Ameba has two stages of development: cyst and trophozoite
|
|
|
- Daniel Evans
- 6 years ago
- Views:
Transcription
1
2 Amebiasis A parasitic disease of worldwide public health importance Second to malaria in mortality due to protozoan parasites Invasive amebiasis results in up to 100,000 deaths / year Amebiasis is infection with Entamoeba histolytica. Approximately million people infected with E. histolytica, develop amebic colitis or extraintestinal abscesses Infection commonly acquired by ingestion of food or water contaminated with E. histolytica cysts Amebiasis is transmitted through gastrointestinal tract.
3 Ameba has two stages of development: cyst and trophozoite
4 Clinical Forms of Amebic Colitis Amebic dysentery diarrhea with visible blood and mucus (+) E. histolytica trophozoites with ingested red blood cells (hematophagous trophozoite) in stools or tissues Sigmoidoscopic examination: inflamed mucosa with or without discrete ulcers Nondysenteric amebic colitis recurrent bouts of diarrhea with or without mucus but no visible blood (+) E. histolytica cysts or trophozoites with no ingested red blood cells (nonhematophagous trophozoite) in stools sigmoidoscopic examination: normal
5 Drug Treatment of Amebic Colitis Tissue Amebicide: active against invasive forms in the tissues; inconsistent activity against cyst forms Drug of Choice: Metronidazole 35-50mg/kg/day TID x 7-10 days Alternative: Tinidazole 50 mg/kg/day (max 2 g) OD x 3 days Secnidazole Luminal agents - eradicate remaining cysts in the intestinal lumen; recommended after giving a tissue amebicide Diloxanide furoate 20 mg/kg/day TID x 10 days Iodoquinol mg/kg/day TID x 20 days Paromomycin mg/kg/day TID x 10 days Tetracycline
6 Four Principal Steps in the Management of Children with Bloody Diarrhea F - Fluids Prevent dehydration with oral or IV rehydration fluids F - Follow-up Re-evaluate clinical status after 48 hrs F - Feeding Continue provision of nutritious food: breastfeeding; small frequent meals A - Antimicrobial therapy Ideally, antimicrobial treatment should be based on suspected or identified specific bacterial pathogen
7 A nitroimidazole Metronidazole Pharmacokinetics Oral metronidazole is readily absorbed and permeates all tissues by simple diffusion. Protein binding is low (<20%) Metabolizing in liver. Excreted mainly in the urine. Pharmacological Effects and Clinical Uses 1. Anti-amebiasis: kills all tissue infections with E. histolytica trophozoites but not cysts. No effect against luminal parasites and so must be used with a luminal amebicide to ensure eradication of the infection. 2. Anti-trichomoniasis: 3. Anti-giardiasis:
8 MOA Metronidazole kills protozoa & is bactericidal for anaerobic bacteria. Metronidazole is a prodrug. It requires reductive activation of the NITRO group. This occurs in sensitive anaerobic protozoa & anaerobic bacteria by Ferredoxins; which are electron transport proteins. These proteins can donate electrons to Metronidazole, which serves as electron acceptor. The reduced product is cytotoxic, it targets DNA & other bio-molecules/proteins, resulting in cell death. Hence, it kills the micro-organisms
9 Adverse Effects and Cautions Nausea, headache, dry mouth, a metallic taste in the mouth. Infrequent: vomiting, diarrhea, rashes, insomnia, neutropenia, Rare: severe CNS toxicity (ataxia, encephalopathy, seizures) drug withdrawal
10 Tinidazole It is a second- generation Nitroimidazole. Congener of Metronidazole It is similar to Metronidazole in spectrum of activity, MOA, absorption, A/E & D/I. It is also effective against cysts of E. histolytica. It is longer acting once daily dose. Short course 2gm daily, single dose-- for 3 days. Secnidazole Longer acting, Single 2gm dose is given
11 Emetine and Dehydroemetine Emetine (an alkaloid derived from ipecac), and dehydroemetine (a synthetic analog), are effective against tissue trophozoites of E. histolytica. Because of major toxicity concerns they have been almost completely replaced by metronidazole. Administered subcutaneously (preferred) or i.m. (but never i.v.) because oral preparations are absorbed erratically. Mechanisms Inhibiting peptidyl-trna transposition inhibiting elongation of peptide chain inhibiting protein synthesis interfering cleavage and breeding of trophozoites
12 Adverse Effects and Cautions Mild when used for 3-5 days. Toxicity increase with length of therapy. 1. Cardiac toxicity: arrhythmias, CHF, hypotension, ECG changes 2. Neuromuscular blockade: muscle weakness and discomfort 3. Local stimulation: pain and tenderness in the area of injection. 4. Gastrointestinal tract discomfort: nausea, vomiting Not be used in patients with cardiac or renal disease, in young children, or in pregnancy. Pharmacological Effects and Clinical Uses kills E. histolytica trophozoites of histolytic tissues but no effect against luminal trophozoites, a luminal amebicide should also be given. Used to treat amebic dysentery for the minimum period because of toxicity. Occasionally as alternative therapies for amebic liver abscess.
13 Chloroquine Tissue Amebicide specially against Amoebic Hepatitis & Liver Abscess. Concentrated in liver; kills trophozoits of E. histolytica Not effective for amebic colitis or luminal amebae because absorbed in upper intestine. Use : Amebic liver abscess. Hepatic amebiasis / abscess; not responding to Metronidazole Not effective in the treatment of intestinal or other extrahepatic amebiasis.
