NHS ENGLAND BOARD PAPER
|
|
|
- Sylvia Alexander
- 5 years ago
- Views:
Transcription
1 NHS ENGLAND BOARD PAPER Paper: PB /05 Title: Items which should not routinely be prescribed in primary care Lead Director: Professor Sir Bruce Keogh, Medical Director Paul Baumann, Chief Financial Officer Purpose of Paper: The Next Steps document, published on 31 March 2017, included as part of the NHS 10 Point Efficiency Plan a commitment to review the appropriateness of aspects of NHS-prescribed products of low clinical value and/or available to the public over the counter (OTC). The NHS England Commissioning Committee has approved a set of proposals which, subject to support from the NHS England Board, will now be consulted on. These propose initially limiting prescribing of 18 low value products costing 141million pa and potentially medicines that are available over the counter for generally time-limited/short term conditions suitable for self-care, costing a further million. In addition, the Department of Health (DH) is consulting on limits on 26million of expenditure on Gluten Free foods. Our consultation will also seek views on other opportunities for prescribing efficiency. The Board is invited to: Approve formal public consultation on products which are considered to be relatively ineffective, unnecessary, inappropriate or unsafe for prescription on the NHS. Page 1 of 9
2 Items which should not routinely be prescribed in primary care A. Background 1. Last year, 1.1billion prescription items were dispensed in primary care at a cost of 9.2billion. Over 90% of prescriptions currently issued are exempt from a prescription charge. It is vital that the NHS achieves the greatest value from the money that it spends, and we know that across England there is significant variation on what is being prescribed and to whom. Often patients are receiving medicines which have been proven to be ineffective or for which there are other more effective and/or cheaper alternatives, and products which it may no longer be appropriate to prescribe on the NHS. 2. NHS England has partnered with NHS Clinical Commissioners (NHSCC) to support Clinical Commissioning Groups (CCGs) in ensuring that they can use their prescribing resources effectively and get the best value from the medicines that their local population uses. CCGs asked for a nationally coordinated approach to the development of commissioning guidance developed with and by CCGs. The aim is that this will lead to more equitable and consistent local decisions 3. In response to calls from GPs and CCGs who were having to take individual decisions about their local formularies, NHSCC surveyed their members during February and March 2017 to assess views amongst CCGs as to whether a range of medicines and other products should be available for prescription on the NHS. 4. NHSCC engaged with NHS England to secure support for their work, which aligns with the objectives of the Medicines Value Programme that we are establishing to pursue savings from the rapidly rising drugs bill whilst improving outcomes for patients. Any savings from implementing the proposals will be reinvested in improving patient care. 5. Together, NHS England and NHSCC established a clinical working group, chaired by representatives of these two organisations, with membership including GPs and pharmacists, CCGs, Royal College of General Practitioners, National Institute for Health and Care Excellence (NICE), Department of Health, the Royal Pharmaceutical Society and others. 6. Initial work has focused on developing guidelines for a list of eighteen products which are relatively ineffective, unnecessary, inappropriate or unsafe for prescription on the NHS. The Department of Health is consulting separately on access to Gluten Free foods. 7. Consideration has also been given to a wider list of medicines which, in addition to being available on prescription, are available over the counter and some of those are either clinically ineffective or used to treat short term conditions which are suitable for self-care. 8. The NHS England Commissioning Committee, on 28 June 2017, received and supported proposals to limit use of medicine in these categories, and agreed that a public consultation should be launched, subject to approval by the full Board. Page 2 of 9
3 9. The commissioning guidance upon which we are consulting will be addressed to CCGs to support them to fulfil their duties around appropriate use of resources. This will need to be taken into account by the CCGs in adopting or amending their own local guidance to their clinicians in primary care. Those clinicians will be expected to have regard to the local and national guidance, but retain their clinical discretion in treating individual patients. 10. It is proposed that the final guidance will be statutory guidance issued under S14ZG of the NHS Act B. Proposed action on medicines which are relatively ineffective, unnecessary, inappropriate or unsafe for prescription on the NHS (initial work: eighteen products) 11. Guidelines have been developed for eighteen products which are relatively ineffective, unnecessary, inappropriate or unsafe for prescription on the NHS. 12. These eighteen products comprise a list of ten products originally compiled by NHSCC, working in conjunction with their member CCGs. To it, we have added a further eight items identified following further discussion within the NHSE/NHSCC clinical working group (denoted by * in the tables below). 13. The eighteen products, which cost the NHS an estimated 141million a year, have been included on the list for at least one of three reasons: Items of low clinical effectiveness, where there is a lack of robust evidence of clinical effectiveness or there are significant safety concerns; Items which are clinically effective but where more cost effective products are available, including some products that have been subject to excessive price inflation; and/or Items which are clinically effective but due to the nature of the product are deemed a low priority for NHS funding. 14. Having identified the eighteen items, the clinical working group has met on three occasions to review each product against the following criteria: o Legal Status i.e. is it prescription only, or is it available over the counter in pharmacies and/or any retail outlet? o Indication i.e. what condition is it used to treat? o Background i.e. a general narrative on the drug incl. pack size, tablet size, whether administered orally etc. o Patent Protection i.e. is the drug still subject to a patent? o Efficacy i.e. is it clinically effective? o Safety i.e. is the drug safe? o Alternative treatments and exceptionality for individuals i.e. do alternatives exist and if so, who would they be used for? o Equalities and Health Inequalities i.e. are there groups of people who would be disproportionately affected? o Financial implications, comprising: Commissioning/funding pathway i.e. how does the NHS pay for the drug? Medicine Cost i.e. how much does the drug cost per item? Page 3 of 9
4 Healthcare Resource Utilisation i.e. what NHS resources would be required to implement a change? Annual Spend i.e. what is the annual spend of the NHS on this item? o Unintended consequences 15. The list of eighteen products is as outlined below. The specific recommendations attached to each, and the rationale for the recommendations, are as outlined in the accompanying consultation document. (i) Items of low clinical effectiveness, where there is a lack of robust evidence of clinical effectiveness or there are significant safety concerns ANNUAL COST ESTIMATED AT 49MILLION Item Co-proxamol Omega 3 Fatty Acid Compounds Lidocaine Plasters Rubefacients* Dosulepin* Glucosamine and Chondroiton* Lutein and antioxidants* Oxycodone and Naloxone* Homeopathy items (see Appendix A) Herbal medicines (ii) Items which are clinically effective but where more cost-effective items are available (this includes items that have been subject to excessive price inflation) ANNUAL COST ESTIMATED AT 89MILLION Item Liothyronine Doxazosin Modified Release (MR) Perindopril Arginine* Fentanyl Immediate Release Tadalafil Once Daily Trimipramine* Paracetamol and Tramadol Combination product* (iii) Items which are clinically effective but, due to the nature of the item, are deemed a low priority for NHS funding Item Some travel vaccines already not permitted on the NHS (see note below) 16. Please note that for Travel Vaccines, this only applies to seven vaccines which are not currently available on the NHS for the purposes of travel but are sometimes prescribed and administered in error due to confusion over vaccine eligibility. Public Health England has agreed to undertake a joint assessment of a further four travel Page 4 of 9
