Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 12 Years and Older
|
|
|
- Logan Hunter
- 5 years ago
- Views:
Transcription
1 Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 12 Years and Older The Canadian Thacic Society recommends a tempary, greater the inhaled cticosteroid () dose in selected patients in response to wsening asthma symptoms. 1 The green yellow red zone framewk describes asthma symptoms that are controlled, partially controlled uncontrolled, respectively. This guideline based recommendation is distinct from the manufacturer s recommended dose in the product monograph. Any the dose requires an individual assessment by the clinician a written asthma action plan. F all examples in this document, the tempary dose does not exceed the manufacturer s recommended maximum dose. As a result, it is not possible to recommend an evidenced based greater the all dosage situations. In the absence of an evidence based option, clinicians exercising clinical judgment may choose a 2 fold the dose. Initiating prednisone in the yellow zone is also an alternative evidence based option. Dose Dose of dose in Fluticasone propionate pmdi Flovent HFA 50 /puff 2 puff ** 125 /puff 1 puff * /puff puffs *** /puff 1 puff * /puff *Although the manufacturer recommends that the usual dose be obtained using 2 puffs from each available strength of Flovent HFA pmdi, one puff multiple puffs may be required to obtain the prescribed dose. **Increasing the dose beyond may be impractical, an frequency to qid dosing may be considered. ***Switching from the 125 to the 250 pmdi will reduce the number of puffs required per dose. Dose Dose of dose in Fluticasone propionate Flovent Diskus /inhalation 1 inh 4 inhalations 250 /inhalation 1 inh inhalations /inhalation 1 inh 0 2 inhalations 0 0 Dose based on 1 inhalation from each available strength of Flovent Diskus. Me than 1 inhalation may be required to obtain the prescribed dose from the patient s inhaler Dose Dose of dose in Fluticasone furoate* Arnuity Ellipta /inhalation 1 inh 2 inhalations /inhalation 1 inh No recommendation** *Dose based on 1 inhalation from each available strength of Arnuity Ellipta. Me than 1 inhalation may be required to obtain the prescribed dose from the lower strength inhaler.. 1
2 Budesonide Pulmict Turbuhaler /inhalation 1 inh /inhalation 1 inh /inhalation 1 inh Dose 4 inhalations 4 inhalations 3 inhalations Total dose 1 dose In Dose based on 1 inhalation from each available strength of Pulmict Turbuhaler. Me than 1 inhalation may be required to obtain the prescribed dose from the patient s inhaler. Beclomethasone pmdi Qvar 50 /puff 1 puff 50 /puff /puff 1 puff /puff Dose 8 puffs Total dose dose In Dose based on 1 puff from each available strength of Qvar inhaler. Me than 1 inhalation may be required to obtain the prescribed dose from the patient s inhaler. Ciclesonide pmdi Alvesco /puff 1 puff * /puff 2 puffs /puff 1 puff /puff 2 puffs /puff Dose using the patient s existing inhaler Total dose dose In *Dose based on 1 puff from each available strength of Alvesco inhaler. Me than 1 inhalation may be required to obtain the prescribed dose from the patient s inhaler. Medication in the Green Zone Dose using the patient s existing inhaler Total dose dose Mometasone Asmanex Twisthaler /inhalation 1 inh 4 inhalations /inhalation 1 inh * 2 inhalations /inhalation 1 inh 2 inhalations /inhalation 1 inh 1 inhalation /inhalation 1 inh *Dose based on 1 puff from each available strength of Asmanex Twisthaler. Me than 1 inhalation may be required to obtain the prescribed dose from the patient s inhaler. 2
3 Advair pmdi Fluticasone/salmeterol* 125/ Dose using additional fluticasone pmdi, Flovent HFA switching to the higher strength of Advair pmdi Add fluticasone 250 /puff pmdi 3 puffs Switch to Advair 250/25 dose In /25 0 Add fluticasone 250 /puff pmdi 0 *Note: Since each puff from the Advair pmdi delivers salmeterol 25, the manufacturer s recommended dose is 2 puffs from each available strength of Advair pmdi in der to obtain 50 of salmeterol at each dose time. Advair Diskus Fluticasone/salmeterol* /50 1 inhalation Dose using additional doses from the fluticasone, Flovent Diskus switch to a higher strength of Advair Diskus combine the 2 strategies Add fluticasone 250 /inhalation, 1 inhalation add fluticasone /inhalation, 3 inhalations fold 5 fold 250/50 1 inhalation 500 Add fluticasone 250 /inhalation, 3 inhalations add fluticasone 500 /inhalation, 1 inhalation plus fluticasone 500, 1 inhalation /50 1 inhalation 0 Add fluticasone 500 /inhalation, 1 inhalation 0 *Note: Since each inhalation from the Advair Diskus delivers salmeterol 50, the manufacturer s recommended dose is 1 inhalations from each available strength of Advair Diskus in der to obtain 50 of salmeterol at each dose time. 3
4 Breo Ellipta Fluticasone furoate/vilanterol* /25 1 inhalation Dose using additional doses from the fluticasone furoate, Arnuity Ellipta switch to a higher strength of Breo Elliipta Add fluticasone furoate /inh, 1 inhalation Switch to Breo Ellipta /25, 1 inhalation /25 1 inhalation *Note: Each inhalation from either strength of Breo Ellipta delivers vilanterol 25, which is the maximum dose Note: Breo Ellipta /25 and /25 are indicated in asthma. Breo Ellipta /25 is not indicted f use in COPD. Zenhale pmdi Mometasone/fmoterol* Dose using Additional mometasone, Asmanex Twisthaler switch to a higher strength of Zenhale pmdi /5 Add mometasone /inhalation 1 inhalation Switch to Zenhale /5 /5 *Note: Since each puff from the Zenhale pmdi delivers fmoterol 5, the manufacturer s recommended dose is 2 puffs from each available strength of Zenhale pmdi in der to obtain 10 of fmoterol at each dose time. Note: The addition of mometasone, Asmanex Twisthaler as a strategy to increase the inhaled cticosteroid dose introduces a different device (breath actuated, dry powder inhaler) and may necessitate patient education on the use of the new device. 4