14 Diloxanide Furoate Diloxanide furoate is a dichloroacetamide derivative. Effective luminal amebicide but is not active against tissue trophozoites. The unabsorbed diloxanide in the gut is the active antiamebic substance. DOC for asymptomatic luminal infections. It is used with a tissue amebicide, usually metronidazole. Adverse Effects: flatulence, nausea, abdominal cramps, rashes, abortion. Precautions: Pregnancy
15 IODOQUINOL Iodoquinol ( Diiodohydroxyquine ) is a halogenated hydroxyquinoline. An effective luminal amobecide used with metronidazole to treat amebic infections. Only effective against trophozoits in lumen. Pharmacokinetics : Poorly understood 90% unabsorbed amebicide. 1 0% absorbed Metabolized to Glucronides, excreted in urine. Half life hrs
16 ADVERSE EFFECTS Diarrhea, anorexia, nausea, vomiting, abdominal pain, headache Iodism : Dermatitis, urticaria, pruritis, fever. Some idoquinol can produce severe neurotoxicity on prolonged use & high doses--- so used with caution CAUTIONS Taken with meals. With caution in: optic neuropathy, Non-amebic Hepatic disease, Renal or Thyroid disease. Contraindication in intolerance to Iodine
17 Paromomycin Aminoglycoside antibiotic. Not significantly absorbed from the GIT. Only as a luminal amebicide and has no effect against extraintestinal amebic infections. Less toxic than other agents inhibiting protein synthesis kill trophozoites Adverse Effects: Abdominal distress and diarrhea
18 Tetracyclines Used as Luminal amebicide. Does not kill bacteria directly but disturbs the symbiosis between normal intestinal flora & E. histolytica. The amebae grow at expense of normal intestinal flora. Tetracyclines are broad spectrum antibiotics & kill these flora leading to death of E. histolytica also. Used in resistant cases
19
PRESCRIBING INFORMATION PRODUCT MONOGRAPH HUMATIN*CAP
 PRESCRIBING INFORMATION PRODUCT MONOGRAPH HUMATIN*CAP (Paromomycin Sulfate Capsules MANUFACTURER STANDARD) 250 mg Capsules ANTIBIOTIC DATE OF PREPARATION January 08, 2001 8250 Décarie Blvd, suite 110 Montréal,
PRESCRIBING INFORMATION PRODUCT MONOGRAPH HUMATIN*CAP (Paromomycin Sulfate Capsules MANUFACTURER STANDARD) 250 mg Capsules ANTIBIOTIC DATE OF PREPARATION January 08, 2001 8250 Décarie Blvd, suite 110 Montréal,
PACKAGE LEAFLET: Information for the patient. METRONIDAZOL Tablets 250 mg Solution for infusion 0.5 % Ovules mg (Metronidazole)
 PACKAGE LEAFLET: Information for the patient METRONIDAZOL Tablets 250 mg Solution for infusion 0.5 % Ovules - 500 mg (Metronidazole) Read this leaflet carefully before you start taking this medicine. Keep
PACKAGE LEAFLET: Information for the patient METRONIDAZOL Tablets 250 mg Solution for infusion 0.5 % Ovules - 500 mg (Metronidazole) Read this leaflet carefully before you start taking this medicine. Keep
Bacillary Dysentery (Shigellosis)
 Bacillary Dysentery (Shigellosis) An acute bacterial disease involving the large and distal small intestine, caused by the bacteria of the genus shigella. Infectious agent Shigella is comprised of four
Bacillary Dysentery (Shigellosis) An acute bacterial disease involving the large and distal small intestine, caused by the bacteria of the genus shigella. Infectious agent Shigella is comprised of four
METRONIDAZOLE. General Monograph, Antibacterial - Antiprotozoal
 METRONIDAZOLE General Monograph, Antibacterial - Antiprotozoal Pharmacology: Metronidazole is amebicidal, trichomonacidal and bactericidal. A chemically reactive, reduced form of metronidazole is thought
METRONIDAZOLE General Monograph, Antibacterial - Antiprotozoal Pharmacology: Metronidazole is amebicidal, trichomonacidal and bactericidal. A chemically reactive, reduced form of metronidazole is thought
Entamoeba histolytica/e. dispar. A. Haghighi,
 Entamoeba histolytica/e. dispar A. Haghighi, Wednesday, February 14, 2018 Classification of Protozoa? The protozoa are generally unicellular and may be divided for convenience, into four distinct groups
Entamoeba histolytica/e. dispar A. Haghighi, Wednesday, February 14, 2018 Classification of Protozoa? The protozoa are generally unicellular and may be divided for convenience, into four distinct groups
Amoebiasis. (Amoebic dysentery)
 Amoebiasis (Amoebic dysentery) Causative agent: Entamoeba histolytica Amoebiasis Harbouring of protozoa E. histolytica inside the body with or without disease only 10% of infected develop disease two types
Amoebiasis (Amoebic dysentery) Causative agent: Entamoeba histolytica Amoebiasis Harbouring of protozoa E. histolytica inside the body with or without disease only 10% of infected develop disease two types
Entamoeba histolytica
 Entamoeba histolytica cosmopolitan distribution no animal reservoirs facultative pathogen most clear the infection spontaneous in 6-12 months with mild or no symptoms can cause a serious invasive disease
Entamoeba histolytica cosmopolitan distribution no animal reservoirs facultative pathogen most clear the infection spontaneous in 6-12 months with mild or no symptoms can cause a serious invasive disease
PERSISTENT DIARRHOEA. IAP UG Teaching slides
 PERSISTENT DIARRHOEA 1 DEFINITION Prolongation of acute diarrhoea / dysentery for more than 14 days Generally associated with weight loss. 2 PROTRACTED DIARRHOEA Prolongation of acute diarrhoea or dysentery
PERSISTENT DIARRHOEA 1 DEFINITION Prolongation of acute diarrhoea / dysentery for more than 14 days Generally associated with weight loss. 2 PROTRACTED DIARRHOEA Prolongation of acute diarrhoea or dysentery
3-Mutualism: It is a relationship between two. organisms; one called. or harmless. 2-Commensalism: It is a relationship in which one
 Host-Parasite relationships Symbiosis: It is the permanent relationship between two dissimilar organisms which depending each others. They are three types 1- Parasitism : It is a relationship between two
Host-Parasite relationships Symbiosis: It is the permanent relationship between two dissimilar organisms which depending each others. They are three types 1- Parasitism : It is a relationship between two
MOVICOL HALF PI December MOVICOL-Half. Powder for Solution (macrogol 3350) Potassium 5.4 mmol/l. Bicarbonate 17 mmol/l
 MOVICOL -Half Powder for Solution (macrogol 3350) Product Name: Product Description: MOVICOL-Half Each sachet of MOVICOL-Half contains: Macrogol 3350 6.563 g Sodium chloride 175.4 mg Sodium bicarbonate
MOVICOL -Half Powder for Solution (macrogol 3350) Product Name: Product Description: MOVICOL-Half Each sachet of MOVICOL-Half contains: Macrogol 3350 6.563 g Sodium chloride 175.4 mg Sodium bicarbonate
Schistosome life cycle.
