU STI Awareness Month specials for students: -Free condoms during April (10 per student) -Free walk-in rapid HIV testing April 22-27 (no appointment needed) -STI Screening
Sexually Transmitted. Diseases
 Sexually Transmitted Diseases How can I get an STD? Many STDs are carried and transmitted through semen and vaginal fluids. Some STDs can be spread through skin to skin contact Mother to child STDs: Signs
Sexually Transmitted Diseases How can I get an STD? Many STDs are carried and transmitted through semen and vaginal fluids. Some STDs can be spread through skin to skin contact Mother to child STDs: Signs
SUCRALFATE TABLETS, USP
 1234567890 10 210002-01 SUCRALFATE TABLETS, USP DESCRIPTION Sucralfate is an -D-glucopyranoside, -D-fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex. It has the following structural formula:
1234567890 10 210002-01 SUCRALFATE TABLETS, USP DESCRIPTION Sucralfate is an -D-glucopyranoside, -D-fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex. It has the following structural formula:
Neomycin B (R 1 =H, R 2 =CH 2 NH 2 ) Neomycin C (R 1 =CH 2 NH 2, R 2 =H) The chemical structure for the active ingredient Polymyxin B Sulfate is:
 MAXITROL * (neomycin and polymyxin B sulfates and dexamethasone ophthalmic ointment) Sterile DESCRIPTION MAXITROL (neomycin and polymyxin B sulfates and dexamethasone ophthalmic ointment) is a multiple
MAXITROL * (neomycin and polymyxin B sulfates and dexamethasone ophthalmic ointment) Sterile DESCRIPTION MAXITROL (neomycin and polymyxin B sulfates and dexamethasone ophthalmic ointment) is a multiple
NEW ZEALAND DATA SHEET
 1. PRODUCT NAME (strength pharmaceutical form) Hydrocortisone Cream 1% (PSM) Topical Cream 1% w/w 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Name and strength of the active substance Hydrocortisone Cream
1. PRODUCT NAME (strength pharmaceutical form) Hydrocortisone Cream 1% (PSM) Topical Cream 1% w/w 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Name and strength of the active substance Hydrocortisone Cream
Timby/Smith: Introductory Medical-Surgical Nursing, 9/e
 Timby/Smith: Introductory Medical-Surgical Nursing, 9/e Chapter 62: Caring for Clients With Sexually Transmitted Diseases Slide 1 Epidemiology Introduction Study of the occurrence, distribution, and causes
Timby/Smith: Introductory Medical-Surgical Nursing, 9/e Chapter 62: Caring for Clients With Sexually Transmitted Diseases Slide 1 Epidemiology Introduction Study of the occurrence, distribution, and causes
13 NONCLINICAL TOXICOLOGY 5.1 Increased Bleeding Time Carcinogenesis, Mutagenesis, Impairment of 5.2 Delayed Healing
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ILEVRO* (nepafenac ophthalmic suspension), 0.3% safely and effectively. See full prescribing information
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ILEVRO* (nepafenac ophthalmic suspension), 0.3% safely and effectively. See full prescribing information
CONTRAINDICATIONS Active ocular infections (4).
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DEXTENZA safely and effectively. See full prescribing information for DEXTENZA. DEXTENZA (dexamethasone
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DEXTENZA safely and effectively. See full prescribing information for DEXTENZA. DEXTENZA (dexamethasone
LUPIN LIMITED SAFETY DATA SHEET. Section 1: Identification. Abacavir Sulfate, Lamivudine and Zidovudine Tablets 300 mg, 150 mg and 300 mg
 LUPIN LIMITED SAFETY DATA SHEET Section 1: Identification Section 1, Identification Material Manufacturer Distributor Abacavir Sulfate, Lamivudine and Zidovudine Tablets 300 mg, 150 mg and 300 mg Lupin
LUPIN LIMITED SAFETY DATA SHEET Section 1: Identification Section 1, Identification Material Manufacturer Distributor Abacavir Sulfate, Lamivudine and Zidovudine Tablets 300 mg, 150 mg and 300 mg Lupin
TOPCORT Cream/Ointment (Mometasone furoate 0.1%)
 Published on: 10 Jul 2014 TOPCORT Cream/Ointment (Mometasone furoate 0.1%) Composition TOPCORT Cream Mometasone Furoate, IP... 0.1% w/w In a cream base... q.s. TOPCORT Ointment Mometasone Furoate, IP...