5 Symbict Turbuhaler Budesonide/fmoterol* /6 1 inhalation /6 1 inhalation /6 2 inhalations /6 2 inhalations Dose using Existing Symbict Turbuhaler Symbict Adjustable Maintenance Dosing Increase to 4 inhalations/day dose /6 1 inhalation /6 1 inhalation /6 2 inhalations /6 2 inhalations Increase to 4 inhalations/day alone, add budesonide 1 inhalation Or continue 2 inh + budesonide 2 inh /6 1 2 inhalations 2 inhalations /6 1 2 inhalations 2 inhalations to to Symbict Maintenance and Reliever Therapy (SMART) In addition to the dose, may take 1 additional dose as needed in response to symptoms. Not me than 6 inhalations on any single occasion. Not me than 8 inhalations per day in total ( and reliever doses).** Up to /day Up to 1600 /day Up to Up to *Dose based on 1 inhalation from each available strength of Symbict Turbuhaler. Not me than 6 inhalations should be taken on any single occasion **If satisfacty relief of asthma symptoms is not achieved with a maximum of 8 inhalations per day, seek medical attention. Reference: 1. Lougheed MD, Lemière C, Ducharme FM, et al. Canadian Thacic Society 2012 guideline update: Diagnosis and management of asthma in preschoolers, children and adults. Can Resp J 2012;19(2): Copyright This document is provided f clinicians, as resource in the management of asthma f persons aged 12 years and older. Determination of the most appropriate dose of inhaled cticosteroid f any individual is the responsibility of the clinician together with the patient. The Ontario Lung Association makes no claims about the appropriateness of the inhaled cticosteroid dose. 5
Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 16 Years and Older
 Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 16 Years and Older The Canadian Thoracic Society and other international asthma
Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 16 Years and Older The Canadian Thoracic Society and other international asthma
APPENDIX 1 Printable point-of-care tables Asthma Action Plan Yellow Zone Formulation Table Region: Europe
 APPENDIX 1 Printable point-of-care tables Asthma Action Plan Yellow Zone Formulation Table Region: Europe Instructions: Print on 8.5 x14 (216 x 279 mm) paper (Legal size) Medication in Green Zone Change
APPENDIX 1 Printable point-of-care tables Asthma Action Plan Yellow Zone Formulation Table Region: Europe Instructions: Print on 8.5 x14 (216 x 279 mm) paper (Legal size) Medication in Green Zone Change
Drug Class Monograph
 Drug Class Monograph Class: Inhaled Corticosteroids Drugs: Aerospan (flunisolide), Advair Diskus, Advair HFA (fluticasone/salmeterol), Alvesco (ciclesonide), Arnuity Ellipta (fluticasone furoate), Asmanex
Drug Class Monograph Class: Inhaled Corticosteroids Drugs: Aerospan (flunisolide), Advair Diskus, Advair HFA (fluticasone/salmeterol), Alvesco (ciclesonide), Arnuity Ellipta (fluticasone furoate), Asmanex
Inhaled Corticosteroid Dose Comparison in Asthma
 This Clinical Resource gives subscribers additional insight related to the Recommendations published in April 2017 ~ Resource #330402 Inhaled Corticosteroid Dose Comparison in Asthma The chart below provides
This Clinical Resource gives subscribers additional insight related to the Recommendations published in April 2017 ~ Resource #330402 Inhaled Corticosteroid Dose Comparison in Asthma The chart below provides
Diagnosis and Management of Asthma
 Supporting Evidence: Diagnosis and Management of Asthma The subdivision of this section is: Appendix B Tables Copyright 2016 by 1 Eleventh Edition/December 2016 Appendix B Asthma Summary Tables Class:
Supporting Evidence: Diagnosis and Management of Asthma The subdivision of this section is: Appendix B Tables Copyright 2016 by 1 Eleventh Edition/December 2016 Appendix B Asthma Summary Tables Class:
Inhaled Corticosteroids Drug Class Prior Authorization Protocol
 Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Inhaled Corticosteroids Drug Class Prior Authorization Protocol
 Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Fasenra) Reference Number: CP.PHAR.## Effective Date: 01.16.18 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Fasenra) Reference Number: CP.PHAR.## Effective Date: 01.16.18 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Alberta Childhood Asthma Pathway for Primary Care
 Asthma Diagnosis Box 1 Diagnosis: Based on symptom pattern, careful and thorough history of symptoms (wheeze, cough, night waking and activity limitations), and assessment of family history of asthma and
Asthma Diagnosis Box 1 Diagnosis: Based on symptom pattern, careful and thorough history of symptoms (wheeze, cough, night waking and activity limitations), and assessment of family history of asthma and
TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder
 TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
Aerospan (flunisolide)
 STRENGTH DOSAGE FORM ROUTE GPID 80mcg/actuation HFA aerosol inhaler w/ Inhaled 35718 8.9 g/canister adapter MANUFACTURER Meda Pharmaceuticals INDICATION Aerospan Inhalation Aerosol is indicated for the
STRENGTH DOSAGE FORM ROUTE GPID 80mcg/actuation HFA aerosol inhaler w/ Inhaled 35718 8.9 g/canister adapter MANUFACTURER Meda Pharmaceuticals INDICATION Aerospan Inhalation Aerosol is indicated for the
Orally Inhaled Corticosteroids to 2022
 Greystone Research Associates 1+603-595-4340 April 2015 Orally Inhaled Corticosteroids to 2022 Drugs, Devices, Markets and Forecasts Contents A Comprehensive Market Analysis Report Scope & Overview 2 Table
Greystone Research Associates 1+603-595-4340 April 2015 Orally Inhaled Corticosteroids to 2022 Drugs, Devices, Markets and Forecasts Contents A Comprehensive Market Analysis Report Scope & Overview 2 Table
A Visual Approach to Simplifying Respiratory Drug Regimens
 A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP 3 Main Categories Inhaled Respiratory Drugs Binds to beta-2 receptors Relaxation of smooth muscles in the lung
A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP 3 Main Categories Inhaled Respiratory Drugs Binds to beta-2 receptors Relaxation of smooth muscles in the lung
benralizumab (Fasenra )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
The Medical Letter. on Drugs and Therapeutics
 The Medical Letter publications are protected by US and international copyright laws. Forwarding, copying or any other distribution of this material is strictly prohibited. For further information call:
The Medical Letter publications are protected by US and international copyright laws. Forwarding, copying or any other distribution of this material is strictly prohibited. For further information call:
Clinical Policy: Dupilumab (Dupixent) Reference Number: ERX.SPA.49 Effective Date:
 Clinical Policy: (Dupixent) Reference Number: ERX.SPA.49 Effective Date: 06.01.17 Last Review Date: 02.19 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Dupixent) Reference Number: ERX.SPA.49 Effective Date: 06.01.17 Last Review Date: 02.19 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
COPD Update: Focus on Intensifying LABA, LAMA and ICS Therapy
 Update: Focus on Intensifying LABA, LAMA and ICS Therapy B.C. Provincial Academic Detailing Service February 2017 Background In Canada, approximately 20 inhaled medications are approved to treat Chronic
Update: Focus on Intensifying LABA, LAMA and ICS Therapy B.C. Provincial Academic Detailing Service February 2017 Background In Canada, approximately 20 inhaled medications are approved to treat Chronic
reslizumab (Cinqair )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Improving Outcomes in COPD
 Neil MacIntyre MD Duke University Durham NC Current treatment guidelines f COPD focus Barriers to providing optimal treatment Diagnosis of COPD EXPOSURE TO RISK FACTORS AND/ OR SYMPTOMS sputum cough dyspnea
Neil MacIntyre MD Duke University Durham NC Current treatment guidelines f COPD focus Barriers to providing optimal treatment Diagnosis of COPD EXPOSURE TO RISK FACTORS AND/ OR SYMPTOMS sputum cough dyspnea
BREEZHALER. Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate)
 Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate) BREEZHALER Please date initial after you have directly observed
Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate) BREEZHALER Please date initial after you have directly observed
BREEZHALER. Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate)
 Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate) BREEZHALER Please date initial after you have directly observed
Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate) BREEZHALER Please date initial after you have directly observed
BREEZHALER. Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate)
 Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate) BREEZHALER Please date initial after you have directly observed
Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate) BREEZHALER Please date initial after you have directly observed
Select Inhaled Respiratory Agents
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
COPD Medications Coverage Summary Non-Insured Health Benefits Coverage SABA Bricanyl turbuhaler Yes Yes
 COPD Medications Coverage Summary Drug Non-Insured Health Benefits Coverage SABA Bricanyl turbuhaler Yes Yes Ventolin MDI + generics Yes Yes Ventolin Diskus NO NO Yukon Pharmacare/Chronic Disease Program
COPD Medications Coverage Summary Drug Non-Insured Health Benefits Coverage SABA Bricanyl turbuhaler Yes Yes Ventolin MDI + generics Yes Yes Ventolin Diskus NO NO Yukon Pharmacare/Chronic Disease Program
Nucala (mepolizumab injection for subcutaneous use)
 Nucala (mepolizumab injection for subcutaneous use) Policy Number: 5.01.612 Last Review: 01/2018 Origination: 02/2016 Next Review: 02/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Nucala (mepolizumab injection for subcutaneous use) Policy Number: 5.01.612 Last Review: 01/2018 Origination: 02/2016 Next Review: 02/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
MDI Bonanza. Dwayne Griffin, DO
 MDI Bonanza Dwayne Griffin, DO Bonanza 3. A MDI costing $200 - $500 per month SISYPHUS MDI Griffin Mountain Evolution of Deliver Systems for COPD in the US 2003 2009 2011 2013 2004 2012 2014 Prescribing
MDI Bonanza Dwayne Griffin, DO Bonanza 3. A MDI costing $200 - $500 per month SISYPHUS MDI Griffin Mountain Evolution of Deliver Systems for COPD in the US 2003 2009 2011 2013 2004 2012 2014 Prescribing
CLINICAL MEDICAL POLICY
 CLINICAL MEDICAL POLICY Policy Name: Xolair (omalizumab) Policy Number: MP-051-MD-DE Responsible Department(s): Medical Management; Clinical Pharmacy Provider Notice Date: 10/01/2017 Original Effective
CLINICAL MEDICAL POLICY Policy Name: Xolair (omalizumab) Policy Number: MP-051-MD-DE Responsible Department(s): Medical Management; Clinical Pharmacy Provider Notice Date: 10/01/2017 Original Effective
1.* Dosage. A. Adults
 Aerosolized Agents (Metered-Dose Inhalers) - Inhaled Anti-Inflammatory Drugs: Corticosteroids [Developed, January 1995; Revised, February, 1997; August 1997; March 1998; February 1999; January 2000; March
Aerosolized Agents (Metered-Dose Inhalers) - Inhaled Anti-Inflammatory Drugs: Corticosteroids [Developed, January 1995; Revised, February, 1997; August 1997; March 1998; February 1999; January 2000; March
A Visual Approach to Simplifying Respiratory Drug Regimens
 A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP October 23, 2017 Learning Objectives Be able to list at least 3 major adverse effects of inhaled medications
A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP October 23, 2017 Learning Objectives Be able to list at least 3 major adverse effects of inhaled medications
Ferris State University College of Pharmacy MPA CE Symposium 2016 Paul Thill, PharmD, BCPS
 Ferris State University College of Pharmacy MPA CE Symposium 2016 Paul Thill, PharmD, BCPS Objectives Categorize the new asthma and COPD inhalers in to existing or newly created categories Discuss the
Ferris State University College of Pharmacy MPA CE Symposium 2016 Paul Thill, PharmD, BCPS Objectives Categorize the new asthma and COPD inhalers in to existing or newly created categories Discuss the
A Visual Approach to Simplifying Respiratory Drug Regimens
 Adverse Effects of Inhaled Medications A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP June 28, 2017 Drug Category Beta 2 agonists antagonists Adverse Effects
Adverse Effects of Inhaled Medications A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP June 28, 2017 Drug Category Beta 2 agonists antagonists Adverse Effects