 Schistosomiasis infects approximately 200 million persons and kills approximately 280,000 annually. Most of the mortality comes from hepatic granulomas and fibrosis Schistosoma japonicum and Schistosoma
Schistosomiasis infects approximately 200 million persons and kills approximately 280,000 annually. Most of the mortality comes from hepatic granulomas and fibrosis Schistosoma japonicum and Schistosoma
Immodium / loprarmide
 Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
Labeled Uses: Treatment of Clostiridum Difficile associated diarrhea (CDAD)
 Brand Name: Dificid Generic Name: fidaxomicin Manufacturer 1,2,3,4,5 : Optimer Pharmaceuticals, Inc. Drug Class 1,2,3,4,5 : Macrolide Antibiotic Uses 1,2,3,4,5 : Labeled Uses: Treatment of Clostiridum
Brand Name: Dificid Generic Name: fidaxomicin Manufacturer 1,2,3,4,5 : Optimer Pharmaceuticals, Inc. Drug Class 1,2,3,4,5 : Macrolide Antibiotic Uses 1,2,3,4,5 : Labeled Uses: Treatment of Clostiridum
CHAPTER FOUR. Intestinal Amebae
 PART II Protozoa CHAPTER FOUR Intestinal Amebae OUTLINE INTESTINAL AMEBAE Entamoeba histolytica/entamoeba dispar (EN-ta-MEE-buh HIS-toe-LIT-I-ka/EN-ta-MEEbuh DIS-par) Entamoeba hartmanni (EN-ta-MEE-buh
PART II Protozoa CHAPTER FOUR Intestinal Amebae OUTLINE INTESTINAL AMEBAE Entamoeba histolytica/entamoeba dispar (EN-ta-MEE-buh HIS-toe-LIT-I-ka/EN-ta-MEEbuh DIS-par) Entamoeba hartmanni (EN-ta-MEE-buh
Cryptosporidium parvum. Cyclospora cayetanensis. Isospora belli. Entamoeba histolytica.
 Cryptosporidium parvum Cyclospora cayetanensis Isospora belli Entamoeba histolytica Giardia lamblia cellular immunity humoral immunity microsporidia Trichomonas vaginalis 12 64 http://www.aids-care.org.tw
Cryptosporidium parvum Cyclospora cayetanensis Isospora belli Entamoeba histolytica Giardia lamblia cellular immunity humoral immunity microsporidia Trichomonas vaginalis 12 64 http://www.aids-care.org.tw
Foodborne Disease in the Region of Peel
 Foodborne Disease in the Region of Peel HIGHLIGHTS The incidence of selected foodborne diseases was generally higher in Peel than in Ontario between 1993 and 22. A higher incidence was observed in Peel
Foodborne Disease in the Region of Peel HIGHLIGHTS The incidence of selected foodborne diseases was generally higher in Peel than in Ontario between 1993 and 22. A higher incidence was observed in Peel
Giardiasis. Table of Contents
 Table of Contents Case Definition... Error! Bookmark not defined. Reporting Requirements... 2 Etiology... Error! Bookmark not defined. Clinical Presentation... Error! Bookmark not defined. Diagnosis...
Table of Contents Case Definition... Error! Bookmark not defined. Reporting Requirements... 2 Etiology... Error! Bookmark not defined. Clinical Presentation... Error! Bookmark not defined. Diagnosis...
MOVICOL Junior Powder for Solution (macrogol 3350)
 MOVICOL Junior Powder for Solution (macrogol 3350) Product Name: MOVICOL Junior Product Description: Each sachet of MOVICOL Junior contains: Macrogol 3350 Sodium chloride Sodium bicarbonate Potassium chloride
MOVICOL Junior Powder for Solution (macrogol 3350) Product Name: MOVICOL Junior Product Description: Each sachet of MOVICOL Junior contains: Macrogol 3350 Sodium chloride Sodium bicarbonate Potassium chloride
SUMMARY OF PRODUCT CHARACTERISTICS. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains the following active ingredients:
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Lacrofarm, powder for oral solution, sachet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains the following active ingredients:
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Lacrofarm, powder for oral solution, sachet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains the following active ingredients:
Non_ pathogenic Amoeba of humans:
 The parasite protozoa Phylum: Sarcomastigophora Sarcodina االميبات Amoebae Non_ pathogenic Amoeba of humans: 1. Entamoeba hartmanii, contain trophozoite and cyst 2. Entamoeba coli, cyst and trophozoite
The parasite protozoa Phylum: Sarcomastigophora Sarcodina االميبات Amoebae Non_ pathogenic Amoeba of humans: 1. Entamoeba hartmanii, contain trophozoite and cyst 2. Entamoeba coli, cyst and trophozoite
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Lacrofarm Junior, powder for oral solution, sachet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet of contains the following active
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Lacrofarm Junior, powder for oral solution, sachet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet of contains the following active
NURSING PROCESS FOCUS: Patients Receiving Amphotericin B (Fungizone, Abelcet)
 NURSING PROCESS FOCUS: Patients Receiving Amphotericin B (Fungizone, Abelcet) ASSESSMENT Prior to administration: Obtain complete health history including allergies, drug history, and possible drug interactions.
NURSING PROCESS FOCUS: Patients Receiving Amphotericin B (Fungizone, Abelcet) ASSESSMENT Prior to administration: Obtain complete health history including allergies, drug history, and possible drug interactions.
MOVICOL Junior Chocolate Flavour Powder for Solution (macrogol 3350)
 MOVICOL Junior Chocolate Flavour Powder for Solution (macrogol 3350) Product Name: MOVICOL Junior Chocolate Flavour Product Description: Each sachet of MOVICOL Junior Chocolate contains: Macrogol 3350
MOVICOL Junior Chocolate Flavour Powder for Solution (macrogol 3350) Product Name: MOVICOL Junior Chocolate Flavour Product Description: Each sachet of MOVICOL Junior Chocolate contains: Macrogol 3350
MOVICOL Lemon-Lime Flavour Powder for Solution (macrogol 3350)
 MOVICOL Lemon-Lime Flavour Powder for Solution (macrogol 3350) Product Name: MOVICOL Lemon-Lime Flavour Product Description: Each sachet of MOVICOL Lemon-Lime contains: Macrogol 3350 Sodium chloride Sodium
MOVICOL Lemon-Lime Flavour Powder for Solution (macrogol 3350) Product Name: MOVICOL Lemon-Lime Flavour Product Description: Each sachet of MOVICOL Lemon-Lime contains: Macrogol 3350 Sodium chloride Sodium
It is insoluble in water, but soluble in methanol and chloroform.
 PRODUCT INFORMATION FASIGYN (Tinidazole) DESCRIPTION Fasigyn (tinidazole) is a derivative of the substituted imidazole group of compounds. It is a pale yellow crystalline solid with the following structural
PRODUCT INFORMATION FASIGYN (Tinidazole) DESCRIPTION Fasigyn (tinidazole) is a derivative of the substituted imidazole group of compounds. It is a pale yellow crystalline solid with the following structural
MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350)
 MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350) NAME OF THE MEDICINE: MOVICOL Liquid Orange Flavour, Concentrate for Oral Solution. DESCRIPTION: A clear colourless solution.
MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350) NAME OF THE MEDICINE: MOVICOL Liquid Orange Flavour, Concentrate for Oral Solution. DESCRIPTION: A clear colourless solution.
