Extracorporeal Shockwave Therapy (ESWT) and Radial Pulse Therapy (RPT) August 2016
|
|
|
- Vivian Patrick
- 5 years ago
- Views:
Transcription
1 Commissioning Policy Extracorporeal Shockwave Therapy (ESWT) and Radial Pulse Therapy (RPT) August 2016 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible: NHS South Worcestershire Clinical Commissioning Group (CCG) NHS Redditch & Bromsgrove Clinical Commissioning Group (CCG) NHS Wyre Forest Clinical Commissioning Group (CCG) Collectively referred to as the Worcestershire CCGs COMMISSIONING SUMMARY Worcestershire Clinical Commissioning Policy Collaboration has undertaken a review of the evidence and circumstances associated with use of Electrocorporeal Shockwave Therapy (ESWT) and Radial Pulse Therapy (RPT) and concluded that for Worcestershire CCGs responsible patients: Services associated with use of either ESWT or RPT are NOT ROUTINELY COMMISSIONED for any indication including plantar fasciitis and Achilles tendinopathy. Further clarity is required in relation to the optimum duration of use and long term benefit and the impact of either treatment on concomitant and future use of other interventions including progression to surgery. Do you need this document in other languages or formats (i.e. large print)? Please contact the Communications Team on
2 Document Details: Version: 1.0 Ratified by (name and date of NHS Redditch & Bromsgrove CCG CET 06/07/16 Committee): NHS South Worcestershire CCG CET 12/07/16 NHS Wyre Forest CCG CET 26/07/16 Date issued: 5 th August 2016 Internal Review Date: Documents will be reviewed as a minimum every 3 years. However, earlier revisions to the policy may be made in light of published updates to local and national evidence of effectiveness and cost effectiveness and/or recommendations and guidelines from local, national and international clinical professional bodies. Lead Executive/Director: Name of originator/author: Target audience: Distribution: Equality & Diversity Impact Assessment Date to Initiate Review: 05/08/2019 Chris Emerson, Director of Contracting Fiona Bates, Medicines Commissioner and Public Health Liaison Patients, GPs, Secondary Care and Primary Care (Community) Providers, Independent Sector Providers GPs, Secondary Care and Primary Care (Community) Providers, Independent Sector Providers 24 February 2016 Key individuals involved in developing the document: Name Mrs Fiona Bates Mrs Helen Bryant Ms Saira Waraich Designation Medicines Commissioning/Public Health, South Worcestershire CCG Senior Commissioning Manager, Redditch and Bromsgrove CCG FY2 Public Health, Worcestershire County Council Circulated to the following individuals/groups for comments: Name Clinical Commissioning Policy Collaborative, which includes: GPs, Commissioners, Medicines Commissioning, Public Health, Patient and Public Representatives Date 8 th March 2016 Version Control: Version No Type of Change Date Description of change ESWT and RPT Commissioning Policy Page 2 of 10
3 Table of Contents 1. Definitions Scope of policy Background Relevant National Guidance and Facts Evidence Review Patient Eligibility Supporting Documents Equality Impact Assessment... 9 ESWT and RPT Commissioning Policy Page 3 of 10
4 1. Definitions 1.1 Exceptional clinical circumstances are clinical circumstances pertaining to a particular patient, which can properly be described as exceptional, when compared to the clinical circumstances of other patients with the same clinical condition and at the same stage of development of that condition (i.e. similar patients). A patient with exceptional clinical circumstances will have clinical features or characteristics which differentiate that patient from other patients in that cohort and result in that patient being likely to obtain significantly greater clinical benefit (than those other patients) from the intervention for which funding is sought. 1.2 A Similar Patient is a patient who is likely to be in the same or similar clinical circumstances as the requesting patient and who could reasonably be expected to benefit from the requested treatment to the same or a similar degree. The existence of more than one similar patients indicates that a decision regarding the commissioning of a service development or commissioning policy is required of the Commissioner. 1.3 An individual funding request (IFR) is a request received from a provider or a patient with explicit support from a clinician, which seeks exceptional funding for a single identified patient for a specific treatment. 1.4 An in-year service development is any aspect of healthcare, other than one which is the subject of a successful individual funding request, which the Commissioner agrees to fund outside of the annual commissioning round. Such unplanned investment decisions should only be made in exceptional circumstances because, unless they can be funded through disinvestment, they will have to be funded as a result of either delaying or aborting other planned developments. 2. Scope of policy 2.1 This policy is part of a suite of locally endorsed Commissioning Policies. Copies of these Commissioning Policies are available on the following website address: This policy applies to all patients for whom the Worcestershire CCGs have responsibility including: People provided with primary medical services by GP practices which are members of any one of the CCGs and People usually resident in any of the areas covered by the CCG's and not provided with primary medical services by any CCG. 2.3 The policy relates to electrocorporeal shockwave therapy treatment and radial pulse therapy used in the management of a variety of pain related conditions including but not limited to plantar fasciitis and Achilles tendinopathy. 2.4 For patients who do not fall within the eligibility criteria set out in the policy but where there is demonstrable evidence that the patient has exceptional clinical circumstances, an Individual Funding Request may be submitted for consideration. The referring clinician should consult the Commissioner s Operational Policy for Individual Funding Requests document for further guidance on this process. For a definition of the term exceptional clinical circumstances, please refer to the Definitions section of this document. ESWT and RPT Commissioning Policy Page 4 of 10
5 3. Background 3.1. The NHS Constitution, which details the principles and values that guide the NHS, has been applied in the agreement of this policy NHS Redditch & Bromsgrove Clinical Commissioning Group, NHS South Worcestershire Clinical Commissioning Group and NHS Wyre Forest Clinical Commissioning Group consider all lives of all patients whom they serve to be of equal value and, in making decisions about funding treatment for patients, will seek not to discriminate on the grounds of sex, age, sexual orientation, ethnicity, educational level, employment, marital status, religion or disability except where a difference in the treatment options made available to patients is directly related a particular patient s clinical condition or is related to the anticipated benefits to be derived from a proposed form of treatment Extracorporeal Shockwave Therapy (ESWT) is a relatively new development that has been trialled in the treatment of chronic heel pain, including but not limited to, pain caused by plantar fasciitis and Achilles tendinopathy In ESWT, acoustic pressure waves are delivered to the focus of pain on the heel, with a view to achieving a reduction in pain. Multiple treatments may be given. The mechanism of action is not yet known, although there are various hypotheses There are various machines manufactured currently that deliver ESWT, which generate shockwaves using electromagnetic, electrohydraulic, or piezoelectric power Radial Pulse Therapy (RPT) is a similar treatment, but can be distinguished from focused ESWT as it differs both in the way it generates and delivers shock waves (using a ballistic source, and delivering pressure pulses more superficially). However, not all clinicians make this distinction ESWT is proposed as a possible non-invasive alternative to surgery where conservative measures such as analgesia, heel supports and physiotherapy have been unsuccessful in significantly improving pain. Avoidance of surgery could prevent associated complications and morbidity for patients, and potentially reduce costs for CCGs, if the effect is maintained. 4. Relevant National Guidance and Facts 4.1 ESWT was evaluated by NICE in 2009 for both plantar fasciitis (IPG 311) and Achilles tendinopathy (IPG 312). At the time, ESWT had been shown to be safe to use in both conditions, but there had not been convincing evidence for its efficacy in treating either cause of heel pain. NICE Guidance therefore stated that ESWT should only be used with special arrangements for clinical governance, consent and audit or research. 4.2 However, further trials and systematic reviews have since been published, which should be taken into account as NICE guidance is no longer representative of the body of data currently available. 4.3 The British Orthopaedic Foot & Ankle Society does not make mention of ESWT or RPT for the management of either plantar fasciitis or Achilles tendinopathy. ESWT and RPT Commissioning Policy Page 5 of 10