Key features and changes to these four components of asthma care include:
 Guidelines for the Diagnosis and Management of Asthma in Adults Clinical Practice Guideline MedStar Health These guidelines are provided to assist physicians and other clinicians in making decisions regarding
Guidelines for the Diagnosis and Management of Asthma in Adults Clinical Practice Guideline MedStar Health These guidelines are provided to assist physicians and other clinicians in making decisions regarding
Long Term Care Formulary RS -29
 RESTRICTED USE Asthma/COPD Management 1 of 6 PROTOCOL: Asthma Glossary of Medication Acronyms: SABA: short-acting beta agonist (e.g. salbutamol) SABD: short-acting bronchodilator (e.g. ipratropium or SABA)
RESTRICTED USE Asthma/COPD Management 1 of 6 PROTOCOL: Asthma Glossary of Medication Acronyms: SABA: short-acting beta agonist (e.g. salbutamol) SABD: short-acting bronchodilator (e.g. ipratropium or SABA)
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Nucala) Reference Number: CP.PHAR.200 Effective Date: 04.01.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy: (Nucala) Reference Number: CP.PHAR.200 Effective Date: 04.01.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Cinqair (reslizumab injection for intravenous use)
 Cinqair (reslizumab injection for intravenous use) Policy Number: 5.02.522 Last Review: 04/2018 Origination: 04/2016 Next Review: 04/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Cinqair (reslizumab injection for intravenous use) Policy Number: 5.02.522 Last Review: 04/2018 Origination: 04/2016 Next Review: 04/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing
 Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
COPD Medicine. No one ever showed me how to use this. Wendy Happel; RRT, COPD Educator Krystal Fedoris; RRT-NPS, BA, COPD Educator
 Medicine. No one ever showed me how to use this. Wendy Happel; RRT, Educator Krystal Fedoris; RRT-NPS, BA, Educator 1 Taking prescriptions correctly Taking prescriptions can be a challenge Busy schedules
Medicine. No one ever showed me how to use this. Wendy Happel; RRT, Educator Krystal Fedoris; RRT-NPS, BA, Educator 1 Taking prescriptions correctly Taking prescriptions can be a challenge Busy schedules
What You Need to Know about Metered-Dose Inhalers and the HFA Propellant
 What You Need to Know about Metered- Inhalers and the HFA Propellant There are a number ways to deliver inhaled medication. They include: Metered-dose inhaler () Metered-dose inhaler with spacer/holding
What You Need to Know about Metered- Inhalers and the HFA Propellant There are a number ways to deliver inhaled medication. They include: Metered-dose inhaler () Metered-dose inhaler with spacer/holding
12/18/2017. Disclosures. Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing
 Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
MANAGING ASTHMA. Nancy Davis, RRT, AE-C
 MANAGING ASTHMA Nancy Davis, RRT, AE-C What is asthma? Asthma is a chronic respiratory disease characterized by episodes or attacks of inflammation and narrowing of small airways in response to asthma
MANAGING ASTHMA Nancy Davis, RRT, AE-C What is asthma? Asthma is a chronic respiratory disease characterized by episodes or attacks of inflammation and narrowing of small airways in response to asthma
Inhaled bronchodilators relax constricted airways and treat the noisy part of asthma: coughing, wheezing, choking and shortness of breath.
 Inhaled bronchodilators relax constricted airways and treat the noisy part of asthma: coughing, wheezing, choking and shortness of breath. AccuNeb inhalation 0.021% solution: 0.63mg/3mL 3-4 times solution
Inhaled bronchodilators relax constricted airways and treat the noisy part of asthma: coughing, wheezing, choking and shortness of breath. AccuNeb inhalation 0.021% solution: 0.63mg/3mL 3-4 times solution
SAMPLE. mg by mouth every day for day(s) Prednisolone. Other Medicine: Medicine Dose How long Directions
 Pediatric Asthma Discharge Prescription and Short-Term Plan The doctor will fill out this form before your child is discharged. Please follow this plan until you see your usual doctor in 3 to 7 days. Hospital
Pediatric Asthma Discharge Prescription and Short-Term Plan The doctor will fill out this form before your child is discharged. Please follow this plan until you see your usual doctor in 3 to 7 days. Hospital
Disclosure. Case. Objectives. Case Continued. Inhalers. Asthma: A GINA Update to the NAEPP 2007 Guidelines 1/20/2015
 Disclosure Asthma: A GINA Update to the NAEPP 2007 Guidelines Robert (RC) Hellinga, Pharm.D. PGY 1 Pharmacy Resident Wolfson Children s Hospital/Baptist Health I do not have a vested interest in or affiliation
Disclosure Asthma: A GINA Update to the NAEPP 2007 Guidelines Robert (RC) Hellinga, Pharm.D. PGY 1 Pharmacy Resident Wolfson Children s Hospital/Baptist Health I do not have a vested interest in or affiliation
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Fluticasone/Salmeterol (Advair Diskus, Advair HFA) Reference Number: CP.PMN.31 Effective Date: 10.01.18 Last Review Date: 07.13.18 Line of Business: Oregon Health Plan See Important Reminder
Clinical Policy: Fluticasone/Salmeterol (Advair Diskus, Advair HFA) Reference Number: CP.PMN.31 Effective Date: 10.01.18 Last Review Date: 07.13.18 Line of Business: Oregon Health Plan See Important Reminder
AIRDUO RESPICLICK (fluticasone-salmeterol) aerosol DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol
 DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
Stepping down asthma treatment guidelines
 Stepping down asthma treatment guidelines The potential for inhaled corticosteroids (ICS) to cause dose-related side-effects has led to asthma management guidelines recommending a dose reduction once asthma
Stepping down asthma treatment guidelines The potential for inhaled corticosteroids (ICS) to cause dose-related side-effects has led to asthma management guidelines recommending a dose reduction once asthma
Using Inhaled Corticosteroids as Needed for Asthma: giving patients relief or leaving them breathless?