Published on: 10 Jul 2014 TOPCORT Cream/Ointment (Mometasone furoate 0.1%) Composition TOPCORT Cream Mometasone Furoate, IP... 0.1% w/w In a cream base... q.s. TOPCORT Ointment Mometasone Furoate, IP...
Prescribing Information
 Prescribing Information Pr DERMOVATE Cream (clobetasol propionate cream, USP) Pr DERMOVATE Ointment (clobetasol propionate ointment, USP) Topical corticosteroid TaroPharma Preparation Date: A Division
Prescribing Information Pr DERMOVATE Cream (clobetasol propionate cream, USP) Pr DERMOVATE Ointment (clobetasol propionate ointment, USP) Topical corticosteroid TaroPharma Preparation Date: A Division
Make Love Not Warts Genital Warts
 Patricia Wong, MD 735 Cowper Street Palo Alto, CA 94301 650-473-3173 www.patriciawongmd.com Make Love Not Warts Genital Warts Genital warts are the most common sexually transmitted disease. The lifetime
Patricia Wong, MD 735 Cowper Street Palo Alto, CA 94301 650-473-3173 www.patriciawongmd.com Make Love Not Warts Genital Warts Genital warts are the most common sexually transmitted disease. The lifetime
In Canada and around the world, the trend is clear: sexually transmitted infections (STIs) are on the rise.
 Adapted From: Sexually Transmitted Infections Pamphlet. Public Health Agency of Canada, 2007 In Canada and around the world, the trend is clear: sexually transmitted infections (STIs) are on the rise.
Adapted From: Sexually Transmitted Infections Pamphlet. Public Health Agency of Canada, 2007 In Canada and around the world, the trend is clear: sexually transmitted infections (STIs) are on the rise.
INVELTYS (loteprednol etabonate ophthalmic suspension) 1%, for topical ophthalmic use Initial U.S. Approval: 1998
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use INVELTYS safely and effectively. See full prescribing information for INVELTYS. INVELTYS (loteprednol
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use INVELTYS safely and effectively. See full prescribing information for INVELTYS. INVELTYS (loteprednol
Coly-Mycin S Otic with Neomycin and Hydrocortisone (colistin sulfate neomycin sulfate thonzonium bromide hydrocortisone acetate otic suspension)
 Coly-Mycin S Otic with Neomycin and Hydrocortisone (colistin sulfate neomycin sulfate thonzonium bromide hydrocortisone acetate otic suspension) DESCRIPTION Coly-Mycin S Otic with Neomycin and Hydrocortisone
Coly-Mycin S Otic with Neomycin and Hydrocortisone (colistin sulfate neomycin sulfate thonzonium bromide hydrocortisone acetate otic suspension) DESCRIPTION Coly-Mycin S Otic with Neomycin and Hydrocortisone
FULL PRESCRIBING INFORMATION: CONTENTS*
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OTIPRIO safely and effectively. See full prescribing information for OTIPRIO. OTIPRIO (ciprofloxacin
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OTIPRIO safely and effectively. See full prescribing information for OTIPRIO. OTIPRIO (ciprofloxacin
PRODUCT INFORMATION VANIQA EFLORNITHINE CREAM, 11.5%
 NAME OF THE MEDICINE eflornithine hydrochloride PRODUCT INFORMATION VANIQA EFLORNITHINE CREAM, 11.5% Chemically, eflornithine hydrochloride (±)-2-(difluoromethyl) ornithine monohydrochloride monohydrate.
NAME OF THE MEDICINE eflornithine hydrochloride PRODUCT INFORMATION VANIQA EFLORNITHINE CREAM, 11.5% Chemically, eflornithine hydrochloride (±)-2-(difluoromethyl) ornithine monohydrochloride monohydrate.
The only biologic approved to treat SLE: now with multiple delivery options
 The only biologic approved to treat SLE: now with multiple delivery options BENLYSTA (belimumab) Autoinjector SC Prefilled syringe IV Intravenous infusion Consider the options: visit Belimumab.com INDICATION
The only biologic approved to treat SLE: now with multiple delivery options BENLYSTA (belimumab) Autoinjector SC Prefilled syringe IV Intravenous infusion Consider the options: visit Belimumab.com INDICATION