5 vaccines which are eligible for prescription currently, with a view to whether they should be removed from NHS availability without unintended public health consequences. At present, there is considerable confusion at prescriber level as to what is or is not allowed to be prescribed at NHS expense in this area. 17. Based on a review of the criteria in paragraph 14, NHS England proposes to make one or more of the following recommendations to CCGs for each product: Advise CCGs that prescribers should not initiate [item] in primary care for any new patient; Advise CCGs to support prescribers in de-prescribing [item] for all patients and ensure the availability of relevant services to facilitate this change; Advise CCGs that if, in exceptional circumstances, there is a clinical need for [item] to be prescribed in primary care, this should be undertaken in a cooperation arrangement with a multi-disciplinary team and/or other healthcare professional; Advise CCGs that all prescribing for [item] should be carried out by a specialist; and Advise CCGs that [item] should not be routinely prescribed in primary care but may be prescribed in specific circumstances. 18. Subject to NHS England Board approval, we intend to put these recommendations to national consultation with CCGs, patients, clinicians, professional and other stakeholder bodies, from July to October This consultation is intended to provide a consistent, national framework in the context of which, local CCGs will be able to decide to implement the national clinical commissioning guidance, with due regard to both local circumstances and their own impact assessments. C. Action on over the counter medicines 19. As part of this review of medicines which could be considered to be of a low clinical value, NHS Clinical Commissioners and NHS England have identified a number of products and conditions which fall into wider categories that need to be considered. The NHS in England spends approximately 645million on medicines which can otherwise be purchased over the counter from a pharmacy and/or other outlets such as petrol stations or convenience stores. 20. These include products that: Can be purchased over the counter, and sometimes at a lower cost than that that would be incurred by the NHS; Treat a condition that is considered to be self-limiting and so does not need treatment as it will heal/be cured of its own accord; and/or Treat a condition which lends itself to self-care, ie that the person suffering does not normally need to seek medical care and/or treatment for the condition. 21. These conditions include the following, which in most cases are minor and/or selflimiting conditions: Diarrhoea Constipation Acute Pain Cold sores Teething Nappy rash Page 5 of 9
6 Athlete s foot Fever Oral and vaginal thrush Head lice Insect bites and stings Conjunctivitis Contact dermatitis Sore throat Headache Indigestion and heartburn (Dyspepsia) Mouth ulcers Haemorrhoids Ear wax Warts and verrucae Soft tissue injury/musculoskeletal joint injury Viral upper respiratory tract infections Scabies Ring worm Mild acne Minor burns and scalds 22. Approximately 3200 different products, which can be purchased over the counter, are prescribed on the NHS for the above conditions. Examples include: Painkillers (analgesics) and medicines for fever (antipyretics), such as paracetamol and ibuprofen Antifungal creams Nasal sprays Coughs and cold remedies Sunscreens Laxatives Lubricating eye drops Eczema creams and ointments Antiviral creams Ear wax removal liquid 23. These product categories include: Medicines which are relatively clinically ineffective; Medicines which are clinically effective but suitable for self-care and used to treat acute conditions; (i) Medicines which are relatively clinically ineffective 24. Some medicines which are available both on prescription and OTC are recognised as being relatively clinically ineffective. There are likely to be a significant number of individual products within the 3,200 which fit within this category. Our consultation document will consult on the criteria to be used in identifying which of the 3,200 products should be recommended as generally not prescribed, and give respondents the opportunity to comment on any individual products. (ii) Medicines which are clinically effective but suitable for self-care and used to treat generally short term/time-limited conditions; 25. A broader list of medicines which are available both on prescription and OTC are regarded as being clinically effective but used to treat conditions which are suitable for self-care and are episodic and which do not require ongoing or long term treatment. We have estimated NHS expenditure of up to 100m p.a. on those prescribing instances where a medicine has only been prescribed once within a six month period Page 6 of 9
7 for an individual patient. Within that, over 50m pa is accounted for by spending on medicines used to treat minor and/or self-limiting conditions. By self-limiting, we mean conditions which without treatment to alleviate symptoms, would heal of their own accord, for example the common cold. Whilst many conditions suitable for self-care are self-limiting, some are not. Some examples of self-care conditions which are not self-limiting and which would not resolve without treatment include hayfever (antihistamine used to treat) or vitamin D insufficiency (vitamin D used to treat). The draft consultation document consults on restricting in this category of prescribing too. 26. It also seeks views on the appropriateness of prescribing in the far bigger category of 545million pa, which it is estimated is spent on two or more prescriptions for the same condition in a six month period that could have been purchased over the counter. 27. The draft consultation document is attached as an Annex to this Board paper. Recommendations 28. The Board is asked to: Approve formal public consultation on the proposals outlined in this paper. Page 7 of 9
8 Homeopathy APPENDIX A 1. NHS England s view is that, at best, homeopathy is a placebo and a misuse of scarce NHS funds which could better be devoted to treatments that work. 2. Data on the residual use and cost of homeopathy on the NHS are hard to come by. A recent Freedom of Information request by a third party suggested that at least 578,000 has been spent on prescribed homeopathy over the past five years, with the total cost being higher than that when the cost of consultations was factored in. 3. As well as primary care prescribing, there are two homeopathic hospitals affiliated to NHS Trusts in Bristol and at University College London Hospitals (UCLH). In both cases, CCGs have sought to restrict provision. 4. Bristol CCG is the lead commissioner for homeopathy for 13 CCGs, and University Hospitals Bristol is the lead secondary care provider (but subcontracts the service). Homeopathy referrals are now subject to Individual Prior Approval, which have reduced volumes significantly but not completely. 5. A recent NHS England London review sought clarity on commissioning of homeopathy services in the region. This found that no CCG commissions homeopathic treatments; however the Royal London Hospital for Integrated Medicine (RLHIM) may be providing homeopathy as part of a wider package of treatment. CCGs commission a range of treatment pathways from UCLH provided from the RLHIM site. 6. There are a number of complexities that limit UCLH s ability to identify the total system cost of providing homeopathy: RLHIM provides an outpatient service with a standard outpatient tariff that covers a number of different treatments and therapies, one of which is homeopathy; In the majority of cases, homeopathy is provided as part of a wider package of treatment rather than in isolation; and Currently this means UCLH are unable to accurately say how many patients have received homeopathy alone. 7. In addition to consulting on initiatives in primary care prescribing of homeopathy, it is therefore also proposed that we explore including a ban on reimbursing homeopathic treatments in the NHS Standard Contract. Page 8 of 9
9 APPENDIX B Gluten Free Foods 1. Gluten Free (GF) foods, costing 26 million a year, are subject to a separate consultation which was run by the Department of Health, concluding in June DH has still to respond to this consultation. 2. Staple GF foods, e.g. bread, flour, pasta, oats etc, are available on prescription to patients diagnosed with gluten sensitivity enteropathies. A limited number of nonstaple foods are also available, including crackers, biscuits and pizza bases. 3. Unlike other foodstuffs, gluten containing products are not necessary for a healthy diet and patients with gluten sensitivity can safely exclude it from their diet and still eat healthily without purchasing special foods. Patients can safely eat meat, fish, vegetables, fruit, rice and most dairy products, and there are a wide variety of products now on the market, eg rice cakes, crackers etc to allow patients to complement their GF diets safely and obtain all their nutritional requirements. 4. GF foods were made available on prescription in the late 1960s when the availability of such foods on general sale was extremely limited. GF foods are now readily available in large supermarkets and therefore the ability of patients to obtain these foods without a prescription has greatly increased. However, some GF foods, in particular bread, are still more expensive than non-gf foods. 5. Research into the prescribing position in England, as at March 2016, has shown that out of 209 CCGs in England, 112 have adopted policies to restrict the provision of GF foods. This includes 18 CCGs that have stopped the prescribing of all GF foods, and a further 12 that are considering or consulting on changes to GF prescribing. Of the CCGs that have made restrictions to the type of GF food available, most have restricted to either bread and flour, or bread, flour and pasta. Types of GF food prescribed in 2015 Cost and Items Type Net Ingredient Cost (NIC) Items dispensed Biscuits 1,086, ,300 Bread 16,710, ,900 Cakes/Pastries Cereals 912, ,800 Cooking Aids Grains/Flours 132,100 23,500 Mixes (flour/bread) 4,328, ,300 Pasta 2,554, ,100 TOTALS 25,727,200 1,678, NHS England supports this consultation. Page 9 of 9