 Using Inhaled Corticosteroids as Needed for Asthma: giving patients relief or leaving them breathless? Lindsay Thomas, Pharm.D. PGY2 Ambulatory Care Resident Department of Pharmacotherapy and Pharmacy
Using Inhaled Corticosteroids as Needed for Asthma: giving patients relief or leaving them breathless? Lindsay Thomas, Pharm.D. PGY2 Ambulatory Care Resident Department of Pharmacotherapy and Pharmacy
Clinical Policy: Mepolizumab (Nucala) Reference Number: ERX.SPA.214 Effective Date:
 Clinical Policy: (Nucala) Reference Number: ERX.SPA.214 Effective Date: 07.01.16 Last Review Date: 02.19 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Nucala) Reference Number: ERX.SPA.214 Effective Date: 07.01.16 Last Review Date: 02.19 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Question I was one of the first dry power devices available in the US Flovent, Serevent and Advair are all available in this device
 What Device am I Class Side Effects History Potpourri Monitoring Tools 10 10 10 10 10 20 20 20 20 20 30 30 30 30 30 40 40 40 40 40 50 50 50 50 50 WHAT KIND OF DEVICE AM I? I was one of the first dry power
What Device am I Class Side Effects History Potpourri Monitoring Tools 10 10 10 10 10 20 20 20 20 20 30 30 30 30 30 40 40 40 40 40 50 50 50 50 50 WHAT KIND OF DEVICE AM I? I was one of the first dry power
Asthma. Definition. Symptoms
 Asthma Definition Asthma is a condition in which your airways narrow and swell and produce extra mucus. This can make breathing difficult and trigger coughing, wheezing and shortness of breath. For some
Asthma Definition Asthma is a condition in which your airways narrow and swell and produce extra mucus. This can make breathing difficult and trigger coughing, wheezing and shortness of breath. For some
Allergies and Asthma 5/21/2013. Objectives. Allergic Rhinitis (AR): Risk Factor for ASTHMA. Rhinitis and Asthma
 Allergies and Asthma Presented By: Dr. Fadwa Gillanders, Pharm.D Clinical Pharmacy Specialist May 2013 Objectives Understand the relationship between asthma and allergic rhinitis Understand what is going
Allergies and Asthma Presented By: Dr. Fadwa Gillanders, Pharm.D Clinical Pharmacy Specialist May 2013 Objectives Understand the relationship between asthma and allergic rhinitis Understand what is going
Clinical Policy: Dupilumab (Dupixent) Reference Number: ERX.SPA.49 Effective Date:
 Clinical Policy: (Dupixent) Reference Number: ERX.SPA.49 Effective Date: 06.01.17 Last Review Date: 02.19 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Dupixent) Reference Number: ERX.SPA.49 Effective Date: 06.01.17 Last Review Date: 02.19 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: Omalizumab (Xolair) Reference Number: ERX.SPA.141 Effective Date: Last Review Date: 08.17
 Clinical Policy: (Xolair) Reference Number: ERX.SPA.141 Effective Date: 03.01.14 Last Review Date: 08.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Xolair) Reference Number: ERX.SPA.141 Effective Date: 03.01.14 Last Review Date: 08.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Preschool Asthma What you need to know in 10 minutes
 Preschool Asthma What you need to know in 10 minutes Alan Kaplan MD CCFP(EM) FCFP Family Physician Airways Group of Canada Respiratory Medicine section CFPC Faculty/Presenter Disclosure Faculty: Alan Kaplan
Preschool Asthma What you need to know in 10 minutes Alan Kaplan MD CCFP(EM) FCFP Family Physician Airways Group of Canada Respiratory Medicine section CFPC Faculty/Presenter Disclosure Faculty: Alan Kaplan
US max daily dose i Food and Drug Administration [2]
![US max daily dose i Food and Drug Administration [2] US max daily dose i Food and Drug Administration [2]](/thumbs/88/115964542.jpg) APPENDIX 2 etable 1. Regulatory limits on total daily dose for asthma medications Drugs Canadian max daily dose i Health Canada [1] US max daily dose i Food and Drug Administration [2] European max daily
APPENDIX 2 etable 1. Regulatory limits on total daily dose for asthma medications Drugs Canadian max daily dose i Health Canada [1] US max daily dose i Food and Drug Administration [2] European max daily
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Dupixent) Reference Number: CP.PHAR.336 Effective Date: 05.01.17 Last Review Date: 02.19 Line of Business: Commercial, HIM, Medicaid Coding Implications Revision Log See Important Reminder
Clinical Policy: (Dupixent) Reference Number: CP.PHAR.336 Effective Date: 05.01.17 Last Review Date: 02.19 Line of Business: Commercial, HIM, Medicaid Coding Implications Revision Log See Important Reminder
Reference Guide for Caring for Pediatric Patients with Asthma
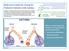 Reference Guide for Caring for Pediatric Patients with Asthma Co-Chair: Nancy Cantey Banasiak, DNP, PPCNP-BC, APRN Co-Chair: Deborah Hickman, DNP, APRN-CNP, CPNP-PC, NNP-BC Asthma and Allergy SIG Members
Reference Guide for Caring for Pediatric Patients with Asthma Co-Chair: Nancy Cantey Banasiak, DNP, PPCNP-BC, APRN Co-Chair: Deborah Hickman, DNP, APRN-CNP, CPNP-PC, NNP-BC Asthma and Allergy SIG Members
MEDICAL COVERAGE GUIDELINES ORIGINAL EFFECTIVE DATE: 07/05/18 SECTION: DRUGS LAST REVIEW DATE: LAST CRITERIA REVISION DATE: ARCHIVE DATE:
 CINQAIR (reslizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
CINQAIR (reslizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Drug Effectiveness Review Project Summary Report
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Stakeholder Comments and Ontario Drug Policy Research Network (ODPRN) Response:
 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca info@odprn.ca Stakeholder Comments and Ontario Drug Policy Research Network (ODPRN) Response: Reimbursement Options Report April 7 th, 2014 2 COMMENT We
30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca info@odprn.ca Stakeholder Comments and Ontario Drug Policy Research Network (ODPRN) Response: Reimbursement Options Report April 7 th, 2014 2 COMMENT We
Correct Use of Inhaler Devices
 PL Detail-Document #300206 This PL Detail-Document gives subscribers additional insight related to the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER February 2014 Correct Use of