6 4.4 NICE have issued draft interventional procedure guidance for use of ESWT for Achilles tendinopathy due for final publication in September The draft recommendations are: a. The evidence on extracorporeal shockwave therapy (ESWT) for Achilles tendinopathy raises no major safety concerns. Current evidence on efficacy of the procedure is inconsistent and limited in quality and quantity. Therefore, ESWT for Achilles tendinopathy should only be used with special arrangements for clinical governance, consent and audit or research. b. Clinicians wishing to do ESWT for Achilles tendinopathy should take the following actions. Inform the clinical governance leads in their NHS trusts. Ensure that patients understand the uncertainty about the procedure's efficacy and provide them with clear written information. In addition, the use of NICE's information for the public is recommended. Audit and review clinical outcomes of all patients having ESWT for Achilles tendinopathy. c. NICE encourages further research into ESWT for Achilles tendinopathy. Studies should clearly describe patient selection, treatment protocols, use of local anaesthesia and the type and duration of energy applied (see section 3). Studies should include validated outcome measures and have a minimum of 1 year of follow-up. NICE may update the guidance on publication of further evidence. 4.5 Plantar Fasciitis: Pain caused by wear and tear of plantar fascia (tissue in the sole of the foot) due to overuse, injury, or biomechanical predisposition 10% lifetime prevalence More common in athletes, high BMI, and year-olds Causes 80% of all heel pain No association with gender 80% of people will have complete resolution of symptoms within one year. 90% of cases will resolve with non-surgical management Usual Management: First-line: analgesia, arch support, heel cushion, rest, plantar fascia stretches Second-line: corticosteroid injection Third-line: surgical release of plantar fascia (plantar fasciotomy) 4.5 Achilles Tendinopathy: Heel pain resulting from damage to the Achilles tendon, caused by injury, overuse, or biomechanical predisposition. It can be categorised according to the part of the Achilles tendon that is affected i.e. 'midportion' AT affects the middle of the tendon, whereas 'insertional' AT occurs where the tendon inserts into the heel. 6% cumulative lifetime incidence in people who are not physically active; as much as 50% in elite athletes. Most common in men, especially athletes between years of age. Usual Management: First-line: analgesia, rest, stretching exercises, strength training, physiotherapy, heel support. Not suitable for corticosteroid injection Second-line: surgical removal of adhesions/nodules or tendon decompression (up to 25% of refractory Achilles tendinopathy patients progress to surgery) 5. Evidence Review 5.1 Two robust and most recent systematic reviews were available and analysed. Two further systematic reviews undertaken by the York Centre for Reviews and Dissemination were published prior to this and were also analysed. Two of the ESWT and RPT Commissioning Policy Page 6 of 10
7 reviews incorporated ESWT (and RPT) for plantar fasciitis and 3 of the reviews incorporated ESWT for Achilles tendinopathy. The review by Speed (2013) includes patients with either plantar fasciitis or Achilles tendinopathy, primarily incorporating randomised controlled trials and assessed as high quality. The review by Mani-Babu (2014) was specific to Achilles tendinopathy and included a mix of randomised controlled trials and prospective studies, assessed as 50% high quality. Al-Abbad (2013) review focussed on Achilles tendinopathy with overall evidence assessed as satisfactory and concerns regarding the reliability of conclusions reached. Chang (2012) review focussed on plantar fasciitis but appears to be wellconducted and reported comparing different methods/intensity of shockwave delivery. Concerns across all reviews related to the inconsistent definition of ESWT in terms of high dose, low dose and RPT which is sometimes classified as low dose, the number of impulses, and the number of treatment sessions and intervals between; variable patient characteristics in terms of symptoms and use of co-interventions; variable outcome measures. 5.2 Plantar Fasciitis There is high quality evidence available to support the efficacy of high dose ESWT and RPT. There is limited evidence to suggest that ESWT o is comparable to physiotherapy and achieves quicker outcomes. o has a better long term outcome compared with corticosteroid injection. o is comparable to surgery, without the risks of an invasive procedure The available evidence involves short-term intervention of up to 3 sessions over a 3 month period, with follow-up of between 3-6 months. 5.3 Achilles Tendinopathy There is some degree of evidence supporting high dose ESWT in insertional Achilles tendinopathy, whereas low dose evidence is contradictory. There is insufficient evidence for ESWT in midportion Achilles tendinopathy. The available evidence involves short-term intervention of up to 4 sessions over a 4 month period, with medium term follow-up of between 1-2 years 5.4 No other evidence was found to indicate whether ESWT for any indication affects the following: Progression to surgical intervention Reduced need for concomitant intervention/healthcare contacts 5.5 There is insufficient evidence with which to support the use of either ESWT or RPT in clinical practice for either plantar fasciitis or Achilles tendinopathy. There is no clarity regarding the optimum duration of treatment and long-term benefit and the impact on the need for both concomitant and future use of other interventions. It is evident from both trial data assessed and local audits of use that treatment is often undertaken early when there is a high chance of resolution without intervention and that other lifestyle changes may impact on symptom resolution in advance of intervention. There is no evidence from either trial data or local audit data that ESWT/RPT delays the progression to surgical intervention. ESWT and RPT Commissioning Policy Page 7 of 10
8 5.6 A further piece of work reviewed both the need for and the evidence for use of ESWT in diabetic patients: Achilles tendinopathy is reported as more prevalent in diabetic patients but this could not be substantiated in the literature. There is conflicting evidence regarding an association between differences in Achilles tendon structure in the diabetic population. Plantar fasciitis has no association in the literature with diabetes. There are no published trials specifically investigating efficacy of shockwave therapy for Achilles tendinopathy or plantar fasciitis in the diabetic population. Speed et al specifically excludes patients with co-existing disease. There is no sub-group analysis of the diabetic population in any of the published systematic reviews. An earlier meta-analysis of 19 randomised controlled trials for the use of ESWT in plantar fasciitis involved 9 studies which excluded patients with diabetes. Just one cohort study of 225 patients identified 5 diabetic patients in whom treatment with ESWT for plantar fasciitis was significantly less likely to be successful. 6. Patient Eligibility 6.1 Plantar Fasciitis and Achilles Tendinopathy Services involving use of Extracorporeal Shockwave Therapy and Radial Pulse Therapy are not supported for use within the NHS for the Worcestershire CCGs responsible population. 6.2 Other Indications No other indications for use of ESWT or RPT will be funded for use within the NHS for the Worcestershire CCGs responsible population. Any identified new indications for use require submission of a completed new technologies request form for consideration by the Clinical Commissioning Policy Collaboration. 7. Supporting Documents Worcestershire CCPC: Brief Evidence Review of Shockwave Therapy for Heel Pain. October 2015 Worcestershire CCPC: Report Should Shockwave Therapy be Available To Patients with Diabetes Mellitus for Plantar Fasciitis and Achilles Tendinopathy?. May 2016 Worcestershire CCGs: Operational Policy for Individual Funding Requests Worcestershire CCGs: Prioritisation Framework for the Commissioning of Healthcare Services NHS England: Ethical Framework for Priority Setting Resource Allocation NHS England: Individual Funding Requests NHS Constitution, updated 27 th July 2015 ESWT and RPT Commissioning Policy Page 8 of 10