CONSULATION ON ITEMS WHICH SHOULD NOT BE ROUTINELY PRESCRIBED IN PRIMARY CARE FREQUENTLY ASKED QUESTIONS
 CONSULATION ON ITEMS WHICH SHOULD NOT BE ROUTINELY PRESCRIBED IN PRIMARY CARE FREQUENTLY ASKED QUESTIONS Consultation content: 18 products Patient experience Savings Working group Over-the-counter medicines
CONSULATION ON ITEMS WHICH SHOULD NOT BE ROUTINELY PRESCRIBED IN PRIMARY CARE FREQUENTLY ASKED QUESTIONS Consultation content: 18 products Patient experience Savings Working group Over-the-counter medicines
Proposed changes to prescribing in Croydon. Lambeth. NHS prescriptions in Lambeth
 Proposed changes to prescribing in Croydon Lambeth NHS prescriptions in Lambeth 1 NHS Lambeth Clinical Commissioning Group 1. Introduction We are seeking your views on our proposals to make some changes
Proposed changes to prescribing in Croydon Lambeth NHS prescriptions in Lambeth 1 NHS Lambeth Clinical Commissioning Group 1. Introduction We are seeking your views on our proposals to make some changes
Items which should not routinely be prescribed in primary care: A Consultation on guidance for CCGs
 Items which should not routinely be prescribed in primary care: A Consultation on guidance for CCGs Items which should not routinely be prescribed in primary care: A Consultation on guidance for CCGs Version
Items which should not routinely be prescribed in primary care: A Consultation on guidance for CCGs Items which should not routinely be prescribed in primary care: A Consultation on guidance for CCGs Version
Proposed changes to prescribing in Croydon MALARIA TABLETS. Consultation on proposed changes to NHS prescriptions in Southwark
 Proposed changes to prescribing in Croydon MALARIA TABLETS Consultation on proposed changes to NHS prescriptions in Southwark NHS Croydon Clinical Commissioning Group Contents Who are we and what is this
Proposed changes to prescribing in Croydon MALARIA TABLETS Consultation on proposed changes to NHS prescriptions in Southwark NHS Croydon Clinical Commissioning Group Contents Who are we and what is this
Items which should not routinely be prescribed in primary care: Summary of PCN statements PCN to PCN
 Items which should not routinely be prescribed in primary care: Summary of PCN statements PCN 302-2018 to PCN 318-2018 In November 2017, NHS England issued final guidance in relation to items which should
Items which should not routinely be prescribed in primary care: Summary of PCN statements PCN 302-2018 to PCN 318-2018 In November 2017, NHS England issued final guidance in relation to items which should
Prescribing medicine for minor illnesses. Consultation. Information booklet
 Prescribing medicine for minor illnesses Consultation Information booklet Every year, there are a lot of GP appointments for illnesses people can treat by themselves, without having to see a doctor. People
Prescribing medicine for minor illnesses Consultation Information booklet Every year, there are a lot of GP appointments for illnesses people can treat by themselves, without having to see a doctor. People
Prescribing of over the counter medicines is changing
 Prescribing of over the counter medicines is changing NHS England held a consultation on some changes to prescribing medication you can buy over the counter. This medication can be bought over the counter
Prescribing of over the counter medicines is changing NHS England held a consultation on some changes to prescribing medication you can buy over the counter. This medication can be bought over the counter
Medicines for Self Care Policy
 Document purpose This policy describes the commissioning decision for medicines for self-care made by NHS Mansfield and Ashfield CCG and NHS Newark and Sherwood CCG. Version Version 1.0 Final Title Associated
Document purpose This policy describes the commissioning decision for medicines for self-care made by NHS Mansfield and Ashfield CCG and NHS Newark and Sherwood CCG. Version Version 1.0 Final Title Associated
Response ID ANON-788X-RE3D-D
 Response ID ANON-788X-RE3D-D Submitted to Conditions for which over the counter items should not routinely be prescribed in primary care: A consultation on guidance for CCGs Submitted on 2018-03-13 15:11:25
Response ID ANON-788X-RE3D-D Submitted to Conditions for which over the counter items should not routinely be prescribed in primary care: A consultation on guidance for CCGs Submitted on 2018-03-13 15:11:25
Appendix 1 Consultation Document and Survey
 Appendix 1 Consultation Document and Survey Proposals to stop prescribing medicines for minor conditions NHS Blackburn with Darwen Clinical Commissioning Group continuously reviews the services it commissions
Appendix 1 Consultation Document and Survey Proposals to stop prescribing medicines for minor conditions NHS Blackburn with Darwen Clinical Commissioning Group continuously reviews the services it commissions
Hertfordshire and West Essex Sustainability and Transformation Partnership. Welcome
 Welcome What is an STP? Herts Valleys CCG, East and North Hertfordshire CCG and West Essex CCG working together with county and district councils The challenges we face Prioritising health services in
Welcome What is an STP? Herts Valleys CCG, East and North Hertfordshire CCG and West Essex CCG working together with county and district councils The challenges we face Prioritising health services in
Please select which items you would like to share your views on (please select)? NHS England proposes to advise clinical commissioning groups,
 Items which should not routinely be prescribed in primary care: A Consultation on guidance for CCGs Homeopathy Research Institute submission to public consultation by NHS England 21 October 2017 Rachel
Items which should not routinely be prescribed in primary care: A Consultation on guidance for CCGs Homeopathy Research Institute submission to public consultation by NHS England 21 October 2017 Rachel
Promotion of Self Care Briefing Document
 RCCG/GB/17/035 Promotion of Self Care Briefing Document 1. Introduction The proposal aims to promote self-care through the purchasing of medications and products from local pharmacies for the treatment
RCCG/GB/17/035 Promotion of Self Care Briefing Document 1. Introduction The proposal aims to promote self-care through the purchasing of medications and products from local pharmacies for the treatment
Treating minor conditions
 Treating minor conditions Think Pharmacist THE BEST OF HEALTH FOR WEST SUFFOLK Treating minor conditions Minor conditions are generally not a serious health problem and can be treated with Over The Counter
Treating minor conditions Think Pharmacist THE BEST OF HEALTH FOR WEST SUFFOLK Treating minor conditions Minor conditions are generally not a serious health problem and can be treated with Over The Counter
CCG/PH updates FAO Community Pharmacies
 CCG/PH updates FAO Community Pharmacies 1. Stop smoking county update 2.Gluten Free Prescribing East and West CCG 3.Discontinuation of Minor Ailments West CCG 4.Changes to OTC prescribing East CCG 5.changes
CCG/PH updates FAO Community Pharmacies 1. Stop smoking county update 2.Gluten Free Prescribing East and West CCG 3.Discontinuation of Minor Ailments West CCG 4.Changes to OTC prescribing East CCG 5.changes
NHS West Cheshire Clinical Commissioning Group does not fund the prescribing for dental conditions on FP10.
 PRESCRIBING COMMISSIONING POLICY: DENTAL CONDITIONS NHS West Cheshire Clinical Commissioning Group does not fund the prescribing for dental conditions on FP10. Note: Patients who are not eligible for treatment
PRESCRIBING COMMISSIONING POLICY: DENTAL CONDITIONS NHS West Cheshire Clinical Commissioning Group does not fund the prescribing for dental conditions on FP10. Note: Patients who are not eligible for treatment
Items which should not be routinely prescribed in primary care: Consultation Report of Findings NHS England
 Items which should not be routinely prescribed in primary care: Consultation Report of Findings NHS England NHS England INFORMATION READER BOX Directorate Medical Operations and Information Specialised
Items which should not be routinely prescribed in primary care: Consultation Report of Findings NHS England NHS England INFORMATION READER BOX Directorate Medical Operations and Information Specialised
Agenda Item: 16 NHS Norwich CCG Governing Body Tuesday 25 th July 2017
 Agenda Item: 16 NHS Norwich CCG Governing Body Tuesday 25 th July 2017 Subject: Presented By: Submitted To: Purpose of Paper: Self-care for Self-Limiting Conditions Policy Karen Watts, Director of Quality
Agenda Item: 16 NHS Norwich CCG Governing Body Tuesday 25 th July 2017 Subject: Presented By: Submitted To: Purpose of Paper: Self-care for Self-Limiting Conditions Policy Karen Watts, Director of Quality
This guidance applies to all prescribers, both medical and non-medical.