PL Detail-Document #300206 This PL Detail-Document gives subscribers additional insight related to the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER February 2014 Correct Use of
ANTINEOPLASTIC DRUGS CHAPTER 21. Antineoplastic drugs - designed to treat malignancies, now also used to treat diseases with inflammatory component
 ANTINEOPLASTIC DRUGS CHAPTER 21 Antineoplastic drugs - designed to treat malignancies, now also used to treat diseases with inflammatory component Tx of malignancies Antineoplastic drugs: methotrexate
ANTINEOPLASTIC DRUGS CHAPTER 21 Antineoplastic drugs - designed to treat malignancies, now also used to treat diseases with inflammatory component Tx of malignancies Antineoplastic drugs: methotrexate
Breakout Session # A 12:45 1:25. New Methods of Medication Delivery to the Lungs: Say Goodbye to the MDI as we know it
 Breakout Session # A 12:45 1:25 New Methods of Medication Delivery to the Lungs: Say Goodbye to the MDI as we know it Michael J. Welch MD, FAAP, FAAAAI, CPI Co-director, Allergy & Asthma Medical Group
Breakout Session # A 12:45 1:25 New Methods of Medication Delivery to the Lungs: Say Goodbye to the MDI as we know it Michael J. Welch MD, FAAP, FAAAAI, CPI Co-director, Allergy & Asthma Medical Group
STRIVERDI RESPIMAT (olodaterol hcl) aerosol
 STRIVERDI RESPIMAT (olodaterol hcl) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
STRIVERDI RESPIMAT (olodaterol hcl) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
GMMMG Asthma Formulary Inhaler Options August 2017
 Regular Preventer Beclometasone Beclometasone Beclometasone Brand name Qvar Easi-Breathe Clenil Modulite Easyhaler Device MDI MDI DPI Strengths 50 microgram 100 microgram 200 microgram Adult asthma 2 inhalations
Regular Preventer Beclometasone Beclometasone Beclometasone Brand name Qvar Easi-Breathe Clenil Modulite Easyhaler Device MDI MDI DPI Strengths 50 microgram 100 microgram 200 microgram Adult asthma 2 inhalations
First to Market or 505 (b)2 CMC Considerations IPAC-RS/UF Orlando Inhalation Conference Orlando, Florida
 First to Market or 505 (b)2 CMC Considerations IPAC-RS/UF Orlando Inhalation Conference Orlando, Florida Prasad Peri, Ph.D., Branch Chief, ONDQA, FDA March 19, 2014 1 Topics for discussion Introduction
First to Market or 505 (b)2 CMC Considerations IPAC-RS/UF Orlando Inhalation Conference Orlando, Florida Prasad Peri, Ph.D., Branch Chief, ONDQA, FDA March 19, 2014 1 Topics for discussion Introduction
How to Use Inhaled Medications for Asthma and COPD
 How to Use Inhaled Medications for Asthma and COPD This information is not intended to diagnose health problems or to take the place of medical advice or care you receive from your physician or other health
How to Use Inhaled Medications for Asthma and COPD This information is not intended to diagnose health problems or to take the place of medical advice or care you receive from your physician or other health
Asthma medications: Know your options - MayoClinic.com. Asthma medications: Know your options
 MayoClinic.com reprints This single copy is for your personal, noncommercial use only. For permission to reprint multiple copies or to order presentation-ready copies for distribution, use the reprints
MayoClinic.com reprints This single copy is for your personal, noncommercial use only. For permission to reprint multiple copies or to order presentation-ready copies for distribution, use the reprints
The Inflammatory Response. Inflammation in the Airway. The Inflammatory Response. Inflammation in the Airway. Inflammation in the Airway
 The Inflammatory Response RSPT 2217 Gardenhire Chapter 11 A general definition inflammation is the response of vascularized tissue to injury First described in the first century A.D. and revised in the
The Inflammatory Response RSPT 2217 Gardenhire Chapter 11 A general definition inflammation is the response of vascularized tissue to injury First described in the first century A.D. and revised in the
Supplementary materials
 Supplementary materials Table S1 Patient comorbidities by diagnosis Total Asthma n (%) 5348 (148.4) COPD n (%) 4563 (143.4) ACOS n (%) 469 (197.9) No other comorbidities 95 (26.4) 614 (19.3) 27 (11.4)
Supplementary materials Table S1 Patient comorbidities by diagnosis Total Asthma n (%) 5348 (148.4) COPD n (%) 4563 (143.4) ACOS n (%) 469 (197.9) No other comorbidities 95 (26.4) 614 (19.3) 27 (11.4)
DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE UNIFORM FORMULARY BENEFICIARY ADVISORY PANEL
 DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE UNIFORM FORMULARY BENEFICIARY ADVISORY PANEL I. Uniform Formulary Review Process Under 10 U.S.C. 1074g, as implemented by 32
DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE UNIFORM FORMULARY BENEFICIARY ADVISORY PANEL I. Uniform Formulary Review Process Under 10 U.S.C. 1074g, as implemented by 32
Dose. Route. Units. Given. Dose. Route. Units. Given
 Chapter 4 Respiratory Andrew Stanton SALBUTAMOL (in acute asthma) 5 in acute asthma Nebulised (driven by oxygen not air) 4 6 hourly In acute severe asthma not responding to initial treatment or in life-threatening
Chapter 4 Respiratory Andrew Stanton SALBUTAMOL (in acute asthma) 5 in acute asthma Nebulised (driven by oxygen not air) 4 6 hourly In acute severe asthma not responding to initial treatment or in life-threatening
Medications for Managing COPD in Hospice Patients. Jim Joyner, PharmD, CGP Director of Clinical Operations Outcome Resources
 Medications for Managing COPD in Hospice Patients Jim Joyner, PharmD, CGP Director of Clinical Operations Outcome Resources Goal of medications in COPD Decrease symptoms and/or complications Reduce frequency
Medications for Managing COPD in Hospice Patients Jim Joyner, PharmD, CGP Director of Clinical Operations Outcome Resources Goal of medications in COPD Decrease symptoms and/or complications Reduce frequency
THE COPD PRESCRIBING TOOL
 THE COPD PRESCRIBING TOOL Revised edition, 2017 www.bpac.org.nz/copd CLASSIFICATION The COPD prescribing tool This tool provides pharmacological treatment options for patients with COPD based on their