9 8. Equality Impact Assessment Organisation Worcestershire CCGs Department Commissioning Name of lead person Fiona Bates Piece of work being assessed Aims of this piece of work Electrocorporeal Shockwave Therapy (ESWT) and Radial Pulse Therapy (RPT) Commissioning Policy To define the circumstances whether and when ESWT and RPT services will be funded in Worcestershire Date of EIA 07/03/2016 Other partners/stakeholders involved CCPC Who will be affected by this piece of work? Patients with heel pain arising from either plantar fasciitis or Achilles tendinopathy. Single Equality Scheme Strand Gender Race Disability Baseline data and research on the population that this piece of work will affect. What is available? E.g. population data, service user data. What does it show? Are there any gaps? Use both quantitative data and qualitative data where possible. Include consultation with service users wherever possible Achilles tendinopathy is more common in men, who may be affected more by the limitations of this policy; there is no gender difference reported for plantar fasciitis. Achilles tendinopathy and plantar fasciitis are both more common in athletes and there may be varying prevalences of athletes by both race and sport. The increased prevalence of heel pain in athletes will disadvantage the more abled person; 6% cumulative lifetime incidence in people who are not physically active; as much as 50% in elite athletes. Religion/ belief No evidence of a link between religion and heel pain No Sexual No evidence of a general link between sexual orientation and heel pain No orientation Age Plantar fasciitis is more common in athletes (who are likely to be younger) and year olds. Yes Achilles tendinopathy is more common in athletes and year olds. Social deprivation Yes There are associations between social class and levels of physical activity. The Health Survey for England 2008 identified that males in the lowest income quintile had the lowest percentage that met the recommended amount of physical activity. For women, the top quintile demonstrates the highest level of physical activity. When work related activity is removed this disparity with men is increased. Carers No evidence of a link between carers and heel pain No Human rights No impact No Is there likely to be a differential impact? Yes, no, unknown Yes Yes but extent and nature unknown Yes ESWT and RPT Commissioning Policy Page 9 of 10
10 Equality Impact Assessment Action Plan Strand Issue Action required How will you measure the outcome/impact Gender Higher prevalence Achilles tendinopathy in No action cannot influence NA males prevalence. Race Varying prevalences of different sports by No action - cannot influence NA race prevalence Disability Able bodied people who undertake exercise Unable to influence the extent to NA more likely to be disadvantaged by policy which exercise is undertaken Age There are different age groups in whom heel As these indications are largely NA pain is more prevalent. associated with wear and tear, Social Deprivation Level of income can influence the level of physical activity and therefore the likely incidence of heel pain. it is not possible to influence. Unable to influence NA Timescale Lead ESWT and RPT Commissioning Policy Page 10 of 10
Commissioning Policy. Treatment of Snoring. April 2010
 Commissioning Policy Treatment of Snoring April 2010 This commissioning policy applies to patients within: South Worcestershire Clinical Commissioning Group (CCG) Redditch & Bromsgrove Clinical Commissioning
Commissioning Policy Treatment of Snoring April 2010 This commissioning policy applies to patients within: South Worcestershire Clinical Commissioning Group (CCG) Redditch & Bromsgrove Clinical Commissioning
Commissioning Policy. The use of Acupuncture in the Management of Musculoskeletal Pain. July 2013
 Commissioning Policy The use of Acupuncture in the Management of Musculoskeletal Pain July 2013 This commissioning policy applies to patients within: South Worcestershire Clinical Commissioning Group (CCG)
Commissioning Policy The use of Acupuncture in the Management of Musculoskeletal Pain July 2013 This commissioning policy applies to patients within: South Worcestershire Clinical Commissioning Group (CCG)
Reversal of Sterilisation (Men and Women) January 2018
 Commissioning Policy Reversal of Sterilisation (Men and Women) January 2018 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible: NHS South Worcestershire
Commissioning Policy Reversal of Sterilisation (Men and Women) January 2018 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible: NHS South Worcestershire
Commissioning Policy. Exogen Ultrasound Bone Healing System for Long Bone Fractures with Non-Union or Delayed Healing.
 Commissioning Policy Exogen Ultrasound Bone Healing System for Long Bone Fractures with Non-Union or Delayed Healing September 2014 This commissioning policy has been endorsed by and applies to patients
Commissioning Policy Exogen Ultrasound Bone Healing System for Long Bone Fractures with Non-Union or Delayed Healing September 2014 This commissioning policy has been endorsed by and applies to patients
Extracorporeal shockwave therapy for refractory Achilles tendinopathy
 Extracorporeal shockwave therapy for refractory Achilles Issued: August 2009 www.nice.org.uk/ipg312 NHS Evidence has accredited the process used by the NICE Interventional Procedures Programme to produce
Extracorporeal shockwave therapy for refractory Achilles Issued: August 2009 www.nice.org.uk/ipg312 NHS Evidence has accredited the process used by the NICE Interventional Procedures Programme to produce
five or more such episodes in each of the preceding two years
 Commissioning Policy Tonsillectomy (Adults and Children) April 2018 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible: NHS South Worcestershire Clinical
Commissioning Policy Tonsillectomy (Adults and Children) April 2018 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible: NHS South Worcestershire Clinical
Hernia Repair in Adults (18 years and over)
 Commissioning Policy Hernia Repair in Adults (18 years and over) June 2017 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible: NHS South Worcestershire
Commissioning Policy Hernia Repair in Adults (18 years and over) June 2017 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible: NHS South Worcestershire
EXTRACORPOREAL SHOCKWAVE THERAPY (ESWT) INDIVIDUAL FUNDING REQUESTS POLICY
 EXTRACORPOREAL SHOCKWAVE THERAPY (ESWT) INDIVIDUAL FUNDING REQUESTS POLICY Version: 1718.v1 Recommendation by: Date Ratified: 01 November 2017 Name of Originator/Author: Approved by Responsible Committee/Individual:
EXTRACORPOREAL SHOCKWAVE THERAPY (ESWT) INDIVIDUAL FUNDING REQUESTS POLICY Version: 1718.v1 Recommendation by: Date Ratified: 01 November 2017 Name of Originator/Author: Approved by Responsible Committee/Individual:
Management of GALLSTONE Disease and GALLBLADDER Pathology December 2017
 Commissioning Policy Management of GALLSTONE Disease and GALLBLADDER Pathology December 2017 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible: NHS South
Commissioning Policy Management of GALLSTONE Disease and GALLBLADDER Pathology December 2017 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible: NHS South
Commissioning Policy Individual Funding Request
 Commissioning Policy Individual Funding Request Extracorporeal Shockwave Therapy (ESWT) Individual Funding Requests Policy Date Adopted: 6 th February 2017 Version: 1617.1 Individual Funding Request Team
Commissioning Policy Individual Funding Request Extracorporeal Shockwave Therapy (ESWT) Individual Funding Requests Policy Date Adopted: 6 th February 2017 Version: 1617.1 Individual Funding Request Team
Commissioning policy for: Hallux Valgus (bunions)
 Commissioning policy for: Hallux Valgus (bunions) 01 April 2016 VERSION CONTROL Version: 3.0 Ratified by: NHS Warwickshire rth CCG Governing Body Date ratified: 24 March 2016 Name of originator/author:
Commissioning policy for: Hallux Valgus (bunions) 01 April 2016 VERSION CONTROL Version: 3.0 Ratified by: NHS Warwickshire rth CCG Governing Body Date ratified: 24 March 2016 Name of originator/author:
Extra-corporeal Shock Wave Therapy (ESWT) for non-insertional Achilles tendinopathy Inflamed Achilles tendon
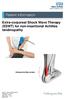 Extra-corporeal Shock Wave Therapy (ESWT) for non-insertional Achilles tendinopathy Inflamed Achilles tendon Source: Trauma & Orthopaedics Reference No: 6459-1 Issue date: 1/2/19 Review date: 1/2/22 Page
Extra-corporeal Shock Wave Therapy (ESWT) for non-insertional Achilles tendinopathy Inflamed Achilles tendon Source: Trauma & Orthopaedics Reference No: 6459-1 Issue date: 1/2/19 Review date: 1/2/22 Page
Musculoskeletal Surgery and Therapeutic Interventions January 2016
 Commissioning Policy Musculoskeletal Surgery and Therapeutic Interventions January 2016 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible: NHS South Worcestershire
Commissioning Policy Musculoskeletal Surgery and Therapeutic Interventions January 2016 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible: NHS South Worcestershire
Extra-corporeal Shock Wave Therapy (ESWT) for plantar fasciitis / heel pain
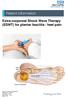 Extra-corporeal Shock Wave Therapy (ESWT) for plantar fasciitis / heel pain Source: Trauma & Orthopaedics Reference No: 6460-1 Issue date: 1/2/19 Review date: 1/2/22 Page 1 of 6 Plantar fasciitis / heel
Extra-corporeal Shock Wave Therapy (ESWT) for plantar fasciitis / heel pain Source: Trauma & Orthopaedics Reference No: 6460-1 Issue date: 1/2/19 Review date: 1/2/22 Page 1 of 6 Plantar fasciitis / heel
Specialised Services Policy:
 Specialised Services Policy: CP35 Cochlear Implants Document Author: Specialised Planner for Women & Children s Services Executive Lead: Director of Planning Approved by: Executive Board Issue Date: 05
Specialised Services Policy: CP35 Cochlear Implants Document Author: Specialised Planner for Women & Children s Services Executive Lead: Director of Planning Approved by: Executive Board Issue Date: 05
Commissioning Policy. Tertiary Treatment for Assisted Conception Services
 Commissioning Policy Tertiary Treatment for Assisted Conception Services April 2013 This commissioning policy applies to patients within: South Worcestershire Clinical Commissioning Group (CCG) Redditch
Commissioning Policy Tertiary Treatment for Assisted Conception Services April 2013 This commissioning policy applies to patients within: South Worcestershire Clinical Commissioning Group (CCG) Redditch
Specialised Services Commissioning Policy. CP29: Bariatric Surgery
 Specialised Services Commissioning Policy CP29: Bariatric Surgery Document Author: Specialist Planner, Cardiothoracic Executive Lead: Director of Planning Approved by: Management Group Issue Date: 12 June
Specialised Services Commissioning Policy CP29: Bariatric Surgery Document Author: Specialist Planner, Cardiothoracic Executive Lead: Director of Planning Approved by: Management Group Issue Date: 12 June
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC)
 BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) Sept 2015 Review: Sept 2018 Bulletin 227: EXOGEN ultrasound bone healing system used for the management of long bone fractures JPC Recommendations:
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) Sept 2015 Review: Sept 2018 Bulletin 227: EXOGEN ultrasound bone healing system used for the management of long bone fractures JPC Recommendations:
Lincolnshire JSNA: Cancer
 What do we know? Summary Around one in three of us will develop cancer at some time in our lives according to our lifetime risk estimation (Sasieni PD, et al 2011). The 'lifetime risk of cancer' is an
What do we know? Summary Around one in three of us will develop cancer at some time in our lives according to our lifetime risk estimation (Sasieni PD, et al 2011). The 'lifetime risk of cancer' is an
A patient s guide to. Radial Extracorporeal Shockwave Therapy (ESWT)
 A patient s guide to Radial Extracorporeal Shockwave Therapy (ESWT) The Foot & Ankle Unit at the Royal National Orthopaedic Hospital is made up of a multi-disciplinary team. The team consists of four specialist
A patient s guide to Radial Extracorporeal Shockwave Therapy (ESWT) The Foot & Ankle Unit at the Royal National Orthopaedic Hospital is made up of a multi-disciplinary team. The team consists of four specialist
Insulin Pumps and Glucose Monitors in Adults Policy
 Insulin Pumps and Glucose Monitors in Adults Policy Version: 2016-19 Ratified by: NHS Leeds West CCG Assurance Committee on; 16 November 2016 NHS Leeds North CCG Governance on Performance and Risk Committee
Insulin Pumps and Glucose Monitors in Adults Policy Version: 2016-19 Ratified by: NHS Leeds West CCG Assurance Committee on; 16 November 2016 NHS Leeds North CCG Governance on Performance and Risk Committee
CATARACT REFERRAL FOR ASSESSMENT OF SURGICAL TREATMENT CRITERIA BASED ACCESS POLICY
 CATARACT REFERRAL FOR ASSESSMENT OF SURGICAL TREATMENT CRITERIA BASED ACCESS POLICY Version: Recommendation by: 1516.V1a Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: MAY 2015
CATARACT REFERRAL FOR ASSESSMENT OF SURGICAL TREATMENT CRITERIA BASED ACCESS POLICY Version: Recommendation by: 1516.V1a Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: MAY 2015
Hip Replacement Surgery Including referral for Surgical Assessment of Osteoarthritis Criteria Based Access Policy
 Hip Replacement Surgery Including referral for Surgical Assessment of Osteoarthritis Criteria Based Access Policy Version: 1617.v6 Recommendation by: Somerset CCG Clinical Commissioning Policy Forum (CCPF)
Hip Replacement Surgery Including referral for Surgical Assessment of Osteoarthritis Criteria Based Access Policy Version: 1617.v6 Recommendation by: Somerset CCG Clinical Commissioning Policy Forum (CCPF)
Draft Falls Prevention Strategy
 Cheshire West & Chester Council Draft Falls Prevention Strategy 2017-2020 Visit: cheshirewestandchester.gov.uk Visit: cheshirewestandchester.gov.uk 02 Cheshire West and Chester Council Draft Falls Prevention
Cheshire West & Chester Council Draft Falls Prevention Strategy 2017-2020 Visit: cheshirewestandchester.gov.uk Visit: cheshirewestandchester.gov.uk 02 Cheshire West and Chester Council Draft Falls Prevention
GROMMET INSERTION RECURRENT ACUTE OTITIS MEDIA (WITHOUT EFFUSION) SECONDARY CARE PRIOR APPROVAL POLICY
 Version: 1718.v1 Ratified by: SCCG COG Date Ratified: 05 April 2017 Name of Originator/Author: Name of Responsible Committee/Individual: IFR SCCG CCPF/ IFR Date issued: 18 April 2017 Review date: Target
Version: 1718.v1 Ratified by: SCCG COG Date Ratified: 05 April 2017 Name of Originator/Author: Name of Responsible Committee/Individual: IFR SCCG CCPF/ IFR Date issued: 18 April 2017 Review date: Target
Enhanced Service Specification. Childhood seasonal influenza vaccination programme 2018/19
 Enhanced Service Specification Childhood seasonal influenza vaccination programme 2018/19 Contents Childhood seasonal influenza vaccination programme... 1 Contents... 4 1 Introduction... 5 2 Background...
Enhanced Service Specification Childhood seasonal influenza vaccination programme 2018/19 Contents Childhood seasonal influenza vaccination programme... 1 Contents... 4 1 Introduction... 5 2 Background...
Cryopreservation for Infertility Associated with Medical Treatment or Surgery
 Commissioning Policy Cryopreservation for Infertility Associated with Medical Treatment or Surgery August 2018 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible:
Commissioning Policy Cryopreservation for Infertility Associated with Medical Treatment or Surgery August 2018 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible:
Case scenarios: Patient Group Directions
 Putting NICE guidance into practice Case scenarios: Patient Group Directions Implementing the NICE guidance on Patient Group Directions (MPG2) Published: March 2014 [updated March 2017] These case scenarios
Putting NICE guidance into practice Case scenarios: Patient Group Directions Implementing the NICE guidance on Patient Group Directions (MPG2) Published: March 2014 [updated March 2017] These case scenarios
Community alcohol detoxification in primary care
 Community alcohol detoxification in primary care 1. Purpose The purpose of this primary care enhanced service is to improve the health and quality of life of people whose health may be compromised by their
Community alcohol detoxification in primary care 1. Purpose The purpose of this primary care enhanced service is to improve the health and quality of life of people whose health may be compromised by their