 1 Prescribing Guidance Gluten Free Foods This guidance applies to all prescribers, both medical and non-medical. NHS Dudley has agreed that the prescribing of gluten-free food for patients with a confirmed
1 Prescribing Guidance Gluten Free Foods This guidance applies to all prescribers, both medical and non-medical. NHS Dudley has agreed that the prescribing of gluten-free food for patients with a confirmed
self care what is self care? caring a pack to start conversations... THE NHS SAYS But can it mean more? mental wellbeing minor ailments
 self care In Dudley borough mental wellbeing what is self care? THE NHS SAYS minor ailments long term conditions caring role of professionals Self care is about keeping fit and healthy, understanding when
self care In Dudley borough mental wellbeing what is self care? THE NHS SAYS minor ailments long term conditions caring role of professionals Self care is about keeping fit and healthy, understanding when
Changing the way we prescribe in Calderdale. Consultation document
 Changing the way we prescribe in Calderdale Consultation document Who are we? Clinical commissioning groups (CCGs) are responsible for planning and buying (commissioning) healthcare. We are made up of
Changing the way we prescribe in Calderdale Consultation document Who are we? Clinical commissioning groups (CCGs) are responsible for planning and buying (commissioning) healthcare. We are made up of
CHOOSING WISELY FOR KINGSTON PROPOSED CHANGES TO LOCAL HEALTHCARE - IVF
 GOVERNING BODY LEAD: Fergus Keegan, Director of Quality, Kingston & Richmond CCGs REPORT AUTHOR: Sue Lear, Acting Deputy Director of Commissioning ATTACHMENT: AGENDA ITEM: D2 RECOMMENDATION: The Governing
GOVERNING BODY LEAD: Fergus Keegan, Director of Quality, Kingston & Richmond CCGs REPORT AUTHOR: Sue Lear, Acting Deputy Director of Commissioning ATTACHMENT: AGENDA ITEM: D2 RECOMMENDATION: The Governing
Proposal to reduce prescribing of medicines and products that can be purchased without a prescription
 Brent CCG Governing Body Proposal to reduce prescribing of medicines and products that can be purchased without a prescription SRO: Janet Cree, Managing Director, Hammersmith and Fulham CCG (this is an
Brent CCG Governing Body Proposal to reduce prescribing of medicines and products that can be purchased without a prescription SRO: Janet Cree, Managing Director, Hammersmith and Fulham CCG (this is an
THE RESPONSIBLE PHARMACIST REGULATIONS
 THE RESPONSIBLE PHARMACIST REGULATIONS A SUMMARY OF THE RESPONSES TO PUBLIC CONSULTATION ON PROPOSALS FOR THE CONTENT OF THE REGULATIONS DH INFORMATION READER BOX Policy HR / Workforce Management Planning
THE RESPONSIBLE PHARMACIST REGULATIONS A SUMMARY OF THE RESPONSES TO PUBLIC CONSULTATION ON PROPOSALS FOR THE CONTENT OF THE REGULATIONS DH INFORMATION READER BOX Policy HR / Workforce Management Planning
NHS WEST ESSEX CLINICAL COMMISSIONING GROUP
 NHS WEST ESSEX CLINICAL COMMISSIONING GROUP Gluten Free Policy Brief Description (max 50 words) Target Audience NHS West Essex CCG Board agreed to restrict the prescription of Gluten Free foods to bread
NHS WEST ESSEX CLINICAL COMMISSIONING GROUP Gluten Free Policy Brief Description (max 50 words) Target Audience NHS West Essex CCG Board agreed to restrict the prescription of Gluten Free foods to bread
Seeking your views on proposed changes to prescribing in Croydon. Tuesday 13 December 2016
 Seeking your views on proposed changes to prescribing in Croydon Tuesday 13 December 2016 Dr Agnelo Fernandes Assistant Clinical Chair Paula Swann Chief Officer NHS financial pressures July 2016 NHS financial
Seeking your views on proposed changes to prescribing in Croydon Tuesday 13 December 2016 Dr Agnelo Fernandes Assistant Clinical Chair Paula Swann Chief Officer NHS financial pressures July 2016 NHS financial
Meeting of Governing Body Date: 7 August 2018 Time: 1.30pm Location: Clevedon Hall, Elton Rd, Clevedon, North Somerset, BS21 7RQ
 Meeting of Governing Body Date: Time: 1.30pm Location: Clevedon Hall, Elton Rd, Clevedon, North Somerset, BS21 7RQ Agenda number: 6.1 Report title: Review of NHS-funded homeopathy services Report Author:
Meeting of Governing Body Date: Time: 1.30pm Location: Clevedon Hall, Elton Rd, Clevedon, North Somerset, BS21 7RQ Agenda number: 6.1 Report title: Review of NHS-funded homeopathy services Report Author:
NHS ENGLAND BOARD PAPER
 NHS ENGLAND BOARD PAPER Paper: PB.15.12.2016/04 Title: Taking the cancer strategy forward: programme update Lead Director: Professor Sir Bruce Keogh, National Medical Director Cally Palmer, National Cancer
NHS ENGLAND BOARD PAPER Paper: PB.15.12.2016/04 Title: Taking the cancer strategy forward: programme update Lead Director: Professor Sir Bruce Keogh, National Medical Director Cally Palmer, National Cancer
Policy for Conditions for which over the counter items should not routinely be prescribed in primary care.
 Cannock Chase Clinical Commissioning Group South East Staffordshire and Seisdon Peninsula Clinical Commissioning Group Stafford and Surrounds Clinical Commissioning Group East Staffordshire Clinical Commissioning
Cannock Chase Clinical Commissioning Group South East Staffordshire and Seisdon Peninsula Clinical Commissioning Group Stafford and Surrounds Clinical Commissioning Group East Staffordshire Clinical Commissioning
Policy for the provision of gluten-free food in East and North Hertfordshire
 Policy for the provision of gluten-free food in East and North Hertfordshire Page 1 of 13 DOCUMENT CONTROL SHEET Document Owner: Document Author(s): Director of Commissioning Stacey Golding, Senior Pharmaceutical
Policy for the provision of gluten-free food in East and North Hertfordshire Page 1 of 13 DOCUMENT CONTROL SHEET Document Owner: Document Author(s): Director of Commissioning Stacey Golding, Senior Pharmaceutical
Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181)
 Putting NICE guidance into practice Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181) Published: July 2014 This costing report accompanies Lipid modification:
Putting NICE guidance into practice Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181) Published: July 2014 This costing report accompanies Lipid modification:
Have your say on local plans to change the range of items available on NHS prescription
 Have your say on local plans to change the range of items available on NHS prescription Engagement runs from Wednesday 4 January 2017 to Wednesday 1 February 2017 What are we planning to do? The organisations
Have your say on local plans to change the range of items available on NHS prescription Engagement runs from Wednesday 4 January 2017 to Wednesday 1 February 2017 What are we planning to do? The organisations
GOVERNING BODY MEETING in Public 22 February 2017 Agenda Item 3.4
 GOVERNING BODY MEETING in Public 22 February 2017 Paper Title Purpose of paper Redesign of Services for Frail Older People in Eastern Cheshire To seek approval from Governing Body for the redesign of services
GOVERNING BODY MEETING in Public 22 February 2017 Paper Title Purpose of paper Redesign of Services for Frail Older People in Eastern Cheshire To seek approval from Governing Body for the redesign of services
A guide to essential gluten-free foods available from the pharmacy
 A guide to essential gluten-free foods available from the pharmacy This information guide has been produced by For further information: This resource has been reviewed by the British Dietetic Association.
A guide to essential gluten-free foods available from the pharmacy This information guide has been produced by For further information: This resource has been reviewed by the British Dietetic Association.