THE COPD PRESCRIBING TOOL Revised edition, 2017 www.bpac.org.nz/copd CLASSIFICATION The COPD prescribing tool This tool provides pharmacological treatment options for patients with COPD based on their
Respiratory Health. Asthma and COPD
 Respiratory Health Asthma and COPD Definition of asthma Working definition by AAH 2014: Chronic lung disease Can be controlled not cured Large variation in lung function Large variation in respiratory
Respiratory Health Asthma and COPD Definition of asthma Working definition by AAH 2014: Chronic lung disease Can be controlled not cured Large variation in lung function Large variation in respiratory
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Nucala) Reference Number: CP.PHAR.200 Effective Date: 04.01.16 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy: (Nucala) Reference Number: CP.PHAR.200 Effective Date: 04.01.16 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Enhancing Patient Care
 Enhancing Patient Care I CAN control my asthma! PHYSICIAN WEB ORIENTATION COMMUNITY PEDIATRIC ASTHMA SERVICE JULY 2014 Sorry we couldn't connect in person This presentation will inform Calgary Zone physicians
Enhancing Patient Care I CAN control my asthma! PHYSICIAN WEB ORIENTATION COMMUNITY PEDIATRIC ASTHMA SERVICE JULY 2014 Sorry we couldn't connect in person This presentation will inform Calgary Zone physicians
Improving Outcomes in COPD. Improving Outcomes in COPD 4/4/2018
 Updates 2018 Neil MacIntyre MD Duke University Durham NC. Barnes PJ. N Engl J Med. 2000;343:269-80. 1 COPD spectrum Proximal predominant (large airways) mucus gland hypertrophy (cough/sputum) reduced respiraty
Updates 2018 Neil MacIntyre MD Duke University Durham NC. Barnes PJ. N Engl J Med. 2000;343:269-80. 1 COPD spectrum Proximal predominant (large airways) mucus gland hypertrophy (cough/sputum) reduced respiraty
II: Moderate Worsening airflow limitations Dyspnea on exertion, cough, and sputum production; patient usually seeks medical
 Table 3.1. Classification of COPD Severity Stage Pulmonary Function Test Findings Symptoms I: Mild Mild airflow limitations +/ Chronic cough and sputum production; patient unaware of abnormal FEV 1 80%
Table 3.1. Classification of COPD Severity Stage Pulmonary Function Test Findings Symptoms I: Mild Mild airflow limitations +/ Chronic cough and sputum production; patient unaware of abnormal FEV 1 80%
Adult Emergency Department Asthma Care Pathway (EDACP)
 Adult Emergency Department Asthma Care Pathway (EDACP) June, 2018 Contact Information Email: info@lungontario.ca Phone: 1-888-344-LUNG (5864) Fax: 416-922-9430 Adult Emergency Department Asthma Care Pathway
Adult Emergency Department Asthma Care Pathway (EDACP) June, 2018 Contact Information Email: info@lungontario.ca Phone: 1-888-344-LUNG (5864) Fax: 416-922-9430 Adult Emergency Department Asthma Care Pathway
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Seebri Neohaler) Reference Number: CP.CPA.150 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Seebri Neohaler) Reference Number: CP.CPA.150 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy
Asthma/COPD Update with Inhaler Workshop
 Asthma/COPD Update with Inhaler Workshop October 8, 2017 Nathan Samsa, DO, Pharm D, RPh, FACOI None Disclosures Agenda Asthma Updates COPD Updates Inhaler Workshop Medication Acronyms SABA: Short acting
Asthma/COPD Update with Inhaler Workshop October 8, 2017 Nathan Samsa, DO, Pharm D, RPh, FACOI None Disclosures Agenda Asthma Updates COPD Updates Inhaler Workshop Medication Acronyms SABA: Short acting
Global Strategy for the Diagnosis, Management and Prevention of COPD 2016 Clinical Practice Guideline. MedStar Health
 Global Strategy f the Diagnosis, Management and Prevention of COPD 2016 Clinical Practice Guideline MedStar Health These guidelines are provided to assist physicians and other clinicians in making decisions
Global Strategy f the Diagnosis, Management and Prevention of COPD 2016 Clinical Practice Guideline MedStar Health These guidelines are provided to assist physicians and other clinicians in making decisions
ASTHMA PRESCRIBING GUIDELINES FOR ADULTS AND CHILDREN OVER 12
 North Hampshire CCG Asthma Prescribing Guidelines June 2015 ASTHMA PRESCRIBING GUIDELINES FOR ADULTS AND CHILDREN OVER 12 These guidelines are based on the British Thoracic Society (BTS) and Scottish Intercollegiate
North Hampshire CCG Asthma Prescribing Guidelines June 2015 ASTHMA PRESCRIBING GUIDELINES FOR ADULTS AND CHILDREN OVER 12 These guidelines are based on the British Thoracic Society (BTS) and Scottish Intercollegiate
Respiratory Inhalers. Identification Guide Version 3
 Respiratory Inhalers Identification Guide Version 3 This booklet has been prepared by NHSGGC Medicines Information. Endorsed by NHSGGC Respiratory Managed Clinical Network, February 2017. Designed by Medical
Respiratory Inhalers Identification Guide Version 3 This booklet has been prepared by NHSGGC Medicines Information. Endorsed by NHSGGC Respiratory Managed Clinical Network, February 2017. Designed by Medical
Common Inhaled Asthma Medications Dose Comparison and Tips for Use
 Detail-Document #210303 This Detail-Document accompanies the related article published in PHARMACIST S LETTER / PRESCRIBER S LETTER March 2005 ~ Volume 21 ~ Number 210303 Common Inhaled Asthma Medications
Detail-Document #210303 This Detail-Document accompanies the related article published in PHARMACIST S LETTER / PRESCRIBER S LETTER March 2005 ~ Volume 21 ~ Number 210303 Common Inhaled Asthma Medications
GINA. At-A-Glance Asthma Management Reference. for adults, adolescents and children 6 11 years. Updated 2017
 GINA At-A-Glance Asthma Management Reference for adults, adolescents and children 6 11 years Updated 2017 This resource should be used in conjunction with the Global Strategy for Asthma Management and
GINA At-A-Glance Asthma Management Reference for adults, adolescents and children 6 11 years Updated 2017 This resource should be used in conjunction with the Global Strategy for Asthma Management and