PFIZER INC. Study Initiation Date and Primary Completion or Completion Dates: March 1991 to August 1991
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Parasite Organism Chart Parasite Description Habitat/Sources of Isolation Blastocystis hominis
 Blastocystis hominis B. hominis has recently been reclassified as a protozoan, of which there are thought to be four separate serologic groups. 1 This organism is transmitted via the fecal-oral route or
Blastocystis hominis B. hominis has recently been reclassified as a protozoan, of which there are thought to be four separate serologic groups. 1 This organism is transmitted via the fecal-oral route or
Many of you asked about this topic
 Many of you asked about this topic Water borne disease: The largest water quality problem in developing countries. CEE 3510 Environmental Quality Engineering According to: Chemical and Engineering News
Many of you asked about this topic Water borne disease: The largest water quality problem in developing countries. CEE 3510 Environmental Quality Engineering According to: Chemical and Engineering News
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Compound Macrogol 13.72 g powder for oral solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet of Compound Macrogol 13.72 g
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Compound Macrogol 13.72 g powder for oral solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet of Compound Macrogol 13.72 g
AXITAB-CV TAB. COMPOSITION :
 AXITAB-CV TAB. COMPOSITION : Each film coated tablet contains: Cefuroxime Axetil I.P. Eq. to Anhydrous 500mg. Potassium Clavulanate Diluted I.P. Eq. to Clavulanic Acid 125mg DESCRIPTION : Cefuroxime Axetil
AXITAB-CV TAB. COMPOSITION : Each film coated tablet contains: Cefuroxime Axetil I.P. Eq. to Anhydrous 500mg. Potassium Clavulanate Diluted I.P. Eq. to Clavulanic Acid 125mg DESCRIPTION : Cefuroxime Axetil
Pathogenic amoebae and ciliate. Dr. Narissara Jariyapan Department of Parasitology Faculty of Medicine Chiang Mai University
 Pathogenic amoebae and ciliate Dr. Narissara Jariyapan Department of Parasitology Faculty of Medicine Chiang Mai University Objectives After the lecture, students must know 1. General morphology of pathogenic
Pathogenic amoebae and ciliate Dr. Narissara Jariyapan Department of Parasitology Faculty of Medicine Chiang Mai University Objectives After the lecture, students must know 1. General morphology of pathogenic
Alberta Health and Wellness Public Health Notifiable Disease Management Guidelines August 2011
 August 2011 Amoebiasis Revision Dates Case Definition Reporting Requirements Remainder of the Guideline (i.e., Etiology to References sections inclusive) Case Definition August 2011 August 2011 October
August 2011 Amoebiasis Revision Dates Case Definition Reporting Requirements Remainder of the Guideline (i.e., Etiology to References sections inclusive) Case Definition August 2011 August 2011 October
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia)
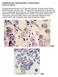 PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
TYPES OF ORGANISM RELATIONSHIPS
 TYPES OF ORGANISM RELATIONSHIPS Normal Flora. Normal flora consists of microorganisms that are normally and consistently found in or on the body in the absence of disease. Symbiosis. This is the close
TYPES OF ORGANISM RELATIONSHIPS Normal Flora. Normal flora consists of microorganisms that are normally and consistently found in or on the body in the absence of disease. Symbiosis. This is the close
2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet of Macrovic Junior powder for oral solution contains the following active ingredients:
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Macrovic Junior powder for oral solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet of Macrovic Junior powder for oral solution
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Macrovic Junior powder for oral solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet of Macrovic Junior powder for oral solution
PACKAGE INSERT USP ANTIBIOTIC
 Pr AMPICILLIN for Injection USP ANTIBIOTIC ACTIONS AND CLINICAL PHARMACOLOGY Ampicillin has a broad spectrum of bactericidal activity against many gram-positive and gramnegative aerobic and anaerobic bacteria.
Pr AMPICILLIN for Injection USP ANTIBIOTIC ACTIONS AND CLINICAL PHARMACOLOGY Ampicillin has a broad spectrum of bactericidal activity against many gram-positive and gramnegative aerobic and anaerobic bacteria.
COMPLETE DIGESTIVE STOOL ANALYSIS - Level 5
 COMPLETE DIGESTIVE STOOL ANALYSIS - Level 5 MACROSCOPIC DESCRIPTION Stool Colour Brown Result Range Brown Markers Colour - Brown is the colour of normal stool. Other colours may indicate abnormal GIT conditions.
COMPLETE DIGESTIVE STOOL ANALYSIS - Level 5 MACROSCOPIC DESCRIPTION Stool Colour Brown Result Range Brown Markers Colour - Brown is the colour of normal stool. Other colours may indicate abnormal GIT conditions.
MESALO Foam (Mesalazine)
 Published on: 10 Jul 2014 MESALO Foam (Mesalazine) Composition MESALO Foam Each actuation delivers: Mesalazine, BP.... 1.0 g White-grayish to slightly reddish-violet, creamy, firm foam. Dosage Form Rectal
Published on: 10 Jul 2014 MESALO Foam (Mesalazine) Composition MESALO Foam Each actuation delivers: Mesalazine, BP.... 1.0 g White-grayish to slightly reddish-violet, creamy, firm foam. Dosage Form Rectal
EMILOK Global. (omeprazole) Composition: Each capsule contains 20 mg omeprazole as enteric-coated
 EMILOK Global (omeprazole) Composition: Each capsule contains 20 mg omeprazole as enteric-coated granules. Properties: Emilok (omeprazole) belongs to the group of proton pump inhibitors, inhibits both
EMILOK Global (omeprazole) Composition: Each capsule contains 20 mg omeprazole as enteric-coated granules. Properties: Emilok (omeprazole) belongs to the group of proton pump inhibitors, inhibits both
SUCRALFATE TABLETS, USP
 1234567890 10 210002-01 SUCRALFATE TABLETS, USP DESCRIPTION Sucralfate is an -D-glucopyranoside, -D-fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex. It has the following structural formula:
1234567890 10 210002-01 SUCRALFATE TABLETS, USP DESCRIPTION Sucralfate is an -D-glucopyranoside, -D-fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex. It has the following structural formula:
SUCRALFATE TABLETS, USP
 1234567890 10 210002 SUCRALFATE TABLETS, USP DESCRIPTION Sucralfate is an -D-glucopyranoside, -D-fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex. It has the following structural formula:
1234567890 10 210002 SUCRALFATE TABLETS, USP DESCRIPTION Sucralfate is an -D-glucopyranoside, -D-fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex. It has the following structural formula:
Antimicrobials; antibacterials, antifungals, antiprotozoans, antivirals, and antihelminthics 6
 Antimicrobials; antibacterials, antifungals, antiprotozoans, antivirals, and antihelminthics 6 Inhibition of Ergosterol Synthesis Azoles Miconazole Triazoles Allylamines For azole-resistant infections
Antimicrobials; antibacterials, antifungals, antiprotozoans, antivirals, and antihelminthics 6 Inhibition of Ergosterol Synthesis Azoles Miconazole Triazoles Allylamines For azole-resistant infections
CASE 2 - PARASITOLOGY ENTAMOEBA HISTOLYTICA DR. SALAMA ABD MEGUID
 CASE 2 - PARASITOLOGY ENTAMOEBA HISTOLYTICA DR. SALAMA ABD MEGUID G/D Host Reserv Infect Stage MOT Division Diagnos Stage Life Cycle Worldwide but especially Africa, China, Latin America and South East
CASE 2 - PARASITOLOGY ENTAMOEBA HISTOLYTICA DR. SALAMA ABD MEGUID G/D Host Reserv Infect Stage MOT Division Diagnos Stage Life Cycle Worldwide but especially Africa, China, Latin America and South East
Children Enteric coated tablet : 1-3 mg/kg per day in divided doses.