Prescribing for Clinical Need Engagement Report
 Prescribing for Clinical Need Engagement Report Executive Summary Proposal Between 3 August 2016 and 9 September 2016 NHS Blackpool Clinical Commissioning Group (CCG) conducted engagement activity to gather
Prescribing for Clinical Need Engagement Report Executive Summary Proposal Between 3 August 2016 and 9 September 2016 NHS Blackpool Clinical Commissioning Group (CCG) conducted engagement activity to gather
Our plan about prescriptions and saving money
 Our plan about prescriptions and saving money Telling us what you think An easy read booklet & questionnaire 1 Who we are We are NHS Croydon Clinical Commissioning Group (CCG). We are responsible for buying
Our plan about prescriptions and saving money Telling us what you think An easy read booklet & questionnaire 1 Who we are We are NHS Croydon Clinical Commissioning Group (CCG). We are responsible for buying
NORTH STRATEGIC NEIGHBOURHOOD FORUM. Peter Howarth, Head of Medicines Management, Tameside & Glossop CCG
 Report To: NORTH STRATEGIC NEIGHBOURHOOD FORUM Date: 16 October 2018 Reporting Officer: Subject: Report Summary: Peter Howarth, Head of Medicines Management, Tameside & Glossop CCG CCG CONSULTATION ON
Report To: NORTH STRATEGIC NEIGHBOURHOOD FORUM Date: 16 October 2018 Reporting Officer: Subject: Report Summary: Peter Howarth, Head of Medicines Management, Tameside & Glossop CCG CCG CONSULTATION ON
Care homes - Homely remedies
 Bulletin 72 August 2014 Community Interest Company Care homes - Homely remedies Care home staff have a recognised duty of care to be able to respond to minor symptoms experienced by residents. A homely
Bulletin 72 August 2014 Community Interest Company Care homes - Homely remedies Care home staff have a recognised duty of care to be able to respond to minor symptoms experienced by residents. A homely
This paper outlines the engagement activity that took place, and provides key themes from the 57 written responses received.
 Agenda item: 5.4 Subject: Presented by: Prepared by: Submitted to: Specialist Fertility Services Dr Dustyn Saint SNCCG Commissioning Team SNCCG Communications and Engagement Team SNCCG Governing Body Date:
Agenda item: 5.4 Subject: Presented by: Prepared by: Submitted to: Specialist Fertility Services Dr Dustyn Saint SNCCG Commissioning Team SNCCG Communications and Engagement Team SNCCG Governing Body Date:
Case scenarios: Patient Group Directions
 Putting NICE guidance into practice Case scenarios: Patient Group Directions Implementing the NICE guidance on Patient Group Directions (MPG2) Published: March 2014 [updated March 2017] These case scenarios
Putting NICE guidance into practice Case scenarios: Patient Group Directions Implementing the NICE guidance on Patient Group Directions (MPG2) Published: March 2014 [updated March 2017] These case scenarios
NHS Sheffield Community Pharmacy Seasonal Flu Vaccination Programme for hard to reach at risk groups (and catch up campaign for over 65s)
 NHS Sheffield Community Pharmacy Seasonal Flu Vaccination Programme for hard to reach at risk groups 2012-13 (and catch up campaign for over 65s) Service Evaluation! Supported by Sheffield!Local!Pharmaceutical!Committee!
NHS Sheffield Community Pharmacy Seasonal Flu Vaccination Programme for hard to reach at risk groups 2012-13 (and catch up campaign for over 65s) Service Evaluation! Supported by Sheffield!Local!Pharmaceutical!Committee!
Q1 Which of the following applies to you?
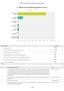 Q1 Which of the following applies to you? Answered: 80 Skipped: 1 I have coeliac disease I am the parent/guard... I am a carer of an adult... I am responding o... I am a clinician, f... I am responding
Q1 Which of the following applies to you? Answered: 80 Skipped: 1 I have coeliac disease I am the parent/guard... I am a carer of an adult... I am responding o... I am a clinician, f... I am responding
GOVERNING BOARD. Assisted Conception (IVF): Review of access criteria. Date of Meeting 21 January 2015 Agenda Item No 13. Title
 GOVERNING BOARD Date of Meeting 21 January 2015 Agenda Item No 13 Title Assisted Conception (IVF): Review of access criteria Purpose of Paper The SHIP (Southampton, Hampshire, Isle of Wight and Portsmouth)
GOVERNING BOARD Date of Meeting 21 January 2015 Agenda Item No 13 Title Assisted Conception (IVF): Review of access criteria Purpose of Paper The SHIP (Southampton, Hampshire, Isle of Wight and Portsmouth)
Care homes - Homely remedies
 Bulletin 72 August 2014 Care homes - Homely remedies Care home staff have a recognised duty of care to be able to respond to minor symptoms experienced by residents. A homely remedy is a medicinal product
Bulletin 72 August 2014 Care homes - Homely remedies Care home staff have a recognised duty of care to be able to respond to minor symptoms experienced by residents. A homely remedy is a medicinal product
HC 963 SesSIon november Department of Health. Young people s sexual health: the National Chlamydia Screening Programme
 Report by the Comptroller and Auditor General HC 963 SesSIon 2008 2009 12 november 2009 Department of Health Young people s sexual health: the National Chlamydia Screening Programme 4 Summary Young people
Report by the Comptroller and Auditor General HC 963 SesSIon 2008 2009 12 november 2009 Department of Health Young people s sexual health: the National Chlamydia Screening Programme 4 Summary Young people
Putting NICE guidance into practice. Resource impact report: Hearing loss in adults: assessment and management (NG98)
 Putting NICE guidance into practice Resource impact report: Hearing loss in adults: assessment and management (NG98) Published: June 2018 Summary This report focuses on the recommendation from NICE s guideline
Putting NICE guidance into practice Resource impact report: Hearing loss in adults: assessment and management (NG98) Published: June 2018 Summary This report focuses on the recommendation from NICE s guideline
Standard Operating Procedure: Early Intervention in Psychosis Access Times
 Corporate Standard Operating Procedure: Early Intervention in Psychosis Access Times Document Control Summary Status: New Version: V1.0 Date: Author/Owner: Rob Abell, Senior Performance Development Manager
Corporate Standard Operating Procedure: Early Intervention in Psychosis Access Times Document Control Summary Status: New Version: V1.0 Date: Author/Owner: Rob Abell, Senior Performance Development Manager
The Society welcomes the opportunity to respond the Department of Health s consultation on generic substitution.
 Lindsey Gilpin Chairman of the English Pharmacy Board Telephone: 020 7572 2519 Fax: 0207 572 2501 e-mail: howard.duff@rpsgb.org 25 th March 2010 Dear Sir/Madam, Consultation on the proposals to implement
Lindsey Gilpin Chairman of the English Pharmacy Board Telephone: 020 7572 2519 Fax: 0207 572 2501 e-mail: howard.duff@rpsgb.org 25 th March 2010 Dear Sir/Madam, Consultation on the proposals to implement
CABINET PROCURING A SUBSTANCE MISUSE & COMMUNITY TREATMENT SERVICE IN RUTLAND
 CABINET Report No: 105/2017 PUBLIC REPORT 16 May 2017 PROCURING A SUBSTANCE MISUSE & COMMUNITY TREATMENT SERVICE IN RUTLAND Report of the Director of Public Health Strategic Aim: Safeguarding Key Decision:
CABINET Report No: 105/2017 PUBLIC REPORT 16 May 2017 PROCURING A SUBSTANCE MISUSE & COMMUNITY TREATMENT SERVICE IN RUTLAND Report of the Director of Public Health Strategic Aim: Safeguarding Key Decision:
Service Specification & Contract Intermediate Stop Smoking Service & Voucher fulfilment - Pharmacy Newcastle
 Service Specification & Contract Intermediate Stop Smoking Service & Voucher fulfilment - Pharmacy Newcastle Contents 1. Agreement period 2. Scope 3. Targets 4. Service outline 5. Support for clients using
Service Specification & Contract Intermediate Stop Smoking Service & Voucher fulfilment - Pharmacy Newcastle Contents 1. Agreement period 2. Scope 3. Targets 4. Service outline 5. Support for clients using
Gluten-free resource pack
 Gluten-free resource pack Contents 1. Frequently asked questions 2. Gluten-free suppliers list 3. Gluten-free menu suggestions 4. Gluten-free checklist (from Coeliac UK) 5. Gluten-free diet on a budget
Gluten-free resource pack Contents 1. Frequently asked questions 2. Gluten-free suppliers list 3. Gluten-free menu suggestions 4. Gluten-free checklist (from Coeliac UK) 5. Gluten-free diet on a budget
The RPS is the professional body for pharmacists in Wales and across Great Britain. We are the only body that represents all sectors of pharmacy.