PERCUTANEOUS TIBIAL NERVE STIMULATION (PTNS) TREATMENT FOR URINARY INCONTINENCE SECONDARY CARE PRIOR APPROVAL POLICY
 PERCUTANEOUS TIBIAL NERVE STIMULATION (PTNS) TREATMENT FOR URINARY INCONTINENCE SECONDARY CARE PRIOR APPROVAL POLICY Version: Recommendation by: 1617v2a Somerset CCG Clinical Commissioning Policy Forum
PERCUTANEOUS TIBIAL NERVE STIMULATION (PTNS) TREATMENT FOR URINARY INCONTINENCE SECONDARY CARE PRIOR APPROVAL POLICY Version: Recommendation by: 1617v2a Somerset CCG Clinical Commissioning Policy Forum
TRIGGER FINGER CRITERIA BASED ACCESS POLICY
 TRIGGER FINGER CRITERIA BASED ACCESS POLICY Version: Discussion and Recommendation by the Somerset CCG Clinical Commissioning Policy Forum 1617.v1b Date: 16 June 2016 Name of Originator/Author: Name of
TRIGGER FINGER CRITERIA BASED ACCESS POLICY Version: Discussion and Recommendation by the Somerset CCG Clinical Commissioning Policy Forum 1617.v1b Date: 16 June 2016 Name of Originator/Author: Name of
BUNION (AND OTHER PAINFUL TOE CONDITION) SURGICAL TREATMENT POLICY PRIOR APPROVAL
 BUNION (AND OTHER PAINFUL TOE CONDITION) SURGICAL TREATMENT POLICY PRIOR APPROVAL Version: 1718.v3 Recommendation by: Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 12 July 2017
BUNION (AND OTHER PAINFUL TOE CONDITION) SURGICAL TREATMENT POLICY PRIOR APPROVAL Version: 1718.v3 Recommendation by: Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 12 July 2017
Musculoskeletal Surgery and Therapeutic Interventions September 2017
 Commissioning Policy Musculoskeletal Surgery and Therapeutic Interventions September 2017 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible: NHS South
Commissioning Policy Musculoskeletal Surgery and Therapeutic Interventions September 2017 This policy applies to patients for whom the following Clinical Commissioning Groups are responsible: NHS South
Alcohol interventions in secondary and further education
 National Institute for Health and Care Excellence Guideline version (Draft for Consultation) Alcohol interventions in secondary and further education NICE guideline: methods NICE guideline Methods
National Institute for Health and Care Excellence Guideline version (Draft for Consultation) Alcohol interventions in secondary and further education NICE guideline: methods NICE guideline Methods
Enhanced Service Specification. Childhood seasonal influenza vaccination programme 2017/18
 Enhanced Service Specification Childhood seasonal influenza vaccination programme 2017/18 2 Enhanced Service Specification Childhood seasonal influenza vaccination programme Version number: 1 First published:
Enhanced Service Specification Childhood seasonal influenza vaccination programme 2017/18 2 Enhanced Service Specification Childhood seasonal influenza vaccination programme Version number: 1 First published:
GOVERNING BODY REPORT
 GOVERNING BODY REPORT DATE OF MEETING: 20th September 2012 TITLE OF REPORT: KEY MESSAGES: NHS West Cheshire Clinical Commissioning Group has identified heart disease as one of its six strategic clinical
GOVERNING BODY REPORT DATE OF MEETING: 20th September 2012 TITLE OF REPORT: KEY MESSAGES: NHS West Cheshire Clinical Commissioning Group has identified heart disease as one of its six strategic clinical
GOVERNING BOARD. Assisted Conception (IVF): Review of access criteria. Date of Meeting 21 January 2015 Agenda Item No 13. Title
 GOVERNING BOARD Date of Meeting 21 January 2015 Agenda Item No 13 Title Assisted Conception (IVF): Review of access criteria Purpose of Paper The SHIP (Southampton, Hampshire, Isle of Wight and Portsmouth)
GOVERNING BOARD Date of Meeting 21 January 2015 Agenda Item No 13 Title Assisted Conception (IVF): Review of access criteria Purpose of Paper The SHIP (Southampton, Hampshire, Isle of Wight and Portsmouth)
3.3.1 Introduction. Nikolaus B.M. Császár and Christoph Schmitz
 3.3 Shock Wave Therapy 55 Nikolaus B.M. Császár and Christoph Schmitz 3.3 Shock Wave Therapy Effectiveness and safety of extracorporeal shock wave therapy for disorders of the tendons: A systematic review
3.3 Shock Wave Therapy 55 Nikolaus B.M. Császár and Christoph Schmitz 3.3 Shock Wave Therapy Effectiveness and safety of extracorporeal shock wave therapy for disorders of the tendons: A systematic review
Specialised Services Policy: CP23 Vagal Nerve Stimulation
 Specialised Services Policy: CP23 Vagal Nerve Stimulation Document Author: Specialist Services Planning Manager for Neurosciences and Complex Conditions Executive Lead: Director of Planning and Performance
Specialised Services Policy: CP23 Vagal Nerve Stimulation Document Author: Specialist Services Planning Manager for Neurosciences and Complex Conditions Executive Lead: Director of Planning and Performance
Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181)
 Putting NICE guidance into practice Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181) Published: July 2014 This costing report accompanies Lipid modification:
Putting NICE guidance into practice Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181) Published: July 2014 This costing report accompanies Lipid modification:
Annual MRI Breast Screening Criteria Based Access Policy Date Adopted: 21st August 2015 Version:
 Annual MRI Breast Screening Criteria Based Access Policy Date Adopted: 21st August 2015 Version: 1516.2 Document Control Title of document Annual MRI Breast Screening Authors job title(s) IFR Manager Document
Annual MRI Breast Screening Criteria Based Access Policy Date Adopted: 21st August 2015 Version: 1516.2 Document Control Title of document Annual MRI Breast Screening Authors job title(s) IFR Manager Document
Commissioning Policy: Coventry and Rugby CCG (CRCCG)
 Introduction The rationale and scope of this policy is contained within the overarching LPP policy, which sets out CRCCG approach to Low Priority Procedures. This document refers specifically to Tonsillectomy/Adenotonsillectomy
Introduction The rationale and scope of this policy is contained within the overarching LPP policy, which sets out CRCCG approach to Low Priority Procedures. This document refers specifically to Tonsillectomy/Adenotonsillectomy
Cataract Policy. (Referral for Assessment of Surgical Treatment)
 Cataract Policy (Referral for Assessment of Surgical Treatment) MAY 2015 Document Control Title of document Cataract Policy Authors name(s) Authors job title(s) IFR Team Directorate(s) IFR Document status
Cataract Policy (Referral for Assessment of Surgical Treatment) MAY 2015 Document Control Title of document Cataract Policy Authors name(s) Authors job title(s) IFR Team Directorate(s) IFR Document status
CABINET PROCURING A SUBSTANCE MISUSE & COMMUNITY TREATMENT SERVICE IN RUTLAND
 CABINET Report No: 105/2017 PUBLIC REPORT 16 May 2017 PROCURING A SUBSTANCE MISUSE & COMMUNITY TREATMENT SERVICE IN RUTLAND Report of the Director of Public Health Strategic Aim: Safeguarding Key Decision:
CABINET Report No: 105/2017 PUBLIC REPORT 16 May 2017 PROCURING A SUBSTANCE MISUSE & COMMUNITY TREATMENT SERVICE IN RUTLAND Report of the Director of Public Health Strategic Aim: Safeguarding Key Decision:
GOVERNING BODY. Kingston Assisted Conception Guidelines
 GOVERNING BODY LEAD: Dr Naeem Iqbal REPORT AUTHOR: Niran Rehill & Livia Royle, Public Health Royal Borough of Kingston / Kingston CCG ATTACHMENT: AGENDA ITEM: 8 H RECOMMENDATION: The Governing Body is
GOVERNING BODY LEAD: Dr Naeem Iqbal REPORT AUTHOR: Niran Rehill & Livia Royle, Public Health Royal Borough of Kingston / Kingston CCG ATTACHMENT: AGENDA ITEM: 8 H RECOMMENDATION: The Governing Body is
Primary Mental Health Services. Engagement for Redesign 2015
 Primary Mental Health Services Engagement for Redesign 2015 Introduction The three Clinical Commissioning Groups (CCGs) across Worcestershire, NHS Wyre Forest CCG, NHS Redditch and Bromsgrove CCG and NHS
Primary Mental Health Services Engagement for Redesign 2015 Introduction The three Clinical Commissioning Groups (CCGs) across Worcestershire, NHS Wyre Forest CCG, NHS Redditch and Bromsgrove CCG and NHS
Barnsley Youth Justice Plan 2017/18. Introduction
 Barnsley Youth Justice Plan 2017/18 Introduction Barnsley s Youth Justice Service sits within the Local Authority s Targeted Youth Support Service. The governance of the provision has changed in 2016/17.
Barnsley Youth Justice Plan 2017/18 Introduction Barnsley s Youth Justice Service sits within the Local Authority s Targeted Youth Support Service. The governance of the provision has changed in 2016/17.