 Ultrafen Tablet/SR Tablet/Suppository/Gel Description Ultrafen is a preparation of Diclofenac is a non-steroidal antiinflammatory agent with marked analgesic, anti-inflammatory and antipyretic properties.
Ultrafen Tablet/SR Tablet/Suppository/Gel Description Ultrafen is a preparation of Diclofenac is a non-steroidal antiinflammatory agent with marked analgesic, anti-inflammatory and antipyretic properties.
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT TRANSISOFT 8.5 g powder for oral solution in sachet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains 8.5 g of macrogol
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT TRANSISOFT 8.5 g powder for oral solution in sachet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains 8.5 g of macrogol
Clinical approach to evaluate infectious diarrhea. Diarrhea. Defect Stool exam Examples mechanism. stool
 Clinical approach to evaluate infectious diarrhea Diarrhea Mechanism Clinical manifestation Having three of more loose or liquid stools per day, or having more stools than normal for that person 1ry Defect
Clinical approach to evaluate infectious diarrhea Diarrhea Mechanism Clinical manifestation Having three of more loose or liquid stools per day, or having more stools than normal for that person 1ry Defect
SUMMARY OF THE PRODUCT CHARACTERISTICS. The content of electrolyte ions per sachet when made up to 125 ml of solution.
 SUMMARY OF THE PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Molaxole powder for oral solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains following active substances Macrogol
SUMMARY OF THE PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Molaxole powder for oral solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each sachet contains following active substances Macrogol
Malaria. benign (mild) malaria
 Malaria Caused by the plasmodium protozoa. Four species of plasmodium causes human malaria: Plasmodium falciparum responsible for nearly all serious complications and deaths. P. vivax P. malariae P. ovale
Malaria Caused by the plasmodium protozoa. Four species of plasmodium causes human malaria: Plasmodium falciparum responsible for nearly all serious complications and deaths. P. vivax P. malariae P. ovale
Pharmaceutical form(s)/strength: Capsules, 200mg, 400mg, Oral suspensions, 90mg/5ml, 180mg/5ml, 36 mg/ml SI/H/PSUR/0002/002 Date of FAR:
 0BCore Safety Profile Active substance: Ceftibuten Pharmaceutical form(s)/strength: Capsules, 200mg, 400mg, Oral suspensions, 90mg/5ml, 180mg/5ml, 36 mg/ml P-RMS: SI/H/PSUR/0002/002 Date of FAR: 14.02.2013
0BCore Safety Profile Active substance: Ceftibuten Pharmaceutical form(s)/strength: Capsules, 200mg, 400mg, Oral suspensions, 90mg/5ml, 180mg/5ml, 36 mg/ml P-RMS: SI/H/PSUR/0002/002 Date of FAR: 14.02.2013
Introductory Clinical Pharmacology Chapter 32 Antiparkinsonism Drugs
 Introductory Clinical Pharmacology Chapter 32 Antiparkinsonism Drugs Dopaminergic Drugs: Actions Symptoms of parkinsonism are caused by depletion of dopamine in CNS Amantadine: makes more of dopamine available
Introductory Clinical Pharmacology Chapter 32 Antiparkinsonism Drugs Dopaminergic Drugs: Actions Symptoms of parkinsonism are caused by depletion of dopamine in CNS Amantadine: makes more of dopamine available
Product Information. Powder for Oral Solution. DESCRIPTION: The ingredients of MOVIPREP ORANGE are contained in two separate sachets:
 Product Information NAME OF THE MEDICINE: MOVIPREP ORANGE MOVIPREP ORANGE Powder for Oral Solution DESCRIPTION: The ingredients of MOVIPREP ORANGE are contained in two separate sachets: Sachet A: Macrogol
Product Information NAME OF THE MEDICINE: MOVIPREP ORANGE MOVIPREP ORANGE Powder for Oral Solution DESCRIPTION: The ingredients of MOVIPREP ORANGE are contained in two separate sachets: Sachet A: Macrogol
Antimalarial Drugs. Munir Gharaibeh, MD, PhD, MHPE Department of Pharmacology Faculty of Medicine October 2014
 Antimalarial Drugs Munir Gharaibeh, MD, PhD, MHPE Department of Pharmacology Faculty of Medicine October 2014 Malaria Annual Global Incidence: 219 million in 2010. Annually, in Africa, I million children
Antimalarial Drugs Munir Gharaibeh, MD, PhD, MHPE Department of Pharmacology Faculty of Medicine October 2014 Malaria Annual Global Incidence: 219 million in 2010. Annually, in Africa, I million children
Flagellates I Genito-urinary & Intestinal flagellates
 Flagellates I Genito-urinary & Intestinal flagellates Dr. Anuluck Junkum PARA 317221 Objective Can describe the morphology, life cycle, pathology, diagnosis and prevention of pathogenic flagellate Classification
Flagellates I Genito-urinary & Intestinal flagellates Dr. Anuluck Junkum PARA 317221 Objective Can describe the morphology, life cycle, pathology, diagnosis and prevention of pathogenic flagellate Classification
Bacteriology Virology Mycology Parasitology Immunology Pathology Epidemiology. Microbe alternate terms Germs, microorganisms, pathogens, agents, bugs
 1 BIO 205 Microbiology for Health Professionals The study of microscopic agents as they relate to human health The causes of infectious disease. The control of infectious disease. Host defenses against
1 BIO 205 Microbiology for Health Professionals The study of microscopic agents as they relate to human health The causes of infectious disease. The control of infectious disease. Host defenses against
PHARMACOLOGY II. Dr Shariq Syed Associate Professor AIKTC, SoP
 PHARMACOLOGY II Dr Shariq Syed Associate Professor AIKTC, SoP INTRODUCTION TO BACTERIA! INTRODUCTION TO BACTERIA! THEY COME IN DIFFERENT SHAPES ANTIMICROBIAL SITES OF ACTION SULPHONAMIDES 1930, Physician/researcher
PHARMACOLOGY II Dr Shariq Syed Associate Professor AIKTC, SoP INTRODUCTION TO BACTERIA! INTRODUCTION TO BACTERIA! THEY COME IN DIFFERENT SHAPES ANTIMICROBIAL SITES OF ACTION SULPHONAMIDES 1930, Physician/researcher