 Royal Pharmaceutical Society 2 Ash Tree Court Woodsy Close Cardiff Gate Business Park Pontprennau Cardiff CF23 8RW Mr Mark Drakeford AM, Chair, Health and Social Care Committee National Assembly for Wales
Royal Pharmaceutical Society 2 Ash Tree Court Woodsy Close Cardiff Gate Business Park Pontprennau Cardiff CF23 8RW Mr Mark Drakeford AM, Chair, Health and Social Care Committee National Assembly for Wales
Should the NHS pay for homeopathy? HAVE YOUR SAY. Public consultation
 Should the NHS pay for homeopathy? HAVE YOUR SAY Public consultation Have your say 1 Setting the scene 1.1 West Kent PCT 1.2 This consultation West Kent Primary Care Trust (PCT) ensures that health services
Should the NHS pay for homeopathy? HAVE YOUR SAY Public consultation Have your say 1 Setting the scene 1.1 West Kent PCT 1.2 This consultation West Kent Primary Care Trust (PCT) ensures that health services
GOVERNING BODY REPORT
 GOVERNING BODY REPORT DATE OF MEETING: 20th September 2012 TITLE OF REPORT: KEY MESSAGES: NHS West Cheshire Clinical Commissioning Group has identified heart disease as one of its six strategic clinical
GOVERNING BODY REPORT DATE OF MEETING: 20th September 2012 TITLE OF REPORT: KEY MESSAGES: NHS West Cheshire Clinical Commissioning Group has identified heart disease as one of its six strategic clinical
Service Level Agreement for the Provision of Level 1 Substance Misuse Services from a Community Pharmacy under contract to NHS Grampian
 Service Level Agreement for the Provision of Level 1 Substance Misuse Services from a Community Pharmacy under contract to NHS Grampian 1. Introduction The provision of Substance Misuse (SM) services through
Service Level Agreement for the Provision of Level 1 Substance Misuse Services from a Community Pharmacy under contract to NHS Grampian 1. Introduction The provision of Substance Misuse (SM) services through
Enhanced Service Specification. Childhood seasonal influenza vaccination programme 2017/18
 Enhanced Service Specification Childhood seasonal influenza vaccination programme 2017/18 2 Enhanced Service Specification Childhood seasonal influenza vaccination programme Version number: 1 First published:
Enhanced Service Specification Childhood seasonal influenza vaccination programme 2017/18 2 Enhanced Service Specification Childhood seasonal influenza vaccination programme Version number: 1 First published:
Referral criteria/red flags/ Exceptions in addition to general exceptions. Self-limiting condition Recommendations Rationale
 Appendix 2: Conditions for which over the counter (OTC) items should not be routinely prescribed in Primary Care. The rationale and recommendations from the NHSE consultation, exceptions and referral criteria.
Appendix 2: Conditions for which over the counter (OTC) items should not be routinely prescribed in Primary Care. The rationale and recommendations from the NHSE consultation, exceptions and referral criteria.
Buprenorphine used in the treatment of opioid dependence: availability and price
 7 th floor Wellington House 133 155 Waterloo Road London SE1 8UG 24 October 2018 Gateway number: L2018-443 Dear Director of Public Health, Buprenorphine used in the treatment of opioid dependence: availability
7 th floor Wellington House 133 155 Waterloo Road London SE1 8UG 24 October 2018 Gateway number: L2018-443 Dear Director of Public Health, Buprenorphine used in the treatment of opioid dependence: availability
NHS Sheffield Community Pharmacy Catch Up Seasonal Flu Vaccination Programme for hard to reach at risk groups
 NHS Sheffield Community Pharmacy Catch Up Seasonal Flu Vaccination Programme for hard to reach at risk groups 2011-12 Service Evaluation Supported by Sheffield Local Pharmaceutical Committee Supporting
NHS Sheffield Community Pharmacy Catch Up Seasonal Flu Vaccination Programme for hard to reach at risk groups 2011-12 Service Evaluation Supported by Sheffield Local Pharmaceutical Committee Supporting
Enhanced Service Specification. Childhood seasonal influenza vaccination programme 2018/19
 Enhanced Service Specification Childhood seasonal influenza vaccination programme 2018/19 Contents Childhood seasonal influenza vaccination programme... 1 Contents... 4 1 Introduction... 5 2 Background...
Enhanced Service Specification Childhood seasonal influenza vaccination programme 2018/19 Contents Childhood seasonal influenza vaccination programme... 1 Contents... 4 1 Introduction... 5 2 Background...
Pharmaceutical System in the UK
 Pharmaceutical System in the UK David Kullman Medicines, i Pharmacy and Industry Group Department of Health PPRI Conference, Vienna 30 September 2011 Disclaimer Conflict of interest: Nothing to disclose
Pharmaceutical System in the UK David Kullman Medicines, i Pharmacy and Industry Group Department of Health PPRI Conference, Vienna 30 September 2011 Disclaimer Conflict of interest: Nothing to disclose
North Somerset Autism Strategy
 North Somerset Autism Strategy Approved by: Ratification date: Review date: September 2017 1 Contents 1 Introduction and background... 3 2 Defining Autism...Error! Bookmark not defined. 3 National and
North Somerset Autism Strategy Approved by: Ratification date: Review date: September 2017 1 Contents 1 Introduction and background... 3 2 Defining Autism...Error! Bookmark not defined. 3 National and
Prescribing Guidelines on Gluten-Free products. Information for GPs
 Prescribing Guidelines on Gluten-Free products Information for GPs This guideline should be used in conjunction with NICE clinical guideline 86 Coeliac disease: recognition and assessment of coeliac disease.
Prescribing Guidelines on Gluten-Free products Information for GPs This guideline should be used in conjunction with NICE clinical guideline 86 Coeliac disease: recognition and assessment of coeliac disease.