DEXA SCAN POLICY CRITERIA BASED ACCESS
 DEXA SCAN POLICY Version: 1718.v2 Recommendation by: Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 01 November 2017 Name of Originator/Author: Approved by Responsible Committee/Individual:
DEXA SCAN POLICY Version: 1718.v2 Recommendation by: Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 01 November 2017 Name of Originator/Author: Approved by Responsible Committee/Individual:
Primary Health Networks
 Primary Health Networks Drug and Alcohol Treatment Activity Work Plan 2016-17 to 2018-19 Drug and Alcohol Treatment Budget Gippsland When submitting this Activity Work Plan 2016-2018 to the Department
Primary Health Networks Drug and Alcohol Treatment Activity Work Plan 2016-17 to 2018-19 Drug and Alcohol Treatment Budget Gippsland When submitting this Activity Work Plan 2016-2018 to the Department
Specialised Services Commissioning Policy: CP34 Circumcision for children
 Specialised Services Commissioning Policy: CP34 Circumcision for children March 2019 Version 3.0 Document information Document purpose Document name Author Policy Circumcision for Children Welsh Health
Specialised Services Commissioning Policy: CP34 Circumcision for children March 2019 Version 3.0 Document information Document purpose Document name Author Policy Circumcision for Children Welsh Health
Royal College of Psychiatrists in Wales Consultation Response
 Royal College of Psychiatrists in Wales Consultation Response RESPONSE OF: RESPONSE TO: THE ROYAL COLLEGE OF PSYCHIATRISTS in WALES The Autism Bill Date: 20 November 2017 The Royal College of Psychiatrists
Royal College of Psychiatrists in Wales Consultation Response RESPONSE OF: RESPONSE TO: THE ROYAL COLLEGE OF PSYCHIATRISTS in WALES The Autism Bill Date: 20 November 2017 The Royal College of Psychiatrists
Clinical Commissioning Policy: Deep Brain Stimulation for Refractory Epilepsy
 Clinical Commissioning Policy: Deep Brain Stimulation for Refractory Epilepsy Reference: NHS England xxx/x/x 1 Clinical Commissioning Policy: Deep Brain Stimulation for Refractory Epilepsy First published:
Clinical Commissioning Policy: Deep Brain Stimulation for Refractory Epilepsy Reference: NHS England xxx/x/x 1 Clinical Commissioning Policy: Deep Brain Stimulation for Refractory Epilepsy First published:
GOVERNING BODY MEETING in Public 22 February 2017 Agenda Item 3.4
 GOVERNING BODY MEETING in Public 22 February 2017 Paper Title Purpose of paper Redesign of Services for Frail Older People in Eastern Cheshire To seek approval from Governing Body for the redesign of services
GOVERNING BODY MEETING in Public 22 February 2017 Paper Title Purpose of paper Redesign of Services for Frail Older People in Eastern Cheshire To seek approval from Governing Body for the redesign of services
GROMMET INSERTION IN ADULTS WITH OTITIS MEDIA WITH EFFUSION (OME) SECONDARY CARE PRIOR APPROVAL POLICY
 SECONDARY CARE PRIOR APPROVAL POLICY Version: Recommendation by: 1617.v3a Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 13 July 2016 Name of Originator/Author: Approved by Responsible
SECONDARY CARE PRIOR APPROVAL POLICY Version: Recommendation by: 1617.v3a Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 13 July 2016 Name of Originator/Author: Approved by Responsible
Specialised Services Policy Position PP151
 Specialised Services Policy Position PP151 Complex Devices: Implantable Cardioverter Defibrillators and Cardiac Resynchronisation Therapy for arrhythmias and heart failure January 2019 Version 1.0 Document
Specialised Services Policy Position PP151 Complex Devices: Implantable Cardioverter Defibrillators and Cardiac Resynchronisation Therapy for arrhythmias and heart failure January 2019 Version 1.0 Document
Full Equality Impact Assessment (EIA)
 Full Equality Impact Assessment (EIA) Service(s) under review: In-vitro fertilisation (IVF) This paper outlines the post engagement Equality Impact Assessment of BHR CCGs proposals on In-vitro fertilisation
Full Equality Impact Assessment (EIA) Service(s) under review: In-vitro fertilisation (IVF) This paper outlines the post engagement Equality Impact Assessment of BHR CCGs proposals on In-vitro fertilisation
EQUALITY IMPACT ASSESSMENT. Business Division/Directorate: Name of Service/Title of Policy or Strategy, Name of Event:
 EQUALITY IMPACT ASSESSMENT Business Division/Directorate: Service: Adult Community Policy: Name of Service/Title of Policy or Strategy, Name of Event: Event: South Yorkshire Service for Deaf People with
EQUALITY IMPACT ASSESSMENT Business Division/Directorate: Service: Adult Community Policy: Name of Service/Title of Policy or Strategy, Name of Event: Event: South Yorkshire Service for Deaf People with
Standard Operating Procedure: Early Intervention in Psychosis Access Times
 Corporate Standard Operating Procedure: Early Intervention in Psychosis Access Times Document Control Summary Status: New Version: V1.0 Date: Author/Owner: Rob Abell, Senior Performance Development Manager
Corporate Standard Operating Procedure: Early Intervention in Psychosis Access Times Document Control Summary Status: New Version: V1.0 Date: Author/Owner: Rob Abell, Senior Performance Development Manager
Specialised Services Policy Position PP104
 Specialised Services Policy Position PP104 Personalised External Aortic Root Support (PEARS) for surgical management of enlarged aortic root (adults) March 2019 Version 1.0 Document information Document
Specialised Services Policy Position PP104 Personalised External Aortic Root Support (PEARS) for surgical management of enlarged aortic root (adults) March 2019 Version 1.0 Document information Document
CARPAL TUNNEL SURGERY CRITERIA BASED ACCESS POLICY
 CARPAL TUNNEL SURGERY CRITERIA BASED ACCESS POLICY Version: 1516.2c Recommendation by: Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 11 May 2016 Name of Originator/Author: Approved
CARPAL TUNNEL SURGERY CRITERIA BASED ACCESS POLICY Version: 1516.2c Recommendation by: Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 11 May 2016 Name of Originator/Author: Approved
COMMISSIONING POLICY
 COMMISSIONING POLICY Morbid Obesity in Adults: Secondary Care Referral Pathway Specialty: General Surgery Diabetic Medicine Respiratory Medicine Version: 2.3 Ratified by (name of Committee): Clinical Senate
COMMISSIONING POLICY Morbid Obesity in Adults: Secondary Care Referral Pathway Specialty: General Surgery Diabetic Medicine Respiratory Medicine Version: 2.3 Ratified by (name of Committee): Clinical Senate
Plantar Fasciitis and Heel Pain
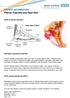 PATIENT INFORMATION Plantar Fasciitis and Heel Pain What is plantar fasciitis? Heel pain and plantar fasciitis Plantar fasciitis causes pain under your heel. It usually goes in time. Treatment may speed
PATIENT INFORMATION Plantar Fasciitis and Heel Pain What is plantar fasciitis? Heel pain and plantar fasciitis Plantar fasciitis causes pain under your heel. It usually goes in time. Treatment may speed
LIPOSUCTION (COSMETIC) INDIVIDUAL FUNDING REQUEST POLICY
 LIPOSUCTION (COSMETIC) INDIVIDUAL FUNDING REQUEST POLICY Version: Recommendation by: 1617.v2a Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 13 July 2016 Name of Originator/Author:
LIPOSUCTION (COSMETIC) INDIVIDUAL FUNDING REQUEST POLICY Version: Recommendation by: 1617.v2a Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 13 July 2016 Name of Originator/Author:
Specialised Services Policy: CP 44 Body Contouring
 Specialised Services Policy: CP 44 Body Contouring Document Author: Specialised Planner Executive Lead: Director of Planning Approved by: Management Group Issue Date: 11 July 2013 Review Date: 01 July
Specialised Services Policy: CP 44 Body Contouring Document Author: Specialised Planner Executive Lead: Director of Planning Approved by: Management Group Issue Date: 11 July 2013 Review Date: 01 July
Meeting of Bristol Clinical Commissioning Group Governing Body
 Meeting of Bristol Clinical Commissioning Group Governing Body To be held on Tuesday 24 February 2015 commencing at 13:30 at the Vassall Centre, Gill Avenue, Bristol, BS16 2QQ Title: OFSTED Report Agenda
Meeting of Bristol Clinical Commissioning Group Governing Body To be held on Tuesday 24 February 2015 commencing at 13:30 at the Vassall Centre, Gill Avenue, Bristol, BS16 2QQ Title: OFSTED Report Agenda
PRIMARY CARE CO-COMMISSIONING COMMITTEE 18 March 2016
 Part 1 Part 2 PRIMARY CARE CO-COMMISSIONING COMMITTEE 18 March 2016 Title of Report Supporting deaf patients to access primary care services Purpose of the Report The report is to provide the co-commissioning
Part 1 Part 2 PRIMARY CARE CO-COMMISSIONING COMMITTEE 18 March 2016 Title of Report Supporting deaf patients to access primary care services Purpose of the Report The report is to provide the co-commissioning
BOTULINUM TOXIN (BOTOX) POLICY HYPERHIDROSIS - PRIOR APPROVAL
 BOTULINUM TOXIN (BOTOX) POLICY HYPERHIDROSIS - PRIOR APPROVAL Version: Ratified by: 1617.v2c SCCG COG Date Ratified: 11 May 2016 Name of Originator/Author: Name of Responsible Committee/Individual: IFR
BOTULINUM TOXIN (BOTOX) POLICY HYPERHIDROSIS - PRIOR APPROVAL Version: Ratified by: 1617.v2c SCCG COG Date Ratified: 11 May 2016 Name of Originator/Author: Name of Responsible Committee/Individual: IFR
Revised Standards. S 1a: The service routinely collects data on age, gender and ethnicity for each person referred for psychological therapy.