MOVIPREP Powder for Solution
 Product Information MOVIPREP Powder for Solution NAME OF THE MEDICINE: MOVIPREP DESCRIPTION: The ingredients of MOVIPREP are contained in two separate sachets: Sachet A: Macrogol 3350 Sodium sulfate anhydrous
Product Information MOVIPREP Powder for Solution NAME OF THE MEDICINE: MOVIPREP DESCRIPTION: The ingredients of MOVIPREP are contained in two separate sachets: Sachet A: Macrogol 3350 Sodium sulfate anhydrous
0BCore Safety Profile. Pharmaceutical form(s)/strength: Losec MUPS tablets 10, 20 mg (OTC) NL/H/PSUR/0058/001 Date of FAR:
 0BCore Safety Profile Active substance: Omeprazole Pharmaceutical form(s)/strength: Losec MUPS tablets 10, 20 mg (OTC) P-RMS: NL/H/PSUR/0058/001 Date of FAR: 13.06.2013 4.2 Posology and method of administration
0BCore Safety Profile Active substance: Omeprazole Pharmaceutical form(s)/strength: Losec MUPS tablets 10, 20 mg (OTC) P-RMS: NL/H/PSUR/0058/001 Date of FAR: 13.06.2013 4.2 Posology and method of administration
Flagyl 500 mg dosage 5 days
 Search Flagyl 500 mg dosage 5 days 2000 mg once daily for 3 days, or 400 mg. three times daily for 5 days, or 500 mg twice daily for 7 to 10 days.. The dosage of metronidazole, therefore,. metronidazole
Search Flagyl 500 mg dosage 5 days 2000 mg once daily for 3 days, or 400 mg. three times daily for 5 days, or 500 mg twice daily for 7 to 10 days.. The dosage of metronidazole, therefore,. metronidazole
INTESTINAL PROTOZOAN INFECTIONS
 INTESTINAL PROTOZOAN INFECTIONS The protozoa that parasitise the human intestine belong to four groups: amoebae, flagellates, ciliates and coccidia. In general, the intestinal amoebae, flagellates and
INTESTINAL PROTOZOAN INFECTIONS The protozoa that parasitise the human intestine belong to four groups: amoebae, flagellates, ciliates and coccidia. In general, the intestinal amoebae, flagellates and
2. QUALITATIVE AND QUANTITATIVE COMPOSITION One sachet contains 5.631g fosfomycin trometamol equivalent to 3.0 g fosfomycin.
 Doctor leaflet 1. NAME OF THE MEDICINAL PRODUCT MONUROL 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One sachet contains 5.631g fosfomycin trometamol equivalent to 3.0 g fosfomycin. Excipients: One sachet
Doctor leaflet 1. NAME OF THE MEDICINAL PRODUCT MONUROL 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One sachet contains 5.631g fosfomycin trometamol equivalent to 3.0 g fosfomycin. Excipients: One sachet
VANLID Capsules (Vancomycin hydrochloride)
 Published on: 22 Sep 2014 VANLID Capsules (Vancomycin hydrochloride) Composition VANLID Capsules Each capsule contains: Vancomycin Hydrochloride IP equivalent to Vancomycin.. 250 mg Approved colours used
Published on: 22 Sep 2014 VANLID Capsules (Vancomycin hydrochloride) Composition VANLID Capsules Each capsule contains: Vancomycin Hydrochloride IP equivalent to Vancomycin.. 250 mg Approved colours used
College of Pharmacy Advanced Clinical Pharmacology Spring 2013 Semester. Antibiotics PCN. Danita 1 Dee Narciso Pharm D
 College of Pharmacy Advanced Clinical Pharmacology Spring 2013 Semester Antibiotics PCN Danita 1 Dee Narciso Pharm D 2 Objectives Recognize the signs and symptoms of infection Become familiar with antibiotics
College of Pharmacy Advanced Clinical Pharmacology Spring 2013 Semester Antibiotics PCN Danita 1 Dee Narciso Pharm D 2 Objectives Recognize the signs and symptoms of infection Become familiar with antibiotics
Alberta Health and Wellness Public Health Notifiable Disease Management Guidelines August 2011
 August 2011 Giardiasis Revision Dates Case Definition Reporting Requirements Remainder of the Guideline (i.e., Etiology to References sections inclusive) Case Definition August 2011 August 2011 October
August 2011 Giardiasis Revision Dates Case Definition Reporting Requirements Remainder of the Guideline (i.e., Etiology to References sections inclusive) Case Definition August 2011 August 2011 October
Thyroid Hormones (T 4 & T 3 )
 1 Thyroid Hormones (T 4 & T 3 ) Normalize growth and development, body temperature, and energy levels. Used as thyroid replacement therapy in hypothyroidism. Thyroxine (T 4 ) is peripherally metabolized
1 Thyroid Hormones (T 4 & T 3 ) Normalize growth and development, body temperature, and energy levels. Used as thyroid replacement therapy in hypothyroidism. Thyroxine (T 4 ) is peripherally metabolized
Case Report Primary Pulmonary Amebiasis Complicated with Multicystic Empyema
 Case Reports in Pulmonology Volume 2016, Article ID 8709347, 4 pages http://dx.doi.org/10.1155/2016/8709347 Case Report Primary Pulmonary Amebiasis Complicated with Multicystic Empyema Ali Zakaria, Bayan
Case Reports in Pulmonology Volume 2016, Article ID 8709347, 4 pages http://dx.doi.org/10.1155/2016/8709347 Case Report Primary Pulmonary Amebiasis Complicated with Multicystic Empyema Ali Zakaria, Bayan
PRUFLOX Tablets (Prulifloxacin)
 Published on: 10 Jul 2014 PRUFLOX Tablets (Prulifloxacin) Composition Each Film coated tablet contains: Prulifloxacin... 600 mg Colours: Red Oxide of Iron & Titanium Dioxide IP Dosage Form Tablet Pharmacology
Published on: 10 Jul 2014 PRUFLOX Tablets (Prulifloxacin) Composition Each Film coated tablet contains: Prulifloxacin... 600 mg Colours: Red Oxide of Iron & Titanium Dioxide IP Dosage Form Tablet Pharmacology
Annex III. Amendments to relevant sections of the Product Information
 Annex III Amendments to relevant sections of the Product Information Note: These amendments to the relevant sections of the product information are the outcome of the referral procedure. The product information
Annex III Amendments to relevant sections of the Product Information Note: These amendments to the relevant sections of the product information are the outcome of the referral procedure. The product information
LESSON ASSIGNMENT. Intestinal Parasites and Antiparasitic Agents.