Commissioning Policy: South Warwickshire CCG (SWCCG)
 Commissioning Policy: South Warwickshire CCG (SWCCG) Treatment Conditions for which over the counter items should not routinely be prescribed in primary care Background During 2017/18 South Warwickshire
Commissioning Policy: South Warwickshire CCG (SWCCG) Treatment Conditions for which over the counter items should not routinely be prescribed in primary care Background During 2017/18 South Warwickshire
Access to clinical trial information and the stockpiling of Tamiflu. Department of Health
 MEMORANDUM FOR THE COMMITTEE OF PUBLIC ACCOUNTS HC 125 SESSION 2013-14 21 MAY 2013 Department of Health Access to clinical trial information and the stockpiling of Tamiflu 4 Summary Access to clinical
MEMORANDUM FOR THE COMMITTEE OF PUBLIC ACCOUNTS HC 125 SESSION 2013-14 21 MAY 2013 Department of Health Access to clinical trial information and the stockpiling of Tamiflu 4 Summary Access to clinical
Homely Remedies Guidance for Care Homes
 Homely Remedies Guidance for Care Homes What is a homely remedy? Homely or household remedy is another name for a non-prescription medicine which is used in a care home for the short term management of
Homely Remedies Guidance for Care Homes What is a homely remedy? Homely or household remedy is another name for a non-prescription medicine which is used in a care home for the short term management of
Care Homes - Homely Remedies Protocol
 Care Homes - Homely Remedies Protocol A homely remedy is a medicine for the short-term treatment of a minor ailment, such as indigestion, a cough, mild to moderate pain or constipation, and can be obtained
Care Homes - Homely Remedies Protocol A homely remedy is a medicine for the short-term treatment of a minor ailment, such as indigestion, a cough, mild to moderate pain or constipation, and can be obtained
Introduction, medicines classification and standard operating procedures
 Sample chapter copyright Pharmaceutical Press chapter 1 Introduction, medicines classification and standard operating procedures Overview Upon completion of this chapter, you should be able to: understand
Sample chapter copyright Pharmaceutical Press chapter 1 Introduction, medicines classification and standard operating procedures Overview Upon completion of this chapter, you should be able to: understand
Ways to Wellbeing. Social Prescribing Programme. Social Impact Report Report produced by:
 Ways to Wellbeing Social Prescribing Programme Social Impact Report 2016-2017 Report produced by: Who We Are Wellbeing Enterprises CIC is an award winning social enterprise recognised for its innovative
Ways to Wellbeing Social Prescribing Programme Social Impact Report 2016-2017 Report produced by: Who We Are Wellbeing Enterprises CIC is an award winning social enterprise recognised for its innovative
Health Scrutiny Panel 6 February 2014
 Agenda Item No: 5 Health Scrutiny Panel 6 February 2014 Report title Infertility Policy Review Wolverhampton Clinical Commissioning Group Cabinet member with lead responsibility Wards affected Accountable
Agenda Item No: 5 Health Scrutiny Panel 6 February 2014 Report title Infertility Policy Review Wolverhampton Clinical Commissioning Group Cabinet member with lead responsibility Wards affected Accountable
Enquiry Severity Immediate or other. Joint or muscle pain - any part of the body. Abdominal pain Acute Non-acute. Acne Anxiety Acute Non-acute
 We operate a hybrid triage system offering a mixture of telephone consultations, on the day emergency consultations with a doctor or pre bookable (3 days in advance) GP / Nurse Practitioner appointments
We operate a hybrid triage system offering a mixture of telephone consultations, on the day emergency consultations with a doctor or pre bookable (3 days in advance) GP / Nurse Practitioner appointments
Guidance on Bulk Prescribing for Care Home Patients
 Guidance on Bulk Prescribing for Care Home Patients Introduction Many patients in care homes taking medicines when required (PRN) can inevitably present problems for the prescriber in determining the quantity
Guidance on Bulk Prescribing for Care Home Patients Introduction Many patients in care homes taking medicines when required (PRN) can inevitably present problems for the prescriber in determining the quantity
Specialised Services Commissioning Policy. CP29: Bariatric Surgery
 Specialised Services Commissioning Policy CP29: Bariatric Surgery Document Author: Specialist Planner, Cardiothoracic Executive Lead: Director of Planning Approved by: Management Group Issue Date: 12 June
Specialised Services Commissioning Policy CP29: Bariatric Surgery Document Author: Specialist Planner, Cardiothoracic Executive Lead: Director of Planning Approved by: Management Group Issue Date: 12 June
Non-Prescription Medicinal Products Containing Codeine: Guidance for Pharmacists on Safe Supply to Patients
 Non-Prescription Medicinal Products Containing Codeine: Guidance for Pharmacists on Safe Supply to Patients Pharmaceutical Society of Ireland Version 3 October 2017 Updates made following the enactment
Non-Prescription Medicinal Products Containing Codeine: Guidance for Pharmacists on Safe Supply to Patients Pharmaceutical Society of Ireland Version 3 October 2017 Updates made following the enactment
1. Introduction. 2. Role of PSI as Pharmacy Regulator
 1. Introduction 1.1. I would like to begin by thanking the Committee for inviting the Pharmaceutical Society of Ireland here today to assist in the Committee s on-going scrutiny of the Cannabis for Medicinal
1. Introduction 1.1. I would like to begin by thanking the Committee for inviting the Pharmaceutical Society of Ireland here today to assist in the Committee s on-going scrutiny of the Cannabis for Medicinal
Report by the Comptroller and. SesSIon January Improving Dementia Services in England an Interim Report
 Report by the Comptroller and Auditor General HC 82 SesSIon 2009 2010 14 January 2010 Improving Dementia Services in England an Interim Report 4 Summary Improving Dementia Services in England an Interim
Report by the Comptroller and Auditor General HC 82 SesSIon 2009 2010 14 January 2010 Improving Dementia Services in England an Interim Report 4 Summary Improving Dementia Services in England an Interim
NHS Diabetes Programme
 NHS Diabetes Programme London Regional Event Vision Through the NHS Diabetes Programme we will aim to slow the future growth in the incidence of diabetes and reduce the rate of complications associated
NHS Diabetes Programme London Regional Event Vision Through the NHS Diabetes Programme we will aim to slow the future growth in the incidence of diabetes and reduce the rate of complications associated
Governing Body (Public) Meeting
 ENCLOSURE: T Agenda Item: 86/13 Governing Body (Public) Meeting DATE: 25 July 2013 Title Recommended action for the Governing Body Expanding the Treatment Access Policy for Bexley That the Governing Body:
ENCLOSURE: T Agenda Item: 86/13 Governing Body (Public) Meeting DATE: 25 July 2013 Title Recommended action for the Governing Body Expanding the Treatment Access Policy for Bexley That the Governing Body:
Health and Wellbeing Board 10 November 2016
 Title Report of Wards All Status Public Urgent Key Enclosures Officer Contact Details Health and Wellbeing Board 10 November 2016 Update on childhood immunisations 0-5 years Dr Andrew Howe - Director of
Title Report of Wards All Status Public Urgent Key Enclosures Officer Contact Details Health and Wellbeing Board 10 November 2016 Update on childhood immunisations 0-5 years Dr Andrew Howe - Director of
PATIENT GROUP DIRECTION PROCEDURE
 PATIENT GROUP DIRECTION PROCEDURE Date approved 2 October 2015 Version 3 Approved by Yvette Oade, Chief Medical Officer Procedure Lead Clinical Governance Lead - Medicines Management Procedure Author Karen
PATIENT GROUP DIRECTION PROCEDURE Date approved 2 October 2015 Version 3 Approved by Yvette Oade, Chief Medical Officer Procedure Lead Clinical Governance Lead - Medicines Management Procedure Author Karen
INFECTIOUS DISEASES & ADMINISTRATION OF MEDICINES IN SCHOOL
 INFECTIOUS DISEASES & ADMINISTRATION OF MEDICINES IN SCHOOL This policy is for use with pupils who have short term medical conditions that require medicine. The needs of those pupils who have long term
INFECTIOUS DISEASES & ADMINISTRATION OF MEDICINES IN SCHOOL This policy is for use with pupils who have short term medical conditions that require medicine. The needs of those pupils who have long term
COPYRIGHTED MATERIAL. Introduction
 1 Introduction Pharmacology is the science of drugs and their effects on biological systems. A drug can be defined as a chemical that can cause a change in a biological system; the important biological