 Revised Standards S 1a: The service routinely collects data on age, gender and ethnicity for each person referred for psychological therapy. S1b: People starting treatment with psychological therapy are
Revised Standards S 1a: The service routinely collects data on age, gender and ethnicity for each person referred for psychological therapy. S1b: People starting treatment with psychological therapy are
Barnet Scrutiny Committee report 13 th October Barnet Sexual Health Strategy Dr Andrew Howe, Director of Public Health
 Barnet Scrutiny Committee report 13 th October 2015 Title Report of Wards Status Urgent Key Enclosures Officer Contact Details Barnet Sexual Health Strategy 2015-2020 Dr Andrew Howe, Director of Public
Barnet Scrutiny Committee report 13 th October 2015 Title Report of Wards Status Urgent Key Enclosures Officer Contact Details Barnet Sexual Health Strategy 2015-2020 Dr Andrew Howe, Director of Public
CONTINUOUS GLUCOSE MONITORING POLICY INDIVIDUAL FUNDING
 CONTINUOUS GLUCOSE MONITORING POLICY INDIVIDUAL FUNDING Version: 1718.v1 Recommendation by: Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 12 July 2017 Name of Originator/Author:
CONTINUOUS GLUCOSE MONITORING POLICY INDIVIDUAL FUNDING Version: 1718.v1 Recommendation by: Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 12 July 2017 Name of Originator/Author:
ifuse implant system for treating chronic sacroiliac joint pain
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology guidance SCOPE ifuse implant system for treating chronic sacroiliac joint pain 1 Technology 1.1 Description of the technology The ifuse
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology guidance SCOPE ifuse implant system for treating chronic sacroiliac joint pain 1 Technology 1.1 Description of the technology The ifuse
Specialised Services Policy: CP91 Extracorporeal Photophoresis (ECP) for the Treatment of Chronic Graft versus Host Disease in Adults
 Specialised Services Policy: Extracorporeal Photophoresis (ECP) for the Treatment of Chronic Graft versus Host Disease in Adults Document Author: Specialised Planner Executive Lead: Director of Planning
Specialised Services Policy: Extracorporeal Photophoresis (ECP) for the Treatment of Chronic Graft versus Host Disease in Adults Document Author: Specialised Planner Executive Lead: Director of Planning
Translation and Interpretation Policy
 Translation and Interpretation Policy Version 1 Ratified By NHS West Cheshire Clinical Commissioning Group Governing Body Date Ratified 16 th November 2017 Author(s) Jonathan Taylor Responsible Committee
Translation and Interpretation Policy Version 1 Ratified By NHS West Cheshire Clinical Commissioning Group Governing Body Date Ratified 16 th November 2017 Author(s) Jonathan Taylor Responsible Committee
Commissioning Policy Individual Funding Request
 Commissioning Policy Individual Funding Request Penile Conditions - Surgical Opinion and Treatment Policy including Circumcision in all male patients over the age of 18 years Prior Approval Policy Date
Commissioning Policy Individual Funding Request Penile Conditions - Surgical Opinion and Treatment Policy including Circumcision in all male patients over the age of 18 years Prior Approval Policy Date
No Smoking Policy. No Smoking Policy Page: Page 1 of 13. Author: Strategic HR Manager Version: 1.3. Date of Approval: 7 October 2015 Status: Final
 No Smoking Policy No Smoking Policy Page: Page 1 of 13 Date of Approval: 7 October 2015 Status: Final Recommended by Approved by HR Senior Management Team Executive Management Team Approval date 7 th October
No Smoking Policy No Smoking Policy Page: Page 1 of 13 Date of Approval: 7 October 2015 Status: Final Recommended by Approved by HR Senior Management Team Executive Management Team Approval date 7 th October
2) Are there opportunities to better promote equality of opportunity for people within the Section 75 equality categories?
 Equality, Good Relations and Human Rights SCREENING The Health and Social Care Board is required to consider the likely equality implications of any policies or decisions. In particular it is asked to
Equality, Good Relations and Human Rights SCREENING The Health and Social Care Board is required to consider the likely equality implications of any policies or decisions. In particular it is asked to
Commissioning Policy TBC 2018
 Commissioning Policy Exogen Ultrasound Bone Healing System for Bone Fractures with Non-Union TBC 2018 This commissioning policy has been endorsed by and applies to patients within: NHS North Staffordshire
Commissioning Policy Exogen Ultrasound Bone Healing System for Bone Fractures with Non-Union TBC 2018 This commissioning policy has been endorsed by and applies to patients within: NHS North Staffordshire
Worcestershire Dementia Strategy
 Worcestershire Dementia Strategy An Easy Read Summary Introduction This is a plan about how we will support people with dementia, their families and carers in Worcestershire. This is called the Worcestershire
Worcestershire Dementia Strategy An Easy Read Summary Introduction This is a plan about how we will support people with dementia, their families and carers in Worcestershire. This is called the Worcestershire
ABDOMINOPLASTY/APRONECTOMY INDIVIDUAL FUNDING REQUEST POLICY
 ABDOMINOPLASTY/APRONECTOMY INDIVIDUAL FUNDING REQUEST POLICY Version: Recommendation by: 1819.v1.3 Date Ratified: June 2018 Name of Originator/Author: Approved by Responsible Committee/Individual: Somerset
ABDOMINOPLASTY/APRONECTOMY INDIVIDUAL FUNDING REQUEST POLICY Version: Recommendation by: 1819.v1.3 Date Ratified: June 2018 Name of Originator/Author: Approved by Responsible Committee/Individual: Somerset
Low back pain and sciatica in over 16s NICE quality standard
 March 2017 Low back pain and sciatica in over 16s NICE quality standard Draft for consultation This quality standard covers the assessment and management of non-specific low back pain and sciatica in young
March 2017 Low back pain and sciatica in over 16s NICE quality standard Draft for consultation This quality standard covers the assessment and management of non-specific low back pain and sciatica in young
Working Better Together on Safeguarding: Annual Reports of the Bradford Safeguarding Children Board (BSCB) and the Safeguarding Adults Board (SAB)
 Report of the Director of Health and Wellbeing and the Director of Children s Services to the meeting of Bradford and Airedale Health and Wellbeing Board to be held on 29 th November 2016. Subject: O Working
Report of the Director of Health and Wellbeing and the Director of Children s Services to the meeting of Bradford and Airedale Health and Wellbeing Board to be held on 29 th November 2016. Subject: O Working
St Helens CCG NHS Funded Treatment for Subfertility Policy 2015/16
 St Helens CCG NHS Funded Treatment for Subfertility Policy 2015/16 1 Standard Operating Procedure St Helens CCG NHS Funded Treatment for Sub Fertility Policy Version 1 Implementation Date May 2015 Review
St Helens CCG NHS Funded Treatment for Subfertility Policy 2015/16 1 Standard Operating Procedure St Helens CCG NHS Funded Treatment for Sub Fertility Policy Version 1 Implementation Date May 2015 Review
This specification should be read in conjunction with the Rotherham Hospice overall contract and schedules.