 LESSON ASSIGNMENT LESSON 2 Intestinal Parasites and Antiparasitic Agents. TEXT ASSIGNMENT Paragraphs 2-1 through 2-7. 2-1. Given an important term pertaining to intestinal parasites and a group of statements,
LESSON ASSIGNMENT LESSON 2 Intestinal Parasites and Antiparasitic Agents. TEXT ASSIGNMENT Paragraphs 2-1 through 2-7. 2-1. Given an important term pertaining to intestinal parasites and a group of statements,
Helicobacter 2008;13:1-6. Am J Gastroent 2007;102: Am J of Med 2004;117:31-35.
 An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
Sulaimani University College of Pharmacy. Medical Parasitology
 Dr. Abdullah A. Hama Microbiology/ parasitology and virology ( Chapter 1) Text book: 7-695-57059-1-978 Sulaimani University College of Pharmacy Medical Parasitology Lec. 2 part 1 Protozoa/ class: Sarcodina
Dr. Abdullah A. Hama Microbiology/ parasitology and virology ( Chapter 1) Text book: 7-695-57059-1-978 Sulaimani University College of Pharmacy Medical Parasitology Lec. 2 part 1 Protozoa/ class: Sarcodina
Giardia lamblia (flagellates)
 Giardia lamblia (flagellates) Dr. Hala Al Daghistani Giardia lamblia (Giardia duodenalis or Giardia intestinalis) is the causative agent of giardiasis and is the only common pathogenic protozoan found
Giardia lamblia (flagellates) Dr. Hala Al Daghistani Giardia lamblia (Giardia duodenalis or Giardia intestinalis) is the causative agent of giardiasis and is the only common pathogenic protozoan found
ccess safe drinking wa r is everyone s right Protozoans that cause diarrheal disease
 ccess safe drinking wa r is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
ccess safe drinking wa r is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
Anas Raed. - Zaid Emad. - Malik Zuhlof
 - 6 - Anas Raed - Zaid Emad - Malik Zuhlof 1 P a g e - This lecture started with the excellent presentation of chronic fatigue syndrome and the role of Vitamin B12 in its treatment by our colleague Osama
- 6 - Anas Raed - Zaid Emad - Malik Zuhlof 1 P a g e - This lecture started with the excellent presentation of chronic fatigue syndrome and the role of Vitamin B12 in its treatment by our colleague Osama
M0BCore Safety Profile
 M0BCore Safety Profile Active substance: Aciclovir Pharmaceutical form(s)/strength: Tablets 200, 400 or 800 mg Dispersible tablets 200, 400 or 800 mg Oral suspensions 200 mg or 400 mg per 5 ml. Freeze
M0BCore Safety Profile Active substance: Aciclovir Pharmaceutical form(s)/strength: Tablets 200, 400 or 800 mg Dispersible tablets 200, 400 or 800 mg Oral suspensions 200 mg or 400 mg per 5 ml. Freeze
Treatment of Inflammatory Bowel Disease. Michael Weiss MD, FACG
 Treatment of Inflammatory Bowel Disease Michael Weiss MD, FACG What is IBD? IBD is an immune-mediated chronic intestinal disorder, characterized by chronic or relapsing inflammation within the GI tract.
Treatment of Inflammatory Bowel Disease Michael Weiss MD, FACG What is IBD? IBD is an immune-mediated chronic intestinal disorder, characterized by chronic or relapsing inflammation within the GI tract.
Contrast Materials Patient Safety: What are contrast materials and how do they work?
 Contrast Materials Patient Safety: What are contrast materials and how do they work? Which imaging exams use contrast materials? How safe are contrast materials? How should I prepare for my imaging procedure
Contrast Materials Patient Safety: What are contrast materials and how do they work? Which imaging exams use contrast materials? How safe are contrast materials? How should I prepare for my imaging procedure
Parasitology. Lab. Amoeba
 Parasitology. Lab. Kingdom : Protista Subkingdom : Protozoa Phylum : Sacromastigophora Subphylum : Sarcodina Superclass : Rhizopoda Class : Lobosea Order : Amoebida Amoeba Protozoa Amoebae geneus Entamoeba
Parasitology. Lab. Kingdom : Protista Subkingdom : Protozoa Phylum : Sacromastigophora Subphylum : Sarcodina Superclass : Rhizopoda Class : Lobosea Order : Amoebida Amoeba Protozoa Amoebae geneus Entamoeba
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Omnilax 10 g powder for oral solution, sachet 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One sachet contains 10 g of macrogol 4000.
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Omnilax 10 g powder for oral solution, sachet 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One sachet contains 10 g of macrogol 4000.
Beta-lactamase production should have no effect on Azithromycin activity.