Effective Date: 4/27/2016 Version: 1.0 Approval By: CCC Clinical Delivery Steering Planned Review Date: 4/27/2017
 Protocol Title: Adult Asthma Protocol Effective Date: 4/27/2016 Version: 1.0 Approval By: CCC Clinical Delivery Steering Planned Review Date: 4/27/2017 1 Purpose & Objective This protocol provides evidence-based
Protocol Title: Adult Asthma Protocol Effective Date: 4/27/2016 Version: 1.0 Approval By: CCC Clinical Delivery Steering Planned Review Date: 4/27/2017 1 Purpose & Objective This protocol provides evidence-based
How to Use Inhaled Medications
 How to Use Inhaled Medications FOR ASTHMA AND COPD introduction Inhaled medications are an important part of controlling and treating asthma and chronic obstructive pulmonary disease (COPD). The medication
How to Use Inhaled Medications FOR ASTHMA AND COPD introduction Inhaled medications are an important part of controlling and treating asthma and chronic obstructive pulmonary disease (COPD). The medication
Stepping-down combination ICS/LABA asthma inhaler therapy: Adults 18yrs
 Step Down guidance Stepping-down combination ICS/LABA asthma inhaler therapy: Adults 18yrs Important Complete asthma control needs to be achieved for at least 12 weeks before attempting to step patients
Step Down guidance Stepping-down combination ICS/LABA asthma inhaler therapy: Adults 18yrs Important Complete asthma control needs to be achieved for at least 12 weeks before attempting to step patients
Nancy Davis, RRT, AE-C
 Nancy Davis, RRT, AE-C Asthma Statistics 25.6 million Americans diagnosed with asthma 6.8 million are children 10.5 million missed school days per year 14.2 lost work days for adults Approximately 10%
Nancy Davis, RRT, AE-C Asthma Statistics 25.6 million Americans diagnosed with asthma 6.8 million are children 10.5 million missed school days per year 14.2 lost work days for adults Approximately 10%
Rapporteur s Public Assessment Report for paediatric studies submitted in accordance with Article 45 of Regulation (EC) No1901/2006, as amended
 Rappteur s Public Assessment Rept f paediatric studies submitted in accdance with Article 45 of Regulation (EC) No1901/2006, as amended Symbict Turbohaler 80/4.5 μg, 160/4.5 μg, 320/9 μg DE/W/046/pdWS/001
Rappteur s Public Assessment Rept f paediatric studies submitted in accdance with Article 45 of Regulation (EC) No1901/2006, as amended Symbict Turbohaler 80/4.5 μg, 160/4.5 μg, 320/9 μg DE/W/046/pdWS/001
QUANTITY LIMIT CRITERIA. BROVANA (arformoterol tartrate) SEREVENT DISKUS (salmeterol) STRIVERDI RESPIMAT (olodaterol)
 Carelirst. +.V Family of health care plans DRUG CLASS COMBINATIONS QUANTITY LIMIT CRITERIA LONG ACTING BETA2-ADRENERGIC AGONIST, ORAL INHALATION BRAND NAME (generic) LONG-ACTING BETA2-ADRENERGIC AGONISTS:
Carelirst. +.V Family of health care plans DRUG CLASS COMBINATIONS QUANTITY LIMIT CRITERIA LONG ACTING BETA2-ADRENERGIC AGONIST, ORAL INHALATION BRAND NAME (generic) LONG-ACTING BETA2-ADRENERGIC AGONISTS:
AT TRIAGE. Alberta Acute Childhood Asthma Pathway: Evidence based* recommendations For Emergency / Urgent Care
 1 1 Should the child be placed into the Pathway? Asthma Clinical Score (PRAM) Inclusion Children 1 year and 18 years of age who present with wheezing and respiratory distress, and have been diagnosed by
1 1 Should the child be placed into the Pathway? Asthma Clinical Score (PRAM) Inclusion Children 1 year and 18 years of age who present with wheezing and respiratory distress, and have been diagnosed by
CAMPER APPLICATION PACKET
 CAMPER APPLICATION PACKET Monday- Friday June 12-16, 2017 Rockport, Texas DEADLINE FOR SUBMITTING ALL FORMS: THE IMPORTANCE OF COMPLETING ALL CAMP FORMS Although it may seem like a lot of paperwork, the
CAMPER APPLICATION PACKET Monday- Friday June 12-16, 2017 Rockport, Texas DEADLINE FOR SUBMITTING ALL FORMS: THE IMPORTANCE OF COMPLETING ALL CAMP FORMS Although it may seem like a lot of paperwork, the
2014 Step Therapy Criteria (List of Step Therapy Criteria)
 Criteria Last Updated: November 1, 2014 2014 Step Therapy Criteria (List of Step Therapy Criteria) PLEASE READ CAREFULLY: IEHP MEDICARE DUALCHOICE (HMO SNP) REQUIRES YOU TO FIRST TRY CERTAIN DRUGS TO TREAT
Criteria Last Updated: November 1, 2014 2014 Step Therapy Criteria (List of Step Therapy Criteria) PLEASE READ CAREFULLY: IEHP MEDICARE DUALCHOICE (HMO SNP) REQUIRES YOU TO FIRST TRY CERTAIN DRUGS TO TREAT
Respiratory Medications and Devices Update 2/15
 Respiratory Medications and Devices Update 2/15 Dewey Hahlbohm, PA-C, AE-C Wendy Brown, Pharm.D., MPAS, PA-C, AE-C Objectives! Review mechanism of action for asthma pharmacologic agents! Describe key patient
Respiratory Medications and Devices Update 2/15 Dewey Hahlbohm, PA-C, AE-C Wendy Brown, Pharm.D., MPAS, PA-C, AE-C Objectives! Review mechanism of action for asthma pharmacologic agents! Describe key patient
Pediatric Asthma: Pharmacotherapy. Joseph Spahn, MD Children s Hospital Colorado & University of Colorado Medical School Aurora, Colorado
 Pediatric Asthma: Pharmacotherapy Joseph Spahn, MD Children s Hospital Colorado & University of Colorado Medical School Aurora, Colorado Pediatric Asthma: Pharmacotherapy Disclosures/Conflicts of Interest:
Pediatric Asthma: Pharmacotherapy Joseph Spahn, MD Children s Hospital Colorado & University of Colorado Medical School Aurora, Colorado Pediatric Asthma: Pharmacotherapy Disclosures/Conflicts of Interest:
INHALERS for COPD INTRODUCTION. Types of inhalers. Inhaler technique. MDIs for COPD WET AEROSOLS. Dr Christopher Worsnop
 INHALERS for COPD Dr Christopher Worsnop Department of Respiratory Medicine Austin Hospital INTRODUCTION Most drugs for COPD are given via inhalers. This reduces the dose that needs to be given and delivers
INHALERS for COPD Dr Christopher Worsnop Department of Respiratory Medicine Austin Hospital INTRODUCTION Most drugs for COPD are given via inhalers. This reduces the dose that needs to be given and delivers
FASENRA (benralizumab)
 FASENRA (benralizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
FASENRA (benralizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