 Care Pathway/Service Commissioner Lead Provider Lead Period Applicability of Module E (Acute Services Requirements) Rotherham Palliative Medicine Service Gail Palmer Fiona Hendry 1 April 2011 31 March
Care Pathway/Service Commissioner Lead Provider Lead Period Applicability of Module E (Acute Services Requirements) Rotherham Palliative Medicine Service Gail Palmer Fiona Hendry 1 April 2011 31 March
Health Scrutiny Panel 6 February 2014
 Agenda Item No: 5 Health Scrutiny Panel 6 February 2014 Report title Infertility Policy Review Wolverhampton Clinical Commissioning Group Cabinet member with lead responsibility Wards affected Accountable
Agenda Item No: 5 Health Scrutiny Panel 6 February 2014 Report title Infertility Policy Review Wolverhampton Clinical Commissioning Group Cabinet member with lead responsibility Wards affected Accountable
Tier 3 and 4 healthy weight and obesity services in Kent
 Tier 3 and 4 healthy weight and obesity services in Kent Model user guide September 2016 1 Background Tier 3 healthy weight services for Kent residents are currently commissioned by Kent County Council
Tier 3 and 4 healthy weight and obesity services in Kent Model user guide September 2016 1 Background Tier 3 healthy weight services for Kent residents are currently commissioned by Kent County Council
Delivering first class mental health care services in south west London Committed to care, creating communities
 Estates Modernisation Programme Delivering first class mental health care services in south west London Committed to care, creating communities Contents Introduction to the Estates Modernisation Programme
Estates Modernisation Programme Delivering first class mental health care services in south west London Committed to care, creating communities Contents Introduction to the Estates Modernisation Programme
Technology appraisal guidance Published: 21 February 2018 nice.org.uk/guidance/ta507
 Sofosbuvir velpatasvir voxilaprevir for treating chronic hepatitis C Technology appraisal guidance Published: 21 February 2018 nice.org.uk/guidance/ta507 NICE 2018. All rights reserved. Subject to Notice
Sofosbuvir velpatasvir voxilaprevir for treating chronic hepatitis C Technology appraisal guidance Published: 21 February 2018 nice.org.uk/guidance/ta507 NICE 2018. All rights reserved. Subject to Notice
PRIMARY CARE CO-COMMISSIONING COMMITTEE 8 SEPTEMBER 2015
 Part 1 Part 2 PRIMARY CARE CO-COMMISSIONING COMMITTEE 8 SEPTEMBER 2015 Title of Report Trafford Palliative care Quality Premium Scheme 2015/16 Purpose of the Report The purpose of the report is to detail
Part 1 Part 2 PRIMARY CARE CO-COMMISSIONING COMMITTEE 8 SEPTEMBER 2015 Title of Report Trafford Palliative care Quality Premium Scheme 2015/16 Purpose of the Report The purpose of the report is to detail
NHS public health functions agreement Service specification No.11 Human papillomavirus (HPV) programme
 NHS public health functions agreement 2018-19 Service specification No.11 Human papillomavirus (HPV) programme 1 NHS public health functions agreement 2018-19 Service specification No.11 Human papillomavirus
NHS public health functions agreement 2018-19 Service specification No.11 Human papillomavirus (HPV) programme 1 NHS public health functions agreement 2018-19 Service specification No.11 Human papillomavirus
Shoulder Impingement Surgery for Subacromial Pain Policy CRITERIA BASED ACCESS
 Shoulder Impingement Surgery for Subacromial Pain Policy CRITERIA BASED ACCESS Version: 1718.v2 Recommendation by: Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 12 July 2017 Name
Shoulder Impingement Surgery for Subacromial Pain Policy CRITERIA BASED ACCESS Version: 1718.v2 Recommendation by: Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 12 July 2017 Name
Commissioning Policy Individual Funding Request
 Commissioning Policy Individual Funding Request Vasectomy Policy Criteria Based Access Policy Date Adopted: 6 th February 2017 Version: 1617.1 Individual Funding Request Team - A partnership between Bristol,
Commissioning Policy Individual Funding Request Vasectomy Policy Criteria Based Access Policy Date Adopted: 6 th February 2017 Version: 1617.1 Individual Funding Request Team - A partnership between Bristol,
Dorset Health Scrutiny Committee
 Dorset Health Scrutiny Committee Date of Meeting 15 June 2018 Officer/Author Diane Bardwell, Dementia Services Review Project Manager, NHS Dorset Clinical Commissioning Group Subject of Report Dementia
Dorset Health Scrutiny Committee Date of Meeting 15 June 2018 Officer/Author Diane Bardwell, Dementia Services Review Project Manager, NHS Dorset Clinical Commissioning Group Subject of Report Dementia
Management of AIDS/HIV Infected Healthcare Workers Policy
 Management of AIDS/HIV Infected Healthcare Workers Policy DOCUMENT CONTROL: Version: 4 Ratified by: Corporate Policy Panel Date ratified: 20 July 2017 Name of originator/author: HR Manager Name of responsible
Management of AIDS/HIV Infected Healthcare Workers Policy DOCUMENT CONTROL: Version: 4 Ratified by: Corporate Policy Panel Date ratified: 20 July 2017 Name of originator/author: HR Manager Name of responsible
GROMMET INSERTION 18 YEARS AND UNDER PERSISTENCE OF BILATERAL OTITIS MEDIA WITH EFFUSION SECONDARY CARE PRIOR APPROVAL POLICY
 SECONDARY CARE PRIOR APPROVAL POLICY Version: 1718.v3 Ratified by: Date Ratified: 05 April 2017 Name of Originator/Author: Name of Responsible Committee/Individual: Somerset CCG Clinical Commissioning
SECONDARY CARE PRIOR APPROVAL POLICY Version: 1718.v3 Ratified by: Date Ratified: 05 April 2017 Name of Originator/Author: Name of Responsible Committee/Individual: Somerset CCG Clinical Commissioning
What can NHS Health Scotland do to reduce health inequalities? Questions for applying the Health Inequalities Action Framework
 What can NHS Health Scotland do to reduce health inequalities? Questions for applying the Health Inequalities Action Framework Introduction Definition: health inequalities are the differences in health
What can NHS Health Scotland do to reduce health inequalities? Questions for applying the Health Inequalities Action Framework Introduction Definition: health inequalities are the differences in health
abcdefghijklmnopqrstu
 Chief Medical Officer Directorate Chief Medical Officer and Secretariat Division abcdefghijklmnopqrstu T: 0131-244 2399 F: 0131-244 2989 E: sandra.falconer@scotland.gsi.gov.uk NHS Board Medical and Nursing
Chief Medical Officer Directorate Chief Medical Officer and Secretariat Division abcdefghijklmnopqrstu T: 0131-244 2399 F: 0131-244 2989 E: sandra.falconer@scotland.gsi.gov.uk NHS Board Medical and Nursing
Commissioning Policy Individual Funding Request
 Commissioning Policy Individual Funding Request Hip Replacement Surgery including referral for Surgical Assessment of Osteoarthritis Criteria Based Access Policy Date Adopted: 1 st June 2016 Version: 1617.1.01
Commissioning Policy Individual Funding Request Hip Replacement Surgery including referral for Surgical Assessment of Osteoarthritis Criteria Based Access Policy Date Adopted: 1 st June 2016 Version: 1617.1.01
Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital
 Please Note: This policy is currently under review and is still fit for purpose. Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital This procedural document supersedes
Please Note: This policy is currently under review and is still fit for purpose. Venous Thromboembolism (VTE) Prevention and Treatment of VTE in Patients Admitted to Hospital This procedural document supersedes
SCHEDULE 2 THE SERVICES. A. Service Specifications
 SCHEDULE 2 THE SERVICES A. Service Specifications Service Specification No. 04/MSKT/0013 Service PAN DORSET FRACTURE LIAISON SERVICE Commissioner Lead CCP for Musculoskeletal & Trauma Provider Lead Deputy
SCHEDULE 2 THE SERVICES A. Service Specifications Service Specification No. 04/MSKT/0013 Service PAN DORSET FRACTURE LIAISON SERVICE Commissioner Lead CCP for Musculoskeletal & Trauma Provider Lead Deputy