 AZIMEX Composition Azimex 250 Capsules Each capsule contains Azithromycin (as dihydrate) 250 mg Capsules Azimex 500 mg Capsules Each capsule contains Azithromycin (as dihydrate) 500 mg Action Azithromycin
AZIMEX Composition Azimex 250 Capsules Each capsule contains Azithromycin (as dihydrate) 250 mg Capsules Azimex 500 mg Capsules Each capsule contains Azithromycin (as dihydrate) 500 mg Action Azithromycin
PRESCRIBING INFORMATION mg primaquine phosphate equivalent to 15 mg primaquine base. Antimalarial
 PRESCRIBING INFORMATION Pr PRIMAQUINE primaquine phosphate tablets USP 26.3 mg primaquine phosphate equivalent to 15 mg primaquine base Antimalarial sanofi-aventis Canada Inc. 2150 St. Elzear Blvd. West
PRESCRIBING INFORMATION Pr PRIMAQUINE primaquine phosphate tablets USP 26.3 mg primaquine phosphate equivalent to 15 mg primaquine base Antimalarial sanofi-aventis Canada Inc. 2150 St. Elzear Blvd. West
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Fexofenadine Cipla 120 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 120 mg fexofenadine
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Fexofenadine Cipla 120 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 120 mg fexofenadine
Annex I: Proposed Core Safety Profile (CSP) 4.3 Contraindications
 Annex I: Proposed Core Safety Profile (CSP) 4.3 Contraindications Hypersensitivity to cefuroxime or to any of the excipients listed in section 6.1. Patients with known hypersensitivity to cephalosporin
Annex I: Proposed Core Safety Profile (CSP) 4.3 Contraindications Hypersensitivity to cefuroxime or to any of the excipients listed in section 6.1. Patients with known hypersensitivity to cephalosporin
33. I will recommend this primer to my colleagues. A. Strongly Agree D. Disagree B. Agree E. Strongly Disagree C. Neither agree nor disagree
 27. The primer increased my ability to recognize foodborne illnesses and increased the likelihood that I will consider such illnesses in my patients. 28. The primer increased my knowledge and skills in
27. The primer increased my ability to recognize foodborne illnesses and increased the likelihood that I will consider such illnesses in my patients. 28. The primer increased my knowledge and skills in
FOODBORNE INFECTIONS. Caroline Charlier-Woerther March 2017
 FOODBORNE INFECTIONS Caroline Charlier-Woerther March 2017 LEARNING OBJECTIVES Know the pathogens involved in diarrheas Know the basics of management of diarrhea Know the main patterns of listeriosis and
FOODBORNE INFECTIONS Caroline Charlier-Woerther March 2017 LEARNING OBJECTIVES Know the pathogens involved in diarrheas Know the basics of management of diarrhea Know the main patterns of listeriosis and
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Calvepen 666 mg Tablets. Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 666 mg of Phenoxymethylpenicillin Calcium equivalent
1 NAME OF THE MEDICINAL PRODUCT Calvepen 666 mg Tablets. Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 666 mg of Phenoxymethylpenicillin Calcium equivalent
SCHEDULING STATUS: S0 For pack sizes of 24 tablets or less. For pack sizes of more than 24 tablets
 SCHEDULING STATUS: S0 For pack sizes of 24 tablets or less S1 For pack sizes of more than 24 tablets PROPRIETARY NAME: AND DOSAGE FORM PANADO MELTABS (Tablets) COMPOSITION: Each tablet contains 500 mg
SCHEDULING STATUS: S0 For pack sizes of 24 tablets or less S1 For pack sizes of more than 24 tablets PROPRIETARY NAME: AND DOSAGE FORM PANADO MELTABS (Tablets) COMPOSITION: Each tablet contains 500 mg
WARNING: TENDON EFFECTS and EXACERBATION OF MYASTHENIA GRAVIS
 DECLESAU (dergrafloxacin) tablets, for oral use DECLESAU (dergrafloxacin) injection, solution for intravenous use WARNING: TENDON EFFECTS and EXACERBATION OF MYASTHENIA GRAVIS Fluoroquinolones, including
DECLESAU (dergrafloxacin) tablets, for oral use DECLESAU (dergrafloxacin) injection, solution for intravenous use WARNING: TENDON EFFECTS and EXACERBATION OF MYASTHENIA GRAVIS Fluoroquinolones, including
Structure of viruses
 Antiviral Drugs o Viruses are obligate intracellular parasites. o lack both a cell wall and a cell membrane. o They do not carry out metabolic processes. o Viruses use much of the host s metabolic machinery.
Antiviral Drugs o Viruses are obligate intracellular parasites. o lack both a cell wall and a cell membrane. o They do not carry out metabolic processes. o Viruses use much of the host s metabolic machinery.
Antituberculous Agents
 Antituberculous Agents Primary or First Line Drugs: Isoniazid (INH) Rifampin Rifadin or Rimactane Ethambutal Streptomycin Pyrazinamide Isoniazid(INH) Most active. Small molecule, water soluble, Structurally
Antituberculous Agents Primary or First Line Drugs: Isoniazid (INH) Rifampin Rifadin or Rimactane Ethambutal Streptomycin Pyrazinamide Isoniazid(INH) Most active. Small molecule, water soluble, Structurally
CHAPTER AT A GLANCE. Science Class (SA-II)- IX
 13 CHAPTER AT A GLANCE Health is a general condition of a preson s mind and body. According to WHO (World Health Organisation) health is a state of physical, metal and social well-being of a person. To
13 CHAPTER AT A GLANCE Health is a general condition of a preson s mind and body. According to WHO (World Health Organisation) health is a state of physical, metal and social well-being of a person. To
My dog or cat has problems with the stomach, intestines or liver... what do I do now?
 Dietary food for the targeted tackling of gastrointestinal and/or liver problems in dogs and cats My dog or cat has problems with the stomach, intestines or liver... what do I do now? reliable and and
Dietary food for the targeted tackling of gastrointestinal and/or liver problems in dogs and cats My dog or cat has problems with the stomach, intestines or liver... what do I do now? reliable and and
Microbial Diseases of the Digestive System
 PowerPoint Lecture Presentations prepared by Mindy Miller-Kittrell, North Carolina State University Packet #13 Chapter #23 Microbial Diseases of the Digestive System Structures of the Digestive System
PowerPoint Lecture Presentations prepared by Mindy Miller-Kittrell, North Carolina State University Packet #13 Chapter #23 Microbial Diseases of the Digestive System Structures of the Digestive System
Protein Synthesis Inhibitors. Ass Prof. Dr. Naza M. Ali 15 Nov 2018 Lec 8
 Protein Synthesis Inhibitors Ass Prof. Dr. Naza M. Ali 15 Nov 2018 Lec 8 These drugs selectively inhibit bacterial protein synthesis. The selectivity is due to the differences between bacterial and human
Protein Synthesis Inhibitors Ass Prof. Dr. Naza M. Ali 15 Nov 2018 Lec 8 These drugs selectively inhibit bacterial protein synthesis. The selectivity is due to the differences between bacterial and human
Package Insert. Constipeg
 Package Insert Constipeg Product Summary 1. Name of the medicinal product Constipeg sachets 2. Qualitative and quantitative composition Each 13.7 g sachet contains PEG polyethylene glycol (macrogol) USNF
Package Insert Constipeg Product Summary 1. Name of the medicinal product Constipeg sachets 2. Qualitative and quantitative composition Each 13.7 g sachet contains PEG polyethylene glycol (macrogol) USNF
SUMMARY OF PRODUCT CHARACTERISTICS 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Fexofenadine hydrochloride 180 mg film-coated tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each film coated tablet contains 180mg
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Fexofenadine hydrochloride 180 mg film-coated tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each film coated tablet contains 180mg
COLAZIDE PRODUCT INFORMATION NAME OF MEDICINE. Structural Formula: CAS
 Colazide Product Information 1/9 COLAZIDE PRODUCT INFORMATION NAME OF MEDICINE Generic Name: balsalazide sodium Structural Formula: CAS 80573-04-2 DESCRIPTION Capsules containing 750 mg balsalazide sodium,
Colazide Product Information 1/9 COLAZIDE PRODUCT INFORMATION NAME OF MEDICINE Generic Name: balsalazide sodium Structural Formula: CAS 80573-04-2 DESCRIPTION Capsules containing 750 mg balsalazide sodium,