1 Introduction Pharmacology is the science of drugs and their effects on biological systems. A drug can be defined as a chemical that can cause a change in a biological system; the important biological
Headache Pathway Case for Change
 Headache Pathway Case for Change November 2017 Table of Contents 1 2 3 4 5 6 7 Executive summary... 3 Background... 4 Introduction... 5 Epidemiology of headache... 5 National strategic context and drivers
Headache Pathway Case for Change November 2017 Table of Contents 1 2 3 4 5 6 7 Executive summary... 3 Background... 4 Introduction... 5 Epidemiology of headache... 5 National strategic context and drivers
SPORTS FOODS, MEDICAL AND PERFORMANCE SUPPLEMENT POLICY
 SPORTS FOODS, MEDICAL AND PERFORMANCE SUPPLEMENT POLICY RA Board Approved Date 7 th November 2013 Page 1 of 14 1. Background The Australian Institute of Sport (AIS) Sports Supplement Program was originally
SPORTS FOODS, MEDICAL AND PERFORMANCE SUPPLEMENT POLICY RA Board Approved Date 7 th November 2013 Page 1 of 14 1. Background The Australian Institute of Sport (AIS) Sports Supplement Program was originally
USE OF UNLICENSED MEDICINES AND OFF-LABEL MEDICINES WHERE A LICENSED MEDICINE IS AVAILABLE
 NHS Scotland Directors of Pharmacy and Scottish Association of Medical Directors USE OF UNLICENSED MEDICINES AND OFF-LABEL MEDICINES WHERE A LICENSED MEDICINE IS AVAILABLE CONSENSUS STATEMENT This consensus
NHS Scotland Directors of Pharmacy and Scottish Association of Medical Directors USE OF UNLICENSED MEDICINES AND OFF-LABEL MEDICINES WHERE A LICENSED MEDICINE IS AVAILABLE CONSENSUS STATEMENT This consensus
HIGHLANDS SURGERY NEWSLETTER
 HIGHLANDS SURGERY NEWSLETTER Spring 2017 Inside This Issue 1 Virtual GP service (babylon) 2 Website Did Not Attend 3 Who to contact when you are not feeling well 4 Who to contact when you are not feeling
HIGHLANDS SURGERY NEWSLETTER Spring 2017 Inside This Issue 1 Virtual GP service (babylon) 2 Website Did Not Attend 3 Who to contact when you are not feeling well 4 Who to contact when you are not feeling
Review of Medicines Act 1968: informal consultation on issues relating to the PLR regime and homeopathy
 Review of Medicines Act 1968: informal consultation on issues relating to the PLR regime and homeopathy Introduction 1. The consolidation and review of the UK medicines legislation (including the Medicines
Review of Medicines Act 1968: informal consultation on issues relating to the PLR regime and homeopathy Introduction 1. The consolidation and review of the UK medicines legislation (including the Medicines
Which? response to the NHS dental services in England Independent Review by Professor Jimmy Steele
 Which? response to the NHS dental services in England Independent Review by Professor Jimmy Steele DATE: 14 July 2009 TO: Rt Hon Andy Burnham MP Secretary of State for Health Department of Health Richmond
Which? response to the NHS dental services in England Independent Review by Professor Jimmy Steele DATE: 14 July 2009 TO: Rt Hon Andy Burnham MP Secretary of State for Health Department of Health Richmond
Self-care aware When should you prescribe?
 Self-care aware When should you prescribe? Over-the-counter medicines can only be sold within their licensed indications and in limited quantities in some cases. Below are some examples of instances when
Self-care aware When should you prescribe? Over-the-counter medicines can only be sold within their licensed indications and in limited quantities in some cases. Below are some examples of instances when
OUR LADY QUEEN OF PEACE R.C. PRIMARY SCHOOL
 OUR LADY QUEEN OF PEACE R.C. PRIMARY SCHOOL Food in School, including Packed Lunch Policy In our school we believe that each person is unique and created to flourish in God s image. We aspire to excellence
OUR LADY QUEEN OF PEACE R.C. PRIMARY SCHOOL Food in School, including Packed Lunch Policy In our school we believe that each person is unique and created to flourish in God s image. We aspire to excellence
Medicines which should not be regularly prescribed in primary care. Consultation. Information booklet
 Medicines which should not be regularly prescribed in primary care Consultation Information booklet About this document When we say primary care, we mean the medical services that people go to first. These
Medicines which should not be regularly prescribed in primary care Consultation Information booklet About this document When we say primary care, we mean the medical services that people go to first. These
Insulin Pumps and Glucose Monitors in Adults Policy
 Insulin Pumps and Glucose Monitors in Adults Policy Version: 2016-19 Ratified by: NHS Leeds West CCG Assurance Committee on; 16 November 2016 NHS Leeds North CCG Governance on Performance and Risk Committee
Insulin Pumps and Glucose Monitors in Adults Policy Version: 2016-19 Ratified by: NHS Leeds West CCG Assurance Committee on; 16 November 2016 NHS Leeds North CCG Governance on Performance and Risk Committee
POLICY BRIEFING. Prime Minister s challenge on dementia 2020 implementation plan
 POLICY BRIEFING Prime Minister s challenge on dementia 2020 implementation plan Date: 14th March 2016 Author: Christine Heron LGiU associate Summary The Prime Minister s challenge on dementia contains
POLICY BRIEFING Prime Minister s challenge on dementia 2020 implementation plan Date: 14th March 2016 Author: Christine Heron LGiU associate Summary The Prime Minister s challenge on dementia contains
GMS Contract in Wales Enhanced Service for Homeless Patients Specification
 Doc 4 GMS Contract in Wales 2008-09 Enhanced Service for Homeless Patients Specification Introduction The purpose of this paper is to provide an enhanced service specification for Local Health Boards when
Doc 4 GMS Contract in Wales 2008-09 Enhanced Service for Homeless Patients Specification Introduction The purpose of this paper is to provide an enhanced service specification for Local Health Boards when
Draft Guidelines on the Sale and Supply of Non- Prescription Medicinal Products from a Retail Pharmacy Business
 Draft Guidelines on the Sale and Supply of Non- Prescription Medicinal Products from a Retail Pharmacy Business To facilitate compliance with Regulations 5(1)(d), 5(1)(h) and 10 of the Regulation of Retail
Draft Guidelines on the Sale and Supply of Non- Prescription Medicinal Products from a Retail Pharmacy Business To facilitate compliance with Regulations 5(1)(d), 5(1)(h) and 10 of the Regulation of Retail
Collation of responses to GW. 1. Please state the definitions that you use for different forms of palliative and end of life services
 Collation of responses to GW 1. Please state the definitions that you use for different forms of palliative and end of life services Palliative care is the active holistic care of patients with advanced
Collation of responses to GW 1. Please state the definitions that you use for different forms of palliative and end of life services Palliative care is the active holistic care of patients with advanced
St. Cuthbert s RC Primary School
 St. Cuthbert s RC Primary School Packed Lunch Policy Head Teacher: R Donnelly Chair of Governors: T Devereux Article 24 Every child has the right to the best possible health. Governments must work to provide
St. Cuthbert s RC Primary School Packed Lunch Policy Head Teacher: R Donnelly Chair of Governors: T Devereux Article 24 Every child has the right to the best possible health. Governments must work to provide
Appendix 1. Cognitive Impairment and Dementia Service Elm Lodge 4a Marley Close Greenford Middlesex UB6 9UG
 Appendix 1 Mr Dwight McKenzie Scrutiny Review Officer Legal and Democratic Services Ealing Council Perceval House 14 16 Uxbridge Road Ealing London W5 2HL Cognitive Impairment and Dementia Service Elm
Appendix 1 Mr Dwight McKenzie Scrutiny Review Officer Legal and Democratic Services Ealing Council Perceval House 14 16 Uxbridge Road Ealing London W5 2HL Cognitive Impairment and Dementia Service Elm
Guidance on Bulk Prescribing for Care Home Residents
 Guidance on Bulk Prescribing for Care Home Residents There are many residents in care homes taking medicines when required, which may present problems for the prescriber in determining the quantity to
Guidance on Bulk Prescribing for Care Home Residents There are many residents in care homes taking medicines when required, which may present problems for the prescriber in determining the quantity to
FreeStyle Libre for glucose monitoring: Interim Position Statement for GPs & Patient FAQ
 North Central London Joint Formulary Committee FreeStyle Libre for glucose monitoring: Interim Position Statement for GPs & Patient FAQ GPs should not prescribe FreeStyle Libre sensors on the NHS until
North Central London Joint Formulary Committee FreeStyle Libre for glucose monitoring: Interim Position Statement for GPs & Patient FAQ GPs should not prescribe FreeStyle Libre sensors on the NHS until
